Attached files
| file | filename |
|---|---|
| EX-99.2 - TRANSCRIPT OF CONFERENCE CALL - QUESTCOR PHARMACEUTICALS INC | d247478dex992.htm |
| EX-99.1 - PRESS RELEASE - QUESTCOR PHARMACEUTICALS INC | d247478dex991.htm |
| 8-K - FORM 8-K - QUESTCOR PHARMACEUTICALS INC | d247478d8k.htm |
 1
NASDAQ:
QCOR
NASDAQ:
QCOR
Third Quarter 2011 Conference Call
Third Quarter 2011 Conference Call
Exhibit 99.3 |
 2
•
•
Telephone replay is available by dialing:
–
U.S.: 800-406-7325
–
International: 303-590-3030
–
Replay Passcode: 4480054
•
•
Telephone replay is available by dialing:
–
U.S.: 800-406-7325
–
International: 303-590-3030
–
Replay Passcode: 4480054
Conference Call Logistics
2
Today’s webcast, accompanying slide presentation and
archived replay is available online at
http://ir.questcor.com/events.cfm. |
 3
Safe Harbor Statement
Note: Except for the historical information contained herein, this press release contains
forward-looking statements that have been made pursuant to the Private Securities
Litigation Reform Act of 1995. These statements relate to future events or our future
financial performance. In some cases, you can identify forward-looking statements by terminology such as
“believes,” “continue,” “could,” “estimates,”
“expects,” “growth,” “may,” “plans,” “potential,” “should,” “substantial” or
“will” or the negative of such terms and other comparable terminology. These
statements are only predictions. Actual events or results may differ materially.
Factors that could cause or contribute to such differences include, but are not limited
to, the following: our reliance on Acthar for substantially all of our net sales and profits; Reductions in vials used
per prescription resulting from changes in treatment regimens by physicians or patient
compliance with physician recommendations; the complex nature of our manufacturing
process, and the potential for supply disruptions or other business disruptions; the
lack of patent protection for Acthar, and the possible FDA approval and market introduction of
competitive products; our ability to generate revenue from sales of Acthar to treat
on-label indications associated with NS and our ability to develop other therapeutic
uses for Acthar; research and development risks, including risks associated with
Questcor's preliminary work in the area of nephrotic syndrome and potential work in the area of systemic lupus
erythematosus, and our reliance on third-parties to conduct research and development and
the ability of research and development to generate successful results; regulatory
changes or other policy actions by governmental authorities and other third parties in
connection with U.S. health care reform or efforts to reduce federal and state government deficits;
our ability to receive high reimbursement levels from third party payers; an increase in the
proportion of our Acthar unit sales comprised of Medicaid-eligible patients and
government entities; our ability to estimate reserves required for Acthar used by
government entities and Medicaid-eligible patients and the impact that unforeseen invoicing of historical
Medicaid sales may have upon our results; our ability to operate within an industry that is
highly regulated at both the Federal and state level; our ability to effectively manage
our growth and our reliance on key personnel; the impact to our business caused by
economic conditions; our ability to protect our proprietary rights; our ability to maintain effective
controls over financial reporting; the risk of product liability lawsuits; unforeseen business
interruptions; volatility in Questcor's monthly and quarterly Acthar shipments and
end-user demand, as well as volatility in our stock price; and other risks
discussed in Questcor's annual report on Form 10-K for the year ended December 31, 2010, and other
documents filed with the Securities and Exchange Commission.
The risk factors and other information contained in these documents should be considered in
evaluating Questcor's prospects and future financial performance.
The risk factors and other information contained in these documents should be considered in
evaluating Questcor's prospects and future financial performance.
