As filed with the Securities and Exchange Commission on
June 18, 2010.
Registration
No. 333-166400
SECURITIES AND EXCHANGE
COMMISSION
Washington, D.C.
20549
Amendment No. 1
to
Form S-1
REGISTRATION
STATEMENT
UNDER
THE SECURITIES ACT OF
1933
AURORA DIAGNOSTICS,
INC.
(Exact Name of Registrant as
Specified in its Charter)
| |
|
|
|
|
Delaware
(State or Other Jurisdiction
of
Incorporation or Organization)
|
|
8071
(Primary Standard
Industrial
Classification Code Number)
|
|
27-2416884
(I.R.S. Employer
Identification Number)
|
11025 RCA
Center Drive, Suite 300
Palm Beach Gardens, FL 33410
(866) 420-5512
(Address, including zip code,
and telephone number, including area code, of registrant’s
principal executive offices)
Gregory
A. Marsh
Chief Financial Officer
11025 RCA Center Drive, Suite 300
Palm Beach Gardens, FL 33410
(866) 420-5512
(Name, address, including zip
code, and telephone number, including area code, of agent for
service)
Copies to:
| |
|
|
J. Mark Ray
Alston & Bird LLP
1201 West Peachtree Street
Atlanta, GA
30309-3424
(404) 881-7000
|
|
Michael Benjamin
Shearman & Sterling LLP
599 Lexington Avenue
New York, NY 10022
(212) 848-4000
|
Approximate date of commencement of proposed sale to the
public: As soon as practicable after this
Registration Statement becomes effective.
If any of the securities being registered on this Form are to be
offered on a delayed or continuous basis pursuant to
Rule 415 under the Securities Act, check the following
box. o
If this Form is filed to register additional securities for an
offering pursuant to Rule 462(b) under the Securities Act,
please check the following box and list the Securities Act
registration statement number of the earlier effective
registration statement for the same
offering. o
If this Form is a post-effective amendment filed pursuant to
Rule 462(c) under the Securities Act, check the following
box and list the Securities Act registration statement number of
the earlier effective registration statement for the same
offering. o
If this Form is a post-effective amendment filed pursuant to
Rule 462(d) under the Securities Act, check the following
box and list the Securities Act registration statement number of
the earlier effective registration statement for the same
offering. o
Indicate by check mark whether the registrant is a large
accelerated filer, an accelerated filer, a non-accelerated
filer, or a smaller reporting company. See the definitions of
“large accelerated filer,” “accelerated
filer” and “smaller reporting company” in Rule
12b-2 of the
Exchange Act. (Check one):
|
|
|
|
| Large
accelerated
filer o
|
Accelerated
filer o
|
Non-accelerated
filer þ
|
Smaller reporting
company o
|
(Do not check if a smaller
reporting company)
The Registrant hereby amends this Registration Statement on
such date or dates as may be necessary to delay its effective
date until the Registrant shall file a further amendment which
specifically states that this Registration Statement shall
thereafter become effective in accordance with Section 8(a)
of the Securities Act of 1933 or until the Registration
Statement shall become effective on such date as the Commission
acting pursuant to said Section 8(a) may determine.
The
information in this prospectus is not complete and may be
changed. Neither we nor the selling stockholders may sell these
securities until the registration statement filed with the
Securities and Exchange Commission is effective. This prospectus
is not an offer to sell these securities, and neither we nor the
selling stockholders are soliciting offers to buy these
securities in any state where the offer or sale is not
permitted.
|
PROSPECTUS
(Subject to Completion)
Issued
June 18, 2010
Shares
Class A Common Stock
Aurora Diagnostics, Inc. is
offering
shares of its Class A common stock and the selling
stockholders are
offering
shares of Class A common stock. We will not receive any
proceeds from the sale of shares by the selling stockholders.
This is our initial public offering and no public market exists
for our shares. We anticipate that the initial public offering
price will be between $ and
$ per share.
We have applied to have our Class A common stock listed
on the NASDAQ Global Market under the symbol
“ARDX.”
Investing in our Class A common stock involves risks.
See “Risk Factors” beginning on page 15.
Price
$
Per Share
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Underwriting
|
|
|
|
Proceeds to
|
|
|
|
Price to
|
|
Discounts and
|
|
Proceeds to
|
|
Selling
|
|
|
|
Public
|
|
Commissions
|
|
Us
|
|
Stockholders
|
|
|
|
Per share
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
Total
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
We and the selling stockholders have granted the underwriters
the right to purchase up to an
additional shares
of Class A common stock to cover over-allotments.
The Securities and Exchange Commission and state securities
regulators have not approved or disapproved these securities or
determined if this prospectus is truthful or complete. Any
representation to the contrary is a criminal offense.
The underwriters expect to deliver the shares of Class A
common stock to purchasers
on ,
2010.
|
|
|
| Morgan
Stanley |
UBS Investment Bank |
Barclays Capital |
|
|
|
| RBC
Capital Markets |
BMO Capital Markets |
Lazard Capital Markets |
,
2010
TABLE OF
CONTENTS
| |
|
|
|
|
|
|
|
Page
|
|
|
|
|
|
|
1
|
|
|
|
|
|
15
|
|
|
|
|
|
35
|
|
|
|
|
|
37
|
|
|
|
|
|
44
|
|
|
|
|
|
44
|
|
|
|
|
|
45
|
|
|
|
|
|
47
|
|
|
|
|
|
49
|
|
|
|
|
|
56
|
|
|
|
|
|
59
|
|
|
|
|
|
84
|
|
|
|
|
|
88
|
|
|
|
|
|
108
|
|
|
|
|
|
115
|
|
|
|
|
|
123
|
|
|
|
|
|
128
|
|
|
|
|
|
135
|
|
|
|
|
|
139
|
|
|
|
|
|
141
|
|
|
|
|
|
145
|
|
|
|
|
|
149
|
|
|
|
|
|
149
|
|
|
|
|
|
150
|
|
|
|
|
|
F-1
|
|
| EX-23.1 |
| EX-23.2 |
You should rely only on the information contained in this
prospectus and in any free writing prospectus. We, the
underwriters and the selling stockholders have not authorized
anyone to provide you with information different from that
contained in this prospectus. We, the underwriters and the
selling stockholders are offering to sell, and seeking offers to
buy, shares of our Class A common stock only in
jurisdictions where offers and sales are permitted. The
information in this prospectus is accurate only as of the date
of this prospectus, regardless of the time of delivery of this
prospectus or any sale of shares of our Class A common
stock.
Until ,
2010 (25 days after the commencement of this offering), all
dealers that buy, sell or trade shares of our Class A
common stock, whether or not participating in this offering, may
be required to deliver a prospectus. This delivery requirement
is in addition to the obligation of dealers to deliver a
prospectus when acting as underwriters and with respect to their
unsold allotments or subscriptions.
For investors outside of the United States, neither we, nor the
selling stockholders or any of the underwriters have done
anything that would permit this offering or possession or
distribution of this prospectus in any jurisdiction where action
for that purpose is required. Persons outside the United States
who come into possession of this prospectus must inform
themselves about, and observe any restrictions relating to, the
offering of the shares of Class A common stock and the
distribution of this prospectus outside of the United States.
INDUSTRY
AND MARKET DATA
Industry and market data used throughout this prospectus were
obtained through company research, surveys and studies conducted
by third parties and industry and general publications. The
information contained in “Prospectus Summary,”
“Management’s Discussion and Analysis of Financial
Condition and Results of Operations,” “Industry”
and “Business” is based on studies, analyses and
surveys including:
|
|
|
| |
•
|
“Laboratory Industry Strategic Outlook: Market Trends and
Analysis 2009” prepared by Washington G-2 Reports, or the
Washington G-2 Report;
|
|
|
|
| |
•
|
“The U.S. Anatomic Pathology Market Forecast & Trends
2010” prepared by Laboratory Economics, or the Laboratory
Economics Report;
|
|
|
|
| |
•
|
“Cancer Facts & Figures 2009” prepared by the
American Cancer Society, or the American Cancer Society Report;
|
|
|
|
| |
•
|
“Trendwatch Chartbook 2000: Trends Affecting Hospitals and
Health Systems” prepared by the Lewin Group, Inc. for the
American Hospital Association and “Trendwatch Chartbook
2009: Trends Affecting Hospitals and Health Systems”
prepared by Avalere Health for the American Hospital
Association, or the AHA Reports; and
|
|
|
|
| |
•
|
“Projections of the Population by Selected Age Groups and
Sex for the United States: 2010 to 2050” prepared by the
Population Division, U.S. Census Bureau, or the U.S. Census
Bureau Report.
|
Information originally published in Washington G-2 Reports
“Laboratory Industry Strategic Outlook: Market Trends
& Analysis 2009” is used herein with the express
written permission of Washington G2 Reports. Copyright
©2010.
www.g2reports.com. While we are not aware of any misstatements
regarding the industry data presented herein, estimates involve
risks and uncertainties and are subject to change based on
various factors, including those discussed under the heading
“Risk Factors.”
TRADEMARKS
AURORA DIAGNOSTICS and CONNECTDX THE INFORMATION GATEWAY and
Design and other trademarks or service marks of Aurora appearing
in this prospectus are our property. All trade names, trademarks
and service marks of other companies appearing in this
prospectus are the property of the respective holders.
PROSPECTUS
SUMMARY
This summary highlights all material information about us and
this offering, but does not contain all of the information that
you should consider before investing in our Class A common
stock. You should read this entire prospectus carefully,
including the “Risk Factors” and the consolidated
financial statements and related notes. This prospectus includes
forward-looking statements that involve risks and uncertainties.
See “Forward-Looking Statements.”
Unless we state otherwise or the context otherwise requires,
the terms “we,” “us,” “our,”
“Aurora Diagnostics” and the “Company” refer
to Aurora Diagnostics, Inc. and our subsidiaries, as well as the
professional associations and professional corporations which
are separate legal entities in certain states that we control
through contractual arrangements. All information in this
prospectus with respect to Aurora Diagnostics, Inc. gives effect
to the reorganization transactions described under
“Prospectus Summary — Reorganization
Transactions” and “Organizational Structure” as
if they had occurred on December 31, 2009. “Aurora
Holdings” refers to our subsidiary “Aurora Diagnostics
Holdings, LLC.” With respect to periods prior to
April 23, 2010, the terms “we,” “us,”
“our,” “Aurora” and the “Company”
refer to Aurora Holdings and its subsidiaries.
Our
Business
We are a specialized diagnostics company providing services that
play a key role in the diagnosis of cancer and other diseases.
Our experienced pathologists deliver comprehensive diagnostic
reports of a patient’s condition and consult frequently
with referring physicians to help determine the appropriate
treatment. Our diagnostic reports often enable the early
detection of disease, allowing referring physicians to make
informed and timely treatment decisions that improve their
patients’ health in a cost-effective manner.
We are a leading specialized diagnostics company in terms of
revenues, focused on the anatomic pathology market. We are
well-positioned in the higher-growth subspecialties of anatomic
pathology, with a leading market position in dermatopathology
and in the women’s health pathology subspecialty, and a
growing market position in urologic pathology, hematopathology
and general surgical pathology. Our strengths in anatomic
pathology are complemented by our specialized clinical and
molecular diagnostics offerings, which enable us to provide a
broad selection of diagnostic services to our referring
physicians, our primary clients.
The majority of our revenues in 2009 were derived from
physicians providing diagnostic services in the non-hospital
outpatient channel of the anatomic pathology market, which in
2008 was one of the fastest-growing and largest channels of that
market. We also maintain 36 exclusive contracts with hospitals
under which we provide inpatient and outpatient professional
anatomic pathology services. We also provide medical director
services and, for some hospitals, technical slide preparation
services.
Our business model builds upon the expertise of our experienced
pathologists to provide seamless, reliable and comprehensive
pathology and molecular diagnostics offerings to referring
physicians. We typically have established, long-standing
relationships with our referring physicians as a result of
focused localized delivery of diagnostic services, personalized
responses and frequent consultations, and flexible information
technology, or IT, solutions that are customizable to our
clients’ needs. Our IT and communications platform enables
us to deliver diagnostic reports to our clients generally within
24 hours of specimen receipt, helping to improve patient
care. In addition, our IT platform enables us to closely track
and monitor volume trends from referring physicians.
The success of our business model and the value of our
specialized diagnostic service offering are reflected in our
significant growth allowing us to reach $171.6 million in
annual revenues in 2009. Through a combination of organic growth
and strategic acquisitions, we have achieved a scale allowing us
to provide diagnostic services to the patients of our
approximately 10,000 referring physicians, generating
approximately 1.6 million accessions in 2009. With 19
primary laboratories across the United States, we have achieved
a national footprint and a leading presence in our local markets
upon which we are continuing to build a more integrated and
larger-scale diagnostics company.
1
Our
Industry
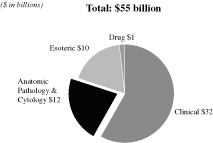
The U.S. diagnostic testing industry had revenues of
approximately $55 billion in 2008 and grew at a rate of
7 percent compounded annually from 2000 to 2008, according
to the Washington G-2 Report. Within the overall industry in
2008, the anatomic pathology market totaled approximately
$13 billion in revenues, or 24 percent of total
industry revenues, according to the Laboratory Economics Report.
Anatomic pathology services involve the diagnosis of cancer and
other medical conditions through the examination of tissues
(histology) and the analysis of cells (cytology) and generally
command higher reimbursement rates, on a per specimen basis,
than clinical pathology services.
According to the Washington G-2 Report, the anatomic pathology
market has expanded more rapidly than the overall industry, with
revenues growing 4.8 percent on a compound annual basis
between 2006 and 2009, compared to 4.5 percent for the rest
of the industry. Excluding growth in esoteric testing, the
remainder of the industry grew at a compound annual rate of only
0.1 percent over the same period. Substantially all of the
revenues for anatomic pathology businesses consist of payments
or reimbursements for specialized diagnostic services rendered
to referring physicians, and these revenues are affected
primarily by changes in case volume, which we refer to as
accession volume, payor mix and reimbursement rates.
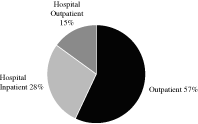
The non-hospital outpatient channel is the largest component of
the anatomic pathology market and has grown more rapidly than
other channels. This channel accounted for $7.6 billion, or
57 percent, of anatomic pathology revenues for the year
ended December 31, 2008, representing 10 percent
growth in 2008 according to the Laboratory Economics Report. The
remainder of the anatomic pathology market is comprised of the
hospital inpatient channel, which accounted for
$3.7 billion or 28 percent, of anatomic pathology
revenues, representing 2 percent growth in 2008, and the
hospital outpatient channel, which accounted for
$1.9 billion, or 15 percent, of anatomic pathology
revenues, representing 4 percent growth in 2008, according
to the Laboratory Economics Report.
We believe that demand for non-hospital outpatient anatomic
pathology services will continue to expand due to the following
trends:
|
|
|
| |
•
|
Aging of the U.S. Population: The number
of individuals
65-years-old
and older will increase to 55 million, or by
36 percent, over the next decade, a percentage rate that is
nearly four times faster than that of the overall population,
according to the U.S. Census Bureau Report. According to
the American Cancer Society Report, the risk of being diagnosed
with cancer increases as individuals age, with an estimated
52 percent of all new cancer cases diagnosed in persons
65-years-old
and older in 2009.
|
|
|
|
| |
•
|
Increasing Incidence of Cancer: The number of
new cancer cases grew by 2 percent between 2007 and 2009 to
approximately 1.5 million new cases according to the
American Cancer Society Report. The number of new skin cancer
cases grew by 15 percent between 2007 and 2009,
representing the highest percentage increase among all diagnosed
cancer types.
|
|
|
|
| |
•
|
Expanding Demand for Non-Hospital Outpatient
Services: The non-hospital outpatient channel of
the anatomic pathology market is expected to continue growing at
a higher percentage rate than the overall industry, principally
driven by patient preference and the cost-effectiveness of
outpatient diagnostic services compared to inpatient diagnostic
services.
|
| |
| |
•
|
Medical Advancements Allowing for Earlier Diagnosis and
Treatment of Disease: Physicians are increasingly
relying on diagnostic testing to help identify the risk of
disease, to detect the symptoms of disease earlier, to aid in
the choice of therapeutic regimen, to monitor patient compliance
and to evaluate treatment results. We believe physicians,
patients and payors increasingly recognize the value of
diagnostic testing as a means to improve health and reduce the
overall cost of health care through early detection.
|
The anatomic pathology market remains highly-fragmented, with
the two largest clinical laboratory companies accounting for
only 14 percent of annual revenues for the market in 2008.
The remaining 86 percent of annual revenues for the market
was comprised of over 13,000 pathologists and numerous
specialized testing companies that offer a relatively narrow
menu of diagnostic tests. In 2008, approximately 70 percent
of pathologists licensed in the U.S. were in private
practice according to the Washington G-2 Report. As a result, we
believe that
2
there are substantial consolidation opportunities in the
anatomic pathology market as smaller operators seek access to
the resources of diagnostics companies with a more comprehensive
selection of services for referring physicians.
Our
Competitive Strengths
We believe that we are distinguished by the following
competitive strengths:
|
|
|
| |
•
|
Leading Market Position in Higher-Growth Subspecialties of
Expanding Industry. We are a leading specialized
diagnostics company, focused on the faster-growing non-hospital
outpatient channel within the anatomic pathology market with
leading market positions in two of the three higher-growth
subspecialties of the market: dermatopathology and women’s
health pathology.
|
| |
| |
•
|
Locally-Focused Business Model with National
Scale. Our business model centers on achieving
significant local market share, which yields operating
efficiencies and national scale when consolidated across all of
our operations. The diagnostic services we provide are designed
specifically to meet the needs of the local markets we serve.
Our national infrastructure enables us to more efficiently
manage our operations, improve productivity and deliver a more
extensive menu of diagnostic services to our local clients. As a
result of our strong local presence and high-quality diagnostic
services, we have established significant loyalty with referring
physicians and key payors in our local markets. In 2009, we
derived more than 85 percent of our revenues from
locally-focused,
in-network
payor contracts.
|
| |
| |
•
|
Experienced, Specialized Pathologists Focused on Client
Service. We believe our pathologists have
long-standing client relationships and provide high-quality
service within their respective local communities. Over
one-third of our pathologists are specialized in
dermatopathology, with the remainder focused on women’s
health pathology, urologic pathology, hematopathology and
general surgical pathology. This alignment of our
pathologists’ specialties with those of the referring
physicians is critical to our ability to retain existing and
attract new clients. Our clinical expertise and frequent
interactions with clients on patient diagnoses enables us to
establish effective consultative and long-term relationships
with referring physicians.
|
| |
| |
•
|
Professional Sales, Marketing and Client Service
Team. We maintain a sales, marketing and client
service team of over 100 professionals who are highly-trained
and organized by subspecialty to better meet the needs of our
referring physicians and their patients. Our sales
representatives are incentivized through compensation plans to
not only secure new physician clients, but also to maintain and
enhance relationships with existing physician clients. As a
result, they have enabled us to expand our geographic market
presence to 30 states and increase market penetration and
market share in our local markets.
|
| |
| |
•
|
Proprietary IT Solutions. Delivery of clinical
information is essential to our business and a critical aspect
of the differentiated service that we provide to our clients. We
have developed scalable IT solutions that maximize the
flexibility,
ease-of-use
and speed of delivery of our diagnostic reports, which has
enabled us to rapidly grow our accession volume and meet the
increasing physician demand for our diagnostic services. We also
monitor referral patterns on a daily basis using our IT
infrastructure, which allows us to respond quickly to referring
physicians through our sales and marketing teams. We achieved
this through the development of a proprietary suite of IT
solutions called ConnectDX that is compatible with most
electronic medical record, or EMR, systems. ConnectDX
incorporates customized interface solutions, low cost and
efficient printer capabilities, compliant web portal capacities,
and proprietary software, all resulting in efficient and
reliable onsite client connections.
|
| |
| |
•
|
Proven Acquisition, Integration and Development
Capabilities. We have significant expertise and a
proven track record of identifying, acquiring and integrating
pathology practices into our diagnostic laboratory network. Our
management team successfully expanded our operations through the
acquisition of 16 anatomic pathology laboratories and one
clinical laboratory and through the development of two de
novo diagnostic laboratories. We have improved the performance
of the laboratories we have acquired by applying our standard
operating procedures, enhancing sales and marketing
capabilities, implementing our IT platform and realizing
efficiencies from our national operations and management. We
believe our
|
3
|
|
|
| |
|
operational platform, expertise and value proposition enable us
to capitalize on the considerable consolidation opportunities in
the highly-fragmented anatomic pathology market.
|
|
|
|
| |
•
|
Experienced Senior Leadership. Our senior
management team has approximately 80 years of combined
experience in the health care industry, including senior
management positions with leading diagnostic companies including
AmeriPath, DIANON Systems and Laboratory Corporation of America,
and collectively have successfully completed over 65
acquisitions and built a number of de novo specialized
diagnostic laboratories. We believe that our management’s
strong reputation, extensive network of industry relationships
and experience in building and growing successful companies in
the industry help us to drive operating performance, hire and
retain talented pathologists and other employees and attract
acquisition candidates.
|
Our
Business Strategy
We intend to achieve growth by pursuing the following strategies:
|
|
|
| |
•
|
Continue to Drive Market Penetration through Sales and
Marketing. We plan to drive organic growth
through our professional sales and marketing organization. Our
63-person
sales and marketing team provides us with broad coverage to
augment and further penetrate existing physician relationships
and to develop new referral relationships. We plan to
strategically add sales professionals to laboratories in markets
that will most benefit from enhanced outreach, increasing our
presence in existing and new markets.
|
| |
| |
•
|
Leverage our IT Platform to Increase Operating
Efficiencies. We believe our IT platform will
allow us to gain market share in our existing subspecialties by
improving productivity and reducing turnaround times. We have
recently introduced an IT solution called doc2MD, a
leading EMR system for dermatology practices for which we have
an exclusive, long-term license. We intend to continue to
develop our internal IT operations into a better-integrated
diagnostic platform, which will improve national coordination
and provide real time visibility into key performance metrics.
In addition, we plan to continue to introduce innovative IT
solutions, interface capabilities and market-specific IT
solutions that enhance our value proposition to referring
physicians.
|
| |
| |
•
|
Expand through Targeted Acquisitions. We plan
to identify and acquire leading laboratories to augment our
organic growth, broaden our geographic presence and enhance our
service offering. We intend to continue to build our business
and enhance our reputation as a preferred acquiror for
independent laboratories. We believe that our recognizable
identity and strong reputation make us a preferred partner for
independent laboratories.
|
| |
| |
•
|
Expand Diagnostic Services Capabilities. We
intend to expand our services in the areas of clinical and
molecular diagnostics to complement our existing anatomic
pathology businesses. We believe we can leverage our depth of
experience and physician relationships to sell these new
diagnostic services in conjunction with our existing testing
services as a comprehensive offering. As a “one-stop”
diagnostic services provider, we would not only better serve our
current clients, but also position ourselves to attract new
business under a more diverse service model.
|
| |
| |
•
|
Develop De Novo Diagnostics Laboratories. We
plan to continue to selectively develop diagnostic laboratories
on a de novo basis, as we have done in certain markets, to
expand our market presence, broaden our service offering and
leverage the capabilities of our existing laboratories and
pathologists.
|
| |
| |
•
|
Expand Contracts with Hospitals in Target
Markets. We intend to continue to develop
additional contracts with hospitals in target markets as part of
a broader strategy to strengthen and grow our outpatient
business and expand our local market share.
|
| |
| |
•
|
Further Expand into Growing Long-Term Care
Market. We have a growing presence providing
clinical diagnostic services to the long-term care markets in
Central and Northern Florida. We intend to expand this regional
coverage into the large South Florida market and replicate our
success in other states with growing long-term care markets. We
believe that our IT solutions, and our ability to meet the rapid
service requirements for the long term care market, provide us
with a significant competitive advantage in these markets.
|
4
Risks
Associated with Our Business
Our business is subject to numerous risks, which are highlighted
in the section entitled “Risk Factors.” These risks
represent challenges to the successful implementation of our
strategy and the growth of our business. Some of these risks are:
|
|
|
| |
•
|
Adverse changes in coverage or reimbursement guidelines and
amounts applicable to our specialized diagnostic services,
including health care reimbursement reform and cost-containment
measures implemented by government agencies or third-party
payors, could reduce our revenues and have a material adverse
impact on our business;
|
|
|
|
| |
•
|
If referring physicians, who are our primary clients, choose to
internalize technologies or technical or professional diagnostic
services functions that we currently use or perform, and we do
not develop new or alternative technologies or functions that
are attractive to our clients, it may reduce the demand for our
specialized diagnostic services and adversely affect our
business; and
|
|
|
|
| |
•
|
Recent and, if enacted, proposed federal or state health care
reform measures could increase our costs, decrease our revenues,
expose us to expanded liability or require us to revise the ways
in which we conduct our business, any of which could adversely
affect our operating results and financial condition.
|
For further discussion of these and other risks you should
consider before making an investment in our Class A common
stock, see the section entitled “Risk Factors”
beginning on page 16.
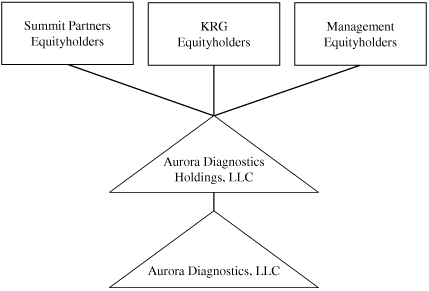
Corporate
History and Organizational Structure
We were incorporated in Delaware on April 23, 2010. We are
a holding company and our principal asset after the completion
of this offering will be our indirect equity interest in Aurora
Holdings. In June 2006, Aurora Holdings was organized as a
limited liability company and was initially capitalized by
affiliates of Summit Partners, affiliates of GSO Capital
Partners and members of our senior management team. In June
2009, GSO Capital Partners’ equity interest in Aurora
Holdings was purchased by an affiliate of KRG Capital Partners.
Prior to giving effect to the reorganization transactions
described below, affiliates of Summit Partners, whom we refer to
as the Summit Partners Equityholders, owned 51 percent of
the economic interest in Aurora Holdings; an affiliate of KRG
Capital Partners, whom we refer to as the KRG Equityholders,
owned 34 percent of the economic interest in Aurora
Holdings and members of our senior management team and
consultants, whom we refer to as the Management Equityholders,
owned 15 percent of the economic interest in Aurora
Holdings. We refer to the Summit Partners Equityholders, the KRG
Equityholders and the Management Equityholders as our Principal
Equityholders. We refer to membership interests in Aurora
Holdings as Aurora Holdings Units.
Reorganization
Transactions
In connection with this offering, we will enter into a series of
transactions as described below that will result in our indirect
acquisition of all of the business and operational control of
Aurora Holdings
and percent
of the Aurora Holdings Units. We refer to this series of
transactions as the Reorganization Transactions.
As part of the Reorganization Transactions, we will indirectly,
through a newly-formed, wholly-owned subsidiary, ARDX Sub, Inc.,
which we refer to as ARDX Sub, acquire Aurora Holdings Units
from certain of the Summit Partners Equityholders and the KRG
Equityholders in exchange for shares of our Class A common
stock, as well as rights, which we refer to as TRA Rights, under
an agreement that we refer to as the Tax Receivable Agreement.
This Tax Receivable Agreement will obligate Aurora Diagnostics,
Inc. to pay to an entity controlled by our Principal
Equityholders 85 percent of certain cash tax savings, if
any, realized by Aurora Diagnostics, Inc. after the completion
of this offering. See “Certain Relationships and Related
Party Transactions — Tax Receivable Agreement.”
Following the Reorganization Transactions, certain of the Summit
Partners Equityholders and all of the Management Equityholders,
whom we refer to as the Aurora Holdings Continuing Members, will
continue to own percent of
the Aurora Holdings Units. See “Organizational
Structure — Reorganization Transactions.” The KRG
Equityholders will not continue to own any Aurora Holdings Units
following the consummation of the
5
Reorganization Transactions. In addition, we will indirectly
acquire all of the business and operational control of Aurora
Holdings, and an immaterial amount of cash, from the Aurora
Holdings Continuing Members in exchange for newly-issued shares
of our Class B common stock. The Aurora Holdings Continuing
Members will also receive rights to distributions that are
calculated in a manner that is similar to the TRA Rights under
the Aurora Holdings limited liability company agreement, which
will be further amended and restated in connection with the
Reorganization Transactions. We refer to the Aurora Holdings
limited liability company agreement, as further amended and
restated, as the Second Amended and Restated Aurora Holdings LLC
Agreement. See “Organizational Structure —
Reorganization Transactions.” Our Class B common stock
is intended to provide voting rights in us to the Aurora
Holdings Continuing Members that reflect their continuing
economic interest in Aurora Holdings after the completion of
this offering.
All of these Reorganization Transactions will be consummated
immediately prior to the closing of this offering. We intend to
account for the Reorganization Transactions using a carryover
basis as the contemplated transactions are exchanges among
entities under common control that do not affect economic
ownership.
Following the completion of the Reorganization Transactions, we
will have two classes of common stock:
|
|
|
| |
•
|
Class A common stock. We will sell shares
of our Class A common stock in this offering. Each share of
our Class A common stock will be entitled to one vote on
matters submitted to a vote of our stockholders. Our
Class A common stock will represent all of the economic
rights in us (including rights to dividends and distributions
upon liquidation, but excluding the return of the par value of
the Class B common stock upon liquidation). Immediately
following consummation of the Reorganization Transactions, but
without giving effect to the sale of shares of our Class A
common stock by us or the selling stockholders in this offering:
|
|
|
|
| |
•
|
the Summit Partners Equityholders will
hold
shares of our Class A common stock;
|
| |
| |
•
|
the KRG Equityholders will
hold
shares of our Class A common stock; and
|
| |
| |
•
|
the Management Equityholders will hold no shares of our
Class A common stock.
|
|
|
|
| |
•
|
Class B common stock. The Aurora Holdings
Continuing Members will be the only holders of our Class B
common stock. Each share of our Class B common stock will
be entitled to one vote on matters submitted to a vote of our
stockholders. Our Class B common stock will only represent
voting rights and will not represent any economic rights in us
(including rights to dividends and distributions upon
liquidation, but excluding the return of the par value of the
Class B common stock upon liquidation). The Aurora Holdings
Continuing Members will hold one share of our Class B common
stock for each Aurora Holdings Unit held by them. Immediately
following consummation of the Reorganization Transactions, but
without giving effect to the purchase of shares of our
Class B common stock with a portion of the net proceeds
that we will receive from this offering:
|
|
|
|
| |
•
|
the Summit Partners Equityholders will
hold
shares of our Class B common stock;
|
| |
| |
•
|
the KRG Equityholders will hold no shares of our Class B
common stock; and
|
| |
| |
•
|
the Management Equityholders will
hold
shares of our Class B common stock.
|
See “Principal and Selling Stockholders” for more
information on the shares of our Class A common stock to be
offered by the selling stockholders in this offering.
6
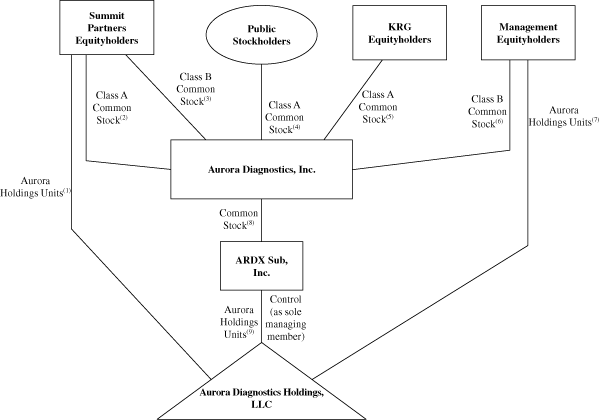
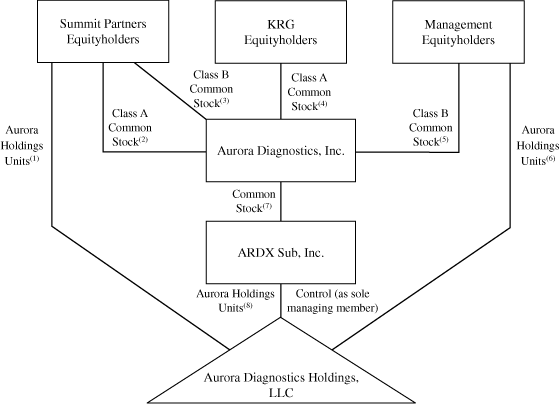
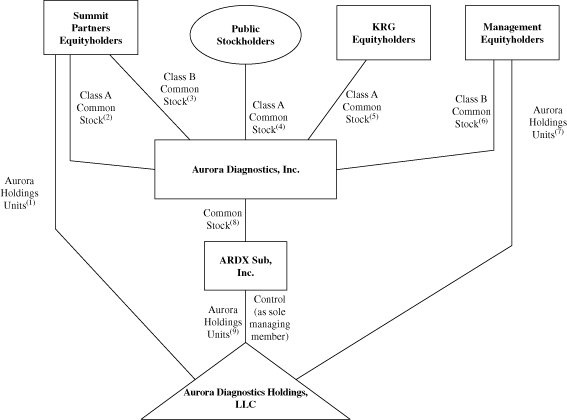
The diagram below depicts our organizational structure
immediately after giving effect to the Reorganization
Transactions, this offering and the application of the net
proceeds that we will receive from this offering.
|
|
|
|
(1)
|
|
The Summit Partners Equityholders
will
hold percent
of the Aurora Holdings Units.
|
|
(2)
|
|
The Summit Partners Equityholders
will
hold percent
of our Class A common stock.
|
|
(3)
|
|
The Summit Partners Equityholders
will
hold percent
of our Class B common stock.
|
|
(4)
|
|
Public stockholders will
hold percent
of our Class A common stock.
|
|
(5)
|
|
The KRG Equityholders will
hold percent
our Class A common stock.
|
|
(6)
|
|
The Management Equityholders will
hold percent
our Class B common stock.
|
|
(7)
|
|
The Management Equityholders will
hold percent
of the Aurora Holdings Units.
|
|
(8)
|
|
We will hold 100 percent of
ARDX Sub common stock.
|
|
(9)
|
|
ARDX Sub will
hold percent
of the Aurora Holdings Units.
|
Following the Reorganization Transactions, and after giving
effect to this offering and the application of the net proceeds
that we will receive from this offering, we will indirectly
hold percent
of the Aurora Holdings Units and will, through ARDX Sub, be the
sole managing member of Aurora Holdings. As the sole managing
member of Aurora Holdings, we will have all business and
operational control of Aurora Holdings and its subsidiaries. We
will consolidate the financial results of Aurora Holdings and
our net income (loss) will be reduced by the noncontrolling
interest expense to reflect the rights of the Aurora Holdings
Continuing Members with respect to their retained Aurora
Holdings Units.
As part of this offering, the Summit Partners Equityholders and
the KRG Equityholders will sell a portion of their shares of our
Class A common stock. We will not receive any of the
proceeds from the sale of shares of our Class A common
stock in this offering by the selling stockholders. Immediately
following the completion of this offering, we will use a portion
of the net proceeds that we will receive from this offering,
along with TRA Rights, to purchase shares of our Class B
common stock and Aurora Holdings Units from the Aurora Holdings
Continuing Members.
7
Tax
Receivable Agreement
In connection with this offering, Aurora Diagnostics, Inc. will
enter into the Tax Receivable Agreement with ARDX Sub, Aurora
Holdings and an entity controlled by our Principal
Equityholders, which we refer to as the Tax Receivable Entity,
to which our Principal Equityholders are contributing the TRA
Rights they receive in the Reorganization Transactions. This Tax
Receivable Agreement will provide for the payment by Aurora
Diagnostics, Inc. to the Tax Receivable Entity of
85 percent of certain cash tax savings, if any, in
U.S. federal, state, local and foreign income tax realized
by Aurora Diagnostics, Inc. after the completion of this
offering as a result of:
|
|
|
| |
•
|
favorable tax attributes associated with amortizable goodwill
and other intangibles held by Aurora Holdings and created by its
previous acquisitions;
|
| |
| |
•
|
any step-up
in tax basis in our share of Aurora Holdings’ assets
resulting from:
|
|
|
|
| |
•
|
the acquisition by us of Aurora Holdings Units from the Aurora
Holdings Continuing Members in exchange for shares of our
Class A common stock or cash, or
|
| |
| |
•
|
payments under the Tax Receivable Agreement to the Tax
Receivable Entity; and
|
|
|
|
| |
•
|
tax benefits related to imputed interest deemed to be paid by us
as a result of the Tax Receivable Agreement.
|
See “Organizational Structure — Reorganization
Transactions.”
Corporate
Information
We were incorporated in Delaware in April 2010 and began
laboratory operations in June 2006. Our executive offices are
located at 11025 RCA Center Drive, Suite 300, Palm Beach
Gardens, FL 33410. Our telephone number is
(866) 420-5512
or
(561) 626-5512.
Our website address is www.auroradx.com. Information included or
referred to on our website is not part of this prospectus.
8
THE
OFFERING
|
|
|
|
Shares of Class A common stock outstanding before this
offering
|
|
shares |
| |
|
Shares of Class A common stock offered by us
|
|
shares |
| |
|
Shares of Class A common stock offered by the selling
stockholders
|
|
shares |
| |
|
Total
|
|
shares |
| |
|
Shares of Class A common stock to be outstanding after this
offering
|
|
shares.
If, immediately after the completion of this offering and the
application of the net proceeds that we will receive from this
offering, all of the Aurora Holdings Continuing Members elected
to exchange their Aurora Holdings Units for shares of our
Class A common
stock, shares
of Class A common stock would be outstanding. |
| |
|
Shares of Class B common stock to be outstanding after this
offering
|
|
shares.
Shares of our Class B common stock have voting but no
economic rights (including rights to dividends and distributions
upon liquidation, but excluding the return of par value upon
liquidation) and will be issued in an amount equal to the number
of Aurora Holdings Units held by the Aurora Holdings Continuing
Members. When an Aurora Holdings Unit is exchanged by an Aurora
Holdings Continuing Member for a share of Class A common
stock, a share of our Class B common stock will be
cancelled. |
| |
|
Over-allotment option to be offered by us
|
|
shares.
All of the net proceeds we receive from any over-allotment
option to be offered by us will be used to acquire additional
Aurora Holdings Units and shares of Class B common stock from
the Aurora Holdings Continuing Members. |
| |
|
Over-allotment option to be offered by the selling stockholders
|
|
shares |
| |
|
Voting Rights
|
|
Each share of our Class A common stock entitles its holder
to one vote per share, representing an aggregate
of percent of the combined voting
power of our common stock upon completion of this offering and
the application of the net proceeds that we will receive from
this offering. |
| |
|
|
|
Each share of our Class B common stock entitles its holder
to one vote per share, representing an aggregate
of percent of the combined voting
power of our common stock upon completion of this offering and
the application of the net proceeds that we will receive from
this offering. |
| |
|
|
|
All classes of our common stock generally vote together as a
single class on all matters submitted to a vote of our
stockholders. Upon completion of this offering, our Class B
common stock will be held exclusively by the Aurora Holdings
Continuing Members. |
| |
|
|
|
See “Description of Capital Stock.” |
9
|
|
|
|
Exchange
|
|
Aurora Holdings Units held by the Aurora Holdings Continuing
Members (along with a corresponding number of shares of our
Class B common stock) may be exchanged with us for shares
of our Class A common stock on a
one-for-one
basis or, in certain circumstances, an equivalent amount of
cash. The Aurora Holdings Continuing Members will
hold
Aurora Holdings Units, or percent
of the outstanding Aurora Holdings Units, following the
completion of this offering and the application of the net
proceeds that we will receive from this offering. |
| |
|
Use of proceeds
|
|
We expect the net proceeds that we will receive from this
offering will be approximately
$ million based on an assumed
initial public offering price of $
per share, which is the midpoint of the price range set forth on
the cover page of this prospectus, after deducting underwriting
discounts and commissions and estimated offering expenses
payable by us and after giving effect to estimates of certain
expenses that we expect to be reimbursed. |
| |
|
|
|
Based on an assumed initial public offering price of
$ per share, which is the midpoint
of the price range set forth on the cover page of this
prospectus, we intend to use $ of
the proceeds from this offering, along with TRA Rights, to
purchase
Aurora Holdings Units
and shares
of our Class B common stock held by the Aurora Holdings
Continuing Members (or $
and
Aurora Holdings Units
and shares
of our Class B common stock if the underwriters exercise
their over-allotment option in full). |
| |
|
|
|
We intend to use the remaining proceeds from this offering for
working capital and general corporate purposes, which may
include future acquisitions. |
| |
|
|
|
We will not receive any of the proceeds from the sale of shares
of Class A common stock by the selling stockholders in this
offering. |
| |
|
|
|
See “Use of Proceeds.” |
| |
|
Risk Factors
|
|
You should read the “Risk Factors” section of this
prospectus for a discussion of factors to consider carefully
before deciding to invest in shares of our Class A common
stock. |
| |
|
Proposed NASDAQ Global Market Symbol
|
|
‘‘ARDX” |
The number of shares of our Class A common stock that will
be outstanding after this offering excludes shares of our
Class A common stock reserved for issuance upon the
exchange of our Class B common stock and Aurora Holdings
Units into Class A common stock.
Unless we indicate otherwise, all information in this prospectus
assumes:
|
|
|
| |
•
|
consummation of the Reorganization Transactions described under
“Prospectus Summary — Reorganization
Transactions” and “Organizational Structure;”
|
| |
| |
•
|
that the underwriters do not exercise their option to purchase
up
to shares
of our Class A common stock from us and the selling
stockholders to cover over-allotments; and
|
| |
| |
•
|
an initial public offering price of
$ per share, which is the midpoint
of the price range set forth on the cover page of this
prospectus.
|
10
Summary
Historical and Pro Forma
Consolidated Financial and Operating Data
The following table sets forth our summary historical and pro
forma consolidated financial data, at the dates and for the
periods indicated.
The historical financial data for each of the three years in the
period ended December 31, 2009, has been derived from our
audited historical consolidated financial statements included
elsewhere in this prospectus. The historical financial data for
the three months ended March 31, 2009 and 2010 and balance
sheet data as of March 31, 2010 have been derived from our
unaudited historical condensed consolidated financial statements
included elsewhere in this prospectus and include, in the
opinion of management, all adjustments, consisting only of
normal recurring adjustments, that management considers
necessary for the fair presentation of the financial information
set forth in those statements.
The summary unaudited pro forma condensed consolidated financial
data have been derived by the application of pro forma
adjustments to our historical consolidated financial statements
included elsewhere in this prospectus. The unaudited pro forma
condensed consolidated statements of operations data for the
year ended December 31, 2009 and the three months ended
March 31, 2010 and balance sheet data as of March 31,
2010 give effect to:
|
|
|
| |
•
|
our 2009 acquisition of South Texas Dermatopathology Lab, P.A.,
as if that acquisition occurred January 1, 2009;
|
|
|
|
| |
•
|
our 2010 acquisitions of Bernhardt Laboratories, Inc., Pinkus
Dermatopathology Laboratory, P.C., and Pathology Solutions,
LLC as if those acquisitions had occurred as of January 1,
2009;
|
|
|
|
| |
•
|
the execution of our new credit facilities;
|
|
|
|
| |
•
|
consummation of the Reorganization Transactions as if they
occurred as of March 31, 2010; and
|
|
|
|
| |
•
|
this offering and the use of the net proceeds that we will
receive from this offering, as if effective on March 31,
2010 for the unaudited pro forma consolidated balance sheet and
January 1, 2009 for the unaudited pro forma consolidated
statement of operations.
|
The summary pro forma financial information is included for
illustrative purposes only and may not accurately reflect our
results of operations or financial position for the periods and
as of the dates described above had the relevant transactions
occurred as of these dates. In addition, the summary pro forma
financial information is based on certain preliminary estimates
which may change materially upon completion of further analysis
and is not necessarily indicative of future results.
You should read this summary historical and pro forma
consolidated financial data together with our consolidated
historical financial statements and the related notes,
“Unaudited Pro Forma Financial Information” and
“Management’s Discussion and Analysis of Financial
Condition and Results of Operations,” in each case,
included elsewhere in this prospectus.
11
Aurora
Diagnostics Holdings LLC
Summary
Consolidated Financial and Operating
Data(1)
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Year Ended December 31,
|
|
|
Three Months Ended March 31,
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Pro Forma,
|
|
|
|
|
|
|
|
|
Pro Forma,
|
|
|
|
|
|
|
|
|
|
|
|
|
|
As Adjusted
|
|
|
|
|
|
|
|
|
As Adjusted
|
|
|
|
|
2007(1)
|
|
|
2008(1)
|
|
|
2009(1)
|
|
|
2009(2)
|
|
|
2009(1)
|
|
|
2010(1)
|
|
|
2010(2)
|
|
|
|
|
(in thousands, except per share data)
|
|
|
|
|
Consolidated Income Statement Data:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net Revenues
|
|
$
|
63,451
|
|
|
$
|
157,850
|
|
|
$
|
171,565
|
|
|
$
|
213,643
|
|
|
$
|
40,947
|
|
|
$
|
46,419
|
|
|
$
|
49,617
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating costs and expenses:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost of services
|
|
|
27,480
|
|
|
|
66,382
|
|
|
|
71,778
|
|
|
|
90,095
|
|
|
|
17,186
|
|
|
|
22,342
|
|
|
|
23,708
|
|
|
Selling, general and administrative expenses
|
|
|
15,172
|
|
|
|
33,194
|
|
|
|
36,854
|
|
|
|
43,543
|
|
|
|
9,177
|
|
|
|
10,820
|
|
|
|
11,119
|
|
|
Provision for doubtful accounts
|
|
|
2,378
|
|
|
|
8,037
|
|
|
|
9,488
|
|
|
|
10,220
|
|
|
|
2,305
|
|
|
|
2,610
|
|
|
|
2,610
|
|
|
Intangible asset amortization expense
|
|
|
5,721
|
|
|
|
14,308
|
|
|
|
14,574
|
|
|
|
16,018
|
|
|
|
3,628
|
|
|
|
3,891
|
|
|
|
4,001
|
|
|
Management fees
|
|
|
644
|
|
|
|
1,559
|
|
|
|
1,778
|
|
|
|
—
|
|
|
|
410
|
|
|
|
476
|
|
|
|
—
|
|
|
Impairment of goodwill and other intangible assets
|
|
|
—
|
|
|
|
—
|
|
|
|
8,031
|
(4)
|
|
|
8,031
|
(4)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Acquisition and business development costs
|
|
|
374
|
|
|
|
676
|
|
|
|
1,074
|
|
|
|
1,074
|
|
|
|
130
|
|
|
|
298
|
|
|
|
298
|
|
|
Equity based compensation expense
|
|
|
—
|
|
|
|
1,164
|
(3)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total operating costs and expenses
|
|
|
51,769
|
|
|
|
125,320
|
|
|
|
143,577
|
|
|
|
168,981
|
|
|
|
32,836
|
|
|
|
40,437
|
|
|
|
41,736
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income from operations
|
|
|
11,682
|
|
|
|
32,530
|
|
|
|
27,988
|
|
|
|
44,662
|
|
|
|
8,111
|
|
|
|
5,982
|
|
|
|
7,881
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other income (expense):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest expense
|
|
|
(7,114
|
)
|
|
|
(21,577
|
)
|
|
|
(18,969
|
)
|
|
|
(17,010
|
)
|
|
|
(4,791
|
)
|
|
|
(3,721
|
)
|
|
|
(4,229
|
)
|
|
Write-off of deferred debt issue
costs(5)
|
|
|
(3,451
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Other income
|
|
|
124
|
|
|
|
125
|
|
|
|
28
|
|
|
|
356
|
|
|
|
29
|
|
|
|
(12
|
)
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total other expense, net
|
|
|
(10,441
|
)
|
|
|
(21,452
|
)
|
|
|
(18,941
|
)
|
|
|
(16,654
|
)
|
|
|
(4,762
|
)
|
|
|
(3,733
|
)
|
|
|
(4,229
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income before income taxes
|
|
|
1,241
|
|
|
|
11,078
|
|
|
|
9,047
|
|
|
|
28,008
|
|
|
|
3,349
|
|
|
|
2,249
|
|
|
|
3,652
|
|
|
Provision for income
taxes(6)
|
|
|
762
|
|
|
|
408
|
|
|
|
45
|
|
|
|
11,203
|
|
|
|
17
|
|
|
|
10
|
|
|
|
1,461
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income
|
|
$
|
479
|
|
|
$
|
10,670
|
|
|
$
|
9,002
|
|
|
|
16,805
|
|
|
$
|
3,332
|
|
|
$
|
2,239
|
|
|
|
2,191
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income available to noncontrolling interest
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income available to common stockholders
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
|
|
|
|
|
|
|
|
|
|
|
$
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income available to common stockholders per common share:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic net income per common share
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
|
|
|
|
|
|
|
|
|
|
|
$
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Diluted net income per common share
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
|
|
|
|
|
|
|
|
|
|
|
$
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Consolidated operating data:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Number of accessions
|
|
|
619
|
|
|
|
1,472
|
|
|
|
1,557
|
|
|
|
1,903
|
|
|
|
376
|
|
|
|
443
|
|
|
|
460
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other financial data:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Adjusted
EBITDA(8)
|
|
$
|
19,086
|
|
|
$
|
52,066
|
|
|
$
|
55,931
|
|
|
$
|
72,271
|
|
|
$
|
12,847
|
|
|
$
|
11,449
|
|
|
$
|
12,983
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
12
Aurora
Diagnostics Holdings LLC
Summary
Consolidated Financial and Operating Data
| |
|
|
|
|
|
|
|
|
|
|
|
March 31, 2010
|
|
|
|
|
|
|
|
Pro Forma,
|
|
|
|
|
Actual(1)
|
|
|
As
Adjusted(7)
|
|
|
|
|
(in thousands)
|
|
|
|
|
Consolidated balance sheet data:
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
$
|
3,222
|
|
|
$
|
|
|
|
Total assets
|
|
|
490,999
|
|
|
|
|
|
|
Working capital, excluding deferred tax items
|
|
|
10,785
|
|
|
|
|
|
|
Long term debt, including current portion
|
|
|
235,188
|
|
|
|
|
|
|
Total equity
|
|
|
227,313
|
|
|
|
|
|
|
Tax receivable arrangement
|
|
|
—
|
|
|
|
|
|
|
|
|
|
(1)
|
|
The summary consolidated financial
data for the years ended December 31, 2007, 2008 and 2009
and the three months ended March 31, 2009 and 2010 are that
of Aurora Holdings prior to the completion of the Reorganization
Transactions, this offering and our recently completed
refinancing.
|
|
|
|
|
(2)
|
|
The pro forma, as adjusted,
consolidated income statement data for the year ended
December 31, 2009 and the three months ended March 31,
2010 gives effect to the following as if they had occurred
January 1, 2009:
|
|
|
|
| |
•
|
our 2009 acquisition of South Texas Dermatopathology Lab, P.A.;
|
|
|
|
| |
•
|
our 2010 acquisitions of Bernhardt Laboratories, Inc., Pinkus
Dermatopathology Laboratory, P.C. and Pathology Solutions,
LLC;
|
|
|
|
| |
•
|
the execution of our new credit facilities;
|
|
|
|
| |
•
|
the consummation of the Reorganization Transactions; and
|
| |
| |
•
|
the use of the net proceeds that we will receive from this
offering.
|
|
|
|
|
(3)
|
|
During 2008, we adopted a new
equity incentive plan, which we refer to as the New Plan, to
replace our original equity incentive plan. The New Plan
provides awards of membership interest units in Aurora Holdings.
These interests are denominated as
Class D-1,
Class D-2,
and
Class D-3 units
of Aurora Holdings. During 2008, we authorized and issued 4,000
D-1 units, 3,000 D-2 units and 3,000 D-3 units of
Aurora Holdings under the New Plan. All membership interest
units in Aurora Holdings issued in 2008 were fully vested as of
December 31, 2008. We recorded compensation expense of
$1.2 million for these awards. There were no other grants
under the New Plan. In connection with the Reorganization
Transactions, the Class D Units of Aurora Holdings issued
under the New Plan will be either exchanged for shares of our
Class A common stock or cancelled without consideration.
|
|
(4)
|
|
As of September 30, 2009, we
tested goodwill and intangible assets for potential impairment
and recorded a non-cash impairment expense of $8.0 million
resulting from a write down of $6.6 million in the carrying
value of goodwill and a write down of $1.4 million in the
carrying value of other intangible assets. The write-down of the
goodwill and other intangible assets related to one reporting
unit. Regarding this reporting unit, we believe events occurred
and circumstances changed that more likely than not reduced the
fair value of the intangible assets and goodwill below their
carrying amounts. These events during 2009 consisted primarily
of the loss of significant customers present at the acquisition
date, which adversely affected the current year and expected
future revenues and operating profit of the reporting unit.
|
|
|
|
|
(5)
|
|
In December 2007, we refinanced our
previous credit facilities. As a result, we wrote off
$3.5 million of unamortized deferred debt issue costs.
|
|
|
|
|
(6)
|
|
Aurora Holdings is a Delaware
limited liability company taxed as a partnership for federal and
state income tax purposes, in accordance with the applicable
provisions of the Internal Revenue Code. Accordingly, Aurora
Holdings was not generally subject to income taxes. The income
attributable to Aurora Holdings was allocated to the members of
Aurora Holdings in accordance with the terms of the existing
Aurora Holdings limited liability company agreement, which we
refer to as the Aurora Holdings LLC Agreement. However, certain
of our subsidiaries are corporations, file separate returns and
are subject to federal and state income taxes. The historical
provision for income taxes for these subsidiaries is reflected
in our consolidated financial statements and includes federal
and state taxes currently payable and changes in deferred tax
assets and liabilities excluding the establishment of deferred
tax assets and liabilities related to the acquisitions. The pro
forma, as adjusted, estimated provision for income taxes assumes
a blended 40 percent effective tax rate, after giving
effect to the Reorganization Transactions and was based on the
federal and state statutory income tax rates in effect during
the respective periods.
|
|
|
|
|
(7)
|
|
The pro forma, as adjusted,
consolidated balance sheet data as of March 31, 2010 gives
effect to the following, as if they were effective as of
March 31, 2010:
|
|
|
|
| |
•
|
the execution of our new credit facilities;
|
|
|
|
| |
•
|
the consummation of the Reorganization Transactions; and
|
|
|
|
| |
•
|
the use of the net proceeds that we will receive from this
offering.
|
13
|
|
|
|
(8)
|
|
The following is a reconciliation
of net income to Adjusted EBITDA:
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months
|
|
|
|
|
Year Ended December 31,
|
|
|
Ended March 31,
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Pro Forma,
|
|
|
Pro Forma,
|
|
|
|
|
|
|
|
|
|
|
|
|
|
As Adjusted
|
|
|
As Adjusted
|
|
|
|
|
2007
|
|
|
2008
|
|
|
2009
|
|
|
2009
|
|
|
2010
|
|
|
|
|
(in thousands)
|
|
|
|
|
Net Income
|
|
$
|
479
|
|
|
$
|
10,670
|
|
|
$
|
9,002
|
|
|
$
|
16,805
|
|
|
$
|
2,192
|
|
|
Interest expense
|
|
|
7,114
|
|
|
|
21,577
|
|
|
|
18,969
|
|
|
|
17,010
|
|
|
|
4,229
|
|
|
Income taxes
|
|
|
762
|
|
|
|
408
|
|
|
|
45
|
|
|
|
11,203
|
|
|
|
1,461
|
|
|
Depreciation and amortization
|
|
|
6,386
|
|
|
|
16,137
|
|
|
|
17,060
|
|
|
|
18,504
|
|
|
|
4,803
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
EBITDA
|
|
|
14,741
|
|
|
|
48,792
|
|
|
|
45,076
|
|
|
|
63,522
|
|
|
|
12,685
|
|
|
Management
fees(A)
|
|
|
644
|
|
|
|
1,559
|
|
|
|
1,778
|
|
|
|
—
|
|
|
|
—
|
|
|
Stock-based compensation
|
|
|
—
|
|
|
|
1,164
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Unusual
charges(B)(C)(D)
|
|
|
3,825
|
|
|
|
676
|
|
|
|
9,105
|
|
|
|
9,105
|
|
|
|
298
|
|
|
Other
|
|
|
(124
|
)
|
|
|
(125
|
)
|
|
|
(28
|
)
|
|
|
(356
|
)
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Adjusted EBITDA, as defined
|
|
$
|
19,086
|
|
|
$
|
52,066
|
|
|
$
|
55,931
|
|
|
$
|
72,271
|
|
|
$
|
12,983
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Adjusted EBITDA is defined as
earnings before interest, taxes, depreciation and amortization
(EBITDA), further adjusted to exclude unusual items and other
adjustments which we believe are appropriate to provide
additional information to investors, enhancing their
understanding of our financial performance and providing them an
important financial metric used to evaluate performance in the
health care industry. We further believe that providing this
information allows our investors greater transparency and a
better understanding of our ability to meet our debt service
obligations and make capital expenditures.
|
|
|
|
|
|
|
Adjusted EBITDA does not represent
net income or cash flow from operations as those terms are
defined by GAAP and does not necessarily indicate whether cash
flows will be sufficient to fund cash needs. Further, our new
credit facilities require that Adjusted EBITDA be calculated for
the most recent four fiscal quarters. As a result, the measure
can be disproportionately affected by a particularly strong or
weak quarter. Further, it may not be comparable to the measure
for any subsequent four-quarter period or any complete fiscal
year.
|
|
|
|
|
|
|
Adjusted EBITDA is not a recognized
measurement under GAAP, and investors should not consider
Adjusted EBITDA as a substitute for measures of our financial
performance as determined in accordance with GAAP, such as net
income and operating income. Because other companies may
calculate Adjusted EBITDA differently than we do, Adjusted
EBITDA may not be comparable to similarly titled measures
reported by other companies. Adjusted EBITDA has other
limitations as an analytical tool when compared to the use of
net income, which we believe is the most directly comparable
GAAP financial measure, including:
|
|
|
|
| |
•
|
Adjusted EBITDA does not reflect the provision of income tax
expense in our various jurisdictions;
|
| |
| |
•
|
Adjusted EBITDA does not reflect the interest expense we incur;
|
| |
| |
•
|
Adjusted EBITDA does not reflect any attribution of costs to our
operations related to our investments and capital expenditures
through depreciation and amortization charges;
|
| |
| |
•
|
Adjusted EBITDA does not reflect the cost of compensation we
provide to our employees in the form of stock option
awards; and
|
| |
| |
•
|
Adjusted EBITDA excludes expenses that we believe are unusual or
non-recurring, but which others may believe are normal expenses
for the operation of a business.
|
|
|
|
|
(A)
|
|
Management fees related to expenses
payable to affiliates. These management fees will terminate
along with our management services agreement upon the completion
of this offering.
|
|
|
|
|
(B)
|
|
During 2007, net income included
the write-off of previously deferred debt issue costs in
connection with the refinancing of our previous credit facility.
|
|
|
|
|
(C)
|
|
During 2009, we recorded a non-cash
impairment charge of $8.0 million related to goodwill and
other intangible assets.
|
|
|
|
|
(D)
|
|
Unusual charges also includes an
add back for acquisition and business development costs as
reported in our consolidated statements of operations.
|
14
RISK
FACTORS
Buying shares of our Class A common stock involves risk.
You should consider carefully the risks and uncertainties
described below, together with all of the other information in
this prospectus, including the financial statements and the
related notes appearing in the back of this prospectus, before
deciding to purchase shares of our Class A common stock.
Risks
Relating to Our Business
Changes
in regulation and policies may adversely affect reimbursement
for diagnostic services and could have a material adverse impact
on our business.
Reimbursement levels for health care services are subject to
continuous and often unexpected changes in policies, and we face
a variety of efforts by government payors to reduce utilization
and reimbursement for diagnostic testing services. Changes in
governmental reimbursement may result from statutory and
regulatory changes, retroactive rate adjustments, administrative
rulings, competitive bidding initiatives, and other policy
changes.
In 2010, the U.S. Congress passed legislation relating to
health care reform, including the Patient Protection and
Affordable Care Act, or PPACA, and the Health Care and Education
Affordability Reconciliation Act of 2010, or HCEARA. While the
comprehensive health reform legislation passed by the
U.S. Congress and signed into law by the President in 2010
did not adversely affect reimbursement for our anatomic
pathology services, this legislation provides for two separate
reductions in the reimbursement rates for our clinical
laboratory services: a “productive adjustment”
(currently estimated to be between 1.1 and 1.4 percent),
and an additional 1.75 percent reduction. Each of these
would reduce the annual Consumer Price Index-based update that
would otherwise determine our reimbursement for clinical
laboratory services. The U.S. Congress has considered, at
least yearly in conjunction with budgetary legislation, changes
to one or both of the Medicare fee schedules under which we
receive reimbursement, which include the physician fee schedule
and the clinical laboratory fee schedule. Further reductions in
reimbursement for Medicare services or changes in policy
regarding coverage of tests may be implemented from time to
time. A substantial portion of our anatomic pathology services
are billed under a single code (CPT 88305) and our revenue and
business may be adversely affected if the reimbursement rate
associated with that code is reduced. Even when reimbursement
rates are not reduced, policy changes add to our costs by
increasing the complexity and volume of administrative
requirements. Medicaid reimbursement, which varies by state, is
also subject to administrative and billing requirements and
budget pressures. Recently, state budget pressures have caused
states to consider several policy changes that may impact our
financial condition and results of operations, such as delaying
payments, reducing reimbursement, restricting coverage
eligibility and service coverage, and imposing taxes on our
services.
Increased
internalization of diagnostic testing by our clients or
patients, including the use of new testing technologies by our
clients or patients, could adversely affect our
business.
Our clients, such as referring physicians and hospitals, may
internalize diagnostic testing or technologies that have
historically been performed by diagnostic laboratory companies
like us. Our industry has experienced a recent market trend in
which physicians and hospitals perform the technical
and/or
professional components of their laboratory testing needs in
their own offices. If this trend continues or becomes more
pronounced and our clients internalize diagnostic testing
functions or technologies that we currently perform or use, and
we do not develop new or alternative functions or technologies
that are attractive to our clients, it may reduce the demand for
our diagnostic testing services and adversely affect our
business.
In addition, advances in technology may lead to the development
of more cost-effective tests that can be performed outside of a
commercial laboratory such as:
|
|
|
| |
•
|
point-of-care
tests that can be performed by physicians in their offices;
|
| |
| |
•
|
tests that can be performed by hospitals in their own
laboratories; or
|
| |
| |
•
|
home testing that can be performed by patients in their homes.
|
15
Any advance in technology could reduce demand for our services
or render them obsolete.
Compliance costs associated with the Clinical Laboratory
Improvement Amendments of 1988, or CLIA, make it
cost-prohibitive for many physicians to operate clinical
laboratories in their offices. However, diagnostic tests
approved or cleared by the U.S. Food and Drug
Administration, or FDA, for home use are automatically deemed to
be “waived” tests under CLIA and may be performed by
our referring physicians and their patients with minimal
regulatory oversight under CLIA. Test kit manufacturers could
seek to increase sales to both our referring physicians and
their patients of test kits approved by the FDA for
point-of-care
testing or home use. Development of such technology and its use
by our clients would reduce the demand for our laboratory-based
testing services and adversely affect our business.
Failure
to timely or accurately bill for our services or collect
outstanding payments could have a material adverse effect on our
business.
Billing for diagnostic services is complex. We bill numerous
payors, including physicians, patients, insurance companies,
Medicare, and Medicaid, according to applicable law, billing
requirements and, as applicable, contractual arrangements. This
complexity is further compounded by rapidly changing
requirements for auditing, external compliance, and internal
compliance policies and procedures.
Most of our bad debt expense in 2009, which totaled
5.5 percent of revenues, resulted from the failure of
patients to pay their bills, including copayments and
deductibles. Failure to timely or correctly bill could lead to
lack of reimbursement for services or an increase in the aging
of our accounts receivable, which could adversely affect our
results of operations. Increases in write-offs of doubtful
accounts, delays in receiving payments, potential retroactive
adjustments, and penalties resulting from audits by payors would
also adversely affect our financial condition. Failure to comply
with applicable laws relating to billing governmental health
care programs could also lead to various penalties, including
exclusion from participation in Medicare or Medicaid programs,
asset forfeitures, civil and criminal fines and penalties, and
the loss of various licenses, certificates, and authorizations
necessary to operate our business, any of which could have a
material adverse effect on our business.
Non-governmental
third-party payors have taken steps to control the utilization
and reimbursement of diagnostic services.
We face efforts by non-governmental third-party payors,
including health plans, to reduce utilization of diagnostic
testing services and reimbursement for diagnostic services. For
instance, third-party payors often use the payment amounts under
the Medicare fee schedules as a reference in negotiating their
payment amounts. As a result, a reduction in Medicare
reimbursement rates could result in a reduction in the
reimbursements we receive from such third-party payors. Changes
in test coverage policies of and reimbursement from other
third-party payors may also occur independently from changes in
Medicare. Such reimbursement and coverage changes in the past
have resulted in reduced prices, added costs and reduced
accession volume and have added more complex and new regulatory
and administrative requirements.
The health care industry has also experienced a trend of
consolidation among health insurance plans, resulting in fewer,
larger health plans with significant bargaining power to
negotiate fee arrangements with health care providers like us.
Some of these health plans, as well as independent physician
associations, have demanded that laboratories accept discounted
fee structures or assume a portion or all of the financial risk
associated with providing diagnostic testing services to their
members through capitated payment arrangements. In addition,
some health plans have limited the preferred provider
organization or
point-of-service
laboratory network to only a single national laboratory to
obtain improved
fee-for-service
pricing. The increased consolidation among health plans also has
increased the potential adverse impact of ceasing to be a
contracted provider with any such insurer. See
“— Failure to participate as a provider with
payors or operating as a non-contracted provider could have a
material adverse effect on our business.”
We expect that efforts to reduce reimbursements, impose more
stringent cost controls and reduce utilization of diagnostic
testing services will continue. These efforts may have a
material adverse effect on our business and results of
operations.
16
Changes
in payor mix may adversely affect reimbursement for diagnostic
services and could have a material adverse impact on our
business.
Most of our services are billed to a party other than the
physician that ordered the test. In 2009, we received
25 percent of our revenues from Medicare and Medicaid and
61 percent of our revenues from non-governmental
third-party payors, including health plans. If we bill a higher
percentage of our services to payors who reimburse at rates
lower than our current payors, our results of operations and
financial condition would suffer.
Integration
of our operations with newly acquired businesses may be
difficult and costly.
Since our inception, we have acquired 17 existing diagnostic
services businesses. We expect to evaluate potential strategic
acquisitions of diagnostic services and other businesses that
might augment our existing specialized diagnostic testing
services. These acquisitions have involved and could continue to
involve the integration of a separate company that previously
operated independently and had different systems, processes and
cultures. As such, we have not yet completed the integration of
several of our past acquisitions. In particular, many of our
operations, such as our laboratory information systems and
billing systems, are not yet standardized and some aspects of
the
day-to-day
operations of our laboratories continue to be conducted on a
decentralized basis.
The process of integrating businesses we acquire may
substantially disrupt both our existing businesses and the
businesses we acquire. This disruption may divert management
from the operation of our business or may cause us to lose key
employees or clients. Additionally, we may have difficulty
consolidating facilities and infrastructure, standardizing
information and other systems and coordinating
geographically-separated facilities and workforces, resulting in
a decline in the quality of services.
Any past or future acquisitions, and the related integration
efforts, may be difficult, costly or unsuccessful. In each case,
our existing business and the businesses we acquire may be
adversely affected. Even if we are able to successfully
integrate businesses we have acquired, we may not be able to
realize the benefits that we expect from them.
Businesses
we acquire may have significant unknown or contingent
liabilities that could adversely impact our operating
results.
Businesses we acquire may have unknown or contingent liabilities
or liabilities that are in excess of the amounts that we
originally estimated. Although we generally seek indemnification
from the sellers of businesses we acquire for matters that are
not properly disclosed to us, we may not successfully obtain
indemnification. Even in cases where we are able to obtain
indemnification, we may be subject to liabilities greater than
the contractual limits of our indemnification or the financial
resources of the indemnifying party. In the event that we are
responsible for liabilities substantially in excess of any
amounts recovered through rights to indemnification, this could
adversely impact our operating results.
Failure
of our IT or communication systems, or the failure of these
systems to keep pace with technological advances or changes in
regulation and policies related to our IT or communication
systems, could adversely impact our business.
Our laboratory operations depend significantly on the
uninterrupted performance of our IT and communication systems.
Sustained system failures or interruption of our systems in one
or more of our laboratories could disrupt our ability to process
laboratory requisitions, handle client service, perform testing,
provide our reports or test results in a timely manner, or bill
the appropriate party for our services.
Our efforts to invest in new or improved IT systems and billing
systems may be costly, and require time and resources for
implementation. While we have begun implementing a plan to
standardize and improve our laboratory information systems and
billing systems, we expect that it will take several years to
complete full implementation. Our efforts to invest in new or
improved IT systems and billing systems may not ultimately be
successful, and our failure to properly implement our plan to
standardize and improve our laboratory information systems and
billing systems could adversely impact our business.
17
Public and private initiatives to create electronic medical
record standards and to mandate standardized coding systems for
the electronic exchange of information, including test orders
and test results, could require costly modifications to our
existing IT systems. We expect that any standards that might be
adopted or implemented would allow us adequate time to comply
with such standards. However, any failure or delay in
implementing standards may result in a loss of clients and
business opportunities, which could adversely impact our
business.
Failure
to adequately safeguard data, including patient data that is
subject to regulations related to patient privacy, could
adversely impact our business.
The success of our business depends on our ability to obtain,
process, analyze, maintain and manage data, including sensitive
information such as patient data. If we do not adequately
safeguard that information and it were to become available to
persons or entities that should not have access to it, our
business could be impaired, our reputation could suffer and we
could be subject to fines, penalties and litigation. Although we
have implemented security measures, our infrastructure is
vulnerable to computer viruses, break-ins and similar disruptive
problems caused by our clients or others that could result in
interruption, delay or cessation of service. Break-ins, whether
electronic or physical, could potentially jeopardize the
security of confidential client and supplier information stored
physically at our locations or electronically in our computer
systems. Such an event could damage our reputation, cause us to
lose existing clients and deter potential clients. It could also
expose us to liability to parties whose security or privacy has
been infringed, to regulatory actions by the Centers for
Medicare & Medicaid Services, or CMS, part of the United
States Department of Health and Human Services, or HHS, or to
civil or criminal sanctions. The occurrence of any of the
foregoing events could adversely impact our business.
The American Recovery and Reinvestment Act of 2009 imposed
additional obligations on health care entities with respect to
data privacy and security, including new notifications in case
of a breach of privacy and security standards. We are unable to
predict the extent to which these new obligations may prove
technically difficult, time-consuming or expensive to implement.
Failure
to attract and retain experienced and qualified personnel could
adversely affect our business.
Our success depends on our ability to attract, retain and
motivate experienced anatomic pathologists, histotechnologists,
cytotechnologists, skilled laboratory and IT staff, experienced
sales representatives and other personnel. Competition for these
employees is strong, and if we are not able to attract and
retain qualified personnel it would have a material adverse
effect on our business.
We are dependent on the expertise of our local medical directors
and our executive officers. The loss of these individuals could
have a material adverse effect on our business.
Our sales representatives have developed and maintain close
relationships with a number of health care professionals, and
our specialized approach to marketing our services positions our
sales representatives to have a deep knowledge of the needs of
the referring physicians they serve. Given the nature of the
relationships we seek to develop with our clients, losses of
sales representatives may cause us to lose clients.
Changes
in medical treatment, reimbursement rates and other market
conditions in the dermatopathology market could adversely affect
our business.
We derive a significant portion of our revenues from our
dermatopathology subspecialty, which makes us particularly
sensitive to changes in medical treatment, reimbursement rates
and other market conditions in the dermatopathology market. Our
revenues are particularly sensitive to changes that affect the
number of or reimbursement for dermatopathology-related
services. In 2009, we derived approximately 47 percent of
our revenues from our dermatopathology subspecialty services,
primarily from biopsies of the skin. If there is a significant
development in the prevention of skin cancer, or an adverse
development in the reimbursement rate for skin biopsies, it
could have a material adverse effect on our business.
18
Failure
to adequately scale our infrastructure to meet demand for our
diagnostic services or to support our growth could create
capacity constraints and divert resources, resulting in a
material adverse effect on our business.
Increases in demand for our diagnostic services, including
unforeseen or significant increases in demand due to client or
accession volume, could strain the capacity of our personnel and
infrastructure. Any strain on our personnel or infrastructure
could lead to inaccurate test results, unacceptable turn-around
times or client service failures. Furthermore, although we are
not currently subject to these capacity constraints, if demand
increases for our diagnostic services, we may not be able to
scale our personnel or infrastructure accordingly. Any failure
to handle increases in demand, including increases due to client
or accession volumes, could lead to the loss of established
clients and have a material adverse effect on our business.
We intend to expand by establishing laboratories in additional
geographic markets. In addition to acquisition or development
costs, this will require us to spend considerable time and money
to expand our infrastructure and to hire and retain experienced
anatomic pathologists, histotechnologists, cytotechnologists,
skilled laboratory and IT staff, experienced sales
representatives, client service associates and other personnel
for our additional laboratories. We will also need federal,
state and local certifications, as well as supporting
operational, logistical and administrative infrastructure. Even
after new laboratories are operational, it may take time for us
to derive the same economies of scale we have in our existing
laboratories. Moreover, we may suffer reduced economies of scale
in our existing laboratories as we seek to balance the amount of
work allocated to each facility and expand those laboratories.
An expansion of our laboratories or systems could divert
resources, including the focus of our management, away from our
current business.
Failure
to effectively continue or manage our strategic and organic
growth could cause our growth rate to decline.
Our business strategy includes continuing to selectively acquire
existing diagnostic services businesses. Since our inception, we
have acquired 17 existing diagnostic services businesses. To
continue this strategic growth, we will need to continue to
identify appropriate businesses to acquire and successfully
undertake the acquisition of these businesses on reasonable
terms. Consolidation and competition within our industry, among
other factors, may make it difficult or impossible to identify
businesses to acquire on a timely basis, or at all. In
particular, the competition to acquire independent private labs
and pathology groups has increased. In addition to historical
competitors such as national lab companies, regional hospital
centers and specialty lab companies, a number of private equity
firms have recently made initial investments in the laboratory
industry and may become potential competitors to our efforts to
source new acquisitions. Our inability to continue our strategic
growth would cause our growth rate to decline and could have a
material adverse effect on our business.
We also seek to continue our organic growth through the
expansion of our sales force, the development of de novo
laboratories, strategic extension of our operations into markets
such as long-term care, and the inclusion of new clinical and
molecular tests in our testing menu. Because of limitations in
available capital and competition within our industry, among
other factors, we may not be able to implement any or all of
these organic growth strategies on a reasonable schedule, or at
all. Our failure to continue our organic growth would cause our
growth rate to decline and could have a material adverse effect
on our business.
Our revenues have grown from $3.5 million in 2006 to
$171.6 million in 2009. To manage our growth, we must
continue to implement and improve our operational and financial
systems and to expand, train, manage and motivate our employees.
We may not be able to effectively manage the expansion of our
operations, and our systems, procedures or controls may not be
adequate to support our operations. Our management may not be
able to rapidly scale the infrastructure necessary to exploit
the market opportunity for our services. Our inability to manage
growth could have a material adverse effect on our business.
Failure
to participate as a provider with payors or operating as a
non-contracted provider could have a material adverse effect on
our business.
The health care industry has experienced a trend of
consolidation among health care insurers, resulting in fewer,
larger insurers with significant bargaining power in negotiating
fee arrangements with health care providers
19
like us. Managed care providers often restrict their contracts
to a small number of laboratories that may be used for tests
ordered by physicians in the managed care provider’s
network. If we do not have a contract with a managed care
provider, we may be unable to gain those physicians as clients,
and it could adversely affect our business.
In cases in which we do contract with a specified insurance
company as a participating provider, we are considered
“in-network,”
and the reimbursement of third-party payments is governed by
contractual relationships.
In cases in which we do not have a contractual relationship with
an insurance company or we are not an approved provider for a
government program, we have no contractual right to collect for
our services and such payors may refuse to reimburse us for our
services. This could lead to a decrease in accession volume and
a corresponding decrease in our revenues. In instances where we
are an
out-of-network
provider, reductions in reimbursement rates for
non-participating providers could also adversely affect us.
Third-party payors with whom we do not participate as a
contracted provider may also require that we enter into
contracts, which may have pricing and other terms that are
materially less favorable to us than the terms under which we
currently operate. While accession volume may increase as a
result of these contracts, our revenues per accession may
decrease.
Use of our diagnostic services as a non-participating provider
also typically results in greater copayments for the patient
unless we elect to treat them as if we were a participating
provider in accordance with applicable law. Treating such
patients as if we were a participating provider may adversely
impact results of operations because we may be unable to collect
patient copayments and deductibles. In some states, applicable
law prohibits us from treating these patients as if we were a
participating provider. As a result, referring physicians may
avoid use of our services, which could result in a decrease in
accession volume and adversely affect revenues.
Failure
to raise additional capital or generate the significant capital
necessary to continue our growth could reduce our ability to
compete and could harm our business.
We expect that our existing cash and cash equivalents, together
with the net proceeds that we will retain from this offering and
availability under our new credit facilities, will be sufficient
to meet our anticipated cash needs until 2012. After that, we
may need to raise additional funds, and we may not be able to
obtain additional debt or equity financing on favorable terms,
if at all. If we raise additional equity financing, our
stockholders may experience significant dilution of their
ownership interests, and the per share value of our Class A
common stock could decline. Furthermore, if we engage in debt
financing, the holders of debt would have priority over the
holders of common stock, and we may be required to accept terms
that restrict our ability to incur additional indebtedness, and
take other actions that would otherwise be in the interests of
our stockholders and force us to maintain specified liquidity or
other ratios, any of which could harm our business, operating
results and financial condition. If we need additional capital
and cannot raise it on acceptable terms, we may not, among other
things, be able to:
|
|
|
| |
•
|
continue to expand our sales and marketing and research and
development organizations;
|
| |
| |
•
|
develop or acquire complementary technologies, services,
products or businesses;
|
| |
| |
•
|
expand operations both organically and through acquisitions;
|
| |
| |
•
|
hire, train and retain employees; or
|
| |
| |
•
|
respond to competitive pressures or unanticipated working
capital requirements.
|
Our failure to do any of these things could seriously harm our
business, financial condition and results of operations.
The
agreement governing our new credit facilities contains, and
future debt agreements may contain, various covenants that limit
our discretion in the operation of our business.
Our agreement and the related instruments governing borrowings
under our new credit facilities contain, and the agreements and
instruments governing any future debt agreements of ours may
contain, various restrictive covenants that, among other things,
require us to comply with or maintain certain financial tests
and ratios and restrict our ability to:
20
|
|
|
| |
•
|
redeem or repurchase stock, pay dividends or make other
distributions;
|
| |
| |
•
|
make certain investments;
|
| |
| |
•
|
create liens;
|
| |
| |
•
|
enter into transactions with affiliates;
|
| |
| |
•
|
make acquisitions;
|
| |
| |
•
|
merge or consolidate;
|
| |
| |
•
|
transfer or sell assets; and
|
| |
| |
•
|
make fundamental changes in our corporate existence and
principal business.
|
In addition, events beyond our control could affect our ability
to comply with and maintain the financial tests and ratios
contained in these documents. Any failure by us to comply with
or maintain all applicable financial tests and ratios and to
comply with all applicable covenants could result in an event of
default with respect to our new term loan and revolving credit
facility or future debt agreements. This could lead to the
acceleration of the maturity of our outstanding loans and the
termination of the commitments to make further extensions of
credit. Even if we are able to comply with all applicable
covenants, the restrictions on our ability to operate our
business at our sole discretion could harm our business by,
among other things, limiting our ability to take advantage of
financing, mergers, acquisitions and other corporate
opportunities.
We may
be unable to obtain, maintain or enforce our intellectual
property rights and may be subject to intellectual property
litigation that could adversely impact our
business.
We may be unable to obtain or maintain adequate proprietary
rights for our products and services or to successfully enforce
our proprietary rights, and we cannot assure you that our
products or methods do not infringe the patents or other
intellectual property rights of third parties. Infringement and
other intellectual property claims and proceedings brought
against us, whether successful or not, could result in
substantial costs and harm our reputation. Such claims and
proceedings can also distract and divert management and key
personnel from other tasks important to the success of our
business. In addition, intellectual property litigation or
claims could force us to do one or more of the following:
|
|
|
| |
•
|
cease developing, performing or selling products or services
that incorporate the challenged intellectual property;
|
| |
| |
•
|
obtain and pay for licenses from the holder of the infringed
intellectual property right, which licenses may not be available
on reasonable terms, or at all;
|
| |
| |
•
|
redesign or reengineer our tests;
|
| |
| |
•
|
change our business processes; and
|
| |
| |
•
|
pay substantial damages, court costs and attorneys’ fees,
including potentially increased damages for any infringement
held to be willful.
|
In the event of an adverse determination in an intellectual
property suit or proceeding, or our failure to license essential
technology, our sales could be harmed
and/or our
costs could increase, which would harm our financial condition.
We
have a limited operating history, which may make it difficult to
accurately evaluate our business and prospects.
We commenced operations in June 2006. As a result, we have a
limited operating history upon which to accurately predict our
potential revenue. Our revenues and income potential and our
ability to expand our business into additional anatomic
pathology specialties and markets is still unproven. As a result
of these factors, the future revenues and income potential of
our business is uncertain. Although we have experienced
significant revenue growth since our inception, we may not be
able to sustain this growth. Any evaluation of our business and
our
21
prospects must be considered in light of these factors and the
risks and uncertainties often encountered by companies in our
stage of development. Our profitability may be adversely
affected as we expand our infrastructure or if we incur
increased selling expenses or other general and administrative
expenses. Some of these risks and uncertainties include our
ability to:
|
|
|
| |
•
|
execute our business model;
|
| |
| |
•
|
create brand recognition;
|
| |
| |
•
|
respond effectively to competition;
|
| |
| |
•
|
manage growth in our operations;
|
| |
| |
•
|
respond to changes in applicable government regulations and
legislation;
|
| |
| |
•
|
access additional capital when required; and
|
| |
| |
•
|
attract and retain key personnel.
|
Current
economic conditions, including the current recession in the
United States and the worldwide economic slowdown, as well as
further disruptions in the financial markets, could adversely
impact our operating results and financial
condition.
The current economic recession in the United States and
worldwide economic slowdown could adversely affect our operating
results and financial condition. Among other things, the
potential decline in federal and state revenues that may result
from these conditions may create additional pressures to contain
or reduce reimbursements for our services from Medicare,
Medicaid and other government sponsored programs. The increased
job losses and elevated unemployment rates in the United States
resulting from the recession could result in a smaller
percentage of our patients being covered by commercial payors
and a larger percentage being covered by lower-paying Medicaid
programs. Employers may also begin to select more restrictive
commercial plans with lower reimbursement rates. To the extent
that payors are adversely affected by a decline in the economy,
we may experience further pressure on commercial rates, delays
in fee collections and a reduction in the amounts we are able to
collect. In addition, if the current turmoil in the financial
markets continues, interest rates may increase and it could be
more difficult to obtain credit in the future. Any or all of
these factors, as well as other consequences of the current
economic conditions which currently cannot be anticipated, could
adversely impact our operating results and financial condition.
Competition
in our industry from existing or new companies and failure to
obtain and retain clients could have a material adverse impact
on our business.
Our success depends on our ability to obtain and retain clients
and maintain accession volume. A reduction in the number of our
clients, or in the tests ordered or specimens submitted by our
clients, without offsetting increases or growth, could impact
our ability to maintain or grow our business and could have a
material adverse effect on our business.
While there has been significant consolidation in recent years
in the diagnostic testing industry, the industry remains
fragmented and highly-competitive both in terms of price and
service. We primarily compete with various clinical test
providers, anatomic pathology practices, hospital-affiliated
laboratories, commercial clinical laboratories and
physician-office laboratories. This competition is based
primarily on price, clinical expertise, quality of service,
client relationships, breadth of testing menu, speed of
turnaround of test results, reporting and IT systems, reputation
in the medical community and ability to employ qualified
personnel. Some of our competitors may have greater technical,
financial and other resources than we do. Our failure to
successfully compete on any of these factors could result in a
loss of clients and adversely affect our ability to grow.
Replication of our business model by competitors may adversely
affect growth and profitability. Barriers to entry in anatomic
pathology markets include the need to form strong relationships
with referring physicians, hire experienced pathologists, make
capital investments and acquire IT. These barriers may not be
sufficient to prevent or deter new entrants to our market, and
competitors could replicate or improve some or all aspects of
our business
22
model and cause us to lose market share in the areas where we
compete or inhibit our growth, which could have a material
adverse effect on our business.
Failure
to acquire rights to new technologies, products or tests, or
discontinuations or recalls of existing technologies, products
or test, could negatively impact our testing volume and net
revenues.
The diagnostic testing industry is characterized by rapid
changes in technology, frequent introductions of new products
and diagnostics tests and evolving industry standards and client
demands for new diagnostic technologies. Other companies or
individuals, including our competitors, may obtain patents or
other property rights that would prevent, limit or interfere
with our ability to develop, perform or sell our tests or
operate our business or increase our costs. Advances in
technology may result in the creation of enhanced diagnostic
tools that enable other laboratories, hospitals, physicians,
patients or third parties to provide specialized diagnostic
services that are superior to ours or more patient-friendly,
efficient or cost-effective. These developments may result in a
decrease in the demand for our tests or cause us to reduce the
prices for our tests. We may be unable to develop or introduce
new tests on our own, which means that our success may depend,
in part, on our ability to license new and improved technologies
on favorable terms. We may be unable to continue to negotiate
acceptable licensing arrangements, and arrangements that we do
conclude may not yield commercially successful diagnostic tests.
If we are unable to acquire rights to these testing methods at
competitive rates, our research and development costs may
increase as a result. In addition, if we are unable to develop
and introduce, or acquire rights to, new tests, technology and
services to expand our testing business, our testing methods may
become outdated when compared with our competition and our
testing volume and revenues may be materially and adversely
affected.
From time to time, manufacturers discontinue or recall reagents,
test kits or instruments we use to perform diagnostic services.
Such discontinuations or recalls could adversely affect our
costs, testing volume and revenues.
Regulatory
Risks
New
and proposed federal or state health care reform measures could
adversely affect our operating results and financial
condition.
The U.S. Congress and state legislatures continue to focus
on health care reform. Together, the recently-enacted PPACA and
HCEARA comprise a broad health care reform initiative. While
this legislation does not adversely affect reimbursement for our
anatomic pathology services, it provides for two separate
reductions in the reimbursement rates for our clinical
laboratory services: a “productive adjustment”
(currently estimated to be between 1.1 and 1.4 percent) and
an additional 1.75 percent reduction. Each of these would
reduce the annual Consumer Price Index-based update that would
otherwise determine our reimbursement for clinical laboratory
services. The effect of the new legislation on the extent of
coverage and reimbursement for new services is uncertain. This
legislation also provides for increases in the number of persons
covered by public and private insurance programs in the U.S.
In addition, several key legislators and appointed and elected
officials have proposed significant reform to the federal health
care system. Some of the reforms call for universal health care
coverage, including the availability of a new
government-sponsored health plan, and tax levies on
laboratories. A number of states, including California,
Colorado, Connecticut, Massachusetts, New York and Pennsylvania,
are contemplating significant reform of their health insurance
markets. These federal and state proposals are still being
debated in the U.S. Congress and various legislatures.
We cannot predict whether the federal and state health care
reform legislation that has been enacted will have a material
impact on us. Further, we cannot predict whether federal or
state governments will enact any additional laws to effect
health care reform and, if any such new laws were enacted, what
their terms would be and whether or in what ways any new laws
would affect us. However, it is possible that new laws could
increase our costs, decrease our revenues, expose us to expanded
liability or require us to revise the ways in which we conduct
our business, any of which could adversely affect our operating
results and financial condition.
23
If we
fail to comply with the complex federal, state and local
government laws and regulations that apply to our business, we
could suffer severe consequences that could adversely affect our
operating results and financial condition.
Our operations are subject to extensive federal, state and local
government regulations, all of which are subject to change.
These government laws and regulations currently include, among
other things:
|
|
|
| |
•
|
the federal Anti-Kickback Statute, which prohibits persons from
knowingly and willfully soliciting, receiving, offering or
providing remuneration, directly or indirectly, in cash or in
kind, in exchange for or to induce either the referral of an
individual for, or the purchase, order or recommendation of, any
goods or service for which payment may be made under
governmental payor programs such as Medicare and Medicaid;
|
| |
| |
•
|
the federal False Claims Act, which prohibits individuals or
entities from knowingly presenting to, or causing to be
presented to, the federal government, claims for payment that
are false or fraudulent;
|
| |
| |
•
|
the Health Insurance Portability and Accountability Act, or
HIPAA, which established comprehensive federal standards with
respect to the use and disclosure of protected health
information;
|
| |
| |
•
|
the Health Information Technology for Economic and Clinical
Health Act, or HITECH Act, which was passed as part of the
American Recovery and Reinvestment Act and which strengthens
many of the requirements applicable to privacy and security,
among other things;
|
| |
| |
•
|
the Stark Law, which prohibits a physician from making a
referral to an entity for certain designated health services
reimbursed by Medicare or Medicaid if the physician (or a member
of the physician’s family) has a financial relationship
with the entity and which also prohibits the submission of any
claim for reimbursement for designated health services furnished
pursuant to a prohibited referral;
|
| |
| |
•
|
the federal Civil Monetary Penalty Law, which prohibits the
offering of remuneration or other inducements to beneficiaries
of federal health care programs to influence the
beneficiaries’ decisions to seek specific governmentally
reimbursable items or services or to choose particular providers;
|
| |
| |
•
|
the Clinical Laboratory Improvement Amendments, which requires
that laboratories be certified by the federal government or by a
federally-approved accreditation agency;
|
| |
| |
•
|
the anti-markup rule, which prohibits a physician or supplier
billing the Medicare program from marking up the price of a
purchased diagnostic service performed by another physician or
supplier who does not “share a practice” with the
billing physician or supplier;
|
| |
| |
•
|
state law equivalents of the above, such as anti-kickback and
false claims laws that may apply to items or services reimbursed
by any third-party payor, including commercial insurers;
|
| |
| |
•
|
state laws that prohibit the splitting or sharing of fees
between physicians and non-physicians;
|
| |
| |
•
|
state laws that govern the manner in which licensed physicians
can be organized to perform and bill for medical services;
|
| |
| |
•
|
the reassignment rules, which preclude Medicare payment for
covered services to anyone other than the patient, physician, or
other person who provided the service, with limited
exceptions; and
|
| |
| |
•
|
state laws that prohibit other specified practices, such as
billing an entity that does not have ultimate financial
responsibility for the service, waiving coinsurance or
deductibles, billing Medicaid a higher charge than the lowest
charge offered to another payor, and placing professionals who
draw blood, or phlebotomists, in the offices of referring
physicians.
|
We believe that we operate in material compliance with these
laws and regulations. However, these laws and regulations are
complex and, among other things, practices that are permissible
under federal law may not be permissible in all states. In
addition, these laws and regulations are subject to
interpretation by courts and enforcement agencies. Our failure
to comply could lead to civil and criminal penalties, exclusion
from participation in Medicare and Medicaid, and possible
prohibitions or restrictions on our laboratories’ ability
to provide diagnostic
24
services, and any such penalties, exclusions, prohibitions or
restrictions could have a material adverse effect on our
arrangements with managed care organizations and private payors.
Governmental investigations of laboratories have been ongoing
for a number of years and are expected to continue. In fact, a
substantial increase in funding of Medicare and Medicaid program
integrity and anti-fraud efforts has been proposed.
Investigations of our laboratories, regardless of their outcome,
could damage our reputation and adversely affect important
business relationships that we have with third parties, as well
as have a material adverse effect on our business.
The laws of many states prohibit business corporations,
including us and our subsidiaries, from owning corporations that
employ physicians, or from exercising control over the medical
judgements or decisions of physicians. These laws and their
interpretations vary from state to state and are enforced by
both the courts and regulatory authorities, each with broad
discretion. The manner in which we operate each practice is
determined primarily by the corporate practice of medicine
restrictions of the state in which the practice is located,
other applicable regulations and commercial considerations.
We believe that we are currently in material compliance with the
corporate practice of medicine laws in each of the states in
which we operate. Nevertheless, it is possible that regulatory
authorities or other parties may assert that we are engaged in
the unauthorized corporate practice of medicine. If such a claim
were successfully asserted in any jurisdiction, we could be
subject to civil and criminal penalties, which could exclude us
from participating in Medicare, Medicaid and other governmental
health care programs, or we could be required to restructure our
contractual and other arrangements.
Failure
to comply with complex federal and state laws and regulations
related to submission of claims for our services could result in
significant monetary damages and penalties and exclusion from
the Medicare and Medicaid programs.
We are subject to extensive federal and state laws and
regulations relating to the submission of claims for payment for
our services, including those that relate to coverage of our
services under Medicare, Medicaid and other governmental health
care programs, the amounts that may be billed for our services
and to whom claims for services may be submitted. Submission of
our claims is particularly complex because we provide both
anatomic pathology services and clinical laboratory tests, which
generally are paid using different reimbursement principles. Our
failure to comply with applicable laws and regulations could
result in our inability to receive payment for our services or
result in attempts by third-party payors, such as Medicare and
Medicaid, to recover payments from us that have already been
made. Submission of claims in violation of certain statutory or
regulatory requirements can result in penalties, including civil
money penalties of up to $10,000 for each item or service billed
to Medicare in violation of the legal requirement, and exclusion
from participation in Medicare and Medicaid. Government
authorities may also assert that violations of laws and
regulations related to submission of claims violate the federal
False Claims Act or other laws related to fraud and abuse,
including submission of claims for services that were not
medically necessary. We could be adversely affected if it was
determined that the services we provided were not medically
necessary and not reimbursable, particularly if it were asserted
that we contributed to the physician’s referrals of
unnecessary services to us. It is also possible that the
government could attempt to hold us liable under fraud and abuse
laws for improper claims submitted by an entity for services
that we performed if we were found to have knowingly
participated in the arrangement that resulted in submission of
the improper claims.
Our
business could be harmed by the loss or suspension of a license
or imposition of a fine or penalties under, or future changes
in, the law or regulations of the Clinical Laboratory
Improvement Amendments or those of Medicare, Medicaid or other
federal, state or local agencies.
The diagnostic testing industry is subject to extensive
regulation, and many of these statutes and regulations have not
been interpreted by the courts. CLIA requires that laboratories
be certified by the federal government or by a
federally-approved accreditation agency every two years. CLIA
mandates specific standards in the areas of personnel
qualifications, administration, proficiency testing, patient
test management, quality control, quality assurance and
inspections. CLIA regulations include special rules applicable
to cytology testing, such as pap smears, including workload
limits, specialized proficiency testing requirements that apply
not just to the laboratory,
25
but also to the individuals performing the tests, specialized
personnel standards and quality control procedures. A laboratory
may be sanctioned based on its failure to participate in an
acceptable proficiency testing program, unsatisfactory
performance in proficiency testing or for prohibited activities
related to proficiency testing, such as failing to test the
proficiency testing samples in the same manner as patient
specimens or communicating with other laboratories regarding
proficiency testing results. The sanction for failure to comply
with CLIA requirements, including proficiency testing
violations, may be suspension, revocation or limitation of a
laboratory’s CLIA certificate, as well as the imposition of
significant fines and criminal penalties. While imposition of
certain CLIA sanctions may be subject to appeal, few, if any,
such appeals have been successful. A CLIA certificate is
necessary to conduct business. As a result, any CLIA sanction or
our failure to renew a CLIA certificate could have a material
adverse effect on our business. Although each laboratory
facility is separately certified by CLIA, if the CLIA
certificate of any our laboratories is revoked, CMS could seek
revocation of our other laboratories’ CLIA certificates
based on their common ownership or operation with the laboratory
facility whose certificate was revoked. Some states have enacted
analogous state laws that are more strict than CLIA.
Changes
in laws and regulations that address billing arrangements for
our services could have a material adverse effect on our
revenues.
While we do not bill referring physicians for our services when
those services are covered under a government program, in some
cases, we do, where permissible, bill referring physicians for
services that are not covered under a government program. Laws
and regulations in several states currently preclude us from
billing referring physicians, either by requiring us to bill
directly the third-party payor or other person ultimately
responsible for payment for the service, or by prohibiting or
limiting the referring physician’s or other
purchaser’s ability to bill a greater amount than the
amount paid for the service. An increase in the number of states
whose laws prevent such arrangements could adversely affect us
by encouraging physicians to furnish such services directly or
by causing physicians to refer services to another laboratory
for testing. Currently, Medicare does not require beneficiaries
to pay coinsurance for clinical laboratory testing or subject
such tests to a deductible. From time to time, legislation has
been proposed that would subject diagnostic services to
coinsurance and deductibles. Such legislation could be enacted
in the future. Legislation subjecting diagnostic services to
coinsurance or deductibles could adversely affect our revenues
given the anticipated difficulty in collecting such amounts from
Medicare beneficiaries. In addition, we could be subject to
potential fraud and abuse violations if adequate procedures to
bill and collect copayments were not established and followed.
We are
increasingly subject to initiatives to recover improper payments
and overpayments and such initiatives could result in
significant monetary damages and penalties and exclusion from
the Medicare and Medicaid programs.
Government payors have increased initiatives to recover improper
payments and overpayments. For example, in March 2005, CMS
initiated a demonstration project using Recovery Audit
Contractors, or RACs, who are paid a contingent fee to detect
and correct improper Medicare payments. As part of their duties,
RACs collect overpayments from Medicare providers, including
those providers who were paid for services that were not
medically necessary or were incorrectly coded. Effective
January 1, 2010, the RAC program will be operated
throughout the United States on a permanent basis, and RACs will
then have authority to pursue improper payments made on or after
October 1, 2007.
Failure
to comply with environmental, health and safety laws and
regulations could adversely affect our ability to operate and
result in fines, litigation or other consequences.
We are subject to licensing and regulation under numerous
federal, state and local laws and regulations relating to the
protection of the environment and human health and safety. Our
use, generation, manufacture, handling, transportation, storage
and disposal of medical specimens, such as human tissue,
infectious and hazardous waste, and radioactive materials, as
well as the health and safety of our laboratory employees, are
covered under these laws and regulations.
In particular, the federal Occupational Safety and Health
Administration has established extensive requirements relating
to workplace safety for health care employers, including certain
laboratories, whose workers may be exposed to blood-borne
pathogens such as HIV and the hepatitis B virus. These
requirements, among other things, require work practice
controls, protective clothing and equipment, training, medical
follow-up,
vaccinations and
26
other measures designed to minimize exposure to, and the
transmission of, blood-borne pathogens. In addition, the
Needlestick Safety and Prevention Act requires, among other
things, that we include in our safety programs the evaluation
and use of engineering controls such as safety needles if found
to be effective at reducing the risk of needlestick injuries in
the workplace.
We cannot entirely eliminate the risk of accidental injury,
contamination or sabotage from working with hazardous materials
or wastes. Our general liability insurance or workers’
compensation insurance policies may not cover damages and fines
arising from biological or hazardous waste exposure or
contamination. In the event of contamination or injury, we could
be held liable for damages or subject to fines in an amount
exceeding our resources, and our operations could be suspended
or otherwise adversely affected.
