Attached files
Table of Contents
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
| x | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended June 30, 2010
OR
| ¨ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15 (d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission File Number: 000-21244
PAREXEL INTERNATIONAL CORPORATION
(Exact name of registrant as specified in its charter)
| Massachusetts | 04-2776269 | |
| (State or other jurisdiction of incorporation or organization) |
(I.R.S. Employer Identification Number) | |
| 195 West Street , Waltham, Massachusetts | 02451 | |
| (Address of principal executive offices) | (Zip Code) | |
Registrant’s telephone number, including area code: (781) 487-9900
Securities Registered Pursuant to Section 12(b) of the Act:
| Title of each class: |
Name of each exchange on which registered: | |
| Common Stock, $.01 par value per share | Nasdaq Global Select Market |
Securities Registered Pursuant to Section 12(g) of the Act:
None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes x No ¨
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ¨ No x
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes x No ¨.
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes ¨ No ¨
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ¨
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act.
| Large Accelerated Filer |
x |
Accelerated Filer |
¨ | |||
| Non-accelerated Filer |
¨ (Do not check if a smaller reporting company) |
Smaller Reporting Company |
¨ | |||
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ¨ No x
The aggregate market value of common stock, $.01 par value per share, held by non-affiliates as of December 31, 2009 was approximately $796.5 million based on the closing price of the registrant’s Common Stock as reported on the Nasdaq Global Select Market on December 31, 2009, the last business day of the registrant’s most recently completed second fiscal quarter. The registrant has assumed that all holders of 10% or more of its Common Stock, if any, are affiliates solely for purposes of calculating the aggregate market value of Common Stock held by non-affiliates. As of August 22, 2010 there were 58,484,834 shares of common stock, $.01 par value per share, outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
Specified portions of the Registrant’s Proxy Statement for the Annual Meeting of Stockholders to be held on December 9, 2010 are incorporated by reference into Part III of this report.
Table of Contents
PAREXEL INTERNATIONAL CORPORATION
2
Table of Contents
This annual report on Form 10-K includes “forward-looking statements” within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), and Section 27A of the Securities Act of 1933, as amended (the “Securities Act”). For this purpose, any statements contained in this report regarding PAREXEL International Corporation’s (“PAREXEL,” the “Company,” “we,” “us,” “ours” or “its”) strategy, future operations, financial position, future revenue, projected costs, prospects, plans, goals, and objectives of management, other than statements of historical facts, are forward-looking statements. The words “anticipates,” “believes,” “estimates,” “expects,” “intends,” “may,” “plans,” “projects,” “will,” “would,” “targets,” “could,” and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. The Company cannot guarantee that it actually will achieve the plans, intentions or expectations expressed or implied in its forward-looking statements. There are a number of important factors that could cause actual results, levels of activity, performance or events to differ materially from those expressed or implied in the forward-looking statements the Company makes. These important factors are described under “Critical Accounting Policies and Estimates” and under “Risk Factors” set forth below. Although the Company may elect to update forward-looking statements in the future, it specifically disclaims any obligation to do so, even if its estimates change, and readers should not rely on forward-looking statements in this document as representing the Company’s views as of any date subsequent to the date of this annual report.
| ITEM 1. | BUSINESS |
GENERAL
PAREXEL is a leading biopharmaceutical services company, providing a broad range of expertise in clinical research, medical communications, consulting, and advanced technology products and services to the worldwide pharmaceutical, biotechnology, and medical device industries. Our primary objective is to provide quality solutions for managing the biopharmaceutical product lifecycle with the goal of reducing the time, risk, and cost associated with the development and commercialization of new therapies. Since our incorporation in 1983, we have developed significant expertise in processes and technologies supporting this strategy. Our product and service offerings include: clinical trials management, data management, biostatistical analysis, medical communications, clinical pharmacology, patient recruitment, regulatory and product development consulting, health policy and reimbursement, performance improvement, industry training and publishing, medical imaging services, ClinPhone® randomization and trial supply management services (“RTSM”), electronic data capture systems (“EDC”), clinical trial management systems (“CTMS”), web-based portals, systems integration, patient diary applications, and other drug development services. We believe that our comprehensive services, depth of therapeutic area expertise, global footprint and related access to patients, and sophisticated information technology, along with our experience in global drug development and product launch services, represent key competitive strengths.
Our services complement the research and development (“R&D”) and marketing functions of pharmaceutical, biotechnology, diagnostics, and medical device companies. Through our clinical research and product launch services, PAREXEL seeks to help clients maximize the return on their significant investments in research and development by reducing the time, risk, and cost of clinical development and launch of new products. For large pharmaceutical and biotechnology companies, outsourcing these types of services to PAREXEL provides those companies with a high quality, variable cost alternative to the fixed costs associated with internal drug development. In addition, these large companies can benefit from PAREXEL’s technical resource pool, broad therapeutic area expertise, other advisory services, and global infrastructure, all of which are designed to expedite parallel, multi-country clinical trials and accelerate time-to-market. For smaller companies, PAREXEL provides access to expertise and a virtual and global network that enables them to develop their new drugs. Our vision is to integrate and build critical mass in the complementary businesses of clinical research, medical communications, drug development and process optimization consulting, as well as related information technology products and integration services. Our goal is to provide significant benefits to sponsor clients through this strategy, namely, a faster and less expensive development and launch process, as well as a clinical development strategy and expertise that support the marketing strategy for new medical products. We believe that the outsourcing of these services has increased in the past and should continue to increase in the future because of several factors, which are placing increased pressure on clients. These factors include the need to more tightly manage costs, capacity limitations, reductions in exclusivity periods, the desire to speed up patient recruitment and reduce development time, increased globalization of clinical trials, productivity issues, upcoming patent expirations, more stringent government regulations, and pricing pressure. With increased levels of investment continuing to be required and with development times being extended, we believe these trends will continue to create opportunities for companies like PAREXEL that are focused on improving the efficiency of the drug development process. Moreover, many of our clients are reassessing how they conduct their R&D activities and are now engaging in outsourcing at a more strategic level. One consequence of this reassessment is that they have started to concentrate their outsourced clinical development activities with a smaller number of providers. We believe that our broad range of offerings, our global presence, our information technology solutions, and our expertise in clinical drug development well position us to participate in these strategic partnerships.
3
Table of Contents
PAREXEL is one of the largest biopharmaceutical services companies in the world, based upon annual service revenue. Headquartered near Boston, Massachusetts, we manage 70 locations and have approximately 9,720 employees throughout 54 countries around the world. We have operations in major healthcare markets around the world, including the United States (“U.S.”), Canada, China, Taiwan, Japan, Singapore, Korea, Malaysia, the Philippines, Thailand, Indonesia, Germany, the United Kingdom (“U.K.”), France, Italy, Spain, Sweden, Australia, South Africa, Argentina, Brazil, Peru, Chile, Mexico, Israel, Norway, Belgium, The Netherlands, Denmark, Finland, India, Russia, Poland, the Czech Republic, Lithuania, Hungary, Croatia, Serbia, Romania, and Ukraine. During Fiscal Year 2010, we derived 65.4% of our service revenue from international operations and 34.6% from the United States. Breakdowns of service revenue by geographic region for previous years can be found in Note 16 to the consolidated financial statements included in Item 8 of this annual report. PAREXEL was incorporated in 1983 as a regulatory affairs consulting firm and is a Massachusetts corporation. Josef H. von Rickenbach, Chairman of the Board and Chief Executive Officer of PAREXEL, was a co-founder. Since our inception, we have executed a focused growth strategy embracing internal expansion as well as strategic acquisitions to expand or enhance our portfolio of services, geographic presence, therapeutic area knowledge, information technology capabilities, and client relationships.
Acquisitions have been, and may continue to be, an important component of PAREXEL’s growth strategy. In August 2008, we completed the acquisition of ClinPhone plc (“ClinPhone”), a London Stock Exchange-traded company incorporated in England and Wales, for approximately $190 million. ClinPhone’s strong clinical technology offering was combined with our Perceptive Informatics business segment to provide an extensive line of products and services throughout the entire clinical development lifecycle. Biopharmaceutical companies have increasingly requested technology solutions and expertise to support the full range of clinical development activities while improving the speed and efficiency of clinical programs. We believe that the broad technological offering that we now provide gives clients a stronger, more comprehensive suite of clinical information technologies.
On February 11, 2008, our Board of Directors approved a two-for-one stock split. The record date for the stock split was February 22, 2008. The stock split was completed on March 3, 2008. All share and per share amounts for all periods presented have been adjusted to reflect the effect of this stock split.
DESCRIPTION OF BUSINESS
We provide a broad range of expertise in clinical research, medical communications, consulting and informatics and advanced technology services to the worldwide pharmaceutical, biotechnology, and medical device industries. We manage the company in three business segments: Clinical Research Services (“CRS”), PAREXEL Consulting and Medical Communications Services (“PCMS”), and Perceptive Informatics (“Perceptive”).
CRS constitutes our core business and includes all phases of clinical research from “first-in-man” trials, where a medicinal entity is tested on human subjects for the first time, through post-marketing studies, after approval by the presiding regulatory body (for example, FDA, EMEA or others). CRS service offerings include clinical trials management and biostatistics, data management, clinical logistics, and clinical pharmacology, as well as related medical advisory, patient recruitment, and investigator site services.
PCMS provides technical expertise and advice in such areas as drug development, regulatory affairs, and good manufacturing practice (“GMP”) compliance consulting. In addition, PCMS provides a full spectrum of market development, product development, and targeted communications services in support of product launch. PCMS consultants also identify alternatives and propose solutions to address clients’ product development, registration, and commercialization issues. Additionally, PCMS provides reimbursement and market access (“RMA”) services.
Perceptive provides information technology solutions designed to improve clients’ product development processes. Perceptive’s portfolio of products and services includes medical imaging services, ClinPhone RTSM, CTMS, EDC, web-based portals, systems integration, and patient diary applications.
The revenue generated by each of our business segments for each of the last three fiscal years is described below under the headings for each segment. The gross profit of each segment for each of the last three fiscal years are described in Note 17 to the consolidated financial statements included in Item 8 of this annual report.
CLINICAL RESEARCH SERVICES (CRS)
Our CRS business segment provides clinical trials management and biostatistics, data management and clinical pharmacology, as well as related medical advisory and investigator site services. This segment generated revenue of $870.7 million, or 77.0%, of our consolidated service revenue in Fiscal Year 2010, $804.2 million, or 76.5%, of our consolidated service revenue in Fiscal Year 2009, and $745.6 million, or 77.3%, of our consolidated service revenue in Fiscal Year 2008.
4
Table of Contents
Our CRS business segment offers complete services for the design, initiation and management of clinical trials programs, a critical element in obtaining regulatory approval for biopharmaceutical products. We have performed services in connection with trials in most therapeutic areas, including Oncology, Cardiology, Infectious Diseases, Neurology, Allergy/Immunology, Endocrinology/Metabolism, Gastroenterology, Obstetrics/Gynecology, Orthopedics, Pediatrics, Psychiatry, and Transplantation. Our multi-disciplinary clinical trials group examines a product’s existing preclinical and clinical data to design clinical trials to provide evidence of the product’s safety and efficacy.
Our CRS business segment can manage many aspects of clinical trials including: study and protocol design; Case Report Form (“CRF”) design of paper or electronic questionnaires used in clinical research; site and investigator recruitment; patient enrollment; study monitoring and data collection; data analysis; report writing; and medical services.
Clinical trials are monitored and conducted by CRS in adherence with Good Clinical Practice (“GCP”). The design of efficient CRFs, detailed operations manuals, and site monitoring by our clinical research associates seek to ensure that clinical investigators and their staff follow established study protocols. We have adopted standard operating procedures that are intended to satisfy regulatory requirements and serve as a tool for controlling and enhancing the quality of our worldwide clinical services.
Clinical trials represent one of the most expensive and time-consuming parts of the overall biopharmaceutical product development process. The information generated during these trials is critical to gaining marketing approval from the United States Food and Drug Administration (the “FDA”), the European Medicines Agency based on the recommendation of the Committee for Medicinal Products for Human Use, and other comparable regulatory agencies as well as market acceptance by clinicians, patients, and third-party payors. CRS clinical trial management services involve many phases of clinical trials, including Phases I, II, III, and IV. See “Government Regulations” below for additional information regarding processes involved in clinical trials.
Early Phase – The Early Phase group of CRS encompasses the early stages of clinical testing, when a product is first evaluated to assess the potential safety and efficacy of the product. These tests vary from “first-in-man” to “dose-ranging” to “proof of concept” studies in Phases I and IIa of development. The Early Phase group of CRS offers clients a one-stop service where studies are performed in healthy volunteers as well as in patients of various disease populations. The support services include project and program management, drug development consulting, medical writing, handling of investigational products, data management, biostatistical and bioanalytical services. Our international network of Early Phase operations includes operations in Berlin, Germany; Baltimore, Maryland (U.S.); Glendale, Culver City and Paramount, California (U.S.); Bloemfontein, George and Port Elizabeth, South Africa; and Harrow, U.K. The bioanalytical laboratory which performs drug analyses in accordance with Good Laboratory Practices (“GLP”), a system of managed controls for laboratory and research organizations to ensure the consistency and reliability of results, is located in Bloemfontein. With these locations, the Early Phase group offers clinical pharmacology services (including bioanalytical services) with a total of 580 dedicated beds (cooperating partners not included) on three continents.
Phase II-III/PACE – The Phase II-III/Peri Approval Clinical Excellence (“PACE”) group of CRS (formerly referred to as “Late Phase”) encompasses the later stages of clinical testing. Through this CRS unit, we assist clients with one or more of the following aspects of clinical trials as described below. CRS performs both full-service and single- or multi-service trials. As a result, our involvement may range from being involved in just one aspect of a clinical trial to all aspects of a clinical trial. These services include the following, the majority of which are also provided by our Early Phase group:
| • | Study Protocol Design – The protocol defines, among other things, the medical issues a study seeks to examine and the statistical tests that will be conducted. Accordingly, the protocol specifies the frequency and type of laboratory and clinical measures that are to be tracked and analyzed, the number of patients required to produce a statistically valid result, the period of time over which they must be tracked and the frequency and dosage of drug administration. The study’s success depends on the protocol’s ability to predict correctly the requirements of the regulatory authorities and to generate data that will satisfy those requirements. |
| • | CRF Design – Once the study protocol has been finalized, a CRF must be developed. The CRF is the critical source document for collecting the necessary clinical data as dictated by the study protocol. It may change at different stages of a trial. CRFs for one patient in a given study may consist of 100 or more pages. |
| • | Site and Investigator Recruitment – The product under investigation is administered to patients usually by third-party physicians, serving as independent contractors, referred to as investigators, at hospitals, clinics, or other locations, referred to as clinical sites. Medical devices are implemented or tested by investigators in similar settings. Potential investigators may be identified and solicited by the product sponsor. A significant portion of a trial’s success depends on the successful identification and recruitment of experienced investigators with an adequate base of patients who satisfy the requirements of the study protocol. We have access to several thousand investigators who have conducted clinical trials for us. We provide additional services at the clinical investigator site to assist physicians and expedite the clinical research process. |
5
Table of Contents
| • | Patient Enrollment – The investigators, usually with our assistance, find and enroll patients suitable for the study. The speed with which trials can be completed is significantly affected by the rate at which patients are enrolled. Prospective patients are required to review information about the drug and its possible side effects, and sign an informed consent form to record their knowledge and acceptance of potential side effects. Patients also undergo a medical examination to determine whether they meet the requirements of the study protocol. Patients then receive the investigational product or a control (for example, a placebo) and are examined by the investigator as specified by the study protocol. Investigators are responsible for administering the products to patients, as well as examining patients and conducting necessary tests. |
| • | Study Monitoring and Data Collection – As patients are examined and tests are conducted in accordance with the study protocol and applicable regulatory requirements, data are recorded on CRFs, either electronically or paper-based. CRFs are transmitted electronically from study sites or collected by specially trained persons known as clinical monitors. Monitors visit sites regularly to ensure that the CRFs are completed correctly and to verify that the study has been conducted in compliance with the protocol and GCP. The monitors send completed CRFs to the study coordination site, where the CRFs are reviewed for consistency and accuracy before their data are entered into an electronic database. We offer several EDC technologies, which significantly enhance both the quality and timeliness of clinical data capture and collection while achieving significant efficiency savings. Our study monitoring and data collection services are designed to comply with the adverse events reporting guidelines and related regulatory requirements of the FDA and other relevant regulatory agencies. As much as 90% of new trials are EDC-based. |
| • | Data Management – Our data management professionals provide a broad array of services to support the accurate collection, organization, validation, and analysis of clinical data. For instance, they assist in the design of CRFs and investigator training manuals to ensure that data are collected in an organized and consistent format in compliance with the study protocol and all applicable regulatory requirements. Databases are designed according to the analytical specifications of the project and the particular needs of the client. Prior to data entry, our personnel screen the data to detect errors, omissions, and other deficiencies in completed CRFs. The use of scanning and imaging of the CRFs and the use of EDC technologies to gather and report clinical data expedites data exchange while minimizing data collection errors by permitting the verification of data integrity in a more timely manner. After the data is entered, the data management team performs an array of data abstraction, data review, medical coding, serious adverse event reconciliations, loading of electronic data (such as laboratory data), database verification, and editing and resolution of data problems. The data is then submitted to the sponsor in a customized format prescribed by the sponsor. Our CRS business segment has extensive experience throughout the world in the creation of scientific databases for all phases of the drug development process, including the creation of customized databases to meet client-specific formats, integrated databases to support new drug application (“NDA”) and equivalent submissions and databases created and maintained in compliance with FDA, European, Asian and other regulatory specifications and requirements. |
| • | Biostatistics and Programming – Our biostatistics professionals assist clients with all phases of drug development, including biostatistical consulting, database design, data analysis, and statistical reporting. These professionals develop and review protocols, design appropriate analysis plans, and design report formats to address the objectives of the study protocol as well as the client’s individual objectives. Working with programming staff, biostatisticians perform appropriate analyses and produce tables, graphs, listings, and other applicable displays of results according to an analysis plan. Our CRS business segment biostatisticians may also represent clients during panel hearings at the FDA and other regulatory agencies. |
| • | Report Writing – A description of the study conducted, along with the statistical analysis of data collected during the trial and other clinical data are presented and summarized in a final report generated for inclusion in a regulatory document. We assist clients with writing reports for inclusion in these documents. |
| • | Medical Services – Throughout the course of a development program, our physicians provide a wide range of medical research and consulting services to improve the efficiency and quality of clinical research, including medical supervision of clinical trials, medical monitoring of patient safety, review and reporting of adverse events, medical writing, and strategy and product development. Our medical services professionals also provide lifecycle drug safety services combining operational pharmacovigilance and pharmacovigilance consulting. Operational pharmacovigilance capabilities cover all phases of clinical development and drug safety for marketed products. |
6
Table of Contents
| • | Project Management – Throughout the entire spectrum of activities described above, our CRS segment provides project management services. These services entail providing overall leadership to our project team, acting as the main client liaison, project planning, managing progress against study goals and deliverables, budget management, progress and metrics reporting, and issue resolution. These project management services are offered on all types of trials – single-service, multi-service, or full-service. |
| • | Clinical Logistics – In association with the clinical trials we conduct, we offer a full range of clinical logistics services that include coordinating investigational drug supply manufacturing, managing import/export requirements, labeling, warehousing, distribution, and inventory control (including the return and destruction of unused trial medication, lab services, and ancillary supplies). |
PAREXEL CONSULTING AND MEDICAL COMMUNICATIONS SERVICES (PCMS)
Our PCMS segment provides technical expertise and advice in such areas as drug development, regulatory affairs, reimbursement, and GMP compliance. It also provides a full spectrum of market development, product development, and targeted communications services in support of product launch. Our PCMS consultants identify alternatives and propose solutions to address client issues associated with product development, registration, and commercialization. Our PCMS unit also provides RMA consulting services. Service revenue from the PCMS business segment represented $121.6 million, or 10.8%, of consolidated service revenue in Fiscal Year 2010, $121.8 million, or 11.6%, of consolidated service revenue in Fiscal Year 2009, and $129.8 million, or 13.5%, of consolidated service revenue in Fiscal Year 2008. We conduct our PCMS operations through four groups:
| • | Integrated Product Development Consulting, |
| • | Strategic Compliance Consulting, |
| • | Medical Communications Services, and |
| • | Reimbursement & Market Access. |
Integrated Product Development Consulting – Our Integrated Product Development (“IPD”) consulting group provides comprehensive product development and regulatory consulting services for pharmaceutical, biotechnology, and medical device companies in major jurisdictions in the U.S., Europe, Japan and emerging markets in Asia, Middle East and North Africa, and Latin America. These services include drug and device development and regulatory strategy design, scientific and technical evaluation, writing and review services, regulatory application (both for clinical trials and for marketing authorizations in dozens of countries) preparation and review, regulatory training for client personnel, and expert liaison with the FDA and other regulatory agencies around the world. Our IPD consulting group works closely with clients to design product development and regulatory strategies and comprehensive registration programs. Our product development and regulatory experts include individuals who have joined us from the biopharmaceutical industry and regulatory agencies such as the FDA and agencies in the UK, Germany, The Netherlands, and France. Our experts review existing published literature and regulatory precedents, evaluate the scientific and technical data of a product, assess the competitive and regulatory environment, identify deficiencies, and define the steps necessary to obtain regulatory approvals in the most expeditious manner. Through these services, we help our clients obtain regulatory approval for particular products or product lines in markets around the world.
Strategic Compliance Consulting – Our Strategic Compliance group offers a range of specialized clinical and manufacturing consulting services for clients in the life sciences industry. These services are designed to help pharmaceutical, biotechnology, and medical device companies achieve and maintain regulatory compliance, product quality, and process excellence. These services include clinical and manufacturing compliance strategy, assistance with regulatory agency enforcement issues, risk management, GCP, GLP and current GMP audits, pre-approval inspection readiness, process optimization, organizational alignment, and training. Our Strategic Compliance group offers its clients experienced regulatory and industry professionals—formerly from the FDA, or from the Quality Departments of major biotech, pharmaceutical, and medical device companies.
Medical Communications Services – Our Medical Communications Services (“MedCom”) group assists biopharmaceutical clients in their efforts to achieve optimal market penetration for their products worldwide through expert medical communications and publications services. MedCom utilizes its expertise in strategic consultancy, market and competitive landscaping, publications planning, scientific writing, managed markets, and regulatory compliance to provide effective and compliant scientific communications to a diverse audience of provider, payer, and patient advocacy group stakeholders. An integrated communications plan can detail external and internal strategies, including communications objectives, target audiences, communications priorities and timing, key messages, key meetings and events, and target publications and media. MedCom supports marketing communication objectives across a broad spectrum of media from publications through interactive technologies. Other services include planning of meetings and exhibits in premier scientific meetings and symposia.
7
Table of Contents
Reimbursement & Market Access – Our Reimbursement and Market Access (“RMA”) group offers clients the ability to understand how changing marketplace dynamics may impact product reimbursement, patient access and commercial success. Our professionals assist biopharmaceutical and medical device companies in developing reimbursement strategies and tactical programs to support products during all phases of development. Our services are designed to provide workable solutions to complex challenges clients face based on emerging payer policy. We provide services in the areas of comparative effectiveness, cost effectiveness analysis, dossier development, budget impact modeling, and public, private and managed markets strategy development. Our services also include designing and implementing reimbursement support help lines and patient assistance programs.
PERCEPTIVE INFORMATICS (PERCEPTIVE)
Our Perceptive segment provides information technology solutions designed to improve the product development processes of our clients. Perceptive’s portfolio of products and services include medical imaging services, ClinPhone RTSM, CTMS, EDC, web-based portals, systems integration, and patient diary applications. Service revenue from the Perceptive business represented $138.7 million, or 12.2%, of consolidated service revenue in Fiscal Year 2010, $124.7 million, or 11.9%, of consolidated service revenue in Fiscal Year 2009, and $88.8 million, or 9.2%, of consolidated service revenue in Fiscal Year 2008.
Through the acquisition of ClinPhone in August 2008, Perceptive became one of the industry’s largest clinical technology providers. Perceptive offers broad and comprehensive access to clinical information technologies and resources, providing clients and service providers with the benefits of an extensive line of products and services throughout the entire clinical development lifecycle.
ClinPhone Randomization & Trial Supply Management (ClinPhone RTSM) – Perceptive provides automated randomization and logistics management through its ClinPhone RTSM solutions. Our services include both Interactive Voice Response (“IVR”) and Interactive Web Response (“IWR”) technologies.
ClinPhone RTSM solutions have been used in over 2,800 clinical trials, helping our clients achieve treatment group balance, eliminate selection bias, and limit the predictability of treatment allocations. This is all designed to comply with the latest regulatory requirements. ClinPhone RTSM allows effective real-time implementation of randomization algorithm modifications required for adaptive trial designs.
Medical Imaging Services – Perceptive offers products and services that allow our clients to apply and manage medical imaging in clinical trials. Clinical study sponsors increasingly rely on imaging as a surrogate endpoint in support of efficacy and safety. Our therapeutic and imaging experts provide a range of capabilities in the application of imaging techniques from early clinical development through peri-approval studies. These services include:
| • | Standardization of imaging and image management at investigative sites |
| • | Image collection at a central location and management of the blinding process |
| • | Development of independent review charters for review and approval by regulatory authorities |
| • | Subcontracting independent reviewers and training these reviewers on the assessment criteria and reviewer roles and responsibilities |
| • | Management of the logistical processes involved in the independent review |
Clinical Trial Management System (CTMS) – We offer CTMS solutions to assist biopharmaceutical companies with the complex process of planning and managing clinical trials. The two software packages that we provide are the IMPACT® and TrialWorks™ Clinical Trail Management Systems. The IMPACT solution provides top global pharmaceutical companies with a flexible solution that includes breadth and depth of features, while the TrialWorks solution is particularly well-suited to the emerging biopharmaceutical companies that demand rapid implementation and low cost.
Electronic Data Capture (EDC) – DataLabs® EDC is one of the industry’s first single data management systems that unifies the functionality of paper data entry (PDE) with the flexibility of electronic data capture (EDC). DataLabs EDC is able to combine data collected on paper with data collected electronically into one easy-to-use electronic clinical data management platform. The collected information feeds into a single database providing clients with fully integrated data. With DataLabs EDC, users are able to design a study, collect data using either method and then clean and manage that data using a single system. DataLabs EDC is based on Microsoft® connected technology and servers, providing end-users with an intuitive, familiar, and easy-to-use experience.
Electronic Patient Reported Outcomes (ePRO) – Patient self-reported data is increasingly playing a key part in efficacy and quality of life assessment, patient recruitment, symptom and safety information and medical compliance monitoring. Our ePRO solutions provide the flexibility to choose between the most commonly used ePRO methods, IVR, Web, PDA and Tablet:
| • | IVR (Interactive Voice Response) – Our leading IVR platform enables ePRO delivery using the subject’s own telephone, making it highly cost-effective and simple to deploy; |
8
Table of Contents
| • | Web/IWR (Interactive Web Response) – The web offers all of the advantages and benefits of IVR as subjects use any PC connected to the Internet to securely access the ePRO application; and |
| • | PDA/Tablet – Depending on the specific characteristics of the protocol, a device-based solution may be best suited for a study. |
E-Clinical Technology Services (eCTS) – Perceptive provides leading solutions to integrate systems and processes to help companies simplify the concurrent use of the multiple technologies involved in clinical trials. Perceptive’s integrations are delivered by our dedicated eCTS experts who have an in-depth understanding of advanced technologies, clinical development processes and validated system integrations.
Perceptive’s integration solutions and services include our Clinical Technology Integration Platform (CTIP), which is a unique environment designed to facilitate seamless two-way exchange of data across different systems via reliable and repeatable integrations. Through our CTIP and dedicated eCTS staff, we can support most integration requirements including validated integrations between our hosted products and key third-party hosted technology solutions.
E-Clinical Suite – Perceptive offers an eClinical suite that is the unique combination of our leading products and services: Perceptive Portal™, DataLabs® EDC, ClinPhone® RTSM, IMPACT® and TrialWorks™ CTMS applications, and ePRO and medical imaging services. We deliver applications that seamlessly integrate and tightly interoperate to increase value and utility to our clients.
INFORMATION TECHNOLOGY
We are committed to investing in information technology designed to help us to provide high quality services, competitive, and cost-effective, client-facing solutions and well-managed internal resources. We have built our information technology solutions by developing proprietary technology as well as purchasing and integrating commercially available information technology solutions that address critical aspects of our business, such as project proposals/budget generation, time information management, revenue and resource forecasting, clinical data entry and management, clinical trial management, project management, quality management, and procurement/expense processing.
We maintain an internal information technology group that is responsible for technological planning, applications development, program management, technical operations, and management of our worldwide computer infrastructure and voice and data networks. Our information systems are designed to function in support of and reinforce all of our policies and procedures while enabling us to respond to the multiple needs of our different clients and regulatory systems. Our systems also enable us to respond quickly to client inquiries regarding progress on projects and, in some cases, to gain direct access to client data on client owned systems.
SALES AND MARKETING
Our sales and marketing personnel carry out our global business development activities. In addition to significant selling experience, most of these individuals have technical and/or scientific backgrounds. Our senior executives and project team leaders also participate in maintaining key client relationships and engaging in business development activities.
Each of our three business segments has a business development team that focuses on its particular market segment. While all teams may work with the same client companies, the individual clients they work with within PAREXEL can vary. In many cases, however, the business segment selling teams work together in order to provide clients with the most appropriate service offering to meet their needs. Moreover, we have developed strategic account management teams to provide clients with a single point of contact and to facilitate cross-selling opportunities.
Each business development employee is generally responsible for a specific client segment or group of clients and for strengthening and expanding an effective relationship with that client. Each individual is responsible for developing his or her client base on our behalf, responding to client requests for information, developing and defending proposals, and making presentations to clients.
Our business development group is supported by our marketing team. Our marketing activities consist primarily of market information development and analysis, strategic planning, competitive analysis, brand management, collateral development, participation in industry conferences, advertising, e-marketing, publications, and website development and maintenance. The marketing team focuses both on supporting the individual business development teams for their specific market segments as well as promoting an integrated marketing strategy and communications plan for PAREXEL as a whole.
9
Table of Contents
CLIENTS
We have in the past derived, and may in the future derive, a significant portion of our service revenue from a core group of major projects or clients. Concentrations of business in the biopharmaceutical services industry are common and we expect to continue to experience such concentration in future years. Our five largest clients accounted for 27%, 28% and 31% of our consolidated service revenue, for the three fiscal years ending on June 30, 2010, 2009, and 2008, respectively. No single client accounted for 10% or more of consolidated service revenue in any of Fiscal Years 2010, 2009 or 2008. However, we expect that client concentrations will rise in the future in conjunction with our increasing number of strategic partnerships.
BACKLOG
Backlog represents anticipated service revenue from work not yet completed or performed under signed contracts, letters of intent, and, in some cases, verbal commitments which are supported by written communications. Once work commences, revenue is generally recognized over the life of the contract as services are provided. Backlog at June 30, 2010 was $2,680.5 million, compared with $2,176.4 million at June 30, 2009. Subject to the matters addressed in the following paragraph, we anticipate that approximately $1,286.6 million of the backlog as of June 30, 2010 will be recognized as service revenue in Fiscal Year 2011.
We believe that our backlog as of any date is not necessarily a meaningful predictor of future results. Projects included in backlog are subject to termination, revision, or delay. As detailed more fully in the “Risk Factors” section of this annual report, clients terminate, delay, or change the scope of projects for a variety of reasons including, among others, the failure of products being tested to satisfy safety requirements, unexpected or undesirable clinical results of the product, clients’ decision to forego a particular study, insufficient patient enrollment or investigator recruitment, or production problems resulting in shortages of the drug. Additionally, we have been entering into an increasing number of strategic partnerships. As a result, we may see an increased number of delays and/or cancellations impacting the conversion of backlog into revenue. This trend may continue in the future. Generally, our contracts can be terminated upon thirty to sixty days notice by the client. We are typically entitled to receive certain fees and, in some cases, a termination fee for winding down a delayed or terminated project.
COMPETITION
We compete with other biopharmaceutical services companies and other clinical research organizations (“CROs”) that provide one or more of the services currently being offered by us. Some of the larger biopharmaceutical services companies, such as Quintiles Transnational Corporation, Covance Inc., Pharmaceutical Product Development Inc., Kendle International Inc., and Icon plc, offer services that compete directly with our services at many levels.
