Attached files
| file | filename |
|---|---|
| EX-5.1 - OPINION - MMRGlobal, Inc. | exhibit5-1.pdf |
| EX-23.1 - CONSENT - MMRGlobal, Inc. | exhibit23-1.pdf |
| EX-5.1 - OPINION - MMRGlobal, Inc. | exh5-1.htm |
| EX-23.1 - CONSENT - MMRGlobal, Inc. | exh23-1.htm |
As filed with the Securities
and Exchange Commission on June 18, 2010
Registration No. 333-________
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
![]()
FORM S-1
![]()
MMRGLOBAL, INC.
|
Delaware (State or other jurisdiction of incorporation or organization) |
7389 (Primary Standard Industrial Classification Code Number) |
33-0892797 (I.R.S. Employer Identification Number) |
468 North Camden Drive, Suite 200
Beverly Hills, California 90210
(310) 476-7002
(Address, including zip code, and telephone number, including
area code, of Registrant's principal executive offices)
![]()
Robert H. Lorsch
Chief Executive Officer
MMRGlobal, Inc.
468 North Camden Drive, Suite 200
Beverly Hills, California 90210
(310) 476-7002
area code, of agent for service)
![]()
Copies to:
Mark L. Skaist, Esq.
Vince Cangolosi, Esq.
Stradling Yocca Carlson & Rauth,
a Professional Corporation
660 Newport Center Drive, Suite 1600
Newport Beach, California 92660
(949) 725-4100
Approximate date of commencement of proposed sale to the public:
As soon as practicable after the effective date of this Registration Statement.
If any of the securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933 check the following box: x
If this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, please check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. o
If this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. o
If this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. o
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated file, or a smaller
reporting company. See the definitions of "large accelerated filer," "accelerated file" and "smaller reporting
company" in Rule 12b-2 of the Exchange Act. (Check one):
|
Large accelerated filer o |
Accelerated filer o |
Non-accelerated filer o
|
Smaller reporting company x |
CALCULATION OF REGISTRATION FEE
|
Title of Each Class of Securities to be Registered |
Amount to |
Proposed Maximum |
Proposed Maximum |
Amount of |
|||||
|
Common Stock, $0.001 par value per share |
60,000,000 |
$0.20 |
$10,000,000 |
$713.00 |
![]()
(1) Pursuant to Rule 416 under the Securities Act, the shares being registered hereunder include such indeterminate number of shares of common stock as may be issuable with respect to the shares being registered hereunder as a result of stock splits, stock dividends or similar transactions.
(2) Estimated solely for the purpose of calculating the amount of the registration fee pursuant to Rule 457(c) of the Securities Act of 1933, using the closing price as reported on the OTC Bulletin Board on June 14, 2010.
The registrant hereby amends this registration statement on such date or dates as may be necessary to delay its effective date until the registrant shall file a further amendment which specifically states that this registration statement shall thereafter become effective in accordance with section 8(a) of the Securities Act of 1933 or until the registration statement shall become effective on such date as the Securities and Exchange Commission acting pursuant to said section 8(a) may determine.
The information in this prospectus is not complete and may be changed. We may not sell these securities until the registration
statement filed with the Securities and Exchange Commission is effective. This prospectus is not an offer to sell these securities
and it is not soliciting an offer to buy these securities in any jurisdiction where the offer or sale is not permitted. Subject to Completion, Dated June 18, 2010 Prospectus

60,000,000 Shares
Common Stock
This prospectus relates to the offer and resale of up to 60,000,000 shares of our common stock, par value $0.001 per share, by the selling stockholder, Dutchess Equity Fund, L.P., or "Dutchess". Of such shares, (i) Dutchess has agreed to purchase 60,000,000 pursuant to the investment agreement dated September 15, 2009, as amended on May 7, 2010, between Dutchess and us, and (ii) 230,800 shares were issued to Dutchess in partial consideration for the preparation of the documents for its investment. Subject to the terms and conditions of such investment agreement, which is referred to in this prospectus as the "Investment Agreement," we have the right to put up to $10.0 million in shares of our common stock to Dutchess. This arrangement is sometimes referred to as an "Equity Line." For more information on the selling stockholder, please see the section of this prospectus entitled "Selling Stockholder".
We will not receive any proceeds from the resale of these shares of common stock offered by Dutchess. We will, however, receive proceeds from the sale of shares to Dutchess pursuant to the Equity Line. When we put an amount of shares to Dutchess, the per share purchase price that Dutchess will pay to us in respect of such put will be determined in accordance with a formula set forth in the Investment Agreement. Generally, in respect of each put, Dutchess will pay us a per share purchase price equal to ninety-four percent (94%) of the average of two lowest daily volume weighted average price ("VWAP") of the Company's common stock during the five consecutive trading day period beginning on the trading day immediately following the date of delivery of the applicable put notice.
Dutchess may sell the shares of common stock from time to time at the prevailing market price on the Over-the Counter (OTC) Bulletin Board, or on an exchange if our shares of common stock become listed for trading on such an exchange, or in negotiated transactions. Dutchess is an "underwriter" within the meaning of the Securities Act of 1933, as amended (the "Securities Act") in connection with the resale of our common stock under the Equity Line. For more information, please see the section of this prospectus entitled "Plan of Distribution".
Our common stock is quoted on the OTC Bulletin Board under the symbol "MMRF". In connection with our name change, we have applied for a new trading symbol, but will continue to use MMRF until we have been granted a new symbol. The last reported sale price of our common stock on the OTC Bulletin Board on June 16, 2010 was $0.19 per share.
Investing in the offered securities involves a high degree of risk, including those risks set forth in the "Risk Factors" section of this prospectus, as well as those set forth in any prospectus supplement.
We will be responsible for all fees and expenses incurred in connection with the preparation and filing of this registration statement, provided, however, we will not be required to pay any underwriters' discounts or commissions relating to the securities covered by the registration statement.
You should read this prospectus and any prospectus supplement carefully before you decide to invest. You should not assume that the information in this prospectus is accurate as of any date other than the date on the front of this document.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or passed upon the accuracy or adequacy of this prospectus. Any representation to the contrary is a criminal offense.
![]()
The date of this prospectus is ____________, 2010
TABLE OF CONTENTS
|
|
Page |
|
|
|
ii |
|
|
|
1 |
|
|
|
9 |
|
|
|
16 |
|
|
|
16 |
|
|
|
18 |
|
|
|
19 |
|
|
MANAGEMENT'S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
|
37 |
|
|
55 |
|
|
|
58 |
|
|
|
63 |
|
|
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT |
|
65 |
|
|
66 |
|
|
|
70 |
|
|
|
73 |
|
|
|
74 |
|
|
|
74 |
|
|
|
74 |
|
|
|
75 |
|
|
|
F-1 |
![]()
This prospectus is part of a registration statement that we filed with the Securities and Exchange Commission. You should rely only on the information contained in this prospectus or to which we have referred you. We have not authorized anyone to provide you with information or to make any representation on behalf of the Company that is different from that contained in this prospectus. You should not rely on any unauthorized information or representation. This prospectus is an offer to sell only the securities offered by this prospectus under circumstances and in jurisdictions where it is lawful to do so. The information in this prospectus is accurate only as of the date of this prospectus, regardless of the date of delivery of this prospectus or of any sales of these securities. Our business, financial condition, results of operations and prospects may have changed since the date of this prospectus. This prospectus may be used only in jurisdictions where it is legal to sell these securities.
![]()
i
CAUTIONARY STATEMENT REGARDING FORWARD LOOKING STATEMENTS
This prospectus includes forward-looking statements. All statements, other than statements of historical fact, contained in this prospectus constitute forward-looking statements. These statements are based on the current expectations, forecasts, and assumptions of our management and are subject to various risks and uncertainties that could cause our actual results to differ materially from those expressed or implied by the forward-looking statements. Forward-looking statements are sometimes identified by language such as "believe," "may," "could," "estimate," "continue," "anticipate," "intend," "should," "plan," "expect," "appear," "future," "likely," "probably," "suggest," "goal," "potential" and similar expressions and may also include references to plans, strategies, objectives, and anticipated future performance as well as other statements that are not strictly historical in nature. The risks, uncertainties, and other factors that could cause our actual results to differ materially from those expressed or implied in this prospectus include, but are not limited to, those noted under the caption "Risk Factors" beginning on page 8 of this prospectus. Readers should carefully review this information as well the risks and other uncertainties described in other filings we may make after the date of this prospectus with the Securities and Exchange Commission.
Readers are cautioned not to place undue reliance on forward-looking statements. They reflect opinions, assumptions, and estimates only as of the date they were made, and we undertake no obligation to publicly update or revise any forward-looking statements in this prospectus, whether as a result of new information, future events or circumstances, or otherwise.
![]()
ii
PROSPECTUS SUMMARY
This summary highlights the information contained elsewhere in this prospectus. Because this is only a summary, it does not contain all of the information that you should consider before buying shares of our common stock. You should read the entire prospectus and any prospectus supplements carefully, especially the sections entitled "Caution Regarding Forward Looking Statements," "Risk Factors" and "Management's Discussion and Analysis of Financial Condition and Results of Operations," together with our financial statements and the related notes included elsewhere in this prospectus and in any prospectus supplements related thereto, before deciding to purchase shares of our common stock.
MMRGlobal, Inc.
Depending upon the context, the terms "MMRGlobal", "MMR Information Systems," "Company," "we," "our" and "us," refers to either MMRGlobal, Inc. (formerly known as MMR Information Systems, Inc.) alone, or MMRGlobal, Inc. and its subsidiaries collectively.
Organizational History
We were incorporated as Favrille in Delaware in 2000, and at its inception and before the Merger on January 27, 2009 (see below), we previously operated under a different management team as a biopharmaceutical company focused on the development and commercialization of targeted immunotherapies for the treatment of cancer and other diseases of the immune system. In May 2008, our ongoing Phase 3 registration trial for our lead product candidate failed to show a statistically significant improvement in the treatment of patients with follicular B-cell non-Hodgkin's lymphoma, and accordingly, we determined to sell all of our equipment related to manufacturing of this product, as well as other personal property in an auction. On September 9, 2008, this auction was consummated and we received $3.2 million in net proceeds from the sale of the assets. With the disposition of all of our equipment and other personal property, we ceased to engage in any operations and became a "shell company" as such term is defined in Rule 12b-2 of the Securities Exchange Act of 1934, as amended, or the Exchange Act.
Agreement and Plan of Merger and Reorganization
On November 8, 2008, we entered into an Agreement and Plan of Merger and Reorganization, or the Merger Agreement, with our wholly-owned subsidiary Montana Merger Sub, Inc., or the Merger Sub, a Delaware corporation, and MMR, pursuant to which Merger Sub would merge with and into MMR, with MMR continuing as the surviving corporation and a wholly-owned subsidiary of our company. We refer to this transaction, which closed on January 27, 2009 and which became effective on February 9, 2009, as the Merger. Following the Merger, the holders of MMR equity prior to the Merger, on a fully diluted basis, owned or had the right to acquire approximately 60.3% of our equity, the holders of our equity prior to the Merger, on a fully diluted basis, owned approximately 33.2% of our equity, and certain beneficiaries under the Creditor Plan (which consist of our former officers and former directors and their affiliates) had the right to own up to approximately 6.5% of our equity. The Creditor Plan is described in Item 7 "Management's Discussion and Analysis of Financial Condition and Results of Operations" and a copy is filed as Exhibit 10.5 to our annual report on Form 10-K for the year ended December 31, 2009..
As a result of the Merger in January 2009, MMR became our wholly-owned subsidiary and its legal name became MyMedicalRecords, Inc. Although we are the legal acquirer, the Merger is accounted for as a reverse acquisition in accordance with U.S. generally accepted accounting principles, or GAAP. Under this method of accounting, Favrille is treated as the "acquired" company for financial reporting purposes. This determination is primarily based on the fact that MMR's former shareholders hold a majority of the equity of the consolidated company, MMR's operations comprise the ongoing operations of the consolidated entity and MMR's senior management and director designees have assumed control of the consolidated company.
In addition, upon the consummation of the Merger on January 27, 2009, MMR became our wholly-owned operating subsidiary and we ceased being a "shell company" as such term is defined in Rule 12b-2 of the Exchange Act. We refer to MyMedicalRecords, Inc. as "MMR" from time to time in this prospectus. MMR was incorporated in Delaware in 2005 and is headquartered in Los Angeles, California.
1
The Merger is being accounted for as a reverse acquisition in accordance with U.S. generally accepted accounting principles. Under this method of accounting, we were treated as the "acquired" company for financial reporting purposes. This determination was primarily based on the fact that MMR's former shareholders hold a majority of the equity of the consolidated company, MMR's operations comprise the ongoing operations of the consolidated entity and MMR's senior management and director designees have assumed control of the consolidated company.
Our Company and Business Subsequent to the Merger
As of the closing date of the Merger on January 27, 2009, we adopted MMR's business of empowering consumers to manage the important records in their life, whether paper-based or digital, and by doing so, to better control and organize their lives overall. We do this through web-based products that facilitate consumer access to medical records and vital documents (such as living wills, birth certificates, insurance policies and important financial records). The following description of our business relates to our current business and operations.
Our principal product, "MyMedicalRecords," is an easy-to-use, secure web-based Personal Health Record system, or PHR, which allows documents, images and voice mail messages to be transmitted in and out of our proprietary system using a variety of methods, including fax and file upload. Our platform converts documents it receives by fax into a Portable Digital Format, or PDF, file. These files and any other uploaded files are stored in the consumer's personal account, which is secured by a unique user ID and password combination. A notification is sent to up to three user e-mail addresses (including to e-mail enabled cell phones) whenever a new record is received. Users can then access their files via the Internet and take advantage of an easy-to-use, customized filing system that allows them to categorize, annotate and file their records for easy access, organization and retrieval. Users can then print, download or e-mail their records from their account, giving our customers greater control over their own personal health and other information, which they can share with health care providers and others as needed. We are currently selling our MyMedicalRecords PHR product primarily through corporations as an employee benefit and to affinity groups as a "value-added" service for their members, as well as directly to retail customers, although we plan to sell through additional sales channels in the future.
In the second half of 2007, we launched our "MyESafeDepositBox" service, which is geared towards small businesses, the financial services, insurance and legal service industries. MyESafeDepositBox is based on the same technology and architecture as our MyMedicalRecords PHR product. However, rather than focusing on medical records, MyESafeDepositBox is designed to provide secure on-line storage for vital financial, legal and insurance documents. MyESafeDepositBox may be used as a virtual on-line "safe" and could serve as an essential part of any household's or business's disaster preparedness plan.
We are now also in the final stages of developing a new product, "MyMedicalRecords Pro" (MMRPro), which we believe will provide physician offices with a powerful and cost-effective solution to the costly and time-consuming problem of digitizing paper-based medical records. When complete, our MMRPro service is expected to combine the scanning of medical records at a physician's office with 24/7 access to these records for both doctors and patients through separate secure web-based portals. We recognize that the critical nature of personal health information requires that these product advances be implemented with the utmost care to protect the privacy and confidentiality of our customers' data. Although we are not a covered entity under the Health Insurance Portability and Accountability Act of 1996, commonly referred to as HIPAA, we consider it important to take into account privacy and security standards and requirements of HIPAA when implementing our products and services and we believe that we meet and/or exceed current HIPAA standards.
We plan to continue to take advantage of the burgeoning consumer health information market and leverage recent and anticipated federal legislation, including by adapting the technology supporting our products to be able to take advantage of government funds in the 2009 economic stimulus package. We believe that as medical costs and insurance costs increase, while benefits decrease, healthcare consumers will require greater control over their personal (and their family's) health and health information. Our MyMedicalRecords PHR product is designed to enable healthcare consumers to store their important medical records and data in one central and secure place where they can manage those records and control how they are accessed and shared. While every healthcare consumer in the U.S. is a potential user of our MyMedicalRecords PHR product, we believe that the product has most appeal to these particular market niches:
- the chronically ill and their families who share information among many doctors;
- consumers with "senior" parents who, in their role as caregivers, want to be sure they have current medical information readily available to react quickly in case of parental illness;
2
- consumers with newborns who will be able to build a complete medical file for the newborn, which can last the newborn's entire life;
- travelers and businesspeople working overseas who want to ensure that they always have access to their medical records in the event of an emergency; and
- corporations seeking to offer their employees a valuable benefit (we believe those with expatriate employees would be particularly interested in our product).
Because of the similarity in functionality between our MyMedicalRecords PHR product and our MyESafeDepositBox product, we market these products through many of the same channels. Our marketing strategy for our MyMedicalRecords PHR product and MyESafeDepositBox product focuses primarily on the following channels:
- to corporate accounts, as an added employee benefit for their employees or bundled or co-branded with their product offerings to their customers;
- through affinity groups (such as alumni organizations and other membership organizations) and discount health benefit membership groups to their members; and
- direct to consumers as a retail subscription product, on-line and through hospitals and doctors.
Our MyMedicalRecords Pro product is designed to combine onsite scanning services with the storage and accessibility features of our MyMedicalRecords PHR product. We plan to market our MyMedicalRecords Pro product to doctors, particularly solo and small group practitioners, who are looking for an easy and inexpensive way to digitize patient medical files and give patients up-to-date access to those records.
For a complete description of our business, please see the section entitled "Our Business".
Summary Financial Data
Because this is only a summary of our financial information, it does not contain all of the financial information that may be important to you. Therefore, you should carefully read all of the information in this prospectus and any prospectus supplement, including the financial statements and their explanatory notes and the section entitled "Management's Discussion and Analysis of Financial Condition and Results of Operations," before making a decision to invest in our common stock. The information contained in the following summary is derived from our financial statements for the quarters ended March 31, 2010 and 2009 and the years ended December 31, 2009 and 2008.
For accounting purposes, the Merger was treated as a reverse acquisition with MMR being the accounting acquirer. Accordingly, the historical financial results prior to the Merger are those of MMR and replace our historical financial results as we existed prior to the Merger. Our results of operations are included in MMR's financial results beginning on January 27, 2009.
| Three Months ended | Fiscal Year ended | |||||||||||
|
March 31,
|
December 31,
|
|||||||||||
|
2010
|
2009
|
2009
|
2008
|
|||||||||
| Total Revenues | $ | 90,164 | $ | 145,787 | $ | 619,249 | $ | 451,355 | ||||
| Cost of Revenues |
113,036
|
106,852
|
456,456
|
409,900
|
||||||||
| Gross Profit (Loss) | (22,872) | 38,935 | 162,793 | 41,455 | ||||||||
| Total Operating Expense |
2,231,586
|
1,574,039
|
7,112,685
|
3,165,256
|
||||||||
| Loss From Operations | $ | (2,254,458) | $ | (1,535,104) | $ | (6,949,892) | $ | (3,123,801) | ||||
| Net Loss | $ | (6,463,358) | $ | (3,876,390) | $ | (10,295,661) | $ | (3,558,096) | ||||
| Net Loss Available to Common Shareholders per share: | ||||||||||||
| Basic and Diluted | $ | (0.03) | $ | (0.04) | $ | (0.08) | $ | (0.06) | ||||
3
Our Principal Executive Offices Our principal executive offices are located at 468 N. Camden Drive, Suite 200, Beverly Hills, California 90210.
Our telephone number is (310) 476-7002 and our website address is www.mmrglobal.com. Information
included or referred to on our website is not a part of this prospectus. Market Data and Industry Information We obtained the market data and industry information contained in this prospectus from internal surveys,
estimates, reports and studies, as appropriate, as well as from market research, publicly available information and industry
publications. Although we believe our internal surveys, estimates, reports, studies and market research, as well as industry
publications are reliable, we have not independently verified such information, and as such, we do not make any
representation as to its accuracy. Summary of the Offering This prospectus relates to the resale of up to 60,000,000 shares of our common stock by Dutchess
(representing 28.47% of
The Investment Agreement is further described below under the heading, "Investment Agreement".
|
Shares of common stock offered by us |
None. |
|
|
|
|
Shares outstanding before the Offering |
210,752,146 |
|
Shares of common stock offered by the Selling Stockholder |
(i) 230,800 shares were issued to Dutchess as a document preparation fee for the Equity Line and are outstanding upon the effective date of the registration statement to which this prospectus relates; and (ii) 60,000,000 shares are issuable to Dutchess pursuant to the terms of the Investment Agreement. |
|
|
|
|
Shares Outstanding following the Offering: |
270,752,146 |
4
|
Offering Price |
To be determined by the prevailing market price for the shares at the time of the sale or in negotiated transactions. |
|
|
|
|
Use of proceeds |
We will not receive any proceeds from the sale of shares by the selling stockholder. However, we will receive proceeds from the Equity Line. See "Use of Proceeds." We intend to use such proceeds for working capital, reduction of indebtedness, acquisitions and other general corporate purposes. |
|
|
|
|
Risk Factors |
An investment in our common stock is speculative and involves substantial risks. You should read the "Risk Factors" section of this prospectus for a discussion of certain factors to consider carefully before deciding to invest in shares of our common stock. |
|
|
|
|
Plan of Distribution |
The shares of common stock covered by this prospectus may be sold by the selling stockholder in the manner described under "Plan of Distribution." |
|
|
|
|
OTC Bulletin Board Symbol |
"MMRF.OB" |
Investment Agreement
We entered into the Investment Agreement with Dutchess on September 15, 2009. We subsequently amended the Investment Agreement on May 7, 2010. Pursuant to the Investment Agreement, Dutchess committed to purchase up to $10,000,000 of our common stock, over the course of 60 months. The aggregate number of shares issuable by us and purchasable by Dutchess under the Investment Agreement is 100,000,000.
We may draw on the facility from time to time, as and when we determine appropriate in accordance with the terms and conditions of the Investment Agreement. The maximum amount that we are entitled to put in any one notice is the greater of (i) 200% of the average daily volume (U.S. market only) of the common stock for the three (3) trading days prior to the date of delivery of the applicable put notice, multiplied by the average of the closing prices for such trading days or (ii) $150,000. The purchase price shall be set at ninety-four percent (94%) of the average of two lowest daily VWAP of our common stock during the Pricing Period. There are put restrictions applied on days between the put notice date and the closing date with respect to that particular put. During such time, we are not entitled to deliver another put notice.
There are circumstances under which we will not be entitled to put shares to Dutchess, including the following:
- we will not be entitled to put shares to Dutchess unless there is an effective registration statement under the Securities Act to cover the resale of the shares by Dutchess;
- we will not be entitled to put shares to Dutchess unless our common stock continues to be quoted on the OTC Bulletin Board, or becomes listed on a national securities exchange;
- we will not be entitled to put shares to Dutchess to the extent that such shares would cause Dutchess'beneficial ownership to exceed 4.99% of our outstanding shares; and
- we will not be entitled to put shares to Dutchess prior to the closing date of the preceding put.
There is no guarantee that we will be able to meet the foregoing conditions or any other conditions under the Investment Agreement or that we will be able to draw down any portion of the amounts available under the Investment Agreement.
5
The Registration Statement covers only 60,000,000 shares of common stock under the Investment Agreement. In order to meet the full allotment of 100,000,000 shares authorized under the Investment Agreement, the Company's common stock would have an assumed VWAP of $0.10 less the 6% discount. However, based on current prices, we have decided to limit ourselves to 53,191,489 shares available, or $10,000,000, based on an assumed VWAP of $0.20 (which is equal to recent market prices). If our share price rises, we will not be able to draw down in excess of the maximum of $10 million. If our share price falls, and we decide to issue more than 60,000,000 shares, we will need to file one or more additional registration statements with the SEC covering those additional shares.
The issuance of our shares of common stock under the Investment Agreement will have no effect on the rights or privileges of existing holders of common stock except that the economic and voting interests of each stockholder will be diluted as a result of the issuance of our shares. Although the number of shares of common stock that stockholders presently own will not decrease, these shares will represent a smaller percentage of our total shares that will be outstanding after any issuances of shares of common stock to Dutchess. If we draw down amounts under the Investment Agreement when our share price is decreasing, we will need to issue more shares to raise the same amount than if our stock price was higher. Such issuances will have a dilutive effect and may further decrease our stock price. An example of the effect of issuing shares when our stock price is comparatively low is set forth below. The net tangible book value per share before and after the distribution will be $(0.043) and $0.005, respectively. The amount of the increase in such net tangible book value per share attributable to the cash payments made by Dutchess will be $0.05. The amount of the immediate dilution from the public offering price which will be absorbed by such purchasers will be $(0.001).
Under the Investment Agreement, the purchase price of the shares to be sold to Dutchess will be at a discount of 6% from the average of the two lowest VWAP of our common stock during the Pricing Period . The table below illustrates an issuance of shares of common stock to Dutchess under the Investment Agreement for a hypothetical draw down amount of $50,000 at an assumed VWAP of $0.20, which is equal to the closing price of our common stock on the OTC Bulletin Board on June 14, 2010.
| Draw Down | Price to be Paid by | Number of Shares | ||||||
|
Amount
|
VWAP
|
% Discount
|
Dutchess
|
to be Issued
|
||||
| $50,000 | $0.20 | 6% | $ 0.188 | 265,957 |
By comparison, if the VWAP of our common stock was lower than $0.20, the number of shares that we would be required to issue in order to have the same draw down amount of $50,000 would be larger, as shown by the following table:
| Draw Down | Price to be Paid by | Number of Shares | ||||||
|
Amount
|
VWAP
|
% Discount
|
Dutchess
|
to be Issued
|
||||
| $50,000 | $0.15 | 6% | $ 0.141 | 354,610 |
Accordingly, the difference of the second example outlined above from the first example outlined above, would be dilution of an additional 88,653 shares issued due to the lower stock price. In effect, a lower price per share of our common stock means a higher number of shares to be issued to Dutchess, which equates to greater dilution of existing stockholders. The effect of this dilution may, in turn, cause the price of our common stock to decrease further, both because of the downward pressure on the stock price that would be caused by a large number of sales of our shares into the public market by Dutchess, and because our existing stockholders may disagree with a decision to sell shares to Dutchess at a time when our stock price is low, and may in response decide to sell additional numbers of shares, further decreasing our stock price.
We are limited in the maximum number of shares that we may be required to obtain funds from the Investment Agreement to 100,000,000 shares. The actual number of shares which may be sold under the Investment Agreement is dependent upon our trading volume and share price, which varies from day to day. If we issue and sell all shares under the Investment Agreement, our stockholders could experience significant downward pressure on the price of our common stock. The following table shows the effect on the number of shares required for an Advance for the value of the full $10 million Investment Agreement, in the event the common stock price declines by 20%, 40% and 60% from the trading price.
6
|
Price Decreases By
|
||||||||
|
20%
|
40%
|
60%
|
||||||
| VWAP during the Pricing Period (as defined above) | $0.20 | $0.16 | $0.12 | $0.08 | ||||
| Purchase Price (defined above as 94% of the VWAP) | $0.188 | $0.150 | $0.113 | $0.075 | ||||
| Number Subject to the Investment Agreement if 100% | ||||||||
| of the Investment Agreement is Executed. | 53,191,489 | 66,489,362 | 88,652,482 | 100,000,000 | ||||
| (maximum | ||||||||
| allowed | ||||||||
| under | ||||||||
| Investment | ||||||||
| Agreement) | ||||||||
At a VWAP of approximately $0.1064, we would exceed the maximum number of shares allowed to be sold under the Investment Agreement.
We entered into the Investment Agreement with the intention to grow our business, which in turn should increase our value. Because of the nature of the Investment Agreement, it appears unlikely that we will be able to draw down the full $10 million without significant positive value being added as a result of the aggregate draw downs. In addition, as reflected above, if our share price declines significantly and we still desire to draw down on the Investment Agreement, we will have to file one or more additional registration statements.
The Investment Agreement further provides that the Company and Dutchess are each entitled to customary indemnification from the other for any losses or liabilities we or it suffers as a result of any breach by the other of any provisions of the Investment Agreement or our registration rights agreement with Dutchess, or as a result of any lawsuit brought by a third-party arising out of or resulting from the other party's execution, delivery, performance or enforcement of the Investment Agreement or the registration rights agreement.
The Investment Agreement also contains representations and warranties of each of the parties. The assertions embodied in those representations and warranties were made for purposes of the Investment Agreement and are subject to qualifications and limitations agreed to by the parties in connection with negotiating the terms of the Investment Agreement. In addition, certain representations and warranties were made as of a specific date, may be subject to a contractual standard of materiality different from what a stockholder or investor might view as material, or may have been used for purposes of allocating risk between the respective parties rather than establishing matters as facts.
Dutchess further agreed that during the term of the Investment Agreement, neither Dutchess nor any of its affiliates, nor any entity managed or controlled by it, will, or cause or assist any person to, enter into or execute any short sale of any shares of our common stock as defined in Regulation SHO promulgated under the Exchange Act.
In connection with the preparation of the Investment Agreement and the registration rights agreement, we paid Dutchess a document preparation fee in the amount of $5,000, and issued Dutchess 230,800 shares of common stock, which equaled $30,000 worth of common stock based on the closing sales price of our common stock on August 13, 2009, the execution date of the non-binding term sheet with Dutchess.
7
Registration Rights Agreement
Pursuant to the terms of a Registration Rights Agreement, dated September 15, 2009, between Dutchess and us, we are obligated to file one or more registration statements with the SEC to register the resale by Dutchess of shares of common stock issued or issuable under the Investment Agreement. We are obligated to file with the SEC an initial registration statement on Form S-1 of which this prospectus forms a part by October 15, 2009, covering the resale of a portion of the 100,000,000 shares of common stock equal to one-third (1/3) of our current public float (where "public float" shall be derived by subtracting the number of shares of common stock held by our officers, directors and "affiliates" (as such term is defined in Rule 144(a)(1) of the 1933 Act) from the total number of shares of our common stock then outstanding). Pursuant to comments made by the SEC on November 9, 2009, we were required to withdraw our initial registration statement and renegotiate certain terms of the Investment Agreement. Following the successful renegotiation of such terms, we were obligated to file a new registration statement on Form S-1 as soon as practicable thereafter. We are obligated to use all commercially reasonable efforts to have the initial registration statement declared effective by the SEC within 180 days after the closing date. After the later of (i) sixty (60) days after the time that Dutchess shall have resold substantially all of the shares registered for resale under the initial registration statement, or (ii) six (6) months after the effective date of the initial registration statement, we are obligated to register for resale another portion of the 100,000,000 equal to one-third (1/3) of our then outstanding public float. This registration process will continue until such time as all 100,000,000 shares of common stock issuable under the Investment Agreement have been registered for resale on effective registration statements. In no event will we be obligated to register for resale more than 100,000,000 shares of common stock.
8
RISK FACTORS
Your investment in our common stock involves a high degree of risk. You should consider the risks described below and the other information contained in this prospectus carefully before deciding to invest in our common stock. If any of the following risks actually occur, our business, financial condition and operating results could be harmed. As a result, the trading price of our common stock could decline, and you could lose a part or all of your investment.
Risks Related to Our Business
Risks Related to Our Business
It is anticipated that we will continue to incur losses and negative cash flow from operations for the foreseeable future and may not be able to continue as a going concern.
We are still a company with a limited operating history although we have generated minimal revenue. While our new business includes personal health care record and storage products that are currently available, we are nevertheless still at an early stage in developing a business model that will enable us to generate significant revenue from the use of our products. Prior to the Merger, MMR financed its operations primarily through private placements of MMR capital stock and from secured loans from The RHL Group, Inc., a wholly-owned affiliate of MMR's founder and Chief Executive Officer, and our current Chairman, President and Chief Executive Officer, Robert H. Lorsch. Although we expect to continue to receive financing from The RHL Group, we have also continued to incur losses from operations and need additional sources of financing to fund our operations until we develop a profitable business. Even with additional funds from The RHL Group, there is no assurance that we will be able to generate sufficient revenue and working capital to fund our operations and create a sustainable going concern. As a result, it is expected that we will continue to incur operating losses for the foreseeable future.
If we fail to obtain additional financing, we will be unable to fund our operations.
We expect that the cash used in our operations will increase for the next several years. As of December 31, 2009, the Company's current liabilities exceeded its current assets by $7.2 million. Furthermore, during the year ended December 31, 2009, the Company incurred losses of $10.3 million. At the current level of borrowing, the Company requires cash of $275,000 per year to service its debt. Furthermore, not including debt service, in order to continue operating its business, the Company uses an average of $225,000 cash per month, or $2.7 million per year. In addition to the above cash burn from operations, the Company will be required to obtain additional financing in order to meet the obligations for installment payments of $621,000 under the Creditor Plan and our obligations under the subordinated secured indebtedness to The RHL Group (which note payable had a balance of $1,545,767 at December 31, 2009), among other debt obligations. Such obligations are currently due and payable pursuant to the terms of the note. At this rate of cash burn, the Company's existing current assets will sustain the business for approximately 5 months.
Although we generate some cash from our operations, we will need additional financing in order to fund operations until we can become cash flow positive, which is not expected to occur in the foreseeable future. For a description and copy of the Creditor Plan see Item 7 "Management's Discussion and Analysis of Financial Condition and Results of Operations" below and Exhibit 10.5.
Other than to meet the Creditor Plan obligations, our future funding requirements will depend on many factors, including:
- The pace of market adoption of current and future products;
- The length of sales cycles and implementation efforts for major corporate accounts;
- The launch of new products; and
- The build up of a sales and service delivery organization.
If additional debt financing is raised in the future, we may be required to grant lenders a security interest in all or a portion of our assets and issue warrants to acquire our equity securities, resulting in dilution to our stockholders. In addition, any such debt financing may involve restrictive covenants, including limitations on our ability to incur additional debt, limitations on our ability to acquire or assign intellectual property rights and other operating restrictions that could adversely impact our ability to conduct our business. Further, our ability to raise funding through the sale of equity securities will be significantly limited by our existing authorized but unissued common stock. Following the Merger, we have a limited amount of available common stock and any changes to our certificate of incorporation to increase our authorized share capital would require a vote of our stockholders, which could be time consuming and costly to obtain.
9
Future additional funding may not be available on acceptable terms, or at all. If we are unable to raise additional capital when required or on acceptable terms, then we may have to significantly delay, scale back or discontinue the development or commercialization of one or more of our products.
We have been, and may continue to be, unable to generate sufficient cash flow to service our debt obligations.
Our ability to make payments on our indebtedness and to fund our operations, working capital and capital expenditures, depends on our ability to generate cash in the future, which is subject to our ability to execute on our business plan, and also to general economic, industry, financial, competitive, operating, and other factors that are beyond our control.
MMR has a secured credit facility with The RHL Group, with all outstanding amounts due thereunder being guaranteed by us. On August 18, 2009, we entered into a Waiver Agreement with The RHL Group, pursuant to which The RHL Group waived MMR's continuing payment default under the credit facility until August 31, 2009, which waiver period will continue automatically until The RHL Group notifies MMR otherwise. Should The RHL Group notify us that the waiver period has been terminated and, as of that time, we continue to be in payment default under the credit facility, The RHL Group would have the right to call any outstanding debt immediately due and payable, as well as exercise any other rights it has as a secured creditor under the credit facility and applicable law. As of December 31, 2009, the aggregate principal amount owed under The RHL Group credit facility was approximately $1,545,767.
As of December 31, 2009, we owed an aggregate of approximately $1,381,377 in principal amount to certain of our former employees and creditors, as evidenced by promissory notes issued to the same in connection with the Merger. We are obligated to make 18 monthly payments to such employees and creditors beginning August 2, 2009. We have not yet made any of the payments due and payable to such employees and creditors, and thus they are entitled to exercise such rights as may be available to them under applicable law.
We cannot assure you that our business will generate sufficient cash flow from operations or that future borrowings will be available to us in an amount sufficient to enable us to pay amounts due on our existing indebtedness or to fund our other liquidity needs. Thus, we may need to refinance all or a portion of our indebtedness on or before maturity. Our ability to refinance our indebtedness or obtain additional financing will depend on, among other things:
- our financial condition at the time;
- restrictions in our outstanding debt instruments; and
- other factors, including the condition of the financial markets.
As a result, we may not be able to refinance any of our indebtedness on commercially reasonable terms, or at all. If we do not generate sufficient cash flow from operations, and additional borrowings or refinancings or other proceeds are not available to us, we may not have sufficient cash to enable us to meet all of our obligations, thereby potentially rendering us insolvent.
The level of our indebtedness could adversely affect our financial condition.
We have significant debt service obligations. As of December 31, 2009, the aggregate principal amount owed under all of our outstanding debt was approximately $2.7 million. The level of our indebtedness could have important consequences. For example, it could:
- increase our vulnerability to adverse economic and industry conditions;
- require us to dedicate a substantial portion of our cash flow from operations to the payment of our indebtedness, thereby reducing the availability of cash to fund working capital and capital expenditures and for other general corporate purposes;
- restrict us from making strategic acquisitions, acquiring new content or exploring other business opportunities;
- limit our ability to obtain financing for working capital, capital expenditures, general corporate purposes or acquisitions;
- place us at a disadvantage compared to our competitors that have less indebtedness; and
- limit our flexibility in planning for, or reacting to, changes in our business and industry.
10
We are a company with limited operating history and currently have only a small management team and staff, which could limit our ability to effectively seize market opportunities and grow our business.
Our operations are subject to all of the risks inherent in a growing business enterprise, including the likelihood of operating losses. As a smaller company with a limited operating history, our success will depend, among other factors, upon how we manage the problems, expenses, difficulties, complications and delays frequently encountered in connection with the growth of a new business, products and channels of distribution, and current and future development, as well as in the competitive emerging healthcare records management business. In addition, as a company with a limited operating history we have only a small management team and staff to grow our business and manage the risks inherent in a growing business enterprise. These factors could limit our ability to effectively seize market opportunities and grow our business.
Any significant limitation or failure of our technology systems that are critical to our operations could constrain our operations.
We are highly dependent upon the use of third-party technology systems to operate our business. Any failures in these systems could disrupt our business and subject us to losses. Moreover, although we have in place certain disaster recovery plans and backup servers, we may experience system delays and interruptions as a result of natural disasters, power failures, acts of war, and third-party failures. Potential system failures and the cost necessary to address them could result in material financial loss or costs, regulatory actions, breach of client contracts, reputational harm or legal claims and liability, which in turn could negatively impact our business prospects, results of operations and financial condition.
We may not be able to maximize our legacy biotechnology assets or protect our proprietary rights, which may hurt our competitive position and future revenues.
We will only be able to protect our proprietary rights from unauthorized use by third parties to the extent that these rights are covered by valid and enforceable patents or are effectively maintained as trade secrets and are otherwise protectable under applicable law. We will attempt to protect our proprietary position by filing and maintaining U.S. and foreign patent applications related to our proprietary products, technology, inventions and improvements that are important to the development of our business.
The patent positions of biotechnology and biopharmaceutical companies involve complex legal and factual questions and, therefore, enforceability cannot be predicted with certainty. Patents, if issued, may be challenged, invalidated or circumvented. Thus, any patents that we own or license from third parties for future products may not provide any protection against competitors. Pending patent applications we may file in the future, or those we may license from third parties, may not result in patents being issued. Also, patent rights may not provide us with proprietary protection or competitive advantages against competitors with similar technology. Furthermore, others may independently develop similar technologies or duplicate any technology that we have developed or we will develop. The laws of some foreign countries may not protect our intellectual property rights to the same extent as do the laws of the United States.
In addition to patents for future and current products, we own a substantial portfolio of assets acquired from Favrille, including data, samples, and other intellectual property rights. We currently seek protection for these assets, in part, through confidentiality and proprietary information agreements. These agreements may not provide meaningful protection or adequate remedies for proprietary technology in the event of unauthorized use, transfer or disclosure of confidential and proprietary information. The parties may not comply with or may breach these agreements. Furthermore, our trade secrets may otherwise become known to, or be independently developed by, competitors.