Failure to comply with federal, state and local laws and
regulations could subject us to denial of the right to conduct
business, fines, criminal penalties
and/or other
enforcement actions which would have a material adverse effect
on our business. In addition, the current environmental, health
and safety requirements applicable to our business, facilities
and employees could be revised to become more stringent, and new
laws and requirements could be adopted in the future. Thus,
compliance with applicable environmental, health and safety laws
and regulations could become both more costly and more difficult
in the future.
Failure
to comply with the Health Insurance Portability and
Accountability Act security and privacy regulations may increase
our costs.
HIPAA and related regulations establish comprehensive federal
standards with respect to the use and disclosure of protected
health information by health plans, health care providers and
health care clearinghouses. Additionally, HIPAA establishes
standards to protect the confidentiality, integrity and
availability of protected health information.
Federal privacy regulations restrict our ability to use or
disclose patient identifiable laboratory data, without patient
authorization for purposes other than payment, treatment or
health care operations, as defined by HIPAA. These privacy and
security regulations provide for significant fines and other
penalties for wrongful use or disclosure of protected health
information, including civil and criminal fines and penalties.
We believe we are in substantial compliance with the privacy
regulations. However, the documentation and process requirements
of the privacy regulations are complex and subject to
interpretation and our efforts in this respect are ongoing. Our
failure to comply with the privacy regulations could subject us
to sanctions or penalties. Although the HIPAA statute and
regulations do not expressly provide for a private right of
damages, we could also incur damages under state laws to private
parties for the wrongful use or disclosure of confidential
health information or other private information. We have
policies and procedures to comply with the HIPAA regulations and
state laws. In addition, we must also comply with
non-U.S. laws
governing the transfer of health care data relating to citizens
of other countries.
Changes
in regulations or failure to follow regulations requiring the
use of “standard transactions” for health care
services issued under the Health Insurance Portability and
Accountability Act could adversely affect our profitability and
cash flows.
Pursuant to HIPAA, the Secretary of HHS has issued final
regulations designed to facilitate the electronic exchange of
information in certain financial and administrative
transactions. HIPAA transaction standards are complex and
subject to differences in interpretation by payors. For
instance, some payors may interpret the standards to require us
to provide certain types of information, including demographic
information not usually provided to us by physicians. As a
result of inconsistent application of transaction standards by
payors or our inability to obtain certain billing information
not usually provided to us by physicians, we could face
increased costs and complexity, a temporary disruption in
receipts and ongoing reductions in reimbursements and revenues.
Any future requirements for additional standard transactions,
such as claims attachments or use of a national provider
identifier, could prove technically difficult, time-consuming or
expensive to implement.
Our
business could be adversely impacted by the Centers for Medicare
& Medicaid Services’ adoption of the new coding set
for diagnoses.
CMS has adopted a new coding set for diagnosis, commonly known
as ICD-10, which significantly expands the coding set for
diagnoses. The new coding set is currently required to be
implemented by October 1, 2013. We
27
may be required to incur significant expense in implementing the
new coding set, and if we do not adequately implement it, our
business could be adversely impacted. In addition, if as a
result of the new coding set physicians fail to provide
appropriate codes for desired tests, we may not be reimbursed
for tests we perform.
We may
be subject to liability claims for damages and other expenses
not covered by insurance that could adversely impact our
operating results.
The provision of diagnostic testing services to patients may
subject us to litigation and liability for damages based on an
allegation of malpractice, professional negligence in the
performance of our treatment and related services, the acts or
omissions of our employees, or other matters. Our exposure to
this litigation and liability for damages increases with growth
in the number of our laboratories and tests performed. Potential
judgments, settlements or costs relating to potential future
claims, complaints or lawsuits could result in substantial
damages and could subject us to the incurrence of significant
fees and costs. Our insurance may not be sufficient or available
to cover these damages, costs or expenses. Our business,
profitability and growth prospects could suffer if we face
negative publicity or if we pay damages or defense costs in
connection with a claim that is outside the scope of any
applicable insurance coverage, including claims related to
contractual disputes and professional and general liability
claims.
Our
insurance costs may increase over the next several years, and
our coverage may not be sufficient to cover claims and
losses.
We maintain a program of insurance coverage against a broad
range of risks in our business. In particular, we maintain
professional liability insurance, subject to deductibles. The
premiums and deductibles under our insurance may increase over
the next several years as a result of general business rate
increases, coupled with our continued growth. We are unable to
predict whether such increases in premiums and deductibles will
occur and the amount of any such increases, but such increases
could adversely impact our earnings. The liability exposure of
operations in the health care services industry has increased,
resulting not only in increased premiums, but also in limited
liability on behalf of the insurance carriers. Our ability to
obtain the necessary and sufficient insurance coverage for our
operations upon expiration of our insurance policies may be
limited, and sufficient insurance may not be available on
favorable terms, if at all. We could be materially and adversely
affected by any of the following:
|
|
|
| |
•
|
our inability to obtain sufficient insurance for our operations;
|
| |
| |
•
|
the collapse or insolvency of one or more of our insurance
carriers;
|
| |
| |
•
|
further increases in premiums and deductibles; and
|
| |
| |
•
|
an inability to obtain one or more types of insurance on
acceptable terms.
|
Risks
Related to Our Organization and Structure
We are
a holding company and our principal asset after completion of
this offering will be our equity interests in Aurora Holdings,
and we are accordingly dependent upon distributions from Aurora
Holdings to pay dividends, if any, taxes and other
expenses.
We are a holding company and, upon completion of the
Reorganization Transactions and this offering, our principal
asset will be our indirect ownership of equity interests in
Aurora Holdings. See “Prospectus Summary —
Reorganization Transactions” and “Organizational
Structure — Reorganization Transactions.” We have
no independent means of generating revenue. We intend to cause
Aurora Holdings to make pro rata distributions to its
unitholders, including us, in an amount sufficient to cover all
applicable taxes payable. We also intend to cause Aurora
Holdings to make distributions to us, including for purposes of
paying corporate and other overhead expenses and payments under
the Tax Receivable Agreement, but our ability to make these
distributions will be limited by restrictions in our debt
agreements. To the extent that we need funds and Aurora Holdings
is restricted from making such distributions under applicable
law or regulation or as a result of the terms in our debt
agreements, or is otherwise unable to provide such funds, it
could adversely affect our liquidity and financial condition.
In addition, we are dependent on our ability to generate
revenues through contractual arrangements with our affiliated
physician practices. Any restructuring of our contractual and
other arrangements with physician practices
28
could result in lower revenues from such practices, increased
expenses in the operation of such practices and reduced
influence over the business decisions of such practices.
Alternatively, some of our existing contracts may not comply
with applicable regulations or be unenforceable, which could
result in the termination of those contracts and an associated
loss of revenue.
We are
controlled by our Principal Equityholders whose interest in our
business may be different than yours, and certain statutory
provisions afforded to stockholders are not applicable to
us.
When this offering is completed, our Principal Equityholders
will beneficially own shares representing, in
aggregate, percent
of the combined voting power of our Class A common stock
and Class B common stock
(or percent
if the underwriters exercise their over-allotment option in
full) after the completion of this offering and the application
of the net proceeds that we will receive from this offering.
Specifically, the Summit Partners Equityholders will, in
aggregate, beneficially
own percent of our common
stock immediately following the completion of this offering, the
KRG Equityholders will, in aggregate, beneficially own
approximately percent of our
common stock immediately following the completion of this
offering, and our Managing Equityholders will, in aggregate,
beneficially own
approximately percent of our
common stock immediately following the completion of this
offering.
Accordingly, our Principal Equityholders can exercise
significant influence over our business policies and affairs,
including the power to nominate our Board of Directors. In
addition, our Principal Equityholders can control any action
requiring the general approval of our stockholders, including
the adoption of amendments to our certificate of incorporation
and bylaws and the approval of mergers or sales of substantially
all of our assets. The concentration of ownership and voting
power of our Principal Equityholders may also delay, defer or
even prevent an acquisition by a third party or other change of
control of our company and may make some transactions more
difficult or impossible without the support of our Principal
Equityholders, even if such events are in the best interests of
noncontrolling stockholders. Moreover, this concentration of
ownership may make it difficult for stockholders other than our
Principal Equityholders to replace management. The concentration
of voting power among our Principal Equityholders may have an
adverse effect on the price of our Class A common stock.
We have opted out of section 203 of the General Corporation
Law of the State of Delaware, or the Delaware General
Corporation Law, which, subject to certain exceptions, prohibits
a publicly-held Delaware corporation from engaging in a business
combination transaction with an interested stockholder for a
period of three years after the interested stockholder became
such. Therefore, after the
lock-up
period expires, our Principal Equityholders are able to transfer
control of us to a third party by transferring their
Class A common stock, which would not require the approval
of our Board of Directors or our other stockholders.
Our certificate of incorporation will provide that the doctrine
of “corporate opportunity” will not apply against our
Principal Equityholders and their respective affiliates in a
manner that would prohibit them from investing in competing
businesses or doing business with our clients. To the extent
they invest in such other businesses, our Principal
Equityholders may have differing interests than our other
stockholders.
We are party to a Registration Rights Agreement with certain of
our Principal Equityholders. Under the Registration Rights
Agreement, our Principal Equityholders have certain registration
rights with respect to our Class A common stock. See
“Certain Relationships and Related Party
Transactions — Registration Rights Agreement.”
See “Principal and Selling Stockholders” and
“Certain Relationships and Related Party Transactions.”
The
U.S. Congress has enacted new legislation that affects the
taxation of our Class A common stock held by or through foreign
entities.
Recently enacted legislation generally will impose a withholding
tax of 30 percent on dividend income from our Class A common
stock and the gross proceeds of a disposition of our Class A
common stock paid to certain foreign entities after
December 31, 2012, unless the foreign entity complies with
certain conditions or an exception applies. See “Certain
U.S. Federal Income and Estate Tax Consequences to Non-U.S.
Holders of Common Stock — Recently-Enacted Federal Tax
Legislation.”
29
We
will be required to pay an affiliate of our Principal
Equityholders for certain tax benefits we may claim (but may
never fully realize) as a result of transactions before and
after this offering.
Beginning in 2006, certain acquisitions by Aurora Holdings have
resulted in an increase in the tax basis of intangible assets,
primarily goodwill, which results in tax amortization deductions
that would not have been available in the absence of those
acquisitions. The Aurora Holdings Units held by the Aurora
Holdings Continuing Members (along with shares of our
Class B common stock) will be exchangeable in the future
for cash or shares of our Class A common stock. These
future exchanges are likely to result in tax basis adjustments
to the assets of Aurora Holdings, which adjustments would also
be allocated to us. The existing tax basis and the anticipated
tax basis adjustments are expected to reduce the amount of tax
that we would otherwise be required to pay in the future.
In connection with this offering, Aurora Diagnostics, Inc. will
enter into the Tax Receivable Agreement with ARDX Sub, Aurora
Holdings and the Tax Receivable Entity that will provide for the
payment by Aurora Diagnostics Inc. to the Tax Receivable Entity
of 85 percent of certain cash tax savings, if any, in
U.S. federal, state, local and foreign income tax realized
by Aurora Diagnostics Inc. after the completion of this offering
as a result of:
|
|
|
| |
•
|
favorable tax attributes associated with amortizable goodwill
and other intangibles held by Aurora Holdings and created by its
previous acquisitions;
|
| |
| |
•
|
any step-up
in tax basis in our share of Aurora Holdings’ assets
resulting from:
|
|
|
|
| |
•
|
the acquisition by us of Aurora Holdings Units from the Aurora
Holdings Continuing Members in exchange for shares of our
Class A common stock or cash, or
|
| |
| |
•
|
payments under the Tax Receivable Agreement to the Tax
Receivable Entity; and
|
|
|
|
| |
•
|
tax benefits related to imputed interest deemed to be paid by us
as a result of the Tax Receivable Agreement.
|
The actual increase in tax basis, as well as the amount and
timing of any payments under the Tax Receivable Agreement, will
vary depending upon a number of factors, including the timing of
exchanges by the Aurora Holdings Continuing Members, the price
of our Class A common stock at the time of the exchange,
the extent to which such exchanges are taxable, the amount and
timing of the taxable income we generate in the future and the
tax rate then applicable and the portion of our payments under
the Tax Receivable Agreement constituting imputed interest or
amortizable basis.
The payments we are required to make under the Tax Receivable
Agreement could be substantial. We expect that, as a result of
the amount of the increases in the tax basis of the tangible and
intangible assets of Aurora Holdings, assuming no material
changes in the relevant tax law and that we earn sufficient
taxable income to realize in full the potential tax benefit
described above, future payments under the Tax Receivable
Agreement in respect of the existing tax attributes will
aggregate $ and range from
approximately $ to
$ per year over the
next years. These amounts reflect only the cash
tax savings attributable to current tax attributes resulting
from past acquisitions described above as well as from the
Reorganization Transactions. Future payments under the Tax
Receivable Agreement in respect of subsequent acquisitions of
Aurora Holdings Units would be in addition to these amounts and
would, if such exchanges took place at
$ per share, which is the midpoint
of the price range set forth on the cover page of this
prospectus, be of substantial magnitude.
In addition, although we do not believe that the Internal
Revenue Service, or IRS, would challenge the tax basis increases
or other benefits arising under the Tax Receivable Agreement,
the Tax Receivable Entity will not reimburse or indemnify us for
any payments previously made if such tax basis increases or
other tax benefits are subsequently disallowed or for any other
claims made by the IRS, except that excess payments made to the
Tax Receivable Entity will be netted against payments otherwise
to be made, if any, after our determination of such excess. As a
result, in such circumstances, we could make payments under the
Tax Receivable Agreement that are greater than our cash tax
savings.
Because we are a holding company with no operations of our own,
our ability to make payments under the Tax Receivable Agreement
is dependent on the ability of Aurora Holdings and its
subsidiaries to make distributions to us. Our debt agreements
will restrict the ability of our subsidiaries to make
distributions to us under some circumstances, which could affect
our ability to make payments under the Tax Receivable Agreement.
More
30
specifically, we will be able to receive distributions to make
payments under the Tax Receivable Agreement with respect to
current year cash tax savings, but such distributions will not
be increased on account of any lump sum payment arising under
the Tax Receivable Agreement. To the extent that we are unable
to make payments under the Tax Receivable Agreement because of
such restrictions, such payments will be deferred and will
accrue interest until paid.
Under the terms of the Tax Receivable Agreement, certain events
may cause the acceleration or modification of our obligation to
make payments to the Tax Receivable Entity. In the event of an
adverse change in tax law or if we choose to terminate the Tax
Receivable Agreement (with the consent of our independent
directors), we would be required to pay a lump sum amount in
lieu of the estimated future payments otherwise provided for in
the Tax Receivable Agreement. The lump sum amount would be based
on assumptions regarding tax rates and would be calculated based
on a present value of the total amount otherwise payable under
the Tax Receivable Agreement. If the assumptions used turn out
to be false, we may pay more or less than the 85 percent of the
cash tax savings that would have been realized by us.
Furthermore, in the event of a change of control, our
successor’s obligations under the Tax Receivable Agreement
would be based on the assumption that the cash tax savings to us
were realized in full. See “Certain Relationships and
Related Party Transactions — Tax Receivable
Agreement.”
Risks
Related to This Offering
We
have broad discretion in the use of the net proceeds that we
will receive from this offering and may not use them in a manner
in which our stockholders would consider
appropriate.
A portion of the net proceeds that we will receive from this
offering will be used to acquire Aurora Holdings Units (and
accompanying shares of our Class B common stock) from the
Aurora Holdings Continuing Members, but we cannot specify with
certainty the particular uses of the remaining net proceeds that
we will receive from this offering. Our management will have
broad discretion in the application of the remaining net
proceeds that we will receive from this offering, including for
any of the purposes described under the heading “Use of
Proceeds” included elsewhere in this prospectus. Our
stockholders may not agree with the manner in which our
management chooses to allocate and spend the net proceeds that
we will receive from this offering. The failure by our
management to apply these funds effectively could have a
material adverse effect on our business. Pending their use, we
may invest the net proceeds that we will receive from this
offering in a manner that does not produce income or that loses
value.
We may
become involved in securities class action litigation that could
harm our reputation and business.
The public equities markets intermittently experience
significant price and volume fluctuations that have affected the
market prices of shares of diagnostic services companies like
ours. Market fluctuations may cause the price of our stock to
decline. In the past, public companies have often been subject
to securities class action litigation following a broad decline
in the market price of their securities. This risk is especially
relevant for us because diagnostic services companies have
experienced significant stock price volatility in recent years.
We may become involved in this type of litigation in the future.
Litigation often is lengthy and costly and may divert
management’s attention and resources, which could adversely
affect our business.
Failure
to manage increased costs, including those related to company
compliance programs, as a result of operating as a public
company may have an adverse effect on our
business.
As a public company, we will incur significant additional
administrative, legal, accounting and other expenses beyond
those of a private company. In addition, the Sarbanes-Oxley Act
of 2002, as well as rules subsequently implemented by the
Securities and Exchange Commission, or the SEC, and the NASDAQ
Global Market in the past several years have imposed numerous
additional requirements on public companies. These requirements
have included the establishment and maintenance of effective
disclosure and financial controls and changes in corporate
governance practices. We will need to devote significant
resources to deal with these public company-associated
requirements, including compliance programs, investor relations
and financial reporting obligations. These rules and regulations
will increase our legal and financial compliance costs and will
make some activities more time-consuming and costly. As a public
company, it will be more difficult and more expensive for us to
obtain director
31
and officer liability insurance, and we may be required to
accept reduced policy limits and coverage or incur substantially
higher costs to obtain the same or similar coverage. If we are
not able to comply with the requirements of the Sarbanes-Oxley
Act of 2002 or if we or our independent registered public
accounting firm identify deficiencies in our internal control
over financial reporting that are deemed to be significant
deficiencies or material weaknesses, the market price of our
Class A common stock could decline. We could also be
subject to sanctions or investigations by the NASDAQ Global
Market, the SEC or other regulatory authorities, which could
adversely affect our reputation, results of operations, and
financial condition.
Failure
to achieve and maintain effective internal control over
financial reporting in accordance with Section 404 of the
Sarbanes-Oxley Act of 2002 could have a material adverse effect
on our business.
As a public company, we will be required to document and test
our internal control procedures in order to satisfy the
requirements of Section 404 of the Sarbanes-Oxley Act of
2002, which will require annual management assessments of the
effectiveness of our internal control over financial reporting.
Our independent registered public accounting firm will also be
required to deliver a report providing its own assessment of the
effectiveness of our internal control over financial reporting.
During the course of our testing, we may identify deficiencies
that we may not be able to remediate in time to meet our
deadline for compliance with Section 404. We will first be
required to comply with the requirements of Section 404 for
our fiscal year ended December 31, 2011. We also may not be
able to conclude on an ongoing basis that our internal control
over financial reporting is effective in accordance with
Section 404, and our independent registered public
accounting firm may not be able to or willing to agree with our
assessment of the effectiveness of our internal control over
financial reporting. Failure to achieve and maintain an
effective internal control environment could harm our operating
results, cause us to fail to meet our reporting obligations or
require that we restate our financial statements for prior
periods, any of which could cause a decline in the market price
of our Class A common stock. Testing and maintaining
internal control over financial reporting will also involve
significant costs and could divert management’s attention
from other matters that are important to our business.
Provisions
in our certificate of incorporation and under Delaware law may
prevent or frustrate attempts by our stockholders to change our
management and hinder efforts to acquire a controlling interest
in us.
Provisions of our certificate of incorporation and bylaws may
discourage, delay or prevent a merger, acquisition or other
change in control that stockholders may consider favorable,
including transactions in which you might otherwise receive a
premium for your shares. These provisions may also prevent or
frustrate attempts by our stockholders to replace or remove our
management. These provisions include:
|
|
|
| |
•
|
a classified board of directors;
|
| |
| |
•
|
limitations on the removal of directors;
|
| |
| |
•
|
advance notice requirements for stockholder proposals and
nominations;
|
| |
| |
•
|
the inability of stockholders to act by written consent or to
call special meetings; and
|
| |
| |
•
|
the ability of our Board of Directors to designate the terms of
and issue new series of preferred stock without stockholder
approval.
|
The affirmative vote of the holders of at least two-thirds of
our shares of capital stock entitled to vote is necessary to
amend or repeal the above provisions of our certificate of
incorporation. In addition, absent approval of our Board of
Directors, our bylaws may only be amended or repealed by the
affirmative vote of the holders of at least two-thirds of our
shares of capital stock entitled to vote.
If an
active, liquid trading market for our Class A common stock
does not develop, you may not be able to sell your shares
quickly or at or above the initial offering price.
Prior to this offering, there has not been a public market for
our Class A common stock. An active and liquid trading
market for our Class A common stock may not develop or be
sustained following the completion of this offering. You
32
may not be able to sell your shares quickly, at or above the
initial offering price or at all if trading in our stock is not
active. The initial public offering price may not be indicative
of prices that will prevail in the trading market. See
“Underwriters” for more information regarding the
factors that will be considered in determining the initial
public offering price.
If you
purchase shares of our Class A common stock in this
offering, you will suffer immediate and substantial dilution of
your investment and may suffer dilution in the
future.
Purchasers of our Class A common stock in this offering
will pay a price per share that substantially exceeds the per
share value of our tangible assets after subtracting our
liabilities and the per share price paid by our existing
stockholders. Accordingly, if you purchase shares at an assumed
initial public offering price of $
per share, you will experience immediate and substantial
dilution of $ per share,
representing the difference between our pro forma net tangible
book value per share after giving effect to this offering at the
assumed initial public offering price and the public offering
price of $ per share. In addition,
purchasers of our Class A common stock in this offering
will have contributed
approximately percent of the
aggregate price paid by all purchasers of our stock but will own
only approximately percent of
our Class A common stock outstanding after this offering.
In the future, we may also acquire other companies or assets,
raise additional needed capital or finance strategic alliances
by issuing equity, which may result in additional dilution to
you.
If
equity research analysts do not publish research or reports
about our business, or if they issue unfavorable commentary or
downgrade our Class A common stock, the price of our
Class A common stock could decline.
The trading market for our Class A common stock will rely
in part on the research and reports that equity research
analysts publish about us and our business. We do not control
these analysts or the content and opinions included in their
reports. Securities analysts may elect not to provide research
coverage of our Class A common stock after the completion
of this offering, and such lack of research coverage may
adversely affect the market price of our Class A common
stock. The price of our stock could decline if one or more
equity research analysts downgrade our stock or if those
analysts issue other unfavorable commentary or cease publishing
reports about us or our business. If one or more equity research
analysts ceases coverage of our company, we could lose
visibility in the market, which in turn could cause our stock
price to decline.
Purchasers
of our Class A common stock could incur substantial
losses.
Our stock price is likely to be volatile. The stock market in
general has experienced extreme volatility that has often been
unrelated to the operating performance of particular companies.
Investors may not be able to sell their Class A common
stock at or above the initial public offering price. The market
price for our Class A common stock may be influenced by
many factors, including:
|
|
|
| |
•
|
changes in health care coverage or reimbursement guidelines and
amounts, including health care reimbursement reform and
cost-containment measures implemented by government agencies;
|
| |
| |
•
|
changes in the structure of health care payment systems;
|
| |
| |
•
|
variations in deductible and coinsurance amounts;
|
| |
| |
•
|
regulatory developments affecting the health care or diagnostic
services industry;
|
| |
| |
•
|
our failure to comply with applicable regulations or increased
investigative or enforcement initiatives by governmental and
other third-party payors;
|
| |
| |
•
|
changes in the payor mix or the mix or cost of our specialized
diagnostic services;
|
| |
| |
•
|
the timing and volume of patient orders and seasonality of our
business;
|
| |
| |
•
|
the timing and cost of our sales and marketing efforts;
|
| |
| |
•
|
litigation involving our company, our industry, or both;
|
| |
| |
•
|
the departure of key personnel;
|
33
|
|
|
| |
•
|
our ability to continue to add new laboratories;
|
| |
| |
•
|
future sales of our Class A common stock;
|
| |
| |
•
|
variations in our financial results or those of companies that
are perceived to be similar to us;
|
| |
| |
•
|
changes in market valuations of companies similar to ours;
|
| |
| |
•
|
changes in recommendations by securities analysts and
investors’ perceptions of us;
|
| |
| |
•
|
changes in our capitalization, including future issuances of our
Class A common stock or the incurrence of additional
indebtedness; and
|
| |
| |
•
|
general economic, industry and market conditions.
|
Shares
eligible for future sale by existing stockholders may adversely
affect our stock price.
Sales of a substantial number of shares of our Class A
common stock in the public market could occur at any time,
particularly after the expiration of the
lock-up
agreements described in the “Underwriters” section of
this prospectus. These sales, or the perception in the market
that the holders of a large number of shares intend to sell
shares, could reduce the market price of our Class A common
stock.
After the closing of this offering, we will
have
outstanding shares of our Class A common stock, after
giving effect to the sale
of shares
of our Class A common stock offered by us and the selling
stockholders in this offering at a purchase price equal to the
assumed initial public offering price of
$ per share, which is the midpoint
of the price range set forth on the cover page of this
prospectus, after deducting the estimated underwriting discount
and commissions and offering expenses payable by us.
This also includes the shares that we are selling in this
offering, which may be resold in the public market immediately.
Of the remaining
shares, shares
are currently restricted as a result of securities laws or
lock-up
agreements but will be available for resale in the public market
as described in the “Shares Eligible for Future
Sale” section of this prospectus.
Our Principal Equityholders own
approximately shares
of our Class A common stock or
approximately percent
of our outstanding Class A common stock. If the Aurora
Holdings Continuing Members exchanged all of their Aurora
Holdings Units (and accompanying shares of our Class B
common stock) for shares of our Class A common stock, the
Aurora Holdings Continuing Members would hold an
additional shares of our
Class A common stock, or
approximately percent of our
outstanding Class A common stock in the aggregate. All of
the shares held by our Principal Equityholders may be sold
without complying with the registration provisions of the
Securities Act, as amended, or the Securities Act, upon
satisfying the conditions of Rule 144 of the Securities
Act. The sale of shares by these Principal Equityholders under
Rule 144 may have an adverse affect on the market price of
our Class A common stock and may inhibit our ability to
manage subsequent equity or debt financing.
If a large number of shares of our Class A common stock or
securities convertible into our Class A common stock are
sold in the public market after they become eligible for sale,
the sales could reduce the trading price of our Class A
common stock and impede our ability to raise future capital.
We
have not paid cash dividends and do not expect to pay dividends
in the future, which means that you may not be able to realize
the value of our shares except through sale.
Although Aurora Holdings has made tax and other distributions to
its members in accordance with the Aurora Holdings LLC
Agreement, we have never declared or paid cash dividends. We
currently expect to retain earnings for our business and do not
anticipate paying dividends on our Class A common stock at
any time in the foreseeable future. Our Board of Directors will
decide whether to pay dividends on our Class A common stock
from time to time in the exercise of its business judgment.
Because we do not anticipate paying dividends in the future, the
only opportunity to realize the value of our Class A common
stock will likely be through an appreciation in value and a sale
of those shares. There is no guarantee that shares of our
Class A common stock will appreciate in value or even
maintain the price at which our stockholders have purchased
their shares.
34
FORWARD-LOOKING
STATEMENTS
This prospectus includes forward-looking
statements. All statements other than statements of
historical facts contained in this prospectus, including
statements regarding our future results of operations and
financial position, business strategy and plans and objectives
for future operations, are forward-looking statements. The words
“believe,” “may,” “might,”
“will,” “estimate,” “continue,”
“anticipate,” “intend,” “expect,”
“plan,” “could,” “would” and
similar expressions are intended to identify forward-looking
statements. We have based these forward-looking statements
largely on our current expectations and projections about future
events and financial trends that we believe may affect our
financial condition, results of operations, business strategy,
short-term and long-term business operations and objectives, and
financial needs. We caution the forward-looking statements in
this prospectus are subject to a number of known and unknown
risks, uncertainties and assumptions that may cause our actual
results, performance or achievements to be materially different
from any future results, performances or achievements expressed
or implied by the forward-looking statements. In addition to the
risks described in “Risk Factors,” factors that could
contribute to these differences include, among other things:
|
|
|
| |
•
|
changes in medical treatment or reimbursement rates or
utilization for our anatomic pathology markets;
|
| |
| |
•
|
competition for our diagnostic services, including the
internalization of testing functions and technologies by our
clients;
|
| |
| |
•
|
changes in payor regulations, policies or payor mix;
|
| |
| |
•
|
the anticipated benefits from acquisitions not being fully
realized or not being realized within the expected time frames;
|
| |
| |
•
|
disruptions or failures of our IT solutions or infrastructure;
|
| |
| |
•
|
loss of key executives and technical personnel;
|
| |
| |
•
|
the failure to maintain relationships with clients, including
referring physicians and hospitals, and with payors;
|
| |
| |
•
|
covenants in our debt agreements;
|
| |
| |
•
|
our substantial amount of indebtedness;
|
| |
| |
•
|
the protection of our intellectual property;
|
| |
| |
•
|
general economic, business or regulatory conditions affecting
the health care and diagnostic testing services industries;
|
| |
| |
•
|
federal or state health care reform initiatives;
|
| |
| |
•
|
violation of, failure to comply with, or changes in federal and
state laws and regulations related to, submission of claims for
our services, fraud and abuse, patient privacy, and billing
arrangements for our services;
|
| |
| |
•
|
attainment of licenses required to test patient specimens from
certain states or the loss or suspension of licenses;
|
| |
| |
•
|
control by our Principal Equityholders;
|
| |
| |
•
|
payments under the Tax Receivable Agreement;
|
| |
| |
•
|
compliance with certain corporate governance requirements and
costs incurred in connection with becoming a public company;
|
| |
| |
•
|
failure to establish and maintain internal controls over
financial reporting; and
|
| |
| |
•
|
the other factors discussed under the heading “Risk
Factors” and elsewhere in this prospectus.
|
Moreover, we operate in a very competitive and rapidly changing
environment, and new risks emerge from
time-to-time.
It is not possible for our management to predict all risks, nor
can we assess the impact of all factors on
35
our business or the extent to which any factor, or combination
of factors, may cause actual results to differ materially from
those contained in any forward-looking statements we may make.
In light of these risks, uncertainties and assumptions, the
forward-looking events and circumstances discussed in this
prospectus may not occur, and actual results could differ
materially and adversely from those anticipated or implied in
the forward-looking statements.
You should not rely upon forward-looking statements as
predictions of future events. Although we believe that the
expectations reflected in the forward-looking statements are
reasonable, we cannot guarantee that the future results, levels
of activity, performance or events and circumstances reflected
in the forward-looking statements will be achieved or occur.
Moreover, neither we nor any other person assumes responsibility
for the accuracy and completeness of the forward-looking
statements. We undertake no obligation to update publicly any
forward-looking statements for any reason after the date of this
prospectus to conform these statements to actual results or
changes in our expectations.
The forward-looking statements in this prospectus speak only as
of the date of this prospectus. You should assume that the
information appearing in this prospectus is accurate only as of
the date on the front cover of this prospectus. Our business,
financial condition, results of operations, or prospects may
have changed since that date. Neither the delivery of this
prospectus nor the sale of the common shares means that
information contained in this prospectus is correct after the
date of this prospectus. Except as otherwise required by
applicable laws, we undertake no obligation to publicly update
or revise any forward-looking statements, the risk factors or
other information described in this prospectus, whether as a
result of new information, future events, changed circumstances
or any other reason after the date of this prospectus.
The Private Securities Litigation Reform Act of 1995 and
Section 27A of the Securities Act do not protect any
statements we make in connection with this offering.
This prospectus also contains market data related to our
business and industry. These market data include projections
that are based on a number of assumptions. While we believe
these assumptions to be reasonable and sound as of the date of
this prospectus, if these assumptions turn out to be incorrect,
actual results may differ from the projections based on these
assumptions. As a result, our markets may not grow at the rates
projected by these data, or at all. The failure of these markets
to grow at these projected rates may have a material adverse
effect on our business, results of operations, financial
condition and the market price of our Class A common stock.
36
ORGANIZATIONAL
STRUCTURE
Structure
Prior to the Reorganization Transactions
We were incorporated in Delaware on April 23, 2010. We are
a holding company, and our principal asset after the completion
of this offering will be our indirect equity interest in Aurora
Holdings. We have not engaged in any business or other
activities except for activities in contemplation of this
offering. We currently expect that our only business or other
activities will be our indirect investment in Aurora Holdings
and our participation in the Reorganization Transactions. Aurora
Holdings and its subsidiaries have historically conducted the
business described in this prospectus. Following the completion
of this offering and the Reorganization Transactions, we expect
to conduct our business through Aurora Holdings and its
subsidiaries.
Aurora Holdings was organized in 2006 as a limited liability
company to act as a holding company for Aurora Diagnostics, LLC
and our other operating subsidiaries. Aurora Holdings was
initially capitalized by affiliates of Summit Partners,
affiliates of GSO Capital Partners and members of our senior
management team. In June 2009, GSO Capital Partners’ equity
interest in Aurora Holdings was purchased by an affiliate of KRG
Capital Partners.
We are authorized to issue two classes of common stock:
Class A common stock and Class B common stock. Each
share of the Class A common stock and Class B common
stock provides the holder with one vote on all matters submitted
to a vote of stockholders; however, the holders of Class B
common stock do not have any of the economic rights (including
rights to dividends and distributions upon liquidation, but
excluding the return of the par value on liquidation) provided
to holders of Class A common stock. All shares of our
common stock generally vote together, as a single class, on all
matters submitted to a vote of stockholders. Prior to giving
effect to the Reorganization Transactions, all of our
outstanding common stock is and will be held by James C. New,
our Chairman, Chief Executive Officer and President.
Prior to the Reorganization Transactions, Aurora Holdings had
131,382 outstanding membership interests in eight classes,
including 21,382
Class A-1
Units, 85,000 Class A Units, 10,000 Class B Units,
5,000 Class C Units, 4,000
Class D-1
Units, 3,000
Class D-2
Units, 3,000
Class D-3
Units and Class X capital of $7.1 million. Prior to
giving effect to the Reorganization Transactions, the Aurora
Holdings Units are owned as follows:
|
|
|
| |
•
|
the Summit Partners Equityholders currently own 51 percent
of the economic interest in Aurora Holdings;
|
|
|
|
| |
•
|
the KRG Equityholders currently own 34 percent of the
economic interest in Aurora Holdings; and
|
|
|
|
| |
•
|
the Management Equityholders currently own 15 percent of
the economic interest in Aurora Holdings.
|
37
The diagram below depicts the organizational structure of Aurora
Holdings prior to the Reorganization Transactions.
Reorganization
Transactions
In connection with this offering, we will enter into the
Reorganization Transactions. As part of the Reorganization
Transactions:
|
|
|
| |
•
|
We will form ARDX Sub and certain entities organized and
controlled, respectively, by the Summit Partners Equityholders
and the KRG Equityholders will merge with and into ARDX Sub, and
ARDX Sub will be the surviving entity in the merger and will
hold all of the Aurora Holdings Units previously owned by those
entities; the stockholders of each of those entities will
receive in the merger an aggregate
of shares
of our Class A common stock and TRA Rights;
|
| |
| |
•
|
ARDX Sub will acquire additional Aurora Holdings Units from
entities organized and controlled by the Summit Partners
Equityholders in exchange for shares of our Class A common
stock and TRA Rights;
|
| |
| |
•
|
Aurora Holdings will enter into the Second Amended and Restated
Aurora Holdings LLC Agreement such that all of its outstanding
Class A-1
Units, Class A Units, Class B Units, Class C
Units,
Class D-1
Units,
Class D-2
Units,
Class D-3
Units and Class X capital will be reclassified as Aurora
Holdings Units (all of which will be of a single class), and the
Aurora Holdings Continuing Members will also receive rights to
distributions that are calculated in a manner that is similar to
the TRA Rights under the Second Amended and Restated Aurora
Holdings LLC Agreement;
|
| |
| |
•
|
ARDX Sub will acquire all business and operational control of
Aurora Holdings, and an immaterial amount of cash, from the
Aurora Holdings Continuing Members in exchange for newly-issued
shares of our Class B common stock;
|
| |
| |
•
|
Certain of the Summit Partners Equityholders and the KRG
Equityholders and all of the Management Equityholders will form
the Tax Receivable Entity;
|
|
|
|
| |
•
|
Certain of the Summit Partners Equityholders and the KRG
Equityholders and all of the Management Equityholders will
contribute TRA Rights for interests in the Tax Receivable
Entity, and we, ARDX Sub and Aurora Holdings will enter into the
Tax Receivable Agreement with the Tax Receivable Entity; and
|
|
|
|
| |
•
|
We will use a portion of the net proceeds that we will receive
from this offering to acquire, through ARDX Sub, a portion of
the Aurora Holdings Units (and a corresponding number of shares
of Class B common stock) held by the Aurora Holdings Continuing
Members in exchange for cash and TRA Rights.
|
All of the Reorganization Transactions will be consummated
immediately prior to the closing of this offering.
38
The diagram below depicts our organizational structure
immediately prior to this offering after giving effect to the
Reorganization Transactions.
|
|
|
|
(1) |
|
The Summit Partners Equityholders will
hold percent of the Aurora Holdings
Units. |
|
|
|
|
(2) |
|
The Summit Partners Equityholders will
hold percent of our Class A common
stock. |
|
|
|
|
(3) |
|
The Summit Partners Equityholders will
hold percent of our Class B common
Stock. |
|
|
|
|
(4) |
|
The KRG Equityholders will
hold percent of our Class A
common stock. |
|
|
|
|
(5) |
|
The Management Equityholders will
hold percent of our Class B
common stock. |
|
|
|
|
(6) |
|
The Management Equityholders will
hold percent of the Aurora Holdings
Units. |
|
|
|
|
(7) |
|
We will hold 100 percent of ARDX Sub common stock. |
|
|
|
|
(8) |
|
ARDX Sub will hold percent of the
Aurora Holdings Units. |
All of these Reorganization Transactions will be consummated
immediately prior to the closing of this offering. We intend to
account for the Reorganization Transactions using a carryover
basis as the contemplated transactions are exchanges among
entities under common control that do not affect economic
ownership.
See “— Holding Company Structure and Tax
Receivable Agreement” and “Certain Relationships and
Related Party Transactions.”
Effect of
the Reorganization Transactions and this Offering
The Reorganization Transactions are intended to create a
corporate holding company that will facilitate public ownership
of, and investment in, us.
As part of this offering, the Summit Partners Equityholders and
KRG Equityholders will
sell shares
of our Class A common stock. We will not receive any of the
proceeds from the sale of shares of our Class A common
stock in this offering by the selling stockholders. Immediately
following the completion of this offering, we will use a portion
of the net proceeds that we will receive from this offering,
along with TRA Rights, to
purchase shares
of our
39
Class B common stock and an equal number of Aurora Holdings
Units from the Aurora Holdings Continuing Members. This will
further reduce the interest of the Aurora Holdings Continuing
Members in us.
Upon completion of the Reorganization Transactions, this
offering and the application of the net proceeds that we will
receive from this offering, our Class A common stock will
be held as follows:
|
|
|
| |
•
|
Our public stockholders will hold an aggregate
of shares of our Class A
common stock
(or shares
if the underwriters exercise their over-allotment option in
full), representing percent
of the combined voting power in us
(or percent if the
underwriters exercise their over-allotment option in full)
and percent of the economic
interest in us (or percent if
the underwriters exercise their over-allotment option in full);
|
| |
| |
•
|
Certain Summit Partners Equityholders will hold an aggregate
of shares
of our Class A common stock
(or shares
if the underwriters exercise their over-allotment option in
full),
representing percent
of the combined voting power in us
(or percent
if the underwriters exercise their over-allotment option in
full) and percent of the
economic interest in us
(or percent
if the underwriters exercise their over-allotment option in
full);
|
| |
| |
•
|
The KRG Equityholders will hold an aggregate
of shares
of our Class A common stock
(or shares
if the underwriters exercise their over-allotment option in
full), representing percent
of the combined voting power in us
(or percent if the
underwriters exercise their over-allotment option in full)
and percent of the economic
interest in us (or percent if
the underwriters exercise their over-allotment option in
full); and
|
| |
| |
•
|
The Management Equityholders will hold no shares of our
Class A common stock.
|
Upon completion of the Reorganization Transactions, this
offering and the application of the net proceeds that we will
receive from this offering, our Class B common stock will
be held as follows:
|
|
|
| |
•
|
Certain of the Summit Partners Equityholders will hold an
aggregate
of shares
of our Class B common stock
(or shares
if the underwriters exercise their over-allotment in full),
representing percent of the
combined voting power in us
(or percent if the
underwriters exercise their over-allotment in full) and none of
the economic interest in us; and
|
| |
| |
•
|
The KRG Equityholders will hold no shares of our Class B
common stock; and
|
| |
| |
•
|
The Management Equityholders will hold an aggregate
of shares
of our Class B common stock
(or shares
if the underwriters exercise their over-allotment in full),
representing percent of the
combined voting power in us
(or percent if the
underwriters exercise their over-allotment in full) and none of
the economic interest in us.
|
Upon completion of the Reorganization Transactions, this
offering and the application of the net proceeds that we will
receive from this offering, the Aurora Holdings Units will be
held as follows:
|
|
|
| |
•
|
We will indirectly, through ARDX Sub, be the sole managing
member of Aurora Holdings, will have all business and
operational control of Aurora Holdings, and will indirectly hold
an aggregate of Aurora
Holdings Units (or
Aurora Holdings Units if the Underwriters exercise their
over-allotment in full), representing 100 percent of the
voting power in Aurora Holdings
and percent of the economic
interest in Aurora Holdings (or
percent if the Underwriters exercise their over-allotment in
full). We will consolidate the financial results of Aurora
Holdings, and our net income (loss) will be reduced by a
noncontrolling interest expense to reflect the entitlement of
the Aurora Holdings Continuing Members to a portion of Aurora
Holdings’ net income (loss);
|
|
|
|
| |
•
|
Certain of the Summit Partners Equityholders, will hold an
aggregate
of
Aurora Holdings Units (or Aurora
Holdings Units if the underwriters exercise their over-allotment
in full), representing none of the voting power in Aurora
Holdings and percent of the
economic interest in Aurora Holdings
(or percent if the underwriters
exercise their over-allotment in full);
|
|
|
|
| |
•
|
The KRG Equityholders will hold no Aurora Holdings
Units; and
|
|
|
|
| |
•
|
The Management Equityholders will hold an aggregate
of
Aurora Holdings Units (or Aurora
Holdings Units if the underwriters exercise their over-allotment
in full), representing none of the voting
|
40
|
|
|
| |
|
power in Aurora Holdings
and percent of the economic
interest in Aurora Holdings (or
percent if the underwriters exercise their over-allotment in
full).
|
The diagram below depicts our organizational structure after
giving effect to the Reorganization Transactions and after
giving effect to this offering and the application of the net
proceeds that we will receive from this offering.
|
|
|
|
(1) |
|
The Summit Partners Equityholders will
hold percent of the Aurora
Holdings Units. |
|
|
|
|
(2) |
|
The Summit Partners Equityholders will
hold percent of our Class A
common stock. |
|
|
|
|
(3) |
|
The Summit Partners Equityholders will
hold percent of our Class B
common stock. |
|
|
|
|
(4) |
|
Public stockholders will hold
percent of our Class A common stock. |
|
|
|
|
(5) |
|
The KRG Equityholders will hold
percent of our Class A common stock. |
|
|
|
|
(6) |
|
The Management Equityholders will
hold percent of our Class B
common stock. |
|
|
|
|
(7) |
|
The Management Equityholders will
hold percent of the Aurora
Holdings Units. |
|
|
|
|
(8) |
|
We will hold 100 percent of ARDX Sub common stock. |
|
|
|
|
(9) |
|
ARDX Sub will hold percent of the
Aurora Holdings Units. |
Holding
Company Structure and Tax Receivable Agreement
We are a holding company and, immediately after the consummation
of the Reorganization Transactions and the completion of this
offering, our principal asset will be our indirect interest in
Aurora Holdings. We do not intend to list our Class B
common stock on any stock exchange.
The Tax Receivable Entity will be formed with our Principal
Equityholders holding its equity interests. Aurora Diagnostics,
Inc. will enter into the Tax Receivable Agreement with ARDX Sub,
Aurora Holdings and the Tax Receivable Entity that will provide
for the payment by Aurora Diagnostics, Inc. to the Tax
Receivable Entity of
41
85 percent of certain cash tax savings, if any, in
U.S. federal, state, local and foreign income tax realized
by Aurora Diagnostics, Inc. after the completion of this
offering as a result of:
|
|
|
| |
•
|
favorable tax attributes associated with amortizable goodwill
and other intangibles held by Aurora Holdings and created by its
previous acquisitions;
|
| |
| |
•
|
any step-up
in tax basis in our share of Aurora Holdings’ assets
resulting from:
|
|
|
|
| |
•
|
the acquisition by us of Aurora Holdings Units from the Aurora
Holdings Continuing Members in exchange for shares of our
Class A common stock or cash, or
|
| |
| |
•
|
payments under the Tax Receivable Agreement to the Tax
Receivable Entity; and
|
|
|
|
| |
•
|
tax benefits related to imputed interest deemed to be paid by us
as a result of the Tax Receivable Agreement.
|
We are entering into the Tax Receivable Agreement because
favorable tax attributes have been or will be made available to
us as a result of transactions before and after the offering.
Our Principal Equityholders believe that the value of these tax
attributes should be considered in determining the value of
their contribution to us. As it may be difficult to determine
the present value of these tax attributes with a reasonable
level of certainty, the Tax Receivable Agreement with the Tax
Receivable Entity will obligate Aurora Diagnostics, Inc. to make
payments to the Tax Receivable Entity of 85 percent of
certain cash tax savings, if any, in U.S. federal, state,
local and foreign income tax realized by Aurora Diagnostics,
Inc. as a result of these attributes. Aurora Diagnostics, Inc.
will retain the benefit of the remaining 15 percent of
these certain cash tax savings.
In addition, future exchanges of Aurora Holdings Units for
shares of our Class A common stock or cash, as well as
payments under the Tax Receivable Agreement, will produce
additional favorable tax attributes to us, which would not be
available in the absence of such exchanges. The Tax Receivable
Agreement therefore will obligate Aurora Diagnostics, Inc. to
make payments to the Tax Receivable Entity of 85 percent of
certain cash tax savings, if any, realized by Aurora
Diagnostics, Inc. as a result of those additional tax
attributes. Aurora Diagnostics, Inc. will also retain the
benefit of the remaining 15 percent of these additional
certain cash tax savings.
Although we do not believe that the IRS would challenge the tax
basis increases or other benefits arising under the Tax
Receivable Agreement, the Tax Receivable Entity will not
reimburse or indemnify us for any payments previously made if
such tax basis increases or other tax benefits are subsequently
disallowed or for any other claims made by the IRS, except that
excess payments made to the Tax Receivable Entity will be netted
against payments otherwise to be made, if any, after our
determination of such excess. As a result, in such
circumstances, we could make payments to the Tax Receivable
Entity under the Tax Receivable Agreement that are greater than
our cash tax savings. See “Certain Relationships and
Related Party Transactions — Tax Receivable
Agreement.”
As a member of Aurora Holdings, ARDX Sub will incur
U.S. federal, state, local and foreign income taxes on its
allocable share of any net taxable income of Aurora Holdings. As
will be authorized by the Second Amended and Restated Aurora
Holdings LLC Agreement and to the extent permitted under our
debt agreements, we intend for our subsidiary to cause Aurora
Holdings to continue to distribute cash to its members (which,
after consummation of the Reorganization Transactions and the
completion of this offering, will consist of ARDX Sub and the
Aurora Holdings Continuing Members) at least to the extent
necessary to provide sufficient funds to each member to pay its
tax liabilities, if any, with respect to the taxable income of
Aurora Holdings.
In addition, the Second Amended and Restated Aurora Holdings LLC
Agreement will permit Aurora Holdings to make cash distributions
to its members (which, after consummation of the Reorganization
Transactions and the completion of this offering, will consist
of ARDX Sub and the Aurora Holdings Continuing Members) that are
calculated in a manner similar to the TRA Rights, which we refer
to as TRA Distributions. These TRA Distributions will enable us
to make payments under the Tax Receivable Agreement. In the
event of a termination or change in law giving rise to a lump
sum payment obligation under the Tax Receivable Agreement
(discussed below), the TRA Distributions will be limited to
amounts that otherwise would have been distributed absent such
event. If such lump sum payment obligation arises, then we alone
will be entitled to receive further TRA Distributions, which we
will use to pay down the lump sum payment obligation.
See “Certain Relationships and Related Party
Transactions — Second Amended and Restated Aurora
Holdings Limited Liability Company Agreement” and
“Management’s Discussion and Analysis of Financial
Condition and Results of Operations — Liquidity and
Capital Resources.”
The obligations resulting from the Tax Receivable Agreement that
will be entered into are expected to be more than offset by the
tax benefits that we will receive in connection with the
Reorganization Transactions and
42
subsequent exchanges. Although not assured, we expect that the
consideration that we will remit under the Tax Receivable
Agreement will not exceed the tax liability that we otherwise
would have been required to pay absent the transfers of tax
attributes indirectly to us as a result of the Reorganization
Transactions and subsequent exchanges.
Rights to receive payments under the Tax Receivable Agreement
may be terminated by the Tax Receivable Entity if, as the result
of an actual or proposed change in law, the existence of the
agreement would cause recognition of ordinary income (instead of
capital gain) in connection with future exchanges of Aurora
Holdings Units for cash or shares of Class A common stock
or would otherwise have material adverse tax consequences to the
Tax Receivable Entity or its owners. There have been legislative
proposals in the U.S. Congress that, if enacted, may result in
such ordinary income recognition. Further, in the event of such
a termination, the Tax Receivable Entity would have the right,
subject to the delivery of an appropriate tax opinion, to
require us to pay a lump sum amount in lieu of the payments
otherwise provided under the agreement. That lump sum amount
would be calculated by increasing the portion of the tax savings
retained by us to 30 percent (from 15 percent) and by
calculating a present value for the total amount that would
otherwise be payable under the agreement, using a discount rate
and assumptions as to income tax rates and as to our ability to
utilize the tax benefits (including the assumption that we will
have sufficient taxable income to fully utilize the tax benefits
and the assumption that all exchanges that have not taken place
will take place as of the effective date of the acceleration,
which will increase the amount of the lump sum payment). If the
assumptions used in this calculation turn out not to be true, we
may pay more or less than the specified percentage of our actual
cash tax savings. This lump sum amount is subordinate to amounts
payable under our new credit facilities and may be paid in cash
or be deferred until all amounts payable under our credit
facilities in existence as of the date of termination of the Tax
Receivable Agreement have been paid, and the deferred amount
will bear interest at a rate of the lesser of 6.5 percent
or 1-year
LIBOR plus 2.0 percent per annum. In view of the foregoing
changes in the calculation of our obligations, we do not expect
that the net impact of any such acceleration upon our overall
financial condition would be materially adverse as compared to
our obligations if laws do not change and the obligations are
not accelerated. It is also possible that the net impact of such
an acceleration would be beneficial to our overall financial
condition. The ultimate impact of a decision by the Tax
Receivable Entity to accelerate will depend on what the ongoing
payments would have been under the Tax Receivable Agreement
absent acceleration, which will depend on various factors.
We also have the right (with the consent of our independent
directors) to terminate the Tax Receivable Agreement. If we
exercise this right, then the Tax Receivable Entity would be
entitled to a lump sum amount in lieu of the payments otherwise
provided under the agreement. That lump sum amount would be
calculated by determining a present value for the total amount
that would otherwise be payable under the agreement, using a
discount rate and assumptions as to income tax rates and as to
our ability to utilize the tax benefits (including the
assumption that we will have sufficient taxable income to fully
utilize the tax benefits and the assumption that all exchanges
that have not taken place will take place as of the date of the
termination, which will increase the amount of the lump sum
payment). If the assumptions used in this calculation turn out
not to be true, we may pay more or less than the specified
percentage of certain cash tax savings realized by us after the
completion of this offering. This lump sum amount must be paid
in cash or be deferred until all amounts payable under our
credit facilities in existence as of the date of termination of
the Tax Receivable Agreement have been paid. Any such
acceleration can occur only at our election. Should we elect to
terminate the Tax Receivable Agreement, we do not expect that
the net impact of any such acceleration upon our overall
financial condition would be materially adverse as compared to
our existing obligations. The ultimate impact of a decision by
the Tax Receivable Entity to accelerate will depend on what the
ongoing payments would have been under the Tax Receivable
Agreement absent acceleration, which will in turn depend on the
various factors mentioned above.
If we default on any of our material obligations under the Tax
Receivable Agreement, then, unless the Tax Receivable Entity
seeks specific performance of the Tax Receivable Agreement, the
Tax Receivable Entity has the option to accelerate payments due
under the Tax Receivable Agreement and require us to make a lump
sum payment representing all past due and future payments under
the Tax Receivable Agreement, discounted to present value.
In addition, the Tax Receivable Agreement provides that, upon
certain mergers, asset sales or other forms of business
combination or certain other changes of control, our or our
successor’s obligations with respect to tax benefits would
be based on certain assumptions, including that we or our
successor would have sufficient taxable income to fully utilize
the deductions arising from the increased tax deductions and tax
basis and other benefits covered by the Tax Receivable
Agreement. As a result, upon a change of control, we could be
required to make payments under the Tax Receivable Agreement
that are greater than the specified percentage of our cash tax
savings.
43
USE OF
PROCEEDS
We estimate that the net proceeds that we will receive from the
sale of the Class A common stock offered by us will be
approximately $ million,
assuming an initial public offering price of
$ per share, which is the midpoint
of the price range set forth on the cover page of this
prospectus, and after deducting estimated underwriting discounts
and commissions and estimated offering expenses payable by us
and after giving effect to estimates of certain expenses that we
expect to be reimbursed. We will not receive any proceeds from
the sale of shares of Class A common stock by the selling
stockholders, including any proceeds resulting from the
underwriters’ exercise of their option to purchase
additional shares from the selling stockholders. A $1.00
increase (decrease) in the assumed initial public offering price
of $ per share would increase
(decrease) the amount of proceeds that we will receive from this
offering by $ million,
assuming the number of shares offered by us, which is set forth
on the cover page of this prospectus, remains the same, and
after deducting the estimated underwriting discounts and
commissions and estimated offering expenses payable by us.
The principal purposes of this offering are to raise capital to
acquire Aurora Holdings Units in connection with this offering
and to increase our capitalization and financial flexibility,
fund our growth, provide a public market for our Class A
common stock and facilitate access to public capital markets. We
intend to use the remainder of the net proceeds that we will
receive from this offering for working capital and other general
corporate purposes, which may include general and administrative
expenditures, sales and marketing expenditures, developing new
products and funding acquisitions and capital expenditures. We
may use a portion of the net proceeds that we will receive from
this offering to acquire businesses, products, services or
technologies. While we do not have commitments for any specific
acquisitions at this time, we would expect such acquisitions to
primarily include businesses that provide pathology diagnostic
testing services or services that we deem complementary to our
existing businesses. Based on an assumed initial public offering
price of $ per share, which is the
midpoint of the price range set forth on the cover page of this
prospectus, we intend to use $ of
the proceeds from this offering to
purchase
Aurora Holdings Units held by the Aurora Holdings Continuing
Members or shares of Class B
common stock (or $
or
Aurora Holdings Units or shares
of Class B common stock if the underwriters exercise their
over-allotment option in full). As a result of this purchase,
certain Aurora Holdings Continuing Members may be obligated to
make filings under Section 16(a) of the Securities Exchange
Act of 1934, as amended, or the Exchange Act, reporting a change
in beneficial ownership. We will have broad discretion in the
way we use the net proceeds that we will receive from this
offering.
The amount of net proceeds from this offering that will be used
to acquire Aurora Holdings Units will be directly related to the
number of shares of Class A common stock that we issue in
this offering. However, the amounts that we actually expend for
the other purposes specified above such as growth, working
capital and general corporate purposes may vary significantly
depending on a number of factors, including changes in our
growth strategy, the amount of our future revenues and expenses
and our future cash flow. As a result, we will retain broad
discretion in the allocation of the net proceeds that we will
receive from this offering among these specified purposes. You
will not have an opportunity to evaluate the economic, financial
or other information on which we base our decisions regarding
the use of the net proceeds that we will receive from this
offering.
Pending the uses described above, we may invest the net proceeds
that we receive from this offering in short-term,
interest-bearing, investment-grade securities.
DIVIDEND
POLICY
Although Aurora Holdings has made tax and other distributions to
its members in accordance with the Aurora Holdings LLC
Agreement, we have never declared or paid cash dividends on our
common or preferred stock. We currently do not anticipate paying
any cash dividends in the foreseeable future. We intend to
retain any earnings to finance the development and expansion of
our business. Any future determination to declare cash dividends
will be made at the discretion of our Board of Directors,
subject to applicable laws, and will depend on our financial
condition, results of operations, contractual restrictions,
capital requirements, general business conditions and other
then-existing factors that our Board of Directors may deem
relevant.
44
CAPITALIZATION
The following table sets forth our actual capitalization as of
March 31, 2010 and on a pro forma, as adjusted, basis to
reflect:
|
|
|
| |
•
|
the Reorganization Transactions described under “Prospectus
Summary — Reorganization Transactions” and
“Organizational Structure;”
|
| |
| |
•
|
the sale
of shares
of our Class A common stock by us in this offering at an
assumed initial public offering price of
$ per share, which is the midpoint
of the price range set forth on the cover page of this
prospectus, after deducting the underwriters’ discounts and
commissions and the estimated offering expenses;
|
|
|
|
| |
•
|
the execution of our new credit facilities; and
|
|
|
|
| |
•
|
the application of the net proceeds that we will receive from
this offering as described under “Use of Proceeds.”
|
The information below is illustrative only and our cash, cash
equivalents and short-term investments and capitalization
following the completion of this offering will be based on the
actual initial public offering price and other terms of this
offering determined at pricing. You should read this table
together with “Management’s Discussion and Analysis of
Financial Condition and Results of Operations” and our
consolidated financial statements and related notes included
elsewhere in this prospectus.
| |
|
|
|
|
|
|
|
|
|
|
|
March 31, 2010
|
|
|
|
|
|
|
|
Pro Forma,
|
|
|
|
|
Actual(1)
|
|
|
as
Adjusted(3)
|
|
|
|
|
(unaudited)
|
|
|
|
|
(in thousands except share data)
|
|
|
|
|
Cash, cash equivalents and short-term investments
|
|
$
|
3,222
|
|
|
$
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Current and long-term
debt(2)
|
|
$
|
235,188
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Aurora Holdings Members’ equity
|
|
|
227,313
|
|
|
|
|
|
|
Stockholders’ Equity:
|
|
|
|
|
|
|
|
|
|
Class A common stock, $0.01 par value per
share; shares
authorized
(actual), shares
issued and outstanding (actual);
and shares
authorized (pro forma as
adjusted), shares
issued and outstanding (pro forma as adjusted)
|
|
|
—
|
|
|
|
|
|
|
Class B common stock, $0.01 par value per
share; shares
authorized
(actual), shares
issued
and shares
outstanding
(actual); shares
authorized (pro forma as
adjusted), shares
issued
and shares
outstanding (pro forma as adjusted)
|
|
|
—
|
|
|
|
|
|
|
Additional paid-in capital
|
|
|
—
|
|
|
|
|
|
|
Accumulated other comprehensive (loss)
|
|
|
—
|
|
|
|
|
|
|
Retained earnings
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total stockholders’ equity of Aurora Diagnostics, Inc.
|
|
|
—
|
|
|
|
|
|
|
Noncontrolling interest
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total stockholders’ equity
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total capitalization
|
|
$
|
462,501
|
|
|
$
|
|
|
|
|
|
|
|
|
|
|
|
|
45
|
|
|
|
(1)
|
|
The actual capitalization
represents Aurora Holdings’ capitalization before giving
effect to the Reorganization Transactions, the refinancing of
our pre-existing credit facilities or the completion of this
offering.
|
|
|
|
|
(2)
|
|
Actual as of March 31, 2010,
includes the current and long term portions of our pre-existing
credit facilities, net of original issue discount, and the fair
value of contingent consideration.
|
|
|
|
|
(3)
|
|
Each $1.00 increase (decrease) in
the assumed initial public offering price of
$ per share, which is the midpoint
of the price range set forth on the cover page of this
prospectus, after deducting the underwriters’ discounts and
commissions and the estimated offering expenses, would increase
(decrease) the amount of pro forma as adjusted cash, cash
equivalents and short-term investments, additional paid-in
capital, total stockholders’ equity, total capitalization
and net proceeds that we will receive from this offering by
approximately $ million,
assuming the number of shares offered by us, as set forth on the
cover page of this prospectus, remains the same and after
deducting the estimated underwriting discounts and commissions
and estimated offering expenses that we must pay and after
giving effect to estimates of certain expenses that we expect to
be reimbursed.
|
The above share data excludes shares of our Class A common
stock reserved for issuance upon the exchange of our
Class B common stock into Class A common stock.
46
DILUTION
If you invest in our Class A common stock, your investment
will be diluted immediately to the extent of the difference
between the public offering price per share of our Class A
common stock and the pro forma net tangible book value per share
of our Class A common stock after this offering. Our pro
forma net tangible book value as of March 31, 2010 was
approximately $ million, or
$ per share of Class A common
stock. Pro forma net tangible book value per share represents
the amount of stockholders’ equity less the net book value
of intangible assets, divided by the number of shares of our
Class A common stock outstanding at that date, after giving
effect to the Reorganization Transactions described under
“Prospectus Summary — Reorganization
Transactions” and “Organizational
Structure — Reorganization Transactions.”
Net tangible book value dilution per share to new investors
represents the difference between the amount per share paid by
purchasers of shares of Class A common stock in this
offering and the pro forma net tangible book value per share of
Class A common stock immediately after the completion of
this offering. After giving effect to our sale of shares of
Class A common stock in this offering at an assumed initial
public offering price of $ per
share, which is the midpoint of the price range set forth on the
cover of this prospectus, and after deducting the underwriting
discounts and commissions and estimated offering expenses
payable by us and after giving effect to estimates of certain
expenses that we expect to be reimbursed, our pro forma as
adjusted net tangible book value as of March 31, 2010 would
have been $ million, or
$ per share. This represents an
immediate increase in net tangible book value of
$ per share to existing
stockholders and an immediate dilution in net tangible book
value of $ per share to investors
purchasing Class A common stock in this offering. Dilution
per share to new investors is determined by subtracting pro
forma net tangible book value per share after this offering from
the assumed initial public offering price per share paid by a
new investor. The following table illustrates the per share
dilution:
| |
|
|
|
Assumed initial public offering price per share
|
|
$
|
|
Pro forma net tangible book value (deficit) per share as of
March 31, 2010 before giving effect to the Tax Receivable
Agreement
|
|
|
|
Pro forma net tangible book value (deficit) per share before the
change attributable to new investors
|
|
|
|
Increase in pro forma net tangible book value per share
attributable to new investors
|
|
|
|
Pro Forma adjusted net tangible book value (deficit) per share
after this offering
|
|
|
|
Dilution per share to new investors
|
|
$
|
A $1.00 increase or decrease in the assumed initial public
offering price of $ per share,
which is the midpoint of the price range set forth on the cover
page of this prospectus, would increase or decrease our pro
forma as adjusted net tangible book value as of March 31,
2010, by approximately
$ million, the pro forma as
adjusted net tangible book value per share after this offering
by $ per share and the dilution in
pro forma as adjusted net tangible book value per share to new
investors in this offering by $
per share, assuming the number of shares offered by us, as set
forth on the cover page of this prospectus, remains the same and
after deducting the estimated underwriting discounts and
commissions and offering expenses payable by us.
The sale
of shares
of Class A common stock to be sold by the selling
stockholders in this offering will reduce the number of shares
held by our Principal Equityholders
to shares,
or percent
of the total shares outstanding, and will increase the number of
shares held by new investors participating in this offering
to shares,
or percent
of the total shares outstanding. In addition, if the
underwriters exercise their over-allotment option in full, the
number of shares held by our Principal Equityholders will be
further reduced
to shares,
or percent
of the total shares outstanding after this offering, and the
number of shares held by new investors participating in this
offering will be further increased
to shares,
or percent
of the total shares outstanding after this offering.
47
The following table summarizes, on the same pro forma basis as
of March 31, 2010, the total number of shares of
Class A common stock and Class B common stock
purchased from us, the total consideration paid to us and the
average price per share paid by the Principal Equityholders, and
by new investors purchasing shares in this offering (amounts in
thousands, except percentages and per share data), after giving
effect to the sale by us and the selling stockholders
of shares
of our Class A common stock in this offering at an assumed
initial public offering price of $
per share, which is the midpoint of the price range set forth on
the cover page of this prospectus, before deducting the
estimated underwriting discounts and commissions and offering
expenses payable by us:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Shares of Class A
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Common Stock and
|
|
|
Total Consideration
|
|
|
|
|
|
|
|
Class B Common Stock Purchased
|
|
|
to Us
|
|
|
Average Price
|
|
|
|
|
Number
|
|
|
Percent
|
|
|
Amount
|
|
|
Percent
|
|
|
per Share
|
|
|
|
|
Principal Equityholders
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
New investors
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
48
UNAUDITED
PRO FORMA FINANCIAL INFORMATION
The unaudited pro forma consolidated balance sheet at
March 31, 2010 and the unaudited pro forma consolidated
statement of operations for the twelve months ended
December 31, 2009 and the three months ended March 31,
2010 give effect to:
|
|
|
| |
•
|
our 2009 acquisition of South Texas Dermatopathology Lab, P.A.,
as if that acquisition had occurred January 1, 2009;
|
|
|
|
| |
•
|
our 2010 acquisitions of Bernhardt Laboratories, Inc., Pinkus
Dermatopathology Laboratory, P.C. and Pathology Solutions,
LLC, as if those acquisitions had occurred as of January 1,
2009;
|
|
|
|
| |
•
|
the execution of our new credit facilities;
|
|
|
|
| |
•
|
consummation of the Reorganization Transactions as if they were
effective as of March 31, 2010; and
|
|
|
|
| |
•
|
this offering and the use of the net proceeds that we will
receive from this offering, as if effective on March 31,
2010 for the unaudited pro forma consolidated balance sheet and
January 1, 2009 for the unaudited pro forma consolidated
statements of operations.
|
The unaudited pro forma financial information has been prepared
by our management and is based on our historical financial
statements and the assumptions and adjustments described herein
and in the notes to the unaudited pro forma financial
information below. We believe the presentation of the unaudited
pro forma financial information is prepared in conformity with
Article 11 of
Regulation S-X
of the Exchange Act.
Our historical financial information for the year ended
December 31, 2009 has been derived from our audited
consolidated financial statements and accompanying notes
included elsewhere in this prospectus. The historical financial
data for the three months ended March 31, 2010 and balance
sheet as of March 31, 2010 have been derived from our
unaudited historical condensed consolidated financial statements
included elsewhere in this prospectus.
We based the pro forma adjustments on available information and
on assumptions that we believe are reasonable under the
circumstances. See the notes to unaudited pro forma financial
information for a discussion of assumptions made. The unaudited
pro forma financial information is presented for informational
purposes and is based on management’s preliminary
estimates, including the preliminary application of our
acquisition accounting. Our final estimates and related
accounting may differ materially from the preliminary estimates.
The unaudited pro forma consolidated statements of operations do
not purport to represent what our results of operations actually
would have been if the transactions set forth above had occurred
on the dates indicated or what our results of operations will be
for future periods.
49
Aurora
Diagnostics Inc.
Unaudited
Pro Forma Consolidated Balance Sheet
March 31,
2010
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
March 31, 2010
|
|
|
|
|
|
|
|
Refinancing
|
|
|
Reorganization
|
|
|
Offering
|
|
|
Pro Forma,
|
|
|
|
|
Historical(1)
|
|
|
Adjustments
|
|
|
Adjustments(6)
|
|
|
Adjustments
|
|
|
As Adjusted
|
|
|
|
|
(in thousands)
|
|
|
|
|
Assets
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Current Assets
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash
|
|
$
|
3,222
|
|
|
$
|
226,625
|
(2)
|
|
$
|
|
|
|
$
|
—(8
|
)
|
|
$
|
|
|
|
|
|
|
|
|
|
|
(227,549
|
)(2)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts receivable, net
|
|
|
21,325
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
Prepaid expenses and other assets
|
|
|
2,877
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
Prepaid income taxes
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
Deferred tax assets
|
|
|
2,220
|
|
|
|
—
|
|
|
|
|
(3)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current assets
|
|
|
29,644
|
|
|
|
(924
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Property and Equipment, net
|
|
|
8,868
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other Assets:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Deferred debt issue costs, net
|
|
|
3,640
|
|
|
|
7,971
|
(2)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(3,640
|
)(2)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Deposits and other noncurrent assets
|
|
|
131
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
Goodwill
|
|
|
328,442
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
Intangible assets, net
|
|
|
120,274
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
452,487
|
|
|
|
4,331
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
490,999
|
|
|
$
|
3,407
|
|
|
$
|
|
|
|
$
|
—
|
|
|
$
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Liabilities and Members’ Equity
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Current Liabilities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Current portion of long-term debt
|
|
$
|
11,597
|
|
|
$
|
5,000
|
(2)
|
|
$
|
—
|
|
|
$
|
|
|
|
$
|
|
|
|
|
|
|
|
|
|
|
2,250
|
(2)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(8,691
|
)(2)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Current portion of fair value of contingent consideration
|
|
|
6,227
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
Accounts payable and accrued expenses
|
|
|
6,577
|
|
|
|
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accrued compensation
|
|
|
7,264
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
Accrued interest and other current liabilities
|
|
|
2,798
|
|
|
|
(58
|
)(2)
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1,824
|
)(2)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current liabilities
|
|
|
34,463
|
|
|
|
(3,323
|
)
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
Tax receivable arrangement
|
|
|
—
|
|
|
|
—
|
|
|
|
|
(3)
|
|
|
|
|
|
|
|
|
|
Deferred tax liabilities, net
|
|
|
11,859
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
Long-term debt, net of current portion
|
|
|
202,925
|
|
|
|
222,750
|
(2)
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(3,375
|
)(2)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(198,209
|
)(2)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1,018
|
(2)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Fair value of contingent consideration, net of current portion
|
|
|
14,439
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
Commitments and contingencies
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Equity
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Members’ equity
|
|
|
227,313
|
|
|
|
(2,296
|
)(2)
|
|
|
(4
|
)
|
|
|
(8
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
(8,500
|
)(2)
|
|
|
(7
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1,018
|
)(2)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(3,640
|
)(2)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Common stockholders’ equity
|
|
|
—
|
|
|
|
|
|
|
|
|
(3)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(4)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Additional paid in capital
|
|
|
|
|
|
|
|
|
|
|
|
(7)
|
|
|
|
|
|
|
|
|
|
Noncontrolling interest
|
|
|
—
|
|
|
|
—
|
|
|
|
|
(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
490,999
|
|
|
$
|
3,407
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
50
|
|
|
|
(1)
|
|
Amounts represent our historical
balance sheet as of March 31, 2010 which was derived from
the unaudited condensed consolidated financial statements
contained elsewhere in the prospectus.
|
|
|
|
|
(2)
|
|
These pro forma adjustments reflect
the application of the net proceeds from our recently completed
refinancing in accordance with the sources and uses below
(dollars in thousands).
|
| |
|
|
|
|
|
Sources
|
|
|
|
|
Cash
|
|
$
|
924
|
|
|
Revolver
|
|
|
5,000
|
|
|
New credit facilities
|
|
|
|
|
|
Term loan, long term portion
|
|
|
222,750
|
|
|
Term loan, current portion
|
|
|
2,250
|
|
|
Original issue discount - term loan (1.5%)
|
|
|
(3,375
|
)
|
|
|
|
|
|
|
|
Total Proceeds
|
|
$
|
227,549
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
Uses
|
|
|
|
|
Repayment of existing term loan, long term portion
|
|
$
|
198,209
|
|
|
Repayment of existing term loan, current portion
|
|
|
8,691
|
|
|
Repayment of accrued interest
|
|
|
1,824
|
|
|
Repayment of Aurora Holdings Class Z capital
|
|
|
8,558
|
|
|
Estimated fees and expenses
|
|
|
10,267
|
|
|
|
|
|
|
|
|
Total Outflows
|
|
$
|
227,549
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The repayment of Class Z capital
includes $58,000 of accrued dividends at a rate of 12 percent as
of March 31, 2010.
|
|
|
|
|
|
|
Our approximate fees and expenses
are summarized as follows:
|
| |
|
|
|
|
|
Commitment fees
|
|
$
|
6,625
|
|
|
Expenses (legal, accounting, etc)
|
|
|
1,346
|
|
|
|
|
|
|
|
|
Total deferred debt issue costs, capitalized
|
|
|
7,971
|
|
|
Prepayment penalty
|
|
|
2,296
|
|
|
|
|
|
|
|
|
Total approximate fees and expenses
|
|
$
|
10,267
|
|
|
|
|
|
|
|
|
|
|
|
|
|
In addition to the prepayment
penalty, as of March 31, 2010 in connection with our recently
completed refinancing we would have written off previously
deferred debt issues costs of $3.6 million and unamortized
original issue discount of $1.1 million.
|
|
|
|
|
(3)
|
|
A number of acquisitions by Aurora
Holdings have resulted in an increase in the tax basis of
intangible assets, primarily goodwill, which results in higher
tax amortization expense compared to book amortization. In
addition, the Reorganization Transactions and future exchanges
of Aurora Holdings Units for our Class A common stock or
cash will result in an increase in our tax basis of intangible
assets. These tax attributes would not have been available to us
in the absence of those transactions. Amortization from the
increase in tax basis will be available, subject to limitations,
to reduce the amount of tax we may be required to pay in the
future. Under the Tax Receivable Agreement, Aurora Diagnostics,
Inc. will agree to pay to the Tax Receivable Entity
85 percent of certain cash tax savings, if any, in U.S.
federal, state, local and foreign income tax realized by Aurora
Diagnostics, Inc. after the completion of this offering as a
result of:
|
|
|
|
| |
•
|
favorable tax attributes associated with amortizable goodwill
and other intangibles held by Aurora Holdings and created by
previous acquisitions;
|
| |
| |
•
|
any step-up
in tax basis in our share of Aurora Holdings’ assets
resulting from:
|
|
|
|
| |
•
|
the acquisition by us of Aurora Holdings Units from the Aurora
Holdings Continuing Members in exchange for shares of our
Class A common stock or cash, or
|
| |
| |
•
|
payments under the Tax Receivable Agreement to the Tax
Receivable Entity; and
|
|
|
|
| |
•
|
tax benefits related to imputed interest deemed to be paid by us
as a result of the Tax Receivable Agreement.
|
|
|
|
|
|
|
On a pro forma basis we estimated
the total realizable tax benefit, excluding future exchanges, as
the result of existing step up in basis to
be million. Therefore we have
recorded a deferred tax asset
of million and a liability
related to the Tax Receivable Agreement
of million representing our
obligation to the Tax Receivable Entity. The remaining
15 percent, or million,
has been recorded as an increase of additional paid-in capital,
a component of common stockholders’ equity.
|
| |
|
|
|
In consideration of the
Reorganization Transactions, the Tax Receivable Agreement
further provides that Aurora Diagnostics, Inc. will pay to the
Tax Receivable Entity 85 percent of Aurora Diagnostics,
Inc.’s actual reduction in income taxes that we realize
related to future exchanges for which we have not included a pro
forma adjustment as we cannot predict the amounts and timing of
such future exchanges, or if these exchanges will occur. In
addition, future exchanges are not a part of the Reorganization
Transactions or this offering.
|
51
|
|
|
|
(4)
|
|
Reflects the reclassification of
the remaining pro forma Aurora Holdings members’ equity to
our common stockholders’ equity.
|
|
|
|
|
(5)
|
|
Following the Reorganization
Transactions and the completion of this offering, we will record
a noncontrolling interest relating to the economic interests of
the Aurora Holdings Continuing Members in Aurora Holdings. This
adjustment reflects our pro forma equity in the economic
interest after giving effect to the noncontrolling interest of
our Principal Equityholders. As discussed under “Prospectus
Summary— Reorganization Transactions” and
“Organizational Structure — Reorganization
Transactions,” we will have all of the business and
operational control of Aurora Holdings.
|
|
|
|
|
(6)
|
|
For purposes of supplemental
disclosure, if 100 percent of the noncontrolling interest
of Aurora Holdings had been exchanged in connection with this
offering, the Tax Receivable Agreement would have increased
$ million for a total of
$ .
|
|
|
|
|
(7)
|
|
Represents the transfer of Aurora
Diagnostics Holdings, LLC retained earnings to additional paid
in capital for Aurora Diagnostics, Inc.
|
|
|
|
|
(8)
|
|
Reflects the application of the
primary proceeds of $ million from this
offering net of estimated fees and expenses of
$ million. The resulting net cash will be used
as described under the heading “Use of Proceeds.”
|
52
Aurora
Diagnostics Inc.
Unaudited
Pro Forma Consolidated Statement of Operations
Year
ended December 31, 2009
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31, 2009
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Pro
|
|
|
Pro
|
|
|
|
|
|
|
|
|
|
|
|
Pro
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Forma
|
|
|
Forma
|
|
|
|
|
|
|
|
|
|
|
|
Forma,
|
|
|
|
|
|
|
|
2009
|
|
|
2010
|
|
|
Acquisition
|
|
|
for
|
|
|
Refinancing
|
|
|
Reorganization
|
|
|
Offering
|
|
|
As
|
|
|
|
|
Historical(1)
|
|
|
Acquisitions(2)
|
|
|
Acquisitions(3)
|
|
|
Adjustments(2)(3)
|
|
|
Acquisitions
|
|
|
Adjustments
|
|
|
Adjustments
|
|
|
Adjustments
|
|
|
Adjusted
|
|
|
|
|
(in thousands)
|
|
|
|
|
Net Revenues
|
|
$
|
171,565
|
|
|
$
|
7,328
|
|
|
$
|
34,750
|
|
|
$
|
—
|
|
|
$
|
213,643
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
213,643
|
|
|
Operating costs and expenses:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost of services
|
|
|
71,778
|
|
|
|
5,200
|
|
|
|
18,956
|
|
|
|
(5,839
|
)(4)
|
|
|
90,095
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
90,095
|
|
|
Selling, general and administrative expenses
|
|
|
36,854
|
|
|
|
1,482
|
|
|
|
7,173
|
|
|
|
(1,966
|
)(4)
|
|
|
43,543
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
43,543
|
|
|
Provision for doubtful accounts
|
|
|
9,488
|
|
|
|
—
|
|
|
|
732
|
|
|
|
—
|
|
|
|
10,220
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
10,220
|
|
|
Intangible asset amortization expense
|
|
|
14,574
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,444
|
(5)
|
|
|
16,018
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
16,018
|
|
|
Management fees
|
|
|
1,778
|
|
|
|
—
|
|
|
|
—
|
|
|
|
422
|
(6)
|
|
|
2,200
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(2,200
|
)(10)
|
|
|
—
|
|
|
Impairment of goodwill and other intangible assets
|
|
|
8,031
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
8,031
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
8,031
|
|
|
Acquisition and business development costs
|
|
|
1,074
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,074
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,074
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total operating costs and expenses
|
|
|
143,577
|
|
|
|
6,682
|
|
|
|
26,861
|
|
|
|
(5,939
|
)
|
|
|
171,181
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(2,200
|
)
|
|
|
168,981
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income from operations
|
|
|
27,988
|
|
|
|
646
|
|
|
|
7,889
|
|
|
|
5,939
|
|
|
|
42,462
|
|
|
|
—
|
|
|
|
—
|
|
|
|
2,200
|
|
|
|
44,662
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other income (expense):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest expense
|
|
|
(18,969
|
)
|
|
|
(52
|
)
|
|
|
29
|
|
|
|
—
|
|
|
|
(18,992
|
)
|
|
|
1,982
|
(8)
|
|
|
—
|
|
|
|
—
|
|
|
|
(17,010
|
)
|
|
Write-off of deferred debt issue costs
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Other income
|
|
|
28
|
|
|
|
—
|
|
|
|
328
|
|
|
|
—
|
|
|
|
356
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
356
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total other expense, net
|
|
|
(18,941
|
)
|
|
|
(52
|
)
|
|
|
357
|
|
|
|
—
|
|
|
|
(18,636
|
)
|
|
|
1,982
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(16,654
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income before income taxes
|
|
|
9,047
|
|
|
|
594
|
|
|
|
8,246
|
|
|
|
5,939
|
|
|
|
23,826
|
|
|
|
1,982
|
|
|
|
—
|
|
|
|
2,200
|
|
|
|
28,008
|
|
|
Provision for income taxes
|
|
|
45
|
|
|
|
215
|
|
|
|
434
|
|
|
|
2,376
|
(7)
|
|
|
3,069
|
|
|
|
793
|
(7)
|
|
|
6,461
|
(9)
|
|
|
880
|
(7)
|
|
|
11,203
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income
|
|
$
|
9,002
|
|
|
$
|
379
|
|
|
$
|
7,812
|
|
|
$
|
3,563
|
|
|
$
|
20,757
|
|
|
$
|
1,189
|
|
|
$
|
(6,461
|
)
|
|
$
|
1,320
|
|
|
|
16,805
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income available to non-controlling interest
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income available to common stockholders
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income available to common stockholders per common share:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic net income per common share
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Diluted net income per common share
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
53
Aurora
Diagnostics Holdings, Inc.
Unaudited
Pro Forma Consolidated Statement of Operations
For the
Three Months Ended March 31, 2010
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
March 31, 2010
|
|
|
|
|
|
|
|
|
|
|
Pro
|
|
|
Pro
|
|
|
|
|
|
|
|
|
|
|
|
Pro
|
|
|
|
|
|
|
|
|
|
|
Forma
|
|
|
Forma
|
|
|
|
|
|
|
|
|
|
|
|
Forma,
|
|
|
|
|
|
|
|
2010
|
|
|
Acquisition
|
|
|
for
|
|
|
Refinancing
|
|
|
Reorganization
|
|
|
Offering
|
|
|
As
|
|
|
|
|
Historical(1)
|
|
|
Acquisitions(3)
|
|
|
Adjustments
|
|
|
Acquisitions
|
|
|
Adjustments
|
|
|
Adjustments
|
|
|
Adjustments
|
|
|
Adjusted
|
|
|
|
|
(in thousands)
|
|
|
|
|
Net Revenues
|
|
$
|
46,419
|
|
|
$
|
3,198
|
|
|
$
|
—
|
|
|
$
|
49,617
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
49,617
|
|
|
Operating costs and expenses:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost of services
|
|
|
22,342
|
|
|
|
1,230
|
|
|
|
136
|
(4)
|
|
|
23,708
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
23,708
|
|
|
Selling, general and administrative expenses
|
|
|
10,820
|
|
|
|
299
|
|
|
|
—
|
|
|
|
11,119
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
11,119
|
|
|
Provision for doubtful accounts
|
|
|
2,610
|
|
|
|
—
|
|
|
|
—
|
|
|
|
2,610
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
2,610
|
|
|
Intangible asset amortization expense
|
|
|
3,891
|
|
|
|
—
|
|
|
|
110
|
(5)
|
|
|
4,001
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
4,001
|
|
|
Management fees
|
|
|
476
|
|
|
|
—
|
|
|
|
32
|
(6)
|
|
|
508
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(508
|
)(10)
|
|
|
—
|
|
|
Impairment of goodwill and other intangible assets
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Acquisition and business development costs
|
|
|
298
|
|
|
|
—
|
|
|
|
—
|
|
|
|
298
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
298
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total operating costs and expenses
|
|
|
40,437
|
|
|
|
1,529
|
|
|
|
278
|
|
|
|
42,244
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(508
|
)
|
|
|
41,736
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income from operations
|
|
|
5,982
|
|
|
|
1,669
|
|
|
|
(278
|
)
|
|
|
7,373
|
|
|
|
—
|
|
|
|
—
|
|
|
|
508
|
|
|
|
7,881
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other income (expense):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest expense
|
|
|
(3,721
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
(3,721
|
)
|
|
|
(508
|
)(8)
|
|
|
—
|
|
|
|
—
|
|
|
|
(4,229
|
)
|
|
Write-off of deferred debt issue costs
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Other income
|
|
|
(12
|
)
|
|
|
12
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total other expense, net
|
|
|
(3,733
|
)
|
|
|
12
|
|
|
|
—
|
|
|
|
(3,721
|
)
|
|
|
(508
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income before income taxes
|
|
|
2,249
|
|
|
|
1,681
|
|
|
|
(278
|
)
|
|
|
3,652
|
|
|
|
(508
|
)
|
|
|
—
|
|
|
|
508
|
|
|
|
3,652
|
|
|
Provision (benefit) for income taxes
|
|
|
10
|
|
|
|
—
|
|
|
|
(111
|
)(7)
|
|
|
(101
|
)
|
|
|
(203
|
)(7)
|
|
|
1,592
|
(9)
|
|
|
203
|
(7)
|
|
|
1,461
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income
|
|
$
|
2,239
|
|
|
$
|
1,681
|
|
|
$
|
(167
|
)
|
|
$
|
3,753
|
|
|
$
|
(305
|
)
|
|
$
|
(1,592
|
)
|
|
$
|
305
|
|
|
|
2,191
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income available to non-controlling interest
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income available to common stockholders
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income available to common stockholders per common share:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic net income per common share
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Diluted net income per common share
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1)
|
|
Amounts represent our historical
statements of operations for the year ended December 31,
2009 (audited) and the three months ended March 31, 2010
(unaudited) which were derived from the financial statements of
Aurora Holdings contained elsewhere in the prospectus.
|
|
|
|
|
(2)
|
|
Amounts represent the historical
audited statement of operations of South Texas Dermatopathology
Lab, P.A. prior to our acquisition on November 21, 2009,
which we refer to as the 2009 Acquisition.
|
|
|
|
|
(3)
|
|
For the year ended
December 31, 2009, amounts represent the historical
unaudited statements of operations of the 2010 Acquisitions
which were funded on December 31, 2009 and were consummated
January 1, 2010 and the historical audited statement of
operations of our acquisition of Pathology Solutions, LLC
completed March 12, 2010. For the three months ended
March 31, 2010, the amounts represent the historical
unaudited statement of operations, prior to our acquisition on
March 12, 2010, of Pathology Solutions, LLC.
|
54
|
|
|
|
(4)
|
|
The pro forma adjustment reflects
the reduction in compensation expense of physicians and former
owners of acquired practices, including salary, bonus and other
compensation, to the amounts that will be paid to these
physicians and former owners in accordance with their post
acquisition employment agreements.
|
|
|
|
|
(5)
|
|
Represents the additional
amortization expense for the identifiable intangible assets,
based on our preliminary acquisition accounting, as if the 2009
and 2010 Acquisitions had occurred on the January 1, 2009.
The identifiable intangible assets related to the 2009 and 2010
Acquisitions total approximately $73.8 million and are
being amortized over periods ranging from 5 to 10 years.
The majority of the identifiable intangible assets relate to
customer relationships. In determining the estimated
amortization periods, we considered the operating history and
customer stability of the acquired practice and industry
information related to customary amortization periods.
|
|
|
|
|
(6)
|
|
Reflects the management fees
payable under a management services agreement with certain of
our Principal Equityholders. In accordance with the management
services agreement, these fees are calculated as 1 percent
of the net revenue of our 2009 and 2010 acquisitions.
|
|
|
|
|
(7)
|
|
Represents the tax effect of the
pro forma adjustments at an effective tax rate of
40 percent and was based on the federal and state statutory
income tax rates in effect during the respective periods.
|
|
|
|
|
(8)
|
|
Represents the difference in
interest expense related to the rates and balances outstanding
under the new credit facilities compared to our term loans
outstanding during the year ended December 31, 2009 and the
three months ended March 31, 2010. Under the new credit
facilities the base interest rate is subject to a LIBOR (London
Interbank Offering Rate) of 2.0 percent. Therefore, we assumed
the London Interbank Offering Rate, or LIBOR, floor of
2 percent plus the credit spread of 4.25 percent or a
total interest rate of 6.25 percent on a combined
$230 million term loan and revolver balance. The
adjustments also reflect the difference in the amortization of
the deferred debt issue cost and original issue discount. The
following summarizes the components of the interest expense
adjustments.
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months
|
|
|
|
|
Year Ended
|
|
|
Ended
|
|
|
|
|
December 31,
|
|
|
March 31,
|
|
|
|
|
2009
|
|
|
2010
|
|
|
|
|
Elimination of amortization of original issue discount
|
|
$
|
305
|
|
|
$
|
81
|
|
|
Amortization of new original issue discount
|
|
|
(563
|
)
|
|
|
(141
|
)
|
|
Elimination of amortization of deferred debt issue costs
|
|
|
1,090
|
|
|
|
291
|
|
|
Amortization of new debt issue costs
|
|
|
(1,329
|
)
|
|
|
(332
|
)
|
|
Elimination of term loan interest expense
|
|
|
16,853
|
|
|
|
3,187
|
|
|
Interest expense related to the new credit facilities
|
|
|
(14,375
|
)
|
|
|
(3,594
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Net reduction (increase) in interest expense
|
|
$
|
1,982
|
|
|
|
(508
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Our new credit facility is subject
to a LIBOR floor of 2.0 percent. At the time LIBOR rates
exceed the 2.0 percent floor, every .125 percent
increase in LIBOR would increase our interest expense
approximately $0.3 million.
|
|
|
|
|
(9)
|
|
Reflects the necessary adjustment
to record our income tax provision at a 40 percent rate due
to our transition to being taxed as a corporation following
completion of the Reorganization Transactions. The
40 percent rate was based on the federal and state
statutory income tax rates in effect during the respective
periods.
|
|
|
|
|
(10)
|
|
Represents the elimination of the
management fees payable under a management services agreement
with certain of our Principal Equityholders. The management
services agreement will be terminated following the completion
of this offering.
|
55
SELECTED
HISTORICAL CONSOLIDATED FINANCIAL DATA
The following selected historical consolidated financial data
should be read in conjunction with the section titled
“Management’s Discussion and Analysis of Financial
Condition and Results of Operations” and the consolidated
financial statements and the related notes included in this
prospectus. The selected historical consolidated financial data
included in this section are not intended to replace the
consolidated financial statements and the related notes included
in this prospectus.
The consolidated statements of operations data for the fiscal
years 2007, 2008 and 2009, and consolidated balance sheets data
as of fiscal year end 2008 and 2009, were derived from Aurora
Holdings’ audited consolidated financial statements that
are included elsewhere in this prospectus. The consolidated
statements of operations data for the period from our inception
in June 2006 through December 31, 2006, and consolidated
balance sheet data as of December 31, 2006 and
December 31, 2007, were derived from Aurora Holdings’
audited consolidated financial statements not included in this
prospectus. The consolidated statements of operations data for
the three months ended March 31, 2009 and 2010 and
consolidated balance sheet data as of March 31, 2010 were
derived from Aurora Holdings’ unaudited condensed
consolidated financial statements included elsewhere in this
prospectus and include, in the opinion of management, all
adjustments, consisting only of normal recurring adjustments,
that management considers necessary for the fair presentation of
the financial information set forth in those statements. The
historical results presented below are not necessarily
indicative of financial results to be achieved in future periods.
The selected historical consolidated financial data does not
give effect to:
|
|
|
| |
•
|
the results of operations of our 2009 acquisition of South Texas
Dermatopathology Lab, P.A. prior to our acquisition on
November 21, 2009;
|
|
|
|
| |
•
|
the results of operations or balance sheet data of our 2010
acquisitions of Bernhardt Laboratories, Inc. and Pinkus
Dermatopathology Laboratory, P.C. prior to our acquisitions
on January 1, 2010 and Pathology Solutions, LLC prior to
our acquisition on March 12, 2010;
|
|
|
|
| |
•
|
the execution of our new credit facilities;
|
|
|
|
| |
•
|
consummation of the Reorganization Transactions; or
|
| |
| |
•
|
this offering and the use of the net proceeds that we will
receive from this offering.
|
56
Aurora
Diagnostics Holdings, LLC
Selected Consolidated Statements of
Operations(1)
Period from June 2006 (inception) to December 31,
2006,
Years ended December 31, 2007, 2008 and 2009 and
Three months ended March 31, 2009 and 2010
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Period from
|
|
|
|
|
|
|
|
|
|
|
June 2006
|
|
|
|
|
|
|
|
|
|
|
(Inception) to
|
|
|
|
|
|
Three Months
|
|
|
|
|
December 31,
|
|
|
Year Ended December 31,
|
|
|
Ended March 31,
|
|
|
|
|
2006
|
|
|
2007
|
|
|
2008
|
|
|
2009
|
|
|
2009
|
|
|
2010
|
|
|
|
|
(in thousands)
|
|
|
|
|
Net Revenues
|
|
$
|
3,487
|
|
|
$
|
63,451
|
|
|
$
|
157,850
|
|
|
$
|
171,565
|
|
|
$
|
40,947
|
|
|
$
|
46,419
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating costs and expenses:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost of services
|
|
|
1,045
|
|
|
|
27,480
|
|
|
|
66,382
|
|
|
|
71,778
|
|
|
|
17,186
|
|
|
|
22,342
|
|
|
Selling, general and administrative expenses
|
|
|
3,035
|
|
|
|
15,172
|
|
|
|
33,194
|
|
|
|
36,854
|
|
|
|
9,177
|
|
|
|
10,820
|
|
|
Provision for doubtful accounts
|
|
|
69
|
|
|
|
2,378
|
|
|
|
8,037
|
|
|
|
9,488
|
|
|
|
2,305
|
|
|
|
2,610
|
|
|
Intangible asset amortization expense
|
|
|
470
|
|
|
|
5,721
|
|
|
|
14,308
|
|
|
|
14,574
|
|
|
|
3,628
|
|
|
|
3,891
|
|
|
Management fees
|
|
|
35
|
|
|
|
644
|
|
|
|
1,559
|
|
|
|
1,778
|
|
|
|
410
|
|
|
|
476
|
|
|
Impairment of goodwill and other intangible assets
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
8,031
|
(3)
|
|
|
—
|
|
|
|
—
|
|
|
Acquisition and business development costs
|
|
|
—
|
|
|
|
374
|
|
|
|
676
|
|
|
|
1,074
|
|
|
|
130
|
|
|
|
298
|
|
|
Equity based compensation expense
|
|
|
—
|
|
|
|
—
|
|
|
|
1,164
|
(2)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total operating costs and expenses
|
|
|
4,654
|
|
|
|
51,769
|
|
|
|
125,320
|
|
|
|
143,577
|
|
|
|
32,836
|
|
|
|
40,437
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income (loss) from operations
|
|
|
(1,166
|
)
|
|
|
11,682
|
|
|
|
32,530
|
|
|
|
27,988
|
|
|
|
8,111
|
|
|
|
5,982
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other income (expense):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest expense
|
|
|
(94
|
)
|
|
|
(7,114
|
)
|
|
|
(21,577
|
)
|
|
|
(18,969
|
)
|
|
|
(4,791
|
)
|
|
|
(3,721
|
)
|
|
Write-off of deferred debt issue costs
|
|
|
—
|
|
|
|
(3,451
|
)(4)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Other income
|
|
|
25
|
|
|
|
124
|
|
|
|
125
|
|
|
|
28
|
|
|
|
29
|
|
|
|
(12
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total other expense, net
|
|
|
(69
|
)
|
|
|
(10,441
|
)
|
|
|
(21,452
|
)
|
|
|
(18,941
|
)
|
|
|
(4,762
|
)
|
|
|
(3,773
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income (loss) before income taxes
|
|
|
(1,236
|
)
|
|
|
1,241
|
|
|
|
11,078
|
|
|
|
9,047
|
|
|
|
3,349
|
|
|
|
2,249
|
|
|
Provision for income
taxes(5)
|
|
|
—
|
|
|
|
762
|
|
|
|
408
|
|
|
|
45
|
|
|
|
17
|
|
|
|
10
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income (loss)
|
|
$
|
(1,236
|
)
|
|
$
|
479
|
|
|
$
|
10,670
|
|
|
$
|
9,002
|
|
|
$
|
3,332
|
|
|
$
|
2,239
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
57
Aurora
Diagnostics Holdings, LLC
Selected
Consolidated Balance Sheet Data
December 31,
2006, 2007, 2008 and 2009 and March 31, 2010
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31,
|
|
|
March 31,
|
|
|
|
|
2006
|
|
|
2007
|
|
|
2008
|
|
|
2009
|
|
|
2010
|
|
|
|
|
|
|
|
(in thousands)
|
|
|
|
|
|
|
|
|
|
|
Consolidated Balance Sheet Data
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and equivalents
|
|
$
|
1,744
|
|
|
$
|
8,558
|
|
|
$
|
7,278
|
|
|
$
|
27,424
|
|
|
$
|
3,222
|
|
|
Total assets
|
|
|
40,180
|
|
|
|
388,339
|
|
|
|
415,516
|
|
|
|
462,744
|
|
|
|
490,999
|
|
|
Working capital, excluding deferred tax items
|
|
|
1,892
|
|
|
|
10,161
|
|
|
|
11,005
|
|
|
|
30,081
|
|
|
|
10,785
|
|
|
Long term debt, including current portion
|
|
|
7,550
|
|
|
|
215,678
|
|
|
|
227,313
|
|
|
|
219,752
|
|
|
|
235,188
|
|
|
Members’ equity
|
|
|
31,334
|
|
|
|
145,077
|
|
|
|
161,176
|
|
|
|
217,064
|
|
|
|
227,313
|
|
|
|
|
|
(1)
|
|
The summary consolidated financial
data for the period from June 2006 (inception) to
December 31, 2006 and the years ended December 31,
2007, 2008 and 2009 and for the three months ended
March 31, 2009 and 2010 is that of Aurora Holdings prior to
the Reorganization Transactions described under “Prospectus
Summary — Reorganization Transactions” and
“Organizational Structure — Reorganization
Transactions.”
|
|
|
|
|
(2)
|
|
During 2008, we adopted the New
Plan to replace our original equity incentive plan. This New
Plan provides awards of membership interest units in Aurora
Holdings. These interests are denominated as
Class D-1,
Class D-2,
and
Class D-3 units
in Aurora Holdings. During 2008, Aurora Holdings authorized and
issued 4,000 D-1 units, 3,000 D-2 units and 3,000
D-3 units of Aurora Holdings. All membership interest units
of Aurora Holdings issued in 2008 were fully vested as of
December 31, 2008. We recorded a compensation expense of
$1.2 million for these awards. There were no other grants
under the New Plan. In connection with the Reorganization
Transactions, the Class D Units of Aurora Holdings issued under
the New Plan will either be exchanged for shares of our
Class A common stock or cancelled without consideration.
|
|
(3)
|
|
As of September 30, 2009, we
tested goodwill and intangible assets for potential impairment
and recorded a non-cash impairment expense of $8.0 million
resulting from a write-down of $6.6 million in the carrying
value of goodwill and a write down of $1.4 million in the
carrying value of other intangible assets. The write-down of the
goodwill and other intangible assets related to one reporting
unit. Regarding this reporting unit, we believe events occurred
and circumstances changed that more likely than not reduced the
fair value of the intangible assets and goodwill below their
carrying amounts. These events during 2009 consisted primarily
of the loss of significant customers present at the acquisition
date, which adversely affected the current year and expected
future revenues and operating profit of the reporting unit.
|
|
(4)
|
|
In December 2007, we refinanced our
previous credit facilities. As a result, we wrote off
$3.5 million of unamortized deferred debt issue costs.
|
|
(5)
|
|
Aurora Holdings is a Delaware
limited liability company taxed as a partnership for federal and
state income tax purposes, in accordance with the applicable
provisions of the Internal Revenue Code. Accordingly, Aurora
Holdings was generally not subject to income taxes. The income
attributable to Aurora Holdings was allocated to the members of
Aurora Holdings in accordance with the terms of the Aurora
Holdings LLC Agreement. However, certain of our subsidiaries are
structured as corporations, file separate returns and are
subject to federal and state income taxes. The historical
provision for income taxes for these subsidiaries is reflected
in our consolidated financial statements and includes federal
and state taxes currently payable and changes in deferred tax
assets and liabilities excluding the establishment of deferred
tax assets and liabilities related to the acquisitions. The pro
forma, as adjusted, provision for income taxes assumes a
40 percent effective tax rate, after giving effect to the
Reorganization Transactions.
|
58
MANAGEMENT’S
DISCUSSION AND ANALYSIS OF
FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The historical consolidated financial data discussed below
reflect the historical results of operations and financial
condition of our subsidiary Aurora Holdings.