We believe that the synergies arising from integrating the products and services offered by our different business units, coupled with our global infrastructure (and resulting rapid access to diverse patient populations), technology products and services, and depth of expertise and experience differentiate us from our competitors. Although there are no guarantees that we will continue to do so, we believe that we compete favorably in all of our business areas and segments, as more fully described below:
CRS
The clinical outsourcing services industry is very fragmented, with several hundred providers offering varying levels of service, skills, and capabilities. Our CRS group primarily competes against in-house departments of pharmaceutical companies, other full service biopharmaceutical services companies, small specialty CROs, and to a small extent, universities, teaching hospitals, and other site organizations. The primary competitors for our CRS business include Quintiles Transnational Corporation, Covance Inc., Pharmaceutical Product Development Inc., Kendle International Inc., and Icon plc.
CRS generally competes on the basis of:
| • | a broad international presence with strategically located facilities; |
| • | the ability to organize and manage large-scale clinical trials on a global basis; |
| • | the ability to recruit investigators and patients expeditiously; |
| • | medical and scientific expertise in a specific therapeutic area; |
| • | quality of services; |
| • | breadth of services; |
10
Table of Contents
| • | the ability to integrate information technology with systems to improve the efficiency of clinical research; |
| • | previous experience with a client or a specific therapeutic area; |
| • | the ability to manage large and complex medical databases; |
| • | the ability to provide statistical and regulatory services; |
| • | financial strength and stability; and |
| • | price. |
We believe CRS’s key competitive strengths are its global footprint and related rapid access to diverse patient populations, therapeutic expertise, technological expertise and its experience in global drug development.
PCMS
Our PCMS segment competes with a large and diverse group of specialty service providers, including major consulting firms with pharmaceutical industry practices, large and small biopharmaceutical services companies, individual consultants, specialty medical communications services companies, and medical communication subsidiaries of large international advertising companies.
We believe that a central feature of our PCMS service offering is our combination of scientific, regulatory and business expertise. We consider PCMS’s key competitive strengths to include a combination of deep expertise in early stage drug development, regulatory strategy and submissions, GMP compliance, business process optimization, reimbursement and market access, and global marketing and communications strategies. We believe that this broad range of capabilities enables us to help our clients get the right product to market in an efficient and effective manner.
PERCEPTIVE
Our Perceptive business competes primarily with biopharmaceutical services companies, information technology companies, and software companies. Companies in this segment compete based on the strength and usability of their technology offerings, their expertise and experience, and their understanding of the clinical development process. Perceptive’s key competitive strength is its combination of technological expertise and knowledge of clinical development that was enhanced by the acquisition of ClinPhone in August 2008. Additionally, Perceptive’s offerings provide substantial synergies to our CRS services.
INTELLECTUAL PROPERTY
Our trademark “PAREXEL,” is of material importance to us. This and other trademarks have been registered in the U.S. and many foreign countries. The duration of trademark registrations varies from country to country. However, trademarks generally may be renewed indefinitely as long as they are in use and/or their registrations are properly maintained, and as long as they have not been found to have become generic.
EMPLOYEES
As of June 30, 2010, we had 9,720 full-time equivalent employees. Approximately 28.1% of the employees are located in North America and approximately 71.9% are located throughout Europe, Asia/Pacific, Africa, and South America. We believe that we have good relationships with our employees.
The success of our business depends upon our ability to attract and retain qualified professional, scientific, and technical staff. The level of competition among employers in the U.S. and overseas for skilled personnel, particularly those with Ph.D., M.D., or equivalent degrees, is high. We believe that our name recognition and our multinational presence, which allows for international transfers, are an advantage in attracting employees. In addition, we believe that the wide range of clinical trials in which we participate allows us to offer broad experience to clinical researchers.
GOVERNMENT REGULATIONS
We provide clinical trial and diverse consulting services to the pharmaceutical, biotechnology, and medical device industries. Lack of success in obtaining approval for the conduct of clinical trials in the countries where we manage clinical trials on behalf of our clients can adversely affect us. We make no guarantees to our clients with regard to successful outcomes of the regulatory process, including the success of clinical trial applications or marketing authorization applications.
Clinical research services provided by PAREXEL in the U.S. are subject to ongoing FDA regulation. We are obligated to comply with FDA requirements governing activities such as obtaining institutional review board (IRB) approval and patient informed consents, verifying qualifications of investigators, reporting patients’ adverse reactions to products, and maintaining
11
Table of Contents
thorough and accurate records. We are also required to ensure that the computer systems we use to process human data from clinical trials are validated in accordance with the FDA’s electronic records regulations, 21 CFR. Part 11, which apply to the pharmaceutical and CRO industries when companies choose to use electronic records in lieu of paper records or electronic signatures in lieu of traditional signatures. We must maintain source documents for each study for specified periods, and such documents may be reviewed according to GCP standards by the study sponsors and the FDA and other agencies (for example the EMA, European Medicines Agency) during audits and inspections. Non-compliance with GCP can result in the disqualification of data collected during a clinical trial and in non-approval or non-clearance of a product application submitted to the FDA or other agencies around the world.
The clinical investigation of new drugs, biologics, and medical devices is highly regulated by government agencies around the world. The standard for the conduct of clinical research and development studies is embodied in GCP, a set of international standards and guidelines which stipulate procedures designed to ensure the quality and integrity of data obtained from clinical testing, and to protect the rights and safety of clinical trial participants. The FDA and many other regulatory authorities require that study results submitted to such authorities be based on studies conducted in compliance with GCP. Effective May 1, 2004, the European Union (“EU”) enacted the Clinical Trials Directive (the “Directive” or “CTD”) in an attempt to harmonize the regulatory requirements of the member states of the EU for the conduct of clinical trials in its territory. The Directive requires sponsors of clinical trials to submit formal applications to national ethics committees and regulatory authorities prior to the initiation of clinical trials in any of the 27 Member States of the EU. Whereas some Member States, prior to the implementation of the Directive, had minimal requirements for clinical trial initiation, all Member States are now subject to the same stringent requirements of the Directive. As in the United States, clinical trials in the EU are expected to be carried out in compliance with detailed requirements for GCP. The international regulatory approval process, in the EU as well as many other countries, includes all of the risks and potential delays associated with the FDA approval process.
Because the FDA’s regulatory requirements have served as the model for much of the regulation of new drug development worldwide, regulatory requirements similar to those of the FDA exist in the other countries in which we operate. Our regulatory capabilities include knowledge of the specific regulatory requirements of numerous countries. We have managed simultaneous regulatory submissions in more than one country for a number of drug sponsors during each of the past ten years. Beginning in 1990, the FDA and corresponding regulatory agencies of the EU and Japan commenced discussions to develop harmonized standards for preclinical and clinical studies and the format and content of applications for new drug approvals through a process known as the International Conference on Harmonisation (“ICH”) of Technical Requirements for Registration of Pharmaceuticals for Human use. Data from multinational studies adhering to GCP are now generally acceptable to the FDA and regulators in Canada, the EU and Japan. The ICH process has sanctioned a single common format for drug and biologic marketing authorization applications, known as the Common Technical Document (“CTD”) in the U.S., Europe, Japan and Canada. On July 1, 2003 the CTD format became mandatory in Europe and Japan and highly recommended by the FDA in the U.S. and by Canadian regulatory authorities. We have developed the expertise to prepare CTDs for our clients in both paper and electronic form.
REGULATION OF DRUGS AND BIOLOGICS
Before a new drug or biologic may be approved and marketed, the drug or biologic must undergo extensive testing and regulatory review in order to determine that the drug or biologic is safe and effective. It is not possible to estimate the time in which preclinical and Phase I, II and III studies will be completed with respect to a given product, although the time period may last many years. Using the U.S. regulatory environment as an example, the stages of this development process are generally as follows:
Preclinical Research (approximately 1 to 3.5 years) – In vitro (“test tube”) and animal studies must be conducted in accordance with GLP to establish the relative toxicity of the drug or biologic over a wide range of doses and to detect any potential to cause a variety of adverse conditions or diseases, including birth defects or cancer. If results warrant continuing development of the drug or biologic, the results of the studies are submitted to the FDA by the manufacturer as part of an Investigational New Drug Application (“IND”), which must be reviewed by the FDA before proposed clinical testing can begin. An IND must include, among other things, preclinical data, chemistry, manufacturing and control information, and an investigational plan, and must become effective before such trials may begin. An IND automatically becomes effective 30 days after receipt by the FDA, unless the FDA, within the 30-day time period, raises concerns or questions relating to one or more proposed clinical trials. In such a case, the IND sponsor and the FDA must resolve any outstanding concerns before the clinical trial can begin. As a result, there can be no assurance that submission of an IND will result in the ability to commence clinical trials.
12
Table of Contents
Clinical Trials (approximately 3.5 to 6 years)
Phase I consists of basic safety and pharmacology testing in approximately 20 to 80 human subjects, usually healthy volunteers or stable patients, and includes studies to evaluate the metabolic and pharmacologic action of the product in humans, how the drug or biologic works, how it is affected by other drugs, how it is tolerated and absorbed, where it goes in the body, how long it remains active, and how it is broken down and eliminated from the body.
Phase II includes basic efficacy (effectiveness) and dose-range testing in a limited patient population (usually 100 to 200 patients) afflicted with a specific disease or condition for which the product is intended for use, further safety testing, evaluation of effectiveness, and determination of optimal dose levels, dose schedules, and routes of administration. If Phase II studies yield satisfactory results and no hold is placed on further studies by the FDA, Phase III studies can commence.
Phase III includes larger scale, multi-center, comparative clinical trials conducted with patients afflicted by a target disease, in order to provide enough data for a valid statistical test of safety and effectiveness required by the FDA and others, and to provide an adequate basis for product labeling.
The FDA receives reports on the progress of each phase of clinical testing and may require the modification, suspension, or termination of clinical trials if, among other things, an unreasonable risk is presented to patients or if the design of the trial is insufficient to meet its stated objective.
NDA or Biologic License Application (“BLA”) Preparation and Submission – Upon completion of Phase III trials, the sponsor assembles the statistically analyzed data from all phases of development, along with the chemistry and manufacturing and pre-clinical data and the proposed labeling, among other things, into a single large document, the NDA (New Drug Application) or BLA in CTD format as of July 1, 2003, which today comprises, on average, roughly 100,000 pages.
FDA Review of NDA or BLA – The FDA carefully scrutinizes data from all phases of development to confirm that the manufacturer has complied with regulations and that the drug or biologic is safe and effective for the specific use (or “indication”) under study. The FDA may refuse to accept the NDA or BLA for filing and substantive review if certain administrative and content criteria are not satisfied. Even after accepting the submission for review, the FDA may also require additional testing or information before approval of an NDA or BLA. The FDA must deny approval of an NDA or BLA if applicable regulatory requirements are not satisfied.
Post-Marketing Surveillance and Phase IV Studies – Federal regulation requires the sponsor to collect and periodically report to the FDA additional safety and efficacy data on the drug or biologic for as long as the manufacturer markets the product (post-marketing surveillance). If the product is marketed outside the U.S., these reports must include data from all countries in which the product is sold. Additional studies (Phase IV) may be required by the FDA as a condition of the product’s approval to assess safety or verify clinical benefit or may be voluntarily undertaken after initial approval to find new uses for the product, to test new dosage formulations, or to confirm selected non-clinical benefits, e.g., increased cost-effectiveness or improved quality of life. Product approval may be withdrawn if compliance with regulatory standards is not maintained or if problems occur following initial marketing. In addition, the FDA and other major regulatory agencies ask sponsor companies to prepare risk management plans for approved and marketed drugs and biologics, aimed at assessing areas of drug risk and plans for managing such risks should they materialize. The passage of the FDA Amendments Act of 2007 imposed additional requirements on sponsors to address drug safety (for example, through a plan called Risk Evaluation and Mitigation Strategies or REMS), to conduct post-marketing studies required by the FDA and to submit clinical trial information, including clinical study results, of investigational and marketed drugs (as well as medical devices) to a databank maintained by the National Institutes of Health and accessible to the public on the Internet (www.clinicaltrials.gov). This was done in order to increase the “public transparency” of clinical results.
REGULATION OF MEDICAL DEVICES
Unless a medical device is exempted from pre-market approval or clearance requirements, which are described below, FDA approval or clearance of the device is required before the product may be marketed in the United States. In order to obtain pre-market clearance for marketing, a manufacturer must demonstrate substantial equivalence to a similar legally marketed product by submitting a pre-market notification, or 510(k), to the FDA. The FDA may require preclinical and clinical data to support a substantial equivalence determination, and there can be no assurance the FDA will find a device substantially equivalent. Clinical trials can take extended periods of time to complete. In addition, if the FDA requires an approved Investigational Device Exemption (“IDE”) before clinical device trials may commence, there can be no guarantee that the agency will approve the IDE. The IDE approval process could also result in significant delays.
13
Table of Contents
After submission of a pre-market notification containing, among other things, any data collected, the FDA may find the device substantially equivalent and the device may be marketed. After a device receives 510(k) clearance, any modification that could significantly affect its safety or effectiveness, or that would constitute a major change in its intended use, requires a new 510(k) clearance or could require approval of a pre-market approval application (“PMA”). If the FDA finds that a device is not substantially equivalent, the manufacturer may request that the FDA make a risk-based classification to place the device in Class I or Class II. However, if a timely request for risk-based classification is not made, or if the FDA determines that a Class III designation is appropriate, a PMA will be required before the device may be marketed.
The PMA approval process is lengthy, expensive, and typically requires, among other things, extensive data from preclinical testing and a well-controlled clinical trial or trials that demonstrate a reasonable assurance of safety and effectiveness. There can be no assurance that review will result in timely, or any, PMA approval. There may also be significant conditions associated with the approval, including limitations on labeling and advertising claims and the imposition of post-market testing, tracking, or surveillance requirements. Even after approval, a new PMA or PMA supplement is required in the event of a modification to the device, its labeling or its manufacturing process.
REGULATION OF PATIENT INFORMATION
The confidentiality, security, use and disclosure of patient-specific information are subject to governmental regulation. Regulations to protect the safety and privacy of human subjects who participate in or whose data are used in clinical research generally require clinical investigators to obtain affirmative informed consent from identifiable research subjects before research is undertaken. Under the Health Insurance Portability and Accountability Act of 1996, or HIPAA, the U.S. Department of Health and Human Services has issued regulations mandating heightened privacy and confidentiality protections for certain types of individually identifiable health information, or protected health information, when used or disclosed by health care providers and other HIPAA-covered entities or business associates that provide services to or perform functions on behalf of these covered entities. HIPAA regulations require individuals’ written authorization before identifiable health information may be used for research, in addition to any required informed consent. HIPAA regulations also specify standards for de-identifying health information so that information can be handled outside of the HIPAA requirements and for creating limited data sets that can be used for research purposes under less stringent HIPAA restrictions.
Outside of the United States, many countries have enacted laws to safeguard the privacy and security of personal information, including individually identifiable health information. The member states of the European Union have adopted a rigorous system of data protection regulations, based upon a framework imposed by the 1995 European Commission Directive on Data Protection, or Privacy Directive. These rules provide broad protections for personal information, including, among other things, notice requirements, limits on the scope and duration for which personal information may be maintained and processed, restrictions on disclosures of personal information, standards for providing individuals with control over the manner in which personal information is processed, and restrictions on transfers of such data to the United States and other countries that the European Union finds to lack “adequate” data protection laws of their own. Health-related information is recognized as a special, sensitive category of personal information, which may generally be processed only pursuant to the affirmative, or opt-in, consent of the individual to whom the information pertains. Violations of these data protection regulations are subject to administrative penalties, civil money penalties, and criminal prosecution, including corporate fines and personal liability.
In order to comply with these laws and regulations, we may need to implement new privacy and security measures, which may require us to make substantial expenditures or cause us to limit the products and services we offer. In addition, if we violate applicable laws, regulations, contractual commitments, or other duties relating to the use, privacy or security of health information, we could be subject to civil liability or criminal penalties and it may be necessary to modify our business practices.
POTENTIAL LIABILITY AND INSURANCE
Our clinical research services focus on the testing of experimental drugs and devices on human volunteers pursuant to study protocols and in accordance with laws and regulations which govern clinical trials. Clinical research involves a risk of liability for personal injury or death to patients due to, among other reasons, possible unforeseen adverse side effects or improper administration of the new drug or medical device. We do not generally provide health care services directly to patients. Rather, our physicians or third party physician investigators are responsible for administering drugs and evaluating the study patients. Many of the patients enrolled in clinical trials are already seriously ill and are at risk of further illness or death.
We believe that the risk of liability to patients in clinical trials is mitigated by various regulatory requirements, including the role of IRBs, the need to obtain each patient’s informed consent, and the oversight by applicable regulatory authorities. The FDA, the Medicines and Healthcare products Regulatory Agency (MHRA) in the U.K., and regulatory authorities in other
14
Table of Contents
countries require each human clinical trial to be reviewed and approved by the IRB at each study site. An IRB is an independent ethics committee that includes both medical and non-medical personnel and is obligated to protect the interests of patients enrolled in the trial. The IRB approves and monitors the protocol and the measures designed to protect patients, such as the requirement to obtain informed consents.
To reduce our potential liability, we generally seek to incorporate indemnity provisions into our contracts with clients to protect us from liability for adverse reactions to the study drug as well as any negligent acts by the study Sponsor and/or third party physician investigators. These indemnity provisions do not, however, protect us against certain of our own actions, such as those involving negligence. Moreover, these indemnities are contractual arrangements that are subject to negotiation with individual clients, and the terms and scope of such indemnities can vary from client to client and from study to study. Finally, the financial performance of these indemnities is not secured, so that we bear the risk that an indemnifying party may not have the financial ability to fulfill its indemnification obligations. We could be materially and adversely affected if we were required to pay damages or incur defense costs in connection with an uninsured claim that is outside the scope of an indemnity or where the indemnity, although applicable, is not performed in accordance with its terms.
We currently maintain a portfolio of insurance coverage, including a professional liability insurance policy, subject to deductibles and coverage limits. There can be no assurance that this insurance coverage will be adequate, or that insurance coverage will continue to be available on terms acceptable to PAREXEL.
AVAILABLE INFORMATION
Our Internet website is http://www.parexel.com. We make available through this website our annual report on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K and amendments to those reports filed pursuant to Sections 13(a) and 15(d) of the Exchange Act. We make these reports available free of charge through our website as soon as reasonably practicable after they have been electronically filed with, or furnished to, the Securities and Exchange Commission (“SEC”). Any materials we file with the SEC may also be read and copied at the SEC’s public reference room located at 100 F Street, N.E. Washington, D.C. 20549. Please call the SEC at 1-800-SEC-0330 for further information on the public reference room. Our SEC filings are also available to the public on the SEC’s Internet website at www.sec.gov.
15
Table of Contents
| ITEM 1A. | RISK FACTORS |
In addition to other information in this report, the following risk factors should be considered carefully in evaluating our company and our business. These important factors could cause our actual results to differ from those indicated by forward-looking statements made in this report, including in the section of this report entitled “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and other forward-looking statements that we may make from time to time. If any of the following risks occur, our business, financial condition, or results of operations would likely suffer.
Additional risks not currently known to us or other factors not perceived by us to present significant risk to our business at this time also may impair our business operations.
Risks Associated with our Business and Operations
The loss, modification, or delay of large or multiple contracts may negatively impact our financial performance.
Our clients generally can terminate their contracts with us upon 30 to 60 days notice or can delay the execution of services. The loss or delay of a large contract or the loss or delay of multiple contracts could adversely affect our operating results, possibly materially. We have in the past experienced large contract cancellations and delays, which have adversely affected our operating results, including cancellations of a late-phase contract during the first quarter of Fiscal Year 2008 and a late-phase contract during the second quarter of Fiscal Year 2007.
Clients terminate or delay their contracts for a variety of reasons, including:
| • | failure of products being tested to satisfy safety requirements; |
| • | failure of products being tested to satisfy efficacy criteria; |
| • | products having unexpected or undesired clinical results; |
| • | client cost reductions as a result of budgetary limit or changing priorities; |
| • | client decisions to forego a particular study, perhaps for economic reasons; |
| • | merger or potential merger related activities involving the client; |
| • | insufficient patient enrollment in a study; |
| • | insufficient investigator recruitment; |
| • | clinical drug manufacturing problems resulting in shortages of the product; |
| • | product withdrawal following market launch; and |
| • | shut down of manufacturing facilities. |
The current economic environment may negatively impact our financial performance as a result of client defaults and other factors.
Our ability to attract and retain clients, invest in and grow our business and meet our financial obligations depends on our operating and financial performance, which, in turn, is subject to numerous factors. In addition to factors specific to our business, prevailing economic conditions and financial, business and other factors beyond our control can also affect us. We cannot anticipate all the ways in which the current economic climate and financial market conditions could adversely impact our business.
We are exposed to risks associated with reduced profitability and the potential financial instability of our clients, many of whom may be adversely affected by volatile conditions in the financial markets, the economy in general and disruptions to the demand for health care services and pharmaceuticals. These conditions could cause clients to experience reduced profitability and/or cash flow problems that could lead them to modify, delay or cancel contracts with us, including contracts included in our current backlog.
Some of our clients do not generate revenue and rely upon equity and debt investments and other external sources of capital to meet their cash requirements. Due to the poor condition of the current global economy and other factors outside of our control, these clients may lack the funds necessary to meet outstanding liabilities to us, despite contractual obligations. For example, in the second quarter of Fiscal Year 2009, one of our biopharma clients informed us that it had encountered funding difficulties when one of its major investors defaulted on a contractual investment commitment, and that as a result the client
16
Table of Contents
would be unable to make payments due to us in connection with an on-going service contract for a large Phase III clinical trial. As a result, we recorded approximately $15.0 million in reserves related to this late-stage trial, including $12.3 million in bad debt reserves. It is possible that similar situations could arise in the future, and such defaults could negatively affect our financial performance, possibly materially.
We face risks arising from the restructuring of our operations.
In October 2009, we adopted a plan to restructure our operations to reduce expenses, better align costs with current and future geographic sources of revenue, and improve operating efficiencies. During Fiscal Year, 2010, we recorded $16.8 million in restructuring charges related to this plan, including approximately $11.6 million in employee separation benefits associated with the elimination of 238 managerial and staff positions and $5.2 million in costs related to the abandonment of certain property leases. Although we expect that all costs associated with the restructuring plan were recorded as of June 30, 2010, if we incur additional restructuring charges, our financial condition and results of operations may suffer.
Restructuring also presents significant potential risks of events occurring that could adversely affect us, including a decrease in employee morale, the failure to achieve targeted cost savings and the failure to meet operational targets and customer requirements due to the loss of employees and any work stoppages that might occur.
The fixed price nature of our contracts could hurt our operating results.
Approximately 90% of our contracts are fixed price. If we fail to accurately price our contracts, or if we experience significant cost overruns that are not recovered from our clients, our gross margins on the contracts would be reduced and we could lose money on contracts. In the past, we have had to commit unanticipated resources to complete projects, resulting in lower gross margins on those projects. We might experience similar situations in the future.
If we are unable to attract suitable willing investigators and volunteers for our clinical trials, our clinical development business might suffer.
The clinical research studies we run in our CRS segment rely upon the ready accessibility and willing participation of physician investigators and volunteer subjects. Investigators are typically located at hospitals, clinics or other sites and supervise administration of the study drug to patients during the course of a clinical trial. Volunteer subjects generally include people from the communities in which the studies are conducted. Our clinical research development business could be adversely affected if we were unable to attract suitable and willing investigators or volunteers on a consistent basis.
If our Perceptive business is unable to maintain continuous, effective, reliable and secure operation of its computer hardware, software and internet applications and related tools and functions, its business will be harmed.
Our Perceptive business involves collecting, managing, manipulating and analyzing large amounts of data, and communicating data via the Internet. In our Perceptive business, we depend on the continuous, effective, reliable and secure operation of computer hardware, software, networks, telecommunication networks, Internet servers and related infrastructure. If the hardware or software malfunctions or access to data by internal research personnel or customers through the Internet is interrupted, our Perceptive business could suffer. In addition, any sustained disruption in Internet access provided by third parties could adversely impact our Perceptive business.
Although the computer and communications hardware used in our Perceptive business is protected through physical and software safeguards, it is still vulnerable to fire, storm, flood, power loss, earthquakes, telecommunications failures, physical or software break-ins, and similar events. And while certain of our operations have appropriate disaster recovery plans in place, we currently do not have redundant facilities everywhere in the world to provide IT capacity in the event of a system failure. In addition, the Perceptive software products are complex and sophisticated, and could contain data, design or software errors that could be difficult to detect and correct. If Perceptive fails to maintain and further develop the necessary computer capacity and data to support the needs of our Perceptive customers, it could result in a loss of or a delay in revenue and market acceptance. Additionally, significant delays in the planned delivery of system enhancements or inadequate performance of the systems once they are completed could damage our reputation and harm our business.
Finally, long-term disruptions in the infrastructure caused by events such as natural disasters, the outbreak of war, the escalation of hostilities, and acts of terrorism (particularly in areas where we have offices) could adversely affect our businesses. Although we carry property and business interruption insurance, our coverage may not be adequate to compensate us for all losses that may occur.
17
Table of Contents
Our business is subject to international economic, political, and other risks that could negatively affect our results of operations or financial position.
We provide most of our services on a worldwide basis. Our service revenue from non-U.S. operations represented approximately 65.4% and 64.4% of total consolidated service revenue for the Fiscal Years 2010 and 2009, respectively. More specifically, our service revenue from operations in Europe, Middle East and Africa represented 48.5% and 50.3% of total consolidated service revenue for the corresponding periods. Our service revenue from operations in the Asia/Pacific region represented 11.8% and 9.1% of total consolidated service revenue for the respective periods. Accordingly, our business is subject to risks associated with doing business internationally, including:
| • | changes in a specific country’s or region’s political or economic conditions, including Western Europe, in particular; |
| • | potential negative consequences from changes in tax laws affecting our ability to repatriate profits; |
| • | difficulty in staffing and managing widespread operations; |
| • | unfavorable labor regulations applicable to our European or other international operations; |
| • | changes in foreign currency exchange rates; |
| • | the need to ensure compliance with the numerous regulatory and legal requirements applicable to our business in each of these jurisdictions and to maintain an effective compliance program to ensure compliance; and |
| • | longer payment cycles of foreign customers and difficulty of collecting receivables in foreign jurisdictions. |
Our operating results are impacted by the health of the North American, European and Asian economies, among others. Our business and financial performance may be adversely affected by current and future economic conditions that cause a decline in business and consumer spending, including a reduction in the availability of credit, rising interest rates, financial market volatility and recession.
If we cannot retain our highly qualified management and technical personnel, our business would be harmed.
We rely on the expertise of our Chairman and Chief Executive Officer, Josef H. von Rickenbach, and our Chief Operating Officer, Mark A. Goldberg, and it would be difficult and expensive to find qualified replacements with the level of specialized knowledge of our products and services and the biopharmaceutical services industry. While we are a party to an employment agreement with Mr. von Rickenbach, it may be terminated by either party upon notice to the counterparty.
In addition, in order to compete effectively, we must attract and retain qualified sales, professional, scientific, and technical operating personnel. Competition for these skilled personnel, particularly those with a medical degree, a Ph.D. or equivalent degrees, is intense. We may not be successful in attracting or retaining key personnel.
Risks Associated with our Financial Results
Our operating results have fluctuated between quarters and years and may continue to fluctuate in the future, which could affect the price of our common stock.
Our quarterly and annual operating results have varied and will continue to vary in the future as a result of a variety of factors. For example, our income from operations totaled $20.1 million for the fiscal quarter ended June 30, 2010, $25.8 million for the fiscal quarter ended March 31, 2010, $18.7 million for the fiscal quarter ended December 31, 2009, and $18.5 million for the fiscal quarter ended September 30, 2009. Factors that cause these variations include:
| • | the level of new business authorizations in particular quarters or years; |
| • | the timing of the initiation, progress, or cancellation of significant projects; |
| • | exchange rate fluctuations between quarters or years; |
| • | restructuring charges; |
| • | seasonality; |
| • | the mix of services offered in a particular quarter or year; |
| • | the timing of the opening of new offices or internal expansion; |
| • | timing, costs and the related financial impact of acquisitions; |
18
Table of Contents
| • | the timing and amount of costs associated with integrating acquisitions; |
| • | the timing and amount of startup costs incurred in connection with the introduction of new products, services or subsidiaries |
| • | the dollar amount of changes in contract scope finalized during a particular period; and |
| • | the amount of any reserves we are required to record. |
Many of these factors, such as the timing of cancellations of significant projects and exchange rate fluctuations between quarters or years, are beyond our control.
If our operating results do not match the expectations of securities analysts and investors, the trading price of our common stock will likely decrease.
Backlog may not result in revenue, and the rate at which this backlog converts into revenue may slow when compared to historical trends.
Our backlog is not necessarily a meaningful predictor of future results because backlog can be affected by a number of factors, including the size and duration of contracts, many of which are performed over several years. Additionally, as described above, contracts relating to our clinical development business are subject to early termination by the client and clinical trials can be delayed or canceled for many reasons, including unexpected test results, safety concerns, regulatory developments or economic issues. Also, the scope of a contract can be reduced significantly during the course of a study. If the scope of a contract is revised, the adjustment to backlog occurs when the revised scope is approved by the client. For these and other reasons, we do not fully realize all of our backlog as net revenue.
In addition, the rate at which our backlog converts into revenue may slow. A slowdown in this conversion rate means that the rate of revenue recognized on contract awards may be slower than what we have experienced in the past, particularly in connection with the ramp-up and initiation of some recently awarded strategic partnerships, which could impact our net revenue and results of operations on a quarterly and annual basis.
Our revenue and earnings are exposed to exchange rate fluctuations, which has substantially affected our operating results.
We conduct a significant portion of our operations in foreign countries. Because our financial statements are denominated in U.S. dollars, changes in foreign currency exchange rates could have and have had a significant effect on our operating results. For example, as a result of year-over-year foreign currency fluctuation, service revenue for the twelve months ended June 30, 2010 was positively impacted by approximately $10.3 million as compared with the same period in the previous year. Exchange rate fluctuations between local currencies and the U.S. dollar create risk in several ways, including:
| • | Foreign Currency Translation Risk. The revenue and expenses of our foreign operations are generally denominated in local currencies, primarily the pound sterling and the Euro, and are translated into U.S. dollars for financial reporting purposes. For Fiscal Years 2010 and 2009, approximately 24.1% and 24.3% of consolidated service revenue was denominated in Euros, respectively. Revenue, denominated in pounds sterling, was 14.6% and 15.6%, respectively. Accordingly, changes in exchange rates between foreign currencies and the U.S. dollar will affect the translation of foreign results into U.S. dollars for purposes of reporting our consolidated results. |
| • | Foreign Currency Transaction Risk. We may be subjected to foreign currency transaction risk when our foreign subsidiaries enter into contracts or incur liabilities denominated in a currency other than the foreign subsidiaries functional (local) currency. To the extent we are unable to shift the effects of currency fluctuations to the clients, foreign exchange rate fluctuations as a result of foreign currency exchange losses could have a material adverse effect on our results of operations. |
Although we try to limit these risks through exchange rate fluctuation provisions stated in our service contracts, or by hedging transaction risk with foreign currency exchange contracts, we do not succeed in all cases. Even in those cases where we are successful, we may still experience fluctuations in financial results from our operations outside of the U.S., and we may not be able to favorably reduce the currency transaction risk associated with our service contracts.
19
Table of Contents
Our effective income tax rate may fluctuate from quarter-to-quarter, which may affect our earnings and earnings per share.
Our quarterly effective income tax rate is influenced by our projected profitability in the various taxing jurisdictions in which we operate. Changes in the distribution of profits and losses among taxing jurisdictions may have a significant impact on our effective income tax rate, which in turn could have a material adverse effect on our net income and earnings per share. Factors that affect the effective income tax rate include, but are not limited to:
| • | the requirement to exclude from our quarterly worldwide effective income tax calculations losses in jurisdictions where no tax benefit can be recognized; |
| • | actual and projected full year pretax income; |
| • | changes in tax laws in various taxing jurisdictions; |
| • | audits by taxing authorities; and |
| • | the establishment of valuation allowances against deferred tax assets if it is determined that it is more likely than not that future tax benefits will not be realized. |
Additionally, recently enacted legislation regarding U.S. international tax laws relating to U.S. foreign tax credits and the expiration of the “look through” rules may affect the amount of international income subject to taxation in the United States and impact our ability to utilize foreign tax credits. These changes may cause fluctuations in our effective income tax rate that could cause fluctuation in our earnings and earnings per share, which could affect our stock price.
Our results of operations may be adversely affected if we fail to realize the full value of our goodwill and intangible assets.