Our success will further depend, in part, on our ability to operate without infringing the proprietary rights of others. If our activities infringe on patents owned by others, we could incur substantial costs in defending ourselves in suits brought against a licensor or us. Should our products or technologies be found to infringe on patents issued to third parties, the manufacture, use and sale of our products could be enjoined, and we could be required to pay substantial damages. In addition, we, in connection with the development and use of our products and technologies, may be required to obtain licenses to patents or other proprietary rights of third parties, which may not be made available on terms acceptable to us.
11
We are highly dependent on our Chief Executive Officer and the loss of him could have a material adverse effect on our business and results of operations. Further, we may not be able to attract qualified officers to replace him or other key management personnel necessary to grow our business.
We are highly reliant on the services of our Chief Executive Officer, Robert H. Lorsch. If Mr. Lorsch left, it could have a material adverse effect on our business and results of operations. Further, we must continue to hire experienced managers to continue to grow our business. As a company with limited operating history, we may have difficulty attracting and retaining new individuals. If we are not successful in attracting management, it could have a material adverse effect on our ability to grow our business, which would adversely affect our results of operations and financial condition.
Our management will continue to be required to devote substantial time to comply with public company regulations.
As a public company, we will incur significant legal, accounting and other expenses. The Sarbanes-Oxley Act of 2002, as well as rules implemented by the SEC, impose various requirements on public companies, even those that are not listed. In light of the Merger and change in our management and business, our new management and other personnel will need to devote a substantial amount of time to these requirements, but do not have recent experience with these requirements. Moreover, these rules and regulations will increase our legal and financial compliance costs and will make some activities more time- consuming and costly.
The Sarbanes-Oxley Act of 2002 requires, among other things, that we maintain effective internal controls for financial reporting and disclosure controls and procedures. In particular, we must perform system and process evaluation and testing of our internal control over financial reporting to allow management and our independent registered public accounting firm to report on the effectiveness of our internal control over financial reporting when required by Section 404 of the Sarbanes-Oxley Act of 2002. Our compliance with Section 404 will require that we incur substantial accounting and related expense and expend significant management efforts. We will need to hire additional accounting and financial staff to satisfy the ongoing requirements of Section 404.
In addition to maintaining the remediations we have made in correcting our previously reported material weaknesses in our internal control over financial reporting, we need to bring our financial reporting procedures up to public-company standards so as to allow management to report on, and, when required by Section 404 of the Sarbanes-Oxley Act of 2002, our independent registered public accountants to attest to, our internal control over financial reporting. As a result, we will need to incur additional expenses and diversion of management's time in this area. We will be required to incur significant costs in documenting, evaluating, testing and remediating our internal control procedures and systems, including the ongoing engagement of our external consultants or additional financial and accounting personnel. Such actions, while necessary and beneficial, will adversely affect our financial results.
We face substantial competition.
While we believe that our MyMedicalRecords products occupies a unique niche in the marketplace, other companies offer similar services that compete for subscriber enrollment and health care professionals from the same patient and physician population, and could compete more directly with us by developing processes and technology functionally similar to those that form our MyMedicalRecords products. MMRPro competes with scanning services that market their services to doctors seeking to convert their historical paper records into electronic files, as well as Electronic Medical Record systems. MMRPro also competes with EMR systems that offer doctors the opportunity to make their entire office paperless. Most of these competitors are larger, more deeply funded companies. To the extent they have more success in obtaining market share and customer acceptance of their products, it could have a negative effect on our ability to grow our business, which could have a material adverse effect on our results of operations and financial condition.
Our growth will depend on our ability to develop our brand and any failure to do so could limit our business prospects, which could have a material adverse effect on our results of operations and financial condition.
We believe that establishing a strong brand will be critical to achieving widespread acceptance and adoption of our products and services. Promoting and positioning our brand will depend largely on the success of our marketing efforts, distribution channels and ability to provide high quality service. Establishing a significant brand presence for an on-line company often requires substantial marketing investment, and many "dot.com" companies have failed to generate the necessary adoption rates even after such a process. Brand promotion activities may not yield increased revenues, and even if they do, any increased revenues may not offset the expenses we incur in building the MyMedicalRecords brand. If we are not successful in building the MyMedicalRecords brand, it could limit our business prospects, which could have a material adverse effect on our results of operations and financial condition.
12
The handling of medical records is highly regulated. Not only could we be subject to substantial liability for mishandling records if we fail to comply with applicable requirements or if third-parties gain unauthorized access to records or servers but our products may need constant revisions or modifications to comply with an increasingly complex regulatory regime.
The proper handling of medical records is subject to extensive state and federal regulations and legal requirements, and we anticipate incurring significant costs to keep informed of and in compliance with such regulations and requirements. The volume and complexity of the regulations is daunting and many have changed substantially in recent years and all are subject to the uncertainties of interpretation. We recognize the critical nature of managing an individuals' health information requires that its products and advances be implemented with the utmost care to protect the privacy and confidentiality of its customers' data. The Health Insurance Portability and Accountability Act of 1996, commonly referred to as HIPAA, requires covered entities to protect the privacy and confidentiality of protected health information, or PHI, of their patients and customers. Although we are not a covered entity (as that term is defined in HIPAA), we consider it important to take into account the Privacy and Security Standards and other requirements of HIPAA when implementing its products and services and believes that it meets and/or exceeds current HIPAA standards.
The Health Information Technology for Economic and Clinical Health Act, commonly referred to as HITECH, enacted on February 17, 2009 as part of the American Recovery and Reinvestment Act, expanded HIPAA's reach beyond that of just covered entities. Now, business associates, defined as an entity that performs a function, activity, or service on behalf of a covered entity and that requires use or access the PHIs of the covered entities, and vendors of Personal Health Records that use or access PHIs must also comply with the Privacy and Security Standards of HIPAA. One of the key obligations under HITECH is the requirement to notify individuals when there has been (or there is a strong possibility of) a breach of the individual's PHI.
Further, our products and services may become subject to greater regulation, particularly at the federal level. We may have to reduce, enhance or remove certain offerings to comply with new regulatory standards. If we are not able to effectively modify our product and service offerings to adapt to stricter standards, it could have a material adverse effect on our ability to grow our business and our results of operations and financial condition.
Because of the types of agreements that we enter into with our customers for our products, there is a time lag between the date of the agreement or license and product launch and revenue generation, which may be significant.
In the ordinary course of our business, we enter into agreements with certain customers that typically provide for customization of our product. The degree of customization can vary from simple modifications required to co-brand to highly tailored product modifications, which require significant development efforts on our part. Because these customer arrangements typically do not provide for payment of revenues until product launch, there is often a time lag between the date we enter into an agreement or license with a particular customer and when we begin generating revenues. This time lag can be significant in some cases a year or longer especially when there is a high degree of customization or translation involved requiring extensive product development efforts. Thus, even if we are successful in negotiating agreements and licenses with many customers to co-brand or private label our products for such customers, because of the nature of our arrangements with our customers, there is a risk that it may be some time before we generate revenues from such arrangements, if at all.
Third parties claiming that we infringe their proprietary rights could cause us to incur significant legal expenses and prevent us from selling our products.
The software and Internet industries are characterized by the existence of a large number of patents, trademarks and copyrights and by frequent litigation based on allegations of patent infringement or other violations of intellectual property rights. In addition to our biotechnology assets, as we expand our product offerings into areas where larger companies with large patent portfolios compete, the possibility of an intellectual property claim against us grows. We could receive claims that we have infringed the intellectual property rights of others, including claims regarding patents, copyrights and trademarks. Any such claim, with or without merit, could result in costly litigation and distract management from day-to-day operations. If we are not successful in defending such claims, we could be required to stop selling our products, pay monetary amounts as damages, enter into royalty or licensing arrangements, or satisfy indemnification obligations that we have with some of our customers.
13
Our executive officers and directors may have interests that are different from, or in addition to, those of our stockholders generally.
Our executive officers and directors may have interests that are different from, or are in addition to, those of our stockholders generally. These interests include having subordinated debt with a balance of $1,545,767 at December 31, 2009 secured by a security interest in substantially all of our assets, the provision and continuation of indemnification and insurance arrangements for our directors, as well as certain other interests described elsewhere in this prospectus. Notably, our current President, Chairman and Chief Executive Officer, Robert H. Lorsch, may be deemed to beneficially control a total of 28.0% of our voting capital stock, as of March 20, 2010, including shares issuable upon exercise of options and warrants held by Mr. Lorsch and The RHL Group. Because of his high percentage of beneficial ownership, Mr. Lorsch may be able to control matters requiring the vote of stockholders, including the election of our board of directors and certain other significant corporate actions. This control could delay, defer or prevent others from initiating a potential merger, takeover or other change in our control, even if these actions would benefit our other stockholders and us. This control could adversely affect the voting and other rights of our stockholders and could depress the market price of our common stock.
Risks Related to Our Common Stock
Our stock price is expected to continue to be volatile, and the market price of our common stock may drop further.
The market price of our common stock could continue to be subject to significant fluctuations. Moreover, the stock markets in general have experienced substantial volatility that has often been unrelated to the operating performance of individual companies. These broad market fluctuations may also adversely affect the trading price of our common stock. In the past, following periods of volatility in the market price of a company's securities, stockholders have often instituted class action securities litigation against those companies. Such litigation, if instituted, could result in substantial costs and diversion of management attention and resources, which could significantly harm our operating results, financial condition, profitability and/or reputation.
We do not expect to pay cash dividends, and accordingly, stockholders must rely on stock appreciation for any return on their investment.
We anticipate that we will retain our earnings, if any, for future growth and therefore do not anticipate paying cash dividends in the future. As a result, only appreciation of the price of our common stock will provide a return to stockholders. Investors seeking cash dividends should not invest in our common stock.
Provisions in our amended and restated certificate of incorporation and amended and restated bylaws and applicable Delaware law may prevent or discourage third parties or our stockholders from attempting to replace our management or influencing significant decisions.
Provisions in our amended and restated certificate of incorporation and amended and restated bylaws may have the effect of delaying or preventing a change in control of our company or our management, even if doing so would be beneficial to our stockholders. These provisions include:
- dividing our Board of Directors into three classes serving staggered three-year terms;
- authorizing our Board of Directors to issue preferred stock without stockholder approval;
- prohibiting cumulative voting in the election of directors;
- prohibiting stockholder actions by written consent;
- limiting the persons who may call special meetings of stockholders;
- prohibiting our stockholders from making certain changes to our certificate of incorporation or bylaws except with 66.7% stockholder approval; and
- requiring advance notice for raising business matters or nominating directors at stockholders' meetings.
We are also subject to provisions of the Delaware corporation law that, in general, prohibit any business combination with a beneficial owner of 15% or more of our common stock for three years unless the holder's acquisition of our stock was approved in advance by our Board of Directors. Together, these charter and statutory provisions could make the removal of management more difficult and may discourage transactions that otherwise could involve payment of a premium over prevailing market prices for our common stock.
There is currently a limited trading market for our common stock, which may limit our stockholders' ability to sell shares of our stock.
Our common stock is currently quoted on the over-the-counter bulletin board, or OTC:BB. Because of the limited trading volume on the OTC:BB of our common stock and the penny stock regulations described below, our investors may not be able to sell their shares due to the absence of a trading market.
14
We may be subject to penny stock regulations and restrictions, which could make it difficult for stockholders to sell their shares of our stock.
SEC regulations generally define "penny stocks" as equity securities that have a market price of less than $5.00 per share or an exercise price of less than $5.00 per share, subject to certain exemptions. If we do not fall within any exemptions from the "penny stock" definition, we will be subject to Rule 15g-9 under the Exchange Act, which regulations are commonly referred to as the "Penny Stock Rules." The Penny Stock Rules impose additional sales practice requirements on broker-dealers prior to selling penny stocks, which may make it burdensome to conduct transactions in our shares. If our shares are subject to the Penny Stock Rules, it may be difficult to sell shares of our stock, and because it may be difficult to find quotations for shares of our stock, it may be impossible to accurately price an investment in our shares. In addition to the Penny Stock Rules, we are unable to utilize the safe harbor provisions of the Forward Looking Statements sections of the Exchange Act. There can be no assurance that our common stock will qualify for an exemption from the Penny Stock Rules, or that if an exemption currently exists, that we will continue to qualify for such exemption. In any event, we are subject to Section 15(b)(6) of the Exchange Act, which gives the SEC the authority to restrict any person from participating in a distribution of a penny stock if the SEC determines that such a restriction would be in the public interest.
The Financial Industry Regulatory Authority, or FINRA, sales practice requirements may also limit a stockholder's ability to buy and sell our stock.
In addition to the Penny Stock Rules described above, FINRA has adopted rules that require that in recommending an investment to a customer, a broker-dealer must have reasonable grounds for believing that the investment is suitable for that customer. Prior to recommending speculative low-priced securities to their non-institutional customers, broker-dealers must make reasonable efforts to obtain information about the customer's financial status, tax status, investment objectives and other information. Under interpretations of these rules, FINRA believes that there is a high probability that speculative low priced securities will not be suitable for at least some customers. FINRA requirements make it more difficult for broker-dealers to recommend that their customers buy our common stock, which may limit your ability to buy and sell our stock and have an adverse effect on the market for our shares.
A significant amount of our issued and outstanding shares of common stock are restricted securities and may not be freely resold to the public. When the restriction on any or all of these shares is lifted, and the shares are sold in the open market, the price of our common stock could be adversely affected.
A significant amount of our issued and outstanding shares of common stock are "restricted securities" as defined under Rule 144 promulgated under the Securities Act of 1933, as amended, and may only be sold pursuant to an effective registration statement or an exemption from registration, if available. Although Rule 144 may not be immediately available to permit resales of such shares, once available, and given the number of shares that would no longer be restricted, sales of shares by our shareholders, whether pursuant to Rule 144 or otherwise, could have an immediate negative effect upon the price of our common stock.
When we issue additional shares in the future, it will likely result in the dilution of our existing stockholders.
Our certificate of incorporation authorizes the issuance of up to 650,000,000 shares of common stock with a $0.001 par value and 5,000,000 preferred shares with a par value of $0.001. As of May 4, 2010, 210,752,146 common shares were issued and outstanding and no shares of preferred stock were issued and outstanding. If we issue any additional shares, such issuance will cause a reduction in the proportionate ownership and voting power of all current stockholders. Further, such issuance may result in a change of control of our corporation.
Moreover, in the past, we issued warrants and options to acquire shares of common stock. As of May 4, 2010, we had warrants, options and convertible notes to purchase an aggregate of 92,849,638 shares of our common stock. In addition, the issuance of any shares for acquisition, licensing or financing efforts, upon conversion of any preferred stock or exercise of warrants and options, pursuant to our equity compensation plans, or otherwise may result in a reduction of the book value and market price of the outstanding shares of our common stock.
Risks Related to this Offering
We are registering the resale of a maximum of 60,000,000 shares of common stock which may be issued to Dutchess under the Equity Line. The resale of such shares by Dutchess could depress the market price of our common stock.
We are registering the resale of a maximum of 60,000,000 shares of common stock under the registration statement of which this prospectus forms a part. The sale of these shares into the public market by Dutchess could depress the market price of our common stock. As of May 4, 2010, there were 210,752,146 shares of our common stock issued and outstanding. In total, we may issue up to 100,000,000 shares to Dutchess pursuant to the Equity Line, meaning that we are obligated to file one or more registration statements covering the 40,000,000 shares not covered by the registration statement of which this prospectus forms a part. The sale of those additional shares into the public market by Dutchess could further depress the market price of our common stock.
15
Existing stockholders could experience substantial dilution upon the issuance of common stock pursuant to the Equity Line.
This prospectus relates to the resale of up to 60,000,000 shares of our common stock by Dutchess. As of May 4, 2010, we had 210,752,146 shares outstanding. The sale by Dutchess of all shares pursuant to this prospectus would result in a substantial dilution to our stockholders' current ownership.
Furthermore, our Equity Line with Dutchess contemplates our issuance of up to 100,000,000 shares of our common stock to Dutchess, subject to certain restrictions and obligations. If the terms and conditions of the Equity Line are satisfied, and we choose to exercise our put rights to the fullest extent permitted and sell 100,000,000 shares of our common stock to Dutchess, our existing stockholders' ownership will be further diluted by such sales.
Dutchess will pay less than the then-prevailing market price for our common stock under the Equity Line.
The common stock to be issued to Dutchess pursuant to the Investment Agreement will be purchased at a 6% discount to the average of two lowest daily VWAP of our common stock during the five consecutive trading day period beginning on the trading day immediately following the date of delivery of a put notice by us to Dutchess, subject to certain exceptions. Therefore, Dutchess has a financial incentive to sell our common stock upon receiving the shares to realize the profit equal to the difference between the discounted price and the market price. If Dutchess sells the shares, the price of our common stock could decrease.
We may not be able to access sufficient funds under the Equity Line when needed.
Our ability to put shares to Dutchess and obtain funds under the Equity Line is limited by the terms and conditions in the Investment Agreement, including restrictions on when we may exercise our put rights, restrictions on the amount we may put to Dutchess at any one time, which is determined in part by the trading volume of our common stock, and a limitation on our ability to put shares to Dutchess to the extent that it would cause Dutchess to beneficial own more than 4.99% of our outstanding shares. In addition, we do not expect the Equity Line to satisfy all of our funding needs, even if we are able and choose to take full advantage of the Equity Line.
We will not receive any proceeds from the resale of our common stock offered by Dutchess. However, we will receive proceeds from the sale of our common stock to Dutchess pursuant to the Investment Agreement. The proceeds from our exercise of the put option pursuant to the Investment Agreement will be used to support the commercialization of our current and future product candidates, for general working capital needs, for the reduction of indebtedness and for other purposes that our board of directors, in its good faith, deems to be in our best interest.
All net proceeds from the sale of the common stock covered by this prospectus will go to the selling stockholder. See "Selling Stockholder" and "Plan of Distribution" described below.
The information provided in the table and discussions below has been obtained from the selling stockholder. The table below identifies the selling stockholder and shows the number of shares of common stock beneficially owned by it before and after this offering, and the numbers of shares offered for resale by the selling stockholder. Our registration of these shares does not necessarily mean that the selling stockholder will sell all or any of their shares of common stock. However, the "Shares Beneficially Owned After Offering" columns in the table assume that all shares covered by this prospectus will be sold by the selling stockholder and that no additional shares of common stock will be bought or sold by the selling stockholder. No estimate can be given as to the number of shares that will be held by the selling stockholder after completion of this offering because the selling stockholder may offer some or all of the shares and, to our knowledge, there are currently no agreements, arrangements or understanding with respect to the sale of any of the shares. In addition, the selling stockholder may have sold, transferred or otherwise disposed of, or may sell, transfer or otherwise dispose of, at any time or from time to time since the date on which it provided the information regarding the shares, all or a portion of the shares of common stock beneficially owned in transactions exempt from the registration requirements of the Securities Act.
16
The following table sets forth the name of the selling stockholder, and if applicable, the nature of any position, office, or other material relationship which the selling stockholder has had, within the past three years, with us or with any of our predecessors or affiliates, the amount of shares of our common stock beneficially owned by the stockholder prior to the offering, the amount being offered for the stockholder's account, the amount being offered for the stockholder's account and the amount to be owned by such stockholder after completion of the offering.
|
|
|
Shares Beneficially Owned |
|
|
|
Shares Beneficially Owned |
||||
|
Beneficial Owner |
|
Shares |
|
% |
|
Shares Being |
|
Shares |
|
% |
|
|
|
|
|
|
|
|
|
|
|
|
|
Dutchess Equity Fund, L.P.(2) |
|
230,800 |
|
* |
|
60,000,000 |
(3) |
230,800 |
|
- |
_____________
* Connotes less than 1.0%.
(1) Beneficial ownership is determined in accordance with Rule 13d-3(d) promulgated by the Securities and Exchange Commission under the Exchange Act, and generally includes voting or investment power with respect to securities. The number and percentage of shares beneficially owned is determined in accordance with Rule 13d-3 of the Exchange Act and is not necessarily indicative of beneficial ownership for any other purpose. Applicable percentage ownership is based on 210,752,146 shares of common stock outstanding as of May 4, 2010. Except as otherwise noted, we believe that the stockholder named in the table has sole voting and investment power with respect to all shares of common stock shown as beneficially owned by it, subject to applicable community property laws.
(2) Dutchess is a Delaware limited partnership. Michael Novielli and Douglas H. Leighton are directors of Dutchess with voting and investment power over the shares.
(3) Represents (i) a portion of the 100,000,000 shares of common stock issuable by us and purchasable by Dutchess under the Investment Agreement equal to one-third (1/3) of our public float as of May 4, 2010. As of that date, our public float was equal to 125,070,597 shares (where "public float" was derived by subtracting the number of shares of common stock held by our officers, directors and "affiliates" (as such term is defined in Rule 144(a)(1) of the 1933 Act) from the total number of shares of our common stock outstanding as of May 4, 2010), and (ii) 230,800 shares of our common stock that were issued to Dutchess as a document preparation fee for the Equity Line and are outstanding upon the effective date of the registration statement to which this prospectus relates.
17
MARKET FOR COMMON EQUITY AND RELATED STOCKHOLDER MATTERS
Market Information
Our common stock traded on The Nasdaq Global Market under the symbol "FVRL" from our initial public offering through the time of our delisting on October 3, 2008. On October 3, 2008, our common stock began trading over-the-counter on the OTC Bulletin Board under the symbol "FVRL.OB". Following the Merger and our name change from Favrille, Inc. to MMR Information Systems, Inc., our common stock began trading on the OTC Bulletin Board under the symbol "MMRF.OB". On June 15, 2010 we changed our name to MMRGlobal, Inc. and have applied with the SEC and FINRA for a symbol change to MMRG.
Holders
As of June 9, 2010, there were approximately 3,536 holders of record of our common stock.
Price Range
The market for our common stock is limited and volatile. The following table sets forth the range of high and low high and low closing prices for our common stock for each of the periods indicated as reported by the OTC Bulletin Board. The prices quoted on the OTC Bulletin Board reflect inter-dealer prices, without retail mark-up, mark-down or commissions. The OTC Bulletin Board prices listed below may not represent actual transaction prices.
|
|
High |
|
Low |
|
|
|
Fiscal year ending December 31, 2010: |
|
|
|
|
|
|
Second Quarter |
|
$0.32 |
|
$0.13 |
|
|
First Quarter |
|
$0.17 |
|
$0.08 |
|
|
Fiscal year ended December 31, 2009: |
|
|
|
|
|
|
Fourth Quarter |
|
$0.15 |
|
$0.07 |
|
|
Third Quarter |
|
$0.15 |
|
$0.08 |
|
|
Second Quarter |
|
$0.35 |
|
$0.10 |
|
|
First Quarter |
|
$0.30 |
|
$0.05 |
|
|
Fiscal year ended December 31, 2008: |
|
|
|
|
|
|
Fourth Quarter |
|
$0.05 |
|
$0.02 |
|
|
Third Quarter |
|
$0.07 |
|
$0.02 |
|
|
Second Quarter |
|
$1.87 |
|
$0.05 |
|
|
First Quarter |
|
$1.97 |
|
$1.24 |
|
Dividends
We have never declared or paid any cash dividends on our capital stock. We currently intend to retain all of our future earnings, if any, to finance the growth and development of our business. We do not intend to pay cash dividends on our common stock for the foreseeable future. Any future determination related to dividend policy will be made at the discretion of our Board of Directors.
18
Securities Authorized for Issuance Under Equity Compensation Plans
The following table sets forth certain information with respect to our equity compensation plans as of December 31, 2009.
|
Plan category
|
Number of securities to be issued upon exercise of outstanding options, warrants and rights (a) |
Weighted-average exercise price of outstanding options, warrants and rights (b) |
Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a)) (c) |
||||
| Equity compensation plans | 3,571,469 | $ 0.11 | 23,428,531 | (1) | |||
| approved by security holders | |||||||
| Equity compensation plans not | |||||||
| approved by security holders |
48,504,024
|
$ 0.72 |
-
|
||||
| Total |
52,075,493
|
$ 0.68
|
23,428,531
|
____________
(1) Includes a total of 27 million shares of our common stock reserved for issuance under our 2001 Equity Incentive Plan.
Organizational History
We were incorporated in Delaware in 2000 under the name "Favrille, Inc." and previously operated as a biopharmaceutical company focused on the development and commercialization of targeted immunotherapies for the treatment of cancer and other diseases of the immune system. In May 2008, our ongoing Phase 3 registration trial for our lead product candidate failed to show a statistically significant improvement in the treatment of patients with follicular B-cell non-Hodgkin's lymphoma, and accordingly, we determined to sell all of our equipment and other personal property in an auction. On September 9, 2008, this auction was consummated and we received $3.2 million in net proceeds from the sale of the assets. With the disposition of all of our equipment and other personal property, we ceased to engage in any operations and became a "shell company" as such term is defined in Rule 12b-2 of the Exchange Act.
Agreement and Plan of Merger and Reorganization
On January 27, 2009, we consummated a reverse subsidiary merger with what was then called MyMedicalRecords.com, Inc., a Delaware corporation, or MMRCI, pursuant to which the holders of MMRCI equity prior to the merger, on a fully diluted basis, owned or had the right to acquire approximately 60.3% of our equity, the holders of our equity prior to the merger, on a fully diluted basis, owned approximately 33.2% of our equity, and certain beneficiaries under a creditor plan (which beneficiaries consist of our former officers and former directors and their affiliates) had the right to own up to approximately 6.5% of our equity. We refer to this business combination as the "Merger" from time to time in this prospectus. In connection with the Merger, we changed our name from "Favrille, Inc." to "MMR Information Systems, Inc.", MMRCI changed its name to "MyMedicalRecords, Inc." and became our wholly owned subsidiary, and we ceased being a "shell company" as such term is defined in Rule 12b-2 of the Exchange Act. We refer to MyMedicalRecords, Inc. as "MMR" from time to time in this prospectus. MMR was incorporated in Delaware in 2005 and is headquartered in Los Angeles, California.
19
The Merger is being accounted for as a reverse acquisition in accordance with U.S. generally accepted accounting principles. Under this method of accounting, we are treated as the "acquired" company for financial reporting purposes. This determination is primarily based on the fact that MMR's former shareholders hold a majority of the equity of the consolidated company, MMR's operations comprise the ongoing operations of the consolidated entity and MMR's senior management and director designees have assumed control of the consolidated company.
Our Company and Business Subsequent to the Merger
As of the closing date of the Merger on January 27, 2009, we adopted MMR's business of empowering consumers to manage the important records in their life, whether paper-based or digital, and by doing so, to better control and organize their lives overall. The following description of our business relates to our current business and operations.
Business Overview
We offer a suite of secure, online products that empower consumers and professionals alike to manage the important records in their life and business, whether paper-based or digital. We are part of a trend where people are increasingly trusting on-line services as a way for them to store and manage critical, sensitive information such as medical records, financial records and vital documents. A series of informative videos on our products can be found at
www.mmrvideos.com.We also continue efforts to maximize the current and future value, if any, of our intellectual property that we acquired from Favrille when the Merger was completed in January 2009. These assets include, but are not limited to, patents, data and samples from the Company's pre-merger clinical vaccine trials, the Specifid vaccine, and the anti-CD20 antibodies.
Products and Services
Our suite of secure, web-based products all are built on proprietary, patent-pending technologies that allows users, including consumers, affinity groups, membership organizations, small businesses, physicians and health care professionals to easily store and organize information.
Our consumer product, "MyMedicalRecords", is an easy-to-use, secure web-based Personal Health Record system, or PHR, which allows documents, images and voice mail messages to be transmitted in and out of our system using a variety of methods, including fax, voice and file upload. Our platform converts documents it receives by fax into Adobe's Portable Digital Format, or PDF, file. These files, as well as voice files and any other uploaded files are stored in the consumer's personal account, which is secured by a unique user ID and password combination. A notification is sent to up to three user e-mail addresses (including to e-mail enabled cell and smartphones) whenever a new record is received. Users can then access their files via the Internet and take advantage of an intuitive, customized filing system that allows them to categorize, annotate and file their records for easy access, organization and retrieval. Users can then print, download, e-mail or fax their records from their account, giving our customers greater control over their own personal health records and other information, which they can share with health care providers and others as they move through the continuum of care. We are currently selling our MyMedicalRecords PHR product primarily through corporations as an employee benefit and to affinity groups as a "value-added" service for their members or clients, as well as directly to retail customers. When sold to employers and/or affinity groups the Company counts members as individuals who have paid access to the MMR system through their employers benefit program or as a member benefit from a respective affinity group. The Company counts users as the individuals in that member group that activate and commence usage of their individual PHR account. We plan to sell through additional sales channels including insurance and financial industries in the future. In addition, the MyMedicalRecords PHR is an important component of the MMRPro system which we are selling to doctor's offices and other healthcare professionals (See section on "MMRPro").
Our "MyESafeDepositBox" service is geared towards small businesses, the financial services, insurance and legal service industries. MyESafeDepositBox is based on the same technology and architecture as our MyMedicalRecords PHR product. However, rather than focusing on storing medical records, MyESafeDepositBox is designed to provide secure on-line storage for medical records and vital financial, legal and insurance documents including wills and advanced directives. MyESafeDepositBox.com may be used as a virtual on-line "safe" and could serve as an essential part of any household's or business's disaster preparedness plan.
20
In September 2009, we introduced our MMRPro product which provides physician offices with a powerful and cost-effective solution to the costly and time-consuming problem of digitizing paper-based medical records, as well as providing doctors the ability to access to those records through a private portal at MyMedicalRecordsMD.com. MMRPro also features an integrated e-Prescribe automated drug order entry system. It also automatically deploys records that patients can view through a free patient portal, MMRPatientView.com. MMRPro also includes a "stimulus program" that allows doctors to earn administrative reimbursements when their patients upgrade from the free MMRPatientView.com portal to a full-featured, paid MyMedicalRecords PHR.
We recognize the critical nature of managing an individuals' health information requires that our products and advances be implemented with the utmost care to protect the privacy and confidentiality of our customers' data. The Health Insurance Portability and Accountability Act of 1996, commonly referred to as HIPAA, requires covered entities to protect the privacy and confidentiality of protected health information, or PHI, of their patients and customers. Although we are not a covered entity (as that term is defined in HIPAA), we consider it important to take into account the Privacy and Security Standards and other requirements of HIPAA when implementing our products and services and believe that we meet and/or exceed current HIPAA standards.
The Health Information Technology for Economic and Clinical Health Act, commonly referred to as HITECH, enacted on February 17, 2009 as part of the American Recovery and Reinvestment Act, expanded HIPAA's reach beyond that of just covered entities. Now, business associates, defined as an entity that performs a function, activity, or service on behalf of a covered entity and that requires use or access the PHIs of the covered entities, and vendors of Personal Health Records that use or access PHIs, must also comply with the Privacy and Security Standards of HIPAA. One of the key obligations under HITECH is the requirement to notify individuals when there has been (or there is a strong possibility of) a breach of the individual's PHI.
While we only rarely function as a business associate to a covered entity, we continue to meet and/or exceed the current HIPAA standards. Further, as a vendor of PHRs, we are implementing policies and procedures and reviewing our relationships with all necessary parties to ensure our compliance with HITECH and its associated regulations.
We plan to continue to take advantage of the burgeoning consumer health information market and leverage recent federal legislation, including the HITECH Act. We believe that the health care reform legislation recently passed by Congress and signed by the President into law represents a significant behavioral shift in how consumers manage their health care because of the requirement that everyone have insurance. We believe this new law represents a "tipping point" in health care that benefits MMRIS because we provide easy-to-use, secure products that offer tools to help people better manage their health information. Health care reform also has significant implications for doctors and we expect that as more doctors look for cost-efficient ways to digitize their patient records, MMRPro will gain continued awareness and acceptance among the medical provider community. We also believe that as medical costs and insurance costs increase, and benefits decrease, healthcare consumers will require greater control over their personal (and their family's) health and health information. The Company recorded significant percentage growth in site traffic during 2009; this is likely attributable to the increased interest in Personal Health Records and, given the far-reaching effects of health care reform, the Company expects visitor growth to increase throughout 2010.
Our MyMedicalRecords PHR product is designed to enable consumers to store their important medical records and data in one central and secure place where they can manage those records and control how they are accessed and shared. While every healthcare consumer in the U.S., as well as other countries that are focused on health information technologies are potential users of our MyMedicalRecords PHR product, we believe that the product has most appeal to these particular market niches:
- the chronically ill and their families who share information among many doctors; this "co-morbid population" represents a disproportionate share of U.S. health care costs;
- individuals with Health Savings Accounts who need to carefully manage their eligible expenses over the course of a year;
- consumers with "senior" parents who, in their role as caregivers, want to be sure they have current medical information readily available to react quickly in case of parental illness;
- consumers with newborns who will be able to build a complete medical file for the newborn, which can last the newborn's entire life;
- employees who are forced to change doctors and other providers when their employer changes health insurance and who therefore need to manage the transfer to medical information to their new providers;
21
- travelers and businesspeople working overseas who want to ensure that they always have access to their medical records in the event of an emergency; and
- corporations seeking to offer their employees a valuable benefit (we believe those with expatriate employees would be particularly interested in our product).
Because of the similarity in functionality between our MyMedicalRecords PHR product and our MyESafeDepositBox product, we market these products through many of the same channels. Our marketing strategy for our MyMedicalRecords PHR product and MyESafeDepositBox product focuses primarily on the following channels:
- to corporate accounts, as an added employee benefit for their employees or bundled or co-branded with their product offerings to their customers;
- to insurance companies who are looking for a valuable benefit to provide risk and accident policyholders both in the United States and abroad;
- through affinity groups (such as alumni organizations and other membership organizations) and discount health benefit membership groups to their members; and
- direct to consumers as a retail subscription product.
Our MMRPro product is an integrated, end-to-end service that gives doctors, particularly solo and small group practitioners, an easy and inexpensive way to scan and digitize patient medical files, manage those files through a virtual patient chart and give patients up-to-date access to those records. MMRPro is being marketed to:
- Doctors, particularly small group and solo practitioners who still do not have any way to digitize the paper in their offices and who do not want to invest the hundreds of thousands of dollars necessary to implement an Electronic Medical Record, or EMR, system;
- Community hospitals and other clinics which do not have the funds or technology resources to invest in an EMR system;
- EMR vendors who are looking for a way to bring Adobe's PDF images into their systems without having to build their own import modules
The Company is an Independent Software Vendor of Kodak, which manufactures the scanner used in the MMRPro system. As a result, MMR can take advantage of and utilize the resources of a national Kodak reseller and distribution channel to sell this product into doctor offices nationwide.
Our Products
MyMedicalRecords - An On-line Personal Health Record (PHR)
Our MyMedicalRecords PHR product, principally accessible at www.mymedicalrecords.com, delivers an easy-to-use, web-based medical records and health information storage, retrieval and management system for consumers. Our MyMedicalRecords PHR product allows for paper-based medical records such as lab reports, radiology reports, MRI reports and progress notes to be transmitted via fax to a secure mailbox that is created when a user enrolls in the service. Our MyMedicalRecords PHR product is designed to allow a user to fax his or her health records or other vital documents into a personal MyMedicalRecords PHR account via a dedicated telephone number, which we refer to as the user's "Lifeline." This nique telephone number is assigned to the user upon enrollment. A user can also upload digital files, such as x-rays, scans or other medical images, as well as other vital documents into his or her account directly from a personal computer through any Internet browser. In addition, the MyMedicalRecords PHR accepts voice messages. Our MyMedicalRecords PHR product allows users to store and segregate information for up to ten family members in a single account.
Healthcare providers can transmit documents to a patient's MyMedicalRecords PHR mailbox without making any changes to existing patient or practice management software and takes advantage of the one piece of equipment found in virtually every doctor's office - a fax machine.
In addition to medical records, our MyMedicalRecords PHR product can be used to provide secure storage for a variety of important confidential records, including:
- patient charts and notes;
- patient medical histories;
22
- vaccination and immunization records;
- lab reports and test results, including any images (or, if offered by a laboratory, links that the user can use to access the laboratory's stored images);
- copies of prescription orders;
- x-ray results and images (either the actual images or, if offered by a radiology provider, links that the user can use to access the provider's stored images); and
- EKG results and images (either the actual images or, if offered by an imaging provider, links that the user can use to access the provider's stored images); and
- birth certificates, living wills, healthcare powers of attorney and advance directives.
In addition, users can store important legal, insurance and financial documents, as well as copies of identification documents such as passports and driver licenses in their MyMedicalRecords PHR accounts.
How it Works
Most providers, regardless of size, have a fax machine in the office. We have based our service on the efficient and effective conversion of faxed documents into PDF files, and the subsequent annotation and filing of these faxes using a simple, intuitive, on-line document management system.
- Upon enrollment, we send every user a Welcome Kit containing a set of stickers to be placed on their medical files at doctors' offices, as well as a sticker for the back of a driver license, both containing emergency access information.
- The user gives their doctor(s) a sticker to be applied on the face of their medical file. The sticker (see below) contains a standing instruction to the doctor's office to fax to the user's personal Lifeline number any documents being placed in the medical file.
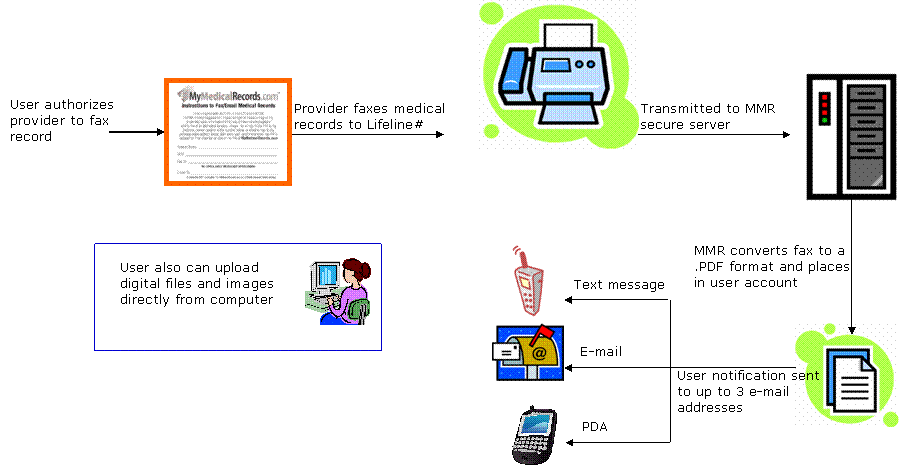
- When our MyMedicalRecords PHR system receives a document by fax transmission, it converts it into a PDF file and, using secure File Transfer Protocol transmission, or FTP, deposits it in the user's account on a secure server.
- When a faxed record or voice mail is received via the user's personal Lifeline number, our MyMedicalRecords PHR system sends a notification message to up to three e-mail addresses (including to e-mail enabled cell phones) provided by the user to inform the user that a new document or voice message has arrived in his or her account.
23
- The user then accesses the web site, www.mymedicalrecords.com, from any Internet-connected computer, and logs in using their unique ID (Lifeline number or a User ID that they create) and password. Users are able to view their faxed records using Adobe's PDF software, which can be downloaded from the Internet free of charge.
- Our MyMedicalRecords PHR product allows users to store, organize and sort records by doctor, date, type of medical record, and, in the event multiple family members have enrolled in the service, by family member. Users also can annotate their filed records with free-form notes and then use a search feature to find any record containing a specific word or phrase. We believe this capability to easily index, file, sort and retrieve medical records adds to the usability of our MyMedicalRecords PHR product.
- The user can also upload images, such as x-rays or EKG results directly into his or her MyMedicalRecords PHR account and can view these images using an Internet browser or other graphics software. Health information stored in the user's MyMedicalRecords PHR account can also be printed, downloaded and e-mailed from the user's e-mail so the user can easily take it to any provider who may be involved in the treatment of his or her medical condition.
