Attached files
| file | filename |
|---|---|
| 8-K - FORM 8-K - ENDO HEALTH SOLUTIONS INC. | d234577d8k.htm |
 ENDO
PHARMACEUTICALS UBS Global Life Sciences Conference
September 21, 2011
Exhibit 99.1
grow. collaborate. innovate.
thrive. |
 grow.
collaborate. innovate. thrive. ©2011 Endo Pharmaceuticals
2
FORWARD LOOKING STATEMENTS
This presentation contains forward-looking statements within the meaning of the Private
Securities Litigation
Reform
Act
of
1995.
Statements
including
words
such
as
“believes,”
“expects,”
“anticipates,”
“intends,”
“estimates,”
“plan,”
“will,”
“may,”
“look forward,”
“intend,”
“guidance,”
“future”
or similar
expressions are forward-looking statements. Because these statements reflect our
current views, expectations and beliefs concerning future events, these
forward-looking statements involve risks and uncertainties. Investors should note
that many factors, as more fully described under the caption “Risk
Factors”
in our Form 10-K, Form 10-Q and Form 8-K filings with the Securities and Exchange
Commission and as otherwise enumerated herein or therein, could affect our future
financial results and could cause our actual results to differ materially from those
expressed in forward-looking statements contained in our Annual Report on Form
10-K. The forward-looking statements in this presentation are qualified by
these risk factors. These are factors that, individually or in the aggregate, could cause our
actual results to differ materially from expected and historical results. We assume no
obligation to publicly update any forward-looking statements, whether as a result
of new information, future developments or otherwise. |
 grow.
collaborate. innovate. thrive. ENDO PHARMACEUTICALS
I.
Our Business
II.
Executing Strategy for Growth
III.
American Medical Systems
IV.Financial
Outlook 3 |
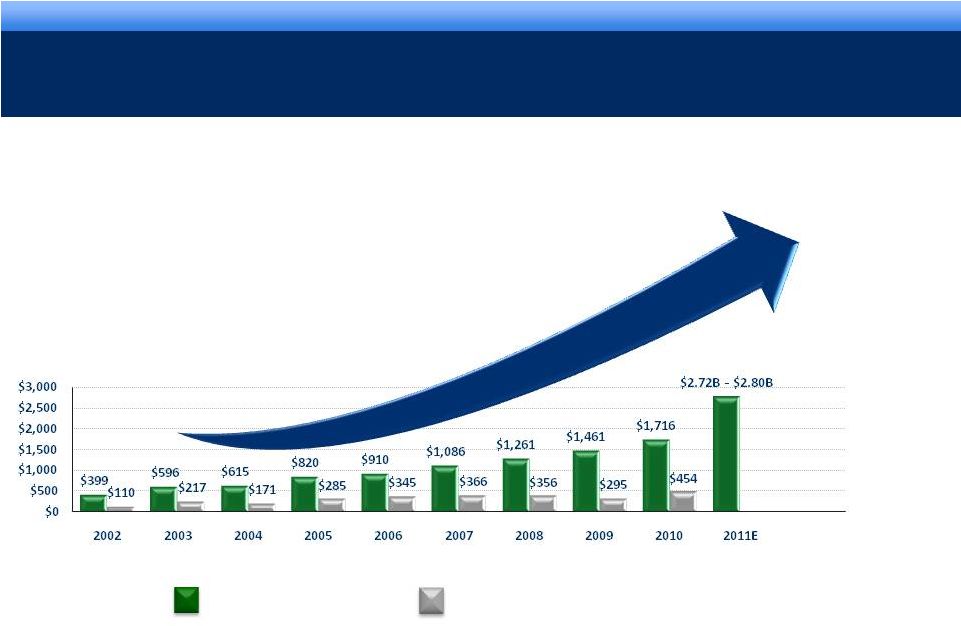 STRONG
OPERATING PERFORMANCE 16% 3-YEAR CAGR FOR REVENUE*
* Revenue CAGR 2007-2010.
4
Revenue
Cash Flow from Operations
$mm
grow. collaborate. innovate. thrive.