As of June 30, 2010, our total assets included $335.3 million of goodwill and net intangible assets. We assess the realizability of our net intangible assets and goodwill annually as well as whenever events or changes in circumstances indicate that these assets may be impaired. These events or circumstances generally include operating losses or a significant decline in earnings associated with the acquired business or asset. Our ability to realize the value of the goodwill and indefinite-lived intangible assets will depend on the future cash flows of these businesses. These cash flows in turn depend in part on how well we have integrated these businesses. If we are not able to realize the value of the goodwill and indefinite-lived intangible assets, we may be required to incur material charges relating to the impairment of those assets.
Our business has experienced substantial expansion in the past and such expansion and any future expansion could strain our resources if not properly managed.
We have expanded our business substantially in the past. For example, in August 2008, we completed the acquisition of ClinPhone, a leading clinical technology organization, for a purchase price of approximately $190 million. Future rapid expansion could strain our operational, human and financial resources. In order to manage expansion, we must:
| • | continue to improve operating, administrative, and information systems; |
| • | accurately predict future personnel and resource needs to meet client contract commitments; |
| • | track the progress of ongoing client projects; and |
| • | attract and retain qualified management, sales, professional, scientific and technical operating personnel. |
If we do not take these actions and are not able to manage the expanded business, the expanded business may be less successful than anticipated, and we may be required to allocate additional resources to the expanded business, which we would have otherwise allocated to another part of our business.
If we are unable to successfully integrate an acquired company, the acquisition could lead to disruptions to our business. The success of an acquisition will depend upon, among other things, our ability to:
| • | assimilate the operations and services or products of the acquired company; |
| • | integrate acquired personnel; |
| • | retain and motivate key employees; |
| • | retain customers; |
| • | identify and manage risks facing the acquired company; and |
| • | minimize the diversion of management’s attention from other business concerns. |
20
Table of Contents
Acquisitions of foreign companies may also involve additional risks, including assimilating differences in foreign business practices and overcoming language and cultural barriers.
In the event that the operations of an acquired business do not meet our performance expectations, we may have to restructure the acquired business or write-off the value of some or all of the assets of the acquired business.
Risks Associated with our Industry
We depend on the pharmaceutical and biotechnology industries, either or both of which may suffer in the short or long term.
Our revenues depend greatly on the expenditures made by the pharmaceutical and biotechnology industries in research and development. In some instances, companies in these industries are reliant on their ability to raise capital in order to fund their research and development projects. Accordingly, economic factors and industry trends that affect our clients in these industries also affect our business. If companies in these industries were to reduce the number of research and development projects they conduct or outsource, our business could be materially adversely affected.
In addition, we are dependent upon the ability and willingness of pharmaceutical and biotechnology companies to continue to spend on research and development and to outsource the services that we provide. We are therefore subject to risks, uncertainties and trends that affect companies in these industries. We have benefited to date from the tendency of pharmaceutical and biotechnology companies to outsource clinical research projects, but any downturn in these industries or reduction in spending or outsourcing could adversely affect our business. For example, if these companies expanded upon their in-house clinical or development capabilities, they would be less likely to utilize our services.
Because we depend on a small number of industries and clients for all of our business, the loss of business from a significant client could harm our business, revenue and financial condition.
The loss of, or a material reduction in the business of, a significant client could cause a substantial decrease in our revenue and adversely affect our business and financial condition, possibly materially. In Fiscal Years 2010, 2009, and 2008, our five largest clients accounted for approximately 27%, 28%, and 31% of our consolidated service revenue, respectively. We expect that a small number of clients will continue to represent a significant part of our consolidated revenue. This concentration may increase as a result of the increasing number of strategic partnerships into which we have been entering with sponsors. Our contracts with these clients generally can be terminated on short notice. We have in the past experienced contract cancellations with significant clients.
We face intense competition in many areas of our business; if we do not compete effectively, our business will be harmed.
The biopharmaceutical services industry is highly competitive and we face numerous competitors in many areas of our business. If we fail to compete effectively, we may lose clients, which would cause our business to suffer.
We primarily compete against in-house departments of pharmaceutical companies, other full service clinical research organizations (“CROs”), small specialty CROs, and, to a lesser extent, universities, teaching hospitals, and other site organizations. Some of the larger CROs against which we compete include Quintiles Transnational Corporation, Covance, Inc., Pharmaceutical Product Development Inc., Kendle Inc., and Icon plc. In addition, our PCMS business competes with a large and fragmented group of specialty service providers, including advertising/promotional companies, major consulting firms with pharmaceutical industry groups and smaller companies with pharmaceutical industry focus. Perceptive competes primarily with CROs, information technology companies and other software companies. Some of these competitors, including the in-house departments of pharmaceutical companies, have greater capital, technical and other resources than we have. In addition, our competitors that are smaller specialized companies may compete effectively against us because of their concentrated size and focus.
In recent years, a number of the large pharmaceutical companies have established formal or informal alliances with one or more CROs relating to the provision of services for multiple trials over extended time periods. Our success depends in part on successfully establishing and maintaining these relationships. If we fail to do so, our revenues and results of operations could be adversely affected, possibly materially.
21
Table of Contents
If we do not keep pace with rapid technological changes, our products and services may become less competitive or obsolete, especially in our Perceptive business.
The biotechnology, pharmaceutical and medical device industries generally, and clinical research specifically, are subject to increasingly rapid technological changes. Our competitors or others might develop technologies, products or services that are more effective or commercially attractive than our current or future technologies, products or services, or render our technologies, products or services less competitive or obsolete. If our competitors introduce superior technologies, products or services and we cannot make enhancements to our technologies, products and services necessary to remain competitive, our competitive position would be harmed. If we are unable to compete successfully, we may lose clients or be unable to attract new clients, which could lead to a decrease in our revenue.
Risks Associated with Regulation or Legal Liabilities
If governmental regulation of the drug, medical device and biotechnology industry changes, the need for our services could decrease.
Governmental regulation of the drug, medical device and biotechnology product development process is complicated, extensive, and demanding. A large part of our business involves assisting pharmaceutical, biotechnology and medical device companies through the regulatory approval process. Changes in regulations that, for example, streamline procedures or relax approval standards, could eliminate or reduce the need for our services. If companies regulated by the United States Food and Drug Administration (the “FDA”) or similar foreign regulatory authorities needed fewer of our services, we would have fewer business opportunities and our revenues would decrease, possibly materially.
In the United States, the FDA and the Congress have attempted to streamline the regulatory process by providing for industry user fees that fund the hiring of additional reviewers and better management of the regulatory review process. In Europe, governmental authorities have approved common standards for clinical testing of new drugs throughout the European Union by adopting standards for Good Clinical Practices (“GCP”) and by making the clinical trial application and approval process more uniform across member states. The FDA has had GCP in place as a regulatory standard and requirement for new drug approval for many years and Japan adopted GCP in 1998.
The United States, Europe and Japan have also collaborated for over 15 years on the International Conference on Harmonisation (“ICH”), the purpose of which is to eliminate duplicative or conflicting regulations in the three regions. The ICH partners have agreed upon a common format (the Common Technical Document) for new drug marketing applications that reduces the need to tailor the format to each region. Such efforts and similar efforts in the future that streamline the regulatory process may reduce the demand for our services.
Parts of our PCMS business advise clients on how to satisfy regulatory standards for manufacturing and clinical processes and on other matters related to the enforcement of government regulations by the FDA and other regulatory bodies. Any reduction in levels of review of manufacturing or clinical processes or levels of regulatory enforcement, generally, would result in fewer business opportunities for our business in this area.
If we fail to comply with existing regulations, our reputation and operating results would be harmed.
Our business is subject to numerous governmental regulations, primarily relating to worldwide pharmaceutical and medical device product development and regulatory approval and the conduct of clinical trials. If we fail to comply with these governmental regulations, it could result in the termination of our ongoing research, development or sales and marketing projects, or the disqualification of data for submission to regulatory authorities. We also could be barred from providing clinical trial services in the future or could be subjected to fines. Any of these consequences would harm our reputation, our prospects for future work and our operating results. In addition, we may have to repeat research or redo trials. If we are required to repeat research or redo trials, we may be contractually required to do so at no further cost to our clients, but at substantial cost to us.
We may lose business opportunities as a result of healthcare reform and the expansion of managed-care organizations.
Numerous governments, including the U.S. government, have undertaken efforts to control growing healthcare costs through legislation, regulation and voluntary agreements with medical care providers and drug companies. In March 2010, the United States Congress enacted healthcare reform legislation intended over time to expand health insurance coverage and impose health industry cost containment measures. This legislation may significantly impact the pharmaceutical industry. The U.S. Congress has also considered and may adopt legislation that could have the effect of putting downward pressure on the prices that pharmaceutical and biotechnology companies can charge for prescription drugs. In addition, various state legislatures and European and Asian governments may consider various types of healthcare reform in order to control growing healthcare costs. We are presently uncertain as to the effects of the recently enacted legislation on our business and are unable to predict what legislative proposals will be adopted in the future, if any.
22
Table of Contents
If these efforts are successful, drug, medical device and biotechnology companies may react by spending less on research and development. If this were to occur, we would have fewer business opportunities and our revenue could decrease, possibly materially. In addition, new laws or regulations may create a risk of liability, increase our costs or limit our service offerings.
In addition to healthcare reform proposals, the expansion of managed-care organizations in the healthcare market and managed-care organizations’ efforts to cut costs by limiting expenditures on pharmaceuticals and medical devices could result in pharmaceutical, biotechnology and medical device companies spending less on research and development. If this were to occur, we would have fewer business opportunities and our revenue could decrease, possibly materially.
We may have substantial exposure to payment of personal injury claims and may not have adequate insurance to cover such claims.
Our CRS business primarily involves the testing of experimental drugs and medical devices on consenting human volunteers pursuant to a study protocol. Clinical research involves a risk of liability for a number of reasons, including, but not limited to:
| • | personal injury or death to patients who participate in the study or who use a product approved by regulatory authorities after the clinical research has concluded; |
| • | general risks associated with clinical pharmacology facilities, including professional malpractice of clinical pharmacology medical care providers; and |
| • | errors and omissions during a trial that may undermine the usefulness of a trial or data from the trial or study. |
In order to mitigate the risk of liability, we seek to include indemnity provisions in our CRS contracts with clients and with investigators. However, we are not able to include indemnity provisions in all of our contracts. In addition, even if we are able to include an indemnity provision in our contracts, the indemnity provisions may not cover our exposure if:
| • | we had to pay damages or incur defense costs in connection with a claim that is outside the scope of an indemnity agreement; or |
| • | a client failed to indemnify us in accordance with the terms of an indemnity agreement because it did not have the financial ability to fulfill its indemnification obligation or for any other reason. |
In addition, contractual indemnifications generally do not protect us against liability arising from certain of our own actions, such as negligence or misconduct.
We also carry insurance to cover our risk of liability. However, our insurance is subject to deductibles and coverage limits and may not be adequate to cover claims. In addition, liability coverage is expensive. In the future, we may not be able to maintain or obtain the same levels of coverage on reasonable terms, at a reasonable cost, or in sufficient amounts to protect us against losses due to claims.
Existing and proposed laws and regulations regarding confidentiality of patients’ information could result in increased risks of liability or increased cost to us or could limit our product and service offerings.
The confidentiality, security, use and disclosure of patient-specific information are subject to governmental regulation. Regulations to protect the safety and privacy of human subjects who participate in or whose data are used in clinical research generally require clinical investigators to obtain affirmative informed consent from identifiable research subjects before research is undertaken. Under the Health Insurance Portability and Accountability Act of 1996, or HIPAA, the U.S. Department of Health and Human Services has issued regulations mandating privacy and security protections for certain types of individually identifiable health information, or protected health information, when used or disclosed by health care providers and other HIPAA-covered entities or business associates that provide services to or perform functions on behalf of these covered entities. HIPAA regulations require individuals’ written authorization before identifiable health information may be used for research, in addition to any required informed consent. HIPAA regulations also specify standards for de-identifying health information so that information can be handled outside of the HIPAA requirements and for creating limited data sets that can be used for research purposes under less stringent HIPAA restrictions. The European Union and its member states, as well as other countries, such as Japan, and state governments in the United States, have adopted and continue to issue new medical privacy and general data protection laws and regulations. In order to comply with these laws and regulations, we may need to implement new privacy and security measures, which may require us to make substantial expenditures or cause us to limit the products and services we offer. In addition, if we violate applicable laws, regulations, contractual commitments, or other duties relating to the use, privacy or security of health information, we could be subject to civil liability or criminal penalties and it may be necessary to modify our business practices.
23
Table of Contents
Failure to achieve and maintain effective internal controls in accordance with Section 404 of the Sarbanes-Oxley Act of 2002, and delays in completing our internal controls and financial audits, could have a material adverse effect on our business and stock price.
If we fail to achieve and maintain effective internal controls, we will not be able to conclude that we have effective internal controls over financial reporting in accordance with Section 404 of the Sarbanes-Oxley Act of 2002. Failure to achieve and maintain an effective internal control environment, and delays in completing our internal controls and financial audits, could cause investors to lose confidence in our reported financial information and us, which could result in a decline in the market price of our common stock, and cause us to fail to meet our reporting obligations in the future, which in turn could impact our ability to raise equity financing if needed in the future. Our Fiscal Year 2009 management assessment revealed a material weakness in our internal controls over financial reporting due to insufficient controls associated with accounting for the ClinPhone business combination, specifically the adoption by ClinPhone of an accounting policy for revenue recognition in accordance with U.S. GAAP for IVR sales contracts with multiple revenue elements and the determination of the fair value of deferred revenue assumed in the business combination. We have changed our internal controls to address this material weakness, but we have not yet tested the effectiveness of our remediation since we have not completed any further acquisitions. There can be no assurance that our remediation will be successful. During the course of our continued testing, we also may identify other significant deficiencies or material weaknesses, in addition to the ones already identified, which we may not be able to remediate in a timely manner or at all.
Risks Associated with Leverage
Our indebtedness may limit cash flow available to invest in the ongoing needs of our business.
As of June 30, 2010, we had $212.5 million principal amount of debt outstanding and remaining borrowing availability of $65.0 million under our revolving line of credit. We may incur additional debt in the future. Our leverage could have significant adverse consequences, including:
| • | requiring us to dedicate a substantial portion of any cash flow from operations to the payment of interest on, and principal of, our debt, which will reduce the amounts available to fund working capital and capital expenditures, and for other general corporate purposes; |
| • | increasing our vulnerability to general adverse economic and industry conditions; |
| • | limiting our flexibility in planning for, or reacting to, changes in our business and the industry in which we compete; and |
| • | placing us at a competitive disadvantage compared to our competitors that have less debt. |
Under the terms of the credit facility we entered into in June 2008, which we refer to as the 2008 Credit Facility, interest rates are fixed based on market indices at the time of borrowing and, depending upon the interest mechanism selected by us, may float thereafter. Some of our other smaller credit facilities also bear interest at floating rates. As a result, the amount of interest payable by us on our borrowings may increase if market interest rates change.
We may not have sufficient funds or may be unable to arrange for additional financing to pay the amounts due under our existing or any future debt. In addition, a failure to comply with the covenants under our existing debt instruments could result in an event of default under those instruments. In the event of an acceleration of amounts due under our debt instruments as a result of an event of default, we may not have sufficient funds or may be unable to arrange for additional financing to repay our indebtedness or to make any accelerated payments.
In addition, the terms of the 2008 Credit Facility provide that upon the occurrence of a change in control, as defined in the credit facility agreement, all outstanding indebtedness under the facility would become due. This provision may delay or prevent a change in control that stockholders may consider desirable.
Moreover, the United States credit markets continue to experience an unprecedented contraction, and it remains very difficult to acquire credit at this time. As a result of the tightened credit markets, we may not be able to obtain additional financing on favorable terms, or at all. If one or more of the financial institutions that supports our $165 million revolving credit facility, which is part of our 2008 Credit Facility, fails, we may not be able to find a replacement, which would negatively impact our ability to borrow the remaining funds available under the $165 million facility.
24
Table of Contents
Moreover, the 2008 Credit Facility will expire in June 2013 and all unpaid principal and interest under the facility will become due at that time. The recent and ongoing turmoil in the credit markets could affect our ability to refinance the 2008 Credit Facility or further increase our funding costs.
Our existing debt instruments contain covenants that limit our flexibility and prevent us from taking certain actions.
The agreement in connection with our 2008 Credit Facility includes a number of significant restrictive covenants. These covenants could adversely affect us by limiting our ability to plan for or react to market conditions, meet our capital needs and execute our business strategy. These covenants, among other things, limit our ability and the ability of our restricted subsidiaries to:
| • | incur additional debt; |
| • | make certain investments; |
| • | enter into certain types of transactions with affiliates; |
| • | make specified restricted payments; and |
| • | sell certain assets or merge with or into other companies. |
These covenants may limit our operating and financial flexibility and limit our ability to respond to changes in our business or competitive activities. Our failure to comply with these covenants could result in an event of default, which, if not cured or waived, could result in our being required to repay these borrowings before their scheduled due date.
Risks Associated with our Common Stock
Our corporate governance structure, including provisions of our articles of organization, by-laws, shareholder rights plan, as well as Massachusetts law, may delay or prevent a change in control or management that stockholders may consider desirable.
Provisions of our articles of organization, by-laws and our shareholder rights plan, as well as provisions of Massachusetts law, may enable our management to resist acquisition of us by a third party, or may discourage a third party from acquiring us. These provisions include the following:
| • | we have divided our board of directors into three classes that serve staggered three-year terms; |
| • | we are subject to Section 8.06 of the Massachusetts Business Corporation Law, which provides that directors may only be removed by stockholders for cause, vacancies in our board of directors may only be filled by a vote of our board of directors, and the number of directors may be fixed only by our board of directors; |
| • | we are subject to Chapter 110F of the Massachusetts General Laws, which may limit the ability of some interested stockholders to engage in business combinations with us; |
| • | our stockholders are limited in their ability to call or introduce proposals at stockholder meetings; and |
| • | our shareholder rights plan would cause a proposed acquirer of 20% or more of our outstanding shares of common stock to suffer significant dilution. |
These provisions could have the effect of delaying, deferring, or preventing a change in control of us or a change in our management that stockholders may consider favorable or beneficial. These provisions could also discourage proxy contests and make it more difficult for stockholders to elect directors and take other corporate actions. These provisions could also limit the price that investors might be willing to pay in the future for shares of our stock.
In addition, our board of directors may issue preferred stock in the future without stockholder approval. If our board of directors issues preferred stock, the rights of the holders of common stock would be subordinate to the rights of the holders of preferred stock. Our board of directors’ ability to issue the preferred stock could make it more difficult for a third party to acquire, or discourage a third party from acquiring, a majority of our stock.
25
Table of Contents
Our stock price has been, and may in the future be volatile, which could lead to losses by investors.
The market price of our common stock has fluctuated widely in the past and may continue to do so in the future. On June 30, 2010, the closing sales price of our common stock on the Nasdaq Global Select Market was $21.68 per share. During the period from June 30, 2005 to June 30, 2010, our common stock traded at prices ranging from a high of $36.16 per share to a low of $6.11 per share. Investors in our common stock must be willing to bear the risk of such fluctuations in stock price and the risk that the value of an investment in our stock could decline.
Our stock price can be affected by quarter-to-quarter variations in a number of factors including, but not limited to:
| • | operating results; |
| • | earnings estimates by analysts; |
| • | market conditions in our industry; |
| • | prospects of healthcare reform; |
| • | changes in government regulations; |
| • | general economic conditions, and |
| • | our effective income tax rate. |
In addition, the stock market has from time to time experienced significant price and volume fluctuations that are not related to the operating performance of particular companies. These market fluctuations may adversely affect the market price of our common stock. Although our common stock has traded in the past at a relatively high price-earnings multiple, due in part to analysts’ expectations of earnings growth, the price of the stock could quickly and substantially decline as a result of even a relatively small shortfall in earnings from, or a change in, analysts’ expectations.
26
Table of Contents
| ITEM 1B. | UNRESOLVED STAFF COMMENTS |
None.
| ITEM 2. | PROPERTIES |
As of June 30, 2010, PAREXEL occupied approximately 2,038,000 square feet of building space in 70 locations in 54 countries. Except for 26,600 square feet of building space in Poitiers, France, we do not own any properties, but utilize space under various leases that expire between 2010 and 2023. Total square feet by region is summarized below:
| Region |
Square Feet | |
| The Americas |
697,000 | |
| Europe, Middle East & Africa |
1,053,000 | |
| Asia/Pacific |
288,000 | |
| Total |
2,038,000 | |
Our largest facilities are located in (i) the United States, where we occupy approximately 642,000 square feet, (ii) Germany, where we occupy approximately 406,000 square feet, (iii) the United Kingdom, where we occupy approximately 268,000 square feet, (iv) South Africa, where we occupy approximately 139,000 square feet, and (v) India, where we occupy approximately 127,000 square feet. Our principal facilities are set forth below:
| Facility |
Sq. Ft. | Use of Facility |
Lease Expirations | |||
| Headquarters in Waltham, MA |
64,000 | CRS, PCMS and Corporate | 2019 | |||
| Berlin, Germany |
351,000 | All Business Segments and General & Administrative | 2016 - 2024 | |||
| Hyderabad, India |
114,000 | All Business Segments and General & Administrative | 2013 - 2014 | |||
| Lowell, MA |
108,000 | All Business Segments and General & Administrative | 2011 | |||
| Billerica, MA |
100,000 | Perceptive and General & Administrative | 2018 | |||
| Uxbridge, UK |
87,000 | CRS, PCMS and General & Administrative | 2022 | |||
| Nottingham, UK |
80,000 | Perceptive and General & Administrative | 2012 - 2015 |
The following table indicates the approximate square footage of property attributable to each of our operating segments and corporate staff:
| Segment |
Total Square Feet | |
| CRS |
977,000 | |
| PCMS |
287,000 | |
| Perceptive |
383,000 | |
| General and Administrative |
391,000 | |
| Total |
2,038,000 | |
We believe that our facilities are adequate for our operations and that additional space will be available at satisfactory terms, if needed.
| ITEM 3. | LEGAL PROCEEDINGS |
PAREXEL periodically becomes involved in various legal proceedings and claims that arise in the ordinary course of business. We believe that no matters currently pending would, in the event of an adverse outcome, have a material impact on our consolidated financial position, results of operations, or liquidity but there can be no assurance that such matters would not, in the event of an adverse outcome, have a material impact on our consolidated financial position, results of operations, or liquidity.
| ITEM 4. | [REMOVED AND RESERVED] |
27
Table of Contents
| ITEM 5. | MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS, AND ISSUER PURCHASES OF EQUITY SECURITIES |
MARKET INFORMATION AND HOLDERS
Our common stock is traded on the Nasdaq Global Select Market under the symbol “PRXL.” The table below shows the high and low sales prices of the common stock for each quarter of the Fiscal Years 2010 and 2009, respectively, on the Nasdaq Global Select Market.
| 2010 | 2009 | |||||||||||
| High | Low | High | Low | |||||||||
| First Quarter |
$ | 16.62 | $ | 11.54 | $ | 36.16 | $ | 24.69 | ||||
| Second Quarter |
$ | 14.65 | $ | 11.78 | $ | 28.84 | $ | 6.11 | ||||
| Third Quarter |
$ | 24.13 | $ | 14.10 | $ | 11.81 | $ | 7.20 | ||||
| Fourth Quarter |
$ | 25.64 | $ | 19.62 | $ | 14.87 | $ | 7.77 | ||||
As of August 23, 2010, there were approximately 141 stockholders of record of our common stock. The number does not include stockholders for which shares were held in a “nominee” or “street” name.
DIVIDENDS
We have never declared or paid any cash dividends on our capital stock, nor do we anticipate paying any cash dividends in the foreseeable future. We intend to retain future earnings for the development and expansion of our business.
Under the terms of the 2008 Credit Facility, which is described in “Lines of Credit” in Item 7 of this annual report, neither we nor any of our subsidiaries may pay any dividend or make any other distribution with respect to any shares of capital stock except that (i) PAREXEL may declare and pay dividends solely in additional shares of our common stock or in accordance with stock option plans or other benefit plans for management or employees of PAREXEL or our subsidiaries and (ii) our subsidiaries may declare and pay dividends ratably with respect to their capital stock. In addition, we only may repurchase stock during any fiscal year in an aggregate amount that does not exceed 30% of the consolidated net income (excluding extraordinary gains and extraordinary non-cash losses) for the preceding fiscal year.
28
Table of Contents
COMPANY STOCK PERFORMANCE GRAPH
The following performance graph and related information shall not be deemed “soliciting material” or to be “filed” with the SEC, nor shall such information be incorporated by reference into any future filing under the Securities Act or the Exchange Act, except to the extent PAREXEL specifically incorporates it by reference.
Our common stock is listed for trading on the Nasdaq Global Select Market under the symbol “PRXL.” The Stock Price Performance Graph set forth below compares the cumulative total stockholder return on our common stock for the period from June 30, 2005 through June 30, 2010, with the cumulative total return of the Nasdaq U.S. Stock Index and the Nasdaq Health Services Index over the same period. The comparison assumes $100 was invested on June 30, 2005 in PAREXEL’s common stock, in the Nasdaq U.S. Stock Index, and in the Nasdaq Health Services Index and assumes reinvestment of dividends, if any.
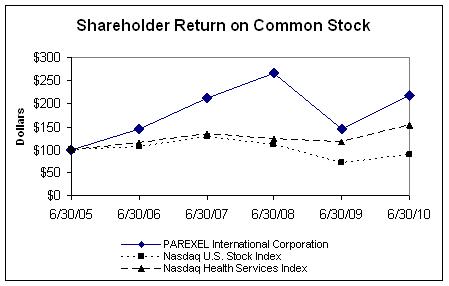
| Fiscal Years Ended June 30, | ||||||||||||||||||
| Total Return Index For: |
2005 | 2006 | 2007 | 2008 | 2009 | 2010 | ||||||||||||
| PAREXEL International Stock |
$ | 100.00 | $ | 145.61 | $ | 212.21 | $ | 265.49 | $ | 145.11 | $ | 218.77 | ||||||
| Nasdaq U.S. Stock Index |
$ | 100.00 | $ | 106.32 | $ | 126.72 | $ | 110.84 | $ | 71.47 | $ | 88.57 | ||||||
| Nasdaq Health Services Index |
$ | 100.00 | $ | 114.71 | $ | 134.51 | $ | 123.42 | $ | 116.98 | $ | 152.65 | ||||||
The stock price performance shown on the graph above is not necessarily indicative of future price performance. Information used in the graph was obtained from The Nasdaq Stock Market, a source believed to be reliable, but we are not responsible for any errors or omissions in such information.
29
Table of Contents
| ITEM 6. | SELECTED FINANCIAL DATA |
The following selected consolidated financial data of PAREXEL for the five years ended June 30, 2010 are derived from our consolidated financial statements. The information set forth below should be read in conjunction with “Management’s Discussion and Analysis of Financial Condition and Results of Operations” included as Item 7 in this annual report and the consolidated financial statements and related footnotes included as Item 8 in this annual report.
| For the fiscal years ended
June 30, (in thousands, except per share data and number of employees) | |||||||||||||||
| 2010(1) | 2009(2) | 2008(3 ) | 2007 | 2006(4 ) | |||||||||||
| OPERATIONS |
|||||||||||||||
| Service revenue |
$ | 1,131,039 | $ | 1,050,755 | $ | 964,283 | $ | 741,955 | $ | 614,947 | |||||
| Income from operations |
$ | 83,109 | $ | 75,644 | $ | 86,666 | $ | 57,566 | $ | 39,855 | |||||
| Net income |
$ | 41,542 | $ | 39,307 | $ | 64,640 | $ | 37,289 | $ | 23,544 | |||||
| Basic earnings per share |
$ | 0.72 | $ | 0.68 | $ | 1.16 | $ | 0.68 | $ | 0.44 | |||||
| Diluted earnings per share |
$ | 0.71 | $ | 0.68 | $ | 1.12 | $ | 0.66 | $ | 0.44 | |||||
| FINANCIAL POSITION |
|||||||||||||||
| Cash and marketable securities |
$ | 107,413 | $ | 96,352 | $ | 51,918 | $ | 96,677 | $ | 92,749 | |||||
| Working capital |
$ | 158,624 | $ | 191,705 | $ | 146,535 | $ | 118,746 | $ | 131,552 | |||||
| Total assets |
$ | 1,220,710 | $ | 1,224,461 | $ | 948,071 | $ | 680,013 | $ | 538,633 | |||||
| Short-term debt |
$ | 32,082 | $ | 32,090 | $ | 66,474 | $ | 30,463 | $ | 498 | |||||
| Long-term obligations |
$ | 283,922 | $ | 353,998 | $ | 112,319 | $ | 29,003 | $ | 37,455 | |||||
| Stockholders’ equity |
$ | 439,555 | $ | 414,745 | $ | 428,091 | $ | 316,616 | $ | 248,763 | |||||
| OTHER DATA |
|||||||||||||||
| Purchases of property and equipment |
$ | 78,959 | $ | 75,181 | $ | 67,067 | $ | 40,855 | $ | 29,763 | |||||
| Depreciation and amortization |
$ | 60,320 | $ | 52,928 | $ | 37,686 | $ | 30,855 | $ | 26,035 | |||||
| Number of employees |
9,720 | 9,275 | 8,050 | 6,485 | 5,600 | ||||||||||
| Weighted average shares used in computing: |
|||||||||||||||
| Basic earnings per share |
58,062 | 57,538 | 55,896 | 54,633 | 53,113 | ||||||||||
| Diluted earnings per share |
58,756 | 57,847 | 57,461 | 56,216 | 54,026 | ||||||||||
| (1) | Income from operations for the year ended June 30, 2010 includes restructuring charges of $0.5 million of accelerated depreciation on abandoned facilities, $5.2 million of facility-related costs and $11.6 million in severance costs; $6.2 million impairment charge on an investment, $4.3 million in legal settlement costs related to a small acquisition which was completed several years ago; the release of $1.1 million in certain reserves due to lower than expected wind-down costs related to the $15 million accrual established in Q2 FY09 for a client contract default (see below); and a $0.4 million asset impairment charge. |
| (2) | Income from operations for the year ended June 30, 2009 includes $15 million in other charges ($12.3 million for bad debt expense and $2.7 million in anticipated wind-down costs and related expenses for service fees, pass-through costs, and investigator fees). These costs were related to the cancellation of a Phase III trial by a small biopharma client that had filed for bankruptcy. See Note 18 to the consolidated financial statements in this annual report on Form 10-K for more detail. |
| (3) | Income from operations for the year ended June 30, 2008 reflects a $0.9 million benefit from changes in restructuring charges related to facilities and severance expenses. |
| (4) | Income from operations for the year ended June 30, 2006 reflects $1.6 million of compensation expense in conjunction with the acquisition of the Perceptive minority interest. Additionally, we recorded a $2.6 million reduction to the existing restructuring reserve as a result of the execution of sub-lease agreements and changes in assumptions of leased facilities, which was offset by $1.8 million in severance-related restructuring expenses incurred during Fiscal Year 2006 in association with the fourth quarter Fiscal Year 2005 restructuring plan. |
30
Table of Contents
| ITEM 7. | MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
OVERVIEW
We are a leading biopharmaceutical services company, providing a broad range of expertise in clinical research, medical communications services, consulting and informatics and advanced technology products and services to the worldwide pharmaceutical, biotechnology, and medical device industries. Our primary objective is to provide solutions for managing the biopharmaceutical product lifecycle with the goal of reducing the time, risk, and cost associated with the development and commercialization of new therapies. Since our incorporation in 1983, we have developed significant expertise in processes and technologies supporting this strategy. Our product and service offerings include: clinical trials management, data management, biostatistical analysis, medical communications, clinical pharmacology, patient recruitment, regulatory and product development consulting, health policy and reimbursement, performance improvement, medical imaging services, ClinPhone® RTSM, IMPACT® and TrialWorks™ CTMS, DataLabs® EDC, web-based portals, systems integration, ePRO, and other drug development consulting services. We believe that our comprehensive services, depth of therapeutic area expertise, global footprint and related access to patients, and sophisticated information technology, along with our experience in global drug development and product launch services, represent key competitive strengths.