- Users also have the ability to fax records out of their MyMedicalRecords PHR account using an integrated Internet fax service. This feature gives users the ability to easily share information with multiple providers with the privacy of web-based faxing that doesn't require them to print out documents and then place them in a physical fax machine. This feature also transforms the PHR into an integrated fax messaging service, which the Company believes provides a significant competitive advantage over other PHRs.
We sell our MyMedicalRecords PHR product direct to consumers on a monthly and annual subscription basis. Current retail pricing for our MyMedicalRecords PHR product for a family that permits storing information for up to 10 people is $99.95 for an annual subscription or $9.95 for a monthly subscription. We also occasionally offer free trials and discounted pricing as a way to incentivize consumer acceptance of our MyMedicalRecords PHR product.
For special key account programs, such as healthcare providers that would like to make the service available to patients, corporations who want to offer the service as a benefit to employees, or affinity groups and other organizations who want to offer the service to their members, we provide different access-based pricing plans. The pricing plans vary based on the number of people in the organization and the expected use of the product across the organization's members.
Additional Product Features
Our MyMedicalRecords PHR product offers users multiple ways to enter their personal medical information, including by fax, voice or digital file upload. We believe this capability makes our MyMedicalRecords PHR product easier to use than other products in the marketplace and makes our product's information storage features more accessible to potential customers. In addition to the core document conversion, storage and retrieval capabilities, our MyMedicalRecords PHR product provides a layer of useful, value-added interactive tools to help users better manage their personal and/or their families' medical records. These tools include:
- Ability to Attach Received Faxes to e-mail Notifications. Users can elect to have records attached to the notification e-mail they receive when a new medical record is received into their account. This capability is intended to save users the extra step of logging into their account to view new records, which we believe creates a higher level of convenience and usability.
- Health History. Users can enter their personal health history, including information about doctors (such as a doctor's name, address and specialty), vaccinations and immunizations, hospitalizations/surgeries, allergies and other medical conditions that may affect ongoing healthcare.
- Appointment Calendar Feature. Users are able to take advantage of a calendar feature to schedule and generate reminders about upcoming doctor and other health-related appointments. These reminders appear when a user logs into his or her MyMedicalRecords PHR account and also can be sent to the user's e-mail address (or e-mail enabled cell phone) and imported into Microsoft Outlook.
- Prescription History and Refill Reminder Feature. Users are able to enter their prescriptions, pharmacies and refill dates into their MyMedicalRecords PHR accounts. The system generates reminders about refills, which are visible to users when they log into their accounts and are sent to their e-mail addresses (or e-mail enabled cell phone).
24
- Drug Information and Interaction Database. Users can check for potential interactions across multiple prescriptions and over the counter drugs with this comprehensive database, licensed from Multum. When each user adds a new drug to their MMR PHR prescription history, they can quickly determine if that medication has any kind of negative interaction with other prescriptions they take. This tool is especially vital because drug interactions kill as many as 100,000 Americans each year and consumers who see multiple providers are especially at risk.
- Voice Reminders and Messaging. We believe our MyMedicalRecords PHR product may create more efficient communication between doctors and patients. In addition to using a patient's personal MyMedicalRecords PHR telephone number to fax health information to a patient's secure on-line account, people can use the telephone number to leave a voice message, such as an appointment reminder, in a secure voice mailbox that is only accessible by the MyMedicalRecords PHR user. Users can also take advantage of this feature to leave themselves a reminder message such as for a doctor appointment or prescription refill reminder. Our MyMedicalRecords PHR product is designed to send a user a notification via e-mail when he or she receives a voice message. This gives users a helpful tool that they can access remotely.
- Secondary Passwords on Selected File Folders. Users can assign a second password to four of the file folders in their MyMedicalRecords PHR account. This feature creates an additional layer of security for personal vital or medical documents that a MyMedicalRecords PHR user does not want a doctor to have access to in the event that the user has given the doctor access to the account, or if the user does not want other family members to be able to see selected information.
- Emergency Log-In For Physicians and Other Emergency Response Personnel. Users can create a special password, discretely associated with each family member, which doctors and other emergency response personnel can use to gain access to the particular family member's medical records in the event of a medical emergency. This password grants access to an account but does not allow any additions, changes or deletions to be made to the account. In addition, users can decide which records a doctor will be able to see in an emergency situation. Users can write this password on an emergency sticker they receive when they enroll in our MyMedicalRecords PHR service, which can be affixed to a driver's license or personal ID. Users can even include a photograph in their emergency profile.
- Health Information Library. Users have access to an interactive audio and visual encyclopedia, licensed from a third-party, of over 2,000 health information topics presented in both English and Spanish.
Other Applications for Our MyMedicalRecords PHR Product Technologies
We believe our MyMedicalRecords PHR product technology presents potential market opportunities beyond its core "personal health records" storage and management purpose. In the wake of recent hurricanes, fires and other disasters, a great deal of emphasis has been placed on families having a secure place to store their records and vital documents where they cannot be lost or damaged. Our MyMedicalRecords PHR product addresses this need because it allows for the fax transmission, upload and storage of insurance, financial and other personal documents, as well as a place to store digital files such as photographs. We believe this recent emphasis provides us with an opportunity to expand our core market and use our MyMedicalRecords PHR product technology to create the essential "safe deposit" box for all important documents and records of a family or small business. The MyESafeDeositBox virtual storage product extends the MyMedicalRecords technology into these additional markets.
MyESafeDepositBox - An On-line Secure Document Storage System
Our MyESafeDepositBox product is designed to meet the needs of companies and individuals who are looking for a simple, efficient and economical way to securely store important legal, insurance and financial documents that they cannot afford to lose in a natural disaster such as an earthquake, hurricane, flood or fire. We believe our MyESafeDepositBox product may also serve as a valuable tool for younger consumers who would not otherwise utilize a Personal Health Record storage system, but are looking for a secure way to organize their personal information to take better control over their financial affairs. Pricing for MyESafeDepositBox is similar to the MyMedicalRecords Personal Health Records. It is sold to consumers on a direct subscription basis for $9.95 per month or $99.95 annually, and is made available to corporations and membership groups on an access-fee basis.
Approximately 63% of our revenue over the last two years was generated by our MyMedicalRecords PHR and MyESafeDepositBox products.
25
MyMedicalRecords Pro - An Integrated Scanning and Web-Based Document Management Solution for Medical Providers
MMRPro was launched in late 2009. The product provides physician practices, particularly smaller practices that are still largely or entirely paper based and resistant to making the significant investment required to convert to an Electronic Medical Records, or EMR system, a powerful, integrated solution that enables them to scan, digitize, store, manage, retrieve and share records with patients through a managed "Software As Service" web application created and managed by the Company.
A typical EMR implementation costs well in excess of $100,000 and can take several weeks or months to integrate into a doctor's practice. Worse, during this implementation, a doctor's office is asked to significantly reduce their patient load by as much as 50% which means that the practice loses one-half of its revenue. In a health care economy where patient load is critical to a doctor's financial success, this can be very problematic. MMRPro is sold on a three-year license, which includes all hardware, software and system management; a typical four doctor practice would pay $600 per month or $21,600 over the life of the license.
How It Works
At the core of the MMRPro system is a Kodak Scan Station 520 duplex scanner, configured with a special version of Kodak's Capture Pro document imaging software. This state-of-the art scanner when integrated with the document imaging software is only available as part of the MMRPro system and is not sold as a stand-alone product.

When MMRPro is installed at a doctor practice, office staff creates a unique bar code sheet for each patient. The bar code cover sheet is placed in the patient's chart prior to the patient coming into the office. Each tab in the patient chart also is assigned a unique bar code so that the patient chart can be replicated online; these bar codes can be printed out using standard Avery labels. The labels can then be placed on patient medical records. At the end of an appointment, any paper medical records for the patients, such as progress notes, lab reports, radiology reports and previous medical history that have been reviewed and approved by the doctor, are scanned using the Scan Station 520 and then sorted, according to bar codes, into the correct tabs of a virtual patient chart that has been re-created online where they are securely stored. Doctors can annotate these scanned records with free-form text notes and search those notes later on making it easy to retrieve specific records.
Once documents have been scanned, doctors or their staff can access them from any Internet-connected computer through the MyMedicalRecordsMD doctor portal. In addition, when records are scanned the MMRPro system instantly deploys a copy of the record that the patient can view through a free patient portal, MMRPatientView.com. The Company believes that the ability to instantly share records with patients gives doctors
26
the ability to more efficiently manage and operate their practices because they and their staff are not continually being asked to retrieve patient records. MMRPro also allows for secure messaging between doctor's office and patients, as well as provides both inbound and outbound Internet fax capability, making it easier for a practice to share information with other health providers.
No revenue over the last two years has been generated by our MMRPro product.
Additional MMRPro Features
- Integrated e-Prescribe Module. MMRPro also includes an integrated e-Prescribe module that gives doctors the ability to do automated prescription ordering and refills. The e-Prescribe software gives doctors a choice of all formulations for a given medication, as well as available generic alternatives and shows what patient benefit coverage will be for the prescription. Any special instructions for the patient can also be entered into the module so they are automatically printed on the prescription label. The prescription order can be faxed or automatically transmitted to the pharmacy. In addition, the e-Prescribe module also makes it possible for doctors to keep a drug history on all their patients, as well as check for any interactions and allergies in real-time.
- Integrated Fax Messaging. MMRPro supports both inbound and outbound Internet fax. While fax continues to be the primary way doctors send and receive information such as lab reports, doctors are increasingly concerned about the privacy issues associated with a fax machine in an open area. MMRPro solves the problem by allowing faxes to be initiated from or delivered directly to admin or doctor computer desktops through its integrated fax messaging. Faxes, which are viewable as PDFs, can be assigned to different doctors in the practice and then filed as part of the patient record. Each practice receives one main fax number for the office, and then individual numbers can be assigned to each doctor in the practice.
- Voice messaging. MMRPro also accepts voice messages into its system so doctor can hear voice messages from labs or other providers and then make those part of the patient record.
- Practice-specific branding and content. MMRPro allows providers to create custom content for their practices that patients see when they access records through the MMR Patient View portal. For example, a practice could use this feature to include a message to remind patients to get flu shots or give a wellness tip.
- Secure messaging. With MMRPro, doctors, office staff and patients can exchange messages securely from within the application. Messaging makes it possible for patient questions to be answered, or appointments scheduled, without having to make phone calls.
- Automated Patient Reminder System. MMRPro features a patient calendaring system that automatically can send emails or even make telephone calls reminding patients of upcoming appointments. Freeing office staff from these tasks can greatly improve office efficiency.
The MMRPro "Stimulus Program"
In addition to its product functionality, MMRPro includes a "Stimulus Program" that creates cost-savings and revenue opportunity for its users; in fact, the Company believes its Stimulus Program can generate at least as much money for a doctor practice over a 3 year term as the $44,000 that is possible through the HITECH Act over a five year period.
The Company believes that MMRPro can ultimately eliminate the need for doctor office staff to go into patient charts to retrieve their records. The Company estimates that this can cost a doctor office as much as $50 per occurrence, when you take into account all of the labor and copying costs associated with retrieving a patient record. If MMRPro makes it possible for a doctor office to eliminate having to manually retrieve just one record per day, it would translate to a savings of more than $10,000 in a single year.
What's more, the e-Prescribe module entitles doctors who write prescriptions for Medicaid patients to receive an automatic 2% incentive for using automated order entry and eliminating paper.
In addition, doctors earn administrative reimbursements when their patients upgrade from the free MMRPatientView portal to a full-featured, paid MyMedicalRecords Personal Health Record. The Company shares 30% of the subscription revenue from those upgrades with doctors on a monthly basis and believes that patient acceptance for the MyMedicalRecords PHR will be driven by the benefit of being able to store and manage medical records from all doctors for up to 10 family members, not just the one doctor and one family member capability of the free MMR Patient View Portal.
27
The Market for Our Products
Demand for our medical records products is driven by the U.S. healthcare market and the health information management market. We believe demand for our MyESafeDepositBox product will be driven by consumer and small business focus on disaster preparedness and financial planning.
U.S. Healthcare Market
On March 23, 2010, President Obama signed into law a major overhaul of the United States health care and health insurance industries. As part of this overhaul, we anticipate that there will be an increased demand for the cost savings inherent in the use of electronic medical records, particularly the benefits to patients by having a personal health record. Also in February 17, 2009, President Obama signed into law the American Reinvestment and Recovery Act of 2009, or the ARRA, representing the largest government-driven investment in electronic healthcare technologies. Within ARRA, the Health Information Technology for Economic and Clinical Health Act, or HITECH, provisioned more than $19 billion in incentives to healthcare organizations that modernize their medical records systems through the widespread use of health information technology, or HIT. Throughout the process of healthcare reform, the challenge to rein in healthcare expenses will continue to be the nation's long-term fiscal challenge and this creates opportunities for HIT solutions that are shown to create greater efficiencies in care, safety and costs.
For example, despite the economic downturn, U.S. healthcare spending reached an estimated $2.5 trillion in 2009, an increase of 5.7 percent since 2008. The health share of the nation's gross domestic product, or GDP, reached 17.3 percent, a growth rate in 2009 of 1.1 percent, representing the largest one-year increase in GDP share since 1960. Expenditures in physician and clinical services reached $527.6 billion in 2009 and are expected to grow 6.1 percent to $637 billion by 2017. By 2019, national health spending is projected to nearly double, reaching $4.5 trillion and consuming 19.3 percent of the GDP. (Office of the Actuary, Centers for Medicare and Medicaid Services cited in Health Affairs, published 2/4/2010)
Thus, demand for solutions that help curb the spiraling costs of healthcare spending is expected to accelerate. In 2007, healthcare technology already represented 54 percent of all deals in the healthcare industry totaling $172.9 billion. More recently, a January 2009 study released by Kalorama Information, a leading provider of market research in medical and healthcare markets, projected the EMR market alone will grow 14.1 percent each year through 2012 with consumer demand for access to their personal health information, or PHI, forcing greater use of EMR technology. The researchers estimated that total implementation costs for the EMR rollouts are expected to be between $68 million to $255 billion. (Kalorama Information as cited in FierceHealthIT, 1/5/2009)
We believe the forces between the need to control healthcare spending and HIT mandates to help achieve this create positive market forces for the Company's MMRPro and MyMedicalRecords PHR solutions. Even with the uncertainties healthcare providers face in their day-to-day practice, we anticipate they will focus on spending strategic dollars on health IT as ROI. Moreover, given that 70 percent of small physician practices with four doctors or less still rely on paper-based systems to manage patient charts - and over 50 percent of physician practices in the U.S. consist of one to three doctors - there is tremendous opportunity for niche market share. We believe that MMR's strategic partnership with Kodak to distribute MMRPro as an end-to-end solution throughout Kodak's sales channels further positions the Company to penetrate small-to mid-size physician practices with a cost-effective entry-level EMR system that offers an e-Prescribe module and an integrated patient portal providing patients with timely access to their personal health information, both of which are meaningful use requirements to qualify for stimulus funds. Further value propositions for MMRPro in today's constantly evolving healthcare market is its SaaS platform that can work with any EMR system, its cost effectiveness and ease-of-use in light of the fact that doctors remain concerned about the cost of installing expensive, complex systems that interfere with office workflow, and the Company's response to ensure privacy protections for patients covered under HIPAA's Privacy and Security Rules within HITECH.
Additionally, one of the key challenges to cost containment in the healthcare system is treating those with chronic conditions. About 10 percent of the population accounts for 63 percent of spending on health services, and 21 percent of health spending is for only 1% of the population. (Department of Health and Human Services, AHRQ [MEPS] 2006). What is needed is a Personal Health Record that can be the main source of information between the primary care physician, specialists and the other health entities involved in the individual's care. With coordinated access to a patient's health information, there is continuity of care that can lead to cost reductions by eliminating unnecessary repeat tests and avoiding preventable medical injuries. According to the much cited Rand Corporation study published in 2005, in addition to the estimated $346 billion that could be saved annually through the use of health IT, the study projected that $82 billion to $100 billion in savings could be realized in coordination across the
28
continuum of care. We believe the MyMedicalRecords PHR - Internet-based for 24/7 access, personally-controlled, portable, affordable and easy-to-use - fulfills what surveys reveal consumers are most looking for in a PHR, which is privacy, access to information online, the ability to track immunizations and monitor lab reports, communicate with doctors, and correct mistakes in their records.
Given healthcare's prominence in the national debate and the role of Health IT in cost containment, America's changing demographics and the first wave of baby boomers about to enter Medicare, and the PHR requirement for meaningful use of an EHR, we believe providing a Personal Health Record to consumers will be a growing force in the healthcare system. On March 23, 2010, the President signed into law a major overhaul of the United States health care and health insurance industries. As part of this overhaul, we anticipate that there will be an increased demand for the cost savings inherent in the use of electronic medical records. We believe that we are effectively positioned to quickly take a role in meeting this demand. Furthermore, we expect that the specific use of electronic medical records will continue to be supported by additional Federal legislation and by private healthcare reform where the addition of tens of millions of covered Americans will require cost-effective health IT solutions to fiscally manage safer, coordinated healthcare.
Driver for MyESafeDepositBox Market
The primary market driver for our MyESafeDepositBox product is the need for individuals and businesses to be able to easily, efficiently and securely maintain backup copies of paper-based financial, insurance, legal and other vital business and personal documents.
Even though the market for external data storage got off to a slower start in 2009 because of the economy, it ended on a strong note according to International Data Corporation's, or IDC's, Worldwide Disk Storage Systems' market report for the fourth quarter of 2009. The IDC's Quarterly Tracker published on March 5, 2010 reported that worldwide disk storage capacity grew 33% in the fourth quarter of 2009 and disk storage revenue for the quarter was up 0.2% from the same period a year earlier, to $7.3 billion. At the same time, In-Stat reported on March 9, 2010 that with home networking adoption passing 50% of households in North America and 13% worldwide, the consumer network storage market is seeing continued growth and is expected to grow at a compound annual growth rate of nearly 40% between 2009 and 2014.
Both companies and individuals are continuing to seek solutions that will allow them to safely backup - and quickly restore - any lost data. We believe that our MyESafeDepositBox product meets the need of a sub-set of this vast data storage market, and we intend to target individuals and businesses that want to find a secure, Web-based solution for storing their most critical personal, financial, legal and insurance records, rather than those looking to back up the entire contents of their computer hard drive or corporate network.
In addition, the need for individuals to augment their personal plans for disaster preparedness is reflected in a Red Cross poll that showed seven of 10 respondents were only somewhat prepared for a disaster, with 59% to 73% having no specific evacuation plan. For businesses, the need to implement a disaster recovery plan can be the key to their survival. In the publication, "The Definitive Handbook for Business Management," cited by Hewlett-Packard, it states that between 60% and 90% of companies that don't have a disaster plan find themselves out of business within 24 months of experiencing a major disaster. As natural and man-made disasters continue to grow - from hurricanes, earthquakes, and floods to computer viruses and even terrorist attacks - a common denominator for recovery is access to information and documentation, and we believe our MyESafeDepositBox product meets this need by offering users a safe and easy way to store and access all of their important documents and vital records online.
Drivers for MMRPro Market
Doctors are under increasing pressure to make their offices "digital" as mandated by the Federal Government in order to qualify for the $44,000 in stimulus funds under the HITECH Act. Stimulus Funds from the HITECH Act are paid out over a five year period; for example, a doctor who qualifies for funds in 2010 will receive payments beginning in 2011 through 2015. Despite this, most doctors still are resistant to using Electronic Medical Record systems. In fact, a survey conducted by the New England Journal of Medicine in 2008 showed that A national survey of 2,758 physicians in 2008 found that fewer than one in five doctors have adopted electronic medical record systems in their office practices, with only 4% of doctor practices using full EMR systems and 83% using no EMR system at all. Similarly, a 2009 article in the on-line version of the Harvard Business Review showed that nearly 90% of doctor in practices with four doctors or less do not have Electronic Medical Record Systems. Part of the reason for this low adaptation rate is cost. A reported released by the Congressional Budget Office (CBO) in 2008 cites studies that place the cost of an Electronic Medical Records system at about $25,000 to $45,000 per physician. Annual cost estimates for operating and maintaining the system, which include software licensing fees, technical support, and updating and replacing used equipment, range between about 12 percent and 20 percent of
29
initial costs, or $3,000 to $9,000 per physician per year. So a three-doctor practice may spend as much as $162,000 in the first year to install and maintain a system. This doesn't include the "hidden" costs of lost revenue from having to reduce patient load and also delays in billing while a new system is being implemented.
In addition, the "Meaningful Use" standards that the government plans to use to determine if a doctor practice will receive those stimulus funds are potentially very onerous to doctors, asking them to provide detailed information on patients that includes factors like demographics, body mass index, growth charts and other specific data points.
As a result, the Company sees a significant niche opportunity to provide an efficient, cost-effective system that helps doctors take the required first step to any form of digital office - scanning and digitizing patient records. The Company believes that the simplicity and elegance of MMRPro makes it attractive to the many thousands of medical offices, community hospitals and other health care providers who are still paper-bound and searching for a way to start their digital conversion.
Customers
To date, we have signed contracts and agreements for both our MyMedicalRecords PHR product and our MyESafeDepositBox product in the healthcare, corporate employee benefits and affinity and membership group markets. We have also sold these products directly to retail consumers.
In the healthcare market, we have provided the patients of participating hospitals with the use of our MyMedicalRecords PHR product free of charge when they are discharged from inpatient facilities. We also offer a private label of our MyMedicalRecords PHR product for Medic-Alert, which offers it as an enhancement to their existing PHR service and we are continuing to expand that relationship. We also have corporate clients who provide our MyMedicalRecords PHR product to their employees as a free benefit, and affinity group and membership clients who bundle the product as a "value added" service to their constituents. We receive compensation under these agreements either on a per enrolled account basis or on an access basis in which we are paid based on the number of persons who are eligible to sign up for our service, regardless of the number that actually enrolls. We are also an integrated service provider on the Google Health Network, which allows users to synchronize information between their Google Health account and their MyMedicalRecords PHR account.
In the affinity and membership group area, we have seen double digit growth in the number of users who have access to the MyMedicalRecords Personal Health Record; the Company expects that this trend will continue into the future as these groups create new programs that bundle the MyMedicalRecords PHR and MyESafeDeposit virtual storage vault.
During 2009, our four largest customers (MMR Australia at $171,500, Alexian at $153,916, Medialert at $70,620 and Coverdell at $69,341) accounted for approximately 75% of our total revenue.
Sales & Marketing Strategies
MyMedicalRecords PHR
Our marketing strategy for our MyMedicalRecords PHR product calls for continued focus on four main sales channels:
- Corporate Sales - Employee Benefit Offering. We are pursuing the human resources and benefits market to secure agreements or strategic arrangements providing for companies to offer our product to their employees and members. We believe the user-friendly nature of our MyMedicalRecords PHR product makes it readily acceptable to employees and gives companies a low cost way to demonstrate their employee-friendliness.
- Affinity Groups and Membership Organizations. Our MyMedicalRecords PHR product can be bundled with other health or travel-related services. For example, a travel accident insurance company can include our MyMedicalRecords PHR product in its suite of emergency medical and repatriation services for travelers going abroad. We believe giving users the ability to access their medical records in an emergency situation overseas may add considerable value to an insurance company's travel insurance policies. Additionally, an affinity group, such as an alumni association, may offer our MyMedicalRecords PHR product as a recruitment or renewal tool. We plan on continuing to utilize outside sales representatives who specialize in selling services to these market segments.
- Private Label Branding. Our MyMedicalRecords PHR product is designed to allow site pages to be customized with a corporate, affinity group or membership organization logo and content that is specific to that entity. The technology built into our MyMedicalRecords PHR product also is designed to allow companies, groups or organization to instantly communicate with hundreds or even thousands of employees in the event of an announcement or emergency situation.
30
- Direct to Consumer Marketing. We intend to continue to focus on marketing our MyMedicalRecords PHR product directly to consumers via both on-line and off-line advertising vehicles. To fully monetize our agreement with eMail Frequency LLC, which was announced in September 2009, we are in the process of testing different email creative, both text and HTML, as well as different landing pages to determine the best formula for growing our subscriber base. Our MyMedicalRecords PHR product is currently available to individuals on a monthly or annual subscription basis.
MyESafeDepositBox
Because of the similarity in functionality between our MyESafeDepositBox product and our MyMedicalRecords PHR product, we market these products through many of the same channels. Both products are designed to offer users the ability to fax, upload and store important and private records or documents in a secure electronic environment, safe from fire or flood, and secure from the threat of identity theft. Thus, for example, we market our MyMedicalRecords PHR product to insurance companies as an additional benefit for health insurance policy holders, while at the same time marketing our MyESafeDepositBox product to insurance companies that may offer it to their risk and casualty policy customers. In addition, we market our MyESafeDepositBox to financial institutions and legal service providers as a safe and secure way for their customers to store important and private documents provided by these companies.
MyMedicalRecords Pro
The Company is an Independent Software Vendor for Eastman Kodak, which manufactures the scanner used in the MMRPro system. As a result, MMR can take advantage of and utilize the resources of the national Kodak reseller and distribution channel to sell this product into doctor offices nationwide. MMR already has attended the annual Kodak Executive Summit, which was held in San Antonio this past February, where we had the opportunity to introduce the product to a number of Kodak resellers who expressed great enthusiasm for using the MMRPro solution as a way for them to penetrate the rapidly growing document imaging healthcare vertical. In addition, the Company shared its booth at this past April's Health Information Management Systems Society (HIMSS) show in Atlanta with Kodak personnel, providing further opportunity to create enthusiasm for MMRPro within the Kodak sales channel. Beyond selling the product, the Kodak channel also provides the Company with a national sales installation integration and support network, including help desk and technical support capabilities.
The Company is currently conducting sales training for resellers either in-person or via webinars in order to ensure they have the necessary tools to sufficiently take the MMRPro service into the healthcare vertical.
The Company also plans to use a direct sales force to sell the MMRPro service and has increased its internal sales staff to support the MMRPro rollout. In addition to selling to healthcare providers, the Company believes there is an opportunity to sell the product to current EMR providers who are looking to piggyback on MMRPro because it offers a way for them to import PDF images into their system.
Clinical Research Sales Strategy
Through the Company's strategic relationship with GRS Global, a full-service clinical trials management organization, the Company is designing customized personal and electronic health record products designed to follow patients through clinical trials. The Company believes that such a product offering can reduce the cost of managing clinical trial results and generate long term users of the Company's product. The Company will target biotech companies and pharmaceutical manufactures to purchase this product to obtain better monitoring of patient results and patient involvement.
Additional On-Going Marketing Strategies
In addition to the three main marketing channels discussed above, we have also identified other potential markets for increasing sales of our products:
- Small Businesses. We believe our products could serve as an affordable and easy-to-implement tool for small companies that have not, or do not want to, invest in expensive IT data backup infrastructure.
- Government Agencies. We believe that federal, state and local governments would be interested in our products as they provide a powerful, yet easy to use addition to any family's disaster preparedness plan and we are interested in approaching agencies at all levels to explore the possibility of setting up pilot programs.
31
- Travel Companies and the Travel Industry. We believe that travelers represent a strong market for our products because these products are designed to enable travelers to access to their medical records, or other important documents (copies of passports, credit cards, etc.), in the event of an emergency when they are away from home. We plan to market our products to airlines, hotels, automobile clubs and other-travel related companies and organizations as well as to advertise on travel-related websites to reach this market.
International Licensing
We exploit our intellectual property, technology and our MyMedicalRecords brand internationally. In November 2009, the Company announced its intention to enter into a definitive agreement with Unis-Tonghe Technology (Zhengzhou) Co., Ltd., or UNIS, to form a joint venture to build a customized version of MMR's proprietary PHR services (www.mymedicalrecords.com) and professional document imaging and management solutions in China.
UNIS is a subsidiary of Unisplendour Corporation Limited (SHE: 00938) (www.unis.cn), one of China's leading IT firms. MMR's technology will support a UNIS medical records development project for sale to China's public and private hospitals. China has an initiative to create health information systems to electronically acquire, store and transport individual health records.
Our senior management and technology executives met with counterparts from UNIS-Tonge and Nihilent, MMR's technology partner in India, together for a collaborative session on requirements to integrate the MyMedicalRecords Personal Health Record and MMRPro system (www.mymedicalrecordsmd.com) into a health IT platform that could be deployed throughout China's healthcare market.
In February 2006, the Company entered into a letter agreement with MyMedicalRecords Australia ("MMR AU") granting it a 10 year exclusive right to market and sell the Company's products and services in Australia and New Zealand. This letter agreement was subsequently formalized in the form of a ten year license agreement in October 2007. The Company terminated the license agreement on September 30, 2009.
On January 4, 2010, we entered into a Cooperation Agreement and related documents (the "Cooperation Agreement") with Unis-TongHe Technology (Zhengzhou) Co., Ltd. ("Unis"). Under the Cooperation Agreement, we and Unis agreed to form a joint venture in China for the purpose of deploying our personal health record services and document imaging and management solutions in China. We will own 40% of the joint venture and Unis will own 60% and each party will have the right to designate two members of the joint venture's board of directors, with the fifth member being a Chinese citizen mutually designated by us and Unis. Under the Cooperation Agreement, board actions will require the approval of more than three of the five members of the joint venture's board of directors and no material actions may be taken unless all board members are present and voting at the meeting.
Under the Cooperation Agreement, we will contribute an aggregate of 100 million RMB to the joint venture, based on each party's respective ownership, in the form of intellectual property rights, equipment, brand value, cash and such other consideration as may be agreed to between the parties. Each party's obligation to contribute to the joint venture is subject to a number of conditions, including obtaining all necessary approvals of and licenses from the Chinese government, as well as the joint venture meeting its budget, goals and objectives at the time any contributions are due. Under the Cooperation Agreement, each party's contributions will be made over a period of sixty months.
Principal Suppliers and Supply Contracts
We currently contract with various third-party telecommunications, data center and information technology service providers and developers to develop and maintain our products.
We have a contract with an outside vendor to host one of our product websites and provide fax and voice messaging services and the toll-free numbers used by the Company's customers to access their accounts. We pay our outside vendor a monthly website hosting fee and additional usage fees for fax, voice messaging and toll-free number services. Our contract was effective until September 2009, but automatically renewed, and shall continue to renew, for successive one year periods under its current terms and conditions, unless terminated by either party by written notice no less than 90 days before expiration of the term.
32
We contract with an outside vendor to house, manage and maintain the production servers that host our MyMedicalRecords PHR and MyESafeDepositBox web applications and store user related data. We pay a monthly fee for the management and technical support services required to maintain and operate its servers, which are housed in two separate data centers. Our agreement with this vendor is effective until terminated by either party upon fifteen days prior written notice.
We contract with a third-party for the development and maintenance of the software applications necessary to run our MyMedicalRecords PHR, MyESafeDepositBox and MyMedicalRecords Pro products. Our outside developer supports our software development needs through a team of software engineers, programmers, quality control personnel and testers, who work with our internal product development team on all aspects of application development, design, integration and support of our products. Under our development contract, we own exclusive intellectual property rights over all software applications developed pursuant to the contract. We pay our developer a per project fee for the development services provided and a monthly fee for support and maintenance services. Our agreement with this developer was effective until June 31, 2009 at which time we extended the term of the agreement for another six months, and can automatically continue to extend the term of the agreement for additional six month terms.
On September 16, 2009, we entered into a Licensing and Sales Commission Agreement, or the Licensing Agreement with E-Mail Frequency, LLC, which we refer to as the Licensor, and David T. Loftus, which we refer to as the Consultant. The License Agreement was filed as Exhibit 10.1 to our Quarterly Report on Form 10-Q for the Quarter Ended September 30, 2009. Pursuant to the Licensing Agreement, we agreed to exclusively license from the Licensor the usage of the Licensor's direct marketing database of street addresses, cellular phone numbers, e-mail addresses and other comprehensive data, or the Database. In addition, we engaged the services of the Consultant to assist in the use of the Database. Under the terms of the Licensing Agreement, we paid a $250,000 one-time consulting fee to the Consultant in the form of 2,777,778 shares of restricted common stock, which we refer to as the Consulting Fee. In addition to the Consulting Fee, we will pay the Licensor a percentage of actual revenue received by us from successful sales made pursuant to use of the Database. The Licensing Agreement has a five-year term, but may be terminated by us after the Licensing Agreement has been in effect for one year after the effective date of the Licensing Agreement. Upon such termination, we will be obligated to pay the Licensor its fees owed under the Licensing Agreement for the remainder of the term in addition to eight times the total fees paid to the Licensor over the last three months of use of the Licensing Agreement.
In the event that we are unable to continue to obtain supply, development or maintenance services from any of our suppliers or developers, we believe that we would be able to choose among several other suppliers, developers, or maintenance services to continue the development of our products. In selecting our current supply, development and maintenance vendors with which we have agreements, we received competitive bids from other vendors who would have been able to provide equivalent services. Thus, we believe that the marketplace for such services is broad enough that we would be able to reach commercially reasonable terms for the continued development of our products within the termination periods provided in each agreement.
Disaster Recovery Plan
We have a disaster recovery plan in place that is designed to ensure the safekeeping of records stored in a user's MyMedicalRecords PHR, MyESafeDepositBox, MyMedicalRecordsMD and MMRPatientView accounts, while maintaining continuity of our services should our main server site be affected by a natural or man-made disaster. We have a standby disaster recovery site over 100 miles away from the facility housing our main production servers. We backup the database holding our customers' records to servers housed in this "hot" standby disaster recovery site so that both systems contain identical information on a near-real-time basis. In addition, our main production site has redundancy measures built-in at all levels of the infrastructure and is designed to facilitate maximum availability of our product websites.
Competition
MyMedicalRecords PHR
Our MyMedicalRecords PHR product competes with a number of products and service providers in the consumer health information management marketplace today such as HealthVault, iHealthRecord.org, Google Health, WebMD, and others. In addition, we compete with Internet-portals offered by insurance companies, hospitals and HMOs for their policyholders and patients.
33
Each of our competitors offers varying PHR products and services for on-line storage and access to medical records at varying price points; however, we believe our MyMedicalRecords PHR product offers unique features that distinguish it from those of our competitors. In particular, we believe our MyMedicalRecords PHR product offers greater ease of use and accuracy than our competitors' products because copies of the actual medical records, such as laboratory test results and radiology reports can be faxed or uploaded directly into the user's MyMedicalRecords PHR account, rather than requiring users to input the data themselves, which may result in transcription errors. Our MyMedicalRecords PHR is designed to allow a user to send a medical record into their MyMedicalRecords PHR account from any fax machine, anywhere in the world and also to store and manage those records in a personalized filing system with 16 file folders.
Competitors, including Health Vault and WebMD, offer limited document management capability for personal health records; they have to be scanned and uploaded by the user and they do not allow users to manage stored information with the same sophistication as the MMR PHR. In addition, these services do not offer integrated outbound fax directly from the user account, which means that users of competitive products have to print out and manually handle paper in order share information with other providers along the continuum of care.
In addition, while hospital patient, HMO patient and insurance policyholder Internet-portals allow users to see certain information regarding test results, prescriptions or claims data, and may even give patients the ability to set appointments and communicate with doctors, these portals only allow users to view data from that specific provider, and if a user changes his or her healthcare provider or insurance carrier, the information may not be available in the future. Our MyMedicalRecords PHR product is designed to offer our customers a single secure on-line depository for all of their health information and records, from every provider, so that this information is available any time a MyMedicalRecords PHR user needs to access it.
We also believe the enhanced features offered with our MyMedicalRecords PHR product, such as outbound fax, document management, emergency login, appointment and prescription reminders and voice messaging features, offer consumers unique and attractive advantages that separate our MyMedicalRecords PHR product from the competition. All of these competing services, especially free services like Google and HealthVault, raise consumer awareness about the need for access to personal health information. While this increased awareness may increase the marketability of our MyMedicalRecords PHR product, growth in the consumer health information management marketplace may also attract new entrants. While we believe that greater ease of use and certain enhanced features distinguish our MyMedicalRecords PHR product from those of our competitors, many of our competitors may have greater resources and more experience in this market, and can modify their product offerings to make them more competitive, including attempting to replicate some features of our MyMedicalRecords PHR product. We have also sought to protect our proprietary technology through patents in both the U.S. and overseas. See "- Intellectual Property - Patents" below.
MyESafeDepositBox
Our MyESafeDepositBox product competes with a number of on-line backup and electronic data storage services. The increasing use of external hard drives and flash drives to backup data also has the potential to compete with on-line data storage services such as our MyESafeDepositBox product. In addition, Wells Fargo Bank recently offered a VSafe service to its customers, which has many of the same features and functions as our MyESafeDepositBox product.
We believe that MyESafeDepositBox is a superior product when compared with Wells Fargo's VSafe in that we allow users to fax vital records directly into the MyESafeDepositBox account without having to first scan paper-based records and then upload information into their account. It also permits service providers, such as insurance agents or lenders, to fax documents directly into a user's account. In addition, much like the MyMedicalRecords PHR, the MyESafeDepositBox service also offers outbound fax and emergency login, features which further differentiate us in the marketplace. We also believe the portability of MyESafeDepositBox creates a product advantage in the marketplace because users are not required to do business with any particular bank. Users also benefit from the ability to sort, store and manage their vital records, while also using a free form text search to find records stored in their account with specific annotations. Our ability to provide private label branding affords banks, insurance companies, escrow services and other financial and legal businesses to provide not only a useful product but also reinforces that business's identity.
34
MMRPro
MMRPro competes with scanning services that market their services to doctors seeking to convert their historical paper records into electronic files, as well as Electronic Medical Record systems. Scanning services typically do not provide the doctor with an integrated end-to-end system that not only scans the record, but automatically sorts it by patient and by patient chart tab. Most scanners merely digitize patient records and store them either on a local drive or a Local Area Network drive, which requires the doctors to have an IT consultant manage their on-line records. Since MMRPro is a "Software As Service" model, the scanner records are sent to a web-hosted application with redundant data storage facilities and MMR handles the physical storage and management of patient data in compliance with all HIPAA privacy and security guidelines. This not only relieves doctors of having to worry about their in-house records management, it also allows them to access patient records from any Internet-connected computer as well as to deploy a copy of a record for the patient.
MMRPro also competes with EMR systems that offer doctors the opportunity to make their entire office paperless. However, many doctors, particularly solo and small group practitioners, are resisting EMRs because of the high cost of conversion and the difficulty and expense associated with maintaining an EMR system. MMRPro provides an efficient alternative to a full-blown EMR for many thousands of dollars less cost. MMRPro is designed to allow a doctor to digitize and securely store his or her patient records on-line for a fraction of the cost of an EMR system.
Intellectual Property
Patents
In September 2005, we filed a utility patent application on key elements of our proprietary process with the United States Patent and Trademark Office. In March 2007, we filed a separate application for our Emergency "Read Only" Password service (see discussion above under "- Our Products - MyMedicalRecords - An On-line Personal Health Record (PHR) - Additional Product Features - Emergency Log-In For Physicians and Other Emergency Response Personnel"). In May 2006, we filed a patent application with Australian authorities pursuant to the Patent Cooperation Treaty, which was awarded on May 29, 2008. A second patent application with Australian authorities was awarded on December 24, 2009. A patent application filed pursuant to the Patent Cooperation Treaty was awarded in Singapore on January 30, 2009. We intend to pursue patent protection in other countries where we may offer our products in the future. We also own the source code for our website. We consider patent protection an important element of a sound intellectual property portfolio, although we do not anticipate being able to prevent our competition from using other methods to achieve similar ends.
We intend to pursue patent protection in other countries where we may offer our products in the future. We also own the source code for our website. We consider patent protection an important element of a sound intellectual property portfolio, although we do not anticipate being able to prevent our competition from using other methods to achieve similar ends.
Other Assets
The Company acquired significant Intellectual Property assets from the merger with Favrille and is seeking to exploit and monetize those assets, which include, but are not limited to, data from the Company's pre-merger clinical vaccine trials, the Specifid vaccine, and the anti-CD20 antibodies.
