Attached files
| file | filename |
|---|---|
| EX-21.1 - AEOLUS PHARMACEUTICALS, INC. | v206778_ex21-1.htm |
| EX-31.2 - AEOLUS PHARMACEUTICALS, INC. | v206778_ex31-2.htm |
| EX-31.1 - AEOLUS PHARMACEUTICALS, INC. | v206778_ex31-1.htm |
| EX-32.1 - AEOLUS PHARMACEUTICALS, INC. | v206778_ex32-1.htm |
| EX-23.1 - AEOLUS PHARMACEUTICALS, INC. | v206778_ex23-1.htm |
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington,
D.C. 20549
FORM
10-K
(MARK
ONE)
x ANNUAL
REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF
1934
For
the fiscal year ended September 30, 2010
OR
¨ TRANSITION
REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF
1934
For
the transition period from ____________ to__________
Commission
File Number 0-50481
AEOLUS
PHARMACEUTICALS, INC.
(Exact
name of registrant as specified in its charter)
|
Delaware
|
56-1953785
|
|
|
(State or other jurisdiction of
incorporation or organization)
|
(I.R.S. Employer
Identification No.)
|
|
26361 Crown Valley Parkway, Suite 150
|
||
|
Mission Viejo, California
|
92691
|
|
|
(Address of principal executive offices)
|
(Zip Code)
|
Registrant’s
telephone number, including area code: 949-481-9825
Securities
registered pursuant to Section 12(b) of the Act: None
Securities
registered pursuant to Section 12(g) of the Act:
Common
Stock, $.01 par value per share
(Title
of class)
Indicate
by check mark if the registrant is a well-known seasoned issuer, as defined in
Rule 405 of the Securities Act. Yes ¨ No x
Indicate
by check mark if the registrant is not required to file reports pursuant to
Section 13 or Section 15(d) of the Act. Yes ¨ No x
Indicate
by check mark whether the registrant (1) has filed all reports required to be
filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the
preceding 12 months (or for such shorter period that the registrant was required
to file such reports), and (2) has been subject to such filing requirements for
the past 90 days. Yes x No ¨
Indicate
by check mark whether the registrant has submitted electronically and posted on
its corporate Web site, if any, every Interactive Data File required to be
submitted and posted pursuant to Rule 405 of Regulation S-T (§ 232.405 of this
chapter) during the preceding 12 months (or for such shorter period that the
registrant was required to submit and post such files). Yes ¨ No
¨
Indicate
by check mark if disclosure of delinquent filers pursuant to Item 405 of
Regulation S-K (§ 229.405 of this chapter) is not contained herein,
and will not be contained, to the best of registrant’s knowledge, in definitive
proxy or information statements incorporated by reference in Part III of this
Form 10-K or any amendment to this Form 10-K. ¨
Indicate
by check mark whether the registrant is a large accelerated filer, an
accelerated filer, a non-accelerated filer, or a smaller reporting
company. See the definitions of “large accelerated filer,”
“accelerated filer” and “smaller reporting company” in Rule 12b-2 of the
Exchange Act.
|
Large accelerated filer ¨
|
|
Accelerated filer ¨
|
|
Non-accelerated filer ¨
(Do not check if a smaller reporting company)
|
|
Smaller reporting company x
|
Indicate
by check mark whether the registrant is a shell company (as defined in Rule
12b-2 of the Exchange Act). Yes ¨ No x
The
aggregate market value of the voting common stock held by non-affiliates of the
registrant based upon the average of the bid and asked price on the OTC Bulletin
Board as of March 31, 2010, the last business day of the registrant’s most
recently completed second fiscal quarter, was approximately
$2,513,684. Shares of common stock held by each executive officer and
director and by each other stockholder who owned 10% or more of the outstanding
common stock as of such date have been excluded in that such stockholder might
be deemed to be an affiliate of the registrant. This determination of
affiliate status might not be conclusive for other purposes.
As of
December 29, 2010, the registrant had 59,684,050 outstanding shares of common
stock.
DOCUMENTS
INCORPORATED BY REFERENCE
Portions
of the registrant’s definitive Information Statement to be filed pursuant to
Regulation 14C in connection with the registrant’s Written Consent in Lieu of
the 2011 Annual Meeting of Stockholders are incorporated herein by reference
into Part III hereof.
AEOLUS
PHARMACEUTICALS, INC.
ANNUAL
REPORT ON FORM 10-K
Table
of Contents
|
Page
|
|||
|
PART
I
|
|||
|
Item 1.
|
Business
|
3
|
|
|
Executive
Officers
|
30
|
||
|
Item 1A.
|
Risk
Factors
|
31
|
|
|
Item 1B.
|
Unresolved
Staff Comments
|
45
|
|
|
Item 2.
|
Properties
|
45
|
|
|
Item 3.
|
Legal
Proceedings
|
45
|
|
|
Item 4.
|
(Removed
and Reserved)
|
45
|
|
|
PART
II
|
|||
|
Item 5.
|
Market
for Registrant’s Common Equity, Related Stockholder Matters and Issuer
Purchases of Equity Securities
|
45
|
|
|
Item 6.
|
Selected
Financial Data
|
47
|
|
|
Item 7.
|
Management’s
Discussion and Analysis of Financial Condition and Results of
Operations
|
48
|
|
|
Item 7A.
|
Quantitative
and Qualitative Disclosures About Market Risk
|
54
|
|
|
Item 8.
|
Financial
Statements and Supplementary Data
|
54
|
|
|
Item 9.
|
Changes
in and Disagreements with Accountants on Accounting and Financial
Disclosure
|
75
|
|
|
Item 9A.
|
Controls
and Procedures
|
75
|
|
|
Item 9B.
|
Other
Information
|
76
|
|
|
PART
III
|
|||
|
Item 10.
|
Directors,
Executive Officers and Corporate Governance
|
76
|
|
|
Item 11.
|
Executive
Compensation
|
76
|
|
|
Item 12.
|
Security
Ownership of Certain Beneficial Owners and Management and Related
Stockholder Matters
|
76
|
|
|
Item 13.
|
Certain
Relationships and Related Transactions and Director
Independence
|
76
|
|
|
Item 14.
|
Principal
Accounting Fees and Services
|
76
|
|
|
PART
IV
|
|||
|
Item 15.
|
Exhibits,
Financial Statement Schedules
|
77
|
|
2
PART
I
NOTE
REGARDING FORWARD-LOOKING STATEMENTS
This
Annual Report on Form 10-K contains forward-looking statements within the
meaning of Section 27A of the Securities Act of 1933, as amended, and Section
21E of the Securities Exchange Act of 1934, as amended, that relate to future
events or our future financial performance. You can identify
forward-looking statements by terminology such as “may,” “might,” “will,”
“could,” “should,” “would,” “expect,” “plan,” “anticipate,” “believe,”
“estimate,” “predict,” “intend,” “potential” or “continue” or the negative of
these terms or other comparable terminology. Our actual results might
differ materially from any forward-looking statement due to various risks,
uncertainties and contingencies, including but not limited to those identified
in Item 1A entitled “Risk Factors” beginning on page 31 of this report, as well
as those discussed in our other filings with the Securities and Exchange
Commission (the “SEC”) and the following:
|
|
·
|
our need for, and our ability
to obtain, additional funds;
|
|
|
·
|
our ability to obtain grants
to develop our drug
candidates;
|
|
|
·
|
uncertainties relating to
non-clinical
studies, clinical
trials and regulatory reviews and
approvals;
|
|
|
·
|
uncertainties relating to our
pre-clinical trials and regulatory reviews and
approvals;
|
|
|
·
|
our dependence on a limited
number of therapeutic
compounds;
|
|
|
·
|
the early stage of the
drug
candidates we are
developing;
|
|
|
·
|
the acceptance of any future
products by physicians and
patients;
|
|
|
·
|
competition with and
dependence on collaborative
partners;
|
|
|
·
|
loss of key consultants,
management or scientific
personnel;
|
|
|
·
|
our ability to obtain adequate
intellectual property protection and to enforce these rights;
and
|
|
|
·
|
our ability to avoid
infringement of the intellectual property rights of
others.
|
Although
we believe that the expectations reflected in the forward-looking statements are
reasonable, we cannot guarantee future results, levels of activity, performance
or achievements. We disclaim any intention or obligation to update or
revise any forward-looking statements, whether as a result of new information,
future events or otherwise.
Item
1. Business.
General
Overview
Aeolus
Pharmaceuticals, Inc. (“we,” “us” or the “Company”), a Southern California-based
biopharmaceutical company, is developing a new class of broad spectrum catalytic
antioxidant compounds based on technology discovered at Duke University and
National Jewish Health.
Our lead
compound, AEOL 10150, is entering human clinical trials in oncology, where it
will be used in combination with radiation therapy. AEOL 10150 has
previously been tested in two Phase I clinical trials with no serious adverse
events reported. The compound is also being developed as a medical
countermeasure against the pulmonary sub-syndrome of acute radiation syndrome
(“Pulmonary Acute Radiation Syndrome” or “Lung-ARS”) as well as the
gastrointestinal sub-syndrome of acute radiation syndrome (“GI-ARS”),
both caused by exposure to high levels of radiation due to a radiological
or nuclear event. It is also being developed for use as a
countermeasure for exposure to chemical vesicants such as chlorine gas and
sulfur mustard gas. AEOL 10150 has already performed well in animal
efficacy and safety studies in each of these potential indications. A
significant portion of the funding for the medical countermeasure development
programs to date has come from various government entities. Although
our management expects this funding to continue, there is no guarantee that it
will do so.
We were
incorporated in the State of Delaware in 1994. Our common stock
trades on the OTC Bulletin Board under the symbol “AOLS.” Our
principal executive offices are located at 26361 Crown Valley Parkway, Suite
150 Mission Viejo, California 92691, and our phone number at that address
is (949) 481-9825. Our website address is
www.aeoluspharma.com. However, the information on, or that can be
accessed through, our website is not part of this report. We also
make available free of charge through our website our most recent annual report
on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K, and
any amendments to those reports, as soon as reasonably practicable after such
material is electronically filed with or furnished to the SEC.
Strategy
Our
strategy is to use non-dilutive capital wherever possible to develop our
exciting platform of broad-spectrum catalytic antioxidant compounds in important
unmet indications of national strategic importance. We plan to continue to
leverage that capital, like the investments made by U.S. government agencies,
such as The National Institute of Allergy and Infectious Diseases (“NIAID”) and
National Institutes of Health’s (“NIH”) Countermeasures Against Chemical Threats
(“CounterACT”), in AEOL 10150 as a medical countermeasure, to concurrently
develop these promising compounds for use in significant unmet medical
indications, like oncology. We are currently doing this with AEOL 10150, where
we are leveraging the potential substantial government investment in research
and development of AEOL 10150 as a medical countermeasure to develop the
compound in oncology indications, where it would be used in combination with
radiation therapy.
3
Business
Overview
We
are developing a platform of a new class of broad-spectrum catalytic
antioxidant compounds based on technology discovered at Duke University and
National Jewish Health. These compounds, known as metalloporphyrins, scavenge
reactive oxygen species (“ROS”) at the cellular level, mimicking the effect of
the body’s own natural antioxidant enzyme superoxide dismutase (“SOD”).
While the benefits of antioxidants in reducing oxidative stress are
well-known, research with our compounds indicates that metalloporphyrins can be
used to affect signaling via ROS at the cellular level. In addition, there
is evidence that high-levels of ROS can affect gene expression and this may be
modulated through the use of metalloporphyrins. We believe this could have a
profound beneficial impact on people who have been exposed, or are about to be
exposed, to high-doses of radiation.
Our lead
compound, AEOL 10150, is a metalloporphyrin specifically designed to neutralize
reactive oxygen and nitrogen species. The neutralization of these species
reduces oxidative stress, inflammation, and subsequent tissue damage-signaling
cascades resulting from radiation exposure. We are leveraging the
significant investment made by U.S. government agencies to develop this
promising compound for use in oncology indications, where it would be used in
combination with radiation therapy, and is currently in development for use as
both a therapeutic and prophylactic drug. Data has already been
published showing that AEOL 10150 does not interfere with the therapeutic
benefit of radiation therapy in prostate and lung cancer preclinical
studies. Early next year we expect to release data showing the drug’s
impact, if any, when used in combination with radiation and/or
chemotherapy. In mid-2011 we expect to begin Phase I/II studies in
non-small cell lung cancer (“NSCLC”). Later in 2011 we are expecting to begin a
Phase I/II study in Mesothelioma, where approximately 30% of the patients
succumb to the effects of the radiation therapy.
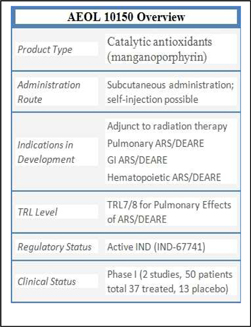
AEOL
10150 is also currently at Technical Readiness Level (“TRL”) 7 as a medical
countermeasure (“MCM”) for GI-ARS and Lung-ARS, both of which are caused by
exposure to high levels of radiation due to a radiological or nuclear
event. To date, this development program has largely been funded by us
through programs at the University of Maryland and Duke University and NIAID and
NIH CounterACT.
In
December 2009, we were informed by the Biomedical Advanced Research and
Development Authority (“BARDA”) that we had been chosen to submit a full
proposal for funding of our Lung-ARS program from its current stage all the way
through U.S. Food and Drug Administration (“FDA”) approval, based on a
summary “white paper” submitted by us earlier in 2009. We submitted
our full proposal in February 2010. We were notified in July 2010
that our proposal had been chosen by BARDA, and then entered into negotiations
for a development contract with the agency. We are awaiting a final
decision from BARDA on the contract. If the contract from BARDA is
awarded, it is expected that it will fund all of the costs to bring AEOL 10150
to FDA approval for that indication.
NIAID’s Radiation/Nuclear
Medical Countermeasures development program is currently testing AEOL 10150 as a
countermeasure for GI-ARS caused by exposure to high levels of radiation due to
a radiological or nuclear event. Similarly, the NIH‘s CounterACT has
tested, and continues to test, AEOL 10150 as a medical countermeasure for
exposure to chemical vesicants such as chlorine gas and mustard gas. In
September 2010, BARDA invited us to submit a full proposal in response to our
“White Paper” for the development of AEOL 10150 as an MCM to chlorine gas
exposure. The proposal seeks funding to take the compound from its
current state to FDA approval over a three year period. We submitted
our full proposal to BARDA in December 2010, and expect a response from BARDA by
the third quarter of 2011.
AEOL
10150 has already performed well in animal safety studies, been well-tolerated
in two human clinical trials, demonstrated efficacy in two species in acute
radiation syndrome (“ARS”) studies and demonstrated statistically significant
survival efficacy in an acute radiation-induced lung injury model. AEOL
10150 has also demonstrated efficacy in validated animal models for GI-ARS,
chlorine gas exposure, and sulfur mustard gas exposure. Efficacy has been
demonstrated in Lung-ARS in both rodent and non-human primate studies (“NHP”),
with AEOL 10150 treated groups showing significantly reduced weight loss,
inflammation, oxidative stress, lung damage, and most importantly,
mortality. Therapeutic efficacy was demonstrated when delivered after
exposure to radiation (24 hours after exposure for mice in the GI-ARS study and
NHPs in the Lung-ARS studies, and two hours after exposure for mice in the
Lung-ARS studies). We are looking at a longer post exposure period in studies
that will begin in early 2011.
We have
an active Investigational New Drug Application (“IND”) on file with the FDA for
AEOL 10150 as a potential treatment for amyotrophic lateral sclerosis (“ALS”).
In the first half of 2011 we plan to file an IND with the oncology division of
the FDA. Extensive toxicology and pharmacology packages are already
in place. We have already completed two Phase 1 safety studies in 50 humans
demonstrating the drug to be safe and well tolerated. Chemistry, Manufacturing,
and Controls (“CMC”) work has been completed, and pilot lots have been prepared
for scaling-up.
We have
two programs underway for the development of our second drug candidate, AEOL
11207, for the treatment of epilepsy and Parkinson’s
disease. These programs are being funded, in part, by private
foundations and government grants.
4
Aeolus’
Catalytic Antioxidant Program
Figure
1
The
findings of research on natural antioxidant enzymes and antioxidant scavengers
support the concept of antioxidants as a broad new class of therapeutic drugs,
if certain limitations noted below could be overcome. We established
our research and development program to explore and exploit the therapeutic
potential of small molecule catalytic antioxidants (Figure 1). We
have achieved our initial research objectives and have begun to extend our
preclinical accomplishments into non-clinical studies, clinical trials and drug
development programs.
Our
catalytic antioxidant program is designed to:
|
|
·
|
Retain
the catalytic mechanism and high antioxidant efficiency of the natural
enzymes, and
|
|
|
·
|
Create
and develop stable and small molecule antioxidants without the limitations
of SOD so that they:
|
|
|
ü
|
Have
broader antioxidant activity,
|
|
|
ü
|
Have
better tissue penetration,
|
|
|
ü
|
Have
a longer life in the body, and
|
|
|
ü
|
Are
not proteins, which are more difficult and expensive to
manufacture.
|
We have
created a class of small molecules that consume reactive oxygen and nitrogen
species catalytically; that is, these molecules are not themselves consumed in
the reaction. Our class of compounds is a group of manganoporphyrins
(an anti-oxidant containing manganese) that retain the benefits of antioxidant
enzymes, are active in animal models of disease and, unlike the body’s own
enzymes, have properties that make them suitable drug development
candidates.
Our most
advanced compound, AEOL 10150 (Figure 1), is a small molecule, broad-based
catalytic antioxidant that has shown the ability to scavenge a broad range of
reactive oxygen species, or free radicals. As a catalytic
antioxidant, AEOL 10150 mimics and thereby amplifies the body’s natural
enzymatic systems for eliminating these damaging compounds. Because
oxygen and nitrogen-derived reactive species are believed to have an important
role in the pathogenesis of many diseases, we believe that our catalytic
antioxidants and AEOL 10150 may have a broad range of potential therapeutic
uses.
AEOL
10150 has shown efficacy in a variety of animal models, including sulfur mustard
gas exposure, as a protectant against radiation exposure, ALS, stroke, radiation
injury, pulmonary diseases, and diabetes. We filed an IND for AEOL
10150 in April 2004, under which clinical trials were conducted as more fully
described below under the heading “AEOL 10150 in Amyotrophic Lateral
Sclerosis.” In 2011 we plan to file an IND with the oncology division
of the FDA. For a more detailed description of antioxidants see the
section below under the heading “Background on Antioxidants.”
AEOL 10150 in Radiation
Therapy
Overview
According
to the American Cancer Society, cancer is the second leading cause of death by
disease, representing one out of every four deaths in the United States with an
approximate 569,000 Americans expected to die of cancer in 2010. In
2010, more than 1.5 million new cancer cases were expected to be diagnosed in
the United States. According to the Radiological Society of North
America, about 50 to 60 percent of cancer patients are treated with radiation at
some time during their disease. The NIH estimates overall costs of
cancer in 2008 in the United States at $228.1 billion, $93.2 billion for direct
medical costs, $18.8 billion for indirect morbidity costs (costs of lost
productivity due to illness) and $116.1 billion for indirect mortality costs
(cost of lost productivity due to premature death).
Combinations
of surgery, chemotherapy and radiation treatments are the mainstay of modern
cancer therapy. Success is often determined by the ability of patients to
tolerate the most aggressive, and most effective, treatment regimens. Radiation
therapy-induced toxicity remains a major factor limiting radiation doses. The
ability to deliver maximal radiation doses for treatment of tumors without
injury to surrounding normal tissue has important implications in oncology
therapeutic outcomes because higher doses of radiation therapy may improve both
local tumor control and patient survival.
Advances
in the tools of molecular and cellular biology have enabled researchers to
develop a better understanding of the underlying mechanisms responsible for
radiation therapy-induced normal tissue injury. For decades ionizing
radiation has been known to increase production of free radicals, which is
reflected by the accumulation of oxidatively damaged cellular
macromolecules.
5
As one
example of radiation-induced damage to adjacent normal tissue, radiation therapy
may injure pulmonary tissue either directly via generation of ROS or indirectly
via the action on parenchymal and inflammatory cells through biological
mediators such as transforming growth factor beta (“TGF B”) and pro-inflammatory
cytokines. Since the discovery of SOD, it has become clear that these
enzymes provide an essential line of defense against ROS. SODs and
SOD mimics, such as AEOL 10150, act by catalyzing the degradation of superoxide
radicals into oxygen and hydrogen peroxide. SODs are localized
intra/extracellularly, are widely expressed throughout the body, and are
important in maintenance of redox status (the balance between oxidation and
reduction). Previous studies have demonstrated that treating
irradiated animal models with SOD delivered by injection of the enzyme through
liposome/viral-mediated gene therapy or insertion of human SOD gene can
ameliorate radiation therapy-induced damage. For an illustrative
example of the radiation therapy reaction see Figure 2.
Figure
2
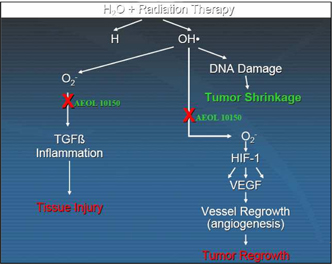
Figure 2 above shows the dual
mechanism of action of radiation therapy and the application of AEOL 10150 to
the process.
In vitro
studies have demonstrated that AEOL 10150 reduces the formation of lipid
peroxides and that it inactivates biologically important ROS molecules such
as superoxide, hydrogen peroxide and peroxynitrite. AEOL
10150 inactivates these ROS by one or two electron oxidation or reduction
reactions in which the oxidation state of the manganese moiety in AEOL
10150 changes. AEOL 10150 is not consumed in the reaction and it
continues to inactivate such ROS molecules as long as it is present at the
target site.
Pre-
clinical studies
Figure
3
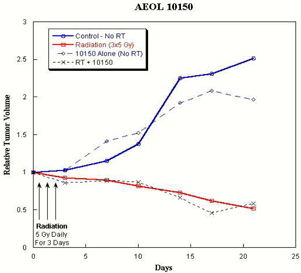
Figure 3. Relative tumor volumes of
human prostate tumor implants in nude mice: Implants of well-vascularized PC3
tumors were grown to substantial size prior to receiving fractionated radiation
(5 Gy daily for three days). AEOL 10150 (7.5 mg/kg/bid) was
administered subcutaneously commencing on the first day of irradiation and
continued for 20 days. Other groups of mice received either no
irradiation, irradiation only or AEOL 10150 without irradiation.
Due to
the similar mechanisms of actions between radiation therapy (in oncology) and
radiation exposure (from nuclear events), we believe that the pre-clinical
studies performed for the development of AEOL 10150 as a potential medical
countermeasure against the effects of Lung-ARS, as described below, also provide
support for the development of AEOL 10150 in oncology, to be used in combination
with radiation therapy.
We have
performed several additional studies specifically for this indication to ensure
the use of an antioxidant in radioprotection of normal adjacent tissue does not
interfere with the efficacy of tumor radiotherapy. A number of
preclinical, in vivo studies have addressed this issue and have demonstrated
that AEOL 10150 does not negatively affect tumor radiotherapy.
In one
study (Vujaskovic, et al. of Duke University), human prostate tumors
(PC3) grown in nude mice to substantial size were fraction irradiated with
5 Gy per day for 3 days for a total of 15 Gy. AEOL 10150 at 7.5
mg/kg/bid was administered subcutaneously on the first day of radiation and
continued for either of two time courses: when tumor volume reached 5 times
the initial volume or for twenty days. The receding tumor volume
curves for irradiation only and for irradiation plus AEOL 10150 were
super-imposable. Therefore AEOL 10150 did not interfere with the
radiation effect on xenogenic prostate tumor.
In
another study of prostate cancer tumors (Gridley, et al of Loma Linda
University), mouse prostate cancer cell line RM-9 was injected
subcutaneously into C57/Bl6 mice, followed by up to 16 days of AEOL 10150
delivered intraperitonealy at 6 mg/kg/day. On day seven, a
single non-fractionated dose of radiation (10 Gy) was
delivered. Therefore, the mice received compound for seven days
prior to radiation. The results of this study demonstrated that
AEOL 10150 does not protect the prostate tumor against radiation, and,
in fact, AEOL 10150 showed a trend towards increasing the effectiveness of
the radiation treatment. The primary effect appears to be in
down-regulation of radiation induced HIF-1 expression and VEGF and
up-regulation of IL-4. Thus, AEOL 10150, through its down-regulation
of VEGF, may inhibit formation of blood vessels (i.e., angiogenisis) required
for tumor re-growth and protects normal tissues from damage induced by radiation
and chemotherapy.
6
Figure
4
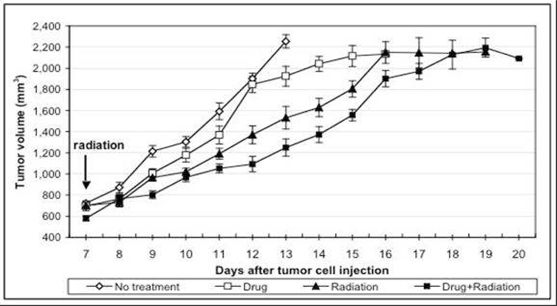
Figure 4 above measures tumor volume
against time after implantation of RM-9 tumor cells and shows that AEOL 10150
treatment resulted in inhibition of tumor re-growth in a study performed by Dr.
Gridley of Loma Linda University. Daily intraperitoneal injections of
AEOL 10150 were initiated on day 1. At 12 days, approximately one
half of each tumor-bearing group and control mice with no tumor were euthanized
for in vitro analyses; remaining mice/group were followed for tumor growth and
euthanized individually when maximum allowed tumor volume was
attained. Each point represents the mean +/- standard error of the
mean. Two-way analysis of the variance for days 8 to 14 revealed that
group and time had highly significant main effects (Ps<0.001) and a group x
time interaction was noted (P<0.001).
Figure
5
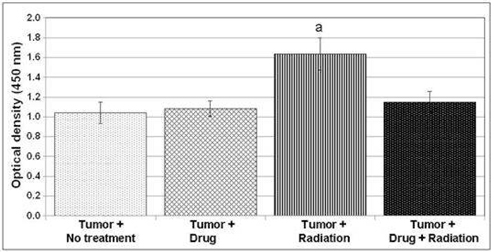
Figure5 above shows the HIF-1
Expression in prostate tumors and the impact of the treatment of AEOL 10150 in a
study by Dr. Gridley of Loma Linda University.
In summary, the data obtained
in these preclinical studies suggest that the post-irradiation, long-term
delivery of AEOL 10150 may be protective against radiation-induced lung injury,
as assessed by histopathology and immunohistochemistry. Oxidative stress,
inflammation and hypoxia, which play important roles in the pathogenesis of
radiation mediated fibrosis, were shown to be reduced in animals treated with
higher doses of AEOL 10150. Studies have also shown that AEOL 10150 does not
adversely affect tumor response to radiation therapy. Thus, treatment with AEOL
10150 does not significantly protect tumors from the cell killing effects of
radiation therapy. This combined with other studies that have shown that AEOL
10150 significantly prevents radiation induced normal tissue injury suggests
that AEOL 10150 has the potential to achieve normal tissue protection without
protection of tumor tissue.
7
Future
Development Plans
We plan
to leverage the substantial investment made by various government agencies in
the development of AEOL 10150 as a potential medical countermeasure against the
effects of Lung-ARS, chlorine gas and GI-ARS to develop AEOL 10150 in oncology,
where it will be used in combination with radiation therapy. We
believe we would be able to use many of the non-clinical studies as support for
efficacy and safety as support of a new drug application (“NDA”) filing for
oncology indications.
We expect
to begin a Phase I/II study in non-small cell lung cancer (“NSCLC”), where AEOL
10150 will be used in combination with radiation therapy, in the first half of
2011. Later in 2011 we are expecting to begin a Phase I/II study in
Mesothelioma, where approximately 30% of the patients succumb to the effects of
the radiation therapy.
Competition
There are
currently three drugs approved for the treatment of the side effects of
radiation therapy. We do not believe that any of these drugs directly
competes with AEOL 10150 in terms of mechanism of action or targeted therapeutic
benefit when used in combination with radiation therapy.
Amifostine
(Ethyol®) is approved by the FDA as a radioprotector. Amifostine (Ethyolâ) is marketed by
MedImmune, Inc. for use in reduction of chemotherapy-induced kidney toxicity
associated with repeated administration of cisplatin in patients with advanced
ovarian cancer and radiation-induced xerostomia (damage to the salivary gland)
in patients undergoing post-operative radiation treatment for head and neck
cancer. MedImmune, Inc. is studying Amifostine in other indications of radiation
therapy. KepivanceTM
(palifermin) is marketed by Amgen, Inc. for use in the treatment of
severe oral mucositis (mouth sores) in patients with hematologic (blood) cancers
who are undergoing high-dose chemotherapy followed by bone transplant. Amgen,
Inc. is also studying Kepivance as an antimucositis agent in patients with head
and neck cancer, non-small cell lung cancer and colon cancer. Salagen Tablets
(pilocarpine hydrochloride) is marketed by Eisai Pharmaceuticals in the United
States as a treatment for the symptoms of xerostomia induced by radiation
therapy in head and neck cancer patients. In addition, there are many drugs
under development to treat the side effects of radiation therapy.
Funding
Options
If we are
awarded the above referenced contract with BARDA, we believe that substantially
all of the costs associated with the preclinical, chemistry, manufacturing, and
controls (“CMC”) and toxicology necessary for the oncology indications, plus a
large safety database in humans, will be covered by the contract. We plan
on funding the clinical efficacy development of AEOL 10150 in oncology using
internally generated funds. If the Company is not awarded a contract
with BARDA, then we would need to raise additional capital or partner with
another firm in order to complete the non-clinical programs noted
above.
AEOL 10150 as a potential
medical countermeasure against the effects of acute radiation syndrome in the
lungs
Overview
During
recent years, the threat of nuclear attack on U.S. soil has increased. The
lack of efficient post-exposure treatments for victims experiencing acute
radiation toxicity presents a serious problem should an attack with a
radiological device occur.
Immediately
after exposure, the most critical components of acute radiation syndrome are the
hematopoietic (bone marrow) and early-onset GI-ARS because symptoms begin very
quickly and can be lethal. However, depending on the level and location of
radiation exposure, much of the lethality of both hematopoietic and early-onset
gastrointestinal syndromes are potentially avoidable with proper treatment,
including supportive care (fluids and antibiotics) and Neupogen, leaving
complications to later responding tissues subsequently becoming a major
problem.
In
situations of accidental exposure, it was initially assumed that a whole-body
dose exceeding 10 Gy was inevitably fatal. However, experience with
nuclear accident victims suggests that when patients survive gastrointestinal
and bone marrow syndromes, respiratory failure becomes the major cause of
death. This effect is known as a delayed effect of acute radiation
exposure (“DEARE”).
Research
has shown that damage associated with the exposure to upper half body
irradiation or total body irradiation is an acute, but delayed, onset of
radiation pneumonitis (inflammation of lung tissue) followed by lung fibrosis
(scarring caused by inflammation). The incidence of radiation pneumonitis rises
very steeply at relatively low radiation doses. A nuclear incident is
likely to result in a wide, inhomogeneous distribution of radiation doses to the
body that allows hematological recovery. But a higher exposure to the thorax
leaves open the risk of serious pulmonary complications.
For the
government, interested in saving as many citizens’ lives as possible, it makes
little sense to provide care to allow people to survive the short-term effects
of radiation exposure following an event, to merely have them die several weeks
or months later due to the delayed effects of radiation
exposure.
8
AEOL
10150 has already performed well in animal safety studies, been well-tolerated
in two human clinical trials, demonstrated efficacy in two species in ARS
studies and demonstrated statistically significant survival efficacy in an
acute radiation-induced lung injury model. AEOL 10150 has also demonstrated
efficacy in validated animal models for GI-ARS, chlorine gas exposure, and
sulfur mustard gas exposure. Efficacy has been demonstrated in both Lung-ARS and
DEARE in both rodent and NHP studies, with AEOL 10150 treated groups showing
significantly reduced weight loss, inflammation, oxidative stress, lung damage,
and most important, mortality. Therapeutic efficacy was demonstrated when
delivered after exposure to radiation (24 hours after exposure for mice in the
GI-ARS studies and NHPs in the Lung-ARS studies, and two hours after
exposure for mice in the Lung-ARS studies). We are looking at a longer post
exposure period in studies that will begin in early 2011.
Pre-
clinical studies
Clinical
experience and experience with nuclear accident victims points out that one of
the primary concerns associated with radiation exposure is an acute, but delayed
onset of radiation pneumonitis with an incidence that rises very steeply at
relatively low radiation doses (to 90-percent occurrence at 11 Gray (“Gy”)). To
evaluate AEOL 10150’s ability to mitigate acute radiation-induced lung
injury, mice were exposed to 15 Gy of upper half body irradiation (“UHBI”) and
subsequently treated with AEOL 10150.
In a
study led by Zeljko Vujaskovic, M.D. Ph.D. at Duke University Medical Center,
C57BL/6 female mice were randomized into six groups. Each of the groups was
paired to include irradiated and non-irradiated groups of animals that were
untreated, treated with a low dose (10 mg/kg) of AEOL 10150, or treated with a
high dose of AEOL 10150 (20 mg/kg). Animals received treatments subcutaneously
beginning 2 hours after irradiation (20 and 40 mg/kg initial loading dose,
respectively) followed by a maintenance dose of half the initial dose three
times per week for 4 weeks. Survival, wet lung weights and body weights,
histopathology, and immunohistochemistry were used to assess lung damage.
Results demonstrate that treatment with AEOL 10150 increased survival (Fig. 6),
maintained body weight (Fig. 7), protected lung tissue (Fig. 8 and 9), and
reduced oxidative stress (via DNA and protein oxidation analysis) compared with
untreated irradiated animals.

Figure 6. Kaplan Meier survival curves for
C57BL/6J mice after upper half body irradiation. The survival data displayed that
there were no deaths in the sham-irradiated animals and animals receiving drug
alone. In contrast, 9/20 (45%) of the animals that received 15 Gy UHBI died
during the 6-week follow-up period. Treatment with low/high doses of AEOL 10150
markedly reduced radiation-induced mortality to only 10%
(2/20).
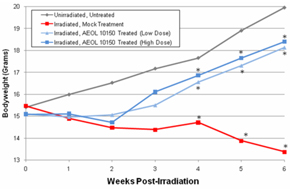
Figure 7: Average body weight changes among
groups. UHBI alone mice
demonstrated significant weight loss beginning 3 weeks post-exposure compared
with UHBI + low/high doses of AEOL 10150 groups.
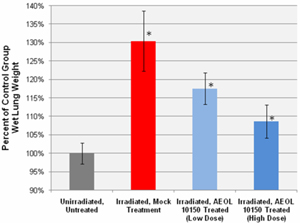
Figure 8. Wet lung weights. Wet lung weights were measured as an
index of pulmonary edema and consolidation. UHBI alone mice had significantly
higher wet lung weights than did the UHBI + low/high doses AEOL10150 groups. *=p
< 0.05
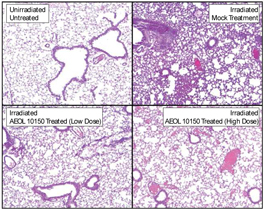
Figure 9. Hematoxylin and Eosin Staining of
Lung Tissue. Lung
histology at 6 weeks revealed a significant decrease in lung structural damage
in UHBI + low/high doses of AEOL 10150 groups, in comparison with UHBI alone.
20x magnification.
9
Data from
a study in which AEOL 10150 was administered to 40 mice that had been exposed to
radiation also show a statistically significant increase in survival rates among
mice that were treated with AEOL 10150 compared to controls. Additionally, mice
receiving AEOL 10150 experienced a reversal in weight loss seen in the untreated
mice. The six month study, led by Zeljko Vujaskovic, M.D. Ph.D. at Duke
University Medical Center, was designed to test the efficacy of AEOL 10150 as a
treatment for damage to the lungs due to exposure to radiation. At 45 days, all
of the animals in the untreated group had either died or been sacrificed based
on animal care rules. The remaining animals that received AEOL 10150 did
not need to be sacrificed based on animal care rules, but a majority were
sacrificed in order to increase the numbers that could be compared to the
untreated animals sacrificed at 45 days, since there would be no untreated
animals for comparison at the end of six months. In addition to the
statistically significant (P< 0.05) survival advantage, statistically
significant differences in body weights and wet lung weights were seen over the
first six weeks of the study. Untreated mice experienced a steady decline in
body weight over the six weeks, while treated animals experienced weight gain
that was just slightly less than that seen in the controls (animals not
receiving radiation). AEOL 10150 also demonstrated statistically significant
reductions in markers for oxidative stress and inflammation – secondary
endpoints for the study.
A number
of other preclinical studies by Zeljko Vujaskovic, MD, PhD; Mitchell Anscher,
MD, et al at Duke University have demonstrated the efficacy of
AEOL 10150 in radioprotection of normal tissue. Chronic administration
of AEOL 10150 by continuous, subcutaneous infusion for 10 weeks has demonstrated
a significant protective effect from radiation-induced lung injury in
rats. Female Fisher 344 rats were randomly divided into four different
dose groups (0, 1, 10 and 30 mg/kg/day of AEOL 10150), receiving either
short-term (one week) or long-term (ten weeks) drug administration via
osmotic pumps. Animals received single dose radiation therapy of 28 Gy
to the right hemithorax. Breathing rates, body weights, histopathology
and immunohistochemistry were used to assess lung damage. For the long
term administration, functional determinants of lung damage 20 weeks
post-radiation were significantly decreased by AEOL 10150. Lung
histology at 20 weeks revealed a significant decrease in structural damage
and fibrosis. Immunohistochemistry demonstrated a significant reduction
in macrophage accumulation, collagen deposition and fibrosis, oxidative
stress and hypoxia in animals receiving radiation therapy along with
AEOL 10150. Figure
10 above shows a
semi-quantitative analyses of lung histology at 20 weeks which revealed a
significant decrease in structural damage and its severity in animals
receiving 10 and 30 mg/kg/day after radiation in comparison to radiation
therapy along with placebo group or radiation therapy along with 1 mg/kg of AEOL
10150 (p = 0.01).
Figure
10
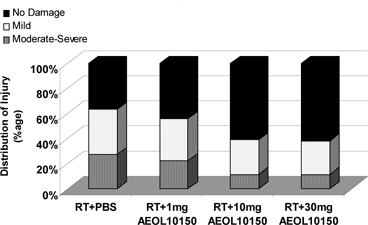
Figure 10 above shows that AEOL 10150 treatment
decreases the severity of damage and increases the percentage of lung tissue
with no damage from radiation therapy (Rabbani et al Int J Rad Oncol Biol Phys
67:573-80, 2007).
Two
additional studies examining the effect of subcutaneous injections of AEOL 10150
on radiation-induced lung injury in rats have been completed. The
compound was administered subcutaneously by a b.i.d. dosing regimen (i.e., 2.5 mg/kg or 5.0
mg/kg) on the first day of radiation and daily for five consecutive
weeks. Radiation was fractionated rather than single dose, with 40 Gy
divided in five 8 Gy doses. Preliminary immunohistologic analyses of
the lung tissue from these two studies showed a
dose dependent decrease in the inflammatory response quantified by the
number of activated macrophages or areas of cell damage. These in vivo
studies employing subcutaneous administration of AEOL 10150, either
by continuous infusion via osmotic pump or BID injection, demonstrate that
AEOL 10150 protects healthy lung tissue from radiation injury delivered
either in a single dose or by fractionated radiation therapy
doses. AEOL 10150 mediates its protective effect(s) by inhibiting a
number of events in the inflammatory cascade induced by radiation
damage.
Additional
in vivo studies have been performed that provide support for
manganoporphyrin antioxidant protection of lung tissue from
radiation. Treatment with a related manganoporphyrin compound, AEOL
10113 significantly improved pulmonary function, decreased histopathologic
markers of lung fibrosis, decreased collagen (hydroxyproline) content,
plasma levels of the profibrogenic cytokine, transforming growth factor beta (TGF-β) and,
as demonstrated by immunohistochemistry of
lung tissue, collagen deposition and TGF-β.
In summary, AEOL 10150 has
consistently shown a protective effect against the harmful effects in radiation,
including when the drug is administered up to 72 hours after
exposure.