You should read the following discussion and analysis of our
financial condition and results of operations together with our
financial statements and related notes appearing in the back of
this prospectus. Some of the information contained in this
discussion and analysis or set forth elsewhere in this
prospectus, including information with respect to plans and
strategy for our business and related financing, includes
forward-looking statements that involve risks and uncertainties.
You should review the section entitled “Risk Factors”
contained in this prospectus for a discussion of important
factors that could cause actual results to differ materially
from the results described in or implied by the forward-looking
statements contained in the following discussion and
analysis.
General
We are a specialized diagnostics company providing services that
play a key role in the diagnosis of cancer and other diseases.
Our experienced pathologists deliver comprehensive diagnostic
reports of a patient’s condition and consult frequently
with referring physicians to help determine the appropriate
treatment. Our diagnostic reports often enable the early
detection of disease, allowing referring physicians to make
informed and timely treatment decisions that improve their
patients’ health in a cost-effective manner. Through our
pathologist-operated laboratory practices, we provide
physician-based general anatomic and clinical pathology,
dermatopathology, molecular diagnostic services and other
esoteric testing services to physicians, hospitals, clinical
laboratories and surgery centers. Our operations consist of one
reportable segment.
The U.S. diagnostic testing industry had revenues of
approximately $55 billion in 2008 and, according to the
Washington G-2 Report, grew at a rate of 7 percent
compounded annually from 2000 to 2008. According to the
Laboratory Economics Report, within the overall industry, the
anatomic pathology market totaled approximately $13 billion
in revenues, or 24 percent of total industry revenues, in
2008. Anatomic pathology services involve the diagnosis of
cancer and other medical conditions through the examination of
tissues (histology) and the analysis of cells (cytology) and
generally command higher reimbursement rates, on a per specimen
basis, than clinical pathology services.
According to the Washington G-2 Report, the anatomic pathology
market has expanded more rapidly than the overall industry, with
revenues growing 4.8 percent on a compound annual basis
between 2006 and 2009, compared to 4.5 percent for the rest
of the industry. Excluding growth in esoteric testing, the
remainder of the industry grew at a compound annual rate of only
0.1 percent over the same period. Substantially all of the
revenues for anatomic pathology businesses consist of payments
or reimbursements for specialized diagnostic services rendered
to referring physicians, and these revenues are affected
primarily by changes in case volume, which we refer to as
accession volume, payor mix and reimbursement rates. Accessions
are measured as the number of patient cases, and each accession
may include multiple specimens. Accession volume varies from
period to period based on the number of referring physicians and
the frequency of their ordering, the relative mix of the
referring physicians’ anatomic pathology specialties, and
the type and number of tests ordered.
The non-hospital outpatient channel is the largest component of
the anatomic pathology market and has grown more rapidly than
other channels. This channel accounted for $7.6 billion, or
57 percent, of anatomic pathology revenues for the year
ended December 31, 2008, representing 10 percent
growth in 2008 according to the Laboratory Economics Report. The
remainder of the anatomic pathology market is comprised of the
hospital inpatient channel, which accounted for
$3.7 billion or 28 percent, of anatomic pathology
revenues, representing 2 percent growth in 2008, and the
hospital outpatient channel, which accounted for
$1.9 billion, or 15 percent, of anatomic pathology
revenues, representing 4 percent growth in 2008, according
to the Laboratory Economics Report.
For the year ended December 31, 2009 we processed
approximately 1.6 million accessions.
59
Corporate
History
We were incorporated in the State of Delaware on April 23,
2010 for purposes of this offering. As described under
“Prospectus Summary — Reorganization
Transactions” and “Organizational Structure,” we
are a holding company and our principal asset after completion
of this offering will be our indirect equity interests in our
subsidiary, Aurora Holdings. Our subsidiary was organized in the
State of Delaware as a limited liability company on June 2,
2006 to develop and operate as a diagnostic services company. We
have grown our business significantly over the last three years,
driven largely by the acquisition of local and regional
pathology laboratories throughout the United States and organic
growth within these acquired operations. We have completed 17
acquisitions of diagnostic services companies and opened
two de novo laboratories, and our revenues have increased
from $3.5 million in 2006 to $171.6 million in 2009.
Statement
of Operations Overview
Net
Revenues
Substantially all of our revenues consist of payments or
reimbursements for specialized diagnostic services rendered to
patients of our referring physicians. Net revenue per accession
is impacted mainly by changes in reimbursement rates and test
and payor mix. Accession volume varies from period to period
based on the referral patterns of our referring physicians and
the frequency of their ordering, the relative mix of the
referring physicians’ anatomic pathology specialties, and
the type and number of tests ordered. Accession volume is also
affected by seasonal trends and generally declines during the
summer and holiday periods. Furthermore, accession volume is
also subject to declines due to weather conditions, such as
severe snow storms and flooding or excessively hot or cold
spells, which can deter patients from visiting our referring
physicians. More recently, we believe the slowdown in the
general economy and increase in unemployment has reduced the
number of patients visiting our referring physician offices,
resulting in a reduction of referrals.
Our billings for services reimbursed by third-party payors,
including Medicare, and patients are based on a
company-generated fee schedule that is generally set at higher
rates than our anticipated reimbursement rates. Our billings to
physicians, which are not reimbursed by third-party payors,
represent less than 10 percent of net revenues and are
billed based on negotiated fee schedules that set forth what we
charge for our services. Reimbursement under Medicare for
specialized diagnostic services is subject to a Medicare
physician fee schedule and, to a lesser degree, a clinical
laboratory fee schedule, both of which are updated annually. Our
billings to insured patients include co-insurance and
deductibles as dictated by the patient’s insurance
coverage. Billings for services provided to uninsured patients
are based on our company-generated fee schedule. Our revenues
are recorded net of the estimated differences between the amount
billed and the estimated payment to be received from third party
payors, including Medicare. We do not have any capitated payment
arrangements, which are arrangements under which we are paid a
contracted per person rate regardless of the services we
provide. We generally provide services on an
in-network
basis, where we perform services for persons within the networks
of payors with which we have contracts. Services performed on an
out-of-network
basis, where we perform services for persons outside of the
networks of payors with which we have contracts, comprised less
than 15 percent of our 2009 revenues. We may face
continuing pressure on reimbursement rates as government payors
and private insurers have taken steps and may continue to take
steps to control the cost, use, and delivery of health care
services, including diagnostic testing services. Changes in
payor mix could lead to corresponding changes in revenues based
on the differences in reimbursement rates.
Compliance with applicable laws and regulations, as well as
internal policies and procedures, adds further complexity and
costs to our operations. Furthermore, we are generally obligated
to bill in the specific manner prescribed by each governmental
payor and private insurer, who may each have different billing
requirements. Reimbursements for anatomic pathology services are
received from governmental payors, such as Medicare and
Medicaid; private insurance, including managed care
organizations and commercial payors; and private payors, such as
physicians and individual patients. For the year ended
December 31, 2009, we derived approximately 61 percent
of revenues from private insurance, including managed care
organizations and commercial payors; approximately
25 percent of revenues from Medicare and Medicaid; and
approximately 14 percent from physicians and individual
patients.
60
In most cases, we provide a global testing service which
includes both the technical slide preparation and professional
diagnosis. We also fulfill requests from physicians for only the
technical component of our services, or TC, which principally
includes technical slide preparation and the non-professional
items associated with our diagnostic services, including
equipment, supplies and technical personnel, or the professional
component of our services, or PC, which principally includes
review and diagnosis by a pathologist. If a physician requires
only the TC services such as slide preparation, we prepare the
slide and then return it to the referring physician for
assessment and diagnosis.
Cost
of Services
Cost of services consists of physician costs, including
compensation, benefits and medical malpractice insurance and
other physician related costs. In addition, cost of services
includes costs related to the technical preparation of specimens
and transcription of reports, depreciation, courier and
distribution costs, and all other costs required to fulfill the
diagnostic service requirements of our referring physicians and
their patients.
Cost of services generally increases with accession volume and
reflects the additional staffing, equipment, supplies and
systems needed to process the increased volume and maintain
client service levels. A major component of cost of services is
physician costs which, for the year ended December 31,
2009, represented 36 percent of our total cost of services.
In the future, we may experience increases in physician costs to
retain existing physicians, to replace departing physicians or
to hire new pathologists to support accession growth. Therefore,
we expect our cost of revenues will continue to increase
commensurate with revenue growth.
Selling,
General and Administrative Expenses
Selling, general and administrative expenses consist primarily
of general lab and corporate overhead, billing, information
technology, accounting, human resources, and sales and marketing
expenses. We expect sales and marketing and IT expenses to
increase faster than revenue as we hire additional personnel and
invest in lab and billing information systems to support
continued same store revenue growth and retain existing customer
relationships. In addition, we expect accounting expenses, which
includes audit and Sarbanes-Oxley Act of 2002 costs, to increase
substantially as a result of our contemplated initial public
offering. As our business matures and we attain a sufficient
size and scope, we expect selling, general and administrative
expenses as a percent of revenue to reduce over time.
Provision
for Doubtful Accounts
The provision for doubtful accounts and the related allowance
are adjusted periodically, based upon an evaluation of
historical collection experience with specific payors for
particular services, anticipated collection levels with specific
payors for new services, industry reimbursement trends, accounts
receivable aging and other relevant factors. The majority of our
provision for doubtful accounts relates to our estimate of
uncollectible amounts from patients who are uninsured or fail to
pay their coinsurance or deductible obligations. Changes in
these factors in future periods could result in increases or
decreases in our provision for doubtful accounts and impact our
results of operations, financial position and cash flows.
In an effort to maximize our collections of accounts receivable,
we take a number of steps to collect amounts, including
coinsurance and deductibles, owed by third party payors,
government payors, referring physicians and patients. The
process generally includes:
|
|
|
| |
•
|
verification of complete insurance information and patient
demographics,
|
|
|
|
| |
•
|
active management and follow up on denials,
|
|
|
|
| |
•
|
delivery of scheduled statements to patients,
and/or
|
|
|
|
| |
•
|
forwarding significant past due accounts to outside collection
agencies.
|
Due to the fact that we operate on multiple billing platforms,
we evaluate collectibility and the related adequacy of our
allowance for doubtful accounts using information from multiple
sources. Not all of our systems produce the same level of
information by payor or by aging classification. However, we
believe that sufficient
61
information exists in each respective billing system to make
reasonable estimates of our provision for doubtful accounts.
Recent
Developments
Class Z
Capital
On March 12, 2010, we issued Class Z capital of Aurora
Holdings to then-existing members of Aurora Holdings for total
consideration of $8.5 million. In the event that the Class
Z capital was repaid within six months of that date, the holders
of the Class Z capital of Aurora Holdings were to receive a
preferred return equal to the initial contribution of the
members holding such membership interests, plus dividends.
Dividends were to accrue on the Class Z capital of Aurora
Holdings at an annual rate of 12 percent for the first
three months and 16 percent for the next three months. In
the event we did not redeem the Class Z capital within six
months from the date it was issued, the Class Z capital of
Aurora Holdings would have converted to
Class A-1
membership interests of Aurora Holdings at the same valuation as
the original
Class A-1
membership interests of Aurora Holdings. On May 26, 2010,
in connection with our recently completed refinancing, we
redeemed all of the outstanding Class Z membership
interests of Aurora Holdings and paid approximately
$0.2 million of accrued dividends.
New
Credit Facilities
On May 26, 2010, we entered into a new credit and guaranty
agreement, which we refer to as our credit agreement. Our credit
agreement provides for a $335.0 million credit facility
with Barclays Bank PLC, as administrative agent and collateral
agent, certain other financial institutions in various arranger
and agent capacities, and the lenders from time to time party to
the credit agreement. Our credit agreement includes a
$225.0 million senior secured term loan facility and a
$110.0 million senior secured revolving credit facility
($50.0 million of which became available upon the closing
of our new credit facility and $60.0 million of which will
be available upon the completion of an offering of equity
satisfying certain criteria under our credit agreement). Upon
completion of this offering, we expect the size of our revolving
credit facility to increase to $110.0 million. We have the
right to increase the commitments under our credit agreement by
up to $50.0 million, provided certain conditions are met.
None of the lenders under our credit agreement has committed or
is obligated to provide any such increase in the commitments.
A portion of the net proceeds of our credit agreement was used
to repay all indebtedness outstanding under our previous first
and second lien credit facilities and to terminate the
agreements associated with such previous credit facilities. We
have recorded a non-cash write-off of any remaining unamortized
original issue discount and debt issue costs related to our
previous credit facilities of approximately $4.4 million
during the quarter ended June 30, 2010, and we were
required to pay a $2.3 million prepayment premium to
terminate our previous credit facilities.
Aurora Diagnostics, LLC, a direct,
wholly-owned
subsidiary of Aurora Holdings, is the borrower under our credit
agreement. The obligations of the borrower under our credit
agreement and any exposure under any interest rate agreement or
other hedging arrangements entered into with any of the lenders
are guaranteed by the guarantors party to our credit agreement,
including Aurora Holdings, certain of its subsidiaries and, upon
completion of the Reorganization Transactions and this offering,
Aurora Diagnostics, Inc. and certain of its subsidiaries. Such
obligations of the borrower and such guarantors are secured by a
first-priority security interest in substantially all of the
assets of the borrower and of each guarantor party thereto.
The loans under our credit agreement will bear interest, at the
option of the borrower, at one of the following rates:
|
|
|
| |
•
|
Base Rate Loans, at a rate per annum equal to:
|
|
|
|
| |
•
|
Barclays Bank PLC’s prime rate,
|
|
|
|
| |
•
|
0.50% plus Federal Funds Effective Rate, and
|
|
|
|
| |
•
|
the LIBOR rate, as adjusted for applicable reserve requirements
(subject to a 2.00% floor) for an interest period of one month
plus 1.00%; plus
|
62
• LIBOR Rate Loans, at a rate per annum
equal to:
|
|
|
| |
•
|
the LIBOR rate, as adjusted for applicable reserve requirements
(subject to a 2.00% floor) determined for the applicable
interest period; plus
|
During the continuance of certain material events of default,
and at the request of the requisite lenders during the
continuance of any other event of default, the loans under our
credit agreement shall bear interest at the otherwise applicable
rate plus an additional 2.00% per annum. An unused commitment
fee equal to either 0.75% or 0.50% per annum (depending on the
amount of the unused portion of the facility at such time) of
the daily average unused portion of the revolving credit
facility is also payable.
Our term loan facility will amortize during the period the loan
is outstanding in quarterly installments that shall each be
equal to 0.25% of the initial principal amount of the term loan
with the balance payable on the maturity date which is the sixth
anniversary date of our credit agreement. The revolving credit
facility will mature, and the revolving commitments relating
thereto will terminate, on the fourth anniversary date of our
credit agreement.
Optional prepayments of borrowings under our credit agreement,
and optional reductions of the unutilized portion of the
revolving credit facility commitments, will be permitted at any
time, in minimum principal amounts, without premium or penalty,
subject to reimbursement of the lenders’ redeployment costs
in the case of a prepayment of LIBOR Rate Loans other than a
prepayment made on the last day of the relevant interest period.
Our credit agreement requires, subject to certain exceptions,
prepayments from excess cash flow and from the proceeds of
certain asset sales, insurance or condemnation and issuances of
certain debt and equity.
Our credit agreement requires us to satisfy specified financial
covenants, including an interest coverage ratio, a total
leverage ratio and a maximum capital expenditures covenant, as
set forth in our credit agreement. In addition, our credit
agreement contains certain representations and warranties and
affirmative and negative covenants which, among other things,
limit the incurrence of additional indebtedness, liens and
encumbrances, distributions, prepayments of other indebtedness,
guarantees, investments, acquisitions, asset sales, mergers and
consolidations, transactions with affiliates, certain changes to
the Reorganization Transactions, certain payments under the Tax
Receivables Agreement, changes to organizational documents and
to material agreements and certain other agreements and other
matters customarily restricted in such agreements.
Our credit agreement contains certain events of default
including payment defaults, cross defaults to certain other
indebtedness in excess of specified amounts, covenant defaults,
breaches of representations and warranties, certain events of
bankruptcy and insolvency, certain ERISA events, judgment
defaults in excess of specified amounts, failure of any guaranty
or security document supporting our credit agreement to be in
full force and effect, failure of certain indebtedness in excess
of specified amounts to be subordinated to the obligations under
our credit agreement, change in control, certain criminal
proceedings, incurrence of indebtedness by affiliated practices
in excess of specified amounts and certain other customary
events of default.
Health
Care Reform
In 2010, the U.S. Congress passed and the President signed
into law the PPACA and HCEARA. Together, the PPACA and HCEARA
comprise a broad health care reform initiative. While this
legislation did not adversely affect reimbursement for our
anatomic pathology services, this legislation provides for two
separate reductions in the reimbursement rates for our clinical
laboratory services: a “productive adjustment”
(currently estimated to be between 1.1 and 1.4 percent),
and an additional 1.75 percent reduction. Each of these
would reduce the annual Consumer Price Index-based update that
would otherwise determine our reimbursement for clinical
laboratory services. For the year ended December 31, 2009,
revenues from clinical lab services were less than
10 percent of our total revenues. Uncertainty also exists
around the extent of coverage and reimbursement for new
services. This legislation also provides for increases in the
number of persons covered by public and private insurance
programs in the U.S.
63
Reorganization
Transactions
In connection with this offering, we will enter into the
Reorganization Transactions described under “Organizational
Structure,” including the creation or acquisition of
amortizable tax assets in connection with this offering and the
Reorganization Transactions and the creation of liabilities in
connection with entering into the Tax Receivable Agreement,
concurrent with the completion of this offering.
Acquisitions
Through March 31, 2010, we have acquired 17 diagnostic
services companies throughout the United States. Our most recent
acquisition was completed on March 12, 2010. The following
summarizes the acquisitions we completed in 2007, 2008, 2009 and
2010.
2007
Acquisitions
During 2007, we acquired substantially all of the assets of
three pathology practices and 100 percent of the equity of
five pathology practices for an aggregate purchase price
(including acquisition costs) of $319.8 million and
additional consideration in the form of contingent notes. The
aggregate purchase price included cash of $306.1 million
and subordinated unsecured contingent notes payable of
$13.7 million. The cash portion of the purchase price was
funded primarily with proceeds from member contributions and
drawings under our previous term loan facilities of
$115.3 million and $190.8 million, respectively.
In connection with one acquisition in 2007, we agreed to assume
up to $4.0 million to be paid to four retired physicians.
The obligation is to be paid over three to five years. As of
December 31, 2009, this acquisition related liability had a
remaining balance of approximately $0.6 million. During
2008 and 2009, we paid $1.9 million and $1.5 million,
respectively, related to this liability.
2008
Acquisition
During 2008, we acquired 100 percent of the equity of one
pathology practice for an aggregate purchase price (including
acquisition costs) of $27.3 million and additional
consideration in the form of contingent notes. The purchase
price was funded primarily with proceeds from the issuance of
Aurora Holdings’ Class A, C, and X membership
interests of $7.3 million and borrowings under our previous
term loan facility of $20.0 million.
2009
Acquisition
In November 2009, we acquired 100 percent of the equity of
one pathology practice for an aggregate cash purchase price of
$15.3 million. In addition, we issued contingent
consideration, payable over three years based on the acquired
practice’s future performance. We have estimated the fair
value of the contingent consideration and recorded a related
liability as of December 31, 2009 of $3.1 million. The
cash portion of the purchase price was funded primarily with
proceeds from the issuance of
Class A-1
membership interests in June 2009. The estimated fair value of
the assets acquired and liabilities assumed in connection with
the 2009 acquisition are preliminary and are expected to be
finalized in 2010.
2010
Acquisitions
On January 1, 2010, we acquired 100 percent of the
equity of two pathology practices for an aggregate cash purchase
price of $17.0 million. These acquisitions were consummated
on January 1, 2010 and, therefore, the cash paid totaling
$17.0 million was included in deposits and other
non-current assets as of December 31, 2009. On
March 12, 2010, we acquired 100 percent of the equity
of a pathology practice for an aggregate cash purchase price of
$22.5 million. Each transaction included contingent
consideration payable over three to five years based on the
acquired practices’ future performance. We have estimated
the fair value of the contingent consideration and recorded a
related liability as of the date of each acquisition. The
maximum amount of the contingent cash consideration, assuming
the acquisition meets the maximum stipulated earnings level, is
$39.9 million payable over three to five years. We funded
the cash portion of the acquisitions using $31.0 million of
cash primarily related to
64
member contributions from the holders of Aurora Holdings
Class A-1 Units and an additional $8.5 million related
to the sale of Aurora Holdings Class Z membership interests.
The following table summarizes the consideration paid for the
acquisitions completed in 2007, 2008, 2009 and 2010.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Subordinated
|
|
Total
|
|
|
|
Cash Paid
|
|
Notes Issued
|
|
Consideration
|
|
|
|
(in thousands)
|
|
|
|
2007 Acquisitions
|
|
$
|
306,116
|
|
|
$
|
13,658
|
|
|
$
|
319,774
|
|
|
2008 Acquisition
|
|
$
|
27,301
|
|
|
$
|
—
|
|
|
$
|
27,301
|
|
|
2009 Acquisition
|
|
$
|
15,340
|
|
|
$
|
—
|
|
|
$
|
15,340
|
|
|
2010 Acquisitions
|
|
$
|
39,475
|
|
|
$
|
—
|
|
|
$
|
39,475
|
|
As a result of the significant number and size of the
acquisitions completed over the last four years, many of the
changes in our consolidated results of operations and financial
position discussed below relate to the acquisitions completed in
2007, 2008, 2009 and 2010.
65
Results
of Operations
The following table outlines, for the periods presented, our
results of operations as a percentage of net revenues.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Year Ended
|
|
|
Three Months Ended
|
|
|
|
|
December 31,
|
|
|
March 31,
|
|
|
|
|
2007
|
|
|
2008
|
|
|
2009
|
|
|
2009
|
|
|
2010
|
|
|
|
|
Net revenues
|
|
|
100.0
|
%
|
|
|
100.0
|
%
|
|
|
100.0
|
%
|
|
|
100.0
|
%
|
|
|
100.0
|
%
|
|
Operating costs and expenses:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost of services
|
|
|
43.3
|
|
|
|
42.1
|
|
|
|
41.8
|
|
|
|
42.0
|
|
|
|
48.1
|
|
|
Selling, general and administrative expenses
|
|
|
23.9
|
|
|
|
21.0
|
|
|
|
21.5
|
|
|
|
22.4
|
|
|
|
23.3
|
|
|
Provision for doubtful accounts
|
|
|
3.7
|
|
|
|
5.1
|
|
|
|
5.5
|
|
|
|
5.6
|
|
|
|
5.6
|
|
|
Intangible asset amortization expense
|
|
|
9.0
|
|
|
|
9.1
|
|
|
|
8.5
|
|
|
|
8.9
|
|
|
|
8.4
|
|
|
Management fees
|
|
|
1.0
|
|
|
|
1.0
|
|
|
|
1.0
|
|
|
|
1.0
|
|
|
|
1.0
|
|
|
Impairment of goodwill and other intangible assets
|
|
|
—
|
|
|
|
—
|
|
|
|
4.7
|
|
|
|
—
|
|
|
|
—
|
|
|
Acquisition and business development costs
|
|
|
0.6
|
|
|
|
0.4
|
|
|
|
0.6
|
|
|
|
0.3
|
|
|
|
0.6
|
|
|
Equity-based compensation expense
|
|
|
—
|
|
|
|
0.7
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total operating costs and expenses
|
|
|
81.5
|
|
|
|
79.4
|
|
|
|
83.6
|
|
|
|
80.2
|
|
|
|
87.1
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income from operations
|
|
|
18.5
|
|
|
|
20.6
|
|
|
|
16.4
|
|
|
|
19.8
|
|
|
|
12.9
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other income (expense):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest expense
|
|
|
(11.2
|
)
|
|
|
(13.7
|
)
|
|
|
(11.1
|
)
|
|
|
(11.7
|
)
|
|
|
(8.0
|
)
|
|
Write-off of deferred debt issue costs
|
|
|
(5.4
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Other income
|
|
|
0.2
|
|
|
|
0.1
|
|
|
|
—
|
|
|
|
0.1
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total other expense, net
|
|
|
(16.4
|
)
|
|
|
(13.6
|
)
|
|
|
(11.1
|
)
|
|
|
(11.6
|
)
|
|
|
(8.0
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income before income taxes
|
|
|
2.1
|
|
|
|
7.0
|
|
|
|
5.3
|
|
|
|
8.2
|
|
|
|
4.8
|
|
|
Provision for income taxes
|
|
|
1.2
|
|
|
|
0.3
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income
|
|
|
0.9
|
%
|
|
|
6.7
|
%
|
|
|
5.3
|
%
|
|
|
8.2
|
%
|
|
|
4.8
|
%
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Our historical consolidated operating results do not reflect:
|
|
|
| |
•
|
the Reorganization Transactions described under “Prospectus
Summary — Reorganization Transactions” and
“Organizational Structure;”
|
|
|
|
| |
•
|
the results of operations of our 2009 and 2010 acquisitions
prior to the effective date of those acquisitions; and
|
|
|
|
| |
•
|
this offering and the application of the net proceeds that we
will receive from this offering.
|
As a result, our historical consolidated operating results may
not be indicative of what our results of operations will be for
future periods.
Comparison
of the Three Months Ended March 31, 2010 and
2009
Net
Revenues
Net revenues increased $5.5 million or 13.4 percent to
$46.4 million for the three months ended March 31,
2010 from $40.9 million for the three months ended
March 31, 2009. Organic revenues decreased
$1.3 million or
66
3.1 percent from $40.9 million to $39.7 million,
and the remaining increase of $6.8 million reflects the
impact of the 2010 and 2009 acquisitions.
Organic accessions grew to approximately 377,000 accessions
compared to approximately 375,000 for the three months ended
March 31, 2009. During the quarter a number of our labs and
their related referral sources were negatively impacted by
severe winter weather, which resulted in lower testing volumes
for us. We estimate that the severe weather resulted in a
reduction of approximately 19,000 accessions and net revenue of
approximately $2.0 million during the quarter ended
March 31, 2010. Excluding the weather impact, we estimate
the organic accession volume would have increased
5.5 percent. The average revenue per accession decreased
3.5 percent from approximately $109 to approximately $105
primarily due to a change in service mix. This change in service
mix resulted primarily from more referring physicians converting
from global pathology services, where we provide both the TC and
PC of the accession, to a TC or PC arrangement, where we receive
only a portion of the revenue. In these cases, our net revenue
per accession has declined due to the fact that we no longer
receive the entire global fee for the service but instead only
receive a portion of the revenue for the service.
We expect the average revenue per accession of our organic
business to continue to decline primarily as the result of
changes in service mix, including our growth in women’s
health pathology services, including clinical tests, which tend
to have lower revenue per accession and, therefore, decrease
slightly our average revenue per accession. In addition, our
growth rates and average revenue per accession may be positively
or negatively impacted by the reimbursement market, service mix
and average revenue per accession of acquisitions completed in
the future.
Our pathology diagnostic testing services accounted for
substantially all of our revenues for the three month periods
ended March 31, 2010 and March 31, 2009.
Cost of
Services
Cost of services for the three months ended March 31, 2010
increased $5.1 million or 29.5 percent to
$22.3 million from $17.2 million for the three months
ended March 31, 2009. Of the total increase,
$3.1 million related to the acquisitions completed in 2010
and 2009 and the remaining $2.0 million related to our
existing business. Of the $2.0 million increase in costs of
services related to our existing business, approximately
$600,000 related to severance costs associated with physician
terminations and approximately $200,000 related to
start-up
costs of our clinical lab in North Carolina. The clinical lab
became operational in March 2010 and will complement our
existing anatomic pathology services, specifically for the
women’s health pathology market. In addition, we hired
additional pathologists to provide diagnostic services to our
referring physicians in the Arizona, Massachusetts and Minnesota
markets.
As a percentage of net revenues, cost of services was
48.1 percent and 42.0 percent for the three months
ended 2010 and 2009, respectively. The major factors negatively
impacting the percentage for 2010 compared to 2009 included the
severance and clinical lab start up costs, as well as the
$2.0 million shortfall in revenue related to the weather.
As a result the gross margin was 51.9 percent and
58.0 percent for the three months ended March 31, 2010
and 2009, respectively. We currently anticipate that our gross
margin will decline slightly due to a combination of lower
average revenue per accession and increased costs related to
pathologist retention and replacement and higher costs and lower
gross margins in our women’s health pathology services,
including clinical tests. Cost of services and our related gross
profit percentages may be positively or negatively impacted by
the market, service mix and unit price dynamics of acquisitions
completed in the future.
Selling,
general and administrative expenses
Selling, general and administrative expenses increased
$1.6 million, or 17.9 percent, to $10.8 million
for the three months ended March 31, 2010 from
$9.2 million for the three months ended March 31,
2009. Of the total increase, $1.4 million related to the
2010 and 2009 acquisitions and $0.2 million related to our
existing business. Of the increase in our existing business, the
majority relates to sales and marketing initiatives including
the increase in the number of sales representatives and our
introduction of doc2MD.
67
Selling, general and administrative expenses, as a percent of
net revenue, increased to 23.3 percent from
22.4 percent for the three months ended March 31,
2009. Selling, general and administrative expenses as a
percentage of revenue increased due to the lower than expected
revenue for the first quarter and the expansion of sales and
marketing activities during the second half of 2009 and the
first quarter of 2010. We expect to make additional investments
in selling, general and administrative expenses in 2010,
including the addition of field sales representatives, marketing
and IT personnel. In addition, we expect accounting, legal,
compliance and other public company related costs to increase
substantially following the completion of this offering.
Provision
for doubtful accounts
Our provision for doubtful accounts increased $0.3 million
or 13.2 percent to $2.6 million from $2.3 million
for the three months ended March 31, 2009. The majority of
the increase relates to the increase in the total net revenue of
$5.5 million. As a percentage of net revenue, the provision
for doubtful accounts was consistent from period to period at
5.6 percent of net revenue.
We expect our consolidated provision for doubtful accounts of
our existing businesses to range between 5.5 percent and
6.0 percent in future. The future provision for doubtful
accounts could be positively or negatively impacted by the bad
debt experience of future acquired laboratories.
Intangible
asset amortization expense (Amortization)
Amortization expense for the three months ended March 31,
2010 increased to $3.9 million from $3.6 million for
the three months ended March 31, 2009 related to increases
in our amortizable intangible assets associated with the 2010
and 2009 acquisitions. We amortize our intangible assets over a
weighted average lives ranging from 4 to 18 years.
Management
fees
Management fees increased $0.1 million to $0.5 million
for the three months ended March 31, 2010 compared to
$0.4 million for the three months ended March 31,
2009. Management fees are based on 1.0 percent of net
revenue plus expenses. The majority of the increase relates to
the increase in our net revenues. Following the completion of
this offering, we will not be obligated to pay management fees.
Acquisition
and business development costs
On January 1, 2009, we adopted a new accounting standard
related to accounting for business combinations using the
acquisition method of accounting (previously referred to as the
purchase method). In connection with this adoption, during 2010
and 2009 we expensed $0.3 million and $0.1 million,
respectively, of transaction costs associated with our completed
acquisitions and business development costs related to our
prospecting and unsuccessful acquisition activity.
Interest
Expense
Interest expense for the three months ended March 31, 2010
decreased to $3.7 million from $4.8 million for the
three months ended March 31, 2009, partially due to lower
outstanding borrowings under our previous term loan, as well as
lower effective interest rates. For the three months ended
March 31, 2010 our previous average term loan balance was
$208.4 million at an effective rate of 7.2 percent
compared to the prior quarter average term loan balance of
$216.7 million and an effective rate of 9.0 percent.
Provision
for Income Taxes
Prior to the completion of this offering, we were a Delaware
limited liability company for federal and state income tax
purposes, in accordance with the applicable provisions of the
Internal Revenue Code. Accordingly, we were generally not
subject to income taxes, and the income attributable to us was
allocated to the members of Aurora Holdings in accordance with
the terms of the Aurora Holdings LLC Agreement. We make tax
distributions to the members in amounts designed to provide such
members with sufficient cash to pay taxes on their allocated
68
income. However, certain of our subsidiaries are structured as
corporations and therefore are subject to federal and state
income taxes.
Upon the completion of this offering and the Restructuring
Transactions, we expect our provision for income taxes to be
more consistent with federal and state statutory rates, or
40 percent.
Comparison
of the Years Ended December 31, 2009 and 2008
Net
Revenues
Net revenues increased $13.7 million or 8.7 percent to
$171.6 million for the year ended December 31, 2009
from $157.9 million for the year ended December 31,
2008. Organic revenues increased $11.5 million or
7.7 percent from $150.2 million to
$161.8 million, and the remaining increase of
$2.2 million reflects the impact of the 2009 and 2008
acquisitions.
Organic accessions grew by approximately 64,000, or
4.5 percent, to approximately 1.5 million for the year
ended December 31, 2009 from approximately 1.4 million
for the year ended December 31, 2008. The organic volume
growth resulted from the expansion of our sales force in late
2008 and early 2009. The average revenue per accession increased
3.0 percent from approximately $106 to approximately $109
resulting from a combination of an increase in reimbursement
(price) and the ordering of additional tests for accessions
related to cervical screenings.
Our pathology diagnostic testing services accounted for
substantially all of our 2009 and 2008 revenues.
Cost of
Services
Cost of services for the year ended December 31, 2009
increased $5.4 million or 8.1 percent to
$71.8 million from $66.4 million for the year ended
December 31, 2008. Cost of services related to our organic
revenue grew 7.2 percent or $4.6 million, primarily
related to our organic growth in accessions of 4.5 percent.
The increase in our cost of services also included approximately
$0.3 million related to
start-up
costs of our clinical lab in North Carolina. The clinical lab
became operational in March 2010 and will complement our
existing anatomic pathology services, specifically for the
women’s health pathology market. The remaining increase in
cost of services was $0.5 million related to the 2009 and
2008 acquisitions.
As a percentage of revenues, cost of services for both periods
was approximately 42.0, percent resulting in a gross margin
of approximately 58.0 percent for both periods. We
currently anticipate our gross margin to decline slightly due to
a combination of lower average revenue per accession and
increased costs related to pathologist retention and replacement
and higher costs and lower gross margins in our women’s
health pathology services, including clinical tests. Cost of
services and our related profit percentages may be positively or
negatively impacted by the market, service mix and unit price
dynamics of acquisitions completed in the future.
Selling,
general and administrative expenses
Selling, general and administrative expenses increased
$3.7 million, or 11.0 percent, to $36.9 million
for the year ended December 31, 2009 from
$33.2 million for the year ended December 31, 2008. Of
the total increase, $0.4 million related to the 2009 and
2008 acquisitions and $3.3 million related to our existing
business. Of the total increase of $3.7 million,
approximately $1.8 million relates to sales and marketing
initiatives including the increase in the number of sales
representatives and our introduction of doc2MD. In
addition, our billing costs increased $0.6 million
primarily related to the 2009 and 2008 acquisitions. For both
periods billing costs were approximately 4.4 percent of net
revenue.
Selling, general and administrative expenses, as a percent of
net revenue, increased slightly to 21.5 percent from
21.0 percent for the year ended December 31, 2008. The
primary reason for this increase was the expansion of sales and
marketing activities in the year ended December 31, 2009.
We expect to make additional investments in selling, general and
administrative expenses in 2010, including the addition of
additional field sales representatives, marketing and IT
personnel. In addition, we expect accounting, legal, compliance
and other public company related costs to increase substantially
following the completion of this offering.
69
Provision
for doubtful accounts
Our provision for doubtful accounts increased $1.5 million
or 18.1 percent to $9.5 million from $8.0 million
for the year ended December 31, 2008. Approximately
$0.7 million relates to the increase in the total net
revenue of $13.7 million. The remainder of the increase of
$0.8 million related to the increase in the overall
provision as a percent of net revenue to 5.5 percent from
5.1 percent for the year ended December 31, 2008. A
major factor contributing to this increase was slower payment
patterns in certain markets such as Michigan and an increase in
our historical write-offs related to uncollectible accounts,
primarily patient accounts.
We expect our consolidated provision for doubtful accounts of
our existing businesses to range between 5.5 percent and
6.0 percent in future. The future provision for doubtful
accounts could be positively or negatively impacted by the bad
debt experience of future acquired laboratories.
Intangible
asset amortization expense (Amortization)
Amortization expense for the year ended December 31, 2009
increased to $14.6 million from $14.3 million for the
year ended December 31, 2008 related to increases in our
amortizable intangible assets associated with the 2009 and 2008
acquisitions. We amortize our intangible assets over a weighted
average lives ranging from 4 to 18 years.
Management
fees
Management fees increased $0.2 million to $1.8 million
for the year ended December 31, 2009 compared to
$1.6 million for the year ended December 31, 2008.
Management fees are based on 1.0 percent of net revenue
plus expenses. The majority of the increase relates to the
increase in our net revenues. Following the completion of this
offering, we will not be obligated to pay management fees.
Impairment
of goodwill and other intangible assets
For purposes of testing goodwill for impairment, each of our
acquired practices is considered a separate reporting unit. To
estimate the fair value of the reporting units, we utilize a
discounted cash flow model as the primary approach to value
supported by a market approach guideline public company method,
or the GPC Method, which is used as a reasonableness test. We
believe that a discounted cash flow analysis is the most
appropriate methodology to test the recorded value of long-term
assets with a demonstrated long-lived value. The results of the
discounted cash flow provide reasonable estimates of the fair
value of the reporting units because this approach is based on
each respective unit’s actual results and reasonable
estimates of future performance, and also takes into
consideration a number of other factors deemed relevant by
management, including but not limited to, expected future market
revenue growth and operating profit margins. We have
consistently used these approaches in determining the value of
goodwill. We consider the GPC Method to be an adequate
reasonableness test which utilizes market multiples of industry
participants to corroborate the discounted cash flow analysis.
We believe this methodology is consistent with the approach that
any strategic market participant would utilize if they were to
value one of our reporting units.
The following assumptions were made by management in determining
the fair value of the reporting units and related intangibles as
of September 30, 2009: (a) the discount rates ranged
between 13.0 percent and 15.0 percent, based on relative size
and perceived risk of the reporting unit; (b) an average
compound annual growth rate of 7.5 percent during the five year
forecast period; and (c) earnings before interest, taxes,
depreciation, and amortization with an average reporting unit
level margin of 38.9 percent. These assumptions are based on:
(a) the actual historical performance of the reporting
units and (b) management’s estimates of future
performance of the reporting units.
We also consider the economic outlook for the health care
services industry and various other factors during the testing
process, including hospital and physician contract changes,
local market developments, changes in third-party payor
payments, and other publicly available information.
As of September 30, 2009, we tested goodwill and intangible
assets for potential impairment and recorded a non-cash
impairment charge of $8.0 million resulting from a write
down of $6.6 million in the carrying value of goodwill and
a write down of $1.4 million in the carrying value of other
intangible assets. The write down of the goodwill and intangible
assets related to one reporting unit. Regarding this reporting
unit, we believe events
70
occurred and circumstances changed that more likely than not
reduced the fair value of the intangible assets and goodwill
below their carrying amounts. These events during 2009 consisted
primarily of the loss of significant customers present at the
acquisition date, which adversely affected the current year and
expected future revenues and operating profit of the reporting
unit.
As of December 31, 2009, we had goodwill and net intangible
assets of $386.4 million. Many factors, including
competition, general economic conditions, health care reform,
third party payment patterns and industry consolidation, could
have a negative impact on one or more of our reporting units
used in evaluating for impairment. Therefore, we may experience
additional impairment charges in future periods.
Acquisition
and business development costs
On January 1, 2009, we adopted a new accounting standard
related to accounting for business combinations using the
acquisition method of accounting (previously referred to as the
purchase method). In connection with this adoption, during 2009,
we expensed $0.6 million of transaction costs associated
with our completed acquisitions, which under the prior
accounting would have been capitalized as part of the purchase
price. In addition, we expensed $0.5 million of business
development costs related to our prospecting and unsuccessful
acquisition activity. Prior year amounts related to unsuccessful
acquisitions and business development activities were
reclassified to acquisition and business development costs from
selling, general and administrative expenses for comparative
purposes.
Equity
based compensation expense
There was no equity based compensation expense recorded for the
year ended December 31, 2009 because no equity awards were
granted during 2009. The prior year amount of $1.2 million
related to equity incentive awards granted in July 2008.
Interest
Expense
Interest expense for the year ended December 31, 2009
decreased to $19.0 million from $21.6 million for the
year ended December 31, 2008, partially due to lower
outstanding borrowings under our previous term loan, as well as
lower effective interest rates. During 2009, we paid down
$8.2 million of our existing term loan facility. In
addition, $125.0 million of the balance outstanding on the
term loan facility had a fixed interest rate, through our
interest rate swap, at an effective interest rate of
9.9 percent, while the remaining average balance of
$88.2 million had interest based on a floating LIBOR rate.
For the year ended December 31, 2009, the average
30 day LIBOR was 0.33 percent compared to
2.66 percent for the year ended December 31, 2008.
Therefore, the majority of the reduction in interest expense
related to the lower effective interest rate on our floating
rate debt.
Other
Income
Other income primarily consists of interest earned on our cash
and cash equivalents.
Provision
for Income Taxes
Prior to the completion of this offering, we were a Delaware
limited liability company for federal and state income tax
purposes, in accordance with the applicable provisions of the
Internal Revenue Code. Accordingly, we were generally not
subject to income taxes and the income attributable to us was
allocated to the members of Aurora Holdings in accordance with
the terms of the Aurora Holdings LLC Agreement. We make tax
distributions to the members in amounts designed to provide such
members with sufficient cash to pay taxes on their allocated
income. However, certain of our subsidiaries are structured as
corporations, and therefore are subject to federal and state
income taxes.
Upon the completion of this offering and the Restructuring
Transactions, we expect our provision for income taxes to be
more consistent with federal and state statutory rates, or
40 percent.
71
Comparison
of the Years Ended December 31, 2008 and 2007
Net
Revenues
Net revenues increased $94.4 million or 148.8 percent
to $157.9 million for the year ended December 31, 2008
from $63.5 million for the year ended December 31,
2007. Organic revenues increased $2.5 million or
11.3 percent from $22.4 million to $25.0 million
and the remaining increase of $91.9 million related to the
2008 and 2007 acquisitions.
Our organic revenue growth of $2.5 million resulted from a
20.0 percent increase in the volume of accessions and a
7.2 percent decrease in the average revenue per accession.
Organic accessions grew approximately 46,000 to approximately
274,000 accessions for the year ended December 31, 2009
compared to 228,000 for the year ended December 31, 2007.
The organic volume growth resulted from the expansion of our
sales force in late 2007 and 2008. The average organic unit
price per accession decreased from $98 to $91 resulting from
certain referring physicians converting from global billing to
PC-only arrangements which have a lower average revenue per
accession.
Our pathology diagnostic testing services accounted for
substantially all of our 2008 and 2007 revenues.
Cost of
Services
Cost of services for the year ended December 31, 2008
increased $38.9 million or 141.6 percent to
$66.4 million from $27.5 million for the year ended
December 31, 2007. Cost of services related to our organic
revenue grew 22.1 percent or $2.1 million, primarily
related to our organic accession growth of 20.0 percent.
Our pathologist costs accounted for $1.0 million of the
increase, which resulted primarily from the addition of a
pathologist to service increased accession volume in certain
growing markets and the replacement of two retiring pathologists
with higher-cost pathologists. The remaining increase in cost of
services of $36.8 million related to the 2008 and 2007
acquisitions.
As a percentage of revenues, cost of services was
42.1 percent for the year ended December 31, 2008
compared to 43.3 percent for the year ended
December 31, 2007, resulting in an increase in gross margin
from 57.0 percent to 58.0 percent between periods.
Selling,
general and administrative expenses
Selling, general and administrative expenses increased
$18.0 million, or 118.8 percent, to $33.2 million
for the year ended December 31, 2008 from
$15.2 million for the comparable period in 2007. Of the
total increase, $16.7 million related to the 2008 and 2007
acquisitions and $1.3 million related to our existing
business.
Of the total increase of $18.0 million, approximately
$4.0 million related to sales and marketing initiatives
including the increase in the number of sales representatives.
In addition, our billing costs increased $4.4 million as a
result of higher revenues in 2008 and increased billing cost as
a percentage of revenues. Billing costs were 4.4 percent
and 4.0 percent of our net revenues for the year ended
December 31, 2008 and 2007, respectively.
As a percent of net revenue, total selling, general and
administrative expenses decreased to 21.0 percent from
23.9 percent in 2007.
Provision
for doubtful accounts
Our provision for doubtful accounts increased $5.7 million
or 238.0 percent to $8.0 million from
$2.4 million for the year ended December 31, 2007.
Approximately $3.5 million relates to the increase in the
total net revenue of $94.4 million. The remainder of the
increase of $2.2 million related to the increase in the
overall provision as a percent of net revenue to
5.0 percent from 3.8 percent for the year ended
December 31, 2007. A major factor contributing to this
increase was the fact that the blended provision for doubtful
accounts as a percent of revenue for the 2008 and 2007
acquisitions was higher than the base business due to their mix
of services and geographic location in which they operate. This
increased our blended company average in 2008.
72
Intangible
asset amortization expense (Amortization)
Amortization expense for the year ended December 31, 2008
increased to $14.3 million from $5.7 million for the
year ended December 31, 2007 due primarily to the
amortization expense related to the identifiable intangible
assets acquired in connection with the 2008 and 2007
acquisitions.
Management
fees
Management fees increased $0.9 million to $1.6 million
for the year ended December 31, 2008 compared to
$0.6 million for the year ended December 31, 2007. The
majority of the increase relates to the increase in our net
revenues.
Acquisition
and business development costs
We expensed $0.7 million and $0.4 million for 2008 and
2007, respectively, of business development costs related to our
prospecting and unsuccessful acquisition activity. Prior year
amounts related to unsuccessful acquisitions and business
development activities were reclassified to acquisition and
business development costs from selling, general and
administrative expenses for comparative purposes.
Equity
based compensation expense
During 2008, we adopted the New Plan to replace our original
equity incentive plan. The New Plan provides awards of Aurora
Holdings membership interest units. These interest units are
designated as
Class D-1,
Class D-2
and
Class D-2
membership interest units of Aurora Holdings. During 2008, the
Company authorized and issued 4,000 D-1 units, 3,000 D-2 units
and 3,000 D-3 units. All membership interest units issued in
2008 were fully vested as of December 31, 2008.
Compensation expense of $1.2 million was recorded for these
awards.
The fair value of each Aurora Holdings’ membership interest
unit granted in 2008 was estimated using the following
assumptions:
| |
|
|
|
|
|
|
|
2008
|
|
|
|
Expected life
|
|
|
3 years
|
|
|
Volatility percentage
|
|
|
20.2
|
%
|
|
Interest rate
|
|
|
3.1
|
%
|
|
Dividends
|
|
|
—
|
|
|
Forfeiture rate
|
|
|
—
|
|
There were no Aurora Holdings’ membership units granted in
2007.
Interest
Expense
Interest expense for the year ended December 31, 2008
increased to $21.6 million from $7.1 million for the
year ended December 31, 2007, primarily due to higher
outstanding balances under our previous term loan, resulting
from borrowings related to our 2007 and 2008 acquisitions.
During 2008, we paid down $7.8 million of our previous term
loan facility. Our average outstanding interest bearing debt
increased to $218.8 million during 2008 from
$70.4 million in 2007.
Other
Income
Other income primarily consists of primarily of interest earned
on cash and cash equivalents.
Provision
for Income Taxes
Prior to the completion of this offering, we were a Delaware
limited liability company for federal and state income tax
purposes, in accordance with the applicable provisions of the
Internal Revenue Code. Accordingly, we were generally not
subject to income taxes and the income attributable to us was
allocated to the members of Aurora
73
Holdings in accordance with the terms of the Aurora Holdings LLC
Agreement. We make tax distributions to the members in amounts
designed to provide such members with sufficient cash to pay
taxes on their allocated income. However, certain of our
subsidiaries are structured as corporations, and therefore are
subject to federal and state income taxes.
Upon the completion of this offering and the Restructuring
Transactions, we expect our provision for income taxes to be
more consistent with federal and state statutory rates, or
40 percent.
Liquidity
and Capital Resources
We are a holding company with no material business operations.
Our principal asset is the equity interests we own in Aurora
Holdings. We conduct all of our business operations through the
direct and indirect subsidiaries of Aurora Holdings.
Accordingly, our only material sources of cash are dividends or
other distributions or payments that are derived from earnings
and cash flow generated by the subsidiaries of Aurora Holdings.
Since inception, we have primarily financed operations through
capital contributions from our Principal Equityholders, long
term debt financings and cash flow from operations.
On May 26, 2010, we entered into a $335.0 million
credit facility with Barclays Bank PLC and certain other
lenders. This new credit facility includes a 6 year
$225.0 million senior secured first lien term loan credit
facility and a $110.0 million senior secured revolving
credit facility ($50.0 million of which became available
upon the closing of our new credit facility and
$60.0 million of which will be available upon the
completion of an offering of equity satisfying certain criteria
under our credit agreement). Upon completion of this offering,
we expect the size of our revolving credit facility to increase
to $110.0 million. See “— Recent
Developments.” In connection with our new credit
facilities, we repaid all amounts outstanding under the credit
facilities we entered into in December 2007 with a syndicate of
lenders. Our previous credit facilities provided for loan
commitments of up to $255.0 million and for the lenders
thereunder to provide financing for us to repay the outstanding
balance of our former term loan facility, fund working capital
and acquire certain businesses. Our previous credit facility,
which we entered in December 2007, included:
|
|
|
| |
•
|
a revolver loan, not in excess of $5.0 million and
|
| |
| |
•
|
term loans, with a first and second lien, not in excess of
$165.0 million and $85.0 million, respectively.
|
For our previous credit facilities, the lenders’ unfunded
term loan commitment expired on April 30, 2008. As of
March 31, 2010, we had $130.4 million and
$76.5 million outstanding under the first lien and second
lien term loans, respectively. The funded, first lien term loan
was subject to quarterly principal payments beginning on
September 30, 2008 through September 30, 2012. Total
principal paid for the years ended December 31, 2009 and
2008 and the three months ended March 31, 2010 was $8.2,
$7.8 and $2.2 million, respectively, including mandatory and
voluntary payments. All loans under our previous term loan
facilities would have matured in December 2012. Our previous
term loan facilities were collateralized by substantially all of
our assets and guaranteed by all of our subsidiaries. For the
revolver and first lien term loan, interest was at the prime
rate plus 3.25 percent or LIBOR plus 4.25 percent. For
the second lien term loan, interest was at the prime rate plus
6.75 percent or LIBOR plus 7.75 percent. As of
March 31, 2010, the effective interest rates were
4.5 percent and 8.0 percent for the first lien and
second lien, respectively. Other principal payments on the term
loans may be due from time to time from annual excess cash flow
and net cash proceeds from either the sale of assets or equity.
The proceeds from our previous term loan facilities were used to
refinance our former term loan facility and acquire two
businesses in December 2007 and one business in March 2008.
On March 21, 2007 in conjunction with an acquisition
transaction, we entered into a subordinated, unsecured
contingent note with a prior owner of one of our acquired
practices. The payment amount is determined by the
practice’s cumulative EBITDA over a three-year period, with
a minimum payment not to be less than $1.0 million and a
maximum payment not to exceed $2.0 million. Payment amounts
include a 5.5 percent interest rate factor, thus we have
recorded the contingent note in the original purchase price at
its minimum payment amount, discounted by the interest rate
factor of 5.5 percent. The original discount of
$0.1 million is being amortized into interest expense over
the term of the contingent note using the interest method.
74
On April 30, 2007 in conjunction with an acquisition
transaction, we entered into a subordinated, unsecured
contingent note with prior owners of one of our acquired
practices. The payment amount is determined by the
practice’s cumulative EBITDA over a five-year period, with
a minimum payment not to be less than $15.0 million and a
maximum payment not to exceed $30.0 million. Payment
amounts include a 5.5 percent interest rate factor, thus we
recorded the contingent note in the original purchase price at
its minimum payment amount, discounted by the interest rate
factor of 5.5 percent. The original discount of
$2.2 million will be amortized into interest expense over
the term of the contingent note using the interest method.
On November 17, 2006, we entered into a subordinated,
unsecured note payable with a prior owner of one of our acquired
practices with the principal amount of $1.0 million to
assist in the financing of an acquisition. The note had a
three-year term and principal payments were due in equal
quarterly installments of $0.08 million. Interest was paid
quarterly at the prime rate.
Long-term debt consists of the following (in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31
|
|
|
March 31
|
|
|
|
|
2008
|
|
|
2009
|
|
|
2010
|
|
|
|
|
Term loan, first
lien(1)
|
|
$
|
140,766
|
|
|
$
|
132,566
|
|
|
$
|
130,366
|
|
|
Term loan, second
lien(1)
|
|
|
76,534
|
|
|
|
76,534
|
|
|
|
76,534
|
|
|
Revolver
loan(1)
|
|
|
1
|
|
|
|
—
|
|
|
|
—
|
|
|
Subordinated unsecured contingent note, dated March 21, 2007
|
|
|
614
|
|
|
|
316
|
|
|
|
316
|
|
|
Subordinated unsecured contingent note, dated April 30, 2007
|
|
|
10,481
|
|
|
|
8,072
|
|
|
|
8,072
|
|
|
Subordinated unsecured contingent note, dated November 17,
2006
|
|
|
320
|
|
|
|
—
|
|
|
|
—
|
|
|
Capital lease obligations
|
|
|
—
|
|
|
|
262
|
|
|
|
252
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
228,716
|
|
|
|
217,750
|
|
|
|
215,540
|
|
|
Less:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Original issue discount, net
|
|
|
(1,403
|
)
|
|
|
(1,098
|
)
|
|
|
(1,018
|
)
|
|
Current portion
|
|
|
(10,010
|
)
|
|
|
(11,596
|
)
|
|
|
11,597
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Long-term debt, net of current portion
|
|
$
|
217,303
|
|
|
$
|
205,056
|
|
|
$
|
202,925
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1)
|
|
Reflects the previous credit
facilities we entered into during December 2007 with a syndicate
of lenders which were repaid using borrowings under our new
credit facilities dated May 26, 2010.
|
As of March 31, 2010, future maturities of long-term debt
are as follows (in thousands):
| |
|
|
|
|
|
Years ending December
31,
|
|
|
|
|
|
|
2010, Remainder
|
|
$
|
9,412
|
|
|
2011
|
|
|
13,144
|
|
|
2012
|
|
|
192,871
|
|
|
2013
|
|
|
64
|
|
|
2014
|
|
|
48
|
|
|
|
|
|
|
|
|
|
|
$
|
215,540
|
|
|
|
|
|
|
|
In connection with the majority of our acquisitions, we have
agreed to pay additional consideration in future periods based
upon the attainment of stipulated levels of operating earnings
by each of the acquired entities, as defined in their respective
agreements. Generally the contingent consideration is payable
over periods ranging from
75
3 to 5 years. For all acquisitions prior to
January 1, 2009, we do not accrue contingent consideration
obligations prior to the attainment of the objectives, and the
amount owed becomes fixed and determinable. For the years ended
December 31, 2007, 2008 and 2009 and for the three months
ended March 31, 2010, we paid contingent consideration
related to acquisitions completed prior to 2009 of
$1.9 million, $12.5 million, $12.7 million and
$11.7 million, respectively.
For acquisitions completed after January 1, 2009, we
estimate the fair value of the contingent consideration and
record a related liability on our balance sheet. As of
March 31, 2010 we have recorded a $20.7 million
liability related to the fair value of the contingent
consideration related to our 2010 and 2009 acquisitions.
As of March 31, 2010, assuming the practices achieve the
maximum level of stipulated operating earnings, as defined in
the agreements, the maximum principal amount of contingent
consideration payable over the next three to five years is
approximately $126.6 million. A lesser amount will be paid
for earnings below the maximum level of stipulated earnings, and
no payments will be made if the practices do not achieve the
minimum level of stipulated earnings as outlined in their
respective agreements. Any such payments in the future for
acquisitions completed prior to January 1, 2009 would be
accounted for as additional purchase price and increase
goodwill. For acquisitions completed during 2009 and 2010,
future payments will be reflected in the change in the fair
value of the contingent consideration.
As of March 31, 2010, we had $3.2 million in cash and
cash equivalents deposited at high quality financial
institutions. Our primary uses of cash are to fund our
operations, service debt, including contingent notes, and
acquire property and equipment. Cash used to fund our operations
excludes the impact of non-cash items, such as the allowance for
doubtful accounts, depreciation, and non-cash stock-based
compensation, and is impacted by the timing of our payments of
accounts payable and accrued expenses and collections of
accounts receivable.
As of March 31, 2010, we had working capital of
$10.8 million. We expect to continue to spend substantial
amounts of capital to grow our business and acquire diagnostics
businesses. We believe the net proceeds that we will receive
from this offering, together with cash from operations and our
current and future debt financings, will be sufficient to fund
our capital requirements through 2012.
After the Reorganization Transactions and the completion of this
offering, we will enter into the Tax Receivable Agreement which
will obligate Aurora Diagnostics, Inc. to pay to the Tax
Receivable Entity 85 percent of certain cash tax savings,
if any, in U.S. federal, state, local and foreign income
tax realized by Aurora Diagnostics, Inc. from existing and
future tax attributes.
The actual amount and timing of any payments under the Tax
Receivable Agreement will vary depending upon a number of
factors. We expect that future payments under the Tax Receivable
Agreement in respect of tax attributes resulting from past
acquisitions, as well as from the Reorganization Transactions,
will aggregate $ million and
range from approximately
$ million to
$ million per year over the
next
years. These amounts reflect only certain cash tax savings
attributable to current tax attributes resulting from past
acquisitions described above, as well as from the Reorganization
Transactions. It is possible that future transactions or events
could increase or decrease the actual tax benefits realized and
the corresponding Tax Receivable Agreement payments from these
tax attributes. Future payments under the Tax Receivable
Agreement in respect of subsequent acquisitions of Aurora
Holdings Units would be in addition to these amounts and would,
if such exchanges took place at the initial public offering
price, be of comparable magnitude.
In connection with the Reorganization Transactions, this
offering and the related Tax Receivable Agreement, we recorded a
deferred tax asset of million
and a liability related to the Tax Receivable Agreement
of million, which represents
our obligations to the Tax Receivable Entity. The remaining 15%
or million was recorded as an
increase to additional paid in capital, a component of
stockholders’ equity.
Because we are a holding company with no operations of our own,
our ability to make payments under the Tax Receivable Agreement
is dependent on the ability of Aurora Holdings and its
subsidiaries to make distributions to us. Subject to certain
exceptions, our debt agreements restrict the ability of our
subsidiaries to make distributions out of net cash provided by
operating activities to us, which could affect our ability to
make payments under the Tax Receivable Agreement.
76
Cash
flows from operating activities
Net cash provided by operating activities during the three
months ended March 31, 2010 was $4.0 million compared
to $5.8 million during the three months ended
March 31, 2009. The decrease related primarily to a
decrease in net income of $1.1 million and an increase in
the change in net accounts receivable of $1.1 million
compared to the prior quarter. Net accounts receivable increased
due to a billing conversion at one of our labs, which resulted
in a delay in collections, and due to the withholding of
payments by Medicare intermediaries for certain periods while
legislative changes were put in place. As of March 31, 2010
our DSO (Days Sales Outstanding) averaged 37 days, up
slightly from 36 days for the prior quarter.
Net cash provided by operating activities during the year ended
December 31, 2009 was $36.4 million compared to
$29.0 million during the year ended December 31, 2008.
The increase related primarily to an increase in net income
after giving effect to the non cash charge for the impairment of
goodwill and intangible assets. As of December 31, 2009 our
DSO averaged 34 days compared to 32 days in 2008.
Net cash provided by operating activities during the year ended
December 31, 2008 was $29.0 million compared to
$9.8 million during the year ended December 31, 2007.
The increase related primarily to an increase in net income,
adjusted for depreciation and amortization related to our 2007
and 2008 acquisitions. As of December 31, 2008 our DSO
averaged 32 days compared to 39 days in 2007. Our DSO
improved in 2008 as the result of a focused effort on improving
our collection efforts at certain billing locations.
Cash flow
used in investing activities
Net cash used in investing activities during the three months
ended March 31, 2010 was $33.5 million compared to
$6.2 million during the three months ended March 31,
2009. Net cash used in investing activities during the three
months ended March 31, 2010 consisted of $0.9 million
of purchases of property and equipment, $11.7 million for
the payment of contingent notes and $20.9 million for our
acquisition completed March 12, 2010, net of cash acquired.
Contingent note payments were higher due to the timing of
payments compared to the quarter ended March 31, 2010. A
number of the practices contingent note payments were determined
and paid in the fourth quarter of 2008 as opposed to the first
quarter of 2009.
Net cash used in investing activities during the year ended
December 31, 2009 was $49.3 million compared to
$46.3 million during the year ended December 31, 2008.
Net cash used in investing activities during the year ended
December 31, 2009 consisted of $3.0 million of
purchases of property and equipment, $16.9 million for
deposits (primarily for acquisitions completed January 1,
2010), $12.7 million for the payment of contingent notes
and $16.7 million for acquisitions, net of cash acquired.
The purchases of equipment during 2009 primarily consist of
$1.0 million related to computer equipment and software,
$1.3 million related to laboratory equipment, and
$0.7 million related to automobiles, furniture and
leasehold improvements. The computer equipment and software
primarily consisted of servers, desktops, laptops, and related
software in support of our headcount growth across all
functional areas. The laboratory equipment primarily consisted
of microscopes and other analysis equipment for our existing
laboratories.
The increase in deposits relates to our payment on
December 31, 2009 of approximately $17.0 million in
cash to acquire 100 percent of the equity of two pathology
practices. These acquisitions were consummated on
January 1, 2010 and therefore included in deposits as of
December 31, 2009.
In connection with the acquisitions, we have agreed to pay
additional consideration in future periods based upon the
attainment of stipulated levels of operating earnings by each of
the acquired entities, as defined in their respective
acquisition agreements. For all acquisitions prior to
January 1, 2009, we do not accrue contingent consideration
obligations prior to the attainment of the objectives, and the
amount owed becomes fixed and determinable. For the year ended
December 31, 2009, we paid contingent consideration of
$12.7 million.
The majority of the cash used for acquisitions for the year
ended December 31, 2009 related to our November 2009
acquisition of 100 percent of the equity of one pathology
practice for an aggregate cash purchase price of
$15.3 million. The cash portion of the purchase price was
funded primarily with proceeds from the June 2009 issuance of
Class A-1
membership interests.
77
Net cash used in investing activities was $46.3 million
during the year ended December 31, 2008 compared to
$303.5 million during the year ended December 31,
2007. Net cash used in investing activities during the year
ended December 31, 2008 consisted of $2.7 million of
purchases of property and equipment, $12.5 million for the
payment of contingent notes and $31.0 million for
acquisitions, net of cash acquired.
The purchases of equipment during 2008 primarily consist of
$1.3 million related to computer equipment and software,
$0.5 million related to laboratory equipment and
$0.9 million related to furniture and leasehold
improvements. The computer equipment and software primarily
consisted of servers, desktops, laptops, and related software in
support of our headcount growth across all functional areas. The
laboratory equipment primarily consisted of microscopes and
other analysis equipment for our existing laboratories.
The majority of the cash used for acquisitions for the year
ended December 31, 2008 related to our 2008 acquisition.
During 2008, we acquired substantially all of the assets of one
pathology practice, for an aggregate purchase price (including
acquisition costs) of $27.3 million and additional
consideration in the form of contingent notes. The purchase
price was funded primarily with proceeds from capital calls on
Aurora Holdings’ Class A, C, and X membership
interests of $7.3 million and our previous term loan
facility of $20.0 million.
The majority of the cash used for acquisitions for the year
ended December 31, 2007 related to the acquisition of eight
pathology practices for an aggregate cash purchase price of
$306.1 million. The cash portion of the purchase price was
funded primarily with contributions from members of Aurora
Holdings and borrowings under our previous term loan facility of
$115.3 million and $190.8 million, respectively.
Cash
flows from financing activities
Net cash provided by financing activities for the three months
ended March 31, 2010 was $5.2 million compared to net
cash used in financing activities of $2.4 million for the
three months ended March 31, 2009.
For the three months ended March 31, 2010, we made
mandatory repayments under our previous term loan of
$2.2 million compared to $1.8 million for the same
quarter of the prior year. For the three months ended
March 31, 2010, we received proceeds from the sale of
Aurora Holdings’ Class Z capital of $8.5 million,
which was offset by tax distributions to the members of the
Aurora Holdings of $0.6 million compared to the prior
quarter, in which we made tax distributions to the members of
Aurora Holdings of $0.5 million.
Net cash provided by financing activities for the year ended
December 31, 2009 was $33.0 million compared to
$16.0 million for the year ended December 31, 2008.
For the year ended December 31, 2009, we received proceeds
of $50.3 million in connection with the issuance of 21,382
Aurora Holdings Class
A-1
membership interests. In connection with the Class
A-1
issuance, we incurred $3.1 million of costs which were
recorded as a reduction in members’ equity. In addition,
during 2009 we made repayments of $8.2 million and
$3.0 million under our previous term loan facilities and
subordinated notes payable, respectively. Also during 2009, in
accordance with the Aurora Holdings LLC Agreement, we made tax
distributions to certain members totaling $2.8 million.
Net cash provided by financing activities for the year ended
December 31, 2008 was $16.0 million compared to
$300.5 million for the year ended December 31, 2007.
For the year ended December 31, 2008, we borrowed
$22.1 million under our previous term loan facility of
which $20.0 million, along with $7.3 million of
contributions from members of Aurora Holdings, was used to
complete the 2008 acquisition. In addition, during 2008 we made
repayments of $7.8 million and $2.9 million under our
previous term loan facilities and subordinated notes payable,
respectively. Also during 2008, in accordance with the Aurora
Holdings LLC Agreement, we made tax distributions to certain
Aurora Holdings’ members totaling $0.4 million. During
2008, we paid $1.8 million of costs related to Aurora
Holdings’ original member investments.
78
Contractual
Obligations and Commitments
The following table sets forth our long-term contractual
obligations and commitments as of March 31, 2010
(in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Payments Due by Period
|
|
|
Contractual
obligations
|
|
2010, Remainder
|
|
|
2011
|
|
|
2012
|
|
|
2013
|
|
|
2014
|
|
|
After 2014
|
|
|
Total
|
|
|
|
|
Term loans
|
|
$
|
6,518
|
|
|
$
|
10,406
|
|
|
$
|
189,975
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
206,900
|
|
|
Capital lease obligations
|
|
|
34
|
|
|
|
50
|
|
|
|
56
|
|
|
|
64
|
|
|
|
48
|
|
|
|
—
|
|
|
|
252
|
|
|
Estimated interest on term
loans(1)
|
|
|
8,908
|
|
|
|
11,484
|
|
|
|
8,303
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
28,695
|
|
|
Subordinated unsecured contingent notes
|
|
|
2,860
|
|
|
|
2,688
|
|
|
|
2,840
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
8,388
|
|
|
Real estate leases
|
|
|
2,090
|
|
|
|
2,716
|
|
|
|
2,213
|
|
|
|
1,895
|
|
|
|
1,475
|
|
|
|
6,270
|
|
|
|
16,659
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
$
|
20,411
|
|
|
$
|
27,344
|
|
|
$
|
203,387
|
|
|
$
|
1,959
|
|
|
$
|
1,523
|
|
|
$
|
6,270
|
|
|
$
|
260,894
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1)
|
|
Estimated interest payments on our
previous term loan facilities represent the majority of future
interest payments, and have been calculated using the effective
interest rates (three month LIBOR, plus credit spread)
multiplied by the outstanding balances as of March 31,
2010, less mandatory repayments, through their maturity dates.
As disclosed elsewhere in this prospectus, our previous term and
revolving loans have been refinanced by our new credit
facilities on May 26, 2010. See “— Recent
Developments.”
|
After the Reorganization Transactions and the completion of this
offering, we will enter into the Tax Receivable Agreement which
will obligate Aurora Diagnostics, Inc. to pay to the Tax
Receivable Entity 85 percent of certain cash tax savings,
if any, in U.S. federal, state, local and foreign income tax
realized by Aurora Diagnostics, Inc. from certain existing and
future tax attributes. See “Certain Relationships and
Related Party Transactions — Tax Receivable
Agreement.”
From time to time, we may enter into contracts with suppliers,
manufacturers, and other third parties under which we may be
required to make minimum payments. The table above does not
reflect any future obligations that may arise due to the
establishment of additional laboratory facilities, including
facility leasing costs, tenant improvements, and other facility
startup and infrastructure costs.
Off
Balance Sheet Arrangements
None.
Critical
Accounting Policies and Estimates
Our consolidated financial statements are prepared in accordance
with GAAP, which requires us to make estimates and assumptions
that affect the reported amounts of assets and liabilities and
disclosure of contingent assets and liabilities at the dates of
the financial statements and the reported amounts of revenues
and expenses during the reported periods. We have provided a
description of all our significant accounting policies in
Note 1 to our audited consolidated financial statements
included in the back of this prospectus. We believe that of
these significant accounting policies, the following may involve
a higher degree of judgment or complexity.
Net
Revenue and Accounts Receivable
Substantially all of our revenues consist of payments or
reimbursements for specialized diagnostic services rendered to
patients of our referring physicians. Our billings for services
reimbursed by third-party payors, including Medicare, and
patients are based on a company-generated fee schedule that is
generally set at higher rates than our anticipated reimbursement
rates. Our billings to physicians are billed based on negotiated
fee schedules that set forth what we charge for our services.
Reimbursement under Medicare for our services is subject to a
79
Medicare physician fee schedule and, to a lesser degree, a
clinical laboratory fee schedule, both of which are updated
annually. Our billings to patients include co-insurance and
deductibles as dictated by the patient’s insurance
coverage. Billings for services provided to uninsured patients
are based on our company-generated fee schedule. We do not have
any capitated payment arrangements, which are arrangements under
which we are paid a contracted per person rate regardless of the
services we provide.
Our net revenues are recorded net of contractual allowances
which represent the estimated differences between the amount
billed and the estimated payment to be received from third party
payors, including Medicare. Adjustments to the estimated
contractual allowances, based on actual receipts from
third-party payors, including Medicare, are recorded upon
settlement. Our billing is currently processed through multiple
systems. We have an ongoing process to evaluate and record our
estimates for contractual allowances based on information
obtained from each of these billing systems. This information
includes aggregate historical billing and contractual
adjustments, billings and contractual adjustments by payor
class, accounts receivable by payor class, aging of accounts
receivable, historical cash collections and related cash
collection percentages by payor and/or aggregate cash
collections compared to gross charges. In addition, we may take
into account the terms and reimbursement rates of our larger
third party payor contracts and allowables under government
programs in determining our estimates. The degree of information
used in making these estimates varies based on the capabilities
and information provided by each of our billing systems.
The process for estimating the ultimate collection of accounts
receivable associated with our services involves significant
assumptions and judgment. The provision for doubtful accounts
and the related allowance are adjusted periodically, based upon
an evaluation of historical collection experience with specific
payors for particular services, anticipated collection levels
with specific payors for new services, industry reimbursement
trends, accounts receivable aging and other relevant factors.
The majority of our provision for doubtful accounts relates to
our estimate of uncollectible amounts from patients who are
uninsured and may fail to pay their coinsurance or deductible
obligations. Changes in these factors in future periods could
result in increases or decreases in our provision for doubtful
accounts and impact our results of operations, financial
position and cash flows.
Annual
Impairment Testing of Goodwill and Intangibles
Goodwill is our single largest asset. We evaluate the
recoverability and measure the potential impairment of our
goodwill annually. The annual impairment test is a two-step
process that begins with the estimation of the fair value of the
reporting unit. For purposes of testing goodwill for impairment,
each of our acquired practices is considered a separate
reporting unit. To estimate the fair value of the reporting
units, we utilize a discounted cash flow model as the primary
approach to value supported by a market approach guideline
public company method, or the GPC Method, which is used as a
reasonableness test. We believe that a discounted cash flow
analysis is the most appropriate methodology to test the
recorded value of long-term assets with a demonstrated
long-lived value. The results of the discounted cash flow
provide reasonable estimates of the fair value of the reporting
units because this approach is based on each respective
unit’s actual results and reasonable estimates of future
performance, and also takes into consideration a number of other
factors deemed relevant by management, including but not limited
to, expected future market revenue growth and operating profit
margins. We have consistently used these approaches in
determining the value of goodwill. We consider the GPC Method as
an adequate reasonableness test which utilizes market multiples
of industry participants to corroborate the discounted cash flow
analysis. We believe this methodology is consistent with the
approach that any strategic market participant would utilize if
they were to value one of our reporting units.
The first step of the goodwill impairment process screens for
potential impairment and the second step measures the amount of
the impairment, if any. Our estimate of fair value considers
(i) the financial projections and future prospects of our
business, including its growth opportunities and likely
operational improvements, and (ii) comparable sales prices,
if available. As part of the first step to assess potential
impairment, we compare our estimate of fair value for the
reporting unit to the book value of the reporting unit. If the
book value is greater than our estimate of fair value, we would
then proceed to the second step to measure the impairment, if
any. The second step compares the implied fair value of goodwill
with its carrying value. The implied fair value is determined by
allocating the fair value of the reporting unit to all of the
assets and liabilities of that unit as if the reporting unit had
80
been acquired in a business combination and the fair value of
the reporting unit was the purchase price paid to acquire the
reporting unit. The excess of the fair value of the reporting
unit over the amounts assigned to its assets and liabilities is
the implied fair value of goodwill. If the carrying amount of
the reporting unit’s goodwill is greater than its implied
fair value, an impairment loss will be recognized in the amount
of the excess.
On a quarterly basis, we perform a review of our business to
determine if events or changes in circumstances have occurred
which could have a material adverse effect on our fair value and
the fair value of our goodwill. If such events or changes in
circumstances were deemed to have occurred, we would perform an
impairment test of goodwill as of the end of the quarter,
consistent with the annual impairment test performed on
September 30, and record any noted impairment loss.
The following assumptions were made by management in determining
the fair value of the reporting units and related intangibles as
of September 30, 2009: (a) the discount rates ranged
between 13.0 percent and 15.0 percent, based on relative size
and perceived risk of the reporting unit; (b) an average
compound annual growth rate of 7.5 percent during the five year
forecast period; and (c) earnings before interest, taxes,
depreciation, and amortization, with an average reporting unit
level margin of 38.9 percent. These assumptions are based on
(a) the actual historical performance of the reporting
units and (b) management’s estimates of future
performance of the reporting units.
We also consider the economic outlook for the healthcare
services industry and various other factors during the testing
process, including hospital and physician contract changes,
local market developments, changes in third-party payor
payments, and other publicly available information.
Intangible assets, acquired as the result of a business
combination, are recognized at fair value, as an asset apart
from goodwill if the asset arises from contractual or other
legal rights, or if it is separable. Intangible assets,
principally representing the fair value of customer
relationships, health care facility agreements and
non-competition agreements acquired, are capitalized and
amortized on the straight-line method over their expected useful
life, which generally ranges from 4 to 18 years.
We recognize impairment losses for intangible assets when events
or changes in circumstances indicate the carrying amount may not
be recoverable. We continually assesses whether an impairment in
the carrying value of the intangible assets has occurred. If the
undiscounted future cash flows over the remaining amortization
period of an intangible asset indicate the value assigned to the
intangible asset may not be recoverable, we reduce the carrying
value of the intangible asset. We would determine the amount of
any such impairment by comparing anticipated discounted future
cash flows from acquired businesses with the carrying value of
the related assets. In performing this analysis, we consider
such factors as current results, trends and future prospects, in
addition to other relevant factors.
As of September 30, 2009, we tested goodwill and intangible
assets for potential impairment and recorded a non-cash
impairment expense of $8.0 million resulting from a write
down of $6.6 million in the carrying value of goodwill and
a write down of $1.4 million in the carrying value of
intangible assets. The write down of the goodwill and intangible
assets related to one reporting unit. Regarding this reporting
unit, we believe events occurred and circumstances changed that
more likely than not reduced the fair value of the intangible
assets and goodwill below their carrying amounts. These events
during 2009 consisted primarily of the loss of significant
customers present at the acquisition date, which adversely
affected the current year and expected future revenues and
operating profit of the reporting unit.
Quantitative
and Qualitative Disclosures about Market Risk
We maintain our cash balances at high quality financial
institutions. The balances in our accounts may periodically
exceed amounts insured by the Federal Deposit Insurance
Corporation, of up to $250,000 at December 31, 2009 and
2008. We do not believe we are exposed to any significant credit
risk and have not experienced any losses.
Our primary exposure to market risk is interest expense
sensitivity due to changes in the underlying prime rate or LIBOR
which determines the all in cost of our interest expense on our
previous term loan facilities. As of
81
December 31, 2009, we had an interest rate swap agreement
that expired January 10, 2010. Therefore subsequent to that
date the majority of our debt will be variable rate debt and our
earnings could be impacted by changes in variable interest
rates. We do not enter into interest rate swap agreements, or
other derivative financial instruments, for trading or
speculative purposes. We plan to periodically review our
exposure to interest rate fluctuations and access strategies to
manage our exposure.
Recent
Accounting Pronouncements
On January 1, 2009, we adopted a new accounting standard
issued by the FASB related to accounting for business
combinations using the acquisition method of accounting
(previously referred to as the purchase method). Among the
significant changes, this standard requires a redefining of the
measurement date of a business combination, expensing direct
transaction costs as incurred, capitalizing in-process research
and development costs as an intangible asset and recording a
liability for contingent consideration at the measurement date
with subsequent re-measurements recorded in the results of
operations. This standard also requires costs for business
restructuring and exit activities related to the acquired
company to be included in the post-combination financial results
of operations and also provides new guidance for the recognition
and measurement of contingent assets and liabilities in a
business combination. In addition, this standard requires
several new disclosures, including the reasons for the business
combination, the factors that contribute to the recognition of
goodwill, the amount of acquisition related third-party expenses
incurred, the nature and amount of contingent consideration, and
a discussion of pre-existing relationships between the parties.
The application of this standard was material for business
combinations completed in 2009. The standard is likely to have a
significant effect on the way we allocate the purchase price of
certain future business combinations, including the recognition
and measurement of assets acquired and liabilities assumed and
the expensing of direct transaction costs and costs to integrate
the acquired business.
On January 1, 2009, we adopted a new accounting standard
issued by the FASB related to the disclosure of derivative
instruments and hedging activities. This standard expanded the
disclosure requirements about an entity’s derivative
financial instruments and hedging activities, including
qualitative disclosures about objectives and strategies for
using derivatives, quantitative disclosures about fair value
amounts of and gains and losses on derivative instruments, and
disclosures about credit-risk-related contingent features in
derivative instruments.
Effective June 30, 2009, we adopted a newly issued
accounting standard related to accounting for and disclosure of
subsequent events in our consolidated financial statements. This
standard provides the authoritative guidance for subsequent
events that was previously addressed only in United States
auditing standards. This standard establishes general accounting
for and disclosure of events that occur after the balance sheet
date but before financial statements are issued or are available
to be issued and requires us to disclose that we have evaluated
subsequent events through the date of the filing or issue date.
This standard does not apply to subsequent events or
transactions that are within the scope of other applicable GAAP
that provide different guidance on the accounting treatment for
subsequent events or transactions. The adoption of this standard
did not have a material impact on our consolidated financial
statements.
In June 2009, the FASB issued the FASB Accounting Standards
Codification, or ASC. The ASC has become the single source of
non-governmental accounting principles generally accepted in the
United States of America, or GAAP, recognized by the FASB in the
preparation of financial statements. We adopted the ASC as of
July 1, 2009. The ASC does not change GAAP and did not have
an effect on our financial position, results of operations or
cash flows.
In June 2009, the FASB issued new accounting guidance on when an
entity should be included in consolidated financial statements.
The new guidance amends the evaluation criteria to identify the
primary beneficiary of a variable interest entity, or VIE, and
requires ongoing reassessment of whether an enterprise is the
primary beneficiary of the VIE. The new guidance significantly
changes the consolidation rules for VIEs including the
consolidation of common structures, such as joint ventures,
equity-method investments, and collaboration arrangements. The
guidance is applicable to all new and existing VIEs. This
standard is effective for us as of
82
January 1, 2010 and we do not expect the impact of its
adoption to be material to our consolidated financial statements.
In January 2010, the FASB issued an amendment to the accounting
standards related to the disclosures about an entity’s use
of fair value measurements. Among these amendments, entities
will be required to provide enhanced disclosures about transfers
into and out of the Level 1(fair value determined based on
quoted prices in active markets for identical assets and
liabilities) and Level 2 (fair value determined based on
significant other observable inputs) classifications, provide
separate disclosures about purchases, sales, issuances and
settlements relating to the tabular reconciliation of beginning
and ending balances of the Level 3 (fair value determined
based on significant unobservable inputs) classification and
provide greater disaggregation for each class of assets and
liabilities that use fair value measurements. Except for the
detailed Level 3 roll-forward disclosures, the new standard
is effective for us for interim and annual reporting periods
beginning after December 31, 2009. The requirement to
provide detailed disclosures about the purchases, sales,
issuances and settlements in the roll-forward activity for
Level 3 fair value measurements is effective for us for
interim and annual reporting periods beginning after
December 31, 2010. We do not expect that the adoption of
this new standard will have a material impact on our
consolidated financial statements.
83
INDUSTRY
Our
Market
The U.S. diagnostic testing industry had revenues of
approximately $55 billion in 2008 and grew at a rate of
7 percent compounded annually from 2000 to 2008, according
to the Washington G-2 Report.
Testing
Markets
The markets within the broader diagnostic testing industry are
defined as anatomic pathology, clinical pathology, and gene
based and other esoteric testing. Anatomic pathology represents
our core focus and, in 2009, we derived approximately
94 percent of our revenues from anatomic pathology
services, approximately 5 percent of our revenues from
clinical pathology services and approximately 1 percent of
our revenues from gene based and other esoteric testing.
|
|
|
| |
•
|
Anatomic Pathology. Anatomic pathology
services involve the diagnosis of cancer and other medical
conditions through the examination of tissues (histology) and
the analysis of cells (cytology). Volume is measured by the
number of accessions, or the number of patient cases, where each
accession may then include multiple specimens. Generally, the
anatomic pathology process involves the preparation of slides by
trained histotechnologists or cytotechnologists and the review
of those slides by anatomic pathologists. Although anatomic
pathologists do not treat patients, they establish a definitive
diagnosis and may also consult with the referring physician. The
anatomic pathology market was $13 billion, or
24 percent, of 2008 total diagnostic testing industry
revenues according to the Laboratory Economics Report with
$7.6 billion derived from the outpatient anatomic pathology
channel. Generally, anatomic pathology services command higher
reimbursement rates, on a per specimen basis, than clinical
pathology services. According to the Washington
G-2 Report
the anatomic pathology market has expanded more rapidly than the
overall industry, with revenues growing 4.8 percent on a
compound annual basis between 2006 and 2009, compared to
4.5 percent for the rest of the industry. Excluding growth
in esoteric testing, the remainder of the industry grew at a
compound annual rate of only 0.1 percent over the same
period.
|
|
|
|
| |
•
|
Clinical Pathology. Clinical pathology
services generally involve chemical testing and analysis of body
fluids using standardized laboratory tests. These tests usually
do not require the expertise of a pathologist and are frequently
routine, automated and performed by large national or regional
clinical laboratory companies and hospital laboratories. This is
the largest diagnostic testing market representing
$34 billion, or 62 percent, of 2008 total industry
revenues according to the Laboratory Economics Report. The
clinical pathology market grew at a rate of 0.2 percent
compounded annually from 2006 to 2008, according to the
Washington G-2 Report. Quest Diagnostics Incorporated and
Laboratory Corporation of America are the largest providers to
the clinical laboratory market.
|
|
|
|
| |
•
|
Gene-Based and Other Esoteric
Testing. Esoteric testing services typically
involve unique and complex genetic and molecular diagnostics
performed by skilled personnel using sophisticated instruments.
As a result, the esoteric testing market is dominated by a
limited number of commercial laboratories. Compound annual
growth rate for the total esoteric testing market was
19.5 percent from 2006 to 2008, according to the Washington
G-2 Report. We believe the esoteric testing market will continue
to demonstrate the fastest growth of any market in the
U.S. diagnostic testing industry.
|
84
The following chart depicts the relative and total size of the
U.S. laboratory testing markets in 2008:
2008 U.S.
Laboratory Testing Markets
Source: Washington G-2 Report
Patient
Channels
The anatomic pathology market on which we focus includes both
inpatient and outpatient channels. We are primarily focused on
the non-hospital outpatient channel of the anatomic pathology
market.
|
|
|
| |
•
|
Outpatient Channel. The outpatient channel of
the anatomic pathology market involves diagnostic testing
performed on tissues and cells of patients who do not reside in
a medical facility during their diagnosis and treatment. The
outpatient channel is divided into the hospital outpatient
channel, which involves patients diagnosed and treated at
hospitals on an outpatient basis, and the non-hospital
outpatient channel, which involves patients diagnosed and
treated at medical facilities other than hospitals on an
outpatient basis. The non-hospital outpatient channel is the
largest component of the anatomic pathology market and has grown
more rapidly than other channels. This channel accounted for
$7.6 billion, or 57 percent, of anatomic pathology
revenues for the year ended December 31, 2008, representing
10 percent growth according to the Laboratory Economics
Report, and is expected to grow at a higher rate than the
overall industry from 2008 to 2012. The hospital outpatient
channel accounted for $1.9 billion, or 15 percent, of
anatomic pathology revenues, representing 4 percent growth
in 2008.
|
|
|
|
| |
•
|
Inpatient Channel. The inpatient channel of
the anatomic pathology market involves diagnostic testing
performed on tissues and cells of patients who reside in a
hospital during their diagnosis and treatment. Typically, the
hospital operates its own clinical and histology laboratories,
but contracts on an exclusive basis with an independent
pathology group. In 2008, according to the Laboratory Economics
Report, the hospital inpatient channel accounted for
$3.7 billion, or 28 percent, of the anatomic pathology
revenues.
|
The following chart depicts the relative and total size of the
outpatient and inpatient channels in 2008:
2008
Anatomic Pathology Laboratory Testing by Channel
Total: $13 billion
Source: Laboratory Economics Report
85
Anatomic
Pathology Subspecialties
The non-hospital outpatient channel of the anatomic pathology
market also includes a number of subspecialties, including
primarily women’s health pathology, urologic pathology and
dermatopathology. All three of these subspecialty testing areas
are among our core competencies, and we derived approximately
80 percent of our revenues, in aggregate, from these three
subspecialties in 2009.
|
|
|
| |
•
|
Women’s Health Pathology. The
women’s health pathology subspecialty, which includes
gynecological pathology and cytopathology, principally involves
diagnostic tests performed on samples such as Pap smears and
cervical biopsies that are primarily provided by referring
physicians in the obstetrics and gynecology areas. In 2008,
according to the Laboratory Economics Report, the women’s
health pathology subspecialty accounted for $2.6 billion,
or 34 percent, of the broader non-hospital outpatient
channel. In particular, the cervical cancer screening market in
the U.S. has tripled over the past 10 years to
$2 billion in 2008, driven by the adoption of more
expensive monolayer liquid-based testing technologies, favorable
changes in Medicare reimbursement and the introduction of
DNA-based human papillomavirus testing for indeterminate Pap
tests. From 1998 to 2007, monolayer technology grew from
10 percent to 92 percent of the women’s health
pathology subspecialty, according to the Washington G-2 Report.
|
|
|
|
| |
•
|
Urologic Pathology. The urologic pathology
subspecialty involves diagnostic tests performed on tissue
specimens such as prostate biopsies that are provided by
referring physicians in the urology area. In particular, the
prostate cancer screening market is large and growing and in
2008, according to the Laboratory Economics Report, represented
$1.7 billion, or 22 percent, of the broader
non-hospital outpatient channel.
|
|
|
|
| |
•
|
Dermatopathology. The dermatopathology
subspecialty primarily involves diagnostic tests performed on
samples such as skin biopsies that are primarily provided by
referring physicians in the dermatology or plastic surgery
areas. In 2008, according to the Laboratory Economics Report,
dermatopathology accounted for $1 billion, or
13 percent, of the broader non-hospital outpatient channel,
and the number of new skin cancer cases grew 15 percent
between 2007 and 2009, according to the Laboratory Economics
Report.
|
|
|
|
| |
•
|
Other. The non-hospital outpatient channel
includes other subspecialty areas such as gastrointestinal
pathology, which involves diagnostic tests performed on samples
such as endoscopic biopsies that are primarily provided by
gastroenterologists, and hematopathology, which involves
diagnostic tests performed on samples such as blood, bone marrow
and lymph node biopsies that are primarily provided by referring
physicians in the hematology and oncology areas. In 2008,
according to the Laboratory Economics Report, these other
subspecialties accounted for $2.3 billion, or
30 percent, in aggregate, of the broader non-hospital
outpatient channel.
|
The following chart depicts the relative and total size of the
U.S. non-hospital outpatient anatomic pathology
subspecialty testing markets in 2008:
Anatomic
Pathology Outpatient Subspecialties
Source: Laboratory Economics Report
86
Our
Opportunity
We believe that demand for diagnostic services in the
non-hospital outpatient channel of the anatomic pathology
market, including those in the women’s health pathology,
urologic pathology and dermatopathology subspecialties, will
continue to expand.
Aging
of the U.S. population
The number of individuals
65-years-old
and older will increase to 55 million, or by
36 percent, over the next decade, a percentage rate that is
nearly four times faster than that of the overall population,
according to the U.S. Census Bureau Report. According to
the American Cancer Society Report, the risk of being diagnosed
with cancer increases as individuals age, with an estimated
52 percent of all new cancer cases diagnosed in persons
65-years-old
and older in 2009.
Increasing
Incidence of Cancer
The number of new cancer cases per year grew by 2 percent
between 2007 and 2009 to approximately 1.5 million new
cases according to the American Cancer Society Report. Within
that total growth, new skin cancer cases per year grew by
15 percent between 2007 and 2009, representing the highest
percentage increase among all diagnosed cancer types and the
largest number of new cases per year of any type of cancer. With
a high cure rate of greater than 99 percent for certain
types of skin cancer, according to American Academy of
Dermatology, the benefits of preventive skin cancer screening
are significant. Screening for cervical cancer within the
women’s health pathology subspecialty grew at a
12 percent compounded annual rate between 1998 and 2008,
and we expect continued innovation in testing techniques to
drive continued rapid growth. Cancers that can be prevented or
detected earlier by screening account for approximately half of
new cancer cases per year. In 2008, approximately
$228 billion was spent in the U.S. for the diagnosis
and treatment of cancer, including $93 billion for direct
medical costs.
Expanding
Demand for Non-Hospital Outpatient Services
The non-hospital outpatient channel of the anatomic pathology
market is expected to continue growing at a higher percentage
rate than the overall industry, principally driven by patient
preference and the cost-effectiveness of outpatient treatment
settings like ambulatory surgery centers compared to inpatient
hospital-based services. In the year ended December 31,
2008, the number of outpatient surgical procedures represented
62 percent of all surgical procedures performed in the
U.S., as compared to 17 percent in 1980, according to the
AHA Reports.
Medical
Advancements Allowing for Earlier Diagnosis and Treatment of
Disease
Physicians are increasingly relying on diagnostic testing to
help identify the risk of disease, to detect the symptoms of
disease earlier, to aid in the choice of therapeutic regimen, to
monitor patient compliance and to evaluate treatment results. We
believe physicians, patients and payors increasingly recognize
the value of diagnostic testing as a means to improve health and
reduce the overall cost of health care through early detection.
Nursing
Home and Home Health Markets
We believe that the aging of the U.S. population is a
natural driver for the demand for long-term care facilities and
home health and that clinical laboratory testing is a critical
service offered by nursing homes. According to the Washington
G-2 Report, this market has grown from $168.7 billion in
2005 to $198.5 billion in 2008, and projected growth is
estimated at 6.0 percent.
87
BUSINESS
Overview
We are a specialized diagnostics company providing services that
play a key role in the diagnosis of cancer and other diseases.
Our experienced pathologists deliver comprehensive diagnostic
reports of a patient’s condition and consult frequently
with referring physicians to help determine the appropriate
treatment. Our diagnostic reports often enable the early
detection of disease, allowing referring physicians to make
informed and timely treatment decisions that improve their
patients’ health in a cost-effective manner.
We are a leading specialized diagnostics company in terms of
revenues, focused on the anatomic pathology market. We are
well-positioned in the higher-growth subspecialties of anatomic
pathology, with a leading market position in dermatopathology
and in the women’s health pathology subspecialty, and a
growing market position in urologic pathology, hematopathology
and general surgical pathology. Our strengths in anatomic
pathology are complemented by our specialized clinical and
molecular diagnostics offerings, which enable us to provide a
broad selection of diagnostic services to our referring
physicians, our primary clients.
The majority of our revenues in 2009 were derived from
physicians providing diagnostic services in the non-hospital
outpatient channel of the anatomic pathology market, which in
2008 was one of the fastest-growing and largest channels of that
market. We also maintain 36 exclusive contracts with hospitals
under which we provide inpatient and outpatient professional
anatomic pathology services. We also provide medical director
services and, for some hospitals, technical slide preparation
services.
Our business model builds upon the expertise of our experienced
pathologists to provide seamless, reliable and comprehensive
pathology and molecular diagnostics offerings to referring
physicians. We typically have established, long-standing
relationships with our referring physicians as a result of
focused localized delivery of diagnostic services, personalized
responses and frequent consultations, and flexible information
technology, or IT, solutions that are customizable to our
clients’ needs. Our IT and communications platform enables
us to deliver diagnostic reports to our clients generally within
24 hours of specimen receipt, helping to improve patient
care. In addition, our IT platform enables us to closely track
and monitor volume trends from referring physicians.
The success of our business model and the value of our
specialized diagnostic service offering are reflected in our
significant growth allowing us to reach $171.6 million in
annual revenues in 2009. Through a combination of organic growth
and strategic acquisitions, we have achieved a scale allowing us
to provide diagnostic services to the patients of our
approximately 10,000 referring physicians, generating
approximately 1.6 million accessions in 2009. With 19
primary laboratories across the United States, we have achieved
a national footprint and a leading presence in our local markets
upon which we are continuing to build a more integrated and
larger-scale diagnostics company.
Our
Competitive Strengths
We believe that we are distinguished by the following
competitive strengths:
|
|
|
| |
•
|
Leading Market Position in Higher-Growth Subspecialties of
Expanding Industry. We are a leading specialized
diagnostics company, focused on the faster-growing non-hospital
outpatient channel within the anatomic pathology market with
leading market positions in two of the three higher-growth
subspecialties of the market: dermatopathology and women’s
health pathology.
|
| |
| |
•
|
Locally-Focused Business Model with National
Scale. Our business model centers on achieving
significant local market share, which yields operating
efficiencies and national scale when consolidated across all of
our operations. The diagnostic services we provide are designed
specifically to meet the needs of the local markets we serve.
Our national infrastructure enables us to more efficiently
manage our operations, improve productivity and deliver a more
extensive menu of diagnostic services to our local clients. As a
result of our strong local presence and high-quality diagnostic
services, we have established significant loyalty with referring
physicians and key payors in our local markets. In 2009, we
derived more than 85 percent of our revenues from
locally-focused,
in-network
payor contracts.
|
88
|
|
|
| |
•
|
Experienced, Specialized Pathologists Focused on Client
Service. We believe our pathologists have
long-standing client relationships and provide high-quality
service within their respective local communities. Over
one-third of our pathologists are specialized in
dermatopathology, with the remainder focused on women’s
health pathology, urologic pathology, hematopathology and
general surgical pathology. This alignment of our
pathologists’ specialties with those of the referring
physicians is critical to our ability to retain existing and
attract new clients. Our clinical expertise and frequent
interactions with clients on patient diagnoses enables us to
establish effective consultative and long-term relationships
with referring physicians.
|
| |
| |
•
|
Professional Sales, Marketing and Client Service
Team. We maintain a sales, marketing and client
service team of over 100 professionals who are highly-trained
and organized by subspecialty to better meet the needs of our
referring physicians and their patients. Our sales
representatives are incentivized through compensation plans to
not only secure new physician clients, but also to maintain and
enhance relationships with existing physician clients. As a
result, they have enabled us to expand our geographic market
presence to 30 states and increase market penetration and
market share in our local markets.
|
| |
| |
•
|
Proprietary IT Solutions. Delivery of clinical
information is essential to our business and a critical aspect
of the differentiated service that we provide to our clients. We
have developed scalable IT solutions that maximize the
flexibility,
ease-of-use
and speed of delivery of our diagnostic reports, which has
enabled us to rapidly grow our accession volume and meet the
increasing physician demand for our diagnostic services. We also
monitor referral patterns on a daily basis using our IT
infrastructure, which allows us to respond quickly to referring
physicians through our sales and marketing teams. We achieved
this through the development of a proprietary suite of IT
solutions called ConnectDX that is compatible with most
electronic medical record, or EMR, systems. ConnectDX
incorporates customized interface solutions, low cost and
efficient printer capabilities, compliant web portal capacities,
and proprietary software, all resulting in efficient and
reliable onsite client connections.
|
|
|
|
| |
•
|
Proven Acquisition, Integration and Development
Capabilities. We have significant expertise and a
proven track record of identifying, acquiring and integrating
pathology practices into our diagnostic laboratory network. Our
management team successfully expanded our operations through the
acquisition of 16 anatomic pathology laboratories and one
clinical laboratory and through the development of two de
novo diagnostic laboratories. We have improved the performance
of the laboratories we have acquired by applying our standard
operating procedures, enhancing sales and marketing
capabilities, implementing our IT platform and realizing
efficiencies from our national operations and management. We
believe our operational platform, expertise and value
proposition enable us to capitalize on the considerable
consolidation opportunities in the highly-fragmented anatomic
pathology market.
|
|
|
|
| |
•
|
Experienced Senior Leadership. Our senior
management team has approximately 80 years of combined
experience in the health care industry, including senior
management positions with leading diagnostic companies including
AmeriPath, DIANON Systems and Laboratory Corporation of America,
and, collectively, have successfully completed over 65
acquisitions and built a number of de novo specialized
diagnostic laboratories. We believe that our management’s
strong reputation, extensive network of industry relationships
and experience in building and growing successful companies in
the industry help us to drive operating performance, hire and
retain talented pathologists and other employees and attract
acquisition candidates.
|
Our
Business Strategy
We intend to achieve growth by pursuing the following strategies:
|
|
|
| |
•
|
Continue to Drive Market Penetration through Sales and
Marketing. We plan to drive organic growth
through our professional sales and marketing organization. Our
63-person
sales and marketing team provides us with broad coverage to
augment and further penetrate existing physician relationships
and to develop new referral relationships. We plan to
strategically add sales professionals to laboratories in markets
that will most benefit from enhanced outreach, increasing our
presence in existing and new markets.
|
89
|
|
|
| |
•
|
Leverage our IT Platform to Increase Operating
Efficiencies. We believe our IT platform will
allow us to gain market share in our existing subspecialties by
improving productivity and reducing turnaround times. We have
recently introduced an IT solution called doc2MD, a
leading EMR system for dermatology practices for which we have
an exclusive, long-term license. We intend to continue to
develop our internal IT operations into a better-integrated
diagnostic platform, which will improve national coordination
and provide real time visibility into key performance metrics.