Beginning in June 2009, we filed various national phase filings from the Patent Cooperation Treaty patent application directed to anti-CD20 monoclonal antibodies, including in the United States, Australia, Brazil, Canada, China, Europe, India, Japan, South Korea and Mexico. MMR Information Systems, Inc. acquired the Patent Cooperation Treaty application and anti-CD20 monoclonal antibody assets through its reverse merger with Favrille, Inc., a biopharmaceutical company, in January 2009.
MMR Information Systems, Inc. also acquired certain intellectual property rights involving B & T cell vaccine technology relating to the FavID vaccine in various stages in the United States and foreign countries through its reverse merger with Favrille, Inc. in January 2009. We make no guarantees as to the status of certain patents and patent applications. However, we are acting to pursue and maintain available intellectual property protection relating to the FavID vaccine intellectual property portfolio in the United States and major foreign markets of interest.
In November 2009, the Company retained Global Research Services LLC, a full-service clinical trials management organization, to assist the Company in its efforts to accelerate bringing to market its anti-CD20 monoclonal antibodies. Anti-CD20 antibodies are useful in treating B-Cell malignancies, including Non-Hodgkin Lymphoma (NHL) and additional B-Cell mediated conditions such as rheumatoid arthritis. Global Research Services specializes in serving sponsors of Phase I-IV research. MMRF's anti-CD20 antibody asset is potentially a
35
candidate for the next generation Rituximab, marketed under the trade name Rituxan(R) in the United States by Biogen Idec and Genentech (wholly owned member of the Roche Group) and under the name MabThera(R) by Roche in the rest of the world except Japan, where it is co-marketed by Chugai and Zenyaku Kogyo Co. Ltd. Rituxan is one of the world's most successful monoclonal antibodies with reported total sales in 2008 in excess of US $5.4 billion.
The Company also filed for extension of the Company's Patent Cooperation Treaty (PCT) patent application on anti-CD20 monoclonal antibodies to the national phase through filings in major European, Asian, North American, and South American markets.
Other Intellectual Property and Trademarks
We own the URL and domain name for the web address www.mymedicalrecords.com. As we continue to develop our website, we intend to register our logo as a service mark and protect the copyright in the initial and any other proprietary content that we develop to support our MyMedicalRecords PHR product. We also own the domain names www.mymedicalrecordsMD.com and www.mmrpatientview.com for use with MyMedicalRecords Pro, and own the domain name www.myesafedepositbox.com for use with our MyESafeDepositBox product.
We also own the source code for a handheld software program, developed to operate on the Palm operating system, which allows Palm users to create a personal medical history on a personal data assistant, or PDA, so that they can have access to this information while traveling and in the event an Internet connection is not available. This product is currently available for purchase as a PDA download through the Palm website.
Research and Development
In 2007, 2008, and 2009, MMR spent $245,162, $147,602, and $269,270 respectively, on research and development activities. From inception through December 31, 2009, MMR has spent a total of approximately $1.6 million relating to research and development activities, including approximately $644,000 in capitalized website development costs. In 2007 and 2008, Favrille spent $34,864,000 and $19,318,000, respectively, on research and development activities.
Properties
As a virtual Internet-based company we have minimal needs for office space to conduct our day-to-day business operations. Our corporate office is located at 468 N. Camden Drive in Beverly Hills, CA. Our mailing address is 2934½ Beverly Glen Cir., #702, Los Angeles CA 90077. The 468 Camden Drive property is leased on a month-to-month basis at rent of $4,600 per month. The 2934½ Beverly Glen Circle address is a post office box.
Employees
On May 29, 2008, Favrille provided a notice under the federal Worker Adjustment and Retraining Notification Act (the "WARN Act") to 132 of our then 144 employees, including six of our then executive officers, that their employment would end on June 6, 2008. Subsequently, we further reduced our operations and as of December 31, 2008 and prior to the Merger, we employed a total of two full-time employees.
Following the Merger and change in our business, as of the date hereof, we employ a total of six full-time employees and regularly use the services of an additional five consultants.
Legal Proceedings
From time to time, we may become involved in various legal proceedings generally incidental to our business. While the result of any litigation contains an element of uncertainty, management presently believes that the outcome of any unknown legal proceeding or claim, individually or combined, will not have a material adverse effect on our business, financial condition or future results of operations. At this time, we are not aware of any pending litigation.
36
On April 30, 2010, the Company filed a petition in the New York Supreme Court against the Lymphoma Research Foundation ("LRF"), a California nonprofit corporation, to enforce the Company's rights to a portion of certain intellectual property and biotechnology assets acquired as a part of the reverse merger with Favrille. LRF has claimed ownership of some of these assets including samples and data which were transferred to LRF in the summer of 2008. Also on April 30, 2010, LRF filed a declaratory relief action in the New York Supreme Court asserting its rights over the aforementioned samples and data. Both the Company and LRF have agreed to mediate the matter.
Reports
We make available free of charge through our website, www.mmrinformationsystems.com, our annual report on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K and amendments to those reports filed or to be furnished pursuant to Section 13(a) of the Securities Exchange Act of 1934 as soon as reasonably practicable after we electronically file such material with, or furnish it to, the SEC. Any information that is included on or linked to our Internet site is not a part of this report or any registration statement that incorporates this report by reference.
You may also read and copy any materials we file with the SEC at the SEC's Public Reference Room at 100 F Street, NE, Washington, DC 20549, on official business days during the hours of 10:00 am to 3:00 pm. Information on the operation of the Public Reference Room may be obtained by calling the SEC at 1-800-SEC-0330. The SEC maintains an Internet site that contains reports, proxy and information statements, and other information regarding issuers that file electronically with the SEC at http://www.sec.gov.
MANAGEMENT'S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION
AND RESULTS OF OPERATIONS
You should read the following discussion of our financial condition and results of operations together with our financial statements and related notes included elsewhere in this prospectus. This discussion may contain forward-looking statements that involve risks and uncertainties. Our actual results may differ materially from those anticipated in these forward-looking statements due to known and unknown risks, uncertainties and other factors, including those risks discussed under "Risk Factors" and elsewhere in this prospectus.
Business overview
Background and Basis of Presentation
As described above, on January 27, 2009, we consummated a transaction with MMR through a merger of our wholly-owned subsidiary with and into MMR pursuant to the terms of the Merger Agreement. In connection with the Merger, MMR became our wholly-owned subsidiary, with the former stockholders of MMR collectively owning (or having the right to acquire) shares of our common stock representing approximately 60.3% of the voting power of our capital stock on a fully diluted basis.
For accounting purposes, the Merger was treated as a reverse acquisition with MMR being the accounting acquirer. Accordingly, the historical financial results prior to the Merger are those of MMR and replace our historical financial results as we existed prior to the Merger. Our results of operations are included in MMR's financial results beginning on January 27, 2009. The following discussion of our financial condition and results of operations has been prepared using the financial statements of our wholly-owned subsidiary, MMR, for the fiscal year ended December 31, 2008 included elsewhere in this prospectus. The results of operations and financial condition for periods before the Merger on January 27, 2009 do not include the balances of Favrille.
MMR was incorporated in Delaware in 2005 and is headquartered in Los Angeles, CA. Effective February 9, 2009, MMR changed its corporate name to MMR Information Systems, Inc., or MMRIS after completion of the reverse merger with Favrille. We provide secure and easy-to-use online PHRs and electronic safe deposit box storage solutions, serving consumers, healthcare professionals, employers, insurance companies, unions and professional organizations and affinity groups. MMRIS enables individuals and families to access their personal and emergency medical records and other important documents, such as birth certificates, passports, insurance policies and wills anytime from anywhere using the Internet. The MMR products are built on proprietary, patented
37
technologies to allow documents, images and voicemail messages to be transmitted and stored in the system using a variety of methods, including fax, phone, or file upload without relying on any specific electronic medical record platform to populate a user's account. The Company's professional offering, MMR Pro, is designed to give physicians' offices an easy and cost-effective solution to digitizing paper-based medical records and sharing them with patients in real time..
Source of Revenues
Our revenues are derived from services, which are comprised of providing electronic access to consumer medical records and other vital documents and from the licensing of its services. We recognize revenue for such services only when the price is fixed or determinable, persuasive evidence of an arrangement exists, the service is performed, and collectability of the resulting receivable is reasonably assured.
Our subscriber revenues consist of annual and monthly recurring retail subscriptions and usage-based fees, which are primarily paid in advance by credit card, and corporate accounts that are based on either an access-fee or actual number of users, and in each case billed in advance at the beginning of each month of service. We defer the portion of annual recurring subscription fees collected in advance and recognize them on a straight line basis over the subscription period.
Revenues from subscriptions accounted for (i) 64.2%, 62.2% and 56.7% of our total revenues in 2009, 2008 and 2007, respectively.
We grant exclusive licenses for the sale and marketing of our services in international territories in consideration of an up-front license fee and an ongoing royalty. The royalty fee is usually a percentage of revenue earned by the licensee and there usually are certain minimum guarantees. License fee revenues received in advance from international licensees for the grant of the license are deferred and recognized over the period covered by the agreement. Minimum guaranteed royalty payments received in advance are deferred and recognized over the period to which the royalty relates. All such revenues are included under "License Fees and Other." In those cases where a license agreement contains multiple deliverables, the agreement is accounted for in accordance with ASC 605-025 (formerly EITF 00-21, "Revenue Arrangements with Multiple Deliverables"). As of the date hereof, we no longer had any active international licensing agreements. Revenues from licensing and other fees accounted for 35.8%, 37.8% and 43.3% of our total revenues in 2009, 2008 and 2007, respectively.
Cost of Revenue
Our cost of revenue includes the cost of maintaining our voice and fax mailboxes, long-distance call transport costs, fax and voice call processing costs, credit card transaction processing costs, web hosting and management fees, website maintenance and support costs, and costs associated with creating and mailing enrollment packages to our subscribers. Cost of revenue also includes customer service costs. We also charge to cost of revenue our direct selling costs, which include commissions paid to sales representatives who sell our wholesale and access based accounts.
Operating Expenses
The largest component of our operating expenses are our general and administrative expenses, which include personnel salaries and benefits, office rent and supplies, insurance costs, fees for legal and professional services, as well as our expenses for corporate telecommunications and internet access not associated with our products. Our operating expenses also include sales and marketing expenses (which include expenses associated with attending trade shows and travel costs, as well as a portion of personnel salaries allocated to sales and marketing activities), as well as technology development expenses (which includes expenses related to research and development as well as a portion of personnel salaries allocated to development activities).
38
Critical Accounting Policies and Estimates
The preparation of financial statements in conformity with U.S. generally accepted accounting principles requires the use of estimates and assumptions that affect the reported amounts of assets and liabilities, revenues and expenses, and related disclosures in the financial statements. Critical accounting estimates and assumptions are those that may be material due to the levels of subjectivity and judgment necessary to account for highly uncertain matters or the susceptibility of such matters to change, and that have a material impact on financial condition or operating performance. We base our estimates and judgments on our experience and on various other factors that we believe to be reasonable under the circumstances. Actual results may differ from these estimates under different assumptions or conditions. We believe the following critical accounting policies used in the preparation of our financial statements require significant judgments and estimates. For additional information relating to these and other accounting policies, see note 2 to our financial statements appearing elsewhere in this prospectus.
Revenue Recognition
In general, we recognize revenue for the provision of such services only when the price is fixed or determinable, persuasive evidence of an arrangement exists, the service is performed and the collectability of the resulting receivable is reasonably assured.
We follow the provisions of Staff Accounting Bulletin ("SAB") No. 104, "Revenue Recognition in Financial Statements" ("SAB 104") and accordingly, we recognize annual recurring subscription fees paid in advance on a straight line basis over the subscription period. In addition, we follow the provisions FASB ASC 605-25 (formerly EITF 00-21, "Revenue Arrangements with Multiple Deliverables") and accordingly, we defer recognition of upfront fees paid under our licensing agreement and recognize them over the period covered by the licensing agreement, and similarly, we defer recognition of minimum guaranteed royalty fees paid in advance under our licensing agreement and recognize them in the period to which they relate.
Accounting for Income Taxes and Uncertain Tax Positions
Deferred income taxes result primarily from temporary differences between financial and tax reporting. Deferred tax assets and liabilities are determined based on the difference between the financial statement basis and tax basis of assets and liabilities using enacted tax rates. Future tax benefits are subject to a valuation allowance when management is unable to conclude that deferred tax assets will more likely than not be realized from the results of operations. At each of the financial statement dates presented, we recorded a full valuation allowance against deferred income taxes due to our limited operating history and net losses recorded since inception. Our estimate for the valuation allowance for deferred tax assets requires management to make significant estimates and judgments about projected future operating results. If actual results differ from these projections or if management's expectations of future results change, it may be necessary to adjust the valuation allowance.
We have generated losses for federal and state income tax reporting since inception. These tax losses are available for carryforward until their expiration. In addition to potential expiration, there are other factors that could limit our ability to use our federal and state tax loss carryforwards. For example, use of prior net operating loss carryforwards can be limited after an ownership change, such as the Merger. Accordingly, it is not certain how much of our existing net operating loss carryforwards will be available for use. In addition, we must generate taxable income in the future in order to use net operating loss carryforwards that have not expired. We are in the process of evaluating the effects of the Merger and change in control of our company under Internal Revenue Code Section 382, which may limit the availability of our net operating loss carryfowards.
Effective January 1, 2007, we began to measure and record uncertain tax positions in accordance with new guidance, which prescribes a threshold for the financial statement recognition and measurement of a tax position taken or expected to be taken in a tax return. Only tax positions meeting the more-likely-than-not recognition threshold at the effective date may be recognized or continue to be recognized upon adoption of this Interpretation. The new guidance also provides guidance on accounting for derecognition, interest and penalties, and classification and disclosure of matters related to uncertainty in income taxes. Accounting for uncertainties in income tax positions involves significant judgments by management.
39
Intangible Assets
We account for website development costs in accordance with ASC 350-50 (formerly EITF 00-2, "Accounting for Website Development Costs") and ASC 985-20 (formerly SFAS No. 86 "Accounting for the Costs of Computer Software to Be Sold, Leased, or Otherwise Marketed "). Pursuant to ASC 350-50 and 985-20, the Company capitalizes internally developed website costs when the website under development has reached technological feasibility. These costs are amortized, typically over an estimated life of five years, using the larger of the amount calculated using the straight-line method or the amount calculated using the ratio between current period gross revenues and the total of current period gross revenues and estimated future gross revenues. At each balance sheet date, we evaluate the unamortized capitalized website costs compared to the net realizable value. The amount by which the unamortized capitalized website costs exceed its net realizable value is written off. The determination of estimated future gross revenues requires the exercise of judgment and assumptions by our management and actual results could vary significantly from such estimates.
Impairment of Long-Lived Assets and Intangibles
We evaluate long-lived assets and identifiable intangible assets with finite useful lives under the provisions of SFAS No. 144, Accounting for the Impairment or Disposal of Long-Lived Assets, and accordingly, management reviews our long-lived assets and identifiable intangible assets with finite useful lives for impairment whenever events or changes in circumstances indicate that the carrying amount of such assets may not be recoverable. We recognize an impairment loss when the sum of the future undiscounted net cash flows expected to be realized from the asset is less than its carrying amount. If an asset is considered to be impaired, the impairment to be recognized is measured by the amount by which the carrying amount of the asset exceeds its fair value. Considerable judgment is necessary to estimate the fair value of the assets and accordingly, actual results could vary significantly from such estimates. Our most significant estimates and judgments relating to the long-lived asset impairments include the timing and amount of projected future cash flows.
Share-Based Compensation
We recognize compensation expense, under the provisions of ASC 718-20 (formerly SFAS No. 123(R), "Share Based Payments"). As a result, we recognize compensation expense in an amount equal to the estimated fair value of stock-based awards and issuances, such as stock options granted to employees and non-employees and restricted stock awards. This estimation of the fair value of each stock-based grant or issuance on the date of grant involves numerous assumptions by management. Although we calculate the fair value of each stock option under the Black Scholes option pricing model, which is a standard option pricing model, this model still requires the use of numerous assumptions. Assumptions used in this model include, among others, the expected life (turnover), volatility of the underlying equity security, and expected dividends. The model and assumptions also attempt to account for changing employee behavior as the stock price changes and capture the observed pattern of increasing rates of exercise as the stock price increases. Because we do not have adequate historic data regarding exercise rates and determined that MMR's options are "plain vanilla," we assigned the expected life equal to the midpoint between the vesting period and the contractual option termination. Further, because there is no public market for MMR's shares, the expected volatility is based on ranges for similar companies. The use of different public company comparables in our assumptions and other assumptions by management in the Black Scholes option pricing model could produce substantially different results.
Recent Accounting Pronouncements
For a description of recent accounting pronouncements, please see note 2(o) to our financial statements appearing elsewhere in this prospectus.
Internal Control over Financial Reporting
In connection with the audit for the years ended December 31, 2009, December 31, 2008 and 2007, our independent registered public accounting firm identified significant deficiencies in our internal control over financial reporting that are material weaknesses. These included the following:
- Inadequate Financial Statement Close Process and Cut-Off. Because we did not perform a timely and efficient close of our financial records and did not require supporting documentation for all closing journal entries, we were required to make a significant amount of post-closing entries to adjust our financial statements to comply with generally accepted accounting principles. To remedy this weakness, we have hired outside accounting consultants. Our outside consultants have been more and more involved with each quarter close and closed the financial records for the quarter ended September 30, 2009. Beginning during the quarter ended March 31, 2009, the Company also began maintaining a more detailed binder with all account reconciliations and support for all closing journal entries.
40
- Lack of Financial Expertise and Accounting Staff. We have very limited accounting personnel and limited access to experts due to our limited cash situation. Because of these limitations, we misapplied accounting literature and authoritative guidance, which lead to errors in our financial statements. To remedy this weakness, the outside accounting consultants have been taking a more active role in application of accounting literature and authoritative language and we expect the consultants to take an active role until we hire a fulltime Controller.
- Lack of Supporting Documentation and Schedules. Because of the small size of our company prior to the Merger and the informal nature of our relationships with many of our consultants and directors, we did not always formally and promptly document equity-related transactions or maintain detailed accounting records supporting all cash transactions. This lack of supporting documentation and schedules led to errors in determining the impact on our financial statements of these transactions. To remedy this weakness, the Company also began maintaining a more detailed binder with all account reconciliations and support for all closing journal entries, including support for equity related transactions.
- Inadequate Journal Entry Review Process. Because of the small size of our company prior to the Merger, our journal entries were not reviewed and authorized by a person who has responsibility for oversight. This lack of oversight could lead to unauthorized entries, management override of controls or the concealment of errors and fraud. To remedy this weakness, we have hired outside accounting consultants who bring in two levels of review and oversight.
In addition to the above, during the six months ended June 30, 2009, our independent registered public accounting firm identified additional material weaknesses regarding our inadequate approval process and documentation of equity grants because we did not formally document all equity related grants and formally approve them in Board meetings or formally document revenue arrangements with third parties. To remedy this weakness, we hired outside consultants who are experts in implementing controls required under the Sarbanes-Oxley rules and regulations; these consultants have better documented our internal control policies and procedures.
During the nine months ended September 30, 2009, our independent registered public accounting firm identified additional material weaknesses regarding the improper accrual of invoices, whereby we did not properly accrue for all outstanding invoices as of September 30, 2009. To remedy this weakness, our Sarbanes-Oxley consultants improved our processes in documenting our internal control policies and procedures.
To further remedy these material weaknesses in addition to our specific remedial steps outlined above, we plan to hire experienced accounting staff with SEC public company experience, including our December 17, 2009 appointment of Ingrid Safranek as our new Chief Financial Officer.
The material weaknesses that were noted in our earlier filings have all been remediated as of December 31, 2009. This was accomplished by devoting substantial time and resources to the completion of the required assessment of our internal controls over financial reporting as well as the subsequent remediation of the material weaknesses that were identified as a result of that assessment. Our efforts involved management, outside consultants and our audit committee, which approved and provided oversight in the execution of the assessment. We intend to allocate a sufficient level of resources to ensure that our internal control structure is maintained and functions properly and effectively.
In light of these material weaknesses disclosed in our earlier filings, our management performed additional analytical procedures, validation processes and engaged services of a third party financial consulting firm to assist management in the preparation of our financial statements which we believe are in accordance with generally accepted accounting principles and the rules and regulations of the Commission. Given the remediation efforts performed in 2009 and our evaluation under the framework in Internal Control-Integrated Framework, our management concluded that our internal control over financial reporting was effective as of December 31, 2009.
41
Factors Affecting Future Results
In the first quarter of 2009, we entered into arrangements with The Latino Coalition, a non-profit organization with 600,000 business members nationwide, to market our product to its members (either on a direct to consumer basis or as an added benefit that its members can offer their employees), and with the United Marketing Group, a leading direct marketer of affinity membership programs and merchandise products, to market our products as a direct to consumer benefit. In addition, in May 2009 at its annual meeting, the National Rifle Association, or the NRA, announced the launch of NRA E-Safe, a private label version of our Electronic Safe Deposit product. The NRA is now gradually rolling out NRA E-Safe as an added benefit to its members. If members of these organizations take advantage of the introductory offers and subscribe for our services, these contracts could become important sources of revenue for our company over time, particularly after the initial introductory periods. The Company is also devoting its resources to the development of MMRPro which began testing in Beta sites in April 2010. We are working with EMR companies and national scanning companies to create a national channel distribution for product rollout.
MMRPro
MMRPro was launched in late 2009. During the first quarter the Company began the process of selling and installing the first group of 10 systems in physician offices. The installation and sales implementation process takes approximately 6 weeks. This encompasses a checklist of 60 items which includes everything from where the system hardware will be located, installed and connected, a detailed analysis of office workflow, and customizing connectivity to any existing office practice management system. After completion, the Company will commence billing for the system. Prior to the end of the second quarter, the Company expects it will have completed the majority of these installations. These initial installations were sold by the Company on a direct basis. The Company received substantial installation support from Kodak and selected Kodak resellers.
Starting in the third quarter the Company will be calling on physician offices throughout the United States through the nationwide Kodak reseller channel.
The Company also plans on building a strategic relationship with hospitals to underwrite the use of MMRPro for staff physicians. This strategic relationship could enable staff physicians to digitize patient charts prior to admissions streamlining the admissions process for the patients to the hospital.
MMRPro "Stimulus Program"
In addition to its product functionality, MMRPro includes a "Stimulus Program" for the health care professional that creates a revenue opportunity for its users from patient upgrades. The Company believes its Stimulus Program can generate at least as much money for the average group practice over a three year term as the $44,000 that is possible through the HITECH Act over a five year period.
Hispanic Marketing
As a result of MMR's relationship with Kodak, Kodak became a sponsor of The Latino Coalition at its annual Economic Summit for Hispanic entrepreneurs on May 5th, 2010. Kodak, MMR and other Coalition sponsors will work together to introduce MMRPro, which includes Kodak's document imaging solutions, to physician groups and hospitals serving the Latino community. MMR's relationship with The Latino Coalition started in the first quarter of 2009. The Latino Coalition is a non-profit organization with nearly 600,000 business members nationwide. Mr. Hector Barreto, one of the members of MMR's Board of Directors, is the Chairman of the Latino Coalition.
Direct Marketing
On September 15, 2009, the Company entered into a into a five year agreement to license an existing 80 million person direct marketing database of street addresses, cellular phone numbers, e-mail addresses and other comprehensive data with E-Mail Frequency, LLC. The agreement allows the Company to market, through the use of this database, its MyMedicalRecords Personal Health Record, MyEsafeDepositBox virtual vault, and MMRPro integrated document management system for physicians and their patients. The Company is continuing to test the database which continues to increase targeted traffic to the Company's websites.
42
Exploiting MMR Biotech Assets and Patents
The Company plans on recruiting scientists and experienced venture capitalists to assist the Company in generating revenue through licensing agreements as would be usual and customary in the biotech industry and which could include milestone payments as the term is generally used in the industry. The Company will also explore partnerships with manufacturers and or investors of and in Biotech products and services. These are assets and other intellectual property that the Company acquired from its reverse merger with Favrille. Those assets include, but are not limited to, data from the Company's pre-merger clinical vaccine trials, the Specifid vaccine, and the anti-CD20 antibodies.
Beginning in June 2009, we filed various national phase filings from the Patent Cooperation Treaty patent application directed to anti-CD20 monoclonal antibodies, including in the United States, Australia, Brazil, Canada, China, Europe, India, Japan, South Korea and Mexico. The Company also acquired certain intellectual property rights involving B & T cell vaccine technology relating to the FavID vaccine in various stages in the United States and foreign countries through its reverse merger with Favrille. The Company makes no guarantees as to the status of certain patents and patent applications. However, we are acting to pursue and maintain available intellectual property protection relating to the Company's patents and filings including but not limited to the FavID vaccine intellectual property portfolio in the United States and major foreign markets of interest.
In November 2009, the Company retained Global Research Services LLC, a full-service clinical trials management organization, to assist the Company in its efforts to accelerate bringing to market its anti-CD20 monoclonal antibodies. Anti-CD20 antibodies are useful in treating B-Cell malignancies, including Non-Hodgkin Lymphoma (NHL) and additional B-Cell mediated conditions such as rheumatoid arthritis. Global Research Services specializes in serving sponsors of Phase I-IV research. MMRF's anti-CD20 antibody asset is potentially a candidate for the next generation Rituximab, marketed under the trade name Rituxan(R) in the United States by Biogen Idec and Genentech (wholly owned member of the Roche Group) and under the name MabThera(R) by Roche in the rest of the world except Japan, where it is co-marketed by Chugai and Zenyaku Kogyo Co. Ltd. Rituxan is one of the world's most successful monoclonal antibodies with reported total sales in 2009 of US $5.6 billion.
The Company has also had certain patents granted from its Personal Health Record products and services which it believes will generate licensing revenue from competitors to the Company.
Insurance and Banking
On March 22, 2010, the Company entered into a Master Services Agreement with Chartis International LLC ("Chartis"). Chartis provides a wide range of insurance solutions and is one of the world's leading property-casualty and general insurance organizations, with operations in over 160 countries and jurisdictions. The Company believes it will provide MMR's secure online Personal Health Record (PHR) to Chartis policyholders worldwide starting prior to the end of this year. Initially, MMR anticipates launching Chartis branded products in the United Kingdom, France, Germany, Japan, South Korea, Singapore, Malaysia, South Africa, Russia, Ukraine, Finland, Hungary, Czech Republic, Poland, Israel, Estonia, Lithuania, Romania and other countries in Europe , Asia and the Commonwealth of Independent States in addition to select countries in Central and South America, including Argentina. Translations of the product are first expected to be available in English, Spanish, French, German, Italian, Japanese and Chinese.
The Company is also in active negotiations with a major financial institution regarding the development of a paperless loan processing and delivery solutions whereby loan documents can be delivered electronically through the Company's MyESafe DepositBox product with resultant completed documents being filed in a permanent on-line MyESafeDepositBox account offered by the financial institution.
China
The Company and Unis-Tonghe Technology Co ("UNIS") entered into a Cooperation Agreement and related documents, which we refer to as the Cooperation Agreement. Under the Cooperation Agreement, the Company and UNIS agreed to form a joint venture in China for the purpose of deploying the Company's personal health record services and document imaging and management solutions as part of a total EMR solution in China. Upon approval of the venture by the Chinese government the Company will own 40% of the joint venture and UNIS will own 60%.
43
The joint Venture plans on developing a comprehensive Electronic Medical Record system in compliance with recent development criteria being proposed by the Chinese government.
As a result of lead times to develop customized EMR solutions in response to government specifications, this effort could take several years. In the meantime the Company has begun to leverage its resources in China with other U.S. partners on the ground in China including Kodak. As a result, the Company anticipates selling MMRPro in conjunction with Kodak resellers throughout the Asia Pacific region in advance of a UNIS MMR EMR product launch. The Company also believes that China will provide one of the earlier Chartis revenue streams.
Clinical Trials Platform
The Company is exploring the development of a service that could enable visitors to the Company's consumer and professional services websites to easily find upcoming and ongoing clinical trials for themselves, family members and friends who may be seeking new channels for treatment of disease. If this development effort is successful, and is successfully commercialized, the Company could receive revenue from the delivery of patients to these trials through existing strategic partnerships and agreements.
We have incurred losses since inception and our ability to achieve profitability in the near term is primarily dependent on our ability to invest capital in our business to increase revenues while controlling and limiting expenses at a rate slower than revenue growth. Some of the current conditions and factors that we expect could have a significant impact on our future results are:
- Our ability to raise capital in order to meet our financial obligations and invest in our business to grow revenues;
- The pace of market adoption of our current and future products;
- Our ability to launch new products;
- Managing costs while building up an effective sales and service delivery organization for our products; and
- Our ability to enter into marketing arrangements with large membership and affinity organizations for our products and maintain and grow subscribers from such arrangements, such as those noted above, particularly after the initial introductory period.
Going Concern
As more fully described in note 1 to the consolidated financial statements appearing elsewhere in this prospectus, our independent registered public accounting firm included an explanatory paragraph in their report on our 2009 financial statements for the year ended December 31, 2009 related to the uncertainty of our ability to continue as a going concern. At March 31, 2010, current liabilities of $10,376,770 exceeded cash and cash equivalents of $878,442.
Management's plan regarding this matter is to continue to utilize its available line of credit with The RHL Group (see Note 3). At March 31, 2010, there was approximately $1,500,000 available under The RHL Group line of credit. Additionally, the Company plans to continue to sell additional debt and equity securities, continue to settle its existing liabilities through issuance of equity securities, explore other debt financing arrangements, continue to increase its existing subscriber and affiliate customer base and sell MMR Pro products to obtain additional cash flow over the next twelve months. There can be no assurance that funds from these sources will be available when needed or, if available, will be on terms favorable to us or to our stockholders. If additional funds are raised by issuing equity securities, the percentage ownership of our stockholders will be reduced, stockholders may experience additional dilution or such equity securities may provide for rights, preferences or privileges senior to those of the holders of our common stock.
Off-Balance Sheet Arrangements
On January 4, 2010, the Company and UNIS entered into a Cooperation Agreement and related documents, which we refer to as the Cooperation Agreement. Under the Cooperation Agreement, the Company and UNIS agreed to form a joint venture in China for the purpose of deploying the Company's personal health record services and document
44
imaging and management solutions in China. The Company will own 40% of the joint venture and UNIS will own 60% and each party will have the right to designate two members of the joint venture's board of directors, with the fifth member being a Chinese citizen mutually designated by the Company and UNIS. Under the Cooperation Agreement, board actions will require the approval of more than three of the five members of the joint venture's board of directors and no material actions may be taken unless all board members are present and voting at the meeting.
Under the Cooperation Agreement, the Company and UNIS will contribute an aggregate of 100 million RMB to the joint venture, based on each party's respective ownership, in the form of intellectual property rights, equipment, brand value, cash and such other consideration as may be agreed to between the parties. Each party's obligation to contribute to the joint venture is subject to a number of conditions, including obtaining all necessary approvals of and licenses from the Chinese government, as well as the joint venture meeting its budget, goals and objectives at the time any contributions are due. Under the Cooperation Agreement, each party's contributions will be made over a period of sixty months.
The foregoing description of the Cooperation Agreement does not purport to be complete and is qualified in its entirety by reference to the Cooperation Agreement, a copy of which is filed as an Exhibit to the Form 10-K.
The Company's entry into the Cooperation Agreement described above constitutes the creation of a direct financial obligation.
Results of Operations
The following table sets forth items in our statements of operations for the periods indicated:
Comparison of Three months Ended March 31, 2010 to Three months Ended March 31, 2009 and
Twelve Months Ended December 31, 2009 to Twelve Months Ended December 31, 2008.
| Three Months ended | Fiscal Year ended | |||||||||||
|
March 31,
|
December 31,
|
|||||||||||
|
2010
|
2009
|
2009
|
2008
|
|||||||||
| Revenues | ||||||||||||
| Subscriber | $ | 90,164 | $ | 92,495 | $ | 397,798 | $ | 280,572 | ||||
| License fees and other |
-
|
53,292
|
221,451
|
170,783
|
||||||||
| Total revenues | 90,164 | 145,787 | 619,249 | 451,355 | ||||||||
| Cost of revenues |
113,036
|
106,852
|
456,456
|
409,900
|
||||||||
| Gross profit (loss) |
(22,872)
|
38,935
|
162,793
|
41,455
|
||||||||
| General and administrative expenses | 1,219,315 | 1,294,405 | 5,440,545 | 2,108,063 | ||||||||
| Sales and marketing expenses | 930,132 | 235,886 | 1,402,870 | 914,162 | ||||||||
| Technology development |
82,139
|
43,748
|
269,270
|
143,031
|
||||||||
| Total Operating Expenses |
2,231,586
|
1,574,039
|
7,112,685
|
3,165,256
|
||||||||
| Loss from operations | (2,254,458) | (1,535,104) | (6,949,892) | (3,123,801) | ||||||||
| Gain on settlement of payables | - | 52,225 | 89,170 | - | ||||||||
| Change in valuation of derivative liabilities | (2,168,480) | (2,150,831) | 633,626 | - | ||||||||
| Interest and other expenses, net |
(2,040,420)
|
(242,680)
|
(4,068,565)
|
(398,675)
|
||||||||
| Net loss | (6,463,358) | (3,876,390) | (10,295,661) | (3,522,476) | ||||||||
| Dividend to Preferred Shareholders |
-
|
-
|
-
|
(35,620)
|
||||||||
| Net Loss Available to Common Shareholders | $ |
(6,463,358)
|
$ |
(3,876,390)
|
$ |
(10,295,661)
|
$ |
(3,558,096)
|
||||
| Total Shares issued and outstanding | 204,409,509 | 122,269,253 | 173,689,343 | 80,629,266 | ||||||||
| Weighted average common shares outstanding | ||||||||||||
| (Basic and Diluted) | 188,263,878 | 109,748,626 | 132,311,386 | 60,911,929 | ||||||||
| EPS Basic and Diluted | $ | (0.03) | $ | (0.04) | $ | (0.08) | $ | (0.06) | ||||
| Stock Price | $ | 0.16 | $ | 0.28 | $ | 0.08 | $ | 0.05 | ||||
| Market Cap | $ | 32,705,521 | $ | 34,235,391 | $ | 13,895,147 | $ | 4,031,463 | ||||
45
Revenues. As a result of the termination of international licenses for the Company's products in September 2009,
revenues decreased by $55,623 for the three months ended March 31, 2010 from $145,787 for the three months ended
March 31, 2009 to 90,164. As a result of these terminated international license agreements, the Company was able to enter
into a Master Agreement with Chartis which the Company believes will have significantly more value than the terminated
international licenses. Cost of revenue. Our cost of revenue increased by $6,184, from $106,852 for the three months ended March 31,
2009 to $113,036 for the three months ended March 31, 2010. We had a gross loss and negative gross margin of 25.4% for
the first quarter of 2010 compared to a gross profit margin of 26.7% for the first quarter of 2009. The decrease in our gross
margin was due to the termination of the MMR Australia license agreement as license fee revenues carried minimal to no
cost. Operating expenses. The following table sets forth the individual components of our operating expenses for the three
months ended March 31, 2010 and 2009:
Operating Expenses.
Three Months ended
March 31,
2010
2009
General and administrative expenses
$
1,219,315
$
1,294,405
Sales and marketing expenses
930,132
235,886
Technology development
82,139
43,748
Total Operating Expenses
$
2,231,586
$
1,574,039
Sales and Marketing. In addition to the non-cash equity based compensation, sales and marketing expenses grew because we attended several trade shows and Health Care Information and Management Systems Society ("HIMSS") conferences and incurred international travel related sales and marketing expenses during first three months of 2010 compared to no trade show or HIMSS attendances or international travel during the same period in 2009. As a result, we incurred a total trade show and international travel-related expenses of approximately $183,000 during the three months ended March 31, 2010, as compared to only approximately $4,000 during the same period in 2009.
Gain on settlement of payables. During the three months ended March 31, 2009, we had a gain on the settlement of payables of $52,225 in connection with settlement agreements entered into with certain vendors relating to liabilities we assumed in connection with the Merger. There was no such gain during the three months ended March 31, 2010.
Change in valuation of derivative liabilities. In November 2007, Favrille issued warrants to purchase 4.4 million shares of common stock in conjunction with a registered direct offering of common stock and warrants. We assumed these outstanding warrants as a result of the Merger on January 27, 2009. The value associated with these warrants was recorded as a liability utilizing the Black-Scholes valuation model, and revalued at the end of each quarter. The change in valuation of the warrants from the beginning to the end of the period is recorded as change in valuation of derivative liabilities. During the three months ended March 31, 2009, the change in valuation of the warrants amounted to $355,993. During the three months ended March 31, 2010, the change in valuation of the warrants amounted to $177,996.
On January 27, 2009, the Company performed an analysis as if all holders of options and warrants exercised their rights under those contracts. This analysis resulted in the conclusion that the Company had inadequate authorized shares to settle 100% of these contracts. Therefore, as shareholder approval would be necessary to increase the number of authorized shares, settlement of these obligations would not be within the control of the Company. Consequently certain non-employee options and warrants were accounted for as derivative liabilities. The event giving rise to this condition was the merger between Favrille, Inc. and MyMedicalRecords.com, Inc. on January 27,
46
2009. On that date, the fair value of the option and warrant contracts subject to this analysis was reclassified from equity to derivative liabilities. On March 31, 2009, these contracts were again valued using the Black-Scholes option valuation model and the difference between the original value at January 27, 2009 of $871,051 and the value at March 31, 2009 of $2,665,889 was recorded as a loss on change in value of derivatives. The loss recorded related to this matter was $1,794,838 and is included in the change in valuation of derivative liabilities for the three months ended March 31, 2009 of $2,150,831.
Also, we had certain non-employee options and warrants outstanding which were accounted for as derivatives as there was a possibility, although remote, that the Company may not have enough authorized shares to settle its 12% Convertible Promissory Note obligations using common stock. The event giving rise to this condition was the first Convertible Note the Company entered into on July 16, 2009. As of July 16, 2009 and for all subsequent options and warrants issued prior to March 31, 2010, the fair value of vested non-employee options and warrant contracts subject to this analysis was reclassified from equity to derivative liabilities. On March 31, 2010, these contracts were again valued using the Black-Scholes option valuation model and the difference between the value at December 31, 2009 of $1,534,824 plus the value of options and warrants granted to non-employees during the three months ended March 31, 2010 of 89,770, including the cancellation of exercised warrants and options of $19,813 and the value at March 31, 2010 of $3,576,430, was recorded as a loss on change in value of derivatives. The loss recorded related to this matter was $1,971,650 and is included in the change in valuation of derivative liabilities for the three months ended March 31, 2010 of $2,168,480.
On May 4, 2010, the condition leading to the $3,576,430 derivative liability (see Note 9) and the $1,971,650 loss on change in value of derivatives for the three months ended March 31, 2010 was remediated and on this date, the derivative liability was reclassified back into equity. As of March 31, 2010, two unrelated third-partied had a principal balance of $100,000 and 250,000 warrants to purchase common stock outstanding in connection with the 12% Convertible Promissory Notes. On May 4, 2010, the two parties converted the $100,000 balance and exercised the 250,000 warrants and received 886,943 shares of common stock. As a result, the condition leading to the $3,576,430 derivative liability was remediated and the derivative liability was reclassified back into equity.
Interest and Other Finance Charges, Net. We had interest and other finance charges, net of $2,040,420 for the three months ended March 31, 2010, an increase of $1,797,740 from $242,680 for the three months ended March 31, 2009. The increase was due to non-cash interest expense of $1,971,363 attributed to the conversion feature and warrant issued with the 12% Convertible Promissory Notes. See Note 11 for a detailed explanation. The Company did not enter into any such 12% Convertible Promissory Notes during the three months ended March 31, 2009.