We are managed through three business segments: Clinical Research Services (“CRS”), PAREXEL Consulting and MedCom Services (“PCMS”) and Perceptive Informatics, Inc. (“Perceptive”).
| • | CRS constitutes our core business and includes all phases of clinical research from Early Phase (encompassing the early stages of clinical testing that range from first-in-man through proof-of-concept studies, formerly referred to as “Clinical Pharmacology”) to Phase II-III and Phase IV, which we call Peri Approval Clinical Excellence (“PACE”). Our services include clinical trials management and biostatistics, data management and clinical pharmacology, as well as related medical advisory, patient recruitment, clinical supply and drug logistics, pharmacovigilance, and investigator site services. |
| • | PCMS provides technical expertise and advice in such areas as drug development, regulatory affairs, and biopharmaceutical process and management consulting. PCMS also provides a full spectrum of market development, product development, and targeted communications services in support of product launch. PCMS consultants identify alternatives and propose solutions to address clients’ product development, registration, and commercialization issues. In addition, PCMS provides health policy consulting, as well as reimbursement and market access (“RMA”) services. |
| • | Perceptive provides information technology solutions designed to improve clients’ product development processes. Perceptive offers a portfolio of products and services that includes medical imaging services, ClinPhone® RTSM, IMPACT® and TrialWorks™ CTMS, DataLabs® EDC, web-based portals, systems integration, and ePRO. |
We conduct a significant portion of our operations in foreign countries. Approximately 65.4% and 64.4% of our consolidated service revenue for the fiscal years ended June 30, 2010 and 2009, respectively, were from non-U.S. operations. Because our financial statements are denominated in U.S. dollars, changes in foreign currency exchange rates can have a significant effect on our operating results. For the Fiscal Year 2010, approximately 24.1% of total consolidated service revenue was denominated in Euros and approximately 14.6% of total consolidated service revenue was denominated in pounds sterling. For the Fiscal Year 2009, approximately 24.3% of total consolidated service revenue was denominated in Euros and approximately 15.6% of total consolidated service revenue was denominated in pounds sterling.
Approximately 90% of our contracts are fixed price, with some variable components, and range in duration from a few months to several years. Cash flows from these contracts typically consist of a down payment required to be paid at the time of contract execution with the balance due in installments over the contract’s duration, usually on a milestone achievement basis. Revenue from these contracts is generally recognized as work is performed. As a result, cash receipts do not necessarily correspond to costs incurred and revenue recognized on contracts.
Generally, our clients can either terminate their contracts with us upon thirty to sixty days notice or delay execution of services. Clients may terminate or delay contracts for a variety of reasons, including: merger or potential merger-related activities involving the client, the failure of products being tested to satisfy safety requirements or efficacy criteria, unexpected or undesired clinical results of the product, client cost reductions as a result of budgetary limits or changing priorities, the client’s decision to forego a particular study, insufficient patient enrollment or investigator recruitment, or clinical drug manufacturing problems resulting in shortages of the product.
31
Table of Contents
ACQUISITIONS
Acquisitions are an important component of our business strategy. We account for acquisitions in accordance with Accounting Standards Codification (“ASC”) 805, “Business Combinations.” Since June 30, 2007, we have completed the following acquisitions:
ClinPhone
In August 2008, we completed the acquisition of ClinPhone, one of the world’s leading clinical technology organizations, for approximately $190 million, comprised of $172 million for the stock of ClinPhone and $18 million as repayment of ClinPhone’s existing debt. We believe that the acquisition of ClinPhone has advanced our position as a clinical technology leader. Biopharmaceutical companies have increasingly requested technology solutions and expertise to support the full range of clinical development activities while improving the speed and efficiency of clinical programs. We believe that the broad technological offering that we now provide gives clients a more comprehensive and robust suite of clinical information technologies.
APEX
In September 2007, we acquired a majority of the outstanding shares of Taiwan-based APEX International Clinical Research Co., Ltd. (“APEX”) and completed the acquisition of all of the outstanding shares of APEX in November 2007 for a total of approximately $55 million. The acquisition strengthened our global capabilities, providing clients with a wider range of clinical research service offerings throughout the Asia-Pacific region, including mainland China, Hong Kong, India, Taiwan, Singapore, Indonesia, South Korea, Malaysia, Thailand, the Philippines, New Zealand, and Australia.
CRITICAL ACCOUNTING POLICIES AND ESTIMATES
This discussion and analysis of our financial condition and results of operations are based on our consolidated financial statements, which have been prepared in accordance with accounting principles generally accepted in the U.S. The preparation of these financial statements requires us to make estimates and assumptions that affect the reported amounts of assets and liabilities, revenues and expenses, and other financial information. On an ongoing basis, we evaluate our estimates and judgments. We base our estimates on historical experience and on various other factors that we believe to be reasonable under the circumstances. Actual results may differ from these estimates under different assumptions or conditions.
We regard an accounting estimate underlying our financial statements as a “critical accounting estimate” if the nature of the estimate or assumption is material due to the level of subjectivity and judgment involved or the susceptibility of such matter to change and if the impact of the estimate or assumption on financial condition or operating performance is material. We believe that the following accounting policies are most critical to aid in fully understanding and evaluating our reported financial results:
REVENUE RECOGNITION
We recognize service revenue when all of the following criteria are met: (i) persuasive evidence of an arrangement exists, (ii) the service offering has been delivered to the client, (iii) the collection of fees is probable, and (iv) amount of fees to be paid by the client is fixed or determinable.
Our CRS client arrangements generally involve multiple service deliverables, where bundled service deliverables are accounted for in accordance with ASC 605-25, “Multiple-Element Arrangements.” We have determined that each of our service deliverables has standalone value and have established objective evidence of fair value for each of our service deliverables based on the price charged when sold to other similar customers. Accordingly, revenues are recognized upon delivery of actual units and when all other revenue recognition criteria are met.
Within Perceptive’s ClinPhone RTSM business, we offer selected software solutions through hosted applications delivered through a standard web-browser. Revenue from hosting service arrangements is recognized ratably over the term of the hosting arrangement, including customary and expected extensions. Fees charged and costs incurred in the setup stage of these arrangements are deferred until the start of the hosting period and are amortized and recognized ratably over the estimated hosting period. Within the CTMS operating unit of Perceptive, software revenue is recognized on a proportional performance basis due to the significant nature of customization of each project. Within the EDC operating unit of Perceptive, revenue is recognized ratably over the contract service period.
Deferred revenue represents amounts billed or cash received in advance of revenue recognized.
32
Table of Contents
Critical management estimates may be involved in the determination of “fair value,” “stand-alone value,” “hosting period,” and other revenue elements. Changes to these elements could affect the amount and timing of revenue recognition.
BILLED ACCOUNTS RECEIVABLE, UNBILLED ACCOUNTS RECEIVABLE AND DEFERRED REVENUE
Billed accounts receivable represent amounts for which invoices have been sent to clients. Unbilled accounts receivable represent amounts recognized as revenue for which invoices have not yet been sent to clients. Deferred revenue represents amounts billed or payments received for which revenue has not yet been earned. We maintain a provision for losses on receivables based on historical collectability and specific identification of potential problem accounts. Critical management estimates may be involved in the determination of “collectability” and the amounts required to be recorded as provisions for losses on receivables.
INCOME TAXES
Our global provision for corporate income taxes is determined in accordance with ASC 740, “Income Taxes,” which requires that deferred tax assets and liabilities be recognized for the effect of temporary differences between the book and tax basis of recorded assets and liabilities. A valuation allowance is established if it is more likely than not that future tax benefits from the deferred tax assets will not be realized. Income tax expense is based on the distribution of profit before tax among the various taxing jurisdictions in which we operate, adjusted as required by the tax laws of each taxing jurisdiction. Changes in the distribution of profits and losses among taxing jurisdictions may have a significant impact on our effective tax rate.
We account for uncertain tax positions in accordance with the provisions of ASC 740, which requires financial statement reporting of the expected future tax consequences of uncertain tax return reporting positions on the presumption that all relevant tax authorities possess full knowledge of those tax reporting positions, as well as all of the pertinent facts and circumstances. In addition, ASC 740 mandates expanded financial statement disclosure about uncertainty in income tax reporting positions.
We are subject to ongoing audits by federal, state and foreign tax authorities that may result in proposed assessments. Our estimate for the potential outcome for any uncertain tax issue is based on judgment. We believe we have adequately provided for any uncertain tax positions in accordance with ASC 740. However, future results may include favorable or unfavorable adjustments to our estimated tax liabilities in the period assessments are made or resolved or when statutes of limitation on potential assessments expire.
GOODWILL
Goodwill represents the excess of the cost of an acquired business over the fair value of the related net assets at the date of acquisition. Under ASC 350, “Intangibles—Goodwill and Other,” goodwill is subject to annual impairment testing or more frequent testing if an event occurs or circumstances change that would more likely than not reduce the carrying value of the reporting unit below its fair value. The impairment testing involves determining the fair market value of each of the reporting units with which the goodwill was associated and comparing that value with the reporting unit’s carrying value. We conducted an impairment assessment in Fiscal Year 2010 using a discounted cash flow analysis at the reporting unit level to determine fair value. This analysis included significant judgment regarding the assumptions used, such as our weighted average cost of capital, revenue growth rates, profit margins, capital expenditures, and other factors that were all based on current strategic forecasts and other financial metrics. As of June 30, 2010, there were no required adjustments to the carrying value of goodwill since the fair value of our net assets were substantially above the carrying value of our net assets at each of our reporting units. Variations over time in the measures and estimates used in the discounted cash flow analysis could result in future impairment of goodwill that could have a material impact to our financial position or our results of operations.
33
Table of Contents
RESULTS OF OPERATIONS
Note 19 to our consolidated financial statements included in this Annual Report on Form 10-K provides a summary of our unaudited quarterly results of operations for the years ended June 30, 2010 and 2009.
ANALYSIS BY SEGMENT
We evaluate our segment performance and allocate resources based on service revenue and gross profit (service revenue less direct costs), while other operating costs are allocated and evaluated on a geographic basis. Accordingly, we do not include the impact of selling, general, and administrative expenses, depreciation and amortization expense, interest income (expense), other income (loss), and income tax expense (benefit) in segment profitability. We attribute revenue to individual countries based upon the number of hours of services performed in the respective countries and inter-segment transactions are not included in service revenue. Furthermore, we have a global infrastructure supporting our business segments and therefore, assets are not identified by reportable segment. Service revenue, direct costs, and gross profit on service revenue for Fiscal Years 2010, 2009, and 2008 were as follows:
| Twelve Months Ended | Increase (Decrease) |
% | |||||||||||
| (in thousands) |
June 30, 2010 | June 30, 2009 | |||||||||||
| Service revenue |
|||||||||||||
| Clinical Research Services |
$ | 870,721 | $ | 804,237 | $ | 66,484 | 8.3 | % | |||||
| PAREXEL Consulting and MedCom Services |
121,652 | 121,785 | (133 | ) | -0.1 | % | |||||||
| Perceptive Informatics, Inc. |
138,666 | 124,733 | 13,933 | 11.2 | % | ||||||||
| Total service revenue |
$ | 1,131,039 | $ | 1,050,755 | $ | 80,284 | 7.6 | % | |||||
| Direct costs |
|||||||||||||
| Clinical Research Services |
$ | 550,167 | $ | 517,250 | $ | 32,917 | 6.4 | % | |||||
| PAREXEL Consulting and MedCom Services |
74,492 | 78,223 | (3,731 | ) | -4.8 | % | |||||||
| Perceptive Informatics, Inc. |
79,908 | 79,590 | 318 | 0.4 | % | ||||||||
| Total direct costs |
$ | 704,567 | $ | 675,063 | $ | 29,504 | 4.4 | % | |||||
| Gross profit |
|||||||||||||
| Clinical Research Services |
$ | 320,554 | $ | 286,987 | $ | 33,567 | 11.7 | % | |||||
| PAREXEL Consulting and MedCom Services |
47,160 | 43,562 | 3,598 | 8.3 | % | ||||||||
| Perceptive Informatics, Inc. |
58,758 | 45,143 | 13,615 | 30.2 | % | ||||||||
| Total gross profit |
$ | 426,472 | $ | 375,692 | $ | 50,780 | 13.5 | % | |||||
| Twelve Months Ended | Increase (Decrease) |
% | |||||||||||
| (in thousands) |
June 30, 2009 | June 30, 2008 | |||||||||||
| Service revenue |
|||||||||||||
| Clinical Research Services |
$ | 804,237 | $ | 745,641 | $ | 58,596 | 7.9 | % | |||||
| PAREXEL Consulting and MedCom Services |
121,785 | 129,804 | (8,019 | ) | -6.2 | % | |||||||
| Perceptive Informatics, Inc. |
124,733 | 88,838 | 35,895 | 40.4 | % | ||||||||
| Total service revenue |
$ | 1,050,755 | $ | 964,283 | $ | 86,472 | 9.0 | % | |||||
| Direct costs |
|||||||||||||
| Clinical Research Services |
$ | 517,250 | $ | 493,879 | $ | 23,371 | 4.7 | % | |||||
| PAREXEL Consulting and MedCom Services |
78,223 | 85,930 | (7,707 | ) | -9.0 | % | |||||||
| Perceptive Informatics, Inc. |
79,590 | 49,590 | 30,000 | 60.5 | % | ||||||||
| Total direct costs |
$ | 675,063 | $ | 629,399 | $ | 45,664 | 7.3 | % | |||||
| Gross profit |
|||||||||||||
| Clinical Research Services |
$ | 286,987 | $ | 251,762 | $ | 35,225 | 14.0 | % | |||||
| PAREXEL Consulting and MedCom Services |
43,562 | 43,874 | (312 | ) | -0.7 | % | |||||||
| Perceptive Informatics, Inc. |
45,143 | 39,248 | 5,895 | 15.0 | % | ||||||||
| Total gross profit |
$ | 375,692 | $ | 334,884 | $ | 40,808 | 12.2 | % | |||||
34
Table of Contents
FISCAL YEAR ENDED JUNE 30, 2010 COMPARED WITH THE FISCAL YEAR ENDED JUNE 30, 2009
For Fiscal Year 2010, we had net income of $41.5 million compared with net income of $39.3 million for Fiscal Year 2009. This increase was due primarily to factors described below. On a fully diluted basis, earnings per share increased to $0.71 in Fiscal Year 2010 from $0.68 for the corresponding period in Fiscal Year 2009.
Revenue
Service revenue increased by $80.3 million, or 7.6%, to $1,131.0 million for Fiscal Year 2010 from $1,050.8 million for Fiscal Year 2009. On a geographic basis, service revenue was distributed as follows (in millions):
| Fiscal Year 2010 | Fiscal Year 2009 | |||||||||||
| Region |
Service Revenue | % of Total | Service Revenue | % of Total | ||||||||
| The Americas |
$ | 449.3 | 39.7 | % | $ | 426.3 | 40.6 | % | ||||
| Europe, Middle East & Africa |
$ | 548.4 | 48.5 | % | $ | 528.9 | 50.3 | % | ||||
| Asia/Pacific |
$ | 133.3 | 11.8 | % | $ | 95.6 | 9.1 | % | ||||
Service revenue in The Americas increased by $23.0 million, or 5.4%; Europe, Middle East & Africa service revenue increased by $19.5 million, or 3.7%; and Asia/Pacific service revenue increased by $37.7 million, or 39.5%. Overall, service revenue was positively impacted by foreign currency exchange rate fluctuations including: $1.3 million in The Americas, $1.1 million in Europe, Middle East & Africa, and $7.9 million in Asia/Pacific.
On a segment basis, CRS service revenue increased by $66.5 million, or 8.3%, to $870.7 million for Fiscal Year 2010 from $804.2 million for Fiscal Year 2009. The growth was attributable to a $47.1 million increase in Phase II-III/PACE, the positive $12.1 million impact of foreign currency exchange rate fluctuations, and a $7.3 million increase in Early Phase. Growth in the Phase II-III/PACE business has been largely due to the impact of strategic partnerships and increased business activity in the Asia/Pacific region as a result of global studies and more work for local biopharmaceutical companies. Growth in Early Phase business was a result of increased demand due largely to improvements in the unit’s sales team and approach.
PCMS service revenue decreased nominally to $121.7 million for Fiscal Year 2010 from $121.8 million for the same period in Fiscal Year 2009. The decline was caused by a $2.4 million decrease in health policy and strategic reimbursement services, which was adversely affected by uncertainty in the regulatory markets due to changes in the U.S. healthcare law, and the $0.7 million negative impact of foreign currency exchange rate fluctuations; partly offset by a $3.0 million increase in the consulting business, including strong growth in compliance consulting work.
Perceptive service revenue increased by $14.0 million, or 11.2%, to $138.7 million for Fiscal Year 2010 from $124.7 million for Fiscal Year 2009. The increase was due to an $18.0 million increase in ClinPhone RTSM and support services, a $2.5 million increase in EDC services, and a $2.2 million increase in medical imaging; partly offset by a $7.6 million decrease in CTMS and installation support services, and the $1.1 million negative impact of foreign currency exchange rate fluctuations. The overall increases are due to the trend toward increasing integration of technology into clinical trials. As of Fiscal Year 2010, Perceptive handled more electronic case report forms than paper-based ones. The decrease in CTMS and integration support services was due, in part, to lower demand for perpetual licenses, replaced by increased demand for hosted solutions.
Reimbursement revenue consists of reimbursable out-of-pocket expenses incurred on behalf of and reimbursable by clients. Reimbursement revenue does not yield any gross profit to us, nor does it have an impact on net income.
Direct Costs
Direct costs increased by $29.5 million, or 4.4%, to $704.6 million for the Fiscal Year 2010 from $675.1 million for the Fiscal Year 2009. As a percentage of total service revenue, direct costs decreased to 62.3% from 64.2% for the respective periods.
On a segment basis, CRS direct costs increased by $32.9 million, or 6.4%, to $550.2 million for Fiscal Year 2010 from $517.3 million for Fiscal Year 2009. The increase was due to higher business activity levels and the $7.1 million negative impact of foreign exchange rate fluctuations. As a percentage of service revenue, CRS direct costs decreased to 63.2% for the Fiscal Year 2010 from 64.3% for the Fiscal Year 2009 due primarily to strong performance in Asia/Pacific, the continued effectiveness of cost controls, and improved productivity and efficiency; partly offset by an unfavorable swing in bonus expense.
35
Table of Contents
PCMS direct costs decreased $3.7 million, or 4.8%, to $74.5 million for Fiscal Year 2010 from $78.2 million for Fiscal Year 2009. This $3.7 million decrease was caused mainly by a lower level of activity in the medical communications business. As a percentage of service revenue, PCMS direct costs decreased to 61.2% from 64.2% for the respective periods. This reduction resulted from efforts by PCMS to implement substantial improvements in business processes, mainly related to the strategic marketing portion of the business and the shedding of certain unprofitable service lines.
Perceptive direct costs increased slightly by $0.3 million, or 0.4%, to $79.9 million for Fiscal Year 2010 from $79.6 million for Fiscal Year 2009. The increase was due to increases in ClinPhone RTSM and support services activities and higher spending in CTMS and integration support services; partly offset by a decrease in other areas. This latter decrease was due largely to the fact that we had no counterparts to a $1.4 million reserve for a customer dispute in medical imaging and $0.6 million associated with the termination of a (pre-acquisition) Perceptive IVR supplier contract, both recorded in Fiscal Year 2009. As a percentage of service revenue, Perceptive direct costs decreased to 57.6% for Fiscal Year 2010 from 63.8% for Fiscal Year 2009.
Selling, General and Administrative
Selling, general and administrative (“SG&A”) expense increased by $35.2 million, or 15.2%, to $267.4 million for Fiscal Year 2010 from $232.2 million for Fiscal Year 2009. This $35.2 million increase was due primarily to a $19.3 million increase in personnel costs (driven in part by bonus expense in Fiscal Year 2010 for which there was no comparable bonus expense in Fiscal Year 2009), a $5.4 million increase in sales-related costs (including commissions), $4.3 million related to the legal settlement costs related to a small acquisition which was completed several years ago, $4.3 million in higher rent and office expenses, and $1.6 million related to the negative impact for foreign currency exchange rate movements. As a percentage of service revenue, SG&A increased to 23.6% for Fiscal Year 2010 from 22.1% for Fiscal Year 2009.
Depreciation and Amortization
Depreciation and amortization (“D&A”) expense increased by $7.4 million, or 14.0%, to $60.3 million for the Fiscal Year 2010 from $52.9 million for the Fiscal Year 2009 primarily due to increased capital expenditures over the past twenty four months. As a percentage of service revenue, D&A expense increased to 5.3% for the Fiscal Year 2010 from 5.0% for the same period in 2009.
Other Charge
In the second quarter of Fiscal Year 2009, we recorded $15 million in reserves for bad debt expense related to impaired accounts receivable and anticipated wind-down costs and related expenses for service fees, pass-through costs, and investigator fees from a small biopharma client that filed for bankruptcy protection. In the second quarter of Fiscal Year 2010, we released $1.1 million of these reserves to reflect lower-than-anticipated close-out costs. See Note 18 to our consolidated financial statements included in this Annual Report on Form 10-K for more information.
Restructuring Charge
For Fiscal Year 2010, we recorded $16.8 million in restructuring charges in association with the 2010 Restructuring Plan, including approximately $11.6 million in employee separation benefits associated with the elimination of 238 managerial and staff positions and $5.2 million in costs related to the abandonment of certain property leases.
Income from Operations
Income from operations increased to $83.1 million for Fiscal Year 2010 from $75.6 million in Fiscal Year 2009. Income from operations as a percentage of service revenue, or operating margin, increased to 7.3% from 7.2% for the respective periods.
Other Expense
Other expense increased by $8.1 million to $19.9 million in Fiscal Year 2010 from $11.8 million in Fiscal Year 2009. The $8.1 million increase was attributable to $9.0 million in miscellaneous expense; partly offset by a $0.9 million increase in interest income, net of interest expense.
Miscellaneous expense for Fiscal Year 2010 of $9.6 million included $7.0 million of losses related to foreign exchange contracts, a $6.1 million reserve for an impaired investment in a French laboratory that filed for bankruptcy protection, and a $0.4 million asset impairment charge; partly offset by $4.4 million in gains on the revaluation of foreign denominated assets/liabilities. Miscellaneous income for Fiscal Year 2009 of $0.6 million consisted of a $3.0 million legal settlement charge related to a contract dispute, $2.3 million for the write-off of certain impaired assets, $0.9 million of losses related to foreign exchange contracts, and $1.4 million in other losses; partly offset by $7.0 million of gains on the revaluation of foreign denominated assets/liabilities.
36
Table of Contents
Taxes
For the Fiscal Year 2010 and 2009, we had an effective income tax rate of 34.2% and 38.4%, respectively. The reduction in the tax rate was primarily attributable to a decrease in the reserves required for uncertain tax positions as of June 30, 2010 and improved profitability in the U.S.
FISCAL YEAR ENDED JUNE 30, 2009 COMPARED WITH THE FISCAL YEAR ENDED JUNE 30, 2008
For Fiscal Year 2009, we had net income of $39.3 million compared with net income of $64.6 million for Fiscal Year 2008. This decrease was due primarily to factors described below. On a fully diluted basis, earnings per share decreased to $0.68 from $1.12 for the corresponding periods. Fiscal Year 2008 included a tax benefit of $0.07 per share on a fully diluted basis, resulting from a decrease in German statutory tax rates.
Revenue
Service revenue increased by $86.5 million, or 9.0%, to $1,050.8 million for Fiscal Year 2009 from $964.3 million for Fiscal Year 2008. On a geographic basis, service revenue was distributed as follows (in millions):
| Fiscal Year 2009 | Fiscal Year 2008 | |||||||||||
| Region |
Service Revenue | % of Total | Service Revenue | % of Total | ||||||||
| The Americas |
$ | 426.3 | 40.6 | % | $ | 377.9 | 39.2 | % | ||||
| Europe, Middle East & Africa |
$ | 528.9 | 50.3 | % | $ | 515.4 | 53.5 | % | ||||
| Asia/Pacific |
$ | 95.6 | 9.1 | % | $ | 71.0 | 7.4 | % | ||||
Service revenue in The Americas increased by $48.4 million, or 12.8%; Europe, Middle East & Africa service revenue increased by $13.5 million, or 2.6%; and Asia/Pacific service revenue increased by $24.6 million, or 34.6%. The increase in Europe, Middle East & Africa was negatively impacted by foreign currency fluctuations of approximately $85.1 million. Excluding that impact, growth in that region would have been approximately 16.5%. The impact of foreign currency fluctuations in the Americas and Asia/Pacific regions was minimal.
On a segment basis, CRS service revenue increased by $58.6 million, or 7.9%, to $804.2 million for Fiscal Year 2009 from $745.6 million for Fiscal Year 2008. This increase was due primarily to $121.0 million in additional revenue in the Phase II-III/PACE portions of the business as a result of continuing strong demand for our services, particularly from the large pharmaceutical segment, and approximately $6.7 million in incremental revenue from the acquisition of APEX, which was acquired in the first quarter of Fiscal Year 2008. These increases were partially offset by the negative impact of foreign currency fluctuations of approximately $52.4 million and a decrease of $16.7 million in our Early Phase business, as a result of lower demand caused by uncertainties in the capital markets that led clients to reduce their research and development spending.
PCMS service revenue decreased by $8.0 million, or 6.2%, to $121.8 million for Fiscal Year 2009 from $129.8 million for the same period in 2008. This decrease was attributable to the $2.5 million impact of the disposition of Barnett Educational Services and targeted withdrawals from certain other unprofitable service lines in the prior year in the MedCom business and $12.4 million related to foreign currency fluctuations. These amounts were partly offset by growth of approximately $6.9 million in PAREXEL Consulting that was driven by increased demand by clients for assistance in the new stricter regulatory environment.
Perceptive service revenue increased by $35.9 million, or 40.4%, to $124.7 million for Fiscal Year 2009 from $88.8 million for Fiscal Year 2008. This increase was due primarily to the acquisition of ClinPhone, which contributed $53.3 million in revenue; partly offset by approximately $13.3 million related to the negative impact of foreign currency fluctuations and a $4.1 million decrease in other parts of the business, primarily in our legacy IVR service and our medical imaging units. The decrease in our legacy IVR product was due primarily to the migration of our clients to the ClinPhone RTSM product.
ClinPhone’s revenue of $53.3 million included the $21.0 million impact of a correction with regard to the accounting treatment for revenue recognition of certain projects involving the ClinPhone RTSM system and acquisition-related deferred revenue. With regard to certain ClinPhone RTSM-related projects, we had been recognizing start-up revenue (and costs) during the start-up period of a study. However, this start-up revenue, totaling approximately $16.9 million in Fiscal Year 2009, should have been deferred and recognized ratably over an estimated “hosting” period. Additionally, we had recognized $4.1 million of acquired deferred revenue on projects for which the work had been completed prior to our acquisition. Since there was no additional work that needed to be done on those projects after the acquisition date, the fair value of this deferred revenue should have been zero at the time of acquisition.
37
Table of Contents
Reimbursement revenue consists of reimbursable out-of-pocket expenses incurred on behalf of and reimbursable by clients. Reimbursement revenue does not yield any gross profit to us, nor does it have an impact on net income.
Direct Costs
Direct costs increased by $45.7 million, or 7.3%, to $675.1 million for the Fiscal Year 2009 from $629.4 million for the Fiscal Year 2008. As a percentage of total service revenue, direct costs decreased to 64.2% from 65.3% for the respective periods.
On a segment basis, CRS direct costs increased by $23.4 million, or 4.7%, to $517.3 million for Fiscal Year 2009 from $493.9 million for Fiscal Year 2008. This increase resulted from approximately $77.6 million in costs (primarily for labor costs) to support growth in the Phase II-III/PACE portions of the business, including $4.3 million in incremental costs related to APEX; partly offset by $9.0 million in lower costs for the Early Phase business as a result of lower business volume, and approximately $45.1 million attributable to the impact of foreign currency fluctuations. As a percentage of service revenue, CRS direct costs decreased to 64.3% for the Fiscal Year 2009 from 66.2% for the Fiscal Year 2008 driven by improved productivity and efficiency in the Phase II-III/PACE portion of the business.
PCMS direct costs decreased $7.7 million, or 9.0%, to $78.2 million for Fiscal Year 2009 from $85.9 million for Fiscal Year 2008. This $7.7 million decrease was caused by approximately $6.1 million in foreign currency fluctuations, a $0.8 million decrease in the consulting business due to productivity improvements, and a $0.7 million reduction in expenses for the MedCom business due to the shedding of Barnett Educational Services and other unprofitable business lines. As a percentage of service revenue, PCMS direct costs decreased to 64.2% from 66.2% for the respective periods.
Perceptive direct costs increased by $30.0 million, or 60.5%, to $79.6 million for Fiscal Year 2009 from $49.6 million for Fiscal Year 2008. Of the total $30.0 million increase, $27.4 million was due to incremental direct costs associated with ClinPhone, $8.3 million was related to incremental labor costs, $1.4 million was related to the recording of a reserve for a customer dispute, and $0.6 million was associated with the termination of a (pre-acquisition) Perceptive supplier contract; offset by $3.7 million related to foreign currency fluctuations and a $4.0 million decrease in other expenses. As a percentage of service revenue, Perceptive direct costs increased to 63.8% for Fiscal Year 2009 from 55.8% for Fiscal Year 2008, due primarily to the decrease in revenue related to the correction in accounting for certain ClinPhone hosted service projects, as described above.
Selling, General and Administrative
Selling, general and administrative (“SG&A”) expense increased by $20.8 million, or 9.8%, to $232.2 million for Fiscal Year 2009 from $211.4 million for Fiscal Year 2008. This $20.8 million increase was due primarily to $34.7 million in incremental expenses resulting from the acquisitions of ClinPhone and APEX and approximately $11.1 million in costs required to support higher business volume, primarily in personnel and facilities costs; offset by $25.0 million attributable to the positive impact of foreign exchange fluctuations. As a percentage of service revenue, SG&A increased slightly to 22.1% for Fiscal Year 2009 as compared with 21.9% for Fiscal Year 2008.
Depreciation and Amortization
Depreciation and amortization (“D&A”) expense increased by $15.2 million, or 40.4%, to $52.9 million for the Fiscal Year 2009 from $37.7 million for the Fiscal Year 2008. The increase was due primarily to $8.9 million in depreciation and amortization expense associated with ClinPhone-related assets and $10.9 million related primarily to the depreciation of fixed assets; partially offset by $4.6 million attributable to foreign exchange fluctuations. As a percentage of service revenue, D&A expense increased to 5.0% for the Fiscal Year 2009 from 3.9% for the same period in 2008.
Other Charge
In the second quarter of Fiscal Year 2009, we recorded $15 million in reserves for bad debt expense related to impaired accounts receivable and anticipated wind-down costs and related expenses for service fees, pass-through costs, and investigator fees from a small biopharma client that filed for bankruptcy protection. See Note 18 to our consolidated financial statements included in this Annual Report on Form 10-K for more information.
Other Income and Expense
We recorded net other expense of $10.9 million for the Fiscal Year 2009 and $1.1 million for the Fiscal Year 2008. This $9.8 million increase was attributable to $9.5 million in interest expense, net of interest income and a decrease of $0.3 million in miscellaneous income. The increase in interest expense is primarily attributable to the increased debt that we borrowed to fund the ClinPhone acquisition. The $0.3 million decrease in miscellaneous income is due to a $7.6 million increase in miscellaneous expenses (including $3.0 million write-off related to a contract dispute, a $2.3 million write-off of certain impaired assets, and a $0.8 million loss related to an investment); partly offset by a $7.3 million increase in foreign exchange gains.
38
Table of Contents
Taxes
For the Fiscal Year 2009 and 2008, we had an effective income tax rate of 38.4% and 23.7%, respectively. The low tax rate for the Fiscal Year 2008 was primarily attributable to a reduction in deferred tax liabilities in that year due, in part, to a decrease in German statutory tax rates.
LIQUIDITY AND CAPITAL RESOURCES
Since our inception, we have financed our operations and growth with cash flow from operations, proceeds from the sale of equity securities, and, more recently, credit facilities to fund business acquisitions and working capital. Investing activities primarily reflect the costs of acquisitions and capital expenditures for information systems enhancements and leasehold improvements. As of June 30, 2010, we had cash, cash equivalents, and marketable securities of approximately $107.4 million.
DAYS SALES OUTSTANDING
Our operating cash flow is heavily influenced by changes in the levels of billed and unbilled receivables and deferred revenue. These account balances as well as days sales outstanding (“DSO”) in accounts receivable, net of deferred revenue, can vary based on contractual milestones and the timing and size of cash receipts. DSO was 49 days and 57 days at June 30, 2010 and June 30, 2009, respectively. The improvement in DSO was primarily due to improved collections. Accounts receivable, net of provision for losses on receivables, totaled $478.9 million ($229.9 million in billed accounts receivable and $249.0 million in unbilled accounts receivable) at June 30, 2010 and $481.3 million ($251.2 million in billed accounts receivable and $230.1 million in unbilled accounts receivable) at June 30, 2009. Deferred revenue was $261.1 million at June 30, 2010 and $266.5 million at June 30, 2009. DSO is calculated by adding the end-of-period balances for billed and unbilled account receivables, net of deferred revenue and the provision for losses on receivables, then dividing the resulting amount by the sum of total revenue plus investigator fees billed for the most recent quarter, and multiplying the resulting fraction by the number of days in the quarter.