|
 4
•
886 paid MS scripts
–
Up 174% YOY
–
Up 18% sequentially
•
60 paid NS scripts
–
Much better than expected
–
Sales force expansion complete
•
Record financial performance
–
2,910 vials
–
$59.8M in net sales
–
$0.35 EPS
•
886 paid MS scripts
–
Up 174% YOY
–
Up 18% sequentially
•
60 paid NS scripts
–
Much better than expected
–
Sales force expansion complete
•
Record financial performance
–
2,910 vials
–
$59.8M in net sales
–
$0.35 EPS
Strong Third Quarter Results |
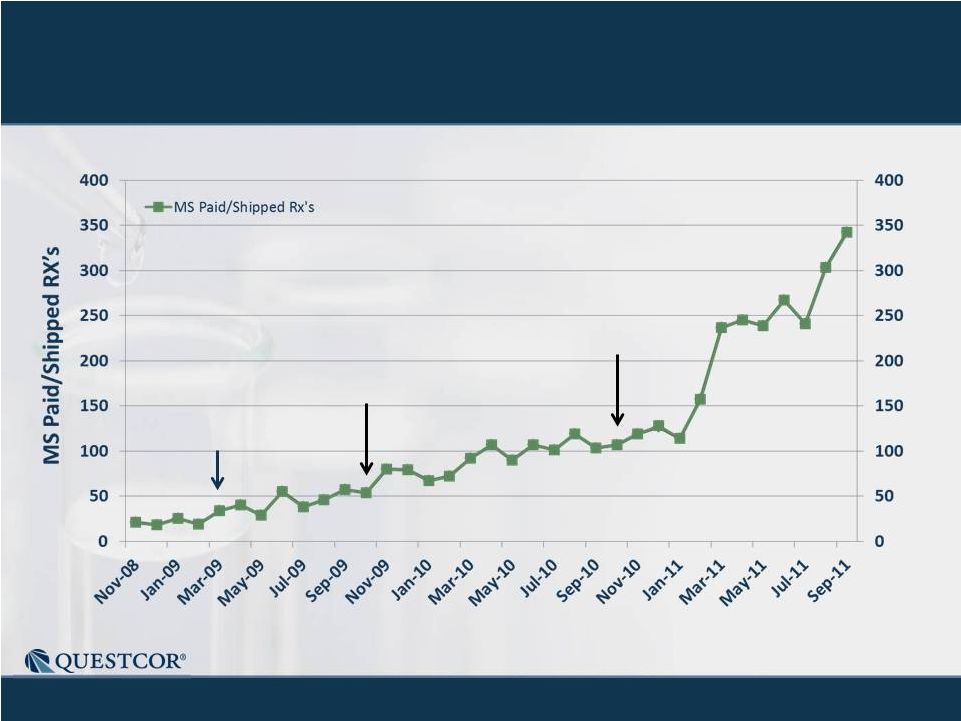
 5
MS
Sales
-
Record
of
Consistent
Growth
New
Paid
Rxs
New
Paid
Rxs
Notes: Historical trend information is not necessarily indicative of future results.
Chart includes "Related Conditions" - diagnoses that are
either alternative descriptions of the condition or are closely related to the medical
condition which is the focus of the chart. 77
15
30
38
Yellow numbers in the bars show the number of MS sales
people making calls at the end of the quarter.
8
24
35
51
69
78
124
141
213
231
304
323
354
508
751
886
Q1 '08
Q2 '08
Q3 '08
Q4 '08
Q1 '09
Q2 '09
Q3 '09
Q4 '09
Q1 '10
Q2 '10
Q3 '10
Q4 '10
Q1 '11
Q2 '11
Q3' 11 |
 6
Monthly MS Sales Have 160% CAGR
15-30 reps
30-38 reps
38-77 reps |
 7
NS Sales –
Off to a Good Start
New Paid Rxs
New Paid Rxs
Notes: Historical trend information is not necessarily indicative of future results.
Chart includes "Related Conditions" - diagnoses that are
either alternative descriptions of the condition or are closely related to the medical
condition which is the focus of the chart. 5
Yellow numbers in the bars show the number of NS sales people
making
calls
at
the
end
of
the
quarter.
Q3’
11
included
expansion
and training of new sales people.
11
4
8
7
18
45
60
Q1 '10
Q2 '10
Q3 '10
Q4 '10
Q1 '11
Q2 '11
Q3 '11 |
 8
MS Sales -
Record of Consistent Growth
New Paid Rxs
New Paid Rxs
Notes: Historical trend information is not necessarily indicative of future results.
Chart includes "Related Conditions" - diagnoses that are
either alternative descriptions of the condition or are closely related to the medical
condition which is the focus of the chart. 77
15
30
38
Yellow numbers in the bars show the number of MS sales
people making calls at the end of the quarter.
8
Q1 '08
Q2 '08
Q3 '08
Q4 '08
Q1 '09
Q2 '09
Q3 '09
Q4 '09
Q1 '10
Q2 '10
Q3 '10
Q4 '10
Q1 '11
Q2 '11
Q3' 11
24
35
51
69
78
124
141
213
231
304
323
354
508
751
886 |
 9
NS Sales –
Off to a Good Start
New Paid Rxs
New Paid Rxs
Notes: Historical trend information is not necessarily indicative of future results.