10
During
fiscal year 2010, we initiated another study in mice to determine the optimal
length of treatment with AEOL 10150 when used as an MCM to Lung-ARS. This study,
led by Zeljko Vujaskovic, M.D. Ph.D. at Duke University, was designed to build
on the previously completed study that demonstrated the efficacy of AEOL 10150
as a treatment for damage to the lungs due to exposure to radiation (described
in detail above), and
determine the most effective duration of delivery for treatment after exposure.
The results from the study showed that treatment for 4 to 10 weeks after
exposure appears to be optimal. Under the BARDA contract, if awarded to us,
additional studies will be performed to further refine the timeline and analyze
whether extending treatment beyond 10 weeks would be beneficial. Treatment for
4, 6 and 10 weeks showed the greatest impact on body weight and lung damage as
shown in figures 11 and 12 below.
|
Figure
11
|
|
Figure
12
|
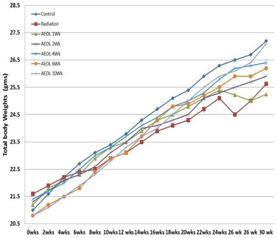 |
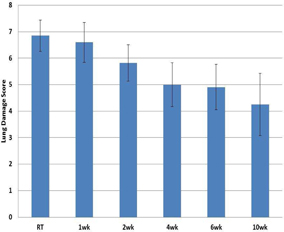 |
Non-
clinical studies
In 2010,
we initiated a study to confirm the efficacy of AEOL 10150 as an MCM to nuclear
and radiological exposure in non-human primates. The study was designed to test
the efficacy of AEOL 10150 as a treatment for Lung-ARS and to begin establishing
an animal model that can be validated and could be utilized by the FDA for
approval of an MCM for Pulmonary Acute Radiation Syndrome under the “Animal
Rule”. The FDA “Animal Rule” enumerates criteria whereby the FDA can rely on
animal efficacy data when “evidence is needed to demonstrate efficacy of new
drugs against lethal or permanently disabling toxic substances when efficacy
studies in humans, ethically cannot be conducted.” The criteria are discussed
below.
Preliminary
results from the study were reported during the fiscal year, showing that AEOL
10150 promotes survival in a non-human primate model of Lung-ARS. The primary
objective of the study was to determine if AEOL 10150 could mitigate
radiation-induced lung injury and enhance survival in rhesus macaques exposed to
whole thorax lung irradiation (“WTLI”) and administered supportive care. Two
cohorts of Non-Human Primates (“NHP”) (total n=13) were exposed to 11.5Gy
LINAC-derived photon radiation in the WTLI protocol. The control cohort had n=6
and AEOL-treated cohort was n=7. This model showed 100% incidence of severe
radiation-induced lung damage. AEOL 10150 was administered subcutaneously at
5mg/kg beginning at day 1 post WTLI and continued as a single, daily injection
for 28 consecutive days. While none of the six control animals survived the 180
day study period, 2 of the 7 (or 28.5%) of the animals receiving drug
survived.
All other
clinical parameters are in process of being analyzed and collated. Some of the
key measurements include: respiratory rate, pulse oximetry for SpO2 values and
histological examination (special stains will be used to determine presence and
degree of fibrosis, alveolar space, cellular infiltration and oxidative damage).
Final results of the study are expected in early 2011.
In
rodents, non-human primates and humans, radiation of the lungs can cause reduced
breathing capacity, pneumonitis, fibrosis, weight loss and death and is
characterized by oxidative stress, inflammation and elevated macrophage counts.
AEOL 10150 has proven to be an effective countermeasure to radiation exposure of
the lungs in mice and rats in published studies such as Rabbani et al Int J Rad
Oncol Biol Phys 67:573-80, 2007, Rabbani et al Free Rad Res 41:1273-82, 2007 and
Gridley et al Anticancer Res 27:3101-9, 2007.
Clinical
studies
We
believe our two previous phase 1 clinical studies can be utilized in any
potential IND and NDA filing with the FDA for AEOL 10150 as an MCM for ARS. At
this time, we do not have any clinical trials underway but are in the process of
planning additional safety studies.
11
Future
Development Plans
Our
objective is to develop AEOL 10150 as an MCM against ARS pulmonary effects, via
the FDA’s “Animal Rule”. This development pathway requires demonstration of the
key study efficacy parameter of AEOL 10150 treatment in two animal models
relevant to the human radiation response and its treatment, demonstration of
safety in humans, demonstration of relevant dosing and administration in humans,
and clear identification of the mechanism of radiation-induced damage to the
lung and its amelioration by the drug candidate.
AEOL
10150 has several distinct advantages as an MCM, including the
following:
|
|
·
|
Demonstrated
survival increase in animal studies when administered 2 hours after
exposure (P<0.05),
|
|
|
·
|
Demonstrated
reduction in lung fibrosis in animal studies up to 24 hours post exposure
(P<0.05),
|
|
|
·
|
Demonstrated
histological improvement in lung tissue post-radiation
exposure,
|
|
|
·
|
Addresses
an unmet medical need as an MCM to
Lung-ARS,
|
|
|
·
|
Established
safety profile in both clinical and pre-clinical
studies,
|
|
|
·
|
Subcutaneous
self-administration possible by exposed individuals during
emergency,
|
|
|
·
|
Rapid
administration, allowing large numbers of patients to be treated
quickly,
|
|
·
|
Stable
for up to 4½ years at 0–8°C and 1 year at room
temperature,
|
|
·
|
Requires
no non-standard storage conditions (i.e., not
photosensitive),
|
|
·
|
Currently
in development as an adjunct to radiation therapy; if approved will
provide a pre-existing distribution and stockpile resource at oncology
centers in the event of a radiological
emergency,
|
|
|
·
|
Demonstrated
potential as both a therapeutic and
prophylactic,
|
|
|
·
|
Demonstrated
potential to address multiple sub-syndromes of
ARS,
|
|
|
·
|
Demonstrated
potential to address sulfur mustard gas and chlorine gas exposure,
and
|
|
|
·
|
Potential
dual use as an adjunct treatment for cancer patients receiving radiation
therapy.
|
We
believe that in order to file an NDA for ARS with the FDA, we will need to
demonstrate efficacy in animal models and demonstrate product safety which is
based upon the FDA’s “Animal Rule”. We also plan on pursuing Fast Track
submission status for this indication, enabling rolling NDA submission
process and a key step in achieving Priority Review, if accepted by the FDA. The
FDA determines within 45 days of a company’s request, made once the complete NDA
is submitted, whether a Priority or Standard Review designation
will be assigned.
The FDA’s
“Animal Rule” enumerates criteria whereby the FDA can rely on animal efficacy
data when evidence is needed to demonstrate efficacy of new drugs against lethal
or permanently disabling toxic substances when efficacy studies in humans cannot
be ethically conducted. The criteria are as follows:
|
|
·
|
Knowledge
of the mechanism of radiation-induced damage to the lung and its
amelioration by the candidate drug.
|
|
|
·
|
Pharmacokinetic
and pharmacodynamic analysis to provide information on relevant dose and
administration schedule.
|
|
|
·
|
Direct
correlation of key study parameters (e.g., survival or major
morbidity) with the desired clinical benefit in
humans.
|
|
|
·
|
Collection
of efficacy data in two species relevant to the human radiation response
and its treatment unless otherwise justified under GLP-compliant
conditions.
|
|
|
·
|
A
Phase 1 safety trial using the same product and formulation as used in the
pivotal trial(s) required.
|
Demonstrate
Efficacy in Animal Models
Our
efficacy plan is designed to accomplish two key goals: the validation of two
animal models for acute radiation-induced lung injury and the generation of
pivotal efficacy data in these species. The efficacy data produced in pivotal
studies using these validated models will provide the data required to
demonstrate efficacy of AEOL 10150 at the dose and schedule proposed for
licensure. A second criterion of the “Animal Rule” is that the models must be
reflective of “real world” conditions to which a human is likely to be exposed.
The proposed models have been designed to reflect these real world conditions.
Initial studies have been conducted with whole thorax exposure models to
irradiate the total lung parenchym, and will be followed by studies with Total
Body Irradiation (“TBI”) with shielding of roughly 5 percent of bone marrow.
This study design mimics real world conditions in which it is anticipated that
many of those exposed to radiation will benefit from some shielding (e.g., from cars, buildings,
etc.), which will protect some bone marrow and allow for survival without a bone
marrow transplant. This shielding approach has been used to develop both murine
and NHP models for GI-ARS and in the NHP models for radiation-induced lung
injury.
Demonstrate
Product Safety
For
product approval under the Animal Rule, we will also demonstrate product safety
using the same product and formulation used in the animal efficacy trials and
proposed for use in humans. Demonstration of safety includes preclinical
demonstration of safety via the standard pre-clinical studies and analyses
methods and Phase I safety trials sufficient to demonstrate product safety in
the target patient population. We believe our safety studies completed as a
therapy for ALS may be utilized to demonstrate safety for this indication. As a
result, we believe it may only be necessary to conduct mutagenicity and
teratogenicity studies for completion of the pre-clinical package required for
NDA filing and product licensure. We also plan to conduct two additional
Phase I clinical safety studies.
12
Competition
Currently
there are no FDA-approved drugs for the treatment of Lung-ARS. We are also not
aware of any other drug candidates that have demonstrated the ability to protect
the lungs from radiation given post exposure, which we believe is a critical
aspect of the development of an MCM against the effects of acute radiation
syndrome. Since the government is interested in saving as many citizens’ lives
as possible, it makes little sense to provide care to allow people to survive
the short-term effects of radiation exposure following an event, only to have
them die several weeks or months later due to the delayed effects of radiation
exposure.
However,
in general, we face significant competition for U.S. government funding for both
development and procurement of an MCM for biological, chemical and nuclear
threats, diagnostic testing systems and other emergency preparedness
countermeasures. The U.S. federal government has currently allocated a
significant amount of research funding to the development of countermeasures
against the effects of radiation exposure. As a result, there are many drug
candidates under development as a possible countermeasure against the effects of
radiation exposure.
Funding
Options
In
October 2010, we were notified that we had been awarded the maximum amount, of
$244,479, under the Qualifying Therapeutic Discovery Grant Program (“QTDP”)
administered by the Internal Revenue Service (“IRS”) and the Department of
Health and Human Services (“HHS”) in support of our development of AEOL 10150 as
an MCM for Lung-ARS.
In
December 2009, we were informed by BARDA that we had been chosen to submit a
full proposal for funding of our Lung-ARS program from its current stage all the
way through FDA approval, based on a summary “white paper” submitted by us
earlier in 2009. We submitted our full proposal in February 2010. We
were notified in July 2010 that our proposal had been chosen by BARDA, and then
entered into negotiations for a development contract with the agency. We
are awaiting a final decision from BARDA on the contract. If the contract
from BARDA is awarded, it is expected that it will fund all of the costs to
bring AEOL 10150 to FDA approval for that indication.
Absent a
contract with BARDA, the Company would be dependent upon NIH-NIAID or internal
funding for the research and development of AEOL 10150 as a medical
countermeasure for Lung-ARS.
AEOL 10150 as a potential
medical countermeasure against the effects of radiation on the gastro-intestinal
tract
Overview
GI-ARS is
a massive, currently untreatable, problem following high-dose, potentially
lethal radiation exposure. Agents that mitigate these effects would reduce
sickness and hopefully prevent fatalities. The intestinal epithelium, a single
layer of cells lining the surface of the GI lumen, is responsible for vital
functions of nutrient absorption, maintaining fluid and electrolyte balance and
protection of the body from bacteria, bacterial toxins and non-absorbed
materials. The functional integrity of the GI system is maintained via incessant
production of epithelial cells from specialized stem cells located in crypts at
the base of the epithelium. High-dose, total-body irradiation can result in a
lethal GI syndrome that results in significant morbidity and mortality within
days consequent to killing of the crypt stem cells and loss of the protective
and absorptive epithelial barrier. There are no FDA-approved drugs or biologics
to treat GI-ARS.
Pre-clinical
studies
The NIH
NIAID’s Radiation/Nuclear Medical Countermeasures development program is
currently testing AEOL 10150 as a countermeasure for GI-ARS. The studies are
being funded by the NIAID and are designed to test the efficacy of AEOL 10150 as
a treatment for damage to the GI tract due to exposure to radiation. The study
protocols call for the examination of both histological and survival endpoints
in mice in a multi-armed vehicle-controlled trial. For the histological portion,
crypt histology will be assessed with crypt number and crypt width being the
primary endpoint. Animals receiving AEOL 10150 began dosing 24 hours after
radiation exposure and receive one dose per day for the remainder of the study.
Preliminary results have demonstrated that AEOL 10150 can effectively increase
regeneration of GI stem cells, reduce the severity and duration of diarrhea and
improve survival when administered at 24 hours after doses of total-body
irradiation that produce the lethal GI syndrome. The studies are being conducted
by Epistem in compliance with criteria of the FDA that are a pre-requisite for
movement of our drug along the pathway for FDA licensure to treat lethally
irradiated persons in the event of a terrorist nuclear act. Epistem operates a
major contract research organization and provides services to identify novel
drugs that can protect or improve the repair of the GI tract following exposure
to irradiation and performs these studies as part of NIH’s program for the
screening of novel agents for bio-defense applications.
13
At a
development meeting held in the fourth quarter of 2010, the Medical
Countermeasures Against Radiological Threats (“MCART”) reviewed the results of
the two mouse studies that have been conducted with AEOL 10150 to date and
concluded:
|
|
1)
|
AEOL
is biologically active as a countermeasure (specifically for
GI-ARS)
|
|
|
2)
|
Based
on the fact that all of the animals in the control group died, the level
of radiation exposure may be too high for the study, and a lower level of
exposure that generates a mortality rate of 50 to 70 percent may be more
appropriate to examine efficacy.
|
|
|
3)
|
Epistem
will conduct a radiation dose range study with AEOL 10150 in which they
will look at exposing animals to radiation between 9 and 12 Gy and compare
control animals to animals treated with AEOL 10150. The study is expected
to begin in the first quarter of 2011 assuming availability of
mice.
|
|
|
4)
|
The
University of Maryland will initiate a study of AEOL 10150 in NHPs during
the second quarter of 2011. Animals will be exposed to radiation between 9
and 12 Gy and control animals will be compared to those receiving
drug.
|
We are
unaware of any published studies of agents that accomplish this enhanced stem
cell regenerative effect while maintaining GI function and improving survival
when administered post irradiation.
Future
Development Plans
In
collaboration with the NIH NIAID, we are planning additional studies to confirm
the efficacy results demonstrated in the study described above. During the first
quarter of fiscal year 2011, we expect NIH NIAID to initiate a third study of
AEOL 10150 in mice. This study will examine the effects of radiation doses from
9 to 11 Gy on the GI tract, as well as the effect that AEOL 10150 has in
mitigating these effects. Studies examining the effects of 9 to 11 Gy of
radiation on the GI tract in non-human primates are expected to begin during the
second quarter of fiscal year 2011. We also expect to perform additional studies
which could be funded by NIH NIAID to optimize dose and duration of delivery,
and to evaluate the window of opportunity for treatment after
exposure.
Upon
completion of these studies we would need to demonstrate efficacy in animal
models and demonstrate product safety based upon the FDA’s “Animal Rule”. We
also plan on pursuing Fast Track submission status for this
indication, enabling rolling NDA submission process and a key step in
achieving Priority Review, if accepted by the FDA. FDA determines within 45 days
of a company’s request, made once the complete NDA is submitted, whether a Priority or Standard Review designation
will be assigned. Under the “Animal Rule,” we would need to complete pivotal
studies in two species relevant to the human radiation response and its
treatment. We believe that these studies can be completed using existing
validated models for both murine and NHP. This study design would also mimic
real world conditions in which it is anticipated that many of those exposed to
radiation will benefit from some shielding (e.g., from cars, buildings,
etc.), which will protect some bone marrow and allow for survival without a bone
marrow transplant.
We will
also demonstrate product safety using the same product and formulation used in
the animal efficacy trials and proposed for use in humans. Demonstration of
safety includes preclinical demonstration of safety via the standard
pre-clinical studies and analyses methods and Phase I safety trials sufficient
to demonstrate product safety in the target patient population. We believe our
safety studies completed as a therapy for ALS may be utilized to demonstrate
safety for this indication. As a result, we believe it may only be necessary to
conduct mutagenicity and teratogenicity studies for completion of the
pre-clinical package required for NDA filing and product licensure. We also
plan to conduct two additional Phase I clinical safety studies in patients
receiving radiation therapy. This will provide safety data in a population
similar to that for which the final product indication will be targeted (i.e., patients receiving high
radiation doses sufficient to cause radiation-related lung damage) for a longer
period of treatment with our compound. A secondary benefit of using this
population is that prophylactic effects of the product can also be demonstrated;
efficacy measures toward achieving this end will be incorporated into the study
design.
Competition
We are
unaware of any compounds that protect crypt cells and that increase survival
when given to animals exposed to radiation at levels greater than 10 Gys and
given after exposure. There are several companies developing drug candidates
that have shown efficacy when given prior to exposure or at lower levels of
radiation.
However,
in general, we face significant competition for U.S. government funding for both
development and procurement of medical countermeasures for biological, chemical
and nuclear threats, diagnostic testing systems and other emergency preparedness
countermeasures. The U.S. federal government has currently allocated a
significant amount of research funding to the development of countermeasures
against the effects of radiation exposure. As a result, there are many drug
candidates under development as a possible countermeasure against the effects of
radiation exposure.
14
Funding
Options
AEOL
10150 as an MCM for GI-ARS is being tested by the NIH NIAID MCART. The NIH NIAID
MCART development program leads the U.S. effort to develop treatments for
radiation sickness following a nuclear terrorist attack. GI-ARS is a potentially
fatal, currently untreatable, problem following high-dose radiation exposure.
Agents that mitigate these effects could reduce sickness and hopefully prevent
fatalities. There are currently no medications approved by the FDA to treat this
syndrome.
AEOL 10150 as a potential
medical countermeasure against the effects of chlorine gas
Overview
Like
sulfur mustard, chlorine gas is a toxic gas that confers airway injury through
primary oxidative stress and secondary inflammation. Chlorine inhalation
was recently used in terrorist/insurgent attacks on military and civilian
populations, and has caused numerous industrial, transportation, swimming pool,
and household accidents, as well as deaths to members of the U.S. military in
the past. Chlorine gas, also known as bertholite, was first used as a
weapon in World War I. Chlorine gas was also used against the local population
and coalition forces in the first Iraq War in the form of chlorine
bombs.
The
increased risk of a terrorist attack in the United States involving chemical
agents has created new challenges for many departments and agencies across the
federal government. Within the HHS, the NIH is taking a leadership role in
pursuing the development of new and improved medical countermeasures designed to
prevent, diagnose, and treat the conditions caused by potential and existing
chemical agents of terrorism. In addition, many of the same chemicals
posing a threat as terrorist agents may also be released from transportation and
storage facilities by industrial accidents or during a natural
disaster. The NIH has developed a comprehensive Countermeasures Against
Chemical Threats (“CounterACT”) Research Network that includes Research Centers
of Excellence, individual research projects, small business innovation research,
contracts and other programs. The CounterACT network is conducting basic,
translational and clinical research aimed at the discovery and/or identification
of better therapeutic and diagnostic medical countermeasures against chemical
threat agents, and their movement through the regulatory process. The
overarching goal of this research program is to enhance our diagnostic and
treatment response capabilities during an emergency.
Another
critical goal of the CounterACT program is to assist in the development of safe
and effective medical countermeasures designed to prevent, diagnose, and treat
the conditions caused by potential and existing chemical agents of terrorism
which can be added to the Nation’s Strategic National Stockpile
(“SNS”). The SNS is maintained by the Centers for Disease Control and
Prevention (“CDC”). The SNS now contains CHEMPACKS which are located in
secure, environmentally controlled areas throughout the United States available
for rapid distribution in case of emergency. The CDC has established a
diagnostic response network for the detection of nerve agents, mustard, cyanide
and toxic metals. The NIH will continue to research, develop and improve
medical products that include chemical antidotes, drugs to reduce morbidity and
mitigate injury, drugs to reduce secondary chemical exposure and diagnostic
tests and assessment tools to be used in mass casualty situations.
Worldwide,
independent of warfare and chemical terrorism, chlorine is the greatest single
cause of major toxic release incidents (16.Davis DS, Dewolf GB, Ferland KA, et
al. Accidental Release of Air Toxins. Park Ridge, New Jersey: NDC;
1989:6-9.). In the U.S., there are about 5-6,000 exposures per year
resulting in, on average, about one death, 10 major, 400-500 moderate, and
3-4,000 minor adverse outcomes. Like mustard, chlorine causes damage to
upper and lower respiratory tracts. While chlorine is an irritant, its
intermediate water solubility may delay emergence of upper airway symptoms for
several minutes. Aqueous decomposition of chlorine gas forms hydrochloric
acid and hypochlorous acid, itself also a product of inflammation. Cell
injury is thought to result from oxidation of functional groups in cell
components, from tissue formation of hydrochloric acid and hypochlorous acid,
and possibly from formation of other ROS. For treatment of acute exposures
in humans, decontamination, supplemental oxygen, treatment of bronchospasm
and/or laryngospasm, and supportive care are the only accepted therapies, while
use of nebulized sodium bicarbonate and parenteral and/or inhaled steroids
remain quite controversial. No specific beneficial therapies are
available. We expect that AEOL 10150 will decrease airway injury,
inflammation, oxidative damage, hyperreactivity and cell proliferation after
acute chlorine gas inhalation in mice and therefore could be a possible
beneficial therapy for chlorine gas inhalation injury to the
airways.
Pre-clinical
studies
Researchers
from National Jewish Health and McGill University have completed a series of
preliminary studies demonstrating that AEOL 10150 protects lungs from chlorine
gas exposure in mice and rats. The primary objective of these studies was
to determine whether administration of AEOL 10150, after exposure, reduces the
severity of acute lung injury and asthma-like symptoms induced by chlorine
gas. AEOL 10150 was given to mice at a 5 mg/kg subcutaneous dose one hour
after chlorine gas exposure (100 ppm for 5 minutes) and repeated every 6
hours. Twenty-four hours after exposure, lung inflammation was assessed by
changes in BAL cellularity and neutrophil influx. AEOL 10150 significantly
reduced (p<0.05, n=6/group) chlorine gas-induced lung inflammation as
measured by BAL fluid cellularity levels by 40% that appeared to be due to
limiting neutrophil influx. AEOL 10150 also significantly attenuated
(p<0.05, n=6) the degree of asthma-like airway reactivity induced by chlorine
gas exposure by 40%. These results indicate that AEOL 10150 can attenuate lung
injury and asthma-like symptoms from chlorine gas exposure and may provide an
effective countermeasure against chlorine gas-induced lung
injury.
15
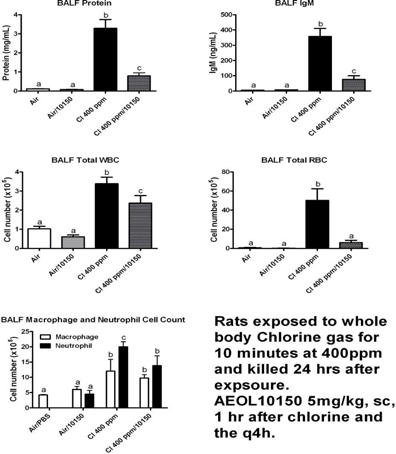
National
Jewish Health replicated the mice studies previously conducted by McGill
University in rats to determine whether AEOL 10150 mitigates lung damage due to
chlorine gas exposure. In the study, 10150 significantly reduced protein, IgM,
white blood cell, red blood cell, macrophage and neutrophil counts in
Broncho-alveolar lavage fluid.
National
Jewish Health’s research is funded by NIH CounterACT and the institute plans to
next run studies to determine whether the initiation of treatment can be
delayed. Additionally, studies will be run to examine the longer term effect of
chlorine gas exposure and AEOL 10150’s ability to mitigate those
effects.
Future
Development Plans
Following
these confirmatory studies, we seek to launch the two pivotal efficacy studies
required for approval by the FDA under the “Animal Rule” as well as complete the
necessary safety studies as further described under the heading AEOL 10150 as a potential medical countermeasure
against the effects of acute radiation syndrome in the lungs – Future
Development Plans – Demonstrate Product Safety.
Competition
There are
currently no effective treatments for chlorine gas exposure and AEOL 10150 is a
major focus of the NIH CounterACT program to identify an effective
treatment.
However,
in general, we face significant competition for U.S. government funding for both
development and procurement of MCMs for biological, chemical and nuclear
threats, diagnostic testing systems and other emergency preparedness
countermeasures. The U.S. federal government has currently allocated a
significant amount of research funding to the development of countermeasures
against bioterrorism. As a result, there are many drug candidates
under development as a possible countermeasure against chemical threat
agents.
Funding
Options
Currently,
the development of AEOL 10150 as a potential MCM against the effects of chlorine
gas is being funded by the NIH CounterACT program.
In May of
2010, we submitted a “White Paper” to BARDA in response to a Broad Agency
Announcement (BAA-BARDA-100-SOL-0012) for advanced research and development of
MCM for chemical, biological, radiological and nuclear threats, seeking
potential countermeasures for chemical agents.
In
September 2010, BARDA invited the Company to submit a full proposal in response
to its “White Paper” for the development of AEOL 10150 as an MCM to chlorine gas
exposure. The proposal seeks funding to take the compound from its current state
to an FDA approval over a four year period. We submitted our full proposal to
BARDA in December 2010, and expect a response from BARDA by the third quarter of
2011.
16
AEOL 10150 as a potential
medical countermeasure against the effects of mustard gas
Overview
Sulfur
mustards, of which mustard gas is a member, are a class of related cytotoxic,
vesicant chemical warfare agents with the ability to form large blisters on
exposed skin and cause pneumonitis and fibrosis in the lungs. In their pure
form most sulfur mustards are colorless, odorless, viscous liquids at room
temperature. When used as warfare agents they are usually yellow-brown in
color and have an odor resembling mustard plants, garlic or
horseradish. Mustard agents, including sulfur mustard, are regulated under
the 1993 Chemical Weapons Convention. Three classes of chemicals are
monitored under this Convention, with sulfur and nitrogen mustard grouped in the
highest risk class, “schedule 1”. However, concerns about its use in a
terrorist attack have led to resurgence in research to develop a protectant
against exposure.
Mustard
gas is a strong vesicant (blister-causing agent). Due to its alkylating
properties, it is also strongly mutagenic (causing damage to the DNA of exposed
cells) and carcinogenic (cancer causing). Those exposed usually suffer no
immediate symptoms. Within 4 to 24 hours the exposure develops into deep,
itching or burning blisters wherever the mustard contacted the skin; the eyes
(if exposed) become sore and the eyelids swollen, possibly leading to
conjunctivitis and blindness. At very high concentrations, if inhaled, it
causes bleeding and blistering within the respiratory system, damaging the
mucous membrane and causing pulmonary edema. Blister agent exposure over
more than 50% body surface area is usually fatal.
The NIH
awarded a five-year, $7.8 million grant to National Jewish Health and the
University of Colorado Health Sciences Center, both in Denver, Colorado. This
Center of Excellence was developed to focus on sulfur mustard toxicity in the
lung and skin and the long-term goal is to develop an effective treatment for
mustard gas induced injury in lung and skin. Members of the Center are
establishing optimal compounds, route and mode of delivery and research projects
are ongoing to determine countermeasures that will help establish specific
interventions needed to treat mustard gas-induced injury. After three years of
research, AEOL 10150 has been identified by the National Jewish Health Center of
Excellence as the lead compound for its center, and research work there has been
focused on further testing and studies of AEOL 10150.
Research
in the area of mustard gas-mediated lung injury has provided experimental
evidence that the mechanisms of these injuries are directly linked to the
formation of reactive oxygen and nitrogen species and that superoxide dismutase
and catalase can improve injury responses. This theory has led to the
hypothesis that the administration of catalytic antioxidant therapy can protect
against mustard gas-induced acute lung and dermal injury. AEOL 10150 has
already been shown to be well tolerated in humans and could be rapidly developed
as a drug candidate in this area pending animal efficacy data.
Researchers
have found that the chemical warfare agent analog, 2-chloroethyl ethyl sulfide
(“CEES”)-induced lung injury could be improved by both exogenous superoxide
dismutase and catalase. Both of these natural enzymes are important
catalytic antioxidants and both of these reactions are exhibited by
metalloporphyrins. CEES-induced lung injury is dependent in part upon blood
neutrophils. Activated neutrophils are an important source of reactive
oxygen species that are known to contribute to lung injury
responses. Antioxidants have also been shown to protect against
CEES-induced dermal injury. Mustard exposure is often associated with
producing acute respiratory distress syndrome (“ARDS”) that requires
supplemental oxygen therapy to maintain adequate tissue
oxygenation.
Pre-clinical
studies
A study
performed by researchers from National Jewish Health demonstrated that AEOL
10150 showed statistically significant protection of lung tissue in animals
exposed to CEES or half-mustard. In a study sponsored by the NIH CounterACT
program, AEOL 10150 was tested along with 19 other compounds to determine
effectiveness in protecting lung tissue against edema and hemorrhage resulting
from exposure to mustard gas.
AEOL
10150 was given to rats one hour after CEES exposure and again 6 hours later.
Eighteen hours after exposure, lung edema and hemorrhage was assessed by changes
in the bronchoalveolar lavage protein and red blood cell levels. AEOL 10150
significantly reduced (p<0.05) mustard gas-induced lung edema and hemorrhage.
These results suggest that AEOL 10150 rescues the lung from mustard gas exposure
and may provide a countermeasure against mustard gas-induced lung injury.
Further studies at National Jewish Health and the University of Colorado showed
that doses in the range of 5 to 30 mg/kg of AEOL 10150 given at one and eight
hours after exposure mitigate both lung and skin injury in animal models. Doses
in the range of 5 to 10 mg/kg/d showed the most potent effect including
significant mitigation as assessed by histopathology and
immunohistochemistry.
Non-clinical
studies
In 2009,
several studies were launched to test the efficacy of AEOL 10150 as a treatment
for damage to the skin and lungs due to exposure to sulfur mustard gas and to
examine potential effective doses, duration of delivery and the window of
opportunity for treatment after exposure. The studies are being conducted
using “whole” sulfur mustard gas at Lovelace Respiratory Research Institute,
another CounterACT Center of Excellence, and using CEES at National Jewish
Health and build on results from previous studies using CEES conducted at
National Jewish Health and the University of Colorado.
The first
whole mustard study was completed in October 2009. The study demonstrated that
AEOL 10150 protects lungs from whole mustard gas exposure in rats. The data
affirmed our earlier studies where AEOL 10150 protected the lung against the
half-mustard, CEES. The primary objective of the studies was to determine
whether administration of AEOL 10150, after exposure, reduces the severity of
acute lung injury induced by mustard gas. AEOL 10150 was given to rats one hour
after sulfur mustard exposure and repeated every 6 hours. Twenty-four hours
after exposure, lung edema was assessed by changes in the bronchoalveolar lavage
(“BAL”) protein levels. AEOL 10150 significantly reduced (p<0.05) mustard
gas-induced lung edema as measured by BAL protein levels. In addition, AEOL
10150 decreased SM-induced increase in the numbers of BAL neutrophils. These
results indicate that AEOL 10150 can attenuate lung injury from mustard gas
exposure and may provide an effective countermeasure against mustard gas-induced
lung injury.
17
In June
2010, National Jewish Health and Lovelace Respiratory Research Institute
reported results from a second whole mustard study confirming that AEOL 10150
protects lungs from whole mustard gas exposure in rats. The primary
objective of this study was to determine whether administration of AEOL 10150,
after exposure, reduces the severity of acute lung injury induced by mustard
gas. AEOL 10150 was given to rats one hour after sulfur mustard vapor
exposure and repeated every 6 hours. Twenty-four hours after exposure, lung
edema was assessed by changes in the bronchoalveolar lavage (BAL) protein
levels. AEOL 10150 significantly reduced (p<0.05) mustard gas-induced lung
edema as measured by bronchoalveolar lavage protein levels. In addition,
AEOL 10150 decreased SM-induced increases in macrophages (p<0.05) and
epithelial cells in BAL fluid (P<0.05). In all three measurements AEOL
10150 provided approximately 100 percent protection – with levels approximating
that of the control animals in the study. These results indicate that AEOL
10150 can attenuate lung injury from mustard gas exposure and may provide an
effective countermeasure against mustard gas-induced lung injury.
Future
Development Plans
Following
these confirmatory studies, we seek to launch the two pivotal efficacy studies
required for approval by the FDA under the “Animal Rule” as well as complete the
necessary safety studies as further described under the heading AEOL 10150 as a potential medical countermeasure
against the effects of acute radiation syndrome in the lungs – Future
Development Plans – Demonstrate Product Safety.
Competition
There are
currently no effective treatments for mustard gas exposure and AEOL 10150 is a
major focus of a sponsored research grant awarded by the NIH CounterAct program
to National Jewish Health to identify an effective treatment.
However,
in general, we face significant competition for U.S. government funding for both
development and procurement of medical countermeasures for biological, chemical
and nuclear threats, diagnostic testing systems and other emergency preparedness
countermeasures. The U.S. federal government has currently allocated a
significant amount of research funding to the development of countermeasures
against bioterrorism. As a result, there are many drug candidates under
development as a possible countermeasure against chemical threat
agents.
Funding
Options
This
development program to date has been funded under the NIH CounterAct Program and
we expect that future efficacy studies necessary for approval by the FDA will
also be funded by the NIH CounterAct program.
AEOL 10150 in Amyotrophic
Lateral Sclerosis
Overview
Amyotrophic
Lateral Sclerosis (“ALS”), commonly referred to as “Lou Gehrig’s disease,” the
most common motor neuron disease, results from progressive degeneration of both
upper and lower motor neurons. Motor Neuron Disease (“MND”) is an all
embracing term used to cover a number of illnesses of the motor neuron. ALS,
Progressive Muscular Atrophy (PMA), Progressive Bulbar Palsy (PBP), Primary
Lateral Sclerosis (PLS) are all subtypes. MND is the generic term for this
disease and is used more frequently in Europe, while ALS is used more frequently
in the U.S.
According
to the ALS Association (“ALSA”), the incidence of ALS is two per 100,000
people. ALS occurs more often in men than women, with typical onset between
40 and 70 years of age. ALS is a progressive disease and approximately 80%
of ALS patients die within five years of diagnosis, with only 10% living more
than 10 years. The average life expectancy is two to five years after
diagnosis, with death from respiratory and/or bulbar muscle failure. The
International Alliance of ALS/MND Associations reports there are over 350,000
patients with ALS/MND worldwide and 100,000 people die from the disease each
year worldwide. In the United States, ALSA reports that there are
approximately 30,000 patients with ALS with 5,600 new patients diagnosed each
year.
Sporadic
(i.e., of unknown
origin) ALS is the most common form, accounting for approximately 90% of
cases. The cause of sporadic ALS is unclear. Familial ALS comprises
the remainder of cases and 5-10% of these patients have a mutated superoxide
dismutase 1 (“SOD1”) gene. More than 90 point mutations have been
identified, all of which appear to associate with ALS, and result in motor
neuron disease in corresponding transgenic mice. SOD mutations have been
observed in both familial and sporadic ALS patients, although the nature of the
dysfunction produced by the SOD1 mutations remains unclear. The clinical
and pathological manifestations of familial ALS and sporadic ALS are
indistinguishable suggesting common pathways in both types of
disease.
18
In
November 2003, the FDA granted orphan drug designation for our ALS drug
candidate. Orphan drug designation qualifies a product for possible funding
to support clinical trials, study design assistance from the FDA during
development and for financial incentives, including seven years of marketing
exclusivity upon FDA approval.
Pre-clinical
studies
John P.
Crow, Ph.D., and his colleagues at the University of Alabama at Birmingham
tested AEOL 10150 in an animal model of ALS (SOD1 mutant G93A transgenic
mice). The experiments conducted by Dr. Crow (now at the University of
Arkansas College of Medicine) were designed to be clinically relevant by
beginning treatment only after the onset of symptoms in the animals is
observed. Twenty-four confirmed transgenic mice were alternately assigned
to either a control group or AEOL 10150-treatment on the day of symptom onset,
which was defined as a noticeable hind-limb weakness. Treatment began on
the day of symptom onset. The initial dose of AEOL 10150 was 5 mg/kg, with
continued treatment at a dose of 2.5 mg/kg once a day until death or near
death.
|
Treatment
|
Age at Symptom
onset mean days
+ SD(range)
|
Survival Interval
mean days +
SD(range)
|
P-value Log-rank (v.
control)
|
P-value
Wilcoxon (v.
control)
|
||||||||||||
|
Control
|
104.8 + 1.43 | 12.8 + 0.79 | ||||||||||||||
| (100-112 | ) | (9-16 | ) | |||||||||||||
|
AEOL 10150
|
106.1 + 1.5 | 32.2 + 2.73 | ||||||||||||||
| (100-115 | ) | (15-46 | ) |
<
0.0001
|
0.0002 | |||||||||||
Table 1. Effect of AEOL 10150 on survival of
G93A transgenic mice
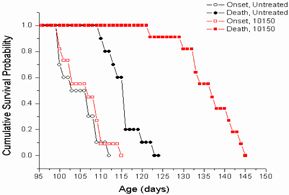
Figure 13.
Table 1
and Figure 13 above show that AEOL 10150 treatment resulted in a greater than
2.5 times mean survival interval, compared to control. AEOL 10150-treated
mice were observed to remain mildly disabled until a day or two before
death. In contrast, control mice experienced increased disability
daily.
Dr. Crow
has repeated the ALS preclinical experiment a total of four times, in each case
with similar results. The efficacy of AEOL 10150 in the G93A mouse model of
ALS has also been evaluated by two additional laboratories. One of these
laboratories verified an effect of AEOL 10150 in prolonging survival of the
G93A mouse, while no beneficial effect of the drug was identified in the other
laboratory.
Future
Development Plans
We do not
currently have any plans to pursue the development of AEOL 10150 for the
treatment of ALS unless we are able to obtain funding specifically for this
purpose.
Competition
Rilutek®
(riluzole), marketed by Sanofi-Aventis SA, is the only commercially approved
treatment for ALS in the United States and the European
Union. Administration of Rilutek prolongs survival of ALS patients by an
average of 60-90 days, but has little or no effect on the progression of muscle
weakness, or quality of life. Rilutek was approved in the United States in
1995, and in 2001 in the European Union. However, there are at least twenty
drug candidates reported to be in clinical development for the treatment of
ALS.
19
In
addition, ALS belongs to a family of diseases called neurodegenerative diseases,
which includes Alzheimer’s, Parkinson’s and Huntington’s disease. Due to
similarities between these diseases, a new treatment for one ailment potentially
could be useful for treating others. There are many companies that are
producing and developing drugs used to treat neurodegenerative diseases other
than ALS.
AEOL
10150 Clinical Development Program
AEOL
10150 has been thoroughly tested for safety, tolerability and pharmacokinetics
with no serious or clinically significant adverse effects observed. To date, 38
patients have received AEOL 10150 in three clinical trials designed to test the
safety and tolerability of the drug candidate.
In
September 2005, we completed a multi-center, double-blind, randomized,
placebo-controlled, Phase I clinical trial. This escalating-dose study was
conducted to evaluate the safety, tolerability and pharmacokinetics of AEOL
10150 administered by twice daily subcutaneous injections in patients with
ALS.
In the
Phase Ia study, 4-5 patients diagnosed with ALS were placed in a dosage cohort
(3 or 4 receiving AEOL 10150 and 1 receiving placebo). Each dose cohort was
evaluated at a separate clinical center. In total, seven separate cohorts
were evaluated in the study, and 25 ALS patients received AEOL 10150. Based
upon an analysis of the data, it was concluded that single doses of AEOL 10150
ranging from 3 mg to 75 mg were safe and well tolerated. In addition, no
serious or clinically significant adverse clinical events were reported, nor
were there any significant laboratory abnormalities. Based upon extensive
cardiovascular monitoring (i.e., frequent
electrocardiograms and continuous Holter recordings for up to 48 hours following
dosing), there were no compound-related cardiovascular
abnormalities.
The most
frequently reported adverse events in this Phase I clinical trial were injection
site reactions, followed by dizziness and headache. Adverse events were
primarily mild in severity, and approximately one-half of the events were
considered to have a possible relationship to the study medication. In
addition, no clinically meaningful findings were noted in the safety,
laboratory, vital sign, the Unified Parkinson’s Disease Rating Scale (“UPDRS”),
functional ALS, or electro cardiogram (“ECG”) data. All cohorts exhibited
dose-related peak plasma drug concentrations and consistent disappearance
half-lives.