CASH FLOWS
Net cash provided by operating activities for Fiscal Year 2010 totaled $157.7 million and was generated by net income of $41.5 million, a $61.9 million increase in accounts payable and other current liabilities, $60.3 million of non-cash charges for depreciation and amortization, $7.0 million of non-cash charges for stock-based compensation, and $4.0 million related to increases in tax liabilities. These sources of cash were offset, in part, by increases of $9.7 million in accounts receivable, a $5.7 million increase in other assets, and a $1.6 million increase in prepaid expenses and other current assets.
Net cash used in investing activities for Fiscal Year 2010 totaled $92.3 million, including $79.0 million of capital expenditures primarily for computer software and hardware, including a major upgrade to our ERP systems, and leasehold improvements and $13.7 million for the purchase of marketable securities in foreign government treasury certificates.
Net cash used in financing activities for Fiscal Year 2010 totaled $57.0 million, and consisted of $63.3 million of net repayments under lines of credit; offset, in part, by $6.8 million in proceeds related to the issuance of common stock in connection with our stock option and employee stock purchase plans.
Net cash provided by operating activities for Fiscal Year 2009 totaled $110.1 million and was generated by net income of $39.3 million and non-cash charges of $103.0 million, including $52.9 million of depreciation and amortization expense, a $23.5 million increase in deferred income taxes, $17.8 million in provisions for doubtful accounts, $7.3 million related to stock-based compensation, and $1.5 million of other charges, These sources of cash were offset by $32.2 million related to changes in operating assets and liabilities – comprised of a $30.4 million decrease in other current liabilities, a $46.3 million increase in other assets (related mainly to ClinPhone), and a $4.0 million decrease in long-term income taxes payable; offset, in part, by a $27.0 million decrease in billed and unbilled accounts receivable, net of deferred revenue, an $18.6 million decrease in prepaid expenses and other current assets, and a $2.9 million increase in accounts payable.
Net cash used in investing activities for Fiscal Year 2009 totaled $265.1 million and consisted primarily of $190.3 million used for acquisitions and $75.2 million related to purchases of property and equipment (primarily computer software and hardware, and leasehold improvements).
Net cash provided by financing activities for Fiscal Year 2009 totaled $211.2 million, and consisted of $206.4 million in borrowings under lines of credit and other arrangements, net of repayments, and $4.8 million from proceeds from the issuance of common stock in connection with the Company’s stock option and employee stock purchase plans.
39
Table of Contents
LINES OF CREDIT
2008 Credit Facility
On June 13, 2008, PAREXEL, certain subsidiaries of PAREXEL, JPMorgan Chase Bank, N.A., as Administrative Agent, J.P. Morgan Europe Limited, as London Agent, and the lenders party thereto (the “Lenders”) entered into an agreement for a credit facility (as amended and restated as of August 14, 2008 and as further amended by the first amendment thereto dated as of December 19, 2008, the “2008 Credit Facility”) in the principal amount of up to $315 million (collectively, the “Loan Amount”). The 2008 Credit Facility consists of an unsecured term loan facility and an unsecured revolving credit facility. Of the total principal amount, up to $150 million is made available through a term loan and up to $165 million is made available through a revolving credit facility. A portion of the revolving loan facility is available for swingline loans of up to $20 million to be made by JP Morgan Chase Bank, N.A. and for letters of credit. We may request the lenders to increase the 2008 Credit Facility by an additional amount of up to $50 million. Such increase may, but is not committed to, be provided.
Borrowings made under the 2008 Credit Facility bear interest, at our determination, at a rate based on the highest of prime, the federal funds rate plus .50% and the one-month Adjusted LIBOR Rate (as defined in the 2008 Credit Facility) plus 1.00% (such highest rate, the “Alternate Base Rate”) plus a margin (not to exceed a per annum rate of .75%) based on the Leverage Ratio (defined below), in which case it is a floating interest rate, or based on LIBOR or EURIBOR plus a margin (not to exceed a per annum rate of 1.75%) based on the Leverage Ratio, in which case the interest rate is fixed at the beginning of each interest period for the balance of the interest period. An interest period is typically one, two, three, or six months. The “Leverage Ratio” is a ratio of the consolidated total debt to consolidated net income before interest, taxes, depreciation and amortization (EBITDA). Loans outstanding under the 2008 Credit Facility may be prepaid at any time in whole or in part without premium or penalty, other than customary breakage costs, if any. The 2008 Credit Facility terminates and any outstanding loans under it mature on June 13, 2013.
Repayment of the principal borrowed under the revolving credit facility (other than a swingline loan) is due on June 13, 2013. Repayment of principal borrowed under the term loan facility is as follows:
| • | 5% of principal borrowed was repaid by June 30, 2009; |
| • | 20% of principal borrowed was repaid by June 30, 2010; |
| • | 20% of principal borrowed must be repaid during the one-year period from July 1, 2010 to June 30, 2011; |
| • | 25% of principal borrowed must be repaid during the one-year period from July 1, 2011 to June 30, 2012; and |
| • | 30% of principal borrowed must be repaid during the one-year period from July 1, 2012 to June 13, 2013. |
All payments of principal on the term loan facility made during each annual period described above are required to be made in equal quarterly installments and to be accompanied by accrued interest thereon. To the extent not previously paid, all borrowings under the term loan facility must be repaid on June 13, 2013. Swingline loans under the 2008 Credit Facility generally must be paid on the first date after such swingline loan is made that is the 15th or last day of a calendar month.
Interest due under the revolving credit facility (other than a swingline loan) and the term loan facility must be paid quarterly for borrowings with an interest rate determined at the Alternate Base Rate. Interest must be paid on the last day of the interest period selected by us for borrowings with an interest rate based on LIBOR or EURIBOR; provided that for interest periods of longer than three months, interest is required to be paid every three months. Interest under swingline loans is payable when principal is required to be repaid.
Our obligations under the 2008 Credit Facility may be accelerated upon the occurrence of an event of default under the 2008 Credit Facility, which includes customary events of default, including payment defaults, defaults in the performance of affirmative and negative covenants, the inaccuracy of representations or warranties, bankruptcy and insolvency related defaults, cross defaults to other material indebtedness, defaults relating to such matters as ERISA and judgments, and a change of control default. Our obligations under the 2008 Credit Facility are guaranteed by certain of our U.S. domestic subsidiaries, and we have guaranteed any obligations of any co-borrowers under the 2008 Credit Facility.
In connection with the 2008 Credit Facility, we agreed to pay a commitment fee on the term loan commitment, payable quarterly calculated as a percentage of the unused amount of the term loan commitments at a per annum rate of 0.30%, and a commitment fee on the revolving loan commitment calculated as a percentage of the unused amount of the revolving loan commitments at a per annum rate of up to 0.375% (based on the Leverage Ratio). To the extent there are letters of credit outstanding under the 2008 Credit Facility, we will pay to the Administrative Agent, for the benefit of the lenders, and to the issuing bank certain letter of credit fees, a fronting fee and additional charges. We also agreed to pay various fees to JPMorgan Chase Bank, N.A. or KeyBank or both.
40
Table of Contents
On August 14, 2008, we drew down approximately $78 million via the revolving credit facility available under the 2008 Credit Facility. This borrowing was our first drawdown under the 2008 Credit Facility, and the funds were used to repay all of our loans under the Amended and Restated Credit Agreement dated as of September 18, 2007, as amended, among the Company and the other parties thereto (the “2007 Credit Facility”), and to terminate all of our commitments thereunder. The proceeds of this borrowing were also used to pay certain fees and out-of-pocket expenses to the Lenders under the 2008 Credit Facility. On August 26, 2008, we drew down an additional amount of approximately $192 million under the 2008 Credit Facility in connection with the closing of the ClinPhone acquisition, pursuant to which we acquired all the issued shares of ClinPhone for approximately $172 million. The proceeds of the borrowing were also used to repay certain indebtedness of ClinPhone owed to HSBC Bank.
As of June 30, 2010, we had $212.5 million in principal amount of debt outstanding under the 2008 Credit Facility, consisting of $100.0 million of principal borrowed under the revolving credit facility and $112.5 million of principal under the term loan, and remaining borrowing availability of approximately $65.0 million under the revolving credit facility. Principal in the amount of $150 million under the 2008 Credit Facility has been hedged with an interest rate swap agreement and carries a fixed interest rate of 4.8%. As of June 30, 2010, our debt under the 2008 Credit Facility, including the $150 million of principal hedged with an interest swap agreement, carried an average interest rate of 4.0%.
The 2008 Credit Facility contains affirmative and negative covenants applicable to us and our subsidiaries, including financial covenants requiring us to comply with maximum leverage ratios, minimum interest coverage ratios, a minimum net worth test (which covenant allows for foreign translation adjustments of up to $50 million in connection with the calculations required under such covenant) and maximum capital expenditures requirements, as well as restrictions on liens, investments, indebtedness, fundamental changes, acquisitions, dispositions of property, making specified restricted payments (including stock repurchases exceeding an agreed to percentage of consolidated net income), and transactions with affiliates. As of June 30, 2010, we were in compliance with all covenants under the 2008 Credit Facility.
Additional Lines of Credit
We have a line of credit with RBS Nederland, NV in the amount of Euro 12.0 million. This line of credit is not collateralized, is payable on demand, and bears interest at an annual rate ranging between 2% and 4%. The line of credit may be revoked or canceled by the bank at any time at its discretion. We primarily entered into this line of credit to facilitate business transactions with the bank. At June 30, 2010, we had Euro 12.0 million available under this line of credit.
We have a line of credit with HSBC UK in the amount of 2.0 million pounds sterling. This line of credit was established by ClinPhone and is guaranteed by PAREXEL International Holding BV. The line is not secured and bears interest at an annual rate ranging between 2% and 4%. At June 30, 2010, we had 2.0 million pounds sterling available under this line of credit.
We have an unsecured line of credit with JP Morgan UK in the amount of $4.5 million that bears interest at an annual rate ranging between 2% and 4%. We primarily entered into this line of credit to facilitate business transactions with the bank. At June 30, 2010, we had $4.5 million available under this line of credit.
We have other foreign lines of credit with banks totaling $2.0 million. These lines of credit are used as overdraft protection and bear interest at annual rates ranging from 2% and 4%. The lines of credit are payable on demand. At June 30, 2010, we had $2.0 million available under these arrangements.
We have a cash pooling arrangement with RBS Nederland, NV. Pooling occurs when debit balances are offset against credit balances and the overall net position is used as a basis by the bank for calculating the overall pool interest amount. Each legal entity owned by PAREXEL and party to this arrangement remains the owner of either a credit (deposit) or a debit (overdraft) balance. Therefore, interest income is earned by legal entities with credit balances, while interest expense is charged to legal entities with debit balances. Based on the pool’s overall balance, the bank then (1) recalculates the overall interest to be charged or earned, (2) compares this amount with the sum of previously charged/earned interest amounts per account and (3) additionally pays/charges the difference. The gross overdraft balance related to this pooling arrangement was $91.0 million and $117.9 million at June 30, 2010 and June 30, 2009, respectively. However, on a net basis, we have surplus cash balances over all accounts for the respective periods.
We have financing agreements with a vendor to finance software purchases. The agreements carry four-year terms and bear annual interest rates ranging between 0% to 3%. As of June 30, 2010, the balance on the promissory notes issued in connection with the financing agreements was $2.9 million.
41
Table of Contents
FINANCING NEEDS
Our primary cash needs are for operating expenses, such as salaries and fringe benefits, hiring and recruiting, business development and facilities, business acquisitions, capital expenditures, and repayment of principal and interest on our borrowings. Our requirements for cash to pay principal and interest on our borrowings will increase significantly in future periods because we borrowed approximately $192 million under the 2008 Credit Facility in August 2008 to finance the acquisition of ClinPhone. Our only committed external source of funds is under our 2008 Credit Facility described above. Our principal source of cash is from the performance of services under contracts with our clients. If we were unable to generate new contracts with existing and new clients or if the level of contract cancellations increased, our revenue and cash flow would be adversely affected (see “Part II, Item 1A – Risk Factors” for further detail). Absent a material adverse change in the level of our new business bookings or contract cancellations, we believe that our existing capital resources together with cash flow from operations and borrowing capacity under existing lines of credit will be sufficient to meet our foreseeable cash needs over the next twelve months and on a longer term basis. Depending upon our revenue and cash flow from operations, it is possible that we will require external funds to repay amounts outstanding under our 2008 Credit Facility upon maturity in 2013.
We expect to continue to acquire businesses to enhance our service and product offerings, expand our therapeutic expertise, and/or increase our global presence. Depending on their size, any future acquisitions may require additional external financing, and we may from time to time seek to obtain funds from public or private issuances of equity or debt securities. We may be unable to secure such financing at all or on terms acceptable to us, as a result of our outstanding borrowings under the 2008 Credit Facility. In addition, under the terms of the 2008 Credit Facility, interest rates are fixed based on market indices at the time of borrowing and, depending upon the interest mechanism selected by us, may float thereafter. As a result, the amount of interest payable by us on our borrowings may increase if market interest rates change.
We made capital expenditures of approximately $79.0 million during the twelve months ended June 30, 2010, primarily for computer software (including the implementation of a large project accounting system), hardware, and leasehold improvements. We expect capital expenditures to total approximately $65 to $70 million in Fiscal Year 2011, primarily for computer software and hardware and leasehold improvements.
On September 9, 2004, our board of directors approved a stock repurchase program authorizing the purchase of up to $20.0 million of our common stock to be repurchased in the open market subject to market conditions. As of June 30, 2010, we had acquired 1,240,828 shares at a total cost of $14.0 million under this program. There were no repurchases made during the twelve months ended June 30, 2010.
DEBT, CONTRACTUAL OBLIGATIONS, CONTINGENT LIABILITIES AND GUARANTEES
The following table summarizes our contractual obligations at June 30, 2010:
| (in thousands) |
Less than 1 year |
1-3 years | 3-5 years | More than 5 years |
Total | ||||||||||
| Debt obligations (principal) |
$ | 32,071 | $ | 183,707 | $ | — | $ | — | $ | 215,778 | |||||
| Operating leases |
51,529 | 75,210 | 50,932 | 85,824 | 263,495 | ||||||||||
| Obligations under capital leases |
552 | 40 | — | — | 592 | ||||||||||
| Purchase obligations* |
27,851 | 18,638 | 1,159 | 232 | 47,880 | ||||||||||
| Total |
$ | 112,003 | $ | 277,595 | $ | 52,091 | $ | 86,056 | $ | 527,745 | |||||
|
* includes commitments to purchase software, hardware, and services. |
|||||||||||||||
The above table does not include approximately $56.3 million of potential tax liabilities from unrecognized tax benefits related to uncertain tax positions. See Note 14 to our consolidated financial statements included in this Annual Report on Form 10-K for more information.
We have letter-of-credit agreements with banks, totaling approximately $5.3 million, guaranteeing performance under various operating leases and vendor agreements. We also have an unsecured facility consisting of a term loan facility for $150 million and a revolving credit facility for $165 million with a group of lenders (including and managed by JPMorgan Chase Bank, N.A.) that is guaranteed by certain of our U.S. subsidiaries.
42
Table of Contents
OFF-BALANCE SHEET ARRANGEMENTS
We have no off-balance sheet arrangements that have or are reasonably likely to have a current or future effect on our financial condition, changes in financial condition, revenues or expenses, results of operations, liquidity, capital expenditures or capital resources that are material to our investors.
RESTRUCTURING PLAN
In October 2009, we adopted a plan to restructure our operations to reduce expenses, better align costs with current and future geographic sources of revenue, and improve operating efficiencies (the “2010 Restructuring Plan”). During Fiscal Year 2010, we recorded $16.8 million in restructuring charges in association with the 2010 Restructuring Plan, including approximately $5.2 million in costs related to the abandonment of certain property leases and $11.6 million in employee separation benefits associated with the elimination of 238 managerial and staff positions. We expect to see incremental savings in Fiscal Year 2011 resulting from this plan.
INFLATION
We believe the effects of inflation generally do not have a material adverse impact on our operations or financial condition.
RECENTLY ISSUED ACCOUNTING STANDARDS
In October 2009, the FASB issued ASU No. 2009-14, “Software (Topic 985): Certain Revenue Arrangements That Include Software Elements—a consensus of the FASB Emerging Issues Task Force (“EITF”)” (formerly EITF 09-3). ASU 2009-14 revises FASB ASC 985-605 to drop from its scope all tangible products containing both software and non-software components that operate together to deliver the products’ functions. It also amends the determination of how arrangement consideration should be allocated to deliverables in a multi-deliverable revenue arrangement. ASU 2009-14 is effective for us in Fiscal Year 2011. Early adoption is permitted with required transition disclosures based on the period of adoption. We do not expect the adoption of this guidance to have a material impact on our consolidated financial statements.
In October 2009, the FASB issued ASU No. 2009-13, “Revenue Recognition (Topic 605): Multiple-Deliverable Revenue Arrangements – a consensus of the FASB Emerging Issues Task Force” (formerly EITF 08-1), which amends the revenue recognition guidance for arrangements with multiple deliverables. ASU 2009-13 addresses how to determine whether an arrangement involving multiple deliverables contains more than one unit of accounting and how to allocate consideration to each unit of accounting in the arrangement. This ASU replaces all references to fair value as the measurement criteria with the term selling price and establishes a hierarchy for determining the selling price of a deliverable. It also eliminated the use of the residual value method for determining the allocation of arrangement consideration. ASU 2009-13 will be effective for us in Fiscal Year 2011. Early adoption is permitted with required transition disclosures based on the period of adoption. We do not expect the adoption of this guidance to have a material impact on our consolidated financial statements.
43
Table of Contents
| ITEM 7A. | QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK |
MARKET RISK
Market risk is the potential loss arising from adverse changes in market rates and prices, such as foreign currency rates, interest rates, and other relevant market rates or price changes. In the ordinary course of business, we are exposed to market risk resulting from changes in foreign currency exchange rates, and we regularly evaluate our exposure to such changes. Our overall risk management strategy seeks to balance the magnitude of the exposure and the costs and availability of appropriate financial instruments.
FOREIGN CURRENCY EXCHANGE RATES AND INTEREST RATES
We derived approximately 65.4% of our consolidated service revenue for the twelve months ended June 30, 2010 from operations outside of the U.S., including 24.1% denominated in Euros and 14.6% denominated in pounds sterling. We derived approximately 64.4% of our consolidated service revenue for the twelve months ended June 30, 2009 from operations outside of the U.S., including 24.3% denominated in Euros and 15.6% denominated in pounds sterling. We do not have significant operations in countries in which the economy is considered to be highly inflationary. Our financial statements are denominated in U.S. dollars. Accordingly, changes in exchange rates between foreign currencies and the U.S. dollar will affect the translation of financial results into U.S. dollars for purposes of reporting our consolidated financial results.
It is our policy to mitigate the risks associated with fluctuations in foreign exchange rates and in market rates of interest. Accordingly, we have instituted foreign currency hedging programs and an interest rate swap program. See Note 4 to our consolidated financial statements included in this Annual Report on Form 10-K for more information on our hedging programs and interest rate swap program.
As of June 30, 2010, the programs with derivatives designated as hedging instruments under ASC 815 were deemed effective and the notional values of the derivatives were approximately $213.8 million, including an interest rate swap agreement with a notional value of $150 million in connection with the borrowings under our 2008 Credit Facility. Under certain circumstances, such as the occurrence of significant differences between actual cash receipts and forecasted cash receipts, the ASC 815 programs could be deemed ineffective. In that event, the unrealized gains and losses related to these derivatives, and currently reported in accumulated other comprehensive income, would be recognized in earnings. As of June 30, 2010, the estimated amount that could be recognized in other income was a loss of approximately $7.0 million, net of tax.
As of June 30, 2010, the notional value of derivatives that were not designated as hedging instruments under ASC 815 was approximately $131.1 million. The potential change in the fair value of these foreign currency exchange contracts that would result from a hypothetical change of 10% in exchange rates would be approximately $13.1 million.
During the twelve months ended June 30, 2010 and 2009, we recorded foreign exchange losses of $2.5 million and foreign exchange gains of $6.1 million, respectively. We acknowledge our exposure to additional foreign exchange risk as it relates to assets and liabilities that are not part of the economic hedge program, but quantification of this risk is difficult to assess at any given point in time.
Our exposure to interest rate changes relates primarily to the amount of our short-term and long-term debt. Short-term debt was $32.1 million at June 30, 2010 and $32.1 million at June 30, 2009. Long-term debt was $183.7 million at June 30, 2010 and $247.1 million at June 30, 2009.
MARKETABLE SECURITIES
During the third quarter of Fiscal Year 2010, we purchased marketable securities in foreign government treasury certificates that are actively traded. We expect to hold these securities to maturity and have elected to account for them under the fair value option as outlined in ASC 825. As of June 30, 2010, the value of these marketable securities was $12.2 million. Since the counterparty is a stable sovereign and due to the relatively short terms of maturity (less than one year), we do not believe that these investments are at high risk of default. Nevertheless, these investments are still at risk for adverse changes in market rates and prices.
44
Table of Contents
| Item 8. | Financial Statements and Supplementary Data |
PAREXEL INTERNATIONAL CORPORATION
CONSOLIDATED STATEMENTS OF INCOME
(in thousands, except per share data)
| For the years ended June 30, | ||||||||||||
| 2010 | 2009 | 2008 | ||||||||||
| Service revenue |
$ | 1,131,039 | $ | 1,050,755 | $ | 964,283 | ||||||
| Reimbursement revenue |
204,836 | 196,126 | 198,687 | |||||||||
| Total revenue |
1,335,875 | 1,246,881 | 1,162,970 | |||||||||
| Costs and expenses: |
||||||||||||
| Direct costs |
704,567 | 675,063 | 629,399 | |||||||||
| Reimbursable out-of-pocket expenses |
204,836 | 196,126 | 198,687 | |||||||||
| Selling, general and administrative |
267,377 | 232,153 | 211,392 | |||||||||
| Depreciation |
49,943 | 43,373 | 33,005 | |||||||||
| Amortization |
10,377 | 9,555 | 4,681 | |||||||||
| Other (benefit) charge |
(1,144 | ) | 15,000 | — | ||||||||
| Restructuring charge (benefit) |
16,810 | (33 | ) | (860 | ) | |||||||
| Total costs and expenses |
1,252,766 | 1,171,237 | 1,076,304 | |||||||||
| Income from operations |
83,109 | 75,644 | 86,666 | |||||||||
| Interest income |
5,077 | 12,718 | 22,018 | |||||||||
| Interest expense |
(15,403 | ) | (23,948 | ) | (23,767 | ) | ||||||
| Other expense, net |
(9,608 | ) | (576 | ) | (251 | ) | ||||||
| Total other expense, net |
(19,934 | ) | (11,806 | ) | (2,000 | ) | ||||||
| Income before provision for income taxes |
63,175 | 63,838 | 84,666 | |||||||||
| Provision for income taxes |
21,633 | 24,531 | 20,026 | |||||||||
| Net income |
$ | 41,542 | $ | 39,307 | $ | 64,640 | ||||||
| Earnings per share: |
||||||||||||
| Basic |
$ | 0.72 | $ | 0.68 | $ | 1.16 | ||||||
| Diluted |
$ | 0.71 | $ | 0.68 | $ | 1.12 | ||||||
| Weighted average shares: |
||||||||||||
| Basic |
58,062 | 57,538 | 55,896 | |||||||||
| Diluted |
58,756 | 57,847 | 57,461 | |||||||||
The accompanying notes are an integral part of the consolidated financial statements.
45
Table of Contents
PAREXEL INTERNATIONAL CORPORATION
CONSOLIDATED BALANCE SHEETS
(in thousands, except share data)
| June 30, 2010 | June 30, 2009 | |||||||
| ASSETS |
||||||||
| Current assets: |
||||||||
| Cash and cash equivalents |
$ | 95,170 | $ | 96,352 | ||||
| Marketable securities |
12,243 | — | ||||||
| Billed and unbilled accounts receivable, net |
478,926 | 481,321 | ||||||
| Prepaid expenses |
32,084 | 24,636 | ||||||
| Deferred tax assets |
28,932 | 21,268 | ||||||
| Income taxes receivable |
— | 7,631 | ||||||
| Other current assets |
8,502 | 16,215 | ||||||
| Total current assets |
655,857 | 647,423 | ||||||
| Property and equipment, net |
187,888 | 170,486 | ||||||
| Goodwill |
248,226 | 247,612 | ||||||
| Other intangible assets, net |
87,114 | 98,799 | ||||||
| Non-current deferred tax assets |
7,193 | 15,385 | ||||||
| Long-term income taxes receivable |
17,737 | 21,308 | ||||||
| Other assets |
16,695 | 23,448 | ||||||
| Total assets |
$ | 1,220,710 | $ | 1,224,461 | ||||
| LIABILITIES AND STOCKHOLDERS’ EQUITY |
||||||||
| Current liabilities: |
||||||||
| Notes payable and current portion of long-term debt |
$ | 32,082 | $ | 32,090 | ||||
| Accounts payable |
34,353 | 31,648 | ||||||
| Deferred revenue |
261,080 | 266,453 | ||||||
| Accrued expenses |
35,994 | 34,937 | ||||||
| Accrued restructuring charges, current portion |
7,760 | 876 | ||||||
| Accrued employee benefits and withholdings |
87,606 | 59,638 | ||||||
| Current deferred tax liabilities |
15,977 | 18,110 | ||||||
| Income tax payable |
2,840 | — | ||||||
| Other current liabilities |
19,541 | 11,966 | ||||||
| Total current liabilities |
497,233 | 455,718 | ||||||
| Long-term debt, net of current portion |
183,707 | 247,083 | ||||||
| Non-current deferred tax liabilities |
32,235 | 44,446 | ||||||
| Long-term accrued restructuring charges, less current portion |
2,318 | 1,268 | ||||||
| Long-term income tax liabilities |
48,232 | 47,881 | ||||||
| Other liabilities |
17,430 | 13,320 | ||||||
| Total liabilities |
781,155 | 809,716 | ||||||
| Stockholders’ equity: |
||||||||
| Preferred stock – $.01 par value; shares authorized: 5,000,000; Series A junior participating preferred stock – 50,000 shares designated, none issued and outstanding |
— | — | ||||||
| Common stock – $.01 par value; shares authorized: 75,000,000 at June 30, 2010 and 2009; shares issued and outstanding: 58,433,717 and 57,782,931 at June 30, 2010 and 2009, respectively |
578 | 572 | ||||||
| Additional paid-in capital |
233,677 | 219,849 | ||||||
| Retained earnings |
246,734 | 205,192 | ||||||
| Accumulated other comprehensive (loss) income |
(41,434 | ) | (10,868 | ) | ||||
| Total stockholders’ equity |
439,555 | 414,745 | ||||||
| Total liabilities and stockholders’ equity |
$ | 1,220,710 | $ | 1,224,461 | ||||
The accompanying notes are an integral part of the consolidated financial statements.
46
Table of Contents
PAREXEL INTERNATIONAL CORPORATION
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY
(in thousands)
| Common Stock | ||||||||||||||||||||||||||
| Number of Shares |
Par Value |
Additional Paid-in Capital |
Retained Earnings |
Accumulated Other Comprehensive Income (Loss) |
Total Stockholders’ Equity |
Comprehensive Income (Loss) |
||||||||||||||||||||
| Balance at June 30, 2007 |
55,131 | $ | 551 | $ | 191,573 | $ | 102,564 | $ | 21,928 | $ | 316,616 | |||||||||||||||
| Shares issued under stock option/employee stock purchase plans |
1,641 | 16 | 12,597 | 12,613 | ||||||||||||||||||||||
| Stock-based compensation |
5,240 | 5,240 | ||||||||||||||||||||||||
| Unrealized loss on derivative instruments, net of taxes |
(913 | ) | (913 | ) | (913 | ) | ||||||||||||||||||||
| Foreign currency translation adjustment |
31,214 | 31,214 | 31,214 | |||||||||||||||||||||||
| Cumulative effect of change in accounting upon adoption of FIN 48 |
(1,324 | ) | (1,324 | ) | ||||||||||||||||||||||
| Cumulative effect of change in accounting of Synchron investment |
5 | 5 | ||||||||||||||||||||||||
| Net income |
64,640 | 64,640 | 64,640 | |||||||||||||||||||||||
| Total comprehensive income |
$ | 94,941 | ||||||||||||||||||||||||
| Balance at June 30, 2008 |
56,772 | $ | 567 | $ | 209,410 | $ | 165,885 | $ | 52,229 | $ | 428,091 | |||||||||||||||
| Shares issued under stock option/employee stock purchase plans |
575 | 6 | 4,837 | 4,843 | ||||||||||||||||||||||
| Stock-based compensation |
7,313 | 7,313 | ||||||||||||||||||||||||
| Vested restricted stock, net of shares surrendered for tax |
436 | (1 | ) | (1,711 | ) | (1,712 | ) | |||||||||||||||||||
| Unrealized gain on derivative instruments, net of taxes |
352 | 352 | 352 | |||||||||||||||||||||||
| Foreign currency translation adjustment |
(63,449 | ) | (63,449 | ) | (63,449 | ) | ||||||||||||||||||||
| Net income |
39,307 | 39,307 | 39,307 | |||||||||||||||||||||||
| Total comprehensive loss |
$ | (23,790 | ) | |||||||||||||||||||||||
| Balance at June 30, 2009 |
57,783 | $ | 572 | $ | 219,849 | $ | 205,192 | $ | (10,868 | ) | $ | 414,745 | ||||||||||||||
| Shares issued under stock option/employee stock purchase plans |
651 | 6 | 6,811 | 6,817 | ||||||||||||||||||||||
| Stock-based compensation |
7,017 | 7,017 | ||||||||||||||||||||||||
| Unrealized loss on derivative instruments, net of taxes |
(6,889 | ) | (6,889 | ) | (6,889 | ) | ||||||||||||||||||||
| Foreign currency translation adjustment |
(23,677 | ) | (23,677 | ) | (23,677 | ) | ||||||||||||||||||||
| Net income |
41,542 | 41,542 | 41,542 | |||||||||||||||||||||||
| Total comprehensive income |
$ | 10,976 | ||||||||||||||||||||||||
| Balance at June 30, 2010 |
58,434 | $ | 578 | $ | 233,677 | $ | 246,734 | $ | (41,434 | ) | $ | 439,555 | ||||||||||||||
The accompanying notes are an integral part of the consolidated financial statements.