In addition, we plan to continue to introduce innovative IT
solutions, interface capabilities and market-specific IT
solutions that enhance our value proposition to referring
physicians.
|
| |
| |
•
|
Expand through Targeted Acquisitions. We plan
to identify and acquire leading laboratories to augment our
organic growth, broaden our geographic presence and enhance our
service offering. We intend to continue to build our business
and enhance our reputation as a preferred acquiror for
independent laboratories. We believe that our recognizable
identity and strong reputation make us a preferred partner for
independent laboratories.
|
| |
| |
•
|
Expand Diagnostic Services Capabilities. We
intend to expand our services in the areas of clinical and
molecular diagnostics to complement our existing anatomic
pathology businesses. We believe we can leverage our depth of
experience and physician relationships to sell these new
diagnostic services in conjunction with our existing testing
services as a comprehensive offering. As a “one-stop”
diagnostic services provider, we would not only better serve our
current clients, but also position ourselves to attract new
business under a more diverse service model.
|
| |
| |
•
|
Develop De Novo Diagnostics Laboratories. We
plan to continue to selectively develop diagnostic laboratories
on a de novo basis, as we have done in certain markets, to
expand our market presence, broaden our service offering and
leverage the capabilities of our existing laboratories and
pathologists.
|
| |
| |
•
|
Expand Contracts with Hospitals in Target
Markets. We intend to continue to develop
additional contracts with hospitals in target markets as part of
a broader strategy to strengthen and grow our outpatient
business and expand our local market share.
|
| |
| |
•
|
Further Expand into Growing Long-Term Care
Market. We have a growing presence providing
clinical diagnostic services to the long-term care markets in
Central and Northern Florida. We intend to expand this regional
coverage into the large South Florida market and replicate our
success in other states with growing long-term care markets. We
believe that our IT solutions, and our ability to meet the rapid
service requirements for the long term care market, provide us
with a significant competitive advantage in these markets.
|
Our
Services
Anatomic pathology typically requires a pathologist to make a
specific diagnosis. Anatomic pathologists are medical doctors
who specialize in the study of disease. Anatomic pathologists do
not treat patients, but rather assist other physicians in
determining the correct diagnosis of their patient’s
ailments. A pathologist’s diagnosis represents a critical
factor in determining a patient’s future care. In addition,
anatomic pathologists may consult with attending physicians
regarding treatment plans. In these capacities, the anatomic
pathologist often serves as the “physician’s
physician,” thereby creating long-term relationships.
Anatomic pathologists perform their duties in laboratories,
including independent free-standing local laboratories, hospital
laboratories, regional and national laboratories, in ambulatory
surgery centers and in a variety of other settings. Referring
physicians take specimens from patients, and those specimens are
transported to a laboratory by courier or an overnight delivery
service. Once received at the laboratory, a specimen is
processed and mounted onto a slide by laboratory technologists
for examination by a pathologist. Once the pathologist examines
a specimen, the pathologist typically records the results of
testing performed in the form of a report to be transmitted to
the referring physician. Since specimens are transportable and
technology facilitates communication, samples can be diagnosed
by a pathologist from a remote location. Therefore, pathologists
are generally not needed
“on-site”
to make a diagnosis. This enhances utilization of available
capacity in outpatient and inpatient laboratories and allows the
practice to service a wider geographic area.
90
An anatomic pathologist must have an understanding of a broad
range of medicine. An anatomic pathologist may perform
diagnostic testing services for a number of subspecialty testing
markets such as dermatopathology, urologic pathology,
women’s health pathology, gastrointestinal pathology,
hematopathology or surgical pathology. While physical
examination or radiology procedures may suggest a diagnosis for
many diseases, the definitive diagnosis is generally established
by the anatomic pathologist.
Sales and
Marketing; Client Service
The selection of a pathologist to perform diagnostic testing
services is primarily made by individual referring physicians.
We maintain a sales and marketing team of 63 professionals who
are highly-trained and organized by subspecialty to better meet
the needs of our referring physicians. We have designed our
compensation structure to incentivize our sales representatives
to not only secure new physician clients, but also to maintain
and enhance relationships with existing physician clients. As a
result, our sales and marketing team has enabled us to expand
our geographic market presence to 30 states by leveraging
our extensive offering of diagnostic services across our markets.
We currently focus our marketing and sales efforts primarily on
dermatologists, urologists and gynecologists, as well as
gastroenterologists, hematologists and oncologists and their
office staff. The physicians on which our marketing and sales
efforts are focused include both non-hospital-based and
hospital-based physicians. Our sales representatives concentrate
on a geographic area based on the number of existing clients and
client prospects, which we identify using several national
physician databases that provide physician address information,
patient demographic information and other data.
At the beginning of a new client relationship, one of our sales
representatives visits a prospective physician client and
describes in detail our differentiated service offerings,
focusing on our experienced pathologists, local presence, rapid
turn-around times, comprehensive diagnostic reports, client
service and IT solutions. Our sales representatives focus on the
specialties offered by their respective divisions, which allows
them not only to discuss our specialized diagnostic services,
but also to describe diagnostic developments and new products
and technologies in their practice areas.
We also maintain a client service team of over 40 professionals
who are highly-trained and organized by subspecialty. Our
dedicated client service team provides ongoing support to our
clients and, in particular, the office staff of our referring
physicians. Our client service team enables us to augment the
long-standing relationships between our pathologists and their
clients to maintain a more stable base of referrals. These
service teams provide our clients with a personal, knowledgeable
and consistent point of contact within our company. Client
service team members coordinate the provision of services,
ensure testing supplies are replenished, answer administrative
and billing questions, and resolve service issues. We believe
these additional client contacts greatly enhance client
satisfaction and strengthen overall client relationships.
Once a client relationship is established, our sales
representatives and client service team provide frequent
follow-up
sales and service calls to ensure we are continuing to meet
physician needs and expectations and to explore other
opportunities for the physician to use our diagnostic services.
For example, once a client relationship is established, our
sales representatives and client service team frequently contact
the client to track testing volume and monitor satisfaction. In
addition, our sales representatives and client service team
frequently conduct client surveys, and our pathologists dialogue
with clients to develop relationships and identify areas in
which our relationships and service levels may be improved or
expanded. We believe that the frequency of these sales calls
allows our sales representatives and client service team to
build and enhance strong relationships with our clients, helping
us to better understand their needs and develop new service
offerings.
We currently focus our marketing and sales efforts by
subspecialty. Our representatives are extensively trained in the
specific subspecialty they service and they are knowledgeable
about our test offerings and new diagnostic technologies
available in the market by subspecialty. This provides
additional value to our physician clients and staff, as our
representatives become a resource to our client’s practice.
Our product offerings which have been developed to meet the
unique needs of each subspecialty are branded under the
following names:
|
|
|
| |
•
|
DermDX (dermatopathology services for the dermatology market);
|
91
|
|
|
| |
•
|
WomensDX (Womens Health Services for the OB/GYN market);
|
| |
| |
•
|
UroDX (for the Urology market);
|
|
|
|
| |
•
|
GastroDX (for the Gastroenterology market);
|
|
|
|
| |
•
|
HemaDX (for the Hematology and Oncology market);
|
|
|
|
| |
•
|
TreatmentDX (drug treatment market); and
|
|
|
|
| |
•
|
CareDX (long term and assisted care markets).
|
The offerings provide a comprehensive test menu so each
physician specialist can order the best test available to make a
accurate diagnosis and appropriate treatment decisions.
Competition
The anatomic pathology market is highly competitive. Competition
in our industry is based on several factors, including price,
clinical expertise, quality of service, client relationships,
breadth of testing menu, speed of turnaround of test results,
reporting and IT systems, reputation in the medical community
and ability to employ qualified personnel. Our competitors
include local and regional pathology groups, national
laboratories, hospital-based pathology groups and specialty
physician groups.
|
|
|
| |
•
|
Local and Regional Pathology Groups. Local and
regional pathology groups typically provide a relatively narrow
menu of test services to community physicians and, in certain
cases, to hospital-based pathologists.
|
| |
| |
•
|
National Laboratory Companies. National
laboratories typically offer a full suite of tests for a variety
of medical professionals, including general practitioners,
hospitals and pathologists. National laboratories have
identified anatomic pathology as a focus area for future growth
and will continue to be a competitive challenge going forward.
|
| |
| |
•
|
Hospital Pathologists. Pathologists working in
hospitals typically provide most of the diagnostic services
required for hospital in-patients and, sometimes, hospital
outpatients. Hospital pathologists act as medical directors for
the hospital’s clinical and histology laboratories.
Typically, hospital pathologists provide these services to
hospitals under exclusive and long-term contractual arrangements.
|
|
|
|
| |
•
|
Specialty Physician Groups. An increasing
number of specialty physician groups (urologists and
gastroenterologists in particular) are building their own
laboratories and in-sourcing pathology services. This trend
represents a significant source of competition, and it could
impact the anatomic pathology landscape in the future. According
to the Laboratory Economics Report, at least 500 specialty
physician groups have set up laboratories between 2005 and 2009.
|
The anatomic pathology market remains highly-fragmented, with
the two largest clinical laboratory companies accounting for
only 14 percent of annual revenues for the market in 2008.
We are one of approximately ten similarly-sized anatomic
pathology companies (in terms of revenues) that comprised, in
aggregate, approximately 14 percent of annual revenues for the
market in 2008. The remaining 72 percent of annual revenues
for the market was comprised of over 13,000 pathologists and
numerous specialized testing companies that offer a relatively
narrow menu of diagnostic services. In 2008, approximately
70 percent of pathologists licensed in the U.S. were
in private practices according to the Washington G-2 Report.
There is an evolving trend among pathologists to form larger
practices to provide a broader range of outpatient and inpatient
services and enhance the utilization of the practices’
pathologists. We believe this trend can be attributed to several
factors, including cost containment pressures by governmental
and other third-party payors, increased competition, managed
care and the increased costs and complexities associated with
operating a medical practice. Moreover, given the current trends
of increasing outpatient services, outsourcing and the
consolidation of hospitals, pathologists are seeking to align
themselves with larger practices that can assist providers in
the evolving health care environment. Larger practices can offer
pathologists certain advantages, such as obtaining and
negotiating contracts with hospitals and other providers,
managed care providers and national clinical laboratories;
marketing and selling of professional services; providing
continuing education and career advancement opportunities;
making available a broad range of specialists with whom to
consult; providing access to capital and business and management
experience; establishing
92
and implementing more efficient and cost effective billing and
collection procedures; and expanding the practice’s
geographic coverage area. Each of these factors support the
pathologists in the efficient management of the complex and
time-consuming non-medical aspects of their practice. As a
result, we believe that there are substantial consolidation
opportunities in the anatomic pathology market as smaller
pathology providers seek access to the resources of diagnostics
companies with a more comprehensive selection of services for
referring physicians.
Seasonality
Our business is affected by seasonal trends and generally
declines during the winter months, the year-end holiday period
and other major holidays. Adverse weather conditions can also
influence our business.
Quality
Assurance
We consider the quality of our diagnostic services to be of
critical importance, and we have established a comprehensive
quality assurance program for our laboratories designed to drive
accurate and timely test results and to ensure the consistent
high quality of our testing services. In addition to the
compulsory proficiency programs and external inspections
required by the Centers for Medicare & Medicaid Services,
or CMS, and other regulatory agencies, we have developed a
variety of internal systems and procedures to emphasize,
monitor, and continuously improve the quality of our operations.
We also participate in numerous externally-administered quality
surveillance programs, and our laboratories are accredited by
the College of American Pathologists, or CAP. CAP is an
independent, non-governmental organization of board-certified
pathologists that accredits laboratories nationwide on a
voluntary basis and that has been accredited by CMS to inspect
laboratories to determine adherence to the CLIA standards. The
CAP accreditation program involves both unannounced
on-site
inspections of our laboratories and our participation in
CAP’s ongoing proficiency testing program. A
laboratory’s receipt of accreditation by CAP satisfies the
Medicare requirement for participation in proficiency testing
programs administered by an external source, one of
Medicare’s primary requirements for reimbursement
eligibility.
Information
Systems
We are focused on implementing IT systems that streamline
internal operations and provide customized IT solutions to meet
the needs of our clients. We offer our IT solutions primarily
through the proprietary system ConnectDx and increasingly
through the doc2MD system for which we have an exclusive,
long-term license.
We developed ConnectDx, a customizable application
platform to provide a gateway for delivering and printing our
reports and communicating with many of our clients. A number of
our IT solutions provide an immediate impact to referring
physicians and their offices. The most common connectivity tools
include:
|
|
|
| |
•
|
Electronic interfaces;
|
| |
| |
•
|
Client EMR interfacing;
|
| |
| |
•
|
Internet report delivery (web portal);
|
| |
| |
•
|
Printed reports;
|
| |
| |
•
|
Patient education document;
|
| |
| |
•
|
Auto fax;
|
| |
| |
•
|
Patient data from clients office system requisitions;
|
| |
| |
•
|
Color remote printing hardware; and
|
| |
| |
•
|
Secure remote printing software.
|
Electronic interfaces provide a means through which we and our
clients can share data efficiently and accurately. These
customized interfaces can transfer patient information like
demographics, requisitions and diagnostic results between our IT
system and the IT system of our referring physicians. It takes
us an average of
93
eight weeks to build and implement a new interface tailored to
the client, whereas it may take our competitors up to eight
months to implement a standard interface.
Two key elements that we believe differentiate ConnectDx
from our competitors’ electronic interfaces are the
relative speed with which we can create and implement customized
solutions for clients and the reduced overhead costs associated
with doing so. Since ConnectDx was created to accommodate
flexibility, customizations such as delivering reports to
specified client system directories or printing multiple copies
of reports at physician offices during particular times each day
are easy to implement. This functional flexibility is achieved
with relatively low cost to us as a result of our IT
solution’s layered and adaptable design. We expect the
number of ConnectDx installations to grow and provide
additional value to our customers. We plan to expand the
products offered through ConnectDx to include utilization
and patient education reports as well as practice-specific
solutions.
We recently acquired an exclusive, long-term license to
doc2MD, an EMR and practice management software that was
specifically designed for dermatology practices, and we have
begun installing this software at select client locations. We
believe that the doc2MD software is a simple,
cost-effective program that allows dermatologists to quickly and
accurately document patient encounters. During 2009, we
conducted a market test for doc2MD with a number of our
dermatology practices and received positive feedback. We believe
that doc2MD will provide us with the platform to drive
our organic growth in the dermatopathology space, further
enabling us to maintain stable relationships with existing
dermatology clients and build relationships with new dermatology
clients.
Most of our IT solutions are implemented on a laboratory by
laboratory basis in connection with our efforts to integrate
acquired laboratories. We have developed, and will continue to
develop, faster integration times of our laboratory information
system, or LIS, offering. After an acquisition, we generally
transition our acquired laboratories to a common accounting
system and software package for financial processing and
reporting within 60 days of closing. Generally, the LIS and
billing platforms of our acquired laboratories, as well as all
their
day-to-day
laboratory operations, continue to operate as they did
pre-acquisition. We bill for our services using the existing
billing systems of the acquired laboratories or, in some
locations, we use an outsourced vendor to provide billing
services.
A significant benefit for acquired laboratories is the linkage
of data between our central database and our laboratories via a
private network. Information feeds from our laboratory systems
to our centralized backend database tally accession details,
client delivery products, practice management interfacing and
sales data. Daily sales volumes are monitored and can be
categorized by channel including product line, or territory. The
centralized client contact system’s ability to alert sales
representatives to changes in client trends within a single day
further enables our sales and client service teams to monitor
existing client retention and grow our client base.
Corporate
Structure
We derive our revenues from owned practices in which we either
directly employ our pathologists through our wholly owned
operating subsidiaries or we contract with an affiliated
practice entity that employs our pathologists. In 2009, we
derived 68% of our revenues from our affiliated practices.
We have long-term management agreements with eleven professional
entities in Michigan, Minnesota, Nevada, Texas, North Carolina,
Florida and Georgia. Pursuant to these management agreements, we
manage and control the non-medical functions of our affiliated
practices, including (i) recruiting, training, employing and
managing the technical and support staff; (ii) developing,
equipping and staffing laboratory facilities; (iii) establishing
and maintaining courier services to transport specimens; (iv)
negotiating and maintaining contracts with hospitals, managed
care organizations and other payors; (v) providing financial
reporting and administration, clerical, purchasing, payroll,
billing and collection, information systems, sales and
marketing, risk management, employee benefits, legal, tax and
accounting services; and (vi) monitoring compliance with
applicable laws and regulations. We receive a management fee
from each of our affiliated practices for the management
services we provide pursuant to the management agreements, each
of which has an initial term of 50 years and cannot be
terminated by our affiliated practice without cause.
Our Board of Directors and management formulate strategies and
policies which are implemented locally on a day-to-day basis by
each of our affiliated practices. Each affiliated practice has a
laboratory director who reports to
94
our Chief Executive Officer and Chief Operating Officer. Our
Chief Operating Officer, with the assistance of the laboratory
directors, supervises all employment and staffing matters
relating to our affiliated practices. We coordinate marketing
activities, administer national managed care and vendor
contracts, and create budgeting, accounting, billing, finance,
personnel and administrative policies and supervise the
implementation thereof.
In Michigan, Minnesota, Texas, Florida, Georgia and North
Carolina, our affiliated practices are owned by one or more
licensed physicians with whom we have entered into contractual
arrangements that govern the physicians’ ownership of our
affiliated practices. The physicians may not vote or transfer
their ownership interests in our affiliated practices or
distribute or encumber the assets of our affiliated practices
without our prior authorization. In addition, we have the
irrevocable and unconditional right to cause the physicians to
transfer their ownership interests in our affiliated practices
to our designee for nominal consideration. Through these
contractual arrangements, we have controlling voting and
financial interests in our affiliated practices. In Nevada, our
affiliated practice is owned by a trust, of which one of our
wholly owned subsidiaries is the sole beneficiary.
In addition, in Alabama, our laboratory is party to a long-term
services agreement with an unaffiliated physician group that
provides services to the laboratory on an exclusive basis. While
we do not have a controlling voting or financial interest in the
physician group, we have the right to consult with the physician
group regarding business decisions and approve or disapprove the
retention or discharge of all employees by the physician group.
The manner in which we acquire and operate a particular practice
is determined primarily by the corporate practice of medicine
restrictions of the state in which the practice is located and
other applicable regulations. We exercise diligence and care in
structuring our practices and arrangements with providers in an
effort to comply with applicable federal and state laws and
regulations, and we believe that our current practices and
arrangements do comply in all material respects with applicable
laws and regulations. However, due to uncertainties in the law,
there can be no assurance that our practices and arrangements
would be deemed to be in compliance with applicable laws and
regulations, and any noncompliance could result in a material
adverse effect on us. See “— Government
Regulation.”
Contracts
and Relationships with Providers
We employ our pathologists, control the practice entities that
employ our pathologists or contract with pathologists on an
independent contractor basis to provide diagnostic services in
our laboratories. While we exercise legal control over our
practices, we do not exercise control over, or otherwise
influence, the medical judgment or professional decisions of any
pathologist associated with our practices.
Our pathologist employment agreements typically have terms of
between 3 and 5 years and generally can be terminated by
either party without cause upon between 90 and 180 days
notice. Our pathologists generally receive a base compensation,
health and welfare benefits generally available to our employees
and, in some cases, annual performance bonuses. Our pathologists
are required to hold a valid license to practice medicine in the
jurisdiction in which they practice. We are responsible for
billing patients, physicians and payors for services rendered by
our pathologists. Most of our pathologist employment agreements
contain restrictive covenants, including non-competition,
non-solicitation and confidentiality covenants.
Our business is dependent on the recruitment and retention of
pathologists, particularly those with subspecialties like
dermatopathology. While we have generally been able to recruit
and retain pathologists in the past, no assurance can be given
that we will be able to continue to do so successfully or on
terms similar to our current arrangements. The relationship
between our pathologists and their respective local medical
communities is important to the operation and continued
profitability of our practices. In the event that a significant
number of pathologists terminate their relationships with us or
become unable or unwilling to continue their employment, our
business could be materially harmed.
We manage and control all of the non-medical functions of our
practices. We are not licensed to practice medicine. The
practice of medicine is conducted solely by the physicians in
our practices.
95
Billing
and Reimbursement
Billing
Billing for diagnostic services is generally highly complex.
Laboratories must bill various payors, such as private insurance
companies, managed care companies, governmental payors such as
Medicare and Medicaid, physicians, hospitals and employer
groups, each of which may have different billing requirements.
Additionally, the audit requirements we must meet to ensure
compliance with applicable laws and regulations, as well as our
internal compliance policies and procedures, add further
complexity to the billing process. Other factors that complicate
billing include:
|
|
|
| |
•
|
variability in coverage and information requirements among
various payors;
|
| |
| |
•
|
missing, incomplete or inaccurate billing information provided
by ordering physicians;
|
| |
| |
•
|
billings to payors with whom we do not have contracts;
|
| |
| |
•
|
disputes with payors as to which party is responsible for
payment; and
|
| |
| |
•
|
disputes with payors as to the appropriate level of
reimbursement.
|
Billing for diagnostic services in connection with governmental
payor programs is subject to numerous federal and state
regulations and other requirements, resulting in additional
costs to us. These additional costs include those related to:
|
|
|
| |
•
|
variability in coverage and information requirements among
various payors;
|
| |
| |
•
|
increased complexity in our billing due to the additional
procedures and processes required by governmental payor programs;
|
| |
| |
•
|
training and education of our employees and customers;
|
| |
| |
•
|
compliance and legal costs; and
|
| |
| |
•
|
costs related to, among other factors, medical necessity denials
and the absence of advance beneficiary notices.
|
Reimbursement
Depending on the billing arrangement and applicable law, the
party that reimburses us for our services may be:
|
|
|
| |
•
|
a third party who provides coverage to the patient, such as an
insurance company, managed care organization or a governmental
payor program;
|
| |
| |
•
|
the physician or other authorized party (such as a hospital or
another laboratory) who ordered the testing service or otherwise
referred the services to us; or
|
| |
| |
•
|
the patient.
|
For the year ended December 31, 2009, we derived
approximately 61 percent of our revenues from private
insurance, including managed care organizations and other
healthcare insurance providers, 25 percent from
governmental payor programs and 14 percent from other
sources.
In 2009, approximately 22 percent of our annual revenues
were derived from the Medicare program, which is overseen by
CMS. Because a large percentage of our revenues are derived from
the Medicare program, the Medicare coverage and reimbursement
rules are significant to our operations. Reimbursement under the
Medicare program for the diagnostic services that we offer is
subject to either the national Medicare clinical laboratory fee
schedule or the national Medicare physician fee schedule, each
of which is subject to geographic adjustments and is updated
annually. The physician fee schedule is designed to set
compensation rates for those medical services provided to
Medicare beneficiaries that require a degree of physician
supervision. Clinical diagnostic laboratory tests furnished to
non-hospital patients are paid according to the clinical
laboratory fee schedule.
96
Most of the services that we provide are anatomic pathology
services, which are reimbursed separately under the Medicare
physician fee schedule, and beneficiaries are responsible for
applicable coinsurance and deductible amounts. The physician fee
schedule is based on assigned relative value units for each
procedure or service, and an annually determined conversion
factor is applied to the relative value units to calculate the
reimbursement. The Sustainable Growth Rate formula used to
calculate the fee schedule conversion factor resulted in
significant decreases in payment levels in recent years,
including a decrease in physician fee schedule payments for 2010
of approximately 21 percent. On December 19, 2009, the
President signed into law a provision that delayed the cut for
two months until March 1, 2010. The reimbursement cut was
delayed until June 17, 2010 and Congress is currently
evaluating a further delay. It is not clear when, or if, the cut
will be enacted and, if so, for how long the cut will be
effective.
Future decreases in the Medicare physician fee schedule are
possible unless the U.S. Congress acts to change the
Sustainable Growth Rate formula used to calculate the fee
schedule payment amounts or continues to mandate freezes or
increases each year. Because the vast majority of our diagnostic
services currently are reimbursed under the physician fee
schedule, changes to the physician fee schedule could result in
a greater impact on our revenues than changes to the Medicare
clinical laboratory fee schedule.
The clinical laboratory fee schedule assigns amounts to specific
procedure billing codes, and each Medicare contractor
jurisdiction has a fee schedule that establishes the price for
each specific laboratory billing code. In addition, Medicare
also sets a cap on the amount that it will pay for any
individual test. Currently, this cap, usually referred to as the
National Limitation Amount, or NLA, is set at a percentage of
the median of all the contractor fee schedule amounts for each
test code. In the past, the U.S. Congress has frequently
lowered the percentage of the median used to calculate the NLA
in order to achieve budget savings. Currently, the NLA ceiling
is set at 74 percent of the medians for established tests
and 100 percent of the median for certain new tests that
were not previously reimbursed. In billing Medicare for clinical
laboratory services, we are required to accept, as payment in
full, the lowest of our actual charge, the fee schedule amount
for the state or local geographical area, or the NLA.
Because we must accept Medicare payment for clinical laboratory
services as payment in full, Medicare beneficiaries have no
coinsurance or deductible amount for clinical laboratory
services, although they do for anatomic pathology services.
However, on several occasions, in recent years, the
U.S. Congress has considered also imposing a
20 percent coinsurance on clinical laboratory fees, which
would require us to bill patients for their portion of these
services. To date, the U.S. Congress has never enacted such
a provision. However, because of the relatively low payment for
many clinical laboratory tests, the cost of billing and
collecting for these services would often exceed the amount
actually received from the patient. Therefore, the imposition of
such a requirement by the U.S. Congress would have the
effect of increasing our costs of billing and collecting.
The federal Social Security Act establishes that these clinical
laboratory fee schedule amounts are to be increased annually
based on the Consumer Price Index for All Urban Consumers, or
the CPI-U, as of June 30 for the previous
12-month
period. The U.S. Congress has frequently legislated that
the CPI-U increase not be implemented. For instance, the
U.S. Congress eliminated the CPI-U update through 2008 in
the Medicare Prescription Drug, Improvement and Modernization
Act of 2003, or MMA. For 2009, the CPI-U update would have been
5.0 percent. However, the Medicare Improvements for
Patients and Providers Act of 2008, or MIPPA, mandated a
0.5 percent cut to the CPI-U for years 2009 through 2013.
Accordingly, the update for 2009 was reduced to
4.5 percent. Because of the falling CPI-U and the
0.5 percent mandatory reduction, the clinical laboratory
fee schedule update for 2010 was -1.9 percent.
The U.S. Congress recently passed, and the President
recently signed into law, comprehensive health care reform
legislation that consisted of the PPACA and the HCEARA. This
legislation imposed additional cuts on the Medicare
reimbursement for clinical laboratories and repealed the
0.5 percent cut that was enacted by MIPPA and replaced it
with a “productivity adjustment” that will reduce the
update in payments for clinical laboratory tests by between 1.1
and 1.4 percent. In addition, this legislation includes a
1.75 percent reduction in reimbursement rates for clinical
laboratories for the years 2011 through 2015.
The payment amounts under the Medicare clinical laboratory fee
schedule are important not only for our reimbursement under
Medicare, but also because the schedule often establishes the
payment amounts set by other third party payors. For example,
state Medicaid programs are prohibited from paying more than the
Medicare fee schedule limit for clinical laboratory services
furnished to Medicaid recipients.
97
The MMA required CMS to conduct a competitive bidding
demonstration project for clinical laboratory tests, with some
exclusions, to determine whether competitive bidding could be
used to provide laboratory services at reduced cost to Medicare,
while continuing to maintain quality and access to care.
Anatomic physician services were not, however, covered by the
demonstration. The initial demonstration project in the
San Diego — Carlsbad — San Marcos,
California area was scheduled to begin on July 1, 2008.
However, the demonstration project was enjoined by an April 2008
preliminary injunction in a lawsuit filed by laboratory
plaintiffs against the Secretary HHS, and the demonstration was
later repealed in July 2008 by the U.S. Congress under
MIPPA. Currently, the court in the lawsuit is considering the
request by laboratories that CMS either return the bid
information or agree not to use it in any rate-setting
activities. The court recently denied the government’s
request to dismiss the lawsuit as moot. Despite the repeal of
the demonstration project, the HHS Secretary could retain the
bid application information to set lower future payment rates
for laboratory services if the court decides against the
laboratory plaintiffs. In addition, the U.S. Congress could
revisit competitive bidding for clinical laboratory services in
light of the current Administration’s efforts to reform the
healthcare system and generate healthcare savings. If
competitive bidding for clinical laboratory services is
implemented at a future time, we could be precluded from billing
the Medicare program for certain clinical laboratory services
furnished to Medicare beneficiaries if we are not a winning
bidder, or, as part of the competitive bidding process, we could
be required to offer reduced payment amounts in order to
participate in the arrangement.
When we provide clinical diagnostic laboratory services to
patients who reside in skilled nursing facilities, we must
comply with special billing rules for Medicare. For skilled
nursing facilities patients who are covered by Part A of
Medicare, which typically applies to patients who have been
discharged from a hospital into the skilled nursing facility, we
must bill, and be paid by, the skilled nursing facility itself.
For other Medicare patients, covered by Part B, we must
bill Medicare directly for the testing services requested.
Because skilled nursing facilities sometimes move between
Part A and Part B coverage, billing appropriately for
these services may be complex and time-consuming.
Government
Regulation
The services that we provide are heavily regulated by both
federal and state governmental authorities. Failure to comply
with the applicable regulations can subject us to significant
civil and criminal penalties, loss of license, or the
requirement that we repay amounts previously paid to us. The
significant areas of regulation are set out below.
Clinical
Laboratory Improvement Amendments of 1988 and State
Regulation
As a diagnostic service provider, each of our laboratory
entities is required to hold certain federal, state and local
licenses, certifications and permits to conduct our business.
Under the Clinical Laboratory Improvement Amendments of 1988, or
CLIA, each laboratory is required to hold a certificate
applicable to the type of work we perform at the laboratory and
to comply with certain CLIA-imposed standards. CLIA regulates
virtually all clinical laboratories by requiring they be
certified by the federal government and comply with various
operational, personnel, facilities administration, quality and
proficiency requirements intended to ensure that their clinical
laboratory testing services are accurate, reliable and timely.
CLIA does not preempt state laws that are more stringent than
federal law.
As part of the renewal process, our laboratories are subject to
survey and inspection every two years to assess compliance with
program standards and may be subject to additional random
inspections. Standards for testing under CLIA are based on the
level of complexity of the tests performed by the laboratory.
Laboratories performing high complexity testing are required to
meet more stringent requirements than laboratories performing
less complex tests. Each of our laboratories holds a CLIA
certificate to perform high complexity testing. CLIA compliance
and certification is also a prerequisite to be eligible to bill
for services provided to governmental payor program
beneficiaries. In addition, each laboratory must also enroll
with an approved proficiency testing program by which it
periodically tests specimens submitted from an outside
proficiency testing organization and then submits its results
back to the organization for grading. Failure to achieve a
passing score on a proficiency test can result in the laboratory
losing its right to perform the test at issue. Failure to comply
with other proficiency testing regulations, such as referring a
proficiency testing specimen to another laboratory for analysis,
can result in the revocation of the laboratory’s license
and of the licenses of other laboratories owned by us.
98
In addition to CLIA requirements, we are subject to various
state laws. CLIA provides that a state may adopt laboratory
regulations that are more stringent than those under federal
law. In some cases, the state programs actually substitute for
the federal CLIA program. In other instances the state’s
regulations may be in addition to the CLIA program. State laws
may require that laboratory personnel meet certain
qualifications, specify certain quality controls or prescribe
record maintenance requirements. Our laboratories are licensed
and accredited by the appropriate state agencies in the states
in which they operate.
Health
Insurance Portability and Accountability Act
Under the administrative simplification provisions of the Health
Insurance Portability and Accountability Act, or HIPAA, HHS has
issued regulations that establish uniform standards governing
the conduct of certain electronic healthcare transactions and
protecting the privacy and security of protected health
information, referred to as PHI, used or disclosed by healthcare
providers and other covered entities. Four principal regulations
with which we are currently required to comply have been issued
in final form under HIPAA: privacy regulations; security
regulations; standards for electronic transactions; and the
national provider identifier, or NPI, regulations. We must also
comply with regulations that require covered entities and
business associates to provide notification after a breach of
unsecured PHI.
The privacy regulations cover the use and disclosure of PHI by
healthcare providers. They also set forth certain rights that an
individual has with respect to his or her PHI maintained by a
healthcare provider, including the right to access or amend
certain records containing PHI or to request restrictions on the
use or disclosure of PHI. The HIPAA privacy regulations, among
other things, restrict our ability to use or disclose PHI in the
form of patient-identifiable laboratory data without written
patient authorization for purposes other than payment,
treatment, or healthcare operations, except for disclosures for
various public policy purposes and other permitted purposes
outlined in the privacy regulations. The privacy regulations
provide for significant fines and other penalties for wrongful
use or disclosure of PHI. In addition, the American Recovery and
Reinvestment Act of 2009, or ARRA, increases the civil monetary
penalty amounts for violations of the HIPAA regulations.
Although the HIPAA statute and regulations do not expressly
provide for a private right of damages, laws in certain states
provide for damages to private parties for the wrongful use or
disclosure of confidential health information or other private
personal information. Moreover, ARRA has created a new right of
action for state attorneys general to sue on behalf of
individuals who are harmed under the HIPAA regulations.
The security regulations establish requirements for safeguarding
the confidentiality, integrity and availability of PHI which is
electronically transmitted or electronically stored. The
security regulations provide for sanctions and penalties for
violations. In addition, ARRA increases the civil monetary
penalty amounts for violations of the HIPAA regulations and
creates a new right of action for state attorneys general. The
HIPAA privacy and security regulations establish a uniform
federal minimum standard and do not supersede state laws that
are more stringent or provide individuals with greater rights
with respect to the privacy or security of, and access to, their
records containing PHI. As a result, we are required to comply
with both HIPAA privacy and security regulations and varying
state privacy and security laws. ARRA also applies the HIPAA
privacy and security provisions and the civil and criminal
penalties associated with violating these provisions to business
associates in the same manner as they apply to covered entities.
In addition, HIPAA imposes standards for electronic
transactions, which establish standards for common healthcare
transactions. In particular, we utilize these standard
transaction sets where required by HIPAA.
Finally, HIPAA also established a new NPI as the standard unique
health identifier for healthcare providers to use in filing and
processing healthcare claims and other transactions.
As part of the HIPAA requirements, certain specified coding sets
are established that must be used for all billing transactions.
Currently, all healthcare providers use a system of diagnosis
coding referred to as the International Classification of
Diseases, 9th edition, or ICD-9. However, HHS, which oversees
HIPAA, has recently established a new requirement that will
require all healthcare entities, including ours, to move to a
new system of diagnosis codes, ICD-10, by October 1, 2013.
ICD-10 utilizes more codes and is considered more complex than
the current system. Because we must often rely on referring
physicians to supply us with the
99
appropriate diagnosis codes, the movement to the new system may
increase billing difficulties if physicians or payors have
difficulty in making the transition to the new codes.
In addition to PHI, the healthcare information of patients often
includes social security numbers and other personal information
that is not of an exclusively medical nature. The consumer
protection laws of a majority of states now require
organizations that maintain such personal information to notify
each individual if their personal information is accessed by
unauthorized persons or organizations so that the individuals
can, among other things, take steps to protect themselves from
identity theft. Penalties imposed by these state consumer
protection laws vary from state to state but may include
significant civil monetary penalties, private litigation and
adverse publicity. In addition, the Federal Trade Commission, or
FTC, has established new Red Flag Rules relating to identity
theft prevention programs, which are considered applicable to
healthcare companies. The compliance date for the Red Flag Rules
has repeatedly been delayed and is currently June 1, 2010.
Under the Red Flag Rules, the FTC is authorized to bring
enforcement actions in federal court for violations and could
impose a $2,500 penalty for each violation of the Red Flag
Rules. Noncompliance could also be penalized through state
enforcement or other civil liability.
Federal
and State Fraud and Abuse Laws
There are a variety of federal laws prohibiting fraud and abuse
involving federal government payment programs, such as Medicare
and Medicaid. These laws are enforced by the federal
U.S. Attorneys and the HHS Office of the Inspector General,
or OIG. In addition, the Medicare program increasingly uses a
variety of contractors to review billings submitted by providers
to the Medicare program. These contractors include Recovery
Audit Contractors, or RACs, and Carrier Error Rate Test, or
CERT, contractors. The RAC program began as a demonstration
program in a limited number of states that was designed to
detect improper Medicare payments to providers and suppliers. It
was expanded to cover all 50 states by Section 302 of
the Tax Relief and Health Care Act of 2006, which makes the RAC
program permanent and requires the HHS Secretary to expand the
program to all 50 states by no later than 2010. In
addition, CMS conducts CERT audits, which also monitor the
accuracy of payments made by Medicare payment contractors. In
either case, any overpayments found by the RAC or CERT
contractors must be repaid to the Medicare program. In some
cases, these overpayments can be used as the basis for an
extrapolation, by which the error rate is applied to a larger
universe of claims, which can result in even higher repayments.
The federal healthcare Anti-Kickback Law prohibits, among other
things, knowingly and willfully offering, paying, soliciting or
receiving remuneration as an inducement for, or in return for,
the purchase, lease or order of any healthcare item or service
reimbursable under a governmental payor program. The definition
of “remuneration” has been broadly interpreted to
include anything of value, including, for example, gifts,
discounts, the furnishing of supplies or equipment, credit
arrangements, payments of cash, waivers of payments, ownership
interests and providing anything at less than its fair market
value. The Anti-Kickback Law is broad, and it prohibits many
arrangements and practices that are lawful in businesses outside
of the healthcare industry.
Recognizing that the Anti-Kickback Law is broad and may
technically prohibit many innocuous or beneficial arrangements
within the healthcare industry, HHS has issued a series of
regulatory “safe harbors.” These safe harbor
regulations set forth certain provisions which, if met, will
assure healthcare providers and other parties that they will not
be prosecuted under the federal Anti-Kickback Law. Although full
compliance with these provisions ensures against prosecution
under the federal Anti-Kickback Law, the failure of a
transaction or arrangement to fit within a specific safe harbor
does not necessarily mean that the transaction or arrangement is
illegal or that prosecution under the federal Anti-Kickback Law
will be pursued. Still, in the absence of an applicable safe
harbor, a violation of the Anti-Kickback Law may occur even if
only one purpose of an arrangement is to induce patient
referrals or purchases or to induce the provision of a
laboratory service reimbursable by a federal healthcare program.
The penalties for violating the Anti-Kickback Law can be severe.
These sanctions include criminal penalties and civil sanctions,
including fines, imprisonment and possible exclusion from the
Medicare and Medicaid programs. Many states have also adopted
laws similar to the federal Anti-Kickback Law, some of which
apply to the referral of patients for healthcare items or
services reimbursed by any source, not only governmental payor
programs.
100
Generally, arrangements that would be considered suspect and
possible violations under the Anti-Kickback Law include
arrangements between a laboratory and a physician (or related
organizations or individuals) in which the laboratory would:
|
|
|
| |
•
|
provide items or services to the physician or other referral
source without charge or for amounts that are less than their
fair market value;
|
| |
| |
•
|
pay the physician or other referral source amounts that are in
excess of the fair market value of items or services that were
provided; or
|
| |
| |
•
|
enter into an arrangement with a physician or other entity
because it is a current or potential referral source.
|
In 2006, the OIG adopted a new safe harbor (and an exception
under the Physician Self-Referral Law, discussed below) that
permitted health care entities, including laboratories, to
provide electronic prescribing and electronic health records, or
EHR, technology to referring physicians. Compliance with the
provisions of these provisions allows us to donate EHR to
physicians without being found to be in violation of either the
Anti-Kickback Law or the Physician Self-Referral Law. Under
these provisions, we are permitted to donate software,
information technology and training services (but not hardware)
that is necessary and used predominately to create, maintain,
transmit or receive EHR, so long as the recipient of the
donation meets certain established criteria. Such technology
must be considered “interoperable” and must have
certain other capabilities established in the regulations. In
addition, the recipient of the donation must pay at least
15 percent of the cost of the service.
From time to time, the OIG issues alerts and other guidance on
certain practices in the healthcare industry. In October 1994,
the OIG issued a Special Fraud Alert on arrangements for the
provision of laboratory services. The Fraud Alert set forth a
number of practices allegedly engaged in by some laboratories
and healthcare providers that raise issues under the
Anti-Kickback Law. These practices include laboratories
providing:
|
|
|
| |
•
|
employees to furnish valuable services for physicians (other
than collecting patient specimens for testing for the
laboratory) that are typically the responsibility of the
physicians’ staff;
|
| |
| |
•
|
free testing to physicians’ managed care patients in
situations where the referring physician benefits from the free
laboratory tests;
|
| |
| |
•
|
free pick-up
and disposal of biohazardous waste for physicians for items
unrelated to a laboratory’s testing services;
|
| |
| |
•
|
general-use facsimile machines or computers to physicians that
are not exclusively used in connection with the laboratory
services; and
|
| |
| |
•
|
free testing for healthcare providers, their families and their
employees, known as professional courtesy testing.
|
The OIG emphasized in the Special Fraud Alert that when one
purpose of an arrangement is to induce referrals of
program-reimbursed laboratory testing, both the laboratory and
the healthcare provider or physician may be liable under the
Anti-Kickback Law and may be subject to criminal prosecution and
exclusion from participation in the Medicare and Medicaid
programs.
The OIG has also expressed concern about the provision of
discounts on laboratory services billed to customers in return
for the referral of more lucrative federal healthcare program
business. In a 1999 Advisory Opinion, the OIG concluded that a
proposed arrangement whereby a laboratory would offer physicians
significant discounts on non-federal healthcare program
laboratory tests might violate the Anti-Kickback Law. The OIG
reasoned that the laboratory could be viewed as providing such
discounts to the physician in exchange for referrals by the
physician of business to be billed by the laboratory to Medicare
at non-discounted rates. The OIG indicated that the arrangement
would not qualify for protection under the discount safe harbor
because Medicare and Medicaid would not get the benefit of the
discount. Subsequently, in 2000 correspondence, the OIG stated
that the Anti-Kickback Law could be violated if there were
linkage between the discount offered to the physician and the
physician’s referrals of tests covered under a federal
healthcare program that would be billed by the laboratory
directly. Where there was evidence of such linkage, the
arrangement would be considered suspect if the charge to the
physician was below the laboratory’s “average fully
loaded costs” of the test.
101
As discussed above, discounts to referral sources raise issues
under the Anti-Kickback Law. In addition, any discounted charge
below the amount that Medicare or Medicaid would pay for a
service also raises issues under Medicare’s
“substantially in excess” provision. The Medicare
statute permits the government to exclude a laboratory from
participation in federal healthcare programs if it charges
Medicare or Medicaid substantially in excess of its usual
charges in the absence of good cause. In 2000, the OIG stated in
informal correspondence that the prohibition was violated only
if the laboratory’s charge to Medicare was substantially
more than the “median non-Medicare/-Medicaid charge.”
On September 15, 2003, the OIG issued a notice of proposed
rulemaking addressing the statutory prohibition. Under the
proposed rule, a provider’s charge to Medicare or Medicaid
would be considered “substantially in excess of [its] usual
charges” if it was more than 120 percent of the
provider’s mean or median charge for the service. The
proposed rule was withdrawn in June 2007. At that time, the OIG
stated that it would continue to evaluate billing patterns of
individuals and entities on a
case-by-case
basis.
In addition to the administrative simplification regulations
discussed above, HIPAA created two new federal crimes:
healthcare fraud and false statements relating to healthcare
matters. The healthcare fraud statute prohibits knowingly and
willfully executing a scheme to defraud any healthcare benefit
program, including private payors. A violation of this statute
is a felony and may result in fines, imprisonment or exclusion
from governmental payor programs such as the Medicare and
Medicaid programs. The false statements statute prohibits
knowingly and willfully falsifying, concealing or covering up a
material fact or making any materially false, fictitious or
fraudulent statement in connection with the delivery of or
payment for healthcare benefits, items or services. A violation
of either of these two statutes is a felony and may result in
fines or imprisonment or exclusion from governmental payor
programs.
Finally, another development affecting the healthcare industry
is the increased use of the False Claims Act and, in particular,
actions brought pursuant to the False Claims Act’s
“whistleblower” or “qui tam” provisions. The
False Claims Act imposes liability on any person or entity that,
among other things, knowingly presents, or causes to be
presented, a false or fraudulent claim for payment by a federal
governmental payor program. Under the False Claims Act, a person
acts knowingly if he has actual knowledge of the information,
acts in deliberate ignorance of the truth or falsity of the
information, or acts in reckless disregard of the truth or
falsity of the information, A claim may be considered false if
any information included on the claim is inaccurate, including
the code reflecting the patient’s diagnosis or the
procedure performed. Similarly, if the service for which the
claim was submitted does not reflect the service that was
ordered and performed, the claim could be considered a false
claim. The qui tam provisions of the False Claims Act allow a
private individual to bring actions on behalf of the federal
government, alleging that the defendant has defrauded the
federal government by submitting a false claim to the federal
government, and they permit such individuals to share in any
amounts paid by the entity to the government in fines or
settlement. In addition, various states have enacted false claim
laws analogous to the federal False Claims Act, although many of
these state laws apply where a claim is submitted to any third
party payor and not merely a governmental payor program. When an
entity is determined to have violated the False Claims Act, it
may be required to pay up to three times the actual damages
sustained by the government, plus civil penalties of between
$5,500 and $11,000 for each separate false claim. Conduct that
violates the False Claims Act may also lead to exclusion from
the Medicare and Medicaid programs. Given the number of claims
likely to be at issue in connection with any billing practice on
which liability might be based, potential damages under the
False Claims Act for even a single inappropriate billing
arrangement could be significant.
On May 20, 2009, the Federal Enforcement and Recovery Act
of 2009, or FERA, became law, significantly amending the federal
False Claims Act. FERA subjects entities to False Claims Act
liability if they knowingly make a false statement in an attempt
to obtain money or property that is to be spent or used on the
government’s behalf or to advance a government program or
interest. Previously, the False Claims Act allowed entities to
be held liable for making false statements only if such
statements were made to “get” a false claim paid or
approved “by the Government.” Because no guidance
exists as to the meaning of the language contained in FERA, the
full scope of this provision will likely be resolved in
case-by-case
litigation. The False Claims Act amendments under FERA also
extend liability for the knowing retention of overpayments. The
overpayment provision imposes liability if an entity
“knowingly and improperly avoids or decreases an obligation
to pay or transmit money or property to the Government.”
This provision has been interpreted to require recipients of
government funds to immediately determine whether they have
received “overpayments” and whether the funds received
should be retained or
102
returned to the government. Under the False Claims Act
amendments, recipients of government funds may also now face
False Claims Act liability for failure to repay, or at least to
reassess, historical overpayments of which they are currently
aware. The enactment of FERA will likely result in heightened
scrutiny and enforcement.
Physician
Referral Prohibitions
Under a federal law directed at “self-referral,”
commonly known as the “Stark Law,” there are
prohibitions, with certain exceptions, on Medicare and Medicaid
payments for laboratory tests referred by physicians who
personally, or through a family member, have an investment
interest in, or a compensation arrangement with, the laboratory
performing the tests. A person who engages in a scheme to
circumvent the Stark Law’s referral prohibition may be
fined up to $100,000 for each such arrangement or scheme. In
addition, any person who presents or causes to be presented a
claim to the Medicare or Medicaid program in violation of the
Stark Law is subject to civil monetary penalties of up to
$15,000 per bill submission, an assessment of up to three times
the amount claimed, and possible exclusion from participation in
federal governmental payor programs. Bills submitted in
violation of the Stark Law may not be paid by Medicare or
Medicaid, and any person collecting any amounts with respect to
any such prohibited bill is obligated to refund such amounts.
Many states also have anti-“self-referral” and other
laws that are not limited to Medicare and Medicaid referrals.
The Stark Law prohibits a laboratory from obtaining payment for
services resulting from a physician’s referrals if there is
any arrangement between the laboratory and the physician or the
physicians’ practice that involves remuneration, unless the
arrangement is protected by an exception to the Stark Law’s
self-referral prohibition or a provision of the Stark Law
stating that the particular arrangement would not result in
“remuneration,” as defined for purposes of the Stark
Law. In addition, under the Stark Law’s provisions, a
request by a pathologist for laboratory and pathology testing
services is not considered a “referral” covered by the
Stark Law, so long as the request meets certain other
requirements.
Other
Government Billing and Payment Policies
Many payors establish specific rules and requirements that
affect how we bill for our services. The most significant of
those payors is the federal Medicare program, because it
accounts for a substantial percentage of our revenues.
Therefore, we must closely follow the policies that are imposed
by Medicare with regard to billing and payment.
Medicare has several ways in which it can limit how it pays for
our services. For example, Medicare can impose either National
Coverage Decisions (NCD) or Local Coverage Decisions
(LCD) for the services that we provide. Under NCDs and
LCDs, Medicare can impose imitations on when it will pay for
certain services, such as requiring particular diagnosis codes
to be submitted from the physician or limiting the number of
units of any particular service. Recently, the Medicare
Administrative Contractors, or MACs, which process and pay
Medicare claims, have considered some LCDs that could limit how
we bill for our services. For example, Palmetto GBA, which
processes Medicare claims in several jurisdictions, has limited
how it reimburses for flow cytometry services, which constitute
a commonly performed pathology service. If that policy is
implemented, or if other MACs adopt a similar policy, then it
could limit the payment we receive for these services.
In addition, Medicare has implemented a policy referred to as
Medically Unbelievable Edits, which limit the number of units of
a service that can be billed at any one time. Some Medically
Unbelievable Edits have already been established for the
services that we offer, and Medicare is considering adding
Medically Unbelievable Edits for some other services that we
offer. Under these edits, Medicare will not pay us for any
services, if we bill for more units of service than are
permitted under the Medically Unbelievable Edits limit
established by Medicare. This could affect the amount that we
are paid for services that we bill to the Medicare Program.
Medicare also regulates who we are permitted to bill for our
services. In most instances, we must bill directly to, and are
paid by, Medicare or its contractors for the services that we
furnish to Medicare beneficiaries. However, because of recent
interpretations by Medicare, ordering physicians are permitted
to order diagnostic services, such as those that we provide,
which the service provider then bills to the physician or his
group practice. The ordering physician, in turn, then bills and
is paid by the Medicare Program. In response to these
arrangements, Medicare has enacted an “anti-markup”
provision that requires physicians to bill Medicare at their
acquisition price for the
103
services, rather than at the higher Medicare fee schedule
amount. However, even under the anti-markup provision,
physicians are permitted to bill Medicare at the full fee
schedule amount if the services were performed by a physician
who “shares a practice” with the ordering physician.
The services will be considered to have been done by a physician
who shares a practice with the ordering physician, so long as
they were done in the same building where the ordering physician
maintains a practice. This interpretation of the anti-markup
provision, as well as other provisions, may lead more physicians
to decide to provide these services “in house,” which
could affect our revenues.
Under Medicare regulations, we are also sometimes required to
bill other entities for the services that we provide. In 1999,
Medicare announced a policy that applies to anatomic pathology
specimens for hospital patients. This policy would require us to
bill the technical component to the hospital and the
professional component to Medicare for all anatomic pathology
services that we provide to hospital patients. However, in 2000,
the U.S. Congress prevented this policy from going into
effect for all “covered hospitals,” which were those
hospitals that had arrangements with independent laboratories in
effect as of July 22, 1999, the date that CMS had first
announced the policy. That “grandfather provision” was
originally scheduled to be effective for two years, but it has
been extended repeatedly by the U.S. Congress and remains
in effect. The “grandfather provision” is currently
scheduled to expire on December 31, 2010.
Education
Requirements of the Deficit Reduction Act of 2005
The federal Deficit Reduction Act of 2005, or DRA, contains,
among other things, requirements concerning False Claims Act
education that state Medicaid programs must impose on
participating providers as well as other entities. The DRA
requires organizations that make or receive Medicaid payments of
at least $5 million annually in a state to establish an
education plan for their employees, managers, contractors and
agents, which must include written policies and detailed
guidance on the federal False Claims Act, state false claims
laws, and the rights and protections afforded whistleblowers
under the False Claims Act and its state counterparts.
Food
and Drug Administration Regulation
Pursuant to its authority under the federal Food, Drug and
Cosmetic Act, or FDCA, the Food and Drug Administration, or FDA,
has regulatory responsibility over instruments, test kits,
reagents and other devices used to perform diagnostic testing by
laboratories such as ours. Specifically, the manufacturers and
suppliers of reagents, which we obtain for use in diagnostic
tests, are subject to regulation by the FDA and are required to,
among other things, register their establishments with the FDA,
to conform manufacturing operations to the FDA’s quality
system regulation, or QSR, and to comply with certain reporting
and other record-keeping requirements. The FDA also regulates
the sale or distribution, in interstate commerce, of products
classified as medical devices under the FDCA, including
in vitro diagnostic test kits. Such devices must undergo
premarket review by the FDA prior to commercialization unless
the device is of a type exempted from such review by statute or
pursuant to the FDA’s exercise of enforcement discretion.
The FDA has not usually attempted to regulate standard
laboratory diagnostic tests developed and validated by
laboratories for their own use, known as laboratory-developed
tests or LDTs; rather, it has stated it will exercise its
enforcement discretion over such tests. However, more recently,
the FDA has expressed some concern about the use of certain
LDTs, including new, sophisticated molecular diagnostics or
genetic testing, and has suggested greater FDA oversight of such
tests may be appropriate. One of our laboratories uses some LDTs
and, therefore, some of the tests that we offer could be subject
to the FDA requirements if the FDA were to act in this area. The
FDA regularly considers the application of additional regulatory
controls over the development and use of LDTs by laboratories
such as ours, and with the recent change in political
administration at the FDA, more robust enforcement, (including
inspections, warning letters, and other agency inquiries) of the
applicable pre- and post-market requirements for LDTs is
possible.
The comprehensive health care reform legislation enacted in 2010
contains a provision that creates a new 2.3 percent excise
tax applicable to the sale of medical devices. Because we
purchase and utilize certain products in our laboratory that
could be considered medical devices, it is possible that some of
these products could be subject
104
to this excise tax. Because this legislation is new and
insufficient guidance exists as to its applicability, we are
currently unable to determine the impact this provision will
have on our business.
State
Requirements
Corporate
Practice of Medicine
Numerous states have enacted laws prohibiting business
corporations, such as us, from practicing medicine and employing
or engaging physicians to practice medicine. These laws are
designed to prevent interference in the medical decision-making
process by anyone who is not a licensed physician. This
prohibition is generally referred to as the prohibition against
the corporate practice of medicine. Violation of this
prohibition may result in civil or criminal fines, as well as
sanctions imposed against us
and/or the
professional through licensing proceedings. We do not employ
physicians in any states where the prohibition against the
corporate practice of medicine applies to our laboratories. Our
arrangements with providers in corporate practice of medicine
states are described above under “— Corporate
Structure” and “— Contracts and
Relationships with Providers.”
Direct
Billing Laws
While we often cannot bill physicians for our services when
those services are covered under a government program, where
permissible, we do in some cases bill referring physicians for
services that are not covered under a government program. Laws
and regulations in several states currently preclude us from
billing referring physicians, either by requiring us to bill
directly the third-party payor or other person ultimately
responsible for the service or by prohibiting or limiting the
referring physician’s or other purchaser’s ability to
“markup” its acquisition cost for that service. An
increase in the number of states that impose similar
restrictions could adversely affect us by encouraging physicians
to furnish such services directly or by causing physicians to
refer services to another laboratory for testing.
State
Laboratory Licensing
In addition to CLIA requirements, we are subject to various
state laws regulating the operation of our laboratories, as well
as the receipt by our laboratories of
out-of-state
specimens. If a laboratory is out of compliance with a
state’s statutory or regulatory standards, the applicable
state agency may suspend, limit, revoke or annul the
laboratory’s license, censure the holder of the license or
assess civil money penalties. In certain instances, statutory or
regulatory noncompliance may also result in a laboratory’s
being found guilty of a misdemeanor.
Each of Florida, New York, Alabama, New Hampshire, Nevada,
Massachusetts and Georgia require in-state laboratories to be
licensed under the laws and regulations of such states. Because
New York State’s regulation of laboratories is more
stringent than the federal CLIA standards, New York State is
exempt from CLIA. However, New York State maintains its own
regulatory framework, which establishes standards for
day-to-day
operation of a clinical laboratory, physical facilities
requirements, equipment and quality control. New York law also
mandates proficiency testing for laboratories licensed under New
York state law, regardless of whether or not such laboratories
are located in New York. Each of our laboratories is licensed
and accredited by the appropriate state agency in the state in
which it operates.
Other
States’ Laboratory Testing
Florida, New York, Pennsylvania, California and Maryland each
require
out-of-state
laboratories that accept specimens from those states to be
licensed. We have obtained licenses in these states and believe
we are in material compliance with applicable licensing laws.
We may become aware from time to time of other states that
require
out-of-state
laboratories to obtain licensure in order to accept specimens
from the state, and it is possible that other states do have
such requirements or will have such requirements in the future.
If we identify any other state with such requirements or if we
are contacted by any other state advising us of such
requirements, we intend to follow instructions from the state
regulators as to how we should comply with such requirements.
105
Physician
Licensing
Many of the states from which we solicit specimens require that
a physician interpreting specimens from that state be licensed
by that particular state, irrespective of where the services are
provided. If our pathologists read specimens from states for
which they are not properly licensed, our pathologists could be
considered to be engaged in the unlicensed practice of medicine
and could face fines or other penalties, which we could be
required to pay on behalf of our pathologists. We are not aware
of any states from which we do not have required pathologist
licenses.
Other
Regulatory Requirements
Our laboratory is subject to federal, state and local
regulations relating to the handling and disposal of regulated
medical waste, hazardous waste and biohazardous waste, including
chemical and biological agents and compounds, blood and bone
marrow samples and other human tissue. Typically, we use outside
vendors who are contractually obligated to comply with
applicable laws and regulations to dispose of such waste. These
vendors are licensed or otherwise qualified to handle and
dispose of such waste. Historically, our costs associated with
handling and disposal of such wastes have not been material.
The Occupational Safety and Health Administration has
established extensive requirements relating to workplace safety
for healthcare employers, including requirements to develop and
implement programs to protect workers from exposure to
blood-borne pathogens by preventing or minimizing any exposure
through needle stick or similar penetrating injuries. We have
instituted policies and procedures to comply with the OSHA
requirements, and we regularly monitor the implementation of our
safety programs.
Compliance
Infrastructure
Compliance with government rules and regulations is a
significant concern throughout our industry, in part due to
evolving interpretations of these rules and regulations. We seek
to conduct our business in compliance with all statutes and
regulations applicable to our operations. To this end, we have
created a Compliance Committee and have designated a Compliance
Officer to assist with reviews of regulatory compliance
procedures and policies throughout our business. Our executive
management team is responsible for the oversight and operation
of our compliance efforts. The Compliance Officer is responsible
for administering and monitoring compliance with our Standards
of Conduct. We provide periodic training programs to our
personnel to promote the observance of our policies, which are
designed to ensure compliance with the statutes and regulations
applicable to us.
Intellectual
Property
Our intellectual property consists primarily of trademarks and
trade secrets. The marks AURORA DIAGNOSTICS and CONNECTDX THE
INFORMATION GATEWAY and Design are our most recognizable
trademarks. Those trademarks are registered with the
U.S. Patent and Trademark Office, or USPTO, along with the
mark CUNNINGHAM PATHOLOGY ASSOCIATES P.A. and Design. We have
submitted applications to the USPTO for registration of the
marks DERMDX, GASTRODX, TREATMENTDX, URODX and WOMEN’SDX.
We maintain a program to protect our trademarks and will
institute legal action where necessary to prevent others from
using or registering confusingly similar trademarks. Our
intellectual property also includes the copyright in and to our
Tiger TCPC software, which is registered with the
U.S. Copyright Office.
Properties
and Facilities
We lease our corporate headquarters at 11025 RCA Center Drive,
Suite 300, Palm Beach Gardens, FL 33410 (approximately
8,500 square feet), and we lease 32 other facilities: 12 in
Florida, one in New York, two in Nevada, one in New Jersey, two
in Arizona, four in Michigan, one in New Hampshire, one in
Massachusetts, one in Minnesota, one in Texas, one in Georgia,
four in Alabama and one in North Carolina. These facilities are
used for laboratory operations, administrative, billing and
collections operations and storage space. The 32 facilities have
lease terms expiring from 2010 to 2019. We also own one
commercial condominium in Florida, which is used as a draw
station and satellite laboratory.
106
Insurance
We maintain liability insurance for our services. As a general
matter, providers of diagnostic services may be subject to
lawsuits alleging medical malpractice or other similar legal
claims. Some of these suits may involve claims for substantial
damages, and the results may be material to our results of
operations and cash flows in the period in which the impact of
such claims is determined or the claims are paid. We believe our
insurance coverage is sufficient to protect us from material
liability for such claims, and we believe that we will be able
to obtain adequate insurance coverage in the future at
acceptable costs. However, we must renew our insurance policies
annually, and we may not be able to maintain adequate liability
insurance in the future on acceptable terms or with adequate
coverage against potential liabilities or at all.
Employees
As of March 31, 2010, we had 986 employees. We employ
73 pathologists, 764 laboratory technicians and staff, 30
corporate office personnel and 103 sales, marketing and client
service personnel. In addition to our 73 employed pathologists,
we have contractual arrangements with a physician practice under
which 16 pathologists exclusively practice medicine as
independent contractors at our Alabama laboratory. None of our
employees are subject to collective bargaining agreements. We
consider our relationships with our employees to be good.
Legal
Proceedings
We are not currently a party to any material legal proceedings.
We may be named in other various claims, disputes, legal actions
and other proceedings involving malpractice, employment and
other matters. A negative outcome in certain of the ongoing
litigation could harm our business, financial condition,
liquidity or results of operations. Further, prolonged
litigation, regardless of which party prevails, could be costly,
divert management’s attention or result in increased costs
of doing business.
107
MANAGEMENT
AND BOARD OF DIRECTORS
The following table sets forth the name, age and position of
each of our executive officers and directors as of June 18,
2010.
| |
|
|
|
|
|
Name
|
|
Age
|
|
Position(s)
|
|
|
|
James C. New
|
|
65
|
|
Chairman of our Board of Directors, Chief Executive Officer and
President
|
|
Martin J. Stefanelli
|
|
49
|
|
Chief Operating Officer, Vice President and Secretary
|
|
Gregory A. Marsh
|
|
49
|
|
Chief Financial Officer, Vice President and Treasurer
|
|
Fred Ferrara
|
|
42
|
|
Chief Information Officer
|
|
Michael J. Null
|
|
40
|
|
Vice President, Sales and Marketing
|
|
Thomas S.
Roberts(1)
|
|
46
|
|
Director
|
|
Christopher
Dean(2)
|
|
36
|
|
Director
|
|
Peter J.
Connolly(3)
|
|
37
|
|
Director
|
|
Mark M.
King(2)
|
|
49
|
|
Director
|
|
Christopher J.
Bock(3)
|
|
40
|
|
Director
|
|
Blair
Tikker(1)
|
|
53
|
|
Director
|
|
James M.
Emanuel(4)
|
|
61
|
|
Director
|
|
|
|
|
(1)
|
|
Member of our Compensation
Committee.
|
|
|
|
|
(2)
|
|
Member of our Nominating and
Corporate Governance Committee.
|
|
|
|
|
(3)
|
|
Member of our Audit Committee.
|
|
|
|
|
(4)
|
|
Member of our Audit Committee,
Nominating and Corporate Governance Committee and Compensation
Committee.
|
| |
|
|
James C. New
Chairman,
Chief Executive Officer
and President
|
|
Mr. New has served as our Chairman, Chief Executive Officer and
President since 2006. Prior to joining us, Mr. New was a
private investor from 2003 to 2006. Mr. New served as the
President, Chief Executive Officer and Chairman of AmeriPath, an
anatomic pathology laboratory company, from January 1996 through
2003. Prior to joining AmeriPath, Mr. New served as the
President, Chief Executive Officer, and a director of
RehabClinics, an outpatient rehabilitation company. The Board
has concluded that Mr. New should serve as a director and our
Chairman because he is our Chief Executive Officer and
President. We and the Board benefit from his extensive
experience in managing anatomic pathology companies. In
addition, Mr. New’s substantial ownership interest in us
aligns his interests with those of our other shareholders.
|
Martin J. Stefanelli
Chief Operating Officer,
Vice President and Secretary
|
|
Mr. Stefanelli has served as our Chief Operating Officer, Vice
President and Secretary since 2006. Prior to joining us, Mr.
Stefanelli served as the President and Chief Operating Officer
of Asterand, a tissue-based research services provider for the
pharmaceutical and biotechnology industry, from 2004 to 2006.
Mr. Stefanelli served as the Executive Vice President and Chief
Operating Officer of AmeriPath, an anatomic pathology laboratory
company, from June 2003 to November 2004, and prior to joining
AmeriPath, Mr. Stefanelli was employed for thirteen years by
DIANON Systems, an anatomic and clinical pathology laboratory
company.
|
108
| |
|
|
Gregory A. Marsh
Chief Financial Officer,
Vice President and Treasurer
|
|
Mr. Marsh has served as our Chief Financial Officer, Vice
President and Treasurer since November 2007. Prior to joining
us, Mr. Marsh served as an executive officer at CardioNet and
PDSHeart, each a cardiovascular diagnostic healthcare provider.
He served as the Chief Financial Officer of PDSHeart from 2003
to 2005 and then Chief Operating Officer from 2005 until March
2007, when the company was acquired by CardioNet. Mr. Marsh
then served as the Chief Financial Officer of CardioNet until
November 2007. From 1996 until 2003, Mr. Marsh was employed by
AmeriPath, an anatomic pathology laboratory company, serving as
Vice President, Chief Financial Officer and Secretary from 2001
to 2003 and Vice President, Corporate Controller from 1996 to
2001.
|
|
|
|
|
Fred Ferrara
Chief Information Officer
|
|
Mr. Ferrara has served as our Chief Information Officer since
2006. Mr. Ferrara served as the Director of Information
Technology at LabCorp Inc., an anatomic pathology laboratory
company, from 2003 until he joined Aurora in 2006. Mr. Ferrara
joined LabCorp upon its acquisition of DIANON Systems, where Mr.
Ferrara had been employed since 1997.
|
|
|
|
|
Michael J. Null
Vice President, Sales
and Marketing
|
|
Mr. Null has served as our Vice President, Sales &
Marketing since April 2007. Prior to joining us, Mr. Null
served as the Vice President of Sales and Marketing at Asterand,
a tissue-based research services provider for the pharmaceutical
and biotechnology industry, from 2001 to 2007. He served as a
senior account manager and business development manager at
Renaissance, a global IT consulting and staffing company, from
1997 to 2002. Prior to joining Renaissance, Mr. Null was
employed for five years by DIANON Systems, an anatomic and
clinical pathology laboratory company.
|
|
|
|
|
Thomas S. Roberts
Director
|
|
Mr. Roberts has served as one of our directors since 2006 and
currently serves as a Managing Director of Summit Partners, a
growth equity firm. Mr. Roberts joined Summit Partners in
1989. Mr. Roberts also served in the past as the Chairman and
Director of AmeriPath, an anatomic pathology laboratory company.
The Board has concluded that Mr. Roberts should serve as a
director because of his significant executive experience as well
as the fact that his relationship with Summit Partners, which
has a substantial ownership interest in us, aligns his interests
with those of our other shareholders.
|
|
|
|
|
Christopher Dean
Director
|
|
Mr. Dean has served as one of our directors since 2006 and
currently serves as a Managing Director of Summit Partners, a
growth equity firm. Mr. Dean joined Summit Partners in 2001.
The Board has concluded that Mr. Dean should serve as a director
because of his significant executive experience as well as the
fact that his relationship with Summit Partners, which has a
substantial ownership interest in us, aligns his interests with
those of our other shareholders.
|
109
| |
|
|
Peter J. Connolly
Director
|
|
Mr. Connolly has served as one of our directors since 2006 and
currently serves as a Principal at Summit Partners, a growth
equity firm. Prior to joining Summit Partners in 2004, Mr.
Connolly was employed by Goldman, Sachs & Co., an
investment banking firm and Deloitte LLP, an accounting and
consulting firm. The Board has concluded that Mrs. Connolly
should serve as a director because of his significant executive
experience as well as the fact that his relationship with Summit
Partners, which has a substantial ownership interest in us,
aligns his interests with those of our other shareholders.
|
Mark M. King
Director
|
|
Mr. King has served as one of our directors since June 2009 and
is a Co-Founder and Managing Director of KRG Capital Partners, a
private equity investment firm. Prior to forming KRG Capital
Partners in 1996, Mr. King led many industry consolidating
transactions and co-founded and was President and Vice Chairman
of Industrial Services Technologies, Inc., a provider of
maintenance services in the refinery, fertilizer and chemicals
industries. The Board has concluded that Mr. King should serve
as a director because of his significant executive experience as
well as the fact that his relationship with KRG Capital
Partners, which has a substantial ownership interest in us,
aligns his interests with those of our other shareholders.
|
Christopher J. Bock
Director
|
|
Mr. Bock has served as one of our directors since June 2009 and
is a Managing Director of KRG Capital Partners, a private equity
investment firm. Mr. Bock joined KRG Capital Partners in
1997. The Board has concluded that Mr. Bock should serve as a
director because of his significant executive experience as well
as the fact that his relationship with KRG Capital Partners,
which has a substantial ownership interest in us, aligns his
interests with those of our other shareholders.
|
Blair Tikker
Director
|
|
Mr. Tikker has served as one of our directors since June 2009
and is a Managing Director of KRG Capital Partners, a private
equity investment firm. Mr. Tikker joined KRG Capital
Partners in 2007. Prior to joining KRG Capital Partners, Mr.
Tikker was employed by a number of hospital systems, physician
groups and managed care companies. Mr. Tikker served as the
CEO of HMS Healthcare, a hospital information systems provider,
from 2001 until 2005. The Board has concluded that Mr. Tikker
should serve as a director because of his significant executive
experience as well as the fact that his relationship with KRG
Capital Partners, which has a substantial ownership interest in
us, aligns his interests with those of our other shareholders.
|
James M. Emanuel
Director
|
|
Mr. Emanuel has served as one of our directors since May 2010.
Mr. Emanuel has engaged in consulting and private investment
activities since his retirement from Lincare, Inc., a national
provider of respiratory therapy services for patients with
pulmonary disorders, where he served as Chief Financial Officer
from January 1984 to June 1997. Mr. Emanuel also served as Chief
Financial Officer and a director of Lincare Holdings Inc. from
November 1990 to June 1997. Mr. Emanuel has served as a director
of SRI/Surgical Express Inc. since 1996 in addition to serving
on private company boards. The Board has concluded that Mr.
Emanuel should serve as a director because of his significant
executive experience.
|
110
Board
Composition and Election of Directors
Board
Composition
Our Board of Directors is currently composed of 8 members,
consisting of seven non-employee members and our current
Chairman, Chief Executive Officer and President, James C. New.
The number of directors on our Board of Directors will be
determined from time to time by resolution of our Board of
Directors.
To supplement the recent election of our sole independent
director Mr. Emanuel, we expect to elect one or more additional
independent directors not more than 90 days after the
completion of this offering and to elect additional independent
directors not more than one year after the completion of this
offering.
Under the NASDAQ Global Market standards, a director will only
qualify as an “independent director” if, in the
opinion of our Board of Directors, that person does not have a
relationship that would interfere with the exercise of
independent judgment in carrying out the responsibilities of a
director.
Our Board of Directors has determined that Mr. Emanuel is an
“independent director” as defined under the NASDAQ
Global Market standards.
There are no family relationships among any of our directors or
executive officers.
Election
and Classification of Directors
In accordance with the terms of our certificate of incorporation
and bylaws, our Board of Directors will be divided into three
classes, Class I, Class II and Class III, with
each class serving staggered three-year terms. Upon the closing
of this offering, the members of the classes will be divided as
follows:
|
|
|
| |
•
|
the Class I directors will be Mr. King,
Mr. Connolly and Mr. New, and their term will expire
at the annual meeting of stockholders to be held in 2011;
|
|
|
|
| |
•
|
the Class II directors will be Mr. Tikker,
Mr. Dean and Mr. Emanuel, and their term will expire
at the annual meeting of stockholders to be held in
2012; and
|
|
|
|
| |
•
|
the Class III directors will be Mr. Bock and
Mr. Roberts, and their term will expire at the annual
meeting of stockholders to be held in fiscal year 2013.
|
At each annual meeting of stockholders, or special meeting in
lieu thereof, upon the expiration of the term of a class of
directors, the successors to such directors will be elected to
serve from the time of his or her election and qualification
until the third annual meeting following his or her election or
special meeting held in lieu thereof. The number of directors
may be changed only by resolution of our Board of Directors or a
66.67 percent vote of the stockholders. Any additional
directorships resulting from an increase in the number of
directors will be distributed among the three classes so that,
as nearly as possible, each class will consist of one-third of
the directors. This classification of our Board of Directors may
have the effect of delaying or preventing changes in control of
management.
Board
Committees
Committees
of our Board of Directors
Our Board of Directors has established three committees prior to
the effectiveness of our registration statement:
|
|
|
| |
•
|
the audit committee;
|
| |
| |
•
|
the nominating and corporate governance committee; and
|
| |
| |
•
|
the compensation committee.
|
Although the NASDAQ Global Market standards require that all
members of our board committees and a majority our Board of
Directors be independent, under special phase-in rules
applicable to initial public offerings, we have twelve months
from the date of listing to comply with these requirements. We
currently expect to achieve compliance with the NASDAQ Global
Market majority board independence and committee independence
111
requirements by electing a new independent director prior to
completion of this offering and adding additional independent
directors to our Board of Directors before the expiration of the
phase-in period and, for affected committees, replacing
directors who are not independent with one or more of the new
independent directors we expect to elect. If we do not comply
with these requirements before the expiration of the phase-in
period, we will be subject to disciplinary sanctions by the
NASDAQ Global Market, which may include suspension of trading in
the common stock or delisting of the common stock from the
NASDAQ Global Market.
We believe that our audit and compensation committees will meet
the current requirements of the Sarbanes-Oxley Act of 2002 and
the NASDAQ Global Market standards as they become applicable to
us and that our nominating and corporate governance committee
will meet the current requirements of the NASDAQ Global Market
standards as they become applicable to us. The audit committee,
compensation committee and nominating and corporate governance
committee will adopt written charters that will be available on
our website prior to completion of the offering.
Audit Committee. Our audit committee’s
responsibilities include:
|
|
|
| |
•
|
selecting and engaging our independent registered public
accounting firm;
|
| |
| |
•
|
evaluating the qualifications, independence and performance of
our independent registered public accounting firm, including
through the receipt and consideration of reports from such firm;
|
| |
| |
•
|
approving (or, as permitted, pre-approving) the audit and
non-audit services to be performed by our independent registered
public accounting firm;
|
| |
| |
•
|
establishing policies regarding hiring employees from our
independent registered public accounting firm and procedures for
the receipt and retention of accounting related complaints and
concerns;
|
| |
| |
•
|
meeting independently with our independent registered public
accounting firm and management;
|
| |
| |
•
|
reviewing the design, implementation, adequacy and effectiveness
of our internal controls over financial reporting, our
disclosure controls and procedures and our critical accounting
policies;
|
| |
| |
•
|
reviewing and discussing with management and the independent
registered public accounting firm the results of our annual
audit and the review of our quarterly unaudited financial
statements;
|
| |
| |
•
|
reviewing, overseeing and monitoring the integrity of our
financial statements and our compliance with legal and
regulatory requirements as they relate to financial statements
or accounting matters;
|
| |
| |
•
|
reviewing with management and our auditors any earnings
announcements and other public announcements regarding our
results of operations;
|
| |
| |
•
|
preparing the audit committee report that the SEC requires in
our annual proxy statement;
|
| |
| |
•
|
reviewing and approving any related party transactions and
reviewing and monitoring compliance with our code of business
conduct and ethics; and
|
| |
| |
•
|
reviewing and evaluating, at least annually, the performance of
the audit committee and its members including compliance of the
audit committee with its charter.
|
The audit committee has the sole and direct responsibility for
appointing, evaluating and retaining our independent auditors
and for overseeing their work. All audit services and all
non-audit services, other than de minimis non-audit services, to
be provided to us by our independent auditors must be approved
in advance by our audit committee.
Our audit committee is comprised of Messrs. Emanuel,
Connolly and Bock. Mr. Emanuel is the chairperson of the audit
committee. Our Board of Directors has determined that
Mr. Emanuel is an independent director for audit committee
service as defined in
Rule 10A-3
under the Exchange Act. Mr. Emanuel has been designated as
the audit committee financial expert, as defined in
Item 407(d) of
Regulation S-K
under the Securities Act. Although the NASDAQ Global Market
standards require that the audit committee be comprised solely
of independent directors, under special phase-in rules
applicable to initial public offerings, we have twelve months
from the date of listing to
112
comply with this requirement. As described more fully under
“Committees of our Board of Directors” above, we
expect to be in compliance before the expiration of the phase-in
period.
Compensation Committee. Our compensation
committee’s responsibilities include:
|
|
|
| |
•
|
reviewing and recommending to our Board of Directors
compensation and benefit plans for our executive officers and
compensation policies for members of our Board of Directors and
Board committees;
|
| |
| |
•
|
reviewing the terms of offer letters and employment agreements
and arrangements with our officers;
|
| |
| |
•
|
setting performance goals for our officers and reviewing their
performance against these goals and setting compensation based
on such review;
|
| |
| |
•
|
evaluating the competitiveness of our executive compensation
plans and periodically reviewing executive succession plans;
|
| |
| |
•
|
administering our benefit plans and the issuance of stock
options and other awards under our equity incentive plans;
|
| |
| |
•
|
preparing the compensation committee report that the SEC
requires in our annual proxy statement;
|
| |
| |
•
|
reviewing and discussing annually with management our
“Compensation Discussion and Analysis” disclosure
required by SEC rules; and
|
| |
| |
•
|
reviewing and evaluating, at least annually, the performance of
the compensation committee and its members including compliance
of the compensation committee with its charter.
|
Our compensation committee is comprised of Messrs. Emanuel
Roberts and Tikker. Mr. Roberts is the chairperson of the
compensation committee. Although the NASDAQ Global Market
standards require that the compensation committee be comprised
solely of independent directors, under special phase-in rules
applicable to initial public offerings, we have twelve months
from the date of listing to comply with this requirement. As
described more fully under “Committees of our Board of
Directors” above, we expect to be in compliance before the
expiration of the phase-in period.
Nominating and Corporate Governance
Committee. Our nominating and corporate
governance committee responsibilities include:
|
|
|
| |
•
|
evaluating the composition, size and governance of our Board of
Directors and its committees and making recommendations
regarding future planning and the appointment of directors to
our committees;
|
| |
| |
•
|
overseeing an annual evaluation of management succession
planning;
|
| |
| |
•
|
administering a policy for considering stockholder nominees for
election to our Board of Directors;
|
| |
| |
•
|
evaluating and recommending candidates for election to our Board
of Directors;
|
| |
| |
•
|
overseeing our Board of Directors’ performance and
self-evaluation process;
|
| |
| |
•
|
reviewing our corporate governance principles and providing
recommendations to our Board of Directors regarding possible
changes; and
|
| |
| |
•
|
reviewing and evaluating, at least annually, the performance of
the nominating/corporate governance committee and its members
including compliance of the nominating/corporate governance
committee with its charter.
|
The nominating and corporate governance committee is also
responsible for reviewing developments in corporate governance
practices, evaluating the adequacy of our corporate governance
practices and reporting and making recommendations to our Board
of Directors concerning corporate governance matters.
Our nominating and corporate governance committee is comprised
of Messrs. Emanuel, Dean and King. Mr. Dean is
the chairperson of the nominating and corporate governance
committee. Although the NASDAQ Global Market standards require
that the nominating and corporate governance committee be
comprised solely of independent directors, under special
phase-in rules applicable to initial public offerings, we have
twelve months
113
from the date of listing to comply with this requirement. As
described more fully under “Committees of our Board of
Directors” above, we expect to be in compliance before the
expiration of the phase-in period.
Compensation
Committee Interlocks and Insider Participation
During 2008 and 2009, we did not have a compensation committee.
Following the completion of this offering, none of our
compensation committee members will be an officer or employed by
us.
Code of
Business Conduct and Ethics
Our Board of Directors has adopted procedures and policies to
comply with the Sarbanes-Oxley Act of 2002 and the rules adopted
by the SEC and the NASDAQ Global Market, including a code of
business conduct and ethics applicable to our officers,
directors and employees. Upon completion of this offering, our
code of conduct and ethics will be available on our website.
Indemnification
of Officers and Directors and Limitation of Liability
Our certificate of incorporation and bylaws limit the liability
of our directors for monetary damages for breach of their
fiduciary duties, except for liability that cannot be eliminated
under the Delaware General Corporation Law. Delaware law
provides that directors of a corporation will not be personally
liable to the corporation or its stockholders for monetary
damages for breach of their fiduciary duties as directors,
except liability for any of the following:
|
|
|
| |
•
|
any breach of their duty of loyalty to the corporation or its
stockholders;
|
| |
| |
•
|
acts or omissions not in good faith or that involve intentional
misconduct or a knowing violation of law;
|
| |
| |
•
|
voting or assenting to unlawful payments of dividends or
unlawful stock repurchases or redemptions as provided in
Section 174 of the Delaware General Corporation Law; or
|
| |
| |
•
|
any transaction from which the director derived an improper
personal benefit.
|
This limitation of liability does not apply to liabilities
arising under the federal securities laws and does not affect
the availability of equitable remedies such as injunctive relief
or rescission.
Our certificate of incorporation and our bylaws provide that we
shall indemnify our directors and executive officers and may
indemnify our other officers and employees and other agents to
the fullest extent permitted by law, as it now exists or may in
the future be amended, against all expenses and liabilities
reasonably incurred in connection with their service for or on
behalf of us. We believe that indemnification under our bylaws
covers at least negligence and gross negligence on the part of
indemnified parties. If the Delaware General Corporation Law is
amended to provide for further limitations on the personal
liability of directors of corporations, then the personal
liability of our directors will be further limited to the
greatest extent permitted by the Delaware General Corporation
Law. Any amendment to or repeal of the provisions of our bylaws
or certificate of incorporation will not eliminate or reduce the
effect of these provisions in respect of any act or failure to
act, or any cause of action, suit or claim that would accrue or
arise, prior to any amendment or repeal or adoption of an
inconsistent provision.
Our bylaws also permit us to secure insurance on behalf of any
officer, director, employee or other agent for any liability
arising out of his or her actions in this capacity, regardless
of whether our bylaws would permit indemnification. We maintain
liability insurance that insures our directors and officers
against losses and that insures us against our obligations to
indemnify our directors and officers.
In connection with this offering, we expect to enter into
separate indemnification agreements with our directors and
executive officers, in addition to indemnification provided for
in our certificate of incorporation and bylaws. These
agreements, among other things, will provide for indemnification
of our directors and executive officers for expenses, judgments,
fines and settlement amounts incurred by this person in any
action or proceeding arising out of this person’s services
as a director or executive officer or at our request. We believe
that these provisions and agreements are necessary to attract
and retain qualified persons as directors and executive officers.
114
EXECUTIVE
COMPENSATION
COMPENSATION
DISCUSSION AND ANALYSIS
In the paragraphs that follow, we provide an overview and
analysis of our compensation program and policies, the material
compensation decisions we have made under those programs and
policies with respect to our named executive officers, and the
material factors that we considered in making those decisions.
Following this Compensation Discussion and Analysis, you will
find a series of tables containing specific data about the
compensation earned or paid in fiscal year 2009 to the following
individuals, whom we refer to as our named executive officers:
|
|
|
| |
•
|
James C. New, Chairman of our Board of Directors, and our Chief
Executive Officer and President;
|
| |
| |
•
|
Gregory A. Marsh, our Chief Financial Officer, Vice President
and Treasurer;
|
| |
| |
•
|
Fred Ferrara, our Chief Information Officer;
|
| |
| |
•
|
Michael J. Null, our Vice President, Sales and
Marketing; and
|
| |
| |
•
|
Martin J. Stefanelli, our Chief Operating Officer, Vice
President and Secretary.
|
Objectives
of our Compensation Program; How We Set Compensation
Our compensation objectives as a privately-held company were to
recruit and retain a talented team of employees to grow and
develop our business and to reward those employees for
accomplishments related to our growth and development.
Historically, we did not have a compensation committee and our
Board of Directors determined the compensation for our Chief
Executive Officer and, based on the recommendations of our Chief
Executive Officer, the rest of our management team. In setting
compensation, Mr. New and our Board of Directors did not
seek to allocate long-term and current compensation, or cash and
non-cash compensation, in any particular percentage. Instead,
they reviewed each element of compensation independently and
determined the appropriate amount for each element, as discussed
below. Neither management nor our Board of Directors engaged a
compensation consultant during fiscal year 2009. Our historical
compensation-setting processes have been effective for a
privately-held company. As we transition to a public company, we
intend to adjust some of our practices to further align the
interests of our management team with our stockholders.
In anticipation of our initial public offering, we have
established a compensation committee. The compensation
committee, with input from our Chief Executive Officer, will set
and determine the compensation of our executive officers in
later years. In this regard, our compensation committee will,
among other things, review and recommend to our Board of
Directors compensation and benefit plans for our executive
officers, review the terms of employment agreements and
arrangements with our officers, set performance goals for our
officers, review their performance against these goals and set
compensation based on such review, evaluate the competitiveness
of our executive compensation plans, periodically review
executive succession plans, and administer our benefit plans and
the issuance of stock options and other awards under our equity
incentive plans. The compensation committee will have sole
authority to evaluate our Chief Executive Officer’s
performance in light of corporate objectives and to set our
Chief Executive Officer’s compensation based on the
achievement of corporate objectives. Within twelve months from
the date of our listing on the NASDAQ Global Market, each member
of the compensation committee will be “independent”
for purposes of the NASDAQ Global Market standards, a
“non-employee director” for purposes of
Rule 16b-3
of the Exchange Act, and an “outside director” for
purposes of Section 162(m) of the Internal Revenue Code.
2009
Elements of Compensation
The key elements of compensation for our named executive
officers in fiscal year 2009 were base salary and annual cash
bonuses. We also are party to an employment agreement with each
of Messrs. New, Marsh, Ferrara, Null and Stefanelli. These
agreements provide the executive with certain benefits as
described in greater detail below.
115
Base Salaries. We intend for base salaries to
reward core competence in the executive role relative to skill,
experience and contributions to us. We negotiated the base
salaries individually with each executive, with a focus on the
executive’s experience in his respective field and expected
contribution to us. In general, we adjust base salaries in
connection with performance reviews
and/or
changes to the scope of a named executive officer’s
responsibilities. In 2009, none of our named executive officers
received salary increases over 2008 levels. The 2009 annual base
salaries for our named executive officers were as follows:
Mr. New, $400,000; Mr. Marsh, $294,000;
Mr. Ferrara, $222,600; Mr. Null, $225,750; and
Mr. Stefanelli, $315,000. The named executive officers have
not received base salary increases in 2010.
Annual Cash Bonuses. Annual bonuses reward our
named executive officers for their contribution to our financial
goals and focus our named executive officers on both short- and
long-term objectives. Annual bonuses are earned based on the
achievement of certain pre-determined performance goals. On an
annual basis, or at the commencement of an executive
officer’s employment with us, our Board of Directors set a
target level of bonus compensation that is structured as a
percentage of such executive officer’s annual base salary.
The target bonuses for each of our executive officers are as
follows, reflected as a percentage of base salary: Mr. New,
100 percent; Mr. Marsh; 50 percent;
Mr. Ferrara, 40 percent; Mr. Null,
35 percent; and Mr. Stefanelli,75 percent. Our
Board of Directors set such target bonuses after negotiation
with each individual and consideration of Mr. New’s
recommendation and the expected role of each of our executives.
The actual amount of the bonus is based on the extent to which
we and the executive meet or exceed predetermined goals under
the performance metrics, which goals are set by our Board of
Directors prior to the beginning of the performance year in
connection with the annual budgetary process, as discussed below.
For 2009, annual cash incentive bonus opportunities for our
named executive officers were based on achieving pre-established
performance goals relating to net revenue, EBITDA, cash flow
from operations, management of acquisition programs (measured by
the acquisition of companies with annualized EBITDA at
pre-determined levels), and management of our covenants in
connection with our credit facilities. Each of these performance
metrics is an important driver of our business and our rationale
for selecting these performance metrics is as follows:
|
|
|
| |
•
|
We select net revenue and EBITDA to focus the executive on
supporting, improving and growing our business;
|
|
|
|
| |
•
|
Cash from operations is a direct measure of our profitability
and financial performance;
|
|
|
|
| |
•
|
Acquisitions of companies with annualized EBITDA at
pre-determined levels focuses the executive on identifying
acquisitions which meet our financial goals and positively
affect our covenant position; and
|
|
|
|
| |
•
|
As discussed elsewhere in this prospectus, our agreement and the
related instruments governing borrowings under our new credit
facilities contain various restrictive covenants that, among
other things, require us to comply with or maintain certain
financial tests and ratios, as well as certain additional
restrictions. Any breach of covenants in our new credit
facilities could result in a default under our new credit
agreement. Accordingly, maintenance of these covenants is
important to us.
|
116
From the above list, our Board of Directors assigned certain
objectives to each named executive officer based on his role
with us, and assigned a weight to each metric, as reflected in
the table below.
| |
|
|
|
Name
|
|
Performance Objective and
Weight
|
|
|
|
Mr. New
|
|
• Net revenue (15%)
|
|
|
|
• EBITDA (50%)
|
|
|
|
• Acquisition Management (10%)
|
|
|
|
• Management of Bank Covenants (25%)
|
|
Mr. Stefanelli
|
|
• Net revenue (15%)
|
|
|
|
• EBITDA (50%)
|
|
|
|
• Acquisition Management (10%)
|
|
|
|
• Management of Bank Covenants (25%)
|
|
Mr. Marsh
|
|
• Net revenue (10%)
|
|
|
|
• Cash flow from Operations (60%)
|
|
|
|
• Acquisition Management (5%)
|
|
|
|
• Management of Bank Covenants (25%)
|
|
Mr. Ferrara
|
|
• Net revenue (50%)
|
|
|
|
• EBITDA (40%)
|
|
|
|
• Cash flow from Operations (10%)
|
|
Mr. Null
|
|
• Net revenue (50%)
|
|
|
|
• EBITDA (40%)
|
|
|
|
• Cash flow from Operations (10%)
|
The following table provides the targets (in millions) for each
of the performance objectives and actual performance in fiscal
year 2009.
| |
|
|
|
|
|
Objective
|
|
Target Goal
|
|
FY 2009 Actual
|
|
|
|
Net Revenue
|
|
$173.8
|
|
$170.9
|
|
EBITDA
|
|
$57.2
|
|
$55.8
|
|
Cash flow from Operations
|
|
$30.6
|
|
$33.4
|
|
Acquisition Management
|
|
Targets’ Annualized
EBITDA of $4.0
|
|
Targets’ Annualized
EBITDA of $6.4
|
|
Management of
covenants(1)
|
|
N/A
|
|
100%
|
|
|
|
|
(1)
|
|
The achievement of the
“Management of Covenants” objective is generally
measured by whether or not we breached any of the covenants
under our credit facilities.
|
With the exception of the “Management of Covenants”
objective, the achievement of which is either zero percent or
one hundred percent, for each increment of one percent that
actual performance falls below our target goal, the
executive’s target bonus would be reduced by ten percent.
Likewise, for each increment of one percent that actual
performance exceeds our target goal, the executive’s target
bonus would be increased by ten percent, with a maximum of
110 percent. For example, if we had achieved
90 percent of our target goal, then the executive would
have received zero percent of his target bonus, and if we had
achieved 110 percent of our target goal, then the executive
would have received two hundred percent of his target bonus.
Linear interpolation is used to determine
117
payouts between the ranges. The following table provides details
regarding the awards earned by named executive officers in 2009.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Target
|
|
Target Bonus
|
|
Actual Bonus Earned
|
|
Actual Bonus Earned
|
|
Name
|
|
Bonus ($)
|
|
(% of Base Salary)
|
|
($)
|
|
(% of Base Salary)
|
|
|
|
Mr. New
|
|
$
|
400,000
|
|
|
|
100
|
%
|
|
$
|
381,000
|
|
|
|
95
|
%
|
|
Mr. Stefanelli
|
|
$
|
236,250
|
|
|
|
75
|
%
|
|
$
|
225,000
|
|
|
|
71
|
%
|
|
Mr. Marsh
|
|
$
|
147,000
|
|
|
|
50
|
%
|
|
$
|
233,000
|
|
|
|
79
|
%
|
|
Mr. Ferrara
|
|
$
|
89,040
|
|
|
|
40
|
%
|
|
$
|
81,000
|
|
|
|
36
|
%
|
|
Mr. Null
|
|
$
|
79,013
|
|
|
|
35
|
%
|
|
$
|
72,000
|
|
|
|
32
|
%
|
In addition to the bonus described above, in June 2009,
Mr. Marsh received a $10,000 cash bonus in recognition of
his role in connection with KRG Capital Partners’
investment in Aurora Holdings and their acquisition of GSO
Capital Partners’ equity interest in Aurora Holdings. This
bonus was not based on any pre-established performance metrics.
Instead, the cash award was determined by our Board of Directors
on a discretionary basis after the closing of the transaction,
based on Mr. Marsh’s performance in connection with,
and the success of, the transaction.
Long-Term Equity Incentives. In prior years
(most recently 2008), our Board of Directors granted certain
equity interests in Aurora Holdings to our named executive
officers pursuant to our New Plan. The New Plan provided for
grants of
Class D-1,
Class D-2
and
Class D-3
Units, which we refer to collectively as the Class D Units,
to our employees. The terms of the Class D Units were
governed by the Aurora Holdings LLC Agreement, and, generally,
the Class D Units represent the right of the holder to
receive a portion of the proceeds in the event of liquidation
of Aurora Holdings. The Class D units generally vested over
a period of time, subject to the named executive officer’s
continued employment with us. All of such units held by our
named executive officers were fully-vested by December 31,
2008. None of our named executive officers received Class D
units, or any other equity grants, in 2009. In connection with
the Reorganization Transactions discussed elsewhere in this
prospectus, the Class D Units held by our named executive
officers will be exchanged for shares of our Class A common
stock or cancelled without consideration.
Other Benefits. Our named executive officers
participate in various health and welfare programs that are
generally made available to all salaried employees. Our named
executive officers also participate in our executive-level life
insurance program. Mr. New receives reimbursements related
to his country club memberships, as well as Company-paid
premiums for an individual life insurance policy.
Employment
Arrangements
We maintain employment agreements with each of Messrs. New,
Marsh, Ferrara, Null and Stefanelli. The employment agreements
guarantee certain benefits, such as bonus and benefit plans, to
the executives during their employment with us. In addition, the
employment agreements provide certain benefits to the executives
upon their termination of employment by us without cause, by the
executive for good reason, or by reason of their death or
disability. For a description of the employment agreements, see
the narrative following the Summary Compensation table and
“Potential Payments upon Termination of Employment”
and “Potential Payments upon Change in Control” later
in this prospectus.
Tax
Treatment of our Compensation Program
Section 162(m) of the Internal Revenue Code places a limit
of $1 million on the amount of compensation that public
companies may deduct in any one year with respect to its named
executive officers. In fiscal year 2009, as a privately-held
company, Section 162(m) of the Internal Revenue Code did
not apply to us. To the extent that we compensate our named
executive officers in excess of the $1 million limit in the
future, our compensation committee will consider whether we
should design our compensation programs to meet the qualified
performance-based requirements. To maintain flexibility in
compensating our executives, we expect that the compensation
committee will reserve the right to use its judgment to
authorize compensation payments that may be subject to the limit
when the compensation committee believes that such payments are
appropriate.
118
Summary
Compensation
The following table sets forth the cash and other compensation
that we paid to our named executive officers, or that was
otherwise earned by our named executive officers, for their
services in all capacities during the last fiscal year.
Summary
Compensation Table
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Non-Equity
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Incentive Plan
|
|
|
All Other
|
|
|
|
|
|
Name and Principal
Position
|
|
Year
|
|
|
Salary ($)
|
|
|
Bonus($)
|
|
|
Compensation
($)(1)
|
|
|
Compensation
($)(2)
|
|
|
Total ($)
|
|
|
|
|
James C. New
|
|
|
2009
|
|
|
|
400,000
|
|
|
|
—
|
|
|
|
381,000
|
|
|
|
45,154
|
|
|
|
826,154
|
|
|
Chairman of our Board of Directors, Chief Executive Officer and
President
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Martin J. Stefanelli
|
|
|
2009
|
|
|
|
315,000
|
|
|
|
—
|
|
|
|
225,000
|
|
|
|
630
|
|
|
|
540,630
|
|
|
Chief Operating Officer, Vice President and Secretary
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Gregory A. Marsh
|
|
|
2009
|
|
|
|
294,000
|
|
|
|
10,000(3
|
)
|
|
|
233,000
|
|
|
|
630
|
|
|
|
537,630
|
|
|
Chief Financial Officer, Vice President and Treasurer
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Fred Ferrara
|
|
|
2009
|
|
|
|
222,600
|
|
|
|
—
|
|
|
|
81,000
|
|
|
|
287
|
|
|
|
303,887
|
|
|
Chief Information Officer
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Michael J. Null
|
|
|
2009
|
|
|
|
225,750
|
|
|
|
—
|
|
|
|
71,915
|
|
|
|
84,261
|
|
|
|
381,926
|
|
|
Vice President, Sales and Marketing
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1)
|
|
Reflects the dollar amount of
annual performance-based bonuses earned by our named executive
officers in 2009.
|
|
|
|
|
(2)
|
|
Reflects premiums paid in
accordance with the executive-level life insurance plan. Also
includes (i) for Mr. New, reimbursement of $19,087
related to country club memberships and $23,295 in premiums paid
for a separate life insurance policy; and (ii) for
Mr. Null, $83,986 relating to our forgiveness of a note
extended to him in connection with his relocation to Florida, as
further described in “Certain Relationships and Related
Party Transactions — Related Party Loans”).
|
|
|
|
|
(3)
|
|
Reflects the $10,000 bonus
Mr. Marsh received in recognition of his role in connection
with KRG Capital Partners’ investment, including KRG
Capital Partners’ acquisition of GSO Capital Partners’
equity interest in Aurora Holdings. For more information
regarding the annual bonuses, see the “— 2009
Elements of Compensation — Annual Cash Bonuses.”
|
119
2009
Grants of Plan-Based Awards
The following table sets forth each plan-based award granted to
our named executive officers during 2009. No equity awards were
granted to our named executive officers during 2009.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Estimated Future Payouts Under
|
|
|
|
|
Non-Equity Incentive Plan
Awards(1)
|
|
|
|
|
Threshold
|
|
|
Target
|
|
|
Maximum
|
|
|
Name
|
|
($)
|
|
|
($)
|
|
|
($)
|
|
|
|
|
Mr. New
|
|
|
—
|
|
|
|
400,000
|
|
|
|
800,000
|
|
|
Mr. Stefanelli
|
|
|
—
|
|
|
|
236,250
|
|
|
|
472,500
|
|
|
Mr. Marsh
|
|
|
—
|
|
|
|
147,000
|
|
|
|
294,000
|
|
|
Mr. Ferrara
|
|
|
—
|
|
|
|
89,040
|
|
|
|
178,080
|
|
|
Mr. Null
|
|
|
—
|
|
|
|
79,013
|
|
|
|
158,026
|
|
|
|
|
|
(1)
|
|
Reflects potential payout
opportunities under the annual bonus plan. The actual amount
earned by each named executive officer is reflected in the
“Non-Equity Incentive Compensation” column of the
Summary Compensation table.
|
Employment Agreements. We maintain employment
agreements with each of Messrs. New, Marsh, Stefanelli,
Ferrara and Null, the term of which will continue until
terminated by the executive or us. Pursuant to the agreements,
the initial annual base salaries are subject to increases from
time to time in the sole discretion of our Board of Directors,
and the executives have the opportunity to earn performance
bonuses on an annual basis as determined by our Board of
Directors. The executives are also entitled to participate in
any employee benefit plans that we may from time to time have in
effect for our executive-level personnel. In addition, the
employment agreements provide certain benefits to the executives
upon their termination of employment by us. For a description of
such benefits, see “— Potential Payments Upon
Termination of Employment,” below.