In
October 2006, we completed a multi-center, double-blind, randomized,
placebo-controlled, Phase Ib clinical trial. This multiple dose study was
conducted to evaluate the safety, tolerability and pharmacokinetics of AEOL
10150 administered by subcutaneous injection and infusion pump in patients with
ALS. Under the multiple dose protocol, three groups of six ALS patients
(four receiving AEOL 10150 and two receiving placebo) were enrolled, based upon
patients who meet the El Escorial criteria for Clinically Definite ALS,
Clinically Probable ALS, Clinically Probable-Laboratory Supported ALS, or
Definite Familial-Laboratory Supported ALS (i.e., Clinically Possible ALS
with an identified SOD gene mutation).
The first
two cohorts of the Phase Ib multiple dose study received a fixed daily dose of
AEOL 10150 twice a day by subcutaneous injection. In the first cohort, each
patient received twice daily subcutaneous injections of 40 mg of AEOL 10150 or
placebo, for six consecutive days, followed by a single subcutaneous injection
on the seventh day, for a total of 13 injections. In the second cohort,
each patient received twice daily subcutaneous injections of 60 mg of AEOL 10150
or placebo, for six consecutive days, followed by a single subcutaneous
injection on the seventh day, for a total of 13 injections.
In
contrast, the third cohort received a weight adjusted dose (i.e., mg per kilogram of body
weight per day) delivered subcutaneously over twenty four hours by continuous
infusion pump. In the third cohort, each patient received AEOL 10150 via
continuous infusion pump for six and one half consecutive days for a total of
2.0 mg per patient kilogram per day. Each patient in all three cohorts
completed the study and follow-up evaluation at 14 days.
The Phase
Ib study was conducted at five academic clinical ALS centers. Male and
female ALS patients, 18 to 70 years of age, who were ambulatory (with the use of
a walker or cane, if needed) and capable of orthostatic blood pressure
assessments were enrolled in the study. Clinical signs/symptoms, laboratory
values, cardiac assessments and pharmacokinetics (PK) were
performed.
Based
upon an analysis of the data, it was concluded that multiple doses of AEOL 10150
for a period of six and one half consecutive days in the amount of 40 mg per
day, 60 mg per day and 2 mg per kilogram per day were safe and well
tolerated. No serious or clinically significant adverse events were reported or
observed. The most frequent adverse events related to study compound were
injection site observations related to compound delivery. There were no
significant laboratory abnormalities. Based upon extensive cardiovascular
monitoring (i.e.,
frequent electrocardiograms and continuous Holter recordings throughout the six
and one half days of dosing), there were no compound-related cardiovascular
abnormalities.
Pharmacokinetic
findings from the Phase Ib study to data are as follows:
|
|
·
|
Increases
in Cmax and AUC(0-8) appear to correlate with increases in dose, but the
correlation is not strong.
|
|
|
·
|
The
mean Cmax for the 40 mg cohort was 1,735 ng/mL; 2,315 ng/mL for the 60 mg
cohort and 1,653 ng/ml for the 2 mg/kg
cohort.
|
|
|
·
|
There
were probable linear correlations between both Cmax and AUC(0-8) and dose
based on body weight.
|
20
|
|
·
|
The
terminal half- life (a measurement of the time period for which a compound
stays in the body) as determined from Day 7 data was approximately 8 to 9
hours.
|
|
|
·
|
Steady-state
occurs within three days of multiple dosing. There was no evidence
for a third longer half- life that would be associated with long term
accumulation. Thus, compound accumulation is not expected beyond the
third day with multiple dosing.
|
|
|
·
|
From
48 hours to the end of the infusion, the plasma concentrations of AEOL
10150 during the infusion showed little variability, indicating a smoother
delivery of the drug than with twice-daily
injections.
|
During
2008, we completed a follow-on Phase I open label compassionate use multiple
dose study of AEOL 10150 in a patient diagnosed with progressive and
debilitating amyotrophic ALS. The study was conducted at the University of
California, Los Angeles by Martina Wiedau-Pazos, M.D., and was designed to
evaluate the safety and efficacy of AEOL 10150 in an ALS patient over an
extended period of time. The patient received a subcutaneous injection of
75mg of AEOL 10150 two times each day for 34 days. Efficacy and safety data
was monitored for the duration of the study. The primary objective of this
study was to assess the clinical efficacy of AEOL 10150 with respect to the
patient’s baseline assessment of functional status. Secondary objectives
included the assessments of muscle strength, respiratory function, quality of
life and safety. The patent’s baseline efficacy results were an ALS
Functional Rating Scale (“ALSFRS-R”) rating of 19, Muscle strength Manual Muscle
Testing Scale (“MMTS”) of 68 and a forced vital capacity (“FVC”) of 30%. The
patient’s results after 2 months were an ALSFRS-R rating of 22, a MMTS rating of
86 and an FVC of 28%. It should be noted that the subject began using breathing
assistance (BiPAP) approximately two weeks after the study started. The patient
discontinued treatment due to nausea and moderately increased liver
transaminases. Other drug-associated adverse events included mild skin
irritation at the injection site and mild urine discoloration.
AEOL
11207
Overview
We have
selected AEOL 11207 as our second development candidate based upon results from
data obtained from our pre-clinical testing of our pipeline drug
candidates. Because of the wide-ranging therapeutic opportunities that the
compound evidenced in diverse pre-clinical models of human diseases, we have not
yet ascertained what the most robust therapeutic use of AEOL 11207 might
be. However, data collected to date suggest that AEOL 11207 may be useful
as a potential once-every-other-day oral therapeutic treatment option for
central nervous system (“CNS”) disorders, most likely Parkinson’s
disease.
Parkinson’s
disease is a common neurodegenerative disorder, second in occurrence among these
disorders only to Alzheimer’s disease. According to the National Parkinson
Foundation, Parkinson’s affects as many as one million people in the United
States, with approximately 60,000 new cases diagnosed in the United States each
year.
Parkinson’s
specifically involves the progressive destruction of the nerves that secrete
dopamine and control the basal ganglia, an area of the brain involved in the
regulation of movement. Dopamine turnover has been shown to elevate the
levels of ROS in the brain. In addition, a street-drug contaminant has
appeared that can cause parkinsonism in drug abusers. The compound
N-methyl-4-phenyl-1, 2, 3, 6tetrahydropyridine (“MPTP”) has been identified in
underground laboratory preparations of a potent analog of meperidine
(Demerol). MPTP-containing powder, sometimes sold as a new “synthetic
heroin,” can be dissolved in water and administered intravenously or taken by
the intranasal route. MPTP has been documented to produce irreversible
chronic Parkinson symptoms in drug abusers. Agents such as MPTP overproduce
ROS in the basal ganglia. Therefore, ROS mediated neuronal dysfunction may
play a key role in the development of Parkinson’s disease. Symptoms of this
disease include tremors, rigidity and bradykinesia (i.e., slowness of
movement). In the more advanced stages, it can cause fluctuations in motor
function, sleep problems and various neuro-psychiatric disorders. A
biological hallmark of Parkinson’s disease is a reduction in brain dopamine
levels. Preventing or slowing the destruction of brain cells that lead to
the depletion of dopamine levels in the brain is an important therapeutic
approach for the treatment of this disease.
Pre-clinical
studies
Data
developed by our scientists and Dr. Manisha Patel at the University of Colorado
Health Sciences Center and Department of Medicine, indicate that when
administered orally, AEOL 11207 is greater than 80% bioavailable, meaning that
it is readily absorbed and reaches both the circulatory system and the brain in
sufficient amounts to demonstrate biological activity. Data developed with AEOL
11207 in a widely used animal model of Parkinson’s disease (the “MPTP model”)
showed that when administered orally, AEOL 11207 crosses the blood brain barrier
and protected dopamine neurons in a dose-dependent manner. Further data suggest
that the compound has a half- life (a measurement of the time period for which a
compound stays in the body) of about 3 days in both the circulatory system and
the brain, and that prior to stopping administration of the compound, the levels
of AEOL 11207 in both the circulatory system and brain reach a steady state (a
valuable measurement of when the levels of the drug in the body remain
substantially constant, neither increasing nor decreasing) after 2 days of
dosing. Data have also been developed that indicate that when dosing of AEOL
11207 is stopped, the compound is excreted from the body.
In
September 2010, Manisha Patel of the University of Colorado was informed by the
Michael J. Fox Foundation that she had been awarded a supplement to her grant.
The funds are for the synthesis of additional quantities of AEOL1114B and
AEOL11203; for the completion of the evaluation of AEOL1114B and AEOL11203’s
effects on MPTP toxicity (TH+ cells in substantia nigra), and behavioral testing
and accumulation of manganese after chronic dosing.
21
Prior to
receiving the funding for this program, we filed a new composition of matter and
use patent for AEOL 11114B and 11203.
Future
Development Plans
For this
and other reasons, we believe that the therapeutic rationale for developing AEOL
11207 as a neuroprotectant, may substantially change the course of therapeutic
treatment options for Parkinson’s disease if AEOL 11207 were to achieve
regulatory approval for commercialization. However, we are unable to determine
at this time that such regulatory approval for AEOL 11207 can be or will be
secured and we will not be able to further develop AEOL 11207 unless funding for
this purpose is obtained.
AEOL
11207 is patent-protected and has the same chemical core structure as AEOL
10150. Because of this common structural feature, it is anticipated that AEOL
11207 will evidence substantially the same safety profile in clinical
evaluations as observed with AEOL 10150, making clinical trial design and
testing of AEOL 11207 more robust and facile. Furthermore, all of our compounds
evidence the ability to scavenge and decrease ROS and reactive nitrogen species
(RNS), all of which are implicated in a variety of CNS diseases.
Funding
Options
The
University of Colorado, our research provider for the development of AEOL 11207
for the treatment of Parkinson’s Disease, received a grant for funding from the
Michael J. Fox Foundation to further test AEOL 11207 and several other of our
compounds.
In
October 2010, we were notified that we had been awarded the maximum amount, of
$244,479, under the QTDP administered by the IRS and the HHS in support of our
development of AEOL 11207 for Parkinson’s Disease.
Background
on Antioxidants
Oxygen
Stress and Disease
Oxygen
plays a pivotal role in supporting life by enabling energy stored in food to be
converted to energy that living organisms can use. The ability of oxygen to
participate in key metabolic processes derives from its highly reactive
nature. This reactivity is necessary for life, but also generates different
forms of oxygen that can react harmfully with living organisms. In the
body, a small proportion of the oxygen we consume is converted to superoxide, a
free radical species that gives rise to hydrogen peroxide, hydroxyl radical,
peroxynitrite and various other oxidants.
Oxygen-derived
free radicals can damage DNA, proteins and lipids resulting in inflammation and
both acute and delayed cell death. The body protects itself from the
harmful effects of free radicals and other oxidants through multiple antioxidant
enzyme systems such as SOD. These natural antioxidants convert the reactive
molecules into compounds suitable for normal metabolism. When too many free
radicals are produced for the body’s normal defenses to convert, “oxidative
stress” occurs with a cumulative result of reduced cellular function and,
ultimately, disease.
Data also
suggests that oxygen-derived free radicals are an important factor in the
pathogenesis of a large variety of diseases, including neurological disorders
such as ALS, Parkinson’s disease, Alzheimer’s disease and stroke, and in
non-neurological disorders such as cancer radiation therapy damage, emphysema,
asthma and diabetes.
Antioxidants
as Therapeutics
Because
of the role that oxygen-derived free radicals play in disease, scientists are
actively exploring the possible role of antioxidants as a treatment for related
diseases. Preclinical and clinical studies involving treatment with SOD,
the body’s natural antioxidant enzyme, or more recently, studies involving
over-expression of SOD in transgenic animals, have shown promise of therapeutic
benefit in a broad range of disease therapies. Increased SOD function
improves outcome in animal models of conditions including stroke,
ischemia-reperfusion injury (a temporary cutoff of blood supply to tissue) to
various organs, harmful effects of radiation and chemotherapy for the treatment
of cancer, and in neurological and pulmonary diseases. Clinical studies
with bovine SOD, under the brand Orgotein, or recombinant human SOD in several
conditions including arthritis and protection from limiting side effects of
cancer radiation or chemotherapy treatment, have also shown promise of
benefit. The major limitations of enzymatic SOD as a therapeutic are
those found with many proteins, most importantly limited cell penetration and
allergic reactions. Allergic reactions led to the withdrawal of Orgotein
from almost every worldwide market.
22
Catalytic
Antioxidants vs. Antioxidant Scavengers
From a
functional perspective, antioxidant therapeutics can be divided into two broad
categories, scavengers and catalysts. Antioxidant scavengers are
compounds where one antioxidant molecule combines with one reactive oxygen
molecule and both are consumed in the reaction. There is a one-to-one
ratio of the antioxidant and the reactive molecule. With catalytic
antioxidants, in contrast, the antioxidant molecule can repeatedly inactivate
reactive oxygen molecules, which could result in multiple reactive oxygen
molecules combining with each antioxidant molecule.
Vitamin
derivatives that are antioxidants are scavengers. The SOD enzymes
produced by the body are catalytic antioxidants. Catalytic
antioxidants are typically much more potent than antioxidant scavengers, in some
instances by a multiple of up to 10,000.
Use of
antioxidant scavengers, such as thiols or vitamin derivatives, has shown promise
of benefit in preclinical and clinical studies. Ethyol, a
thiol-containing antioxidant, is approved for reducing radiation and
chemotherapy toxicity during cancer treatment, and clinical studies have
suggested benefit of other antioxidants in kidney and neurodegenerative
diseases. However, large sustained doses of the compounds are
required as each antioxidant scavenger molecule is consumed by its reaction with
the free radical. Toxicities and the inefficiency of scavengers have
limited the utility of antioxidant scavengers to very specific
circumstances.
Contracts
and Grants
We seek
to advance development of our drug candidates through external funding
arrangements. We may slow down development programs or place them on hold during
periods that are not covered by external funding. We have received external
funding awards for the development of AEOL 10150 as an MCM for acute radiation
syndrome in the Lung and GI, mustard gas and chlorine gas exposure from the
NIH.
On August
31, 2009, we submitted a white paper to BARDA in response to Special
Instructions Amendment 4 to a Broad Agency Announcement (BAA-BARDA-09-34) for
advanced research and development of medical countermeasures for chemical,
biological, radiological and nuclear threats. The purpose of the special
instructions amendment was to specifically solicit solutions for treating
cutaneous and/or pulmonary (life-threatening pneumonitis and fibrosis) injuries
resulting from exposure to ionizing radiation. BARDA is interested in advanced
development and eventual licensure/approval of MCMs for cutaneous and/or
pulmonary injuries resulting from an acute exposure to radiation from a
radiological/nuclear accident or attack, particularly injuries associated with
ARS or DEARE.
On
December 28, 2009, BARDA informed us that it had accepted our white paper for
Lung-ARS and requested a full proposal from the Company.
On
February 24, 2010, we submitted a full proposal to BARDA for the development of
AEOL 10150 as a medical countermeasure for Lung-ARS.
On July
19, 2010, we were notified that BARDA had completed its initial technical
evaluation of proposals received in response to BAA-BARDA-09-34, and initiated
negotiations for a potential contract for the development of AEOL 10150 as an
MCM for Pulmonary Injury Associated with ARS and DEARE.
As of
December 29, 2010, BARDA had not announced an award in response to Special
Instructions Amendment 4 to Broad Agency Announcement (BAA-BARDA-09-34), but we
expect an announcement in early 2011.
On May
31, 2010, we submitted “White Papers” to BARDA for the development of AEOL 10150
as a medical countermeasure for exposure to chlorine gas and sulfur mustard
gas.
On
September 15, 2010, BARDA informed us that it had accepted our white paper for
chlorine gas exposure and requested a full proposal from the
Company. BARDA also informed us that it had not accepted the white
paper for sulfur mustard gas.
On
December 10, 2010, we submitted a full proposal to BARDA for the development of
AEOL 10150 as a medical countermeasure for chlorine gas exposure. We
expect a response to our proposal by the third quarter of 2011.
AEOL
10150 continues to be the subject of research sponsored by NIH CounterACT as an
MCM for chlorine gas and sulfur mustard gas exposure at National Jewish
Health.
In
October 2010, we were notified that we had been awarded the maximum amount, of
$244,479, under the QTDP administered by the IRS and the HHS in support of
our development of AEOL 10150 as an MCM for Lung-ARS.
In
October 2010, we were also notified that we had been awarded the maximum amount,
of $244,479, under the QTDP administered by the IRS and the HHS in support
of our development of AEOL 11207 as a potential treatment for Parkinson’s
Disease.
23
We
continue to actively pursue additional government or foundation sponsored
development contracts and grants and to encourage both governmental,
non-governmental agencies and philanthropic organizations to provide development
funding or to conduct clinical studies of our drug candidates.
Collaborative
and Licensing Arrangements
Duke
Licenses
Through
our wholly owned subsidiary, Aeolus Sciences, Inc., we have obtained exclusive
worldwide rights from Duke University (“Duke”) to products using antioxidant
technology and compounds developed by Dr. Irwin Fridovich and other
scientists at Duke. We must pay royalties to Duke on net product
sales during the term of the Duke licenses, and must make payments upon the
occurrence of development milestones. In addition, we are obligated
under the Duke licenses to pay patent filing, prosecution, maintenance and
defense costs. The Duke licenses are terminable by Duke in the event
of breach by us and otherwise expire when the last licensed patent
expires.
National
Jewish Health
We have
obtained an exclusive worldwide license from the National Jewish Health (the
“NJH”) to develop, make, use and sell products using proprietary information and
technology developed under a previous Sponsored Research Agreement within the
field of antioxidant compounds and related discoveries. We must make
milestone payments to the NJH upon the occurrence of development milestones and
pay royalties on net sales. We are also obligated to pay patent
filing, prosecution, maintenance and defense costs. The NJH agreement
is terminable by the NJH in the event of breach and otherwise expires when the
last licensed patent expires.
In 2009,
we obtained an additional exclusive worldwide license from the NJH to develop,
make, use and sell products using proprietary information and technology
developed at the NJH related to certain compounds as an MCM against mustard gas
exposure. We must make milestone payments to the NJH upon the
occurrence of development milestones and pay royalties on net
sales. We are also obligated to pay patent filing, prosecution,
maintenance and defense costs. The NJH agreement is terminable by the
NJH in the event of breach and otherwise expires when the last licensed patent
expires.
Research
and Development Expenditures
Expenditures
for research and development activities were $1,690,000, $711,000 and $977,000
during the years ended September 30, 2010, 2009 and 2008,
respectively. Research and development expenses for fiscal 2010
related primarily to the advancement of our lead compound, AEOL
10150.
Manufacturing
We
currently do not have the capability to manufacture any of our drug candidates
on a commercial scale. Materials for non-clinical and clinical
studies are produced under contract with third parties. Assuming the
successful development of one or more of our catalytic antioxidant compounds, we
plan to contract with third parties to manufacture them.
Commercialization
Assuming
successful development and FDA approval of one or more of our compounds, to
successfully commercialize our catalytic antioxidant programs, we must seek
corporate partners with expertise in commercialization or develop this expertise
internally. However, we may not be able to successfully commercialize
our catalytic antioxidant technology, either internally or through collaboration
with others.
Marketing
Our
potential catalytic antioxidant products are being developed for large
therapeutic markets. We believe these markets are best approached by
partnering with established biotechnology or pharmaceutical companies that have
broad sales and marketing capabilities. We are pursuing
collaborations of this type as part of our search for development
partners. However, we may not be able to enter into any marketing
arrangements for any of our products on satisfactory terms or at
all.
24
Biodefense
Industry
Market
Overview
The
market for biodefense countermeasures has grown dramatically as a result of the
increased awareness of the threat of global terror activity in the wake of the
September 11, 2001 terrorist attacks. The U.S. government is the
principal source of worldwide biodefense spending. Most U.S. government spending
on biodefense programs is in the form of development funding from NIAID, BARDA
and the Department of Defense (“DoD”) and procurement of countermeasures by
BARDA, the CDC and the DoD. The U.S. government is now the largest source of
development and procurement funding for academic institutions and biotechnology
companies conducting biodefense research or developing vaccines and
immunotherapies directed at potential agents of bioterror or
biowarfare.
We
analyze the biodefense market in three segments; the United States military
market, United States commercial market and non U.S. markets,with the U.S.
government funding representing the vast majority of the worldwide market.
According to the Center for Biosecurity at the University of Pittsburgh Medical
Center the U.S. government’s biodefense military and civilian spending
approximated $8 billion in fiscal 2009 and has averaged around $5.5 billion from
fiscal years 2001 to 2009. Biodefense military and civilian spending in
fiscal year 2010 is expected to be $6 billion.
|
·
|
U.S.
Civilian: The U.S. civilian market includes funds to protect the
U.S. population from biological agents and is largely funded by the
Project BioShield Act of 2004 (“Project BioShield”). Project
BioShield is the U.S. government’s largest biodefense initiative, and
governs and funds with $5.6 billion the procurement of biodefense
countermeasures for the SNS for the period from July 2004 through
2013. A total of $3.7 billion remains in the Project BioShield
Special Reserve Funds for future grants as of March
2009.
|
|
·
|
U.S.
Military: The DoD is responsible for the development and
procurement of countermeasures for the military segment which focuses on
providing protection for military personnel and civilians who are on
active duty. Funding in fiscal year 2009 is expected to be approximately
$737 million compared to estimated spending of $622 million for
2008. We anticipate that annual funding for the programs
through 2013 will continue in a comparable
range.
|
|
·
|
Non-U.S.
Markets: Non-U.S. markets address protection against
biowarfare agents for both civilians and military personnel in foreign
countries. We anticipate that foreign countries will want to procure
biodefense products as they are developed and validated by procurement by
the U.S. government.
|
Project
BioShield and the Pandemic and All-Hazards Preparedness Act
Project
BioShield became law in 2004 and authorizes the procurement of countermeasures
for chemical, biological, radiological and nuclear attacks for the SNS, which is
a national repository of medical assets and countermeasures designed to provide
federal, state and local public health agencies with medical supplies needed to
treat those affected by terrorist attacks, natural disasters, industrial
accidents and other public health emergencies. Project BioShield provided
appropriations of $5.6 billion to be expended over ten years into a special
reserve fund. As of March 2009, a total of $3.7 billion remains
in the Project BioShield Special Reserve Funds for future grants.
The
Pandemic and All-Hazards Preparedness Act, passed in 2006, established BARDA as
the agency responsible for awarding procurement contracts for biomedical
countermeasures and providing development funding for advanced research and
development in the biodefense arena, and supplements the funding available under
Project BioShield for chemical, biological, radiological and nuclear
countermeasures, and provides funding for infectious disease pandemics. Funding
for BARDA is provided by annual appropriations by Congress. Congress also
appropriates annual funding for the CDC for the procurement of medical assets
and countermeasures for the SNS and for NIAID to conduct biodefense research.
This appropriation funding supplements amounts available under Project
BioShield.
Currently,
the U.S. government may, at its discretion, purchase critical biodefense
products for the SNS prior to FDA approval based on Emergency Use Authorization
(“EUA”) enabled under the Project Bioshield legislation. On an ongoing
basis we monitor notices for requests for proposal, grants and other potential
sources of government funding that could potentially support the development of
our drug candidates. Nevertheless, changes in government budgets,
priorities and agendas as well as political pressures could result in a
reduction in overall government financial support for the biodefense sector in
general and/or specifically the drug candidates we are developing. Due to
the current economic downturn, the accompanying fall in tax revenues and the
U.S. government’s efforts to stabilize the economy, the U.S. government may be
forced or choose to reduce or delay spending in the biodefense field, which
could decrease the likelihood of future government contract awards, the
likelihood that the government will exercise its right to extend any of its
existing contracts and/or the likelihood that the government would procure
products from us. (For further information, see “Risk Factors — Risks Related to Our
Dependence on U.S. Government Grants and Contracts — Most of our immediately
foreseeable future revenues are contingent upon grants and contracts from the
U.S. government and we may not achieve sufficient, if any, revenues from these
agreements to attain profitability.”) As a result, further development of
our drug candidates and ultimate product sales to the government, if any, could
be delayed or stopped altogether.
25
Competition
General
Competition
in the pharmaceutical industry is intense and we expect it to
increase. Technological developments in our field of research and
development occur at a rapid rate and we expect competition to intensify as
advances in this field are made. We will be required to continue to
devote substantial resources and efforts to research and development
activities. Our most significant competitors, among others, are fully
integrated pharmaceutical companies and more established biotechnology
companies, which have substantially greater financial, technical, sales,
marketing and human resources than we do. These companies may succeed
in developing and obtaining regulatory approval for competitive products more
rapidly than we can for our drug candidates. In addition, competitors
may develop technologies and products that are, or are perceived as being,
cheaper, safer or more effective than those being developed by us or that would
render our technology obsolete.
We expect
that important competitive factors in our potential product markets will be the
relative speed with which we and other companies can develop products, complete
the clinical testing and approval processes, and supply commercial quantities of
a competitive product to the market. With respect to clinical
testing, competition might result in a scarcity of clinical investigators and
patients available to test our potential products, which could delay
development.
We are
aware of products in research or development by our competitors that address the
diseases and therapies being targeted by us. In addition, there may
be other competitors of whom we are unaware with products which might be more
effective or have fewer side effects than our products and those of our known
competitors.
Antioxidants
Several
companies have explored the therapeutic potential of antioxidant compounds in
numerous indications. Historically, most of these companies have
focused on engineered versions of naturally occurring antioxidant enzymes, but
with limited success, perhaps because the large size of these molecules makes
delivery into the cells difficult. Antioxidant drug research
continues at a rapid pace despite previous clinical setbacks.
Patents
and Proprietary Rights
We
currently license rights to our potential products from third
parties. We generally seek patent protection in the United States and
other jurisdictions for the potential products and proprietary technology
licensed from these third parties. The process for preparing and
prosecuting patents is lengthy, uncertain and costly. Patents may not
issue on any of the pending patent applications owned by us or licensed by us
from third parties. Even if patents issue, the claims allowed might
not be sufficiently broad to protect our technology or provide us protection
against competitive products or otherwise be commercially
valuable. Patents issued to or licensed by us could be challenged,
invalidated, infringed, circumvented or held unenforceable. Even if
we successfully defend our patents for our products, the costs of defense can be
significant.
As of
December 29, 2010, our catalytic antioxidant small molecule technology base is
described in twelve issued United States patents and five United States pending
patent applications. These patents and patent applications belong in
whole or in part to Duke or the NJH and are licensed to us. These
patents and patent applications cover soluble manganic porphyrins as antioxidant
molecules as well as targeted compounds obtained by coupling such antioxidant
compounds to molecules that bind to specific extracellular
elements. The pending U.S. patent applications and issued U.S.
patents include composition of matter claims and method claims for several
series of compounds. Corresponding international patent applications
have been filed, 79 of which have issued as of December 29, 2010. Our
twelve issued patents will expire between 2015 and 2023.
In
addition to patent protection, we rely upon trade secrets, proprietary know-how
and technological advances that we seek to protect in part through
confidentiality agreements with our collaborative partners, employees and
consultants. Our employees and consultants are required to enter into
agreements providing for confidentiality and the assignment of rights to
inventions made by them while in our service. We also enter into
non-disclosure agreements to protect our confidential information furnished to
third parties for research and other purposes.
Government
Regulation
Our
research and development activities and the manufacturing and marketing of our
future products are subject to regulation by numerous governmental agencies in
the United States and in other countries. The FDA and comparable
agencies in other countries impose mandatory procedures and standards for the
conduct of clinical trials and the production and marketing of products for
diagnostic and human therapeutic use. Before obtaining regulatory
approvals for the commercial sale of any of our products under development, we
must demonstrate through preclinical studies and clinical trials that the
product is safe and efficacious for use in each target
indication. The results from preclinical studies and early clinical
trials might not be predictive of results that will be obtained in large-scale
testing. Our clinical trials might not successfully demonstrate the
safety and efficacy of any products or result in marketable
products.
26
The
United States system of drug approvals is considered to be the most rigorous in
the world. It takes an average of 8.5 years for a drug candidate to
move through the clinical and approval phases in the United States according to
a November 2005 study by the Tufts Center for the Study of Drug
Development. Only five in 5,000 drug candidates that enter
preclinical testing make it to human testing and only one of those five is
approved for commercialization. On average, it costs a company $897
million to get one new drug candidate from the laboratory to United States
patients according to a May 2003 report by Tufts Center for the Study of Drug
Development. A November 2006 study by Tufts Center for the Study of
Drug Development reported that the average cost of developing a new
biotechnology product was $1.2 billion and took on average slightly more than
eight years to be approved by the FDA.
The steps
required by the FDA before new drug products may be marketed in the United
States include:
|
|
·
|
completion
of preclinical studies;
|
|
|
·
|
the
submission to the FDA of a request for authorization to conduct clinical
trials on an IND, which must become effective before clinical trials may
commence;
|
|
|
·
|
adequate
and well-controlled Phase I clinical trials which typically involves
normal, healthy volunteers. The tests study a drug candidate’s
safety profile, including the safe dosage range. The studies
also determine how a drug is absorbed, distributed, metabolized and
excreted as well as the duration of its
action;
|
|
|
·
|
adequate
and well-controlled Phase II clinical trials which typically
involve treating patients with the targeted disease with the drug
candidate to assess a drug’s
effectiveness;
|
|
|
·
|
adequate
and well-controlled Phase III clinical trials involving a larger
population of patients with the targeted disease are treated with the drug
candidate to confirm efficacy of the drug candidate in the treatment of
the targeted indication and to identify adverse
events;
|
|
|
·
|
submission
to the FDA of an NDA; and
|
|
|
·
|
review
and approval of the NDA by the FDA before the product may be shipped or
sold commercially.
|
In
addition to obtaining FDA approval for each product, each product manufacturing
establishment must be registered with the FDA and undergo an inspection prior to
the approval of an NDA. Each manufacturing facility and its quality
control and manufacturing procedures must also conform and adhere at all times
to the FDA’s current good manufacturing practices (“cGMP”)
regulations. In addition to preapproval inspections, the FDA and
other government agencies regularly inspect manufacturing facilities for
compliance with these requirements. Manufacturers must expend
substantial time, money and effort in the area of production and quality control
to ensure full technical compliance with these standards.
Preclinical
testing includes laboratory evaluation and characterization of the safety and
efficacy of a drug and its formulation. Preclinical testing results
are submitted to the FDA as a part of an IND which must become effective prior
to commencement of clinical trials. Clinical trials are typically
conducted in three sequential phases following submission of an IND. Phase I
represents the initial administration of the drug to a small group of humans,
either patients or healthy volunteers, typically to test for safety (adverse
effects), dosage tolerance, absorption, distribution, metabolism, excretion and
clinical pharmacology, and, if possible, to gain early evidence of
effectiveness. Phase II involves studies in a small sample of the
actual intended patient population to assess the efficacy of the drug for a
specific indication, to determine dose tolerance and the optimal dose range and
to gather additional information relating to safety and potential adverse
effects. Once an investigational drug is found to have some efficacy
and an acceptable safety profile in the targeted patient population, Phase III
studies are initiated to further establish clinical safety and efficacy of the
therapy in a broader sample of the general patient population, in order to
determine the overall risk-benefit ratio of the drug and to provide an adequate
basis for any physician labeling. During all clinical studies, we
must adhere to GCP standards. The results of the research and product
development, manufacturing, preclinical studies, clinical studies and related
information are submitted in an NDA to the FDA.
The
process of completing clinical testing and obtaining FDA approval for a new drug
is likely to take a number of years and require the expenditure of substantial
resources. If an application is submitted, there can be no assurance
that the FDA will review and approve the NDA. Even after initial FDA
approval has been obtained, further studies, including post-market studies,
might be required to provide additional data on safety and will be required to
gain approval for the use of a product as a treatment for clinical indications
other than those for which the product was initially tested and
approved. Also, the FDA will require post-market reporting and might
require surveillance programs to monitor the side effects of the
drug. Results of post-marketing programs might limit or expand the
further marketing of the products. Further, if there are any
modifications to the drug, including changes in indication, manufacturing
process, labeling or a change in manufacturing facility, an NDA supplement might
be required to be submitted to the FDA.
The rate
of completion of any clinical trials will be dependent upon, among other
factors, the rate of patient enrollment. Patient enrollment is a
function of many factors, including the size of the patient population, the
nature of the trial, the availability of alternative therapies and drugs, the
proximity of patients to clinical sites and the eligibility criteria for the
study. Delays in planned patient enrollment might result in increased
costs and delays, which could have a material adverse effect on us.
Failure
to comply with applicable FDA requirements may result in a number of
consequences that could materially and adversely affect us. Failure
to adhere to approved trial standards and GCPs in conducting clinical trials
could cause the FDA to place a clinical hold on one or more studies which would
delay research and data collection necessary for product
approval. Noncompliance with GCPs could also have a negative impact
on the FDA’s evaluation of an NDA. Failure to adhere to GMPs and
other applicable requirements could result in FDA enforcement action and in
civil and criminal sanctions, including but not limited to fines, seizure of
product, refusal of the FDA to approve product approval applications, withdrawal
of approved applications, and prosecution.
27
Whether
or not FDA approval has been obtained, approval of a product by regulatory
authorities in foreign countries must be obtained prior to the commencement of
marketing of the product in those countries. The requirements
governing the conduct of clinical trials and product approvals vary widely from
country to country, and the time required for approval might be longer or
shorter than that required for FDA approval. Although there are some
procedures for unified filings for some European countries, in general, each
country at this time has its own procedures and requirements. There
can be no assurance that any foreign approvals would be obtained.
In
addition to the regulatory framework for product approvals, we and our
collaborative partners must comply with laws and regulations regarding
occupational safety, laboratory practices, the use, handling and disposition of
radioactive materials, environmental protection and hazardous substance control,
and other local, state, federal and foreign regulation. The impact of
such regulation upon us cannot be predicted and could be material and
adverse.
Legislation
and Regulation Related to Bioterrorism Counteragents
Because
some of our drug candidates are intended for the treatment of diseases that may
result from acts of bioterrorism, they may be subject to the specific
legislation and regulation described below.
Project
BioShield
Project
BioShield provides expedited procedures for bioterrorism related procurement and
awarding of research grants, making it easier for HHS to quickly commit funds to
countermeasure projects. Project BioShield relaxes procedures under the Federal
Acquisition Regulation for procuring property or services used in performing,
administering or supporting biomedical countermeasure research and development.
In addition, if the Secretary of HHS deems that there is a pressing need,
Project BioShield authorizes the Secretary to use an expedited award process,
rather than the normal peer review process, for grants, contracts and
cooperative agreements related to biomedical countermeasure research and
development activity.
Under
Project BioShield, the Secretary of HHS, with the concurrence of the Secretary
of the Department of Homeland Security (“DHS”), and upon the approval of the
President, can contract to purchase unapproved countermeasures for the SNS in
specified circumstances. Congress is notified of a recommendation for a
stockpile purchase after Presidential approval. Project BioShield specifies that
a company supplying the countermeasure to the SNS is paid on delivery of a
substantial portion of the countermeasure. To be eligible for purchase under
these provisions, the Secretary of HHS must determine that there are sufficient
and satisfactory clinical results or research data, including data, if
available, from preclinical and clinical trials, to support a reasonable
conclusion that the countermeasure will qualify for approval or licensing within
eight years. Project BioShield also allows the Secretary of HHS to authorize the
emergency use of medical products that have not yet been approved by the FDA. To
exercise this authority, the Secretary of HHS must conclude that:
|
|
•
|
the agent for which the
countermeasure is designed can cause serious or life-threatening
disease;
|
|
|
•
|
the product may reasonably be
believed to be effective in detecting, diagnosing, treating or preventing
the disease;
|
|
|
•
|
the known and potential benefits
of the product outweigh its known and potential risks;
and
|
|
|
•
|
there is no adequate alternative
to the product that is approved and
available.
|
Although
this provision permits the Secretary of HHS to circumvent the FDA approval
process, its use would be limited to rare circumstances.
Safety
Act
The
Support Anti-Terrorism by Fostering Effective Technologies Act enacted by the
U.S. Congress in 2002 (the “Safety Act”) creates product liability limitations
for qualifying anti-terrorism technologies for claims arising from or related to
an act of terrorism. In addition, the Safety Act provides a process by which an
anti-terrorism technology may be certified as an “approved product” by the DHS
and therefore entitled to a rebuttable presumption that the government
contractor defense applies to sales of the product. The government contractor
defense, under specified circumstances, extends the sovereign immunity of the
United States to government contractors who manufacture a product for the
government. Specifically, for the government contractor defense to apply, the
government must approve reasonably precise specifications, the product must
conform to those specifications and the supplier must warn the government about
known dangers arising from the use of the product.
Public
Readiness and Emergency Preparedness Act
The
Public Readiness and Emergency Preparedness Act enacted by Congress in 2005 (the
“PREP Act”) provides immunity for manufacturers from all claims under state or
federal law for “loss” arising out of the administration or use of a “covered
countermeasure”. However, injured persons may still bring a suit for “willful
misconduct” against the manufacturer under some circumstances. “Covered
countermeasures” include security countermeasures and “qualified pandemic or
epidemic products”. For these immunities to apply, the Secretary of
HHS must issue a declaration in cases of public health emergency or “credible
risk” of a future public health emergency. We cannot predict whether Congress
will fund the relevant PREP Act compensation programs; or whether the necessary
prerequisites for immunity would be triggered with respect to our drug
candidates.
28
Foreign
Regulation
In
addition to regulations in the United States, we will be subject to a variety of
foreign regulations governing clinical trials and commercial sales and
distribution of our products. Whether or not we obtain FDA approval for a
product, we must obtain approval of a product by the comparable regulatory
authorities of foreign countries before we can commence clinical trials or
marketing of the product in those countries. The actual time required to obtain
clearance to market a product in a particular foreign jurisdiction may vary
substantially, based upon the type, complexity and novelty of the pharmaceutical
product candidate and the specific requirements of that jurisdiction. The
requirements governing the conduct of clinical trials, marketing authorization,
pricing and reimbursement vary from country to country.
Reimbursement
and Pricing Controls
In many
of the markets where we could commercialize a drug candidate following
regulatory approval, the prices of pharmaceutical products are subject to direct
price controls by law and to reimbursement programs with varying price control
mechanisms.
In the
United States, there is an increasing focus on drug pricing in recent years.
There are currently no direct government price controls over private sector
purchases in the United States. However, the Veterans Health Care Act
establishes mandatory price discounts for certain federal purchasers, including
the Veterans Administration, Department of Defense and the Public Health
Service; the discounts are based on prices charged to other
customers.
Under the
Medicaid program (a joint federal/state program that provides medical coverage
to certain low income families and individuals), pharmaceutical manufacturers
must pay prescribed rebates on specified drugs to enable them to be eligible for
reimbursement. Medicare (the federal program that provides medical
coverage for the elderly and disabled) generally reimburses for
physician-administered drugs and biologics on the basis of the product’s average
sales price. Outpatient drugs may be reimbursed under Medicare Part
D. Part D is administered through private entities that attempt to negotiate
price concessions from pharmaceutical manufacturers. Various states have adopted
further mechanisms that seek to control drug prices, including by disfavoring
higher priced products and by seeking supplemental rebates from manufacturers.
Managed care has also become a potent force in the marketplace and increases
downward pressure on the prices of pharmaceutical products.
Public
and private health care payors control costs and influence drug pricing through
a variety of mechanisms, including through negotiating discounts with the
manufacturers and through the use of tiered formularies and other mechanisms
that provide preferential access to particular products over others within a
therapeutic class. Payors also set other criteria to govern the uses of a drug
that will be deemed medically appropriate and therefore reimbursed or otherwise
covered. In particular, many public and private health care payors limit
reimbursement and coverage to the uses that are either approved by the FDA or
that are supported by other appropriate evidence, such as published medical
literature, and appear in a recognized compendium. Drug compendia are
publications that summarize the available medical evidence for particular drug
products and identify which uses are supported or not supported by the available
evidence, whether or not such uses have been approved by the FDA.