47
Table of Contents
PAREXEL INTERNATIONAL CORPORATION
CONSOLIDATED STATEMENTS OF CASH FLOWS
(in thousands)
| For the years ended June 30, | ||||||||||||
| 2010 | 2009 | 2008 | ||||||||||
| Cash flow from operating activities: |
||||||||||||
| Net income |
$ | 41,542 | $ | 39,307 | $ | 64,640 | ||||||
| Adjustments to reconcile net income to net cash provided by operating activities: |
||||||||||||
| Depreciation and amortization |
60,320 | 52,928 | 37,686 | |||||||||
| Stock-based compensation |
7,017 | 7,313 | 5,240 | |||||||||
| (Gain) loss on disposal of assets |
(719 | ) | 578 | (136 | ) | |||||||
| Deferred income taxes |
(11,168 | ) | 23,547 | (10,004 | ) | |||||||
| Provision for losses on receivables, net |
2,566 | 17,759 | 1,743 | |||||||||
| Changes in assets and liabilities, net of effects from acquisitions: |
||||||||||||
| Accounts receivable |
(20,873 | ) | (41,294 | ) | (128,234 | ) | ||||||
| Prepaid expenses and other current assets |
(1,746 | ) | 18,616 | (2,951 | ) | |||||||
| Other assets |
(4,997 | ) | (46,335 | ) | (17,472 | ) | ||||||
| Accounts payable |
3,908 | 2,863 | 7,786 | |||||||||
| Deferred revenue |
8,567 | 68,316 | 38,082 | |||||||||
| Other current liabilities |
52,354 | (30,177 | ) | 2,710 | ||||||||
| Long-term income taxes payable, net of long-term income taxes receivable |
15,165 | (3,968 | ) | 19,740 | ||||||||
| Other liabilities |
5,760 | 654 | (3,102 | ) | ||||||||
| Net cash provided by operating activities |
157,696 | 110,107 | 15,728 | |||||||||
| Cash flow from investing activities: |
||||||||||||
| Purchases of marketable securities |
(13,724 | ) | — | (49,000 | ) | |||||||
| Proceeds from sale of marketable securities |
— | — | 49,000 | |||||||||
| Purchases of property and equipment |
(78,959 | ) | (75,181 | ) | (67,067 | ) | ||||||
| Acquisition of businesses |
(32 | ) | (190,250 | ) | (55,388 | ) | ||||||
| Proceeds from sale of assets |
394 | 343 | 1,194 | |||||||||
| Net cash used in investing activities |
(92,321 | ) | (265,088 | ) | (121,261 | ) | ||||||
| Cash flow from financing activities: |
||||||||||||
| Proceeds from issuance of common stock |
6,817 | 4,843 | 12,613 | |||||||||
| Borrowings under lines of credit |
52,000 | 382,961 | 69,000 | |||||||||
| Repayments under lines of credit |
(113,000 | ) | (175,461 | ) | (32,813 | ) | ||||||
| (Repayments) borrowings under long-term debt, net |
(2,377 | ) | 581 | 2,344 | ||||||||
| Tax withholding payments from restricted stock surrenders |
— | (1,712 | ) | — | ||||||||
| Net cash (used in) provided by financing activities |
(56,560 | ) | 211,212 | 51,144 | ||||||||
| Effect of exchange rate changes on cash and cash equivalents |
(9,997 | ) | (11,796 | ) | 9,630 | |||||||
| Net (decrease) increase in cash and cash equivalents |
(1,182 | ) | 44,434 | (44,759 | ) | |||||||
| Cash and cash equivalents at beginning of year |
96,352 | 51,918 | 96,677 | |||||||||
| Cash and cash equivalents at end of year |
$ | 95,170 | $ | 96,352 | $ | 51,918 | ||||||
| Supplemental disclosures of cash flow information |
||||||||||||
| Net cash paid during year for: |
||||||||||||
| Interest paid |
$ | 14,942 | $ | 23,547 | $ | 25,729 | ||||||
| Income taxes, net of refunds |
$ | 21,599 | $ | 35,640 | $ | 32,289 | ||||||
| Supplemental disclosures of investing activities: |
||||||||||||
| Fair value of assets acquired and goodwill |
$ | 32 | $ | 241,617 | $ | 70,505 | ||||||
| Liabilities assumed |
— | (51,367 | ) | (15,117 | ) | |||||||
| Cash paid for acquisitions |
$ | 32 | $ | 190,250 | $ | 55,388 | ||||||
The accompanying notes are an integral part of the consolidated financial statements.
48
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
NOTE 1. DESCRIPTION OF BUSINESS
PAREXEL is a leading biopharmaceutical services company, providing a broad range of expertise in clinical research, medical communications services, consulting and informatics, and advanced technology products and services to the worldwide pharmaceutical, biotechnology, and medical device industries. Our primary objective is to provide solutions for managing the biopharmaceutical product lifecycle with the goal of reducing the time, risk, and cost associated with the development and commercialization of new therapies. Since our incorporation in 1983, we have developed significant expertise in processes and technologies supporting this strategy. Our product and service offerings include: clinical trials management, data management, biostatistical analysis, medical communications, clinical pharmacology, patient recruitment, regulatory and product development consulting, health policy and reimbursement, performance improvement, industry training and publishing, medical imaging services, ClinPhone RTSM, CTMS, EDC, web-based portals, systems integration, patient diary applications, and other drug development consulting services. We believe that our comprehensive services, depth of therapeutic area expertise, global footprint and related access to patients, and sophisticated information technology, along with our experience in global drug development and product launch services, represent key competitive strengths.
NOTE 2. SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Basis of Presentation and Principles of Consolidation
The consolidated financial statements include the accounts of PAREXEL International Corporation, our wholly-owned and majority-owned subsidiaries. All significant inter-company accounts and transactions have been eliminated.
Use of Estimates
We prepare our financial statements in conformity with U.S. generally accepted accounting principles which require us to make estimates and assumptions that affect the amounts reported in the financial statements. Estimates are used in accounting for, among other items, revenue recognition, allowance for credit losses on receivables, valuation of derivative instruments, periodic impairment reviews of goodwill and intangible assets, and the valuation of long-term assets. Our estimates are based on the facts and circumstances available at the time estimates are made, historical experience, risk of loss, general economic conditions, trends, and assessments of the probable future outcomes of these matters. Actual results could differ from those estimates. Estimates and assumptions are reviewed periodically, and the effects of changes, if any, are reflected in the statement of operations in the period in which they are determined.
Fair Values of Financial Instruments
The fair value of our cash and cash equivalents, marketable securities, accounts receivable, accounts payable, and debt approximates the carrying value of these financial instruments because of the short-term nature of any maturities. We determine the estimated fair values of other financial instruments, including equity and risk management instruments, using available market information and valuation methodologies, primarily discounted cash flow analysis or input from independent investment bankers.
Revenue Recognition
We derive revenue from the delivery of service or software solutions to clients in the worldwide pharmaceutical, biotechnology, and medical device industries. In general, we recognize revenue when all of the following conditions are satisfied: (1) there is persuasive evidence of an arrangement; (2) the service offering has been delivered to the client; (3) the collection of fees is probable; and (4) the amount of fees to be paid by the client is fixed or determinable. Revenue recognition treatment of each business segment is described below.
Deferred revenue represents amounts billed or cash received in advance of revenue recognized. Unbilled accounts receivable represent revenue recognized in excess of amounts billed.
Reimbursable out-of-pocket expenses are reflected in our Consolidated Statements of Income under “Reimbursement revenue” and “Reimbursable out-of-pocket expenses.”
As is customary in our industry, we routinely subcontract on behalf of our clients with independent physician investigators in connection with clinical trials. The related investigator fees are not reflected in our Service revenue, Reimbursement revenue, Reimbursable out-of-pocket expenses, or Direct costs, since such fees are reimbursed by clients on a “pass through basis,” without risk or reward to us. The amounts of these investigator fees were $200.9 million, $189.2 million, and $167.0 million for the fiscal years ended June 30, 2010, 2009, and 2008, respectively.
49
Table of Contents
CRS and PCMS Service Revenues
Service revenues in our CRS and PCMS businesses are derived principally from fee-for-service or fixed-price executory contracts, which typically involve competitive bid awards and multi-year terms. Client billing schedules and payment arrangements are prescribed under negotiated contract terms. Contract provisions do not provide for rights of return or refund, but normally include rights of cancellation with notice, in which case services delivered through the cancellation date are due and payable by the client, including certain costs to conclude the trial or study.
Revenue from fee-for-service contracts generally is recognized as units of output are delivered. Revenue on fixed-price contracts is measured by applying a proportional performance model using output units, such as site or investigator recruitment, patient enrollment, data management, or other deliverables common, for example, to our Phase II-III/PACE business. Performance-based output units are pre-defined in contracts and revenue is recognized based upon actual units of completion. Revenue related to changes in contract scope, which are subject to client approval, is recognized when realization is assured and amounts are reasonably determinable.
Our client arrangements generally involve multiple service deliverables, where bundled service deliverables are accounted for in accordance with ASC 605-25, “Multiple-Element Arrangements.” We have determined that each of our service deliverables has standalone value and have established objective evidence of fair value for each of our service deliverables based on the price charged when sold to other similar customers. Accordingly, revenues are recognized for the separate elements upon delivery of actual units of completion and when all other revenue recognition criteria are met.
Perceptive Service Revenue
Service revenue is derived principally from the delivery of software solutions through our Perceptive business segment. Software solutions include Clinphone RTSM, CTMS, and EDC.
Within Perceptive’s Clinphone RTSM business, we offer selected software solutions through a hosted application delivered through a standard web-browser. We recognize revenue from application hosting services in accordance with ASC 985-605, “Revenue Recognition in the Software Industry” and ASC 605-25, “Multiple-Element Arrangements.” Revenue resulting from these hosting services consists of three stages: set-up (client specification and workflow), hosting and support services, and closeout reporting. We recognize revenue from all stages of a project ratably over the hosting period, including customary and expected extensions. Fees charged and costs incurred in the set-up stage are deferred until the start of the hosting period and are amortized and recognized ratably over the estimated hosting period. Deferred costs include incremental direct costs and certain indirect costs associated with the trial and application setup. These costs include salary and benefits associated with direct labor costs incurred during trial setup, as well as third-party subcontract fees and other contract labor costs. In the event of a contract cancellation by a client, all deferred revenue is recognized and all deferred setup costs are expensed. To the extent that termination-related fees are payable under the contract, such fees are recognized in the period of termination.
Within the CTMS operating unit of the Perceptive business segment, software revenue is recognized on a proportional performance basis in accordance with ASC 985-605, “Revenue Recognition in the Software Industry” and the relevant guidance provided by ASC 605-35, “Construction-Type and Certain Production-Type Contracts,” due to the significant nature of customization of each project.
Within the EDC operating unit of the Perceptive business segment, revenue is recognized ratably over the contract service period.
Cash and Cash Equivalents
We consider all highly liquid investments purchased with original maturities of 90 days or less to be cash equivalents.
Marketable Securities
We account for investments in debt and equity securities in accordance with ASC 320, “Investments – Debt and Equity Securities.” Our short-term investments are classified as held-to-maturity based on our positive intent and ability to hold the securities to maturity. Securities held at June 30, 2010 are foreign government treasury certificates with original maturities over 90 days but less than one year and we have elected to account for these investments under the fair value option under ASC 825 to better represent the value of our assets. Income related to these securities is reported as a component of interest income in our Consolidated Statements of Income. As of June 30, 2010, the value of these marketable securities was $12.2 million.
50
Table of Contents
Concentration of Credit Risk
Financial instruments, which may potentially expose PAREXEL to concentrations of credit risk, include trade accounts receivable. However, we maintain reserves for potential credit losses based on historic collectability and specific identification of potential problem accounts. Such losses, in the aggregate, have not exceeded management expectations. In Fiscal Year 2010, our largest client accounted for 8% of consolidated service revenue. In each of Fiscal Years 2009 and 2008, our largest client accounted for 9% of consolidated service revenue.
We have approximately 5 different counterparties in our derivative contracts, which include interest rate swaps and foreign currency hedges. Each of these counterparties is in the financial services industry and is subject to the credit risks inherent to that industry.
Our marketable securities are invested in foreign government treasury certificates. Since the counterparty is a stable sovereign entity and due to the relatively short terms of maturity (less than one year), we do not believe that these investments are at high risk of default. Nevertheless, these investments are still at risk for adverse changes in market rates and prices.
Billed Accounts Receivable, Unbilled Accounts Receivable and Deferred Revenue
Billed accounts receivable represent amounts for which invoices have been sent to clients. Unbilled accounts receivable represent amounts recognized as revenue for which invoices have not yet been sent to clients. Deferred revenue represents amounts billed or payments received for which revenue has not yet been earned. We maintain a provision for losses on receivables based on historical collectability and specific identification of potential problem accounts.
Property and Equipment
Property and equipment is stated at cost. Depreciation is provided using the straight-line method based on estimated useful lives of 40 years for buildings, 3 to 8 years for computer software and hardware, and 5 years for office furniture, fixtures and equipment. Leasehold improvements are amortized over the lesser of the estimated useful lives of the improvements or the remaining lease term. Charges resulting from the amortization of assets recorded under capital leases are included with depreciation expense. Repair and maintenance costs are expensed as incurred.
Development of Software for Internal Use
PAREXEL accounts for the costs of computer software developed or obtained for internal use in accordance with ASC 350-40, “Internal-Use Software.” We capitalize costs of materials, consultants and payroll and payroll related costs for employees incurred in developing internal-use software. These costs are included in computer software in Note 6 below. Costs incurred during the preliminary project and post-implementation stages are charged to expense.
Research and Development Costs
The Company incurs ongoing research and development costs related to core technologies used internally as well as software and technology sold externally. Unless eligible for capitalization, these costs are expensed as incurred. Research and development expense was $21.0 million, $21.4 million, and $9.2 million in Fiscal Years 2010, 2009, and 2008, respectively, and is included in selling, general and administrative expenses in the consolidated statements of income.
Goodwill
PAREXEL follows the provisions of ASC 350, “Intangibles – Goodwill and Other.” Under this statement, goodwill as well as certain other intangible assets, determined to have an indefinite life, are not amortized. Instead, these assets are evaluated for impairment at least annually or more frequently if an event occurs or circumstances change that would more likely than not reduce the carrying value of the reporting unit below its fair value. Fair values are established primarily using a discounted cash flow methodology, which is based on strategic business plans and long-term forecasts. We have performed our annual impairment test, with no evidence of impairment of our goodwill balance for Fiscal Years 2010 and 2009.
51
Table of Contents
The changes in the carrying amount of goodwill balances for Fiscal Years 2010 and 2009 were as follows (in thousands):
| Carrying amount as of June 30, 2008 |
$ | 147,664 | ||
| FY 2009 |
||||
| ClinPhone acquisition |
117,394 | |||
| Effect of changes in exchange rates used for translation |
(17,446 | ) | ||
| Carrying amount as of June 30, 2009 |
$ | 247,612 | ||
| FY 2010 |
||||
| ClinPhone acquisition adjustments |
7,347 | |||
| Effect of changes in exchange rates used for translation |
(6,734 | ) | ||
| Carrying amount as of June 30, 2010 |
$ | 248,226 | ||
During Fiscal Year 2010, ClinPhone acquisition adjustments reflect final purchase accounting adjustments recorded in the first quarter, including the finalization of tax accounts.
PAREXEL records goodwill to the business segment affected by the transaction; balances at June 30, 2010 were:
| Goodwill by segment (in thousands): |
|||
| Clinical Research Services |
$ | 121,340 | |
| PAREXEL Consulting and MedCom Services |
4,449 | ||
| Perceptive Informatics, Inc. |
122,437 | ||
| Total Goodwill |
$ | 248,226 | |
Intangible Assets
As of June 30, 2010 intangible assets consisted of the following (in thousands):
| Intangible Asset |
Weighted Average Useful Life (years) |
Cost | Accumulated Amortization/Effect of Exchange Rate Changes |
Net | |||||||
| Customer relationships & backlog |
12.7 | $ | 79,560 | $ | 27,058 | $ | 52,502 | ||||
| Non-compete agreements |
2.9 | 1,688 | 1,688 | — | |||||||
| Technology & other intangibles |
8.0 | 26,330 | 10,071 | 16,259 | |||||||
| Tradename* |
NA | 22,158 | 3,805 | 18,353 | |||||||
| Total intangible assets |
$ | 129,736 | $ | 42,622 | $ | 87,114 | |||||
| * | The tradename acquired in the ClinPhone acquisition has an indefinite useful life. |
As of June 30, 2009, intangible assets consisted of the following (in thousands):
| Intangible Asset |
Weighted Average Useful Life (years) |
Cost | Accumulated Amortization/Effect of Exchange Rate Changes |
Net | |||||||
| Customer relationships & backlog |
12.7 | $ | 79,560 | $ | 21,731 | $ | 57,829 | ||||
| Non-compete agreements |
2.9 | 1,688 | 1,176 | 512 | |||||||
| Technology & other intangibles |
8.0 | 26,330 | 5,330 | 21,000 | |||||||
| Tradename* |
NA | 22,158 | 2,700 | 19,458 | |||||||
| Total intangible assets |
$ | 129,736 | $ | 30,937 | $ | 98,799 | |||||
| * | The tradename acquired in the ClinPhone acquisition has an indefinite useful life. |
52
Table of Contents
The changes in the carrying amounts of intangible assets for Fiscal Years 2010 and 2009 were as follows (in thousands):
| Carrying amount as of June 30, 2008 |
$ | 34,608 | ||
| FY 2009 |
||||
| ClinPhone acquisition |
90,571 | |||
| Amortization |
(9,555 | ) | ||
| Effect of changes in rates used for translation |
17,783 | |||
| Carrying amount as of June 30, 2009 |
$ | 98,799 | ||
| FY 2010 |
||||
| Amortization |
(10,376 | ) | ||
| Effect of changes in rates used for translation |
(1,309 | ) | ||
| Carrying amount as of June 30, 2010 |
$ | 87,114 | ||
Estimated amortization expense for the next five years is as follows (in thousands):
| FY 2011 | FY 2012 | FY 2013 | FY 2014 | FY 2015 | ||||
| $9,451 | $8,238 | $7,860 | $7,294 | $6,445 |
Income Taxes
Deferred tax assets and liabilities are recorded for the expected future tax consequences of temporary differences between the carrying amounts and the tax bases of assets and liabilities. Deferred tax assets are recognized for the estimated future tax benefits of deductible temporary differences and tax operating loss and credit carryforwards and are presented net of valuation allowances. Valuation allowances are established in jurisdictions where it is more likely than not that the benefits of the associated deferred tax assets will not be realized. Deferred income tax expense represents the change in the net deferred tax asset and liability balances. Interest and penalties are recognized as a component of income tax expense.
Foreign Currency
Assets and liabilities of PAREXEL’s international operations are translated into U.S. dollars at exchange rates that are in effect on the balance sheet date and equity accounts are translated at historical exchange rates. Income and expense items are translated at average exchange rates in effect during the year. Translation adjustments are accumulated in other comprehensive income (loss) as a separate component of stockholders’ equity in the consolidated balance sheet. Transaction gains and losses are included in other income, net in the consolidated statements of operations. Transaction (losses) gains were $(2.5) million, $6.1 million, and $(1.3) million in Fiscal Years 2010, 2009, and 2008, respectively.
Earnings Per Share
Earnings per share has been calculated in accordance with ASC 260, “Earnings per Share.” Basic earnings per share is computed by dividing net income for the period by the weighted average number of common shares outstanding during the period. Diluted earnings per share is computed by dividing net income by the weighted average number of common shares plus the dilutive effect of outstanding stock options and shares issuable under the employee stock purchase plan. We do not have any participating securities outstanding nor do we have more than one class of common stock.
Recently Issued Accounting Standards
In October 2009, the FASB issued ASU No. 2009-14, “Software (Topic 985): Certain Revenue Arrangements That Include Software Elements—a consensus of the FASB Emerging Issues Task Force (“EITF”)” (formerly EITF 09-3). ASU 2009-14 revises FASB ASC 985-605 to drop from its scope all tangible products containing both software and non-software components that operate together to deliver the products’ functions. It also amends the determination of how arrangement consideration should be allocated to deliverables in a multi-deliverable revenue arrangement. ASU 2009-14 is effective for us in Fiscal Year 2011. Early adoption is permitted with required transition disclosures based on the period of adoption. We do not expect the adoption of this guidance to have a material impact on our consolidated financial statements.
In October 2009, the FASB issued ASU No. 2009-13, “Revenue Recognition (Topic 605): Multiple-Deliverable Revenue Arrangements – a consensus of the FASB Emerging Issues Task Force” (formerly EITF 08-1), which amends the revenue recognition guidance for arrangements with multiple deliverables. ASU 2009-13 addresses how to determine whether an arrangement involving multiple deliverables contains more than one unit of accounting and how to allocate consideration to each unit of accounting in the arrangement. This ASU replaces all references to fair value as the measurement criteria with
53
Table of Contents
the term selling price and establishes a hierarchy for determining the selling price of a deliverable. It also eliminated the use of the residual value method for determining the allocation of arrangement consideration. ASU 2009-13 will be effective for us in Fiscal Year 2011. Early adoption is permitted with required transition disclosures based on the period of adoption. We do not expect the adoption of this guidance to have a material impact on our consolidated financial statements.
NOTE 3. ACQUISITIONS
We account for our acquisitions using the purchase method in accordance with ASC 805, “Business Combinations.” The results of operations of each acquisition have been included in the accompanying consolidated financial statements as of the dates of the acquisition.
ClinPhone – On August 14, 2008, we acquired ClinPhone plc (“ClinPhone”), a company traded on the London Stock Exchange, for approximately $190 million. By combining ClinPhone with our Perceptive Informatics segment, Perceptive is now one of the industry’s largest providers of telecommunications and web-based (“eClinical”) technologies for clinical research. The combined business offers access to a broad array of eClinical technologies and resources, providing clients and service providers with the benefits of an extensive line of products and services throughout the entire clinical development lifecycle.
The components of the purchase price allocation were as follows (in thousands):
| Purchase Price: | Cash paid, net of cash acquired |
$ | 185,306 | |||
| Transaction costs |
4,927 | |||||
| Total |
$ | 190,233 | ||||
| Allocations: | Fair value of assets acquired |
|||||
| Accounts receivable |
$ | 18,416 | ||||
| Other current assets |
2,236 | |||||
| Property and equipment |
12,796 | |||||
| Goodwill |
124,722 | |||||
| Tradename |
22,158 | |||||
| In-process research and development |
224 | |||||
| Other intangible assets |
68,413 | |||||
| Liabilities assumed |
||||||
| Accounts payable |
(8,628 | ) | ||||
| Current liabilities |
(14,693 | ) | ||||
| Deferred revenue |
(478 | ) | ||||
| Other liabilities |
(34,933 | ) | ||||
| Net assets acquired |
$ | 190,233 | ||||
APEX – In September 2007, we acquired a majority of the outstanding shares of Taiwan-based APEX International Clinical Research Co., Ltd. (“APEX”) and completed the acquisition of all the outstanding shares of APEX in November 2007 for a total of approximately $55 million. The acquisition strengthened our global capabilities, providing clients with a wide range of clinical research service offerings throughout the Asia-Pacific region, including mainland China, Hong Kong, India, Taiwan, Singapore, Indonesia, South Korea, Malaysia, Thailand, the Philippines, New Zealand, and Australia.
The components of the purchase price allocation were as follows (in thousands):
| Purchase Price: | Cash paid, net of cash acquired |
$ | 53,427 | |||
| Transaction costs |
1,984 | |||||
| Total |
$ | 55,411 | ||||
| Allocations: | Fair value of assets acquired |
|||||
| Accounts receivable |
$ | 4,010 | ||||
| Other current assets |
860 | |||||
| Property and equipment |
2,740 | |||||
| Goodwill |
52,000 | |||||
| Other intangible assets |
10,918 | |||||
| Liabilities assumed |
||||||
| Accounts payable |
(750 | ) | ||||
| Current liabilities |
(10,042 | ) | ||||
| Deferred revenue |
(4,325 | ) | ||||
| Net assets acquired |
$ | 55,411 | ||||
54
Table of Contents
NOTE 4 – DERIVATIVES
It is our policy to mitigate the risks associated with fluctuations in foreign exchange rates and in market rates of interest. Accordingly, we have instituted foreign currency hedging programs and an interest rate swap program that are accounted for in accordance with ASC 815, “Derivatives and Hedging.”
| • | Our foreign denominated intercompany debt and accounts receivable hedging program is a cash flow hedge program designed to minimize foreign currency volatility. The objective of the program is to reduce variability of cash flows with respect to forecasted billing for services provided outside of the service contract host currency and the foreign exchange exposure related to payment of invoices for services provided in executing the customer contract. We primarily utilize forward exchange contracts and purchased currency options with maturities of no more than 12 months that qualify as cash flow hedges. These are intended to offset the effect of exchange rate fluctuations as services are performed and billed, and are generally expected to be reclassified to earnings in the next 12 months as the underlying transactions occur. |
| • | Under our interest rate hedging program, we swap the difference between fixed and variable interest amounts calculated by reference to an agreed-upon notional principal amount, at specified intervals. The objective of this program is to reduce the variability of cash flows related to fluctuations in market rates of interest. We initially entered into swap agreements for intervals of up to 3 years, of which no amounts are expected to come due in the next 12 months. |
Occasionally, we enter into other foreign currency exchange contracts to offset the impact of currency fluctuations for other currencies and intercompany billings. These hedges include cash flow hedges similar to those described above and other derivative instruments, but may involve other denominations or counterparties. These contracts are not accounted for as hedges in accordance with ASC 815.
The following table presents the notional amounts and fair values of our derivatives as of June 30, 2010 and June 30, 2009 (in thousands). All asset and liability amounts are reported in other current assets and other current liabilities.
| June 30, 2010 | June 30, 2009 | |||||||||||||
| Notional Amount |
Asset (Liability) |
Notional Amount |
Asset (Liability) |
|||||||||||
| Derivatives designated as hedging instruments under ASC 815 |
| |||||||||||||
| Interest rate contracts |
$ | 150,000 | $ | (5,062 | ) | $ | 150,000 | $ | (5,381 | ) | ||||
| Foreign exchange contracts |
63,849 | (4,996 | ) | 54,459 | 5,584 | |||||||||
| Total designated derivatives |
$ | 213,849 | $ | (10,058 | ) | $ | 204,459 | $ | 203 | |||||
| Derivatives not designated as hedging instruments under ASC 815 |
||||||||||||||
| Cross-currency interest rate swap contracts |
$ | 41,969 | $ | (4,161 | ) | $ | 48,256 | $ | (3,736 | ) | ||||
| Foreign exchange contracts |
89,138 | (1,027 | ) | 148,830 | 5,500 | |||||||||
| Total non-designated derivatives |
$ | 131,107 | $ | (5,188 | ) | $ | 197,086 | $ | 1,764 | |||||
| Total derivatives |
$ | 344,956 | $ | (15,246 | ) | $ | 401,545 | $ | 1,967 | |||||
We record the effective portion of any change in the fair value of derivatives designated as hedging instruments under ASC 815 to other comprehensive income on the balance sheet, net of deferred taxes and any ineffective portion to other income (expense) on the income statement. The amounts recognized for the twelve months ended June 30, 2010 and 2009 in other comprehensive income are presented below (in thousands):
| Twelve Months Ended | ||||||||
| June 30, 2010 | June 30, 2009 | |||||||
| Derivatives designated as hedging instruments under ASC 815 |
| |||||||
| Interest rate contracts, net |
$ | 208 | $ | (3,031 | ) | |||
| Foreign exchange contracts, net |
(7,097 | ) | 3,379 | |||||
| Total designated derivatives |
$ | (6,889 | ) | $ | 348 | |||
During Fiscal Years 2010 and 2009, we reclassified approximately $0.4 million into other expense and $0.4 million into other income, respectively, as a result of contract settlements. During both periods, there were no amounts recorded to reflect ineffective portions of any hedges. The estimated net amount of the existing losses that are expected to be reclassified into earnings within the next twelve months is $7.4 million.
55
Table of Contents
The change in the fair value of derivatives not designated as hedging instruments under ASC 815 is recorded to other expense, net on the income statement. The amounts recognized for the twelve months ended June 30, 2010 and 2009 are presented below (in thousands):
| Twelve Months Ended | ||||||||
| June 30, 2010 | June 30, 2009 | |||||||
| Derivatives NOT designated as hedging instruments under ASC 815 |
| |||||||
| Interest rate contracts |
$ | (4,161 | ) | $ | — | |||
| Foreign exchange contracts |
(2,791 | ) | (876 | ) | ||||
| Total non-designated derivatives |
$ | (6,952 | ) | $ | (876 | ) | ||
NOTE 5. BILLED AND UNBILLED ACCOUNTS RECEIVABLE
Accounts receivable at June 30, 2010 and 2009 consisted of the following (in thousands):
| 2010 | 2009 | |||||||
| Billed |
$ | 238,430 | $ | 270,081 | ||||
| Unbilled |
253,511 | 234,459 | ||||||
| Provision for losses on receivables |
(13,015 | ) | (23,219 | ) | ||||
| Total |
$ | 478,926 | $ | 481,321 | ||||
The decrease in the provision for losses on receivables was due primarily to the Fiscal Year 2010 fourth-quarter charge off of the reserve (and associated billed receivables) related to the small biopharma client that had filed for bankruptcy in the second quarter of Fiscal Year 2009.
NOTE 6. PROPERTY AND EQUIPMENT
Property and equipment at June 30, 2010 and 2009 consisted of the following (in thousands):
| 2010 | 2009 | |||||||
| Owned assets: |
||||||||
| Computer software |
$ | 168,534 | $ | 129,906 | ||||
| Computer and office equipment |
79,310 | 71,572 | ||||||
| Leasehold improvements |
81,097 | 72,503 | ||||||
| Medical equipment |
17,045 | 16,202 | ||||||
| Furniture and fixtures |
22,534 | 23,605 | ||||||
| Buildings |
4,472 | 5,140 | ||||||
| Office equipment & other assets |
19,249 | 18,686 | ||||||
| Total |
392,241 | 337,614 | ||||||
| Less: accumulated depreciation |
(204,353 | ) | (167,288 | ) | ||||
| Property and equipment, net |
$ | 187,888 | $ | 170,326 | ||||
| Assets held under capital leases: |
||||||||
| Computer software |
1,603 | 1,603 | ||||||
| Less: accumulated amortization |
(1,603 | ) | (1,443 | ) | ||||
| Total |
— | 160 | ||||||
| Total |
$ | 187,888 | $ | 170,486 | ||||
Depreciation and amortization expense relating to property and equipment, including amortization of assets recorded under capital leases, was $49.9 million, $43.4 million, and $33.0 million for the years ended June 30, 2010, 2009, and 2008, respectively.
During the year ended June 30, 2010, we retired $2.0 million of fully-depreciated assets. During the year ended June 30, 2009, we retired $4.6 million of fully-depreciated assets.
56
Table of Contents
During the years ended June 30, 2010 and 2009, we recorded impairment expense of $0.4 million and $2.3 million, respectively, related to the write-off of certain computer software assets that we had developed internally.
NOTE 7. RESTRUCTURING CHARGES
In October 2009, we adopted a plan to restructure our operations to reduce expenses, better align costs with current and future geographic sources of revenue, and improve operating efficiencies (the “2010 Restructuring Plan”). For Fiscal Year 2010, we recorded $16.8 million in restructuring charges related to the 2010 Restructuring Plan, including approximately $11.6 million in employee separation benefits associated with the elimination of 238 managerial and staff positions and $5.2 million in costs related to the abandonment of certain property leases.
The Pre-2010 Plans reflect the reserves related to various restructuring plans adopted by us since Fiscal Year 2005.
Changes in the restructuring accrual during Fiscal Years 2010, 2009, and 2008 are summarized below:
| (in thousands) |
Balance at June 30, 2009 |
Provisions/ Adjustments |
Payments/Foreign Currency Exchange |
Balance at June 30, 2010 | ||||||||||
| 2010 Restructuring Plan |
||||||||||||||
| Employee severance costs |
$ | — | $ | 11,618 | $ | (6,397 | ) | $ | 5,221 | |||||
| Facilities-related charges |
— | 5,117 | (1,780 | ) | 3,337 | |||||||||
| Other charges |
— | 75 | (21 | ) | 54 | |||||||||
| Pre-2010 Plans |
||||||||||||||
| Facilities-related charges |
2,144 | — | (678 | ) | 1,466 | |||||||||
| Total |
$ | 2,144 | $ | 16,810 | $ | (8,876 | ) | $ | 10,078 | |||||
| Balance at June 30, 2008 |
Provisions/ Adjustments |
Payments/Foreign Currency Exchange |
Balance at June 30, 2009 | |||||||||||
| Pre-2010 Plans |
||||||||||||||
| Facilities-related charges |
$ | 5,244 | $ | (33 | ) | $ | (3,067 | ) | $ | 2,144 | ||||
| Total |
$ | 5,244 | $ | (33 | ) | $ | (3,067 | ) | $ | 2,144 | ||||
| Balance at June 30, 2007 |
Provisions/ Adjustments |
Payments/Foreign Currency Exchange |
Balance at June 30, 2008 | |||||||||||
| Pre-2010 Plans |
||||||||||||||
| Employee severance costs |
$ | 223 | $ | (125 | ) | $ | (98 | ) | $ | 0 | ||||
| Facilities-related charges |
10,084 | (735 | ) | (4,105 | ) | 5,244 | ||||||||
| Total |
$ | 10,307 | $ | (860 | ) | $ | (4,203 | ) | $ | 5,244 | ||||
NOTE 8. CREDIT ARRANGEMENTS
2008 Credit Facility
On June 13, 2008, PAREXEL, certain subsidiaries of PAREXEL, JPMorgan Chase Bank, N.A., as Administrative Agent, J.P. Morgan Europe Limited, as London Agent, and the lenders party thereto (the “Lenders”) entered into an agreement for a credit facility (as amended and restated as of August 14, 2008 and as further amended by the first amendment thereto dated as of December 19, 2008, the “2008 Credit Facility”) in the principal amount of up to $315 million (collectively, the “Loan Amount”). The 2008 Credit Facility consists of an unsecured term loan facility and an unsecured revolving credit facility. Of the total principal amount, up to $150 million is made available through a term loan and up to $165 million is made available through a revolving credit facility. A portion of the revolving loan facility is available for swingline loans of up to $20 million to be made by JP Morgan Chase Bank, N.A. and for letters of credit. We may request the lenders to increase the 2008 Credit Facility by an additional amount of up to $50 million. Such increase may, but is not committed to, be provided.