Potential
Payments upon Termination of Employment
Payments Made Upon Any Termination of
Employment. Regardless of the manner in which a
named executive officer’s employment terminates, he is
entitled to receive amounts earned during his term of employment
including accrued but unpaid base salary through the date of
termination, accrued but unpaid annual bonus, unreimbursed
employment-related expenses owed to the executive officer under
our policies and accrued but unpaid vacation pay. The executive
is also entitled to all accrued benefits under any of our
employee benefit programs (in accordance with the terms of such
programs). These payments do not differ from payments made upon
termination to all employees.
Payments Made Upon Termination Without Cause or Good
Reason. Each of the employment agreements
provides that if the executive is terminated without Cause, or
the executive terminates his employment with us for Good Reason,
the executive will be entitled to receive:
|
|
|
| |
•
|
his base salary for a specified period (in the case of
Mr. New, 24 months, in the case of
Messrs. Stefanelli, Ferrara and Null, 12 months, and
in the case of Mr. Marsh, 12 months if his termination
occurs prior to a “qualifying transaction” (as defined
below) or 18 months if his termination occurs within one
year following a “qualifying transaction”), payable in
equal installments in accordance with our regular payroll
practices;
|
|
|
|
| |
•
|
in the case of Mr. New, an amount equal to two times the
average of his previous three annual bonuses, payable in
installments in accordance with our regular payroll practices;
|
|
|
|
| |
•
|
any unpaid bonus for the previous fiscal year and a pro rata
portion of his bonus for the then-current fiscal year; and
|
|
|
|
| |
•
|
in the case of Mr. New, continued health care coverage for
a period of 24 months.
|
120
Cause generally means the executive’s (i) conviction
or plea of no contest for or indictment on a felony or a crime
involving moral turpitude or the commission of any other act or
omission involving dishonesty or fraud, which involves a
material matter, with respect to us or any of our customers or
suppliers, (ii) substantial and repeated failure to perform
his duties, (iii) gross negligence or willful misconduct
that is harmful to us, (iv) conduct tending to bring us
into substantial public disgrace or disrepute (not applicable to
Mr. Null) and (v) breach of the restrictive covenants
in the employment agreement.
Good Reason generally means, without the executive’s prior
written consent, (i) a reduction in, or failure to pay when
due, the executive’s base salary, (ii) a material
diminution in the executive’s titles or duties inconsistent
with his position, (iii) failure to pay any annual bonus
when due and, in the case of Mr. New, any reduction in his
annual bonus opportunity, (iv) a material reduction in the
employee benefits offered to the executive that is not also
applicable to our other executive employees and (v) a
change in the executive’s principal office to a location
more than 50 miles from Palm Beach Gardens, Florida.
Restrictive Covenants. Each of the agreements
contains confidentiality and customer and employee
nonsolicitation covenants that apply during the executive’s
employment with us and for a certain period of time after his
termination of employment (24 months in the case of
Mr. New, and 12 months in the case of
Messrs. Marsh, Stefanelli, Ferrara and Null).
The following table summarizes the approximate value of the
termination payments and benefits that each of our named
executive officers would receive if he had terminated employment
at the close of business on December 31, 2009. The table
does not include certain amounts that the named executive
officer would be entitled to receive under certain plans or
arrangements that do not discriminate in scope, terms or
operation, in favor of our executive officers and that are
generally available to all salaried employees, such as our
401(k) plan.
Summary
of Termination Payments and Benefits
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
In Connection
|
|
|
|
|
|
|
|
|
|
|
|
|
|
with a
|
|
|
|
|
|
|
|
|
|
|
|
Before Change
|
|
Change in
|
|
|
|
|
|
|
|
|
|
|
|
in Control
|
|
Control
|
|
|
|
|
|
|
|
|
|
|
|
Termination
|
|
Termination
|
|
For Cause/
|
|
|
|
|
|
|
|
|
|
w/o Cause or for
|
|
w/o Cause or for
|
|
Voluntary
|
|
|
|
|
|
Name
|
|
Benefit
|
|
Good Reason ($)
|
|
Good Reason ($)
|
|
Termination ($)
|
|
Death ($)
|
|
Disability ($)
|
|
|
|
Mr. New
|
|
Continued Base
Salary(1)
|
|
|
800,000
|
|
|
|
800,000
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
Continued Health and Dental
Coverage(2)
|
|
|
53,044
|
|
|
|
53,044
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
2x Average
Bonus(3)
|
|
|
360,667
|
|
|
|
360,667
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
Pro-Rated
Bonus(4)
|
|
|
381,000
|
|
|
|
381,000
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
Transaction
Bonus(5)
|
|
|
—
|
|
|
|
400,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
|
|
|
1,594,711
|
|
|
|
1,994,711
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Mr. Stefanelli
|
|
Continued Base
Salary(1)
|
|
|
315,000
|
|
|
|
315,000
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
Pro-Rated
Bonus(4)
|
|
|
225,000
|
|
|
|
225,000
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Total
|
|
|
|
|
540,000
|
|
|
|
540,000
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Mr. Marsh
|
|
Continued Base
Salary(1)
|
|
|
294,000
|
|
|
|
441,000
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
Pro-Rated
Bonus(4)
|
|
|
233,000
|
|
|
|
233,000
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
Transaction
Bonus(5)
|
|
|
—
|
|
|
|
220,500
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Total
|
|
|
|
|
527,000
|
|
|
|
894,500
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Mr. Ferrara
|
|
Continued Base
Salary(1)
|
|
|
222,600
|
|
|
|
222,600
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
Pro-Rated
Bonus(4)
|
|
|
81,000
|
|
|
|
81,000
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Total
|
|
|
|
|
303,600
|
|
|
|
303,600
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Mr. Null
|
|
Continued Base
Salary(1)
|
|
|
225,750
|
|
|
|
225,750
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
Pro-Rated
Bonus(4)
|
|
|
72,000
|
|
|
|
72,000
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Total
|
|
|
|
|
297,750
|
|
|
|
297,750
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
(1)
|
|
Reflects an amount equal to the
applicable multiple of the executive’s then-current base
salary, payable in installments over 24 months, in the case
of Mr. New, or 12 months, in the case of
Messrs. Marsh, Stefanelli, Ferrara and Null.
Mr. Marsh’s multiple of salary is 1x, in the event of
|
121
|
|
|
|
|
|
his termination of employment
prior to a “qualifying transaction”, or 1.5x, in the
event of his termination of employment within one year following
the effective date of a “qualifying transaction.”
|
|
|
|
|
(2)
|
|
Reflects Company-paid Consolidated
Omnibus Budget Reconciliation Act of 1986, or COBRA, payments
for medical and dental coverage based on 2009 rates for
24 months.
|
| |
|
(3)
|
|
Reflects an amount equal to two
times the average of the bonuses Mr. New received in 2006
(on an annualized basis), 2007 and 2008, payable in installments
over 24 months.
|
| |
|
(4)
|
|
Reflects a pro-rated bonus for the
year in which the executive terminates employment. The
pro-ration is based on the executive’s and our performance
relative to the pre-approved objectives.
|
|
|
|
|
(5)
|
|
Reflects 100 percent of
Mr. New’s annual base salary and 50 percent of
the sum of Mr. Marsh’s current annual base salary plus
his current target bonus, payable in a lump sum.
|
Potential
Payments upon Change in Control
In connection with a “qualifying transaction”,
Mr. New would be entitled to receive a lump sum payment
equal to 100 percent of his then-current annual base salary, and
Mr. Marsh would be entitled to receive a lump sum payment
equal to 50 percent of the sum of his then-current annual
base salary plus his then-current target bonus, regardless of
whether their employment was terminated. If a “qualifying
transaction” had occurred on December 31, 2009,
Mr. New and Mr. Marsh would have received $400,000 and
$220,500, respectively. A “qualifying transaction”
generally means either (i) the sale or other disposition of
all or substantially all of our assets and the assets of our
subsidiaries, taken as a whole, or (ii) a transaction or
series of related transactions the result of which is that the
holders of our outstanding voting securities immediately prior
to such transaction are (after giving effect to such
transaction) no longer, in the aggregate, the “beneficial
owners” (as such term is defined in
Rule 13d-3
and
Rule 13d-5
promulgated under the Exchange Act) of more than 50 percent
of the voting power of our outstanding voting securities, and
Summit Partners and the KRG Capital Partners, in the aggregate,
are no longer entitled to appoint a majority of the managers to
the board of managers of Aurora Holdings (excluding a public
offering and certain other issuances by us).
Mr. Marsh’s benefit will terminate immediately prior
to this offering.
None of our other named executive officers would have received
any payments if a change in control had occurred on
December 31, 2009, absent their termination of employment.
Equity
Incentive Plan
We maintain our New Plan, which provides for grants of
Class D units of Aurora Holdings to our employees. The
terms of the Class D units of Aurora Holdings were governed
by the Aurora Holdings LLC Agreement and, generally, the
Class D units of Aurora Holdings are entitled to
distributions following distributions to which the Class A,
Class A-1,
Class B and Class C units of Aurora Holdings are
entitled. Our Board of Directors administers the New Plan. The
Class D units generally vest over a period of time, subject
to the employee’s continued employment with us. We will not
grant any further awards under the New Plan. In connection with
the Reorganization Transactions, the Class D units of
Aurora Holdings held by our employees will either be exchanged
for shares of our Class A common stock or cancelled without
consideration. See “Prospectus Summary —
Reorganization Transactions” and “Organizational
Structure — Reorganization Transactions.”
Director
Compensation
The members of our Board of Directors did not receive any cash,
equity or any other compensation for services rendered during
2009.
Risk
Assessment
Prior to the commencement of this offering, we will conduct a
risk assessment of our compensation policies and practices for
all employees, including non-executive officers, to determine
whether such policies and practices create risks that are
reasonably likely to have a material adverse effect on us.
122
PRINCIPAL
AND SELLING STOCKHOLDERS
The following table sets forth information regarding the
beneficial ownership of our Class A common stock and
Class B common stock, as
of 2010,
after giving effect to the Reorganization Transactions but prior
to this offering, by the following individuals or groups:
|
|
|
| |
•
|
each of our directors;
|
| |
| |
•
|
each of our named executive officers;
|
| |
| |
•
|
all of our directors and executive officers as a group;
|
| |
| |
•
|
each person, or group of affiliated persons, whom we know
beneficially owns more than 5 percent of our outstanding
common stock; and
|
| |
| |
•
|
each selling stockholder.
|
See “Prospectus Summary — Reorganization
Transactions” and “Organizational
Structure — Reorganization Transactions.”
Based on an assumed initial public offering price of
$ per share, which is the midpoint
of the price range set forth on the cover page of this
prospectus, we intend to use $ of
the proceeds from this offering, along with TRA Rights, to
purchase
Aurora Holdings Units
and shares
of our Class B common stock held by the Aurora Holdings
Continuing Members (or $
and
Aurora Holdings Units
and shares
of our Class B common stock if the underwriters exercise
their over-allotment option in full). The beneficial ownership
after the completion of this offering reflects this application
of proceeds.
We have determined beneficial ownership in accordance with the
rules of the SEC. Except as indicated by the footnotes below, we
believe, based on the information furnished to us, that the
persons and entities named in the table below have sole voting
and investment power with respect to all shares of Class A
common stock and Class B common stock that they
beneficially own, subject to applicable community property laws.
We have based our calculation of the percentage of beneficial
ownership
on shares
of our Class A common stock outstanding as
of ,
which assumes:
|
|
|
| |
•
|
the Reorganization Transactions;
|
| |
| |
•
|
an initial public offering price of
$ per share, which is the midpoint
of the price range set forth on the cover page of this
prospectus; and
|
| |
| |
•
|
the application of the net proceeds we will receive from this
offering.
|
The following table assumes the underwriters’
over-allotment option is not exercised:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Percent of Combined
|
|
|
|
|
Class A Common
Stock(1)
|
|
|
Class B Common
Stock(1)
|
|
|
Voting
Power(2)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Number
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
of
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Number of
|
|
|
|
|
|
|
|
|
Shares
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Shares
|
|
|
Percent
|
|
|
|
|
|
After
|
|
|
|
|
|
Number of
|
|
|
Percent
|
|
|
Number
|
|
|
Number
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Prior to
|
|
|
Prior to
|
|
|
Number
|
|
|
this
|
|
|
Percent
|
|
|
Shares
|
|
|
Prior to
|
|
|
of
|
|
|
of Shares
|
|
|
Percent
|
|
|
Prior to
|
|
|
After
|
|
|
|
|
this
|
|
|
this
|
|
|
of Shares
|
|
|
Offering
|
|
|
After this
|
|
|
Prior to this
|
|
|
this
|
|
|
Shares
|
|
|
After this
|
|
|
After this
|
|
|
this
|
|
|
this
|
|
|
Name and Address of Beneficial
Owner(3)(4)
|
|
Offering(1)
|
|
|
Offering
|
|
|
Offered
|
|
|
(1)
|
|
|
Offering
|
|
|
Offering(1)
|
|
|
Offering
|
|
|
Offered
|
|
|
Offering(1)
|
|
|
Offering
|
|
|
Offering
|
|
|
Offering
|
|
|
|
|
Summit Ventures
VI-A,
L.P.(5)
|
|
|
|
|
|
|
|
%
|
|
|
|
|
|
|
|
|
|
|
|
%
|
|
|
|
|
|
|
|
%
|
|
|
|
|
|
|
|
%
|
|
|
|
|
|
|
|
%
|
|
|
|
%
|
|
Summit Ventures
VI-B,
L.P.(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Summit Partners VI (GP),
L.P.(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Summit VI Entrepreneurs Fund,
L.P.(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Summit VI Advisors Fund,
L.P.(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Summit Investors VI,
L.P.(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Summit Partners Private Equity Fund
VII-A,
L.P.(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Summit Partners Private Equity Fund
VII-B,
L.P.(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
123
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Percent of Combined
|
|
|
|
|
Class A Common
Stock(1)
|
|
|
Class B Common
Stock(1)
|
|
|
Voting
Power(2)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Number
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
of
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Number of
|
|
|
|
|
|
|
|
|
Shares
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Shares
|
|
|
Percent
|
|
|
|
|
|
After
|
|
|
|
|
|
Number of
|
|
|
Percent
|
|
|
Number
|
|
|
Number
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Prior to
|
|
|
Prior to
|
|
|
Number
|
|
|
this
|
|
|
Percent
|
|
|
Shares
|
|
|
Prior to
|
|
|
of
|
|
|
of Shares
|
|
|
Percent
|
|
|
Prior to
|
|
|
After
|
|
|
|
|
this
|
|
|
this
|
|
|
of Shares
|
|
|
Offering
|
|
|
After this
|
|
|
Prior to this
|
|
|
this
|
|
|
Shares
|
|
|
After this
|
|
|
After this
|
|
|
this
|
|
|
this
|
|
|
Name and Address of Beneficial
Owner(3)(4)
|
|
Offering(1)
|
|
|
Offering
|
|
|
Offered
|
|
|
(1)
|
|
|
Offering
|
|
|
Offering(1)
|
|
|
Offering
|
|
|
Offered
|
|
|
Offering(1)
|
|
|
Offering
|
|
|
Offering
|
|
|
Offering
|
|
|
|
|
Summit Partners PE VII,
L.P.(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
KRG Capital Fund IV,
L.P.(6)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
KRG Capital Fund
IV-A,
L.P.(6)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
KRG Capital Fund IV (PA),
L.P.(6)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
KRG Capital Fund IV (FF),
L.P.(6)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
KRG Co-Investment,
L.L.C.(6)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Directors and Executive Officers:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
James C. New
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Martin J. Stefanelli
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Fred Ferrara
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Michael Null
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Gregory A. Marsh
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Thomas S.
Roberts(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Christopher
Dean(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Peter J.
Connolly(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Mark M.
King(6)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Christopher J.
Bock(6)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Blair
Tikker(6)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
James M. Emanuel
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
All Directors and Executive Officers as a group (5 persons)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
*
|
|
Less than 1 percent.
|
|
(1)
|
|
The Summit Partners Equityholders
and the Management Equityholders each hold Aurora Holdings Units
and an equal number of shares of Class B common stock. Each
of the Summit Partners Equityholders and the Management
Equityholders has the right at any time to exchange any Aurora
Holdings Units (and a corresponding number of shares of
Class B common stock) for shares of Class A common
stock on a
one-for-one
basis. See “Description of Capital Stock.”
|
|
(2)
|
|
Percentage of total voting power
represents voting power with respect to all shares of our
Class A common stock and Class B common stock, voting
together as a single class. Our Class B common stock does
not have any of the economic rights (including rights to
dividends and distributions upon liquidation, but excluding the
return of par value upon liquidation) associated with our
Class A common stock. See “Description of Capital
Stock.”
|
|
(3)
|
|
Unless otherwise specified, the
address of each beneficial owner listed in the table below is
c/o Aurora
Diagnostics, Inc. 11025 RCA Center Drive, Suite 300, Palm
Beach Gardens, FL 33410.
|
|
(4)
|
|
Beneficial ownership is determined
in accordance with
Rule 13d-3
of the Exchange Act and generally includes voting and investment
power with respect to securities, subject to community property
laws, where applicable.
|
|
(5)
|
|
Represents
(a) shares
of Class B common stock held by Summit Ventures VI-A, L.P.;
(b) shares
of Class A common stock held by Summit Ventures VI-B, L.P.;
(c) shares
of Class A common stock held by Summit Partners VI (GP),
L.P.;
(d) shares
of Class B common stock held by Summit VI Entrepreneurs
Fund, L.P.;
(e) shares
of Class B common stock held by Summit VI Advisors Fund,
L.P.;
(f) shares
of Class B common stock held by Summit Investors VI, L.P.;
(g) shares
of Class B common stock held by Summit Partners Private
Equity
Fund VII-A,
L.P.;
(h) shares
of Class A common stock held by Summit Partners Private
Equity
Fund VII-B,
L.P.; and
(i) shares
of Class A common stock held by Summit Partners PE VII,
L.P. Summit Partners, L.P. is the managing member of Summit
Partners VI (GP), LLC, which is the general partner of Summit
Partners VI (GP), L.P., which is the general partner of each of
Summit Ventures VI-A, L.P., Summit Ventures VI-B, L.P., Summit
VI Advisors Fund, L.P., Summit VI Entrepreneurs Fund, L.P. and
Summit Investors VI, L.P. Summit Partners, L.P. is also the
managing member of Summit Partners PE VII, LLC, which is the
general partner of Summit Partners PE VII, L.P., which is the
general partner of each of Summit Partners Private Equity
Fund VII-A,
L.P. and Summit Partners Private Equity
Fund VII-B,
L.P. Summit Partners, L.P., through an investment committee
currently composed of Bruce R. Evans and Martin J. Mannion, has
voting and dispositive authority over the shares held by each of
these entities and therefore beneficially owns such shares.
Decisions of the investment committee are made by a majority
vote of its members. Gregory M. Avis, John R. Carroll, Peter Y.
Chung, Scott C. Collins, Christopher J. Dean, Bruce R. Evans,
Charles J. Fitzgerald, Craig D.
|
124
|
|
|
|
|
|
Frances, Walter G. Kortschak,
Sotiris T.F. Lyritzis, Martin J. Mannion, Harrison B. Miller,
Kevin P. Mohan, Thomas S. Roberts, E. Roe Stamps, Joseph F.
Trustey and Stephen G. Woodsum are the members of Summit Master
Company, LLC, which is the general partner of Summit Partners,
L.P., and each disclaims beneficial ownership of the shares held
by Summit Partners. The address of each of the Summit Partners
entities is 222 Berkeley Street, 18th Floor, Boston, MA 02116.
|
|
(6)
|
|
Represents
(a) shares
of Class A common stock held by KRG Capital Fund IV,
L.P.;
(b) shares
of Class A common stock held by KRG Capital
Fund IV-A,
L.P.;
(c) shares
of Class A common stock held by KRG Capital Fund IV
(PA), L.P.;
(d) shares
of Class A common stock held by KRG Capital Fund IV
(FF), L.P.; and
(e) shares
of Class A common stock held by KRG Co-Investment, L.L.C.
KRG Capital Management, L.P. is the general partner of each of
KRG Capital Fund IV, L.P., KRG Capital
Fund IV-A,
L.P., KRG Capital Fund IV (PA), L.P. and KRG Capital
Fund IV (FF), L.P. KRG Capital Management, L.P., through an
eleven (11) person investment committee with respect to the
Class IV series of funds, including Mark M. King,
Christopher J. Bock and Blair J. Tikker, has voting and
dispositive authority over the shares held by each of KRG
Capital Fund IV, L.P., KRG Capital
Fund IV-A,
L.P., KRG Capital Fund IV (PA), L.P., KRG Capital
Fund IV (FF), L.P. and KRG Co-Investment L.L.C. and,
therefore, beneficially owns such shares. Decisions of the
investment committee are made by a vote of the majority of its
members and no individual member of the investment committee has
voting or dispositive authority over the shares. Mark M. King,
Christopher J. Bock and Blair J. Tikker are members of KRG
Capital, LLC with respect to the Class IV series of funds,
which is the general partner of KRG Capital Management, L.P.,
and each disclaims beneficial ownership of the shares held by
KRG Capital Management, L.P. Affiliates of Mark M. King and
Christopher J. Bock, individually, are members of KRG
Co-Investment, LLC, and each disclaims beneficial ownership of
the shares held by KRG Capital Management, L.P. The address of
each of the KRG Capital Partners entities is 1515 Arapahoe
Street, Tower 1, Suite 1500, Denver, CO 80202.
|
The following table assumes the underwriters’
over-allotment option is exercised:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Percent of Combined
|
|
|
|
|
Class A Common
Stock(1)
|
|
|
Class B Common
Stock(1)
|
|
|
Voting
Power(2)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Number
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
of
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Number of
|
|
|
|
|
|
|
|
|
Shares
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Shares
|
|
|
Percent
|
|
|
|
|
|
After
|
|
|
|
|
|
Number of
|
|
|
Percent
|
|
|
Number
|
|
|
Number
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Prior to
|
|
|
Prior to
|
|
|
Number
|
|
|
this
|
|
|
Percent
|
|
|
Shares
|
|
|
Prior to
|
|
|
of
|
|
|
of Shares
|
|
|
Percent
|
|
|
Prior to
|
|
|
After
|
|
|
|
|
this
|
|
|
this
|
|
|
of Shares
|
|
|
Offering
|
|
|
After this
|
|
|
Prior to this
|
|
|
this
|
|
|
Shares
|
|
|
After this
|
|
|
After this
|
|
|
this
|
|
|
this
|
|
|
Name and Address of Beneficial
Owner(3)(4)
|
|
Offering(1)
|
|
|
Offering
|
|
|
Offered
|
|
|
(1)
|
|
|
Offering
|
|
|
Offering(1)
|
|
|
Offering
|
|
|
Offered
|
|
|
Offering(1)
|
|
|
Offering
|
|
|
Offering
|
|
|
Offering
|
|
|
|
|
Summit Ventures VI-A,
L.P.(5)
|
|
|
|
|
|
|
|
%
|
|
|
|
|
|
|
|
|
|
|
|
%
|
|
|
|
|
|
|
|
%
|
|
|
|
|
|
|
|
%
|
|
|
|
|
|
|
|
%
|
|
|
|
%
|
|
Summit Ventures VI-B,
L.P.(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Summit Partners VI (GP),
L.P.(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Summit VI Entrepreneurs Fund,
L.P.(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Summit VI Advisors Fund,
L.P.(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Summit Investors VI,
L.P.(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Summit Partners Private Equity Fund VII-A,
L.P.(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Summit Partners Private Equity Fund VII-B,
L.P.(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Summit Partners PE VII,
L.P.(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
125
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Percent of Combined
|
|
|
|
|
Class A Common
Stock(1)
|
|
|
Class B Common
Stock(1)
|
|
|
Voting
Power(2)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Number
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
of
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Number of
|
|
|
|
|
|
|
|
|
Shares
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Shares
|
|
|
Percent
|
|
|
|
|
|
After
|
|
|
|
|
|
Number of
|
|
|
Percent
|
|
|
Number
|
|
|
Number
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Prior to
|
|
|
Prior to
|
|
|
Number
|
|
|
this
|
|
|
Percent
|
|
|
Shares
|
|
|
Prior to
|
|
|
of
|
|
|
of Shares
|
|
|
Percent
|
|
|
Prior to
|
|
|
After
|
|
|
|
|
this
|
|
|
this
|
|
|
of Shares
|
|
|
Offering
|
|
|
After this
|
|
|
Prior to this
|
|
|
this
|
|
|
Shares
|
|
|
After this
|
|
|
After this
|
|
|
this
|
|
|
this
|
|
|
Name and Address of Beneficial
Owner(3)(4)
|
|
Offering(1)
|
|
|
Offering
|
|
|
Offered
|
|
|
(1)
|
|
|
Offering
|
|
|
Offering(1)
|
|
|
Offering
|
|
|
Offered
|
|
|
Offering(1)
|
|
|
Offering
|
|
|
Offering
|
|
|
Offering
|
|
|
|
|
KRG Capital Fund IV,
L.P.(6)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
KRG Capital Fund IV-A,
L.P.(6)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
KRG Capital Fund IV(PA),
L.P.(6)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
KRG Capital Fund IV(FF),
L.P.(6)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
KRG Co-Investment,
L.L.C.(6)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Directors and Executive Officers:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
James C. New
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Martin J. Stefanelli
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Fred Ferrara
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Michael Null
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Gregory A. Marsh
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Thomas S.
Roberts(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Christopher
Dean(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Peter J.
Connolly(5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Mark M.
King(6)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Christopher J.
Bock(6)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Blair
Tikker(6)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
James M. Emanuel
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
All Directors and Executive Officers as a group (5 persons)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
*
|
|
Less than 1 percent.
|
|
(1)
|
|
The Summit Partners Equityholders
and the Management Equityholders each hold Aurora Holdings Units
and an equal number of shares of Class B common stock. Each
of the Summit Partners Equityholders and the Management
Equityholders has the right at any time to exchange any Aurora
Holdings Units (and a corresponding number of shares of
Class B common stock) for shares of Class A common
stock on a
one-for-one
basis. See “Description of Capital Stock.”
|
|
(2)
|
|
Percentage of total voting power
represents voting power with respect to all shares of our
Class A common stock and Class B common stock, voting
together as a single class. Our Class B common stock does
not have any of the economic rights (including rights to
dividends and distributions upon liquidation, but excluding the
return of par value upon liquidation) associated with our
Class A common stock. See “Description of Capital
Stock.”
|
|
(3)
|
|
Unless otherwise specified, the
address of each beneficial owner listed in the table below is
c/o Aurora
Diagnostics, Inc. 11025 RCA Center Drive, Suite 300, Palm
Beach Gardens, FL 33410.
|
|
(4)
|
|
Beneficial ownership is determined
in accordance with
Rule 13d-3
of the Exchange Act and generally includes voting and investment
power with respect to securities, subject to community property
laws, where applicable.
|
|
(5)
|
|
Represents
(a) shares
of Class B common stock held by Summit Ventures VI-A, L.P.;
(b) shares
of Class A common stock held by Summit Ventures VI-B, L.P.;
(c) shares
of Class A common stock held by Summit Partners VI (GP),
L.P.;
(d) shares
of Class B common stock held by Summit VI Entrepreneurs
Fund, L.P.;
(e) shares
of Class B common stock held by Summit VI Advisors Fund,
L.P.;
(f) shares
of Class B common stock held by Summit Investors VI, L.P.;
(g) shares
of Class B common stock held by Summit Partners Private
Equity
Fund VII-A,
L.P.;
(h) shares
of Class A common stock held by Summit Partners Private
Equity
Fund VII-B,
L.P.; and
(i) shares
of Class A common stock held by Summit Partners PE VII,
L.P. Summit Partners, L.P. is the managing member of Summit
Partners VI (GP), LLC, which is the general partner of Summit
Partners VI (GP), L.P., which is the general partner of each of
Summit Ventures VI-A, L.P., Summit Ventures VI-B, L.P., Summit
VI Advisors Fund, L.P., Summit VI Entrepreneurs Fund, L.P. and
Summit Investors VI, L.P. Summit Partners, L.P. is also the
managing member of Summit Partners PE VII, LLC, which is the
general partner of Summit Partners PE VII, L.P., which is the
general partner of each of Summit Partners Private Equity
Fund VII-A,
L.P. and Summit Partners Private Equity
Fund VII-B,
L.P. Summit Partners, L.P., through an investment committee
currently composed of Bruce R. Evans and Martin J. Mannion, has
voting and dispositive authority over the shares held by each of
these entities and therefore beneficially owns such shares.
Decisions of the investment committee are made by a majority
vote of its members. Gregory M. Avis, John R. Carroll, Peter Y.
Chung, Scott C. Collins, Christopher J. Dean, Bruce R. Evans,
Charles J. Fitzgerald, Craig D. Frances, Walter G. Kortschak,
Sotiris T.F. Lyritzis, Martin J. Mannion, Harrison B. Miller,
Kevin P. Mohan, Thomas S. Roberts, E. Roe Stamps, Joseph F.
Trustey and Stephen G. Woodsum are the members of Summit Master
Company, LLC, which is the general partner of
|
126
|
|
|
|
|
|
Summit Partners, L.P., and each
disclaims beneficial ownership of the shares held by Summit
Partners. The address of each of the Summit Partners entities is
222 Berkeley Street, 18th Floor, Boston, MA 02116.
|
|
(6)
|
|
Represents
(a) shares
of Class A common stock held by KRG Capital Fund IV,
L.P.;
(b) shares
of Class A common stock held by KRG Capital
Fund IV-A,
L.P.;
(c) shares
of Class A common stock held by KRG Capital Fund IV
(PA), L.P.;
(d) shares
of Class A common stock held by KRG Capital Fund IV
(FF), L.P.; and
(e) shares
of Class A common stock held by KRG Co-Investment, L.L.C.
KRG Capital Management, L.P. is the general partner of each of
KRG Capital Fund IV, L.P., KRG Capital
Fund IV-A,
L.P., KRG Capital Fund IV (PA), L.P. and KRG Capital
Fund IV (FF), L.P. KRG Capital Management, L.P., through an
eleven (11) person investment committee with respect to the
Class IV series of funds, including Mark M. King,
Christopher J. Bock and Blair J. Tikker, has voting and
dispositive authority over the shares held by each of KRG
Capital Fund IV, L.P., KRG Capital
Fund IV-A,
L.P., KRG Capital Fund IV (PA), L.P., KRG Capital
Fund IV (FF), L.P. and KRG Co-Investment L.L.C. and,
therefore, beneficially owns such shares. Decisions of the
investment committee are made by a vote of the majority of its
members and no individual member of the investment committee has
voting or dispositive authority over the shares. Mark M. King,
Christopher J. Bock and Blair J. Tikker are members of KRG
Capital, LLC with respect to the Class IV series of funds,
which is the general partner of KRG Capital Management, L.P.,
and each disclaims beneficial ownership of the shares held by
KRG Capital Management, L.P. Affiliates of Mark M. King and
Christopher J. Bock, individually, are members of KRG
Co-Investment, LLC, and each disclaims beneficial ownership of
the shares held by KRG Capital Management, L.P. The address of
each of the KRG Capital Partners entities is 1515 Arapahoe
Street, Tower 1, Suite 1500, Denver, CO 80202.
|
Relationship
with Selling Stockholders
All of the shares offered by the selling stockholders were
issued to them in the Reorganization Transactions. For
additional information with respect to the selling stockholders
and their relationship with us please see “Certain
Relationships and Related Party Transactions,”
“Prospectus Summary — Reorganization
Transactions” and “Organizational
Structure — Reorganization Transactions.”
127
CERTAIN
RELATIONSHIPS AND RELATED PARTY TRANSACTIONS
In addition to the cash and equity compensation arrangements of
our directors and executive officers discussed above under
“Executive Compensation — Compensation Discussion
and Analysis,” the following is a description of
transactions since January 1, 2007, to which we have been a
party in which the amount involved exceeded or will exceed
$120,000 and in which any of our directors, executive officers,
beneficial holders of more than 5 percent of our capital
stock, or entities affiliated with them, had or will have a
direct or indirect material interest.
Reorganization
Agreement
In connection with the Reorganization Transactions, we and ARDX
Sub will enter into a reorganization agreement with Aurora
Holdings and our Principal Equityholders which governs the
Reorganization Transactions. In addition, under the
reorganization agreement, the Aurora Holdings Continuing Members
will subscribe for a number of shares of our Class B common
stock equal to the number of Aurora Holdings Units they own, at
price equal to the par value per share of Class B common
stock. For further information, see “Prospectus
Summary — Reorganization Transactions” and
“Organizational Structure — Reorganization
Transactions.”
Second
Amended and Restated Aurora Holdings Limited Liability Company
Agreement
In connection with the Reorganization Transactions, the Aurora
Holdings Continuing Members, ARDX Sub and Aurora Holdings will
enter into the Second Amended and Restated Aurora Holdings LLC
Agreement. In consideration for agreeing to enter into the
Second Amended and Restated Aurora Holdings LLC Agreement, the
Aurora Holdings Continuing Members will receive rights to
distributions that are calculated in a manner that is similar to
the TRA Rights. As a result of the Reorganization Transactions
and in accordance with the terms of the Second Amended and
Restated Aurora Holdings LLC Agreement, we will indirectly
operate our business through Aurora Holdings and its
subsidiaries. With ARDX Sub as the sole managing member of
Aurora Holdings, we will indirectly have all business and
operational control over Aurora Holdings. As such, we, through
our officers and directors, will be responsible for all business
and operational decisions of Aurora Holdings and the
day-to-day
management of Aurora Holdings’ and its subsidiaries’
business.
The holders of Aurora Holdings Units, including ARDX Sub, will
generally incur U.S. federal, state, local and foreign
income taxes on their proportionate share of any net taxable
income of Aurora Holdings. Net profits and net losses of Aurora
Holdings will generally be allocated to its members pro rata in
accordance with the percentages of their respective Aurora
Holdings Units, though certain non-pro rata adjustments will be
made to reflect tax depreciation, amortization and other
allocations. The Second Amended and Restated Aurora Holdings LLC
Agreement will provide for cash distributions to Aurora
Holdings’ members if the taxable income of Aurora Holdings
will give rise to taxable income for any of its members.
Generally, these tax distributions will be computed based on our
estimate of the highest amount of net taxable income of Aurora
Holdings allocable to any such holder of Aurora Holdings Units
multiplied by an assumed tax rate equal to the highest effective
marginal combined U.S. federal, state and local income tax
rate prescribed for an individual or corporate resident in New
York, New York. These tax distributions indirectly received by
us will be used to pay our taxes.
In addition, the Second Amended and Restated Aurora Holdings LLC
Agreement will permit Aurora Holdings to make TRA Distributions
to its members. These TRA Distributions will be calculated in a
manner similar to the TRA Rights. These TRA Distributions will
enable us to make payments under the Tax Receivable Agreement.
In the event of a termination or change in law giving rise to a
lump sum payment obligation under the Tax Receivable Agreement
(discussed in “— Tax Receivable Agreement”
below), the TRA Distributions will be limited to amounts that
otherwise would have been distributed absent such event. If such
lump sum payment obligation arises, then we alone will be
entitled to receive further TRA Distributions, which we will use
to pay down the lump sum payment obligation.
In addition, to the extent permitted under our debt agreements,
Aurora Holdings may make distributions indirectly to us, without
pro rata distributions to other Aurora Holdings members, in
order to pay:
|
|
|
| |
•
|
consideration, if any, for redemption, repurchase or other
acquisition of Aurora Holdings Units to the extent such cash is
used to redeem, repurchase or otherwise acquire our Class A
common stock;
|
128
|
|
|
| |
•
|
operating, administrative and other similar costs incurred by
us, including payments on indebtedness and preferred stock
issued by us, to the extent we use the proceeds from the
issuance to pay expenses (in either case only to the extent
economically equivalent indebtedness or preferred stock were not
issued by Aurora Holdings to us);
|
| |
| |
•
|
payments representing interest with respect to payments not made
when due under the terms of the Tax Receivable
Agreement; and
|
| |
| |
•
|
other payments related to:
|
|
|
|
| |
•
|
legal, tax, accounting and other professional fees and expenses;
|
| |
| |
•
|
judgments, settlements, penalties, fines or other costs and
expenses in respect of any claims involving us; and
|
| |
| |
•
|
other fees and expenses related to the maintenance of our
existence or any securities offering, investment or acquisition
transaction authorized by our Board of Directors.
|
The Second Amended and Restated Aurora Holdings LLC Agreement
will provide that, except as otherwise determined by us, and, at
any time we issue a share of our Class A common stock or
any other equity security, other than pursuant to an employee
benefit plan or shareholder rights plan, the net proceeds
received by us with respect to such issuance, if any, shall be
concurrently invested indirectly in Aurora Holdings (unless such
shares were issued by us solely to fund our ongoing operations
or the purchase of Aurora Holdings Units or to pay our expenses
or other obligations), and Aurora Holdings shall issue to ARDX
Sub one Aurora Holdings Unit or other economically equivalent
equity interest. Conversely, if at any time, any shares of our
Class A common stock are redeemed, repurchased or otherwise
acquired, Aurora Holdings shall redeem, repurchase or otherwise
acquire an equal number of Aurora Holdings Units held by ARDX
Sub, upon the same terms and for the same price, as the shares
of our Class A common stock are redeemed, repurchased or
otherwise acquired.
In accordance with the terms of the Second Amended and Restated
Aurora Holdings LLC Agreement, the Aurora Holdings Continuing
Members will generally have the right to exchange their Aurora
Holdings Units (and corresponding shares of our Class B
common stock) with ARDX Sub for shares of our Class A
common stock on a
one-for-one
basis or, in certain circumstances, an equivalent amount of
cash, subject to customary conversion rate adjustments for stock
splits, stock dividends and reclassifications. As the Aurora
Holdings Continuing Members exchange their Aurora Holdings
Units, our membership interests in Aurora Holdings will be
correspondingly increased. In connection with any proposed
exchange by an Aurora Holdings Continuing Member, we may, in
certain circumstances, elect to directly or indirectly through
ARDX Sub purchase and acquire the applicable Aurora Holdings
Units and corresponding Class B common stock by paying
either:
|
|
|
| |
•
|
with the permission of the exchanging Aurora Holdings
Continuing Member, cash in an amount equal to the fair market
value (determined by the volume weighted average price of the
shares of Class A common stock on the date of purchase
(ignoring the price paid in connection with any block trades of
100,000 or more shares) of the shares of Class A common
stock the Aurora Holdings Continuing Member would have received
in the proposed exchange; or
|
|
|
|
| |
•
|
the number of shares of Class A common stock the member
would have received in the proposed exchange.
|
Unless we make such an election, we have no obligation to the
exchanging member or Aurora Holdings with respect to the
proposed exchange, other than to ensure that we have shares of
our Class A common stock to deliver to the exchanging
Aurora Holdings Continuing Member.
Under the Second Amended and Restated Aurora Holdings LLC
Agreement, the members have agreed that the Summit Partners
Equityholders
and/or one
or more of their respective affiliates are permitted to engage
in business activities or invest in or acquire businesses which
may compete with our business or do business with any client of
ours.
Under the Second Amended and Restated Aurora Holdings LLC
Agreement, Aurora Holdings will indemnify certain of its
members, including us, against any and all losses and expenses
related thereto incurred by reason of the fact that such person
was a member of Aurora Holdings. In the event that losses are
incurred as a result of a
129
member’s fraud or willful misconduct, such member is not
entitled to indemnification under the Second Amended and
Restated Aurora Holdings LLC Agreement.
Aurora Holdings may be dissolved only upon the first to occur of:
|
|
|
| |
•
|
the sale of substantially all of its assets, or
|
|
|
|
| |
•
|
the voluntary agreement of us and the holders of at least
50 percent of the Aurora Holdings Units held by the Aurora
Holdings Continuing Members.
|
Upon dissolution, Aurora Holdings will be liquidated and the
proceeds from any liquidation will be applied and distributed in
the following manner:
|
|
|
| |
•
|
first, to creditors (including to the extent permitted by law,
creditors who are members) in satisfaction of the liabilities of
Aurora Holdings;
|
|
|
|
| |
•
|
second, to establish cash reserves for contingent or unforeseen
liabilities;
|
|
|
|
| |
•
|
third, to the members for unpaid amounts due under the Second
Amended and Restated Aurora Holdings LLC Agreement in respect of
the members’ tax liabilities and TRA Distributions; and
|
|
|
|
| |
•
|
fourth, to the members in proportion of their interests in
Aurora Holdings.
|
For further information, see “Prospectus
Summary — Reorganization Transactions” and
“Organizational Structure — Reorganization
Transactions.”
Tax
Receivable Agreement
Prior to this offering, we intend to complete the Reorganization
Transactions described under “Prospectus
Summary — Reorganization Transactions” and
“Organizational Structure — Reorganization
Transactions.” Also, as described under “Use of
Proceeds,” we intend to use a portion of the proceeds from
this offering to purchase Aurora Holdings Units from certain of
our Principal Equityholders. The purchases of these Aurora
Holdings Units are likely to result in tax basis adjustments to
the assets of Aurora Holdings, and these tax basis adjustments
will be allocated to us. In addition, the Aurora Holdings Units
held by the Aurora Holdings Continuing Members will be
exchangeable in the future for cash or shares of our
Class A common stock. These future exchanges are likely to
result in tax basis adjustments to the assets of Aurora
Holdings, which adjustments would also be allocated to us. Both
the existing tax basis and the anticipated tax basis adjustments
are expected to reduce the amount of tax that we would otherwise
be required to pay in the future.
Aurora Diagnostics, Inc. will enter into the Tax Receivable
Agreement with ARDX Sub, Aurora Holdings and the Tax Receivable
Entity. The Tax Receivable Agreement will generally provide for
the annual payment by Aurora Diagnostics, Inc. to the Tax
Receivable Entity of 85 percent of certain cash tax
savings, if any, in U.S. federal, state, local and foreign
income tax realized by Aurora Diagnostics, Inc. after the
completion of this offering as a result of:
|
|
|
| |
•
|
favorable tax attributes associated with amortizable goodwill
and other intangibles held by Aurora Holdings and created by its
previous acquisitions;
|
| |
| |
•
|
any step-up
in tax basis in our share of Aurora Holdings’ assets
resulting from:
|
|
|
|
| |
•
|
the acquisition by us of Aurora Holdings Units from the Aurora
Holdings Continuing Members in exchange for shares of our
Class A common stock or cash, or
|
| |
| |
•
|
payments under the Tax Receivable Agreement to the Tax
Receivable Entity; and
|
|
|
|
| |
•
|
tax benefits related to imputed interest deemed to be paid by us
as a result of the Tax Receivable Agreement.
|
The actual increase in tax basis, as well as the amount and
timing of any payments under the Tax Receivable Agreement, will
vary depending upon a number of factors, including the timing of
exchanges by the Aurora Holdings Continuing Members, as
applicable, the price of our Class A common stock at the
time of the exchange, the extent to which such exchanges are
taxable, the amount and timing of the taxable income we generate
in the future and the tax rate then applicable, and our use of
the portion of our payments under the Tax Receivable Agreement
constituting
130
imputed interest or amortizable tax basis. We expect that, as a
result of the amount of the increases in the tax basis of the
tangible and intangible assets of Aurora Holdings, assuming no
material changes in the relevant tax law and that we earn
sufficient taxable income to realize in full the potential tax
benefit described above, future payments under the Tax
Receivable Agreement in respect of tax attributes resulting from
past acquisitions described above as well as from the
Reorganization Transactions will aggregate
$ million and range from
approximately $ million to
$ million per year over the
next years. These amounts reflect
only certain cash tax savings attributable to current tax
attributes resulting from past acquisitions described above as
well as from the Reorganization Transactions. It is possible
that future transactions or events could increase or decrease
the actual tax benefits realized and the corresponding Tax
Receivable Agreement payments from these tax attributes. Future
payments under the Tax Receivable Agreement in respect of
subsequent acquisitions of Aurora Holdings Units would be in
addition to these amounts and would, if such exchanges took
place at the initial public offering price, be of substantial
magnitude.
Although we do not believe that the IRS would challenge the tax
basis increases or other benefits arising under the Tax
Receivable Agreement, the Tax Receivable Entity will not
reimburse or indemnify us for any payments previously made if
such tax basis increases or other tax benefits are subsequently
disallowed or for any other claims made by the IRS, except that
excess payments made to the Tax Receivable Entity will be netted
against payments otherwise to be made, if any, after our
determination of such excess. As a result, in such
circumstances, we could make payments under the Tax Receivable
Agreement that are greater than our actual cash tax savings.
Because we are a holding company with no operations of our own,
our ability to make payments under the Tax Receivable Agreement
is dependent on the ability of Aurora Holdings and its
subsidiaries to make distributions to us. Our debt agreements
will restrict the ability of our subsidiaries to make
distributions to us under some circumstances, which could affect
our ability to make payments under the Tax Receivable Agreement.
More specifically, we will be able to receive TRA Distributions
to make payments under the Tax Receivable Agreement with respect
to current year cash tax savings, but such distribution will not
be increased on account of any lump sum payment arising under
the Tax Receivable Agreement. To the extent that we are unable
to make payments under the Tax Receivable Agreement because of
such restrictions, such payments will be deferred and will
accrue interest until paid.
Rights to receive payments under the Tax Receivable Agreement
may be terminated by the Tax Receivable Entity if, as the result
of an actual or proposed change in law, the existence of the
agreement would cause recognition of ordinary income (instead of
capital gain) in connection with future exchanges of Aurora
Holdings Units for cash or shares of Class A common stock
or would otherwise have material adverse tax consequences to the
Tax Receivable Entity or its owners. There have been legislative
proposals in the U.S. Congress that, if enacted, may result in
such ordinary income recognition. Further, in the event of such
a termination, the Tax Receivable Entity would have the right,
subject to the delivery of an appropriate tax opinion, to
require us to pay a lump sum amount in lieu of the payments
otherwise provided under the agreement. That lump sum amount
would be calculated by increasing the portion of the tax savings
retained by us to 30 percent (from 15 percent) and by
calculating a present value for the total amount that would
otherwise be payable under the agreement, using a discount rate
and assumptions as to income tax rates and as to our ability to
utilize the tax benefits (including the assumption that we will
have sufficient taxable income to fully utilize the tax benefits
and the assumption that all exchanges that have not taken place
will take place as of the effective date of the acceleration,
which will increase the amount of the lump sum payment). If the
assumptions used in this calculation turn out not to be true, we
may pay more or less than the specified percentage of our actual
cash tax savings. This lump sum amount is subordinate to amounts
payable under our credit facilities and may be paid in cash or
be deferred until all amounts payable under our credit
facilities in existence as of the date of termination of the Tax
Receivable Agreement have been paid and the deferred amount will
bear interest at a rate of the lesser of 6.5 percent or
1-year LIBOR
plus 2.0 percent per annum. In view of the foregoing
changes in the calculation of our obligations, we do not expect
that the net impact of any such acceleration upon our overall
financial condition would be materially adverse as compared to
our obligations if laws do not change and the obligations are
not accelerated. It is also possible that the net impact of such
an acceleration would be beneficial to our overall financial
condition. The ultimate impact of a decision by the Tax
Receivable Entity to accelerate will depend on what the ongoing
payments would have been under the Tax Receivable Agreement
absent acceleration, which will depend on various factors.
We also have the right (with the consent of our independent
directors) to terminate the Tax Receivable Agreement. If we
exercise this right, then the Tax Receivable Entity would be
entitled to a lump sum amount in lieu
131
of the payments otherwise provided under the agreement. That
lump sum amount would be calculated by determining a present
value for the total amount that would otherwise be payable under
the agreement, using a discount rate and assumptions as to
income tax rates and as to our ability to utilize the tax
benefits (including the assumption that we will have sufficient
taxable income to fully utilize the tax benefits and the
assumption that all exchanges that have not taken place will
take place as of the date of the termination, which will
increase the amount of the lump sum payment). If the assumptions
used in this calculation turn out not to be true, we may pay
more or less than the specified percentage of certain cash tax
savings realized by us after the completion of this offering.
This lump sum amount must be paid in cash or be deferred until
all amounts payable under our credit facilities in existence as
of the date of termination of the Tax Receivable Agreement have
been paid. Any such acceleration can occur only at our election.
Should we elect to terminate the Tax Receivable Agreement, we do
not expect that the net impact of any such acceleration upon our
overall financial condition would be materially adverse as
compared to our existing obligations. The ultimate impact of a
decision by the Tax Receivable Entity to accelerate will depend
on what the ongoing payments would have been under the Tax
Receivable Agreement absent acceleration, which will in turn
depend on the various factors mentioned above.
If we default on any of our material obligations under the Tax
Receivable Agreement, then, unless the Tax Receivable Entity
seeks specific performance of the Tax Receivable Agreement, the
Tax Receivable Entity has the option to accelerate payments due
under the Tax Receivable Agreement and require us to make a lump
sum payment representing all past due and future payments under
the Tax Receivable Agreement, discounted to present value.
In addition, the Tax Receivable Agreement provides that, upon
certain mergers, asset sales or other forms of business
combination or certain other changes of control, our or our
successor’s obligations with respect to tax benefits would
be based on certain assumptions, including that we or our
successor would have sufficient taxable income to fully utilize
the deductions arising from the increased tax deductions and tax
basis and other benefits covered by the Tax Receivable
Agreement. As a result, upon a change of control, we could be
required to make payments under the Tax Receivable Agreement
that are greater than the specified percentage of our cash tax
savings.
Purchase
of Aurora Holdings Units and Class B Common Stock
Immediately following the completion of this offering, we will
use approximately
$ million
of the net proceeds that we will receive from this offering to
purchase Aurora Holdings Units
(and a corresponding number of shares of Class B common
stock) from certain of our Principal Equityholders.
See “Prospectus Summary — Reorganization
Transactions”, “Organizational Structure —
Reorganization Transactions and “Use of Proceeds.”
Related
Party Loans
On November 17, 2006, we entered into master promissory
notes with each of James C. New, our Chairman, Chief Executive
Officer and President, Martin J. Stefanelli, our Chief Operating
Officer, Vice President and Secretary, Michael Null, our Vice
President-Sales and Marketing, and Fred Ferrara, our Chief
Information Officer, under which these executive officers were
obligated to repay us, as of March 31, 2010, an amount in
aggregate of approximately $1.9 million plus accrued
interest. These arrangements were entered into to facilitate the
purchases of Class C Aurora Holdings Units by these
executive officers and were secured by a pledge of such units,
as evidenced by a pledge agreement between us and these
executive officers.
On March 7, 2008, we entered into promissory notes with
each of the same executive officers, under which these executive
officers were obligated to repay us, as of March 31, 2010,
an amount in aggregate of approximately $350,000 plus accrued
interest. These notes were secured by a pledge of Class X
capital of Aurora Holdings then held by each these executive
officers, which was evidenced by a pledge agreement between us
and these executive officers. These arrangements were entered
into to facilitate the purchases of Aurora Holdings’
Class X capital by these executive officers.
In accordance with the terms of these promissory notes, we
required the prepayment of all amounts due thereunder from our
executive officers in connection with or prior to this offering.
All such amounts were prepaid by the executive officers on
April 28, 2010.
132
In connection with our employment of Michael Null, our Vice
President-Sales and Marketing, we entered into a letter
agreement with Mr. Null in May 2008 and a related
promissory note issued by Mr. Null on October 21,
2008, in the aggregate amount of $100,000, to facilitate
Mr. Null’s relocation to our corporate headquarters.
Pursuant to the letter agreement, a portion of the principal and
interest under the note was forgiven each month, and
Mr. Null was entitled to a
gross-up for
taxes accruing as a result thereof. The balance of the note and
all accrued interest were forgiven on April 28, 2010.
On June 2, 2006, we entered into a former term loan
facility with a party related to a former member of Aurora
Holdings, GSO Capital Partners, with total available borrowings
up to $145.0 million. This term loan facility required the
related party to provide financing in connection with our
operations and certain business acquisitions and had a maturity
date of June 2, 2013. GSO Capital Partners extended credit
to us in the form of:
|
|
|
| |
•
|
term loans in aggregate principal amounts not in excess of
$140.0 million, and
|
|
|
|
| |
•
|
revolving loans, in aggregate principal amounts not in excess of
$5.0 million.
|
This former term loan facility was collateralized by
substantially all of Aurora Holdings’ assets and guaranteed
by all of its subsidiaries. Interest was paid in arrears at
LIBOR plus 3.75%. The interest rate was determined at the date
of funding the term loans and was fixed for a period of three to
six months at our option. After this initial interest rate
period, the loan converted to a variable rate note at the
greater of prime rate or the federal funds’ effective rate
plus 0.50% plus a factor of 2.75%. Principal payments on the
loans were due as net cash proceeds from either the sale of
assets or the sale of equity was available.
During 2007, we borrowed an additional $128.8 million under
this facility.
In December 2007, we entered into other former term loan
facilities with a syndicate of lenders providing for a loan
commitment up to $255.0 million. These former term loan
facilities provided, among other things, funds for the repayment
of the outstanding balance of the term loan facility with our
related party described above. In connection with our repayment
of the term loan facility with our related party, that loan
facility was terminated.
Registration
Rights Agreement
We are party to a Registration Rights Agreement with certain of
our Principal Equityholders. Under the Registration Rights
Agreement, our Principal Equityholders have certain registration
rights with respect to our Class A common stock.
Management
Services Agreement
On June 2, 2006, we, through a wholly-owned subsidiary of
Aurora Holdings, and two members of Aurora Holdings, Summit
Partners and GSO Capital Partners, entered into a Management
Services Agreement, or the Management Services Agreement. On
June 12, 2009, the Management Services Agreement was
amended to substitute KRG Capital Partners for GSO Capital
Partners. The Management Services Agreement called for the
members of Aurora Holdings and their affiliates to provide
certain financial and management advisory services in connection
with the general business planning and forecasting and
acquisition and divestiture strategies. In exchange for the
services, we paid an annual fee equal to 1.0% of revenues, plus
expenses to Aurora Holdings’ members. As of
December 31, 2008 and 2009, $0.6 million and
$0.4 million, respectively, of management fees under the
Management Services Agreement are reflected in accounts payable
and accrued expenses in the accompanying consolidated balance
sheets. The consolidated statement of operations includes
management fees of $0.6 million, $1.6 million and
$1.8 million for the respective years ended
December 31, 2007, 2008 and 2009. During 2007, 2008 and
2009, we paid management fees totaling $0.4 million,
$1.3 million and $1.9 million, respectively. The
Management Services Agreement will be terminated prior to the
completion of this offering.
Indemnification
of Officers and Directors
Our certificate of incorporation and bylaws provide that we will
indemnify each of our directors and officers to the fullest
extent permitted by the Delaware General Corporation Law.
133
Further, in connection with this offering, we expect to enter
into indemnification agreements with each of our directors and
officers, and we have purchased a policy of directors’ and
officers’ liability insurance that insures our directors
and officers against the cost of defense, settlement or payment
of a judgment under certain circumstances. For further
information, see “Management and Board of
Directors — Indemnification of Officers and Directors
and Limitation of Liability.”
Board of
Directors
Prior to the completion of this offering, entities affiliated
with the Summit Partners Equityholders had the right to appoint
three of our directors and the KRG Equityholders had the right
to appoint three of our directors. These rights terminate upon
the closing of this offering, and while the nominees of the
Summit Partners Equityholders and the KRG Equityholder will
remain on our Board of Directors following the completion of
this offering, we will have no contractual obligation to retain
them.
Related
Person Transaction Review Procedures
Our Board of Directors has adopted a written policy and
procedures, effective upon completion of this offering, for
review and approval or ratification of transactions involving us
and “related persons” (our directors and executive
officers and their immediate family members or our shareholders
owning five percent or greater of our outstanding Class A
common stock and their immediate family members). The policy
covers any related person transaction that meets the minimum
threshold for disclosure in a proxy statement under the
SEC’s rules. Upon completion of this offering, a copy of
our procedures may be found on our Web site, www.auroradx.com.
Related person transactions must be approved in advance by our
nominating and corporate governance committee whenever possible
or ratified as promptly as possible thereafter. We will disclose
in our proxy statement any transactions that are found to be
directly or indirectly material to a related person.
Prior to entering into a transaction with us, a related person
must provide the details of the transaction to our Chief
Financial Officer, including the relationship of the person to
us, the dollar amount involved, and whether the related person
or his or her family member has or will have a direct or
indirect interest in the transaction. Our Chief Financial
Officer will evaluate the transaction to determine if we or the
related person has a direct or indirect material interest in the
transaction. If so, then our Chief Financial Officer will notify
our Chief Executive Officer and submit the facts of the
transaction to our nominating and corporate governance committee
for its review. Our nominating and corporate governance
committee may approve a transaction only if these review
procedures have been followed and our nominating and corporate
governance committee determines that the transaction is not
detrimental to us and does not violate our policies.
134
DESCRIPTION
OF CAPITAL STOCK
The following description of our capital stock and provisions of
our certificate of incorporation and bylaws are summaries.
Because these are only summaries, they do not contain all the
information that may be important to you. For a complete
description, you should refer to our certificate of
incorporation and bylaws, copies of which have been filed with
the SEC as exhibits to our registration statement, as well as
the relevant portions of the Delaware General Corporation Law.
The description of our capital stock reflects changes to our
capital structure that will occur upon the completion of this
offering.
Capital
Stock
Our authorized capital stock consists
of shares
of Class A common stock, par value $0.01 per
share, shares
of Class B common stock, par value $0.01 per share,
and shares
of preferred stock, par value $0.01 per share. In connection
with the Reorganization Transactions, we will amend our
certificate of incorporation as part of the Reorganization
Transactions so as to provide for the appropriate number of
authorized shares of Class A common stock and Class B
common stock to complete this offering and the Reorganization
Transactions.
Immediately following the Reorganization Transactions, we will
have approximately holders of
record of our Class A common stock
and holders of record of our
Class B common stock. Of the authorized shares of our
capital stock, shares of our
Class A common stock will be issued and
outstanding, shares
of our Class B common stock will be issued and outstanding
and no shares of preferred stock will be issued and outstanding,
based on an assumed initial public offering price of
$ per share, which is the midpoint
of the price range set forth on the cover page of this
prospectus.
After the completion of this offering and the application of the
net proceeds that we will receive from this offering, we expect
to
have shares
of our Class A common stock
outstanding, shares
of our Class B common stock outstanding, and no shares of
preferred stock outstanding, based on an assumed initial public
offering price of $ per share,
which is the midpoint of the price range set forth on the cover
page of this prospectus.
Common
Stock
Voting.
Holders of our Class A common stock and Class B common
stock are entitled to one vote on all matters submitted to
stockholders for their vote or approval. The holders of our
Class A common stock and Class B common stock vote
together as a single class on all matters submitted to
stockholders for their vote or approval, except with respect to
the amendment of certain provisions of our certificate of
incorporation that would alter or change the powers, preferences
or special rights of the Class B common stock so as to
affect them adversely, which amendments must be approved by a
majority of the votes entitled to be cast by the holders of the
shares affected by the amendment, voting as a separate class, or
as otherwise required by applicable law.
Upon completion of this offering and the application of the net
proceeds that we will receive from this offering, our Principal
Equityholders will control
approximately percent of the
combined voting power of our common stock. Accordingly, our
Principal Equityholders can exercise significant influence over
our business policies and affairs and can control any action
requiring the general approval of our stockholders, including
the adoption of amendments to our certificate of incorporation
and bylaws and the approval of mergers or sales of substantially
all of our assets. The concentration of ownership and voting
power of our Principal Equityholders may also delay, defer or
even prevent an acquisition by a third party or other change of
control of our company and may make some transactions more
difficult or impossible without the support of our Principal
Equityholders, even if such events are in the best interests of
noncontrolling stockholders.
Dividends.
The holders of Class A common stock are entitled to receive
dividends when, as, and if declared by our Board of Directors
out of legally available funds. The holders of our Class B
common stock will not have any right to receive dividends other
than dividends consisting of shares of our Class B common
stock paid proportionally with respect to each outstanding share
of our Class B common stock.
135
Liquidation
or Dissolution.
Upon our liquidation or dissolution, the holders of our
Class A common stock will be entitled to share ratably in
those of our assets that are legally available for distribution
to stockholders after payment of liabilities and subject to the
prior rights of any holders of preferred stock then outstanding.
The holders of our Class B common stock will not have any
right to receive a distribution upon a liquidation or
dissolution of our company other than the par value of their
Class B common stock.
Transferability
and Exchange.
Subject to the terms of the Second Amended and Restated Aurora
Holdings LLC Agreement, the Aurora Holdings Continuing Members
may exchange their Aurora Holdings Units (and corresponding
shares of our Class B common stock) with ARDX Sub for
shares of our Class A common stock. Each such exchange will
be on a
one-for-one
basis, subject to customary conversion rate adjustments for
stock splits, stock dividends and reclassifications. In
connection with any proposed exchange by an Aurora Holdings
Continuing Member, we may, with the agreement of the exchanging
Aurora Holdings Continuing Member, elect to acquire (or cause
ARDX Sub to acquire) the applicable Aurora Holdings Units and
corresponding Class B common stock by paying cash in an
amount equal to the fair market value of the shares of
Class A common stock (determined by the volume weighted
average price of the shares of Class A common stock on the
date of the exchange) the member would have received in the
proposed exchange.
Other
Provisions.
None of the Class A common stock or Class B common
stock has any pre-emptive or other subscription rights. There
will be no redemption or sinking fund provisions applicable to
the Class A common stock or Class B common stock. Upon
the completion of this offering, all outstanding shares of
Class A common stock and Class B common stock will be
validly issued, fully paid and non-assessable. When no Aurora
Holdings Units remain exchangeable into shares of our
Class A common stock, our Class B common stock will be
cancelled.
Preferred
Stock
We are authorized to issue up
to shares
of preferred stock. Our Board of Directors is authorized,
subject to limitations prescribed by Delaware law and our
certificate of incorporation, to determine the terms and
conditions of the preferred stock, including whether the shares
of preferred stock will be issued in one or more series, the
number of shares to be included in each series and the powers,
designations, preferences and rights of the shares. Our Board of
Directors is also authorized to designate any qualifications,
limitations or restrictions on the shares without any further
vote or action by the stockholders. The issuance of preferred
stock may have the effect of delaying, deferring or preventing a
change in control of our company and may adversely affect the
voting and other rights of the holders of our Class A
common stock and Class B common stock, which could have an
adverse impact on the market price of our Class A common
stock. We have no current plan to issue any shares of preferred
stock following the completion of this offering.
Corporate
Opportunity
Our certificate of incorporation provides that the doctrine of
“corporate opportunity” will not apply against certain
of our Principal Equityholders, or any of our directors who are
employees of such Principal Equityholders, in a manner that
would prohibit them from investing in competing businesses or
doing business with our clients. In addition, under the Second
Amended and Restated Aurora Holdings LLC Agreement, we have
agreed that the Aurora Holdings Continuing Members
and/or one
or more of their respective affiliates are permitted to engage
in business activities or invest in or acquire businesses which
may compete with our business or do business with any client of
ours.
In the event that any Principal Equityholder acquires knowledge
of a potential transaction or matter which may be a corporate
opportunity for itself and us, we do not have any expectancy in
that corporate opportunity, and the Principal Equityholder does
not have any duty to communicate or offer that corporate
opportunity to us and may pursue or acquire that corporate
opportunity for itself or direct that opportunity to another
person. In addition, if a director or officer of our company who
is also a director, officer, member, manager or employee of any
Principal Equityholder acquires knowledge of a potential
transaction or matter which may be a corporate opportunity for
us
136
and a Principal Equityholder, we will not have any expectancy in
that corporate opportunity unless that corporate opportunity is
expressly offered to such person solely in his or her capacity
as a director or officer of our company.
In recognition that we may engage in material business
transactions with the Principal Equityholders
and/or one
or more of their respective affiliates from which we are
expected to benefit, our amended and restated certificate of
incorporation will provide that any of our directors or officers
who are also directors, officers, stockholders, members,
managers
and/or
employees of any Principal Equityholder will have fully
satisfied and fulfilled his or her fiduciary duty to us and our
stockholders with respect to such transaction, if:
|
|
|
| |
•
|
the transaction was approved, after being made aware of the
material facts of the relationship between us and the Principal
Equityholder and the material terms and facts of the
transaction, by (i) an affirmative vote of a majority of
the members of our Board of Directors who do not have a material
financial interest in the transaction (“Interested
Persons”) or (ii) an affirmative vote of a majority of
the members of a committee of our Board of Directors consisting
of members who are not Interested Person;
|
| |
| |
•
|
the transaction was fair to us at the time we entered into the
transaction; or
|
| |
| |
•
|
the transaction was approved by an affirmative vote of the
holders of a majority of shares of our common stock.
|
See “Risk Factors — We are controlled by our
Principal Equityholders whose interest in our business may be
different than yours, and certain statutory provisions afforded
to stockholders are not applicable to us.”
Anti-Takeover
Effects of Provisions of our Certificate of Incorporation and
Bylaws and Delaware Law
Some provisions of Delaware law, our certificate of
incorporation and our bylaws contain provisions that could make
the following transactions more difficult: an acquisition of us
by means of a tender offer; an acquisition of us by means of a
proxy contest or otherwise; or the removal of our incumbent
officers and directors. It is possible that these provisions
could make it more difficult to accomplish, or could deter,
transactions that stockholders may otherwise consider to be in
their best interest or in our best interests, including
transactions that might result in a premium over the market
price for our shares.
These provisions, summarized below, are intended to discourage
coercive takeover practices and inadequate takeover bids. These
provisions are also designed to encourage persons seeking to
acquire control of us to first negotiate with our Board of
Directors. We believe that the benefits of the increased
protection of our potential ability to negotiate with the
proponent of an unfriendly or unsolicited proposal to acquire or
restructure us outweigh the disadvantages of discouraging these
proposals because negotiation of these proposals could result in
an improvement of their terms.
Undesignated Preferred Stock. Our Board of
Directors has the ability to authorize undesignated shares of
our preferred stock, which allows our Board of Directors to
issue shares of our preferred stock with voting or other rights
or preferences that could impede the success of any unsolicited
attempt to change control of our company. This ability may have
the effect of deferring hostile takeovers or delaying changes in
control or management of our company.
Stockholder Meetings. Our bylaws provide that
a special meeting of stockholders may be called only by our
Chairman, Chief Executive Officer or President, or by a
resolution adopted by a majority of our Board of Directors.
Requirements for Advance Notification of Stockholder
Nominations and Proposals. Our bylaws establish
advance notice procedures with respect to stockholder proposals
and the nomination of candidates for election as directors,
other than nominations made by or at the direction of our Board
of Directors or a committee of our Board of Directors.
Elimination of Stockholder Action by Written
Consent. Our certificate of incorporation and
bylaws eliminate the right of stockholders to act by written
consent without a meeting.
Election and Removal of Directors. Our Board
of Directors is divided into three classes. The directors in
each class will serve for a three-year term, with one class
being elected each year by our stockholders. Once elected,
directors may be removed only for cause and only by the
affirmative vote of at least 66.67 percent of our
outstanding common stock. For more information, see
“Management and Board of Directors — Board
Composition and Election of Directors.” This system of
electing and removing directors may discourage a third party
from making a
137
tender offer or otherwise attempting to obtain control of us
because it generally makes it more difficult for stockholders to
replace a majority of the directors.
Delaware Anti-Takeover Statute. We have opted
out of Section 203 of the Delaware General Corporation Law,
which prohibits persons deemed “interested
stockholders” from engaging in a “business
combination” with a Delaware corporation for three years
following the date these persons become interested stockholders.
Generally, an “interested stockholder” is a person
who, together with affiliates and associates, owns, or within
three years prior to the determination of interested stockholder
status did own, 15 percent or more of a corporation’s
voting stock. Generally, a “business combination”
includes a merger, asset or stock sale, or other transaction
resulting in a financial benefit to the interested stockholder.
Amendment of Certain Provisions in Our Organizational
Documents. The amendment of any of the above
provisions, except for the provision relating to undesignated
shares of our preferred stock, would require approval by holders
of at least 66.67 percent of our then outstanding common
stock.
The provisions of the Delaware General Corporation Law and our
certificate of incorporation and bylaws could have the effect of
discouraging others from attempting hostile takeovers and, as a
consequence, they may also inhibit temporary fluctuations in the
market price of our Class A common stock that often result
from actual or rumored hostile takeover attempts. Such
provisions may also have the effect of preventing changes in our
management. It is possible that these provisions could make it
more difficult to accomplish transactions that stockholders may
otherwise deem to be in their best interests.
Limitations
of Liability and Indemnification Matters
We have adopted provisions in our certificate of incorporation
that limit the liability of our directors for monetary damages
for breach of their fiduciary duties, except for liability that
cannot be eliminated under the Delaware General Corporation Law.
Delaware law provides that directors of a corporation will not
be personally liable for monetary damages for breach of their
fiduciary duties as directors, except liability for any of the
following: any breach of their duty of loyalty to the
corporation or the stockholders; acts or omissions not in good
faith or that involve intentional misconduct or a knowing
violation of law; unlawful payments of dividends or unlawful
stock repurchases or redemptions as provided in Section 174
of the Delaware General Corporation Law; or any transaction from
which the director derived an improper personal benefit. This
limitation of liability does not apply to liabilities arising
under the federal securities laws and does not affect the
availability of equitable remedies such as injunctive relief or
rescission.
Our certificate of incorporation and bylaws also provide that we
will indemnify our directors and executive officers, and that we
may indemnify our other officers and employees and other agents,
to the fullest extent permitted by law. We believe that
indemnification under our bylaws covers at least negligence and
gross negligence on the part of indemnified parties. Our bylaws
also permit us to secure insurance on behalf of any officer,
director, employee or other agent for any liability arising out
of his or her actions in such capacity, regardless of whether
our bylaws would permit indemnification. Upon completion of this
offering, we expect that we will have entered into separate
indemnification agreements with our directors and executive
officers, in addition to the indemnification provided for in our
charter documents. These agreements, among other things, provide
for indemnification of our directors and executive officers for
expenses, judgments, fines and settlement amounts incurred by
any such person in any action or proceeding arising out of such
person’s services as a director or executive officer or at
our request.
Transfer
Agent and Registrar
The transfer agent and registrar for our Class A common
stock is .
NASDAQ
Global Market
We have applied to have our Class A common stock the NASDAQ
Global Market under the symbol ‘‘ARDX.”
138
SHARES ELIGIBLE
FOR FUTURE SALE
Prior to this offering, there has been no market for our
Class A common stock, and we cannot assure you that a
liquid trading market for our Class A common stock will
develop or be sustained after this offering. Future sales of
substantial amounts of our Class A common stock, in the
public market after this offering, or the anticipation of those
sales, could adversely affect market prices prevailing from time
to time and could impair our ability to raise capital through
sales of our equity securities. These factors could also make it
more difficult to raise funds through future offerings of
Class A common stock.
Sales of
Restricted Shares
Upon the completion of this offering, we will have outstanding
an aggregate of
approximately shares
of our Class A common stock, based on an assumed initial
public offering price of $ per
share, which is the midpoint of the price range set forth on the
cover page of this prospectus. Of these
shares, shares
of our Class A common stock to be sold in this offering,
or shares
if the underwriters exercise their over-allotment option in
full, will be freely tradable without restriction or further
registration under the Securities Act, unless the shares are
held by any of our affiliates, as that term is defined in
Rule 144 of the Securities Act. All remaining shares were
issued and sold by us in private transactions and are eligible
for public sale only if registered under the Securities Act or
sold in accordance with Rule 144 or Rule 701, each of
which is discussed below.
In addition, upon the completion of this offering and the
application of the net proceeds that we will receive from this
offering, the Aurora Holdings Continuing Members will own an
aggregate of Aurora Holdings Units
and shares of our
Class B common stock. Pursuant to the terms of the Second
Amended and Restated Aurora Holdings LLC Agreement and our
certificate of incorporation, the Aurora Holdings Continuing
Members could from time to time exchange their Aurora Holdings
Units (and corresponding shares of our Class B common
stock) with ARDX Sub for shares of our Class A common stock
on a
one-for-one
basis. In connection with any proposed exchange by an Aurora
Holdings Continuing Member, we may, in certain circumstances,
elect to acquire (or cause ARDX Sub to acquire) the applicable
Aurora Holdings Units and corresponding Class B common
stock by paying either:
|
|
|
| |
•
|
with the permission of the exchanging Aurora Holdings Continuing
Member, cash in an amount equal to the fair market value on the
date of the exchange (determined by the volume weighted average
price of the shares of Class A common stock on the date of
the exchange) of the shares of Class A common stock the
member would have received in the proposed exchange; or
|
|
|
|
| |
•
|
the number of shares of Class A common stock the member
would have received in the proposed exchange.
|
Shares of our Class A common stock issuable to the Aurora
Holdings Continuing Members upon an exchange of Aurora Holdings
Units would be considered “restricted securities,” as
that term is defined in Rule 144.
Except with respect to shares of our Class A common stock
offered by our selling stockholders in this offering, and Aurora
Holdings Units that we will purchase with the proceeds received
by us in the offering, including shares of our Class A
common stock to be sold by the selling stockholders and Aurora
Holdings Units that we will purchase if the underwriters
exercise their over-allotment option, all of our officers and
directors and substantially all of our stockholders are subject
to lock-up
agreements under which they have agreed not to transfer or
dispose of, directly or indirectly, any shares of our
Class A common stock or any securities convertible into or
exercisable or exchangeable for shares of our Class A
common stock (including Aurora Holdings Units), for a period of
180 days after the date of this prospectus, which is
subject to extension in some circumstances, as discussed below.
As a result of the
lock-up
agreements described below and the provisions of Rule 144
and Rule 701 under the Securities Act, the shares of our
Class A common stock (excluding the shares to be sold in
this offering) will be available for sale in the public market
as follows, based on an assumed initial public offering price of
$ per share, which is the midpoint
of the price range set forth on the cover page of this
prospectus:
|
|
|
| |
•
|
shares
will be eligible for sale on the date of this
prospectus; and
|
| |
| |
•
|
shares
will be eligible for sale as more particularly and except as
described below, beginning after expiration of the
lock-up
period pursuant to Rule 144 or Rule 701.
|
139
We expect the
remaining shares
to become eligible for future sale in the public market pursuant
to Rule 144 at varying times after 180 days from the
date of this prospectus.
Rule 144
In general, under Rule 144, beginning 90 days after
the date of this prospectus, a person who is not our affiliate
and has not been our affiliate for the previous three months,
and who has beneficially owned shares of our Class A common
stock for at least six months, may sell all such shares. An
affiliate or a person who has been our affiliate within the
previous 90 days, and who has beneficially owned shares of
our Class A common stock for at least six months, may sell
within any three-month period a number of shares that does not
exceed the greater of:
|
|
|
| |
•
|
one percent of the number of shares of our Class A
common stock then outstanding, which will equal
approximately shares
immediately after this offering; and
|
| |
| |
•
|
the average weekly trading volume of our Class A common
stock on the NASDAQ Global Market during the four calendar weeks
preceding the filing of a notice on Form 144 with respect
to the sale.
|
All sales under Rule 144 are subject to the availability of
current public information about us. Sales under Rule 144
by affiliates or persons who have been affiliates within the
previous 90 days are also subject to manner of sale
provisions and notice requirements. Upon completion of the
180-day
lock-up
period, subject to any extension of the
lock-up
period under circumstances described below,
approximately shares
our outstanding restricted securities will be eligible for sale
under Rule 144.
Rule 701
In general, under Rule 701 of the Securities Act, any of
our employees, consultants or advisors who purchased shares from
us in connection with a qualified compensatory stock plan or
other written agreement are eligible to resell those shares
90 days after the effective date of this offering in
reliance on Rule 144, but without compliance with the
holding period contained in Rule 144, and, in the case of
non-affiliates, without the availability of current public
information. Subject to the
lock-up
period,
approximately shares
of our Class A common stock will be eligible for sale in
accordance with Rule 701.
Lock-up
Agreements
We, the selling stockholders, all of our directors and officers
and the holders of
approximately percent of our
outstanding stock on a fully diluted basis immediately prior to
this offering have agreed to enter into
lock-up
agreements as described below under “Underwriters.”
Registration
Agreement
We are party to a Registration Agreement with certain of our
Principal Equityholders that grants registration rights to such
Principal Equityholders. Under certain circumstances, these
persons can require us to file registrations statements that
permit them to re-sell their shares. See “Certain
Relationships and Related Party Transactions —
Registration Agreement.”
140
CERTAIN
U.S. FEDERAL INCOME AND ESTATE TAX CONSEQUENCES TO
NON-U.S.
HOLDERS OF COMMON STOCK
General
The following is a general discussion of certain material
U.S. federal income and estate tax consequences of the
ownership and disposition of our Class A common stock that
may be relevant to you if you are a
non-U.S. holder
(as defined below) that acquires our Class A common stock
pursuant to this offering. This discussion is limited to
non-U.S. holders
who hold our Class A common stock as a capital asset within
the meaning of Section 1221 of the Internal Revenue Code of
1986, as amended, which we refer to as the Internal Revenue Code.
This discussion does not address all aspects of
U.S. federal income and estate taxation that may be
relevant to you in light of your particular circumstances, and
does not address any foreign, state or local tax consequences.
Furthermore, this discussion does not consider specific facts
and circumstances that may be relevant to a particular
non-U.S. holder’s
tax position, specific rules that may apply to certain
non-U.S. holders,
including banks, insurance companies, or other financial
institutions, partnerships or other pass-through entities,
holders of 10 percent or more of our common stock,
U.S. expatriates, dealers and traders in securities, or
special tax rules that may apply to a
non-U.S. holder
that holds our Class A common stock as part of a straddle,
hedge or conversion transaction. This discussion is based on
provisions of the Internal Revenue Code, Treasury regulations
and administrative and judicial interpretations as of the date
of this prospectus. All of these are subject to change, possibly
with retroactive effect, or different interpretations. If you
are considering buying our Class A common stock, you should
consult your own tax advisor about current and possible future
tax consequences of owning and disposing of our Class A
common stock in your particular situation.
For purposes of this discussion, a
“non-U.S. holder”
is a beneficial owner of our Class A common stock if that
person is any of the following for U.S. federal income tax
purposes:
|
|
|
| |
•
|
a nonresident alien individual within the meaning of
Section 7701(b) of the Internal Revenue Code;
|
| |
| |
•
|
a foreign corporation within the meaning of Section 7701(a)
of the Internal Revenue Code or other foreign entity taxable as
a foreign corporation under U.S. federal income tax
law; or
|
| |
| |
•
|
a foreign estate or trust within the meaning of
Section 7701(a) of the Internal Revenue Code.
|
If an entity treated as a partnership for U.S. federal
income tax purposes holds shares of our Class A common
stock, the tax treatment of a partner generally will depend on
the status of the partner and upon the activities of the
partnership. If you are a partner of a partnership holding
shares of our Class A common stock, we suggest you consult
your own tax advisor.
Distributions
If distributions are paid on the shares of our Class A
common stock, these distributions generally will constitute
dividends for U.S. federal income tax purposes to the
extent paid from our current or accumulated earnings and
profits, as determined under U.S. federal income tax
principles, and then will constitute a return of capital that is
applied against your tax basis in our Class A common stock
to the extent these distributions exceed those earnings and
profits. Distributions in excess of our current and accumulated
earnings and profits and your tax basis in our Class A
common stock (determined on a share by share basis) will be
treated as a gain from the sale or exchange of our Class A
common stock, the treatment of which is discussed below.
Dividends paid to a
non-U.S. holder
that are not effectively connected with the conduct of a
U.S. trade or business of the
non-U.S. holder
will be subject to U.S. federal withholding tax at a
30 percent rate or, if an income tax treaty applies and
certain information reporting requirements are satisfied, a
lower rate specified by the treaty.
Non-U.S. holders
should consult their tax advisors regarding their entitlement to
benefits under a relevant tax treaty.
The U.S. federal withholding tax generally is imposed on
the gross amount of a distribution, regardless of whether we
have sufficient earnings and profits to cause the distribution
to be a dividend for U.S. federal income tax purposes.
However, we may elect to withhold on less than the gross amount
of the distribution if we determine that the distribution is not
paid out of our current or accumulated earnings and profits,
based on our reasonable estimates.
141
A
non-U.S. holder
eligible for a reduced rate of U.S. federal withholding tax
under a tax treaty may establish entitlement to the benefit of a
reduced rate of withholding under such tax treaty by timely
filing a properly completed IRS
Form W-8BEN
(or a successor form) with us prior to the payment of a
dividend. A
non-U.S. holder
eligible for a reduced rate of U.S. federal withholding tax
under a tax treaty may obtain a refund of any excess amounts
withheld by filing an appropriate claim for a refund together
with the required information with the IRS.
Dividends that are effectively connected with a
non-U.S. holder’s
conduct of a trade or business within the United States and, if
an applicable tax treaty so provides, are also attributable to a
non-U.S. holder’s
U.S. permanent establishment, are exempt from
U.S. federal withholding tax if the
non-U.S. holder
furnishes to us or our paying agent a properly completed IRS
Form W-8ECI
(or successor form) containing the
non-U.S. holder’s
taxpayer identification number. However, dividends exempt from
U.S. federal withholding tax because they are
“effectively connected” or attributable to a
U.S. permanent establishment under an applicable tax treaty
are subject to U.S. federal income tax on a net income
basis at the regular graduated U.S. federal income tax
rates. Any such effectively connected dividends received by a
foreign corporation may, under certain circumstances, be subject
to an additional “branch profits tax” at a
30 percent rate or a lower rate if specified by an
applicable tax treaty.
Gain on
Disposition of our Class A Common Stock
A
non-U.S. holder
generally will not be subject to U.S. federal income tax or
withholding tax with respect to gain recognized on a sale or
other disposition of our Class A common stock unless one of
the following applies:
|
|
|
| |
•
|
The gain is effectively connected with a
non-U.S. holder’s
conduct of a trade or business within the United States and, if
an applicable tax treaty so provides, the gain is also
attributable to a
non-U.S. holder’s
U.S. permanent establishment. In such a case, unless an
applicable tax treaty provides otherwise, the
non-U.S. holder
generally will be taxed on its net gain derived from the sale at
regular graduated U.S. federal income tax rates, and in the
case of a foreign corporation, may also be subject to an
additional branch profits tax as described in “—
Distributions” above.
|
| |
| |
•
|
A
non-U.S. holder
who is an individual holds our Class A common stock as a
capital asset and is present in the United States for 183 or
more days in the taxable year of the sale or other disposition,
and certain other conditions are met. In such a case, the
non-U.S. holder
will be subject to a flat 30 percent tax on the gain
derived from the sale, which may be offset by certain
U.S. capital losses realized in the taxable year of the
sale or other disposition.
|
| |
| |
•
|
At any time during the shorter of the
5-year
period ending on the date of a sale or other disposition of our
stock or the period that the
non-U.S. holder
held our Class A common stock, our company is classified as
a United States Real Property Holding Corporation and, if our
Class A common stock is treated as “regularly traded
on an established securities market,” only if the
non-U.S. holder
owns or is treated as owning more than 5 percent of our
Class A common stock at any time within such period. A
United States Real Property Holding Corporation is generally
defined as a corporation, the fair market value of whose real
property interests equals or exceeds 50 percent of the fair
market value of its U.S. real property interests, its
interests in real property located outside the United States and
any other of its assets used or held for use in a trade or
business. Our company believes it is not and does not anticipate
becoming a United States Real Property Holding Corporation for
U.S. federal income tax purposes.
|
Information
Reporting and Backup Withholding Tax
We must report annually to the IRS and to each
non-U.S. holder
the amount of dividends paid to that holder and the tax withheld
with respect to those dividends. These information reporting
requirements apply even if withholding was not required.
Pursuant to an applicable tax treaty or other agreement, copies
of the information returns reporting those dividends and
withholding may also be made available to the tax authorities in
the country in which the
non-U.S. holder
resides.
Under certain circumstances, Treasury regulations require
information reporting and backup withholding (currently at a
rate of 28 percent), on certain payments on common stock. A
non-U.S. holder
of our Class A common stock that fails to certify its
non-U.S. holder
status in accordance with applicable Treasury regulations or
otherwise
142
establish an exemption may be subject to information reporting
and this backup withholding tax on payments of dividends.
Payment of the proceeds of a sale of our Class A common
stock by or through a U.S. office of a broker is subject to
both information reporting and backup withholding unless the
non-U.S. holder
certifies to the payor in the manner required as to its
non-U.S. status
under penalties of perjury or otherwise establishes an
exemption. As a general matter, information reporting and backup
withholding will not apply to a payment of the proceeds of a
sale of our Class A common stock by or through a foreign
office of a foreign broker effected outside the United States.
However, information reporting requirements, but not backup
withholding, will apply to payment of the proceeds of a sale of
our Class A common stock by or through a foreign office of
a broker effected outside the United States if that broker is:
|
|
|
| |
•
|
a U.S. person,
|
| |
| |
•
|
a foreign person that derives 50 percent or more of its
gross income for specified periods from the conduct of a trade
or business in the United States,
|
| |
| |
•
|
a “controlled foreign corporation” as defined in the
Internal Revenue Code, or
|
| |
| |
•
|
a foreign partnership that at any time during its tax year
either (1) has one or more U.S. persons that, in the
aggregate, own more than 50 percent of the income or
capital interests in the partnership or (2) is engaged in
the conduct of a trade or business in the United States.
|
Information reporting requirements will not apply to the payment
of the proceeds of a sale of our Class A common stock if
the broker receives a statement from the owner, signed under
penalty of perjury, certifying such owner’s
non-U.S. status
or an exemption is otherwise established (generally, such
certification is made on IRS Form W-8BEN).
Non-U.S. holders
should consult their own tax advisors regarding the application
of the information reporting and backup withholding rules to
them.
Amounts withheld under the backup withholding rules do not
constitute a separate U.S. federal income tax. Rather, any
amounts withheld under the backup withholding rules will be
refunded or allowed as a credit against the holder’s
U.S. federal income tax liability, if any, provided the
required information and appropriate claim for refund is timely
filed with the IRS.
Recently-Enacted Federal Tax Legislation
On March 18, 2010, President Obama signed the “Hiring
Incentives to Restore Employment (HIRE) Act,” or the HIRE
Act. The HIRE Act includes a revised version of a bill
introduced in late October 2009 in both the House and the
Senate, the “Foreign Account Tax Compliance Act of
2009” or the FATCA Bill.
Under the HIRE Act, foreign financial institutions (which
include hedge funds, private equity funds, mutual funds,
securitization vehicles and any other investment vehicles
regardless of their size) must comply with new information
reporting rules with respect to their U.S. account holders and
investors (which would include certain equity and debt holders
of such institutions, as well as certain account holders that
foreign entities with U.S. owners) or confront a new withholding
tax on U.S.-source payments made to them. A foreign financial
institution or other foreign entity that does not comply with
the HIRE Act’s reporting requirements generally will be
subject to a new 30 percent withholding tax with respect to any
“withholdable payments” made after December 31,
2012. For this purpose, withholdable payments are U.S.-source
payments otherwise subject to nonresident withholding tax
(including dividends paid on our Class A common stock) and
also include the entire gross proceeds from the sale of any
equity or debt instruments of U.S. issuers (including the gross
proceeds from the disposition of our Class A common stock).
The new HIRE Act’s withholding tax will apply regardless of
whether the payment would otherwise be exempt from U.S.
nonresident withholding tax (e.g., under the portfolio interest
exemption or as capital gain). The U.S. Treasury is authorized
to provide rules for implementing the HIRE Act’s
withholding regime with the existing nonresident withholding tax
rules. The HIRE Act also imposes new information reporting
requirements and increases related penalties for U.S. persons.
Absent any applicable exception, this legislation also generally
will impose a withholding tax of 30 percent on dividend income
from our Class A common stock paid to a foreign entity that
is not a foreign financial institution
143
unless such entity provides the withholding agent with a
certification identifying the substantial U.S. owners of
the entity, which generally includes any United States persons
who directly or indirectly own more than 10 percent of the
entity.
Withholding under the HIRE Act will not apply to withholdable
payments made directly to foreign governments, international
organizations, foreign central banks of issue and individuals,
and the U.S. Treasury is authorized to provide additional
exceptions.
As noted above, the new HIRE Act’s withholding and
information reporting requirements generally will apply to
withholdable payments made after December 31, 2012. You are
urged to consult with your tax advisors regarding these new
provisions.
U.S.
Federal Estate Tax
Common stock owned or treated as owned by an individual who is
not a citizen or resident, as defined for U.S. federal
estate tax purposes, of the United States at the time of death
will be included in that individual’s gross estate for
U.S. federal estate tax purposes and may be subject to
U.S. federal estate tax, unless an applicable estate tax
treaty provides otherwise.
The foregoing discussion is a summary of certain material
U.S. federal income and estate tax consequences of the
ownership and disposition of our Class A common stock by
non-U.S. holders.
You are urged to consult your own tax advisor with respect to
the particular tax consequences to you of ownership and
disposition of our Class A common stock, including the
effect of any state, local,
non-U.S. or
other tax laws.
144
UNDERWRITERS
Under the terms and subject to the conditions contained in an
underwriting agreement dated the date of this prospectus, the
underwriters named below, for whom Morgan Stanley &
Co. Incorporated, UBS Securities LLC, and Barclays Capital Inc.
are serving as the representatives and joint book-running
managers, have severally agreed to purchase, and the selling
stockholders have agreed to sell to them, severally, the number
of shares indicated below:
| |
|
|
|
|
|
Number of
|
|
Underwriter
|
|
Shares
|
|
|
|
Morgan Stanley & Co. Incorporated
|
|
|
|
UBS Securities LLC
|
|
|
|
Barclays Capital Inc.
|
|
|
|
RBC Capital Markets Corporation
|
|
|
|
BMO Capital Markets Corp.
|
|
|
|
Lazard Capital Markets LLC
|
|
|
|
Total
|
|
|
The underwriters and the representatives are collectively
referred to as the “underwriters” and the
“representatives,” respectively. The underwriters are
offering the shares of Class A common stock subject to
their acceptance of the shares from us and the selling
stockholders and subject to prior sale. The underwriting
agreement provides that the obligations of the several
underwriters to pay for and accept delivery of the shares of
Class A common stock offered by this prospectus are subject
to the approval of certain legal matters by their counsel and to
certain other conditions. The underwriters are obligated to take
and pay for all of the shares of Class A common stock
offered by this prospectus if any such shares are taken.
However, the underwriters are not required to take or pay for
the shares covered by the underwriters’ over-allotment
option described below. If an underwriter defaults, the
underwriting agreement provides that the purchase commitments of
the non-defaulting underwriters may be increased.
The underwriters initially propose to offer part of the shares
of Class A common stock directly to the public at the
initial public offering price listed on the cover page of this
prospectus and part to certain dealers at a price that
represents a concession not in excess of
$ a share under the initial public
offering price. Any underwriter may allow a concession not in
excess of $ a share to other
underwriters or to certain dealers. After the initial offering
of the shares of Class A common stock, the offering price
and other selling terms may from time to time be varied by the
representatives.
We and the selling stockholders have granted to the underwriters
an option, exercisable for 30 days from the date of this
prospectus, to purchase up
to
additional shares of our Class A common stock at the
initial public offering price listed on the cover page of this
prospectus, less underwriting discounts and commissions. The
underwriters may exercise this option solely for the purpose of
covering over-allotments, if any, made in connection with the
offering of the shares of Class A common stock offered by
this prospectus. To the extent the option is exercised, each
underwriter will become obligated, subject to certain
conditions, to purchase about the same percentage of the
additional shares of Class A common stock as the number
listed next to the underwriter’s name in the preceding
table bears to the total number of shares of Class A common
stock listed next to the names of all underwriters in the
preceding table. If the underwriters’ option is exercised
in full, the total price to the public would be
$ , the total underwriters’
discounts and commissions paid by us would be
$ , and the total proceeds to us
would be $ .
145
The following table shows the per share and total underwriting
discounts and commissions that we and the selling stockholders
are to pay to the underwriters in connection with this offering.
These amounts are shown assuming both no exercise and full
exercise of the underwriters’ option to purchase additional
shares of our Class A common stock.
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Per Share
|
|
|
Total
|
|
|
|
|
No
|
|
|
Full
|
|
|
No
|
|
|
Full
|
|
|
|
|
Exercise
|
|
|
Exercise
|
|
|
Exercise
|
|
|
Exercise
|
|
|
|
|
Underwriting discounts and commissions paid by us
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
Underwriting discounts and commissions paid by the selling
stockholders
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
Total
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
The expenses of this offering payable by us, not including
underwriting discounts and commissions, are estimated to be
approximately $ , which includes
legal, accounting and printing costs and various other fees
associated with the registration and listing of our Class A
common stock, giving effect to the reimbursement of certain
expenses by the selling stockholders and the underwriters. The
underwriters and the selling stockholders have agreed to
reimburse us for a portion of our expenses.
The underwriters have informed us and the selling stockholders
that they do not intend sales to discretionary accounts to
exceed five percent of the total number of shares of
Class A common stock offered by them.
We have applied to have our Class A common stock listed on
the NASDAQ Global Market under the symbol “ARDX.”
We, the selling stockholders, all of our directors and officers
and the holders of
approximately percent
of our outstanding Class A common stock and Aurora Holdings
Units on a fully diluted basis immediately prior to this
offering have agreed that, subject to certain exceptions,
without the prior written consent of Morgan Stanley &
Co. Incorporated, UBS Securities LLC, and Barclays Capital Inc.
on behalf of the underwriters, we and they will not, during the
period ending 180 days after the date of this prospectus:
|
|
|
| |
•
|
offer, pledge, sell, contract to sell, sell any option or
contract to purchase, purchase any option or contract to sell,
grant any option, right or warrant to purchase, lend, or
otherwise transfer or dispose of, directly or indirectly, any
shares of Class A common stock or any securities
convertible into or exercisable or exchangeable for Class A
common stock or any Aurora Holdings Units;
|
| |
| |
•
|
enter into any swap or other arrangement that transfers to
another, in whole or in part, any of the economic consequences
of ownership of the Class A common stock or any securities
convertible into or exercisable or exchangeable for the
Class A common stock or any Aurora Holdings Units; or
|
| |
| |
•
|
file any registration statement with the SEC relating to the
offering of any shares of Class A common stock or any
securities convertible into or exercisable or exchangeable for
Class A common stock or any Aurora Holdings Units;
|
whether any such transaction described in the first two bullet
points above is to be settled by delivery of Class A common
stock, Aurora Holdings Units or such other securities, in cash
or otherwise. In addition, we and each such person agrees that,
without the prior written consent of Morgan Stanley &
Co. Incorporated, UBS Securities LLC, and Barclays Capital Inc.
on behalf of the underwriters, we and they will not, during the
period ending 180 days after the date of this prospectus,
make any demand for, or exercise any right with respect to, the
registration of any shares of Class A common stock or any
security convertible into or exercisable or exchangeable for
Class A common stock.
146
Subject to certain exceptions, the
lock-up
restrictions described in the immediately preceding paragraph do
not apply to us or the holders referenced above, as follows:
|
|
|
| |
•
|
the sale of shares Class A common stock to the underwriters;
|
| |
| |
•
|
the issuance by us of shares of Class A common stock upon
the exercise of an option or warrant, or the conversion of a
security outstanding on the date of this prospectus, of which
the underwriters have been advised in writing;
|
| |
| |
•
|
transactions by such holders relating to shares of Class A
common stock, Aurora Holdings Units or other securities acquired
in open market transactions after the completion of the offering
of the shares;
|
| |
| |
•
|
the transfer of shares of Class A common stock or any
security convertible into or exercisable or exchangeable for
Class A common stock as a bona fide gift;
|
| |
| |
•
|
distributions by such holders of shares of Class A common
stock or any security convertible into Class A common stock
to limited partners or stockholders of the transferor and
transfers of shares of Class A common stock to an affiliate
(as defined under the Securities Act);
|
| |
| |
•
|
the transfer of shares of Class A common stock that occur
pursuant to a will, other testamentary document or applicable
laws of descent;
|
| |
| |
•
|
transfers of shares of Class A common stock pursuant to a
qualified domestic order or in connection with a divorce
settlement;
|
| |
| |
•
|
the establishment of a trading plan pursuant to
Rule 10b5-1
under the Exchange Act for the transfer of shares of our
Class A common stock, provided that such plan does not
provide for the transfer of Class A common stock during
this 180-day
restricted period;
|
| |
| |
•
|
any issuance or transfer in connection with the Reorganization
Transactions;
|
| |
| |
•
|
any sale of Aurora Holdings Units (along with a corresponding
number of shares of Class B common stock) to us for cash
using the net proceeds we will receive in this offering; or
|
| |
| |
•
|
any exchange of Aurora Holdings Units and a corresponding number
of shares of Class B common stock for shares of
Class A common stock, or exercise by us of our related
purchase right;
|
provided that, in the case of each of the fourth, fifth, sixth,
seventh and ninth types of transactions described above, each
recipient or transferee agrees to be subject to the restrictions
described in the preceding paragraph and that no filing under
Section 16(a) of the Exchange Act, reporting a change in
beneficial ownership of shares of Class A common stock, is
required or voluntarily made in connection with these
transactions during this
180-day
restricted period. In addition, in the case of each of the third
and eleventh types of transactions described above, no filing
under Section 16(a) of the Exchange Act is required or
voluntarily made in connection with these transactions during
this 180-restricted period.