Net loss. As a result of the foregoing, we had a net loss of $6,463,658 for the three months ended March 31, 2010 compared to a net loss of $3,876,390 for the three months ended March 31, 2009.
Fiscal Year Ended December 31, 2009 as Compared to the Fiscal Year Ended December 31, 2008
The following table sets forth items in our statements of operations for the periods indicated:
| Fiscal Year ended | ||||||
|
December 31,
|
||||||
|
2009
|
2008
|
|||||
| Revenues | ||||||
| Subscriber | $ | 397,798 | $ | 280,572 | ||
| License fees and other |
221,451
|
170,783
|
||||
| Total revenues | 619,249 | 451,355 | ||||
| Cost of revenues |
456,456
|
409,900
|
||||
| Gross profit (loss) |
162,793
|
41,455
|
||||
| General and administrative expenses | 5,440,545 | 2,108,063 | ||||
| Sales and marketing expenses | 1,402,870 | 914,162 | ||||
| Technology development |
269,270
|
143,031
|
||||
| Total Operating Expenses |
7,112,685
|
3,165,256
|
||||
| Loss from operations | (6,949,892) | (3,123,801) | ||||
47
| Gain on settlement of payables | 89,170 | - | ||||
| Change in valuation of derivative liabilities | 633,626 | - | ||||
| Interest and other expenses, net |
(4,068,565)
|
(398,675)
|
||||
| Net loss | $ |
(10,295,661)
|
$ |
(3,522,476)
|
Revenues. Revenues increased $167,894, or 37.2%, to $619,249 for the year ended December 31, 2009 from $451,355 for the year ended December 31, 2008 primarily due to significant growth in both subscriber revenues, which resulted from an 8% average monthly growth rate in the number of subscribers during 2009 from our affiliate customers and growth in our current corporate customer base, as well as growth in license and other fees revenues, primarily due to higher royalty payments and platform access fees from our international licensees.
Cost of revenue. Our cost of revenue increased to $456,456, or 11.4%, for the year ended December 31, 2009 from $409,900 for the year ended December 31, 2008, primarily as a result of the added costs for telephone numbers associated with the larger number of subscribers, and the need to support two separate web platforms for two different customer groups. We had a gross margin of 26.3% for the year ended December 31, 2009, as compared to 9.2% for 2008. The primary reasons for the improvement in our gross profit and margin in 2009 were because our revenues increased sufficiently to cover the fixed cost components of our costs of revenue and there was an increase in minimum guaranteed royalty payments with no corresponding cost increase, demonstrating the scalability of our business.
Operating expenses. The following table sets forth the individual components of our operating expenses for the year ended December 31, 2009 and 2008:
| Fiscal Year ended | ||||||
|
December 31,
|
||||||
|
2010
|
2009
|
|||||
| General and administrative expenses | $ | 5,440,545 | $ | 2,108,063 | ||
| Sales and marketing expenses | 1,402,870 | 914,162 | ||||
| Technology development |
269,270
|
143,031
|
||||
| Total Operating Expenses | $ |
7,112,685
|
$ |
3,165,256
|
||
Operating expenses increased 124.7% to $7,112,685 for the year ended December 31, 2009, from $3,165,256 for the year ended December 31, 2008. This was primarily due to the increase in our general and administrative expenses, which increased 158.1%, in large part because of increased legal and audit related fees of approximately $1.9 million, a significant portion of which are expenses specifically associated with the Merger not expected to impact future periods.
In addition, we also incurred general and administrative expenses of approximately $350,000 associated with salary and share based compensation related to Favrille employees assumed as a result of the Merger, which are non-recurring expenses not expected to impact future periods.
Sales and marketing expenses increased by 53.5% in 2009 as compared to 2008 primarily due to additional consultants hired in 2009 to support our sales growth, specifically the anticipated growth from MMRPro.
Technology development expenses increased by 88.3% in 2009 as compared to 2008 due primarily to increased expenses associated with the development of our MMRPro product, which is a non-recurring expense not expected to recur in future periods.
Non-cash related expenses which are included in operating expenses for the year ended December 31, 2009 total $2,155,083 and are made up of primarily of approximately $422,000 relating to the issuance of warrants as payment for services, $880,000 for share based compensation relating to stock options issued during the year, and $440,000 relating to restricted stock issued as payment for services, $221,000 relating to a loss on stock settlement of liabilities, and $100,000 relating to a write-off of advances due from a related party.
48
Gain on settlement of payables. During the year ended December 31, 2009, we had a gain on the settlement of payables of $89,170 in connection with settlement agreements entered into with certain vendors relating to liabilities we assumed in connection with the Merger. There was no such gain during the year ended December 31, 2008.
Change in valuation of derivative liabilities. In November 2007, Favrille issued warrants to purchase 4.4 million shares of common stock in conjunction with a registered direct offering of common stock and warrants. We assumed these outstanding warrants as a result of the Merger on January 27, 2009. The value associated with these warrants was recorded as a liability utilizing the Black-Scholes valuation model. From January 27, 2009 to December 31, 2009, the value of these warrants increased by $88,998. We recorded the change in valuation as a change in valuation of warrants during the year ended December 31, 2009.
Also, we had certain non-employee options and warrants outstanding which were accounted for as derivatives as we had inadequate authorized shares to settle 100% of these contracts in the event they were all to be exercised. The event giving rise to this condition was the merger between Favrille and MyMedicalRecords.com, Inc. on January 27, 2009. On that date, the fair value of the option and warrant contracts subject to this analysis was reclassified from equity to derivative liabilities. The condition was remedied on June 13, 2009 when certain of the option and warrant holders indefinitely waived their rights under those contracts. On that date, the derivative liabilities were reclassified back into equity. On June 13, 2009, these contracts were again valued using the Black-Scholes option valuation model and the difference between the original value at January 27, 2009 of $871,052 and the value at June 13, 2009 of $1,377,033 was recorded as a loss on change in value of derivatives for the year ended December 31, 2009. The loss recorded for this matter was $505,980 for the year ended December 31, 2009. There was no such activity during the year ended December 31, 2008.
Also, we had certain non-employee options and warrants outstanding which were accounted for as derivatives as there was a possibility, although remote, that the Company may not have enough authorized shares to settle its 12% Convertible Promissory Note obligations using common stock. The event giving rise to this condition was the first Convertible Note the Company entered into on July 16, 2009. As of July 16, 2009 and for all subsequent options and warrants issued prior to September 30, 2009, the fair value of the option and warrant contracts subject to this analysis was reclassified from equity to derivative liabilities. On December 31, 2009, these contracts were again valued using the Black-Scholes option valuation model and the difference between the original value at July 16, 2009 plus contracts granted between July 16, 2009 and December 31, 2009 of $2,664,125, including the cancellation of exercised warrants and options of $95,790 and the value at December 31, 2009 of $1,534,824, was recorded as a gain on change in value of derivatives. The gain recorded related to this matter was $1,239,529 included in the change in valuation of derivative liabilities for the twelve months ended December 31, 2009. There was no such change in valuation during the twelve months ended December 31, 2008.
The remaining component of the change is valuation of derivative liability for the twelve months ended December 31, 2009 is attributed to the change in the fair value of the conversion feature of the 12% Convertible Promissory Note for those that have not been converted as of December 31, 2009. This change in the fair value of the conversion feature resulted in a $10,925 loss for the twelve months ended December 31, 2009.
The change in valuation of derivative amounts impacting our statement of operations for the year ending December 31, 2009 was a non- cash expense.
Interest and Other Finance Charges, Net. We had net interest and other financing charges of $4,068,565 for the year ended December 31, 2009, an increase of $3,669,890 from $398,675 for the year ended December 31, 2008. The increase was primarily as a result of common stock and warrants issued during 2009 in exchange for the Company to continue to defer payments to their debtors and consultants of $774,217, to The RHL Group for waiving MMR's payment default under the Third Amended Note of $1,131,852, and to Robert H. Lorsch, the Company's Chairman, Chief Executive Officer and President, in consideration of a personal guarantee. The increase was also driven by $1,959,472 of interest associated with the 12% Convertible Promissory Notes and attached warrants entered during 2009, as well as $200,000 of amortization of The RHL Group loan origination fee during 2009. There were no such fees during 2008.
49
Included in interest and other expenses, net, for the year ended December 31, 2009 were non-cash related expenses of approximately $2.0 million relating to interest on convertible notes payable, as well as approximately $1.9 million relating to the issuance of common stock and warrants in exchange for our vendors and other debt holders agreeing to continue to defer payment of their outstanding balances.
Net loss. As a result of the foregoing, we had a net loss of $10,295,661 for the twelve months ended December 31, 2009 compared to a net loss of $3,558,096 for the twelve months ended December 31, 2008.
Liquidity and Capital Resources
As of March 31, 2010, the Company's current liabilities exceeded its current assets by $8.9 million. Furthermore, during the three months ended March 31, 2010, the Company incurred losses of $6.5 million. At the current level of borrowing, the Company requires cash of $275,000 per year to service its debt. Furthermore, not including debt service, in order to continue operating its business, the Company uses an average of $225,000 cash per month, or $2.7 million per year. At this rate of cash burn, the Company's existing current assets will sustain the business for approximately five months.
In addition to the above cash burn from operations, the Company will be required to obtain additional financing in order to meet the obligations for installment payments of $621,000 under the Creditor Plan and our obligations under the subordinated secured indebtedness to The RHL Group (which note payable had a balance of $1,473,934 at March 31, 2010), among other debt obligations. Such obligations are currently due and payable pursuant to the terms of the note.
To finance its activities, the Company has relied on the issuance of stock and debt to the RHL Group. At December 31, 2009, the Company had a line of credit with the RHL Group in the amount of $3 million. Availability under this line of credit was $1.5 million. However, as part of the April 2009 Restructuring Agreement, The RHL Group is only obligated to advance an additional $100,000. Additionally, during 2010, the Company issued $1,142,300 of convertible debt. The Company expects to continue offering a limited amount of convertible debt during the remainder of 2010. The Company also expects sales from its new MMR Pro to generate revenue and gross profit that will significantly improve its monthly and annual cash burn from operations.
Cash Flows for the Three Months Ended March 31, 2010 compared to Three Months Ended March 31, 2009
Net cash used in operating activities for the three months ended March 31, 2010 was $1,087,749. The reconciliation of net loss of $6,463,358 to net cash used in operating activities for the three months ended March 31, 2010 included the following non-cash charges: $656,539 for share-based compensation, $2,168,480 for change in valuation of derivative liabilities, $1,971,363 for interest on the 12% Convertible Promissory Notes, $20,371 for depreciation and amortization, $352,437 for common stock issued as payment for services, $85,163 for warrants issued for services, and $21,250 for a third-party loan commitment fee amortization. We also had decreases in accounts receivables of $3,175, prepaid expenses and other current assets of $108,322, and increases in accounts payable and accrued expenses of $136,769 contributing to increased operating cash flow, offset by decreased operating cash flow of $29,022 for increases in inventory and $119,541 for decreases in related party payables.
Net cash used in operating activities for the three months ended March 31, 2009 was $1,105,361. The reconciliation of net loss of $3,876,390 to net cash used in operating activities for the three months ended March 31, 2009 included non-cash charges of $318,313 for share-based compensation and $2,150,831 for change in valuation of derivative liabilities, offset by a gain on the settlement of payables of $52,225, as well as increases of $615,266 in accounts payable and accrued expenses and $165,000 in restricted cash.
Our investing activities resulted in a net cash outflow of $20,005 for the three months ended March 31, 2010 relating to MMRPro software development spending. During the three months ended March 31, 2009, our investing activities resulted in a net cash inflow of $1,037,212 in connection with $1,050,506 in cash acquired from the Merger, offset by $6,924 in spending for the acquisition of property and equipment as well as $6,370 in capitalized website development costs.
50
Our financing activities resulted in a net cash inflow of $1,498,430 for the three months ended March 31, 2010 in connection with $1,142,300 in proceeds from the 12% Convertible Promissory Notes, $354,195 from warrant exercises and $3,779 from stock option exercises. During the three months ended March 31, 2009, our cash provided from financing activities was $17,866, which primarily represented borrowings from the line of credit with The RHL Group, Inc.
As of March 31, 2010, we had cash and cash equivalents of $878,442 compared to $25,496 at March 31, 2009.
Cash Flows for the Year Ended December 31, 2009 compared to Year Ended December 31, 2008
Net cash used in operating activities for the year ended December 31, 2009 was $2,416,980. The net loss for the year ended December 31, 2009 of $10,295,661 included non-cash operating activities of $2,819,637 for share based compensation, $1,951,522 for interest on the 12% Convertible Promissory Notes which were paid in the form of equity, $220,792 for loss on stock settlement of liabilities, $78,163 for depreciation and amortization, $100,000 for a write-off of advances due from a related party and $200,000 for The RHL Group loan commitment fee amortization. In addition, accounts payable and accrued expenses increased by $1,920,521 and related party payables increased by $1,304,916. These numbers are offset by a gain on the settlement of payables of $89,170 and $633,626 for change in valuation of derivative liabilities. Significant non-cash activities for the year ended December 31, 2009 included prepaid loan commitment fees for $230,000, prepaid expenses paid with common stock of $504,184, accounts payable paid with common stock of $323,533, the related party line of credit paid with common stock of $477,220 and related party payables paid with common stock of $254,846.
Net cash used in operating activities for the year ended December 31, 2008 was $1,789,861. The reconciliation of net loss of $3,522,476 to net cash used in operating activities for the year ended December 31, 2008 included increases of $258,047 in a related party payable and $483,537 in accounts payable and accrued expenses, and non-cash charges of $397,939 for restricted common stock issued for services, $148,777 for depreciation and amortization, and $197,000 for impairment of intangible assets.
Our investing activities resulted in a net cash inflow of $775,977 for the year ended December 31, 2009 in connection with $1,050,506 in cash acquired from the Merger, offset by $107,087 in spending for intangibles assets for international patent filing fee applications and $275,533 in investments in MMRPro website development costs. Our investing activities resulted in a net cash outflow of $1,340 for the year ended December 31, 2008 in connection with the purchase of property and equipment.
Our financing activities resulted in a net cash inflow of $2,170,522 for the year ended December 31, 2009 resulting from $211,000 in proceeds from the line of credit with The RHL Group, $125,000 in proceeds from an unrelated third-party promissory note, $1,373,000 in proceeds from the 12% Convertible Promissory Notes, $388,343 from warrant exercises and $76,316 from stock option exercises. Cash provided from financing activities for the year ended December 31, 2008 was $1,855,530. Financing activities primarily included proceeds generated from the sale of preferred stock and net proceeds from draw downs on our line of credit from The RHL Group, Inc., a significant stockholder wholly-owned by Robert H. Lorsch, our Chairman, Chief Executive Officer and President.
As of December 31, 2009, we had cash and cash equivalents of $487,766 compared to $75,779 at December 31, 2008.
Description of Indebtedness
The RHL Group
On April 29, 2009, the Company agreed to restructure our secured credit facility with The RHL Group and entered into a Secured Credit Restructuring Agreement with The RHL Group and Robert H. Lorsch, our Chairman, Chief Executive Officer and President (the "Restructuring Agreement"), issued The RHL Group a Third Amended and Restated Note (the "Third Amended Note"), and we agreed to guaranty MMR's obligations under the Third Amended Note (the "Guaranty").
51
The Third Amended Note amended and restated the August 2008 Second Amended and Restated Promissory Note Agreement (the "RHL Note"). The Third Amended Note matures November 30, 2009, and bears interest at the lesser of 10% or the highest rate then permitted by law, and is secured (similar to the previous RHL Note) by the Security Agreement. Although the reserve credit line has been increased to $3,000,000, The RHL Group is only obligated to make a minimum of $100,000 of loans, advances and guarantees under the Third Amended Note within 30 days of execution pursuant to the Restructuring Agreement. The RHL Group was due to receive, as an origination fee ("Fee"), a promissory note for $200,000, bearing interest at 10% per annum and due on demand. On August 17, 2009, the Company paid the $200,000 Fee by granting 2,800,000 shares of common stock to The RHL Group to the value of $364,000 and recorded a loss on issuance of $164,000. The Fee was recorded as a deferred financing cost and is being amortized over the Third Amended Note maturity period. As of December 31, 2009, the remaining unamortized origination fee was $0.
On August 18, 2009, the Company and The RHL Group entered into a Waiver Agreement, pursuant to which, in consideration of The RHL Group's waiver of MMR's payment default under the Third Amended Note, the Company granted to The RHL Group a warrant to purchase an aggregate of 11,039,378 shares of the Company's common stock, with an exercise price equal to $0.13 per share, the closing price of the Company's common stock on the date immediately preceding the date of grant. Under the Waiver Agreement, The RHL Group agreed to waive MMR's payment default until August 31, 2009, which waiver period will automatically continue until The RHL Group notifies MMR otherwise.
On August 17, 2009, in consideration of a personal guaranty given by Robert H. Lorsch, the Company's Chairman, Chief Executive Officer and President, to a vendor, to guarantee payments due to the vendor from the Company for services rendered, the Company granted Mr. Lorsch (i) a warrant to purchase 706,605 shares of Company common stock, at an exercise price of $0.13 per share, the closing price of the Company's common stock on the date immediately preceding the date of grant, and (ii) 141,321 shares of Company common stock.
The RHL Group Note payable had a balance of $1,473,934 at March 31, 2010. The components of the Group Note payable and the related balance sheet presentation as of March 31, 2010 are: (1) $976,924, which is included in the line of credit, related party; (2) $283,694, which is related to credit card charges and a vendor guaranty, is included in accounts payable and accrued expenses; and (3) $213,316, which is related to deferred salary and consulting expenses is included in related party payables. Total interest expense on this note for the three months ended March 31, 2010 and 2009 amounted to $36,304 and $20,434, respectively. The unpaid interest balance as of March 31, 2010 and December 31, 2009 was $81,458 and $45,155, respectively.
In addition, under the Restructuring Agreement, The RHL Group agreed to use commercially reasonable efforts to raise additional financing from third parties, and agreed to extend the maturity of the Third Amended Note for an additional six-month term if we are in full compliance with our covenants and other obligations under the Third Amended Note, on terms to be negotiated at such time. Additionally, Mr. Lorsch agreed to exercise all of his outstanding options prior to May 1, 2009 (which were exercised April 30, 2009), which resulted in an $113,220 reduction in principal owing The RHL Group. Finally, as a condition to agreeing to restructure our secured credit arrangement, we also terminated the Allonge. Thus, if at any time after June 1, 2009 we are not in compliance with our covenants under the Third Amended Note or Security Agreement, The RHL Group may, but is not obligated to, declare an event of default. In conjunction with the Third Amended Note, MMR was required to maintain certain financial covenants, including the requirement that MMR have at least $100,000 in cash in its bank accounts or such other amount as necessary to maintain operations through the subsequent thirty (30) days and timely pay any obligations due respecting payroll and all associated payroll taxes on and after June 1, 2009. As of March 31, 2010, MMR was in compliance with its financial covenants under the terms of the Third Amended Note.
On May, 12, 2010, MMR issued The RHL Group and Robert H. Lorsch, our Chairman, Chief Executive Officer and President, a Fourth Amended and Restated Secured Promissory Note (the "Fourth Amended Note"), and the Company agreed to guaranty MMR's obligations under the Fourth Amended Note (the "Guaranty"). The Fourth Amended Note amends and restates the April 29, 2009 Third Amended and Restated Secured Promissory Note Agreement (the "RHL Note"). The Fourth Amended Note matures April 29, 2011, and bears interest at the lesser of 10% or the highest rate then permitted by law, and is secured (similar to the previous RHL Note) by the Security Agreement. The reserve credit line of the Fourth Amended Noted remained at $3,000,000. In connection with the Fourth Amended Note, The RHL Group received a $200,000 loan origination fee ("Fee") due on May 12, 2010. In the event the Fourth Amended Note is renewed for an additional six-month term, The RHL Group is due an
52
additional $200,000 loan origination fee. In addition, The RHL Group received 2,614,684 warrants to purchase shares of the Company's common stock at an exercise price of $0.21 per share. The warrants are fully vested and be exercisable in cash or in a cashless exercise at any time prior to their fourth anniversary of issuance.
Promissory Notes
On October 1, 2008, concurrent with the execution of the term sheet for the Merger, Favrille advanced MMR $100,000 pursuant to a promissory note issued by MMR to Favrille dated September 30, 2008 (the "First Bridge Note"), which was to be used solely for paying MMR's out of pocket expenses incurred in connection with the Merger. On November 10, 2008 (the first business day after execution of the Merger Agreement), Favrille advanced MMR an additional $500,000 pursuant to a promissory note issued by MMR to Favrille (the "Second Bridge Note,") and on December 22, 2008, January 8, 2009 and January 15, 2009, Favrille advanced MMR an additional $100,000, $35,000 and $50,000, respectively, pursuant to promissory notes issued by MMR to Favrille (the "Supplemental Bridge Notes" and together with the First Bridge Note and the Second Bridge Note, the "Promissory Notes") in each case, solely for MMR's use in paying its out of pocket expenses incurred in connection with the Merger and its operating expenses. Principal outstanding under the Promissory Notes accrued interest at the applicable federal rate of interest per annum, or the maximum rate permissible by the laws in California relating to permissible rates of interest on commercial loans, whichever is less. Because the Merger occurred, the principal and interest under the Promissory Notes has automatically been treated as a contribution to capital to MMR and the Promissory Notes have been fully discharged in accordance with their terms.
On June 11, 2009, the Company entered into a promissory note agreement to borrow $125,000 with an unrelated third-party. The promissory note is due and payable July 30, 2011, and bears interest of 12% per annum payable quarterly beginning on January 1, 2010. The proceeds of the loan will be used by the Company for the filing of international patent applications to expand and perfect the patent rights of the Company in the Anti-CD20 Antibodies outside the United States. The unrelated third-party also received, as a commitment fee, $30,000, due and payable by August 15, 2009. The commitment fee was payable at the Company's option in cash or warrants to acquire 375,000 shares of our common stock at an exercise price per share equal to the lesser of (i) $0.15 per share and (ii) the weighted average trading price for a share of common stock for the ten consecutive trading days preceding the date on which the warrant is exercised. The warrants have a four year term. On July 29, 2009, we elected to pay the commitment fee through the issuance of warrants exercisable into 375,000 shares of our common stock at an exercise price of $0.14 per share. The warrants vested immediately. These warrants were valued using the Black-Scholes option pricing model. The total value of these warrants amounted to $32,399, all of which was recorded as a reduction to the commitment fee payable in July 2009, with a loss on settlement payables for $2,399.
On various dates between July 16, 2009 and March 17, 2010, the Company also entered into seventeen different Note and Warrant Subscription Agreements (the "Note Agreements") with eleven separate individuals (the "Note Purchasers"). Pursuant to the terms of the Note Agreements, these individuals purchased convertible notes in an aggregate amount of $2,623,175 (the "Notes"). The Notes carry an annual interest rate of 12%. The Notes are convertible at the option of the Note Purchasers into a number of shares of the Company's common stock, par value $0.001 per share, equal to the product of eighty percent (80%) multiplied by the arithmetic average of the dollar volume-weighted average price ("VWAP") of the Company's common stock for the ten (10) consecutive trading days ending on the day that is three (3) trading days prior to the applicable conversion date. As of March 31, 2010 all but two of the individuals converted their notes totaling $2,523,175 in exchange for a total of 35,649,729 shares of the Company's common stock.
Commitments and Contingencies
Under the terms of an agreement with an investor who purchased $500,000 of MMR's Series B Preferred shares in 2006, the Company entered into an agreement to invest $250,000 in a joint venture with the investor to establish an entity to market and sell the Company's services in the countries of Japan, China, Korea, Taiwan and Thailand. The Company has paid $100,000 to date of this amount which the Company expensed during the 3 months ended June 30, 2009. The Company has since terminated its relationship with the investor. To date, no joint venture has been formalized or incorporated and no operations have commenced. In September 2007, The RHL Group provided the investor with a guarantee that the Company would meet its obligations under this agreement in exchange for 300,000 shares of MMR common stock valued at $39,000. As consideration for renewing the guarantee when it expired in September 2008, in January 2009, MMR issued The RHL Group 100,000 shares of MMR common stock, which became 328,174 shares of MMRIS common stock upon the closing of the Merger,
53
valued at $5,000. This expense has been reflected in general and administrative expenses in the accompanying consolidated statement of operations for the twelve months ended December 31, 2009. On January 6, 2010, the Company entered into a settlement agreement with the investor to provide for full and final settlement of any and all claims pertaining to any license rights for MMR-Asia to market, sell and sublicense MMRIS and MMR products and services. As part of this settlement agreement, the Company granted 1,388,889 shares of common stock to the investor.
On August 17, 2009, Robert H. Lorsch, the Company's Chairman, Chief Executive Officer and President, agreed to guarantee $282,641in payments due to the vendor from the Company for services rendered. On August 17, 2009, in consideration of a personal guaranty given by Mr. Lorsch, the Company granted Mr. Lorsch (i) a warrant to purchase 706,605 shares of Company common stock, at an exercise price of $0.13 per share, the closing price of the Company's common stock on the date immediately preceding the date of grant, and (ii) 141,321 shares of Company common stock.
For additional information relating to this and other commitments and contingencies, please see the notes to our financial statements appearing elsewhere in this prospectus.
Off-Balance Sheet Arrangements
On January 4, 2010, we entered into a Cooperation Agreement and related documents (the "Cooperation Agreement") with Unis-TongHe Technology (Zhengzhou) Co., Ltd. ("Unis"). Under the Cooperation Agreement, we and Unis agreed to form a joint venture in China for the purpose of deploying our personal health record services and document imaging and management solutions in China. We will own 40% of the joint venture and Unis will own 60% and each party will have the right to designate two members of the joint venture's board of directors, with the fifth member being a Chinese citizen mutually designated by us and Unis. Under the Cooperation Agreement, board actions will require the approval of more than three of the five members of the joint venture's board of directors and no material actions may be taken unless all board members are present and voting at the meeting.
Under the Cooperation Agreement, we will contribute an aggregate of 100 million RMB to the joint venture, based on each party's respective ownership, in the form of intellectual property rights, equipment, brand value, cash and such other consideration as may be agreed to between the parties. Each party's obligation to contribute to the joint venture is subject to a number of conditions, including obtaining all necessary approvals of and licenses from the Chinese government, as well as the joint venture meeting its budget, goals and objectives at the time any contributions are due. Under the Cooperation Agreement, each party's contributions will be made over a period of sixty months.
The Company's entry into the Cooperation Agreement described above constitutes the creation of a direct financial obligation.
54
Executive officers and directors
Our executive officers and directors and their respective ages as of May 4, 2010 are as set forth below:
|
Name |
Age |
Position |
|
Robert H. Lorsch |
60 |
Chairman of the Board; President and |
|
Hector V. Barreto, Jr. |
49 |
Director, Compensation Committee Chair and Audit Committee |
|
David A. Boyden |
49 |
Director |
|
Douglas H. Helm |
68 |
Director, Nomination and Corporate Governance Committee Chair |
|
George Rebensdorf |
55 |
Director, Audit and Nomination and Corporate Governance Committee |
|
Bernard Stolar |
63 |
Director, Audit Committee Chair Compensation Committee |
|
Jack Zwissig |
61 |
Director, Compensation and Nomination and Corporate Governance Committee |
|
Naj Allana |
48 |
Executive Vice President of Technology and Product Development |
|
Ingrid Safranek |
37 |
Vice President of Finance and Chief Financial Officer |
Robert H. Lorsch, Chairman of the Board; President and Chief Executive Officer.
Since 2005, Mr. Lorsch has served as the Chairman, President and Chief Executive Officer of MMR Information Systems, Inc. He is also Chairman and Chief Executive Officer of The RHL Group, Inc., a private equity and business management consulting firm Mr. Lorsch formed in April 1998. In 1994, he co-founded SmarTalk TeleServices, Inc., leading the company through a successful public offering in 1996 and building it into one of the largest providers of prepaid telecommunications products and services, serving as its Chairman and Chief Executive Officer until February 1998, following which SmarTalk moved its headquarters from Los Angeles to Dublin, Ohio with different management following its acquisition of ConQuest Telecommunication Services Corp. The combined company's assets subsequently were liquidated and sold to AT&T in March 1999. In 1986, Mr. Lorsch founded the Lorsch Creative Network, a consulting company that developed marketing, advertising and interactive sales promotions campaigns for nationally and internationally recognized clients. In 1998 Lorsch Creative Network became The RHL Group, Inc. Mr. Lorsch also served on the Personal Health Record Steering Committee of the Healthcare Information and Management Systems Society from 2006 to 2007. Mr. Lorsch is a Member of the Board of Trustees of the California Science Center where the Robert H. Lorsch Family Pavilion stands as a gateway to the Science Center. He is also a Member of the Board of Governors, Cedars-Sinai Medical Center; Member of the Board and of the Executive Committee of D.A.R.E. America; and Member of the Board of the Sheriff's Youth Foundation, and has received numerous honors and awards, including D.A.R.E. America's "Future of America Award"; the Muscular Dystrophy Association's "Humanitarian of the Year Award"; and the Starlight Children's Foundation's "Golden Wish Award." Mr. Lorsch was also awarded the Private Sector Initiatives Citation, or C-Flag, from the White House during the Reagan Administration for his commitment to raising millions of dollars for financing state and local earthquake preparedness education. Following the Merger, Mr. Lorsch continues to serve as Chairman of the Board of MMR Information Systems, Inc. and acts as its President and Chief Executive Officer.55
We believe that Mr. Lorsch's qualifications to continue to serve on our Board of Directors include his 40 years' experience as chief executive officer of various successful corporations he founded, his knowledge in the area of business management consulting, his experience as member of numerous organization committees, his position as our current President and Chief Executive Officer, and his direct responsibility for all areas of our operations.
Hector V. Barreto, Jr., Director. Since July 2006, Mr. Barreto has been the owner and operator of the consulting company Barreto Associates. Mr. Barreto has also worked as a marketing and strategic planning advisor for our wholly-owned subsidiary MMR since August 2006. Prior to forming Barreto Associates, Mr. Barreto was unanimously approved by the U.S. Senate in July 2001 to serve as the Administrator of the Small Business Administration, a position he held until July 2006. From October 1995 until his appointment to the Small Business Administration in July 2001, Mr. Barreto worked as a broker dealer specializing in retirement plans for TELACU/Barreto Financial Services. Mr. Barreto currently serves on the California Commission for Economic Development and is the Chairman of The Latino Coalition, a non-profit, non-partisan organization based in Washington D.C. Prior to the Merger, Mr. Barreto served as a director of MMR Information Systems, Inc. Mr. Barreto received a B.A. in Business Administration from Rockhurst University.
We believe that Mr. Barreto's qualifications to continue to serve on our Board of Directors include his consulting experience, his knowledge in the area of marketing and strategic planning, and his 5 years' experience as the Administrator of the Small Business Administration.
David A. Boyden, Director. Mr. Boyden has been in the healthcare and life science industry for 25 years. Mr. Boyden currently serves as President of Mirador Strategies Inc., a company he founded in January 2005 to provide consulting services focusing on commercialization strategy for life science companies. Prior to founding Mirador, Mr. Boyden was a Senior Brand Director at Amgen, Inc., a biotechnology company that develops, manufactures and markets human therapeutics, where he worked from April 1991 to November 2004 in a number of senior sales and marketing positions. Mr. Boyden received a B.A. in Biochemistry from U.C. San Diego and a M.B.A. in Marketing from the Anderson School of Management at U.C.L.A.
We believe that Mr. Boyden's qualifications to continue to serve on our Board of Directors include his 25 years' experience in the healthcare and life science industry, his 5 years' experience as president of a consulting services company that focuses on commercialization strategy for life science companies, and his vast knowledge in the area of marketing.
Douglas H. Helm, Director. Mr. Helm is the managing member of Helm+Mann LLC, a global consultancy. From 2002 to 2009 he was associated with Employers Direct Corporation in Agoura Hills, California, serving in various capacities including marketing consultant, Vice President and Chief Marketing & Sales Officer of Employers Direct Insurance Company and Chief Operating Officer of its benefit subsidiary, Plenary Insurance Services, until its sale in Oct. 2009. Mr. Helm has over 40 years of experience in insurance and information services and has worked internationally in Russia, China and Europe. Mr. Helm majored in Labor Economics at the University of Washington and received a J.D. from Northwestern School of Law in 1973. Mr. Helm is a member of the Oregon State Bar.
We believe that Mr. Helm's qualifications to continue to serve on our Board of Directors include his 40 years' experience in insurance and information services, his experience in marketing and consulting, his experience in the positions he has held as vice president, chief marketing & sales officer and chief operating officer, and his legal background.
George Rebensdorf, Director. Since 1996, Mr. Rebensdorf has served as Chief Executive Officer of The Rebensdorf Group, Inc., a company providing management consulting, investment banking and financial advisory services to the telecommunications and emerging technology industries. Mr. Rebensdorf is also currently a managing member of E/W Capital, LLC, a financial advisory and investment banking firm. Prior to forming The Rebensdorf Group, from 1995 to 1996, Mr. Rebensdorf served as Senior Vice President, External Affairs of MIDCOM Communications, Inc., where he managed mergers, acquisition, and corporate finance matters for the company. From 1987 to 1995, Mr. Rebensdorf was a general partner of Telenational Communications, Ltd., a diversified telecom services provider, offering services in the United States, Europe, South America and Asia. Following the Merger, Mr. Rebensdorf continues to serve as a director of MMR Information Systems, Inc., a position he has held since 2005. Mr. Rebensdorf graduated magna cum laude from Arizona State University with a B.A. in English, and holds a J.D. from Creighton University School of Law.
We believe that Mr. Rebensdorf's qualifications to continue to serve on our Board of Directors include his experience as chief executive officer of a company that provides management consulting, his experience as managing member of an investment banking and financial advisory firm, and his legal background.
56
Bernard Stolar, Director. Mr. Stolar currently serves as a consultant to the video games industry and is a marketing and strategic planning advisor for our wholly-owned subsidiary, MMR. From February 2007 to September 2008, Mr. Stolar served as Games Industry Evangelist for Google, Inc., where his responsibilities included building in-game advertising. From February 2006 until its purchase by Google, Inc. in February 2007, Mr. Stolar was the Chairman of the Board of Adscape Media. Prior to this, from January 2002 to November 2002, Mr. Stolar was President and Chief Operating Officer of BAM! Entertainment, where he helped transform the company from a content provider for hand-held electronics into a developer and marketer of interactive entertainment for next generation video game consoles. From January 2000 until the division was sold in April 2001, Mr. Stolar served as President of Mattel Interactive, where he was responsible for all of Mattel's software, on-line and computer-enhanced toys. Mr. Stolar also served as President and Chief Operating Officer of Sega of America, Inc. from June 1996 to October 1999 and as an Executive Vice President with Sony Computer Entertainment of America from 1994 to June 1996. Following the Merger, Mr. Stolar continues to serve as a director of MMR Information Systems, Inc., a position he has held since 2005.
We believe that Mr. Stolar's qualifications to continue to serve on our Board of Directors include his marketing and strategic planning experience, his experience as chairman of the board of Adscape Media, and his experience as president and chief operating officer for various companies.
Jack Zwissig, Director. Since 1992, Mr. Zwissig has served as Chief Executive Officer of Zwissig and Associates, a consulting and executive leadership training firm that primarily concentrates on the designing and implementing of corporate culture change, including mergers and acquisitions. Throughout his tenure as head of Zwissig and Associates, Mr. Zwissig has offered consulting, marketing and advertising services to some of America's leading corporations and has led numerous corporate teambuilding workshops and seminars, both in the U.S. and abroad. Prior to the Merger, Mr. Zwissig served as a director of MMR Information Systems, Inc. Mr. Zwissig received a B.S. in Marketing and Management and a M.B.A. from Santa Clara University.
We believe that Mr. Zwissig's qualifications to continue to serve on our Board of Directors include his 18 years' experience as chief executive officer of a consulting and executive leadership training firm, his knowledge in the area of marketing and management, and his business background.
Naj Allana, Executive Vice President of Technology and Product Development. Mr. Allana served as Chief Financial Officer and Chief Technical Officer of MMR from August 2005 until December 2010, the date in which he was appointed to Executive Vice President of Technology and Product Development. Mr. Allana is also currently a partner with AMD Associates, LLC, a management consulting and temporary- staffing firm, where he has worked since February 2002. Prior to joining MMR, from April 2004 to May 2005, Mr. Allana was the Chief Financial Officer and Chief Operating Officer for Alterna, Inc., a manufacturer of high-end hair care products. Mr. Allana has 20 years of cross-industry experience with significant expertise in finance, accounting and information systems. Mr. Allana has a M.S. in Advanced Accounting and Costing from the University of Pune, India (formerly the University of Poona), and a M.B.A. from the U.C.L.A. Anderson School of Management. Prior to the Merger, Mr. Allana served as a director of MMR prior to the Company's merger with Favrille.
Ingrid Safranek, Vice President of Finance and Chief Financial Officer. Ms. Safranek was appointed as the Chief Financial Officer on December 17, 2009. She has been a Certified Public Accountant in California since 2006. She worked for Deloitte & Touche, LLP from 2002 to 2006, where she was part of the audit teams for large and small, private and public clients such as Computer Sciences Corporation, Infonet (later acquired by British Telecom), Candle! Corporation (later acquired by IBM), Primedia, Inc., Gold Circle Entertainment, and the Performing Arts Center, among others. Ms. Safranek's focus was on the technology, media and entertainment industries. She also owned Goldstein Enterprises, a management consulting firm that served numerous clients by providing them with business practices analyses and software application development in order to streamline day-to-day operations and maximize efficiency. Among her clients as owner of Goldstein Enterprises are Nestle USA, Warner Bros. Studios and RJR Fashion Fabrics. Ms. Safranek received a B.A. in Business Economics with a minor in Accounting from U.C.L.A.
57
The following table summarizes the compensation earned by each "named executive officer" of MMR and Favrille for the past two fiscal years, determined on the basis of rules adopted by the SEC relating to "smaller reporting companies."
Summary Compensation Table
|
Non-Equity |
All |
||||||||||||
|
Stock |
Option |
Incentive Plan |
Other |
||||||||||
|
Year |
Salary |
Awards (1) |
Awards (1) |
Compensation |
Compensation |
Totals |
|||||||
|
Robert Lorsch |
2009 |
$175,000 |
$387,372 |
(2) |
$1,978,330 |
(3) |
- |
$36,000 |
(4) |
$2,576,702 |
|||
|
President and |
2008 |
$120,000 |
- |
- |
- |
$380,082 |
(4) |
$500,082 |
|||||
|
Chief Executive Officer |
|||||||||||||
|
Naj Allana |
2009 |
$122,078 |
(5) |
$84,842 |
(6) |
- |
- |
$54,372 |
(7) |
$261,292 |
|||
|
EVP of Technology |
2008 |
$199,020 |
(5) |
$17,324 |
(6) |
- |
- |
$31,483 |
(7) |
$247,827 |
|||
|
and Product Development |
|||||||||||||
|
Ingrid Safranek |
2009 |
$3,000 |
- |
- |
- |
$0 |
$3,000 |
||||||
|
Chief Financial |
2008 |
$0 |
- |
- |
- |
$0 |
$0 |
||||||
|
Officer |
|||||||||||||
|
Richard Teich |
2009 |
$105,750 |
(8) |
$49,200 |
(9) |
- |
- |
$22,900 |
(10) |
$177,850 |
|||
|
2008 |
$101,625 |
(8) |
$9,100 |
(9) |
- |
- |
$19,981 |
(10) |
$130,706 |
- Option and stock awards above are disclosed at their aggregate grant date fair values as calculated under FASB ASC 718 (formerly SFAS 123(R)). Assumptions made for the purpose of computing these amounts are discussed in Note 2 of the Notes to the Consolidated Financial Statements included in our annual report on Form 10-K for the fiscal year ended December 31, 2009.