©2011 Endo Pharmaceuticals
Sustaining our Growth |
 grow.
collaborate. innovate. thrive. ©2011 Endo Pharmaceuticals
5
Through Organic Growth
•
Diversified business lines to maximize
growth
•
Enhanced commercial model driving
growth via strategic resource deployment
•
Invested in R&D portfolio yielding a
diversified pipeline of products and a
recent FDA approval
•
Bolstered management team by adding
expertise and experience in managing and
growing a larger enterprise
5
ENDO’S TRANSFORMATION
Through Strategic Growth
•
With Indevus, we secured a position in
urology
•
HealthTronics gave us an established
presence in Devices & Services and critical
mass in urology
•
Penwest strengthened our pain business
enhancing profitability & flexibility in the
opioid franchise
•
Qualitest brings critical mass to our
generics business & strengthens our pain
portfolio
•
AMS establishes scale in devices
and services and solidifies our position
covering the urology market |
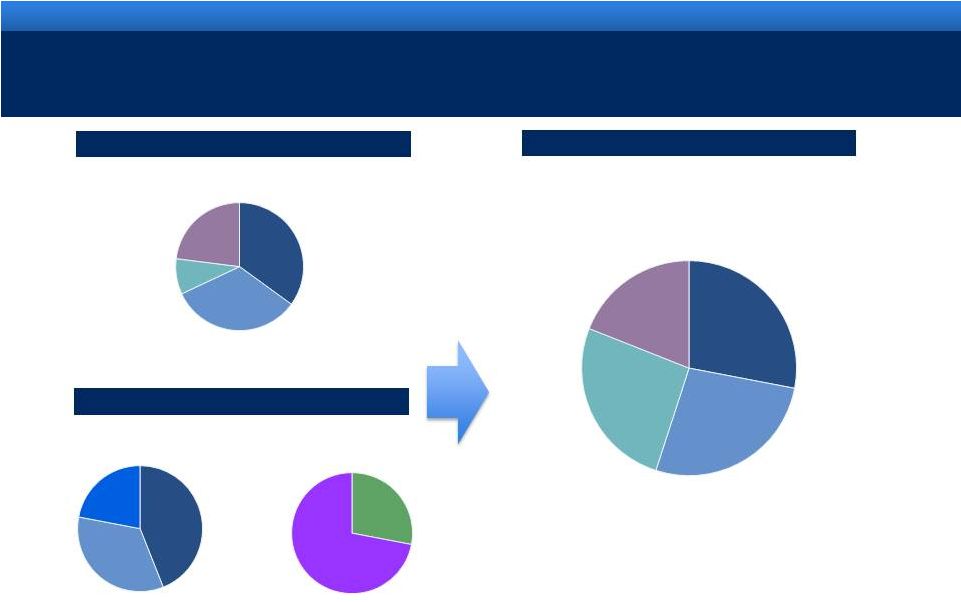 grow.
collaborate. innovate. thrive. ©2011 Endo Pharmaceuticals
6
DIVERSIFIED HEALTHCARE SOLUTIONS COMPANY
Lidoderm®
Generic
Men’s
Health
TTM 6/30/11: $2.25B
(1)
TTM 6/30/11: $0.54B
(2)
TTM 6/30/11: $2.79B
(3)
Revenue mix
Endo
AMS
Pro Forma
1.
Represents TTM of pro forma 2010 acquisitions (Qualitest, HealthTronics and Penwest,
excluding AMS) 2.
Includes $1.1MM of revenue from the uterine health business, which was divested during the
first quarter 2010. 3.
Includes full year of AMS acquisition
Other Branded
Devices &
Services
Women’s
Health
BPH
Lidoderm®
Generic
Other Branded
Devices &
Services
Revenue mix
Revenue mix
International
US
35%
33%
9%
23%
28%
27%
26%
19%
28%
72%
22%
34%
44% |
 grow.
collaborate. innovate. thrive. ©2011 Endo Pharmaceuticals
7
ENDO’S INTEGRATED BUSINESS MODEL
Branded
Pharmaceuticals
Generics
Devices
& Services
Pain
Urology
Oncology
Endocrinology |
 grow.
collaborate. innovate. thrive. ©2011 Endo Pharmaceuticals
STRONG CORE BUSINESS SUPPORTING GROWTH
8 |
 grow.