Different
pricing and reimbursement schemes exist in other countries. In the European
Union, governments influence the price of pharmaceutical products through their
pricing and reimbursement rules and control of national health care systems that
fund a large part of the cost of those products to consumers. Some jurisdictions
operate positive and negative list systems under which products may only be
marketed once a reimbursement price has been agreed. Other member states allow
companies to fix their own prices for medicines, but monitor and control company
profits. The downward pressure on health care costs in general, particularly
prescription drugs, has become very intense. As a result, increasingly high
barriers are being erected to the entry of new products. In addition, in some
countries cross-border imports from low-priced markets exert a commercial
pressure on pricing within that country.
Regulations
Regarding Government Contracting
We may
become a government contractor in the United States and elsewhere which would
mean that we would be subject to various statutes and regulations that govern
the procurement of goods and services by agencies of the United States and other
countries, including the Federal Acquisition Regulation. These governing
statutes and regulations can impose stricter penalties than those normally
applicable to commercial contracts, such as criminal and civil damages liability
and suspension and debarment from future government contracting. In addition,
pursuant to various statutes and regulations, our government contracts may be
subject to unilateral termination or modification by the government for
convenience in the United States and elsewhere, detailed auditing requirements
and accounting systems, statutorily controlled pricing, sourcing and
subcontracting restrictions and statutorily mandated processes for adjudicating
contract disputes.
29
Hazardous
Materials and Select Agents
Our
development and manufacturing processes involve the use of hazardous materials,
including chemicals and radioactive materials, and produce waste products.
Accordingly, we are subject to federal, state and local laws and regulations
governing the use, manufacture, storage, handling and disposal of these
materials. In addition to complying with environmental and occupational health
and safety laws, we must comply with special regulations relating to biosafety
administered by the CDC, HHS and the DoD.
Other
Regulations
In the
United States and elsewhere, the research, manufacturing, distribution, sale and
promotion of drug and biological products are subject to regulation by various
federal, state and local authorities in addition to the FDA, including the
Centers for Medicare and Medicaid Services; other divisions of HHS, such as the
Office of Inspector General: the U.S. Department of Justice and individual U.S.
Attorney offices within the Department of Justice and state and local
governments. For example, sales, marketing and scientific and educational grant
programs must comply with the anti-kickback and fraud and abuse provisions of
the Social Security Act, the False Claims Act, the privacy provisions of the
Health Insurance Portability and Accountability Act and similar state laws.
Pricing and rebate programs must comply with the Medicaid rebate requirements of
the Omnibus Budget Reconciliation Act of 1990 and the Veterans Health Care Act
of 1992. All of these activities are also potentially subject to federal and
state consumer protection and unfair competition laws.
CPEC,
LLC
We were
previously developing bucindolol for the treatment of heart failure, but
development was discontinued in 1999. Commercial rights to bucindolol
are owned by CPEC, LLC, a limited liability company (“CPEC”), of which we own
35% and Indevus Pharmaceuticals, Inc. owns 65%.
During
fiscal 2008, CPEC received a milestone payment from ARCA of
$500,000. The milestone payment was triggered by the acceptance by
the FDA of an NDA for bucindolol. Future milestone payments and
royalty payments to us and CPEC, if any, while provided for under the agreement
between CPEC and ARCA, cannot be assured or guaranteed. Also as a
result of the filing of the NDA with the FDA, we are obligated to pay $412,500
in the form of cash or stock at our election to the majority owner of CPEC who
in turn paid the original licensors of bucindolol per the terms of the 1994
Purchase Agreement of CPEC. On November 6, 2009, we issued 1,099,649 shares to
the majority owner of CPEC to satisfy our obligation.
During
fiscal 2009, we sold our holdings of ARCA generating a gain of
$133,000. In addition during fiscal 2009, ARCA received a Complete
Response letter from the FDA for its NDA for bucindolol for the treatment
of patients with chronic heart failure. In the Complete Response letter,
the FDA stated that it cannot approve the NDA in its current form and specifies
additional actions and information required for approval of the bucindolol
NDA.
Employees
At
December 29, 2010, we had two full-time employees and no part time employees.
None of our employees is represented by a labor union. In addition to
our employees, we utilize several consultants to perform key functions for the
Company.
Executive
Officers
Our
executive officers and their ages as of December 29, 2010 were as
follows:
|
Name
|
Age
|
Position(s)
|
||
|
David
Cavalier
|
41
|
Chairman
of the Board
|
||
|
John
L. McManus
|
46
|
President,
Chief Executive Officer, Chief Financial Officer and
Secretary
|
||
|
Brian
J. Day, Ph.D.
|
50
|
Chief
Scientific Officer
|
David C. Cavalier has
been the Chairman of our Board since April 30, 2004, and became a full time
employee of the Company in November 2009. Since 2001, he has been a Principal
and the Chief Operating Officer of Xmark Opportunity Partners, LLC, a manager of
a family of private investment funds. From 1995 to 1996, Mr. Cavalier
worked for Tiger Real Estate, a $785 million private investment fund sponsored
by Tiger Management Corporation. Mr. Cavalier began his career in 1994 in the
Investment Banking Division of Goldman, Sachs & Co. working on debt and
equity offerings for public and private real estate companies. He received a
B.A. from Yale University and an M.Phil.
from Oxford University.
30
John L.
McManus. Mr. McManus began as a consultant to the Company in
June 2005 as President. He became employed as our President and Chief
Operating Officer in July 2006 and was appointed President and Chief Executive
Officer in March 2007. Mr. McManus, who received his degree in
business administration from the University of Southern California in 1986, is
the founder and president of McManus Financial Consultants, Inc. (“MFC”), which
provides strategic, financial and investor relations advice to senior
managements and boards of directors of public companies, including advice on
mergers and acquisitions. These companies have a combined value of
over $25 billion. He has served as president of MFC since
1997. In addition, Mr. McManus previously served as Vice President,
Finance and Strategic Planning to Spectrum Pharmaceuticals, Inc. where he had
primary responsibility for restructuring Spectrum’s operations and finances,
including the design of strategic and financial plans to enhance Spectrum’s
corporate focus, and leading the successful implementation of these
plans. The implementation of these plans led to an increase in
Spectrum’s market value from $1 million to more than $125 million at the time of
Mr. McManus’ departure.
Brian J. Day, Ph.D. Dr. Day
is a part-time consultant and was appointed our Chief Scientific Officer in
September 2004. Dr. Day has extensive training in both pharmacology
and toxicology with over 14 years of experience. Since 1994 he has
helped guide the design and synthesis of metalloporphyrins and has discovered a
number of their novel activities in biological systems. Dr. Day has
authored over 70 original scientific publications and served as a consultant to
biotechnology companies for over 10 years. He is an active member of
a number of scientific societies including the American Chemical Society,
Society for Free Radicals in Biology and Medicine, and Society of Toxicology,
where he served on the Board of Publications. Dr. Day has been at the
NJH since 1997 and currently is a Professor in the Environmental and
Occupational Health Sciences Division. He is one of our scientific
co-founders and an inventor on a majority of the catalytic antioxidant program’s
patents.
Item
1A. Risk Factors.
You
should carefully consider the following information about risks described below,
together with the other information contained in this annual report on
Form 10-K and in our other filings with the SEC, before you decide to buy or
maintain an investment in our common stock. We believe the risks
described below are the risks that are material to us as of the date of this
annual report. If any of the following risks actually occur, our
business, financial condition, results of operations and future growth prospects
would likely be materially and adversely affected. In these
circumstances, the market price of our common stock could decline, and you may
lose all or part of your investment.
Risks
Related to Our Business
We
have operated at a loss and will likely continue to operate at a loss for the
foreseeable future.
We have
incurred significant losses over the past five years, including net losses of
approximately $25.9 million, $2.3 million and $3.0 million for the years ended
September 30, 2010, 2009 and 2008, respectively, and we had an accumulated
deficit of approximately $182.7 million as of September 30, 2010. Our
operating losses have been due primarily to our expenditures for research and
development on our drug candidates and for general and administrative expenses
and our lack of significant revenues, and during 2010 due to $21.3 million in
warrant liability charges related to warrants issued to investors in
financings. We are likely to continue to incur operating losses until
such time, if ever, that we generate significant recurring
revenues. We anticipate it will take a minimum of four years (and
possibly longer) for us to generate recurring revenues, since we expect that it
will take at least that long before the development of any of our licensed or
other current potential products is completed, marketing approvals are obtained
from the FDA and commercial sales of any of these products can
begin.
We
need substantial additional funding to continue our operations and may be unable
to raise capital when needed, or at all, which would force us to delay, curtail
or eliminate our clinical programs and our product development
programs.
We need
to raise substantial additional capital to fund our operations and clinical
trials and continue our research and development. In addition, we may
need to raise substantial additional capital to enforce our proprietary rights,
defend, in litigation or otherwise, any claims that we infringe third party
patents or other intellectual property rights; and commercialize any of our
products that may be approved by the FDA or any international regulatory
authority.
As of
September 30, 2010, we had cash of approximately $2,355,000. In
addition, we received $336,885 in November 2010 from the IRS for the
QTDP. On December 28, 2010, we raised gross proceeds of $1,000,000
through the sale of our common stock and warrants to two institutional investors
as a result of these investors exercising an option for additional investment
that they received in our August 2010 financing. We expect to use
these funds, including any additional funds received pursuant to the issuance of
additional Common Stock and the exercise of outstanding warrants to purchase our
capital stock, to continue the development of our drug candidates, to expand the
development of our drug pipeline and for working capital.
We
believe we have adequate financial resources to fund our current operations into
the second quarter of fiscal year 2012. However, in order to fund
on-going cash requirements beyond that point, or to further accelerate or expand
our programs, we will need to raise additional funds. We are
continuously considering strategic and financial options available to us,
including public or private equity offerings, debt financings and collaboration
arrangements. If we raise additional funds by issuing securities, our
stockholders will experience dilution of their ownership
interest. Debt financings, if available, may involve restrictive
covenants and require significant interest payments. If we do not
receive additional financing to fund our operations beyond the second quarter of
fiscal 2012, we would have to discontinue some or all of our activities, merge
with or sell, lease or license some or all of our assets to another company, or
cease operations entirely, and our stockholders might lose all or part of their
investments.
31
In
addition, if our catalytic antioxidant program shows scientific progress, we
will need significant additional funds to move therapies through the preclinical
stages of development and clinical trials. If we are unable to raise
the amount of capital necessary to complete development and reach
commercialization of any of our catalytic antioxidant products, we will need to
delay or cease development of one or more of these products or partner with
another company for the development and commercialization of these
products.
Our
independent registered public accounting firm has expressed substantial doubt
about our ability to continue as a going concern.
In its
audit opinion issued in connection with our consolidated balance sheets as of
September 30, 2010 and 2009 and our consolidated statements of operations,
stockholder’s equity and cash flows for the years ended September 30, 2010, 2009
and 2008, our independent registered public accounting firm has expressed
substantial doubt about our ability to continue as a going concern given our
recurring net losses, negative cash flows from operations and working capital
deficiency. The accompanying financial statements have been prepared
on a going concern basis, which contemplates the realization of assets and the
satisfaction of liabilities and commitments in the normal course of
business. The financial statements do not include any adjustments
relating to the recoverability and classification of recorded asset amounts or
amounts of liabilities that might be necessary should we be unable to continue
in existence.
We
have a history of operating losses, expect to continue to incur substantial
losses and may never become profitable.
We have
no products approved for commercialization in the United States or
abroad. Our drug candidates are still being developed, and all but
our AEOL 10150 candidate are still in early stages of
development. Our drug candidates will require significant additional
development, clinical trials, regulatory clearances or approvals by the FDA and
additional investment before they can be commercialized in the United
States.
Our
likelihood for achieving profitability will depend on numerous factors,
including success in:
|
|
·
|
developing
our existing drug candidates and developing and testing new drug
candidates;
|
|
|
·
|
carrying
out our intellectual property
strategy;
|
|
|
·
|
establishing
our competitive position;
|
|
|
·
|
achieving
third-party collaborations;
|
|
|
·
|
receiving
regulatory approvals;
|
|
|
·
|
manufacturing
and marketing products; and
|
|
|
·
|
receiving
government funding and identifying new government funding
opportunities.
|
Many of
these factors will depend on circumstances beyond our control. We
cannot guarantee that we will achieve sufficient revenues for
profitability. Even if we do achieve profitability, we cannot
guarantee that we can sustain or increase profitability on a quarterly or annual
basis in the future. If revenues grow more slowly than we anticipate,
or if operating expenses exceed our expectations or cannot be adjusted
accordingly, then our business, results of operations, financial condition and
cash flows will be materially and adversely affected.
The
current turmoil affecting the financial markets and the possibility that
financial institutions may consolidate or cease operations has resulted in a
tightening in the credit markets, a low level of liquidity in many financial
markets and extreme volatility in fixed income, credit, currency and equity
markets. As a result, there can be no assurances that we will be
successful in obtaining sufficient financing on commercially reasonable terms or
at all. Our requirements for additional capital may be substantial
and will be dependent on many factors, including the success of our research and
development efforts, our ability to commercialize and market products, our
ability to successfully pursue our licensing and collaboration strategy, the
receipt of government funding, competing technological developments, costs
associated with the protection of our intellectual property and any future
change in our business strategy.
As of
September 30, 2010, we had an accumulated deficit of $182.7 million from our
research, development and other activities. We have not generated
material revenues from product sales and do not expect to generate product
revenues sufficient to support us for at least several more years.
Our
research and development (“R&D”) activities are at an early stage and
therefore might never result in viable products.
Our
catalytic antioxidant program is in the early stages of development, involves
unproven technology, requires significant further R&D and regulatory
approvals and is subject to the risks of failure inherent in the development of
products or therapeutic procedures based on innovative
technologies. These risks include the possibilities
that:
|
|
·
|
any
or all of these proposed products or procedures are found to be unsafe or
ineffective or otherwise fail to receive necessary regulatory
approvals;
|
32
|
·
|
the
proposed products or procedures are not economical to market or do not
achieve broad market acceptance;
|
|
·
|
third
parties hold proprietary rights that preclude us from marketing the
proposed products or procedures;
and
|
|
·
|
third
parties market a superior or equivalent
product.
|
Further,
the timeframe for commercialization of any product is long and uncertain because
of the extended testing and regulatory review process required before marketing
approval can be obtained. There can be no assurance that we will be
able to successfully develop or market any of our proposed products or
procedures.
If
our products are not successfully developed and eventually approved by the FDA,
we may be forced to reduce or terminate our operations.
All of
our drug candidates are at various stages of development and must be approved by
the FDA or similar foreign governmental agencies before they can be
marketed. The process for obtaining FDA and foreign regulatory
approval is both time-consuming and costly, with no certainty of a successful
outcome. This process typically requires extensive preclinical and
clinical testing, which may take longer or cost more than we anticipate, and may
prove unsuccessful due to numerous factors. Drug candidates that may
appear to be promising at early stages of development may not successfully reach
the market for a number of reasons. The results of preclinical and
initial clinical testing of these drug candidates may not necessarily indicate
the results that will be obtained from later or more extensive
testing. A number of companies in the pharmaceutical and
biotechnology industries have suffered significant setbacks in advanced clinical
trials, even after obtaining promising results in earlier trials.
Numerous
factors could affect the timing, cost or outcome of our drug development
efforts, including the following:
|
·
|
Difficulty
in securing research laboratories to conduct research activities;
|
|
·
|
Difficulty
in securing centers to conduct
trials;
|
|
·
|
Difficulty
in enrolling patients in conformity with required protocols or projected
timelines;
|
|
·
|
Unexpected
adverse reactions by patients in
trials;
|
|
·
|
Difficulty
in obtaining clinical supplies of the
product;
|
|
·
|
Changes
in the FDA’s or other regulatory body’s requirements for our testing
during the course of that testing;
|
|
·
|
Inability
to generate statistically significant data confirming the efficacy of the
product being tested;
|
|
·
|
Modification
of the drug during testing; and
|
|
·
|
Reallocation
of our limited financial and other resources to other clinical
programs.
|
It is
possible that none of the products we develop will obtain the regulatory
approvals necessary for us to begin commercializing them. The time
required to obtain FDA and other approvals is unpredictable but often can take
years following the commencement of clinical trials, depending upon the nature
of the drug candidate. Any analysis we perform of data from clinical
activities is subject to confirmation and interpretation by regulatory
authorities, which could delay, limit or prevent regulatory
approval. Any delay or failure in obtaining required approvals could
have a material adverse effect on our ability to generate revenues from the
particular drug candidate and we may not have the financial resources to
continue to develop our drug candidates and, as a result, may have to terminate
our operations.
If
we do not reach the market with our products before our competitors offer
products for the same or similar uses, or if we are not effective in marketing
our products, our revenues from product sales, if any, will be
reduced.
We face
intense competition in our development activities. Many of our
competitors are fully integrated pharmaceutical companies and more established
biotechnology companies, which have substantially greater financial, technical,
sales and marketing and human resources than we do. These companies
might succeed in obtaining regulatory approval for competitive products more
rapidly than we can for our products. In addition, competitors might
develop technologies and products that are less expensive and perceived to be
safer or more effective than those being developed by us, which could impair our
product development and render our technology obsolete.
We
are and expect to remain dependent upon collaborations with third parties for
the development of new products, and adverse events involving these
collaborations could prevent us from developing and commercializing our drug
candidates and achieving profitability.
We
currently license from third parties, and do not own, rights under patents and
certain related intellectual property for the development of our drug
candidates. In addition, we expect to enter into agreements with
third parties to license rights to our drug candidates. We might not
be able to enter into or maintain these agreements on terms favorable to us, if
at all. Further, if any of our current licenses were to expire or
terminate, our business, prospects, financial condition and results of
operations could be materially and adversely affected.
33
Our
research and development activities rely on technology licensed from third
parties, and termination of any of those licenses would result in loss of
significant rights to develop and market our products, which would impair our
business, prospects, financial condition and results of operations.
We have
exclusive worldwide rights to our antioxidant small molecule technology through
license agreements with Duke University and the National Jewish
Health. Each license generally may be terminated by the licensor if
we fail to perform our obligations under the agreement, including obligations to
develop the compounds and technologies under license. If terminated,
we would lose the right to develop the products, which could adversely affect
our business, prospects, financial condition and results of
operations. The license agreements also generally require us to meet
specified milestones or show reasonable diligence in development of the
technology. If disputes arise over the definition of these
requirements or whether we have satisfied the requirements in a timely manner,
or if any other obligations in the license agreements are disputed by the other
party, the other party could terminate the agreement, and we could lose our
rights to develop the licensed technology.
If new
technology is developed from these licenses, we may be required to negotiate
certain key financial and other terms, such as royalty payments, for the
licensing of this future technology with these research institutions, and it
might not be possible to obtain any such license on terms that are satisfactory
to us, or at all.
We
now rely, and will continue to rely, heavily on third parties for product and
clinical development, manufacturing, marketing and distribution of our
products.
We
currently depend heavily and will depend heavily in the future on third parties
for support in product development, clinical development, manufacturing,
marketing and distribution of our products. The termination of some
or all of our existing collaborative arrangements, or our inability to establish
and maintain collaborative arrangements, could have a material adverse effect on
our ability to continue or complete clinical development of our
products.
We rely
on contract clinical research organizations (“CROs”) for various aspects of our
clinical development activities including clinical trial monitoring, data
collection and data management. As a result, we have had and continue
to have less control over the conduct of clinical trials, the timing and
completion of the trials, the required reporting of adverse events and the
management of data developed through the trial than would be the case if we were
relying entirely upon our own staff. Although we rely on CROs to
conduct our clinical trials, we are responsible for confirming that each of our
clinical trials is conducted in accordance with the investigational plan and
protocol. Moreover, the FDA and foreign regulatory agencies require
us to comply with good clinical practices (“GCPs”) for conducting, recording and
reporting the results of clinical trials to assure that the data and results are
credible and accurate and that the trial participants are adequately
protected. Our reliance on third parties does not relieve us of these
responsibilities and requirements.
The third
parties on which we rely may have staffing difficulties, may undergo changes in
priorities or may become financially distressed, adversely affecting their
willingness or ability to conduct our trials. We may experience
unexpected cost increases that are beyond our control. Any failure of
such CROs to successfully accomplish clinical trial monitoring, data collection
and data management and the other services they provide for us in a timely
manner and in compliance with regulatory requirements could have a material
adverse effect on our ability to complete clinical development of our products
and obtain regulatory approval. Problems with the timeliness or
quality of the work of a CRO may lead us to seek to terminate the relationship
and use an alternate service provider. However, making such changes
may be costly and would likely delay our trials, and contractual restrictions
may make such a change difficult or impossible. Additionally, it may
be difficult to find a replacement organization that can conduct our trials in
an acceptable manner and at an acceptable cost.
If
we do not receive the grant by the Biomedical Advanced Research and Development
Authority (“BARDA”) for advanced research and development of medical
countermeasures for chemical, biological, radiological and nuclear threats, we
would be dependent upon grants from NIH for continued development of AEOL 10150
for Lung-ARS, or we would need to curtail our development program in this area
significantly and we may be placed at a competitive disadvantage in addressing
this market opportunity.
On March
4, 2009, BARDA issued a Special Instructions Amendment 4 to a Broad Agency
Announcement (BAA-BARDA-09-34) for advanced research and development of medical
countermeasures (MCM) for chemical, biological, radiological and nuclear
threats. We submitted a white paper to this request on August 31,
2009, and on December 28, 2009, we were invited by BARDA to submit a full
proposal. On February 24, 2010, we submitted a full proposal to
BARDA. On July 19, 2010, we were notified by BARDA of the initiation
of negotiations for a contract for the development of AEOL 10150 as an MCM for
Lung-ARS. The timing for an award under this announcement
remains uncertain, but is expected in early 2011. There can be no
assurance that we will be awarded a grant under that announcement or if we
receive an award, that it will not include substantial conditions, that we can
satisfy any of the conditions or that we can begin to receive any proceeds from
any such award within any specific period of time.
Necessary
reliance on the Animal Rule in conducting trials is time-consuming and
expensive.
To obtain
FDA approval for our drug candidate for a bioterrorism indication under current
FDA regulations, we are required to utilize animal model studies for efficacy
and provide animal and human safety data under the “Animal Rule.” For
many of the biological and chemical threats, animal models are not yet
available, and as such we are developing, or will have to develop, appropriate
animal models, which is a time-consuming and expensive research
effort. Further, we may not be able to sufficiently demonstrate the
animal correlation to the satisfaction of the FDA, as these corollaries are
difficult to establish and are often unclear. The FDA may decide that
our data are insufficient for approval and require additional preclinical,
clinical or other studies, refuse to approve our products, or place restrictions
on our ability to commercialize those products. Further, other
countries do not, at this time, have established criteria for review and
approval of these types of products outside their normal review process; i.e., there is no “Animal
Rule” equivalent, and consequently there can be no assurance that we will be
able to make a submission for marketing approval in foreign countries based on
such animal data.
34
Additionally,
few facilities in the U.S. and internationally have the capability to test
animals with radiation, nerve agents, or other lethal biotoxins or chemical
agents or otherwise assist us in qualifying the requisite animal
models. We have to compete with other biodefense companies for access
to this limited pool of highly specialized resources. We therefore
may not be able to secure contracts to conduct the testing in a predictable
timeframe, cost-effectively or at all.
Even
if we succeed in commercializing our drug candidates, they may not become
profitable and manufacturing problems or side effects discovered at later stages
can further increase costs of commercialization.
We cannot
assure you that any drugs resulting from our research and development efforts
will become commercially available. Even if we succeed in developing
and commercializing our drug candidates, we may never generate sufficient or
sustainable revenues to enable us to be profitable. Even if
effective, a product that reaches the market may be subject to additional
clinical trials, changes to or re-approvals of our manufacturing facilities or a
change in labeling if we or others identify side effects or manufacturing
problems after a product is on the market. This could harm sales of
the affected products and could increase the cost and expenses of
commercializing and marketing them. It could also lead to the
suspension or revocation of regulatory approval for the products.
We and
our CMOs will also be required to comply with the applicable FDA current cGMP
regulations. These regulations include requirements relating to
quality control and quality assurance as well as the corresponding maintenance
of records and documentation. Manufacturing facilities are subject to
inspection by the FDA. These facilities must be approved to supply
licensed products to the commercial marketplace. We and our contract
manufacturers may not be able to comply with the applicable cGMP requirements
and other FDA regulatory requirements. Should we or our contract
manufacturers fail to comply, we could be subject to fines or other sanctions or
could be prohibited from marketing any products we develop.
Political
or social factors may delay or impair our ability to market our products and our
business may be materially adversely affected.
Products
developed to treat diseases caused by, or to combat the threat of, bioterrorism
will be subject to changing political and social environments. The
political and social responses to bioterrorism have been
unpredictable. Political or social pressures may delay or cause
resistance to bringing our products to market or limit pricing of our products,
which would harm our business. Changes to favorable laws, such as
Project BioShield, could have a material adverse effect on our business,
prospects, financial condition and results of operations.
Legislation
limiting or restricting liability for medical products used to fight
bioterrorism is new, and we cannot be certain that any such protection will
apply to our products or if applied what the scope of any such coverage will
be.
The U.S.
Public Readiness Act was signed into law in December 2005 (the “Public Readiness
Act”) and creates general immunity for manufacturers of countermeasures,
including security countermeasures (as defined in Section 319F-2(c)(1)(B) of the
Public Readiness Act), when the U.S. Secretary of Health and Human Services
issues a declaration for their manufacture, administration or
use. The declaration is meant to provide general immunity from all
claims under state or federal law for loss arising out of the administration or
use of a covered countermeasure. Manufacturers are excluded from this
protection in cases of willful misconduct. There can be no assurance
that the Secretary of Health and Human Services will make declarations that
would cover any of our other drug candidates or that the U.S. Congress will not
act in the future to reduce coverage under the Public Readiness Act or to repeal
it altogether.
Upon a
declaration by the Secretary of Health and Human Services, a compensation fund
would be created to provide “timely, uniform and adequate compensation to
eligible individuals for covered injuries directly caused by the administration
or use of a covered countermeasure.” The “covered injuries” to which
the program applies are defined as serious physical injuries or
death. Individuals are permitted to bring a willful misconduct action
against a manufacturer only after they have exhausted their remedies under the
compensation program. A willful misconduct action could be brought
against us if an individual(s) has exhausted his or her remedies under the
compensation program, which could thereby expose us to
liability. Furthermore, there is no assurance that the Secretary of
Health and Human Services will issue a declaration under the Public Readiness
Act to establish a compensation fund. We may also become subject to
standard product liability suits and other third party claims if products we
develop which fall outside of the Public Readiness Act cause injury or if
treated individuals subsequently become infected or otherwise suffer adverse
effects from such products.
Healthcare
reform measures and other statutory or regulatory changes could adversely affect
our business.
The
pharmaceutical and biotechnology industries are subject to extensive regulation,
and from time to time legislative bodies and governmental agencies consider
changes to such regulations that could have significant impact on industry
participants. For example, in light of certain highly-publicized safety issues
regarding certain drugs that had received marketing approval, the U.S. Congress
is considering various proposals regarding drug safety, including some which
would require additional safety studies and monitoring and could make drug
development more costly. We are unable to predict what additional legislation or
regulation, if any, relating to safety or other aspects of drug development may
be enacted in the future or what effect such legislation or regulation would
have on our business.
35
The
business and financial condition of pharmaceutical and biotechnology companies
are also affected by the efforts of governments, third-party payors and others
to contain or reduce the costs of healthcare to consumers. In the United States
and various foreign jurisdictions there have been, and we expect that there will
continue to be, a number of legislative and regulatory proposals aimed at
changing the healthcare system, such as proposals relating to the reimportation
of drugs into the U.S. from other countries (where they are then sold at a lower
price) and government control of prescription drug pricing. The pendency or
approval of such proposals could result in a decrease in our share price or
limit our ability to raise capital or to obtain strategic collaborations or
licenses.
The
current disruptions in the financial markets could affect our ability to obtain
additional debt financing on favorable terms (or at all) and have other adverse
effects on us.
The
United States credit markets have recently experienced historic dislocations and
liquidity disruptions which have caused financing to be unavailable in many
cases and even if available caused spreads on prospective debt financings to
widen considerably. These circumstances have materially impacted
liquidity in the debt markets, making financing terms for borrowers able to find
financing less attractive, and in many cases have resulted in the unavailability
of certain types of debt financing. Continued uncertainty in the
credit markets may negatively impact our ability to access additional debt
financing on favorable terms or at all. In addition, Federal
legislation to deal with the current disruptions in the financial markets could
have an adverse affect on our financial condition and results of
operations.
We
will need to enter into collaborative arrangements for the manufacturing and
marketing of our drug candidates, or we will have to develop the expertise,
obtain the additional capital and invest the resources to perform those
functions internally.
We do not
have the staff or facilities to manufacture or market any of the drug candidates
being developed in our catalytic antioxidant program. As a result, we
will need to enter into collaborative arrangements to develop, commercialize,
manufacture and market products that we expect to emerge from our catalytic
antioxidant program, or develop the expertise within our Company. We
might not be successful in entering into such third party arrangements on terms
acceptable to us, if at all. If we are unable to obtain or retain
third-party manufacturing or marketing on acceptable terms, we may be delayed in
our ability to commercialize products, which could have a material adverse
effect on our business, prospects, financial condition and results of
operations. Substantial additional funds and personnel would be
required if we needed to establish our own manufacturing or marketing
operations. We may not be able to obtain adequate funding or
establish these capabilities in a cost-effective or timely manner, which could
have a material adverse effect on our business, prospects, financial condition
and results of operations.
A
failure to obtain or maintain patent and other intellectual property rights
would allow others to develop and sell products similar to ours, which could
impair our business, prospects, financial condition and results of
operations.
The
success of our business depends, in part, on our ability to establish and
maintain adequate protection for our intellectual property, whether owned by us
or licensed from third parties. We rely primarily on patents in the
United States and in other key markets to protect our intellectual
property. If we do not have adequate patent protection, other
companies could develop and sell products that compete directly with ours,
without incurring any liability to us. Patent prosecution,
maintenance and enforcement on a global basis are time-consuming and expensive,
and many of these costs must be incurred before we know whether a product
covered by the claims can be successfully developed or marketed.
Even if
we expend considerable time and money on patent prosecution, a patent
application may never issue as a patent. We can never be certain that
we were the first to invent the particular technology or that we were the first
to file a patent application for the technology because patent applications in
the United States and elsewhere are not typically published for public
inspection for at least 18 months from the date when they are
filed. It is always possible that a competitor is pursuing a patent
for the same invention in the United States as we are and has an earlier
invention date. In some jurisdictions outside of the United States,
priority of invention is determined by the earliest effective filing date, not
the date of invention. Consequently, if a third party pursues the
same invention and has an earlier filing date, patent protection outside the
United States would be unavailable to us. Also, outside the United
States, an earlier date of invention cannot overcome a date of publication that
precedes the earliest effective filing date. Accordingly, the
patenting of our proposed products would be precluded outside the United States
if a prior publication anticipates the claims of a pending application, even if
the date of publication is within a year of the filing of the pending
application.
Even if
patents issue, the patent claims allowed might not be sufficiently broad to
offer adequate protection for our technology against competitive
products. Patent protection differs from country to country, giving
rise to increased competition from other products in countries where patent
coverage is either unavailable, weak or not adequately enforced, if enforced at
all. Once a patent issues, we still face the risk that others will
try to design around our patent or will try to challenge the validity of the
patent. The cost of defending against a challenge to one or more of
our patents could be substantial and even if we prevailed, there could be no
assurance that we would recover damages.
36
If
a third party were to bring an infringement claim against us, we would incur
significant costs in our defense; if the claim were successful, we would need to
develop non-infringing technology or obtain a license from the successful patent
holder, if available.
Our
business also depends on our ability to develop and market products without
infringing on the proprietary rights of others or being in breach of our license
agreements. The pharmaceutical industry is characterized by a large
number of patents, patent filings and frequent and protracted litigation
regarding patent and other intellectual property rights. Many
companies have numerous patents that protect their intellectual property
rights. Third parties might assert infringement claims against us
with respect to our drug candidates and future products. If
litigation were required to determine the validity of a third party’s claims, we
could be required to spend significant time and financial resources, which could
distract our management and prevent us from furthering our core business
activities, regardless of the outcome. If we did not prevail in the
litigation, we could be required to pay damages, license a third party’s
technology, which may not be possible on terms acceptable to us, or at all, or
discontinue our own activities and develop non-infringing technology, any of
which could prevent or significantly delay pursuit of our development
activities.
Protection
of trade secret and confidential information is difficult, and loss of
confidentiality could eliminate our competitive advantage.
In
addition to patent protection, we rely on trade secrets, proprietary know-how
and confidential information to protect our technology. We use
confidentiality agreements with our employees, consultants and collaborators to
maintain the proprietary nature of this technology. However,
confidentiality agreements can be breached by the other party, which would make
our trade secrets and proprietary know-how legally available for use by
others. There is generally no adequate remedy for breach of
confidentiality obligations. In addition, the competitive advantage
afforded by trade secrets is limited because a third party can independently
discover or develop something identical to our own trade secrets or know-how,
without incurring any liability to us.
In
addition, if our current or former employees, consultants or collaborators were
to use information improperly obtained from others (even if unintentional), we
may be subject to claims as to ownership and rights in any resulting know-how or
inventions.
If
we cannot retain or hire qualified personnel or maintain our collaborations, our
programs could be delayed and may be discontinued.
As of
December 29, 2010, we had two full-time employees. We utilize
consultants to assist with our operations and are highly dependent on the
services of our executive officers. We do not maintain “key person”
life insurance on any of our personnel. We also are dependent on our
collaborators for our research and development activities. The loss
of key executive officers or collaborators could delay progress in our research
and development activities or result in their termination entirely.
We
believe that our future success will depend in large part upon our ability to
attract and retain highly skilled scientific and managerial
personnel. We face intense competition for these kinds of personnel
from other companies, research and academic institutions, government entities
and other organizations. If we fail to identify, attract and retain
personnel, we may be unable to continue the development of our drug candidates,
which would have a material adverse effect on our business, prospects, financial
condition and results of operations.
We
face the risk of product liability claims which could exceed our insurance
coverage and deplete our cash resources.
The
pharmaceutical and biotechnology industries expose us to the risk of product
liability claims alleging that use of our drug candidates caused an injury or
harm. These claims can arise at any point in the development,
testing, manufacture, marketing or sale of pharmaceutical products and may be
made directly by patients involved in clinical trials of our products, by
consumers or healthcare providers or by organizations selling our
products. Product liability claims can be expensive to defend, even
if the product did not actually cause the alleged injury or harm.
Insurance
covering product liability claims becomes increasingly expensive as a product
candidate moves through the development pipeline to
commercialization. We have limited product liability insurance
coverage for our clinical trials and this coverage may not be sufficient to
cover us against some or all potential losses due to liability, if any, or to
the expenses associated with defending against liability claims. A
product liability claim successfully asserted against us could exceed our
insurance coverage, require us to use our own cash resources and have a material
adverse effect on our business, financial condition and results of
operations.
In
addition, some of our licensing and other agreements with third parties require
or might require us to maintain product liability insurance. If we
cannot maintain acceptable amounts of coverage on commercially reasonable terms
in accordance with the terms set forth in these agreements, the corresponding
agreements would be subject to termination.
37
The
costs of compliance with environmental, safety and similar laws could increase
our cost of doing business or subject us to liability in the event of
noncompliance.
Our
business is subject to regulation under state and federal laws regarding
occupational safety, laboratory practices, environmental protection and the use,
generation, manufacture, storage and disposal of hazardous
substances. We may be required to incur significant costs in the
future to comply with existing or future environmental and health and safety
regulations. Our research activities involve the use of hazardous
materials, chemicals and radioactive compounds. Although we believe
that our procedures for handling such materials comply with applicable state and
federal regulations, we cannot eliminate the risk of contamination or injury
from these materials. In the event of contamination, we could be
liable for any resulting damages, which could have a material adverse effect on
our business, financial condition and results of operations.
We
are subject to intense competition that could materially impact our operating
results.
We may be
unable to compete successfully against our current or future
competitors. The pharmaceutical, biopharmaceutical and biotechnology
industry is characterized by intense competition and rapid and significant
technological advancements. Many companies, research institutions and
universities are working in a number of areas similar to our primary fields of
interest to develop new products. There also is intense competition
among companies seeking to acquire products that already are being
marketed. Many of the companies with which we compete have or are
likely to have substantially greater research and product development
capabilities and financial, technical, scientific, manufacturing, marketing,
distribution and other resources than at least some of our present or future
strategic partners or licensees.
As a
result, these competitors may:
|
·
|
Succeed
in developing competitive products sooner than us or our strategic
partners or licensees;
|
|
·
|
Obtain
FDA and other regulatory approvals for their products before approval of
any of our products;
|
|
·
|
Obtain
patents that block or otherwise inhibit the development and
commercialization of our drug
candidates;
|
|
·
|
Develop
products that are safer or more effective than our
products;
|
|
·
|
Devote
greater resources to marketing or selling their
products;
|
|
·
|
Introduce
or adapt more quickly to new technologies or scientific
advances;
|
|
·
|
Introduce
products that render our products
obsolete;
|
|
·
|
Withstand
price competition more successfully than us or our strategic partners or
licensees;
|
|
·
|
Negotiate
third-party strategic alliances or licensing arrangements more
effectively; or
|
|
·
|
Take
advantage of other opportunities more
readily.
|
Currently,
there are three drugs approved as radiation protection
agents. Amifostine (Ethyol®) is marketed by MedImmune, Inc. for use
in reduction of chemotherapy-induced kidney toxicity and radiation-induced
xerostomia (damage to the salivary gland). KepivanceTM (palifermin)
is marketed by Amgen, Inc. for use in the treatment of severe oral mucositis
(mouth sores) in patients with hematologic (blood) cancers. Salagen
Tablets (pilocarpine hydrochloride) is marketed by MGI Pharma in the United
States as a treatment for the symptoms of xerostomia induced by radiation
therapy in head and neck cancer patients. However, there are also
many companies working to develop pharmaceuticals that act as a radiation
protection agent.
Acceptance
of our products in the marketplace is uncertain, and failure to achieve market
acceptance will harm our business.
Even if
approved for marketing, our products may not achieve market
acceptance. The degree of market acceptance will depend upon a number
of factors, including:
|
|
·
|
the
receipt of regulatory approvals for the indications that we are
studying;
|
|
|
·
|
the
establishment and demonstration in the medical community of the safety,
clinical efficacy and cost-effectiveness of our products and their
potential advantages over existing therapeutic
products;
|
|
|
·
|
marketing
and distribution support;
|
|
|
·
|
the
introduction, market penetration and pricing strategies of competing and
future products; and
|
|
|
·
|
coverage
and reimbursement policies of governmental and other third-party payors
such as insurance companies, health maintenance organizations and other
plan administrators.
|
Physicians,
patients, payors or the medical community in general may be unwilling to accept,
purchase, utilize or recommend any of our products.
We
may be required to pay milestone and other payments relating to the
commercialization of our products.
Our
agreements by which we acquired rights to our drug candidates provide for
milestone payments by us upon the occurrence of certain regulatory filings and
approvals related to the acquired products. In the event that we
successfully develop our drug candidates, these milestone payments could be
significant. In addition, our agreements require us to pay a royalty
interest on worldwide sales. Also, any future license, collaborative
or other agreements we may enter into in connection with our development and
commercialization activities may require us to pay significant milestone,
license and other payments in the future.
38
We are continually evaluating our
business strategy, and may modify this strategy in light of developments in our
business and other factors.
We
continue to evaluate our business strategy and, as a result, may modify this
strategy in the future. In this regard, we may, from time to time, focus our
development efforts on different drug candidates or may delay or halt the
development of our drug candidates. In addition, as a result of
changes in our strategy, we may also change or refocus our existing drug
discovery, development, commercialization and manufacturing
activities.
Our short-term investments,
marketable securities and restricted investments are subject to certain risks
which could materially adversely affect our overall financial
position.
We invest
our cash in accordance with an established internal policy and customarily in
instruments which historically have been highly liquid and carried relatively
low risk. However, the capital and credit markets have been experiencing extreme
volatility and disruption. Over the past few years, the volatility and
disruption have reached unprecedented levels. We maintain a portfolio of
investments in short-term investments, marketable debt securities and restricted
investments, which are recorded at fair value. Certain of these transactions
expose us to credit risk in the event of default of the issuer. To minimize our
exposure to credit risk, we invest in securities with strong credit ratings.
Should any of our short-term investments, marketable securities or restricted
investments lose value or have their liquidity impaired, it could materially and
adversely affect our overall financial position by imperiling our ability to
fund our operations and forcing us to seek additional financing sooner than we
would otherwise. Such financing may not be available on commercially attractive
terms or at all.