- Mr. Lorsch was granted 141,321 shares of restricted common stock on August 17, 2009 with a grant date fair value of $18,372 as compensation for a personal guarantee. Also, on January 27, 2009, The RHL Group, Inc., a California corporation wholly-owned by Mr. Lorsch, was issued 328,174 shares of restricted common stock with a grant date fair value of $5,000 as compensation for extension of a guarantee. In addition, The RHL Group, Inc. was granted 2,800,000 shares of restricted common stock on August 17, 2009 with a grant date fair value of $364,000 as payment of a loan origination fee.
- On August 17, 2009, Mr. Lorsch and The RHL Group, Inc. were granted an aggregate total of 11,745,983 warrants to purchase shares of the Company's common stock at an exercise price of $0.125 per share with an aggregate total grant date fair value of $1,131,852. This amount was recorded as interest expense during the year ended December 31, 2009, as the Company granted these warrants as consideration for Mr. Lorsch guarantying monies owed to a vendor and The RHL Group, Inc.'s waiver of MMR's payment default under the Third Amended Note. On August 9, 2009, Mr. Lorsch was granted 9,000,000 stock options to purchase shares of the Company's common stock at an exercise price of $0.125 per share with an aggregate total grant date fair value of $846,478.
58
- During the year ended December 31, 2009, other compensation includes $36,000 of auto allowance paid in 2009. During the year ended December 31, 2008, other compensation includes $283,082 of consulting fees paid to The RHL Group, Inc. For more information relating to the consulting agreement with The RHL Group, Inc., see Item 13 "Certain Relationships and Related Party Transactions, and Director Independence," $57,000 of auto allowance paid in 2008 (which includes $21,000 deferred in 2007), and approximately $40,000 of insurance premiums for 2008, including finance charges on the financing of the premium payments, on The RHL Group, Inc.'s Directors' & Officers' insurance policy, which supplements coverage under MMR's Directors' & Officers' policy.
- Mr. Allana received $122,078 and $114,300 in cash as salary for the years ended December 31, 2009 and 2008, respectively. We deferred payment of $10,275 and $84,720 for the years ended December 31, 2009 and 2008, respectively, until such payments could be made without jeopardizing our ability to continue as a going concern.
- During the year ended December 31, 2009, Mr. Allana was issued 707,016 shares of restricted common stock on August 6, 2009 with a grant date fair value of $84,842 as consideration for continuing to defer a portion of his salary. During the year ended December 31, 2008, Mr. Allana was issued 100,000 shares of restricted common stock on February 15, 2008 and 232,440 shares of restricted common stock on November 1, 2008 as consideration for continuing to defer a portion of his salary. The aggregate grant date fair value for restricted common stock issued in 2008 amounted to $17,324.
- During the year ended December 31, 2009, other compensation includes $41,450 in consulting fees for services rendered (paid on an hourly-fee basis) during the months of July through December 2009, when Mr. Allana's salary and time commitment were reduced to accommodate our cash situation, as well as $12,922 in interest earned on deferred compensation for 2009. During the fiscal year ended December 31, 2008, other compensation includes $15,000 in consulting fees for services rendered (paid on an hourly-fee basis) during the months of August through October 2008, when Mr. Allana's salary and time commitment were reduced to accommodate our cash situation. Of the $15,000 in fees, $3,750 were paid in cash and the balance of $11,250 remained unpaid as of December 31, 2008. Other compensation also includes $6,574 of interest earned on deferred compensation for 2008, as well as $10,000 of auto allowance (all of which was deferred).
- Mr. Teich, a key employee of the Company, received $105,750 and $81,625 in cash as salary for the years ended December 31, 2009 and 2008, respectively. We deferred payment of $2,693 and $20,000 for the years ended December 31, 2009 and 2008, respectively, until such payments could be made without jeopardizing our ability to continue as a going concern.
- During the year ended December 31, 2009, Mr. Teich was issued 410,000 shares of restricted common stock on August 6, 2009 with a grant date fair value of $49,200 as consideration for continuing to defer a portion of his salary. During the year ended December 31, 2008, Mr. Teich was issued 50,000 shares of restricted common stock on February 15, 2008 and 160,000 shares of restricted common stock on November 1, 2008 as consideration for continuing to defer a portion of his salary. The aggregate grant date fair value for restricted common stock issued in 2008 amounted to $9,100.
- During the year ended December 31, 2009, other compensation includes $18,011 in consulting fees for services rendered (paid on an hourly-fee basis) during the months of July through December 2009, when Mr. Teich's salary and time commitment were reduced to accommodate our cash situation, as well as $4,889 in interest earned on deferred compensation for 2009. During the year ended December 31, 2008, other compensation included $12,825 in consulting fees for services rendered (paid on an hourly-fee basis) during the months of June through October 2008, when Mr. Teich either received a reduced salary or did not receive a salary to accommodate our cash situation. Includes $5,188 of interest earned on deferred compensation for 2008, as well as a $2,000 auto allowance in 2008.
59
Outstanding Equity Awards at Fiscal Year-End
The following table provides information on all outstanding equity awards held by our named executive officers as of December 31, 2009. Information for MMR named executive officers is provided on an as converted basis as if the Merger had occurred prior to that time.
|
Option/Warrant Awards |
||||||||
|
Number of |
Equity Incentive |
Option/ |
Option/ |
|||||
|
Robert Lorsch |
11,745,983 |
- |
$0.125 |
8/18/2014 |
||||
|
President and |
2,700,000 |
6,000,000 |
$0.125 |
1/27/2014 |
||||
|
Chief Executive Officer |
||||||||
|
Naj Allana |
164,087 |
- |
$0.046 |
9/1/2011 |
||||
|
EVP of Technology and |
||||||||
|
Product Development |
||||||||
There were no outstanding equity awards for Ingrid Safranek, Vice President of Finance and Chief Financial Officer and Richard Teich, a key employee of the Company, as of December 31, 2009.
Employment Agreements
We have entered into an employment agreement with our Chairman, President and Chief Executive Officer, Robert H. Lorsch, with an initial term ending on December 31, 2011, subject to successive automatic extension unless we or Mr. Lorsch elect not to extend. Under the terms of his agreement, Mr. Lorsch shall serve as both our President and Chief Executive Officer and President and Chief Executive Officer of our wholly-owned subsidiary, MMR. The agreement provides for a base salary of $15,000 per month, subject to an upward increase and with an annual bonus and stock option grants in such amounts, if any, as the Board of Directors may determine in its sole discretion. Mr. Lorsch receives a monthly auto allowance, reimbursement of certain life insurance proceeds, and reimbursement for certain other insurance coverage, and is entitled to participate in benefits generally made to our senior executives.
Mr. Lorsch may terminate the agreement upon 30 days written notice without reason or for good reason (as defined in the agreements) if we fail to cure acts or omissions constituting good reason within 30 days. If Mr. Lorsch's employment is terminated by us for cause or voluntarily by Mr. Lorsch without good reason, he will not be entitled to receive any severance payments or benefits under the employment agreement. If Mr. Lorsch's employment is terminated by us without cause or voluntarily by Mr. Lorsch for good reason, Mr. Lorsch will be entitled to one year of salary at his then current rate of pay, including all monthly benefits, and the pro rata portion of the annual bonus otherwise due Mr. Lorsch. Mr. Lorsch is not entitled to any severance payments or benefits under the agreement if he is terminated for cause or voluntarily resigns without good reason. In the event of his disability, Mr. Lorsch would be entitled to receive compensation equal to 40% of his base salary as then in effect. Mr. Lorsch's employment agreement includes provisions that prohibit Mr. Lorsch from disclosing our confidential information and trade secrets and competing with us during the term of his employment agreement or soliciting our employees for 12 months following termination of employment.
60
We also have entered into a consulting agreement with The RHL Group, Inc., which is wholly-owned by Mr. Lorsch, that provides for a monthly fee of $25,000 plus reimbursement of expenses including medical insurance.
We also have approved an employment agreement with our former Chief Financial Officer and Senior Vice President and current Executive Vice President Technology and Product Development, Naj Allana. Under the terms of his proposed agreement, Mr. Allana shall serve both as our Chief Financial Officer and Senior Vice President and as Chief Financial Officer, Senior Vice President and Chief Technical Officer of our wholly-owned subsidiary, MMR. The agreement provides for a base salary of $15,845 per month, subject to an upward increase. In addition to his base salary, Mr. Allana is entitled to receive a commission with respect to certain of our accounts.
Mr. Allana's employment agreement was effective until February 15, 2010 and automatically renews for successive 12 month periods unless terminated at least 120 days prior to the end of the term. If we terminate Mr. Allana's employment, other than for misconduct (as defined in the employment agreement), we would continue to pay Mr. Allana his monthly salary for the remainder of the then current term, or if done in less than 120 days preceding the end of the term, then through the end of the next 12 month term.
On January 26, 2010, we entered into an employment agreement with Ingrid Safranek as our Vice President, Chief Financial Officer and Secretary. Under the employment agreement, Ms. Safranek receives a base salary, subject to annual increases as determined by the board of directors, certain benefits as set forth in the employment agreements, and an annual bonus at the discretion of the board of directors. Ms. Safranek's employment agreement is in effect until June 15, 2010, and shall automatically renew for successive one-year periods, unless extended or otherwise terminated at least 60 days prior to the end of the term.
If the Company terminates Ms. Safranek's employment, unless due to misconduct (as defined in the employment agreement), the Company must continue to pay Ms. Safranek her monthly salary for two (2) months, if such termination occurs within the first year of employment; for six (6) months, if such termination occurs between the first year and the last day of the second year of employment; and for twelve (12) months, if such termination occurs after the last day of the second year of employment. For purposes of clarification, a change in title or diminution of responsibilities, regardless of the level of materiality, shall not be considered a termination under the provisions of this Agreement.
61
Director Compensation
The following table sets forth certain information with respect to the compensation paid to our non-employee directors for the fiscal year ended December 31, 2009:
|
Fees |
|||||||||||||||||
|
or Paid in |
Option |
Stock |
Other |
||||||||||||||
|
Year |
Cash (1) |
Awards (2) |
Awards (2) |
Compensation (3) |
Totals |
||||||||||||
|
Hector V. Barreto, Jr. |
2009 |
$ |
26,000 |
$ |
151,188 |
(4) |
$ |
96,490 |
(10) |
$ |
50,000 |
$ |
323,678 |
||||
|
2008 |
$ |
- |
$ |
- |
$ |
15,250 |
(10) |
$ |
50,000 |
$ |
65,250 |
||||||
|
David A. Boyden |
2009 |
$ |
17,500 |
$ |
27,608 |
(5) |
$ |
- |
$ |
- |
$ |
45,108 |
|||||
|
2008 |
$ |
- |
$ |
- |
$ |
- |
$ |
- |
$ |
- |
|||||||
|
Douglas H. Helm |
2009 |
$ |
21,500 |
$ |
31,059 |
(6) |
$ |
- |
$ |
- |
$ |
52,559 |
|||||
|
2008 |
$ |
- |
$ |
- |
$ |
- |
$ |
- |
$ |
- |
|||||||
|
George Rebensdorf |
2009 |
$ |
22,500 |
$ |
243,007 |
(7) |
$ |
120,000 |
(11) |
$ |
34,133 |
$ |
419,640 |
||||
|
2008 |
$ |
- |
$ |
- |
$ |
21,250 |
(11) |
$ |
50,000 |
$ |
71,250 |
||||||
|
Bernard Stolar |
2009 |
$ |
18,000 |
$ |
156,019 |
(8) |
$ |
- |
$ |
55,868 |
$ |
229,887 |
|||||
|
2008 |
$ |
- |
$ |
- |
$ |
15,250 |
(12) |
$ |
51,025 |
$ |
66,275 |
||||||
|
Jack Zwissig |
2009 |
$ |
18,000 |
$ |
149,117 |
(9) |
$ |
55,000 |
(13) |
$ |
- |
$ |
222,117 |
||||
|
2008 |
$ |
- |
$ |
- |
$ |
22,500 |
(13) |
$ |
- |
$ |
22,500 |
||||||
________________
- All director fees earned during 2009 were deferred. As a result, none of the above fees were paid, and are reflected as a component of related party payables in the consolidated balance sheet as of December 31, 2009.
- Option and stock awards above are disclosed at their aggregate grant date fair values as calculated under FASB ASC 718 (formerly SFAS 123(R)). Assumptions made for the purpose of computing these amounts are discussed in Note 2 of the Notes to the Consolidated Financial Statements included in our annual report on Form 10-K for the fiscal year ended December 31, 2009.
- Other compensation represents additional fees earned by each respective director for consulting services performed.
- Mr. Barreto was granted 255,000 stock options to purchase shares of our common stock on May 21, 2009 with an exercise price of $0.179 per share, and 1,200,000 stock options on August 6, 2009 with an exercise price of $0.125 per share. The aggregate grant date fair value of these options amounted to $151,188.
- Mr. Boyden was granted 200,000 stock options to purchase shares of our common stock on May 21, 2009 with an exercise price of $0.179 per share. The aggregate grant date fair value of these options amounted to $27,608.
- Mr. Helm was granted 225,000 stock options to purchase shares of our common stock on May 21, 2009 with an exercise price of $0.179 per share. The aggregate grant date fair value of these options amounted to $31,059.
- Mr. Rebensdorf was granted 360,000 stock options to purchase shares of our common stock on May 21, 2009 with an exercise price of $0.179 per share, and 2,000,000 stock options on August 6, 2009 with an exercise price of $0.125 per share. The aggregate grant date fair value of these options amounted to $243,007.
62
- Mr. Stolar was granted 290,000 stock options to purchase shares of our common stock on May 21, 2009 with an exercise price of $0.179 per share, and 1,200,000 stock options on August 6, 2009 with an exercise price of $0.125 per share. The aggregate grant date fair value of these options amounted to $156,019.
- Mr. Zwissig was granted 240,000 stock options to purchase shares of our common stock on May 21, 2009 with an exercise price of $0.179 per share, and 1,200,000 stock options on August 6, 2009 with an exercise price of $0.125 per share. The aggregate grant date fair value of these options amounted to $149,117.
- During the year ended December 31, 2009, Mr. Barreto was granted an aggregate total of 830,379 shares of restricted common stock as payment for services, which had an aggregate total grant date fair value of $96,490. During the year ended December 31, 2008, Mr. Barreto was granted an aggregate total of 155,000 shares of restricted common stock as payment for services rendered, which had an aggregate grant date fair value of $15,250.
- During the year ended December 31, 2009, Mr. Rebensdorf was granted an aggregate total of 1,000,000 shares of restricted common stock as payment for services, which had an aggregate total grant date fair value of $120,000. During the year ended December 31, 2008, Mr. Rebensdorf was granted an aggregate total of 175,000 shares of restricted common stock as payment for services rendered, which had an aggregate grant date fair value of $21,250.
- During the year ended December 31, 2008, Mr. Stolar was granted an aggregate total of 155,000 shares of restricted common stock as payment for services, which had an aggregate total grant date fair value of $15,250.
- During the year ended December 31, 2009, Mr. Zwissig was granted an aggregate total of 508,857 shares of restricted common stock as payment for services, which had an aggregate total grant date fair value of $55,000. During the year ended December 31, 2008, Mr. Zwissig was granted an aggregate total of 250,000 shares of restricted common stock as payment for services rendered, which had an aggregate grant date fair value of $22,500.
Our Board of Directors currently consists of seven (7) persons and is divided into three classes, with each class serving a staggered three-year term. Pursuant to our Amended and Restated Certificate of Incorporation, our Class I directors are to be elected at our Annual Meeting, which is the first annual meeting of stockholders following the Merger. None of the individuals designated by us or Favrille, Inc. were members of our Board of Directors immediately prior to the Merger. Upon the consummation of the Merger, the following individuals were appointed to the three classes of directors with terms expiring as follows, with Robert H. Lorsch serving as Chairman:
- Douglas H. Helm - 2010 annual meeting
- Jack Zwissig - 2010 annual meeting
- David A. Boyden - 2011 annual meeting
- George Rebensdorf - 2011 annual meeting
- Hector V. Barreto, Jr. - 2012 annual meeting
- Bernard Stolar - 2012 annual meeting
- Robert H. Lorsch - 2012 annual meeting
Corporate Governance Principles
We are committed to maintaining the highest standards of business conduct and corporate governance. We have adopted a Code of Business Conduct and Ethics for our directors, officers and employees. Our Certificate of Incorporation, Bylaws and the Board of Directors committee charters provide additional framework for our corporate governance principles.
63
Our business, property and affairs are managed under the direction of the Board of Directors. The Board of Directors selects the senior management team, which is charged with the day-to-day operations of the Company's business. Members of the Board of Directors are kept informed of the Company's business through discussions with the Chief Executive Officer, other senior officers and the Company's counsel, by reviewing materials requested by them or otherwise provided to them and by participating in meetings of the Board of Directors and its committees. Having selected the senior management team, the Board of Directors acts as an advisor and counselor to senior management, monitors its performance and proposes or makes changes to the senior management team when it deems necessary or appropriate.
Board Leadership Structure and Role in Risk Oversight
Robert H. Lorsch holds the positions of Chairman, President, and Chief Executive Officer at our Company. He has held these positions since 2005, during which time he has served our Company well. We believe that it is in the best interests of our stockholders to have Mr. Lorsch serve as Chairman of our Board of Directors. Mr. Lorsch's combined role as Chairman and Chief Executive Officer allows for a clear focus for the chain of command to execute our Company's strategic plan. Pursuant to our bylaws, our Board of Directors determines how the powers of our Company shall be exercised and how our business shall be conducted. Thus, our Board of Directors has the necessary flexibility to determine whether the positions of Chairman and Chief Executive Officer should be held by the same person or by separate persons, based on our Company's leadership needs. Moreover, six of our seven directors are deemed independent under NASDAQ's listing standards. The independent directors and Mr. Lorsch effectively oversee our Company's management. Accordingly, we do not find it necessary to require our Chairman to be an independent director. Our corporate governance guidelines, along with Mr. Lorsch's track record in leading our Company for the last 5 years, ensure that our Company will continue to be served well by him serving as our Chairman, President, and Chief Executive Officer.
Our management is responsible for risk management on a day-to-day basis. Our Board of Directors oversees the risk management activities of management directly and through its committees by discussing with management the policies and practices utilized by management in assessing and managing risks and providing input on those policies and practices.
Independence of Directors
Although our common stock is no longer listed on the NASDAQ Global Market, our Board of Directors determined that each of the following directors would be deemed "independent" under NASDAQ Stock Market LLC rules: Messrs. Barreto, Boyden, Helm, Rebensdorf, Stolar and Zwissig. These persons represent a majority of our Board of Directors. Mr. Lorsch, the Chairman of our Board and our President and Chief Executive Officer would not be deemed independent. Messrs. Stolar, Barreto and Rebensdorf are members of our Compensation Committee and Mr. Barreto is the Chairman. Messrs. Lorsch, Rebensdorf and Stolar are members of our Executive Committee and Mr. Lorsch is the Chairman. Messrs. Helm, Zwissig and Rebensdorf are members of our Nominating and Corporate Governance Committee and Mr. Helm is the Chairman. Messrs. Stolar, Barreto and Rebensdorf are members of our Audit Committee but would not be deemed to be independent under the more stringent independence requirements for Audit Committee members set forth in NASDAQ Stock Market LLC Rule 5605(2). Mr. Stolar is Chairman of the Audit Committee.
Compensation Committee Interlocks and Insider Participation
No member of the Compensation Committee of our Board of Directors during fiscal year 2008 served as an officer, former officer or employee of us or any of our subsidiaries. During fiscal year 2009, no executive officer of us served as a member of the compensation committee of any other entity, one of whose executive officers served as a member of our Board of Directors or Compensation Committee, and no executive officer of us served as a member of the board of directors of any other entity, one of whose executive officers served as a member of our Compensation Committee.
64
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
The following table sets forth the beneficial ownership of our common stock as of April 19, 2010, by:
- each of our current directors;
- each of our current executive officers named in the Summary Compensation Table;
- all of our current directors and executive officers as a group; and
- each person or entity (or group of affiliated persons or entities) who is known by us to own beneficially more than 5% of the outstanding shares of common stock.
|
Name and Address of Beneficial Owner(1) |
Number of Shares of |
Percentage |
|
|
Directors and Named Executive Officers |
|||
|
Robert H. Lorsch (3) |
56,656,668 |
27.7% |
|
|
Hector V. Barreto, Jr. (4) |
2,734,192 |
1.3% |
|
|
David A. Boyden (5) |
69,094 |
* |
|
|
Douglas H. Helm (6) |
77,731 |
* |
|
|
George Rebensdorf (7) |
2,928,395 |
1.4% |
|
|
Bernard Stolar (8) |
1,865,904 |
* |
|
|
Jack Zwissig (9) |
2,677,866 |
1.3% |
|
|
Ingrid Safranek (10) |
625,000 |
* |
|
|
Naj Allana (11) |
3,843,864 |
1.9% |
|
|
All Executive Officers and Directors as a group |
71,478,714 |
35.0% |
|
|
(9 Persons) (12) |
|||
|
5% Stockholders |
|||
|
The RHL Group, Inc. (13) |
44,347,444 |
21.7% |
|
|
Robert H. Lorsch (14) |
12,309,224 |
6.0% |
|
|
David Loftus (15) |
25,049,347 |
12.3% |
|
|
Sherry Hackett (16) |
13,727,778 |
6.7% |
|
__________
*
- The business address of each director and executive officer listed is c/o MMR Information Systems, Inc., 2934@frac12; Beverly Glen Circle, Suite 702, Los Angeles, CA 90077.
- This table is based upon information supplied by officers, directors, principal stockholders, and Schedules 13D and 13G filed with the SEC. Beneficial ownership is determined in accordance with the rules of the SEC. Applicable percentage ownership is based on 202,314,696 shares of common stock outstanding as of December 31, 2009. Shares of common stock subject to options, warrants and convertible notes exercisable or convertible within 60 days after December 31, 2009, are deemed outstanding for computing the ownership percentage of the person holding such options, warrants or notes, but are not deemed outstanding for computing the ownership percentage of any other person. Except as otherwise noted, we believe that each of the stockholders named in the table have sole voting and investment power with respect to all shares of common stock shown as beneficially owned by them, subject to applicable community property laws.
65
- Consists of (i) 12,309,224 shares of common stock held directly by Mr. Lorsch and 44,347,444 shares of common stock held directly by The RHL Group, which is wholly owned and controlled by Mr. Lorsch, (ii) a fully vested warrant held by Mr. Lorsch to purchase 706,605 shares of common stock and a fully vested warrant held by The RHL Group, to purchase 11,039,378 shares of common stock, and (iii) stock options held by Mr. Lorsch to purchase 8,700,000 shares of common stock that are vested and exercisable within 60 days after December 31, 2009.
- Includes 532,180 shares subject to options exercisable within 60 days after December 31, 2009.
- Includes 64,455 shares subject to options exercisable within 60 days after December 31, 2009.
- Includes 72,512 shares subject to options exercisable within 60 days after December 31, 2009.
- Includes 1,062,923 shares subject to options exercisable within 60 days after December 31, 2009.
- Includes 543,460 shares subject to options exercisable within 60 days after December 31, 2009.
- Includes 691,433 shares subject to options exercisable within 60 days after December 31, 2009.
- Includes 50,000 shares subject to options exercisable within 60 days after December 31, 2009.
- Includes 164,087 shares subject to options exercisable within 60 days after December 31, 2009.
- Includes 23,627,032 shares subject to options and warrants exercisable within 60 days after December 31, 2009.
- Includes a fully vested warrant to purchase 11,039,378 shares of common stock.
- Includes stock options to purchase 8,700,000 shares of common stock that are vested and exercisable within 60 days after December 31, 2009 and a fully vested warrant to purchase 706,605 shares of common stock.
- Share purchased in conjunction with the 12% Convertible Promissory Notes.
- Share purchased in conjunction with the 12% Convertible Promissory Notes.
CERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONS
Certain Current Related-Person Transactions
Other than compensation agreements and other arrangements with our executive officers and directors and the transactions described below, during our last three fiscal years, there has not been, and there is not currently proposed, any transaction or series of similar transactions to which we were or will be a party in which the amount involved exceeded or will exceed the lesser of $120,000 or one percent of the average of our total assets at year-end for the last two completed fiscal years and in which any of our directors, nominees for director, executive officers, holders of more than five percent of any class of our voting securities or any member of the immediate family of the foregoing persons had or will have a direct or indirect material interest.
See "Management's Discussion and Analysis of Financial Condition and Results of Operations-Liquidity and Capital Resources- Description of Indebtedness" elsewhere in this prospectus for a description of certain of our debt financing transactions with The RHL Group.
Our President, Chairman and Chief Executive Officer, Robert H. Lorsch, is also the Chief Executive Officer of The RHL Group, Inc. and owns all of the capital stock of The RHL Group, Inc. Mr. Lorsch directly, and indirectly through The RHL Group, Inc., beneficially owns approximately 28.0% our total outstanding voting stock. The RHL Group, Inc. has loaned money to MMR pursuant to a secured note.
The RHL Group is an investment holding company which provides consulting, operational and technical services to the Company, which we refer to as the RHL Services. As part of the RHL Services, the RHL Group provides the Company with unrestricted access to its internal business and relationship contact database of more than 10,000 persons and entities, which includes clients of the RHL Group and other individuals which may hold value to the Company. The RHL Group also provides infrastructure support to the Company, including allowing the Company
66
unlimited access to its facilities, equipment, and data, information management and server systems. In addition to allowing the Company the use of its office support personnel, the RHL Group also has consented to allow the Company to utilize the full-time services of Mr. Lorsch as the Company's President, Chairman and Chief Executive Officer, which requires substantial time and energy away from his required duties as the RHL Group's Chairman and Chief Executive Officer. In addition, the RHL Group has made its President, Kira Reed Lorsch, available as the Company's spokesperson, for personal appearances, tradeshows and hosting the Company's product videos. Ms. Reed Lorsch also helps manage the Company's social networking activities.
As disclosed above, we have also entered into a consulting agreement with The RHL Group that provides for a monthly fee of $25,000 plus reimbursement of expenses including medical insurance.
During 2005 and 2006 we occupied space pursuant to a sublease from The RHL Group., pursuant to which we paid an aggregate $315,000 in rent in 2006. From August 2006 through August 2008, we leased space to The RHL Group at no cost at our former Santa Monica office space, which we have since vacated. From August 2008 to May 2009, we occupied space pursuant to a sublease from Robert H. Lorsch, for which we paid $3,000 per month plus utilities. The original term of the lease expired on April 15, 2009, and was renewed on a month-to-month basis until May 31, 2009 when we moved to our new offices in Beverly Hills. The owners of the new office space are not a related party.
We have consulting agreements with our current directors Hector V. Barreto, Jr. and Bernard Stolar pursuant to which Mr. Barreto and Mr. Stolar provide marketing and strategic planning advice and actively seek strategic partnerships and alliances with other entities to market our products. Under the terms of these agreements, we pay Mr. Barreto and Mr. Stolar $50,000 a year payable monthly, and commissions equal to 1% of all revenue generated through their efforts. The agreement with Mr. Barreto is effective until August 2009 and the agreement with Mr. Stolar is effective until November 2009. In the past, both Mr. Barreto and Mr. Stolar have received shares of our common stock as consideration for deferral of payment. Each agreement will automatically renew each successive year until terminated by either party upon 30 days prior written notice.
MMR also has a consulting agreement, as amended, with The Rebensdorf Group, Inc., or TRGI, which is owned by our current director George Rebensdorf. Mr. Rebensdorf also serves as its Chief Executive Officer. Pursuant to the agreement, TRGI provides financial advisory services and assists in negotiations in connection with our efforts to raise funds through private placement transactions. In addition to the initial retainer fee of $30,000 we paid upon execution of the original letter agreement, we have agreed to pay TRGI (a) a $4,167 monthly retainer (commencing May 1, 2009), (b) for transactions with parties introduced by TRGI, a success fee equal to 8% of the value (payable 4% in cash, 4% in warrants), (c) for material assistance in closing transactions with parties not introduced by TRGI, an advisory fee equal to 2% of the value (payable in cash), and (d) an annual grant of 100,000 options, vesting monthly over two-years from the grant date, with an exercise price equal to fair market value on the grant date. Payment of the monthly retainer may be deferred, in which case it would be payable to TRGI pro rata upon payment of any other deferred management or consulting fees. The letter agreement, as amended, is effective until terminated by either party, with or without cause, upon 10 days prior written notice.
In connection with the execution of an addendum to the letter agreement with TRGI on May 21, 2009, we granted Mr. Rebensdorf stock options to acquire 100,000 shares of our common stock at an exercise price of $0.179 per share, which options vest monthly over 2 years and expire on May 21, 2014.
The Company also incurred $31,172 and $0 during the three months ended March 31, 2010 and 2009, respectively, towards finder's fees from George Rebensdorf, a director. Included in related party payables at March, 31, 2010 and December 31, 2009 was $56,633 and $71,333, respectively, with respect to these services. In addition, on January 27, 2010, the Company granted Mr. Rebensdorf 4,000,000 options to purchase common stock in the company at an exercise price of $0.10 per share in consideration for director services. The options vest in three annual installments beginning January 27, 2011.
We also have an oral agreement with our current director Jack Zwissig to provide individual executive coaching services to our management team. Mr. Zwissig receives compensation in the form of stock as determined by our Board of Directors commensurate with the services performed. The agreement with Mr. Zwissig is on a month-to-month basis and continues until terminated by either party. In addition, on January 27, 2010, the Company granted Mr. Zwissig 2,000,000 options to purchase common stock in the company at an exercise price of $0.10 per share in consideration for director services. The options vest in three annual installments beginning January 27, 2011.
67
The Company incurred $73,868 and $51,025 during the year ended December 31, 2009 and 2008, respectively, towards marketing consulting services from Bernard Stolar, a director. Included in related party payables at December 31, 2009 and 2008 was $106,368 and $76,025, respectively, with respect to these services. In addition, on August 6, 2009, the Company granted the Mr. Stolar 1,200,000 options to purchase common stock in the company at an exercise price of $0.125 per share in consideration for consulting and other services. The options vest in eight quarterly installments beginning on the grant date.
The Company incurred $12,500 and $17,500 during the three months ended March 31, 2010 and 2009, respectively, towards marketing consulting services from Bernard Stolar, a director. Included in related party payables at March 31, 2010 and December 31, 2009 was $116,417 and $106,368, respectively, with respect to these services. In addition, on January 27, 2010, the Company granted the Mr. Stolar 4,000,000 options to purchase common stock in the company at an exercise price of $0.10 per share in consideration for director services. The options vest in three annual installments beginning January 27, 2011.
The Company also incurred $76,000 and $50,000 during the year ended December 31, 2009 and 2008, respectively, towards marketing consulting services from Hector Barreto, a director. Included in related party payables at December 31, 2009 and 2008 was $15,500 and $75,000, respectively, with respect to these services. In the first quarter of 2009, the Company entered into an agreement with The Latino Coalition, a non-profit organization in which Mr. Barreto is also the Chairman, to market the Company's product to its members. As of December 31, 2009, the Company has paid The Latino Coalition $16,000 as part of the agreement. In addition, on August 6, 2009, the Company granted Mr. Barreto 1,200,000 options to purchase common stock in the company at an exercise price of $0.125 per share in consideration for consulting and other services. The options vest in eight quarterly installments beginning on the grant date.
The Company also incurred $12,500 during the three months ended March 31, 2010 and 2009, towards marketing consulting services from Hector Barreto, a director. Included in related party payables at March, 31, 2010 and December 31, 2009 was $42,500 and $23,500, respectively, with respect to these services. In addition, on January 27, 2010, the Company granted the Mr. Barreto 4,000,000 options to purchase common stock in the company at an exercise price of $0.10 per share in consideration for director services. The options vest in three annual installments beginning January 27, 2011. Additionally, on January 21, 2010, the Company granted to Mr. Barreto 50,000 shares of common stock, valued at $5,500 in consideration for sales and marketing services. In the first quarter of 2009, the Company entered into an agreement with The Latino Coalition, a non-profit organization in which Mr. Barreto is also the Chairman, to market the Company's product to its members. During the three months ended March 31, 2010 and 2009, the Company paid $0 and $2,000, respectively, to The Latino Coalition for services.
In August 2006, the Company entered into a month-to-month lease with The RHL Group for the use of furniture and art for the Company's offices, for a total of $1,000 per month which terminated on August 15, 2008. The Company incurred expenses of $0 and $7,500 during the year ended December 31, 2009 and 2008, respectively.
In August 2008, the Company entered into an eight month lease with its Chairman and Chief Executive Officer for the use of office space for a total of $3,000 per month plus a share of utilities. This lease was renewed in April 2009 on a month-to-month basis and terminated in May 2009.
In February 2006, the Company entered into a letter agreement with MyMedicalRecords.com.au, or MMR AU granting it a 10 year exclusive right to market and sell the Company's products and services in Australia and New Zealand. The controlling shareholder of MMR AU was, at the time of the signing of the initial letter agreement, a shareholder of the Company. This letter agreement was subsequently formalized in the form of a ten year license agreement in October 2007, at which time the controlling shareholder of MMR AU was no longer a shareholder of the Company. The Company terminated the license agreement on September 30, 2009.
The Company received $50,000 from MMR AU upon the signing of the letter agreement in February 2006 towards set up costs of the platform to be used by customers of MMR AU. The Company completed the setup of the platform for MMR AU in 2007, at which time it started to recognize this amount into revenue. The Company also received $150,000 in 2007 from MMR AU towards the minimum guarantee payment for years 2 and 3 of the license term. This amount has been deferred and is being recognized as income based on the number of months elapsed. On September 30, 2009 the license agreement was terminated and the balance remaining in deferred revenue of $32,082 was recognized as license fee revenue. During the year ended December 31, 2009 and 2008, the Company amortized $48,332 and $84,375, respectively, of these amounts as license fee and other revenues in the accompanying statement of operations. The Company has accounted for this transaction under ASC 605-025 (formerly EITF 00-21, "Revenue Arrangements with Multiple Deliverables").
68
The Company contracts with a significant vendor for the development and maintenance of the software applications necessary to run our MyMedicalRecords PHR, MyESafeDepositBox and MyMedicalRecords Pro products. Our outside developer supports our software development needs through a team of software engineers, programmers, quality control personnel and testers, who work with our internal product development team on all aspects of application development, design, integration and support of our products. This vendor is also a shareholder. For the years ended December 31, 2009 and 2008, the total expenses relating to this shareholder amounted to $369,405 and $132,877, respectively. In addition, the Company capitalized software development costs for the years ended December 31, 2009 and 2008, of $272,532 and $0, respectively. As of December 31, 2009, the total amounts due to the shareholder and included in related party payables amounted to $617,796. For the three months ended March 31, 2010 and 2009, the total expenses relating to this shareholder amounted to $12,533 and $30,825, respectively. In addition, the Company capitalized $20,005 of software development costs for the three months ended March 31, 2009. As of March 31, 2010 and December 31, 2009, the total amounts due to the shareholder and included in related party payables amounted to $630,359 and $617,796, respectively.
On January 6, 2010, the Company entered into a settlement agreement with an MMR-Asia investor to provide for full and final settlement of any and all claims pertaining to any license rights for MMR-Asia to market, sell and sublicense MMRIS and MMR products and services. Refer to the Guarantee provided by The RHL Group section within footnote 10, Commitments and Contingencies for further details. As part of this settlement agreement, the Company granted 1,388,889 shares of common stock to the investor. The value of these shares, of $138,889, was included in related party payables as of December 31, 2009.
The Company incurred costs of $323,278 and $8,324 during the years ended December 31, 2009 and 2008, and $37,895 and $116,122 during the three months ended March 31, 2010 and 2009, respectively, toward consulting services from Audit Prep Services, LLC. Michael T. Psomas, member of Audit Prep Services, LLC, is a shareholder of MMRIS. At March 31, 2010 and December 31, 2009 the Company had $71,884 and $64,568 due to Audit Prep Services, LLC, which was recorded as a related party payable. At December 31, 2008, the Company had $8,324 due to Audit Prep Services, LLC, which was included in accounts payable. On March 8, 2009, the Company satisfied the payable of $8,324 to Audit Prep Services, LLC through the issuance of 100,000 options with a strike price of $0.175 to Mr. Psomas. On July 29, 2009, the Company issued an additional 304,826 options with a strike price of $0.125 and a value of $21,502 to Mr. Psomas in satisfaction of a portion of its liability to Audit Prep Services, LLC due at that time. All options vested immediately on the grant date. Additionally, on July 4, 2009 and September 4, 2009, the Company granted to Mr. Psomas 500,000 and 770,000 shares of common stock, respectively, valued at $137,000 in consideration for amounts due to Audit Prep Services, LLC at the times of issuance. On February 11, 2010, the Company granted to Mr. Psomas 674,000 shares of common stock, valued at $60,660 in consideration for amounts due to Audit Prep Services, LLC at the time of issuance.
On September 16, 2009, we entered into a Licensing and Sales Commission Agreement, or the Licensing Agreement with E-Mail Frequency, LLC, which we refer to as the Licensor, and David T. Loftus, which we refer to as the Consultant. The License Agreement was filed as Exhibit 10.1 to our Quarterly Report on Form 10-Q for the Quarter Ended September 30, 2009. Pursuant to the Licensing Agreement, we agreed to exclusively license from the Licensor the usage of the Licensor's direct marketing database of street addresses, cellular phone numbers, e-mail addresses and other comprehensive data, or the Database. In addition, we engaged the services of the Consultant to assist in its use of the Database. Under the terms of the Licensing Agreement, we paid a $250,000 one-time consulting fee to the Consultant in the form of 2,777,778 shares of restricted common stock, which we refer to as the Consulting Fee. In addition to the Consulting Fee, we will pay the Licensor a percentage of actual revenue received by us from successful sales made pursuant to use of the Database. The Licensing Agreement has a five-year term, but may be terminated by us after the Licensing Agreement has been in effect for one year after the effective date of the Licensing Agreement. Upon such termination, we will be obligated to pay the Licensor its fees owed under the Licensing Agreement for the remainder of the term in addition to eight times the total fees paid to the Licensor over the last three months of use of the Licensing Agreement. The $250,000 one-time licensee fee was recorded as a prepaid consulting fee and included in the prepaid expenses and other current assets as of December 31, 2009, less amortization of $12,500 included in operating expensed for the year ended December 31, 2009. Furthermore, during September and November 2009, the Company entered into 12% Convertible Promissory Notes with Mr. Loftus for a principal amount totaling $600,000 and warrants to purchase the Company's common stock. On the dates of the investments, Mr. Loftus immediately converted the 12% Convertible Promissory Notes into shares of common stock and exercised the attached warrants and received a total 13,410,962 shares of common stock.
69
In addition, the Company incurred a total of $0 during the three months ended March 31, 2010 and 2009, respectively, towards marketing services and consulting services from E-Mail Frequency, LLC and David Loftus. Included in related party payables at March 31, 2010 and December 31, 2009 was $12,533 and $50,577, respectively, in respect to these services. Furthermore, on January 6, 2010, the Company entered into 12% Convertible Promissory Notes with Mr. Loftus for a principal amount totaling $400,000 and warrants to purchase the Company's common stock. On the date of the investment, Mr. Loftus immediately converted the 12% Convertible Promissory Notes into shares of common stock and exercised the attached warrants and received a total 8,860,606 shares of common stock.
The securities above were issued to each of the foregoing in reliance upon exemptions from registration pursuant to Section 4(2) under the Securities Act of 1933, as amended, and the rules promulgated thereunder. At the time of their issuance, the securities granted above are restricted securities for purposes of the Securities Act and the certificates representing such securities shall bear legends to that effect. The exercise/conversion prices of the securities described above were equal to the closing price of our common stock as of the date of grant.