collaborate. innovate. thrive. ©2011 Endo Pharmaceuticals
TESTOSTERONE REPLACEMENT THERAPY
An estimated 14 million men suffer with hypogonadism in US
Approximately 9% are treated
Market TRx Net Sales: >$1.2B
Growth Rate: approximately 20% CAGR last 5 years
Gels have the greatest utilization in the TRT market: ~72%
PCPs account for majority of TRT prescriptions:
More than 60% through Primary Care Physicians
Approximately 30% through specialty (Urologists/Endocrinologists)
Method of Payment for TRT class:
Approximately 80% Commercial
Approximately 12% Government
Remainder from Cash Payment
9 |
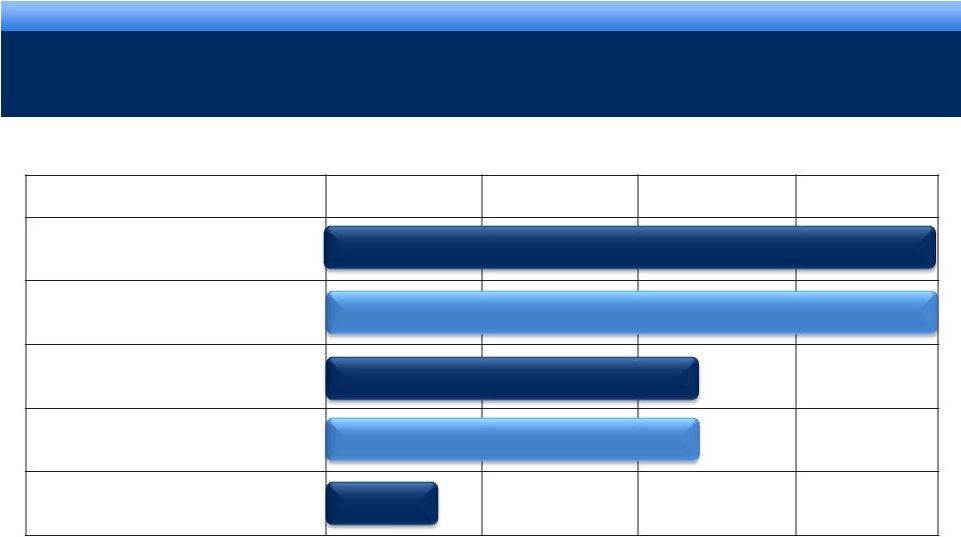 grow.
collaborate. innovate. thrive. DEVELOPMENT PIPELINE*
* There can be no assurance that any of these development programs will be
successful or if successful, the products will ultimately be approved by FDA. **
Granted orphan drug designation *** Licensed from Orion Corporation for joint
development and commercialization 10
Phase I
NDA
Phase II
Phase III
Pending
Update Pending
AVEED
TM
Long Acting Injectable Testosterone
Urocidin
TM
Bladder Cancer
Octreotide Implant
Acromegaly**
Androgen Receptor Agonist***
Castration Resistant Prostate Cancer
OPANA®
ER
Formulation designed to be crush-resistant
©2011 Endo Pharmaceuticals |
 grow.
collaborate. innovate. thrive. ©2011 Endo Pharmaceuticals
11
Combined Generics Business
•
Solid commercial base entering 2011
•
Anticipate to file 14 ANDAs in 2011
•
50 ANDAs currently under review by FDA
•
Expect 14 ANDA approvals in 2011
–
5 ANDA approvals YTD
We expect our combined generics business to grow over 15% over the 2010-
12 period driven by launches from the development portfolio
2011 Generic Development
14 ANDA
Approvals
50 Current
ANDA
Reviews
14 ANDA
Filings
STRONG GENERICS BUSINESS WITH A ROBUST PIPELINE |
 grow.
collaborate. innovate. thrive. ©2011 Endo Pharmaceuticals
12
AMERICAN MEDICAL SYSTEMS ACQUISITION |
 grow.