Our insurance policies are expensive
and protect us only from some business risks, which could leave us exposed to
significant, uninsured liabilities.
We do not
carry insurance for all categories of risk that our business may encounter. We
currently maintain general liability, property, auto, workers’ compensation,
products liability, fiduciary and directors’ and officers’ insurance policies.
We do not know, however, if we will be able to maintain existing insurance with
adequate levels of coverage. For example, the premiums for our directors’ and
officers’ insurance policy have increased in the past and may increase in the
future, and this type of insurance may not be available on acceptable terms or
at all in the future. Any significant uninsured liability may require us to pay
substantial amounts, which would adversely affect our cash position and results
of operations.
We
may have a limitation on the use of net operating loss carryforwards and tax
credits.
Our
ability to utilize our net operating loss carryforwards, or NOLs, and tax
credits may be limited if we undergo or have undergone an ownership change, as
defined in section 382 of the Internal Revenue Code, as a result of changes in
the ownership of outstanding stock. An ownership change generally occurs if the
percentage of stock owned by one or more stockholders who own, directly or
indirectly, 5% or more of the value of our outstanding stock (or are otherwise
treated as 5% stockholders under section 382 and the regulations promulgated
thereunder) has increased by more than 50 percentage points over the lowest
percentage of our outstanding stock owned by these stockholders at any time
during the testing period, which is generally the three-year period preceding
the potential ownership change. In the event of an ownership change, section 382
imposes an annual limitation on the amount of post-ownership change taxable
income a corporation may offset with pre-ownership change NOLs.
We
are exposed to risks if we are unable to comply with changes to laws affecting
public companies, including the Sarbanes-Oxley Act of 2002 and the Dodd-Frank
Act of 2010, and also to increased costs associated with complying with such
laws.
Laws and
regulations affecting public companies in the U.S., including the provisions of
the Sarbanes-Oxley Act of 2002 and the Dodd-Frank Act of 2010, will cause us to
incur increased costs as we evaluate the implications of new rules and respond
to new requirements. Delays or a failure to comply with the new laws,
rules and regulations could result in enforcement actions, the assessment of
other penalties and civil suits. These laws and regulations make it
more expensive for us under indemnities provided by the Company to our officers
and directors and may make it more difficult for us to obtain certain types of
insurance, including liability insurance for directors and officers; as such, we
may be forced to accept reduced policy limits and coverage or incur
substantially higher costs to obtain the same or similar
coverage. The impact of these events could also make it more
difficult for us to attract and retain qualified persons to serve on our Board
of Directors, or as executive officers. We may be required to hire
additional personnel and utilize additional outside legal, accounting and
advisory services — all of which could cause our general and administrative
costs to increase beyond what we currently have planned.
We
have reported a material weakness in the effectiveness of our internal control
over financial reporting, and if we cannot maintain effective internal controls
or provide reliable financial and other information, investors may lose
confidence in our SEC reports.
In this
Annual Report on Form 10-K, we are reporting a material weakness in the
effectiveness of our internal control over financial reporting related to
adequate segregation of duties and financial reporting close processes, which
are described in more detail below under the heading “Controls and Procedures.”
Based on these material weaknesses, we are also reporting in this Annual Report
on Form 10-K that our disclosure controls and procedures were not effective as
of September 30, 2010.
Effective
internal control over financial reporting and disclosure controls and procedures
are necessary for us to provide reliable financial and other reports and
effectively prevent fraud. If we cannot maintain effective internal
control or disclosure controls and procedures, or provide reliable financial or
SEC reports or prevent fraud, investors may lose confidence in our SEC reports,
our operating results and the trading price of our common stock could suffer and
we may become subject to litigation.
39
Our
corporate compliance program cannot guarantee that we are in compliance with all
potentially applicable regulations.
The
development, manufacturing, pricing, sales, coverage and reimbursement of our
products, together with our general operations, are subject to extensive
regulation by federal, state and other authorities within the United States and
numerous entities outside of the United States. While we have
developed and instituted a corporate compliance program based on what we believe
are the current best practices, we cannot provide any assurance that
governmental authorities will find that our business practices comply with
current or future administrative or judicial interpretations of potentially
applicable laws and regulations. If we fail to comply with any of
these laws and regulations, we could be subject to a range of regulatory
actions, including suspension or termination of clinical trials, the failure to
approve a product candidate, restrictions on our products or manufacturing
processes, withdrawal of products from the market, significant fines, or other
sanctions or litigation.
We
may become involved in the future in legal proceedings that, if adversely
adjudicated or settled, could materially impact our financial
condition.
As a
pharmaceutical company, we may become a party to litigation in the ordinary
course of our business, including, among others, matters alleging product
liability, patent or other intellectual property rights infringement, patent
invalidity or breach of commercial contract. In general, litigation
claims can be expensive and time consuming to bring or defend against and could
result in settlements or damages that could significantly impact results of
operations and financial condition.
We
are prohibited from taking certain actions and entering into certain
transactions without the consent of certain holders of our Common Stock and
warrants.
For as
long as certain warrants that were issued on October 6, 2009, July 30, 2010 and
August 12, 2010 are outstanding, we are prohibited from taking certain actions
or entering into certain transactions without the prior consent of the warrant
holders of the warrants issued on October 6, 2009, July 30, 2010 and August 12,
2010 (the “2009/2010 Warrant Holders”). Under these warrants, we are
also prohibited from selling the Company to an entity other than one that is
publicly traded.
Even
though our board of directors may determine that any of these actions are in our
best interest or the best interest of our stockholders, we may be unable to
complete them if we do not get the approval of the 2009/2010 Warrant
Holders, who may withhold consent to any transaction in their sole
discretion. The interests of the 2009/2010 Warrant Holders may differ
from those of our stockholders generally. If we are unable to obtain
consent from the 2009/2010 Warrant Holders, we may be unable to complete actions
or transactions that our board of directors determines are in the best interest
of the Company and its stockholders.
Risks
Related to Our Dependence on U.S. Government Grants and Contracts
Most
of our immediately foreseeable future revenues are contingent upon grants and
contracts from the U.S. government and we may not achieve sufficient, if any,
revenues from these agreements to attain profitability.
For the
foreseeable future, we believe our main customer, if any, will be national
governments, primarily the U.S. government. There can be no
assurances that we will receive any grants. The process of obtaining
government contracts is lengthy and uncertain and we will have to compete with
other companies for each contract. There can be no assurances that we
will be awarded any contracts to supply the U.S. or other governments with our
drug candidates as such awards may be made, in whole or in part, to our
competitors. If the U.S. government makes significant future contract
awards for the supply to the U.S. emergency stockpile of a competing product,
our business will be harmed and it is unlikely that we will ultimately be able
to supply that particular treatment or product to foreign governments or other
third parties. Further, changes in government budgets and agendas, or
advances by our competitors, may result in a decreased and de-prioritized
emphasis on procuring the biodefense products we are developing.
Due to
the current economic downturn, the accompanying fall in tax revenues and the
U.S. government’s efforts to stabilize the economy, the U.S. government may be
forced or choose to reduce or delay spending in the biodefense field, which
could decrease the likelihood of future government contract awards or that the
government would procure products from us.
The
U.S. government’s determination to award any contracts may be challenged by an
interested party, such as another bidder, at the Government Accountability
Office (“GAO”) or in federal court. If such a challenge is
successful, a contract may be terminated.
The laws
and regulations governing the procurement of goods and services by the U.S.
government provide procedures by which other bidders and other interested
parties may challenge the award of a government contract. If we are
awarded a government contract, such challenges or protests could be filed even
if there are not any valid legal grounds on which to base the
protest. If any such protests are filed, the government agency may
decide to suspend our performance under the contract while such protests are
being considered by the GAO or the applicable federal court, thus potentially
delaying delivery of goods and services and payment. In addition, we
could be forced to expend considerable funds to defend any potential
award. If a protest is successful, the government may be ordered to
terminate the contract and reselect bids. The government could even
be directed to award a potential contract to one of the other
bidders.
40
Our
business may become subject to audit by the U.S. government and a negative audit
could adversely affect our business.
U.S.
government agencies such as the Defense Contract Audit Agency (the “DCAA”),
routinely audit and investigate government contractors. These
agencies review a contractor’s performance under its contracts, cost structure
and compliance with applicable laws, regulations and standards.
The DCAA
also reviews the adequacy of, and a contractor’s compliance with, its internal
control systems and policies, including the contractor’s purchasing, property,
estimating, compensation and management information systems. Any
costs found to be improperly allocated to a specific contract will not be
reimbursed, while such costs already reimbursed must be refunded. If
an audit uncovers improper or illegal activities, we may be subject to civil and
criminal penalties and administrative sanctions, including:
|
|
·
|
termination
of contracts;
|
|
|
·
|
forfeiture
of profits;
|
|
|
·
|
suspension
of payments;
|
|
|
·
|
fines;
and
|
|
|
·
|
suspension
or prohibition from conducting business with the U.S.
government.
|
In
addition, we could suffer serious reputational harm if allegations of
impropriety were made against us.
Laws
and regulations affecting government contracts make it more costly and difficult
for us to successfully conduct our business.
We must
comply with numerous laws and regulations relating to the formation,
administration and performance of government contracts, which can make it more
difficult for us to retain our rights under these contracts. These
laws and regulations affect how we conduct business with government
agencies. Among the most significant government contracting
regulations that affect our business are:
|
|
·
|
the
Federal Acquisition Regulations, and agency-specific regulations
supplemental to the Federal Acquisition Regulations, which comprehensively
regulate the procurement, formation, administration and performance of
government contracts;
|
|
|
·
|
the
business ethics and public integrity obligations, which govern conflicts
of interest and the hiring of former government employees, restrict the
granting of gratuities and funding of lobbying activities and incorporate
other requirements such as the Anti-Kickback Act and Foreign Corrupt
Practices Act;
|
|
|
·
|
export
and import control laws and regulations;
and
|
|
|
·
|
laws,
regulations and executive orders restricting the use and dissemination of
information classified for national security purposes and the exportation
of certain products and technical
data.
|
Foreign
governments typically also have laws and regulations governing contracts with
their respective agencies. These foreign laws and regulations could
affect how we conduct business and, in some instances, impose added costs on our
business. Any changes in applicable laws and regulations could
restrict our ability to obtain contracts, which could limit our ability to
conduct our business and materially adversely affect our revenues and results of
operations.
Because
we depend on clinical research centers and other contractors for clinical and
non-clinical testing, including testing under the Animal Rule, and for certain
research and development activities, the results of our clinical trial,
non-clinical animal efficacy studies, and research and development activities
are largely beyond our control.
The
nature of studies, clinical trials and our business strategy of outsourcing
substantially all of our research and development and manufacturing work require
that we rely on clinical research centers and other contractors to assist us
with research and development, clinical and non-clinical testing (including
animal efficacy studies under the Animal Rule), patient enrollment and other
activities. As a result, our success depends largely on the success
of these third parties in performing their responsibilities. Although
we prequalify our contractors and believe that they are fully capable of
performing their contractual obligations, we cannot directly control the
adequacy and timeliness of the resources and expertise that they apply to these
activities. Furthermore, we have to compete with other biodefense
companies for access to this limited pool of highly specialized
resources. If our contractors do not perform their obligations in an
adequate and timely manner or we are unable to enter into contracts with them
because of prior commitments to our competitors, the pace of clinical or
non-clinical development, regulatory approval and commercialization of our drug
candidates could be significantly delayed and our prospects could be adversely
affected.
41
Data
obtained from clinical trials is susceptible to varying interpretations, which
could delay, limit or prevent regulatory clearances.
Data
already obtained, or obtained in the future, from pre-clinical studies,
non-clinical studies and clinical trials does not necessarily predict the
results that will be obtained from later pre-clinical studies and clinical
trials. Moreover, pre-clinical and clinical data is susceptible to varying
interpretations, which could delay, limit or prevent regulatory approval.
A number of companies in the pharmaceutical industry have suffered significant
setbacks in advanced clinical trials, even after promising results in earlier
trials. The failure to adequately demonstrate the safety and effectiveness
of an intended product under development could delay or prevent regulatory
clearance of the drug candidate, which would result in delays to
commercialization and could materially harm our business. Our studies and
clinical trials may not demonstrate sufficient levels of safety and efficacy
necessary to obtain the requisite regulatory approvals for our drugs, and our
proposed drugs may not be approved for marketing.
We may
encounter delays or rejections based on additional government regulation from
future legislation or administrative action or changes in FDA policy during the
period of development, clinical trials and FDA regulatory review. We may
encounter similar delays in foreign countries. If any of our products are
approved for commercialization, sales of the products outside the U.S. would be
subject to foreign regulatory approvals that vary from country to country.
The time required to obtain approvals from foreign countries may be shorter or
longer than that required for FDA approval, and requirements for foreign
licensing may differ from FDA requirements. We may be unable to obtain
requisite approvals from the FDA or foreign regulatory authorities, and even if
obtained, such approvals may not be on a timely basis, or they may not cover the
uses that we request.
Even if
we do ultimately receive FDA approval for any of our drug candidates, these drug
candidates will be subject to extensive ongoing regulation, including
regulations governing manufacturing, labeling, packaging, testing, dispensing,
prescription and procurement quotas, record keeping, reporting, handling,
shipment and disposal of any such drug. Failure to obtain and maintain
required registrations or to comply with any applicable regulations could
further delay or preclude development and commercialization of our drugs and
subject us to enforcement action.
Government
contracts customarily contain provisions that give the government substantial
rights and remedies, many of which are not typically found in commercial
contracts, including provisions that allow the government to:
|
|
•
|
terminate existing contracts, in
whole or in part, for any reason or no
reason;
|
|
|
•
|
unilaterally reduce or modify
contracts or subcontracts, including equitable price
adjustments;
|
|
|
•
|
cancel multi-year contracts and
related orders if funds for contract performance for any subsequent year
become unavailable;
|
|
|
•
|
decline to exercise an option to
renew a contract;
|
|
|
•
|
exercise an option to purchase
only the minimum amount specified in a
contract;
|
|
|
•
|
decline to exercise an option to
purchase the maximum amount specified in a
contract;
|
|
|
•
|
claim rights to products,
including intellectual property, developed under the
contract;
|
|
|
•
|
take actions that result in a
longer development timeline than
expected;
|
|
|
•
|
audit and object to the
contractor’s contract-related costs and fees, including allocated indirect
costs;
|
|
|
•
|
direct the course of a
development program in a manner not chosen by the government
contractor;
|
|
|
•
|
suspend or debar the contractor
from doing business with the government or a specific government
agency;
|
|
|
•
|
pursue criminal or civil remedies
under the False Claims Act and False Statements Act;
and
|
|
|
•
|
control or prohibit the export of
products.
|
Generally,
government contracts contain provisions permitting unilateral termination or
modification, in whole or in part, at the government’s convenience. Under
general principles of government contracting law, if the government terminates a
contract for convenience, the terminated company may recover only its incurred
or committed costs, settlement expenses and profit on work completed prior to
the termination.
If the
government terminates a contract for default, the defaulting company is entitled
to recover costs incurred and associated profits on accepted items only and may
be liable for excess costs incurred by the government in procuring undelivered
items from another source. Some government contracts grant the
government the right to use, for or on behalf of the U.S. government, any
technologies developed by the contractor under the government contract. If we
were to develop technology under a contract with such a provision, we might not
be able to prohibit third parties, including our competitors, from using that
technology in providing products and services to the government.
42
Risks
Related to Owning Our Stock
Our
principal stockholders own a significant percentage of our outstanding common
stock and are, and will continue to be, able to exercise significant influence
over our affairs.
As of
December 29, 2010, Xmark Opportunity Partners, LLC (“Xmark”) possessed voting
power over 41,458,327 shares, or 69.5%, of our outstanding common stock as of
such date, through its management of Goodnow Capital, L.L.C. (“Goodnow”), Xmark
Opportunity Fund, L.P., Xmark Opportunity Fund, Ltd. and Xmark JV Investment
Partners, LLC (collectively, the “Xmark Funds”), and through a voting trust
agreement by and among Biomedical Value Fund, L.P., Biomedical Value Fund, Ltd.,
Xmark Opportunity Partners, LLC and the Company (the “Xmark Voting
Trust”) with respect to 1,000,000 shares. As a result, Xmark is able
to determine a significant part of the composition of our board of directors,
holds significant voting power with respect to matters requiring stockholder
approval and is able to exercise significant influence over our
operations. The interests of Xmark may be different than the
interests of other stockholders on these and other matters. This
concentration of ownership also could have the effect of delaying or preventing
a change in our control or otherwise discouraging a potential acquirer from
attempting to obtain control of us, which could reduce the price of our common
stock.
David
Cavalier, an employee and our Chairman of the Board of Directors, and Xmark with
which he is affiliated, possess voting power of 69.5% of our outstanding common
stock as of December 29, 2010. Accordingly, Mr. Cavalier
currently has, and will continue to have, a significant influence over the
outcome of all corporate actions requiring stockholder approval.
Also as
of December 29, 2010, Efficacy Capital Ltd. (“Efficacy Capital”) owned 9,800,000
shares, or 16.4%, of our outstanding common stock as of such date, through its
management of Efficacy Biotech Master Fund Ltd. As a result, Efficacy
Capital is able to determine a part of the composition of our board of
directors, holds significant voting power with respect to matters requiring
stockholder approval and is able to exercise significant influence over our
operations. The interests of Efficacy Capital may be different than
the interests of other stockholders on these and other matters. This
concentration of ownership could also have the effect of delaying or preventing
a change in our control or otherwise discouraging a potential acquirer from
attempting to obtain control of us, which could reduce the price of our common
stock.
Our
executive officers and directors and holders of greater than five percent of our
outstanding common stock, together with entities that may be deemed affiliates
of, or related to, such persons or entities, beneficially owned greater than
85.9% of our outstanding common stock as of December 29, 2010. As a result,
these stockholders, acting together, may be able to control our management and
affairs and matters requiring stockholder approval, including the election of
directors and approval of significant corporate transactions, such as mergers,
consolidations or the sale of substantially all of our assets. The interests of
our current major stockholders may not always coincide with the interests of
other stockholders and they may take actions to advance their respective
interests to the detriment of other stockholders.
We
may need to sell additional shares of our common stock, preferred stock or other
securities to meet our capital requirements and these future sales could cause
dilution and adversely affect our stock price.
Sales of
substantial amounts of capital stock, or the perception that such sales could
occur, could adversely affect the prevailing market price of the common stock
and our ability to raise capital. We may issue additional common
stock in future financing transactions or as incentive compensation for our
executive management and other key personnel, consultants and
advisors. Issuing any equity securities would be dilutive to the
equity interests represented by our then-outstanding shares of common
stock. The market price for our common stock could decrease as the
market takes into account the dilutive effect of any of these
issuances.
In
the event of the conversion of our preferred stock and exercises of currently
outstanding options and warrants, the ownership interests of our current
stockholders could be substantially diluted, which would reduce the market price
of our common stock and could make it more difficult for us to raise funds in
the future.
As of
December 29, 2010, we had 59,684,050shares of common stock
outstanding. We may grant to our employees, directors and consultants
options to purchase shares of our common stock under our 2004 Stock Option
Plan. In addition, as of December 29, 2010, options to purchase
8,012,565 shares were outstanding at exercise prices ranging from $0.29 to
$51.25 per share, with a weighted average exercise price of $1.30 per share, and
3,912,159 shares were reserved for issuance under the 2004 Stock Option
Plan. In addition, as of December 29, 2010, warrants to purchase
68,226,667 shares of common stock were outstanding at exercise prices ranging
from $0.28 to $2.50 per share, with a weighted exercise price of $0.34 per
share. We have also reserved 475,087 shares of common stock for the
conversion of our outstanding Series B Preferred stock.
In
connection with prior collaborations and financing transactions, we also issued
Series B preferred stock and a promissory note convertible into Series B
preferred stock to affiliates of Elan Corporation, plc
(“Elan”). These securities generally are exercisable and convertible
at the option of the Elan affiliates. The exercise or conversion of
all or a portion of these securities would dilute the ownership interests of our
stockholders.
Our
common stock is not listed on a national exchange, is illiquid and is
characterized by low and/or erratic trading volume, and the per share price of
our common stock has fluctuated from $0.15 to $0.70 during the last two
years.
Our
common stock is quoted on the OTC Bulletin Board under the symbol
“AOLS.” An active public market for our common stock is unlikely to
develop as long as we are not listed on a national securities
exchange. Even if listed, the market for our stock may be impaired
because of the limited number of investors, the significant ownership stake of
Efficacy Capital and Xmark (through its management of Goodnow and the Xmark
Funds), and our small market capitalization, which is less than that authorized
for investment by many institutional investors.
43
Historically,
the public market for our common stock has been characterized by low and/or
erratic trading volume, often resulting in price volatility. The
market price of our common stock is subject to wide fluctuations due to factors
that we cannot control, including the results of preclinical and clinical
testing of our products under development, decisions by collaborators regarding
product development, regulatory developments, market conditions in the
pharmaceutical and biotechnology industries, future announcements concerning our
competitors, adverse developments concerning proprietary rights, public concern
as to the safety or commercial value of any products and general economic
conditions.
Furthermore,
the stock market has experienced significant price and volume fluctuation
unrelated to the operating performance of particular companies. These
market fluctuations can adversely affect the market price and volatility of our
common stock.
If
registration rights that we have previously granted are exercised, or if we
grant additional registration rights in the future, the price of our common
stock may be adversely affected.
Upon
receiving notice from Xmark, we are obligated to register with the SEC shares of
common stock and the common stock underlying the warrants to purchase common
stock held by the Xmark. If these securities are registered with the
SEC, they may be sold in the open market. In addition, upon receiving
notice from Elan, we are obligated to register with the SEC shares of common
stock underlying the Series B preferred stock, warrants to purchase Series B
preferred stock and a promissory note held by the Elan affiliates. If
these securities are registered with the SEC, they may be sold in the open
market. We expect that we also will be required to register any
securities sold in future private financings. The sale of a
significant amount of shares in the open market, or the perception that these
sales may occur, could cause the trading price of our common stock to decline or
become highly volatile.
Anti-takeover
provisions in our charter documents and under Delaware law could make an
acquisition of us, which may be beneficial to our stockholders, more difficult
and may prevent attempts by our stockholders to replace or remove our current
management.
Provisions
in our amended and restated certificate of incorporation and bylaws may delay or
prevent an acquisition of us or a change in our management. These
provisions include a prohibition on actions by written consent of our
stockholders and the ability of our board of directors to issue preferred stock
without stockholder approval. In addition, because we are
incorporated in Delaware, we are governed by the provisions of Section 203 of
the Delaware General Corporation Law, which prohibits stockholders owning in
excess of 15% of our outstanding voting stock from merging or combining with
us. These provisions may frustrate or prevent any attempts by our
stockholders to replace or remove our current management by making it more
difficult for stockholders to replace members of our board of directors, which
is responsible for appointing the members of our management.
We
do not expect to pay cash dividends on our common stock for the foreseeable
future.
We have
never paid cash dividends on our common stock and do not anticipate that any
cash dividends will be paid on the common stock for the foreseeable
future. The payment of any cash dividend by us will be at the
discretion of our board of directors and will depend on, among other things, our
earnings, capital, regulatory requirements and financial
condition. Furthermore, the terms of some of our financing
arrangements directly limit our ability to pay cash dividends on our common
stock.
We
may experience significant and unpredictable changes to the liability for
warrants and record significant gains or losses to our statement of operations
in each period the warrants are outstanding.
In June
2008, the Financial Accounting Standards Board ("FASB") ratified what was
originally referred to as Emerging Issues Task Force (“EITF”) Issue No. 07-5
recently codified by FASB as Account Standard Codification Topic 815, which
contains guidance for determining whether an Instrument (or embedded feature) is
indexed to an entity’s own stock. Equity-linked instruments (or
embedded features) that otherwise meet the definition of a derivative are not
accounted for as derivatives if certain criteria are met, one of which is that
the instrument (or embedded feature) must be indexed to the entity’s own
stock. The Company adopted this guidance on October 1, 2009 and began
applying its provisions to outstanding instruments as of that date. A
number of our outstanding warrants to purchase common stock are impacted by this
guidance. As a result, liability recorded for a number of our outstanding
warrants may increase or decrease, sometimes dramatically, from
quarter-to-quarter. An increase in warrant liability for a period will result in
a corresponding charge to our statement of operations for such period and a
decrease in warrant liability for a period will result in a corresponding gain
to our statement of operations for such period. The Company’s outstanding
warrants will continue to be revalued at each balance sheet date, which could
result in significant and unpredictable changes to our reported liabilities and
significant additional gains or losses charged to the statement of operations
for each period regardless of any changes to the Company’s working capital,
liquidity, or business operations.
44
Item
1B. Unresolved Staff Comments.
None.
Item
2. Properties.
None.
Item
3. Legal Proceedings.
None.
Item
4. (Removed and Reserved).
PART
II
Item
5. Market for Registrant’s Common Equity, Related Stockholder Matters
and Issuer Purchases of Equity Securities.
(a)
Price Range of Common Stock
Our
common stock is traded on the OTC Bulletin Board under the symbol
“AOLS.” The following sets forth the quarterly high and low trading
prices as reported by the OTC Bulletin Board for the periods
indicated. These prices are based on quotations between dealers,
which do not reflect retail mark-up, markdown or commissions, and do not
necessarily represent actual transactions.
|
High
|
Low
|
|||||||
|
Fiscal
Year Ended September 30, 2009
|
||||||||
|
October
1, 2008 through December 31, 2008
|
$ | 0.46 | $ | 0.20 | ||||
|
January
1, 2009 through March 31, 2009
|
$ | 0.46 | $ | 0.15 | ||||
|
April
1, 2009 through June 30, 2009
|
$ | 0.40 | $ | 0.20 | ||||
|
July
1, 2009 through September 30, 2009
|
$ | 0.39 | $ | 0.22 | ||||
|
Fiscal
Year Ending September 30, 2010
|
||||||||
|
October
1, 2009 through December 31, 2009
|
$ | 0.55 | $ | 0.24 | ||||
|
January
1, 2010 through March 31, 2010
|
$ | 0.48 | $ | 0.30 | ||||
|
April
1, 2010 through June 30, 2010
|
$ | 0.49 | $ | 0.25 | ||||
|
July
1, 2010 through September 30, 2010
|
$ | 0.56 | $ | 0.31 | ||||
(b)
Approximate Number of Equity Security Holders
As of
November 30, 2010 the number of record holders of our common stock was 83, and
we estimate that the number of beneficial owners was approximately
2,300.
(c)
Dividends
We have
never paid a cash dividend on our common stock and we do not anticipate paying
cash dividends on our common stock in the foreseeable future. If we pay a cash
dividend on our common stock, we also must pay the same dividend on an as
converted basis on our Series B preferred stock. In addition, under the terms of
the warrants to purchase up to 61,822,749 shares of our common stock issued to
Xmark Opportunity Partners, LLC or its affiliates (“Xmark”) in three
transactions (on each of October 6, 2009, July 30, 2010 and August 11, 2010),
and any additional warrants issued pursuant to the put and/or option right
granted in our August 2010 financing, if we were to pay a dividend on our common
stock, the exercise price of these warrants would be reset from $0.28 per share
or $0.50 per share, as applicable, to $0.01 per share and the warrant holders
would also be entitled receive any such dividend paid.
Moreover,
any additional preferred stock to be issued and any future credit facilities
might contain restrictions on our ability to declare and pay dividends on our
common stock. We plan to retain all earnings, if any, for the foreseeable future
for use in the operation of our business and to fund future
growth.
45
(d)
Equity Compensation Plan and
Additional Equity Information as of September 30, 2010
|
Plan category
|
(a)Number of
securities to be
issued upon
exercise of
outstanding
options,
warrants and
rights
|
(b)Weighted-
average
exercise price
of
outstanding
options,
warrants and
rights
|
(c)Number of
securities remaining
available for future
issuance under
equity compensation
plans (excluding
securities reflected
in column (a))
|
|||||||||
|
Equity
compensation plans approved by our stockholders:
|
||||||||||||
|
2004
Stock Option Plan
|
6,035,441 | $ | 0.47 | 3,964,659 | ||||||||
|
1994
Stock Option Plan
|
1,927,124 | $ | 3.89 | 0 | ||||||||
|
Equity
compensation plans and securities not approved by our
stockholders:
|
||||||||||||
|
Warrants
to purchase Common Stock Issued to National Securities
|
50,000 | $ | 0.38 |
Not
applicable
|
||||||||
|
Warrants
to Purchase Common Stock Issued to Dan Delmonico
|
50,000 | $ | 0.49 |
Not
applicable
|
||||||||
|
Warrants
to Purchase Common Stock Issued to Brookstreet Securities
Corporation
|
250,000 | $ | 1.50 |
Not applicable
|
||||||||
|
Total
– Common Stock
|
8,312,465 | 3,964,659 | ||||||||||
|
Convertible
Promissory Note convertible into shares of Series B Preferred Stock Issued
to Elan Pharma International Limited (as of September 30,
2010)(1)(2)
|
73,659 | $ | 9.00 | 2,940 | ||||||||
|
Total
– Series B Preferred Stock
|
73,569 | 2,940 | ||||||||||
(1) As
of September 30, 2010, each share of Series B preferred stock was convertible
into one share of common stock.
(2) The
conversion value of the note will increase by its 11% interest rate until its
maturity on February 8, 2011.
Description
of Equity Compensation Plans and Equity Securities Not Approved by Our
Stockholders
The
warrants to purchase shares of our common stock issued to Brookstreet Securities
Corporation (“Brookstreet”) have not been approved by our
stockholders. In May 2006, we entered into an agreement with
Brookstreet to provide us with financial advisory services for a one-year
period. For these services, we issued five warrants each to purchase
up to 50,000 shares of our common stock with an exercise price of $0.50, $1.00,
$1.50, $2.00 and $2.50 and vesting dates of on May 24, 2006, August 22, 2006,
November 20, 2006, February 18, 2007 and May 19, 2007,
respectively. The warrants are exercisable for five years from the
date of grant and contain standard adjustment provisions in the event the
Company declares a stock dividend or engages in a recapitalization,
reclassification or reorganization of its capital stock. In addition, the
Company is required to give Brookstreet advance notice of a change in control of
the Company during the term of the warrants.
The
warrants to purchase shares of our common stock issued to Dan Delmonico in
September 2009 have not been approved by our stockholders. In
consideration for services provided by Mr. Delmonico to the Company, the Company
issued three warrants each to purchase up to 20,000, 15,000 and 15,000 shares of
our common stock with an exercise price of $0.39, $.50 and $0.60,
respectively. The warrants are exercisable for five years from the
date of grant and contain standard adjustment provisions in the event the
Company declares a stock dividend or engages in a recapitalization,
reclassification or reorganization of its capital stock.
The
warrants to purchase shares of our common stock issued to National Securities
Corporation (“NSC”) have not been approved by our stockholders. In
January 2010, we entered into an agreement with NSC pursuant to which we
retained NSC as a non-exclusive financial advisor for the period from January 6,
2010 through January 6, 2011. For these services, we issued a warrant to
purchase up to 50,000 shares of our common stock with an exercise price of
$0.38. The warrant is exercisable for five years from the date of
grant and contains standard adjustment provisions in the event the Company
declares a stock dividend or engages in a recapitalization, reclassification or
reorganization of its capital stock.
The
convertible promissory note convertible into shares of Series B Preferred Stock
issued to Elan Pharma International Limited (“Elan”) has not been approved by
our stockholders. We issued the note to Elan in exchange for certain
money borrowed from Elan.
46
(e)
Recent Sales of Unregistered Securities
None
(f)
Purchase of Equity Securities by the Issuer and Affiliated
Purchases
None.
Performance
Graph
We are a
smaller reporting company, as defined by Rule 12b-2 of the Securities Exchange
Act of 1934, as amended, and are not required to provide the information
required under this item.
Item
6. Selected Financial Data.
You
should read the following selected financial data in conjunction with our
consolidated financial statements and the notes to those statements and
“Management’s Discussion and Analysis of Financial Condition and Results of
Operations” included elsewhere in this Annual Report on Form 10-K. We
derived the consolidated statements of operations data for the five fiscal years
ended September 30, 2010 and the related consolidated balance sheet data at
those dates from our audited consolidated financial
statements. Except for the consolidated statements of operations for
the fiscal years ended September 30, 2007 and 2006 and the consolidated balance
sheet data at September 30, 2008, 2007 and 2006, each of these consolidated
financial statements is included elsewhere in this Annual Report on Form
10-K.
Statement
of Operations Data:
|
Year Ended September 30,
|
||||||||||||||||||||
|
2010
|
2009
|
2008
|
2007
|
2006
|
||||||||||||||||
|
(in
thousands, except per share data)
|
||||||||||||||||||||
|
Revenue:
|
||||||||||||||||||||
|
Grant
income and contract revenue
|
$ | — | — | $ | — | $ | — | $ | 92 | |||||||||||
|
Costs
and expenses:
|
||||||||||||||||||||
|
Research
and development
|
1,690 | 711 | 977 | 1,381 | 3,480 | |||||||||||||||
|
General
and administrative
|
1,954 | 1,292 | 1,540 | 1,919 | 2,216 | |||||||||||||||
|
Total
costs and expenses
|
3,644 | 2,003 | 2,517 | 3,300 | 5,696 | |||||||||||||||
|
Loss
from operations
|
(3,644 | ) | (2,003 | ) | (2,517 | ) | (3,300 | ) | (5,604 | ) | ||||||||||
|
Other
income (expenses), net
|
(21,347 | ) | 144 | (405 | ) | 225 | (118 | ) | ||||||||||||
|
Interest
income (expense), net
|
(878 | ) | (437 | ) | (51 | ) | 51 | (6 | ) | |||||||||||
|
Net
loss
|
(25,869 | ) | (2,296 | ) | (2,973 | ) | (3,024 | ) | (5,728 | ) | ||||||||||
|
Preferred
stock dividend and accretion
|
— | — | — | — | (81 | ) | ||||||||||||||
|
Net
loss attributable to common stockholders
|
$ | (25,869 | ) | (2,296 | ) | $ | (2,973 | ) | $ | (3,024 | ) | $ | (5,809 | ) | ||||||
|
Basic
net loss per share attributable to common stockholders
|
$ | (0.46 | ) | (0.07 | ) | $ | (0.09 | ) | $ | (0.10 | ) | $ | (0.31 | ) | ||||||
|
Diluted
net loss per share attributable to common stockholders
|
$ | (0.46 | ) | (0.07 | ) | $ | (0.11 | ) | $ | (0.10 | ) | $ | (0.31 | ) | ||||||
|
Weighted
average common shares outstanding:
|
||||||||||||||||||||
|
Basic
|
56,817 | 34,789 | 31,953 | 30,239 | 18,926 | |||||||||||||||
|
Diluted
|
56,817 | 34,789 | 32,217 | 30,239 | 18,926 | |||||||||||||||
47
Balance
Sheet Data:
|
September 30,
|
||||||||||||||||||||
|
2010
|
2009
|
2008
|
2007
|
2006
|
||||||||||||||||
|
(in
thousands)
|
||||||||||||||||||||
|
Cash
and cash equivalents and marketable securities
|
2,355 | 646 | 399 | 1,727 | 3,324 | |||||||||||||||
|
Working
capital (deficiency)
|
781 | 5 | (1,336 | ) | 1,538 | 1,581 | ||||||||||||||
|
Total
assets
|
2,433 | 811 | 1,120 | 1,931 | 3,554 | |||||||||||||||
|
Long-term
portion of capital lease obligations and notes payable
|
— | 1,194 | 266 | 483 | — | |||||||||||||||
|
Total
liabilities
|
29,169 | 1,968 | 2,157 | 751 | 1,847 | |||||||||||||||
|
Total
stockholders’ equity (deficit)
|
(26,736 | ) | (1,157 | ) | (1,037 | ) | 1,180 | 1,707 | ||||||||||||
Item
7. Management’s Discussion and Analysis of Financial Condition and
Results of Operations.
Introduction
You
should read the following discussion in conjunction with our consolidated
financial statements and the notes appearing elsewhere in this Annual Report on
Form 10-K. The following discussion contains forward-looking statements that
involve risks and uncertainties. Our actual results could differ materially from
those anticipated in the forward-looking statements as a result of various
factors, including those discussed in Item 1A - “Risk Factors” and elsewhere in
this Annual Report on Form 10-K.
Overview
We are
developing a new class of catalytic antioxidant compounds as a medical
countermeasure against biological, chemical and radiological weapons as well as
for diseases and disorders of the central nervous system, respiratory system,
autoimmune system and oncology. Our initial target indications are as a
protective agent against the effects of acute radiation syndrome, sulfur mustard
gas exposure and chlorine gas exposure. We have reported positive safety results
from two Phase I clinical trials of AEOL 10150, our lead drug candidate, with no
serious adverse events noted.
We had
net losses of approximately $25,869,000 and $2,296,000 for the fiscal years
ended September 30, 2010 and 2009, respectively. We had an accumulated deficit
of approximately $182,711,000 at September 30, 2010. We have not yet generated
any revenue from product sales and do not expect to receive any product revenue
in the foreseeable future, if at all.
We have
not had any recurring revenue and therefore we have relied on public or private
equity offerings, debt financings, collaboration arrangements and grants to
finance our operations.
Corporate
Matters
On August
1, 2008, we completed a private placement pursuant to which we agreed to sell to
certain investors (the “August 2008 Investors”) units comprised of our senior
unsecured convertible notes (the “Notes”), in an aggregate principal amount of
up to $5,000,000, which bore interest at a rate of 7% per year and matured on
the 30-month anniversary of their date of issuance, and warrants to purchase up
to an aggregate of 10,000,000 additional shares of our common stock, each with
an initial exercise price of $0.50 per share, subject to adjustment as provided
in the warrants. Each unit (collectively, the “August 2008 Units”) is comprised
of $1,000 in Note principal and warrants to purchase up to 2,000 shares of our
common stock, and had a purchase price of $1,000. On August 1, 2008, we sold and
issued to the August 2008 Investors an aggregate of 500 August 2008 Units
comprised of Notes in the aggregate principal amount of $500,000 and warrants to
purchase up to 1,000,000 shares of our common stock for an aggregate purchase
price of $500,000 (the “August 2008 Financing”). On each of September 4, 2008,
October 1, 2008, November 3, 2008 and December 1, 2008, we sold and issued to
the August 2008 Investors an aggregate of 125 August 2008 Units comprised of
Notes in the aggregate principal amount of $125,000 and warrants to purchase up
to 250,000 shares of our common stock for an aggregate purchase price of
$125,000. In total, we sold and issued Notes in the aggregate principal amount
of $1,000,000 and warrants to purchase up to 2,000,000 shares of our common
stock for an aggregate purchase price of $1,000,000.
On March
30, 2009, we completed a private placement of 5,357,143 shares of our common
stock and warrants to purchase up to an aggregate of 13,392,857 shares of common
stock with an initial exercise price of $0.35 per share and a five year term for
aggregate net proceeds of $1,409,000 (the “March 2009
Financing”).
48
On
October 6, 2009, we completed a private placement of 5,892,857 shares of our
common stock and warrants to purchase up to an aggregate of 11,785,714
additional shares of common stock, with an initial exercise price of $0.28 per
share and a seven year term for aggregate proceeds of $1,650,000 (the “October
2009 Financing”). The warrants contain a “cashless exercise” feature that allows
the holder to exercise the warrants without a cash payment to the Company under
certain circumstances; contain a dividend participation right which allows the
holder to receive any cash dividends paid on the Common Stock without exercising
the warrant and contain a provision that provides for the reduction of the
exercise price to $0.01 in the event of any such payment of cash dividends by
the Company or upon a change of control and contain anti-dilution provisions in
the event of a stock dividend or split, dividend payment or other issuance,
reorganization, recapitalization or similar event. We also granted
the investors in the October 2009 Financing (the “October 2009 Investors”) the
option to acquire, collectively, up to an additional 5,892,857 units (the
“Additional Units”), comprised of an aggregate of 5,892,857 shares of our common
stock and warrants to purchase up to an aggregate of 11,785,714 additional
shares of common stock at the per Additional Unit purchase price of $0.28 (the
“October 2009 Call Option”). In addition, the October 2009 Investors granted us
the option to require the October 2009 Investors, severally and not jointly, to
acquire up to 5,892,857 Additional Units, less any Additional Units acquired
under the October 2009 Call Option, at the per Additional Unit purchase price of
$0.28 (the “October 2009 Put Option”).