Borrowings made under the 2008 Credit Facility bear interest, at our determination, at a rate based on the highest of prime, the federal funds rate plus .50% and the one-month Adjusted LIBOR Rate (as defined in the 2008 Credit Facility) plus 1.00% (such highest rate, the “Alternate Base Rate”) plus a margin (not to exceed a per annum rate of .75%) based on the Leverage Ratio (defined below), in which case it is a floating interest rate, or based on LIBOR or EURIBOR plus a margin (not to exceed a per annum rate of 1.75%) based on the Leverage Ratio, in which case the interest rate is fixed at the beginning of each interest period for the balance of the interest period. An interest period is typically one, two, three, or six months. The “Leverage Ratio” is a ratio of the consolidated total debt to consolidated net income before interest, taxes, depreciation and
57
Table of Contents
amortization (EBITDA). Loans outstanding under the 2008 Credit Facility may be prepaid at any time in whole or in part without premium or penalty, other than customary breakage costs, if any. The 2008 Credit Facility terminates and any outstanding loans under it mature on June 13, 2013.
Repayment of the principal borrowed under the revolving credit facility (other than a swingline loan) is due on June 13, 2013. Repayment of principal borrowed under the term loan facility is as follows:
| • | 5% of principal borrowed was repaid by June 30, 2009; |
| • | 20% of principal borrowed was repaid by June 30, 2010; |
| • | 20% of principal borrowed must be repaid during the one-year period from July 1, 2010 to June 30, 2011; |
| • | 25% of principal borrowed must be repaid during the one-year period from July 1, 2011 to June 30, 2012; and |
| • | 30% of principal borrowed must be repaid during the one-year period from July 1, 2012 to June 13, 2013. |
All payments of principal on the term loan facility made during each annual period described above are required to be made in equal quarterly installments and to be accompanied by accrued interest thereon. To the extent not previously paid, all borrowings under the term loan facility must be repaid on June 13, 2013. Swingline loans under the 2008 Credit Facility generally must be paid on the first date after such swingline loan is made that is the 15th or last day of a calendar month.
Interest due under the revolving credit facility (other than a swingline loan) and the term loan facility must be paid quarterly for borrowings with an interest rate determined at the Alternate Base Rate. Interest must be paid on the last day of the interest period selected by us for borrowings with an interest rate based on LIBOR or EURIBOR; provided that for interest periods of longer than three months, interest is required to be paid every three months. Interest under swingline loans is payable when principal is required to be repaid.
Our obligations under the 2008 Credit Facility may be accelerated upon the occurrence of an event of default under the 2008 Credit Facility, which includes customary events of default, including payment defaults, defaults in the performance of affirmative and negative covenants, the inaccuracy of representations or warranties, bankruptcy and insolvency related defaults, cross defaults to other material indebtedness, defaults relating to such matters as ERISA and judgments, and a change of control default. Our obligations under the 2008 Credit Facility are guaranteed by certain of our U.S. domestic subsidiaries, and we have guaranteed any obligations of any co-borrowers under the 2008 Credit Facility.
In connection with the 2008 Credit Facility, we agreed to pay a commitment fee on the term loan commitment, payable quarterly calculated as a percentage of the unused amount of the term loan commitments at a per annum rate of 0.30%, and a commitment fee on the revolving loan commitment calculated as a percentage of the unused amount of the revolving loan commitments at a per annum rate of up to 0.375% (based on the Leverage Ratio). To the extent there are letters of credit outstanding under the 2008 Credit Facility, we will pay to the Administrative Agent, for the benefit of the lenders, and to the issuing bank certain letter of credit fees, a fronting fee and additional charges. We also agreed to pay various fees to JPMorgan Chase Bank, N.A. or KeyBank or both.
On August 14, 2008, we drew down approximately $78 million via the revolving credit facility available under the 2008 Credit Facility. This borrowing was our first drawdown under the 2008 Credit Facility, and the funds were used to repay all of our loans under the Amended and Restated Credit Agreement dated as of September 18, 2007, as amended, among the Company and the other parties thereto (the “2007 Credit Facility”), and to terminate all of our commitments thereunder. The proceeds of this borrowing were also used to pay certain fees and out-of-pocket expenses to the Lenders under the 2008 Credit Facility. On August 26, 2008, we drew down an additional amount of approximately $192 million under the 2008 Credit Facility in connection with the closing of the ClinPhone acquisition, pursuant to which we acquired all the issued shares of ClinPhone for approximately $172 million. The proceeds of the borrowing were also used to repay certain indebtedness of ClinPhone owed to HSBC Bank.
As of June 30, 2010, we had $212.5 million in principal amount of debt outstanding under the 2008 Credit Facility, consisting of $100.0 million of principal borrowed under the revolving credit facility and $112.5 million of principal under the term loan, and remaining borrowing availability of approximately $65.0 million under the revolving credit facility. Principal in the amount of $150 million under the 2008 Credit Facility has been hedged with an interest rate swap agreement and carries a fixed interest rate of 4.8%. As of June 30, 2010, our debt under the 2008 Credit Facility, including the $150 million of principal hedged with an interest swap agreement, carried an average interest rate of 4.0%.
The 2008 Credit Facility contains affirmative and negative covenants applicable to us and our subsidiaries, including financial covenants requiring us to comply with maximum leverage ratios, minimum interest coverage ratios, a minimum net worth test (which covenant allows for foreign translation adjustments of up to $50 million in connection with the calculations required under such covenant) and maximum capital expenditures requirements, as well as restrictions on liens, investments, indebtedness, fundamental changes, acquisitions, dispositions of property, making specified restricted payments (including stock repurchases exceeding an agreed to percentage of consolidated net income), and transactions with affiliates. As of June 30, 2010, we were in compliance with all covenants under the 2008 Credit Facility.
58
Table of Contents
Additional Lines of Credit
We have a line of credit with RBS Nederland, NV in the amount of Euro 12.0 million. This line of credit is not collateralized, is payable on demand, and bears interest at an annual rate ranging between 2% and 4%. The line of credit may be revoked or canceled by the bank at any time at its discretion. We primarily entered into this line of credit to facilitate business transactions with the bank. At June 30, 2010, we had Euro 12.0 million available under this line of credit.
We have a line of credit with HSBC UK in the amount of 2.0 million pounds sterling. This line of credit was established by ClinPhone and is guaranteed by PAREXEL International Holding BV. The line is not secured and bears interest at an annual rate ranging between 2% and 4%. At June 30, 2010, we had 2.0 million pounds sterling available under this line of credit.
We have an unsecured line of credit with JP Morgan UK in the amount of $4.5 million that bears interest at an annual rate ranging between 2% and 4%. We primarily entered into this line of credit to facilitate business transactions with the bank. At June 30, 2010, we had $4.5 million available under this line of credit.
We have other foreign lines of credit with banks totaling $2.0 million. These lines of credit are used as overdraft protection and bear interest at annual rates ranging from 2% and 4%. The lines of credit are payable on demand. At June 30, 2010, we had $2.0 million available under these arrangements.
We have a cash pooling arrangement with RBS Nederland, NV. Pooling occurs when debit balances are offset against credit balances and the overall net position is used as a basis by the bank for calculating the overall pool interest amount. Each legal entity owned by PAREXEL and party to this arrangement remains the owner of either a credit (deposit) or a debit (overdraft) balance. Therefore, interest income is earned by legal entities with credit balances, while interest expense is charged to legal entities with debit balances. Based on the pool’s overall balance, the bank then (1) recalculates the overall interest to be charged or earned, (2) compares this amount with the sum of previously charged/earned interest amounts per account and (3) additionally pays/charges the difference. The gross overdraft balance related to this pooling arrangement was $91.0 million and $117.9 million at June 30, 2010 and June 30, 2009, respectively. However, on a net basis, we have surplus cash balances over all accounts for the respective periods.
We have financing agreements with a vendor to finance software purchases. The agreements carry four-year terms and bear annual interest rates ranging between 0% to 3%. As of June 30, 2010, the balance on the promissory notes issued in connection with the financing agreements was $2.9 million.
NOTE 9. STOCKHOLDERS’ EQUITY
On February 11, 2008, our Board of Directors approved a two-for-one stock split. The record date for the stock split was February 22, 2008. The stock split was completed on March 3, 2008.
On September 9, 2004, the Board of Directors approved a stock repurchase program authorizing the purchase of up to $20.0 million of our common stock to be repurchased in the open market subject to market conditions. Unless terminated earlier by resolution of the Board of Directors, this repurchase program will expire when the entire amount authorized has been fully utilized. Through June 30, 2010, we had acquired 1,240,828 shares at a total cost of $14.0 million under this program. No stock was repurchased during Fiscal Year 2010.
NOTE 10. EARNINGS PER SHARE
Basic earnings per share is computed by dividing net income for the period by the weighted average number of common shares outstanding during the period. Diluted earnings per share is computed by dividing net income by the weighted average number of common shares plus the dilutive effect of outstanding common stock equivalents. Outstanding options to purchase approximately 1,740,894 shares were excluded from the calculation of diluted earnings per share for the year ended June 30, 2010 because they were anti-dilutive. Outstanding options to purchase approximately 1,388,000 shares were excluded from the calculation of diluted earnings per share for the year ended June 30, 2009 because they were anti-dilutive. Outstanding options to purchase approximately 67,000 shares were excluded from the calculation of diluted earnings per share for the year ended June 30, 2008 because they were anti-dilutive. We also excluded all unvested restricted stock from the calculation of basic and diluted earnings per share because they were anti-dilutive.
59
Table of Contents
The following table outlines the basic and diluted earnings per common share computations:
| Years ended June 30, | |||||||||
| (in thousands, except per share data) |
2010 | 2009 | 2008 | ||||||
| Net income attributable to common shares |
$ | 41,542 | $ | 39,307 | $ | 64,640 | |||
| Weighted average number of shares outstanding, used in computing basic earnings per share |
58,062 | 57,538 | 55,896 | ||||||
| Dilutive common stock equivalents |
694 | 309 | 1,565 | ||||||
| Weighted average shares used in computing diluted earnings per share |
58,756 | 57,847 | 57,461 | ||||||
| Basic earnings per share |
$ | 0.72 | $ | 0.68 | $ | 1.16 | |||
| Diluted earnings per share |
$ | 0.71 | $ | 0.68 | $ | 1.12 | |||
NOTE 11. ACCUMULATED OTHER COMPREHENSIVE INCOME (LOSS)
Comprehensive income (loss) has been calculated by PAREXEL in accordance with ASC 220, “Comprehensive Income.” The reconciliation of the components of accumulated other comprehensive income (loss) was as follows:
| (in thousands) |
Foreign currency translation |
Unrealized gain (loss) on derivative instruments |
Total | |||||||||
| Balance as of June 30, 2007 |
$ | 21,498 | $ | 430 | $ | 21,928 | ||||||
| Changes during Fiscal Year 2008 |
31,214 | (913 | ) | 30,301 | ||||||||
| Balance as of June 30, 2008 |
$ | 52,712 | $ | (483 | ) | $ | 52,229 | |||||
| Changes during Fiscal Year 2009 |
(63,449 | ) | 352 | (63,097 | ) | |||||||
| Balance as of June 30, 2009 |
$ | (10,737 | ) | $ | (131 | ) | $ | (10,868 | ) | |||
| Changes during Fiscal Year 2010 |
(23,677 | ) | (6,889 | ) | (30,566 | ) | ||||||
| Balance as of June 30, 2010 |
$ | (34,414 | ) | $ | (7,020 | ) | $ | (41,434 | ) | |||
The unrealized gain (loss) on derivative instruments is net of taxes of $(3.0) million in Fiscal Year 2010, $0.2 million in Fiscal Year 2009, and $(0.6) million in Fiscal Year 2008 and net of amounts reclassified to other expense, net, of $(0.4) million in Fiscal Year 2010, $0.4 million in Fiscal Year 2009, and $(0.1) million in Fiscal Year 2008.
NOTE 12. STOCK AND EMPLOYEE BENEFIT PLANS
Stock-Based Compensation
We account for stock-based compensation under ASC 718, “Compensation-Stock Compensation.” The stock option compensation cost calculated under the fair value approach is recognized over the vesting period of the stock options (generally over four years). All stock option grants are subject to graded vesting as services are rendered. The fair value for granted options is estimated at the time of the grant using the Black-Scholes option-pricing model. Expected volatilities are based on implied and historical volatilities and PAREXEL uses historical data to estimate option exercise behavior. The following weighted average assumptions were used in the Black-Scholes option-pricing model for awards issued during the respective periods:
| For the years ended June 30, | |||||||||
| 2010 | 2009 | 2008 | |||||||
| Dividend yield |
0.0 | % | 0.0 | % | 0.0 | % | |||
| Expected volatility |
55.4 | % | 55.5 | % | 35.2 | % | |||
| Risk-free interest rate |
2.32 | % | 2.04 | % | 3.54 | % | |||
| Expected term (in years) |
5.00 | 5.37 | 5.00 | ||||||
60
Table of Contents
For the last three fiscal years, we recognized the following stock-based compensation expense:
| For the years ended June 30, | |||||||||
| (in thousands) |
2010 | 2009 | 2008 | ||||||
| Direct costs related |
$ | 2,062 | $ | 2,241 | $ | 1,625 | |||
| Selling, general and administrative related |
4,955 | 5,072 | 3,615 | ||||||
| Total stock-based compensation |
$ | 7,017 | $ | 7,313 | $ | 5,240 | |||
For Fiscal Years 2010, 2009, and 2008, the tax benefit related to stock compensation expense that we recognized was $2.3 million, $2.5 million, and $1.7 million, respectively. As of June 30, 2010, unearned stock-based compensation expense related to unvested awards (stock options and restricted stock) was approximately $15.7 million, which will be recognized over a weighted-average period of 2.5 years.
Stock Options
The Compensation Committee of the Board of Directors is responsible for administration of PAREXEL’s stock option plans and determines the term of each option, the option exercise price, the number of option shares granted, and the rate at which options become exercisable.
In December 2007, we adopted the 2007 Stock Incentive Plan (“2007 Plan”), which provides for the grant of incentive stock options, non-statutory stock options, stock appreciation rights, restricted stock, restricted stock units and other stock-based award grants of up to an aggregate of 2,000,000 shares of common stock to employees, officers, directors, consultants, and advisors. The granting of awards under the 2007 Plan is discretionary and the individuals who may become participants and receive awards under the 2007 Plan, and the number of shares they may acquire, are not determinable.
In September 2005, we adopted the 2005 Stock Incentive Plan (“2005 Plan”), which provides for the grant of incentive stock options, non-statutory stock options, stock appreciation rights, restricted stock, restricted stock units and other stock-based award grants of up to an aggregate of 2,000,000 shares of common stock to employees, officers, directors, consultants, and advisors. The granting of awards under the 2005 Plan is discretionary and the individuals who may become participants and receive awards under the 2005 Plan, and the number of shares they may acquire, are not determinable.
In September 2001, we adopted the 2001 Stock Incentive Plan (“2001 Plan”), which provides for the grant of incentive and non-qualified stock options for the purchase of up to an aggregate of 2,000,000 shares of common stock to employees, officers, directors, consultants, and advisors (and any individuals who have accepted an offer for employment) of PAREXEL. Options under the 2001 Plan expire no more than ten years from the date of grant and the expiration date and vesting period may vary at the Board of Directors’ discretion.
The following table summarizes information related to stock option activity for the respective periods:
| For the years ended June 30, | |||||||||
| (in thousands, except per share data) |
2010 | 2009 | 2008 | ||||||
| Weighted-average fair value of options granted per share |
$ | 7.09 | $ | 6.84 | $ | 8.40 | |||
| Intrinsic value of options exercised |
$ | 5,228 | $ | 7,230 | $ | 27,666 | |||
| Cash received from options exercised |
$ | 4,884 | $ | 3,265 | $ | 11,341 | |||
Stock option activity for the year ended June 30, 2010 was:
| Number of Options | Weighted-Average Exercise Price | |||||
| Balance on June 30, 2009 |
3,977,392 | $ | 14.72 | |||
| FY 2010 |
||||||
| Granted |
562,613 | $ | 14.20 | |||
| Exercised |
(518,906 | ) | $ | 9.41 | ||
| Canceled |
(167,700 | ) | $ | 15.64 | ||
| Outstanding on June 30, 2010 |
3,853,399 | $ | 15.33 | |||
61
Table of Contents
Options that were outstanding, exercisable, and expected to vest as of June 30, 2010 are as follows:
| Number of Options |
Weighted-Average Exercise Price |
Weighted- Average Remaining Contractual Life In Years |
Aggregate Intrinsic Value (In Thousands) | |||||||
| Outstanding |
3,853,399 | $ | 15.33 | 5.17 | $ | 28,213 | ||||
| Exercisable |
1,922,136 | $ | 14.11 | 3.82 | $ | 15,754 | ||||
| Expected to vest |
1,684,813 | $ | 16.71 | 6.41 | $ | 10,660 | ||||
Restricted Stock
PAREXEL uses restricted stock awards (“RSAs”) and restricted stock units (“RSUs”), granted under the plans described above, as a component of compensation for executive officers and non-employee members of the Board of Directors. In Fiscal Year 2010, we granted RSAs that will vest at the end of a three-year service period and RSUs that will vest at the end of three-year service and two-year service periods. The fair values of the 2010 restricted stock awards and restricted stock units were based upon the closing stock prices on the day of the grants. Restricted stock activity for the year ended June 30, 2010 was:
| Shares | Weighted-Average Grant- Date Fair Value | |||||
| Balance on June 30, 2009 |
136,842 | $ | 31.71 | |||
| FY 2010 |
||||||
| RSAs Granted |
270,300 | $ | 13.04 | |||
| RSAs Forfeited |
(28,500 | ) | $ | 19.14 | ||
| RSUs Granted |
28,500 | $ | 20.33 | |||
| Outstanding June 30, 2010 |
407,142 | $ | 19.40 | |||
Employee Stock Purchase Plan
In March 2000, the Board of Directors adopted the 2000 Employee Stock Purchase Plan (the “2000 Purchase Plan”). The 2000 Purchase Plan was amended in May 2005 for offering periods commencing on or after June 1, 2005 to purchase common stock at 95% of the fair market value of the stock on the last day of each purchase period (as defined by the Purchase Plan). In January 2008, the Purchase Plan was further amended to include the automatic enrollment of contributions whereby an eligible employee’s compensation would be reduced and automatic enrollment contributions made on his/her behalf unless an affirmative election not to do so was made. An aggregate of approximately 1,800,000 shares may be issued under the 2000 Purchase Plan.
The following table summarizes the purchases under the 2000 Purchase Plan for the last three fiscal years:
| Shares Purchased | Average Purchase Price | ||||
| Fiscal Year 2010 |
131,880 | $ | 14.65 | ||
| Fiscal Year 2009 |
174,302 | $ | 9.05 | ||
| Fiscal Year 2008 |
57,184 | $ | 22.24 | ||
401(k)
PAREXEL sponsors an employee savings plan (“the Plan”) as defined by Section 401(k) of the Internal Revenue Code of 1986, as amended. The Plan covers substantially all employees in the U.S. who elect to participate. Participants have the opportunity to invest on a pre-tax basis in a variety of mutual fund options and PAREXEL stock. We match 100% of each participant’s voluntary contributions up to 3% of gross salary per payroll period subject to an annual cap of $3,000. PAREXEL contributions vest to the participants in 20% increments for each year of employment and become fully vested after five years of continuous employment. Our contributions to the Plan were approximately $4.4 million, $4.1 million, and $3.1 million for the Fiscal Years 2010, 2009, and 2008, respectively.
62
Table of Contents
NOTE 13. FAIR VALUE MEASUREMENTS
We apply the provisions of ASC 820, “Fair Value Measurements and Disclosures.” ASC 820 defines fair value and provides guidance for measuring fair value and expands disclosures about fair value measurements. ASC 820 enables the reader of financial statements to assess the inputs used to develop those measurements by establishing a hierarchy for ranking the quality and reliability of the information used to determine fair values. ASC 820 requires that assets and liabilities carried at fair value be classified and disclosed in one of the following three categories:
| • | Level 1 – Unadjusted quoted prices in active markets that are accessible to the reporting entity at the measurement date for identical assets and liabilities. |
| • | Level 2 – Inputs other than quoted prices in active markets for identical assets and liabilities that are observable either directly or indirectly for substantially the full term of the asset or liability. Level 2 inputs include the following: |
| • | quoted prices for similar assets and liabilities in active markets |
| • | quoted prices for identical or similar assets or liabilities in markets that are not active |
| • | observable inputs other than quoted prices that are used in the valuation of the asset or liabilities (e.g., interest rate and yield curve quotes at commonly quoted intervals) |
| • | inputs that are derived principally from or corroborated by observable market data by correlation or other means |
| • | Level 3 – Unobservable inputs for the assets or liability (i.e., supported by little or no market activity). Level 3 inputs include management’s own assumption about the assumptions that market participants would use in pricing the asset or liability (including assumptions about risk). |
The following table sets forth by level, within the fair value hierarchy, our assets (liabilities) carried at fair value as of June 30, 2010:
| Level 1 | Level 2 | Level 3 | Total | |||||||||||
| Marketable Securities |
$ | 12,243 | $ | — | $ | — | $ | 12,243 | ||||||
| Interest Rate Derivative Instruments |
— | (9,223 | ) | — | (9,223 | ) | ||||||||
| Foreign Currency Exchange Contracts |
— | (6,023 | ) | — | (6,023 | ) | ||||||||
| Total |
$ | 12,243 | $ | (15,246 | ) | — | $ | (3,003 | ) | |||||
The marketable securities are held in foreign government treasury certificates that are actively traded.
Interest rate derivative instruments are measured at fair value using the mark-to-market valuation technique. The valuation is based on the estimate of net present value of the expected cash flows using relevant mid-market observable data inputs and based on the assumption of no unusual market conditions or forced liquidation.
Foreign currency exchange contracts are measured at fair value using the market-to-market method valuation technique. The inputs to this technique utilize current foreign currency exchange forward market rates published by third-party leading financial news and data providers. This is observable data that represent the rates that the financial institution uses for contracts entered into at that date; however, they are not based on actual transactions so they are classified as Level 2.
As of June 30, 2010, there were no transfers between Level 1, Level 2, or Level 3. Additionally, there were no changes in the valuation techniques used to determine the fair values of our Level 2 or Level 3 assets or liabilities.
The carrying value of our short-term and long-term debt approximates fair value because all of the debt bears variable rate interest.
NOTE 14. INCOME TAXES
Domestic and foreign income (loss) before income taxes for the three years ended June 30 were as follows:
| (in thousands) |
2010 | 2009 | 2008 | |||||||||
| Domestic |
$ | (65 | ) | $ | (34,413 | ) | $ | (3,540 | ) | |||
| Foreign |
63,240 | 98,251 | 88,206 | |||||||||
| $ | 63,175 | $ | 63,838 | $ | 84,666 | |||||||
63
Table of Contents
Provisions for income taxes for the three years ended June 30 were as follows:
| (in thousands) |
2010 | 2009 | 2008 | |||||||||
| Current: |
||||||||||||
| Federal |
$ | (5,284 | ) | $ | (5,600 | ) | $ | 11,269 | ||||
| State |
1,336 | (53 | ) | 1,464 | ||||||||
| Foreign |
31,539 | 30,753 | 19,964 | |||||||||
| 27,591 | 25,100 | 32,697 | ||||||||||
| Deferred: |
||||||||||||
| Federal |
4,605 | (138 | ) | (15,477 | ) | |||||||
| State |
(383 | ) | (106 | ) | 72 | |||||||
| Foreign |
(10,180 | (325 | ) | 2,734 | ||||||||
| (5,958 | ) | (569 | ) | (12,671 | ) | |||||||
| $ | 21,633 | $ | 24,531 | $ | 20,026 | |||||||
Our consolidated effective income tax rate differed from the U.S. federal statutory income tax rate as set forth below:
| (in thousands) |
2010 | % | 2009 | % | 2008 | % | |||||||||||||||
| Income tax expense computed at the federal statutory rate |
$ | 22,111 | 35.0 | % | $ | 22,343 | 35.0 | % | $ | 29,633 | 35.0 | % | |||||||||
| State income taxes, net of federal benefit |
311 | 0.5 | % | 423 | 0.7 | % | 530 | 0.6 | % | ||||||||||||
| Foreign rate differential |
(2,674 | ) | -4.2 | % | (6,179 | ) | -9.7 | % | (5,345 | ) | -6.3 | % | |||||||||
| Change in valuation allowances |
(479 | ) | -0.8 | % | 1,585 | 2.5 | % | (9,540 | ) | -11.3 | % | ||||||||||
| Change in reserves |
(1,467 | ) | -2.3 | % | 941 | 1.5 | % | 9,457 | 11.2 | % | |||||||||||
| Research and development |
(2,705 | ) | -4.3 | % | (2,766 | ) | -4.3 | % | (1,738 | ) | -2.1 | % | |||||||||
| Non-deductible losses |
1,828 | 2.9 | % | 2,185 | 3.4 | % | 0 | 0.0 | % | ||||||||||||
| Other non-deductible expenses |
3,715 | 5.9 | % | 3,371 | 5.3 | % | 1,387 | 1.6 | % | ||||||||||||
| Statutory tax rate changes |
143 | 0.2 | % | 181 | 0.3 | % | (6,332 | ) | -7.4 | % | |||||||||||
| Other |
850 | 1.3 | % | 2,447 | 3.7 | % | 1,974 | 2.4 | % | ||||||||||||
| $ | 21,633 | 34.2 | % | $ | 24,531 | 38.4 | % | $ | 20,026 | 23.7 | % | ||||||||||
Provision has not been made for U.S. or additional foreign taxes on undistributed earnings of foreign subsidiaries as those earnings have been indefinitely reinvested. Undistributed earnings of foreign subsidiaries that have been indefinitely reinvested are approximately $252 million and $234 million at June 30, 2010 and 2009, respectively. It is not practical to estimate the amount of income taxes payable on the earnings that are indefinitely reinvested in foreign operations.
Significant components of our net deferred tax assets as of June 30, 2010 and 2009 were as follows:
| (in thousands) |
2010 | 2009 | ||||||
| Deferred tax assets: |
||||||||
| U.S. loss carryforwards |
$ | 2,648 | $ | 10,525 | ||||
| Foreign loss carryforwards |
8,172 | 4,457 | ||||||
| Accrued expenses |
21,488 | 9,460 | ||||||
| Tax credit carryforwards |
16,900 | 17,895 | ||||||
| Provision for losses on receivables |
2,516 | 6,691 | ||||||
| Deferred compensation |
5,608 | 3,733 | ||||||
| Deferred revenue |
8,185 | 10,112 | ||||||
| Intercompany loans |
4,801 | 5,910 | ||||||
| Other |
765 | 897 | ||||||
| Gross deferred tax assets |
71,083 | 69,680 | ||||||
| Deferred tax asset valuation allowance |
(21,670 | ) | (31,445 | ) | ||||
| Total deferred tax assets |
49,413 | 38,235 | ||||||
| Deferred tax liabilities: |
||||||||
| Property and equipment |
(4,393 | ) | (10,636 | ) | ||||
| Revenue recognition |
(18,913 | ) | (18,711 | ) | ||||
| Intangible assets |
(35,492 | ) | (30,665 | ) | ||||
| Other |
(2,702 | ) | (4,126 | ) | ||||
| Total deferred tax liabilities |
(61,500 | ) | (64,138 | ) | ||||
| Net deferred tax liabilities |
$ | (12,087 | ) | $ | (25,903 | ) | ||
64
Table of Contents
The net deferred tax assets and liabilities included in the consolidated balance sheets as of June 30, 2010 and 2009 were as follows:
| (in thousands) |
2010 | 2009 | ||||||
| Current deferred tax assets |
$ | 28,932 | $ | 21,268 | ||||
| Non-current deferred tax assets |
7,193 | 15,385 | ||||||
| Current deferred tax liabilities |
(15,977 | ) | (18,110 | ) | ||||
| Non-current deferred tax liabilities |
(32,235 | ) | (44,446 | ) | ||||
| $ | (12,087 | ) | $ | (25,903 | ) | |||
At June 30, 2010, state, federal and foreign loss carryforwards of $70.7 million, $29.8 million and $36.7 million, respectively, were available to offset future liabilities for income taxes. Included in the state and federal loss carryforwards are $12.0 million which were attributable to deductions from the exercise of equity awards. The benefit from these deductions will be recorded as a credit to additional paid-in capital if and when realized through a reduction of taxes paid in cash. Use of these loss carryforwards is limited based on the future income of certain subsidiaries. The state and federal net operating losses expire in the years 2011 through 2029. Of the non-U.S. loss carryforwards, $4.9 million will expire between 2013 and 2020; the remainder does not expire. We also have U.S. foreign tax credit carryforwards of $22.6 million which expire in the years 2015 through 2020. Included in the U.S. foreign tax credit carryforwards are $5.8 million which were attributable to deductions from the exercise of equity awards. The benefit from these credits will be recorded as a credit to additional paid-in capital if and when realized through a reduction of taxes paid in cash.
A valuation allowance has been established for certain future income tax benefits related to loss carryforwards, foreign tax credit carryforwards and temporary tax adjustments based on an assessment that it is more likely than not that these benefits will not be realized. In Fiscal Year 2010, the valuation allowance decreased principally as a result of a reduction in non-U.S. loss carryforwards for which it is more likely than not that the benefits will not be realized.
As of June 30, 2009, we had $58.3 million of gross unrecognized tax benefits of which $15.6 million would impact the effective tax rate if recognized. As of June 30, 2010 we had $56.3 million of gross unrecognized tax benefits of which $19.2 million would impact the effective tax rate if recognized. This reserve primarily relates to exposures for income tax matters such as changes in the jurisdiction in which income is taxable and taxation of certain investments. The decrease in gross unrecognized tax benefits is primarily composed of a $6.2 million increase resulting from changes in reserves established in conjunction with the acquisition of ClinPhone Group Limited in Fiscal Year 2009, a $4.5 million decrease resulting from changes in foreign currency exchange rates, neither of which impacted the effective tax rate, a $3.7 million decrease resulting from the expiration of U.S. and international statutes and a $1.3 million decrease which reflects the resolution of foreign examinations.
Unrecognized tax benefits represent favorable positions we have taken, or expect to take, on tax returns. These positions have reduced, or are expected to reduce, our income tax liability on our tax returns and financial statements. As a result of the uncertainty associated with these positions, we have established a liability that effectively reverses the previous recognition of the tax benefits, making them “unrecognized.” Our unrecognized income tax benefits, excluding accrued interest and penalties, are as follows:
| (in thousands) |
2010 | 2009 | 2008 | ||||||||
| Balance at beginning of year |
$ | 58,310 | $ | 63,184 | $ | 44,353 | |||||
| Additions related to tax positions in prior years |
7,557 | 1,777 | 6,839 | ||||||||
| Additions related to tax positions in the current year |
5 | 1,131 | 11,992 | ||||||||
| Reductions related to tax positions in prior years |
(4,488 | ) | (3,691 | ) | — | ||||||
| Reductions related to settlements with tax authorities |
(1,299 | ) | (4,091 | ) | — | ||||||
| Reductions related to the expiration of statutes |
(3,740 | ) | — | — | |||||||
| Balance at end of year |
$ | 56,345 | $ | 58,310 | $ | 63,184 | |||||
As of June 30, 2010, we anticipate that the liability for unrecognized tax benefits for uncertain tax positions could change by up to $2.1 million in the next twelve months as a result of the expiration of statutes. This change is composed primarily of reserves associated with the jurisdiction in which income is taxable and taxation of certain investments.
Our historical practice has been, and continues to be, to recognize interest and penalties related to income tax matters in income tax expense. As of June 30, 2009, $9.4 million of interest and penalties were included in our liability for unrecognized tax benefits. Income tax expense recorded through June 30, 2010 includes approximately $0.6 million of interest and penalties. As of June 30, 2010, $8.9 million of interest and penalties were included in our liability for unrecognized tax benefits.
65
Table of Contents
In April 2008, we were granted a tax holiday from the government of India for our operation in Hyderabad, India, which is engaged in providing information technology services to support our global clinical trial services. This tax holiday was granted for a fifteen-year period and expires in April 2023. The income tax benefit of this holiday was $2.1 million, $0.9 million, and $0.0 million in Fiscal Years 2010, 2009, and 2008, respectively. For the same periods, the per share effects of this holiday were $0.04, $0.01, and $0.00, respectively, on a fully diluted basis.
PAREXEL is subject to U.S. federal income tax, as well as income tax in multiple state, local and foreign jurisdictions. All material state and local income tax matters through 2005 have been concluded. All material federal income tax matters have been concluded through 2005. Substantially all material foreign income tax matters have been concluded for all years through 2000.