Creditor Plan Warrants
Pursuant to the terms and conditions of the Creditor Plan, on January 27, 2009, we issued warrants to acquire 9,999,992 shares of our common stock at an exercise price of $0.12 per share, which expire on January 26, 2014 to certain of our former officers, former directors and their affiliates who were willing to take such equity as partial or full payment for outstanding liabilities. This includes warrants to acquire an aggregate 4,222,834 shares of our common stock at an exercise price of $0.12 per share, which expire on January 26, 2014 to John Longenecker, Ph.D, Tamara Seymour, and Dan Gold our former Chief Executive Officer, former Chief Financial Officer and formed Chief Scientific Officer, respectively.
This section summarizes certain of our authorized and outstanding securities and certain of the provisions of our amended and restated certificate of incorporation and our amended and restated bylaws, each as amended to date.
Description of capital stock
The following description of our capital stock and provisions of our certificate of incorporation and bylaws are summaries and are qualified by reference to our certificate of incorporation and bylaws, respectively, copies of which have been filed with the SEC and are incorporated by reference herein.
General background
Our certificate of incorporation authorizes the issuance of up to 650,000,000 shares of common stock with a $0.001 par value and 5,000,000 preferred shares with a par value of $0.001. As of May 4, 2010, we had issued and outstanding 210,752,146 shares of common stock, held by approximately 3,536 stockholders of record.
Common stock
Except as required by law, holders of our common stock are entitled to vote on all matters as a single class, and each holder of common stock is entitled to one vote for each share of common stock owned. Holders of common stock do not have cumulative voting rights.
Holders of our common stock are entitled to receive ratably any dividends that may be declared by the board of directors out of legally available funds, subject to any preferential dividend rights of any outstanding preferred stock. Upon any liquidation, dissolution, or winding up of the Company, holders of our common stock are entitled to share ratably in all assets remaining available for distribution to stockholders after the payment of all debts and other liabilities and subject to the prior rights of any outstanding preferred stock.
70
Holders of our common stock have no preemptive, subscription, redemption or conversion rights. Our outstanding shares of common stock are validly issued, fully paid and nonassessable. The rights, preferences and privileges of holders of common stock are subject to, and may be adversely affected by, the rights of holders of shares of any series of preferred stock which we may designate and issue in the future without further stockholder approval.
Our common stock is not currently traded on any securities exchange and instead is quoted on the OTC Bulletin Board under the symbol "MMRF.OB."
Preferred stock
Our board of directors is authorized without further stockholder approval to issue from time to time up to an aggregate of 5,000,000 shares of preferred stock in one or more series and to fix or alter the designations, preferences, rights, qualifications, limitations or restrictions of the shares of each series, including the dividend rights, dividend rates, conversion rights, voting rights, term of redemption including sinking fund provisions, redemption price or prices, liquidation preferences and the number of shares constituting any series or designations of such series without further vote or action by the stockholders. No shares of preferred stock have been issued or are outstanding.
The issuance of preferred stock may have the effect of delaying, deferring or preventing a change in control of the Company without further action by the stockholders and may adversely affect the market price of and voting, economic and other rights of, holders of our common stock.
Anti-takeover effects of provisions of Delaware Law and our Charter and Bylaws
We are subject to the provisions of Section 203 of the DGCL. In general, Section 203 prohibits a publicly-held Delaware corporation from engaging in a "business combination" with an "interested stockholder" for a period of three years after the date of the transaction in which the person became an interested stockholder, unless either:
- prior to the date at which the person becomes an interested stockholder, the board of directors approves such transaction or business combination;
- the stockholder acquires more than 85% of the outstanding voting stock of the corporation (excluding shares held by directors who are officers or held in certain employee stock plans) upon consummation of such transaction; or
- the business combination is approved by the board of directors and by the holders of two-thirds of the outstanding voting stock of the corporation (excluding shares held by the interested stockholder) at a meeting of stockholders (and not by written consent).
For purposes of Section 203, "interested stockholder" is a person who, together with affiliates and associates, owns (or within three years prior, did own) 15% or more of the corporation's voting stock. A "business combination" includes a merger, asset sale or other transaction resulting in a financial benefit to such interested stockholder.
71
In addition, certain provisions of our certificate of incorporation and bylaws may be deemed to have an anti-takeover effect and may delay, defer or prevent a tender offer or takeover attempt that a stockholder might consider in its best interest, including those attempts that might result in a premium over the market price for the shares held by our stockholders. These provisions include:
- dividing our Board of Directors into three classes serving staggered three-year terms;
- authorizing our Board of Directors to issue preferred stock without stockholder approval;
- prohibiting cumulative voting in the election of directors;
- prohibiting stockholder actions by written consent;
- limiting the persons who may call special meetings of stockholders;
- prohibiting our stockholders from making certain changes to our certificate of incorporation or bylaws except with 66.7% stockholder approval; and
- requiring advance notice for raising business matters or nominating directors at stockholders' meetings.
The combination of the charter and bylaws provisions summarized above will make it more difficult for our existing stockholders to replace our board of directors as well as for another party to obtain control of us by replacing our board of directors. Since our board of directors has the power to retain and discharge our officers, these provisions could also make it more difficult for existing stockholders or another party to effect a change in management. In addition, the authorization of undesignated preferred stock makes it possible for our board of directors to issue preferred stock with voting or other rights or preferences that could impede the success of any attempt to change our control.
These provisions may have the effect of deterring hostile takeovers or delaying changes in our control or management. These provisions are intended to enhance the likelihood of continued stability in the composition of our board of directors and in the policies they implement, and to discourage certain types of transactions that may involve an actual or threatened change of our control. These provisions are designed to reduce our vulnerability to an unsolicited acquisition proposal. The provisions also are intended to discourage certain tactics that may be used in proxy fights. However, such provisions could have the effect of discouraging others from making tender offers for our shares and, as a consequence, they also may inhibit fluctuations in the market price of our shares that could result from actual or rumored takeover attempts. Such provisions may also have the effect of preventing changes in our management.
Transfer agent and registrar
The transfer agent and registrar for our common stock is BNY Mellon Shareowner Services LLC.
72
The purpose of this prospectus is to permit the selling stockholder to offer and resell up to 60,000,000 shares of our common stock at such times and at such places as it chooses. To the extent required, we may amend and supplement this prospectus from time to time to describe a specific plan of distribution. The decision to sell any shares offered pursuant to this prospectus is within the sole discretion of the selling stockholder.
The distribution of the common stock by the selling stockholder may be effected from time to time in one or more transactions. Any of the common stock may be offered for sale, from time to time, by the selling stockholder at prices and on terms then obtainable, at fixed prices, at prices then prevailing at the time of sale, at prices related to such prevailing prices, or in negotiated transactions at negotiated prices or otherwise. The common stock may be sold by one or more of the following:
• On the OTC Bulletin Board or any other national common stock exchange or automated quotation system on which our common stock is traded, which may involve transactions solely between a broker-dealer and its customers which are not traded across an open market and block trades.
• Through one or more dealers or agents (which may include one or more underwriters), including, but not limited to:
• Block trades in which the broker or dealer as principal and resale by such broker or dealer for its account pursuant to this prospectus.
• Purchases by a broker or dealer as principal and resale by such broker or dealer for its account pursuant to this prospectus.
• Ordinary brokerage transactions.
• Transactions in which the broker solicits purchasers.
• Directly to one or more purchasers.
• A combination of these methods.
Dutchess and any broker-dealers who act in connection with the sale of its shares are "underwriters" within the meaning of the Securities Act, and any discounts, concessions or commissions received by them and profit on any resale of the shares as principal may be deemed to be underwriting discounts, concessions and commissions under the Securities Act. Because the selling stockholder is an "underwriter" within the meaning of the Securities Act, it will be subject to the prospectus delivery requirements of the Securities Act, including Rule 172 thereunder.
The selling stockholder or its underwriters, dealers or agents may sell the common stock to or through underwriters, dealers or agents, and such underwriters, dealers or agents may receive compensation in the form of discounts or concessions allowed or reallowed. Underwriters, dealers, brokers or other agents engaged by the selling stockholder may arrange for other such persons to participate. Any fixed public offering price and any discounts and concessions may be changed from time to time. Underwriters, dealers and agents who participate in the distribution of the common stock may be deemed to be underwriters within the meaning of the Securities Act, and any discounts or commissions received by them or any profit on the resale of shares by them may be deemed to be underwriting discounts and commissions thereunder. The proposed amounts of the common stock, if any, to be purchased by underwriters and the compensation, if any, of underwriters, dealers or agents will be set forth in a prospectus supplement.
73
Unless granted an exemption by the SEC from Regulation M under the Exchange Act, or unless otherwise permitted under Regulation M, the selling stockholder will not engage in any stabilization activity in connection with our common stock, will furnish each broker or dealer engaged by the selling stockholder and each other participating broker or dealer the number of copies of this prospectus required by such broker or dealer, and will not bid for or purchase any common stock of our or attempt to induce any person to purchase any of the common stock other than as permitted under the Exchange Act.
We will not receive any proceeds from the sale of these shares of common stock offered by the selling stockholder. We shall use our reasonable efforts to prepare and file with the SEC such amendments and supplements to the registration statement and this prospectus as may be necessary to keep such registration statement effective and to comply with the provisions of the Securities Act with respect to the disposition of the common stock covered by the registration statement for the period required to effect the distribution of such common stock.
We are paying certain expenses (other than commissions and discounts of underwriters, dealers or agents) incidental to the offering and sale of the common stock to the public. If we are required to update this prospectus during such period, we may incur additional expenses in excess of the amount estimated above. We have agreed to indemnify the selling stockholders against certain losses, claims, damages and liabilities, including liabilities under the Securities Act and the Exchange Act, subject to certain exceptions.
In order to comply with certain state securities laws, if applicable, the common stock will be sold in such jurisdictions only through registered or licensed brokers or dealers. In certain states the shares of common stock may not be sold unless they have been registered or qualified for sale in such state or an exemption from registration or qualification is available and is complied with.
CHANGES AND DISAGREEMENTS WITH ACCOUNTANTS
On December 22, 2009, the Company selected Rose, Snyder & Jacobs, or RSJ as the Company's new independent registered public accounting firm for the Company. The Company requested, and RSJ provided, a reaudit of the prior year's financial statements in addition to the completion of the 2009 audit of the Company's financial statements. The Company requested such reaudit in order to be able to request from RSJ permission to use such audited financial statements in the Company's future filings with the Commission. The engagement of RSJ was approved by the Audit Committee and subsequently ratified by the Board. During the Company's fiscal years ended December 31, 2007 and December 31, 2008 and through December 18, 2009, the Company did not consult with RSJ regarding any of the matters or events set forth in Item 304(a)(2)(i) or Item 304(a)(2)(ii) of Regulation S-K.
Also on December 22, 2009, the Company dismissed SingerLewak LLP, or Singer as the Company's independent registered public accounting firm. This action was approved by the Audit Committee of the Board and subsequently ratified by the Board.
During the fiscal years ended December 31, 2007, and December 31, 2008, respectively, and in the subsequent interim periods ending September 30, 2009 and through December 22, 2009, there were no disagreements between the Company and Singer on any matter of accounting principles or practices, financial statement disclosure or auditing scope or procedure, which disagreements, if not resolved to the satisfaction of Singer, would have caused Singer to make reference to the subject matter of the disagreement in their reports on the financial statements for such years.
Selected legal matters with respect to the validity of the securities offered by this prospectus will be passed upon for us by Stradling Yocca Carlson & Rauth, a Professional Corporation, Newport Beach, California.
The financial statements as of and for the years ended December 31, 2009 and 2008, which are included in this prospectus, have been audited by Rose, Snyder & Jacobs, an independent registered public accounting firm, as stated in their report appearing herein. Such financial statements have been so included in reliance upon the report of such firm given upon their authority as experts in accounting and auditing.
74
WHERE YOU CAN FIND MORE INFORMATION
We have filed a registration statement on Form S-1 with the SEC for the stock offered pursuant to this prospectus. This prospectus does not contain all of the information set forth in the registration statement and the exhibits and schedules thereto. Statements contained in this prospectus as to the contents of any contract or other document referred to are not necessarily complete and in each instance reference is made to the copy of such contract or other document filed as an exhibit to the registration statement, each such statement being qualified in all respects by such reference. For further information with respect to us and the common stock offered hereby, please refer to the registration statement and its exhibits and schedules for further information relating to us and our common stock.
We are subject to the information and periodic reporting requirements of the Securities Exchange Act of 1934 and in accordance therewith file reports, proxy statements and other information with the SEC. Such reports, proxy statements, other information and a copy of the registration statement may be inspected by anyone without charge and copies of these materials may be obtained upon the payment of the fees prescribed by the SEC, at the Public Reference Room maintained by the SEC at Room 1580, 100 F Street, N.E., Washington, D.C. 20549. You may obtain information on the operation of this public reference room by calling 1-800-SEC-0330. The Registration Statement, including all exhibits and schedules and amendments, has been filed with the SEC through the Electronic Data Gathering Analysis and Retrieval system and is available to the public from the SEC's web site at http://www.sec.gov.
75
MMR INFORMATION SYSTEMS, INC.
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
|
|
Page |
|
|
|
|
Audited Annual Financial Statements of MMR Information Systems, Inc. |
|
|
Report of Independent Registered Accounting Firm |
F-2 |
|
Consolidated Balance Sheets at December 31, 2008 and 2009 |
F-3 |
|
Consolidated Statements of Operations for the Years Ended December 31, 2008 and 2009 |
F-4 |
|
Consolidated Statements Stockholders' Equity (Deficit) for the Years Ended December 31, 2008 and 2009 |
F-5 |
|
Consolidated Statements of Cash Flows for the Years Ended December 31, 2008 and 2009 |
F-6 |
|
Notes to Consolidated Financial Statements |
F-7 |
|
Unaudited Interim Consolidated Financial Statements of MMR Information Systems, Inc. |
|
|
Consolidated Balance Sheets at March 31, 2010 (unaudited) and December 31, 2009 |
F-36 |
|
Consolidated Statements of Operations — Three Months Ended March 31, 2010 (unaudited) and March 31, 2009 (unaudited) |
F-37 |
|
Consolidated Statement of Stockholders' Deficit — Three Months Ended March 31, 2010 (unaudited) |
F-38 |
|
Consolidated Statements of Cash Flows — Three Months Ended March 31, 2010 (unaudited) and March 31, 2009 (unaudited) |
F-39 |
|
Notes to Consolidated Financial Statements (unaudited) |
F-40 |
F-1
REPORT OF INDEPENDENT REGISTERED ACCOUNTING FIRM To the Board of Directors and Shareholders of We have audited the accompanying consolidated balance sheets of MMR Information Systems, Inc. (a Delaware
corporation) and Subsidiary as of December 31, 2009 and 2008, and the related consolidated statements of operations,
stockholders' deficit and cash flows for the years then ended. These consolidated financial statements are the
responsibility of the Company's management. Our responsibility is to express an opinion on these consolidated financial
statements based on our audit. We conducted our audit in accordance with the standards established by the Public Company Accounting Oversight
Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance
about whether the financial statements are free of material misstatement. The Company is not required to have, nor
were we engaged to perform, an audit of its internal control over financial reporting. Our audit included consideration of
internal control over financial reporting as a basis for designing audit procedures that are appropriate in the
circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company's internal control
over financial reporting. Accordingly, we express no such opinion. An audit also includes examining, on a test basis,
evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used
and significant estimates made by management, as well as evaluating the overall financial statement presentation. We
believe that our audit provides a reasonable basis for our opinion In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the
consolidated financial position of MMR Information Systems, Inc. and Subsidiary as of December 31, 2009 and 2008,
and the consolidated results of their operations and their cash flows for the years then ended, in conformity with
accounting principles generally accepted in the United States of America. The accompanying consolidated financial statements have been prepared assuming that the
Company will continue as a going concern. As discussed in Note 1 to the financial statements, the Company has
incurred significant operating losses and negative cash flows from operations during the year ended December 31,
2009. These conditions raise substantial doubt about the Company's ability to continue as a going concern.
Management's plans regarding those matters also are described in Note 1. The financial statements do not include any
adjustments that might result from the outcome of this uncertainty /s/ Rose, Snyder & Jacobs Rose, Snyder & Jacobs Encino, California March 29, 2010 F-2
MMR INFORMATION SYSTEMS, INC. The accompanying notes are an integral part of these financial statements F-3
MMR INFORMATION SYSTEMS, INC. The accompanying notes are an integral part of these financial statements F-4
MMR INFORMATION SYSTEMS, INC. The accompanying notes are an integral part of these financial statements F-5
MMR INFORMATION SYSTEMS, INC. The accompanying notes are an integral part of these financial statements F-6
MMR INFORMATION SYSTEMS, INC. 1. NATURE OF OPERATIONS AND BASIS OF PRESENTATION On January 27, 2009, MMR Information Systems, Inc. or MMRIS, (formerly known as Favrille, Inc.), a Delaware corporation formed
in January 2000, and hereinafter referred to as the Company) consummated a transaction with MyMedicalRecords, Inc.
(formerly known as mymedicalrecords.com, Inc., a Delaware corporation formed in 2005, or MMR) through a merger of a wholly-owned
subsidiary of MMRIS with and into MMR pursuant to the terms of the Agreement and Plan of Merger and Reorganization dated
November 8, 2008, or the Merger). Pursuant to the terms of the Merger, all of the outstanding common and preferred stock of MMR
was cancelled and the former stockholders of MMR received an aggregate of 79,812,116 shares of MMRIS common stock. In addition,
MMRIS assumed the obligations of MMR under its outstanding stock options and warrants, which, pursuant to the terms of the Merger,
represented the right to receive an aggregate 12,787,080 shares of MMRIS common stock at the effective time of the Merger. In connection
with the Merger, MMR became a wholly-owned subsidiary of the Company, with the former stockholders of MMR collectively owning shares
of the Company's common and preferred stock representing approximately 60.3% of the voting power of the Company's outstanding capital
stock. For accounting purposes, the Merger was treated as a reverse acquisition with MMR being the accounting acquirer. Accordingly, the
historical financial results prior to the Merger are those of MMR. The results of operations for MMRIS are included in the Company's
consolidated financial results beginning on January 27, 2009. The presentation of consolidated statements of stockholders' deficit reflects the historical stockholders' deficit of MMR
through January 26, 2009. The effect of the issuance of shares of MMRIS common stock in connection with the Merger and the inclusion of
MMRIS's outstanding shares of common stock at the time of the Merger on January 27, 2009 is reflected in the year ended December
31, 2009. The Company, headquartered in Los Angeles, California, seeks to empower consumers and medical professionals by facilitating access
to consumer medical records and associated vital documents (such as living wills, birth certificates and insurance policies). The Company
offers consumers an easy-to-use, secure web-based product that allows documents, images and voice mail messages to be transmitted in
and out of the Company's proprietary system using a variety of methods, including fax, file upload and email. Principles of Consolidation The consolidated financial statements include the accounts of MMRIS, and its wholly-owned subsidiary MMR. All intercompany
transactions have been eliminated upon consolidation. Basis of Presentation and Going Concern The accompanying financial statements are prepared in accordance with U.S. generally accepted accounting principles, or GAAP. On July 1, 2009, MMRIS adopted Financial Accounting Standards Board, or FASB, Accounting Standards
Codification, or ASC 105-10 (formerly Statement of Financial Accounting Standards, or SFAS, No. 168, The FASB Accounting Standards
Codification and Hierarchy of Generally Accepted Accounting Principles, a replacement of FASB Statement No. 162). ASC 105-10
establishes the FASB ASC as the sole source of authoritative accounting principles recognized by the FASB to be applied in preparation of
financial statements in conformity with generally accepted accounting principles in the United States of America. New accounting standards
issued subsequent to June 30, 2009 are communicated by the FASB through Accounting Standards Updates, or ASU's. The ASC is
effective for interim and annual reporting periods ending after September 15, 2009. The Company adopted ASC 105-10 during
the year ended December 31, 2009 and revised its referencing of GAAP accounting standards in these financial statements to reflect the new
standards. F-7
Going Concern As of December 31, 2009, the Company's current liabilities exceeded its current assets by $7.2 million. Furthermore, during the year
ended December 31, 2009, the Company incurred losses of $10.3 million. At December 31, 2009 and December 31, 2008, the Company had $487,776 and $75,779, respectively, in cash and cash equivalents.
Historically, the Company issued capital stock and received funds from The RHL Group, Inc. (a significant shareholder wholly-owned by the
Company's Chairman and Chief Executive Officer) to operate its business. Although, the Company received additional funding from The RHL
Group pursuant to the Third Amended and Restated Note dated April 29, 2009, it nevertheless will still be required to obtain additional
financing in order to meet installment payment obligations resulting from settlement payments with various creditors, which we refer to as the
Creditor Plan and the previously existing obligations under the subordinated secured indebtedness to The RHL Group, which note payable
had a balance of $1,545,767 at December 31, 2009. As a result of the above, there is uncertainty about the Company's ability to continue as a going concern. Management's plan regarding this matter is to continue to utilize its available line of credit with The RHL Group (see Note 3). At
December 31, 2009, there was approximately $1,450,000 available under The RHL Group line of credit. Additionally, the Company plans to
continue to sell additional debt and equity securities, continue to settle its existing liabilities through issuance of equity securities, explore
other debt financing arrangements, continue to increase its existing subscriber and affiliate customer base and sell MMR Pro products to
obtain additional cash flow over the next twelve months. There can be no assurance that funds from these sources will be available when
needed or, if available, will be on terms favorable to us or to our stockholders. If additional funds are raised by issuing equity securities, the
percentage ownership of our stockholders will be reduced, stockholders may experience additional dilution or such equity securities may
provide for rights, preferences or privileges senior to those of the holders of our common stock. If the Company is unable to utilize its available line of credit with The RHL Group, or obtain suitable alternative debt financing, it may
adversely affect the Company's ability to execute its business plan and continue as a going concern. 2. SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (a) MANAGEMENT'S USE OF ESTIMATES The preparation of financial statements in accordance with GAAP requires management to make estimates and assumptions that
affect the reported amounts of assets and liabilities at the date of the financial statements and the reported amounts of net revenue and
expenses during each reporting period. On an ongoing basis, management evaluates its estimates, including those related to revenue
recognition, allowances for doubtful accounts, the valuation of deferred income taxes, tax contingencies, long-lived and intangible assets,
valuation of derivative liabilities and stock-based compensation. These estimates are based on historical experience and on various other
factors that it believes to be reasonable under the circumstances. Actual results could differ from those estimates. (b) CASH AND CASH EQUIVALENTS The Company considers cash equivalents to be only those investments that are highly liquid, readily convertible to cash and with
maturities of 90 days or less at the purchase date. The Company maintains its cash in bank deposit accounts which, at times, may exceed
federally insured limits. The Company has not experienced any losses in such accounts and believes it is not exposed to any significant credit
risk on cash. (c) TRADE AND OTHER RECEIVABLES Receivables represent claims against third parties that will be settled in cash. The carrying value of receivables, net of an allowance
for doubtful accounts, if any, represents their estimated net realizable value. The allowance for doubtful accounts, if any, is estimated based
on historical collection trends, type of customer, the age of outstanding receivables and existing economic conditions. If events or changes in
circumstances indicate that specific receivable
F-8
balances may be impaired, further consideration is given to the collectability of those balances
and the allowance is adjusted accordingly. Past due receivable balances are written-off when collection efforts have been unsuccessful in
collecting the amount due. (d) FAIR VALUE OF FINANCIAL INSTRUMENTS ASC 820-10 (formerly SFAS No. 157, Fair Value Measurements ) requires entities to disclose the fair value of financial
instruments, both assets and liabilities recognized and not recognized on the balance sheet, for which it is practicable to estimate fair value.
ASC 820-10 defines the fair value of a financial instrument as the amount at which the instrument could be exchanged in a current
transaction between willing parties. As of December 31, 2008 and 2009, the carrying value of accounts receivable, related party payables and
receivables, deposits, accounts payable and accrued expenses, compensation payable, severance liabilities, and deferred revenues
approximates fair value due to the short-term nature of such instruments. The carrying value of short-term debt approximates fair value as the
related interest rates approximate rates currently available to the Company. The Company utilizes ASC 820-10 for valuing financial assets and liabilities. ASC 820-10 defines fair value, establishes a framework for
measuring fair value and generally accepted accounting principles and expands disclosures about fair value measurements. This standard
applies in situations where other accounting pronouncements either permit or require fair value measurements. ASC 820-10 does not require
any new fair value measurements. ASC 820-10 also establishes a fair value hierarchy which requires an entity to maximize the use of observable inputs and minimize the
use of unobservable inputs when measuring fair value. The standard describes three levels of inputs that may be used to measure fair value: Level 1: Quoted prices in active markets for identical or similar assets and liabilities. Level 2: Quoted prices for identical or similar assets and liabilities in markets that are not active or observable inputs other than
quoted prices in active markets for identical or similar assets and liabilities. Level 3: Unobservable inputs that are supported by little or no market activity and that are significant to the fair value of the
assets or liabilities. In determining the appropriate fair value of the option, warrant and conversion feature liabilities, the Company used
Level 3 inputs for its valuation methodology. The Company applied the Black-Scholes model to value the derivative, option, warrant and
conversion feature liabilities. See Note 12 for the inputs used in the Black-Scholes option valuation model. F-9
(e) PROPERTY AND EQUIPMENT Property and equipment are stated at cost. Equipment under capital leases is stated at the present value of the minimum lease
payments. Depreciation is calculated using the straight-line method, based upon the following estimated useful lives: Furniture and Fixtures: 5 Years Computer Equipment: 5 Years When items are retired or disposed of, income is charged or credited for the difference between the net book value of the asset and the
proceeds realized thereon. Expenditures for maintenance and repairs are charged to operations as incurred while renewals and betterments are capitalized. All property and equipment, along with all other assets of the Company, are pledged as collateral for a line of credit from a related party
(see Note 3 - Related Party Note Payable). (f) INTANGIBLE ASSETS Intangible assets are comprised of website development costs, domain names and patents. The Company accounts for website
development costs in accordance with ASC 350-50 (formerly Emerging Issues Task Force ("EITF") No. 00-2, "Accounting for Website
Development Costs") and ASC 985-20 (formerly SFAS No. 86 "Accounting for the Costs of Computer Software to Be Sold, Leased, or
Otherwise Marketed"). Pursuant to ASC 350-50 and 985-20, the Company capitalizes internally developed website costs when the
website under development has reached technological feasibility. These costs are amortized, typically over an estimated life of five years,
using the larger of the amount calculated using the straight-line method or the amount calculated using the ratio between current period gross
revenues and the total of current period gross revenues and estimated future gross revenues. At each balance sheet date, the Company
evaluates the unamortized capitalized website costs compared to the net realizable value. The amount by which the unamortized capitalized
website costs exceed its net realizable value is written off. During the year ended December 31, 2008, we concluded that our capitalized
website development costs were impaired by $197,000, which has been included in general and administrative expenses in the
accompanying statements of operations. During the year ended December 31, 2009, there were no impairment charges recorded. The Company accounts for domain names and patents in accordance with ASC 350-30 and ASC 360 (formerly SFAS No. 142,
"Goodwill and Other Intangible Assets"). The Company capitalizes patent costs representing legal fees associated with filing patent
applications and amortizes them on a straight-line basis. The Company is in the process of evaluating the patents' estimated useful life and
will begin amortizing the patents when they are brought to the market. Identifiable intangible assets are amortized over their estimated useful
lives as follows: Website Development Costs: 5 Years Domain Name: 5 Years (g) IMPAIRMENT OF LONG-LIVED ASSETS AND INTANGIBLES The Company accounts for long-lived assets, which include property and equipment and identifiable intangible assets with finite
useful lives (subject to amortization), in accordance with ASC 350-30 (formerly SFAS No. 144 "Accounting for the Impairment or Disposal
of Long-Lived Assets"). ASC 350-30 requires that long-lived assets be reviewed for impairment whenever events or changes in
circumstances indicate that the carrying amount of an asset may not be recoverable. Recoverability is measured by comparing the carrying
amount of an asset to the expected future undiscounted net cash flows generated by the asset. If it is determined that the asset may not be
recoverable, or if the carrying amount of an asset exceeds its estimated future undiscounted cash flows, an impairment charge is recognized
to the extent of the difference between the fair value and the asset's carrying amount. The Company had no impairment charges during the
years ended December 31, 2009 and 2008. F-10
(h) REVENUE RECOGNITION The Company's revenues are derived from services, which are comprised of providing electronic access to consumer medical
records and other vital documents and from the licensing of its services. Revenue is recognized only when the price is fixed or determinable,
persuasive evidence of an arrangement exists, the service is performed, and collectability of the resulting receivable is reasonably
assured. The Company's subscriber revenues consist of annual and monthly recurring retail subscriptions and usage-based fees, which are
primarily paid in advance by credit card, and corporate accounts that are based on either an access-fee or actual number of users, and in
each case billed in advance at the beginning of each month of service. The Company defers the portions of annual recurring subscription
fees collected in advance and recognizes them on a straight line basis over the subscription period. The Company grants exclusive licenses for the sale and marketing of its services in international territories in consideration of an up-front
license fee and an ongoing royalty. The royalty fee is usually a percentage of revenue earned by the licensee and there usually are certain
minimum guarantees. License fee revenues received in advance from international licensees for the grant of the license are deferred and
recognized over the period covered by the agreement. Minimum guaranteed royalty payments received in advance are deferred and
recognized over the period to which the royalty relates. All such revenues are included under "License Fees and Other." In those cases
where a license agreement contains multiple deliverables, the agreement is accounted for in accordance with ASC 605-025 (formerly EITF
00-21, "Revenue Arrangements with Multiple Deliverables"). (i) INCOME TAXES AND UNCERTAIN TAX POSITIONS The Company accounts for income taxes in accordance with ASC 740-10 (formerly SFAS No. 109,
"Accounting for Income Taxes"). The Company recognizes deferred tax assets and liabilities to reflect the
estimated future tax effects, calculated at the tax rate expected to be in effect at the time of realization. A valuation
allowance related to a deferred tax asset is recorded when it is more likely than not that some portion of the deferred tax
asset will not be realized. Deferred tax assets and liabilities are adjusted for the effects of the changes in tax laws and
rates of the date of enactment. ASC 740-10 prescribes a recognition threshold that a tax position is required to meet before being recognized in the financial statements
and provides guidance on recognition, measurement, classification, interest and penalties, accounting in interim periods, disclosure and
transition issues. The Company classifies interest and penalties as a component of interest and other expenses. To date, there have been no
interest or penalties assessed or paid. The Company measures and records uncertain tax positions by establishing a threshold for the financial statement recognition and
measurement of a tax position taken or expected to be taken in a tax return. Only tax positions meeting the more-likely-than-not recognition
threshold at the effective date may be recognized or continue to be recognized. (j) ADVERTISING The Company expenses advertising costs as incurred. Advertising expense for the periods ended December 31, 2009 and 2008
was $808 and $22,960, respectively. (k) SHARE-BASED COMPENSATION The Company accounts for share-based compensation in accordance with ASC 718-20 (formerly SFAS No. 123(R), "Share
Based Payments"). The Company applies ASC 718-20 in accounting for stock-based awards issued to employees in order to recognize
compensation expense related to the fair value of employee share-based awards, including stock options and restricted stock. Determining
the fair value of options at the grant date requires judgment, including estimating the expected term that stock options will be outstanding
prior to exercise, the associated volatility and the expected dividends. Judgment is also required in estimating the amount of share-based
F-11
awards expected to be forfeited prior to vesting. If actual forfeitures differ significantly from these estimates, share-based compensation
expense could be materially impacted. Options issued to non-employees are accounted for in accordance with ASC 505-50 (formerly EITF No. 96-18 "Accounting for Equity
Instruments That Are Issued to Other Than Employees for Acquiring, or in Conjunction with Selling, Goods or Services," and
EITF No. 00-18 "Accounting Recognition for Certain Transactions involving Equity Instruments Granted to Other Than Employees"). Options issued
to non-employees are treated the same as those issued to employees with the exception of determination of the measurement date. The
measurement date is the earlier of (1) the date at which a commitment for performance by the counterparty to earn the equity instruments is
reached, or (2) the date at which the counterparty's performance is complete. Options granted to consultants are valued at their respective
measurement dates, and recognized as expense based on the portion of the total consulting services provided during the applicable period.
As further consulting services are provided in future periods, the Company will revalue the associated options and recognize additional
expense based on their then current values. The fair value of each stock option is estimated on the date of grant using the Black-Scholes option pricing model. Assumptions are
determined at the time of grant. Grants of stock options during the years ended December 31, 2009 and 2008 were valued using the
following assumptions. The assumptions used in the Black-Scholes models are based upon the following data: (1) The Company uses the contractual life of the
underlying non-employee warrants as the expected life; the expected life of the employee options used in this calculation is the period of time
the options are expected to be outstanding and has been determined based on historical exercise experience. (2) In the absence of an
extensive public market for the Company's shares, the expected stock price volatility of the underlying shares over the expected term of the
option or warrant was taken at approximately the mid-point of the range of historical volatilities of similar companies at the various grant
dates. (3) The risk free interest rate is based on published U.S. Treasury Department interest rates for the expected terms of the underlying
options or warrants. (4) Expected dividends are based on historical dividend data and expected future dividend activity. (5) The expected
forfeiture rate is based on historical forfeiture activity and assumptions regarding future forfeitures based on the composition of current
grantees. (l) NET INCOME/LOSS PER SHARE The Company applies the guidance of ASC 260-010 (formerly SFAS No. 128, "Earnings Per Share") for calculating the basic
and diluted loss per share. Basic loss per share is calculated by dividing the net loss attributable to common shareholders by the weighted
average number of common shares outstanding. Diluted loss per share is computed similar to basic loss per share except that the
denominator is increased to include the number of additional common shares that would have been outstanding if the potential shares had
been issued and if the additional shares were dilutive. Common equivalent shares are excluded from the computation of net loss per share if
their effect is anti-dilutive. All potential common shares were excluded from the computation of diluted net loss per common share for the years ended December
31, 2009 and 2008 because they were anti-dilutive due to the Company's net loss position. Stock options, warrants and convertible notes
excluded from the computation totaled 54,647,822 shares for the year ended December 31, 2009 and 12,130,784 shares for the year ended
December 31, 2008. F-12
(m) RESEARCH, DEVELOPMENT AND ENGINEERING Research, development and engineering costs are expensed as incurred and presented as technology development in the
accompanying consolidated statements of operations. Costs for software development relating to our website incurred subsequent to
establishing technological feasibility, in the form of a working model, are capitalized and amortized over their estimated useful lives. (n) CONCENTRATIONS For the year ended December 31, 2009 and 2008, there were four and three customers, respectively, representing a combined
75.2% and 70.3%, respectively, of the Company's revenue. (o) RECENT ACCOUNTING PRONOUNCEMENTS In August 2009, the FASB issued ASU No. 2009-05, Measuring Liabilities at Fair Value,
which provides additional guidance on how companies should measure liabilities at fair value under ASC 820. The ASU clarifies that the
quoted price for an identical liability should be used. However, if such information is not available, a entity may use, the quoted price of an
identical liability when traded as an asset, quoted prices for similar liabilities or similar liabilities traded as assets, or another valuation
technique (such as the market or income approach). The ASU also indicates that the fair value of a liability is not adjusted to reflect the
impact of contractual restrictions that prevent its transfer and indicates circumstances in which quoted prices for an identical liability or quoted
price for an identical liability traded as an asset may be considered level 1 fair value measurements. The ASU is effective
October 1, 2009. The adoption of this standard did not have a material impact on the Company's financial statements. In September 2009, the FASB issued ASU No. 2009-12, Investments in Certain Entities That Calculate
Net Asset Value per Share (or Its Equivalent), that amends ASC 820 to provide guidance on measuring the fair value of certain
alternative investments such as hedge funds, private equity funds and venture capital funds. The ASU indicates that, under certain
circumstance, the fair value of such investments may be determined using net asset value (NAV) as a practical expedient, unless it is
probable the investment will be sold at something other than NAV. In those situations, the practical expedient cannot be used and disclosure
of the remaining actions necessary to complete the sale is required. The ASU also requires additional disclosures of the attributes of all
investments within the scope of the new guidance, regardless of whether an entity used the practical expedient to measure the fair value of
any of its investments. This ASU is effective October 1, 2009. The adoption of this standard did not have a material impact on the
Company's financial statements. In October 2009, the FASB issued ASU No. 2009-13, Multiple-Deliverable Revenue Arrangements — a
consensus of the FASB Emerging Issues Task Force, that provides amendments to the criteria for separating consideration in
multiple-deliverable arrangements. As a result of these amendments, multiple-deliverable revenue arrangements will be separated in more
circumstances than under existing U.S. GAAP. The ASU does this by establishing a selling price hierarchy for determining the selling price of
a deliverable. The selling price used for each deliverable will be based on vendor-specific objective evidence if available, third-party evidence
if vendor-specific objective evidence is not available, or estimated selling price if neither vendor-specific objective evidence nor third-party
evidence is available. A vendor will be required to determine its best estimate of selling price in a manner that is consistent with that used to
determine the price to sell the deliverable on a standalone basis. This ASU also eliminates the residual method of allocation and will require
that arrangement consideration be allocated at the inception of the arrangement to all deliverables using the relative selling price method,
which allocates any discount in the overall arrangement proportionally to each deliverable based on its relative selling price. Expanded
disclosures of qualitative and quantitative information regarding application of the multiple-deliverable revenue arrangement guidance are
also required under the ASU. The ASU does not apply to arrangements for which industry specific allocation and measurement guidance
exists, such as long-term construction contracts and software transactions. The ASU is effective beginning January 1,
2011. The Company is currently evaluating the impact of this standard on the Company's financial statements. F-13
In October 2009, the FASB issued ASU No. 2009-14, Certain Revenue Arrangements That Include Software Elements —
a consensus of the FASB Emerging Issues Task Force, that reduces the types of transactions that fall within the current scope of
software revenue recognition guidance. Existing software revenue recognition guidance requires that its provisions be applied to an entire
arrangement when the sale of any products or services containing or utilizing software when the software is considered more than incidental
to the product or service. As a result of the amendments included in ASU No. 2009-14, many tangible products and services that rely
on software will be accounted for under the multiple-element arrangements revenue recognition guidance rather than under the software
revenue recognition guidance. Under the ASU, the following components would be excluded from the scope of software revenue recognition
guidance: the tangible element of the product, software products bundled with tangible products where the software components and
non-software components function together to deliver the product's essential functionality, and undelivered components that relate to
software that is essential to the tangible product's functionality. The ASU also provides guidance on how to allocate transaction consideration
when an arrangement contains both deliverables within the scope of software revenue guidance (software deliverables) and deliverables not
within the scope of that guidance (non-software deliverables). The ASU is effective beginning January 1, 2011. The Company is
currently evaluating the impact of this standard on the Company's financial statements. In January 2010, the FASB issued ASU No. 2010-6, Improving Disclosures About Fair Value Measurements, that
amends existing disclosure requirements under ASC 820 by adding required disclosures about items transferring into and out of levels 1 and
2 in the fair value hierarchy; adding separate disclosures about purchase, sales, issuances, and settlements relative to level 3
measurements; and clarifying, among other things, the existing fair value disclosures about the level of disaggregation. This ASU is effective
for the first quarter of 2010, except for the requirement to provide level 3 activity of purchases, sales, issuances, and settlements on a gross
basis, which is effective beginning the first quarter of 2011. The Company is currently evaluating the impact of this standard on the
Company's financial statements. In June 2009, the FASB issued additional guidance related to ASC 860, Transfers and Servicing, which seeks to
improve the relevance and comparability of the information that a reporting entity provides in its financial statements about transfers of
financial assets; the effects of the transfer on its financial position, financial performance, and cash flows; and a transferor's continuing
involvement, if any, in transferred financial assets. ASC 860 eliminates the concept of a qualifying special-purpose entity, creates more
stringent conditions for reporting a transfer of a portion of a financial asset as a sale, clarifies other sale-accounting criteria, and changes the
initial measurement of a transferor's interest in transferred financial assets. ASC 860 is effective for interim and annual reporting periods
beginning after November 15, 2009. The Company has not completed its evaluation of ASC 860, but does not expect the adoption to
have a material impact on its financial statements. In June 2009, the FASB issued additional guidance related to ASC 810, Consolidation, which requires an enterprise to determine
whether its variable interest or interests give it a controlling financial interest in a variable interest entity. The primary beneficiary of a variable
interest entity is the enterprise that has both (1) the power to direct the activities of a variable interest entity that most significantly
impact the entity's economic performance, and (2) the obligation to absorb losses of the entity that could potentially be significant to the
variable interest entity or the right to receive benefits from the entity that could potentially be significant to the variable interest entity. ASC
810 also amends FASB Interpretation No 46(R) to require ongoing reassessments of whether an enterprise is the primary beneficiary of a
variable interest entity. ASC 810 is effective for interim and annual reporting periods beginning after November 15, 2009. The
Company has not completed its evaluation of ASC 810, but does not expect the adoption to have a material impact on its financial
statements. In May 2009, the FASB issued additional guidance related to ASC 855, Subsequent Events. ASC 855 establishes that
management must evaluate, as of each reporting period, events or transactions that occur for potential recognition or disclosure in the
financial statements and the circumstances under which an entity should recognize events or transactions occurring after the balance sheet
date through the date that the financial statements are issued or available to be issued. It also requires the disclosure of the date through
which an entity has evaluated subsequent events. The Company adopted ASC 855 for the period ending June 30, 2009. The required
disclosures are included in Note 16, "Subsequent Events". F-14
In April 2009, the FASB issued additional guidance related to ASC 820, Fair Value Measurements and Disclosures. ASC 820
provisions define fair value, establish a framework for measuring fair value and expand disclosure requirements. The new guidance
emphasizes that even if there has been a significant decrease in the volume and level of activity, the objective of a fair value measurement
remains the same. Fair value is the price that would be received to sell an asset or paid to transfer a liability in an orderly transaction (that is,
not a forced liquidation or distressed sale) between market participants. The guidance provides a number of factors to consider when
evaluating whether there has been a significant decrease in the volume and level of activity for an asset or liability in relation to normal market
activity. In addition, when transactions or quoted prices are not considered orderly, adjustments to those prices based on the weight of
available information may be needed to determine the appropriate fair value. The guidance also requires increased disclosures. The
Company adopted the new guidance for the period ending June 30, 2009, and it did not have a material impact on the Consolidated Financial
Statements. The required disclosures are included in Note 2, "Fair Value of Financial Instruments". In April 2009, the FASB issued additional guidance related to ASC 825, Financial Instruments. ASC 825 establishes additional
disclosure requirements of fair values for certain financial instruments in the interim financial statements. The Company adopted the new
guidance for the period ending June 30, 2009. The required disclosures are included in Note 2, "Fair Value of Financial
Instruments". 3. RELATED PARTY NOTE PAYABLE On July 31, 2007, MMR entered into a promissory note agreement and a security agreement with The RHL Group, a corporation wholly
owned by Robert H. Lorsch, the Chairman and CEO of the Company, to borrow up to $100,000 under a revolving line of credit. This
agreement was subsequently superseded by an amended and restated promissory note on August 23, 2007 under the terms of which the line
amount was increased to $1 million. On April 29, 2009, the Company agreed to restructure MMR's secured credit facility with The RHL Group
and entered into a Secured Credit Restructuring Agreement with MMR, The RHL Group and Mr. Lorsch, or the Restructuring Agreement.