Notwithstanding the foregoing, in the event that any
Class A common stock or Aurora Holdings Units held by
either the Summit Partners Equityholders or the KRG
Equityholders are released from such
lock-up
restrictions by the representatives, a pro rata portion of
shares of the Class A common stock or Aurora Holdings Units
held by the other of the Summit Partners Equityholders or the
KRG Equityholders will be immediately and fully released from
any remaining
lock-up
restrictions.
The 180-day
restricted period described in the preceding paragraphs will be
extended if:
|
|
|
| |
•
|
during the last 17 days of the
180-day
restricted period, we issue an earnings release or material news
or a material event relating to our company occurs; or
|
| |
| |
•
|
prior to the expiration of the
180-day
restricted period, we announce that we will release earnings
results during the
16-day
period beginning on the last day of the
180-day
period;
|
in which case the restrictions described in the preceding
paragraphs will continue to apply until the expiration of the
18-day
period beginning on the issuance of the earnings release or the
occurrence of the material news or material event.
147
In order to facilitate this offering of the Class A common
stock, the underwriters may engage in transactions that
stabilize, maintain or otherwise affect the price of the
Class A common stock. Specifically, the underwriters may
sell more shares than they are obligated to purchase under the
underwriting agreement, creating a short position. A short sale
is covered if the short position is no greater than the number
of shares available for purchase by the underwriters under the
over-allotment option. The underwriters can close out a covered
short sale by exercising the over-allotment option or purchasing
shares in the open market. In determining the source of shares
to close out a covered short sale, the underwriters will
consider, among other things, the open market price of shares
compared to the price available under the over-allotment option.
The underwriters may also sell shares in excess of the
over-allotment option, creating a naked short position. The
underwriters must close out any naked short position by
purchasing shares in the open market. A naked short position is
more likely to be created if the underwriters are concerned that
there may be downward pressure on the price of the Class A
common stock in the open market after pricing that could
adversely affect investors who purchase in this offering. In
addition, to stabilize the price of the Class A common
stock, the underwriters may bid for, and purchase, shares of
Class A common stock in the open market. Finally, the
underwriting syndicate may reclaim selling concessions allowed
to an underwriter or a dealer for distributing the Class A
common stock in this offering if the syndicate repurchases
previously distributed Class A common stock to cover
syndicate short positions or to stabilize the price of the
Class A common stock. These activities may raise or
maintain the market price of the Class A common stock above
independent market levels or prevent or retard a decline in the
market price of the Class A common stock. The underwriters
are not required to engage in these activities and may end any
of these activities at any time.
We, the selling stockholders and the underwriters have agreed to
indemnify each other against certain liabilities, including
liabilities under the Securities Act, or to contribute to
payments the underwriters may be required to make in connection
with such liabilities.
A prospectus in electronic format may be made available on the
websites maintained by one or more underwriters. The
underwriters may agree to allocate a number of shares to
underwriters for sale to their online brokerage account holders.
Internet distributions will be allocated by Morgan
Stanley & Co. Incorporated to underwriters that may
make Internet distributions on the same basis as other
allocations.
European
Economic Area
In relation to each Member State of the European Economic Area
which has implemented the Prospectus Directive, from and
including the date on which the Prospectus Directive is
implemented in that Member State, each representative and
underwriter has not made and will not make an offer of our
Class A common stock to the public in that Member State,
except that it may, with effect from and including such date,
make an offer of our Class A common stock to the public in
that Member State:
|
|
|
| |
•
|
at any time to legal entities which are authorized or regulated
to operate in the financial markets or, if not so authorized or
regulated, whose corporate purpose is solely to invest in
securities;
|
| |
| |
•
|
at any time to any legal entity which has two or more of
(1) an average of at least 250 employees during the
last financial year; (2) a total balance sheet of more than
€43,000,000 and (3) an annual net turnover of more
than €50,000,000, as shown in its last annual or
consolidated accounts; or
|
| |
| |
•
|
at any time in any other circumstances which do not require the
publication by us of a prospectus pursuant to Article 3 of
the Prospectus Directive.
|
For the purposes of the above, the expression an “offer of
our Class A common stock to the public” in relation to
any shares of Class A common stock in any Member State
means the communication in any form and by any means of
sufficient information on the terms of the offer and the
Class A common stock to be offered so as to enable an
investor to decide to purchase or subscribe shares of the
Class A common stock, as the same may be varied in that
Member State by any measure implementing the Prospectus
Directive in that Member State, and the expression Prospectus
Directive means Directive 2003/71/EC and includes any relevant
implementing measure in that Member State.
United
Kingdom
Each representative and underwriter has only communicated or
caused to be communicated and will only communicate or cause to
be communicated an invitation or inducement to engage in
investment activity (within the
148
meaning of Section 21 of the Financial Services and Markets
Act of 2000) in connection with the issue or sale of shares
of the Class A common stock in circumstances in which
Section 21(1) of such Act does not apply to us, and it has
complied and will comply with all applicable provisions of such
Act with respect to anything done by it in relation to any
shares of the Class A common stock in, from or otherwise
involving the United Kingdom.
Certain
Relationships
From time to time, certain of the underwriters
and/or their
respective affiliates have directly and indirectly engaged in
various financial advisory, investment banking and commercial
banking services for us and our affiliates, for which they
received customary compensation, fees and expense reimbursement.
In particular, affiliates of Morgan Stanley & Co.
Incorporated, UBS Securities LLC, Barclays Capital Inc.,
RBC Capital Markets Corporation and BMO Capital
Markets Corp., underwriters in this offering, are lenders under
our new credit facilities. Our new secured credit facilities
were negotiated on an arms’ length basis and contains
customary terms pursuant to which the lenders receive customary
fees. In addition, from time to time, certain of the
underwriters and their affiliates may effect transactions for
their own account or the account of customers, and hold on
behalf of themselves or their customers, long or short positions
in our debt or equity securities or loans, and may do so in the
future.
Lazard Frères & Co. referred this transaction to
Lazard Capital Markets LLC and will receive a referral fee from
Lazard Capital Markets LLC in connection with this referral.
Pricing
of the Offering
Prior to this offering, there has been no public market for our
Class A common stock. The initial public offering price
will be determined by negotiations among us, the selling
stockholders and the representatives of the underwriters. Among
the factors to be considered in determining the initial public
offering price will be our future prospects and those of our
industry in general, our sales, earnings and certain other
financial and operating information in recent periods, and the
price to earnings ratios, price to sales ratios and market
prices of securities and certain financial and operating
information of companies engaged in activities similar to ours.
The estimated initial public offering price range set forth on
the cover page of this preliminary prospectus is subject to
change as a result of market conditions and other factors.
LEGAL
MATTERS
Alston & Bird LLP will pass upon the legality of the
shares of our Class A common stock to be issued in this
offering. Shearman & Sterling LLP will pass upon legal
matters in connection with this offering on behalf of the
underwriters.
EXPERTS
The consolidated financial statements of Aurora Holdings, at
December 31, 2008 and 2009 and for the three years ended
December 31, 2009, appearing in this prospectus and
registration statement have been audited by
McGladrey & Pullen, LLP, independent registered public
accounting firm, as set forth in their report thereon appearing
elsewhere herein, and are included in reliance on their report
given on their authority as experts in accounting and auditing.
The consolidated financial statements of South Texas
Dermatopathology Laboratory, P.A. and Subsidiaries at
November 20, 2009 and for the period from January 1,
2009 through November 20, 2009 appearing in this prospectus
and registration statement have been audited by
McGladrey & Pullen, LLP, independent accountants, as
set forth in their report thereon appearing elsewhere herein,
and are included in reliance on their report given on their
authority as experts in accounting and auditing.
The financial statements of Twin Cities Dermatopathology, P.A.
at March 7, 2008 and December 31, 2007 and for the
period from January 1, 2008 through March 7, 2008 and
for the year ended December 31, 2007 appearing in this
prospectus and registration statement have been audited by
McGladrey & Pullen, LLP, independent
149
accountants, as set forth in their report thereon appearing
elsewhere herein, and are included in reliance on their report
given on their authority as experts in accounting and auditing.
The financial statements of Laboratory Medicine Consultants,
Ltd. at December 10, 2007 and for the period from
January 1, 2007 through December 10, 2007 appearing in
this prospectus and registration statement have been audited by
McGladrey & Pullen, LLP, independent accountants, as
set forth in their report thereon appearing elsewhere herein,
and are included in reliance on their report given on their
authority as experts in accounting and auditing.
The consolidated financial statements of Greensboro Pathology
Associates, P.A. and Subsidiary at October 4, 2007 and for
the period from January 1, 2007 through October 4,
2007 appearing in this prospectus and registration statement
have been audited by McGladrey & Pullen, LLP,
independent accountants, as set forth in their report thereon
appearing elsewhere herein, and are included in reliance on
their report given on their authority as experts in accounting
and auditing.
The consolidated financial statements of Mark &
Kambour, M.D., P.A. and Subsidiary at October 11, 2007
and for the period from January 1, 2007 through
October 11, 2007 appearing in this prospectus and
registration statement have been audited by
McGladrey & Pullen, LLP, independent accountants, as
set forth in their report thereon appearing elsewhere herein,
and are included in reliance on their report given on their
authority as experts in accounting and auditing.
The financial statements of Cunningham Pathology, LLC at
April 30, 2007 and for the period from January 1, 2007
through April 30, 2007 appearing in this prospectus and
registration statement have been audited by
McGladrey & Pullen, LLP, independent accountants, as
set forth in their report thereon appearing elsewhere herein,
and are included in reliance on their report given on their
authority as experts in accounting and auditing.
The financial statements of Pathology Solutions, LLC at
March 12, 2010 and for the period from January 1, 2010
through March 12, 2010 and for the years ended
December 31, 2009, 2008 and 2007 appearing in this
prospectus and registration statement have been audited by
McGladrey & Pullen, LLP, independent accountants, as set
forth in their report thereon appearing elsewhere herein, and
are included in reliance on their report given on their
authority as experts in accounting and auditing.
WHERE YOU
CAN FIND MORE INFORMATION
We have filed with the SEC a registration statement on
Form S-1
under the Securities Act with respect to our Class A common
stock offered hereby. This prospectus, which forms part of the
registration statement, does not contain all of the information
set forth in the registration statement and the exhibits and
schedules to the registration statement. Some items are omitted
in accordance with the rules and regulations of the SEC. For
further information about us and our Class A common stock,
we refer you to the registration statement and the exhibits and
schedules to the registration statement filed as part of the
registration statement. Statements contained in this prospectus
as to the contents of any contract or other document filed as an
exhibit are qualified in all respects by reference to the actual
text of the exhibit. You may read and copy the registration
statement, including the exhibits and schedules to the
registration statement, at the SEC’s Public Reference Room
at 100 F Street, N.E., Washington, D.C. 20549.
You can obtain information on the operation of the Public
Reference Room by calling the SEC at
1-800-SEC-0330.
In addition, the SEC maintains an Internet site at
www.sec.gov that contains reports, proxy and information
statements and other information regarding issuers that file
electronically with the SEC and from which you can
electronically access the registration statement, including the
exhibits and schedules to the registration statement.
As a result of the offering, we will become subject to the full
informational requirements of the Securities Exchange Act of
1934, as amended. We will fulfill our obligations with respect
to such requirements by filing periodic reports and other
information with the SEC. We intend to furnish our stockholders
with annual reports containing financial statements certified by
an independent registered public accounting firm. We also
maintain an Internet site at www.auroradx.com. Our
internet site is not a part of this prospectus.
150
INDEX TO
FINANCIAL STATEMENTS
Aurora
Diagnostics Holdings, LLC*
Consolidated Financial Statements
As of December 31, 2008 and 2009 and for the years ended
December 31, 2007, 2008 and 2009
| |
|
|
|
|
|
|
|
Page
|
|
|
|
|
|
|
F-4
|
|
|
|
|
|
F-5
|
|
|
|
|
|
F-6
|
|
|
|
|
|
F-7
|
|
|
|
|
|
F-8
|
|
|
|
|
|
F-9
|
|
Aurora
Diagnostics Holding, LLC*
Unaudited Consolidated Financial Statements
As of March 31, 2010 and for the three months ended
March 31, 2010
South
Texas Dermatopathology Laboratory, P.A. and Subsidiaries
Consolidated Financial Statements
| |
|
|
|
|
|
|
|
|
F-42
|
|
|
|
|
|
F-43
|
|
|
|
|
|
F-44
|
|
|
|
|
|
F-45
|
|
|
|
|
|
F-46
|
|
|
|
|
|
F-47
|
|
Twin
Cities Dermatopathology, P.A.
Financial Statements
| |
|
|
|
|
|
|
|
|
F-53
|
|
|
|
|
|
F-54
|
|
|
|
|
|
F-55
|
|
|
|
|
|
F-56
|
|
|
|
|
|
F-57
|
|
|
|
|
|
F-58
|
|
F-1
Laboratory
Medicine Consultants, LTD.
Financial Statements
| |
|
|
|
|
|
|
|
Page
|
|
|
|
|
|
|
F-62
|
|
|
|
|
|
F-63
|
|
|
|
|
|
F-64
|
|
|
|
|
|
F-65
|
|
|
|
|
|
F-66
|
|
|
|
|
|
F-67
|
|
Greensboro
Pathology Associates, P.A. and Subsidiary
Consolidated Financial Statements
| |
|
|
|
|
|
|
|
|
F-72
|
|
|
|
|
|
F-73
|
|
|
|
|
|
F-74
|
|
|
|
|
|
F-75
|
|
|
|
|
|
F-76
|
|
|
|
|
|
F-77
|
|
Mark &
Kambour, M.D., P.A. and Subsidiary
Consolidated Financial Statements
| |
|
|
|
|
|
|
|
|
F-82
|
|
|
|
|
|
F-83
|
|
|
|
|
|
F-84
|
|
|
|
|
|
F-85
|
|
|
|
|
|
F-86
|
|
|
|
|
|
F-87
|
|
Cunningham
Pathology, LLC
Financial Statements
F-2
Pathology
Solutions, LLC
Financial Statements
| |
|
|
|
|
|
|
|
|
F-99
|
|
|
|
|
|
F-100
|
|
|
|
|
|
F-101
|
|
|
|
|
|
F-102
|
|
|
|
|
|
F-103
|
|
|
|
|
|
|
|
|
|
|
|
F-108
|
|
|
|
|
|
* |
|
The financial statements of Aurora Diagnostics, Inc. have been
omitted as the entity has not commenced operations and has no
activities except in connection with its formation. The members
of Aurora Diagnostics Holdings, LLC will exchange their equity
interests for equity interests in Aurora Diagnostics, Inc. in
connection with the Company’s Reorganization Transactions
described in the Registration Statement. In accordance with
Article 3-05
of
Regulation S-X
and SAB Topic I.J, the financial statements of significant
acquisitions are presented in the Registration Statement and
include the required periods preceding the date of acquisition
by Aurora Diagnostics Holdings, LLC. |
F-3
Report of
Independent Registered Public Accounting Firm
To the Members
Aurora Diagnostic Holdings, LLC
We have audited the accompanying consolidated balance sheets of
Aurora Diagnostic Holdings, LLC as of December 31, 2009 and
2008, and the related consolidated statements of operations,
members’ equity and comprehensive income, and cash flows
for each of the three years in the period ended
December 31, 2009. Our audits also included the financial
statement schedule of Aurora Diagnostic Holdings, LLC. These
financial statements and financial statement schedule are the
responsibility of the Company’s management. Our
responsibility is to express an opinion on these financial
statements based on our audits.
We conducted our audits in accordance with the standards of the
Public Company Accounting Oversight Board (United States). Those
standards require that we plan and perform the audit to obtain
reasonable assurance about whether the financial statements are
free of material misstatement. An audit includes examining, on a
test basis, evidence supporting the amounts and disclosures in
the financial statements. An audit also includes assessing the
accounting principles used and significant estimates made by
management, as well as evaluating the overall financial
statement presentation. We believe that our audits provide a
reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred
to above present fairly, in all material respects, the financial
position of Aurora Diagnostic Holdings, LLC as of
December 31, 2009 and 2008 and the results of its
operations and its cash flows for each of the three years in the
period ended December 31, 2009, in conformity with
U.S. generally accepted accounting principles. Also, in our
opinion, the related financial statement schedule, when
considered in relation to the basic consolidated financial
statements taken as a whole, presents fairly in all material
respects the information set forth therein.
As discussed in Note 2 of the consolidated financial
statements, Aurora Diagnostics Holding, LLC changed its method
of accounting for business combinations during the year ended
December 31, 2009.
/s/ McGladrey & Pullen, LLP
Ft. Lauderdale, Florida
March 26, 2010
F-4
Aurora
Diagnostics Holdings, LLC
| |
|
|
|
|
|
|
|
|
|
|
|
December 31,
|
|
|
|
|
2008
|
|
|
2009
|
|
|
|
|
($ in thousands)
|
|
|
Assets
|
|
|
|
|
|
|
|
|
|
Current Assets
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
$
|
7,278
|
|
|
$
|
27,424
|
|
|
Accounts receivable, net
|
|
|
15,256
|
|
|
|
16,106
|
|
|
Prepaid expenses and other assets
|
|
|
1,894
|
|
|
|
2,031
|
|
|
Prepaid income taxes
|
|
|
—
|
|
|
|
133
|
|
|
Deferred tax assets
|
|
|
2,099
|
|
|
|
2,026
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current assets
|
|
|
26,527
|
|
|
|
47,720
|
|
|
|
|
|
|
|
|
|
|
|
|
Property and Equipment, net
|
|
|
6,702
|
|
|
|
7,580
|
|
|
|
|
|
|
|
|
|
|
|
|
Other Assets:
|
|
|
|
|
|
|
|
|
|
Deferred debt issue costs, net
|
|
|
5,022
|
|
|
|
3,932
|
|
|
Deposits and other noncurrent assets
|
|
|
215
|
|
|
|
17,149
|
|
|
Goodwill
|
|
|
250,340
|
|
|
|
271,725
|
|
|
Intangible assets, net
|
|
|
126,710
|
|
|
|
114,638
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
382,287
|
|
|
|
407,444
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
415,516
|
|
|
$
|
462,744
|
|
|
|
|
|
|
|
|
|
|
|
|
Liabilities and Members’ Equity
|
|
|
|
|
|
|
|
|
|
Current Liabilities
|
|
|
|
|
|
|
|
|
|
Current portion of long-term debt
|
|
$
|
10,010
|
|
|
$
|
11,596
|
|
|
Current portion of fair value of contingent consideration
|
|
|
—
|
|
|
|
804
|
|
|
Accounts payable and accrued expenses
|
|
|
3,042
|
|
|
|
4,850
|
|
|
Accrued compensation
|
|
|
5,319
|
|
|
|
7,124
|
|
|
Accrued interest
|
|
|
3,442
|
|
|
|
3,047
|
|
|
Current portion of acquisition related liability
|
|
|
1,456
|
|
|
|
592
|
|
|
Fair value of derivative
|
|
|
—
|
|
|
|
125
|
|
|
Income taxes payable
|
|
|
164
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current liabilities
|
|
|
23,433
|
|
|
|
28,138
|
|
|
Long-term debt, net of current portion
|
|
|
217,303
|
|
|
|
205,056
|
|
|
Acquisition related liability, net of current portion
|
|
|
656
|
|
|
|
—
|
|
|
Deferred tax liabilities, net
|
|
|
10,375
|
|
|
|
10,190
|
|
|
Fair value of contingent consideration, net of current portion
|
|
|
—
|
|
|
|
2,296
|
|
|
Fair value of derivative
|
|
|
2,573
|
|
|
|
—
|
|
|
Commitments and Contingencies
|
|
|
|
|
|
|
|
|
|
Members’ Equity
|
|
|
|
|
|
|
|
|
|
Member Contributions
|
|
|
|
|
|
|
|
|
|
Class A member units, 85,000 units issued and outstanding
|
|
$
|
146,250
|
|
|
$
|
146,250
|
|
|
Class A-1 member units, 21,382 units issued and outstanding as
of December 31, 2009 and no units issued and outstanding as
of December 31, 2008
|
|
|
—
|
|
|
|
50,282
|
|
|
Class B member units, 10,000 units issued and outstanding
|
|
|
(412
|
)
|
|
|
(2,333
|
)
|
|
Class C member units, 5,000 units issued and outstanding
|
|
|
1,870
|
|
|
|
1,870
|
|
|
Class D member units, 10,000 issued and outstanding
|
|
|
1,164
|
|
|
|
316
|
|
|
Class X, no member units authorized under LLC agreement
|
|
|
6,708
|
|
|
|
6,708
|
|
|
Accumulated Other Comprehensive Loss
|
|
|
(2,573
|
)
|
|
|
(125
|
)
|
|
Equity Transaction Costs
|
|
|
(1,750
|
)
|
|
|
(4,825
|
)
|
|
Retained Earnings
|
|
|
9,919
|
|
|
|
18,921
|
|
|
|
|
|
|
|
|
|
|
|
|
Total Members’ Equity
|
|
$
|
161,176
|
|
|
$
|
217,064
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
415,516
|
|
|
$
|
462,744
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Consolidated Financial Statements.
F-5
Aurora
Diagnostics Holdings, LLC
Years
Ended December 31, 2007, 2008 and 2009
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2007
|
|
|
2008
|
|
|
2009
|
|
|
|
|
($ in thousands)
|
|
|
|
|
Net Revenues
|
|
$
|
63,451
|
|
|
$
|
157,850
|
|
|
$
|
171,565
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating costs and expenses:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost of services
|
|
|
27,480
|
|
|
|
66,382
|
|
|
|
71,778
|
|
|
Selling, general and administrative expenses
|
|
|
15,172
|
|
|
|
33,194
|
|
|
|
36,854
|
|
|
Provision for doubtful accounts
|
|
|
2,378
|
|
|
|
8,037
|
|
|
|
9,488
|
|
|
Intangible asset amortization expense
|
|
|
5,721
|
|
|
|
14,308
|
|
|
|
14,574
|
|
|
Management fees
|
|
|
644
|
|
|
|
1,559
|
|
|
|
1,778
|
|
|
Impairment of goodwill and other intangible assets
|
|
|
—
|
|
|
|
—
|
|
|
|
8,031
|
|
|
Acquisition and business development costs
|
|
|
374
|
|
|
|
676
|
|
|
|
1,074
|
|
|
Equity based compensation expense
|
|
|
—
|
|
|
|
1,164
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total operating costs and expenses
|
|
|
51,769
|
|
|
|
125,320
|
|
|
|
143,577
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income from operations
|
|
|
11,682
|
|
|
|
32,530
|
|
|
|
27,988
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other income (expense):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest expense
|
|
|
(7,114
|
)
|
|
|
(21,577
|
)
|
|
|
(18,969
|
)
|
|
Write-off of deferred debt issue costs
|
|
|
(3,451
|
)
|
|
|
—
|
|
|
|
—
|
|
|
Other income
|
|
|
124
|
|
|
|
125
|
|
|
|
28
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total other expense, net
|
|
|
(10,441
|
)
|
|
|
(21,452
|
)
|
|
|
(18,941
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income before income taxes
|
|
|
1,241
|
|
|
|
11,078
|
|
|
|
9,047
|
|
|
Provision for income taxes
|
|
|
762
|
|
|
|
408
|
|
|
|
45
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income
|
|
$
|
479
|
|
|
$
|
10,670
|
|
|
$
|
9,002
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Consolidated Financial Statements.
F-6
Aurora
Diagnostics Holdings, LLC
Years
Ended December 31, 2007, 2008 and 2009
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Member
|
|
|
Other
|
|
|
Equity
|
|
|
Retained
|
|
|
Total
|
|
|
|
|
Member
|
|
|
Contributions
|
|
|
Comprehensive
|
|
|
Transaction
|
|
|
(Deficit)
|
|
|
Members’
|
|
|
|
|
Units
|
|
|
(Distributions)
|
|
|
Income (Loss)
|
|
|
Costs
|
|
|
Earnings
|
|
|
Equity
|
|
|
|
|
|
|
|
|
|
|
($ in thousands)
|
|
|
|
|
|
|
|
|
|
|
Balance, December 31, 2006
|
|
|
100,000
|
|
|
$
|
32,564
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
(1,230
|
)
|
|
$
|
31,334
|
|
|
Member contributions
|
|
|
—
|
|
|
|
116,705
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
116,705
|
|
|
Equity transaction costs
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(2,000
|
)
|
|
|
—
|
|
|
|
(2,000
|
)
|
|
Member notes receivable
|
|
|
—
|
|
|
|
(1,441
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(1,441
|
)
|
|
Net income
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
479
|
|
|
|
479
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, December 31, 2007
|
|
|
100,000
|
|
|
|
147,828
|
|
|
|
—
|
|
|
|
(2,000
|
)
|
|
|
(751
|
)
|
|
|
145,077
|
|
|
Member contributions
|
|
|
—
|
|
|
|
7,379
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
7,379
|
|
|
Adjustment to equity transaction costs
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
250
|
|
|
|
—
|
|
|
|
250
|
|
|
Equity compensation
|
|
|
10,000
|
|
|
|
1,164
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,164
|
|
|
Fair value of derivative
|
|
|
—
|
|
|
|
—
|
|
|
|
(2,573
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
(2,573
|
)
|
|
Member notes receivable
|
|
|
—
|
|
|
|
(379
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(379
|
)
|
|
Tax distributions
|
|
|
—
|
|
|
|
(412
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(412
|
)
|
|
Net income
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
10,670
|
|
|
|
10,670
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, December 31, 2008
|
|
|
110,000
|
|
|
|
155,580
|
|
|
|
(2,573
|
)
|
|
|
(1,750
|
)
|
|
|
9,919
|
|
|
|
161,176
|
|
|
Contributions from members
|
|
|
21,382
|
|
|
|
50,322
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
50,322
|
|
|
Equity transaction costs
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(3,075
|
)
|
|
|
—
|
|
|
|
(3,075
|
)
|
|
Fair value of derivative
|
|
|
—
|
|
|
|
—
|
|
|
|
2,448
|
|
|
|
—
|
|
|
|
—
|
|
|
|
2,448
|
|
|
Tax distributions
|
|
|
—
|
|
|
|
(2,809
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(2,809
|
)
|
|
Net income
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
9,002
|
|
|
|
9,002
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, December 31, 2009
|
|
|
131,382
|
|
|
$
|
203,093
|
|
|
$
|
(125
|
)
|
|
$
|
(4,825
|
)
|
|
$
|
18,921
|
|
|
$
|
217,064
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Consolidated Financial Statements.
F-7
Aurora
Diagnostics Holdings, LLC
Years
Ended December 31, 2007, 2008 and 2009
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2007
|
|
|
2008
|
|
|
2009
|
|
|
|
|
($ in thousands)
|
|
|
|
|
Cash Flows From Operating Activities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income
|
|
$
|
479
|
|
|
$
|
10,670
|
|
|
$
|
9,002
|
|
|
Adjustments to reconcile net income to net cash provided by
operating activities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Depreciation and amortization
|
|
|
6,386
|
|
|
|
16,137
|
|
|
|
17,060
|
|
|
Amortization of deferred debt issue costs
|
|
|
224
|
|
|
|
978
|
|
|
|
1,090
|
|
|
Amortization of original issue discount on debt
|
|
|
22
|
|
|
|
274
|
|
|
|
305
|
|
|
Deferred income taxes
|
|
|
(885
|
)
|
|
|
(1,094
|
)
|
|
|
(1,568
|
)
|
|
Equity based compensation
|
|
|
—
|
|
|
|
1,164
|
|
|
|
—
|
|
|
Write-off of deferred debt issue costs
|
|
|
3,451
|
|
|
|
—
|
|
|
|
—
|
|
|
Impairment of goodwill and other intangible assets
|
|
|
—
|
|
|
|
—
|
|
|
|
8,031
|
|
|
Changes in assets and liabilities, net of working capital
acquired in business combinations:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(Increase) decrease in:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts receivable
|
|
|
(113
|
)
|
|
|
(1,048
|
)
|
|
|
(287
|
)
|
|
Prepaid expenses
|
|
|
(1,110
|
)
|
|
|
(256
|
)
|
|
|
64
|
|
|
Increase (decrease) in:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts payable and accrued expenses
|
|
|
(1,481
|
)
|
|
|
401
|
|
|
|
1,815
|
|
|
Accrued compensation
|
|
|
(485
|
)
|
|
|
1,445
|
|
|
|
1,544
|
|
|
Accrued interest
|
|
|
1,796
|
|
|
|
1,566
|
|
|
|
(395
|
)
|
|
Taxes payable
|
|
|
1,543
|
|
|
|
(1,267
|
)
|
|
|
(298
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash provided by operating activities
|
|
|
9,827
|
|
|
|
28,970
|
|
|
|
36,363
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash Flows From Investing Activities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Purchase of property and equipment
|
|
|
(2,135
|
)
|
|
|
(2,746
|
)
|
|
|
(2,961
|
)
|
|
Increase in deposits and other noncurrent assets
|
|
|
(60
|
)
|
|
|
31
|
|
|
|
(16,934
|
)
|
|
Payment of contingent notes
|
|
|
(1,929
|
)
|
|
|
(12,531
|
)
|
|
|
(12,668
|
)
|
|
Businesses acquired, net of cash acquired
|
|
|
(299,357
|
)
|
|
|
(31,026
|
)
|
|
|
(16,698
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash used in investing activities
|
|
|
(303,481
|
)
|
|
|
(46,272
|
)
|
|
|
(49,261
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash Flows From Financing Activities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Borrowings under former term loan facility
|
|
|
128,835
|
|
|
|
—
|
|
|
|
—
|
|
|
Repayments under former term loan facility
|
|
|
(135,385
|
)
|
|
|
—
|
|
|
|
—
|
|
|
Repayments of subordinated notes payable
|
|
|
(327
|
)
|
|
|
(2,916
|
)
|
|
|
(3,045
|
)
|
|
Net borrowings under revolver
|
|
|
25
|
|
|
|
(24
|
)
|
|
|
(1
|
)
|
|
Borrowings under new term loan facilities
|
|
|
201,300
|
|
|
|
22,100
|
|
|
|
—
|
|
|
Repayments under new term loan facilities
|
|
|
—
|
|
|
|
(7,800
|
)
|
|
|
(8,200
|
)
|
|
Equity transaction costs
|
|
|
—
|
|
|
|
(1,750
|
)
|
|
|
(3,075
|
)
|
|
Payment of deferred debt issuance and public offering costs
|
|
|
(9,244
|
)
|
|
|
(176
|
)
|
|
|
(148
|
)
|
|
Contributions from members, net of tax distributions
|
|
|
115,264
|
|
|
|
6,588
|
|
|
|
47,513
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash provided by financing activities
|
|
|
300,468
|
|
|
|
16,022
|
|
|
|
33,044
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net increase (decrease) in cash
|
|
|
6,814
|
|
|
|
(1,280
|
)
|
|
|
20,146
|
|
|
Cash and cash equivalents, beginning
|
|
|
1,744
|
|
|
|
8,558
|
|
|
|
7,278
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents, ending
|
|
$
|
8,558
|
|
|
$
|
7,278
|
|
|
$
|
27,424
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Supplemental Disclosures of Cash Flow Information
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash interest payments
|
|
$
|
5,184
|
|
|
$
|
20,736
|
|
|
$
|
17,857
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash tax payments, including member tax distributions
|
|
$
|
163
|
|
|
$
|
3,325
|
|
|
$
|
4,577
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Supplemental Schedule of Noncash Investing and Financing
Activities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Notes receivable for membership interests
|
|
$
|
1,441
|
|
|
$
|
379
|
|
|
$
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Capital lease obligations
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
280
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Consolidated Financial Statements.
F-8
Aurora
Diagnostics Holdings, LLC
|
|
|
Note 1.
|
Nature of
Business and Significant Accounting Policies
|
Nature of Business: Aurora Diagnostics
Holdings, LLC and subsidiaries (the “Company”) was
organized in the State of Delaware as a limited liability
company on June 2, 2006 to operate as a diagnostic services
company. The practices provide physician-based general anatomic
and clinical pathology, dermapathology, molecular diagnostic
services and other esoteric testing services to physicians,
hospitals, clinical laboratories and surgery centers. The
Company’s operations consist of one reportable segment.
The Company operates in a highly regulated industry. The manner
in which licensed physicians can organize to perform and bill
for medical services is governed by state laws and regulations.
Businesses like the Company often are not permitted to employ
physicians or to own corporations that employ physicians or to
otherwise exercise control over the medical judgments or
decisions of physicians.
In states where the Company is not permitted to directly own a
medical services provider or for other commercial reasons, it
performs only non-medical administrative and support services,
does not represent to the public or its clients that it offers
medical services and does not exercise influence or control over
the practice of medicine. In those states, the Company conducts
business through entities that it controls, and it is these
affiliated entities that employ the physicians who practice
medicine. In such states, the Company generally enters into a
contract that restricts the owner of the affiliated entity from
transferring their ownership interests in the affiliated entity
and otherwise provides the Company or its designee with a
controlling voting or financial interest in the affiliated
entity and its laboratory operations. This controlling financial
interest generally is obtained pursuant to a long-term
management services agreement between the Company and the
affiliated entity. Under the management services agreement, the
Company exclusively manages all aspects of the operation other
than the provision of medical services. Generally, the
affiliated entity has no operating assets because the Company
acquired all of its operating assets at the time it acquired the
related laboratory operations. In accordance with the relevant
literature, these affiliated entities are included in the
consolidated financial statements of Aurora Diagnostics
Holdings, LLC.
Summary
of Significant Accounting Policies
Principles of Consolidation: The
accompanying consolidated financial statements of the Company
include the accounts of Aurora Diagnostics Holdings, LLC, its
wholly-owned subsidiaries and companies in which the Company has
a controlling financial interest by means other than the direct
record ownership of voting stock. All accounts and transactions
between the entities have been eliminated in consolidation.
Accounting Estimates: The preparation
of financial statements in conformity with accounting principles
generally accepted in the United States of America requires
management to make estimates and assumptions that affect the
reported amounts of assets and liabilities, disclosure of
contingent assets and liabilities and the reported amounts of
revenues and expenses. Due to the inherent uncertainties in this
process, actual results could differ from those estimates.
Fair Value of Financial Instruments: On
January 1, 2008, the Company adopted a new standard related
to the accounting for financial assets and liabilities and items
that are recognized or disclosed at fair value in the financial
statements on a recurring basis, at least annually.
On January 1, 2009, the Company adopted authoritative
guidance for its nonfinancial assets and liabilities that are
measured at fair value on a nonrecurring basis. The adoption of
the guidance did impact the Company’s disclosure
requirements related to the Company’s 2009 acquisition as
described in Note 2 and the impairment of the
Company’s intangible assets and goodwill described in
Note 5.
In August 2009, the Financial Accounting Standards Board
(“FASB”) issued an amendment to the accounting
standards related to the measurement of liabilities that are
recognized or disclosed at fair value. This standard clarifies
how a company should measure the fair value of liabilities and
that restrictions preventing the transfer of a liability should
not be considered as a factor in the measurement of liabilities
within the scope of this standard. This
F-9
Aurora
Diagnostics Holdings, LLC
Notes to Consolidated Financial Statements —
(Continued)
standard became effective October 1, 2009. The adoption of
this standard did not have a material impact on the
Company’s consolidated financial statements.
The fair value accounting standards clarify the definition of
fair value, prescribes methods for measuring fair value,
establishes a fair value hierarchy based on the inputs used to
measure fair value, and expands disclosures about fair value
measurements. The three-tier fair value hierarchy, which
prioritizes the inputs used in the valuation methodologies, is
described below with Level 1 having the highest priority
and Level 3 having the lowest.
Level 1: Quoted prices in active markets
for identical assets or liabilities.
Level 2: Quoted prices for similar assets
or liabilities in active markets, quoted prices for identical or
similar instruments in markets that are not active, and
model-derived valuations in which all significant inputs are
observable in active markets.
Level 3: Valuations derived from
valuation techniques in which one or more significant inputs are
unobservable.
Revenue Recognition and Accounts
Receivable: The Company recognizes revenue at
the time services are performed. Unbilled receivables are
recorded for services rendered during, but billed subsequent to,
the reporting period. Revenue is reported at the estimated
realizable amounts from patients, third-party payors and others
for services rendered. Revenue under certain third-party payor
agreements is subject to audit and retroactive adjustments.
Provisions for estimated third-party payor settlements and
adjustments are estimated in the period the related services are
rendered and adjusted in future periods as final settlements are
determined. The provision for doubtful accounts and the related
allowance are adjusted periodically, based upon an evaluation of
historical collection experience with specific payors for
particular services, anticipated collection levels with specific
payors for new services, industry reimbursement trends, and
other relevant factors. Changes in these factors in future
periods could result in increases or decreases in the
Company’s provision for doubtful accounts and impact its
results of operations, financial position and cash flows. In
2007, 2008 and 2009, approximately 28%, 28% and 25%,
respectively, of the Company’s consolidated net revenues
were generated by Medicare and Medicaid programs.
Segment Reporting: The Company operates
throughout the United States in one reportable segment, the
medical laboratory industry. Medical laboratories offer a broad
range of testing services to the medical profession. The
Company’s testing services are categorized based upon the
nature of the test: general anatomic pathology,
dermatopathology, molecular diagnostic services and other
esoteric testing services to physicians, hospitals, clinical
laboratories and surgery centers. Our revenues consist of
payments or reimbursements for these services. Revenues from
private insurance, including managed care organizations and
commercial payors, Medicare and Medicaid and physicians and
individual patients represented approximately 61%, 25%, and 14%,
respectively, of revenue for the year ended December 31,
2009.
Cash and Cash Equivalents: The Company
considers deposits and investments that have original maturities
of less than three months, when purchased, to be cash
equivalents. The Company maintains its cash balances at high
quality financial institutions. The Company’s balances in
its accounts may periodically exceed amounts insured by the
Federal Deposit Insurance Corporation, of up to $250,000 at
December 31, 2008 and 2009. The Company does not believe it
is exposed to any significant credit risk and has not
experienced any losses.
Property and Equipment: Property and
equipment is stated at cost. Routine maintenance and repairs are
charged to expense as incurred, while costs of betterments and
renewals are capitalized. Depreciation is calculated on a
straight-line basis, over the estimated useful lives of the
respective assets, which range from 3 to 15 years.
Leasehold improvements are amortized over the shorter of the
term of the related lease, or the useful life of the asset.
Goodwill: Goodwill represents the
excess of cost over the fair value of net tangible and
identifiable intangible assets acquired. The Company reviews
goodwill for impairment at the reporting unit level annually or,
when events or circumstances dictate, more frequently. The
impairment review for goodwill consists of a two- step process
of first determining the fair value of the reporting unit and
comparing it to the carrying value of the net assets
F-10
Aurora
Diagnostics Holdings, LLC
Notes to Consolidated Financial Statements —
(Continued)
allocated to the reporting unit. If the fair value of the
reporting unit exceeds the carrying value, no further analysis
or write-down of goodwill is required. If the fair value of the
reporting unit is less than the carrying value of the net
assets, the implied fair value of the reporting unit is
allocated to all the underlying assets and liabilities,
including both recognized and unrecognized tangible and
intangible assets, based on their fair value. If necessary,
goodwill is then written down to its implied fair value.
Intangible Assets: Intangible assets,
acquired as the result of a business combination, are recognized
at fair value, as an asset apart from goodwill if the asset
arises from contractual or other legal rights, or if it is
separable. Intangible assets, principally representing the fair
value of customer relationships, health care facility agreements
and non-competition agreements acquired, are capitalized and
amortized on the straight-line method over their expected useful
life, which generally ranges from 4 to 18 years.
Long-Lived Assets: The Company
recognizes impairment losses for long-lived assets when events
or changes in circumstances indicate the carrying amount of an
asset may not be recoverable. The Company continually assesses
whether an impairment in the carrying value of the intangible
assets has occurred. If the undiscounted future cash flows over
the remaining amortization period of an intangible asset
indicate the value assigned to the intangible asset may not be
recoverable, the Company reduces the carrying value of the
intangible asset. The Company would determine the amount of any
such impairment by comparing anticipated discounted future cash
flows from acquired businesses with the carrying value of the
related assets. In performing this analysis, the Company
considers such factors as current results, trends and future
prospects, in addition to other relevant factors.
Distributions to Members and Allocation of Profits and
Losses: Profits and losses are allocated to
the members in accordance with certain provisions contained in
the Company’s Amended and Restated Limited Liability
Company Agreement, dated June 12, 2009 (the “LLC
Agreement”). Distributions are also made in accordance with
the terms of the LLC Agreement.
Equity-Based Compensation: During 2008,
the Company adopted a new Equity Incentive Plan (the “New
Plan”) to replace the Company’s original Equity
Incentive Plan. The original Equity Incentive Plan for the grant
of membership options to key employees, directors and
consultants to purchase Class A membership interests was
cancelled upon adoption of the New Plan. No options had been
issued under the original plan. This New Plan provides awards of
membership interest units in the Company. These interests are
denominated as
Class D-1,
Class D-2,
and
Class D-3 units.
During 2008, the Company authorized and issued 4,000
D-1 units; 3,000 D-2 units; and 3,000 D-3 units.
Compensation expense for awards and related tax effects are
recognized as they vest. All membership interest units issued in
2008 were fully vested as of December 31, 2008.
The fair value of each membership interest unit granted is
valued using a Black-Scholes-Merton pricing model at the time
the units are granted. That amount is fully amortized at the
time the units vest. The valuation technique incorporates
assumptions for the expected volatility of the unit price, the
expected term of the unit, expected dividends, forfeitures and
risk-free interest rate for the expected term of the unit.
Expected volatility is based on historical volatility of an
appropriate industry sector index. An appropriate industry
sector index is one that is representative of the industry
sector in which the nonpublic entity operates. The expected term
of the unit is based on historical experience and represents the
time periods units actually remain outstanding. The risk-free
interest rate takes into account the time-value of money. The
risk-free rate for periods within the contractual life of the
option is based on the U.S. Treasury yield curve in effect
at time of grant. The Company estimated forfeitures based on
historical experience and shall revise this estimate in
subsequent periods if actual forfeitures differ from those
estimated.
F-11
Aurora
Diagnostics Holdings, LLC
Notes to Consolidated Financial Statements —
(Continued)
The fair value of each membership interest unit granted in 2008
was estimated using the following assumptions:
| |
|
|
|
|
|
2008
|
|
|
|
Expected life
|
|
3 years
|
|
Volatility percentage
|
|
20.2%
|
|
Interest rate
|
|
3.1%
|
|
Dividends
|
|
—
|
|
Forfeiture rate
|
|
—
|
There were no membership interest units granted or forfeited
during the year ended December 31, 2009.
Deferred Debt Issue Costs: During 2007,
the Company incurred approximately $2.9 million of debt
issue costs associated with the former term loan facility and
$0.4 million in costs paid to the former lender in
connection with the new term loan facility. In December 2007,
the Company obtained a new term loan facility and used a portion
of the proceeds to pay off and terminate the former term loan
facilities. As a result, the Company wrote off $3.5 million
of unamortized deferred debt issue costs.
During 2007 and 2008, the Company incurred approximately
$5.9 million and $0.2 million, respectively, of debt
issue costs in connection with its new term loan facilities.
These deferred debt issue costs are being amortized to interest
expense using the effective interest method. Amortization
expense was $0.2 million, $1.0 million and
$1.1 million for the years ended December 31, 2007,
2008 and 2009, respectively.
Deferred debt issue costs as of December 31, 2008 and 2009
consist of the following (in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
2008
|
|
|
2009
|
|
|
|
|
Deferred debt issue costs
|
|
$
|
6,080
|
|
|
$
|
6,080
|
|
|
Less accumulated amortization
|
|
|
(1,058
|
)
|
|
|
(2,148
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Deferred debt issue costs, net
|
|
$
|
5,022
|
|
|
$
|
3,932
|
|
|
|
|
|
|
|
|
|
|
|
Income Taxes: The Company is a Delaware
limited liability company for federal and state income tax
purposes, in accordance with the applicable provisions of the
Internal Revenue Code. Accordingly, the Company is generally not
subject to income taxes, the income attributable to the limited
liability company is distributed to the members in accordance
with the terms of the operating agreement. In addition, tax
distributions related to the income allocated to each member are
paid out quarterly. However, certain of the Company’s
subsidiaries are structured as corporations, file separate
returns, and therefore are subject to federal and state income
taxes. The provision for income taxes, for these subsidiaries,
is reflected in the Company’s consolidated financial
statements and includes federal and state taxes currently
payable and changes in deferred tax assets and liabilities
excluding the establishment of deferred tax assets and
liabilities related to the acquisitions. Deferred income taxes
represent the estimated future tax effects resulting from
temporary difference between financial statements carrying
values and the tax reporting basis of the related assets and
liabilities. The Company does not recognize a tax benefit,
unless the Company concludes that it is more likely than not
that the benefit will be sustained on audit by the taxing
authority based solely on the technical merits of the associated
tax position. If the recognition threshold is met, the Company
recognizes a tax benefit measured at the largest amount of the
tax benefit that the Company believes is greater than 50% likely
to be realized. The Company records interest and penalties in
income tax expense.
Derivative Financial Instruments: The
Company uses derivative financial instruments to manage its
interest rate risk. The Company records derivatives as either an
asset or liability measured at its fair value. The fair value is
based upon quoted market prices obtained from third-party
institutions. Changes in fair value of derivatives are recorded
each period in current earnings or other comprehensive income,
depending on whether a derivative is
F-12
Aurora
Diagnostics Holdings, LLC
Notes to Consolidated Financial Statements —
(Continued)
designated as part of a hedge transaction and, if it is, the
type of hedge transaction based on the specific qualifying
conditions as prescribed by the FASB standards on accounting for
derivative instruments and hedging activities. If it is
determined the derivative ceases to be a highly effective hedge,
the Company discontinues hedge accounting and any deferred gains
or losses are recorded in the consolidated statements of
operations.
Concentration of Credit Risk: Financial
instruments that potentially subject the Company to
concentrations of credit risk are cash and accounts receivable.
The Company’s policy is to place cash in highly-rated
financial institutions. Concentration of credit risk with
respect to accounts receivable is mitigated by the diversity of
the Company’s payers and their dispersion across many
different geographic regions. While the Company has receivables
due from federal and state governmental agencies, the Company
does not believe such receivables represent a credit risk since
the related healthcare programs are funded by federal and state
governments, and payment is primarily dependent on submitting
appropriate documentation.
Reclassifications: Certain prior year
amounts have been reclassified to conform to the 2009
presentation. The results of these reclassifications had no
effect on consolidated members’ equity or net income.
New Accounting Standards: On
January 1, 2009, the Company adopted a new accounting
standard issued by the FASB related to accounting for business
combinations using the acquisition method of accounting
(previously referred to as the purchase method). Among the
significant changes, this standard requires a redefining of the
measurement date of a business combination, expensing direct
transaction costs as incurred, capitalizing in-process research
and development costs as an intangible asset and recording a
liability for contingent consideration at the measurement date
with subsequent re-measurements recorded in the results of
operations. This standard also requires costs for business
restructuring and exit activities related to the acquired
company to be included in the post-combination financial results
of operations and also provides new guidance for the recognition
and measurement of contingent assets and liabilities in a
business combination. In addition, this standard requires
several new disclosures, including the reasons for the business
combination, the factors that contribute to the recognition of
goodwill, the amount of acquisition related third-party expenses
incurred, the nature and amount of contingent consideration, and
a discussion of pre-existing relationships between the parties.
The application of this standard was material for business
combinations completed in 2009, as further described in
Note 2. The standard is likely to have a significant effect
on the way the Company allocates the purchase price of certain
future business combinations, including the recognition and
measurement of assets acquired and liabilities assumed and the
expensing of direct transaction costs and costs to integrate the
acquired business.
On January 1, 2009, the Company adopted a new accounting
standard issued by the FASB related to the disclosure of
derivative instruments and hedging activities. This standard
expanded the disclosure requirements about an entity’s
derivative financial instruments and hedging activities,
including qualitative disclosures about objectives and
strategies for using derivatives, quantitative disclosures about
fair value amounts of and gains and losses on derivative
instruments, and disclosures about credit-risk-related
contingent features in derivative instruments.
Effective June 30, 2009, the Company adopted a newly issued
accounting standard related to accounting for and disclosure of
subsequent events in its consolidated financial statements. This
standard provides the authoritative guidance for subsequent
events that was previously addressed only in United States
auditing standards. This standard establishes general accounting
for and disclosure of events that occur after the balance sheet
date but before financial statements are issued or are available
to be issued and requires the Company to disclose that it has
evaluated subsequent events through the date of the filing or
issue date. This standard does not apply to subsequent events or
transactions that are within the scope of other applicable GAAP
that provide different guidance on the accounting treatment for
subsequent events or transactions. The adoption of this standard
did not have a material impact on the Company’s
consolidated financial statements.
In June 2009, the FASB issued the FASB Accounting Standards
Codification (the “ASC”). The ASC has become the
single source of non-governmental accounting principles
generally accepted in the United States of
F-13
Aurora
Diagnostics Holdings, LLC
Notes to Consolidated Financial Statements —
(Continued)
America (“GAAP”) recognized by the FASB in the
preparation of financial statements. The Company adopted the ASC
as of July 1, 2009. The ASC does not change GAAP and did
not have an effect on the Company’s financial position,
results of operations or cash flows.
In June 2009, the FASB issued new accounting guidance on when an
entity should be included in consolidated financial statements.
The new guidance amends the evaluation criteria to identify the
primary beneficiary of a variable interest entity, or
“VIE”, and requires ongoing reassessment of whether an
enterprise is the primary beneficiary of the VIE. The new
guidance significantly changes the consolidation rules for VIEs
including the consolidation of common structures, such as joint
ventures, equity-method investments, and collaboration
arrangements. The guidance is applicable to all new and existing
VIEs. This standard is effective for the Company as of
January 1, 2010 and the Company does not expect the impact
of its adoption to be material to its consolidated financial
statements.
In January 2010, the FASB issued an amendment to the accounting
standards related to the disclosures about an entity’s use
of fair value measurements. Among these amendments, entities
will be required to provide enhanced disclosures about transfers
into and out of the Level 1(fair value determined based on
quoted prices in active markets for identical assets and
liabilities) and Level 2 (fair value determined based on
significant other observable inputs) classifications, provide
separate disclosures about purchases, sales, issuances and
settlements relating to the tabular reconciliation of beginning
and ending balances of the Level 3 (fair value determined
based on significant unobservable inputs) classification and
provide greater disaggregation for each class of assets and
liabilities that use fair value measurements. Except for the
detailed Level 3 roll-forward disclosures, the new standard
is effective for the Company for interim and annual reporting
periods beginning after December 31, 2009. The requirement
to provide detailed disclosures about the purchases, sales,
issuances and settlements in the roll-forward activity for
Level 3 fair value measurements is effective for the
Company for interim and annual reporting periods beginning after
December 31, 2010. The Company does not expect that the
adoption of this new standard will have a material impact on its
consolidated financial statements.
Subsequent Events: The Company has
evaluated all subsequent events through the date of issuance of
the consolidated financial statements.
2007
Acquisitions
During 2007, the Company acquired substantially all of the
assets of three pathology practices, and 100% of the equity of
five pathology practices for an aggregate purchase price
(including acquisition costs) of $319.8 million and
additional consideration in the form of contingent notes. The
aggregate purchase price included cash of $306.1 million
and subordinated unsecured notes payable of $13.7 million.
The cash portion of the purchase price was funded primarily with
proceeds from member contributions and the Company’s
current and former term loan facilities of $115.3 million
and $190.8 million, respectively.
In connection with one acquisition in 2007, the Company agreed
to assume up to $4.0 million to be paid to four retired
physicians. The obligation is to be paid over three to five
years. As of December 31, 2009, this acquisition related
liability has a remaining balance of approximately
$0.6 million. During 2008 and 2009, the Company paid
$1.9 million and $1.5 million, respectively, related
to this liability.
2008
Acquisitions
During 2008, the Company acquired 100 percent of the equity
of one pathology practice, for an aggregate purchase price
(including acquisition costs) of $27.3 million and
additional consideration in the form of contingent notes. The
purchase price was funded primarily with proceeds from the
issuance of Class A, C, and X membership interests of
$7.3 million and the Company’s new term loan facility
of $20.0 million.
F-14
Aurora
Diagnostics Holdings, LLC
Notes to Consolidated Financial Statements —
(Continued)
During 2008, the Company finalized its allocation of the
purchase price for the 2008 acquisition and 2007 acquisitions
resulting in an adjustment to goodwill of approximately
$1.1 million related to professional fees and other
acquisition costs.
2009
Acquisitions
On December 31, 2009, the Company paid cash totaling
$17.0 million to acquire 100% of the equity of two
pathology practices. These acquisitions were consummated on
January 1, 2010 and therefore, the cash paid totaling
$17.0 million was included in Deposits and other
non-current assets in the accompanying consolidated balance
sheet as of December 31, 2009.
In November 2009, the Company acquired 100% of the equity of one
pathology practice for an aggregate cash purchase price of
$15.3 million. In addition, the Company issued contingent
consideration, payable over three years based on the
acquiree’s future performance. The Company has estimated
the fair value of the contingent consideration and recorded a
related liability in the accompanying consolidated balance
sheets as of December 31, 2009 of $3.1 million. The
cash portion of the purchase price was funded primarily with
proceeds from the issuance of
Class A-1
membership interests. The estimated fair value of the assets
acquired and liabilities assumed in connection with the 2009
acquisition are preliminary and are expected to be finalized in
2010.
On January 1, 2009, the Company adopted a new accounting
standard issued by the FASB related to accounting for business
combinations using the acquisition method of accounting
(previously referred to as the purchase method). In connection
with this adoption, during 2009, the Company has expensed
$0.6 million of transaction costs associated with its
completed acquisitions and business development costs in the
accompanying consolidated statements of operations.
Goodwill is calculated as the purchase premium after adjusting
for the fair value of net assets acquired and represents the
strategic benefits, including enhanced financial and operational
scale, market diversification, leveraged combined networks and
improved competitive positioning, arising from the integration
of the acquired entities.
The following table summarizes the consideration for the
acquisitions made in 2007, 2008 and 2009. Total consideration
for 2009 acquisitions excludes contingent consideration payable,
with a fair value of approximately $3.1 million (in
thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Date
|
|
Cash
|
|
|
Subordinated
|
|
|
Total
|
|
|
Location
|
|
Acquired
|
|
Paid
|
|
|
Notes Issued
|
|
|
Consideration
|
|
|
|
|
Michigan
|
|
February 2, 2007
|
|
$
|
16,319
|
|
|
$
|
—
|
|
|
$
|
16,319
|
|
|
Georgia
|
|
March 21, 2007
|
|
|
2,925
|
|
|
|
897
|
|
|
|
3,822
|
|
|
Alabama
|
|
April 30, 2007
|
|
|
43,320
|
|
|
|
12,761
|
|
|
|
56,081
|
|
|
Michigan
|
|
May 30, 2007
|
|
|
16,723
|
|
|
|
—
|
|
|
|
16,723
|
|
|
North Carolina
|
|
October 4, 2007
|
|
|
144,663
|
|
|
|
—
|
|
|
|
144,663
|
|
|
Florida
|
|
October 11, 2007
|
|
|
18,704
|
|
|
|
—
|
|
|
|
18,704
|
|
|
Nevada
|
|
December 10, 2007
|
|
|
49,336
|
|
|
|
—
|
|
|
|
49,336
|
|
|
Florida
|
|
December 10, 2007
|
|
|
14,126
|
|
|
|
—
|
|
|
|
14,126
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total 2007 Acquisitions
|
|
|
|
$
|
306,116
|
|
|
$
|
13,658
|
|
|
$
|
319,774
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Minnesota
|
|
March 7, 2008
|
|
|
27,301
|
|
|
|
—
|
|
|
|
27,301
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total 2008 Acquisitions
|
|
|
|
$
|
27,301
|
|
|
$
|
—
|
|
|
$
|
27,301
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Texas
|
|
November 20, 2009
|
|
|
15,340
|
|
|
|
—
|
|
|
|
15,340
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total 2009 Acquisitions
|
|
|
|
$
|
15,340
|
|
|
$
|
—
|
|
|
$
|
15,340
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
F-15
Aurora
Diagnostics Holdings, LLC
Notes to Consolidated Financial Statements —
(Continued)
Contingent
Consideration
In connection with the acquisitions, the Company has agreed to
pay additional consideration in future periods, based upon the
attainment of stipulated levels of operating earnings by each of
the acquired entities, as defined in their respective
agreements. For all acquisitions prior to January 1, 2009,
the Company does not accrue contingent consideration obligations
prior to the attainment of the objectives and the amount owed
becomes fixed and determinable. For the years ended
December 31, 2007, 2008 and 2009, the Company paid
consideration under contingent notes of $1.9 million,
$12.5 million and $12.7 million, respectively.
As of December 31, 2009, assuming the practices, including
the one acquired in November 2009, achieve the maximum level of
stipulated operating earnings, the maximum principal amount of
contingent consideration payable, over the next three to five
years is approximately $101 million. A lesser amount will
be paid for earnings below the maximum level of stipulated
earnings or no payments will be made if the practices do not
achieve the minimum level of stipulated earnings as outlined in
their respective agreements. Any such payments in the future for
acquisitions completed prior to January 1, 2009 would be
accounted for as additional purchase price and increase
goodwill. For acquisitions completed during 2009, future
payments will be reflected in the change in the fair value of
the contingent consideration.
The following tables summarize the estimated aggregate fair
value of the assets acquired and liabilities assumed in
connection with the acquisitions in 2008 and 2009 (in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
2008
|
|
|
2009
|
|
|
|
|
Cash
|
|
$
|
531
|
|
|
$
|
162
|
|
|
Accounts receivable
|
|
|
622
|
|
|
|
563
|
|
|
Other assets
|
|
|
23
|
|
|
|
73
|
|
|
Property and equipment
|
|
|
108
|
|
|
|
125
|
|
|
Intangible assets
|
|
|
9,610
|
|
|
|
3,922
|
|
|
Goodwill
|
|
|
17,275
|
|
|
|
15,327
|
|
|
|
|
|
|
|
|
|
|
|
|
Assets acquired
|
|
|
28,169
|
|
|
|
20,172
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts payable and accrued expenses
|
|
|
254
|
|
|
|
12
|
|
|
Accrued compensation
|
|
|
614
|
|
|
|
264
|
|
|
Fair value of contingent consideration
|
|
|
—
|
|
|
|
3,100
|
|
|
Deferred tax liabilities
|
|
|
—
|
|
|
|
1,456
|
|
|
|
|
|
|
|
|
|
|
|
|
Liabilities assumed
|
|
|
868
|
|
|
|
4,832
|
|
|
|
|
|
|
|
|
|
|
|
|
Net assets acquired
|
|
$
|
27,301
|
|
|
$
|
15,340
|
|
|
|
|
|
|
|
|
|
|
|
|
Net assets acquired
|
|
$
|
27,301
|
|
|
$
|
15,340
|
|
|
Less:
|
|
|
|
|
|
|
|
|
|
Cash acquired
|
|
|
(531
|
)
|
|
|
(162
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Cash paid for acquisitions, net of cash acquired
|
|
|
26,770
|
|
|
|
15,178
|
|
|
Acquisition costs and acquisition-related liability paid
|
|
|
4,256
|
|
|
|
1,520
|
|
|
|
|
|
|
|
|
|
|
|
|
Total businesses acquired and related costs, net of cash acquired
|
|
$
|
31,026
|
|
|
$
|
16,698
|
|
|
|
|
|
|
|
|
|
|
|
F-16
Aurora
Diagnostics Holdings, LLC
Notes to Consolidated Financial Statements —
(Continued)
Pro-Forma
Information (Unaudited)
The accompanying consolidated financial statements include the
results of operations of the acquisitions from the date acquired
through December 31, 2009.
The following unaudited pro forma information presents the
consolidated results of the Company’s operations and the
results of the 2009 acquisition for the years ended
December 31, 2008 and 2009, after giving effect to
amortization, depreciation, interest, income tax, and the
reduced level of certain specific operating expenses (primarily
compensation and related expenses attributable to former owners)
as if the acquisition had been consummated on January 1,
2008. Such unaudited pro forma information is based on
historical unaudited financial information with respect to the
2009 acquisition and does not include operational or other
changes which might have been effected by the Company. The
unaudited pro forma information for the years ended
December 31, 2008 and 2009 presented below is for
illustrative purposes only and is not necessarily indicative of
results which would have been achieved or results which may be
achieved in the future (in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
Pro Forma December 31,
|
|
|
|
2008
|
|
2009
|
|
|
|
Net Revenues
|
|
$
|
165,282
|
|
|
$
|
178,849
|
|
|
|
|
|
|
|
|
|
|
|
|
Net Income
|
|
$
|
11,847
|
|
|
$
|
10,662
|
|
|
|
|
|
|
|
|
|
|
|
The following unaudited pro forma information presents the
consolidated results of the Company’s operations and the
results of the 2008 acquisition for the years ended
December 31, 2007 and 2008, after giving effect to
amortization, depreciation, interest, income tax, and the
reduced level of certain specific operating expenses (primarily
compensation and related expenses attributable to former owners)
as if the acquisition had been consummated on January 1,
2007. Such unaudited pro forma information is based on
historical unaudited financial information with respect to the
2008 acquisition and does not include operational or other
changes which might have been effected by the Company. The
unaudited pro forma information for the years ended
December 31, 2007 and 2008 presented below is for
illustrative purposes only and is not necessarily indicative of
results which would have been achieved or results which may be
achieved in the future (in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
Pro Forma December 31,
|
|
|
|
2007
|
|
2008
|
|
|
|
Net Revenues
|
|
$
|
72,032
|
|
|
$
|
159,252
|
|
|
|
|
|
|
|
|
|
|
|
|
Net Income
|
|
$
|
3,041
|
|
|
$
|
11,076
|
|
|
|
|
|
|
|
|
|
|
|
F-17
Aurora
Diagnostics Holdings, LLC
Notes to Consolidated Financial Statements —
(Continued)
|
|
|
Note 3.
|
Property
and Equipment
|
Property and equipment as of December 31, 2008 and 2009
consists of the following (in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Estimated Useful
|
|
|
|
|
|
|
|
|
|
|
Life (Years)
|
|
|
2008
|
|
|
2009
|
|
|
|
|
Laboratory, office and data processing equipment
|
|
|
5
|
|
|
$
|
4,026
|
|
|
$
|
5,524
|
|
|
Building and leasehold improvements
|
|
|
5 – 15
|
|
|
|
2,626
|
|
|
|
3,009
|
|
|
Furniture and fixtures
|
|
|
5
|
|
|
|
478
|
|
|
|
550
|
|
|
Software
|
|
|
3
|
|
|
|
1,714
|
|
|
|
2,504
|
|
|
Vehicles
|
|
|
3
|
|
|
|
331
|
|
|
|
431
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
9,175
|
|
|
|
12,018
|
|
|
Less accumulated depreciation
|
|
|
|
|
|
|
(2,506
|
)
|
|
|
(4,993
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
6,669
|
|
|
|
7,025
|
|
|
Construction in progress
|
|
|
|
|
|
|
33
|
|
|
|
555
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
6,702
|
|
|
$
|
7,580
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Depreciation expense was $0.7 million, $1.8 million
and $2.5 million for the years ended December 31,
2007, 2008 and 2009, respectively.
|
|
|
Note 4.
|
Accounts
Receivable
|
Accounts receivable consist of the following as of
December 31, 2008 and 2009 (in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
2008
|
|
|
2009
|
|
|
|
|
Accounts receivable
|
|
$
|
23,453
|
|
|
$
|
24,659
|
|
|
Less: Allowance for doubtful accounts
|
|
|
(8,197
|
)
|
|
|
(8,553
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Accounts receivable, net
|
|
$
|
15,256
|
|
|
$
|
16,106
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Note 5.
|
Goodwill
and Intangible Assets
|
The following table presents adjustments to goodwill during 2008
and 2009 (in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
2008
|
|
|
2009
|
|
|
|
|
Goodwill, beginning of period
|
|
$
|
219,429
|
|
|
$
|
250,340
|
|
|
Acquisitions
|
|
|
17,275
|
|
|
|
15,327
|
|
|
Contingent notes*
|
|
|
12,531
|
|
|
|
12,668
|
|
|
Goodwill impairment
|
|
|
—
|
|
|
|
(6,610
|
)
|
|
Other acquisition costs
|
|
|
1,105
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
Goodwill, end of period
|
|
$
|
250,340
|
|
|
$
|
271,725
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
*
|
|
Related to acquisitions completed
prior to January 1, 2009.
|
F-18
Aurora
Diagnostics Holdings, LLC
Notes to Consolidated Financial Statements —
(Continued)
Intangible assets as of December 31, 2008 and 2009 and the
related accumulated amortization are set forth in the table
below (in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Average
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Amortization
|
|
|
December 31, 2008
|
|
|
|
|
Range
|
|
Period
|
|
|
|
|
|
Accumulated
|
|
|
|
|
|
|
|
(Years)
|
|
(Years)
|
|
|
Cost
|
|
|
Amortization
|
|
|
Net
|
|
|
|
|
Amortizing intangible assets:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Customer relationships
|
|
7 – 10
|
|
|
9
|
|
|
$
|
102,810
|
|
|
$
|
(14,796
|
)
|
|
$
|
88,014
|
|
|
Health care facility agreements
|
|
4 – 18
|
|
|
15
|
|
|
|
41,370
|
|
|
|
(4,642
|
)
|
|
|
36,728
|
|
|
Noncompete agreements
|
|
5
|
|
|
5
|
|
|
|
3,030
|
|
|
|
(1,062
|
)
|
|
|
1,968
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total intangible assets
|
|
|
|
|
|
|
|
$
|
147,210
|
|
|
$
|
(20,500
|
)
|
|
$
|
126,710
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Average
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Amortization
|
|
|
December 31, 2008
|
|
|
|
|
Range
|
|
Period
|
|
|
|
|
|
Accumulated
|
|
|
|
|
|
|
|
(Years)
|
|
(Years)
|
|
|
Cost
|
|
|
Amortization
|
|
|
Net
|
|
|
|
|
Amortizing intangible assets:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Customer relationships
|
|
7 – 10
|
|
|
9
|
|
|
$
|
103,089
|
|
|
$
|
(23,414
|
)
|
|
$
|
79,675
|
|
|
Health care facility agreements
|
|
4 – 18
|
|
|
14
|
|
|
|
41,370
|
|
|
|
(7,951
|
)
|
|
|
33,419
|
|
|
Noncompete agreements
|
|
4 – 5
|
|
|
5
|
|
|
|
3,098
|
|
|
|
(1,554
|
)
|
|
|
1,544
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total intangible assets
|
|
|
|
|
|
|
|
$
|
147,557
|
|
|
$
|
(32,919
|
)
|
|
$
|
114,638
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Amortization expense related to intangible assets was
$5.7 million, $14.3 million and $14.6 million for
the years ended December 31, 2007, 2008 and 2009,
respectively.
For purposes of testing goodwill for impairment each of the
Company’s acquired practices is considered a separate
reporting unit. To estimate the fair value of the reporting
units, the Company utilizes a discounted cash flow model as the
primary approach to value supported by a market approach
guideline public company method (the “GPC Method”)
which is used as a reasonableness test. The Company believes
that a discounted cash flow analysis is the most appropriate
methodology to test the recorded value of long-term assets with
a demonstrated long-lived value. The results of the discounted
cash flow provide reasonable estimates of the fair value of the
reporting units because this approach is based on each
respective unit’s actual results and reasonable estimates
of future performance, and also takes into consideration a
number of other factors deemed relevant by management, including
but not limited to, expected future market revenue growth and
operating profit margins. The Company has consistently used
these approaches in determining the value of goodwill. The
Company considers the GPC Method as an adequate reasonableness
test which utilizes market multiples of industry participants to
corroborate the discounted cash flow analysis. The Company
believes this methodology is consistent with the approach that
any strategic market participant would utilize if they were to
value one of the Company’s reporting units.
The following assumptions were made by management in determining
the fair value of the reporting units and related intangibles as
of September 30, 2009: (a) the discount rates ranged
between 13.0% and 15.0%, based on relative size and perceived
risk of the reporting unit (b) an average compound annual
growth rate (“CAGR”) of 7.5% during the five year
forecast period; and (c) earnings before interest, taxes,
depreciation, and amortization (“EBITDA”), with an
average reporting unit level margin of 38.9%. These assumptions
are based on: (a) the
F-19
Aurora
Diagnostics Holdings, LLC
Notes to Consolidated Financial Statements —
(Continued)
actual historical performance of the reporting units and
(b) management’s estimates of future performance of
the reporting units.
The Company also considers the economic outlook for the
healthcare services industry and various other factors during
the testing process, including hospital and physician contract
changes, local market developments, changes in third-party payor
payments, and other publicly available information.
As of September 30, 2009, the Company tested goodwill and
intangible assets for potential impairment and recorded a
non-cash impairment expense of $8.0 million resulting from
a write down of $6.6 million in the carrying value of
goodwill and a write down of $1.4 million in the carrying
value of intangible assets. The write down of the goodwill and
intangible assets related to one reporting unit. Regarding this
reporting unit, the Company believes events occurred and
circumstances changed that more likely than not reduced the fair
value of the intangible assets and goodwill below their carrying
amounts. These events during 2009 consisted primarily of the
loss of significant customers present at the acquisition date,
which adversely affected the current year and expected future
revenues and operating profit of the reporting unit.
As of December 31, 2009, estimated future amortization
expense is as follows (in thousands):
| |
|
|
|
|
Year Ending
|
|
|
|
|
December 31,
|
|
|
|
|
|
|
2010
|
|
$
|
14,999
|
|
|
2011
|
|
|
14,683
|
|
|
2012
|
|
|
13,829
|
|
|
2013
|
|
|
13,411
|
|
|
2014
|
|
|
12,430
|
|
|
Thereafter
|
|
|
45,286
|
|
|
|
|
|
|
|
|
|
|
$
|
114,638
|
|
|
|
|
|
|
|
|
|
|
Note 6.
|
Accounts
Payable and Accrued Expenses
|
Accounts payable and accrued expenses as of December 31,
2008 and 2009 consist of the following (in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
2008
|
|
|
2009
|
|
|
|
|
Accounts payable
|
|
$
|
837
|
|
|
$
|
2,945
|
|
|
Accrued management fees
|
|
|
560
|
|
|
|
430
|
|
|
Other accrued expenses
|
|
|
1,645
|
|
|
|
1,475
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
3,042
|
|
|
$
|
4,850
|
|
|
|
|
|
|
|
|
|
|
|
In 2006, the Company entered into its former term loan facility
with a related party (the “Former Lender”) with total
available borrowings up to $145.0 million. The agreement
was executed on June 2, 2006, and had a maturity date of
June 2, 2013. The agreement called for the Former Lender to
provide financing in connection with the operations and certain
business acquisitions by the Company. The Former Lender extended
credit in the form of: (a) term loans in aggregate
principal amounts not in excess of $140.0 million, and
(b) revolving loans, in aggregate principal amounts not in
excess of $5.0 million. The agreement was collateralized by
substantially all of the Company’s assets and guaranteed by
all of the Company’s subsidiaries. Interest was paid in
arrears at LIBOR plus
F-20
Aurora
Diagnostics Holdings, LLC
Notes to Consolidated Financial Statements —
(Continued)
3.75%. The interest rate was determined at the date of funding
the term notes and was fixed for a period of three to six months
at the option of the Company. After this initial interest rate
period, the loan converted to a variable rate note at the
greater of prime rate or the Federal funds’ effective rate
plus 0.50% plus a factor of 2.75%. Principal payments on the
loans were due as net cash proceeds from either the sale of
assets or the sale of equity was available. During 2007, the
Company borrowed an additional $128.8 million under this
facility.
In December 2007, the Company entered into a new term loan
facility with a syndicate of lenders (the “Lenders”)
providing for a loan commitment up to $255.0 million. The
agreement called for the Lenders to provide financing to repay
the outstanding balance of the former term loan facility, fund
working capital and make acquisitions of certain businesses. The
Lenders’ commitment includes: (a) a revolver loan, not
in excess of $5.0 million and (b) a term loan, with a
first and second lien, not in excess of $165.0 million and
$85.0 million, respectively. The Lenders’ unfunded
term loan commitment expired on April 30, 2008. As of
December 31, 2009, the Company has $132.6 million and
$76.5 million outstanding under the first lien and second
lien, respectively. The funded, first lien term loan, balance as
of April 30, 2008 was subject to quarterly principal
payments beginning on September 30, 2008 through
September 30, 2012. Total principal paid for the years
ended December 31, 2008 and 2009 was $7.8 and
$8.2 million, respectively, including mandatory and
voluntary payments.
All loans under the new term loan facility mature in December
2012. The new term loan facility is collateralized by
substantially all of the Company’s assets and guaranteed by
all of the Company’s subsidiaries. For the revolver and
first lien term loan, interest is at the prime rate plus 3.25%
or LIBOR plus 4.25%. For the second lien term loan, interest is
at the prime rate plus 6.75% or LIBOR plus 7.75%. As of
December 31, 2009, the effective interest rates were 4.5%
and 8.0% for the first lien and second lien, respectively. Other
principal payments on the term loans may be due from time to
time from annual excess cash flow and net cash proceeds from
either the sale of assets or equity. The proceeds from this term
loan facility were used to refinance the Company’s former
term loan facility and acquire two businesses in December 2007
and one business in March 2008.
The new term loan facility was issued with an original issue
discount of $1.7 million. The original issue discount is
being amortized into loan interest expense using the effective
interest method. For the years ended December 31, 2008 and
2009, the Company recorded $0.3 million and
$0.3 million, respectively, of amortization as interest
expense.
The new term loan facility requires the Company to comply on a
quarterly basis with certain financial covenants, including a
fixed charge coverage ratio calculation, a maximum total
leverage ratio calculation, a maximum senior debt leverage ratio
calculation and minimum level of earnings before interest,
taxes, depreciation and amortization, (“EBITDA”) which
become more restrictive over time. In addition, the new term
loan facility includes negative covenants restricting or
limiting the Company’s ability to, without prior approval
of the lenders, among other things, incur, assume or permit to
exist additional indebtedness or guarantees; incur liens and
engage in sale leaseback transactions; make loans and
investments; declare dividends, make payments or redeem or
repurchase capital stock; engage in mergers, acquisitions and
other business combinations; prepay, redeem or purchase certain
indebtedness; amend or otherwise alter terms of our
indebtedness; sell assets; transaction with affiliates and alter
the business it conducts. As of December 31, 2009, the
Company is in compliance with all loan covenants.
On March 21, 2007 in conjunction with an acquisition
transaction, the Company entered into a subordinated, unsecured
contingent note with a prior owner of one of the Company’s
acquired practices. The payment amount is determined by the
practice’s cumulative EBITDA over a three-year period, with
a minimum payment not to be less than $1.0 million and a
maximum payment not to exceed $2.0 million. Payment amounts
include a 5.5% interest rate factor, thus the Company has
recorded the contingent note in the original purchase price at
its minimum payment amount, discounted by the interest rate
factor of 5.5%. The original discount of $0.1 million is
being amortized into interest expense over the term of the
contingent note using the interest method.
F-21
Aurora
Diagnostics Holdings, LLC
Notes to Consolidated Financial Statements —
(Continued)
On April 30, 2007 in conjunction with an acquisition
transaction, the Company entered into a subordinated, unsecured
contingent note with prior owners of one of the Company’s
acquired practices. The payment amount is determined by the
practice’s cumulative EBITDA over a five-year period, with
a minimum payment not to be less than $15.0 million and a
maximum payment not to exceed $30.0 million. Payment
amounts include a 5.5% interest rate factor, thus the Company
recorded the contingent note in the original purchase price at
its minimum payment amount, discounted by the interest rate
factor of 5.5%. The original discount of $2.2 million will
be amortized into interest expense over the term of the
contingent note using the interest method.
On November 17, 2006, the Company entered into a
subordinated, unsecured note payable with a prior owner of one
of the Company’s acquired practices with the principal
amount of $1.0 million to assist in the financing of an
acquisition. The note had a three-year term and principal
payments were due in equal quarterly installments of
$0.08 million. Interest was paid quarterly at the prime
rate.
Long-term debt consists of the following as of December 31,
2008 and 2009 (in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
2008
|
|
|
2009
|
|
|
|
|
Term loan, first lien
|
|
$
|
140,766
|
|
|
$
|
132,566
|
|
|
Term loan, second lien
|
|
|
76,534
|
|
|
|
76,534
|
|
|
Revolver loan
|
|
|
1
|
|
|
|
—
|
|
|
Subordinated unsecured contingent note dated March 21, 2007
|
|
|
614
|
|
|
|
316
|
|
|
Subordinated unsecured contingent note dated April 30, 2007
|
|
|
10,481
|
|
|
|
8,072
|
|
|
Subordinated unsecured note payable dated November 17, 2006
|
|
|
320
|
|
|
|
—
|
|
|
Capital lease obligations
|
|
|
—
|
|
|
|
262
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
228,716
|
|
|
|
217,750
|
|
|
Less:
|
|
|
|
|
|
|
|
|
|
Original issue discount, net
|
|
|
(1,403
|
)
|
|
|
(1,098
|
)
|
|
Current portion
|
|
|
(10,010
|
)
|
|
|
(11,596
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Long-term debt, net of current portion
|
|
$
|
217,303
|
|
|
$
|
205,056
|
|
|
|
|
|
|
|
|
|
|
|
As of December 31, 2009, future maturities of long-term
debt are as follows (in thousands):
| |
|
|
|
|
Year Ending
|
|
|
|
|
December 31,
|
|
|
|
|
|
|
2010
|
|
$
|
11,596
|
|
|
2011
|
|
|
13,145
|
|
|
2012
|
|
|
192,899
|
|
|
2013
|
|
|
64
|
|
|
2014
|
|
|
46
|
|
|
|
|
|
|
|
|
|
|
$
|
217,750
|
|
|
|
|
|
|
|
Interest
Rate
Derivative:
In January 2008, the Company entered into a
2-year
interest rate swap transaction which involves the exchange of
floating for fixed rate interest payments without the exchange
of the underlying principal amount. The interest rate swap has a
notional amount of $125.0 million and a fixed rate of
interest of 3.57%. The swap resets every 90 days and ended
on January 10, 2010. The Company receives interest on the
notional amount if the LIBOR rate
F-22
Aurora
Diagnostics Holdings, LLC
Notes to Consolidated Financial Statements —
(Continued)
exceeds 3.57% and pays interest if the LIBOR rate is below
3.57%. For the years ended December 31, 2008 and 2009, the
interest settlement amount was $0.09 million and
$3.3 million, respectively, which is reflected in interest
expense in the consolidated statement of operations. The change
in fair value of the derivative instrument of $2.6 million
and $2.4 million, for the years ended December 31,
2008 and 2009, respectively, is recognized in other
comprehensive income. Accrued interest includes a swap
receivable of $0.3 million and a swap payable of
$0.9 million as of December 31, 2008 and 2009,
respectively.
|
|
|
Note 8.
|
Related
Party Transactions
|
Acquisition Target Consulting
Agreement: On June 2, 2006, and as
subsequently amended on August 1, 2009, the Company and an
entity owned by two members of the Company entered into a
professional services agreement to provide certain acquisition
target identification consulting services to the Company. In
exchange for these services, the entity will be paid a monthly
retainer of $0.02 million, plus reimbursable expenses. The
entity also earns a success fee of $0.05 million for each
identified acquisition consummated by the Company. During the
years ended December 31, 2007, 2008 and 2009, a total of
approximately $0.5 million, $0.3 million and
$0.3 million respectively, was paid to the entity. There
was a remaining payable of $0.02 million as of
December 31, 2009.
Management and Financial Advisory
Agreement: On June 2, 2006, the Company,
through its wholly-owned subsidiary, and two members of the
Company entered into a management services agreement (the
“Agreement”). On June 12, 2009 the Agreement was
amended to substitute a new member for one of the original
members. The Agreement calls for the members and their
affiliates to provide certain financial and management advisory
services in connection with the general business planning and
forecasting and acquisition and divestiture strategies of the
Company. In exchange for the services, the Company will pay an
annual fee equal to 1.0% of revenues, plus expenses to the
members (“Management Fees”).
As of December 31, 2008 and 2009, $0.6 million and
$0.4 million, respectively, of these Management Fees are
reflected in accounts payable and accrued expenses in the
accompanying consolidated balance sheets. The consolidated
statement of operations includes Management Fees of
$0.6 million, $1.6 million and $1.8 million for
the respective years ended December 31, 2007, 2008 and
2009. During 2007, 2008 and 2009, the Company paid management
fees totaling $0.4 million, $1.3 million and
$1.9 million, respectively.
Facilities Lease Agreements: The
Company leases five of its facilities from entities owned by
physician employees or affiliated physicians who are also former
owners of the acquired practices. The leases provide for monthly
aggregate payments of $0.07 million and expire in August
and September 2011, March 2012, December 2013, and April 2017.
Rent expense incurred and paid to the related entities was
$0.3 million, $0.6 million and $0.7 million for
the years ended December 31, 2007, 2008 and 2009,
respectively.
Former Term Loan Facility: The
Company’s former term loan facility, as described in
Note 7, was with a financial entity owned by a member of
the Company. The rates and terms of the former term loan
facility were comparable with arms’ length third party
transactions.
Unsecured Promissory Note: On
October 21, 2008, the Company entered into an unsecured
promissory note with an officer and member of the Company. The
note is a two-year note and accrues interest at 3.2%. The
remaining balance of the note receivable at December 31,
2009 was $0.04 million.
The Company has multiple classes of membership interests,
including A,
A-1, B, C,
X, D-1, D-2 and D-3 units. Profits and losses are generally
allocated among the members in accordance with the methodology
for computing capital accounts as described in the
Company’s LLC Agreement. Proceeds distributable to the
members in connection with the liquidation, or payable in
connection with a sale, of the Company are distributed or paid
based on the number and class of units and interests held by
each member. The various interests and units share in the
proceeds for a liquidation
and/or sale
in different relative amounts based on the aggregate amount of
the
F-23
Aurora
Diagnostics Holdings, LLC
Notes to Consolidated Financial Statements —
(Continued)
distributions
and/or
payments. The Class A,
A-1, C and X
interests receive priority distributions
and/or
payments equal to the amount of their original contributed
capital. Following these priority distributions, any amounts
that are distributed or payable to the various membership
interests is based on the allocations set forth in the LLC
Agreement.
As of December 31, 2009, the Company has authorized 85,000
Class A membership units, all of which are issued and
outstanding; 10,000 Class B membership units, all issued
and outstanding; and 5,000 Class C membership units, all
issued and outstanding. In connection with the acquisition
during 2008, the Company issued Class X capital for total
consideration of $7.1 million. As discussed in
Note 13, during 2008, the Company issued 10,000 equity
incentive interests in the classes of D-1, D-2, and D-3. During
2009, the Company issued 21,382
A-1 class
membership interests for total consideration of
$50.3 million. In connection with the A -1 issuance the
Company incurred $3.1 million of costs which were recorded
as a reduction in members’ equity.
Pursuant to subscription agreements with certain executives of
the Company, 5,000 Class C membership units were issued
during 2006 at $6.8376 per unit, for a total initial capital
contribution of $0.03 million. During 2007, the executives
made total capital contributions of $2.9 million, of which
$1.4 million was paid in cash, with the remaining balances
to be paid under promissory notes from the executives. These
notes receivable for membership interests bear interest at 5%,
are collateralized by the membership units and have a due date
of June 2, 2011. During 2008, the executives made total
capital contributions of $0.8 million, of which
$0.4 million was paid in cash, with the remaining balances
to be paid under promissory notes from the executives. These
notes receivable for membership interests bear interest at 5%,
are collateralized by the membership units and have a due date
of March 7, 2013. Interest of $0.05 million,
$0.1 million, and $0.1 million on the executive notes
receivable was credited to contributed capital as earned during
the years ended December 31, 2007, 2008 and 2009,
respectively.
During 2007, the Company accrued $2.0 million of equity
transaction costs in connection with the member contributions
for the A membership interests. During 2008, the Company
renegotiated and paid $1.75 million to settle this
liability. The $0.25 million revision of the original
estimate was recorded as an increase to members’ equity
during 2008.
The following is a table detailing the changes in members’
equity by type of membership interest for the years ended
December 31, 2007, 2008 and 2009 (in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Class A
|
|
|
Class A-1
|
|
|
Class B
|
|
|
Class C
|
|
|
Class D-1
|
|
|
Class X
|
|
|
Members’ Equity
|
|
|
|
|
Balance, December 31, 2006
|
|
$
|
32,162
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
402
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
32,564
|
|
|
Member contributions
|
|
|
113,823
|
|
|
|
—
|
|
|
|
—
|
|
|
|
2,882
|
|
|
|
—
|
|
|
|
—
|
|
|
|
116,705
|
|
|
Member notes receivable
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(1,441
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
(1,441
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, December 31, 2007
|
|
|
145,985
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,843
|
|
|
|
—
|
|
|
|
—
|
|
|
|
147,828
|
|
|
Member contributions
|
|
|
265
|
|
|
|
—
|
|
|
|
—
|
|
|
|
54
|
|
|
|
1,164
|
|
|
|
7,060
|
|
|
|
8,543
|
|
|
Member notes receivable
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(27
|
)
|
|
|
—
|
|
|
|
(352
|
)
|
|
|
(379
|
)
|
|
Tax distributions
|
|
|
—
|
|
|
|
—
|
|
|
|
(412
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(412
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, December 31, 2008
|
|
|
146,250
|
|
|
|
—
|
|
|
|
(412
|
)
|
|
|
1,870
|
|
|
|
1,164
|
|
|
|
6,708
|
|
|
|
155,580
|
|
|
Contributions from members
|
|
|
—
|
|
|
|
50,322
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
50,322
|
|
|
Tax distributions
|
|
|
—
|
|
|
|
(40
|
)
|
|
|
(1,921
|
)
|
|
|
—
|
|
|
|
(848
|
)
|
|
|
—
|
|
|
|
(2,809
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, December 31, 2009
|
|
$
|
146,250
|
|
|
$
|
50,282
|
|
|
$
|
(2,333
|
)
|
|
$
|
1,870
|
|
|
$
|
316
|
|
|
$
|
6,708
|
|
|
$
|
203,093
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Note 10.
|
Commitments
and Contingencies
|
During the ordinary course of business, the Company has become
and may in the future become subject to pending and threatened
legal actions and proceedings. The Company may have liability
with respect to its employees and its pathologists. Medical
malpractice claims are generally covered by insurance. While the
F-24
Aurora
Diagnostics Holdings, LLC
Notes to Consolidated Financial Statements —
(Continued)
Company believes the outcome of any such pending legal actions
and proceedings, individually or in the aggregate, will not have
a material adverse effect on the Company’s financial
condition, results of operations or liquidity, if the Company is
ultimately found liable under any medical malpractice claims,
there can be no assurance the Company’s medical malpractice
insurance coverage will be adequate to cover any such liability.
The Company may also, from time to time, be involved with legal
actions related to the acquisition of and affiliation with
physician practices, the prior conduct of such practices, or the
employment (and restriction on competition of ) physicians.
There can be no assurance any costs or liabilities for which the
Company becomes responsible in connection with such claims or
actions will not be material or will not exceed the limitations
of any applicable indemnification provisions or the financial
resources of the indemnifying parties. During 2009, the Company
received a claim for a refund in the amount of $0.2 million
related to payments received for services provided. The Company
believes that the claim is without merit and intends to
vigorously defend its position. The ultimate outcome of this
claim cannot presently be determined, however in
Management’s opinion, the likelihood of a material adverse
outcome is remote. Accordingly, adjustments, if any, that might
result from the resolution of this matter have not been
reflected in the consolidated financial statements as of
December 31, 2009.
Healthcare Regulatory Environment: The
healthcare industry, in general, and the services the Company
provides are subject to extensive federal and state laws and
regulations. Additionally, a portion of the Company’s
revenue is from payments by government-sponsored health
programs, including Medicare, and is subject to audit and
adjustments by applicable regulatory agencies. Failure to comply
with any of these laws or regulations, the results of increased
regulatory audits and adjustments, or change in the
interpretation of the coding of services or the amounts payable
for the Company’s services under these programs could have
a material adverse effect on the Company’s financial
position and results of operations.
Employment Agreements: The Company has
employment agreements with its executive officers and certain
physician employees, the terms of which expire at various times
through December 2014. Such agreements provide for minimum
salary levels that may be adjusted annually for
cost-of-living
changes, and may contain incentive bonuses that are payable if
specified management goals are attained. Under certain of the
agreements, in the event employment is terminated (other than
voluntarily by the employee or the Company for cause or upon the
death of the employee), the Company is committed to pay certain
benefits, including specified monthly severance for periods from
six months to two years from the date of termination. Certain
employment agreements were modified in 2008 for the equity
incentive awards described in Note 13.
Self-Insured Health Benefits: Effective
June 1, 2009, the Company began providing health care
benefits to the majority of its employees through a partially
self-insured plan. The Company records its estimate of the
ultimate cost of, and reserves for, health care benefits based
on computations using the Company’s loss history as well as
industry statistics. In determining its reserves, the Company
includes reserves for estimated claims incurred but not
reported. The amount reserved for estimated claims was
$0.8 million as of December 31, 2009. The ultimate
cost of health care benefits will depend on actual costs
incurred to settle the claims and may differ from the amounts
reserved by the Company for those claims.
Operating Leases: The Company leases
various office and medical laboratory facilities and equipment
under non-cancelable lease agreements with terms ranging from
two to ten years and expiring through April 2017. The terms of
some of the facility leases call for the Company to pay for
certain taxes or common utility charges. Rent expense including
these taxes and common utility charges was $1.1 million,
$2.5 million, and $2.7 million for the years ended
December 31, 2007, 2008, and 2009, respectively. Rent
expense associated with operating leases that include scheduled
rent increases and tenant incentives are recorded on a
straight-line basis over the term of the lease.
F-25
Aurora
Diagnostics Holdings, LLC
Notes to Consolidated Financial Statements —
(Continued)
Aggregate future minimum annual rentals under the lease
agreements as of December 31, 2009 are as follows (in
thousands):
| |
|
|
|
|
Year Ending
|
|
|
|
|
December 31,
|
|
|
|
|
|
|
2010
|
|
$
|
2,389
|
|
|
2011
|
|
|
2,300
|
|
|
2012
|
|
|
1,974
|
|
|
2013
|
|
|
1,657
|
|
|
2014
|
|
|
1,239
|
|
|
Thereafter
|
|
|
6,219
|
|
|
|
|
|
|
|
|
|
|
$
|
15,778
|
|
|
|
|
|
|
|
|
|
|
Note 11.
|
Fair
Value of Financial Instruments
|
Recurring
Fair Value Measurements
The fair value of the Company’s interest rate swap
agreements was recorded as a liability of approximately
$2.6 million and $0.1 million, as of December 31,
2008 and 2009, respectively. The interest rate swap is the only
derivative financial instrument. The fair value of the
derivative instrument was estimated by obtaining quotations from
the financial institution that is a counter party to the
instrument. The LIBOR swap rate is observable at commonly quoted
intervals for the full term of the swap and therefore is
considered a Level 2 item. The fair value is an estimate of
the net amount that the Company would have to pay on
December 31, if the agreements were canceled or transferred
to other parties.
Non-Recurring
Fair Value Measurements
Certain assets that are measured at fair value on a
non-recurring basis, including property and equipment and
intangible assets, are adjusted to fair value only when the
carrying values are greater than their fair values. As described
in Note 5, Goodwill and Intangible Assets, the Company
completed its annual impairment evaluation and recorded a write
off of goodwill and intangibles related to one reporting unit to
reflect its current estimated fair value. The fair value was
derived with fair value models utilizing unobservable inputs
that therefore is considered a Level 3 item.
As of December 31, 2008 and 2009 the carrying amounts of
cash, accounts receivable, accounts payable, accrued interest
and accrued expenses approximate fair value based on the short
maturity of these instruments.
| |
|
|
|
|
|
|
|
|
|
|
|
2008
|
|
2009
|
|
|
|
Long-term debt (including current portion of long-term debt)
|
|
$
|
228,716
|
|
|
$
|
217,750
|
|
The Company uses quoted market prices and yields for the same or
similar types of borrowings in active markets when available to
determine the fair value of the Company’s debt. The fair
values of all other debt were estimated using the present value
of expected future cash flows which incorporates the
Company’s risk profile. These fair values are considered
Level 2 items.
F-26
Aurora
Diagnostics Holdings, LLC
Notes to Consolidated Financial Statements —
(Continued)
The provision for income taxes for certain of the Company’s
subsidiaries structured as corporations for the years ended
December 31, 2007, 2008 and 2009 consist of the following
(in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2007
|
|
|
2008
|
|
|
2009
|
|
|
|
|
Current:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Federal
|
|
$
|
1,496
|
|
|
$
|
1,143
|
|
|
$
|
1,074
|
|
|
State
|
|
|
151
|
|
|
|
359
|
|
|
|
539
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current provision
|
|
|
1,647
|
|
|
|
1,502
|
|
|
|
1,613
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Deferred:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Federal
|
|
|
(885
|
)
|
|
|
(1,053
|
)
|
|
|
(1,458
|
)
|
|
State
|
|
|
—
|
|
|
|
(41
|
)
|
|
|
(110
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total deferred benefit
|
|
|
(885
|
)
|
|
|
(1,094
|
)
|
|
|
(1,568
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total provision for income taxes
|
|
$
|
762
|
|
|
$
|
408
|
|
|
$
|
45
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
A reconciliation of the provision for income taxes with amounts
determined by applying the statutory U.S. federal income
tax rate to income before income taxes for four of the
Company’s subsidiaries structured as corporations consists
primarily of state income taxes and permanent differences.
The following is a summary of the Company’s deferred tax
assets and liabilities as of December 31, 2008 and 2009,
respectively (in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
2008
|
|
|
2009
|
|
|
|
|
Accrued wages
|
|
$
|
18
|
|
|
$
|
25
|
|
|
Allowance for doubtful accounts
|
|
|
1,586
|
|
|
|
1,809
|
|
|
Acquisition related liability
|
|
|
495
|
|
|
|
192
|
|
|
|
|
|
|
|
|
|
|
|
|
Current deferred tax assets:
|
|
$
|
2,099
|
|
|
$
|
2,026
|
|
|
|
|
|
|
|
|
|
|
|
|
Noncurrent deferred tax assets:
|
|
|
|
|
|
|
|
|
|
Acquisition related liability
|
|
|
223
|
|
|
|
9
|
|
|
Noncurrent deferred tax liabilities:
|
|
|
|
|
|
|
|
|
|
Property and equipment
|
|
|
(163
|
)
|
|
|
(225
|
)
|
|
Change from cash to accrual basis of accounting by the
businesses acquired
|
|
|
(783
|
)
|
|
|
(517
|
)
|
|
Intangible assets acquired
|
|
|
(9,652
|
)
|
|
|
(9,457
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Noncurrent deferred tax liabilities, net
|
|
$
|
(10,375
|
)
|
|
$
|
(10,190
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Total deferred tax liabilities, net
|
|
$
|
(8,276
|
)
|
|
$
|
(8,164
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Note 13.
|
Equity
Based Compensation
|
During 2008, the Company issued equity incentive units
(“Units”) in accordance with the new Equity Incentive
Plan. The units represent the right of the holder to receive a
portion of proceeds in the event of liquidation of the company
as defined in the LLC Agreement. The 10,000 units issued in
July 2008 were fully vested as of December 31, 2008. The
units have no exercise price and no expiration. Compensation
cost of $1.2 million has been
F-27
Aurora
Diagnostics Holdings, LLC
Notes to Consolidated Financial Statements —
(Continued)
recognized in the results of operations as of December 31,
2008. The weighted average value is $120 per unit. During 2009,
no equity incentive units were issued or forfeited.
|
|
|
Note 14.
|
Defined
Contribution Plan
|
Effective March 1, 2007, the Company established a salary
deferral plan under Section 401(k) of the Internal Revenue
Code. The plan allows eligible employees to defer up to 100% of
their compensation in accordance with IRS guidelines. Such
deferrals accumulate on a tax deferred basis until the employee
withdraws the funds. The Company is required to match a portion
of the employees’ contribution. For 2007, 2008 and 2009,
the rate of Company match was 25%, up to $1,000 per
participating employee. Total expense recorded for the
Company’s match was $0.06 million for 2007,
$0.1 million for 2008 and $0.1 million for 2009. Also,
in connection with certain acquisitions, the Company assumed the
responsibility under certain defined contribution plans. Total
expense recorded for the Company’s match to these plans was
$0.4 million for 2007, $0.06 million for 2008 and
$0.08 million for 2009.
|
|
|
Note 15.
|
Subsequent
Events
|
On March 12, 2010, the Company issued Class Z capital
to existing members for total consideration of
$8.5 million. In the event the Company completes a
qualifying capital raise or debt refinancing within six months,
the Class Z interests will receive a preferred return equal
to the members’ initial contribution plus interest.
Interest accrues at an annual rate of 12% for the first three
months and 16% for the next three months and is payable upon a
qualifying capital raise or debt refinancing. In the event the
Company does not complete a qualifying capital raise or debt
refinancing within six months, the Class Z membership
interests will convert to
Class A-1
membership interests at the same valuation as the original
Class A-1
membership interests.
Subsequent to year end, the Company paid contingent note amounts
of $6.3 million related to acquisitions completed prior to
January 1, 2009. These contingent note payments resulted in
increases to goodwill.
On January 1, 2010, the Company acquired 100% of the equity
of two pathology practices for an aggregate cash purchase price
of $17.0 million. These acquisitions were consummated on
January 1, 2010 and therefore, the cash paid totaling
$17.0 million was included in Deposits and other
non-current assets as of December 31, 2009. On
March 12, 2010 the Company acquired 100% of the membership
interests of a pathology practice for an aggregate cash purchase
price of $22.5 million. In each transaction, the Company
issued contingent consideration payable over three to five years
based on the acquirees’ future performance. The maximum
amount of the deferred contingent cash consideration is
$32.8 million payable over three to five years. The Company
funded the cash portion of the acquisitions using
$31.0 million cash primarily related to
Class A-1
member contributions and an additional $8.5 million related
to Class Z member contributions. The Company will estimate
the fair value of the contingent consideration and record a
related liability as of the date of each acquisition, once the
estimated fair values of the assets acquired and liabilities
assumed in connection with these acquisitions are finalized.