In
addition, in connection with the October 2009 Financing, the August 2008
Investors agreed to convert all $1,000,000 of the Notes into common stock at a
conversion rate of $0.35 per share and to exchange their remaining option to
purchase an additional $4,000,000 in Notes for warrants to purchase up to
14,285,714 shares of common stock in substantially the same of form and terms of
the warrants issued in the October 2009 Financing, including an initial exercise
price of $0.28 per share. As consideration for the August 2008 Investors to
convert the Notes, we agreed to exchange warrants to purchase up to 2,000,000
shares of common stock issued to the August 2008 Investors in connection with
the sale of the Notes, warrants to purchase up to 2,150,000 shares of common
stock issued to certain investors and one of their affiliates in connection with
a financing completed in November 2005 and warrants to purchase up to 13,392,857
shares of common stock issued to certain investors in connection with the March
2009 Financing for warrants to purchase up to an aggregate of 17,542,857 shares
of our common stock in substantially the same form and terms of the warrants
issued in the October 2009 Financing, including an initial exercise price of
$0.28 per share, subject to adjustment as provided in the warrants.
On
December 24, 2009, we entered into an amendment (the “Amendment”) to the
purchase agreement entered into in connection with the October 2009 Financing,
pursuant to which we agreed to lower the conversion price of the Notes from
$0.35 per share to $0.28 per share and as a result, issued to the October 2009
Investors an additional 714,286 shares of our common stock upon conversion of
the Notes (the “Issuance”). The Amendment was executed to resolve a
misunderstanding regarding one of the financing terms between us and the October
2009 Investors. We did not receive any proceeds from the Issuance. The fair
value of our common stock on the date of issuance was $343,000 and was charged
to the statement of operations as interest expense.
On July
25, 2010, we gave written notice to the October 2009 Investors that we were
exercising the October 2009 Put Option in full, effective July 30, 2010. As
a result of the exercise, we received $1.65 million in gross proceeds from the
October 2009 Investors in exchange for the sale and issuance of an aggregate of
5,892,857 Additional Units, comprised of an aggregate of 5,892,857 shares of our
common stock and warrants to purchase up to an aggregate of 11,785,714
additional shares of common stock at a purchase price of $0.28. Net cash
proceeds from the exercise of the October 2009 Put Option were approximately
$1.6 million after legal costs associated with the exercise and subsequent
issuance of stock and warrants.
On August
11, 2010, we completed a private placement of 2,500,000 units (the “August 2010
Units”), comprised of an aggregate of 2,500,000 shares of common stock and
warrants to purchase up to an aggregate of 1,875,000 additional shares of common
stock (the “August 2010 Warrants”), with an initial exercise price of $0.50 per
share, subject to adjustment as provided in the August 2010 Warrants, with each
August 2010 Unit representing one share of common stock and an August 2010
Warrant to purchase 0.75 share of common stock, at a purchase price of $0.40 per
August 2010 Unit for aggregate gross proceeds of $1,000,000. The
August 2010 Warrants are exercisable for a seven year period from their date of
issuance; contain a “cashless exercise” feature that allows the holder to
exercise the August 2010 Warrants without a cash payment to us under certain
circumstances; contain a dividend participation right which allows the holder to
receive any cash dividends paid on the common stock without exercising the
August 2010 Warrant; contain a provision that provides for the reduction of the
exercise price to $0.01 in the event of any such payment of cash dividends by us
or upon a change of control; and contain anti-dilution provisions in the event
of a stock dividend or split, dividend payment or other issuance,
reorganization, recapitalization or similar event.
In
connection with our August 2010 financing, we also granted the investors in the
August 2010 financing (the “August 2010 Investors”) the option to acquire,
collectively, up to an additional 2,500,000 units, comprised of an aggregate of
2,500,000 shares of common stock and warrants to purchase up to an aggregate of
1,875,000 additional shares of common stock at an exercise price of $0.50 per
share (the “August 2010 Call Option”). In addition, the August 2010
Investors granted us the option to require these August 2010 Investors,
severally and not jointly, to acquire up to 2,500,000 additional units, less any
additional units acquired under the August 2010 Call Option, at the per
additional unit purchase price of $0.40 (the “August 2010 Put Option”). On
December 28, 2010, the investors exercised their Call Option from the August
2010 financing. Under the terms of the option, the Company received
$1 million in cash in exchange for the issuance of 2,500,000 shares of common
stock and 1,875,000 warrants to purchase common stock at an exercise price of
$0.50 per share.
49
Results
of Operations
Fiscal
Year Ended September 30, 2010 Compared to Fiscal Year Ended September 30,
2009
We had a
net loss of $25,869,000, which included a non-cash charge of $21,347,000 related
to increases in the fair value of warrants, for the fiscal year ended September
30, 2010, versus a net loss of $2,296,000 for fiscal year ended September 30,
2009.
We did
not generate any revenue during fiscal 2010 or fiscal 2009.
Research
and Development
Research
and development (“R&D”) expenses increased by about $979,000, or 138%, to
approximately $1,690,000 for the fiscal year ended September 30, 2010 from
approximately $711,000 for the fiscal year ended September 30,
2009. R&D expenses were higher during the fiscal year ended
September 30, 2010 versus September 30, 2009 due to an increase in
research activities to support our lung acute radiation syndrome (“ARS”)
development program and the retention of a consultant to assist in the writing
of proposals for government funding. For the fiscal year ended
September 30, 2010, consultant expenses increased by about $207,000 due to
costs associated with the aforementioned consultant. Preclinical fees increased
about $692,000 over the comparable period in 2009 due to increased animal
studies to support our ARS development program. The increase also reflected the
initiation of production of a compound for oncology studies anticipated to begin
in fiscal year 2011, for which manufacturing expenses were about $110,000. We
currently have seven development programs in progress: the study of our drug
candidates as a potential countermeasure against the effects of sulfur mustard
gas on the lung and skin, as a protectant against the effects of radiation on
the lungs and on the gastro-intestinal tract, as a countermeasure against the
effects of chlorine gas, as a potential treatment for epilepsy and for the
potential treatment for Parkinson’s disease.
In
December 2009, we were informed by the Biologic Advanced Research and
Development Authority (BARDA) that it had been chosen to submit a full proposal
for funding of our Lung ARS program from its current stage to the U.S. Food and
Drug Administration (“FDA”) for approval, based on a summary “white paper”
submitted by us earlier in 2009. In July of 2010, we were informed by
BARDA that our proposal had been chosen, and we entered into negotiations with
BARDA for a contract to fund the development of AEOL 10150 as a medical
countermeasure for pulmonary sub-syndrome of acute radiation
syndrome.
R&D
expenses for our antioxidant program have totaled approximately $37,212,000 from
inception through September 30, 2010. Because of the uncertainty of
our research and development and clinical studies, we are unable to predict the
total level of spending on the program or the program completion
date. However, we expect R&D expenses during fiscal year 2011
will be higher than fiscal 2010 if we are awarded the BARDA
contract. We anticipate that much of the increase in R&D spending
should be reimbursed under that contract, if awarded. If the contract
is not awarded, R&D expenses are expected to be in line with the level of
expenditures in 2010.
General
and Administrative
General
and administrative (“G&A”) expenses include corporate costs required to
support our company, our employees and consultants and our
stockholders. These costs include personnel and outside costs in the
areas of legal, human resources, investor relations and
finance. Additionally, we include in general and administrative
expenses such costs as rent, repair and maintenance of equipment, depreciation,
utilities, information technology and procurement costs that we need to support
the corporate functions listed above.
G&A
expenses increased approximately $662,000, or 51%, to approximately $1,954,000
for the fiscal year ended September 30, 2010 from about $1,292,000 for the
fiscal year ended September 30, 2009. Salaries and wages increased by about
$138,000 due to the hiring of a new employee during the first quarter of fiscal
2010. Consulting stock expense increased by about $247,000 as a result of a
higher level of grant activity to employees, consultants and directors. Investor
relations (“IR”) expenses increased by $133,000, due to increased IR-related
activities performed by outside consultants. Legal fees increased by
$126,000.
We
incurred interest expense of approximately $878,000 for the fiscal year ended
September 30, 2010 compared to interest expense of about $441,000 for the
fiscal year ended September 30, 2009. The increase in interest expense in
fiscal year 2010 reflects about $437,000 incurred in the first quarter of fiscal
year 2010, mostly due to charges as a result of the conversion of the Notes on
October 6, 2009 as part of our October 2009 Financing. In the second, third
and fourth quarters of 2010, interest expense dropped significantly mostly due
to the conversion of the Notes into equity.
As
previously disclosed, certain of our warrants to purchase common stock were
deemed to be a liability upon adoption of a new accounting pronouncement on
October 1, 2009. Subsequent changes to the fair market value resulted in an
offsetting charge in the statements of operations of approximately $21,347,000
for the fiscal year ended September 30, 2010. The warrant liability and
revaluations have not and will not have any impact on our working capital,
liquidity or business operations.
50
Fiscal
Year Ended September 30, 2009 Compared to Fiscal Year Ended September 30,
2008
We had a
net loss of $2,296,000 for the fiscal year ended September 30, 2009, versus a
net loss of $2,973,000 for the fiscal year ended September 30,
2008.
We did
not generate any revenues during fiscal 2009 or fiscal 2008.
Research
and Development
R&D
expenses decreased $266,000, or 27%, to $711,000 for fiscal 2009 from $977,000
for fiscal 2008. The lower level of R&D expenses during the
current period reflects a lower level of manufacturing and research
activities. During fiscal 2008, we were in the process of
manufacturing small quantities of our drug candidates to support our development
program, whereas during fiscal 2009, we had no manufacturing underway and were
only running stability studies on existing drug supplies resulting in a decrease
of $105,000 in manufacturing expenses. Research expenses also
declined during fiscal 2009 as more of our research programs were funded by
external grants. During fiscal 2009, outside research expenses were
$240,000 compared to $458,000 during fiscal 2008.
General
and Administrative
G&A
expenses decreased $248,000, or 16%, to $1,292,000 for fiscal year 2009 from
$1,540,000 for fiscal year 2008. G&A expenses were lower during
fiscal year 2009 versus fiscal year 2008 due to a decline in stock based
compensation expense, Board of Directors expense and legal
fees. Stock based compensation expense decreased by $50,000 as
a result of lower valuations assigned to our stock option grants in the current
year when compared to the prior year. Board of Directors expense
decreased by $101,000 as the Board of Directors adopted a stock based
compensation plan in July 2008 whereas during the prior year, the Board of
Directors received cash and stock based compensation. Legal fees
declined by $84,000 due to management’s continued efforts to reduce costs by
performing regulatory and compliance activities in-house.
During
fiscal year 2009, we recorded a gain on the sale of our holdings of Arca
Biopharma, Inc. (“ARCA”) common stock of $133,000.
During
fiscal 2008, CPEC LLC (“CPEC”) received a milestone payment from
ARCA. In 2003, CPEC, of which we own 35%, out-licensed all rights to
a potential therapeutic compound referred to as “bucindolol” to
ARCA. During fiscal 2008, CPEC received a milestone payment of
$500,000 as a result of ARCA filing a New Drug Application for
bucindolol. We recorded $175,000 of income during fiscal 2008 as a
result of our equity ownership of CPEC. Also as a result of the
filing of the New Drug Application with the FDA, we were obligated to pay
$413,000 in the form of cash or stock, at our election, to the majority owner of
CPEC who in turn paid the original licensors of bucindolol per the terms of the
1994 Purchase Agreement of CPEC. On November 6, 2009, we issued
1,099,649 shares to the majority owner of CPEC to satisfy the
obligation.
We
incurred net interest expense of $437,000 during fiscal 2009 compared to net
interest expense of $51,000 for fiscal 2008. The change reflects
higher interest expense as a result of the issuance of the Notes which had an
interest rate of 7% as well as the related amortization of debt issuance costs
and a note issuance discount. Interest expense also increased as a
result of a higher average balance of our note payable with Elan Corporation,
plc. Interest income on our investments also decreased due to a lower
level of investable assets as well as a lower level of interest rates during
fiscal 2009 versus fiscal 2008.
During
fiscal 2008, as a result of the issuance of the Notes, we were required to lower
the exercise price of 4,687,000 warrants previously issued in a financing the
Company conducted in November 2005 (the “November 2005 Financing”) and in the
2007 Financing to $0.35 per share, the conversion price of the
Notes. As a result of the change in the exercise price, these
warrants were revalued, resulting in an increase in the value of $118,000, which
was charged to the statement of operations. During fiscal 2009, as a result of
our March 2009 Financing, we were required to further lower the exercise price
of 4,687,000 warrants issued in the November 2005 Financing and in the 2007
Financing to $0.28 per share, the purchase price of the common stock and
warrants issued in our March 2009 Financing. As a result of the
change in the exercise price, these warrants were revalued, resulting in an
increase in the value of $38,000, which was charged to the statement of
operations.
During
fiscal 2009, we recorded a gain of $49,000 related to the net increase in the
market value of our trading securities. During fiscal 2008, we
recorded an “other-than-temporary” impairment charge of $49,000 based upon
reduced market values of these securities.
We do not
have any revenue, and therefore we rely on investors, grants, collaborations and
licensing of our compounds to finance our operations. As of September 30, 2010,
we had approximately $2,355,000 of cash and cash equivalents, an increase of
$1,709,000 from September 30, 2009. The increase in cash reflects gross
proceeds of $4,300,000 from the issuance of equity in October 2009, July 2010
and August 2010, offset by an increase in expenses from operations. We believe
we have adequate financial resources to conduct operations into the second
quarter of fiscal year 2012 but in order to fund on-going operating cash
requirements beyond that point, or to further accelerate or expand our programs,
we need to raise significant additional funds.
51
We
incurred significant losses from operations of approximately $25,869,000
(including a non-cash charge for increases in the value of warrants of
$21,347,000) and $2,296,000, and cash outflows from operations of approximately
$2,446,000 and $1,923,000, for the fiscal years ended September 30, 2010 and
2009, respectively. Our ongoing future cash requirements will depend on numerous
factors, particularly the progress of our catalytic antioxidant program and
clinical trials and our ability to negotiate and complete collaborative
agreements or out-licensing arrangements. In order to help fund our on-going
operating cash requirements, we intend to seek new collaborations for our
antioxidant research program that include initial cash payments and on-going
research support. In addition, we might sell additional shares of our stock
and/or convertible debentures and explore other strategic and financial
alternatives, including a merger with another company, the sale of stock and/or
debt, the establishment of new collaborations for current research programs,
that include initial cash payments and ongoing research support and the
out-licensing of our compounds for development by a third party.
As a
result of the October 2009 Financing and the Amendment, both of which are
discussed more fully above under “Corporate Matters” and in Note G—Stockholders’
Equity of our consolidated financial statements included in Item 8 below, we
were able to raise a total of about $1.65 million. On July 30,
2010, we announced the exercise of the October 2009 Put Option with Xmark
Opportunity Fund, L.P., Xmark Opportunity Fund, Ltd. and Xmark JV Investment
Partners, LLC (collectively, the “Xmark Funds”), under which we raised $1.65
million. In addition, on August 11, 2010, we completed a common stock and
warrant financing for gross proceeds of $1 million, with the option to call
another $1 million during the first quarter of fiscal 2011. The proceeds from
these financings are expected to enable us to continue operations into the
second quarter of fiscal 2012.
There are
significant uncertainties as to our ability to access potential sources of
capital. We may not be able to enter into any collaboration on terms acceptable
to us, or at all, due to conditions in the pharmaceutical industry or in the
economy in general or based on the prospects of our catalytic antioxidant
program. Even if we are successful in obtaining collaboration for our
antioxidant program, we may have to relinquish rights to technologies, product
candidates or markets that we might otherwise develop ourselves. These same
risks apply to any attempt to out-license our compounds.
Similarly,
due to market conditions, the illiquid nature of our stock and other possible
limitations on equity offerings, we may not be able to sell additional
securities or raise other funds on terms acceptable to us, if at all. Any
additional equity financing, if available, could result in substantial dilution
to existing stockholders.
Our
forecast of the period of time through which our financial resources will be
adequate to support our operations is forward-looking information, and actual
results could vary.
Contractual
Obligations
Our
contractual obligations (in thousands) as of September 30, 2010 were as
follows:
|
Payments due by period
|
||||||||||||||||||||
|
Contractual Obligations
|
Total
|
Less
than 1
Year
|
1-3
Years
|
3-5
Years
|
More
than 5
Years
|
|||||||||||||||
|
Short
and long-term debt
|
$
|
663
|
$
|
663
|
$
|
—
|
$
|
—
|
$
|
—
|
||||||||||
|
Capital
lease obligations
|
—
|
—
|
—
|
—
|
—
|
|||||||||||||||
|
Operating
leases
|
—
|
—
|
—
|
—
|
—
|
|||||||||||||||
|
Purchase
obligations
|
850
|
850
|
—
|
—
|
—
|
|||||||||||||||
|
Total
|
$
|
1,513
|
$
|
1,513
|
$
|
—
|
$
|
—
|
$
|
—
|
||||||||||
Off
Balance Sheet Arrangements
We do not
have any off-balance sheet arrangements that have or are reasonably likely to
have a current or future effect on our financial condition, changes in financial
condition, revenues or expenses, results of operations, liquidity, capital
expenditures or capital resources as defined under the rules of Securities and
Exchange Commission (“SEC”) Release No. FR-67. We do not have any operating or
capital leases.
52
Relationship
with Goodnow Capital, LLC and Xmark Opportunity Partners, LLC
In July
2003, we initiated a series of transactions that led to our corporate
reorganization and recapitalization. We obtained an aggregate of $8.0 million in
secured bridge financing in the form of convertible promissory notes we issued
to Goodnow Capital, LLC (“Goodnow”). A portion of this financing allowed us to
pay our past due payables and become current. We used the remainder for our
operations, including a toxicology study for our catalytic antioxidant compounds
under development as a treatment for ALS.
We
completed our corporate reorganization on November 20, 2003. The reorganization
involved the merger of our former parent company into one of our wholly owned
subsidiaries. Subsequent to our 2003 reorganization, we completed a number of
equity and debt financings, the majority of which included Xmark as investors.
As of September 30, 2010, Xmark Opportunity Partners, LLC, through its
management of Goodnow and the Xmark Funds, and through the Xmark Voting Trust
and options held by David Cavalier, an affiliate of Xmark and the Chairperson of
our Board of Directors, had voting power over 69.5% of our outstanding common
stock and had beneficial ownership, calculated based on SEC requirements, of
approximately 67.8% of our common stock. As a result of this significant
ownership, Xmark Opportunity Partners, LLC and its affiliates is able to control
future actions voted on by our stockholders.
In
addition, under the terms of the warrants to purchase up to 61,822,749 shares of
our common stock issued to Xmark on October 6, 2009 as well as subsequent
warrant issuances on July 30, 2010 and August 11, 2010 and any additional
warrants issued pursuant to the put and/or call right granted in our August 2010
financing (collectively, the “Xmark Warrants”), if we were to pay a dividend on
our common stock the exercise price of these warrants would be reset from $0.28
per share or $0.50 per share, as applicable, to $0.01 per share and the
warrantholders would also receive any such dividend paid. The Xmark Warrants
also contain a provision that provides for the reduction of the exercise price
to $0.01 upon a change of control and anti-dilution provisions in the event of a
stock dividend or split, dividend payment or other issuance, reorganization,
recapitalization or similar event. In addition, the Xmark Warrants, among other
restrictions, prohibit the sale of our company to an entity other than one that
is publicly traded.
Critical
Accounting Policies and Estimates
Our
consolidated financial statements have been prepared in accordance with
accounting principles generally accepted in the United States of America, which
require us to make estimates and judgments that affect the reported amounts of
assets, liabilities, revenues, expenses and related disclosure of contingent
assets and liabilities. We evaluate our estimates, judgments and the policies
underlying these estimates on a periodic basis as the situation changes, and
regularly discuss financial events, policies, and issues with our independent
registered public accounting firm and members of our audit committee. We
routinely evaluate our estimates and policies regarding revenue recognition;
clinical trial, preclinical, manufacturing and patent related liabilities;
license obligations; inventory; intangible assets; share-based payments; and
deferred tax assets.
Warrant
Liability
On
October 1, 2009, the Company adopted new accounting guidance originally
referred to as Emerging Issues Task Force 07-5, recently codified by FASB as
Account Standards Codification (“ASC”) Topic 815. The guidance revised
previously existing guidance for determining whether an Instrument (or Embedded
Feature) is indexed to an entity’s own stock. Equity-linked instruments (or
embedded features) that otherwise meet the definition of a derivative are not
accounted for as derivatives if certain criteria are met, one of which is that
the instrument (or embedded feature) must be indexed to the entity’s own stock.
The Company applied the new guidance to outstanding instruments as of
October 1, 2009. The fair value of the warrants affected by the new
guidance at the dates of issuance totaled $8,282,000 and was initially recorded
as a component of additional paid-in capital. Upon adoption of the new guidance,
the Company recorded a decrease to the opening balance of additional-paid-in
capital of $8,142,000 and recorded a decrease to accumulated deficit totaling
$4,353,000, representing the decrease in the fair value of the warrants from the
date of issuance to October 1, 2009. The fair value of the warrants at
October 1, 2009 of $3,789,000 was classified as a liability in the balance
sheet as of that date.
Increases
or decreases in fair value of the warrants are included as a component of other
income (expenses) in the accompanying statement of operations for the respective
period. As of September 30, 2010, the liability for warrants increased to
approximately $27,549,000, resulting in an additional charge to the statements
of operations for the fiscal year ended September 30, 2010 of approximately
$21,347,000. The warrant liability and revaluations have not and will not have
any impact on the Company’s working capital, liquidity or business
operations.
53
Item
7A. Quantitative and Qualitative Disclosures About Market
Risk.
Market
Risk
Our
exposure to market risk is presently limited to the interest rate sensitivity of
our cash and cash equivalents, which is affected by changes in the general level
of U.S. interest rates. However, we believe that we are not subject to any
material market risk exposure and do not expect that changes in interest rates
would have a material effect upon our financial position. A hypothetical 10%
change in interest rates would not have a material effect on our Statement of
Operations or Cash Flows for the fiscal year ended September 30, 2010. We
do not have any foreign currency or other derivative financial instruments. Our
debt bears interest at a fixed rate.
Item
8. Financial Statements and Supplementary Data.
INDEX
TO FINANCIAL STATEMENTS
|
Page
|
|
|
Report
of Independent Registered Public Accounting Firm
|
55
|
|
Consolidated
Balance Sheets – As of September 30, 2010 and 2009
|
56
|
|
Consolidated
Statements of Operations – For the fiscal years ended September 30, 2010,
2009 and 2008
|
57
|
|
Consolidated
Statements of Stockholders’ Equity (Deficit) – For the fiscal years ended
September 30, 2010, 2009 and 2008
|
58
|
|
Consolidated
Statements of Cash Flows – For the fiscal years ended September 30, 2010,
2009 and 2008
|
59
|
|
Notes
to Consolidated Financial Statements
|
60
|
54
REPORT
OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the
Board of Directors
Aeolus
Pharmaceuticals, Inc.
We have
audited the accompanying consolidated balance sheets of Aeolus Pharmaceuticals,
Inc. (the “Company”) as of September 30, 2010 and 2009, and the related
consolidated statements of operations, stockholders’ equity (deficit), and cash
flows for each of the years ended September 30, 2010, 2009 and
2008. These financial statements are the responsibility of the
Company’s management. Our responsibility is to express an opinion on
these financial statements based on our audits.
We
conducted our audits in accordance with the standards of the Public Company
Accounting Oversight Board (United States). Those standards require
that we plan and perform the audits to obtain reasonable assurance about whether
the financial statements are free of material misstatement. The
Company is not required to have, nor were we engaged to perform, an audit of its
internal control over financial reporting. Our audits included
consideration of internal control over financial reporting as a basis for
designing audit procedures that are appropriate in the circumstances, but not
for the purpose of expressing an opinion on the effectiveness of the Company’s
internal control over financial reporting. Accordingly, we express no
such opinion. An audit also includes examining, on a test basis,
evidence supporting the amounts and disclosures in the financial statements,
assessing the accounting principles used and significant estimates made by
management, as well as evaluating the overall financial statement presentation.
We believe that our audits provide a reasonable basis for our
opinion.
In our
opinion, the consolidated financial statements referred to above present fairly,
in all material respects, the consolidated financial position of Aeolus
Pharmaceuticals, Inc. as of September 30, 2010 and 2009, and the consolidated
results of its operations and its cash flows for each of the years ended
September 30, 2010, 2009 and 2008, in conformity with accounting principles
generally accepted in the United States of America.
The
accompanying consolidated financial statements have been prepared assuming that
the Company will continue as a going concern. As discussed in Note B
of the consolidated financial statements, the Company has suffered recurring
losses, negative cash flows from operations and management believes that the
Company does not currently possess sufficient working capital to fund its
operations past the second quarter of fiscal 2012. These matters
raise substantial doubt about the Company’s ability to continue as a going
concern. Management’s plans in regard to these matters are also
described in Note B. The consolidated financial statements do not include any
adjustments that might result from the outcome of this uncertainty.
/s/
Haskell & White LLP
HASKELL
& WHITE LLP
Irvine,
California
December
28, 2010
55
AEOLUS
PHARMACEUTICALS, INC.
CONSOLIDATED
BALANCE SHEETS
(DOLLARS
IN THOUSANDS, EXCEPT SHARE DATA)
|
September 30,
|
||||||||
|
2010
|
2009
|
|||||||
|
ASSETS
|
||||||||
|
Current
assets:
|
||||||||
|
Cash
and cash equivalents
|
$ | 2,355 | $ | 646 | ||||
|
Prepaids
and other current assets
|
46 | 133 | ||||||
|
Total
current assets
|
2,401 | 779 | ||||||
|
Investment
in CPEC LLC
|
32 | 32 | ||||||
|
Total
assets
|
$ | 2,433 | $ | 811 | ||||
|
LIABILITIES
AND STOCKHOLDERS’ EQUITY (DEFICIT)
|
||||||||
|
Current
liabilities:
|
||||||||
|
Accounts
payable and accrued expenses
|
$ | 957 | $ | 774 | ||||
|
Short-term
debt
|
663 | — | ||||||
|
Total
current liabilities
|
1,620 | 774 | ||||||
|
Warrant
liability
|
27,549 | — | ||||||
|
Senior
convertible notes to related parties, net (redemption value of $1,000,000
as of September 30, 2009)
|
— | 600 | ||||||
|
Long-term
note payable
|
— | 594 | ||||||
|
Total
liabilities
|
29,169 | 1,968 | ||||||
|
Commitments
and Contingencies (Notes E and J)
|
||||||||
|
Stockholders’
equity (deficit):
|
||||||||
|
Preferred
stock, $.01 par value per share, 10,000,000 shares
authorized:
|
||||||||
|
Series
B nonredeemable convertible preferred stock, 600,000 shares
authorized; 475,087 shares issued and outstanding as of
September 30, 2010 and 2009
|
5 | 5 | ||||||
|
Common
stock, $.01 par value per share, 200,000,000 shares authorized; 56,817,177
and 37,563,392 shares issued and outstanding at September 30, 2010 and
2009, respectively
|
568 | 376 | ||||||
|
Additional
paid-in capital
|
155,402 | 159,657 | ||||||
|
Accumulated
deficit
|
(182,711 | ) | (161,195 | ) | ||||
|
Total
stockholders’ equity (deficit)
|
(26,736 | ) | (1,157 | ) | ||||
|
Total
liabilities and stockholders’ equity (deficit)
|
$ | 2,433 | $ | 811 | ||||
The
accompanying notes are an integral part of these consolidated financial
statements.
56
AEOLUS
PHARMACEUTICALS, INC.
CONSOLIDATED
STATEMENTS OF OPERATIONS
(IN
THOUSANDS, EXCEPT SHARE DATA)
|
Fiscal Year Ended September 30,
|
||||||||||||
|
2010
|
2009
|
2008
|
||||||||||
|
Revenue
|
||||||||||||
|
Grant
income
|
$ | — | $ | — | $ | — | ||||||
|
Costs
and expenses:
|
||||||||||||
|
Research
and development
|
1,690 | 711 | 977 | |||||||||
|
General
and administrative
|
1,954 | 1,292 | 1,540 | |||||||||
|
Total
costs and expenses
|
3,644 | 2,003 | 2,517 | |||||||||
|
Loss
from operations
|
(3,644 | ) | (2,003 | ) | (2,517 | ) | ||||||
|
Equity
in income of CPEC LLC ($175 dividend received in 2008)
|
— | — | 175 | |||||||||
|
Interest
expense
|
(878 | ) | (441 | ) | (93 | ) | ||||||
|
Interest
income
|
— | 4 | 42 | |||||||||
|
Warrant
repricing charges
|
(38 | ) | (118 | ) | ||||||||
|
Warrant
liability charges
|
(21,347 | ) | — | — | ||||||||
|
Collaboration
expense
|
— | — | (413 | ) | ||||||||
|
Gain
(loss) on marketable investments
|
— | 49 | (49 | ) | ||||||||
|
Gain
on sale of investments, available for sale
|
— | 133 | — | |||||||||
|
Net
loss
|
$ | (25,869 | ) | $ | (2,296 | ) | $ | (2,973 | ) | |||
|
Basic
net loss per common share
|
$ | (0.46 | ) | $ | (0.07 | ) | $ | (0.09 | ) | |||
|
Diluted
net loss per common share
|
$ | (0.46 | ) | $ | (0.07 | ) | $ | (0.11 | ) | |||
|
Weighted
average common shares outstanding:
|
||||||||||||
|
Basic
|
56,817 | 34,789 | 31,953 | |||||||||
|
Diluted
|
56,817 | 34,789 | 32,217 | |||||||||
The
accompanying notes are an integral part of these consolidated financial
statements.
57
AEOLUS
PHARMACEUTICALS, INC.
CONSOLIDATED
STATEMENTS OF STOCKHOLDERS’ EQUITY (DEFICIT)
(Dollars
in thousands)
|
Series B Preferred Stock
|
Common Stock
|
Additional
|
Unrealized
|
Accumulated
|
Total Stockholders’
|
|||||||||||||||||||||||||||
|
Shares
|
Par Value
|
Shares
|
Value
|
Paid-in Capital
|
Losses
|
Deficit
|
Equity (Deficit)
|
|||||||||||||||||||||||||
|
Balance
at September 30, 2007
|
475,087 | $ | 5 | 31,952,749 | $ | 320 | $ | 156,781 | $ | 0 | $ | (155,926 | ) | $ | 1,180 | |||||||||||||||||
|
Sale
of Senior Convertible Notes and warrants, net of issuance costs of
$156,000
|
— | — | — | — | 451 | — | — | 451 | ||||||||||||||||||||||||
|
Stock-based
compensation
|
— | — | — | — | 341 | — | — | 341 | ||||||||||||||||||||||||
|
Unrealized
loss on marketable securities held for sale
|
— | — | — | — | — | (36 | ) | — | (36 | ) | ||||||||||||||||||||||
|
Net
loss for the fiscal year ended September 30, 2008
|
— | — | — | — | — | — | (2,973 | ) | (2,973 | ) | ||||||||||||||||||||||
|
Balance
at September 30, 2008
|
475,087 | 5 | 31,952,749 | 320 | 157,573 | (36 | ) | (158,899 | ) | (1,037 | ) | |||||||||||||||||||||
|
Sale
of Senior Convertible Notes and warrants
|
— | — | — | — | 323 | — | — | 323 | ||||||||||||||||||||||||
|
Stock-based
compensation
|
— | — | — | — | 283 | — | — | 283 | ||||||||||||||||||||||||
|
Issuance
of common stock pursuant to a consulting agreement
|
— | — | 78,125 | 1 | 24 | — | — | 25 | ||||||||||||||||||||||||
|
Payment
of interest on Senior Convertible Notes in the form of common
stock
|
— | — | 175,375 | 2 | 61 | — | — | 63 | ||||||||||||||||||||||||
|
Sale
of common stock pursuant to stock offering, net of issuance costs of
$91,000
|
— | — | 5,357,143 | 54 | 1,393 | — | — | 1,446 | ||||||||||||||||||||||||
|
Unrealized
gain on marketable securities held for sale
|
— | — | — | — | — | 36 | — | 36 | ||||||||||||||||||||||||
|
Net
loss for the fiscal year ended September 30, 2009
|
— | — | — | — | — | — | (2,296 | ) | (2,296 | ) | ||||||||||||||||||||||
|
Balance
at September 30, 2009
|
475,087 | 5 | 37,563,392 | 376 | 159,657 | 0 | (161,195 | ) | (1,157 | ) | ||||||||||||||||||||||
|
Impact
of adoption of Accounting Guidance (Note C)
|
— | — | — | — | (8,142 | ) | — | 4,353 | (3,789 | ) | ||||||||||||||||||||||
|
Common
stock sales, net of issuance costs
|
— | — | 14,285,714 | 143 | 1,535 | — | — | 1677.7 | ||||||||||||||||||||||||
|
Senior
convertible note conversion
|
— | — | 3,571,429 | 35 | 1,307 | — | — | 1,342 | ||||||||||||||||||||||||
|
Senior
convertible note interest payment
|
— | — | 36,993 | — | 12 | — | — | 12 | ||||||||||||||||||||||||
|
Exercise
of warrants
|
— | — | 260,000 | 3 | 70 | — | — | 73 | ||||||||||||||||||||||||
|
Issuance
of shares to Endo as milestone payment
|
— | — | 1,099,649 | 11 | 402 | — | — | 413 | ||||||||||||||||||||||||
|
Issuance
of warrants to a consultant
|
— | — | — | — | 29 | — | — | 29 | ||||||||||||||||||||||||
|
Options
issued to consultants
|
— | — | — | — | 533 | — | — | 533 | ||||||||||||||||||||||||
|
Net
loss for the fiscal year ended September 30, 2010
|
(25,869 | ) | (25,869 | ) | ||||||||||||||||||||||||||||
|
Balance
at September 30, 2010
|
475,087 | $ | 5 | 56,817,177 | $ | 568 | $ | 155,403 | $ | 0 | $ | (182,711 | ) | $ | (26,736 | ) | ||||||||||||||||
The
accompanying notes are an integral part of these consolidated financial
statements.
58
AEOLUS
PHARMACEUTICALS, INC.
CONSOLIDATED
STATEMENTS OF CASH FLOWS
(IN
THOUSANDS)
|
Fiscal Year Ended September 30,
|
||||||||||||
|
2010
|
2009
|
2008
|
||||||||||
|
Cash
flows from operating activities:
|
||||||||||||
|
Net
loss
|
(25,869 | ) | (2,296 | ) | (2,973 | ) | ||||||
|
Adjustments
to reconcile net loss to net cash used in operating
activities:
|
||||||||||||
|
Depreciation
|
1 | — | — | |||||||||
|
Noncash
compensation
|
548 | 283 | 341 | |||||||||
|
Noncash
interest and financing costs
|
825 | 433 | 89 | |||||||||
|
Warrant
repricing charges
|
— | 38 | 118 | |||||||||
|
Equity
income in CPEC LLC
|
— | — | (175 | ) | ||||||||
|
Change
in fair value of warrants
|
21,347 | — | — | |||||||||
|
Noncash
consulting and license fee
|
13 | 25 | — | |||||||||
|
(Gain)
loss on marketable investments
|
— | (49 | ) | 49 | ||||||||
|
(Gain)
on sale of investments, available for sale
|
— | (133 | ) | — | ||||||||
|
Change
in assets and liabilities:
|
||||||||||||
|
Prepaid
expenses and other assets
|
94 | (7 | ) | 15 | ||||||||
|
Accounts
payable and accrued expenses
|
595 | (217 | ) | 723 | ||||||||
|
Net
cash used in operating activities
|
(2,446 | ) | (1,923 | ) | (1,813 | ) | ||||||
|
Cash
flows from investing activities:
|
||||||||||||
|
Proceeds
from dividend from CPEC LLC
|
— | — | 175 | |||||||||
|
Purchase
of equipment
|
(7 | ) | — | — | ||||||||
|
Purchase
of investments and marketable securities
|
— | 751 | (525 | ) | ||||||||
|
Net
cash provided by (used in) investing activities
|
(7 | ) | 751 | (350 | ) | |||||||
|
Cash
flows from financing activities:
|
||||||||||||
|
Proceeds
from the issuance of Senior Convertible Notes and Warrants
|
— | 375 | 625 | |||||||||
|
Costs
related to the issuance of Senior Convertible Notes and
Warrants
|
— | — | (156 | ) | ||||||||
|
Proceeds
from short term note payable
|
— | 3 | 372 | |||||||||
|
Repayments
of short term note payable
|
— | (368 | ) | (6 | ) | |||||||
|
Proceeds
from issuance of common stock and warrants
|
4,300 | 1,500 | — | |||||||||
|
Proceeds
from the exercise of warrants
|
73 | — | — | |||||||||
|
Costs
related to the issuance of common stock and warrants
|
(211 | ) | (91 | ) | — | |||||||
|
Net
cash provided by financing activities
|
4,162 | 1,419 | 835 | |||||||||
|
Net
increase (decrease) in cash and cash equivalents
|
1,709 | 247 | (1,328 | ) | ||||||||
|
Cash
and cash equivalents at beginning of year
|
646 | 399 | 1,727 | |||||||||
|
Cash
and cash equivalents at end of year
|
2,355 | 646 | $ | 399 | ||||||||
|
Supplemental
disclosure of cash flow information:
|
||||||||||||
|
Non-cash
payments of interest
|
878 | — | 4 | |||||||||
|
Supplemental
disclosure of non-cash investing and financing activities:
|
||||||||||||
|
Issuance
of warrants to a consultant
|
15 | — | — | |||||||||
|
Notes
|
— | 63 | — | |||||||||
The
accompanying notes are an integral part of these consolidated financial
statements.
59
AEOLUS
PHARMACEUTICALS, INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER
30, 2010
A.
Business Overview
Aeolus
Pharmaceuticals, Inc., a Delaware corporation, is a biopharmaceutical company
that is developing a new class of broad spectrum catalytic antioxidant compounds
based on technology discovered at Duke University and National Jewish
Health. The Company’s lead compound, AEOL 10150, is entering human
clinical trials in oncology, where it will be used in combination with radiation
therapy. AEOL 10150 has previously been tested in two Phase I clinical
trials with no serious adverse events reported. The compound is also
being developed as a medical countermeasure against the pulmonary
sub-syndrome of acute radiation syndrome (“Pulmonary Acute Radiation
Syndrome” or “Lung-ARS”) as well as the
gastrointestinal sub-syndrome of acute radiation syndrome (“GI-ARS”), both
caused by exposure to high levels of radiation due to a radiological or nuclear
event. It is also being developed for use as a countermeasure for
exposure to chemical vesicants such as chlorine gas and sulfur mustard
gas. AEOL 10150 has already performed well in animal efficacy and
safety studies in each of these potential indications. The
“Company” or “Aeolus” refers collectively to Aeolus Pharmaceuticals, Inc. and
its wholly owned subsidiary, Aeolus Sciences, Inc., a Delaware corporation. The
Company’s initial target indications are as a protective agent against the
effects of acute radiation syndrome, sulfur mustard gas exposure and chlorine
gas exposure.
Funding
of the research on the Company’s lead compound, AEOL 10150, for use as a medical
countermeasure comes from a number of sources. Development of the compound for
lung effects of acute radiation exposure has largely been funded by Aeolus
through programs at the University of Maryland and Duke University. In
December 2009, the Company was informed by the Biologic Advanced Research
and Development Authority (“BARDA”) that it had been chosen to submit a full
proposal for funding of its pulmonary sub-syndrome of acute radiation syndrome
(“Lung-ARS”) program from its current stage to the U.S. Food and Drug
Administration (“FDA”) for approval, based on a summary “white paper” submitted
earlier in 2009 by the Company. In July of 2010, the Company was
informed by BARDA that its proposal had been chosen, and the Company entered
into negotiations with BARDA for a contract to fund the development of AEOL
10150 as a medical countermeasure for Lung-ARS.
Since
March 2009, AEOL 10150 has also been under development as a medical
countermeasure for the gastrointestinal
effects of Acute Radiation Syndrome (“GI-ARS”). The GI-ARS program has been
completely funded to date by the National Institutes of Health-National
Institute of Allergy and Infectious Disease (“NIH-NIAID”). Additional NIH-NIAID
funded studies of AEOL 10150 in GI-ARS are expected to begin before the end of
the fiscal year.