Chart includes "Related Conditions" - diagnoses that are
either alternative descriptions of the condition or are closely related to the medical
condition which is the focus of the chart. 5
Yellow numbers in the bars show the number of NS sales people
making calls at the end of the quarter. Q3 ‘11 included expansion
and training of new sales people. |
 10
•
Specialty Sales Force
–
Main focus on MS (~80%), 15% on NS, 5% on IS
–
77 representatives, 13 regional managers, one national director
•
Nephrology Sales Force
–
Focus 100% on nephrotic syndrome
–
28 representatives, 4 regional managers, one national director
•
Combined Forces are calling on
–
>4,000 neurologists
–
>3,000 nephrologists
–
about 100 key children’s hospitals
•
Specialty Sales Force
–
Main focus on MS (~80%), 15% on NS, 5% on IS
–
77 representatives, 13 regional managers, one national director
•
Nephrology Sales Force
–
Focus 100% on nephrotic syndrome
–
28 representatives, 4 regional managers, one national director
•
Combined Forces are calling on
–
>4,000 neurologists
–
>3,000 nephrologists
–
about 100 key children’s hospitals
Total Acthar Sales Force |
 11
•
Treatment of Resistant Glomerular Diseases with ACTH Gel: A
Prospective Trial [G. Appel, et al]
•
Response of Anti-PLA2R to Adrenocorticotropic hormone (ACTH)
Gel in Primary Membranous Nephropathy [L. Beck, et al]
•
Adrenocortocortropin (ACTH) gel suppresses renal
tubulointerstitial inflammation and injury by direct stimulation
of
the melanocortin 1 receptor [R. Gong, et al]
•
Advanced Diabetic Nephropathy with Nephrotic Range Proteinuria:
Long-Term Efficacy of Subcutaneous Adrenocorticotrophic
Hormone (ACTH )Therapy on Proteinuria and Urinary Vascular
Endothelial Growth Factor (VEGF) levels [J. Tumlin, et al]
•
Treatment of Resistant Glomerular Diseases with ACTH Gel: A
Prospective Trial [G. Appel, et al]