F-28
Aurora
Diagnostics Holdings, LLC
Consolidated
Condensed Balance Sheets
| |
|
|
|
|
|
|
|
|
|
|
|
March 31,
|
|
|
December 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
|
|
(unaudited)
|
|
|
|
|
|
|
|
($ in thousands)
|
|
|
|
|
Assets
|
|
|
|
|
|
|
|
|
|
Current Assets
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
$
|
3,222
|
|
|
$
|
27,424
|
|
|
Accounts receivable, net
|
|
|
21,325
|
|
|
|
16,106
|
|
|
Prepaid expenses and other assets
|
|
|
2,877
|
|
|
|
2,031
|
|
|
Prepaid income taxes
|
|
|
—
|
|
|
|
133
|
|
|
Deferred tax assets
|
|
|
2,220
|
|
|
|
2,026
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current assets
|
|
|
29,644
|
|
|
|
47,720
|
|
|
|
|
|
|
|
|
|
|
|
|
Property and Equipment, net
|
|
|
8,868
|
|
|
|
7,580
|
|
|
|
|
|
|
|
|
|
|
|
|
Other Assets:
|
|
|
|
|
|
|
|
|
|
Deferred debt issue costs, net
|
|
|
3,640
|
|
|
|
3,932
|
|
|
Deposits and other noncurrent assets
|
|
|
131
|
|
|
|
17,149
|
|
|
Goodwill
|
|
|
328,442
|
|
|
|
271,725
|
|
|
Intangible assets, net
|
|
|
120,274
|
|
|
|
114,638
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
452,487
|
|
|
|
407,444
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
490,999
|
|
|
$
|
462,744
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Liabilities and Members’ Equity
|
|
|
|
|
|
|
|
|
|
Current Liabilities
|
|
|
|
|
|
|
|
|
|
Current portion of long-term debt
|
|
$
|
11,597
|
|
|
$
|
11,596
|
|
|
Current portion of fair value of contingent consideration
|
|
|
6,227
|
|
|
|
804
|
|
|
Accounts payable and accrued expenses
|
|
|
6,577
|
|
|
|
4,850
|
|
|
Accrued compensation
|
|
|
7,264
|
|
|
|
7,124
|
|
|
Accrued interest and other current liabilities
|
|
|
2,798
|
|
|
|
3,764
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current liabilities
|
|
|
34,463
|
|
|
|
28,138
|
|
|
Long-term debt, net of current portion
|
|
|
202,925
|
|
|
|
205,056
|
|
|
Deferred tax liabilities, net
|
|
|
11,859
|
|
|
|
10,190
|
|
|
Fair value of contingent consideration, net of current portion
|
|
|
14,439
|
|
|
|
2,296
|
|
|
Members’ Equity
|
|
|
227,313
|
|
|
|
217,064
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
490,999
|
|
|
$
|
462,744
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Consolidated Condensed Financial Statements.
F-29
Aurora
Diagnostics Holdings, LLC
Consolidated
Condensed Statements of Operations
Three Months Ended March 31, 2010 and 2009
(unaudited)
| |
|
|
|
|
|
|
|
|
|
|
|
2010
|
|
|
2009
|
|
|
|
|
($ in thousands)
|
|
|
|
|
Net Revenues
|
|
$
|
46,419
|
|
|
$
|
40,947
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating costs and expenses:
|
|
|
|
|
|
|
|
|
|
Cost of services
|
|
|
22,342
|
|
|
|
17,186
|
|
|
Selling, general and administrative expenses
|
|
|
10,820
|
|
|
|
9,177
|
|
|
Provision for doubtful accounts
|
|
|
2,610
|
|
|
|
2,305
|
|
|
Intangible asset amortization expense
|
|
|
3,891
|
|
|
|
3,628
|
|
|
Management fees
|
|
|
476
|
|
|
|
410
|
|
|
Acquisition and business development costs
|
|
|
298
|
|
|
|
130
|
|
|
|
|
|
|
|
|
|
|
|
|
Total operating costs and expenses
|
|
|
40,437
|
|
|
|
32,836
|
|
|
|
|
|
|
|
|
|
|
|
|
Income from operations
|
|
|
5,982
|
|
|
|
8,111
|
|
|
|
|
|
|
|
|
|
|
|
|
Other income (expense):
|
|
|
|
|
|
|
|
|
|
Interest expense
|
|
|
(3,721
|
)
|
|
|
(4,791
|
)
|
|
Other income / expense
|
|
|
(12
|
)
|
|
|
29
|
|
|
|
|
|
|
|
|
|
|
|
|
Total other expense, net
|
|
|
(3,733
|
)
|
|
|
(4,762
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Income before income taxes
|
|
|
2,249
|
|
|
|
3,349
|
|
|
Provision for income taxes
|
|
|
10
|
|
|
|
17
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income
|
|
$
|
2,239
|
|
|
$
|
3,332
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Consolidated Condensed Financial Statements.
F-30
Aurora
Diagnostics Holdings, LLC
Consolidated
Condensed Statement of Members’ Equity and Comprehensive
Income
Three Months Ended March 31, 2010 (unaudited)
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Member
|
|
|
Other
|
|
|
Equity
|
|
|
|
|
|
Total
|
|
|
|
|
Member
|
|
|
Contributions
|
|
|
Comprehensive
|
|
|
Transaction
|
|
|
Retained
|
|
|
Members’
|
|
|
|
|
Units
|
|
|
(Distributions)
|
|
|
Income (Loss)
|
|
|
Costs
|
|
|
Earnings
|
|
|
Equity
|
|
|
|
|
($ in thousands)
|
|
|
|
|
Balance, December 31, 2009
|
|
|
131,382
|
|
|
$
|
203,093
|
|
|
$
|
(125
|
)
|
|
$
|
(4,825
|
)
|
|
$
|
18,921
|
|
|
$
|
217,064
|
|
|
Components of comprehensive income
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
2,239
|
|
|
|
2,239
|
|
|
Fair value of derivative
|
|
|
—
|
|
|
|
—
|
|
|
|
125
|
|
|
|
—
|
|
|
|
—
|
|
|
|
125
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total comprehensive income
|
|
|
—
|
|
|
|
—
|
|
|
|
125
|
|
|
|
—
|
|
|
|
2,239
|
|
|
|
2,364
|
|
|
Contributions from members
|
|
|
—
|
|
|
|
8,500
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
8,500
|
|
|
Tax distributions
|
|
|
—
|
|
|
|
(556
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(556
|
)
|
|
Dividend distributions
|
|
|
—
|
|
|
|
(59
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(59
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, March 31, 2010
|
|
|
131,382
|
|
|
$
|
210,978
|
|
|
$
|
—
|
|
|
$
|
(4,825
|
)
|
|
$
|
21,160
|
|
|
$
|
227,313
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Consolidated Condensed Financial Statements.
F-31
Aurora
Diagnostics Holdings, LLC
Consolidated
Condensed Statements of Cash Flows
Three Months Ended March 31, 2010 and 2009
(unaudited)
| |
|
|
|
|
|
|
|
|
|
|
|
2010
|
|
|
2009
|
|
|
|
|
($ in thousands)
|
|
|
|
|
Cash Flows From Operating Activities
|
|
|
|
|
|
|
|
|
|
Net income
|
|
$
|
2,239
|
|
|
$
|
3,332
|
|
|
Adjustments to reconcile net income to net cash provided by
operating activities:
|
|
|
|
|
|
|
|
|
|
Depreciation and amortization
|
|
|
4,693
|
|
|
|
4,196
|
|
|
Amortization of deferred debt issue costs
|
|
|
291
|
|
|
|
262
|
|
|
Amortization of original issue discount on debt
|
|
|
81
|
|
|
|
73
|
|
|
Deferred income taxes
|
|
|
(464
|
)
|
|
|
—
|
|
|
Changes in assets and liabilities, net of working capital
acquired in business combinations:
|
|
|
|
|
|
|
|
|
|
(Increase) decrease in:
|
|
|
|
|
|
|
|
|
|
Accounts receivable
|
|
|
(2,105
|
)
|
|
|
(1,016
|
)
|
|
Prepaid expenses
|
|
|
(270
|
)
|
|
|
(297
|
)
|
|
Increase (decrease) in:
|
|
|
|
|
|
|
|
|
|
Accounts payable and accrued expenses
|
|
|
330
|
|
|
|
48
|
|
|
Accrued compensation
|
|
|
(293
|
)
|
|
|
(606
|
)
|
|
Accrued interest
|
|
|
(789
|
)
|
|
|
(18
|
)
|
|
Taxes payable
|
|
|
336
|
|
|
|
(142
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Net cash provided by operating activities
|
|
|
4,049
|
|
|
|
5,832
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash Flows From Investing Activities
|
|
|
|
|
|
|
|
|
|
Purchase of property and equipment
|
|
|
(882
|
)
|
|
|
(341
|
)
|
|
Increase in deposits and other noncurrent assets
|
|
|
42
|
|
|
|
12
|
|
|
Payment of contingent notes
|
|
|
(11,703
|
)
|
|
|
(5,510
|
)
|
|
Businesses acquired, net of cash acquired
|
|
|
(20,949
|
)
|
|
|
(386
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Net cash used in investing activities
|
|
|
(33,492
|
)
|
|
|
(6,225
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Cash Flows From Financing Activities
|
|
|
|
|
|
|
|
|
|
Repayments of subordinated notes payable
|
|
|
(11
|
)
|
|
|
(80
|
)
|
|
Net borrowings under revolver
|
|
|
—
|
|
|
|
(1
|
)
|
|
Repayments under new term loan facilities
|
|
|
(2,200
|
)
|
|
|
(1,800
|
)
|
|
Deferred financing costs
|
|
|
(492
|
)
|
|
|
—
|
|
|
Contributions from members, net of tax distributions
|
|
|
7,944
|
|
|
|
(504
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Net cash provided by (used in) financing activities
|
|
|
5,241
|
|
|
|
(2,385
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Net (decrease) in cash
|
|
|
(24,202
|
)
|
|
|
(2,778
|
)
|
|
Cash and cash equivalents, beginning
|
|
|
27,424
|
|
|
|
7,278
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents, ending
|
|
$
|
3,222
|
|
|
$
|
4,500
|
|
|
|
|
|
|
|
|
|
|
|
|
Supplemental Disclosures of Cash Flow Information
|
|
|
|
|
|
|
|
|
|
Cash interest payments
|
|
$
|
4,096
|
|
|
$
|
4,762
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash tax payments, including member tax distributions
|
|
$
|
716
|
|
|
$
|
687
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Consolidated Condensed Financial Statements.
F-32
Aurora
Diagnostics Holdings, LLC
|
|
|
Note 1.
|
Nature of
Business and Significant Accounting Policies
|
Nature of Business: Aurora Diagnostics
Holdings, LLC and subsidiaries (the “Company”) was
organized in the State of Delaware as a limited liability
company on June 2, 2006 to operate as a diagnostic services
company. The Company’s practices provide physician-based
general anatomic and clinical pathology, dermapathology,
molecular diagnostic services and other esoteric testing
services to physicians, hospitals, clinical laboratories and
surgery centers. The Company’s operations consist of one
reportable segment.
The Company operates in a highly regulated industry. The manner
in which licensed physicians can organize to perform and bill
for medical services is governed by state laws and regulations.
Businesses like the Company often are not permitted to employ
physicians or to own corporations that employ physicians or to
otherwise exercise control over the medical judgments or
decisions of physicians.
In states where the Company is not permitted to directly own a
medical services provider or for other commercial reasons, it
performs only non-medical administrative and support services,
does not represent to the public or its clients that it offers
medical services and does not exercise influence or control over
the practice of medicine. In those states, the Company conducts
business through entities that it controls, and it is these
affiliated entities that employ the physicians who practice
medicine. In such states, the Company generally enters into a
contract that restricts the owner of the affiliated entity from
transferring their ownership interests in the affiliated entity
and otherwise provides the Company or its designee with a
controlling voting or financial interest in the affiliated
entity and its laboratory operations. This controlling financial
interest generally is obtained pursuant to a long-term
management services agreement between the Company and the
affiliated entity. Under the management services agreement, the
Company exclusively manages all aspects of the operation other
than the provision of medical services. Generally, the
affiliated entity has no operating assets because the Company
acquired all of its operating assets at the time it acquired the
related laboratory operations. In accordance with the relevant
literature, these affiliated entities are included in the
consolidated financial statements of Aurora Diagnostics
Holdings, LLC.
The accompanying consolidated balance sheet as of
December 31, 2009, which was derived from the audited
financial statements as of December 31, 2009 of Aurora
Diagnostics Holdings, LLC and the accompanying unaudited
condensed consolidated financial statements as of and for the
period ended March 31, 2010 have been prepared by the
Company in accordance with accounting principles generally
accepted in the United States of America for interim financial
reporting. Accordingly, they do not include all of the
information and related footnotes that would normally be
required by accounting principles generally accepted in the
United States of America for complete financial reporting. These
unaudited condensed consolidated financial statements should be
read in conjunction with the Company’s audited financial
statements for the year ended December 31, 2009.
The accompanying unaudited condensed consolidated financial
statements include all adjustments (consisting of a normal and
recurring nature) that management considers necessary for a fair
statement of financial information for the interim periods.
Interim results are not necessarily indicative of the results
that may be expected for the remainder of the year ending
December 31, 2010.
Recent Accounting Pronouncements: In
February 2010, the FASB issued FASB Accounting Standards Update
2010-09,
Subsequent Events (Topic 855): Amendments to Certain
Recognition and Disclosure Requirements. Topic 855 removes
the requirement for a U.S. Securities and Exchange
Commission (“SEC”) filer to disclose a date in both
issued and revised financial statements. Revised financial
statements include financial statements revised as a result of
either correction of an error or retrospective application of
U.S. GAAP. This update was effective upon issuance for the
Company. The Company’s adoption of this update did not have
a significant impact upon its financial statements.
In January 2010, the FASB issued FASB Accounting Standards
Update
2010-06,
Fair Value Measurements and Disclosures (Topic 820):
Improving Disclosures about Fair Value Measurements. This
update provides amendments to Topic 820 that will provide for
more robust disclosures about the: (1) different classes of
assets and liabilities measured at fair value,
(2) valuation techniques and inputs used, (3) activity
in Level 3 fair value
F-33
Aurora
Diagnostics Holdings, LLC
Notes to
Consolidated Condensed Financial
Statements — (Continued)
measurements, and (4) transfers between Levels 1, 2,
and 3. This update is effective for interim and annual reporting
periods beginning after December 15, 2009 and the Company
adopted this update on January 1, 2010. The adoption of
this update did not have a significant impact upon the
Company’s financial statements.
2010
Acquisitions
On January 1, 2010, the Company acquired 100% of the equity
of two pathology practices for an aggregate cash purchase price
of $17.0 million. These acquisitions were funded on
December 31, 2009 and therefore, the cash paid totaling
$17.0 million was included in Deposits and other
non-current assets as of December 31, 2009. On
March 12, 2010 the Company acquired 100% of the membership
interests of a pathology practice for an aggregate cash purchase
price of $22.5 million. In each transaction, the Company
issued contingent consideration payable over three to five years
based on the acquirees’ future performance. The Company has
estimated the fair value of the contingent consideration and
recorded a related liability in the accompanying consolidated
condensed balance sheets as of March 31, 2010 of
$17.5 million. The Company funded the cash portion of the
acquisitions using $31.0 million cash primarily related to
Class A-1
member contributions and an additional $8.5 million related
to Class Z member contributions.
Intangible assets, acquired as the result of a business
combination, are recognized at fair value, as an asset apart
from goodwill if the asset arises from contractual or other
legal rights, or if it is separable. Intangible assets,
principally representing the fair value of customer
relationships, health care facility agreements and
non-competition agreements acquired, are capitalized and
amortized on the straight-line method over their useful life,
which generally ranges from 4 to 18 years. The estimated
value of the assets acquired, liabilities assumed, and
contingent consideration for the 2010 acquisitions are
preliminary and are expected to be finalized in 2010.
During 2010, the Company expensed $0.3 million of
transaction costs associated with its completed acquisitions and
business development.
The following table summarizes the consideration for the
acquisitions made in 2010, excluding contingent consideration
payable with a fair value of approximately $17.5 million
(in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
Date
|
|
|
Cash
|
|
|
Location
|
|
Acquired
|
|
|
Paid
|
|
|
|
|
Florida
|
|
|
January 1, 2010
|
|
|
$
|
7,975
|
|
|
Michigan
|
|
|
January 1, 2010
|
|
|
|
9,000
|
|
|
New Jersey
|
|
|
March 12, 2010
|
|
|
|
22,500
|
|
|
|
|
|
|
|
|
|
|
|
|
Total 2010 Acquisitions
|
|
|
|
|
|
$
|
39,475
|
|
|
|
|
|
|
|
|
|
|
|
Assuming the 2010 acquisitions achieve maximum earnings during
the consideration period, the maximum contingent consideration
payable would be $39.9 million.
Contingent
Consideration
In connection with the acquisitions, the Company has agreed to
pay additional consideration in future periods, based upon the
attainment of stipulated levels of operating earnings by each of
the acquired entities, as defined in their respective
agreements. For all acquisitions prior to January 1, 2009,
the Company does not accrue contingent consideration obligations
prior to the attainment of the objectives and the amount owed
becomes fixed and determinable. For the three months ended
March 31, 2010 and 2009, the Company paid consideration
under contingent notes of $11.7 million and
$5.5 million, respectively.
F-34
Aurora
Diagnostics Holdings, LLC
Notes to
Consolidated Condensed Financial
Statements — (Continued)
As of March 31, 2010, assuming the practices achieve the
maximum level of stipulated operating earnings, the maximum
principal amount of contingent consideration payable, over the
next three to five years is approximately $126.6 million. A
lesser amount will be paid for earnings below the maximum level
of stipulated earnings or no payments will be made if the
practices do not achieve the minimum level of stipulated
earnings as outlined in their respective agreements. Any such
payments in the future for acquisitions completed prior to
January 1, 2009 would be accounted for as additional
purchase price and increase goodwill. For acquisitions completed
after December 31, 2008, future payments will be reflected
in the change in the fair value of the contingent consideration.
The following tables summarize the estimated aggregate fair
value of the assets acquired and liabilities assumed in
connection with the acquisitions in 2010 (in thousands):
| |
|
|
|
|
|
Cash
|
|
$
|
1,865
|
|
|
Accounts receivable
|
|
|
3,114
|
|
|
Other assets
|
|
|
84
|
|
|
Property and equipment
|
|
|
1,208
|
|
|
Intangible assets
|
|
|
9,527
|
|
|
Goodwill
|
|
|
45,014
|
|
|
|
|
|
|
|
|
Assets acquired
|
|
|
60,812
|
|
|
|
|
|
|
|
|
Accounts payable and accrued expenses
|
|
|
1,398
|
|
|
Accrued compensation
|
|
|
433
|
|
|
Fair value of contingent consideration
|
|
|
17,549
|
|
|
Deferred tax liabilities
|
|
|
1,939
|
|
|
|
|
|
|
|
|
Liabilities assumed
|
|
|
21,319
|
|
|
|
|
|
|
|
|
Net assets acquired
|
|
$
|
39,493
|
|
|
|
|
|
|
|
|
Net assets acquired
|
|
$
|
39,493
|
|
|
Less:
|
|
|
|
|
|
Cash acquired
|
|
|
(1,865
|
)
|
|
Deposits and other noncurrent assets
|
|
|
(16,975
|
)
|
|
|
|
|
|
|
|
Cash paid for acquisitions, net of cash acquired
|
|
|
20,653
|
|
|
Acquisition costs and acquisition-related liability paid
|
|
|
296
|
|
|
|
|
|
|
|
|
Total businesses acquired and related costs, net of cash
acquired
|
|
$
|
20,949
|
|
|
|
|
|
|
|
Acquired Accounts receivable is net of $0.6 million
Allowance for doubtful accounts.
Pro-Forma
Information (Unaudited)
The accompanying consolidated condensed financial statements
include the results of operations of the acquisitions from the
date acquired through March 31, 2010, including net revenue
of $5.2 million and net income of $0.6 million.
The following unaudited pro forma information presents the
consolidated results of the Company’s operations and the
results of the 2010 acquisitions for the period ended
March 31, 2010, after giving effect to amortization,
depreciation, interest, income tax, and the reduced level of
certain specific operating expenses (primarily compensation and
related expenses attributable to former owners) as if the
acquisition had been consummated on January 1, 2010. Such
unaudited pro forma information is based on historical unaudited
financial information with respect to the 2010 acquisitions and
does not include operational or other changes which might have
been
F-35
Aurora
Diagnostics Holdings, LLC
Notes to
Consolidated Condensed Financial
Statements — (Continued)
effected by the Company. The unaudited pro forma information for
the period ended March 31, 2010 presented below is for
illustrative purposes only and is not necessarily indicative of
results which would have been achieved or results which may be
achieved in the future (in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
Pro Forma
|
|
|
Pro Forma
|
|
|
|
|
March 31,
|
|
|
March 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
|
|
Net Revenues
|
|
$
|
49,617
|
|
|
$
|
50,538
|
|
|
|
|
|
|
|
|
|
|
|
|
Net Income
|
|
$
|
3,753
|
|
|
$
|
5,295
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Note 3.
|
Accounts
Receivable
|
Accounts receivable consist of the following as of
March 31, 2010 and December 31, 2009 (in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
March 31,
|
|
|
December 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
|
|
Accounts receivable
|
|
$
|
30,947
|
|
|
$
|
24,659
|
|
|
Less: Allowance for doubtful accounts
|
|
|
(9,622
|
)
|
|
|
(8,553
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Accounts receivable, net
|
|
$
|
21,325
|
|
|
$
|
16,106
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Note 4.
|
Goodwill
and Intangible Assets
|
The following table presents adjustments to goodwill during 2010
and 2009 (in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
March 31,
|
|
|
December 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
|
|
Goodwill, beginning of period
|
|
$
|
271,725
|
|
|
$
|
250,340
|
|
|
Acquisitions
|
|
|
45,014
|
|
|
|
15,327
|
|
|
Contingent notes*
|
|
|
11,703
|
|
|
|
12,668
|
|
|
Goodwill impairment
|
|
|
—
|
|
|
|
(6,610
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Goodwill, end of period
|
|
$
|
328,442
|
|
|
$
|
271,725
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
* |
|
Related to acquisitions completed prior to January 1, 2009. |
Intangible assets as of March 31, 2010 and
December 31, 2009 and the related accumulated amortization
are set forth in the table below (in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted Average
|
|
|
March 31, 2010
|
|
|
|
|
Range
|
|
Amortization Period
|
|
|
|
|
|
Accumulated
|
|
|
|
|
|
|
|
(Years)
|
|
(Years)
|
|
|
Cost
|
|
|
Amortization
|
|
|
Net
|
|
|
|
|
Amortizing intangible assets:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Customer relationships
|
|
7 – 10
|
|
|
9
|
|
|
$
|
111,867
|
|
|
$
|
(26,287
|
)
|
|
$
|
85,580
|
|
|
Health care facility agreements
|
|
4 – 18
|
|
|
14
|
|
|
|
41,370
|
|
|
|
(8,778
|
)
|
|
|
32,592
|
|
|
Noncompete agreements
|
|
4 – 5
|
|
|
5
|
|
|
|
3,848
|
|
|
|
(1,746
|
)
|
|
|
2,102
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total intangible assets
|
|
|
|
|
|
|
|
$
|
157,085
|
|
|
$
|
(36,811
|
)
|
|
$
|
120,274
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
F-36
Aurora
Diagnostics Holdings, LLC
Notes to
Consolidated Condensed Financial
Statements — (Continued)
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted Average
|
|
|
December 31, 2009
|
|
|
|
|
Range
|
|
Amortization Period
|
|
|
|
|
|
Accumulated
|
|
|
|
|
|
|
|
(Years)
|
|
(Years)
|
|
|
Cost
|
|
|
Amortization
|
|
|
Net
|
|
|
|
|
Amortizing intangible assets:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Customer relationships
|
|
7 – 10
|
|
|
9
|
|
|
$
|
103,089
|
|
|
$
|
(23,414
|
)
|
|
$
|
79,675
|
|
|
Health care facility agreements
|
|
4 – 18
|
|
|
14
|
|
|
|
41,370
|
|
|
|
(7,951
|
)
|
|
|
33,419
|
|
|
Noncompete agreements
|
|
4 – 5
|
|
|
5
|
|
|
|
3,098
|
|
|
|
(1,554
|
)
|
|
|
1,544
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total intangible assets
|
|
|
|
|
|
|
|
$
|
147,557
|
|
|
$
|
(32,919
|
)
|
|
$
|
114,638
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
As of March 31, 2010, estimated future amortization expense
is as follows (in thousands):
| |
|
|
|
|
Year Ending
|
|
|
|
|
December 31,
|
|
|
|
|
|
|
Remainder of 2010
|
|
$
|
12,020
|
|
|
2011
|
|
|
15,711
|
|
|
2012
|
|
|
14,857
|
|
|
2013
|
|
|
14,439
|
|
|
2014
|
|
|
13,458
|
|
|
2015
|
|
|
12,616
|
|
|
Thereafter
|
|
|
37,173
|
|
|
|
|
|
|
|
|
|
|
$
|
120,274
|
|
|
|
|
|
|
|
|
|
|
Note 5.
|
Accounts
Payable and Accrued Expenses
|
Accounts payable and accrued expenses as of March 31, 2010
and December 31, 2009 consist of the following (in
thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
March 31,
|
|
|
December 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
|
|
Accounts payable
|
|
$
|
3,166
|
|
|
$
|
2,945
|
|
|
Accrued management fees
|
|
|
713
|
|
|
|
430
|
|
|
Other accrued expenses
|
|
|
2,698
|
|
|
|
1,475
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
6,577
|
|
|
$
|
4,850
|
|
|
|
|
|
|
|
|
|
|
|
Approximately $0.6 million of the increase in Other accrued
expenses relates to the practices acquired in 2009 and 2010.
In 2006, the Company entered into a credit facility (the
“2006 Credit Facility”) with a related party (the
“Former Lender”) with total available borrowings up to
$145.0 million. The 2006 Credit Facility was executed on
June 2, 2006 and had a maturity date of June 2, 2013.
The 2006 Credit Facility called for the Former Lender to provide
financing in connection with the operations and certain business
acquisitions by the Company. The Former Lender extended credit
in the form of (a) term loans in aggregate principal
amounts not in excess of $140.0 million,
F-37
Aurora
Diagnostics Holdings, LLC
Notes to
Consolidated Condensed Financial
Statements — (Continued)
and (b) revolving loans in aggregate principal amounts not
in excess of $5.0 million. The 2006 Credit Facility was
collateralized by substantially all of the Company’s assets
and guaranteed by all of the Company’s subsidiaries.
Interest was paid in arrears at LIBOR plus 3.75%. The interest
rate was determined at the date of funding the term loans and
was fixed for a period of three to six months at the option of
the Company. After this initial interest rate period, the loan
converted to a variable rate note at the greater of prime rate
or the Federal funds’ effective rate plus 0.50% plus a
factor of 2.75%. Principal payments on the loans were due as net
cash proceeds from either the sale of assets or the sale of
equity was available. During 2007, the Company borrowed an
additional $128.8 million under this facility.
In December 2007, the Company entered into a new term loan
facility (the “2007 Credit Facility”) with a syndicate
of lenders (the “Lenders”) providing for a loan
commitment up to $255.0 million. The 2007 Credit Facility
called for the Lenders to provide financing to repay the
outstanding balance of the 2006 Credit Facility, fund working
capital and acquire certain businesses. The Lenders’
commitment included: (a) a revolver loan, not in excess of
$5.0 million and (b) a term loan, with a first and
second lien, not in excess of $165.0 million and
$85.0 million, respectively. The Lenders’ unfunded
term loan commitment expired on April 30, 2008. As of
December 31, 2009, the Company has $130.4 million and
$76.5 million outstanding under the first lien and second
lien, respectively. The funded, first lien term loan, balance as
of April 30, 2008 was subject to quarterly principal
payments beginning on September 30, 2008 through
September 30, 2012. Total mandatory principal payments for
the periods ended March 31, 2010 and 2009 were $2.2 and
$1.8 million, respectively.
All loans under the 2007 Credit Facility mature in December
2012. The 2007 Credit Facility is collateralized by
substantially all of the Company’s assets and guaranteed by
all of the Company’s subsidiaries. For the revolver and
first lien term loan, interest is at the prime rate plus 3.25%
or LIBOR plus 4.25%. For the second lien term loan, interest is
at the prime rate plus 6.75% or LIBOR plus 7.75%. As of
March 31, 2010, the effective interest rates were 4.5% and
8.0% for the first lien and second lien, respectively. Other
principal payments on the term loans may be due from time to
time from annual excess cash flow and net cash proceeds from
either the sale of assets or equity. The proceeds from the 2007
Credit Facility were used to refinance the Company’s 2006
Credit Facility and acquire two businesses in December 2007 and
one business in March 2008.
The 2007 Credit Facility was issued with an original issue
discount of $1.7 million. The original issue discount is
being amortized into loan interest expense using the effective
interest method. For the three months ended March 31, 2010
and 2009, the Company recorded $0.08 million and
$0.07 million, respectively, of amortization as interest
expense.
The 2007 Credit Facility requires the Company to comply on a
quarterly basis with certain financial covenants, including a
fixed charge coverage ratio calculation, a maximum total
leverage ratio calculation, a maximum senior debt leverage ratio
calculation and minimum level of earnings before interest,
taxes, depreciation and amortization, (“EBITDA”) which
become more restrictive over time. In addition, the 2007 Credit
Facility includes negative covenants restricting or limiting the
Company’s ability to, without prior approval of the
lenders, among other things, incur, assume or permit to exist
additional indebtedness or guarantees; incur liens and engage in
sale leaseback transactions; make loans and investments; declare
dividends, make payments or redeem or repurchase capital stock;
engage in mergers, acquisitions and other business combinations;
prepay, redeem or purchase certain indebtedness; amend or
otherwise alter terms of our indebtedness; sell assets; transact
with affiliates and alter the business it conducts. As of
March 31, 2010, the Company is in compliance with all loan
covenants.
On March 21, 2007 in conjunction with an acquisition
transaction, the Company entered into a subordinated, unsecured
contingent note with a prior owner of one of the Company’s
acquired practices. The payment amount is determined by the
practice’s cumulative EBITDA over a three-year period, with
a minimum payment not to be less than $1.0 million and a
maximum payment not to exceed $2.0 million. Payment amounts
include a 5.5% interest rate factor, thus the Company has
recorded the contingent note in the original purchase price at
its minimum payment amount, discounted by the interest rate
factor of 5.5%. The original discount of $0.1 million is
being amortized into interest expense over the term of the
contingent note using the interest method.
F-38
Aurora
Diagnostics Holdings, LLC
Notes to
Consolidated Condensed Financial
Statements — (Continued)
On April 30, 2007 in conjunction with an acquisition
transaction, the Company entered into a subordinated, unsecured
contingent note with prior owners of one of the Company’s
acquired practices. The payment amount is determined by the
practice’s cumulative EBITDA over a five-year period, with
a minimum payment not to be less than $15.0 million and a
maximum payment not to exceed $30.0 million. Payment
amounts include a 5.5% interest rate factor, thus the Company
recorded the contingent note in the original purchase price at
its minimum payment amount, discounted by the interest rate
factor of 5.5%. The original discount of $2.2 million will
be amortized into interest expense over the term of the
contingent note using the interest method.
On November 17, 2006, the Company entered into a
subordinated, unsecured note payable with a prior owner of one
of the Company’s acquired practices with the principal
amount of $1.0 million to assist in the financing of an
acquisition. The note had a three-year term and principal
payments were due in equal quarterly installments of
$0.08 million. Interest was paid quarterly at the prime
rate.
Long-term debt consists of the following as of March 31,
2010 and December 31, 2009 (in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
March 31,
|
|
|
December 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
|
|
Term loan, first lien
|
|
$
|
130,366
|
|
|
$
|
132,566
|
|
|
Term loan, second lien
|
|
|
76,534
|
|
|
|
76,534
|
|
|
Subordinated unsecured contingent note dated March 21, 2007
|
|
|
316
|
|
|
|
316
|
|
|
Subordinated unsecured contingent note dated April 30, 2007
|
|
|
8,072
|
|
|
|
8,072
|
|
|
Capital lease obligations
|
|
|
252
|
|
|
|
262
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
215,540
|
|
|
|
217,750
|
|
|
Less:
|
|
|
|
|
|
|
|
|
|
Original issue discount, net
|
|
|
(1,018
|
)
|
|
|
(1,098
|
)
|
|
Current portion
|
|
|
(11,597
|
)
|
|
|
(11,596
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Long-term debt, net of current portion
|
|
$
|
202,925
|
|
|
$
|
205,056
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Note 7.
|
Related
Party Transactions
|
Acquisition Target Consulting
Agreement: On June 2, 2006, and as
subsequently amended on August 1, 2009, the Company and an
entity owned by two members of the Company entered into a
professional services agreement to provide certain acquisition
target identification consulting services to the Company. In
exchange for these services, the entity will be paid a monthly
retainer of $0.02 million, plus reimbursable expenses. The
entity also earns a success fee of $0.05 million for each
identified acquisition consummated by the Company. During the
periods ended March 31, 2010 and 2009, a total of
approximately $0.1 million and $0.07 million,
respectively, was paid to the entity. There was a remaining
payable of $0.01 and $0.02 million as of March 31,
2010 and December 31, 2009, respectively.
Management and Financial Advisory
Agreement: On June 2, 2006, the Company,
through its wholly-owned subsidiary, and two members of the
Company entered into a management services agreement (the
“Agreement”). On June 12, 2009 the Agreement was
amended to substitute a new member for one of the original
members. The Agreement calls for the members and their
affiliates to provide certain financial and management advisory
services in connection with the general business planning and
forecasting and acquisition and divestiture strategies of the
Company. In exchange for the services, the Company will pay an
annual fee equal to 1.0% of revenues, plus expenses to the
members (“Management Fees”).
As of March 31, 2010 and December 31, 2009,
$0.7 million and $0.4 million, respectively, of these
Management Fees are reflected in accounts payable and accrued
expenses in the accompanying consolidated
F-39
Aurora
Diagnostics Holdings, LLC
Notes to
Consolidated Condensed Financial
Statements — (Continued)
balance sheets. The consolidated condensed statement of
operations includes Management Fees of $0.5 million,
$0.4 million for the respective periods ended
March 31, 2010 and 2009. During the periods ended
March 31, 2010 and 2009, the Company did not pay any
management fees.
Facilities Lease Agreements: The
Company leases seven of its facilities from entities owned by
physician employees or affiliated physicians who are also former
owners of the acquired practices. The leases provide for monthly
aggregate base payments of $0.09 million and expire in
August and September 2011, March and December 2012, December
2013, December 2014, and April 2017. Rent expense incurred and
paid to the related entities was $0.4 million and
$0.2 million for the periods ended March 31, 2010 and
2009, respectively.
Former Term Loan Facility: The
Company’s former term loan facility, as described in
Note 6, was with a financial entity owned by a member of
the Company. The rates and terms of the former term loan
facility were comparable with arms’ length third party
transactions.
Unsecured Promissory Note: On
October 21, 2008, the Company entered into an unsecured
promissory note with an officer and member of the Company. The
note is a two-year note and accrues interest at 3.2%. The
remaining balance of the note receivable at March 31, 2010
was $0.03 million.
On March 12, 2010, the Company issued Class Z capital
to existing members for total consideration of
$8.5 million. In the event the Company completes a
qualifying capital raise or debt refinancing within six months,
the Class Z interests will receive a preferred return equal
to the members’ initial contribution plus dividends.
Dividends accrue at an annual rate of 12% for the first three
months and 16% for the next three months and are payable upon a
qualifying capital raise or debt refinancing. In the event the
Company does not complete a qualifying capital raise or debt
refinancing within six months, the Class Z membership
interests will convert to
Class A-1
membership interests at the same valuation as the original
Class A-1
membership interests. On May 26, 2010 the Company completed
a qualifying debt refinancing. On that date, the members were
paid the preferred return equal to their original contribution
of $8.5 million plus dividends of $0.2 million.
|
|
|
Note 9.
|
Commitments
and Contingencies
|
During the ordinary course of business, the Company has become
and may in the future become subject to pending and threatened
legal actions and proceedings. The Company may have liability
with respect to its employees and its pathologists. Medical
malpractice claims are generally covered by insurance. While the
Company believes the outcome of any such pending legal actions
and proceedings, individually or in the aggregate, will not have
a material adverse effect on the Company’s financial
condition, results of operations or liquidity, if the Company is
ultimately found liable under any medical malpractice claims,
there can be no assurance the Company’s medical malpractice
insurance coverage will be adequate to cover any such liability.
The Company may also, from time to time, be involved with legal
actions related to the acquisition of and affiliation with
physician practices, the prior conduct of such practices, or the
employment (and restriction on competition of ) physicians.
There can be no assurance any costs or liabilities for which the
Company becomes responsible in connection with such claims or
actions will not be material or will not exceed the limitations
of any applicable indemnification provisions or the financial
resources of the indemnifying parties. During 2009, the Company
received two claims for refunds in the amount of
$1.2 million related to payments received for services
provided. In June 2010, the Company settled both claims for a
total of $0.3 million. These settlements are reflected as a
reduction of net revenue in the consolidated condensed financial
statements as of March 31, 2010.
Healthcare Regulatory Environment: The
healthcare industry, in general, and the services the Company
provides are subject to extensive federal and state laws and
regulations. Additionally, a portion of the Company’s
revenue is from payments by government-sponsored health
programs, including Medicare, and is subject to audit and
adjustments by applicable regulatory agencies. Failure to comply
with any of these laws or regulations, the
F-40
Aurora
Diagnostics Holdings, LLC
Notes to
Consolidated Condensed Financial
Statements — (Continued)
results of increased regulatory audits and adjustments, or
change in the interpretation of the coding of services or the
amounts payable for the Company’s services under these
programs could have a material adverse effect on the
Company’s financial position and results of operations.
|
|
|
Note 10.
|
Fair
Value of Financial Instruments
|
Non-Recurring
Fair Value Measurements
Certain assets that are measured at fair value on a
non-recurring basis, including property and equipment and
intangible assets, are adjusted to fair value only when the
carrying values are greater than their fair values. The fair
value was derived with fair value models utilizing unobservable
inputs that therefore is considered a Level 3 item.
Fair
Value of Other Financial Instruments
As of March 31, 2010 and December 31, 2009 the
carrying amounts of cash, accounts receivable, accounts payable,
accrued interest and accrued expenses approximate fair value
based on the short maturity of these instruments.
| |
|
|
|
|
|
|
|
|
|
|
|
2010
|
|
2009
|
|
|
|
Long-term debt (including current portion of long-term debt)
|
|
$
|
215,540
|
|
|
$
|
217,750
|
|
The Company uses quoted market prices and yields for the same or
similar types of borrowings in active markets when available to
determine the fair value of the Company’s debt. The fair
values of all other debt were estimated using the present value
of expected future cash flows which incorporates the
Company’s risk profile. These fair values are considered
Level 2 items.
|
|
|
Note 11.
|
Subsequent
Events
|
On April 29, 2010, Aurora Diagnostics, Inc. filed a
registration statement on
Form S-1
with the Securities and Exchange Commission to initiate an
initial public offering. In connection with the offering, Aurora
Diagnostics, Inc. will enter into certain Reorganization
Transactions, including the creation or acquisition of
amortizable tax assets in connection with the offering and the
Reorganization Transactions, and the creation of liabilities in
connection with entering into a Tax Receivable Agreement
concurrent with the completion of the offering. These
transactions are more fully described in the
Form S-1.
In connection with the offering, the Company created Aurora
Diagnostics, Inc. as an incorporated entity in Delaware on
April 23, 2010.
On May 26, 2010, the Company entered into a new
$335.0 million credit facility with Barclays Bank PLC and
certain other lenders. This new credit facility includes a
$225.0 million senior secured first lien term loan facility
with a maturity date of 6 years after the closing of the
new financing. The new credit facility also includes a
$110.0 million senior secured first lien revolving credit
facility ($50.0 million of which became available upon the
closing of the new credit facility and $60.0 million of
which will be available upon the completion of an initial public
offering within nine months of closing the new credit facility)
that matures 4 years after the closing of the new
financing. The Company’s new term loan facility bears
interest, at the Company’s option, at a rate initially
equal to the prime rate plus 3.25 percent per annum or
LIBOR (subject to a floor of 2.00 percent) plus
4.25 percent per annum. The Company’s new credit
facility was used to refinance the 2007 Credit Facility and to
redeem the Company’s Class Z capital plus accrued
dividends and will also be used for acquisitions, working
capital and general corporate purposes. The Company will record
a non-cash write-off of any remaining unamortized original issue
discount and debt issue costs of approximately $4.4 million
related to its existing credit facilities during the period
ending June 30, 2010. In connection with the new credit
facility, the Company repaid all amounts outstanding under the
2007 Credit Facility and incurred a $2.3 million prepayment
penalty.
F-41
Independent
Auditor’s Report
To the Members
Aurora Diagnostics Holdings, LLC
Palm Beach Gardens, Florida
We have audited the accompanying consolidated balance sheet of
South Texas Dermatopathology Laboratory, P.A. and Subsidiaries
as of November 20, 2009, and the related consolidated
statement of operations, stockholders’ equity and cash
flows for the period from January 1, 2009 to
November 20, 2009. These financial statements are the
responsibility of the Company’s management. Our
responsibility is to express an opinion on these financial
statements based on our audit.
We conducted our audit in accordance with auditing standards
generally accepted in the United States of America. Those
standards require that we plan and perform the audit to obtain
reasonable assurance about whether the financial statements are
free of material misstatement. An audit includes examining, on a
test basis, evidence supporting the amounts and disclosures in
the financial statements. An audit also includes assessing the
accounting principles used and significant estimates made by
management, as well as evaluating the overall financial
statement presentation. We believe that our audit provides a
reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred
to above present fairly, in all material respects, the financial
position of South Texas Dermatopathology Laboratory, P.A. and
Subsidiaries as of November 20, 2009, and the results of
their consolidated operations and their cash flows for the
period from January 1, 2009 to November 20, 2009 in
conformity with accounting principles generally accepted in the
United States of America.
West Palm Beach, Florida
April 9, 2010
F-42
South
Texas Dermatopathology Laboratory, P.A. and
Subsidiaries
Consolidated
Balance Sheet
November 20, 2009
| |
|
|
|
|
|
|
|
($ in thousands)
|
|
|
|
|
Assets
|
|
Current Assets
|
|
|
|
|
|
Cash
|
|
$
|
162
|
|
|
Accounts receivable, net
|
|
|
563
|
|
|
Cash surrender value of life insurance
|
|
|
195
|
|
|
Other assets
|
|
|
67
|
|
|
Total current assets
|
|
|
987
|
|
|
Property and Equipment, net
|
|
|
1,034
|
|
|
|
|
|
|
|
|
|
|
$
|
2,021
|
|
|
|
|
|
|
|
|
|
|
Liabilities and Stockholders’ Equity
|
|
Current Liabilities
|
|
|
|
|
|
Notes payable
|
|
$
|
967
|
|
|
Accounts payable
|
|
|
14
|
|
|
Accrued compensation
|
|
|
205
|
|
|
Income tax payable
|
|
|
139
|
|
|
Deferred tax liabilities, current portion
|
|
|
143
|
|
|
|
|
|
|
|
|
Total current liabilities
|
|
|
1,468
|
|
|
|
|
|
|
|
|
Deferred Income Taxes, less current portion
|
|
|
321
|
|
|
|
|
|
|
|
|
Commitments and Contingencies
|
|
|
|
|
|
Stockholders’ Equity
|
|
|
|
|
|
Common Shares, $1 par value per share, 100,000 shares
authorized and 1,000 shares issued and outstanding
|
|
|
1
|
|
|
Additional paid-in capital
|
|
|
3
|
|
|
Retained earnings
|
|
|
228
|
|
|
|
|
|
|
|
|
Total stockholders’ equity
|
|
|
232
|
|
|
|
|
|
|
|
|
|
|
$
|
2,021
|
|
|
|
|
|
|
|
See Notes to Consolidated Financial Statements.
F-43
South
Texas Dermatopathology Laboratory, P.A. and
Subsidiaries
Consolidated
Statement of Operations
For the Period From January 1, 2009
through November 20, 2009
| |
|
|
|
|
|
|
|
($ in thousands)
|
|
|
|
|
Net Revenues
|
|
$
|
7,328
|
|
|
|
|
|
|
|
|
Operating costs and expenses:
|
|
|
|
|
|
Cost of services
|
|
|
5,127
|
|
|
Selling, general and administrative expenses
|
|
|
1,482
|
|
|
Depreciation expense
|
|
|
73
|
|
|
|
|
|
|
|
|
Total operating costs and expenses
|
|
|
6,682
|
|
|
|
|
|
|
|
|
Income from operations
|
|
|
646
|
|
|
Interest expense
|
|
|
(52
|
)
|
|
|
|
|
|
|
|
Income before income taxes
|
|
|
594
|
|
|
Provision for income taxes
|
|
|
215
|
|
|
|
|
|
|
|
|
Net income
|
|
$
|
379
|
|
|
|
|
|
|
|
See Notes to Consolidated Financial Statements.
F-44
South
Texas Dermatopathology Laboratory, P.A. and
Subsidiaries
Consolidated
Statement of Stockholders’ Equity
For the Period From January 1, 2009
through November 20, 2009
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Common Stock
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Outstanding
|
|
|
Additional
|
|
|
|
|
|
Total
|
|
|
|
|
|
|
|
Par
|
|
|
Paid-In
|
|
|
Retained
|
|
|
Shareholders’
|
|
|
|
|
Shares
|
|
|
Value
|
|
|
Capital
|
|
|
Earnings
|
|
|
Equity
|
|
|
|
|
($ in thousands, except shares)
|
|
|
|
|
Balance, December 31, 2008
|
|
|
1,000
|
|
|
$
|
1
|
|
|
$
|
3
|
|
|
$
|
(151
|
)
|
|
$
|
(147
|
)
|
|
Net income
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
379
|
|
|
|
379
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, November 20, 2009
|
|
|
1,000
|
|
|
$
|
1
|
|
|
$
|
3
|
|
|
$
|
228
|
|
|
$
|
232
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Consolidated Financial Statements.
F-45
South
Texas Dermatopathology, Laboratory P.A. and
Subsidiaries
Consolidated
Statement of Cash Flows
For the period from January 1, 2009
through November 20, 2009
| |
|
|
|
|
|
|
|
($ in thousands)
|
|
|
|
|
Cash Flows From Operating Activities
|
|
|
|
|
|
Net income
|
|
$
|
379
|
|
|
Adjustments to reconcile net income to net cash provided by
operating activities:
|
|
|
|
|
|
Depreciation
|
|
|
73
|
|
|
Deferred income taxes
|
|
|
66
|
|
|
Changes in assets and liabilities:
|
|
|
|
|
|
Increase in:
|
|
|
|
|
|
Accounts receivable
|
|
|
(81
|
)
|
|
Other assets
|
|
|
(46
|
)
|
|
Increase in:
|
|
|
|
|
|
Income tax payable
|
|
|
139
|
|
|
Accounts payable
|
|
|
14
|
|
|
Accrued compensation
|
|
|
22
|
|
|
|
|
|
|
|
|
Net cash provided by operating activities
|
|
|
566
|
|
|
|
|
|
|
|
|
Cash Flows From Investing Activities
|
|
|
|
|
|
Purchase of property and equipment
|
|
|
(54
|
)
|
|
|
|
|
|
|
|
Net cash used in investing activities
|
|
|
(54
|
)
|
|
|
|
|
|
|
|
Cash Flows From Financing Activities
|
|
|
|
|
|
Repayment of debt
|
|
|
(412
|
)
|
|
|
|
|
|
|
|
Net cash used in financing activities
|
|
|
(412
|
)
|
|
|
|
|
|
|
|
Net increase in cash
|
|
|
100
|
|
|
|
|
|
|
|
|
Cash:
|
|
|
|
|
|
Beginning
|
|
|
62
|
|
|
|
|
|
|
|
|
Ending
|
|
$
|
162
|
|
|
|
|
|
|
|
|
Supplemental Disclosures of Cash Flow Information
|
|
|
|
|
|
Cash interest payments
|
|
$
|
52
|
|
|
|
|
|
|
|
|
Cash tax payments
|
|
$
|
10
|
|
|
|
|
|
|
|
See Notes to Consolidated Financial Statements.
F-46
South
Texas Dermatopathology, Laboratory P.A. and Subsidiaries
|
|
|
Note 1.
|
Nature of
Business and Significant Accounting Policies
|
Nature of Business: South Texas
Dermatopathology Laboratory, P.A. (the “Company”) was
organized in the State of Texas as a professional business
corporation in 1983, to operate as a pathology laboratory
company.
The Company engages in the business of providing anatomic
pathology and cytology services to physicians.
A summary of the Company’s significant accounting policies
follows:
Principles of Consolidation: The
accompanying consolidated financial statements of the Company
include the accounts of South Texas Dermatopathology Laboratory,
P.A., S&D Texas Holdings, LLC, and STD Capital, L.P. All
accounts and transactions between the entities have been
eliminated in consolidation.
S & D Texas Holdings, LLC (“S &
D”) has been consolidated based on S & D being a
variable interest entity (“VIE”) and the Company being
the primary beneficiary. S & D owns commercial real
estate in Texas which is held for lease. Accounting principles
generally accepted in the United States of America requires an
entity to consolidate a VIE under certain conditions having to
do with the VIE’s activities, related risks and rewards,
and the equity investment of its owners. Management of the
Company has determined that the conditions requiring
consolidation of a VIE have been met. As of November 20,
2009, total assets and total liabilities of S & D were
approximately $925,000 and approximately $1,057,000,
respectively. The Company will reconsider its determination of
whether an entity is a VIE and who the primary beneficiary is if
certain events occur that are likely to cause a change in the
original determinations.
Accounting Estimates: The preparation
of financial statements in conformity with accounting principles
generally accepted in the United States of America requires
management to make estimates and assumptions that affect the
reported amounts of assets and liabilities, disclosure of
contingent assets and liabilities and the reported amounts of
revenues and expenses. Due to the inherent uncertainties in this
process, actual results could differ from those estimates.
Fair value of Financial Instruments: On
January 1, 2008, the Company adopted a new standard related
to the accounting for financial assets and liabilities and items
that are recognized or disclosed at fair value in the financial
statements on a recurring basis, at least annually.
On January 1, 2009, the Company adopted authoritative
guidance for its nonfinancial assets and liabilities that are
measured at fair value on a nonrecurring basis. The adoption of
the guidance did not materially impact the Company’s
disclosure requirements.
In August 2009, the Financial Accounting Standards Board
(“FASB”) issued an amendment to the accounting
standards related to the measurement of liabilities that are
recognized or disclosed at fair value.
This standard clarifies how a company should measure the fair
value of liabilities and that restrictions preventing the
transfer of a liability should not be considered as a factor in
the measurement of liabilities within the scope of this
standard. This standard became effective October 1, 2009.
The adoption of this standard did not have a material impact on
the Company’s consolidated financial statements.
The fair value accounting standards clarify the definition of
fair value, prescribes methods for measuring fair value,
establishes a fair value hierarchy based on the inputs used to
measure fair value, and expands disclosures about fair value
measurements. The three-tier fair value hierarchy, which
prioritizes the inputs used in the valuation methodologies, is
described below with Level 1 having the highest priority
and Level 3 having the lowest.
Level 1: Quoted prices in active markets
for identical assets or liabilities.
Level 2: Quoted prices for similar assets
or liabilities in active markets, quoted prices for identical or
similar instruments in markets that are not active, and
model-derived valuations in which all significant inputs are
observable in active markets.
F-47
South
Texas Dermatopathology, Laboratory P.A. and Subsidiaries
Notes to
Consolidated Financial
Statements — (Continued)
Level 3: Valuations derived from
valuation techniques in which one or more significant inputs are
unobservable.
Revenue Recognition and Accounts
Receivable: The Company recognizes revenue at
the time services are performed. Unbilled receivables are
recorded for services rendered during, but billed subsequent to,
the reporting period. Revenue is reported at the estimated
realizable amounts from patients, third-party payors and others
for services rendered. Revenue under certain third-party payor
agreements is subject to audit and retroactive adjustments.
Provisions for estimated third-party payor settlements and
adjustments are estimated in the period the related services are
rendered and adjusted in future periods as final settlements are
determined. The provision for doubtful accounts and the related
allowance are adjusted periodically, based upon an evaluation of
historical collection experience with specific payors for
particular services, anticipated collection levels with specific
payors for new services, industry reimbursement trends, and
other relevant factors. Changes in these factors in future
periods could result in increases or decreases in the
Company’s provision for doubtful accounts and impact its
results of operations and financial position. For the period
ended November 20, 2009, Medicare and BlueCross/Blue Shield
provided approximately 45% and 18% of the Company’s net
revenues, respectively.
Cash: The Company considers deposits
and investments that have original maturities of less than three
months, when purchased, to be cash equivalents. Daily bank
deposits are held in noninterest bearing accounts with a
financial institution that are insured by the Federal Deposit
Insurance Corporation’s (“FDIC”). These amounts
are guaranteed by the FDIC up to $250,000 as of
November 20, 2009. The Company has not experienced any
losses on such accounts.
Property and Equipment: Property and
equipment is stated at cost. Routine maintenance and repairs are
charged to expense as incurred, while costs of betterments and
renewals are capitalized. Depreciation is calculated on a
straight-line basis, over the estimated useful lives of the
respective assets, which range from 3 to 39 years.
Income Taxes: Deferred taxes are
provided on the liability method whereby deferred tax assets are
recognized for deductive temporary differences, and deferred tax
liabilities are recognized for taxable temporary differences.
Temporary differences are the differences between the reported
amounts of assets and liabilities and their tax bases. Deferred
tax assets are reduced by a valuation allowance when, in the
opinion of management, it is more likely than not that some
portion of all of the deferred tax assets will be realized.
Deferred tax assets and liabilities are adjusted for the effects
of changes in taxes laws and rates on the date of enactment. The
Company does not recognize a tax benefit, unless the Company
concludes that it is more likely than not that the benefit will
be sustained on audit by the taxing authority based solely on
the technical merits of the associated tax position. If the
recognition threshold is met, the Company recognizes a tax
benefit measured at the largest amount of the tax benefit that
the Company believes is greater than 50% likely to be realized.
The Company records interest and penalties in income tax expense.
Concentration of Credit Risk: Financial
instruments that potentially subject the Company to
concentrations of credit risk are cash and accounts receivable.
Concentration of credit risk with respect to accounts receivable
is mitigated by the diversity of the Company’s payors.
While the Company has receivables due from federal and state
governmental agencies, the Company does not believe such
receivables represent a credit risk since the related healthcare
programs are funded by federal and state governments and payment
is primarily dependent on submitting appropriate documentation.
New Accounting Standards: Effective
June 30, 2009, the Company adopted a newly issued
accounting standard related to accounting for and disclosure of
subsequent events in its consolidated financial statements. This
standard provides the authoritative guidance for subsequent
events that was previously addressed only in United States
auditing standards. This standard establishes general accounting
for and disclosure of events that occur after the balance sheet
date but before financial statements are issued or are available
to be issued and requires the Company to disclose that it has
evaluated subsequent events through the date of the filing or
issue date. This standard does not apply to subsequent events or
transactions that are within the scope of other applicable GAAP
that
F-48
South
Texas Dermatopathology, Laboratory P.A. and Subsidiaries
Notes to
Consolidated Financial
Statements — (Continued)
provide different guidance on the accounting treatment for
subsequent events or transactions. The adoption of this standard
did not have a material impact on the Company’s
consolidated financial statements.
In June 2009, the FASB issued the FASB Accounting Standards
Codification (the “ASC”). The ASC has become the
single source of non-governmental accounting principles
generally accepted in the United States of America
(“GAAP”) recognized by the FASB in the preparation of
financial statements. The Company adopted the ASC as of
July 1, 2009. The ASC does not change GAAP and did not have
an effect on the Company’s consolidated financial position,
results of operations or cash flows.
In June 2009, the FASB issued new accounting guidance on when an
entity should be included in consolidated financial statements.
The new guidance amends the evaluation criteria to identify the
primary beneficiary of a variable interest entity, or
“VIE,” and requires ongoing reassessment of whether an
enterprise is the primary beneficiary of the VIE. The new
guidance significantly changes the consolidation rules for VIEs
including the consolidation of common structures, such as joint
ventures, equity-method investments, and collaboration
arrangements. The guidance is applicable to all new and existing
VIEs. This standard is effective for the Company as of
January 1, 2010 and the Company does not expect the impact
of its adoption to be material to its consolidated financial
statements.
Subsequent Events: The Company has
evaluated all subsequent events through April 9, 2010, the
date of issuance of the consolidated financial statements.
|
|
|
Note 2.
|
Accounts
Receivable
|
Accounts receivable consist of the following as of
November 20, 2009 (in thousands):
| |
|
|
|
|
|
Accounts receivable and unbilled receivables
|
|
$
|
843
|
|
|
Less: allowance for bad debt
|
|
|
(280
|
)
|
|
|
|
|
|
|
|
Accounts receivable, net
|
|
$
|
563
|
|
|
|
|
|
|
|
|
|
|
Note 3.
|
Property
and Equipment
|
Property and equipment as of November 20, 2009 consist of
the following (in thousands):
| |
|
|
|
|
|
|
|
|
|
Estimated Useful
|
|
|
|
|
|
|
Life (Years)
|
|
|
|
|
|
|
Buildings and improvements
|
|
5 – 39
|
|
$
|
954
|
|
|
Vehicles
|
|
5 – 7
|
|
|
31
|
|
|
Furniture and fixtures
|
|
7
|
|
|
158
|
|
|
Laboratory, office and data processing equipment
|
|
3 – 5
|
|
|
631
|
|
|
Software
|
|
3
|
|
|
47
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1,821
|
|
|
Less accumulated depreciation
|
|
|
|
|
(984
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
837
|
|
|
Land
|
|
|
|
|
197
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
1,034
|
|
|
|
|
|
|
|
|
|
Depreciation expense was approximately $73,000 for the period
from January 1, 2009 to November 20, 2009.
F-49
South
Texas Dermatopathology, Laboratory P.A. and Subsidiaries
Notes to
Consolidated Financial
Statements — (Continued)
On April 25, 2005, the Company entered into a mortgage note
for the purchase of its building and land. The agreement was
collateralized by substantially all of the Company’s assets
and guaranteed by owners of the Company. Interest was paid in
arrears at 5.5%, and the note’s original maturity date was
April 25, 2020. In December 2009, the physician employee
stockholders paid off the mortgage note in full.
|
|
|
Note 5.
|
Related
Party Transactions
|
The Company has employment agreements with certain of its
stockholders who are physician employees which expire at various
times. Bonuses are discretionary and are based upon the
reasonable value of the physicians’ contribution. Under
certain of the agreements, in the event employment is terminated
due to death, retirement or disability, the employee will
receive an additional compensation for up to 60 days. For
the period from January 1, 2009 through November 20,
2009, the Company paid salaries and bonuses to stockholders who
are physician employees of approximately $1.24 million and
$2.16 million, respectively.
|
|
|
Note 6.
|
Commitments
and Contingencies
|
During the ordinary course of business, the Company may become
subject to legal actions and proceedings. The Company may have
liability with respect to its employees and its pathologists.
Medical malpractice claims are generally covered by insurance.
While the Company believes the outcome of any such legal actions
and proceedings, individually or in the aggregate, will not have
a material adverse effect on the Company’s financial
condition, results of operations or liquidity, if the Company is
ultimately found liable under any medical malpractice claims,
there can be no assurance the Company’s medical malpractice
insurance coverage will be adequate to cover any such liability.
Healthcare Regulatory Environment: The
healthcare industry, in general, and the services the Company
provides, are subject to extensive federal and state laws and
regulations. Additionally, a portion of the Company’s
revenue is from payments by government-sponsored health
programs, including Medicare, and is subject to audit and
adjustments by applicable regulatory agencies. Failure to comply
with any of these laws or regulations, the results of increased
regulatory audits and adjustments, or change in the
interpretation of the coding of services or the amounts payable
for the Company’s services under these programs could have
a material adverse effect on the Company’s financial
position and results of operations.
|
|
|
Note 7.
|
Fair
Value of Financial Instruments
|
Certain assets that are measured at fair value on a
non-recurring basis, including property and equipment are
adjusted to fair value only when the carrying values are greater
than their fair values.
As of November 20, 2009 the carrying amounts of cash,
accounts receivable, accounts payable, and accrued compensation
approximate their fair value based on the short maturity of
these instruments.
F-50
South
Texas Dermatopathology, Laboratory P.A. and Subsidiaries
Notes to
Consolidated Financial
Statements — (Continued)
The provision for income taxes for the period ended
November 20, 2009 consists of the following (in thousands):
| |
|
|
|
|
|
Current:
|
|
|
|
|
|
Federal
|
|
$
|
139
|
|
|
State
|
|
|
10
|
|
|
|
|
|
|
|
|
Total current provision
|
|
|
149
|
|
|
|
|
|
|
|
|
Deferred:
|
|
|
|
|
|
Federal
|
|
|
65
|
|
|
State
|
|
|
1
|
|
|
|
|
|
|
|
|
Total deferred benefit
|
|
|
66
|
|
|
|
|
|
|
|
|
Total provision for income taxes
|
|
$
|
215
|
|
|
|
|
|
|
|
Temporary differences between the financial statement carrying
amounts and tax basis of assets and liabilities that give rise
to significant portions of the deferred tax assets and
liabilities are summarized below as of November 20, 2009:
| |
|
|
|
|
|
Accrued wages
|
|
$
|
72
|
|
|
Allowance for doubtful accounts
|
|
|
(197
|
)
|
|
Other
|
|
|
(18
|
)
|
|
|
|
|
|
|
|
Current deferred tax liabilities, net
|
|
|
(143
|
)
|
|
Non current deferred tax liabilities:
|
|
|
|
|
|
Property and equipment
|
|
$
|
(321
|
)
|
|
Noncurrent deferred tax liabilities, net
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
(321
|
)
|
|
Total deferred tax liabilities, net
|
|
$
|
(464
|
)
|
|
|
|
|
|
|
|
|
|
Note 9.
|
Employee
Benefit Plan
|
The Company has a profit sharing 401(k) Plan covering
substantially all of its employees. Eligible participants may
elect to defer a percentage of their compensation not to exceed
the maximum amount of elective contribution determined by law.
The employer may elect to make a qualified matching contribution
for a plan year. Contributions are based on the sole discretion
of the Company. During the year ended November 20, 2009,
the Company contributed approximately $4,500 to the plan on
behalf of its employees.
F-51
South
Texas Dermatopathology, Laboratory P.A. and Subsidiaries
Notes to
Consolidated Financial
Statements — (Continued)
|
|
|
Note 10.
|
Cash
Surrender Value Life Insurance
|
The Company purchased insurance on the lives of its two
physician stockholders. As beneficiary, the Company receives the
cash surrender value if the policy is terminated and, upon death
of the insured, receives all of the benefits payable. The cash
value of the life insurance policies was approximately $195,000
as of November 20, 2009. Subsequent to November 2009, the
policies were terminated and the cash surrender value was
distributed to the physician employee stockholders.
|
|
|
Note 11.
|
Subsequent
Event
|
On November 20, 2009, the Company was acquired from its
stockholders by Aurora Diagnostics Holdings, LLC.
F-52
Independent
Auditor’s Report
To the Members
Aurora Diagnostics Holdings, LLC
Palm Beach Gardens, Florida
We have audited the accompanying balance sheets of Twin Cities
Dermatopathology, P.A. as of March 7, 2008 and
December 31, 2007, and the related statements of
operations, stockholder’s equity, and cash flows for the
period from January 1, 2008 through March 7, 2008 and
for the year ended December 31, 2007. These financial
statements are the responsibility of the Company’s
management. Our responsibility is to express an opinion on these
financial statements based on our audits.
We conducted our audits in accordance with auditing standards
generally accepted in the United States of America. Those
standards require that we plan and perform the audit to obtain
reasonable assurance about whether the financial statements are
free of material misstatement. An audit includes examining, on a
test basis, evidence supporting the amounts and disclosures in
the financial statements. An audit also includes assessing the
accounting principles used and significant estimates made by
management, as well as evaluating the overall financial
statement presentation. We believe that our audits provide a
reasonable basis for our opinion.
In our opinion, the financial statements referred to above
present fairly, in all material respects, the financial position
of Twin Cities Dermatopathology, P.A. as of March 7, 2008
and December 31, 2007 and, the results of its operations
and its cash flows for the period January 1, 2008 through
March 7, 2008 and for the year then ended December 31,
2007 in conformity with accounting principles generally accepted
in the United States of America.
West Palm Beach, Florida
April 9, 2010
F-53
Twin
Cities Dermatopathology, P.A.
| |
|
|
|
|
|
|
|
|
|
|
|
March 7, 2008
|
|
December 31,
2007
|
|
|
|
($ in thousands)
|
|
|
|
Assets
|
|
|
|
|
|
|
|
|
|
Current Assets
|
|
|
|
|
|
|
|
|
|
Cash
|
|
$
|
892
|
|
|
$
|
1,413
|
|
|
Accounts receivable, net
|
|
|
620
|
|
|
|
565
|
|
|
Prepaid expenses
|
|
|
9
|
|
|
|
9
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current assets
|
|
|
1,521
|
|
|
|
1,987
|
|
|
Property and Equipment, net
|
|
|
106
|
|
|
|
95
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
,1,627
|
|
|
$
|
2,082
|
|
|
|
|
|
|
|
|
|
|
|
|
Liabilities and Stockholder’s Equity
|
|
|
|
|
|
|
|
|
|
Current Liabilities
|
|
|
|
|
|
|
|
|
|
Accounts payable
|
|
$
|
81
|
|
|
$
|
53
|
|
|
Accrued compensation
|
|
|
249
|
|
|
|
228
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current liabilities
|
|
|
330
|
|
|
|
281
|
|
|
|
|
|
|
|
|
|
|
|
|
Stockholder’s Equity
|
|
|
|
|
|
|
|
|
|
Common Shares, $ .01 par value per share, 5,000 shares
authorized and 500 shares issued and outstanding
|
|
|
—
|
|
|
|
—
|
|
|
Retained earnings
|
|
|
1,297
|
|
|
|
1,801
|
|
|
|
|
|
|
|
|
|
|
|
|
Total stockholder’s equity
|
|
|
1,297
|
|
|
|
1,801
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
1,627
|
|
|
$
|
2,082
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Financial Statements.
F-54
Twin
Cities Dermatopathology, P.A.
Statements of Operations
Period from January 1, 2008 through March 7, 2008
and For the Year Ended December 31, 2007
| |
|
|
|
|
|
|
|
|
|
|
|
January 1, 2008
|
|
|
|
|
|
Through
|
|
Year Ended
|
|
|
|
March 7, 2008
|
|
December 31,
2007
|
|
|
|
($ in thousands)
|
|
|
|
Net Revenues
|
|
$
|
1,293
|
|
|
$
|
8,409
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating costs and expenses:
|
|
|
|
|
|
|
|
|
|
Cost of services
|
|
|
562
|
|
|
|
2,633
|
|
|
Selling, general and administrative expenses
|
|
|
98
|
|
|
|
411
|
|
|
Depreciation expense
|
|
|
7
|
|
|
|
88
|
|
|
|
|
|
|
|
|
|
|
|
|
Total operating costs and expenses
|
|
|
667
|
|
|
|
3,132
|
|
|
|
|
|
|
|
|
|
|
|
|
Income from operations
|
|
|
626
|
|
|
|
5,277
|
|
|
Interest income
|
|
|
3
|
|
|
|
23
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income
|
|
$
|
629
|
|
|
$
|
5,300
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Financial Statements.
F-55
Twin
Cities Dermatopathology, P.A.
Statement of Stockholder’s
Equity
For the period from January 1, 2008 through March 7,
2008
and For the Year Ended December 31, 2007
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Common Stock Outstanding
|
|
|
|
Total
|
|
|
|
|
|
Par
|
|
Retained
|
|
Stockholder’s
|
|
|
|
Shares
|
|
Value
|
|
Earnings
|
|
Equity
|
|
|
|
($ in thousands, except shares)
|
|
|
|
Balance, December 31, 2006
|
|
|
500
|
|
|
$
|
—
|
|
|
$
|
1,550
|
|
|
$
|
1,550
|
|
|
Distributions
|
|
|
—
|
|
|
|
—
|
|
|
|
(5,049
|
)
|
|
|
(5,049
|
)
|
|
Net income
|
|
|
—
|
|
|
|
—
|
|
|
|
5,300
|
|
|
|
5,300
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, December 31, 2007
|
|
|
500
|
|
|
|
—
|
|
|
|
1,801
|
|
|
|
1,801
|
|
|
Distributions
|
|
|
—
|
|
|
|
—
|
|
|
|
(1,133
|
)
|
|
|
(1,133
|
)
|
|
Net income
|
|
|
—
|
|
|
|
—
|
|
|
|
629
|
|
|
|
629
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, March 7, 2008
|
|
|
500
|
|
|
$
|
—
|
|
|
$
|
1,297
|
|
|
$
|
1,297
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Financial Statements.
F-56
Twin
Cities Dermatopathology, P.A.
Statements of Cash Flows
For the period from January 1, 2008 through March 7,
2008
and For the Year Ended December 31, 2007
| |
|
|
|
|
|
|
|
|
|
|
|
January 1, 2008
|
|
|
|
|
|
Through
|
|
Year Ended
|
|
|
|
March 7, 2008
|
|
December 31,
2007
|
|
|
|
($ in thousands)
|
|
|
|
Cash Flows From Operating Activities
|
|
|
|
|
|
|
|
|
|
Net income
|
|
$
|
629
|
|
|
$
|
5,300
|
|
|
Adjustments to reconcile net income to net cash provided by
operating activities:
|
|
|
|
|
|
|
|
|
|
Depreciation
|
|
|
7
|
|
|
|
88
|
|
|
Allowance for bad debt
|
|
|
67
|
|
|
|
16
|
|
|
Changes in assets and liabilities:
|
|
|
|
|
|
|
|
|
|
(Increase) Decrease in:
|
|
|
|
|
|
|
|
|
|
Accounts receivable
|
|
|
(122
|
)
|
|
|
(11
|
)
|
|
Prepaid expenses
|
|
|
—
|
|
|
|
8
|
|
|
Increase in:
|
|
|
|
|
|
|
|
|
|
Accounts payable
|
|
|
28
|
|
|
|
20
|
|
|
Accrued compensation
|
|
|
21
|
|
|
|
32
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash provided by operating activities
|
|
|
630
|
|
|
|
5,453
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash Flows From Investing Activities
|
|
|
|
|
|
|
|
|
|
Purchase of property and equipment
|
|
|
(18
|
)
|
|
|
(42
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Net cash used in investing activities
|
|
|
(18
|
)
|
|
|
(42
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Cash Flows From Financing Activities
|
|
|
|
|
|
|
|
|
|
Distributions
|
|
|
(1,133
|
)
|
|
|
(5,049
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Net cash used in financing activities
|
|
|
(1,133
|
)
|
|
|
(5,049
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Net increase (decrease) in cash
|
|
|
(521
|
)
|
|
|
362
|
|
|
Cash, beginning
|
|
|
1,413
|
|
|
|
1,051
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash, ending
|
|
$
|
892
|
|
|
$
|
1,413
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Financial Statements.
F-57
Twin
Cities Dermatopathology, P.A.
|
|
|
Note 1.
|
Nature of
Business and Significant Accounting Policies
|
Nature of Business: Twin Cities
Dermatopathology, P.A. (the “Company”) was organized
in the State of Minnesota as a professional business corporation
in January 1990 to operate as a pathology laboratory.
The Company engages in the business of providing anatomic
pathology and cytology services to physicians in the state of
Minnesota.
A summary of the Company’s significant accounting policies
follows:
Accounting Estimates: The preparation
of financial statements in conformity with accounting principles
generally accepted in the United States of America requires
management to make estimates and assumptions that affect the
reported amounts of assets and liabilities, disclosure of
contingent assets and liabilities and the reported amounts of
revenues and expenses. Due to the inherent uncertainties in this
process, actual results could differ from those estimates.
Fair value of Financial
Instruments: The Company’s financial
instruments consist mainly of cash, accounts receivable, and
accounts payable. The carrying amounts of the Company’s
cash, accounts receivable, and accounts payable approximate fair
value due to the short-term nature of these instruments.
Revenue Recognition and Accounts
Receivable: The Company recognizes revenue at
the time services are performed. Unbilled receivables are
recorded for services rendered during, but billed subsequent to,
the reporting period. Revenue is reported at the estimated
realizable amounts from patients, third-party payors and others
for services rendered. Revenue under certain third-party payor
agreements is subject to audit and retroactive adjustments.
Provisions for estimated third-party payor settlements and
adjustments are estimated in the period the related services are
rendered and adjusted in future periods as final settlements are
determined. The provision for doubtful accounts and the related
allowance are adjusted periodically, based upon an evaluation of
historical collection experience with specific payors for
particular services, anticipated collection levels with specific
payors for new services, industry reimbursement trends, and
other relevant factors. Changes in these factors in future
periods could result in increases or decreases in the
Company’s provision for doubtful accounts and impact its
results of operations and financial position. For the period
ended March 7, 2008 and year ended December 31, 2007,
Medicare provided 15% and 20% of net revenues, respectively, and
Blue Cross/Blue Shield, provided 15% of net revenues in each
period, respectively.
Cash: The Company considers deposits
and investments that have original maturities of less than three
months, when purchased, to be cash equivalents. Daily bank
deposits are held in noninterest-bearing accounts with a
financial institution that guaranteed by the Federal Deposit
Insurance Corporation’s (“FDIC”) up to $100,000
as of March 7, 2008. The Company has not experienced any
losses on such accounts.
Property and Equipment: Property and
equipment is stated at cost. Routine maintenance and repairs are
charged to expense as incurred, while costs of betterments and
renewals are capitalized. Depreciation is calculated on a
straight-line basis, over the estimated useful lives of the
respective assets, which range from 3 to 10 years.
Leasehold improvements are amortized over the shorter of the
term of the related lease, or the useful life of the asset.
Income Taxes: The Company is a
subchapter S Corporation for federal and state income tax
purposes, in accordance with the applicable provisions of the
Internal Revenue Code. Accordingly, the Company is generally not
subject to income taxes. The income attributable to the Company
is distributed to the stockholder.
Concentration of Credit Risk: Financial
instruments that potentially subject the Company to
concentrations of credit risk are cash and accounts receivable.
Concentration of credit risk with respect to accounts receivable
is mitigated by the diversity of the Company’s payors.
While the Company has receivables due from federal and state
governmental agencies, the Company does not believe such
receivables represent a credit risk since the related healthcare
programs are funded by federal and state governments and payment
is primarily dependent on submitting appropriate documentation.
F-58
Twin
Cities Dermatopathology, P.A.
Notes to
Financial Statements — (Continued)
Recent Accounting Pronouncements: In
July 2006, the FASB issued Interpretation No. 48,
Accounting for Uncertainty in Income Taxes — an
interpretation of FASB Statement 109 (“FIN 48”).
FIN 48 clarifies the accounting for uncertainty in income
taxes recognized in an enterprise’s financial statements in
accordance with SFAS 109, Accounting for Income
Taxes. FIN 48 prescribes a comprehensive model for
recognizing, measuring, presenting and disclosing in the
financial statements tax positions taken or expected to be taken
on a tax return. If there are changes in net assets as a result
of application of FIN 48, these will be accounted for as an
adjustment to the opening balance of retained earnings.
Additional disclosures about the amounts of such liabilities
will also be required. In October 2008, the FASB delayed the
effective date of FIN 48 for certain nonpublic enterprises
to annual financial statements for fiscal years beginning after
December 15, 2008. The Company was not required to adopt
FIN 48 for these financial statements. The Company does not
expect the adoption of this statement will materially impact its
financial statements.
In September 2006, Statement of Financial Accounting Standards
No. 157, Fair Value Measurements
(“SFAS 157”) applies to all assets and
liabilities being measured and reported on a fair value basis.
SFAS 157 requires new disclosure that establishes a
framework for measuring fair value in GAAP, and expands
disclosure about fair value measurements. In February 2008, the
FASB issued FASB Staff Position (“FSP”)
No. 157-2,
Effective Date of FASB Statement No. 157, which
deferred the effective date of SFAS 157 for one year for
non-financial assets and non-financial liabilities, except for
items recognized or disclosed at fair value in the financial
statements on a recurring basis for fiscal years beginning after
November 15, 2008.
SFAS 157 establishes a hierarchy ranking the quality and
reliability of the information used to determine fair values.
The statement requires assets and liabilities carried at fair
value will be classified and disclosed in one of the three
following categories:
a. Level 1: Quoted market prices in
active markets for identical assets or liabilities.
b. Level 2: Observable market based
inputs or unobservable inputs corroborated by market data.
c. Level 3: Unobservable inputs not
corroborated by market data.
The Company does not expect the adoption of this Statement will
have a material impact on its financial position or results of
operations.
|
|
|
Note 2.
|
Accounts
Receivable
|
Accounts receivable consist of the following as of (in
thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
March 7, 2008
|
|
December 31,
2007
|
|
|
|
Accounts receivable and unbilled receivables
|
|
$
|
818
|
|
|
$
|
696
|
|
|
Less: allowance for bad debts
|
|
|
(198
|
)
|
|
|
(131
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Accounts receivable, net
|
|
$
|
620
|
|
|
$
|
565
|
|
|
|
|
|
|
|
|
|
|
|
F-59
Twin
Cities Dermatopathology, P.A.
Notes to
Financial Statements — (Continued)
|
|
|
Note 3.
|
Property
and Equipment
|
Property and equipment consist of the following as of (in
thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Estimated Useful
|
|
March 7,
|
|
December 31,
|
|
|
|
Life (Years)
|
|
2008
|
|
2007
|
|
|
|
Laboratory, office and data processing equipment
|
|
|
5
|
|
|
$
|
686
|
|
|
$
|
669
|
|
|
Leasehold improvements
|
|
|
10
|
|
|
|
34
|
|
|
|
34
|
|
|
Furniture and fixtures
|
|
|
5
|
|
|
|
70
|
|
|
|
69
|
|
|
Software
|
|
|
3
|
|
|
|
181
|
|
|
|
181
|
|
|
|
|
|
|
|
|
|
971
|
|
|
|
953
|
|
|
Less accumulated depreciation
|
|
|
|
|
|
|
(865
|
)
|
|
|
(858
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
106
|
|
|
$
|
95
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Depreciation expense was approximately $7,000 for the period
from January 1, 2008 to March 7, 2008 and $88,000 for
the year ended December 31, 2007.
|
|
|
Note 4.
|
Related
Party Transactions
|
The Company has an employment agreement with its sole
stockholder, who is also a physician employee, the terms of
which renew automatically for consecutive twelve-month periods.
The agreement provides for a minimum salary level, adjusted
annually for
cost-of-living
changes. For the period from January 1, 2008 through
March 7, 2008 and year ended December 31, 2007, the
Company paid salaries of approximately $67,000 and $373,000,
respectively, under this agreement.
|
|
|
Note 5.
|
Commitments
and Contingencies
|
During the ordinary course of business, the Company may become
subject to legal actions and proceedings. The Company may have
liability with respect to its employees and its pathologists.
Medical malpractice claims are generally covered by insurance.
While the Company believes the outcome of any such legal actions
and proceedings, individually or in the aggregate, will not have
a material adverse effect on the Company’s financial
condition, results of operations or liquidity, if the Company is
ultimately found liable under any medical malpractice claims,
there can be no assurance the Company’s medical malpractice
insurance coverage will be adequate to cover any such liability.
Healthcare Regulatory Environment: The
healthcare industry, in general, and the services the Company
provides, are subject to extensive federal and state laws and
regulations. Additionally, a portion of the Company’s
revenue is from payments by government-sponsored health
programs, including Medicare, and is subject to audit and
adjustments by applicable regulatory agencies. Failure to comply
with any of these laws or regulations, the results of increased
regulatory audits and adjustments, or change in the
interpretation of the coding of services or the amounts payable
for the Company’s services under these programs could have
a material adverse effect on the Company’s financial
position and results of operations.
Operating Leases: The Company leases
office and medical laboratory facilities and equipment under
non-cancelable lease agreements with terms ranging from two to
ten years and expiring through 2011. The terms of some of the
facility leases call for the Company to pay for certain taxes or
common utility charges. Rent expense including these taxes and
common utility charges was approximately $9,200 for the period
from January 1, 2008 through March 7, 2008 and $55,000
for the year ended December 31, 2007. Rent expense
associated with operating leases that include scheduled rent
increases and tenant incentives is recorded on a straight-line
basis over the term of the lease.
F-60
Twin
Cities Dermatopathology, P.A.
Notes to
Financial Statements — (Continued)
Aggregate future minimum annual rentals under the lease
agreements are as of March 7, 2008 are as follows (in
thousands):
| |
|
|
|
|
|
2009
|
|
$
|
55
|
|
|
2010
|
|
|
55
|
|
|
2011
|
|
|
55
|
|
|
2012
|
|
|
14
|
|
|
|
|
|
|
|
|
|
|
$
|
179
|
|
|
|
|
|
|
|
|
|
|
Note 6.
|
Employee
Benefit Plan
|
The Company has a defined contribution plan covering
substantially all of its employees. Eligible participants may
elect to defer a percentage of their compensation not to exceed
the maximum amount of elective contribution determined by law.
The Company may elect to make a matching contribution based on a
percentage determined by the Company of the employee’s
annual elective contributions or a percentage of the
employee’s annual compensation. The Company contributed
approximately $1,200 and $114,000 for the period from
January 1, 2008 to March 7, 2008 and for the year
ended December 31, 2007, respectively, to the plan.
|
|
|
Note 7.
|
Subsequent
Events
|
On March 7, 2008, the Company was acquired from its
stockholder by Aurora Diagnostics Holdings, LLC.
F-61
To the Members
Aurora Diagnostics Holdings, LLC
Palm Beach Gardens, Florida
We have audited the accompanying balance sheet of Laboratory
Medicine Consultants, LTD. (the “Company”) as of
December 10, 2007, and the related statement of operations
and stockholders’ deficit and cash flows for the period
from January 1, 2007 to December 10, 2007. These
financial statements are the responsibility of the
Company’s management. Our responsibility is to express an
opinion on these financial statements based on our audit.
We conducted our audit in accordance with auditing standards
generally accepted in the United States of America. Those
standards require that we plan and perform the audit to obtain
reasonable assurance about whether the financial statements are
free of material misstatement. An audit includes examining, on a
test basis, evidence supporting the amounts and disclosures in
the financial statements. An audit also includes assessing the
accounting principles used and significant estimates made by
management, as well as evaluating the overall financial
statement presentation. We believe that our audit provides a
reasonable basis for our opinion.
In our opinion, the financial statements referred to above
present fairly, in all material respects, the financial position
of Laboratory Medicine Consultants, LTD. as of December 10,
2007, and the results of its operations and its cash flows for
the period from January 1, 2007 to December 10, 2007
in conformity with accounting principles generally accepted in
the United States of America.
West Palm Beach, Florida
April 9, 2010
F-62
Laboratory
Medicine Consultants, LTD.
Balance
Sheet
December 10, 2007
| |
|
|
|
|
|
|
|
($ in thousands)
|
|
Assets
|
|
|
|
|
|
Current Assets
|
|
|
|
|
|
Cash
|
|
$
|
56
|
|
|
Accounts receivable, net
|
|
|
3,258
|
|
|
|
|
|
|
|
|
Total current assets
|
|
|
3,314
|
|
|
Property and Equipment, net
|
|
|
490
|
|
|
Deferred Tax Assets
|
|
|
2,308
|
|
|
|
|
|
|
|
|
|
|
$
|
6,112
|
|
|
|
|
|
|
|
|
Liabilities and Members’ Equity
|
|
|
|
|
|
Current Liabilities
|
|
|
|
|
|
Notes payable
|
|
$
|
—
|
|
|
Accounts payable
|
|
|
309
|
|
|
Post employment obligation, current portion
|
|
|
1,888
|
|
|
Accrued compensation
|
|
|
150
|
|
|
Income taxes payable
|
|
|
51
|
|
|
Deferred tax liabilities
|
|
|
951
|
|
|
Total current liabilities
|
|
|
3,349
|
|
|
|
|
|
|
|
|
Post employment obligation, less current portion
|
|
|
5,146
|
|
|
|
|
|
|
|
|
Commitments and Contingencies
|
|
|
|
|
|
Stockholders’ Deficit
|
|
|
|
|
|
Common Shares, $10.00 par value, 3,225 shares
authorized;
|
|
|
25
|
|
|
2,500 shares issued and outstanding
|
|
|
|
|
|
Retained earnings
|
|
|
(2,408
|
)
|
|
|
|
|
|
|
|
Total stockholders’ deficit
|
|
|
(2,383
|
)
|
|
|
|
|
|
|
|
|
|
$
|
6,112
|
|
|
|
|
|
|
|
See Notes to Financial Statements.
F-63
Laboratory
Medicine Consultants, LTD.
Statement
of Operations
For the period from January 1, 2007 through
December 10, 2007
| |
|
|
|
|
|
|
|
($ in thousands)
|
|
Net Revenues
|
|
$
|
17,562
|
|
|
Operating costs and expenses:
|
|
|
|
|
|
Cost of services
|
|
|
14,272
|
|
|
Selling, general and administrative expenses
|
|
|
1,314
|
|
|
Depreciation expense
|
|
|
191
|
|
|
|
|
|
|
|
|
Total operating costs and expenses
|
|
|
15,777
|
|
|
|
|
|
|
|
|
Income from operations
|
|
|
1,785
|
|
|
|
|
|
|
|
|
Interest expense
|
|
|
14
|
|
|
Income before income taxes
|
|
|
1,771
|
|
|
Provision for income taxes
|
|
|
650
|
|
|
|
|
|
|
|
|
Net income
|
|
$
|
1,121
|
|
|
|
|
|
|
|
See Notes to Financial Statements.
F-64
Laboratory
Medicine Consultants, LTD.
Statement
of Stockholders’ Deficit
For the period from January 1, 2007 through
December 10, 2007
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Common Stock
|
|
|
|
|
|
|
|
Outstanding
|
|
|
|
|
|
|
|
|
|
Par
|
|
Accumulated
|
|
Total Stockholders’
|
|
|
|
Shares
|
|
Value
|
|
Deficit
|
|
Deficit
|
|
|
|
($ in thousands, except shares)
|
|
|
|
Balance, December 31, 2006
|
|
|
2,500
|
|
|
$
|
25
|
|
|
$
|
(3,529
|
)
|
|
$
|
(3,504
|
)
|
|
Net income
|
|
|
—
|
|
|
|
—
|
|
|
|
1,121
|
|
|
|
1,121
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, December 10, 2007
|
|
|
2,500
|
|
|
$
|
25
|
|
|
$
|
(2,408
|
)
|
|
$
|
(2,383
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Financial Statements.
F-65
Laboratory
Medicine Consultants, LTD.
Statement
of Cash Flows
For the period from January 1, 2007 through
December 10, 2007
| |
|
|
|
|
|
|
|
($ in thousands)
|
|
Cash Flows From Operating Activities
|
|
|
|
|
|
Net income
|
|
$
|
1,121
|
|
|
Adjustments to reconcile net income to net cash provided by
|
|
|
|
|
|
operating activities:
|
|
|
|
|
|
Depreciation
|
|
|
191
|
|
|
Deferred income taxes
|
|
|
599
|
|
|
Changes in assets and liabilities:
|
|
|
|
|
|
Increase in:
|
|
|
|
|
|
Accounts receivable
|
|
|
411
|
|
|
Increase (decrease) in:
|
|
|
|
|
|
Accounts payable
|
|
|
(636
|
)
|
|
Post employment obligation
|
|
|
(687
|
)
|
|
Accrued compensation
|
|
|
(123
|
)
|
|
Income taxes payable
|
|
|
51
|
|
|
|
|
|
|
|
|
Net cash provided by operating activities
|
|
|
927
|
|
|
|
|
|
|
|
|
Cash Flows From Investing Activities
|
|
|
|
|
|
Purchase of property and equipment
|
|
|
(99
|
)
|
|
|
|
|
|
|
|
Net cash used in investing activities
|
|
|
(99
|
)
|
|
|
|
|
|
|
|
Cash Flows From Financing Activities
|
|
|
|
|
|
Repayment of notes payable
|
|
|
(908
|
)
|
|
|
|
|
|
|
|
Net cash used in financing activities
|
|
|
(908
|
)
|
|
|
|
|
|
|
|
Net decrease in cash
|
|
|
(80
|
)
|
|
Cash:
|
|
|
|
|
|
Beginning
|
|
|
136
|
|
|
|
|
|
|
|
|
Ending
|
|
$
|
56
|
|
|
|
|
|
|
|
|
Supplemental Disclosures of Cash Flow Information
|
|
|
|
|
|
Cash interest payments
|
|
$
|
14
|
|
|
|
|
|
|
|
|
Cash tax payments
|
|
$
|
—
|
|
|
|
|
|
|
|
See Notes to Financial Statements.
F-66
Laboratory
Medicine Consultants, LTD.
|
|
|
Note 1.
|
Nature of
Business and Significant Accounting Policies
|
Nature of Business: The Company is an
independent multi-specialty anatomic and clinical pathology
laboratory located in Las Vegas, Nevada. As an independent
laboratory, in operation since 1969, the Company serves
independent physicians and hospitals throughout Nevada.
The Company is organized under Nevada law as a C corporation for
tax purposes.
A summary of the Company’s significant accounting policies
follows:
Accounting Estimates: The preparation
of financial statements in conformity with accounting principles
generally accepted in the United States of America requires
management to make estimates and assumptions that affect the
reported amounts of assets and liabilities, disclosure of
contingent assets and liabilities and the reported amounts of
revenues and expenses. Due to the inherent uncertainties in this
process, actual results could differ from those estimates.
Fair Value of Financial
Instruments: The Company’s financial
instruments consist mainly of cash, accounts receivable and
accounts payable. The carrying amounts of the Company’s
cash, accounts receivable and accounts payable approximate fair
value due to the short-term nature of these instruments.
Revenue Recognition and Accounts
Receivable: The Company recognizes revenue at
the time services are performed. Unbilled receivables are
recorded for services rendered during, but billed subsequent to,
the reporting period. Revenue is reported at the estimated
realizable amounts from patients, third-party payors and others
for services rendered. Revenue under certain third-party payor
agreements is subject to audit and retroactive adjustments.
Provisions for estimated third-party payor settlements and
adjustments are estimated in the period the related services are
rendered and adjusted in future periods as final settlements are
determined. The provision for doubtful accounts and the related
allowance are adjusted periodically, based upon an evaluation of
historical collection experience with specific payors for
particular services, anticipated collection levels with specific
payors for new services, industry reimbursement trends, and
other relevant factors. Changes in these factors in future
periods could result in increases or decreases in the
Company’s provision for doubtful accounts and impact its
results of operations and financial position. For the period
ending December 10, 2007, Medicare and Cigna provided 15%
and 12% of the Company’s net revenues, respectively.
Cash: The Company considers deposits
and investments that have original maturities of less than three
months, when purchased, to be cash equivalents. Daily bank
deposits are held in noninterest bearing accounts with a
financial institution that are insured by the Federal Deposit
Insurance Corporation’s (“FDIC”). These amounts
are guaranteed by the FDIC up to $100,000 as of
December 10, 2007. The Company has not experienced any
losses on such accounts.
Property and Equipment: Property and
equipment is stated at cost. Routine maintenance and repairs are
charged to expense as incurred, while costs of betterments and
renewals are capitalized. Depreciation is calculated on a
straight-line basis, over the estimated useful lives of the
respective assets, which range from 5 to 8 years. Leasehold
improvements are amortized over the shorter of the term of the
related lease, or the useful life of the asset.
Income Taxes: Deferred taxes are
provided on the liability method whereby deferred tax assets are
recognized for deductive temporary differences, and deferred tax
liabilities are recognized for taxable temporary differences.
Temporary differences are the differences between the reported
amounts of assets and liabilities and their tax bases. Deferred
tax assets are reduced by a valuation allowance when, in the
opinion of management, it is more likely than not that some
portion of all of the deferred tax assets will be realized.
Deferred tax assets and liabilities are adjusted for the effects
of changes in taxes laws and rates on the date of enactment.
Concentration of Credit Risk: Financial
instruments that potentially subject the Company to
concentrations of credit risk are cash and accounts receivable.
Concentration of credit risk with respect to accounts receivable
is mitigated by the diversity of the Company’s payors.
While the Company has receivables due from federal and state
F-67
Laboratory
Medicine Consultants, LTD.
Notes to
Financial Statements — (Continued)
governmental agencies, the Company does not believe such
receivables represent a credit risk since the related healthcare
programs are funded by federal and state governments and payment
is primarily dependent on submitting appropriate documentation.
Recent Pending Accounting
Pronouncements: In July 2006, the FASB issued
Interpretation No. 48, Accounting for Uncertainty in
Income Taxes — an interpretation of FASB Statement 109
(“FIN 48”). FIN 48 clarifies the
accounting for uncertainty in income taxes recognized in an
enterprise’s financial statements in accordance with
SFAS 109, Accounting for Income Taxes. FIN 48
prescribes a comprehensive model for recognizing, measuring,
presenting and disclosing in the financial statements tax
positions taken or expected to be taken on a tax return. If
there are changes in net assets as a result of application of
FIN 48, these will be accounted for as an adjustment to the
opening balance of retained earnings. Additional disclosures
about the amounts of such liabilities will also be required. In
October 2008, the FASB delayed the effective date of FIN 48
for certain nonpublic enterprises to annual financial statements
for fiscal years beginning after December 15, 2008. The
Company was not required to adopt FIN 48 for these
financial statements. The Company does not expect the adoption
of this statement will have a material impact on its financial
statements.
In September 2006, the Statement of Financial Accounting
Standards No. 157, Fair Value Measurements
(“SFAS 157”), applies to all assets and
liabilities being measured and reported on a fair value basis.
SFAS 157 requires new disclosure that establishes a
framework for measuring fair value in GAAP, and expands
disclosure about fair value measurements. In February 2008, the
FASB issued FASB Staff Position (“FSP”)
No. 157-2,
Effective Date of FASB Statement No. 157, which
defers the effective date of SFAS 157 for one year for
non-financial assets and non-financial liabilities, except for
items recognized or disclosed at fair value in the financial
statements on a recurring basis for fiscal years beginning after
November 15, 2008.
SFAS 157 establishes a hierarchy ranking the quality and
reliability of the information used to determine fair values.
The statement requires assets and liabilities carried at fair
value will be classified and disclosed in one of the three
following categories:
|
|
|
| |
•
|
Level 1: Quoted market prices in active
markets for identical assets or liabilities.
|
| |
| |
•
|
Level 2: Observable market based inputs
or unobservable inputs corroborated by market data.
|
| |
| |
•
|
Level 3: Unobservable inputs not
corroborated by market data.
|
The Company does not expect the adoption of this Statement will
have a material impact on its financial position or results of
operations.
|
|
|
Note 2.
|
Accounts
Receivable
|
Accounts receivable as of December 10, 2007 consists of the
following (in thousands):
| |
|
|
|
|
|
Accounts receivable and unbilled receivables
|
|
$
|
4,863
|
|
|
Less: allowance for doubtful accounts
|
|
|
(1,605
|
)
|
|
|
|
|
|
|
|
Accounts receivable, net
|
|
$
|
3,258
|
|
|
|
|
|
|
|
F-68
Laboratory
Medicine Consultants, LTD.
Notes to
Financial Statements — (Continued)
|
|
|
Note 3.
|
Property
and Equipment
|
Property and equipment as of December 10, 2007 consists of
the following (in thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
Estimated Useful
|
|
|
|
|
|
Life (Years)
|
|
|
|
Laboratory, office and data processing equipment
|
|
|
5-7
|
|
|
$
|
1,138
|
|
|
Leasehold improvements
|
|
|
8
|
|
|
|
224
|
|
|
Furniture and fixtures
|
|
|
5
|
|
|
|
169
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1,531
|
|
|
Less accumulated depreciation
|
|
|
|
|
|
|
(1,041
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
490
|
|
|
|
|
|
|
|
|
|
|
|
Depreciation expense was approximately $191,000 for the period
ended December 10, 2007.
|
|
|
Note 4.
|
Post
Employment Obligation
|
As of December 10, 2007, the Company has a salary
continuation program with its four retired former stockholders
and remaining active stockholders. Under the terms of the
Company’s Amended and Restated Stockholders Agreement, the
stockholders, upon retirement, death or other specified events,
are entitled to 25% of a full stockholder salary and
distribution, 1.75% of the Company’s adjusted gross annual
revenues, health insurance, and malpractice “tail”
insurance coverage for four years following retirement. The
equity stockholders are eligible for the benefit after
completing their sixth year as a stockholder and are fully
vested after the tenth year. The Company calculates the present
value of these future payments based on average stockholder
salary and distribution levels, longevity and a discount rate
(7.35% as of December 10, 2007). The Company accrued an
aggregate of $7 million related to this post employment
benefit as of December 10, 2007, of which $1.9 million
is payable within one year.
During the period ended December 10, 2007, the Company paid
in full and terminated a $700,000 line of credit with a bank and
a $207,872 commercial term loan. Total interest expense for the
period ended December 10, 2007 was approximately $24,000.
These debt agreements were secured by substantially all of the
Company’s assets.
|
|
|
Note 6.
|
Employee
Benefit Plan
|
The Company has a defined contribution plan covering
substantially all of its employees. Eligible participants may
elect to defer a percentage of their compensation not to exceed
the maximum amount of elective contribution determined by law.
The employer may elect to make a qualified matching contribution
for a plan year. The qualified matching contribution is the
lesser of a percentage determined by the Company of the
participant’s elective contributions for that plan year or
a percentage of the employee’s compensation for that plan
year. For the period ended December 10, 2007, the Company
did not make any contributions towards the defined contribution
plan.
|
|
|
Note 7.
|
Commitments
and Contingencies
|
During the ordinary course of business, the Company has become
and may in the future become subject to pending and threatened
legal actions and proceedings. The Company may have liability
with respect to its employees and its pathologists. Medical
malpractice claims are generally covered by insurance. While the
Company believes the outcome of any such pending legal actions
and proceedings, individually or in the aggregate, will not have
a material adverse effect on the Company’s financial
condition, results of operations or liquidity, if the
F-69
Laboratory
Medicine Consultants, LTD.
Notes to
Financial Statements — (Continued)
Company is ultimately found liable under any medical malpractice
claims, there can be no assurance the Company’s medical
malpractice insurance coverage will be adequate to cover any
such liability.
Healthcare Regulatory Environment: The
healthcare industry, in general, and the services the Company
provides, are subject to extensive federal and state laws and
regulations. Additionally, a portion of the Company’s
revenue is from payments by government-sponsored health
programs, including Medicare, and is subject to audit and
adjustments by applicable regulatory agencies. Failure to comply
with any of these laws or regulations, the results of increased
regulatory audits and adjustments, or change in the
interpretation of the coding of services or the amounts payable
for the Company’s services under these programs could have
a material adverse effect on the Company’s financial
position and results of operations.
Employment Agreements: The Company has
employment agreements with its physician employees, the terms of
which expire at various times. Such agreements provide for
minimum salary levels including various benefits, as well as for
discretionary bonus compensations. Under certain of the
agreements, in the event employment is terminated, the employee
is entitled to receive only the compensation accrued but unpaid
as of the date of termination. The Company paid bonuses of
approximately $61,000 to its employees for the period ended
December 10, 2007, under the agreements.
Self-Insured Health Benefits: During
2007, the Company provided health care benefits to the majority
of its employees through a partially self-insured plan. The
Company records its estimate of the ultimate cost of, and
reserves for, health care benefits based on computations using
the Company’s loss history as well as industry statistics.
In determining its reserves, the Company includes reserves for
estimated claims incurred but not reported. The amount reserved
for estimated claims was $69,583 as of December 10, 2007
which is included in accounts payable in the accompanying
balance sheet. The ultimate cost of health care benefits will
depend on actual costs incurred to settle the claims and may
differ from the amounts reserved by the Company for those claims.
Operating Leases: The Company leases
its laboratory and business office from an unrelated entity
under a 20 year lease that began in October 2000 and ends
on May 31, 2020. Rent expense including taxes and common
utility charges was approximately $220,000 for the period ended
December 10, 2007.
Aggregate future minimum annual rentals under the lease
agreements as of December 10, 2007 are as follows (in
thousands):
| |
|
|
|
|
Year Ended
|
|
|
|
|
December 10,
|
|
|
|
|
2008
|
|
$
|
202
|
|
|
2009
|
|
|
202
|
|
|
2010
|
|
|
202
|
|
|
2011
|
|
|
202
|
|
|
2012
|
|
|
202
|
|
|
Thereafter
|
|
|
1,617
|
|
|
|
|
|
|
|
|
|
|
$
|
2,627
|
|
|
|
|
|
|
|
F-70
Laboratory
Medicine Consultants, LTD.