For the
past several years, AEOL 10150 has been the subject of research and development
as a countermeasure for mustard and chlorine gas exposure through funding by NIH
CounterACT. In May 2010, the Company submitted white papers to BARDA
proposing long-term funding of AEOL 10150 as both a chlorine and mustard gas
countermeasure from the current stages of development for FDA approval. In
September 2010, the Company was informed that its white paper for chlorine had
been chosen by BARDA for a full proposal, but that its proposal for mustard gas
had not been chosen. The chlorine gas proposal was submitted to BARDA
on December 16, 2010.
The
Company has a second compound, AEOL 11207, which has been the subject of
research and development as a treatment for Parkinson’s disease and epilepsy.
Currently, this research work is being funded through grants to Dr. Manisha
Patel at the University of Colorado from the Michael J. Fox Foundation and
Citizens United for Research in Epilepsy (CURE).
As of
September 30, 2010, Aeolus also owned a 35.0% interest in CPEC LLC, a
Delaware limited liability company (“CPEC”).
The
Company’s primary operations are located in Mission Viejo,
California.
B.
Liquidity
The
Company has incurred significant losses from operations of approximately
$25,869,000 (including a non-cash charge for increases in valuation of warrants
of $21,347,000), $2,296,000 and $2,973,000 and cash outflows from operations of
approximately $2,446,000, $1,923,000 and $1,813,000 for the fiscal years ended
September 30, 2010, 2009 and 2008, respectively. The Company expects to
incur additional losses and negative cash flow from operations for several more
years.
As a
result of the October 2009 Financing and the Amendment, both of which are
discussed more fully in Note G– Stockholders’ Equity, the Company was able to
raise a total of about $1.65 million during the first quarter of fiscal year
2010. On July 30, 2010, the Company announced the exercise of
the October 2009 Put Option with Xmark Funds discussed more fully in Note G –
Stockholders’ Equity, under which the Company raised gross proceeds of $1.65
million. In addition, on August 11, 2010, the Company completed a common
stock and warrant financing for gross proceeds of $1 million, with the option to
call another $1 million during the first quarter of fiscal 2011. During the
first quarter of fiscal 2011, Xmark Funds called its option, resulting in gross
proceeds of $1 million to the Company. Cash on hand, plus the
proceeds from the December 2010 option call is expected to enable the Company to
continue operations into the second quarter of fiscal 2012. The level
of research and development activity will depend heavily upon the successful
execution of a contract with BARDA for lung ARS. If the Company is
successful in obtaining the contract, research and development activities will
increase significantly. If the Company is not successful in obtaining
the contract, spending on lung ARS development by the Company will be halted,
resulting in lower research and development expenses.
60
If the
Company is unable to obtain additional funding for its operations beyond the
second quarter of 2012, it will need to eliminate or substantially limit some or
all of its activities, merge with another company, sell, lease or license some
or all of its assets, or cease operations entirely. There can be no assurance
that the Company will be able to obtain additional financing on favorable terms,
or at all, or that the Company will be able to merge with another Company or
sell, lease or license any or all of its assets.
C.
Summary of Significant Accounting Policies
Basis
of Presentation
The
consolidated financial statements include the accounts of Aeolus and its wholly
owned subsidiary. All significant intercompany accounts and transactions have
been eliminated. The Company uses the equity method to account for its 35.0%
ownership interest in CPEC.
Use
of Estimates
The
preparation of financial statements in conformity with accounting principles
generally accepted in the United States of America requires management to make
estimates and assumptions that affect the reported amounts of assets and
liabilities and disclosures of contingent assets and liabilities at the date of
the financial statements and the reported amounts of revenues and expenses
during the reporting period. Actual results could differ from those
estimates.
Cash
and Cash Equivalents
The
Company invests available cash in short-term bank deposits, money market funds,
commercial paper and U.S. government securities. Cash and cash equivalents
include investments with maturities of three months or less at the date of
purchase. The carrying value of cash and cash equivalents approximate their fair
market value at September 30, 2010 and 2009 due to their short-term
nature.
Investments
Investments
consist of equity securities and auction rate securities, each of
investment-grade quality, which have an original maturity dates greater than 90
days. These investments are recorded at fair value and accounted for
as available-for-sale securities. The unrealized gain (loss) during
the period is recorded within accumulated other comprehensive loss unless it is
determined to be other-than-temporary. During the year ended
September 30, 2009, the Company recorded a net unrealized gain on investments of
$36,000 and a realized gain on the auction-rate securities of $49,000. During
the year ended September 30, 2008, the Company recorded a net unrealized loss on
investments of $36,000 and also recorded another than temporary impairment on
the auction-rate securities of $49,000.
Revenue
Recognition
Grant
income is recognized as revenue as work under the grant is performed and the
related expenses are incurred.
Research
and Development
Research
and development costs are expensed in the period incurred. Payments related to
the acquisition of in-process research and development are expensed due to the
stage of development of the acquired compound or technology at the date of
acquisition.
Income
Taxes
Deferred
tax assets and liabilities are determined based on the difference between the
financial statement and tax basis of assets and liabilities using enacted tax
rates in effect for the year in which the differences are expected to affect
taxable income. Valuation allowances are established when necessary to reduce
net deferred tax assets to the amounts expected to be realized.
61
Net
Loss Per Common Share
The
Company computes basic net loss per weighted average share attributable to
common stockholders using the weighted average number of shares of common stock
outstanding during the period. The Company computes diluted net income (loss)
per weighted average share attributable to common stockholders using the
weighted average number of shares of common and dilutive potential common shares
outstanding during the period. Potential common shares outstanding consist of
stock options, convertible debt, warrants and convertible preferred stock using
the treasury stock method and are excluded if their effect is anti-dilutive.
Diluted weighted average common shares excluded incremental shares of
approximately 75,762,878 shares for the fiscal year
ended September 30, 2010 issuable upon the exercise or conversion of
convertible debt, stock options to purchase common stock, convertible preferred
stock and warrants to purchase common stock. These shares were excluded due to
their anti-dilutive effect as a result of the Company’s net loss for the fiscal
year ended September 30, 2010. Diluted weighted average common
shares included incremental shares issuable upon conversion of the Notes, but
excluded incremental shares of approximately 36,954,000 and 20,010,000
respectively for the fiscal year 2009 and 2008, due to their anti-dilutive
effect as a result of the Company’s net loss for the fiscal year 2009 and
2008.
Accounting
for Stock-Based Compensation
The
Company recognizes stock based compensation expense in the statement of
operations based upon the fair value of the equity award amortized over the
vesting period.
Segment
Reporting
The
Company currently operates in only one segment.
Warrant
Liability
On
October 1, 2009, the Company adopted new accounting guidance originally
referred to as Emerging Issues Task Force 07-5, recently codified by FASB as
Account Standards Codification (“ASC”) Topic 815. The guidance revised
previously existing guidance for determining whether an Instrument (or Embedded
Feature) is indexed to an entity’s own stock. Equity-linked instruments (or
embedded features) that otherwise meet the definition of a derivative are not
accounted for as derivatives if certain criteria are met, one of which is that
the instrument (or embedded feature) must be indexed to the entity’s own stock.
The Company applied the new guidance to outstanding instruments as of
October 1, 2009. The fair value of the warrants affected by the new
guidance at the dates of issuance totaled $8,282,000 and was initially recorded
as a component of additional paid-in capital. Upon adoption of the new guidance,
the Company recorded a decrease to the opening balance of additional-paid-in
capital of $8,142,000 and recorded a decrease to accumulated deficit totaling
$4,353,000, representing the decrease in the fair value of the warrants from the
date of issuance to October 1, 2009. The fair value of the warrants at
October 1, 2009 of $3,789,000 was classified as a liability in the balance
sheet as of that date.
Increases
or decreases in fair value of the warrants are included as a component of other
income (expenses) in the accompanying statement of operations for the respective
period. As of September 30, 2010, the liability for warrants increased to
approximately $27,549,000, resulting in an additional charge to the statements
of operations for the fiscal year ended September 30, 2010 of approximately
$21,347,000. The warrant liability and revaluations have not and will not have
any impact on the Company’s working capital, liquidity or business
operations.
D.
Investments
Investments
in Auction-Rate Securities
In fiscal
year 2008, the Company invested in auction-rate securities with a par value of
$525,000. The auction-rate securities were debt obligations secured
by student loans, which loans are generally guaranteed by the U.S. Government
under the Federal Family Education Loan Program (FFELP). In addition
to the U.S. government guarantee on such student loans, many of the securities
also had separate insurance policies guaranteeing both the principal and accrued
interest. The final maturity dates of the auction-rate securities
which the Company owned was between 2029 and 2038. Liquidity
for these securities had historically been provided by an auction process that
resets the applicable interest rate at pre-determined intervals for up to 35
days. In the past, the auction process had generally allowed
investors to obtain immediate liquidity if so desired by selling the securities
at their face amounts. However, disruptions in the credit markets
adversely affected the auction market for these types of
securities. From February 26, 2008 to January 2, 2009, all auctions
scheduled with respect to the Company’s auction-rate securities failed to
close.
The
Company recorded an “other-than-temporary” impairment charge during the fiscal
year ended September 30, 2008 of $49,000 based upon reduced market values as
determined based upon investment statements received from with UBS Financial
Services, Inc. (“UBS”). The Company also recorded these investments
as available for sale and accordingly recorded their value on the balance sheet
at market value as determined by investment statements provided by
UBS.
In
October 2008, the Company entered into an agreement (the “UBS Agreement”) with
UBS for which the Company received the right (“UBS Put Option”) to sell all four
of the auction rate securities back to UBS at par, at its sole discretion,
anytime during the period from January 2, 2009 through January 4, 2011, and gave
UBS the right to purchase these auction rate securities or sell them on the
Company’s behalf at par any time after the execution of the UBS Agreement
through January 4, 2011. The UBS Put Option was not transferable,
tradable or marginable, and was not listed or quoted on any securities exchange
or any electronic communications network. Since the UBS Agreement is
a legally enforceable firm commitment, the UBS Put Option was recognized as a
financial asset at fair value in the financial statements, and accounted for
separately from the associated securities as a noncurrent asset. Since the
Company intended to and subsequently exercised the UBS Put Option on January 2,
2009, the Company did not have the intent to hold the associated auction rate
securities until recovery or maturity. Therefore, the Company classified these
securities as trading which required changes in the fair value of these
securities to be recorded in current period earnings. As a result of
this transfer, the Company recognized in the Statement of Operations a change in
market value of $65,000, reflecting a reversal of the related temporary
valuation allowance that was previously recorded in other comprehensive
loss.
62
Prior to
entering into the UBS Agreement, the Company recorded the auction rate
securities as investments available-for-sale. The Company recorded
unrealized gains and losses on the available-for-sale debt securities in
accumulated other comprehensive income in the shareholders’ equity section of
the balance sheets. Such an unrealized loss did not reduce net income
for the applicable accounting period.
The
Company measured the UBS Put Option at fair value and thus recorded income of
approximately $114,000 and recorded a corresponding long term investment during
the first quarter of fiscal year 2009.
Following
the reclassification of the auction rate securities to trading, the Company
recorded an additional charge to the Statement of Operations of $16,000 to
reduce the value of the auction rate securities, offset by the gain on the UBS
Put Option of $16,000.
On
January 2, 2009, the Company exercised its rights under the UBS Put Option and
sold its four auction rate securities for their par value of $525,000 to
UBS.
Investment
in ARCA Biopharma, Inc.
The
Company also held an investment in equity securities of ARCA BioPharma, Inc.
(“ARCA”) until April 2009. The aggregate carrying amount of the
holdings in ARCA was zero as of September 30, 2010 and 2009,
respectively. The Company accounted for the investment on cost basis
and reviewed the investment for impairment whenever there are identified events
or changes in circumstances that may have a significant adverse effect on the
fair value of the investment. During the fiscal years ended September
30, 2010, 2009 and 2008, the Company did not record an impairment on the
investment in ARCA. During April 2009, the Company sold all of its
23,377 shares of ARCA generating net proceeds of $226,000 and a net gain of
$133,000.
Investment
in CPEC LLC
The
Company uses the equity method to account for its 35.0% ownership interest in
CPEC. During fiscal 2003, CPEC licensed bucindolol, a drug previously
under development by the Company for the treatment of heart failure, to ARCA in
return for possible future royalty and milestone payments. During
fiscal 2008, CPEC declared and paid a dividend of which the Company received
$175,000. The dividend was paid upon receipt of a milestone payment
by CPEC from ARCA, which was triggered upon the filing of a New Drug Application
for bucindolol with the FDA. Also as a result of the filing of the
New Drug Application with the FDA, the Company was obligated to pay $413,000,
which the Company elected to pay in the form of stock to the majority owner of
CPEC who in turn paid the original licensors of bucindolol per the terms of the
1994 Purchase Agreement of CPEC. On November 6, 2009, we issued 1,099,649
shares to the majority owner of CPEC to satisfy the obligation.
CPEC had
$91,000 of net assets at each of September 30, 2010 and 2009. Aeolus’
share of CPEC’s net assets is included in other assets and the Company has no
operations or activities unrelated to the out licensing of
bucindolol.
E.
Commitments
The
Company acquires assets still in development and enters into research and
development arrangements with third parties that often require milestone and
royalty payments to the third party contingent upon the occurrence of certain
future events linked to the success of the asset in
development. Milestone payments may be required, contingent upon the
successful achievement of an important point in the development life-cycle of
the pharmaceutical product (e.g., approval of the product
for marketing by a regulatory agency). If required by the
arrangement, the Company may have to make royalty payments based upon a
percentage of the sales of the pharmaceutical product in the event that
regulatory approval for marketing is obtained. Because of the
contingent nature of these payments, they are not included in the table of
contractual obligations.
These
arrangements may be material individually, and in the unlikely event that
milestones for multiple products covered by these arrangements were reached in
the same period, the aggregate charge to expense could be material to the
results of operations in any one period. In addition, these
arrangements often give Aeolus the discretion to unilaterally terminate
development of the product, which would allow Aeolus to avoid making the
contingent payments; however, Aeolus is unlikely to cease development if the
compound successfully achieves clinical testing objectives.
63
F.
Notes Payable
Senior
Convertible Notes to Related Parties
On
August 1, 2008, the Company entered into a Securities Purchase Agreement
(the “SCN Purchase Agreement”) with three accredited institutional
investors (the “August 2008 Investors”) pursuant to which the Company
agreed to sell to the August 2008 Investors units comprised of senior unsecured
convertible notes of the Company (the “Notes”), in an aggregate principal amount
of up to $5,000,000, which bear interest at a rate of 7% per year and mature on
the 30-month anniversary of their date of issuance, and warrants to purchase up
to an aggregate of 10,000,000 additional shares of the Company’s common stock
(the “August 2008 Warrant Shares”), each with an initial exercise price of $0.50
per share, subject to adjustment as provided in the warrants (the “August 2008
Warrants”). Each unit (collectively, the “August 2008 Units”) is comprised of
$1,000 in Note principal and August 2008 Warrants to purchase up to 2,000 shares
of the Company’s common stock, and has a purchase price of $1,000.
On
August 1, 2008, pursuant to the SCN Purchase Agreement, the Company sold
and issued to the August 2008 Investors an aggregate of 500 August 2008 Units
comprised of Notes in the aggregate principal amount of $500,000 and August 2008
Warrants to purchase up to 1,000,000 shares of common stock for an aggregate
purchase price of $500,000 (the “SCN Financing”).
On each
of September 4, 2008, October 1, 2008, November 3, 2008 and
December 1, 2008, the Company sold and issued to the August 2008 Investors
an aggregate of 125 August 2008 Units comprised of Notes in the aggregate
principal amount of $125,000 and August 2008 Warrants to purchase up to 250,000
shares of common stock for an aggregate purchase price of $125,000 (the
“Subsequent Financings”).
The Notes
issued in the SCN Financing and the Subsequent Financings had an initial
conversion price of $0.35 per share, subject to adjustment as provided in the
Notes. In addition, the August 2008 Investors had the option to purchase up to
an additional 4,000 August 2008 Units, in one or more closings at their sole
option at any time on or before December 31, 2013.
Interest
on the Notes accrued at the rate of 7.0% per annum from the date of issuance,
and was payable semi-annually, on January 31 and July 31 of each year.
Interest was payable, at the Company’s sole election, in cash or shares of
common stock, to holders of Notes on the record date for such interest payments,
with the record dates being each January 15 and July 15 immediately
preceding an interest payment date. The effective interest rate of the Notes,
including the effect of the amortization of the embedded conversion feature and
the note discount, was 39.4%.
The net
proceeds to the Company from the sale of 1,000 August 2008 Units in the SCN
Financing and Subsequent Financing, after deducting for expenses, were
approximately $844,000. The Company used the net proceeds to fund the
development of AEOL 10150 and to fund ongoing operations of the Company.
Offering costs of the private placement were $156,000 and were allocated to the
Notes and August 2008 Warrants based upon their respective fair values. The
offering costs attributed to the Notes in the amount of $100,000 were
capitalized as Debt Issuance Costs. The Debt Issuance Costs were amortized over
the life of the Notes in the SCN Financing.
Pursuant
to the Securities Purchase and Exchange Agreement dated October 6, 2009
(the “October 2009 Purchase Agreement”), as more fully described in Note G
– Stockholders’ Equity, the holders of the Notes agreed to convert all
$1,000,000 in principal amount of the Notes into common stock at a conversion
rate of $0.35 per share and to exchange their remaining option to purchase an
additional $4,000,000 in Notes for warrants to purchase up to 14,285,714 shares
of common stock with an initial exercise price of $0.28 per share, subject to
adjustment as provided in the warrants.
On
December 24, 2009, the Company entered into an amendment (the “Amendment”)
to the October 2009 Purchase Agreement, pursuant to which the Company
agreed to lower the conversion price of the Notes from $0.35 per share to $0.28
per share and as a result, issued an additional 714,286 shares of common stock
to the former holders of the Notes. The Amendment was executed to resolve a
misunderstanding regarding one of the financing terms in the October 6,
2009 financing between the Company and the investors in the financing. The
Company did not receive any proceeds from the issuance. As a result of the
Amendment and the issuance of the additional shares, the Company recorded a
charge of $343,000 in the Statement of Operations as interest expense for the
value of the shares issued on the date of issuance.
Affiliates
of Xmark Opportunity Partners, LLC were the sole investors in the SCN Financing.
Together with its affiliates, Xmark Opportunity Partners, LLC beneficially owned
approximately 52% of the Company’s outstanding common stock prior to the SCN
Financing. Xmark Opportunity Partners, LLC is the sole manager of Goodnow
Capital, LLC (“Goodnow”) and possesses sole power to vote and direct the
disposition of all securities of the Company held by Goodnow. Goodnow has the
right to designate up to two directors for election to the Company’s Board of
Directors, pursuant to the terms of a purchase agreement between Goodnow and the
Company. David C. Cavalier, a current director of the Company and Managing
Partner of Xmark Opportunity Partners LLC, is President of Goodnow. The
transaction was evaluated by the Company’s management and the Board of Directors
for fairness to ensure the terms were reasonable given the related party nature
of the SCN Financing by providing an option for non-related party investors to
participate in the transaction.
64
Elan
Note Payable
In
August 2002, Aeolus borrowed $638,000 from Elan Corporation, plc (“Elan”)
pursuant to a promissory note. The note payable accrued interest at 10%
compounded semi-annually. The note was convertible at the option of Elan into
shares of the Company’s Series B non-voting convertible preferred stock (“Series
B Stock”) at a rate of $43.27 per share. The original note matured on
December 21, 2006. However, in February 2007, the Company and Elan
terminated the note, the Company paid $300,000 in cash to Elan, Elan
forgave $225,000 of the note payable and Elan and the Company entered into a new
two-year note payable in the amount of $453,000 under substantially the same
terms as the original note. In February 2009, the Company and Elan agreed
to amend the new note payable to extend its maturity date from February 7,
2009 to February 7, 2011 and increased the interest rate of the convertible
promissory note from 10% to 11% effective February 7, 2009. As of the date
of the amendment, an aggregate of $553,000 in principal and interest was
outstanding under the convertible promissory note. In the event of an event of
default under the convertible promissory note, Elan may demand immediate payment
of all amounts outstanding under the note. For purposes of the note, an event of
default includes, among other items, a default in the payment of the note
principal or interest when due and payable, an uncured breach by the Company of
its obligations to Elan pursuant the agreements under which the convertible
promissory note was issued, an inability of the Company to pay its debts in the
normal course of business, the cessation of business activities by the Company
(other than as a result of a merger or consolidation with a third party) without
Elan’s prior written consent and the appointment of a liquidator, receiver,
administrator, examiner, trustee or similar officer of the Company or over all
or substantially all of its assets under the law.
During
the term of the note payable, Elan has the option to convert the note into
shares of Series B Stock at a rate of $9.00 per share. Upon the maturity of the
note payable, Aeolus has the option to repay the note either in cash or in
shares of Series B Stock and warrants having a then fair market value equal to
the amount due under the note; provided that the fair market value used for
calculating the number of shares to be issued will not be less than $13.00 per
share. As of September 30, 2010 and 2009, the outstanding balance,
including interest, on the note payable to Elan was approximately $663,000 and
594,000, respectively.
G.
Stockholders’ Equity (Deficit)
Basis
of Presentation
Preferred
Stock
The
Certificate of Incorporation of Aeolus authorizes the issuance of up to
10,000,000 shares of Preferred Stock, at a par value of $.01 per share. The
Board of Directors has the authority to issue Preferred Stock in one or more
series, to fix the designation and number of shares of each such series, and to
determine or change the designation, relative rights, preferences, and
limitations of any series of Preferred Stock, without any further vote or action
by the stockholders of the Company.
In
January 2001, Aeolus issued to Elan 28,457 shares of Series B Stock. In February
2002, the Company issued 58,883 shares of Series B Stock and 480,000 shares of
common stock to Elan in exchange for the retirement of a $1,400,000 note payable
to Elan. In May 2002, the Company sold 416,204 shares of Series B Stock to Elan
for $3,000,000. On January 14, 2005, Elan converted 28,457 shares of the Series
B Stock into 28,457 shares of common stock. As of September 30, 2010, 475,087
shares of Series B Stock were outstanding. Each share of Series B Stock is
convertible into one share of common stock.
Common
Stock
August-December
2008 Financing
In
connection with the Senior Convertible Note (“SCN”) Financing, Aeolus issued
warrants to purchase 2,000,000 shares at an exercise price of $0.50 per share
with a five year term. The fair value of the warrants issued on August 1,
2008 was estimated to be $282,000 using the Black-Scholes option pricing model
with the following assumptions: dividend yield of 0%; expected volatility of
128% risk free interest rate of 3.2%; and an expected life of five years. The
fair value of the warrants issued on September 4, 2008 was estimated to be
$53,000 using the Black-Scholes option pricing model with the following
assumptions: dividend yield of 0%; expected volatility of 130% risk free
interest rate of 3.0%; and an expected life of five years. The fair value of the
warrants issued on October 1, 2008 was estimated to be $93,000 using the
Black-Scholes option pricing model with the following assumptions: dividend
yield of 0%; expected volatility of 133% risk free interest rate of 2.9%; and an
expected life of five years. The fair value of the warrants issued on
November 3, 2008 was estimated to be $76,000 using the Black-Scholes option
pricing model with the following assumptions: dividend yield of 0%; expected
volatility of 139% risk free interest rate of 2.7%; and an expected life of five
years. The fair value of the warrants issued on December 1, 2008 was
estimated to be $75,000 using the Black-Scholes option pricing model with the
following assumptions: dividend yield of 0%; expected volatility of 138% risk
free interest rate of 1.7%; and an expected life of five years.
In
addition, as a result of the SCN Financing, the Company was required to lower
the exercise price of 4,687,000 warrants issued in prior financings of the
Company completed in November 2005 and May 2007 to $0.35 per share, the
conversion price of the Notes. As a result of the change in the exercise price,
these warrants were revalued, resulting in an increase in the value of $118,000
which was charged to the statement of operations during the fourth quarter of
fiscal year 2008.
65
March
2009 Financing
On
March 30, 2009, Aeolus entered into a Securities Purchase Agreement (the
“Purchase Agreement”) with two accredited institutional investors (the
“March 2009 Investors”) pursuant to which the Company sold and issued to
the March 2009 Investors in a private placement an aggregate of 5,357,143
units (the “March 2009 Units”), comprised of an aggregate of 5,357,143
shares of common stock of the Company and warrants to purchase up to an
aggregate of 13,392,857 additional shares of common stock (the “March 2009
Warrants”), with an initial exercise price of $0.35 per share, subject to
adjustment as provided in the March 2009 Warrants, with each
March 2009 Unit representing one share of common stock and a
March 2009 Warrant to purchase two-and-one-half shares of common stock, at
a purchase price of $0.28 per March 2009 Unit for aggregate gross proceeds
of $1,500,000 (collectively, the “March 2009 Financing”). The
March 2009 Warrants are exercisable for a five year period from their date
of issuance; contain a “cashless exercise” feature that allows the holder to
exercise the March 2009 Warrants without a cash payment to the Company
under certain circumstances; contain a dividend participation right which allows
the holder to receive any cash dividends paid on the Company’s common stock
without exercising the March 2009 Warrant and contain a provision that
provides for the reduction of the exercise price to $0.01 in the event of any
such payment of cash dividends by the Company; and contain standard
anti-dilution provisions that provide for the adjustment of the exercise price
and the number of shares of common stock that can be purchased in the event of a
financing at a price per share below the exercise price, a stock dividend or
split, dividend payment or other issuance, reorganization, recapitalization or
similar event.
The fair
value of the March 2009 Warrants was estimated to be $4,129,000 using the
Black-Scholes option pricing model with the following assumptions: dividend
yield of 0%; risk free interest rate of 1.7%; expected volatility of 164%; and
an expected life of five years.
Offering
costs of the March 2009 Financing were $91,000 resulting in net proceeds to
the Company of approximately $1.4 million. The Company has used, and intends to
continue to use, the net proceeds from the March 2009 Financing to finance
the development of AEOL 10150 and to fund ongoing operations of the Company.
Affiliates of Xmark Opportunity Partners, LLC were the sole investors in the
March 2009 Financing.
As a
result of the March 2009 Financing, the Company was required to lower the
exercise price of 4,687,000 warrants previously issued in November 2005 and
May 2007 to $0.28 per share, the purchase price of the March 2009
Units. As a result of the change in the exercise price, these warrants were
revalued resulting in an increase in the value of $38,000, which was charged to
the statement of operations during the second quarter of fiscal year
2009.
October
2009 Financing
On
October 6, 2009, the Company entered into the October 2009 Purchase
Agreement with several accredited institutional investors (the
“October 2009 Investors”) pursuant to which the Company sold and issued to
the October 2009 Investors in a private placement an aggregate of 5,892,857
units (the “October 2009 Units”), comprised of an aggregate of 5,892,857 shares
of common stock (the “October 2009 Shares”) and warrants to purchase up to
an aggregate of 11,785,714 additional shares of common stock (the
“October 2009 Warrants”), with an initial exercise price of $0.28 per
share, subject to adjustment as provided in the October 2009 Warrants, with
each October 2009 Unit representing one share of common stock and a
October 2009 Warrant to purchase two shares of common stock, at a purchase
price of $0.28 per October 2009 Unit for aggregate gross proceeds of $1,650,000
(collectively, the “October 2009 Financing”). The October 2009
Warrants are exercisable for a seven year period from their date of issuance;
contain a “cashless exercise” feature that allows the holder to exercise the
October 2009 Warrants without a cash payment to the Company under certain
circumstances; contain a dividend participation right which allows the holder to
receive any cash dividends paid on the common stock without exercising the
October 2009 Warrant and contain a provision that provides for the
reduction of the exercise price to $0.01 in the event of any such payment of
cash dividends by the Company or upon a change of control and contain
anti-dilution provisions in the event of a stock dividend or split, dividend
payment or other issuance, reorganization, recapitalization or similar
event.
The
Company also granted to the October 2009 Investors the option to acquire,
collectively, up to an additional 5,892,857 October 2009 Units (the “Additional
Units”), comprised of an aggregate of 5,892,857 shares of common stock and
warrants to purchase up to an aggregate of 11,785,714 additional shares of
common stock at the per Additional Unit purchase price of $0.28 (the “October
2009 Call Option”). In addition, the October 2009 Investors granted to the
Company the option to require these October 2009 Investors, severally and
not jointly, to acquire up to 5,892,857 Additional Units, less any Additional
Units acquired under the October 2009 Call Option, at the per Additional Unit
purchase price of $0.28 (the “October 2009 Put Option”). The October 2009 Call
Option was exercisable at any time, and from time to time, on or prior to
June 30, 2010. The October 2009 Put Option was exercisable at any time from
June 30, 2010 to July 30, 2010. On July 30, 2010, the Company
exercised the October 2009 Put Option in full for $1.65 million in gross cash
proceeds and issued 5,892,857 shares of common stock and 11,785,714 warrants to
the October 2009 Investors.
In
addition, the October 2009 Investors agreed to convert all $1,000,000 in
principal amount of the Notes into common stock of the Company at a conversion
rate of $0.35 per share (the “Conversion Shares”), which was subsequently
lowered to $0.28 as discussed below, and to exchange their remaining option to
purchase an additional $4,000,000 in Notes for warrants to purchase up to
14,285,714 shares of common stock in substantially the same of form and terms of
the October 2009 Warrants issued in the October 2009 Financing,
including an initial exercise price of $0.28 per share, subject to adjustment as
provided in the warrants (the “Note Warrants”). As consideration for the
October 2009 Investors to convert the Notes, the Company agreed to exchange
warrants to purchase up to 2,000,000 shares of common stock issued to the
October 2009 Investors in connection with the sale of the Notes, warrants
to purchase up to 2,150,000 shares of common stock issued to the
October 2009 Investors and one of their affiliates in connection with a
financing completed in November 2005 and warrants to purchase up to
13,392,857 shares of common stock issued to the October 2009 Investors in
connection with a financing completed in March 2009 (collectively, the
“Prior Warrants”) for warrants to purchase up to an aggregate of 17,542,857
shares of common stock in substantially the same form and terms of the
October 2009 Warrants issued in the October 2009 Financing, including
an initial exercise price of $0.28 per share, subject to adjustment pursuant to
the warrants (the “Exchange Warrants”) (collectively, the
“Conversion”).
66
In
connection with the October 2009 Financing and the Conversion, the Company
also entered into a Registration Rights Agreement (the “October 2009 Rights
Agreement”) with the October 2009 Investors. In addition, the
October 2009 Investors agreed to terminate the Company’s Registration
Rights Agreements dated November 21, 2005 and March 30, 2009. Pursuant
to the October 2009 Rights Agreement, the Company agreed to file one or
more registration statements (collectively, the “Registration Statements”) with
the Securities and Exchange Commission (the “SEC”) covering the resale of the
October 2009 Shares, the Conversion Shares and all shares of common stock
issuable upon exercise of the October 2009 Warrants, the Note Warrants and
the Exchange Warrants (collectively, the “Registrable Securities”) upon demand
of the holders of a majority of the Registrable Securities (a “Demand
Registration”). Such holders have the right to two Demand Registrations, subject
to certain exceptions. In the event the holders exercise their right to a Demand
Registration, the Company has agreed to file a Registration Statement to
register the resale of the Registrable Securities within a certain number of
days after the request and to use commercially reasonable efforts to cause the
Registration Statement to be declared effective by the SEC as soon as
practicable after the filing thereof. The Company also agreed to use its
commercially reasonable efforts to keep the Registration Statements effective
for a specified period.
The net
proceeds to the Company from the October 2009 Financing, after deducting
for expenses, were approximately $1.6 million. The Company has used, and intends
to continue to use, the net proceeds from the October 2009 Financing to
finance animal efficacy studies in Acute Radiation Syndrome, the development of
AEOL 10150 and ongoing operations of the Company.
The fair
value of the October 2009 Warrants, the Note Warrants and the Exchange
Warrants was estimated to be $10,585,000 using the Black-Scholes option pricing
model with the following assumptions: dividend yield of 0%; expected volatility
of 93%; risk free interest rate of 2.9%; and an expected life of seven years.
The fair value of the Prior Warrants cancelled on October 6, 2009 was
$3,352,000. The proceeds from the October 2009 Financing were allocated
based upon the relative fair values of the October 2009 Warrants and the
October 2009 Shares. Due to the anti-dilution provisions of the
October 2009 Warrants, the Note Warrants and the Exchange Warrants, these
warrants were deemed to be a liability under current accounting guidance and as
a result the warrant liability was increased by $7,233,000 of which $6,213,000
was recorded as a charge to the Statement of Operations and $1,020,000 of
proceeds from the October 2009 Financing was allocated to the value of the
October 2009 Warrants.
Affiliates
of Xmark Opportunity Partners, LLC were the sole investors in the
October 2009 Financing and, together with the Company, were the sole
participants in the Conversion. Together with its affiliates, Xmark Opportunity
Partners, LLC beneficially owned approximately 71% of the Company’s outstanding
common stock prior to the October 2009 Financing and the Conversion. Xmark
Opportunity Partners, LLC is the sole manager of Goodnow and possesses sole
power to vote and direct the disposition of all securities of the Company held
by Goodnow. Goodnow has the right to designate up to two directors for election
to the Company’s Board of Directors pursuant to the terms of a purchase
agreement between Goodnow and the Company. David C. Cavalier, a current
employee, director and Chairman of the Board of the Company, and Managing
Partner of Xmark Opportunity Partners LLC, is President of Goodnow.
On
December 24, 2009, the Company entered into an amendment (the “Amendment”)
to the October 2009 Purchase Agreement pursuant to which the Company agreed
to lower the conversion price of the Notes from $0.35 per share to $0.28 per
share and as a result, issued to the investors in the Company’s
October 2009 Financing an additional 714,286 shares of the Company’s common
stock upon conversion of the Notes (the “Issuance”). The Agreement was executed
to resolve a misunderstanding regarding one of the financing terms between the
Company and the October 2009 Investors. The Company did not receive any
proceeds from the Issuance. The fair value of the common stock on the date of
issuance was $343,000 and was charged to the Statement of Operations as interest
expense.
On
July 30, 2010, the Company exercised the October 2009 Put Option. As a
result of the exercise, the Company received $1.65 million in gross proceeds
from the investors in exchange for 5,892,857 additional Units (the “Additional
Units”), comprised of an aggregate of 5,892,857 shares of common stock and
warrants to purchase up to an aggregate of 11,785,714 additional shares of
common stock at a purchase price of $0.28 per share.
Net cash
proceeds from the exercise of the October 2009 Put Option were approximately
$1.6 million after legal costs associated with the exercise and subsequent
issuance of stock and warrants.
67
On
August 12, 2010, the Company announced an additional financing with certain
existing investors (the “August 2010 Investors”). Under the terms of the
agreement, the Company received $1 million in gross proceeds in exchange for the
issuance of 2.5 million shares of common stock and warrants to purchase up to
1,875,000 shares at an exercise price of $0.50 per share. The Company also
granted to the August 2010 Investors the option to acquire, collectively,
up to an additional 2,500,000 units, comprised of an aggregate of 2,500,000
shares of common stock and warrants to purchase up to an aggregate of 1,875,000
additional shares of common stock at an exercise price of $0.50 (the “August
2010 Call Option”). In addition, the August 2010 Investors granted to the
Company the option to require these August 2010 Investors, severally and
not jointly, to acquire up to 2,500,000 additional units, less any additional
units acquired under the August 2010 Call Option, at the per additional unit
purchase price of $0.40 (the “August 2010 Put Option”). On December 28, 2010,
the investors exercised their Call Option and the company received $1 million in
proceeds in exchange for 2,500,000 common shares and 1,875,000
warrants.
Dividends
The
Company has never paid a cash dividend on its common stock and does not
anticipate paying cash dividends on its common stock in the foreseeable
future. If we pay a cash dividend on our common stock, we also must
pay the same dividend on an as converted basis on our Series B preferred stock.
In addition, under the terms of the warrants to purchase up to 61,822,749 shares
of our common stock issued to Xmark Opportunity Partners, LLC or its affiliates
(“Xmark”) in three transactions (on each of October 6, 2009, July 30, 2010 and
August 11, 2010), and any additional warrants issued pursuant to the put and/or
option right granted in our August 2010 financing, if we were to pay a dividend
on our common stock, the exercise price of these warrants would be reset from
$0.28 per share or $0.50 per share, as applicable, to $0.01 per share and the
warrant holders would also be entitled receive any such dividend
paid.
Warrants
As of
September 30, 2010, warrants to purchase an aggregate of 66,901,667 shares of
common stock were outstanding. Details of the warrants for common stock
outstanding at September 30, 2010 were as follows:
|
Number of Shares
|
Exercise Price
|
Expiration Date
|
||||||
| 350,000 | $ | 0.28 |
November
2010
|
|||||
| 1,926,668 | $ | 0.28 |
May
2012
|
|||||
| 43,614,285 | $ | 0.28 |
October
2016
|
|||||
| 20,000 | $ | 0.39 |
September
2014
|
|||||
| 50,000 | $ | 0.35 |
May
2011
|
|||||
| 15,000 | $ | 0.50 |
September
2014
|
|||||
| 15,000 | $ | 0.60 |
September
2014
|
|||||
| 7,000,000 | $ | 0.75 |
June
2011
|
|||||
| 11,785,714 | $ | 0.28 |
July
2017
|
|||||
| 1,875,000 | $ | 0.50 |
August
2017
|
|||||
| 50,000 | $ | 0.50 |
May
2011
|
|||||
| 50,000 | $ | 1.00 |
May
2011
|
|||||
| 50,000 | $ | 1.50 |
May
2011
|
|||||
| 50,000 | $ | 2.00 |
May
2011
|
|||||
| 50,000 | $ | 0.38 |
April
2020
|
|||||
| 66,901,667 | $ | 0.34 | ||||||
68
As of
September 30, 2009, warrants for common stock outstanding were as
follows:
|
Number of Shares
|
Exercise Price
|
Expiration Date
|
||||||
| 2,500,000 | $ | 0.28 |
November
2010
|
|||||
| 2,186,668 | $ | 0.28 |
May
2012
|
|||||
| 13,392,857 | $ | 0.35 |
March
2014
|
|||||
| 20,000 | $ | 0.39 |
September
2014
|
|||||
| 50,000 | $ | 0.50 |
May
2011
|
|||||
| 1,000,000 | $ | 0.50 |
August
2013
|
|||||
| 250,000 | $ | 0.50 |
September
2013
|
|||||
| 250,000 | $ | 0.50 |
October
2013
|
|||||
| 250,000 | $ | 0.50 |
November
2013
|
|||||
| 250,000 | $ | 0.50 |
December
2013
|
|||||
| 15,000 | $ | 0.50 |
Setpember
2014
|
|||||
| 15,000 | $ | 0.60 |
September
2014
|
|||||
| 7,000,000 | $ | 0.75 |
June
2011
|
|||||
| 50,000 | $ | 1.00 |
May
2011
|
|||||
| 50,000 | $ | 1.50 |
May
2011
|
|||||
| 50,000 | $ | 2.00 |
May
2011
|
|||||
| 50,000 | $ | 2.50 |
May
2011
|
|||||
| 27,379,525 | $ | 0.46 | ||||||
As of
September 30, 2008, warrants for common stock outstanding were as
follows:
|
Number of Shares
|
Exercise Price
|
Expiration Date
|
||||||
| 50,000 | $ | 0.35 |
May
2011
|
|||||
| 2,500,000 | $ | 0.35 |
November
2010
|
|||||
| 2,186,668 | $ | 0.35 |
May
2012
|
|||||
| 1,000,000 | $ | 0.50 |
August
2013
|
|||||
| 250,000 | $ | 0.50 |
Setpember
2013
|
|||||
| 7,000,000 | $ | 0.75 |
June
2011
|
|||||
| 50,000 | $ | 1.00 |
May
2011
|
|||||
| 50,000 | $ | 1.50 |
May
2011
|
|||||
| 50,000 | $ | 2.00 |
May
2011
|
|||||
| 50,000 | $ | 2.50 |
May
2011
|
|||||
| 410,400 | $ | 2.50 |
April
2009
|
|||||
| 1,641,600 | $ | 4.00 |
April
2009
|
|||||
| 1,759 | $ | 19.9 |
October
2008
|
|||||
| 15,240,427 | $ | 1.02 | ||||||
69
|
Weighted Average
|
|||||||||||||
|
Remaining Contractual
|
Aggregrate Intrinsic
|
||||||||||||
|
Number of Shares
|
Exercise Price
|
Term (in years)
|
Value
|
||||||||||
|
Outstanding
at 9/30/2007
|
11,936,668 | $ | 0.58 |
3.7
years
|
$ | 1,078 | |||||||
|
Granted
|
1,250,000 | $ | 0.50 |
4.9
years
|
|||||||||
|
Exercised
|
- | $ | - | ||||||||||
|
Cancelled
|
- | $ | - | ||||||||||
|
Forfeited
|
- | ||||||||||||
|
Outstanding
at 9/30/2008
|
13,186,668 | $ | 0.57 |
2.9
years
|
$ | 797 | |||||||
|
Granted
|
14,192,857 | $ | 0.36 |
4.5
years
|
$ | 536 | |||||||
|
Exercised
|
- |
|
|||||||||||
|
Cancelled
|
- | $ | - | ||||||||||
|
Forfeited
|
- | ||||||||||||
|
Outstanding
at 9/30/2009
|
27,379,525 | $ | 0.46 |
3.3
years
|
$ | 1,051 | |||||||
|
Granted
|
57,324,999 | $ | 0.29 |
6.2
years
|
$ | 15,634 | |||||||
|
Exercised
|
(260,000 | ) | $ | 0.28 |
1.6
years
|
$ | 47 | ||||||
|
Cancelled
|
(17,542,857 | ) | $ | 0.36 |
3.0
years
|
$ | 3,534 | ||||||
|
Forfeited
|
$ | - |
|
||||||||||
|
Outstanding
at 9/30/2010
|
66,901,667 | $ | 0.34 |
5.5
years
|
$ | 16,278 | |||||||
|
Exercisable
at 9/30/2010
|
66,901,667 | $ | 0.34 |
5.5
years
|
$ | 16,278 | |||||||
H.