collaborate. innovate. thrive. ©2011 Endo Pharmaceuticals
13
AMS –
STRATEGIC RATIONALE
Furthers stated strategy to respond to
the changing economics that drive the
U.S. healthcare environment
•
Evolving from a product-driven company to a healthcare
solutions provider
•
Healthcare reform puts a premium on providing cost-
effective health solutions
•
Balances Endo’s integrated business model
Strengthens Endo’s core urology
franchise
•
AMS gives Endo scale in urology therapeutic space with
approximately $1BN in sales
•
AMS enhances Endo’s ability to offer patients solutions
through the entire course of urology treatment options
•
AMS has solid market share in several niche categories
that have grown in the mid-to-high single digit range over
the long term
Brings scale to devices and services
business
•
Generally shorter, less expensive product development
•
Favorable pricing and reimbursement flexibility
•
Higher degree of cash flow sustainability supported by
life cycle management through innovation, less product
substitution risk and continued volume-driven market
growth
By acquiring a high-growth asset, will
diversify and boost revenue and
earnings’
streams
•
Advances growth on several metrics and is immediately
accretive to earnings on a cash basis by $0.60 in 2012.
•
Further diversifies Endo across Branded Pharmaceuticals,
Generic and Devices & Services segments
•
AMS has a robust product portfolio and a strong product
cadence over the next 12 months |
 grow.
collaborate. innovate. thrive. ©2011 Endo Pharmaceuticals
14
Leading Provider of Devices to Pelvic Health Space
•
Men’s Health: Solutions for male incontinence and erectile
dysfunction prostheses
•
Women’s Health: Solutions for female incontinence and pelvic
floor repair
•
Benign Prostatic Hyperplasia (BPH) Therapy: Laser treatments
Strong Product Portfolio Supported by Robust Future
Product Cadence
•
Solid market share in several categories that have historically
grown in the mid-to-high single digit range over the long-term
•
Near-term growth driven by new product launches and
initiatives such as the GreenLight XPS console and MoXy fiber
Seasoned Management Team and High Quality Assets
•
Team has significant experience running a growing devices
business
•
Four global facilities –
two in the US (Minnetonka, MN and San
Jose, CA) and two in Europe (Bruekelen, Netherlands and
Athlone, Ireland)
14
AMS COMPANY PROFILE
Revenue by Segment
FY 2010
Revenue by Geography
FY 2010
Other 1%
Men's Health 45%
BPH 21%
Women's Health 33%
International 27%
United States 73% |
 grow.
collaborate. innovate. thrive. ©2011 Endo Pharmaceuticals
15
AMS –
PRODUCT PORTFOLIO
Men’s Health
BPH
Women’s Health
Product
Description
AMS 800®
Artificial Urinary
Sphincter
InVance®
Less invasive treatment
for moderate
incontinence
AdVance®
Less invasive treatment
for mild to moderate
incontinence
UroLume®
Endoprosthesis for non-
surgical candidates
AMS 700®
Semi-rigid malleable
prostheses/inflatable
prostheses for treatment
of Erectile Dysfunction
Product
Description
Monarc®
Self-fixating, subfascial
hammock for treatment of
stress incontinence
MiniArc®
Single-Incision Sling for
stress incontinence
Elevate®
Transvaginal pelvic floor
repair system (requires no
external incisions)
Product
Description
GreenLight™
Photo Vaporization / laser
therapy designed to
remove prostatic tissue
StoneLight®
Laser treatment of urinary
stones
SureFlex™
Fiber optic line designed
for holmium laser
lithotripsy
TherMatrx®
Less invasive tissue
ablation technique for
men not yet to the point
of urethral obstruction |
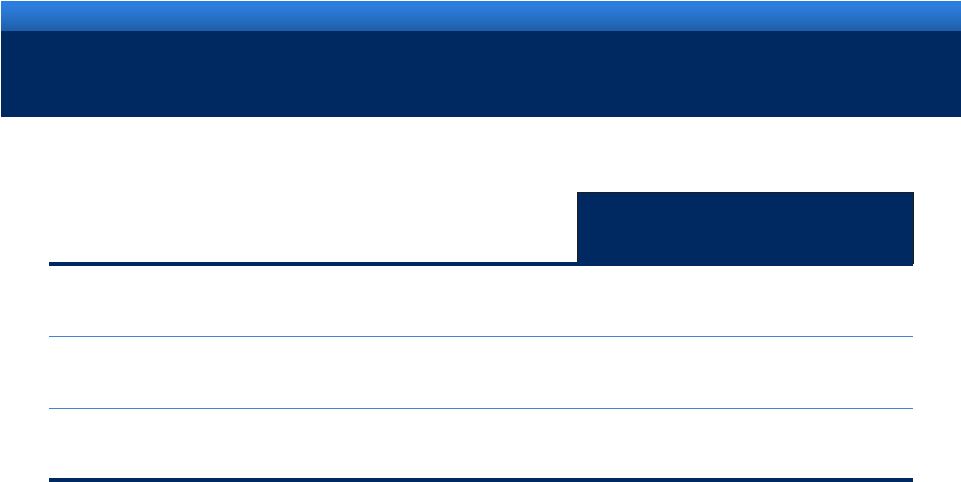 grow.