Notes to
Financial Statements — (Continued)
The provision for income taxes for the period ended
December 10, 2007 consists of the following:
| |
|
|
|
|
|
Current:
|
|
|
|
|
|
Federal
|
|
$
|
51
|
|
|
State
|
|
|
—
|
|
|
|
|
|
|
|
|
Total current provision
|
|
|
51
|
|
|
|
|
|
|
|
|
Deferred:
|
|
|
|
|
|
Federal
|
|
|
599
|
|
|
State
|
|
|
—
|
|
|
|
|
|
|
|
|
Total deferred provision
|
|
|
599
|
|
|
|
|
|
|
|
|
Total provision for income taxes
|
|
$
|
650
|
|
|
|
|
|
|
|
Temporary differences between the financial statement carrying
amounts and tax basis of assets and liabilities that give rise
to significant portions of the deferred tax assets and
liabilities are summarized below as of December 10, 2007:
| |
|
|
|
|
|
Allowance for doubtful accounts
|
|
$
|
(1,108
|
)
|
|
Accrued wages
|
|
|
75
|
|
|
Accounts payable
|
|
|
82
|
|
|
|
|
|
|
|
|
Current deferred tax liabilities, net
|
|
|
(951
|
)
|
|
|
|
|
|
|
|
Property and equipment
|
|
|
(84
|
)
|
|
Post-employment obligation
|
|
|
2,392
|
|
|
|
|
|
|
|
|
Noncurrent deferred tax assets, net
|
|
|
2,308
|
|
|
|
|
|
|
|
|
Total deferred tax assets, net
|
|
$
|
1,357
|
|
|
|
|
|
|
|
|
|
|
Note 9.
|
Subsequent
Events
|
On December 10, 2007, the Company was acquired from its
stockholders by Aurora Diagnostics Holdings, LLC. In conjunction
with the sale to Aurora Diagnostics Holdings, LLC, the portion
of the Company’s post employment benefit program for active
(non-retired) stockholders, and its related obligation of
$3.3 million, was terminated.
Subsequent to December 10, 2007, the Company was notified
of claims made by certain retired stockholders relating to the
Company’s post-employment benefit program and the sale of
the Company to Aurora Diagnostics Holdings, LLC. The Company
intends to vigorously defend its position. The ultimate outcome
of this litigation cannot presently be determined. Accordingly,
adjustments, if any, that might result from the resolution of
this matter have not been reflected in the accompanying
financial statements.
F-71
Independent
Auditor’s Report
To the Members
Aurora Diagnostics Holdings, LLC
Palm Beach Gardens, Florida
We have audited the accompanying consolidated balance sheet of
Greensboro Pathology Associates, P.A. and Subsidiary as of
October 4, 2007, and the related consolidated statement of
operations, stockholders’ equity and cash flows for the
period from January 1, 2007 to October 4, 2007. These
financial statements are the responsibility of the
Company’s management. Our responsibility is to express an
opinion on these financial statements based on our audit.
We conducted our audit in accordance with auditing standards
generally accepted in the United States of America. Those
standards require that we plan and perform the audit to obtain
reasonable assurance about whether the financial statements are
free of material misstatement. An audit includes examining, on a
test basis, evidence supporting the amounts and disclosures in
the financial statements. An audit also includes assessing the
accounting principles used and significant estimates made by
management, as well as evaluating the overall financial
statement presentation. We believe that our audit provides a
reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred
to above present fairly, in all material respects, the financial
position of Greensboro Pathology Associates, P.A. and Subsidiary
as of October 4, 2007, and the results of their operations
and their cash flows for the period from January 1, 2007 to
October 4, 2007 in conformity with accounting principles
generally accepted In the United States of America.
West Palm Beach, Florida
April 9, 2010
F-72
Greensboro
Pathology Associates, P.A. and Subsidiary
| |
|
|
|
|
|
|
|
($ in thousands)
|
|
Assets
|
|
Current Assets
|
|
|
|
|
|
Cash
|
|
$
|
2,386
|
|
|
Accounts receivable, net
|
|
|
4,069
|
|
|
Other assets
|
|
|
73
|
|
|
|
|
|
|
|
|
Total current assets
|
|
|
6,528
|
|
|
|
|
|
|
|
|
Property and Equipment, net
|
|
|
2,163
|
|
|
|
|
|
|
|
|
|
|
$
|
8,691
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Liabilities and Stockholders’ Equity
|
|
|
|
|
|
Current Liabilities
|
|
|
|
|
|
Note payable
|
|
$
|
2,566
|
|
|
Accounts payable
|
|
|
460
|
|
|
Accrued compensation
|
|
|
1,913
|
|
|
|
|
|
|
|
|
Total current liabilities
|
|
|
4,939
|
|
|
|
|
|
|
|
|
Commitments and Contingencies
|
|
|
|
|
|
Stockholders’ Equity
|
|
|
|
|
|
Common Shares, $10.00 par value per share,
10,000 shares authorized; 80 shares issued and
outstanding
|
|
|
1
|
|
|
Additional paid-in capital
|
|
|
7
|
|
|
Retained earnings
|
|
|
3,744
|
|
|
|
|
|
|
|
|
Total stockholders’ equity
|
|
|
3,752
|
|
|
|
|
|
|
|
|
|
|
$
|
8,691
|
|
|
|
|
|
|
|
See Notes to Consolidated Financial Statements.
F-73
Greensboro
Pathology Associates, P.A. and Subsidiary
For the
Period From January 1, 2007 through October 4,
2007
| |
|
|
|
|
|
|
|
($ in thousands)
|
|
Net Revenues
|
|
$
|
25,968
|
|
|
|
|
|
|
|
|
Operating costs and expenses:
|
|
|
|
|
|
Cost of service
|
|
|
19,478
|
|
|
Selling, general and administrative expenses
|
|
|
2,185
|
|
|
Depreciation expense
|
|
|
422
|
|
|
|
|
|
|
|
|
Total operating costs and expenses
|
|
|
22,085
|
|
|
|
|
|
|
|
|
Income from operations
|
|
|
3,883
|
|
|
Interest expense
|
|
|
(139
|
)
|
|
|
|
|
|
|
|
Net income
|
|
$
|
3,744
|
|
|
|
|
|
|
|
See Notes to Consolidated Financial Statements.
F-74
Greensboro
Pathology Associates, P.A. and Subsidiary
For the
Period From January 1, 2007 through October 4,
2007
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Additional
|
|
|
|
Total
|
|
|
|
Common Stock Outstanding
|
|
Paid-In
|
|
Retained
|
|
Stockholders’
|
|
|
|
Shares
|
|
Par Value
|
|
Capital
|
|
Earnings
|
|
Equity
|
|
|
|
($ in thousands, except shares)
|
|
|
|
Balance, December 31, 2006
|
|
|
80
|
|
|
$
|
1
|
|
|
$
|
7
|
|
|
$
|
3,240
|
|
|
$
|
3,248
|
|
|
Distributions
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(3,240
|
)
|
|
|
(3,240
|
)
|
|
Net income
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
3,744
|
|
|
|
3,744
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, October 4, 2007
|
|
|
80
|
|
|
$
|
1
|
|
|
$
|
7
|
|
|
$
|
3,744
|
|
|
$
|
3,752
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Consolidated Financial Statements.
F-75
Greensboro
Pathology Associates, P.A. and Subsidiary
For the
period from January 1, 2007 through October 4,
2007
| |
|
|
|
|
|
|
|
($ in thousands)
|
|
Cash Flows From Operating Activities
|
|
|
|
|
|
Net income
|
|
$
|
3,744
|
|
|
Adjustments to reconcile net income to net cash provided by
operating activities:
|
|
|
|
|
|
Depreciation
|
|
|
422
|
|
|
Allowance for bad debt
|
|
|
1,213
|
|
|
Changes in assets and liabilities:
|
|
|
|
|
|
Increase (decrease) in:
|
|
|
|
|
|
Accounts receivable
|
|
|
(893
|
)
|
|
Other assets
|
|
|
23
|
|
|
Increase (decrease) in:
|
|
|
|
|
|
Accounts payable
|
|
|
(283
|
)
|
|
Accrued compensation
|
|
|
1,141
|
|
|
|
|
|
|
|
|
Net cash provided by operating activities
|
|
|
5,367
|
|
|
|
|
|
|
|
|
Cash Flows From Investing Activities
|
|
|
|
|
|
Purchase of property and equipment
|
|
|
(188
|
)
|
|
|
|
|
|
|
|
Net cash used in investing activities
|
|
|
(188
|
)
|
|
|
|
|
|
|
|
Cash Flows From Financing Activities
|
|
|
|
|
|
Repayment of debt
|
|
|
(367
|
)
|
|
Distribution
|
|
|
(3,240
|
)
|
|
|
|
|
|
|
|
Net cash used in financing activities
|
|
|
(3,607
|
)
|
|
|
|
|
|
|
|
Net increase in cash
|
|
|
1,572
|
|
|
Cash:
|
|
|
|
|
|
Beginning
|
|
|
814
|
|
|
|
|
|
|
|
|
Ending
|
|
$
|
2,386
|
|
|
|
|
|
|
|
|
Supplemental Disclosures of Cash Flow Information
|
|
|
|
|
|
Cash interest payments
|
|
$
|
137
|
|
|
|
|
|
|
|
See Notes to Consolidated Financial Statements.
F-76
Greensboro
Pathology Associates, P.A. and Subsidiary
|
|
|
Note 1.
|
Nature of
Business and Significant Accounting Policies
|
Nature of Business: Greensboro
Pathology Associates, P.A. and Subsidiary (the
“Company”) was organized in the State of North
Carolina as a professional business corporation during February
1998 to operate as a pathology laboratory. During 2004, the
Company elected to be treated as an S-Corporation.
The Company engages in the business of providing anatomic
pathology and cytology services to physicians in the State of
North Carolina.
A summary of the Company’s significant accounting policies
follows:
Principles of Consolidation: The
accompanying consolidated financial statements of the Company
include the accounts of Greensboro Pathology Associates, P.A.
and its wholly-owned subsidiary. All accounts and transactions
between the entities have been eliminated in consolidation.
Accounting Estimates: The preparation
of financial statements in conformity with accounting principles
generally accepted in the United States of America requires
management to make estimates and assumptions that affect the
reported amounts of assets and liabilities, disclosure of
contingent assets and liabilities and the reported amounts of
revenues and expenses. Due to the inherent uncertainties in this
process, actual results could differ from those estimates.
Fair Value of Financial
Instruments: The Company’s financial
instruments consist mainly of cash, accounts receivable,
accounts payable and note payable. The carrying amounts of the
Company’s cash, accounts receivable and accounts payable
approximate fair value due to the short-term nature of these
instruments.
At October 4, 2007, approximately $2.6 million of the
Company’s note payable bears interest at a variable market
rate, and thus, the carrying amounts approximate fair value. As
of October 4, 2007, $2.1 million of the note payable
outstanding was subjected to an interest rate swap agreement
(derivative) which is reflected at fair value in the financial
statements of approximately $19,000 included in other assets on
the accompanying consolidated balance sheet as of
October 4, 2007. The interest rate swap agreement was
terminated subsequent to October 4, 2007, in conjunction
with the repayment of the note payable as further discussed in
Notes 4 and 8.
Revenue Recognition and Accounts
Receivable: The Company recognizes revenue at
the time services are performed. Unbilled receivables are
recorded for services rendered during, but billed subsequent to,
the reporting period. Revenue is reported at the estimated
realizable amounts from patients, third-party payors and others
for services rendered. Revenue under certain third-party payor
agreements is subject to audit and retroactive adjustments.
Provisions for estimated third-party payor settlements and
adjustments are estimated in the period the related services are
rendered and adjusted in future periods as final settlements are
determined. The provision for doubtful accounts and the related
allowance are adjusted periodically, based upon an evaluation of
historical collection experience with specific payors for
particular services, anticipated collection levels with specific
payors for new services, industry reimbursement trends, and
other relevant factors. Changes in these factors in future
periods could result in increases or decreases in the
Company’s provision for doubtful accounts and impact its
results of operations and financial position. For the period
ending October 4, 2007, Medicare and BlueCross/Blue Shield
provided 19% and 28% of the Company’s net revenues,
respectively.
Cash: The Company considers deposits
and investments that have original maturities of less than three
months, when purchased, to be cash equivalents. Daily bank
deposits are held in noninterest bearing accounts with a
financial institution that are insured by the Federal Deposit
Insurance Corporation’s (“FDIC”). These amounts
are guaranteed by the FDIC up to $100,000 as of October 4,
2007. The Company has not experienced any losses on such
accounts.
Property and Equipment: Property and
equipment is stated at cost. Routine maintenance and repairs are
charged to expense as incurred, while costs of betterments and
renewals are capitalized. Depreciation is calculated
F-77
Greensboro
Pathology Associates, P.A. and Subsidiary
Notes to
Consolidated Financial
Statements — (Continued)
on a straight-line basis, over the estimated useful lives of the
respective assets, which range from 3 to 11 years.
Leasehold improvements are amortized over the shorter of the
term of the related lease, or the useful life of the asset.
Income Taxes: The Company is a
subchapter S Corporation for federal and state income tax
purposes, in accordance with the applicable provisions of the
Internal Revenue Code. Accordingly, the Company is generally not
subject to income taxes. The income attributable to the Company
is distributed to the stockholders in accordance with their
proportionate share of earnings.
Derivative Financial Instruments: The
Company utilizes FASB Statement No. 133, Accounting for
Derivative Instruments and Hedging Activities. This
Statement requires that an entity recognize all derivatives as
either assets or liabilities in the balance sheet and measure
those instruments at fair value. During 2007, the Company
entered into an interest rate swap agreement with the bank to
eliminate the impact of changes in interest rates on its debt.
The Company has recorded the fair value of the swap agreement in
other assets on the accompanying consolidated balance sheet as
of October 4, 2007, and as additional financing expense
related to the changes in the carrying amount of the swap
agreement in the accompanying consolidated statement of income
for the period from January 1, 2007 through October 4,
2007.
Provided that the Company does not default on its financing
arrangements, continues its schedule of debt service payments
through the loan maturity dates and does not settle the interest
rate swap agreement, there will be no future cash outlay or
recovery for the fair value of the swap agreement.
Concentration of Credit Risk: Financial
instruments that potentially subject the Company to
concentrations of credit risk are cash and accounts receivable.
Concentration of credit risk with respect to accounts receivable
is mitigated by the diversity of the Company’s payors.
While the Company has receivables due from federal and state
governmental agencies, the Company does not believe such
receivables represent a credit risk since the related healthcare
programs are funded by federal and state governments and payment
is primarily dependent on submitting appropriate documentation.
Recent Accounting Pronouncements: In
July 2006, the FASB issued Interpretation No. 48, Accounting
for Uncertainty in Income Taxes — an interpretation of
FASB Statement 109 (“FIN 48”). FIN 48
clarifies the accounting for uncertainty in income taxes
recognized in an enterprise’s financial statements in
accordance with SFAS 109, Accounting for Income Taxes.
FIN 48 prescribes a comprehensive model for recognizing,
measuring, presenting and disclosing in the financial statements
tax positions taken or expected to be taken on a tax return. If
there are changes in net assets as a result of application of
FIN 48, these will be accounted for as an adjustment to the
opening balance of retained earnings. Additional disclosures
about the amounts of such liabilities will also be required. In
October 2008, the FASB delayed the effective date of FIN 48
for certain nonpublic enterprises to annual financial statements
for fiscal years beginning after December 15, 2008. The
Company was not required to adopt FIN 48 for these
consolidated financial statements. The Company does not expect
the adoption of this statement will impact its financial
statements.
In September 2006, Statement of Financial Accounting Standards
No. 157, Fair Value Measurements
(“SFAS 157”) applies to all assets and
liabilities being measured and reported on a fair value basis.
SFAS 157 requires new disclosure that establishes a
framework for measuring fair value in GAAP, and expands
disclosure about fair value measurements. In February 2008, the
FASB issued FASB Staff Position (“FSP”)
No. 157-2,
Effective Date of FASB Statement No. 157, which defers the
effective date of SFAS 157 for one year for non-financial
assets and non-financial liabilities, except for items
recognized or disclosed at fair value in the financial
statements on a recurring basis for fiscal years beginning after
November 15, 2008.
F-78
Greensboro
Pathology Associates, P.A. and Subsidiary
Notes to
Consolidated Financial
Statements — (Continued)
SFAS 157 establishes a hierarchy ranking the quality and
reliability of the information used to determine fair values.
The statement requires assets and liabilities carried at fair
value will be classified and disclosed in one of the three
following categories:
|
|
|
| |
•
|
Level 1: Quoted market prices in active
markets for identical assets or liabilities.
|
| |
| |
•
|
Level 2: Observable market based inputs
or unobservable inputs corroborated by market data.
|
| |
| |
•
|
Level 3: Unobservable inputs not
corroborated by market data.
|
The Company does not expect the adoption of this Statement will
have a material impact on its financial position or results of
operations.
|
|
|
Note 2.
|
Accounts
Receivable
|
Accounts receivable consists of the following as of
October 4, 2007 (in thousands):
| |
|
|
|
|
|
Accounts receivable and unbilled receivables
|
|
$
|
7,932
|
|
|
Less: allowance for bad debt
|
|
|
(3,863
|
)
|
|
|
|
|
|
|
|
Accounts receivable, net
|
|
$
|
4,069
|
|
|
|
|
|
|
|
|
|
|
Note 3.
|
Property
and Equipment
|
Property and equipment as of October 4, 2007 consists of
the following (in thousands):
| |
|
|
|
|
|
|
|
|
|
Estimated Useful
|
|
|
|
|
|
Life (Years)
|
|
|
|
|
|
Leasehold improvements
|
|
11
|
|
$
|
1,646
|
|
|
Furniture and fixtures
|
|
7
|
|
|
3,934
|
|
|
Laboratory, office and data processing equipment
|
|
3 – 5
|
|
|
1,217
|
|
|
Software
|
|
3
|
|
|
320
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
7,117
|
|
|
Less accumulated depreciation
|
|
|
|
|
(4,954
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
2,163
|
|
|
|
|
|
|
|
|
|
Depreciation expense was approximately $422,000 for the period
from January 1, 2007 to October 4, 2007.
In 2002, the Company entered into a note payable with a
financial institution. The note was secured by all tangible
assets of the Company and accrued interest at LIBOR plus 2%. In
July 2004, the Company entered into an interest rate swap
agreement with a fixed effective rate of 6.42%. Principal and
fixed rate interest were paid monthly. The note was paid in full
with proceeds from the sale of the Company to Aurora Diagnostics
Holdings, LLC as further discussed in Note 8.
|
|
|
Note 5.
|
Related
Party Transactions
|
The Company has employment agreements with certain of its
stockholders’ who are physician employees which expire at
various times. Bonuses are discretionary and may be paid to make
total annual compensation equal to the reasonable value of the
physicians’ contribution. Under certain of the agreements,
in the event employment is terminated due to death, retirement
or disability, the employee will receive an additional
compensation for up to one
F-79
Greensboro
Pathology Associates, P.A. and Subsidiary
Notes to
Consolidated Financial
Statements — (Continued)
year. For the period from January 1, 2007 through
October 4, 2007, the Company paid salaries and bonuses of
approximately $2.4 million and $6.2 million,
respectively.
|
|
|
Note 6.
|
Commitments
and Contingencies
|
During the ordinary course of business, the Company may become
subject to legal actions and proceedings. The Company may have
liability with respect to its employees and its pathologists.
Medical malpractice claims are generally covered by insurance.
While the Company believes the outcome of any such legal actions
and proceedings, individually or in the aggregate, will not have
a material adverse effect on the Company’s financial
condition, results of operations or liquidity, if the Company is
ultimately found liable under any medical malpractice claims,
there can be no assurance the Company’s medical malpractice
insurance coverage will be adequate to cover any such liability.
Healthcare Regulatory Environment: The
healthcare industry, in general, and the services the Company
provides, are subject to extensive federal and state laws and
regulations. Additionally, a portion of the Company’s
revenue is from payments by government-sponsored health
programs, including Medicare, and is subject to audit and
adjustments by applicable regulatory agencies. Failure to comply
with any of these laws or regulations, the results of increased
regulatory audits and adjustments, or change in the
interpretation of the coding of services or the amounts payable
for the Company’s services under these programs could have
a material adverse effect on the Company’s financial
position and results of operations.
Operating Leases: The Company leases an
office and medical laboratory facility under a non-cancelable
lease agreement maturing December 31, 2012. The terms of
the facility lease calls for the Company to pay for certain
taxes or common utility charges. Rent expense including these
taxes and common utility charges was approximately $454,000 for
the period ended October 4, 2007. Rent expense associated
with operating leases that include scheduled rent increases and
tenant incentives is recorded on a straight-line basis over the
term of the lease.
Aggregate future minimum annual rentals under the lease
agreements as of October 4, 2007 are as follows (in
thousands):
| |
|
|
|
|
Year Ending
|
|
|
|
|
October 4,
|
|
|
|
|
|
|
2008
|
|
$
|
630
|
|
|
2009
|
|
|
639
|
|
|
2010
|
|
|
654
|
|
|
2011
|
|
|
674
|
|
|
2012
|
|
|
869
|
|
|
|
|
|
|
|
|
|
|
$
|
3,466
|
|
|
|
|
|
|
|
Employment Agreements: The Company has
employment agreements with its physician employees, the terms of
which expire at various times. Such agreements provide for
minimum salary levels determined by the board of directors.
Bonuses are discretionary and may be paid to make total annual
compensation equal to the reasonable value of the
physicians’ contribution. Under certain of the agreements,
in the event employment is terminated due to death, retirement
or disability, the employee will receive an additional
compensation for up to one year.
|
|
|
Note 7.
|
Employee
Benefit Plan
|
The Company has a profit sharing 401(K) Plan covering
substantially all of its employees. Eligible participants may
elect to defer a percentage of their compensation not to exceed
the maximum amount of elective contribution determined by law.
The employer may elect to make a qualified matching contribution
for a plan year. The
F-80
Greensboro
Pathology Associates, P.A. and Subsidiary
Notes to
Consolidated Financial
Statements — (Continued)
Company guarantees 3% of compensation to each employee under the
401(K) Plan. The profit sharing contribution is discretionary by
the Company. For the period from January 1, 2007 to
October 4, 2007, the Company contributed approximately
$651,000 towards the profit sharing 401 (K).
On October 4, 2007, the Company was acquired from its
stockholders’ by Aurora Diagnostics Holdings, LLC.
F-81
Independent
Auditor’s Report
To the Members
Aurora Diagnostics Holdings, LLC
Palm Beach Gardens, Florida
We have audited the accompanying consolidated balance sheet of
Mark & Kambour, M.D., P.A. and Subsidiary as of
October 11, 2007, and the related consolidated statement of
operations, stockholders’ equity and cash flows for the
period from January 1, 2007 to October 11, 2007. These
financial statements are the responsibility of the
Company’s management. Our responsibility is to express an
opinion on these financial statements based on our audit.
We conducted our audit in accordance with auditing standards
generally accepted in the United States of America. Those
standards require that we plan and perform the audit to obtain
reasonable assurance about whether the financial statements are
free of material misstatement. An audit includes examining, on a
test basis, evidence supporting the amounts and disclosures in
the financial statements. An audit also includes assessing the
accounting principles used and significant estimates made by
management, as well as evaluating the overall financial
statement presentation. We believe that our audit provides a
reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred
to above present fairly, in all material respects, the financial
position of Mark & Kambour, M. D., P.A. and Subsidiary
as of October 11, 2007, and the results of their operations
and their cash flows for the period from January 1, 2007 to
October 11, 2007 in conformity with accounting principles
generally accepted in the United States of America.
West Palm Beach, Florida
April 9, 2010
F-82
Mark &
Kambour, M.D., P.A. and Subsidiary
Consolidated
Balance Sheet
October 11, 2007
| |
|
|
|
|
|
|
|
($ in thousands)
|
|
|
|
Assets
|
|
Current Assets
|
|
|
|
|
|
Cash
|
|
$
|
992
|
|
|
Accounts receivable, net
|
|
|
1,754
|
|
|
Other assets
|
|
|
72
|
|
|
Total current assets
|
|
|
2,818
|
|
|
Deposits
|
|
|
23
|
|
|
Property and Equipment, net
|
|
|
321
|
|
|
|
|
|
|
|
|
|
|
$
|
3,162
|
|
|
|
|
|
|
|
|
Liabilities and Stockholders’ Equity
|
|
Current Liabilities
|
|
|
|
|
|
Notes payable
|
|
$
|
—
|
|
|
Accounts payable
|
|
|
214
|
|
|
Accrued compensation
|
|
|
204
|
|
|
|
|
|
|
|
|
Total current liabilities
|
|
|
418
|
|
|
|
|
|
|
|
|
Commitments and Contingencies
|
|
|
|
|
|
Stockholders’ Equity
|
|
|
|
|
|
Common shares, $1.00 par value per share, 1,000 shares
authorized; 1,000 shares issued and outstanding
|
|
|
1
|
|
|
Additional paid-in capital
|
|
|
14
|
|
|
Retained earnings
|
|
|
2,729
|
|
|
Total stockholders’ equity
|
|
|
2,744
|
|
|
|
|
|
|
|
|
|
|
$
|
3,162
|
|
|
|
|
|
|
|
See Notes to Consolidated Financial Statements.
F-83
Mark &
Kambour, M.D., P.A. and Subsidiary
Consolidated
Statement of Operations
For the Period From January 1, 2007 through
October 11, 2007
| |
|
|
|
|
|
|
|
($ in thousands)
|
|
|
|
Net Revenues
|
|
$
|
8,284
|
|
|
Operating costs and expenses:
|
|
|
|
|
|
Cost of services
|
|
|
4,596
|
|
|
Selling, general and administrative expenses
|
|
|
523
|
|
|
Depreciation expense
|
|
|
76
|
|
|
|
|
|
|
|
|
Total operating costs and expenses
|
|
|
5,195
|
|
|
|
|
|
|
|
|
Income from operations
|
|
|
3,089
|
|
|
Interest expense
|
|
|
10
|
|
|
|
|
|
|
|
|
Net income
|
|
$
|
3,079
|
|
|
|
|
|
|
|
See Notes to Consolidated Financial Statements.
F-84
Mark &
Kambour, M.D., P.A. and Subsidiary
Consolidated
Statement of Stockholders’ Equity
For the Period From January 1, 2007 through
October 11, 2007
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Common Stock
|
|
|
|
|
|
|
|
|
|
Outstanding
|
|
Additional
|
|
|
|
Total
|
|
|
|
|
|
Par
|
|
Paid-In
|
|
Retained
|
|
Shareholders’
|
|
|
|
Shares
|
|
Value
|
|
Capital
|
|
Earnings
|
|
Equity
|
|
|
|
($ in thousands, except shares)
|
|
|
|
Balance, December 31, 2006
|
|
|
1,000
|
|
|
$
|
1
|
|
|
$
|
14
|
|
|
$
|
1,074
|
|
|
$
|
1,089
|
|
|
Distributions
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(1,424
|
)
|
|
|
(1,424
|
)
|
|
Net income
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
3,079
|
|
|
|
3,079
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, October 11, 2007
|
|
|
1,000
|
|
|
$
|
1
|
|
|
$
|
14
|
|
|
$
|
2,729
|
|
|
$
|
2,744
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Consolidated Financial Statements.
F-85
Mark &
Kambour, M.D., P.A. and Subsidiary
Consolidated
Statement of Cash Flows
For the period from January 1, 2007
through October 11, 2007
| |
|
|
|
|
|
|
|
($ in thousands)
|
|
|
|
Cash Flows From Operating Activities
|
|
|
|
|
|
Net income
|
|
$
|
3,079
|
|
|
Adjustments to reconcile net income to net cash provided by
operating activities:
|
|
|
|
|
|
Depreciation
|
|
|
76
|
|
|
Allowance for bad debt
|
|
|
152
|
|
|
Changes in assets and liabilities:
|
|
|
|
|
|
Increase in:
|
|
|
|
|
|
Accounts receivable
|
|
|
(843
|
)
|
|
Other assets
|
|
|
(34
|
)
|
|
Deposits
|
|
|
(20
|
)
|
|
Increase in:
|
|
|
|
|
|
Accounts payable
|
|
|
120
|
|
|
Accrued compensation
|
|
|
127
|
|
|
Net cash provided by operating activities
|
|
|
2,657
|
|
|
Cash Flows From Investing Activities
|
|
|
|
|
|
Purchase of property and equipment
|
|
|
(67
|
)
|
|
Net cash used in investing activities
|
|
|
(67
|
)
|
|
Cash Flows From Financing Activities
|
|
|
|
|
|
Repayment of note payable
|
|
|
(224
|
)
|
|
Distributions
|
|
|
(1,424
|
)
|
|
Net cash used in financing activities
|
|
|
(1,648
|
)
|
|
Net increase in cash
|
|
|
942
|
|
|
Cash:
|
|
|
|
|
|
Beginning
|
|
|
50
|
|
|
Ending
|
|
$
|
992
|
|
|
|
|
|
|
|
|
Supplemental Disclosures of Cash Flow Information
|
|
|
|
|
|
Cash interest payments
|
|
$
|
10
|
|
|
|
|
|
|
|
See Notes to Consolidated Financial Statements.
F-86
Mark &
Kambour, M.D., P.A. and Subsidiary
|
|
|
Note 1.
|
Nature of
Business and Significant Accounting Policies
|
Nature of Business: Mark &
Kambour, M.D., P.A. and Subsidiary (the
“Company”) was organized in the State of Florida as a
sub-chapter S corporation
during May 1989 to operate as a pathology laboratory company.
The Company engages in the business of providing anatomic
pathology services to physicians in the State of Florida.
A summary of the Company’s significant accounting policies
follows:
Principles of Consolidation: The
accompanying consolidated financial statements of the Company
include the accounts of Mark & Kambour M.D., P.A., and
its wholly-owned subsidiary, MA Courier Services, Inc. All
accounts and transactions between the entities have been
eliminated in consolidation.
Accounting Estimates: The preparation
of financial statements in conformity with accounting principles
generally accepted in the United States of America requires
management to make estimates and assumptions that affect the
reported amounts of assets and liabilities, disclosure of
contingent assets and liabilities and the reported amounts of
revenues and expenses. Due to the inherent uncertainties in this
process, actual results could differ from those estimates.
Fair Value of Financial
Instruments: The Company’s financial
instruments consist mainly of cash, accounts receivable and
accounts payable. The carrying amounts of the Company’s
cash, accounts receivable and accounts payable approximate fair
value due to the short-term nature of these instruments.
Revenue Recognition and Accounts
Receivable: The Company recognizes revenue at
the time services are performed. Unbilled receivables are
recorded for services rendered during, but billed subsequent to,
the reporting period. Revenue is reported at the estimated
realizable amounts from patients, third-party payors and others
for services rendered. Revenue under certain third-party payor
agreements is subject to audit and retroactive adjustments.
Provisions for estimated third-party payor settlements and
adjustments are estimated in the period the related services are
rendered and adjusted in future periods as final settlements are
determined. The provision for doubtful accounts and the related
allowance are adjusted periodically, based upon an evaluation of
historical collection experience with specific payors for
particular services, anticipated collection levels with specific
payors for new services, industry reimbursement trends, and
other relevant factors. Changes in these factors in future
periods could result in increases or decreases in the
Company’s provision for doubtful accounts and impact its
results of operations and financial position. For the period
ending October 11, 2007, Medicare and BlueCross/Blue Shield
provided 22% and 13% of the Company’s net revenues,
respectively.
Cash: The Company considers deposits
and investments that have original maturities of less than three
months, when purchased, to be cash equivalents. Daily bank
deposits are held in noninterest bearing accounts with a
financial institution that are insured by the Federal Deposit
Insurance Corporation’s (“FDIC”). These amounts
are guaranteed by the FDIC up to $100,000 as of October 11,
2007. The Company has not experienced any losses on such
accounts.
Property and Equipment: Property and
equipment is stated at cost. Routine maintenance and repairs are
charged to expense as incurred, while costs of betterments and
renewals are capitalized. Depreciation is calculated on a
straight-line basis, over the estimated useful lives of the
respective assets, which range from 3 to 7 years.
Income Taxes: The Company is a
subchapter S Corporation for federal and state income tax
purposes, in accordance with the applicable provisions of the
Internal Revenue Code. Accordingly, the Company is generally not
subject to income taxes. The income attributable to the Company
is distributed to the stockholders in accordance with their
proportionate share of earnings.
Concentration of Credit Risk: Financial
instruments that potentially subject the Company to
concentrations of credit risk are cash and accounts receivable.
Concentration of credit risk with respect to accounts receivable
is mitigated by the diversity of the Company’s payors.
While the Company has receivables due from federal and state
F-87
Mark &
Kambour, M.D., P.A. and Subsidiary
Notes to
Consolidated Financial
Statements — (Continued)
government agencies, the Company does not believe such
receivables represent a credit risk since the related healthcare
programs are funded by federal and state governments and payment
is primarily dependent on submitting appropriate documentation.
Recent Accounting Pronouncements: In
July 2006, the FASB issued Interpretation No. 48, Accounting
for Uncertainty in Income Taxes — an interpretation of
FASB Statement 109 (“FIN 48”). FIN 48
clarifies the accounting for uncertainty in income taxes
recognized in an enterprise’s financial statements in
accordance with SFAS 109, Accounting for Income Taxes.
FIN 48 prescribes a comprehensive model for recognizing,
measuring, presenting and disclosing in the financial statements
tax positions taken or expected to be taken on a tax return. If
there are changes in net assets as a result of application of
FIN 48, these will be accounted for as an adjustment to the
opening balance of retained earnings. Additional disclosures
about the amounts of such liabilities will also be required. In
October 2008, the FASB delayed the effective date of FIN 48
for certain nonpublic enterprises to annual financial statements
for fiscal years beginning after December 15, 2008. The
Company was not required to adopt FIN 48 for these
consolidated financial statements. The Company does not expect
the adoption of this statement will impact its financial
statements.
In September 2006, Statement of Financial Accounting Standards
No. 157, Fair Value Measurements
(“SFAS 157”) applies to all assets and
liabilities being measured and reported on a fair value basis.
SFAS 157 requires new disclosure that establishes a
framework for measuring fair value in GAAP, and expands
disclosure about fair value measurements. In February 2008, the
FASB issued FASB Staff Position (“FSP”)
No. 157-2,
Effective Date of FASB Statement No. 157, which defers the
effective date of SFAS 157 for one year for non-financial
assets and non-financial liabilities, except for items
recognized or disclosed at fair value in the financial
statements on a recurring basis for fiscal years beginning after
November 15, 2008.
SFAS 157 establishes a hierarchy ranking the quality and
reliability of the information used to determine fair values.
The statement requires assets and liabilities carried at fair
value will be classified and disclosed in one of the three
following categories:
|
|
|
| |
•
|
Level 1: Quoted market prices in active
markets for identical assets or liabilities.
|
| |
| |
•
|
Level 2: Observable market based inputs
or unobservable inputs corroborated by market data.
|
| |
| |
•
|
Level 3: Unobservable inputs not
corroborated by market data.
|
The Company does not expect the adoption of this Statement will
have a material impact on its financial position or results of
operations.
|
|
|
Note 2.
|
Accounts
Receivable
|
Accounts receivable as of October 11, 2007 consist of the
following (in thousands):
| |
|
|
|
|
|
Accounts receivable and unbilled receivables
|
|
$
|
2,139
|
|
|
Less: Allowance for bad debt
|
|
|
(386
|
)
|
|
|
|
|
|
|
|
Accounts receivable, net
|
|
$
|
1,754
|
|
|
|
|
|
|
|
F-88
Mark &
Kambour, M.D., P.A. and Subsidiary
Notes to
Consolidated Financial
Statements — (Continued)
|
|
|
Note 3.
|
Property
and Equipment
|
Property and equipment as of October 11, 2007 consist of
the following (in thousands):
| |
|
|
|
|
|
|
|
|
|
Estimated Useful
|
|
|
|
|
|
|
Life (Years)
|
|
|
|
|
|
|
Furniture and fixtures
|
|
7
|
|
$
|
12
|
|
|
Laboratory, office and data processing equipment
|
|
5 – 7
|
|
|
578
|
|
|
Software
|
|
3
|
|
|
78
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
669
|
|
|
Less accumulated depreciation
|
|
|
|
|
(348
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
321
|
|
|
|
|
|
|
|
|
|
Depreciation expense was approximately $76,000 for the period
from January 1, 2007 to October 11, 2007.
During the period ended October 11, 2007, the Company paid
in full a commercial term loan with outstanding balance of
$224,016. Interest expense for the period ended October 11,
2007 was approximately $9,700. The note payable was secured by
substantially all the Company’s assets.
|
|
|
Note 5.
|
Related
Party Transaction
|
The Company has employment agreements with certain of its
stockholders’ who are physician employees of which expire
at various times. Bonuses are discretionary and may be paid
based upon the reasonable value of the physicians’
contribution. For the period from January 1, 2007 through
October 11, 2007, the Company paid salaries and bonuses to
these physicians of approximately $475,000.
|
|
|
Note 6.
|
Commitments
and Contingencies
|
During the ordinary course of business, the Company may become
subject to legal actions and proceedings. The Company may have
liability with respect to its employees and its pathologists.
Medical malpractice claims are generally covered by insurance.
While the Company believes the outcome of any such legal actions
and proceedings, individually or in the aggregate, will not have
a material adverse effect on the Company’s financial
condition, results of operations or liquidity, if the Company is
ultimately found liable under any medical malpractice claims,
there can be no assurance the Company’s medical malpractice
insurance coverage will be adequate to cover any such liability.
Healthcare Regulatory Environment: The
healthcare industry, in general, and the services the Company
provides, are subject to extensive federal and state laws and
regulations. Additionally, a portion of the Company’s
revenue is from payments by government-sponsored health
programs, including Medicare, and is subject to audit and
adjustments by applicable regulatory agencies. Failure to comply
with any of these laws or regulations, the results of increased
regulatory audits and adjustments, or change in the
interpretation of the coding of services or the amounts payable
for the Company’s services under these programs could have
a material adverse effect on the Company’s financial
position and results of operations.
Employment Agreements: The Company has
employment agreements with its physician employees, the terms of
which expire at various times. Such agreements provide for
minimum salary levels determined by the board of directors.
Bonuses are discretionary and may be paid based upon the
reasonable value of the physicians’ contribution. Under
certain of the agreements, in the event employment is terminated
due to death, retirement or disability, the employee will
receive additional compensation for up to one year.
F-89
Mark &
Kambour, M.D., P.A. and Subsidiary
Notes to
Consolidated Financial
Statements — (Continued)
Operating Leases: As of
October 11, 2007, the Company leases an office and medical
laboratory facility under a
month-to-month
agreement until a new facility is completed. On May 29,
2007, the Company entered into a construction agreement with a
contractor for a new leased facility in Coral Gables, FL. Total
costs are expected to be approximately $425,000. Through
October 11, 2007, no payments have been made by the Company
under the agreement.
In May 2007, the Company executed a non-cancelable lease to
begin September 21, 2007, maturing September 30, 2012
for the new facility. The terms of the facility lease call for
the Company to pay for certain taxes or common utility charges.
Rent expense including these taxes and common utility charges
was approximately $48,000 for the period ended October 11,
2007.
Aggregate future minimum annual rentals under the lease
agreements as of October 11, 2007 are as follows (in
thousands):
| |
|
|
|
|
Year Ending
|
|
|
|
|
October 11,
|
|
|
|
|
|
|
2008
|
|
$
|
117
|
|
|
2009
|
|
|
121
|
|
|
2010
|
|
|
126
|
|
|
2011
|
|
|
127
|
|
|
2012
|
|
|
95
|
|
|
|
|
|
|
|
|
|
|
$
|
586
|
|
|
|
|
|
|
|
On October 11, 2007, the Company was acquired from its
stockholders’ by Aurora Diagnostics Holdings, LLC.
F-90
Independent
Auditor’s Report
To the Members
Aurora Diagnostics Holdings, LLC
Palm Beach Gardens, Florida
We have audited the accompanying balance sheet of Cunningham
Pathology, LLC as of April 30, 2007, and the related
statement of operations and members’ equity and cash flows
for the period from January 1, 2007 to April 30, 2007.
These financial statements are the responsibility of the
Company’s management. Our responsibility is to express an
opinion on these financial statements based on our audit.
We conducted our audit in accordance with auditing standards
generally accepted in the United States of America. Those
standards require that we plan and perform the audit to obtain
reasonable assurance about whether the financial statements are
free of material misstatement. An audit includes examining, on a
test basis, evidence supporting the amounts and disclosures in
the financial statements. An audit also includes assessing the
accounting principles used and significant estimates made by
management, as well as evaluating the overall financial
statement presentation. We believe that our audit provides a
reasonable basis for our opinion.
In our opinion, the financial statements referred to above
present fairly, in all material respects, the financial position
of Cunningham Pathology, LLC as of April 30, 2007, and the
results of its operations and its cash flows for the period from
January 1, 2007 through April 30, 2007 in conformity
with accounting principles generally accepted in the United
States of America.
West Palm Beach, Florida
April 9, 2010
F-91
| |
|
|
|
|
|
|
|
($ in thousands)
|
|
|
|
Assets
|
|
|
|
|
|
Current Assets
|
|
|
|
|
|
Cash
|
|
$
|
642
|
|
|
Accounts receivable, net
|
|
|
1,558
|
|
|
Prepaid expenses
|
|
|
93
|
|
|
|
|
|
|
|
|
Total current assets
|
|
|
2,293
|
|
|
Property and Equipment, net
|
|
|
2,422
|
|
|
Deposits
|
|
|
18
|
|
|
|
|
|
|
|
|
|
|
$
|
4,733
|
|
|
|
|
|
|
|
|
Liabilities and Members’ Equity
|
|
|
|
|
|
Current Liabilities
|
|
|
|
|
|
Accounts payable
|
|
$
|
252
|
|
|
Accrued compensation
|
|
|
272
|
|
|
Mortgage payable
|
|
|
1,157
|
|
|
Line of credit
|
|
|
—
|
|
|
|
|
|
|
|
|
Total current liabilities
|
|
|
1,681
|
|
|
Commitments and Contingencies
|
|
|
|
|
|
Members’ Equity
|
|
|
3,052
|
|
|
|
|
|
|
|
|
|
|
$
|
4,733
|
|
|
|
|
|
|
|
See Notes to Financial Statements.
F-92
| |
|
|
|
|
|
|
|
($ in thousands)
|
|
|
|
Net Revenues
|
|
$
|
7,520
|
|
|
Operating costs and expenses:
|
|
|
|
|
|
Cost of services
|
|
|
4,806
|
|
|
Selling, general and administrative expenses
|
|
|
584
|
|
|
Depreciation expense
|
|
|
176
|
|
|
|
|
|
|
|
|
Total operating costs and expenses
|
|
|
5,566
|
|
|
Income from operations
|
|
|
1,954
|
|
|
|
|
|
|
|
|
Other income (expense):
|
|
|
|
|
|
Interest expense
|
|
|
(20
|
)
|
|
Other income
|
|
|
5
|
|
|
Total other expense
|
|
|
(15
|
)
|
|
|
|
|
|
|
|
Net income
|
|
|
1,939
|
|
|
Members’ Equity, Beginning of Year
|
|
|
2,329
|
|
|
Distributions
|
|
|
(1,216
|
)
|
|
|
|
|
|
|
|
Members’ Equity, End of Year
|
|
$
|
3,052
|
|
|
|
|
|
|
|
See Notes to Financial Statements.
F-93
| |
|
|
|
|
|
|
|
($ in thousands)
|
|
|
|
Cash Flows From Operating Activities
|
|
|
|
|
|
Net income
|
|
$
|
1,939
|
|
|
Adjustments to reconcile net income to net cash provided by
operating activities:
|
|
|
|
|
|
Depreciation
|
|
|
176
|
|
|
Allowance for bad debt
|
|
|
13
|
|
|
Changes in assets and liabilities:
|
|
|
|
|
|
(Increase) decrease in:
|
|
|
|
|
|
Accounts receivable
|
|
|
(332
|
)
|
|
Prepaid expenses
|
|
|
247
|
|
|
Increase in:
|
|
|
|
|
|
Accounts payable
|
|
|
(61
|
)
|
|
Accrued compensation
|
|
|
129
|
|
|
Net cash provided by operating activities
|
|
|
2,111
|
|
|
Cash Flows From Investing Activities
|
|
|
|
|
|
Purchase of property and equipment
|
|
|
(45
|
)
|
|
|
|
|
|
|
|
Net cash used in investing activities
|
|
|
(45
|
)
|
|
|
|
|
|
|
|
Cash Flows From Financing Activities
|
|
|
|
|
|
Repayment of line of credit
|
|
|
(205
|
)
|
|
Principal payments on mortgage
|
|
|
(10
|
)
|
|
Distributions paid
|
|
|
(1,210
|
)
|
|
Net cash used in financing activities
|
|
|
(1,425
|
)
|
|
|
|
|
|
|
|
Net increase in cash
|
|
|
641
|
|
|
Cash:
|
|
|
|
|
|
Beginning
|
|
|
1
|
|
|
|
|
|
|
|
|
Ending
|
|
$
|
642
|
|
|
|
|
|
|
|
|
Supplemental Schedule of Noncash Investing and Financing
Activities
|
|
|
|
|
|
Member distribution of property
|
|
$
|
6
|
|
|
|
|
|
|
|
|
Interest paid
|
|
$
|
22
|
|
|
|
|
|
|
|
See Notes to Financial Statements.
F-94
Cunningham
Pathology, LLC
|
|
|
Note 1.
|
Nature of
Business and Significant Accounting Policies
|
Nature of Business: Cunningham
Pathology, LLC (the “Company”) was organized in the
State of Delaware as a limited liability company on
December 10, 1998 to operate as a pathology laboratory
company.
The Company engages in the business of providing anatomic
pathology and cytology services to physicians in the state of
Alabama.
A summary of the Company’s significant accounting policies
follows:
Variable Interest Entity: Accounting
principles generally accepted in the United States of America
require the consolidation of the financial statements of a
variable interest entity (“VIE”) with the financial
statements of its primary beneficiary. These consolidation
requirements apply to the Company and management of the Company
has elected to adopt the guidance. An entity is considered a VIE
when equity investors in the entity do not have the
characteristics of a controlling financial interest or when the
equity at risk is not sufficient for the entity to finance its
activities without additional subordinated financial support.
The primary beneficiary of a VIE is the entity that is subject
to a majority of the risk of loss or entitled to receive a
majority of the VIE’s residual returns, or both. In
determining whether it is the primary beneficiary of a VIE, a
company with a variable interest must also treat a variable
interest held by a related party the same as its own interest.
The Company has consolidated the financial statements of
Cunningham Investment Group, LLC, the entity from which the
Company leases certain of its facilities, in the accompanying
financial statements as required by this guidance based on the
Company being the primary beneficiary. The Company has included
$1,160,000 of assets and $1,157,173 of liabilities attributed to
Cunningham Investment Group, LLC in the consolidated financial
statements at April 30, 2007. The Company will reconsider
its determination of whether an entity is a VIE and who the
primary beneficiary is if certain events occur that are likely
to cause a change in the original determinations.
Accounting Estimates: The preparation
of financial statements in conformity with accounting principles
generally accepted in the United States of America requires
management to make estimates and assumptions that affect the
reported amounts of assets and liabilities, disclosure of
contingent assets and liabilities and the reported amounts of
revenues and expenses. Due to the inherent uncertainties in this
process, actual results could differ from those estimates.
Fair Value of Financial
Instruments: The Company’s financial
instruments consist mainly of cash, accounts receivable and
accounts payable. The carrying amounts of the Company’s
cash, accounts receivable and accounts payable approximate fair
value due to the short-term nature of these instruments.
Revenue Recognition and Accounts
Receivable: The Company recognizes revenue at
the time services are performed. Unbilled receivables are
recorded for services rendered during, but billed subsequent to,
the reporting period. Revenue is reported at the estimated
realizable amounts from patients, third-party payors and others
for services rendered. Revenue under certain third-party payor
agreements is subject to audit and retroactive adjustments.
Provisions for estimated third-party payor settlements and
adjustments are estimated in the period the related services are
rendered and adjusted in future periods as final settlements are
determined. The provision for doubtful accounts and the related
allowance are adjusted periodically, based upon an evaluation of
historical collection experience with specific payors for
particular services, anticipated collection levels with specific
payors for new services, industry reimbursement trends, and
other relevant factors. Changes in these factors in future
periods could result in increases or decreases in the
Company’s provision for doubtful accounts and impact its
results of operations and financial position. For the period
ending April 30, 2007, Medicare and BlueCross/Blue Shield
provided 19% and 57% of the Company’s net revenues,
respectively.
Cash: The Company considers deposits
and investments that have original maturities of less than three
months, when purchased, to be cash equivalents. Daily bank
deposits are held in non-interest bearing accounts with a
financial institution that are insured by the Federal Deposit
Insurance Corporation (“FDIC”). These amounts are
F-95
Cunningham
Pathology, LLC
Notes to
Financial Statements — (Continued)
guaranteed by the FDIC up to $100,000 as of April 30, 2007.
The Company has not experienced any losses on such accounts.
Property and Equipment: Property and
equipment is stated at cost. Routine maintenance and repairs are
charged to expense as incurred, while costs of betterments and
renewals are capitalized. Depreciation is calculated on a
straight-line basis, over the estimated useful lives of the
respective assets, which range from 3 to 7 years. Leasehold
improvements are amortized over the shorter of the term of the
related lease, or the useful life of the asset.
Income Taxes: The Company is a Delaware
limited liability company for federal and state income tax
purposes, in accordance with the applicable provisions of the
Internal Revenue Code. Accordingly, the Company is generally not
subject to income taxes, the income attributable to the limited
liability company is distributed to the members in accordance
with the terms of the operating agreement.
Concentration of Credit Risk: Financial
instruments that potentially subject the Company to
concentrations of credit risk are cash and accounts receivable.
Concentration of credit risk with respect to accounts receivable
is mitigated by the diversity of the Company’s payors.
While the Company has receivables due from federal and state
governmental agencies, the Company does not believe such
receivables represent a credit risk since the related healthcare
programs are funded by federal and state governments and payment
is primarily dependent on submitting appropriate documentation.
Recent Accounting Pronouncements: In
July 2006, the FASB issued Interpretation No. 48,
Accounting for Uncertainty in Income Taxes — an
interpretation of FASB Statement 109 (“FIN 48”).
FIN 48 clarifies the accounting for uncertainty in income
taxes recognized in an enterprise’s financial statements in
accordance with SFAS 109, Accounting for Income
Taxes. FIN 48 prescribes a comprehensive model for
recognizing, measuring, presenting and disclosing in the
financial statements tax positions taken or expected to be taken
on a tax return. If there are changes in net assets as a result
of application of FIN 48, these will be accounted for as an
adjustment to the opening balance of retained earnings.
Additional disclosures about the amounts of such liabilities
will also be required. In October 2008, the FASB delayed the
effective date of FIN 48 for certain nonpublic enterprises
to annual financial statements for fiscal years beginning after
December 15, 2008. The Company was not required to adopt
FIN 48 for these financial statements. The Company does not
expect the adoption of this statement will have a material
impact on its financial statements.
In September 2006, the Statement of Financial Accounting
Standards No. 157, Fair Value Measurements
(“SFAS 157”), applies to all assets and
liabilities being measured and reported on a fair value basis.
SFAS 157 requires new disclosure that establishes a
framework for measuring fair value in GAAP, and expands
disclosure about fair value measurements. In February 2008, the
FASB issued FASB Staff Position (“FSP”)
No. 157-2,
Effective Date of FASB Statement No. 157, which
defers the effective date of SFAS 157 for one year for
non-financial assets and non-financial liabilities, except for
items recognized or disclosed at fair value in the financial
statements on a recurring basis for fiscal years beginning after
November 15, 2008.
SFAS 157 establishes a hierarchy ranking the quality and
reliability of the information used to determine fair values.
The statement requires assets and liabilities carried at fair
value will be classified and disclosed in one of the three
following categories:
|
|
|
| |
•
|
Level 1: Quoted market prices in active
markets for identical assets or liabilities.
|
| |
| |
•
|
Level 2: Observable market based inputs
or unobservable inputs corroborated by market data.
|
| |
| |
•
|
Level 3: Unobservable inputs not
corroborated by market data.
|
The Company does not expect the adoption of this Statement will
have a material impact on its financial position or results of
operations.
F-96
Cunningham
Pathology, LLC
Notes to
Financial Statements — (Continued)
|
|
|
Note 2.
|
Accounts
Receivable
|
Accounts receivable as of April 30, 2007 consist of the
following (in thousands):
| |
|
|
|
|
|
Accounts receivable and unbilled receivables
|
|
$
|
3,938
|
|
|
Less: allowance for doubtful accounts
|
|
|
(2,380
|
)
|
|
|
|
|
|
|
|
Accounts receivable, net
|
|
$
|
1,558
|
|
|
|
|
|
|
|
|
|
|
Note 3.
|
Property
and Equipment
|
Property and equipment as of April 30, 2007 consists of the
following (in thousands):
| |
|
|
|
|
|
|
|
|
|
Estimated Useful
|
|
|
|
|
|
|
Life (Years)
|
|
|
|
|
|
|
Laboratory, office and data processing equipment
|
|
5–7
|
|
$
|
2,869
|
|
|
Building
|
|
39
|
|
$
|
1,191
|
|
|
Leasehold improvements
|
|
8
|
|
|
293
|
|
|
Furniture and fixtures
|
|
5
|
|
|
188
|
|
|
Software
|
|
3
|
|
|
230
|
|
|
Vehicles
|
|
5
|
|
|
194
|
|
|
|
|
|
|
|
4,965
|
|
|
Less accumulated depreciation
|
|
|
|
|
(3,018
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1,947
|
|
|
|
|
|
|
|
473
|
|
|
|
|
|
|
|
2,422
|
|
|
|
|
|
|
|
|
|
Depreciation expense was approximately $176,000 for the
four-month period ended April 30, 2007.
During June 2006, the Company entered into a $1 million
line of credit with a bank with interest at LIBOR plus 2%. On
March 15, 2007, the outstanding balance of approximately
$205,000 under the line of credit was paid in full and the line
was terminated.
On February 23, 1999, the Company entered into a mortgage
note for the purchase of its building and land. The agreement
was collateralized by substantially all of the Company’s
assets. Interest was paid in arrears at 6.875% and the
note’s original maturity date was February 23, 2014. The
Company will continue to make monthly payments at $11,800 with a
final balloon payment of $613,168 due on the maturity date.
|
|
|
Note 5.
|
Employee
Benefit Plan
|
The Company has a defined contribution plan covering
substantially all of its employees. Eligible participants may
elect to defer a percentage of their compensation not to exceed
the maximum amount of elective contribution determined by law.
The employer may elect to make a qualified matching contribution
for a plan year. The qualified matching contribution is the
lesser of a percentage determined by the Company of the
participant’s elective contributions for that plan year or
a percentage of the employee’s compensation for that plan
year. For the period ended April 30, 2007, the Company
contributed approximately $536,000 toward the defined
contribution plan.
F-97
Cunningham
Pathology, LLC
Notes to
Financial Statements — (Continued)
|
|
|
Note 6.
|
Related
Party Transactions
|
The Company leases office space from Cunningham Investment Group
LLC, which is owned by the physician employees. The lease
provides for monthly aggregate minimum payments of approximately
$18,000 and will expire in April 2009. Rent expense incurred and
paid to the related entity was approximately $72,000 for the
period ending April 30, 2007.
|
|
|
Note 7.
|
Commitments
and Contingencies
|
During the ordinary course of business, the Company may become
subject to legal actions and proceedings. The Company may have
liability with respect to its employees and its pathologists.
Medical malpractice claims are generally covered by insurance.
While the Company believes the outcome of any such legal actions
and proceedings, individually or in the aggregate, will not have
a material adverse effect on the Company’s financial
condition, results of operations or liquidity, if the Company is
ultimately found liable under any medical malpractice claims,
there can be no assurance the Company’s medical malpractice
insurance coverage will be adequate to cover any such liability.
Healthcare Regulatory Environment: The
healthcare industry, in general, and the services the Company
provides, are subject to extensive federal and state laws and
regulations. Additionally, a portion of the Company’s
revenue is from payments by government-sponsored health
programs, including Medicare, and is subject to audit and
adjustments by applicable regulatory agencies. Failure to comply
with any of these laws or regulations, the results of increased
regulatory audits and adjustments, or change in the
interpretation of the coding of services or the amounts payable
for the Company’s services under these programs could have
a material adverse effect on the Company’s financial
position and results of operations.
Employment Agreements: The Company has
employment agreements with its physician employees, the terms of
which expire at various times. Such agreements provide for
minimum salary levels including various benefits, as well as for
discretionary bonus compensations. Under certain of the
agreements, in the event employment is terminated (other than
voluntarily by the employee or the Company for cause or upon the
death of the employee), the Company is committed to pay a full
monthly salary for a period of three months. The Company paid
bonuses of approximately $275,000 to its physician employees for
the period ended April 30, 2007.
Operating Leases: The Company leases
various office and medical laboratory facilities and equipment
under non-cancelable lease agreements with terms ranging from
two to ten years and expiring through 2010. The terms of the
facility leases call for the Company to pay for certain taxes or
common utility charges. Rent expense including these taxes and
common utility charges was approximately $119,000 for the period
ended April 30, 2007. Rent expense associated with
operating leases that include scheduled rent increases and
tenant incentives is recorded on a straight-line basis over the
term of the lease.
Aggregate future minimum annual rentals under the lease
agreements as of April 30, 2007 are as follows (in
thousands):
| |
|
|
|
|
|
Year Ended
April 30,
|
|
|
|
|
2008
|
|
$
|
251
|
|
|
2009
|
|
|
242
|
|
|
2010
|
|
|
12
|
|
|
|
|
|
|
|
|
|
|
$
|
505
|
|
|
|
|
|
|
|
|
|
|
Note 8.
|
Subsequent
Events
|
On April 30, 2007, the Company was acquired from its
members by Aurora Diagnostics Holdings, LLC. Payments in excess
of the guaranteed minimum under the promissory notes are
contingent on the performance of the Company.
F-98
Independent
Auditor’s Report
To the Members
Aurora Diagnostics Holdings, LLC
Palm Beach Gardens, Florida
We have audited the accompanying balance sheets of Pathology
Solutions, LLC as of March 12, 2010, December 31,
2009, December 31, 2008, and December 31, 2007 and the
related statements of operations, member’s equity, and cash
flows for the period from January 1, 2010 through
March 12, 2010 and for the years ended December 31,
2009, December 31, 2008, and December 31, 2007. These
financial statements are the responsibility of the
Company’s management. Our responsibility is to express an
opinion on these financial statements based on our audits.
We conducted our audits in accordance with auditing standards
generally accepted in the United States of America. Those
standards require that we plan and perform the audit to obtain
reasonable assurance about whether the financial statements are
free of material misstatements. An audit includes examining, on
a test basis, evidence supporting the amounts and disclosures in
the financial statements. An audit also includes assessing the
accounting principles used and significant estimates made by
management, as well as evaluating the overall financial
statement presentation. We believe that our audits provide a
reasonable basis for our opinion.
In our opinion, the financial statements referred to above
present fairly, in all material respects, the financial position
of Pathology Solutions, LLC as of March 12, 2010,
December 31, 2009, December 31, 2008, and
December 31, 2007 and, the results of its operations and
its cash flows for the period January 1, 2010 through
March 12, 2010 and for the years ended December 31,
2009, December 31, 2008, and December 31, 2007 in
conformity with accounting principles generally accepted in the
United States of America.
West Palm Beach, Florida
June 18, 2010
F-99
Pathology
Solutions, LLC
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
March 12,
|
|
December 31,
|
|
December 31,
|
|
December 31,
|
|
|
|
2010
|
|
2009
|
|
2008
|
|
2007
|
|
|
|
($ in thousands)
|
|
|
|
Assets
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Current Assets
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash
|
|
$
|
487
|
|
|
$
|
675
|
|
|
$
|
2,368
|
|
|
$
|
165
|
|
|
Investments in trading securities
|
|
|
—
|
|
|
|
616
|
|
|
|
459
|
|
|
|
—
|
|
|
Accounts receivable, net
|
|
|
2,525
|
|
|
|
2,586
|
|
|
|
2,162
|
|
|
|
1,384
|
|
|
Other current assets
|
|
|
30
|
|
|
|
33
|
|
|
|
10
|
|
|
|
16
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current assets
|
|
|
3,042
|
|
|
|
3,910
|
|
|
|
4,999
|
|
|
|
1,565
|
|
|
Property and Equipment, net
|
|
|
712
|
|
|
|
693
|
|
|
|
371
|
|
|
|
213
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
3,754
|
|
|
$
|
4,603
|
|
|
$
|
5,370
|
|
|
$
|
1,778
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Liabilities and Members’ Equity
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Current Liabilities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts payable
|
|
$
|
463
|
|
|
$
|
346
|
|
|
$
|
502
|
|
|
$
|
112
|
|
|
Accrued compensation
|
|
|
103
|
|
|
|
44
|
|
|
|
16
|
|
|
|
41
|
|
|
Other accrued expenses
|
|
|
30
|
|
|
|
10
|
|
|
|
296
|
|
|
|
235
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current liabilities
|
|
|
596
|
|
|
|
400
|
|
|
|
815
|
|
|
|
387
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Members’ Equity
|
|
|
3,158
|
|
|
|
4,203
|
|
|
|
4,555
|
|
|
|
1,391
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
3,754
|
|
|
$
|
4,603
|
|
|
$
|
5,370
|
|
|
$
|
1,778
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Financial Statements.
F-100
Pathology
Solutions, LLC
Period from January 1, 2010 through March 12,
2010
and For the Years Ended December 31, 2009, 2008 and
2007
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Period
|
|
|
|
|
|
|
|
|
|
|
|
|
|
January 1, 2010
|
|
|
|
|
|
|
|
|
|
|
|
|
|
through
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
|
|
March 12, 2010
|
|
|
December 31, 2009
|
|
|
December 31, 2008
|
|
|
December 31, 2007
|
|
|
|
|
($ in thousands)
|
|
|
|
|
Net Revenues
|
|
$
|
3,198
|
|
|
$
|
16,464
|
|
|
$
|
8,191
|
|
|
$
|
4,444
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating costs and expenses:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost of services
|
|
|
1,177
|
|
|
|
6,923
|
|
|
|
2,925
|
|
|
|
1,923
|
|
|
Selling, general and administrative expenses
|
|
|
299
|
|
|
|
2,500
|
|
|
|
1,062
|
|
|
|
702
|
|
|
Depreciation expense
|
|
|
53
|
|
|
|
176
|
|
|
|
95
|
|
|
|
50
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total operating costs and expenses
|
|
|
1,529
|
|
|
|
9,599
|
|
|
|
4,082
|
|
|
|
2,675
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income from operations
|
|
|
1,669
|
|
|
|
6,865
|
|
|
|
4,109
|
|
|
|
1,769
|
|
|
Nonoperating income (expense):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest income
|
|
|
—
|
|
|
|
29
|
|
|
|
19
|
|
|
|
4
|
|
|
Other income (expense)
|
|
|
12
|
|
|
|
323
|
|
|
|
7
|
|
|
|
(8
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other income
|
|
|
12
|
|
|
|
352
|
|
|
|
26
|
|
|
|
(4
|
)
|
|
Net income
|
|
$
|
1,681
|
|
|
$
|
7,217
|
|
|
$
|
4,135
|
|
|
$
|
1,765
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Statements of Members’ Equity and Comprehensive
Income
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, beginning
|
|
$
|
4,203
|
|
|
$
|
4,555
|
|
|
$
|
1,391
|
|
|
$
|
77
|
|
|
Contributions
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
50
|
|
|
Distributions
|
|
|
(2,726
|
)
|
|
|
(7,569
|
)
|
|
|
(911
|
)
|
|
|
(505
|
)
|
|
Net income
|
|
|
1,681
|
|
|
|
7,217
|
|
|
|
4,135
|
|
|
|
1,765
|
|
|
Accumulated other comprehensive income
|
|
|
—
|
|
|
|
—
|
|
|
|
(60
|
)
|
|
|
4
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, ending
|
|
$
|
3,158
|
|
|
$
|
4,203
|
|
|
$
|
4,555
|
|
|
$
|
1,391
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
F-101
Pathology
Solutions, LLC
Statements of Cash Flows
Period from January 1, 2010 through March 12, 2010
and For the Years Ended December 31, 2009, 2008 and 2007
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Period
|
|
|
|
|
|
|
|
|
|
|
|
|
|
January 1, 2010
|
|
|
|
|
|
|
|
|
|
|
|
|
|
through
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
Year Ended
|
|
|
|
|
March 12, 2010
|
|
|
December 31, 2009
|
|
|
December 31, 2008
|
|
|
December 31, 2007
|
|
|
|
|
($ in thousands)
|
|
|
|
|
Cash Flows From Operating Activities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income
|
|
$
|
1,681
|
|
|
|
7,217
|
|
|
|
4,135
|
|
|
|
1,765
|
|
|
Adjustments to reconcile net income to net cash provided by
operating activities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Depreciation
|
|
|
53
|
|
|
|
176
|
|
|
|
95
|
|
|
|
50
|
|
|
Net unrealized loss (gain) on trading securities
|
|
|
34
|
|
|
|
(67
|
)
|
|
|
21
|
|
|
|
—
|
|
|
Allowance for bad debt
|
|
|
(128
|
)
|
|
|
(56
|
)
|
|
|
230
|
|
|
|
213
|
|
|
Changes in assets and liabilities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(Increase) Decrease in:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts receivable
|
|
|
189
|
|
|
|
(369
|
)
|
|
|
(1,007
|
)
|
|
|
(1,364
|
)
|
|
Other assets
|
|
|
2
|
|
|
|
(23
|
)
|
|
|
6
|
|
|
|
(16
|
)
|
|
Increase (Decrease) in:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts payable
|
|
|
117
|
|
|
|
(155
|
)
|
|
|
390
|
|
|
|
100
|
|
|
Accrued compensation
|
|
|
59
|
|
|
|
28
|
|
|
|
(25
|
)
|
|
|
22
|
|
|
Other accrued expenses
|
|
|
20
|
|
|
|
(287
|
)
|
|
|
2
|
|
|
|
192
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash provided by operating activities
|
|
|
2,027
|
|
|
|
6,464
|
|
|
|
3,847
|
|
|
|
962
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash Flows From Investing Activities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net trading securities
|
|
|
582
|
|
|
|
(89
|
)
|
|
|
(480
|
)
|
|
|
—
|
|
|
Purchase of property and equipment
|
|
|
(71
|
)
|
|
|
(499
|
)
|
|
|
(253
|
)
|
|
|
(165
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash provided by (used in) investing activities
|
|
|
511
|
|
|
|
(588
|
)
|
|
|
(733
|
)
|
|
|
(165
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash Flows From Financing Activities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Payments on line of credit
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(180
|
)
|
|
Capital contributions
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
50
|
|
|
Distributions to members
|
|
|
(2,726
|
)
|
|
|
(7,569
|
)
|
|
|
(911
|
)
|
|
|
(505
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash used in financing activities
|
|
|
(2,726
|
)
|
|
|
(7,569
|
)
|
|
|
(911
|
)
|
|
|
(635
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net increase (decrease) in cash
|
|
|
(188
|
)
|
|
|
(1,693
|
)
|
|
|
2,203
|
|
|
|
162
|
|
|
Cash, beginning
|
|
|
675
|
|
|
|
2,368
|
|
|
|
165
|
|
|
|
3
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash, ending
|
|
$
|
487
|
|
|
|
675
|
|
|
|
2,368
|
|
|
|
165
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
See Notes to Financial Statements.
F-102
Pathology
Solutions, LLC
|
|
|
Note 1.
|
Nature of
Business and Significant Accounting Policies
|
Nature of Business: Pathology
Solutions, LLC (the “Company”) was organized on
March 4, 2005 in the State of New Jersey as a limited
liability company to operate as an anatomic pathology laboratory.
The Company engages in the business of providing anatomic
pathology services to physicians in the Mid-Atlantic region of
the United States.
A summary of the Company’s significant accounting policies
follows:
Accounting estimates: The preparation
of financial statements in conformity with accounting principles
generally accepted in the United States of America requires
management to make estimates and assumptions that affect the
reported amounts of assets and liabilities, disclosure of
contingent assets and liabilities and the reported amounts of
revenues and expenses. Due to the inherent uncertainties in this
process, actual results could differ from those estimates.
Cash: The Company considers deposits
and investments that have original maturities of less than three
months, when purchased, to be cash equivalents. Daily bank
deposits are held in noninterest bearing accounts with a
financial institution that are insured by the Federal Deposit
Insurance Corporation’s (“FDIC”). The Company has
not experienced any losses on such accounts.
Fair value of financial
instruments: The Company has adopted Fair
Value Measurement and Disclosure guidance for all assets and
liabilities that are recognized or disclosed at fair value in
the financial statements. The guidance defines fair value as the
price that would be received upon sale of an asset or paid upon
transfer of a liability in an orderly transaction between market
participants at the measurement date and in the principle or
most advantageous market for that asset or liability. The fair
value should be calculated based on assumptions that market
participants would use in pricing the asset or liability, not on
assumptions specific to the entity.
In addition to defining fair value, the guidance expands the
disclosure requirements around fair value and establishes a fair
value hierarchy for valuation inputs. The hierarchy prioritizes
the inputs into three levels based on the extent to which inputs
used in measuring fair value or observable in the market. Each
fair value measurement is reported in one of the three levels,
which is determined by the lowest level input that is
significant to the fair value measurement in its entirety. These
levels are:
Level 1: Quoted market prices in active
markets for identical assets or liabilities.
Level 2: Observable market based inputs
or unobservable inputs that are corroborated by market data.
Level 3: Unobservable inputs that are not
corroborated by market data.
Certain assets and liabilities are measured at fair value on a
nonrecurring basis; that is instruments are not measured at fair
value on an ongoing basis but are subject to fair value
adjustments in certain circumstances (for example, when there is
evidence of impairment).
In determining the appropriate levels, the Company performs a
detailed analysis of the assets and liabilities that are subject
to the guidance. Valuations performed maximize the use of
observable inputs and minimize the use of unobservable inputs.
At each reporting period, all financial assets and liabilities
approximated fair value due to the short-term nature of these
accounts.
Investments in marketable equity
securities: The Company’s investments in
marketable equity securities are recorded at fair value and are
classified as trading. Fair value is based on quoted market
prices, level 1 inputs in the fair value hierarchy.
Management determines the appropriate classification of the
securities at the time they are acquired and evaluates the
appropriateness of such classifications at each balance sheet
date. At each balance sheet date, unrealized gains and losses
from trading securities are recognized in income or loss until
realized from sales. During 2009, the Company had a realized
gain on sale of marketable equity securities of approximately
$230,000 which is included in the other income on the
accompanying statement of operations.
F-103
Pathology
Solutions, LLC
Notes to Financial Statements — (Continued)
Revenue recognition and accounts
receivable: The Company recognizes revenue at
the time services are performed. Unbilled receivables are
recorded for services rendered during, but billed subsequent to,
the reporting period. Revenue is reported at the estimated
realizable amounts from patients, third-party payors and others
for services rendered. Revenue under certain third-party payor
agreements is subject to audit and retroactive adjustments.
Provisions for estimated third-party payor settlements and
adjustments are estimated in the period the related services are
rendered and adjusted in future periods as final settlements are
determined. The provision for doubtful accounts and the related
allowance are adjusted periodically, based upon an evaluation of
historical collection experience with specific payors for
particular services, anticipated collection levels with specific
payors for new services, industry reimbursement trends, and
other relevant factors. Changes in these factors in future
periods could result in increases or decreases in the
Company’s provision for doubtful accounts and impact its
results of operations and financial position. Blue Cross/Blue
Shield and Medicare provided the following of the Company’s
net revenues for the periods ended:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
March 12,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
|
|
Blue Cross/Blue Shield
|
|
|
33
|
%
|
|
|
27
|
%
|
|
|
29
|
%
|
|
|
28
|
%
|
|
Medicare
|
|
|
13
|
%
|
|
|
31
|
%
|
|
|
28
|
%
|
|
|
21
|
%
|
Property and equipment: Property and
equipment is stated at cost. Routine maintenance and repairs are
charged to expense as incurred, while costs of betterments and
renewals are capitalized. Depreciation is calculated on a
straight-line basis, over the estimated useful lives of the
respective assets, which range from 3 to 7 years. Leasehold
improvements are amortized over the shorter of the term of the
related lease, or the useful life of the asset.
Distributions to members and allocation of profits and
losses: Profits and losses are allocated to
the members in accordance with certain provisions contained in
the Company’s Limited Liability Company Agreement.
Distributions are also made in accordance with the terms of the
LLC Agreement.
Income taxes: The Company is a New
Jersey limited liability company for federal and state income
tax purposes, in accordance with the applicable provisions of
the Internal Revenue Code. Accordingly, the Company is generally
not subject to income taxes. The income attributable to the
limited liability company is distributed to the members in
accordance with the terms of the operating agreement.
The Financial Accounting Standards Board issued new guidance on
accounting for uncertainty in income taxes. The Company adopted
this new guidance for the year ended December 31, 2009.
Management evaluated the Company’s tax positions and
concluded that the Company had taken no uncertain tax positions
that require adjustment to the financial statements to comply
with the provisions of this guidance. With few exceptions, the
Company is no longer subject to income tax examinations by the
U.S. federal, state or local tax authorities for years
before 2006.
Concentration of credit risk: Financial
instruments that potentially subject the Company to
concentrations of credit risk are cash and accounts receivable.
Concentration of credit risk with respect to accounts receivable
is mitigated by the diversity of the Company’s payors.
While the Company has receivables due from federal and state
governmental agencies, the Company does not believe such
receivables represent a credit risk since the related healthcare
programs are funded by federal and state governments and payment
is primarily dependent on submitting appropriate documentation.
Subsequent events: The Company has
evaluated all subsequent events
through ,
2010, the date of issuance of the consolidated financial
statements.
F-104
Pathology
Solutions, LLC
Notes to Financial Statements — (Continued)
|
|
|
Note 2.
|
Accounts
Receivable
|
Accounts receivable consist of the following as of (in
thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
March 12,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
|
2010
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
|
|
Accounts receivable and unbilled receivables
|
|
$
|
2,805
|
|
|
$
|
2,994
|
|
|
$
|
2,625
|
|
|
$
|
1,618
|
|
|
Less: allowance for doubtful accounts
|
|
|
(208
|
)
|
|
|
(408
|
)
|
|
|
(464
|
)
|
|
|
(234
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts receivable, net
|
|
$
|
2,525
|
|
|
$
|
2,586
|
|
|
$
|
2,162
|
|
|
$
|
1,384
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Note 3.
|
Property
and Equipment
|
Property and equipment consist of the following as of (in
thousands):
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Estimated
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
useful life
|
|
March 12,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
|
(years)
|
|
2010
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
|
|
Transportation equipment
|
|
5
|
|
$
|
142
|
|
|
$
|
127
|
|
|
$
|
35
|
|
|
$
|
17
|
|
|
Leasehold improvements
|
|
5
|
|
|
73
|
|
|
|
69
|
|
|
|
—
|
|
|
|
—
|
|
|
Furniture and fixtures
|
|
7
|
|
|
42
|
|
|
|
42
|
|
|
|
7
|
|
|
|
3
|
|
|
Equipment
|
|
3 - 5
|
|
|
801
|
|
|
|
762
|
|
|
|
481
|
|
|
|
252
|
|
|
Software
|
|
3
|
|
|
57
|
|
|
|
42
|
|
|
|
21
|
|
|
|
19
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1,115
|
|
|
|
1,042
|
|
|
|
544
|
|
|
|
291
|
|
|
Less accumulated depreciation
|
|
|
|
|
(402
|
)
|
|
|
(349
|
)
|
|
|
(173
|
)
|
|
|
(79
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
712
|
|
|
$
|
693
|
|
|
$
|
371
|
|
|
$
|
213
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Depreciation expense was approximately $53,000 for the period
from January 1, 2010 to March 12, 2010 and $176,000,
$95,000, and $50,000 for the years ended December 31, 2009,
2008, and 2007, respectively.
|
|
|
Note 4.
|
Commitments
and Contingencies
|
During the ordinary course of business, the Company may become
subject to legal actions and proceedings. The Company may have
liability with respect to its employees and its pathologists.
Medical malpractice claims are generally covered by insurance.
While the Company believes the outcome of any such legal actions
and proceedings, individually or in the aggregate, will not have
a material adverse effect on the Company’s financial
condition, results of operations or liquidity, if the Company is
ultimately found liable under any medical malpractice claims,
there can be no assurance the Company’s medical malpractice
insurance coverage will be adequate to cover any such liability.
Healthcare regulatory environment: The
healthcare industry, in general, and the services the Company
provides, are subject to extensive federal and state laws and
regulations. Additionally, a portion of the Company’s
revenue is from payments by government-sponsored health
programs, including Medicare, and is subject to audit and
adjustments by applicable regulatory agencies. Failure to comply
with any of these laws or regulations, the results of increased
regulatory audits and adjustments, or change in the
interpretation of the coding of services or the amounts payable
for the Company’s services under these programs could have
a material adverse effect on the Company’s financial
position and results of operations.
F-105
Pathology
Solutions, LLC
Notes to Financial Statements — (Continued)
Employment Agreements: The Company has
employment agreements with its physician employees, the terms
which expire at various times. Such agreements provide for
minimum salary levels including various benefits.
Operating leases: The Company leased an
office and medical laboratory facility under a non-cancelable
lease agreement from January 2007 through May 2009. In June
2009, the Company entered into a lease at a new facility with
annual renewable terms expiring in September 2012. The terms of
the facility lease call for the Company to pay for certain taxes
or common utility charges. Rent expense including these taxes
and common utility charges was approximately $30,000 for the
period from January 1, 2010 through March 12, 2010 and
$80,000, $50,000, and $45,000 for the years ended
December 31, 2009, 2008, and 2007, respectively. Rent
expense associated with operating leases that include scheduled
rent increases and tenant incentives is recorded on a
straight-line basis over the term of the lease.
Aggregate future minimum annual rentals under the lease
agreement are as follows (in thousands):
| |
|
|
|
|
|
Year Ending December
31,
|
|
|
|
|
|
|
2010
|
|
$
|
68
|
|
|
2011
|
|
|
72
|
|
|
2012
|
|
|
56
|
|
|
|
|
|
|
|
|
|
|
$
|
275
|
|
|
|
|
|
|
|
|
|
|
Note 5.
|
Related
Party Transactions
|
The Company utilizes certain services of entities owned by the
majority member for management fees. Total cost of these
services was approximately $0 for the period from
January 1, 2010 through March 12, 2010 and $485,000,
$68,000, and $20,000 for the years ended December 31, 2009,
2008, and 2007, respectively. These related party amounts are
included in selling, general and administrative expenses in the
accompanying statements of operations.
|
|
|
Note 6.
|
Employee
Benefit Plans
|
The Company maintained a defined benefit pension plan covering
substantially all of the Company’s eligible employees which
was established on January 1, 2006 and terminated
March 15, 2009. Benefits were based on the years of service
and compensation. The Company’s plan was funded in
conformity with the funding requirements of the Employee
Retirement Income Security Act “ERISA” and the
Internal Revenue Code. The Company made contributions to the
plan of approximately $233,000 for the period from
January 1, 2009 through March 15, 2009 and $238,000
and $46,000 for the years ended December 31, 2008 and 2007,
respectively.
A summary of fair value of plan assets, projected benefit
obligations, and funded status at December 31, 2009, 2008
and 2007 is as follows:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
|
|
Fair value of plan assets
|
|
$
|
—
|
|
|
$
|
524,210
|
|
|
$
|
284,700
|
|
|
Projected benefit obligations
|
|
|
—
|
|
|
|
580,660
|
|
|
|
281,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Funded status at end of years
|
|
$
|
—
|
|
|
$
|
(56,450
|
)
|
|
$
|
3,700
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
F-106
Pathology
Solutions, LLC
Notes to Financial Statements — (Continued)
Amounts recognized in the balance sheets consist of:
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31,
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
|
2009
|
|
|
2008
|
|
|
2007
|
|
|
|
|
Current assets
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
3,700
|
|
|
Current liabilities
|
|
|
—
|
|
|
|
56,450
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
—
|
|
|
$
|
56,450
|
|
|
$
|
3,700
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
On February 18, 2009, the Company adopted a resolution to
terminate the pension plan as of March 15, 2009. All assets
of the plan were distributed on June 17, 2009, resulting in
a settlement gain of $137,900 which was utilized to offset
amounts previously recorded in other comprehensive income. The
assets associated with the defined benefit pension plan were
contributed to the Company’s Profit Sharing Plan during the
year ended December 31, 2009.
The Profit Sharing Plan covers substantially all of its
employees. Eligible participants are 21 years of age and
have worked at least 1,000 hours during a consecutive
12 month period. The Company may elect to make a
discretionary contribution in each plan year. For the period
ended March 12, 2010 and for the year ended
December 31, 2009, the Company contributed approximately $0
and $145,000, respectively, to the Profit Sharing Plan.
|
|
|
Note 7.
|
Subsequent
Events
|
On March 12, 2010, the Company was acquired by Aurora
Diagnostics Holdings, LLC in exchange for $22.5 million and
certain unsecured contingent promissory notes with maximum
principal payment amounts of $11.5 million contingent on
the performance of the Company.
F-107
Aurora
Diagnostics Holdings, LLC
Years Ended December 31, 2007, 2008 and 2009
(in thousands)
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Charged to
|
|
|
|
Net
|
|
|
|
|
|
Beginning
|
|
Statement of
|
|
Other
|
|
Write Offs and
|
|
Ending
|
|
Description
|
|
Balance
|
|
Operations
|
|
Increases(1)
|
|
Other Adjustments
|
|
Balance
|
|
|
|
Allowance for Doubtful Accounts:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Year ended December 31, 2007
|
|
$
|
421
|
|
|
$
|
2,378
|
|
|
$
|
4,743
|
|
|
$
|
(1,256
|
)
|
|
$
|
6,286
|
|
|
Year ended December 31, 2008
|
|
$
|
6,286
|
|
|
$
|
8,037
|
|
|
$
|
81
|
|
|
$
|
(6,207
|
)
|
|
$
|
8,197
|
|
|
Year ended December 31, 2009
|
|
$
|
8,197
|
|
|
$
|
9,488
|
|
|
$
|
240
|
|
|
$
|
(9,372
|
)
|
|
$
|
8,553
|
|
|
|
|
|
(1) |
|
Represents the Allowance for Doubtful Accounts recorded in
connection with the application of acquisition accounting for
the 2007, 2008 and 2009 acquisitions. |
F-108
Shares
Class A Common
Stock
Prospectus
, 2010
Joint Book-Running Managers
Morgan Stanley
UBS Investment Bank
Barclays Capital
Co-Managers
RBC Capital Markets
BMO Capital Markets
Lazard Capital
Markets
Until ,
2010 all dealers that buy, sell or trade the common stock may be
required to deliver a prospectus, regardless of whether they are
participating in this offering. This is in addition to the
dealers’ obligation to deliver a prospectus when acting as
underwriters and with respect to their unsold allotments or
subscriptions.
PART II
INFORMATION
NOT REQUIRED IN PROSPECTUS
|
|
|
Item 13.
|
Other
Expenses of Issuance and Distribution.
|
Estimated expenses payable in connection with the sale of the
common stock in this offering are as follows:
| |
|
|
|
|
|
SEC registration fee
|
|
$
|
10,695
|
|
|
FINRA filing fee
|
|
|
15,500
|
|
|
Listing fee
|
|
|
*
|
|
|
Printing and engraving expenses
|
|
|
*
|
|
|
Legal fees and expenses
|
|
|
*
|
|
|
Accounting fees and expenses
|
|
|
*
|
|
|
Transfer agent and registrar fees and expenses
|
|
|
*
|
|
|
Blue Sky fees and expenses
|
|
|
*
|
|
|
Miscellaneous
|
|
|
*
|
|
|
Total
|
|
|
*
|
|
|
|
|
|
*
|
|
To be filed by Amendment.
|
We will bear all of the expenses shown above.
|
|
|
Item 14.
|
Indemnification
of Directors and Officers.
|
We are incorporated under the laws of the State of Delaware.
Section 145 of the Delaware General Corporation Law
provides that a Delaware corporation may indemnify any persons
who are, or are threatened to be made, parties to any
threatened, pending or completed action, suit or proceeding,
whether civil, criminal, administrative or investigative (other
than an action by or in the right of such corporation), by
reason of the fact that such person was an officer, director,
employee or agent of such corporation, or is or was serving at
the request of such person as an officer, director, employee or
agent of another corporation or enterprise. The indemnity may
include expenses (including attorneys’ fees), judgments,
fines and amounts paid in settlement actually and reasonably
incurred by such person in connection with such action, suit or
proceeding, provided that such person acted in good faith and in
a manner he or she reasonably believed to be in or not opposed
to the corporation’s best interests and, with respect to
any criminal action or proceeding, had no reasonable cause to
believe that his or her conduct was illegal. A Delaware
corporation may indemnify any persons who are, or are threatened
to be made, a party to any threatened, pending or completed
action or suit by or in the right of the corporation by reason
of the fact that such person was a director, officer, employee
or agent of such corporation, or is or was serving at the
request of such corporation as a director, officer, employee or
agent of another corporation or enterprise. The indemnity may
include expenses (including attorneys’ fees) actually and
reasonably incurred by such person in connection with the
defense or settlement of such action or suit provided such
person acted in good faith and in a manner he or she reasonably
believed to be in or not opposed to the corporation’s best
interests except that no indemnification is permitted without
judicial approval if the officer or director is adjudged to be
liable to the corporation. Where an officer or director is
successful on the merits or otherwise in the defense of any
action referred to above, the corporation must indemnify him or
her against the expenses which such officer or director has
actually and reasonably incurred. Our certificate of
incorporation and bylaws provide for the indemnification of our
directors and officers to the fullest extent permitted under the
Delaware General Corporation Law. We currently maintain a
directors’ and officers’ liability insurance policy.
II-1
Section 102(b)(7) of the Delaware General Corporation Law
permits a corporation to provide in its certificate of
incorporation that a director of the corporation shall not be
personally liable to the corporation or its stockholders for
monetary damages for breach of fiduciary duties as a director,
except for liability for any:
|
|
|
| |
•
|
transaction from which the director derives an improper personal
benefit;
|
| |
| |
•
|
act or omission not in good faith or that involves intentional
misconduct or a knowing violation of law;
|
| |
| |
•
|
unlawful payment of dividends or redemption of shares; or
|
| |
| |
•
|
breach of a director’s duty of loyalty to the corporation
or its stockholders.
|
Our certificate of incorporation and bylaws include such a
provision. Expenses incurred by any officer or director in
defending any such action, suit or proceeding in advance of its
final disposition shall be paid by us upon delivery to us of an
undertaking, by or on behalf of such director or officer, to
repay all amounts so advanced if it shall ultimately be
determined that such director or officer is not entitled to be
indemnified by us.
Section 174 of the Delaware General Corporation Law
provides, among other things, that a director, who willfully or
negligently approves of an unlawful payment of dividends or an
unlawful stock purchase or redemption, may be held liable for
such actions. A director who was either absent when the unlawful
actions were approved, or dissented at the time, may avoid
liability by causing his or her dissent to such actions to be
entered in the books containing minutes of the meetings of the
Board of Directors at the time such action occurred or
immediately after such absent director receives notice of the
unlawful acts.
As permitted by the Delaware General Corporation Law, we expect
to enter into indemnity agreements with each of our directors
and executive officers, that require us to indemnify such
persons against any and all expenses (including attorneys’
fees), witness fees, damages, judgments, fines, settlements and
other amounts incurred (including expenses of a derivative
action) in connection with any action, suit or proceeding,
whether actual or threatened, to which any such person may be
made a party by reason of the fact that such person is or was a
director, an officer or an employee of our company or any of its
affiliated enterprises. Under these agreements, we would not be
required to provide indemnification for matters including:
|
|
|
| |
•
|
indemnification for any proceeding with respect to the unlawful
payment of remuneration to the director or officer;
|
| |
| |
•
|
indemnification for proceedings involving a final judgment that
the director or officer is required to disgorge profits from the
purchase or sale of our securities or a final judgment that the
director’s or officer’s conduct was in bad faith,
knowingly fraudulent or deliberately dishonest or constituted
willful misconduct or a breach of his or her duty of loyalty;
|
| |
| |
•
|
indemnification for proceedings or claims brought by an officer
or director against us or any of our directors, officers,
employees or agents, except for claims to establish a right of
indemnification or proceedings or claims approved by our Board
of Directors or required by law;
|
| |
| |
•
|
indemnification for settlements the director or officer enters
into without our written consent; or
|
| |
| |
•
|
indemnification in violation of any undertaking required by the
Securities Act or in any registration statement that we file.
|
The indemnification agreements also set forth procedures that
will apply in the event of a claim for indemnification
thereunder.
At present, there is no pending litigation or proceeding
involving any of our directors or executive officers as to which
indemnification is required or permitted, and we are not aware
of any threatened litigation or proceeding that may result in a
claim for indemnification.
We have an insurance policy covering our officers and directors
with respect to liabilities, including liabilities arising under
the Securities Act or otherwise.
II-2
We plan to enter into an underwriting agreement which provides
that the underwriters are obligated, under some circumstances,
to indemnify our directors, officers and controlling persons
against specified liabilities, including liabilities under the
Securities Act.
Reference is made to the following documents filed as exhibits
to this registration statement regarding relevant
indemnification provisions described above and elsewhere in this
prospectus:
|
|
|
| |
•
|
Form of Certificate of Incorporation of Aurora Diagnostics,
Inc.; and
|
| |
| |
•
|
Form of By-laws of Aurora Diagnostics, Inc.
|
|
|
|
Item 15.
|
Recent
Sales of Unregistered Securities.
|
Set forth below is information regarding shares of common stock
issued by us within the past three years. Also included is the
consideration, if any, received by us for such shares and
information relating to the section of the Securities Act, or
rule of the SEC, under which we claimed an exemption from
registration.
(a) Issuances of Capital Stock
On April 23, 2010, we issued 100 shares of our common
stock to James C. New, our Chairman, Chief Executive Officer and
President, for nominal consideration of $100. No underwriters
were involved in the foregoing sale of securities. The
securities were issued to a U.S. investor in reliance upon
the exemption from the registration requirements of the
Securities Act, as set forth in Section 4(2) under the
Securities Act relating to sales by an issuer not involving any
public offering, to the extent an exemption from such
registration was required. In connection with the Reorganization
Transactions, these shares will be cancelled.
In connection with the Reorganization Transactions, we will
issue shares
of our Class A common stock to the Summit Partners
Equityholders
and shares
of our Class A common stock to the KRG Equityholders. The
securities will be issued to U.S. investors in reliance
upon the exemption from the registration requirements of the
Securities Act, as set forth in Section 4(2) under the
Securities Act relating to sales by an issuer not involving any
public offering, to the extent an exemption from such
registration is required.
In connection with the Reorganization Transactions, we will
issue shares
of our Class B common stock to the Summit Partners
Equityholders
and shares
of our Class B common stock to the Management
Equityholders. The securities will be issued to
U.S. investors in reliance upon the exemption from the
registration requirements of the Securities Act, as set forth in
Section 4(2) under the Securities Act relating to sales by
an issuer not involving any public offering, to the extent an
exemption from such registration is required.
|
|
|
Item 16.
|
Exhibits
and Financial Statement Schedules.
|
(a) Exhibits: The list of exhibits is set
forth immediately following the signature pages to this
Registration Statement and is incorporated herein by reference.
(b) Financial Statements
Schedules: Schedule II—Valuation and
Qualifying Accounts on
page F-85
is incorporated herein by reference.
* (f) The undersigned registrant hereby undertakes to
provide to the underwriters at the closing specified in the
underwriting agreement, certificates in such denominations and
registered in such names as required by the underwriters to
permit prompt delivery to each purchaser.
* (h) Insofar as indemnification for liabilities arising
under the Securities Act of 1933 may be permitted to
directors, officers and controlling persons of the registrant
pursuant to the foregoing provisions, or otherwise, the
registrant has been advised that in the opinion of the
Securities and Exchange Commission such indemnification is
against public policy as expressed in the Act and is, therefore,
unenforceable. In the event that a claim for indemnification
against such liabilities (other than the payment by the
registrant of expenses incurred or paid by a director, officer
or controlling person of the registrant in the successful
defense of any action, suit or proceeding) is asserted by such
director, officer or controlling person in connection with the
securities being registered, the
II-3
registrant will, unless in the opinion of its counsel the matter
has been settled by controlling precedent, submit to a court of
appropriate jurisdiction the question whether such
indemnification by it is against public policy as expressed in
the Act and will be governed by the final adjudication of such
issue.
* (i) The undersigned registrant hereby undertakes that:
|
|
|
| |
•
|
For purposes of determining any liability under the Securities
Act of 1933, the information omitted from the form of prospectus
filed as part of this registration statement in reliance upon
Rule 430A and contained in a form of prospectus filed by
the registrant pursuant to Rule 424(b)(1) or (4) or
497(h) under the Securities Act shall be deemed to be part of
this registration statement as of the time it was declared
effective.
|
| |
| |
•
|
For the purpose of determining any liability under the
Securities Act of 1933, each post-effective amendment that
contains a form of prospectus shall be deemed to be a new
registration statement relating to the securities offered
therein, and the offering of such securities at that time shall
be deemed to be the initial bona fide offering thereof.
|
II-4
SIGNATURES
Pursuant to the requirements of the Securities Act of 1933, the
registrant has duly caused this registration statement to be
signed on its behalf by the undersigned, thereunto duly
authorized in the City of Palm Beach Gardens, State of Florida
on June 18, 2010.
AURORA DIAGNOSTICS, INC.
By: Gregory A. Marsh, its Chief Financial Officer
Pursuant to the requirements of the Securities Act of 1933, as
amended, this registration statement has been signed below by
the following persons in the capacities and on the dates
indicated.
| |
|
|
|
|
|
|
|
Name
|
|
Title
|
|
Date
|
|
|
|
|
|
|
|
|
|
*
James
C. New
|
|
Chief Executive Officer and
Director (principal executive officer)
|
|
June 18, 2010
|
|
|
|
|
|
|
|
/s/ Gregory
A. Marsh
Gregory
A. Marsh
|
|
Chief Financial Officer
(principal financial and accounting officer)
|
|
June 18, 2010
|
|
|
|
|
|
|
|
*
Thomas
S. Roberts
|
|
Director
|
|
June 18, 2010
|
|
|
|
|
|
|
|
*
Christopher
Dean
|
|
Director
|
|
June 18, 2010
|
|
|
|
|
|
|
|
*
Peter
J. Connolly
|
|
Director
|
|
June 18, 2010
|
|
|
|
|
|
|
|
*
Mark
M. King
|
|
Director
|
|
June 18, 2010
|
|
|
|
|
|
|
|
*
Christopher
J. Bock
|
|
Director
|
|
June 18, 2010
|
|
|
|
|
|
|
|
*
Blair
Tikker
|
|
Director
|
|
June 18, 2010
|
|
|
|
|
|
|
*/s/ Gregory A. Marsh
Gregory
A. Marsh
Attorney in Fact
|
|
|
|
|
II-5
SIGNATURES
AND POWER OF ATTORNEY
I, the undersigned director of Aurora Diagnostics, Inc., hereby
severally constitute and appoint Gregory A. Marsh and James C.
New (with full power to act alone), my true and lawful
attorney-in-fact and agent, with full power of substitution and
resubstitution for him and in his name, place and stead, and in
any and all capacities, to sign any and all amendments
(including post-effective amendments) to this Registration
Statement, and any other registration statement for the same
offering pursuant to Rule 462(b) under the Securities Act
of 1933, and to file the same, with all exhibits thereto and
other documents in connection therewith, with the Securities and
Exchange Commission, granting unto said attorney-in-fact and
agent full power and authority to do and perform each and every
act and thing requisite or necessary to be done in and about the
premises, as full to all intents and purposes as he might or
could do in person, hereby ratifying and confirming all that
said attorney-in-fact and agent, or his substitute or
substitutes, may lawfully do or cause to be done by virtue
hereof.
Pursuant to the requirements of the Securities Act of 1933, as
amended, this registration statement has been signed below by
the following person in the capacities and on the dates
indicated.
| |
|
|
|
|
|
|
|
Name
|
|
Title
|
|
Date
|
|
|
|
|
|
|
|
|
|
/s/ James
M. Emanuel
James
M. Emanuel
|
|
Director
|
|
June 18, 2010
|
II-6
EXHIBIT INDEX
| |
|
|
|
|
Exhibit
|
|
|
|
|
|
Number
|
|
|
|
Description
|
|
|
|
1.1*
|
|
—
|
|
Form of Underwriting Agreement
|
|
3.1*
|
|
—
|
|
Certificate of Incorporation of Aurora Diagnostics, Inc.
|
|
3.2*
|
|
—
|
|
Bylaws of Aurora Diagnostics, Inc.
|
|
4.1*
|
|
—
|
|
Specimen Stock Certificate of Aurora Diagnostics, Inc.’s
Class A Common Stock, par value $0.01 per share
|
|
5.1*
|
|
—
|
|
Opinion of Alston & Bird LLP
|
|
10.1*
|
|
—
|
|
Form of Registration Rights Agreement of Aurora Diagnostics,
Inc., by and among Aurora Diagnostics, Inc. and the other
parties named therein
|
|
10.2*
|
|
—
|
|
Form of Tax Receivable Agreement, among Aurora Diagnostics,
Inc., ARDX Sub, Inc., Aurora Diagnostics Holdings, LLC and the
other parties named therein
|
|
10.3*
|
|
—
|
|
Form of Second Amended and Restated Limited Liability Company
Agreement of Aurora Diagnostics Holdings, LLC, by and among
Aurora Diagnostics Holdings, LLC and the other parties named
therein
|
|
10.4*
|
|
—
|
|
Form of Reorganization Agreement, by and among Aurora
Diagnostics, Inc., ARDX Sub, Inc., Aurora Diagnostics Holdings,
LLC and the other parties named therein
|
|
10.5*
|
|
—
|
|
Form of Credit and Guaranty Agreement, by and among Aurora
Diagnostics LLC as Borrower, Aurora Diagnostics Holdings, LLC
and certain subsidiaries and affiliates of Aurora Diagnostics,
LLC as Guarantors, the various lenders party thereto, Barclays
Bank PLC as Administrative Agent and Collateral Agent, Barclays
Capital, Morgan Stanley Senior Funding, Inc. and UBS Securities
LLC as Joint Lead Arrangers and Joint Bookrunners, Morgan
Stanley Senior Funding, Inc. as Syndication Agent and UBS
Securities LLC as Documentation Agent.
|
|
21*
|
|
—
|
|
List of subsidiaries of Registrant
|
|
23.1
|
|
—
|
|
Consent of McGladrey & Pullen, LLP
|
|
23.2
|
|
—
|
|
Consent of McGladrey & Pullen, LLP
|
|
24.1
|
|
—
|
|
Power of Attorney (included in Part II of this registration
statement)
|
|
|
|
|
*
|
|
To be filed by amendment.
|