Stock-Based Compensation
As an
integral component of a management and employee retention program designed to
motivate, retain and provide incentive to the Company’s management, employees
and key consultants, the Board of Directors approved the 2004 Stock Option Plan
(the “2004 Plan”) and reserved 10,000,000 shares of common stock for issuance
under the 2004 Plan. As of September 30, 2009, 3,964,659 shares were available
to be granted under the 2004 Plan. The exercise price of the incentive stock
options (“ISOs”) granted under the 2004 Plan must not be less than the fair
market value of the common stock as determined on the date of the grant. The
options may have a term up to 10 years. Options typically vest immediately or up
to one year following the date of the grant. Under the Company’s 1994
Stock Option Plan (the “1994 Plan”), incentive stock options or non-qualified
stock options to purchase 2,500,000 shares of Aeolus’ common stock may be
granted to employees, directors and consultants of the Company. As of September 30, 2010, there were no shares available to be
granted under the 1994 Plan. The exercise price of the ISOs granted
under the 1994 Plan must not be less than the fair market value of the common
stock as determined on the date of the grant. The options may have a term up to
10 years. Options typically vest over three years following the date of the
grant.
Below is
a summary of stock option activity for the last three fiscal years ended
September 30th:
70
|
Weighted Average
|
|||||||||||||
|
Remaining Contractual
|
Aggregrate Intrinsic
|
||||||||||||
|
Number of Shares
|
Exercise Price
|
Term (in years)
|
Value
|
||||||||||
|
Outstanding
at 9/30/2007
|
3,873,617 | $ | 2.72 |
7.3
years
|
$ | 2 | |||||||
|
Granted
|
475,000 | $ | 0.35 |
10.0
years
|
|||||||||
|
Exercised
|
- | ||||||||||||
|
Cancelled
|
(113,336 | ) | $ | 0.87 | |||||||||
|
Forfeited
|
- | ||||||||||||
|
Outstanding
at 9/30/2008
|
4,235,281 | $ | 2.50 |
6.7
years
|
$ | 46 | |||||||
|
Granted
|
1,992,750 | $ | 0.33 |
10.0
years
|
117 | ||||||||
|
Exercised
|
- | ||||||||||||
|
Cancelled
|
(51,766 | ) | $ | 32.36 | |||||||||
|
Forfeited
|
- | ||||||||||||
|
Outstanding
at 9/30/2009
|
6,176,265 | $ | 1.58 |
7.0
years
|
$ | 138 | |||||||
|
Granted
|
2,210,000 | $ | 0.40 |
10.0
years
|
363 | ||||||||
|
Exercised
|
- | - | |||||||||||
|
Cancelled
|
(41,361 | ) | $ | 37.06 | - | ||||||||
|
Forfeited
|
(423,000 | ) | $ | 0.62 |
|
41 | |||||||
|
Outstanding
at 9/30/2010
|
7,921,904 | $ | 1.12 |
7.0
years
|
$ | 874 | |||||||
|
Exercisable
at 9/30/2010
|
6,172,743 | $ | 1.32 |
6.2
years
|
$ | 592 | |||||||
Stock
options granted to consultants during fiscal year 2010, 2009 and 2008 were fully
vested when issued or vested over a twelve month period. Stock option
expense for stock options granted to consultants was $73,000, $69,000, and
$77,000 for fiscal year 2010, 2009, and 2008, respectively. For the
fiscal years 2010, 2009 and 2008, all stock options were issued at or above fair
market value of a share of common stock. The weighted-average
grant-date fair value of options granted during fiscal years 2010, 2009 and 2008
was $0.35, $0.30, and $0.35, respectively.
A summary
of the status of non-vested shares for the fiscal years ended September 30
was:
|
Nonvested
at September 30, 2007
|
537,500 | |||
|
Granted
|
475,000 | |||
|
Vested
|
(701,667 | ) | ||
|
Nonvested
at September 30, 2008
|
310,833 | |||
|
Granted
|
1,992,750 | |||
|
Vested
|
(1,003,842 | ) | ||
|
Nonvested
at September 30, 2009
|
1,299,741 | |||
|
Granted
|
2,210,000 | |||
|
Vested
|
(1,715,580 | ) | ||
|
Forfeited
|
(45,000 | ) | ||
|
Nonvested
at September 30, 2010
|
1,749,161 |
The total
deferred compensation expense for outstanding stock options was $603,000 as of
September 30, 2010, which will be recognized over a weighted average period of
ten months. The total fair value of shares vested during fiscal
years 2010, 2009 and 2008 was $527,000, $321,000 and $341,000,
respectively.
71
The
details of stock options for the fiscal year ended September 30, 2010 are
as follows:
|
Options Outstanding
|
Options Exercisable
|
||||||||||||||||||||||
|
Weighted
|
|||||||||||||||||||||||
|
Number
|
Average
|
Number
|
Weighted
|
||||||||||||||||||||
|
Range of
|
Outstanding at
|
Weighted
|
Remaining
|
Exercisable at
|
Average
|
||||||||||||||||||
|
Exercise
|
September 30,
|
Average
|
Contractual
|
September 30,
|
Exercise
|
||||||||||||||||||
|
Prices
|
2010
|
Exercise Price
|
Life (in years)
|
2010
|
Price
|
||||||||||||||||||
| $ | 0.29-$0.32 | 1,526,250 | $ | 0.30 | 8.47 | 1,522,083 | $ | 0.30 | |||||||||||||||
| $ | 0.33-$0.45 | 2,928,500 | $ | 0.40 | 9.44 | 1,183,506 | $ | 0.39 | |||||||||||||||
| $ | 0.55-$0.75 | 768,111 | $ | 0.65 | 6.16 | 768,111 | $ | 0.65 | |||||||||||||||
| $ | 0.78-$0.90 | 794,335 | $ | 0.87 | 5.66 | 794,335 | $ | 0.87 | |||||||||||||||
| $ | 0.91-$1.50 | 1,382,019 | $ | 1.46 | 3.00 | 1,382,019 | $ | 1.46 | |||||||||||||||
| $ | 1.52 | 2,500 | $ | 1.52 | 4.00 | 2,500 | $ | 1.52 | |||||||||||||||
| $ | 1.55-$2.10 | 211,750 | $ | 1.85 | 3.97 | 211,750 | $ | 1.85 | |||||||||||||||
| $ | 2.50-$15 | 237,881 | $ | 6.66 | 2.68 | 237,881 | $ | 6.66 | |||||||||||||||
| 15.50-31.88 | 70,558 | $ | 29.00 | 0.17 | 70,558 | $ | 29.00 | ||||||||||||||||
| $ | 0.29-$31.88 | 7,921,904 | $ | 1.12 | 7.00 | 6,172,743 | $ | 1.32 | |||||||||||||||
Stock-based
compensation expense recognized in the statement of operations is as follows (in
thousands):
|
For the fiscal year ended
|
||||||||||||
|
September 30,
|
||||||||||||
|
2010
|
2009
|
2008
|
||||||||||
|
Research
and Development Expenses
|
56 | 38 | 45 | |||||||||
|
General
and Administrative Expenses
|
492 | 245 | 296 | |||||||||
| 548 | 283 | 341 | ||||||||||
The fair
value of the options associated with the above compensation expense was
determined at the date of the grant using the Black-Scholes option pricing model
with the following weighted average assumptions:
|
For the fiscal year ended September 30,
|
||||||||||||
|
2010
|
2009
|
2008
|
||||||||||
|
Dividend
yield
|
0 | % | 0 | % | 0 | % | ||||||
|
Expected
volatility
|
93 | % | 111 | % | 197% - 198 | % | ||||||
|
Risk-free
interest rate
|
3.07 | % | 3.00 | % | 3.8% - 4.6 | % | ||||||
|
Expected
option life after shares are vested
|
10
years
|
9.3
years
|
10
years
|
|||||||||
I.
Income Taxes
As of
September 30, 2010 and 2009, the Company had federal net operating loss (“NOL”)
carry-forwards of $109,695,000 and $105,303,000, respectively and state
operating loss carry-forwards of $33,106,000 and $28,715,000, respectively. The
use of these federal and state NOL carry-forwards might be subject to limitation
under the rules regarding a change in stock ownership as determined by the
Internal Revenue Code (the “Code”). The Company may have had a change of control
under Section 382 of the Code during fiscal 2004 and 2006; however, a complete
analysis of the limitation of the NOL carry-forwards will not be completed until
the time the Company projects it will be able to utilize such NOLs. The federal
net operating and the state net operating losses began to expire in 2010.
Additionally, the Company had federal research and development carry-forwards as
of September 30, 2010 and 2009 of $3,132,000 and $3,078,000, respectively. The
Company had state research and development carry-forwards as of September 30,
2010 and 2009 of $424,000 and $369,000, respectively.
72
Significant
components of the Company’s deferred tax assets at September 30, 2010 and 2009
consisted of the following (in thousands):
|
2010
|
2009
|
|||||||
|
Net
operating loss carry-forwards
|
$
|
40,223
|
$
|
38,341
|
||||
|
Research
and development credit carry-forwards
|
3,556
|
3,446
|
||||||
|
Accrued
payroll related liabilities
|
2,626
|
2,542
|
||||||
|
Note
discount
|
—
|
322
|
||||||
|
Impairment
on marketable securities
|
—
|
—
|
||||||
|
Total
deferred tax assets
|
46,405
|
44,651
|
||||||
|
Deferred
tax liabilities
|
—
|
—
|
||||||
|
Valuation
allowance for deferred assets
|
(46,405
|
)
|
(44,651
|
)
|
||||
|
Net
deferred tax asset
|
$
|
—
|
$
|
—
|
||||
Due to
the uncertainty surrounding the realization of the favorable tax attributes in
future tax returns, all of the deferred tax assets have been fully offset by a
valuation allowance. The change in the valuation allowance is primarily a result
of the net operating loss carry-forwards.
Taxes
computed at the statutory federal income tax rate of 34% are reconciled to the
provision for income taxes as follows (dollars in thousands):
|
2010
|
2009
|
2008
|
||||||||||
|
Effective
income tax rate
|
0
|
%
|
0
|
%
|
0
|
%
|
||||||
|
United
States Federal income tax at statutory rate
|
$
|
(8,796)
|
$
|
(781
|
)
|
$
|
(1,011
|
)
|
||||
|
State
income taxes (net of federal benefit)
|
(232)
|
(75
|
)
|
(142
|
)
|
|||||||
|
Warrant
expense
|
7,258
|
13
|
40
|
|||||||||
|
Change
in valuation reserves
|
1,703
|
857
|
1,145
|
|||||||||
|
Other
|
67
|
(14
|
)
|
(32
|
)
|
|||||||
|
Provision
for income taxes
|
$
|
—
|
$
|
—
|
$
|
—
|
||||||
On
October 1, 2007, the Company adopted new accounting guidance which altered the
framework for recognizing income tax contingencies. Previously, the focus was on
the subsequent liability recognition for estimated losses from tax contingencies
where such losses were probable and the related amounts could be reasonably
estimated. Under this new guidance, a contingent tax asset (i.e., an uncertain tax
position) may only be recognized if it is more likely than not that it will
ultimately be sustained upon audit. The Company has evaluated its tax positions
for all jurisdictions and all years for which the statute of limitations remains
open and determined that no additional liability for unrecognized tax benefits
and interest was necessary.
J.
Agreements
Duke
Licenses
Aeolus
has obtained exclusive worldwide licenses (the “Duke Licenses”) from Duke
University (“Duke”) to develop, make, have made, use and sell products using
certain technology in the field of free radical and antioxidant research,
developed by certain scientists at Duke. Future discoveries in the
field of antioxidant research from these scientists’ laboratories at Duke are
also covered by the Duke Licenses. The Duke Licenses require Aeolus to use its
best efforts to pursue development of products using the licensed technology and
compounds. These efforts are to include the manufacture or production
of products for testing, development and sale. Aeolus is also obligated to use
its best efforts to have the licensed technology cleared for marketing in the
United States by the U.S. Food and Drug Administration and in other countries in
which Aeolus intends to sell products using the licensed
technology. Aeolus will pay royalties to Duke on net product sales
during the terms of the Duke Licenses, and milestone payments upon certain
regulatory approvals and annual sales levels. In addition, Aeolus is obligated
under the Duke Licenses to pay all or a portion of patent prosecution,
maintenance and defense costs. Unless earlier terminated, the Duke
Licenses continue until the expiration of the last to expire issued patent on
the licensed technology.
National
Jewish Medical and Research Center Agreements
Aeolus
has an exclusive worldwide license (“NJH License”) from National Jewish Health
(“NJH”) to develop, make, have made, use and sell products using certain
technology developed by certain scientists at NJH. The NJH License
requires Aeolus to use commercially reasonable efforts to diligently pursue the
development and government approval of products using the licensed
technology. Aeolus will be obligated to pay royalties to NJH on net
product sales during the term of the NJH License and a milestone payment upon
regulatory approval, if obtained. In addition, Aeolus is obligated
under the NJH License to pay all or a portion of patent prosecution, maintenance
and defense costs. Unless earlier terminated, the NJH License
continues until the expiration of the last to expire issued patent on the
licensed technology.
73
Elan
Corporation, plc
In May
2002, the Company entered into a collaboration transaction with affiliates of
Elan Corporation, plc for the development of the Company’s catalytic antioxidant
compounds as a treatment for tissue damage from cancer radiation and
chemotherapy. Although Elan and the Company terminated this
collaboration in January 2003, the Company will pay Elan a royalty on net sales
of its catalytic antioxidant products sold, if any, for the prevention and
treatment of radiation-induced and chemotherapy-induced tissue
damage.
K.
Quarterly Financial Data (unaudited)
|
First
Quarter
|
Second
Quarter
|
Third
Quarter
|
Fourth
Quarter
|
Total
Year
|
||||||||||||||||
|
(in thousands, except per share amounts)
|
||||||||||||||||||||
|
Fiscal
2010
|
||||||||||||||||||||
|
Total
revenue
|
$
|
—
|
$
|
—
|
$
|
—
|
$
|
—
|
$
|
—
|
||||||||||
|
Net
loss attributable to common stockholders
|
$
|
(15,276
|
)
|
$
|
6,918
|
$
|
(4,623
|
)
|
$
|
(12,888
|
)
|
$
|
(25,869
|
)
|
||||||
|
Basic
net loss per common share attributable to common
stockholders
|
$
|
(0.33
|
)
|
$
|
0.14
|
$
|
(0.10
|
)
|
$
|
(0.24
|
)
|
$
|
(0.53
|
)
|
||||||
|
Diluted
net loss per common share attributable to common
stockholders
|
$
|
(0.33
|
)
|
$
|
0.07
|
$
|
(0.10
|
)
|
$
|
(0.10
|
)
|
$
|
(0.46
|
)
|
||||||
|
Fiscal
2009
|
||||||||||||||||||||
|
Total
revenue
|
$
|
—
|
$
|
—
|
$
|
—
|
$
|
—
|
$
|
—
|
||||||||||
|
Net
loss attributable to common stockholders
|
$
|
(459
|
)
|
$
|
(665
|
)
|
$
|
(426
|
)
|
$
|
(746
|
)
|
$
|
(2,296
|
)
|
|||||
|
Basic
net loss per common share attributable to common
stockholders
|
$
|
(0.01
|
)
|
$
|
(0.02
|
)
|
$
|
(0.01
|
)
|
$
|
(0.03
|
)
|
$
|
(0.07
|
)
|
|||||
|
Diluted
net loss per common share attributable to common
stockholders
|
$
|
(0.02
|
)
|
$
|
(0.02
|
)
|
$
|
(0.01
|
)
|
$
|
(0.02
|
)
|
$
|
(0.07
|
)
|
|||||
L.
Subsequent Events
In
October 2010, we were notified that we had been awarded the maximum amount, of
$244,479, under the QTDP administered by the IRS and the HHS in support of
our development of AEOL 10150 as an MCM for Lung-ARS. In November
2010, we received $244,479 from the IRS as full payment for the Lung-ARS
award.
In
October 2010, we were also notified that we had been awarded the maximum amount,
of $244,479, under the QTDP administered by the IRS and the HHS in support
of our development of AEOL 11207 as a potential treatment for Parkinson’s
Disease. In November 2010, we received $92,406 from the IRS as an
initial payment for the Parkinson’s program award. We expect to
receive the balance of $152,073 after we file our 2010 fiscal year tax return
and submit the request for payment to the IRS.
On
December 1, 2010, the Company entered into a consulting contract with Brian Day,
PhD. Under the terms of the agreement, Dr. Day will serve as Chief
Scientific Officer to the Company and will be entitled to receive a monthly
consulting fee of $11,500 for an initial term of one year. Dr. Day’s monthly
consulting fee will increase to $12,500 when and if, during the term of the
Consulting Agreement, the Company obtains at least $5,000,000 in funding through
either a capital raising transaction, partnership or contract award. In
addition, Dr. Day was granted options to purchase 50,000 shares of common stock
and is eligible for a $30,000 payment and a stock option to purchase an
additional 25,000 shares of common stock if the Company: (a) executes definitive
agreements representing the earliest to occur of: (i) a development or
partnership with another life sciences company for the joint development or
commercialization of any of the Company’s owned or in-licensed patent rights or
(ii) engages in a merger, business combination, reorganization, recapitalization
or other transaction which results in the stockholders of the Company who own at
least 50% of the Company’s voting control immediately prior to such transaction
owning less than 50% of the surviving entity’s voting control immediately after
such transaction, and/or a sale, transfer, lease or other disposition in any
transaction or series of transactions of all or substantially all of the assets
of the Company, or (b) files an Investigational New Drug application for a new
compound in the Company’s drug candidate pipeline with the FDA.
74
On
December 28, 2010, Xmark exercised its “call option” from the August 2010
financing. Under the terms of the option, the Company received $1
million in cash in exchange for the issuance of 2,500,000 shares of common stock
and 1,875,000 warrants to purchase common stock at an exercise price of $0.50
per share.
Management
has evaluated for subsequent events through the date these financial statements
were filed and determined that no other material subsequent events have
occurred.
Item
9. Changes in and Disagreements with Accountants on Accounting and Financial
Disclosure.
None
Item
9A. Controls and Procedures.
As of the
end of the period covered by this Annual Report on Form 10-K, we carried out an
evaluation, under the supervision and with the participation of our management,
including our President and Chief Executive Officer (our Principal Executive
Officer) and Chief Financial Officer (our Principal Financial and Accounting
Officer), of the effectiveness of our disclosure controls and procedures
required by Rule 13a-15 under the Securities Exchange Act of 1934, as amended
(the “Exchange Act”). Based upon that evaluation, our Principal
Executive Officer and Principal Financial and Accounting Officer have concluded
that our disclosure controls and procedures were not effective as of September
30, 2010 to provide reasonable assurance that information required to be
disclosed by us in reports that we file or submit under the Exchange Act is
recorded, processed, summarized and reported within the time periods specified
in SEC rules and forms because of the material weakness discussed
below.
Management’s
Report on Internal Control over Financial Reporting
Our
management is responsible for establishing and maintaining adequate internal
control over financial reporting as defined in Rule 13a-15 (f) and 15d-15 (f) of
the Exchange Act. Our internal control over financial reporting is a
process designed to provide reasonable assurance to our management and Board of
Directors regarding the reliability of financial reporting and the preparation
of financial statements for external purposes in accordance with generally
accepted accounting principles and includes policies and procedures
that:
|
|
¨
|
pertain
to the maintenance of records that, in reasonable detail, accurately and
fairly reflect the transactions and dispositions of our
assets;
|
|
|
¨
|
provide
reasonable assurance that transactions are recorded as necessary to permit
preparation of financial statements in accordance with generally accepted
accounting principles, and that our receipts and expenditures are being
made only in accordance with authorizations of our management and
directors; and
|
|
|
¨
|
provide
reasonable assurance regarding prevention or timely detection of
unauthorized acquisition, use, or disposition of our assets that could
have a material effect on our financial
statements.
|
Because
of its inherent limitations, internal control over financial reporting may not
prevent or detect misstatements, errors or fraud. Also, projections
of any evaluations of effectiveness to future periods are subject to the risk
that controls may become inadequate because of changes in conditions, or that
the degree of compliance with the policies or procedures may
deteriorate.
Our
management assessed the effectiveness of our internal control over financial
reporting as of September 30, 2010. In making this assessment, our
management used criteria set forth by the Committee of Sponsoring Organizations
of the Treadway Commission (“COSO”) in an Internal Control Integrated
Framework. As a result of the lack of segregation of duties and
weaknesses in financial reporting close processes, management
has determined that material weaknesses in internal control over financial
reporting related to the segregation of duties existed as of September 30, 2010,
and based on the criteria set forth by COSO, concluded that our internal control
over financial reporting was not effective as of September 30,
2010.
A
“material weakness,” as defined by the Public Company Accounting Oversight Board
(PCAOB) is a deficiency, or a combination of deficiencies, in internal control
over financial reporting, such that there is a reasonable possibility that a
material misstatement of our annual or interim financial statements will not be
prevented or detected on a timely basis.
To
address the material weakness, management has established mitigating controls to
minimize the potential for material misstatements in the financial
statements. Management has determined that given our size, level of
operations and financial resources, it is not practicable for us to eliminate
the segregation of controls weakness and therefore management has established
mitigating controls to minimize the impact of the lack of segregation of
duties.
This
Annual Report on Form 10-K does not include an attestation report of our
registered public accounting firm regarding internal control over financial
reporting. Management’s report was not subject to attestation by our registered
public accounting firm pursuant to rules of the SEC that permit the Company
to provide only management’s report in this Annual Report on Form
10-K.
75
During
the most recent fiscal quarter, there were no significant changes in our
internal control over financial reporting or in other factors that materially
affected or are reasonably likely to materially affect our internal control over
financial reporting.
Item
9B. Other Information.
None.
PART
III
Certain
information required by Part III is omitted from this Annual Report on Form 10-K
because we expect to file a definitive information statement in connection with
our Written Consent in Lieu of 2011 Annual Meeting of Stockholders (the
“Information Statement”) within 120 days after the end of our fiscal year
pursuant to Regulation 14C promulgated under the Exchange Act, and the
information included therein is incorporated herein by reference to the extent
provided below.
Item
10. Directors, Executive Officers and Corporate
Governance.
The
information set forth under the headings “Proposal
No. 1: Election of Directors,” “Corporate Governance” and
“Section 16(a) Beneficial Ownership Reporting Compliance” in our
Information Statement is incorporated herein by reference. The
information required by this Item 10 concerning our executive officers is set
forth under the heading “Executive Officers” located at the end of Part I Item 1
of this Annual Report on Form 10-K.
Code
of Ethics.
We have
adopted a code of ethics that applies to our principal executive officer,
principal financial officer, principal accounting officer and persons performing
similar functions. We have posted the text of Code of Ethics on our
Internet website at www.aeoluspharma.com.
A copy of the Code of Ethics can also be obtained free of charge by
writing to John L. McManus, Aeolus Pharmaceuticals, Inc., 26361 Crown
Valley Parkway, Suite 150 Mission Viejo, CA 92691.
Item
11. Executive Compensation.
The
information set forth under the headings “Director Compensation” and “Executive
Compensation” in our Information Statement is incorporated herein by
reference.
Item
12. Security Ownership of Certain Beneficial Owners and Management
and Related Stockholder Matters.
The
information set forth under the headings “Security Ownership of Certain
Beneficial Owners and Management” in our definitive Information Statement is
incorporated herein by reference.
Item
13. Certain Relationships and Related Transactions, and Director
Independence.
The
information set forth under the headings “Information Concerning the Board of
Directors and its Committees” and “Certain Related Transactions” in our
Information Statement is incorporated herein by reference.
Item
14. Principal Accounting Fees and Services.
The
information set forth under the heading “Independent Registered Public
Accounting Firm — Fees” in our Information Statement is incorporated herein by
reference.
76
PART
IV
Item
15. Exhibits and Financial Statement Schedules.
(a) The
following financial statement schedules and exhibits are filed as part of this
report or incorporated herein by reference:
(1) Financial
Statement Schedules.
All
financial statement schedules for which provision is made in Regulation S-X are
omitted because they are not required under the related instructions, are
inapplicable, or the required information is given in the financial statements,
including the notes thereto.
(2) Exhibits.
|
Incorporated by Reference To
|
||||||||||
|
Exhibit
Number
|
Description of Document
|
Registrant’s
Form
|
Dated
|
Exhibit
Number
|
Filed
Herewith
|
|||||
|
2.1
|
Agreement
and Plan of Merger and Reorganization dated September 16, 2003 between
Incara, Inc. and Incara Pharmaceuticals Corporation
|
S-4
|
09/19/03
|
2.1
|
||||||
|
3.1
|
Certificate
of Incorporation, as amended
|
10-Q
|
06/30/04
|
3.1
|
||||||
|
3.2
|
Certificate
of Amendment of Amended and Restated Certificate of
Incorporation
|
8-K
|
3/27/06
|
3.1
|
||||||
|
3.3
|
Certificate
of Amendment of Amended and Restated Certificate of
Incorporation
|
8-K
|
10/27/06
|
3.1
|
||||||
|
3.4
|
Bylaws,
as amended
|
8-K
|
10/25/05
|
3.1
|
||||||
|
3.5
|
Certificate
of Designations, Preferences and Rights of Series A Convertible Preferred
Stock of the Company dated November 18, 2005
|
8-K
|
11/23/05
|
3.1
|
||||||
|
4.1
|
Form
of Common Stock Certificate
|
10-Q
|
06/30/04
|
4.1
|
||||||
|
4.2
|
Form
of Series B Preferred Stock Certificate
|
S-4
|
09/19/03
|
4.8
|
||||||
|
4.3
|
Form
of Warrant to Purchase Common Stock dated June 5, 2006.
|
8-K
|
6/5/06
|
10.3
|
||||||
|
4.4
|
Registration
Rights Agreement dated May 22, 2007 by and among the Company and each of
the Purchasers whose names appear on the Schedule attached
thereto.
|
8-K
|
5/22/07
|
4.1
|
||||||
|
4.5
|
Registration
Rights Agreement dated October 6, 2009 by and among the Company and the
investors whose names appear on the signature pages
thereof.
|
8-K
|
10/6/09
|
4.1
|
||||||
|
4.6
|
Form
of Warrant to Purchase Common Stock dated May 22, 2007.
|
8-K
|
5/22/07
|
10.2
|
||||||
|
4.7
|
Form
of Warrant to Purchase Common Stock
|
8-K
|
10/6/09
|
10.2
|
||||||
|
10.1*
|
License
Agreement between Duke University and Aeolus Pharmaceuticals, Inc., dated
July 21, 1995
|
S-1
|
12/08/95
|
10.4
|
||||||
|
10.2
|
Amended
and Restated Limited Liability Company Agreement of CPEC LLC dated July
15, 1999, among CPEC LLC, Intercardia, Inc. and Interneuron
Pharmaceuticals, Inc.
|
8-K
|
07/23/99
|
10.42
|
||||||
|
10.3
|
Assignment,
Assumption and License Agreement dated July 15, 1999, between CPEC LLC and
Intercardia, Inc.
|
8-K
|
07/23/99
|
10.43
|
||||||
|
10.4*
|
License
Agreement dated January 19, 2001 between Incara Pharmaceuticals
Corporation and Incara Development, Ltd.
|
10-Q
|
12/31/00
|
10.59
|
||||||
|
10.5*
|
License
Agreement dated January 19, 2001 between Elan Corporation, plc, Elan
Pharma International Ltd. and Incara Development, Ltd.
|
10-Q
|
12/31/00
|
10.60
|
||||||
77
|
Incorporated by Reference To
|
||||||||||
|
Exhibit
Number
|
Description of Document
|
Registrant’s
Form
|
Dated
|
Exhibit
Number
|
Filed
Herewith
|
|||||
|
10.6
|
Registration
Rights Agreement dated December 21, 2000 among Incara Pharmaceuticals
Corporation, Elan International Services, Ltd. and Elan Pharma
International Ltd.
|
10-Q
|
12/31/00
|
10.62
|
||||||
|
10.7
|
Agreement
and Amendment, effective as of January 22, 2001, by and among Incara
Pharmaceuticals Corporation, Elan International Services, Ltd. and Elan
Pharma International Limited
|
10-Q
|
03/31/01
|
10.64
|
||||||
|
10.8
|
Second
Agreement and Amendment, effective as of January 22, 2001, by and among
Incara Pharmaceuticals Corporation, Elan International Services, Ltd. and
Elan Pharma International Limited
|
10-Q
|
03/31/01
|
10.65
|
||||||
|
10.9
|
Third
Agreement and Amendment, effective as of January 22, 2001, by and among
Incara Pharmaceuticals Corporation, Elan International Services, Ltd. and
Elan Pharma International Limited
|
8-K
|
06/01/01
|
10.66
|
||||||
|
10.10
|
Agreement
and Fourth Amendment, effective February 13, 2002, by and among Incara
Pharmaceuticals Corporation, Elan International Services, Ltd., Elan
Pharma International Limited and Elan Pharmaceutical Investments III,
Ltd.
|
10-Q
|
12/31/01
|
10.75
|
||||||
|
10.11*
|
License
Agreement dated June 25, 1998 between Duke University and Aeolus
Pharmaceuticals, Inc.
|
10-Q
|
03/31/02
|
10.82
|
||||||
|
10.12*
|
License
Agreement dated May 7, 2002 between Duke University and Aeolus
Pharmaceuticals, Inc.
|
10-Q
|
03/31/02
|
10.83
|
||||||
|
10.13*
|
License
Agreement dated November 17, 2000 between National Jewish Medical and
Research Center and Aeolus Pharmaceuticals, Inc.
|
10-Q
|
12/31/00
|
10.56
|
||||||
|
10.14*
|
Securities
Purchase Agreement dated as of May 15, 2002, among Incara Pharmaceuticals
Corporation, Aeolus Pharmaceuticals, Inc., Elan Pharma International
Limited and Elan International Services, Ltd.
|
8-K
|
07/03/02
|
10.84
|
||||||
|
10.15*
|
Development
and Option Agreement dated May 15, 2002, among Elan Pharma International
Limited, Incara Pharmaceuticals Corporation and Aeolus Pharmaceuticals,
Inc.
|
8-K
|
07/03/02
|
10.85
|
||||||
|
10.16
|
Amended
and Restated Registration Rights Agreement dated as of May 15, 2002, among
Incara Pharmaceuticals Corporation, Elan International Services, Ltd. and
Elan Pharma International Limited
|
8-K
|
07/03/02
|
10.86
|
||||||
|
10.17
|
Amendment
No. 1 to License Agreement dated May 14, 2002, between Aeolus
Pharmaceuticals, Inc. and Duke University (amending License Agreement
dated July 21, 1995)
|
8-K
|
07/03/02
|
10.87
|
||||||
|
10.18
|
Amendment
No. 1 to License Agreement dated May 14, 2002, between Aeolus
Pharmaceuticals, Inc. and Duke University (amending License Agreement
dated June 25, 1998)
|
8-K
|
07/03/02
|
10.88
|
||||||
|
10.19
|
Amendment
No. 1 to License Agreement dated May 14, 2002, between Aeolus
Pharmaceuticals, Inc. and National Jewish Medical and Research Center
(amending License Agreement dated November 17, 2000)
|
8-K
|
07/03/02
|
10.89
|
||||||
|
10.20
|
Registration
Rights Agreement dated September 16, 2003 among Incara Pharmaceuticals
Corporation, Incara, Inc. and Goodnow Capital, L.L.C.
|
S-4
|
09/19/03
|
10.101
|
||||||
78
|
Incorporated by Reference To
|
||||||||||
|
Exhibit
Number
|
Description of Document
|
Registrant’s
Form
|
Dated
|
Exhibit
Number
|
Filed
Herewith
|
|||||
|
10.21
|
Letter
dated May 17, 2004 from Elan International Services, Limited and Elan
Pharma International Limited to Incara Pharmaceuticals
Corporation
|
10-Q
|
06/30/04
|
10.106
|
||||||
|
10.22+
|
Aeolus
Pharmaceuticals, Inc. 1994 Stock Option Plan, as amended
|
10-Q
|
06/30/04
|
10.109
|
||||||
|
10.23+
|
Aeolus
Pharmaceuticals, Inc. Amended and Restated 2004 Stock Incentive
Plan
|
S-8
POS
|
3/31/08
|
99.1
|
||||||
|
10.24+
|
Amended
and Restated Employment Agreement dated July 30, 2010 between Aeolus
Pharmaceuticals, Inc. and John L. McManus
|
8-K
|
7/14/06
|
10.1
|
||||||
|
10.25+
|
Letter
Agreement dated July 10, 2006 between Aeolus Pharmaceuticals, Inc. and
McManus & Company, Inc.
|
8-K
|
7/10/06
|
10.2
|
||||||
|
10.26+
|
Form
of Indemnity Agreement
|
8-K
|
2/18/05
|
10.118
|
||||||
|
10.27
|
Terms
of Outside Director Compensation
|
10-K
|
12/17/04
|
10.114
|
||||||
|
10.28+
|
Form
of Incentive Stock Option Agreement
|
10-Q
|
2/8/05
|
10.115
|
||||||
|
10.29+
|
Form
of Nonqualified Stock Option Agreement
|
10-Q
|
2/8/05
|
10.116
|
||||||
|
10.30
|
Subscription
Agreement dated June 5, 2006 by and between the Company and the investors
whose names appear on the signature pages thereof.
|
8-K
|
6/5/06
|
10.1
|
||||||
|
10.31
|
Board
Observer Letter dated June 5, 2006 by and among the Company and Efficacy
Biotech Master Fund Ltd.
|
8-K
|
6/5/06
|
10.6
|
||||||
|
10.32+
|
Consulting
Agreement, dated December 1, 2010, between Aeolus Pharmaceuticals, Inc.
and Brian J. Day
|
8-K
|
12/3/10
|
10.1
|
||||||
|
10.33
|
Registration
Rights Agreement dated August 11, 2010 by and among Aeolus
Pharmaceuticals, Inc. and the investors listed therein
|
8-K
|
8/12/10
|
4.1
|
||||||
|
10.34
|
Securities
Purchase Agreement dated August 11, 2010 by and among Aeolus
Pharmaceuticals, Inc. and the investors listed therein
|
8-K
|
8/12/10
|
10.1
|
||||||
|
10.35
|
Form
of Warrant pursuant to Securities Purchase Agreement dated August 11, 2010
by and among Aeolus Pharmaceuticals, Inc. and the investors listed
therein
|
8-K
|
8/12/10
|
10.2
|
||||||
|
10.36
|
Convertible
Promissory Note dated February 7, 2007 issued by Aeolus Pharmaceuticals,
Inc. to Elan Pharma International Ltd.
|
S-1
|
6/4/07
|
10.43
|
||||||
|
10.37
|
Amendment
No. 1 To Convertible Promissory Note dated February 7, 2009 by and between
Aeolus Pharmaceuticals, Inc. and Elan Pharma International
Limited
|
8-K
|
3/16/09
|
10.1
|
||||||
|
10.38+
|
Form
of Restricted Share Award Agreement
|
S-8
POS
|
3/31/08
|
99.2
|
||||||
|
10.39
|
Securities
Purchase and Exchange Agreement dated October 6, 2009 by and among the
Company and the investors whose names appear on the signature pages
thereof
|
8-K
|
10/6/09
|
10.1
|
||||||
|
14.1
|
Aeolus
Pharmaceuticals, Inc. Code of Ethics for Chief Executive Officer and
Senior Financial Officers, as amended on December 13, 2004
|
8-K
|
12/14/04
|
10.113
|
||||||
|
21.1
|
List
of Subsidiaries
|
X
|
||||||||
|
23.1
|
Consent
of Haskell & White LLP, Independent Registered Public Accounting
Firm
|
X
|
||||||||
|
31.1
|
Certification
of the Principal Executive Officer pursuant to Rule 13a-14(a)
and 15d-14(a)
|
X
|
||||||||
79
|
Incorporated by Reference To
|
||||||||||
|
Exhibit
Number
|
Description of Document
|
Registrant’s
Form
|
Dated
|
Exhibit
Number
|
Filed
Herewith
|
|||||
|
31.2
|
Certification
of the Principal Financial and Accounting Officer pursuant to Rule
13a-14(a) and 15d-14(a)
|
X
|
||||||||
|
32.1
|
Certification
by the Principal Executive Officer and Principal Financial and Accounting
Officer pursuant to 18 U.S.C. 1350 as adopted pursuant to Section 906 of
the Sarbanes-Oxley Act of 2002
|
X
|
||||||||
* The
Company has received confidential treatment of certain portions of this
agreement which have been omitted and filed separately with the U.S. Securities
and Exchange Commission.
+ Indicates
management contract or compensatory plan or arrangement.
80
SIGNATURES
Pursuant
to the requirements of Section 13 or 15(d) of the Securities Exchange Act of
1934, the registrant has duly caused this report to be signed on its behalf by
the undersigned, thereunto duly authorized.
|
AEOLUS
PHARMACEUTICALS, INC.
|
||
|
By:
|
/s/
John L. McManus
|
|
|
John L. McManus
President
and Chief Executive Officer
|
||
|
Date:
|
December
29, 2010
|
Pursuant
to the requirements of the Securities Exchange Act of 1934, this report has been
signed below by the following persons on behalf of the registrant and in the
capacities and on the dates indicated.
|
Signatures
|
Title
|
Date
|
||
|
/s/
John L. McManus
|
President
and Chief Executive Officer
|
December
29, 2010
|
||
|
John
L. McManus
|
(Principal
Executive Officer)
|
|||
|
Chief
Financial Officer, Treasurer and Secretary
|
||||
|
(Principal
Financial and Accounting Officer)
|
||||
|
/s/
David C. Cavalier
|
Chairman
of the Board of Directors
|
December
29, 2010
|
||
|
David
C. Cavalier
|
||||
|
|
Director
|
December
29, 2010
|
||
|
John
M. Farah, Jr., Ph.D.
|
||||
|
|
Director
|
December
29, 2010
|
||
|
Joseph
J. Krivulka
|
||||
|
/s/
Amit Kumar, Ph.D.
|
Director
|
December
29, 2010
|
||
|
Amit
Kumar, Ph.D.
|
||||
|
|
Director
|
December
29, 2010
|
||
|
Michael
E. Lewis, Ph.D.
|
||||
|
/s/
Chris A. Rallis
|
Director
|
December
29, 2010
|
||
|
Chris
A. Rallis
|
||||
|
/s/
Peter D. Suzdak, Ph.D.
|
Director
|
December
29, 2010
|
||
|
Peter
D. Suzdak, Ph.D.
|
81