collaborate. innovate. thrive. ©2011 Endo Pharmaceuticals
16
2011 ENDO GUIDANCE
Guidance
Revenue range
$2.72B -
$2.80B
Adjusted diluted EPS range
$4.55 -
$4.65
Reported (GAAP) diluted EPS range
$2.22 -
$2.32 |
 ENDO
PHARMACEUTICALS grow. collaborate. innovate. thrive. |
 grow.
collaborate. innovate. thrive. ©2011 Endo Pharmaceuticals
18
18
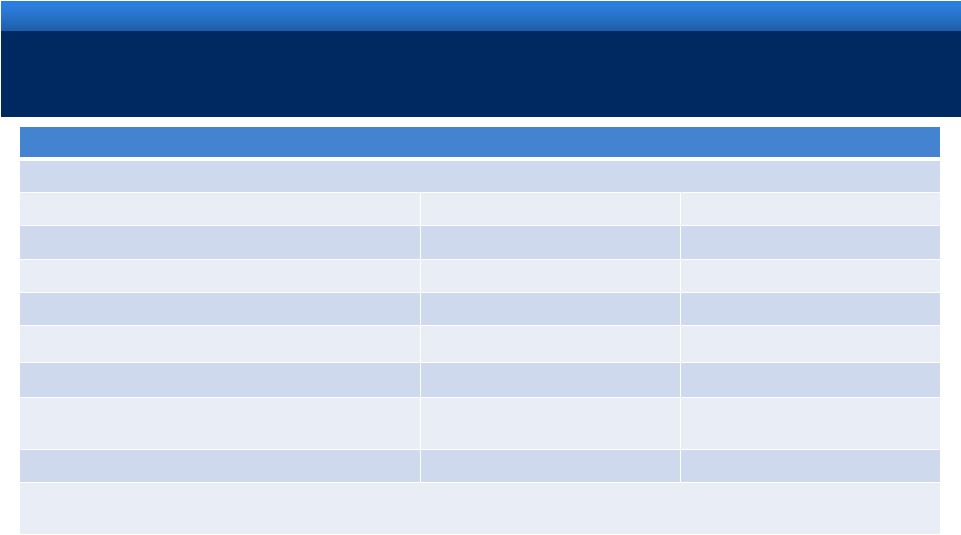
RECONCILIATION OF NON-GAAP MEASURES
18
For an explanation of Endo’s reasons for using non-GAAP measures, see Endo’s
Current Report on Form 8-K filed today with the Securities and Exchange Commission
Reconciliation of Projected GAAP Diluted Earnings Per Share to Adjusted Diluted Earnings Per
Share Guidance for the Year Ending December 31, 2011 Lower End of Range
Upper End of Range
Projected GAAP diluted income per common share
$2.22
$2.32
Upfront and milestone-related payments to partners
$0.27
$0.27
Amortization of commercial intangible assets and inventory step-up
$1.94
$1.94
Acquisition and integration costs related to recent acquisitions.
$0.40
$0.40
Interest expense adjustment for ASC 470-20 and the amortization of
the premium on debt acquired from Indevus
$0.16
$0.16
Tax effect of pre-tax adjustments at the applicable tax rates and
certain other expected cash tax savings as a result of recent
acquisitions
($0.44)
($0.44)
Diluted adjusted income per common share guidance
$4.55
$4.65
The company's guidance is being issued based on certain assumptions including:
•Certain of the above amounts are based on estimates and there
can be no assurance that Endo will achieve these results
•Includes all completed business development transactions as of
September 21, 2011 |
 ENDO
PHARMACEUTICALS UBS Global Life Sciences Conference
September 21, 2011
grow. collaborate. innovate.
thrive. |