NOTE 15. DEBT, COMMITMENTS, CONTINGENCIES AND GUARANTEES
We lease facilities under operating leases that include renewal and escalation clauses. Total rent expense, net of sublease income, was $49.1 million, $48.5 million, and $44.3 million for Fiscal Years 2010, 2009, and 2008, respectively. Future minimum debt obligations, lease payments under non-cancelable leases, and purchase commitments due are as follows:
| (in thousands) |
FY 2011 | FY 2012 | FY 2013 | FY 2014 | FY 2015 | Thereafter | Total | ||||||||||||||
| Debt obligations (principal) |
$ | 32,071 | $ | 38,443 | $ | 145,264 | $ | — | $ | — | $ | — | $ | 215,778 | |||||||
| Operating leases |
52,081 | 41,413 | 33,837 | 28,143 | 22,789 | 85,824 | 264,087 | ||||||||||||||
| Purchase commitments* |
27,851 | 12,999 | 5,639 | 825 | 334 | 232 | 47,880 | ||||||||||||||
| Total |
$ | 112,003 | $ | 92,855 | $ | 184,740 | $ | 28,968 | $ | 23,123 | $ | 86,056 | $ | 527,745 | |||||||
| * | includes commitments to purchase software, hardware, and services. |
We have letter-of-credit agreements with banks, totaling approximately $5.3 million, guaranteeing performance under various operating leases and vendor agreements. We also have an unsecured facility consisting of a term loan facility for $150 million and a revolving credit facility for $165 million with a group of lenders (including and managed by JPMorgan Chase Bank, N.A.) that is guaranteed by certain of our U.S. subsidiaries.
PAREXEL periodically becomes involved in various claims and lawsuits that are incidental to its business. In June 2010, we recorded $4.3 million in legal settlement costs related to a small acquisition that was completed several years ago. We believe, after consultation with counsel, that no other matters currently pending would, in the event of an adverse outcome, have a material impact on our consolidated financial position, results of operations, or liquidity.
NOTE 16. GEOGRAPHIC INFORMATION
Financial information by geographic area for the three years ended June 30 were as follows:
| (in thousands) |
2010 | 2009 | 2008 | ||||||
| Service revenue: |
|||||||||
| The Americas |
$ | 449,357 | $ | 426,284 | $ | 377,857 | |||
| Europe, Middle East & Africa |
548,412 | 528,914 | 515,445 | ||||||
| Asia/Pacific |
133,270 | 95,557 | 70,981 | ||||||
| Total |
$ | 1,131,039 | $ | 1,050,755 | $ | 964,283 | |||
| Income from operations: |
|||||||||
| The Americas |
$ | 21,387 | $ | 3,565 | $ | 16,079 | |||
| Europe, Middle East & Africa |
39,704 | 61,353 | 62,394 | ||||||
| Asia/Pacific |
22,018 | 10,726 | 8,193 | ||||||
| Total |
$ | 83,109 | $ | 75,644 | $ | 86,666 | |||
| Tangible long-lived assets: |
|||||||||
| The Americas |
$ | 110,147 | $ | 78,075 | $ | 58,470 | |||
| Europe, Middle East & Africa |
66,702 | 83,024 | 82,800 | ||||||
| Asia/Pacific |
11,039 | 9,387 | 12,281 | ||||||
| Total |
$ | 187,888 | $ | 170,486 | $ | 153,551 | |||
66
Table of Contents
The following countries represented greater than 10% of consolidated service revenue for the three years ended June 30:
| (in thousands) |
2010 | 2009 | 2008 | ||||||
| Service revenue: |
|||||||||
| United States |
$ | 391,277 | $ | 375,051 | $ | 333,917 | |||
| Germany |
182,288 | 171,163 | 179,890 | ||||||
| United Kingdom |
165,340 | 163,486 | 143,930 | ||||||
NOTE 17. SEGMENT INFORMATION
PAREXEL is managed through three business segments:
| • | CRS constitutes our core business and includes Phase I-IV clinical trials management and biostatistics, data management and clinical pharmacology, as well as related medical advisory and investigator site services. |
| • | PCMS provides technical expertise and advice in such areas as drug development, regulatory affairs, and good manufacturing practices (“GMP”) compliance. PCMS also provides a full spectrum of market development, product development, targeted communications, and strategic reimbursement advisory services in support of product launch. |
| • | Perceptive provides information technology solutions designed to improve the product development processes of our clients. Perceptive offers a portfolio of products and services that includes medical imaging services, ClinPhone® randomization and trial supply management (“RTSM”), DataLabs® electronic data capture (“EDC”), IMPACT® and TrialWorks™ clinical trials management systems (“CTMS”), web-based portals, systems integration, and electronic patient reported outcomes (“ePRO”). |
We evaluate our segment performance and allocate resources based on service revenue and gross profit (service revenue less direct costs), while other operating costs are allocated and evaluated on a geographic basis. Accordingly, we do not include the impact of selling, general, and administrative expenses, depreciation and amortization expense, other income (expense), and income tax expense in segment profitability. We attribute revenue to individual countries based upon the revenue earned in the respective countries; however, inter-segment transactions are not included in service revenue. Furthermore, PAREXEL has a global infrastructure supporting its business segments, and therefore, assets are not identified by reportable segment.
| (in thousands) |
CRS | PCMS | PERCEPTIVE | TOTAL | ||||||||
| Service revenue: |
||||||||||||
| 2010 |
$ | 870,721 | $ | 121,652 | $ | 138,666 | $ | 1,131,039 | ||||
| 2009 |
804,237 | 121,785 | 124,733 | 1,050,755 | ||||||||
| 2008 |
745,641 | 129,804 | 88,838 | 964,283 | ||||||||
| Gross profit on service revenue: |
||||||||||||
| 2010 |
$ | 320,554 | $ | 47,160 | $ | 58,758 | $ | 426,472 | ||||
| 2009 |
286,987 | 43,562 | 45,143 | 375,692 | ||||||||
| 2008 |
251,762 | 43,874 | 39,248 | 334,884 | ||||||||
NOTE 18. OTHER CHARGE & INVESTMENT IMPAIRMENT
In the second quarter of Fiscal Year 2009, we received notification from a small biopharma client that the client would be unable to make payments due to us in connection with an on-going service contract for a large Phase III clinical trial. The client advised us that it encountered funding difficulties when one of its major investors defaulted on a contractual investment commitment to the client. The client informed us that, following the default, it had substantive discussions with two potential commercialization partners and its remaining investors, but was unable to secure additional funding for the trial. The client has since filed for bankruptcy protection. As a result, we recorded $15 million in reserves in the second quarter of Fiscal Year 2009, consisting of $12.3 million in bad debt expense and $2.7 million in anticipated wind-down costs and related expenses for service fees, pass-through costs, and investigator fees. In the second quarter of Fiscal Year 2010, we released $1.1 million of these reserves to reflect lower-than-anticipated close-out costs.
In January 2010, we were informed that a French laboratory, in which we had a direct and indirect investment, filed for bankruptcy protection due to the poor market conditions. We evaluated the investment and recorded a $6.1 million impairment reserve in miscellaneous expense for Fiscal Year 2010, reflecting our total investment in the entity.
67
Table of Contents
NOTE 19. QUARTERLY OPERATING RESULTS (UNAUDITED)
The following is a summary of unaudited quarterly results of operations for the years ended June 30, 2010 and 2009:
| For the year ended June 30, 2010 | |||||||||||||||
| in thousands, except per share data |
First Quarter |
Second Quarter |
Third Quarter |
Fourth Quarter |
Total Year | ||||||||||
| Service revenue |
$ | 259,763 | $ | 284,731 | $ | 291,244 | $ | 295,301 | $ | 1,131,039 | |||||
| Gross profit |
92,934 | 106,494 | 113,475 | 113,569 | 426,472 | ||||||||||
| Income from operations |
18,476 | 18,711 | 25,824 | 20,098 | 83,109 | ||||||||||
| Net income |
12,441 | 3,450 | 12,783 | 12,868 | 41,542 | ||||||||||
| Diluted earnings per share |
$ | 0.21 | $ | 0.06 | $ | 0.22 | $ | 0.22 | $ | 0.71 | |||||
| For the year ended June 30, 2009 | |||||||||||||||
| in thousands, except per share data |
First Quarter |
Second Quarter |
Third Quarter |
Fourth Quarter |
Total Year | ||||||||||
| Service revenue |
$ | 263,046 | $ | 275,846 | $ | 264,457 | $ | 247,406 | $ | 1,050,755 | |||||
| Gross profit |
91,682 | 98,551 | 98,676 | 86,783 | 375,692 | ||||||||||
| Income from operations |
21,993 | 7,710 | 26,419 | 19,522 | 75,644 | ||||||||||
| Net income |
13,619 | 5,208 | 14,204 | 6,276 | 39,307 | ||||||||||
| Diluted earnings per share |
$ | 0.23 | $ | 0.09 | $ | 0.25 | $ | 0.11 | $ | 0.68 | |||||
68
Table of Contents
Management’s Report on Internal Control over Financial Reporting
The management of PAREXEL International Corporation is responsible for establishing and maintaining adequate internal control over financial reporting for the Company. Internal control over financial reporting is defined in Rule 13a-15(f) or 15d-15(f) promulgated under the Securities Exchange Act of 1934 as a process designed by, or under the supervision of, the Company’s principal executive and principal financial officers and effected by the Company’s board of directors, management and other personnel, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles and includes those policies and procedures that:
| • | Pertain to the maintenance of records that in reasonable detail accurately and fairly reflect the transactions and dispositions of the assets of the Company; |
| • | Provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the Company are being made only in accordance with authorizations of management and directors of the Company; and |
| • | Provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of the Company’s assets that could have a material effect on the financial statements. |
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
The Company’s management assessed the effectiveness of the Company’s internal control over financial reporting as of June 30, 2010. In making this assessment, the Company’s management used the criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission (COSO) in Internal Control-Integrated Framework.
Based on the assessment, management concluded that, as of June 30, 2010, the Company’s internal control over financial reporting is effective based on those criteria.
The Company’s independent registered public accounting firm, Ernst & Young LLP, has issued an audit report on the Company’s internal control over financial reporting. This report appears on page 71.
| /s/ Josef H. von Rickenbach |
/s/ James F. Winschel, Jr. | |||
| Josef H. von Rickenbach Chairman of the Board and Chief Executive Officer (principal executive officer) |
James F. Winschel, Jr. Senior Vice President and Chief Financial Officer (principal financial officer) | |||
69
Table of Contents
Report of Independent Registered Public Accounting Firm
The Board of Directors and Stockholders of PAREXEL International Corporation
We have audited the accompanying consolidated balance sheets of PAREXEL International Corporation as of June 30, 2010 and 2009, and the related consolidated statements of income, stockholders’ equity, and cash flows for each of the three years in the period ended June 30, 2010. Our audits also included the financial statement schedule listed in the Index at Item 15. These financial statements and schedule are the responsibility of the Company’s management. Our responsibility is to express an opinion on these financial statements and schedule based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the financial statements referred to above present fairly, in all material respects, the consolidated financial position of PAREXEL International Corporation at June 30, 2010 and 2009, and the consolidated results of its operations and its cash flows for each of the three years in the period ended June 30, 2010, in conformity with U.S. generally accepted accounting principles. Also, in our opinion, the related financial statement schedule, when considered in relation to the basic financial statements taken as a whole, presents fairly in all material respects the information set forth therein.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States), PAREXEL International Corporation’s internal control over financial reporting as of June 30, 2010, based on criteria established in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission and our report dated August 27, 2010 expressed an unqualified opinion thereon.
/s/ Ernst & Young LLP
Boston, Massachusetts
August 27, 2010
70
Table of Contents
Report of Independent Registered Public Accounting Firm
The Board of Directors and Stockholders of PAREXEL International Corporation
We have audited PAREXEL International Corporation’s internal control over financial reporting as of June 30, 2010, based on criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (the COSO criteria). PAREXEL International Corporation’s management is responsible for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting included in the accompanying Management’s Report on Internal Control over Financial Reporting. Our responsibility is to express an opinion on the Company’s internal control over financial reporting based on our audit.
We conducted our audit in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects. Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, testing and evaluating the design and operating effectiveness of internal control based on the assessed risk, and performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
In our opinion, PAREXEL International Corporation maintained, in all material aspects, effective internal control over financial reporting as of June 30, 2010, based on the COSO criteria.
We have also audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States), the consolidated balance sheets as of June 30, 2010 and June 30, 2009 and the related consolidated statements of income, stockholders’ equity, and cash flows for each of the three years in the period ended June 30, 2010 of PAREXEL International Corporation and our report dated August 27, 2010 expressed an unqualified opinion thereon.
/s/ Ernst & Young LLP
Boston, Massachusetts
August 27, 2010
71
Table of Contents
| Item 9. | Changes in and Disagreements with Accountants on Accounting and Financial Disclosure |
Not applicable
| Item 9A. | Controls and Procedures |
| (a) | Evaluation of Disclosure Controls and Procedures |
Management conducted an evaluation, under the supervision and with the participation of our Chief Executive Officer and Chief Financial Officer, of the effectiveness of the design and operation of our disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) under the Securities Exchange Act of 1934, as amended) as of June 30, 2010. Based on the evaluation of our disclosure controls and procedures as of June 30, 2010, our Chief Executive Officer and Chief Financial Officer concluded that, as of such date, PAREXEL’s disclosure controls and procedures were effective at the reasonable assurance level.
Management’s Report on Internal Control over Financial Reporting and the independent registered public accounting firm’s attestation report on our internal control over financial reporting required under Item 308 of Regulation S-K have been included in Item 8.
| (b) | Changes in Internal Control Over Financial Reporting |
No change in our internal control over financial reporting (as defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act) occurred during the fiscal quarter ended June 30, 2010 that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
| Item 9B. | Other Information |
Not applicable.
72
Table of Contents
| Item 10. | Directors, Executive Officers and Corporate Governance |
Information with respect to this item may be found under the captions “Elections of Directors,” “Corporate Governance,” “Executive Officers” and “Section 16(a) Beneficial Ownership Reporting Compliance” in the Proxy Statement for the Company’s 2010 Annual Meeting of Stockholders. Such information is incorporated herein by reference.
CODE OF ETHICS
PAREXEL has adopted a code of business conduct and ethics applicable to all of its employees, including our principal executive officer and principal financial officer. The code of business conduct and ethics is available on our website (www.parexel.com) under the category “Investor Relations-Corporate Governance.”
| Item 11. | Executive Compensation |
Information with respect to this item may be found under the captions “Directors’ Compensation,” “Compensation Committee Interlocks and Insider Participation,” “Executive Compensation,” “Employment and Change of Control Agreements” and “Compensation Committee Report” in the Proxy Statement for the Company’s 2010 Annual Meeting of Stockholders. Such information is incorporated herein by reference.
| Item 12. | Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters |
Information with respect to this item may be found under the caption “Security Ownership of Certain Beneficial Owners and Management” and “Equity Compensation Plan Information” in the Proxy Statement for the Company’s 2010 Annual Meeting of Stockholders. Such information is incorporated herein by reference.
| Item 13. | Certain Relationships and Related Transactions, and Director Independence |
Information with respect to this item may be found under the captions “Certain Relationships and Related Transactions” in the Proxy Statement for the Company’s 2010 Annual Meeting of Stockholders. Such information is incorporated herein by reference.
| Item 14. | Principal Accounting Fees and Services |
Information with respect to this item may be found under the caption “Fees Paid to Independent Registered Public Accounting Firm” in the Proxy Statement for the Company’s 2010 Annual Meeting of Stockholders. Such information is incorporated herein by reference.
73
Table of Contents
| Item 15. | Exhibits, Financial Statement Schedules |
(a) The following documents are filed as part of this report:
(1) FINANCIAL STATEMENTS
The following financial statements and supplementary data are included in Item 8 of this annual report:
| 70-71 | ||
| Consolidated Statements of Income for each of the three years ended June 30, 2010, 2009 and 2008 |
45 | |
| 46 | ||
| 47 | ||
| Consolidated Statements of Cash Flows for each of the three years ended June 30, 2010, 2009 and 2008 |
48 | |
| 49 | ||
| (2) FINANCIAL STATEMENT SCHEDULES |
||
| Schedule II – Valuation and Qualifying Accounts and Reserves |
||
| 76 | ||
All other schedules are omitted because they are not applicable or the required information is shown in the consolidated financial statements or Notes thereto.
(3) EXHIBITS
The list of Exhibits filed as a part of this Annual Report on Form 10-K are set forth on the Exhibit Index immediately preceding such Exhibits, and is incorporated herein by this reference.
74
Table of Contents
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| PAREXEL INTERNATIONAL CORPORATION | ||||||||
| By: | /s/ Josef H. von Rickenbach |
Dated: August 27, 2010 | ||||||
| Josef H. von Rickenbach |
||||||||
| Chairman of the Board and Chief Executive Officer | ||||||||
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
| Signatures |
Title(s) |
Date | ||
| /s/ Josef H. von Rickenbach Josef H. von Rickenbach |
Chairman of the Board and Chief Executive Officer (principal executive officer) |
August 27, 2010 | ||
| /s/ A. Dana Callow, Jr. A. Dana Callow, Jr. |
Director |
August 27, 2010 | ||
| /s/ Patrick J. Fortune Patrick J. Fortune |
Director |
August 27, 2010 | ||
| /s/ Eduard E. Holdener Eduard E. Holdener |
Director |
August 27, 2010 | ||
| /s/ Christopher J. Lindop Christopher J. Lindop |
Director |
August 27, 2010 | ||
| /s/ Richard L. Love Richard L. Love |
Director |
August 27, 2010 | ||
| /s/ James F. Winschel, Jr. James F. Winschel, Jr. |
Senior Vice President and Chief Financial Officer (principal financial and accounting officer) |
August 27, 2010 | ||
| /s/ Ellen M. Zane Ellen M. Zane |
Director |
August 27, 2010 | ||
75
Table of Contents
EXHIBIT INDEX
SCHEDULE II
PAREXEL INTERNATIONAL CORPORATION
VALUATION AND QUALIFYING ACCOUNTS AND RESERVES
| (in thousands) |
Balance at beginning of year |
Charged to costs and expenses |
Other adjustments* |
Balance at end of year | |||||||||
| Provision for losses on receivables |
|||||||||||||
| Year ended June 30, 2008 |
$ | 3,716 | $ | 2,349 | $ | (606 | ) | $ | 5,459 | ||||
| Year ended June 30, 2009 |
$ | 5,459 | $ | 14,543 | $ | 3,217 | $ | 23,219 | |||||
| Year ended June 30, 2010 |
$ | 23,219 | $ | 2,567 | $ | (12,770 | ) | $ | 13,015 | ||||
| * | Other adjustments denote the effects of foreign currency exchange, write-offs, and recoveries. |
| (in thousands) |
Balance at beginning of year |
Charged to income tax expense |
Other adjustments* |
Balance at end of year | ||||||||||
| Valuation allowance for deferred tax assets |
||||||||||||||
| Year ended June 30, 2008 |
$ | 47,588 | $ | (9,540 | ) | $ | (13,519 | ) | $ | 24,529 | ||||
| Year ended June 30, 2009 |
$ | 24,529 | $ | 2,872 | $ | 4,044 | $ | 31,445 | ||||||
| Year ended June 30, 2010 |
$ | 31,445 | $ | (1,739 | ) | $ | (8,036 | ) | $ | 21,670 | ||||
| * | Other adjustments denote the effects of foreign currency exchange, write-offs, recoveries, acquisitions and certain reclassifications related to ASC 740. |
76
Table of Contents
EXHIBIT INDEX
| EXHIBIT NO. |
DESCRIPTION | |
| 3.1 | Amended and Restated Articles of Organization of the Company, as amended. (filed as Exhibit 3.1 to the Company’s Quarterly Report on Form 10-Q for the quarterly period ended December 31, 2006 and incorporated herein by this reference). | |
| 3.2 | Amended and Restated By-laws of the Company, as amended (filed as Exhibit 3.2 to the Company’s Quarterly Report on Form 10-Q for the quarterly period ended September 30, 2009 and incorporated herein by this reference). | |
| 4.1 | Specimen certificate representing the Common Stock of the Company (filed as Exhibit 4.1 to the Company’s Registration Statement on Form S-1 (File No. 33-97406) and incorporated herein by this reference). | |
| 4.2 | Rights Agreement dated March 27, 2003 between the Corporation and Equiserve Trust Company, N.A., as Rights Agent, which includes as Exhibit A the Form of Certificate of Vote of Series A Junior Participating Preferred Stock, as Exhibit B the Form of Rights Certificate and as Exhibit C the Summary of Rights to Purchase Common Stock (filed as Exhibit 4.1 to the Company’s Current Report on Form 8-K/A dated March 31, 2003 and incorporated herein by this reference). | |
| 10.1* | Form of Stock Option Agreement of the Company (filed as Exhibit 10.9 to the Company’s Registration Statement on Form S-1 (File No. 333-1188) and incorporated herein by reference). | |
| 10.2* | Form of Stock Option Agreement under the Company’s Second Amended and Restated 1995 Stock Plan (filed as Exhibit 99.1 to the Company’s current Report on Form 8-K dated September 2, 2004 and incorporated herein by this reference). | |
| 10.3* | Form of Stock Option Agreement for Executive Officers under the Company’s Second Amended and Restated 1995 Stock Plan (filed as Exhibit 10.3 to the Company’s Annual Report on Form 10-K for the year ended June 30, 2004 and incorporated herein by this reference). | |
| 10.4* | Form of Stock Option Agreement under the Company’s 2001 Stock Incentive Plan (filed as Exhibit 10.4 to the Company’s Annual Report on Form 10-K for the year ended June 30, 2004 and incorporated herein by this reference). | |
| 10.5* | Second Amended and Restated 1995 Stock Plan of the Company (filed as Exhibit 10.9 to the Company’s Annual Report on Form 10-K for the year ended June 30, 1998 and incorporated herein by this reference). | |
| 10.6* | 1995 Non-Employee Director Stock Option Plan of the Company (filed as Exhibit 10.14 to the Company’s Registration Statement on Form S-1 (File No. 33-97406) and incorporated herein by this reference). | |
| 10.7.1* | 2001 Stock Incentive Plan of the Company (filed as Exhibit 10.7.1 to the Company’s Annual Report on Form 10-K for the year ended June 30, 2006 and incorporated herein by this reference). | |
| 10.7.2* | Amendment No. 1 to 2001 Stock Incentive Plan of the Company (filed as Exhibit 10.7.2 to the Company’s Annual Report on Form 10-K for the year ended June 30, 2006 and incorporated herein by this reference). | |
| 10.8* | 2005 Stock Incentive Plan of the Company (filed as Exhibit 10.8 to the Company’s Annual Report on Form 10-K for the year ended June 30, 2006 and incorporated herein by this reference). | |
| 10.9* | Form of Restricted Stock Agreement for non-employee directors under the Company’s 2005 Stock Incentive Plan (filed as Exhibit 10.2 to the Company’s Quarterly Report on Form 10-Q for the quarter ended December 31, 2009 and incorporated herein by this reference). | |
| 10.10* | Form of Restricted Stock Agreement for executive officers under the Company’s 2005 Stock Incentive Plan (filed as Exhibit 10.3 to the Company’s Quarterly Report on Form 10-Q for the quarter ended December 31, 2009 and incorporated herein by this reference). | |
| 10.11* | Corporate Plan for Retirement of the Company (filed as Exhibit 10.16 to the Company’s Registration Statement on Form S-1 (File No. 33-97406) and incorporated herein by this reference). | |
| 10.12.1 | Lease dated June 14, 1991 between 200 West Street Limited Partnership and the Company (filed as Exhibit 10.19 to the Company’s Annual Report on Form 10-K for the year ended June 30, 2000 and incorporated herein by this reference). | |
| 10.12.2 | First Amendment dated as of January 3, 1992 to the Lease dated June 14, 1991 between 200 West Street Limited Partnership and the Company (filed as Exhibit 10.25 to the Company’s Registration Statement on Form S-1 (File No. 33-97406) and incorporated herein by this reference). | |
| 10.12.3 | Second Amendment dated as of June 28, 1993 to the Lease dated June 14, 1991 between 200 West Street Limited Partnership and the Company (filed as Exhibit 10.28 to the Company’s Registration Statement on Form S-1 (File No. 33-97406) and incorporated herein by this reference). | |
77
Table of Contents
| EXHIBIT NO. |
DESCRIPTION | |
| 10.12.4 | Third Amendment to Lease dated November 17, 1998 between Boston Properties Limited Partnership and the Company (filed as Exhibit 10.1 to the Company’s Quarterly Report on Form 10-Q for the quarter ended December 31, 1998 and incorporated herein by this reference). | |
| 10.12.5 | Fifth Amendment dated as of June 29, 2007 to the lease dated June 14, 1991 by and between Boston Properties Limited Partnership and PAREXEL International, LLC (filed as Exhibit 10.12.5 to the Company’s Annual Report on Form 10-K for the year ended June 30, 2007 and incorporated herein by this reference). | |
| 10.13* | 1998 Non-Qualified, Non-Officer Stock Option Plan, as amended (filed as Exhibit 10.16 to the Company’s Annual Report on Form 10-K for the year ended June 30, 1999 and incorporated herein by this reference). | |
| 10.14* | Amended and Restated Employment Agreement dated April 15, 2008 between Josef H. von Rickenbach and the Company (filed as Exhibit 10.1 to the Company’s Quarterly Report on Form 10-Q for the quarter ended March 31, 2008 and incorporated herein by this reference). | |
| 10.15.1 | Lease dated November 17, 1998 between Boston Properties Limited Partnership and the Company (filed as Exhibit 10.2 to the Company’s Quarterly Report on Form 10-Q for the quarter ended December 31, 1998 and incorporated herein by this reference). | |
| 10.15.2 | First Amendment dated as of June 29, 2007 to the Lease dated November 17, 1998 by and between Boston Properties Limited Partnership and PAREXEL International LLC (filed as Exhibit 10.15.2 to the Company’s Annual Report on Form 10-K for the year ended June 30, 2007 and incorporated herein by this reference). | |
| 10.16* | Employment Agreement, dated February 21, 2005, between Ulf Schneider and PAREXEL GmbH (filed as Exhibit 10.1 to the Company’s Current Report on Form 8-K dated February 21, 2005 and incorporated herein by this reference). | |
| 10.17 | Fourth Amendment dated August 28, 2000 to the Lease dated November 17, 1998 between Boston Properties Limited Partnership and the Company (filed as Exhibit 10.22 to the Company’s Annual Report on Form 10-K for the year ended June 30, 2000 and incorporated herein by this reference). | |
| 10.18.1* | Amended and Restated Change of Control/Severance Agreement, dated as of April 15, 2008, by and between the Company and James F. Winschel, Jr. (filed as Exhibit 10.2 to the Company’s Quarterly Report on Form 10-Q for the quarter ended March 31, 2008 and incorporated herein by this reference). | |
| 10.19.1* | Amended and Restated Change of Control/Severance Agreement, dated as of October 31, 2008, by and between the Company and Mark A. Goldberg (filed as Exhibit 10.1 to the Company’s Current Report on Form 8-K dated October 31, 2008 and incorporated herein by reference). | |
| 10.20 | PAREXEL International Nonqualified Deferred Compensation Plan (filed as Exhibit 10.21 to the Company’s Annual Report on Form 10-K for the year ended June 30, 2002 and incorporated herein by this reference). | |
| 10.21 | ABN – AMRO Cash Pooling Agreement, dated as of September 14, 2001 (filed as Exhibit 10.21 to the Company’s Annual Report on Form 10-K for the year ended June 30, 2003 and incorporated herein by this reference). | |
| 10.22 | Lease dated April 10, 2002 between API (No. 23) Limited, Arlington Property Investments Limited, PAREXEL International Limited and the Company (filed as Exhibit 10.22 to the Company’s Annual Report on Form 10-K for the year ended June 30, 2003 and incorporated herein by this reference). | |
| 10.23* | Amended and Restated Change of Control/Severance Agreement, dated as of April 15, 2008, by and between the Company and Douglas A. Batt (filed as Exhibit 10.26 to the Company’s Quarterly Report on Form 10-Q for the quarter ended March 31, 2008 and incorporated herein by this reference). | |
| 10.24* | Change of Control/Severance Agreement, dated as of April 15, 2008, by and between PAREXEL International Corporation and Kurt A. Brykman (filed as Exhibit 10.5 to the Company’s Quarterly Report on Form 10-Q for the quarter ended March 31, 2008 and incorporated herein by reference). | |
| 10.25 | Amended and Restated Credit Agreement, dated as of September 18, 2007, between and among the Company and PAREXEL International Holding B.V., as Borrowers, certain subsidiaries of the Borrowers, the Lenders party thereto, JPMorgan Chase Bank, National Association, as Administrative Agent, J.P. Morgan Europe Limited, as London Agent, J.P. Morgan Securities Inc., as Sole Bookrunner and Sole Lead Arranger, ABN Amro Bank N.V., as Syndication Agent and Wachovia Bank, N.A., as Documentation Agent (filed as Exhibit 10.1 to the Company’s Quarterly Report on Form 10-Q for the quarter ended September 30, 2007 and incorporated herein by reference). | |
78
Table of Contents
| EXHIBIT NO. |
DESCRIPTION | |
| 10.26 | Credit Agreement dated as of June 13, 2008, as amended by the First Amendment dated as of July 10, 2008, and as amended and restated as of August 14, 2008, by and among the Company, certain subsidiaries of the Company, JPMorgan Chase Bank, N.A., as Administrative Agent, J.P. Morgan Europe Limited, as London Agent, KeyBank National Association, as Syndication Agent, HSBC Bank USA, N.A., RBS Citizens, N.A. and Fifth Bank, as Co-Documentation Agents, and the lenders party thereto (filed as Exhibit 10.1 to the Company’s Current Report on Form 8-K dated September 2, 2008 and incorporated herein by reference). | |
| 10.26.1 | Amendment and Waiver dated August 11, 2008, by and among the Company, JPMorgan Chase Bank, N.A., as Administrative Agent, and certain lenders to the Credit Agreement dated as of June 13, 2008 (as amended, restated or otherwise modified from time to time), by and among the Company, certain subsidiaries of the Company, the Administrative Agent, J.P. Morgan Europe Limited, as London Agent, and Keybank National Association, as Syndication Agent (filed as Exhibit 10.2 to the Company’s Current Report on Form 8-K dated August 13, 2008 and incorporated herein by reference). | |
| 10.26.2 | First Amendment dated as of December 19, 2008 to the Credit Agreement dated as of June 13, 2008, as amended and restated as of August 14, 2008 (as amended, restated or otherwise modified from time to time, the “Credit Agreement”), among the Company, PAREXEL INTERNATIONAL HOLDING B.V., a subsidiary of the Company that is a private company with limited liability (besloten vennootschap met beperkte aansprakelijkheid) (the “Dutch Borrower”), PAREXEL INTERNATIONAL HOLDING UK LIMITED, a subsidiary of the Company that is a company incorporated in England and Wales (“Bidco”; and together with the Company, the Dutch Borrower and other Persons who are or hereafter are designated as Borrowers pursuant to Section 2.21 of the Credit Agreement, the “Borrowers”), the Subsidiaries of the Borrowers party thereto, the lenders party thereto (the “Lenders”), JPMORGAN CHASE BANK, N.A., as Administrative Agent, J.P. MORGAN EUROPE LIMITED, as London Agent, and KEYBANK NATIONAL ASSOCIATION, as Syndication Agent (filed as Exhibit 10.1 to the Company’s Current Report on Form 8-K dated December 24, 2008 and incorporated herein by reference). | |
| 10.27* | 2007 Stock Incentive Plan (filed as Exhibit 10.1 to the Company’s Quarterly Report on Form 10-Q for the quarter ended December 31, 2007 and incorporated herein by reference). | |
| 10.28* | Perceptive Informatics President Long-Term Discretionary Incentive Plan (filed as Exhibit 10.1 to the Company’s Quarterly Report on Form 10-Q for the quarterly period ended March 31, 2010 and incorporated herein by this reference) | |
| 10.29* | Change of Control/severance Agreement, dated as of November 5, 2009, by and between the Company and Ulf Schneider (filed as Exhibit 10.1 to the Company’s Current Report on Form 8-K dated November 5, 2009, and incorporated herein by this reference). | |
| 10.30* | Form of Restricted Stock Unit Agreement for executive officers under the Company’s 2007 Stock Incentive Plan. | |
| 21.1 | List of subsidiaries of the Company | |
| 23.1 | Consent of Ernst & Young LLP | |
| 31.1 | CEO certification pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 | |
| 31.2 | CFO certification pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 | |
| 32.1 | CEO certification pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 | |
| 32.2 | CFO certification pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 | |
| * | denotes management contract or any compensatory plan, contract or arrangement |
79