•
Response of Anti-PLA2R to Adrenocorticotropic hormone (ACTH)
Gel in Primary Membranous Nephropathy [L. Beck, et al]
•
Adrenocortocortropin (ACTH) gel suppresses renal
tubulointerstitial inflammation and injury by direct stimulation
of
the melanocortin 1 receptor [R. Gong, et al]
•
Advanced Diabetic Nephropathy with Nephrotic Range Proteinuria:
Long-Term Efficacy of Subcutaneous Adrenocorticotrophic
Hormone (ACTH )Therapy on Proteinuria and Urinary Vascular
Endothelial Growth Factor (VEGF) levels [J. Tumlin, et al]
www.asn-online.org
ASN Annual Meeting 2011: Acthar-Related Abstracts
|
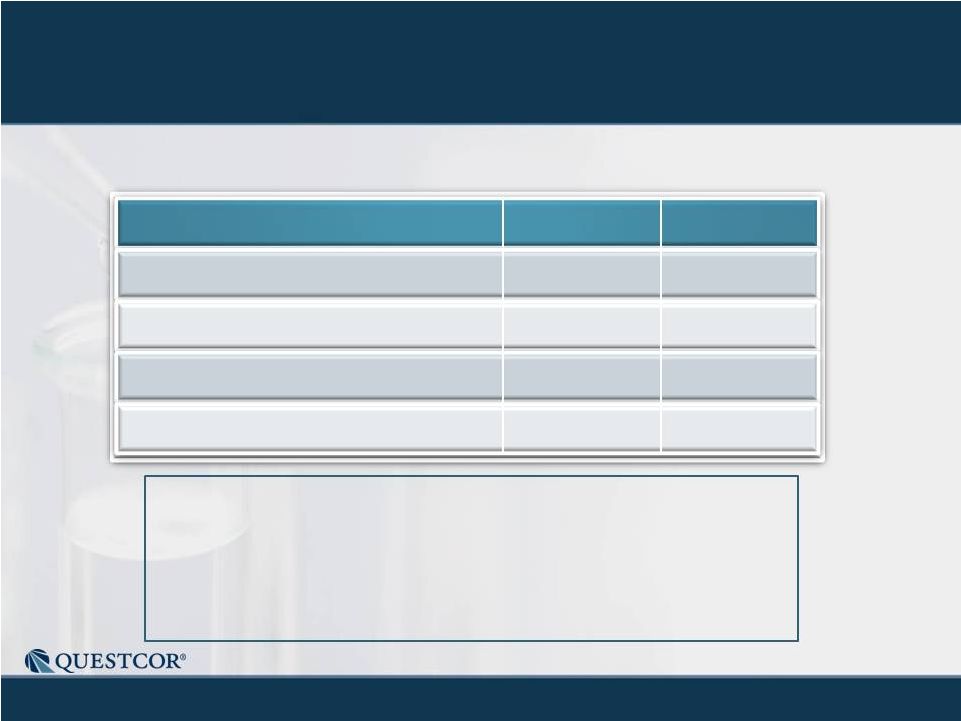 12
Record Net Sales (up 91%) and Solid Earnings (EPS up 94%)
Record Net Sales (up 91%) and Solid Earnings (EPS up 94%)
•
Third quarter vials shipped: 2,910
•
Third quarter cash flow from operations: $32.6M
•
Channel inventory estimated to be within historic range
•
Medicaid reserves continue to appear adequate
•
No shares repurchased
Net Sales ($M)
Gross Margin
Operating Income ($M)
Fully Diluted, GAAP EPS
$59.8
94%
$33.6
$0.35
$31.3
93%
$16.8
$0.18
Q3-2011
Q3-2010
12
Q3-2011 Financial Results |
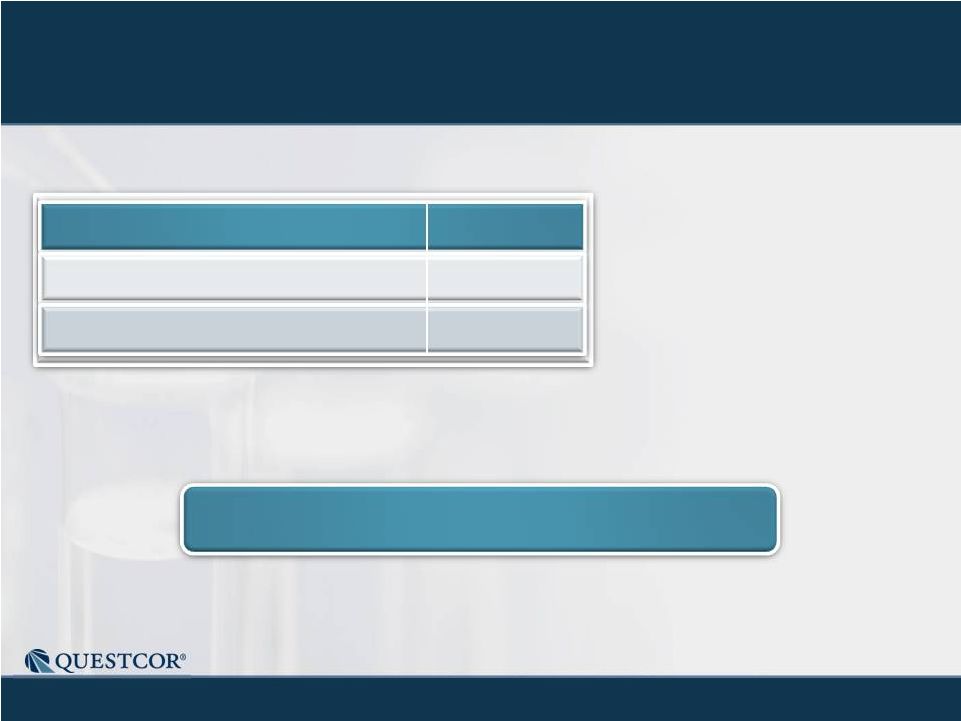 13
*After return of $78 million of cash to shareholders through
share repurchases.
Cash / ST Investments
Accounts Receivable
$180M*
$24M
10/21/11
Debt-free balance sheet
Debt-free balance sheet
Questcor is Cash Flow Positive |
 14
Acthar has sustainable competitive
advantages-without FDA approval risk
Acthar is approved for 19 indications-many
in large markets with sizable unmet need
Sales in MS and NS are growing rapidly,
yet market penetration is low
High margins provide good operating
leverage
Profitable, cash flow positive, no debt
Investment Highlights |
 15
NASDAQ:
QCOR
NASDAQ:
QCOR
Third Quarter 2011 Conference Call
Third Quarter 2011 Conference Call |