MMR issued The RHL Group a Third Amended and Restated Note, or the Third Amended Note, and the Company agreed to guaranty MMR's
obligations under the Third Amended Note. The Third Amended Note amends and restates the previous The RHL Group note, matured November 30, 2009, and bears interest at the
lesser of 10% or the highest rate then permitted by law, and is secured (similar to the previous note) by the Security Agreement. Although the
reserve credit line has been increased to $3,000,000, The RHL Group is only obligated to make a minimum of $100,000 of loans, advances
and guarantees under the Third Amended Note within 30 days of execution pursuant to the Restructuring Agreement. The RHL Group Note
payable had a balance of $1,545,767 at December 31, 2009. The components of the Group Note payable and the related balance sheet
presentation as of December 31, 2009 are: (1) $940,620, which is included in the line of credit, related party payable; (2) $295,484, which is
related to credit card charges and a vendor guaranty, is included in accounts payable and accrued expenses; and (3) $309,663, which is
related to deferred salary and consulting expenses is included in related party payables. The RHL Group was due to receive, as an
origination fee, a promissory note for $200,000, bearing interest at 10% per annum and due on demand. On August 17, 2009, the Company
paid the $200,000 origination fee by granting 2,800,000 shares of common stock to The RHL Group with a value of $364,000 and recorded a
loss on issuance of $164,000. The origination fee was recorded as a deferred financing cost and is being amortized over the Third Amended
Note maturity period. As of December 31, 2009, the remaining unamortized origination fee was $0, and the Fee of $200,000 is included in
interest expense for the year ended December 31, 2009. Total interest expense on this note for the year ended December 31, 2009 and 2008 amounted to $327,110 and $62,442, respectively.
The unpaid interest balance as of December 31, 2009 and December 31, 2008 was $45,155 and $24,963, respectively. In conjunction with the loan agreement noted above, MMR was required to maintain certain financial covenants, including the
requirement that MMR have at least $100,000 in cash in its bank accounts or such other amount as necessary to maintain operations through
the subsequent thirty (30) days and timely pay any obligations due respecting payroll and all associated payroll taxes on and after June 1,
2009. F-15
On August 18, 2009, the Company and The RHL Group entered into a Waiver Agreement, pursuant to which, in consideration of The
RHL Group's waiver of MMR's payment default under the Third Amended Note, the Company granted to The RHL Group a warrant to
purchase an aggregate of 11,039,378 shares of the Company's common stock, with an exercise price equal to $0.13 per share, the closing
price of the Company's common stock on the date immediately preceding the date of grant. Under the Waiver Agreement, The RHL Group
agreed to waive MMR's payment default until August 31, 2009, which waiver period will automatically continue until The RHL Group notifies
MMR otherwise. As of December 31, 2009, MMR was in compliance with its financial covenants under the terms of the Third Amended
Note. The RHL Note matured on November 30, 2009. The Company is currently in negotiations with The RHL Group for renewal of the
agreement. 4. PREPAID EXPENSES AND OTHER CURRENT ASSETS Prepaid expenses and other current assets include the following. 5. PROPERTY AND EQUIPMENT Property and equipment, stated at cost, at December 31, 2009 and 2008 consisted of the following. F-16
Included in computers and related equipment at December 31, 2009 and 2008 are assets under capital leases of $28,565. Included in
accumulated depreciation and amortization at December 31, 2009 and 2008 is $17,085 and $11,372, respectively, related to assets under
capital leases. Amortization expense related to assets under capital leases for the years ended December 2009 and 2008 amounted to
$5,713. Total depreciation expense was $17,517 and $17,935 for the years ended December 31, 2009 and 2008, respectively. 6. INTANGIBLE ASSETS Intangible assets as of December 31, 2009 and 2008 consisted of the following. Amortization expense for the years ended December 31, 2009 and 2008 amounted to $60,647 and $130,843, respectively. The
Company is in the process of evaluating the patents' estimated useful life and will begin amortizing the patents when they are brought to the
market. Estimated amortization expense for each of the next five succeeding years and thereafter is as follows: F-17
7. ACCOUNTS PAYABLE AND ACCRUED EXPENSES 8. ADVANCES FROM AFFILIATE On October 1, 2008, Favrille advanced MMR $100,000 pursuant to a promissory note dated September 30, 2008, which was to be used
solely for paying MMR's out of pocket expenses incurred in connection with the Merger. On November 10, 2008 (the first business day after
execution of the Merger Agreement), Favrille advanced MMR an additional $500,000 pursuant to a promissory note issued by MMR to
Favrille, and on December 22, 2008 an additional $100,000 pursuant to promissory notes issued by MMR to Favrille in each case, solely for
MMR's use in paying its out of pocket expenses incurred in connection with the Merger and its operating expenses. As of December 31,
2008, the total notes payable outstanding from Favrille amounted to $701,322, which included accrued interest of $1,322. Subsequent to the year ended December 31, 2008, the Company borrowed an additional $35,000 and $50,000 on January 8, 2009 and
January 15, 2009, respectively, under the same terms as the aforementioned promissory notes. Principal outstanding under the Promissory Notes accrued interest at the applicable federal rate of interest per annum, or the maximum
rate permissible by the laws in California relating to permissible rates of interest on commercial loans, whichever is less. Pursuant to the
terms of the Merger Agreement, upon occurrence of the Merger, the principal and interest under the Promissory Notes was automatically
treated as a contribution to capital to MMR and the Promissory Notes were fully discharged. 9. INCOME TAXES The items accounting for the difference between income taxes computed at the federal statutory rate and the provision for income taxes
were as follows: F-18
Significant components of deferred tax assets and (liabilities) are as follows: The Company files income tax returns in the United States ("Federal") and California ("State") jurisdictions. The
Company is subject to Federal and State income tax examinations by tax authorities for all years since it's inception. At December 31, 2009, the Company had Federal and State net operating loss carry forwards available to offset future taxable income of
$17,978,844 and $17,876,046, respectively. These carry forwards will begin to expire in the years ending December 31, 2025 and December
31, 2015, respectively. These net operating losses may be subject to various limitations on utilization based on ownership changes under
Internal Revenue Code Section 382 as a result of the Merger, and the Company is in the process of evaluating the impact of this before any
losses are used to offset future taxable income. The Company periodically evaluates the likelihood of the realization of deferred tax assets, and adjusts the carrying amount of the
deferred tax assets by a valuation allowance to the extent the future realization of the deferred tax assets is not judged to be more likely than
not. The Company considers many factors when assessing the likelihood of future realization of our deferred tax assets, including recent
cumulative earnings experience by taxing jurisdiction, expectations of future taxable income or loss, the carry-forward periods available to us
for tax reporting purposes, and other relevant factors. At December 31, 2009, based on the weight of available evidence, including cumulative losses in recent years and expectations of future
taxable income, the Company determined that it was more likely than not that its deferred tax assets would not be realized. According, the
Company has recorded a $7,940,696 valuation allowance, or 100% of its cumulative deferred tax assets. As a result of the implementation of certain provisions of ASC 740, Income Taxes, (formerly FIN 48, Accounting for Uncertainty
in Income Taxes - An Interpretation of FASB Statement No. 109), the Company performed an analysis of its previous tax filings and
determined that there were no positions taken that it considered uncertain. Therefore, there were no unrecognized tax benefits as of
December 31, 2009. Future changes in the unrecognized tax benefit are not expected to have an impact on the effective tax rate due to the existence of the
valuation allowance. The Company estimates that the unrecognized tax benefit will not change within the next twelve months. The Company
will continue to classify income tax penalties and interest, if any, as part of interest and other expenses in its consolidated statements of
operations. There is no interest or penalties accrued as of December 31, 2009. The following table summarizes the open tax years for each major jurisdiction: Jurisdiction Open Tax Years Federal 2005 - 2009 California State 2005 - 2009 F-19
As the Company has significant net operating loss carryforwards, even if certain of the Company's tax positions were disallowed, it is not
foreseen that the Company would have to pay any taxes in the near future. Consequently, the Company does not calculate the impact of
interest or penalties on amounts that might be disallowed. 10. COMMITMENTS AND CONTIGENCIES Leases The Company leases certain facilities and equipment under non-cancelable capital and operating leases, which expire at various
dates through 2011. Total rent expense for the year ended December 31, 2009 and 2008 was $64,850 and $179,335, respectively. Future
minimum lease payments as of December 31, 2009, under non-cancelable operating leases (with initial or remaining lease terms in excess of
one year) and future minimum capital lease payments are as follows: In April 2009, the Company renewed its lease with its Chairman and Chief Executive Officer on a month-to-month basis for use of office
space for a total of $3,000 per month. The Company terminated this lease in May 2009. Effective May 1, 2009, the Company entered into a lease agreement to lease additional office space in Beverly Hills, California. The lease
is month-to-month and requires a monthly payment of $7,667 commencing in June 2009. Guarantee provided by The RHL Group Under the terms of an agreement with an investor who purchased $500,000 of MMR's Series B Preferred shares in 2006, the
Company entered into an agreement to invest $250,000 in a joint venture with the investor to establish an entity to market and sell the
Company's services in the countries of Japan, China, Korea, Taiwan and Thailand. The Company has paid $100,000 to date of this amount
which the Company has expensed during the year ended December 31, 2009 as the Company has since terminated its relationship with the
investor. To date, no joint venture has been formalized or incorporated and no operations have commenced. In September 2007, The RHL
Group, provided the investor with a guarantee that the Company would meet its obligations under this agreement in exchange for 300,000
shares of restricted MMR common stock valued at $39,000. As consideration for renewing the guarantee when it expired in September 2008,
in January 2009, MMR issued The RHL Group 100,000 shares of MMR common stock, which became 328,174 shares of MMRIS common
stock upon the closing of the Merger, valued at approximately $5,000. This expense has been reflected in general and administrative
expenses in the accompanying consolidated statement of operations for the year ended December 31, 2009. On January 6, 2010, the
Company entered into a settlement agreement with the investor to provide for full and final settlement of any and all claims pertaining to any
license rights for MMR-Asia to market, sell and sublicense MMRIS and MMR products and services. As part of this settlement agreement, the
Company granted 1,388,889 shares of common stock to the investor in January 2010. F-20
Guarantee provided by Robert H. Lorsch On August 17, 2009, Robert H. Lorsch, the Company's Chairman, Chief Executive Officer and President, agreed to guarantee $282,641in
payments due to a vendor for services rendered to the Company. On August 17, 2009, in consideration of a personal guaranty given by Mr.
Lorsch, the Company granted Mr. Lorsch (i) a warrant to purchase 706,605 shares of Company common stock, at an exercise price of $0.13
per share, the closing price of the Company's common stock on the date immediately preceding the date of grant, and (ii) 141,321 shares of
Company common stock with a value of $18,372. Employment Agreements The Company has employment agreements with its Chairman, President and Chief Executive Officer, Robert H. Lorsch, its Vice
President of Finance and Chief Financial Officer, Ingrid Safranek, and its Executive Vice President of Technology and Product Development, Naj
Allana. Under each employment agreement, the executive officers receive a base salary, subject to annual increases as determined by the
board of directors, certain benefits as set forth in the employment agreements, and an annual bonus at the discretion of the board of
directors. The current term of Mr. Lorsch's employment agreement is effective until December 31, 2011 and will automatically renew for successive
12 month periods unless terminated at least 60 days prior to the end of the term. The employment agreement may be terminated by the
Company without cause (as defined in the agreement) upon 30 days written notice or for cause if Mr. Lorsch fails to cure the acts or
omissions constituting cause within 30 days. If Mr. Lorsch's employment is terminated by the Company for cause or voluntarily by Mr. Lorsch
without good reason, he will not be entitled to receive any severance payments or benefits under the employment agreement. If Mr. Lorsch's
employment is terminated by the Company without cause or voluntarily by Mr. Lorsch for good reason, Mr. Lorsch will be entitled to one year
of salary at his then current rate of pay, including all monthly benefits, and the pro rata portion of the annual bonus otherwise due Mr. Lorsch.
Mr. Lorsch's employment agreement includes provisions that prohibit Mr. Lorsch from disclosing the Company's confidential information
and trade secrets and competing with the Company during the term of his employment agreement or soliciting the Company's employees for
18 months following termination of employment. We also have approved an employment agreement with our former Chief Financial Officer and Senior Vice President and current
Executive Vice President of Technology and Product Development,
Naj Allana. Under the terms of his proposed agreement, Mr. Allana shall serve both as our Chief
Financial Officer and Senior Vice President and as Chief Financial Officer, Senior Vice President and Chief Technical Officer of our
wholly-owned subsidiary, MMR. The agreement provides for a base salary of $15,845 per month, subject to an upward increase. In addition to his
base salary, Mr. Allana is entitled to receive a commission with respect to certain of our accounts. Mr. Allana's employment agreement was effective until February 15, 2010 and automatically
renews for successive 12 month periods unless terminated at least 120 days prior to the end of the term. If we terminate
Mr. Allana's employment, other than for misconduct (as defined in the employment agreement) or unauthorized use or
disclosure of material confidential information of the Company, we would continue to pay Mr. Allana his monthly salary
for the months remaining through the current 12 month term, or if done 120 days preceding the end of the term, then
through the end of the next 12 month term. On January 26, 2010, we entered into an employment agreement with Ingrid Safranek as our Vice President, Chief Financial Officer and
Secretary. Under the employment agreement, Ms. Safranek receives a base salary, subject to annual increases as determined by the board
of directors, certain benefits as set forth in the employment agreements, and an annual bonus at the discretion of the board of directors. Ms.
Safranek's employment agreement is in effect until June 15, 2010, and shall automatically renew for successive one-year periods, unless
extended or otherwise terminated at least 60 days prior to the end of the term. F-21
If the Company terminates Ms. Safranek's employment, unless due to misconduct (as defined in the employment agreement), the
Company must continue to pay Ms. Safranek her monthly salary for two (2) months, if such termination occurs within the first year of
employment; for six (6) months, if such termination occurs between the first year and the last day of the second year of employment; and for
twelve (12) months, if such termination occurs after the last day of the second year of employment. For purposes of clarification, a change in
title or diminution of responsibilities, regardless of the level of materiality, shall not be considered a termination under the provisions of this
Agreement. Litigation Matters From time to time, the Company is involved in various legal proceedings generally incidental to its business. While the result of any
litigation contains an element of uncertainty, management presently believes that the outcome of any known, pending or threatened legal
proceeding or claim, individually or combined, will not have a material adverse effect on the Company's financial statements. 11. STOCKHOLDERS' DEFICIT Preferred Stock During the year ended December 31, 2008, MMR issued 468,869 shares of Series C Preferred Stock (which became 1,538,702
shares of MMRIS common stock subsequent to the Merger) at $1.33 per share for proceeds of $621,047 (net of $2,548 incurred directly
towards raising capital). From October 2007 to October 2008, MMR, as an incentive to investors, issued one share of common stock with each share of Series B
or Series C Preferred stock purchased. The Company issued 468,869 shares of Common Stock (which became 1,538,702 shares of MMRIS
Common Stock subsequent to the Merger) during the year ending December 31, 2008 valued at $35,620. Because the common shares were
issued as an inducement to invest and were not issued to all purchasers of Series B and Series C Preferred Stock, the common stock has
been accounted for as a distribution to the purchasers of the common shares and has been reflected as a deemed dividend in the Company's
financial statements during the year ended December 31, 2008. As of December 31, 2008, there were 3,332,694; 1,263,750; and 1,580,082 shares of MMR's Series A, Series B and Series C Preferred
Stock, respectively, issued and outstanding. Immediately prior to the Merger, the outstanding MMR preferred stock was converted to MMR
common stock, representing an aggregate of 6,176,526 shares, which converted into MMRIS common stock in the Merger. As a result, any
purchases of preferred stock in the accompanying consolidated balance sheets and consolidated statements of stockholders' deficit has
been retroactively reflected as common stock. As of December 31, 2009, there were no shares of MMR preferred stock issued and
outstanding. The Company has 5,000,000 shares of MMRIS preferred stock authorized. As of December 31, 2009, there were no shares of MMRIS
preferred stock issued and outstanding. Common Stock During the year ended December 31, 2008, MMR issued 4,713,940 shares of common stock (which became 15,469,969 shares of
MMRIS common stock subsequent to the Merger), as consideration for goods and services from both employees and non-employees valued
at $397,939. In August 2008, MMR issued 5,000,000 shares of common stock to The RHL Group as consideration for renewal of the revolving line of
credit agreement through July 31, 2009, consisting of 586,947 new shares of common stock (which became 1,926,202 shares of MMRIS
common stock subsequent to the Merger) and 4,413,053 shares of Treasury stock (14,482,453 shares of MMRIS common stock subsequent
to the Merger), valued at $50,000. Pursuant to the Merger Agreement, MMRIS agreed to issue (or reserve for issuance) an aggregate 92,599,196 shares of MMRIS
common stock to the stockholders of MMR and the holders of options and warrants to acquire MMR common stock at the time of the
Merger. F-22
Immediately prior to the Merger on January 27, 2009, and including the conversion of 6,176,526 shares of MMR preferred stock into
MMR common stock, the issuance of 100,000 shares of MMR common stock to The RHL Group for renewal of a guarantee (see Note 10),
MMR had 24,320,100 shares of common stock issued and outstanding. At the effective time of the Merger, and in accordance with the terms
of the Merger Agreement, these shares of outstanding MMR common stock automatically converted into 79,812,087 shares of MMRIS
common stock. Immediately prior to the Merger, there were 41,254,550 shares of MMRIS common stock issued and outstanding. On July 10, 2009, the
stockholders of the Company approved an increase to the authorized shares of common stock. As a result, the Company is authorized to
issue 650,000,000 shares of common stock. On March 13, 2009, the Company approved the issuance of 1,202,587 shares of MMRIS common stock including 1,145,324 shares of
Treasury stock, as replacement for options to acquire 1,202,587 shares of common stock. This resulted in an additional expense of $1,466
recorded during the year ended December 31, 2009. Warrants for Purchase of Preferred B shares On April 1, 2007, MMR granted 200,000 warrants to purchase Series B Preferred Stock of MMR at an exercise price of $2.00. The
warrants were issued to the largest distributor of employee assistance services in the US as an incentive to market and sell MMR's services
into their customer base. The related services agreement was for five years, and the customer never bought any services from MMR. Upon closing of the Merger on January 27, 2009, these warrants converted into warrants to purchase 656,346 shares of MMRIS common
stock at an exercise price of $0.61 per share. There was no additional expense recorded during the year ended December 31, 2009 resulting
from the modification of these warrants. F-23
12. EQUITY TRANSACTIONS Stock Option Activity A summary of option activity for the years ended December 31, 2008 and 2009 is presented below. Options granted by MMR prior to
the date of the Merger of January 27, 2009 have been retroactively restated to reflect the conversion ratio of MMR to MMRIS shares. The aggregate intrinsic value in the table above is before applicable income taxes and is calculated based on the difference between the
exercise price of the options and the quoted price of the common stock as of the reporting date. A summary of the activity of the Company's nonvested options for the years ended December 31, 2008 and 2009 is presented
below. F-24
As of December 31, 2009, total unrecognized stock-based compensation expense related to nonvested stock options was $1,120,496,
which is expected to be recognized by the year ended December 31, 2011. Total stock option expense recorded during the years ended
December 31, 2009 and 2008 amounted to $887,577 and $15,785, respectively, and is reflected in operating expenses in the accompanying
consolidated statements of operations. The following table summarizes information about stock options outstanding and exercisable at December 31, 2009. Warrants On November 7, 2007, in conjunction with the registered direct offering of an aggregate of 7.4 million shares, the Company
issued warrants to purchase up to 4.4 million shares of the Company's common stock at an exercise price of $2.77 per share. The warrants
are exercisable as of the date of grant through November 7, 2012. The Company valued the warrants using a Black-Scholes pricing
model. ASC 815-40 (formerly EITF 00-19, "Accounting for Derivative Financial Instruments Indexed to, and Potentially Settled in, a
Company's Own Stock") requires freestanding contracts that are settled in a Company's own stock, including common stock warrants to
be designated as an equity instrument, asset or liability. A contract designated as an asset or a liability must be carried at fair value until
exercised or expired, with any changes in fair value recorded in the results of operations. ASC 815-40 (formerly SFAS No. 133,
"Accounting for Derivative Instruments and Hedging Activities") was applied to determine that the liability is accounted for as a
derivative (see annual report on Form 10-K for the year ended December 31, 2008, filed on April 15, 2009 for a more detailed discussion).
The valuation of these warrants amounted to $0 and $88,998 as of December 31, 2008 and 2009. The increase in fair value of $88,998 is
included in change in valuation of derivative liabilities in the accompanying consolidated statements of operations for the year ended
December 31, 2009. Immediately prior to the Merger, MMRIS issued warrants to acquire 9,999,992 shares of MMRIS's common stock at an exercise price of
$0.12 per share, which expire on January 26, 2014, to certain former officers, former directors and their affiliates who were willing to take
such equity as partial or full payment for outstanding liabilities. These warrants vested immediately and were valued on the date of grant
using the Black-Scholes option pricing model using the assumptions as described in Note 2. As this transaction occurred immediately prior to
the Merger, the resulting gain on the issuance of warrants was reflected in the statement of operations of MMRIS. There is no impact of this
transaction recorded on the accompanying consolidated statement of operations for the year ended December 31, 2009. On March 1, 2009, the Company granted to a consultant 500,000 warrants to purchase shares of the Company's common stock at an
exercise price of $0.15 per share. The warrants vest over a six month period and have a contractual life of five years. These warrants were
valued using the Black-Scholes option pricing model using the assumptions as described in Note 2. As of September 1, 2009, the date these
warrants fully vested, the total value of these warrants amounted to $34,741 which was recorded as stock based compensation expense
during the year ended December 31, 2009. On March 1, 2009, the Company granted an additional 1,250,000 warrants to this consultant with
an exercise price of $0.15 per share, conditional upon the consultant being hired as full time employee by November 1, 2009. As the
consultant was not hired, the warrants were cancelled and there was no expense recorded for these warrants. F-25
On March 8, 2009, the Company granted to a group of consultants an aggregate total of 600,000 warrants to purchase shares of the
Company's common stock at an exercise price of $0.175 per share. Of the warrants granted, 100,000 vested immediately upon grant and
500,000 vest over a two year period. These warrants were valued using the Black-Scholes option pricing model using the assumptions as
described in Note 2. The total value of these warrants amounted to $23,544, of which $12,918 related to the 100,000 warrants which was
recorded as a reduction of outstanding accounts payable during the year ended December 31, 2009, and $10,626 related to the 500,000
warrants which was recorded in sales and marketing expense for the year ended December 31, 2009. On March 13, 2009, the Company granted to a group of vendors an aggregate total of 1,300,000 warrants to purchase shares of the
Company's common stock at an exercise price of $0.175 per share. These warrants all vested immediately. These warrants were valued
using the Black-Scholes option pricing model using the assumptions as described in Note 2. The total value of these warrants amounted to
$173,636, which was recorded as interest expense during the year ended December 31, 2009, as the Company granted these warrants as
consideration for the vendors agreeing to defer payment of their outstanding liabilities. On April 6, 2009, the Company granted to a customer 50,000 warrants to purchase shares of the Company's common stock at an
exercise price of $0.23 per share. The warrants vested immediately. These warrants were valued using the Black-Scholes option pricing
model using the assumptions as described in Note 2. The total value of these warrants amounted to $8,490, all of which was recorded as a
reduction to subscriber revenues during the year ended December 31, 2009. On May 13, 2009, the Company granted to a group of consultants an aggregate total of 275,000 warrants to purchase shares of the
Company's common stock at an exercise price of $0.19 per share. Of the warrants granted, 75,000 vested immediately upon grant and
200,000 vest over a six month period. These warrants were valued using the Black-Scholes option pricing model using the assumptions as
described in Note 2. The total amount recorded as operating expense relating to these warrants during the year ended December 31, 2009
amounted to $22,734. On July 29, 2009, the Company elected to pay a $30,000 loan commitment fee through the issuance of warrants exercisable into 375,000
shares of the Company's common stock at an exercise price of $0.14 per share. The warrants vested immediately. These warrants were
valued using the Black-Scholes option pricing model using the assumptions as described in Note 2. The total value of these warrants
amounted to $32,399, all of which was recorded as a reduction to the commitment fee payable in July 2009, with a loss on settlement of
payables for $2,399. On July 29, 2009, the Company granted to a consultant 304,826 warrants to purchase shares of the Company's common stock at an
exercise price of $0.125 per share. The warrants vested immediately. These warrants were valued using the Black-Scholes option pricing
model using the assumptions as described in Note 2. The total value of these warrants amounted to $29,481, all of which was recorded as a
reduction of outstanding accounts payable during the year ended December 31, 2009. On August 11, 2009, the Company granted to a service provider 1,000,000 warrants to purchase shares of the Company's common stock
at an exercise price of $0.125 per share. These warrants all vested immediately. These warrants were used valued using the Black-Scholes
option pricing model using the assumptions as described in Note 2. The total value of these warrants amounted to $96,637, which was
recorded as interest expense during the year ended December 31, 2009, as the Company granted these warrants as consideration for the
service provider agreeing to defer payment of their outstanding liabilities. On August 17, 2009, the Company granted to Robert Lorsch and The RHL Group a total of 11,745,983 warrants to purchase shares of
the Company's common stock at an exercise price of $0.13 per share. These warrants all vested immediately. These warrants were used
valued using the Black-Scholes option pricing model using the assumptions as described in Note 2. The total value of these warrants
amounted to $1,131,852, which was recorded as interest expense during the year ended December 31, 2009, as the Company granted these
warrants as consideration for Robert Lorsch guarantying monies owed to a vendor and The RHL Group's waiver of MMR's payment default
under the Third Amended Note. F-26
During 2009, the Company granted to five unrelated third-parties, or the Holders a total of 8,658,000 warrants to purchase shares of the
Company's common stock in connection with the 12% Convertible Promissory Note, or the Convertible Notes, at an exercise price equal to
the lesser of (i) the product of 50% multiplied by the arithmetic average of the dollar volume-weighted average price, or VWAP, of the
common stock for the 10 consecutive trading days ending on the day that is 3 Trading Days prior to the applicable exercise date or (ii) $0.15.
The total value of these warrants amounted to $839,012, which was bifurcated from the Convertible Notes and accounted for as a discount
on debt. The discount is being accreted over the initial term of the Convertible Notes using the interest method and upon exercise the entire
unamortized discount is recognized into additional paid in capital. See Note 14 below. The following summarizes the total warrants outstanding and exercisable as of December 31, 2009. The weighted average fair value of warrants granted during the year ended December 31, 2009 amounted $0.10. There were 8,683,000
warrants exercised during the year ended December 31, 2009. Of the total warrants exercised, 8,308,000 warrants were granted in
connection with the Convertible Notes while the remaining 375,000 were granted in connection with the $30,000 loan commitment fee. Restricted Stock Program Under the Restricted Stock Program, a restricted stock award is an offer by the Company to sell to an eligible person shares that are
subject to restrictions relating to the sale or transfer of the shares. A committee appointed by the Board to administer the program or the
Board itself shall determine to whom an offer will be made, the number of shares the person may purchase, the price to be paid and the
restriction to which the shares shall be subject. The offer must be accepted by the eligible person within thirty days from the date of the offer
evidenced by the Restricted Stock Purchase Agreement. The purchase price of shares shall not be less than 85% of the fair market value of
such shares on the issue date, with the provision that the purchase price for a 10% stockholder shall not be less than 110% of such fair
market value. Shares are either fully and immediately vested upon issuance, or may vest in installments upon attainment of specified
performance objectives. During the year ended December 31, 2009, the Company issued 13,049,774 shares of common stock in consideration of goods and
services from both employees and non-employees valued at $1,328,000. During the year ended December 31, 2008, MMR issued 4,713,940
shares of restricted MMR common stock for services and interest expense on the line of credit with The RHL Group. These shares of MMR
became 15,469,969 shares of MMRIS subsequent to the merger. The total value of these restricted shares amounted to $397,939, of which
$187,939 is recorded in general administrative expenses and $210,000 is recorded in interest and other expenses, net on the accompanying
consolidated statement of operations for the year ended December 31, 2008. For purposes of financial statement presentation, the
issuances of shares for the period of January 1, 2008 through June 29, 2008 were valued at $0.15 per share. Issuances of shares for the
period of June 29, 2008 through November 7, 2008 were valued at $0.01 per share. Issuances of shares for the period of November 7, 2008
through December 31, 2008 were valued at $0.05 per share. F-27
Stock Bonus Program Under the Stock Bonus Program, shares are issued as a bonus for past services rendered pursuant to the Stock Bonus Agreement.
Stock bonuses may be awarded upon satisfaction of specified performance goals pursuant to the Performance Stock Bonus Agreement. No
shares were issued under the Stock Bonus Program during the years ended December 31, 2009 or 2008. Derivative Liabilities In accordance with ASC 815-40 (formerly EITF 00-19, "Accounting for Derivative Financial Instruments Indexed to, and Potentially
Settled in, a Company's Own Stock"), on January 27, 2009, the Company performed an analysis as if all holders of options and warrants
exercised their rights under those contracts. This analysis resulted in the conclusion that the Company had inadequate authorized shares to
settle 100% of these contracts. Therefore, as shareholder approval would be necessary to increase the number of authorized shares,
settlement of these obligations would not be within the control of the Company. Consequently certain non-employee options and warrants
were accounted for as derivative liabilities in accordance with ASC 815-40 (formerly SFAS No. 133, "Accounting for Derivative Instruments
and Hedging Activities"). The event giving rise to this condition was the merger between Favrille, Inc. and MyMedicalRecords.com, Inc. on January 27, 2009. On
that date, the fair value of 3,436,458 vested non-employee options and 15,218,890 vested warrant contracts subject to this analysis was
reclassified from equity to derivative liabilities. The condition was remedied on June 13, 2009 when certain of the option and warrant holders
waived their rights under those contracts until such time that the Company had 250 million or more shares authorized. On March 31, 2009
and June 13, 2009, these contracts were again valued using the Black-Scholes option valuation model and the difference between the
original value at January 27, 2009 of $871,052 and the values at March 31, 2009 and June 13, 2009 of $2,665,889 and $1,377,033,
respectively, was recorded as a gain on change in value of derivatives for the three months ended June 30, 2009 and a loss on change in
value of derivatives for the six months ended June 30, 2009. On June 13, 2009, the derivative liabilities were reclassified back into equity
resulting in a gain of $1,288,856 for the three months ended June 30, 2009 and a loss of $505,980 for the six months ended June 30, 2009. On July 16, 2009, the Company entered into a 12% Convertible Promissory Note with an unrelated third-party for a principal amount
totaling $30,000. Subsequently, the Company entered into identical 12% Convertible Promissory Notes ("the Convertible Notes") with four
other unrelated third-parties for an additional $530,000. Each Convertible Note is convertible into shares of common stock by dividing (i) the
then outstanding balance of such note by (ii) the product of eighty percent (80%) multiplied by the arithmetic average of the
VWAP of the common stock for the ten (10) consecutive trading days ending on the day that is three (3) trading days prior to
the applicable conversion date. In addition, the Company granted a total of 3,110,000 warrants to purchase shares of the Company's
common stock in connection with the Convertible Notes at an exercise price equal to the lesser of (i) the product of 50% multiplied by the
arithmetic VWAP of the common stock for the 10 consecutive trading days ending on the day that is 3 Trading Days prior to the applicable
exercise date or (ii) $0.15. Such arrangement resulted in the possibility that the Company may not have enough authorized shares to settle
these obligations using common stock. Instead, there is the possibility, although remote, that it might need to settle one or more of these
contracts with cash. Consequently certain non-employee options and warrants were accounted for as derivative liabilities as of July 16,
2009. The event giving rise to this condition was the first Convertible Note the Company entered into on July 16, 2009. As of July 16, 2009 and
for all subsequent options and warrants issued prior to December 31, 2009, the fair value of 2,421,789 vested non-employee options and
33,976,760 vested warrant contracts subject to this analysis was reclassified from equity to derivative liabilities. On December 31, 2009,
these contracts were again valued using the Black-Scholes option valuation model and the difference between the original value at July 16,
2009 plus contracts granted between July 16, 2009 and December 31, 2009 of $2,664,125, including the cancellation of exercised warrants
and options of $95,790 and the value at December 31, 2009 of $1,534,824, was recorded as a gain on change in value of derivatives. The
gain recorded related to this matter was $1,239,529 included in the change in valuation of derivative liabilities for the twelve months ended
December 31, 2009. Included within the $1,239,529 gain was a $8,445 loss due to the de-recognition of options and warrants exercised
between July 16, 2009 and December 31, 2009. F-28
None of the derivatives liabilities were designated as hedging instruments. The following is a reconciliation of the derivative liability:
The inputs used for the Black-Scholes option valuation model were as follows: 13. NOTES PAYABLE The Notes payable balance at December 31, 2008 and 2009 consisted of the following: The Company is in communication with the above debt holders whose maturity dates have passed, and intends to repay the notes
payable in the short-term as the Company's cash flow improves. F-29
Future maturities with respect to these notes payable as of December 31, 2009 were as follows: 14. CONVERTIBLE PROMISSORY NOTES On various dates in 2009, the Company issued Convertible Promissory Notes ("Notes") with accredited investors for an aggregate of
$1,373,000 principal amount. The Notes pay interest at a stated rate of twelve percent (12%) per annum. The Notes are convertible at the option of the holders into common stock at a variable conversion price of eighty percent (80%) multiplied
by the arithmetic average of the volume-weighted average trading price of the Common Stock for the ten (10) consecutive trading days
ending on the day that is three (3) trading days prior to the applicable conversion date, subject to anti-dilution and other customary
adjustments. In connection with the Notes, the Company also issued warrants to purchase an aggregate of 8,658,000 shares of common stock. The
term of the warrants is three years and the exercise price is the lesser of (i) the product of fifty percent (50%) multiplied by the arithmetic
average of the volume-weighted average trading price of the Common Stock for the ten (10) consecutive trading days ending on the day that
is three (3) trading days prior to the applicable exercise date, or (ii) $0.15, subject to anti-dilution and other customary adjustments. Under the terms of the agreements, principal amounts owed under the Notes become due and payable December 31, 2009, June 30,
2010 and July 31, 2010, provided that, upon ten (10) days' prior written notice to the Holder, the Company may, in its sole discretion, extend
the maturity date to June 30, 2010 and December 31, 2010. By virtue of the Notes' variable conversion ratio, a possibility exists, although remote, that the Notes may be convertible into shares of
Common Stock that exceed the currently authorized limit. Consequently, a possibility exists that the Company might be placed in a position of
net settling the Notes for an amount that exceeds their face value. This resulted in the conversion feature being a derivative under Accounting
Standards Codification ("ASC") 815-10 "Derivatives and Hedging." Additionally, as a result of the above, all outstanding options and warrants issued to non-employees, including the warrants issued in
connection with the Notes, are also derivatives as they too could require net settlement in the absence of adequate authorized shares. The fair value of the conversion feature of the Notes and related warrants was derived using the Black-Scholes option valuation model,
resulting in an aggregate fair value of $1,964,585. For two notes issued in November 2009, the fair value of the conversion feature of the
Notes and related warrants exceeded the face value of the Notes by $778,520 and such amount was recorded as a deferred financing cost.
The fair value of the conversion feature of the Notes and related warrants, net of the deferred financing cost, was deducted from the Notes,
resulting in an initial value ascribed to the Notes of $186,934. The initial value of the Notes is being accreted to their face value over the initial
term of the Notes using the interest method. Upon conversion, the entire unamortized discount from the face value of the Notes is recognized
as interest expense. F-30
The following table summarizes the Convertible Promissory Notes and discounts upon issuance: On September 15, 2009, November 5, 2009, November 19, 2009 and December 10, 2009, the same day these particular Notes were
issued, four of the Note holders converted their Notes, in the aggregate face amount of $1,193,000, into 17,991,265 shares of Common
Stock. On these dates the entire conversion feature and warrant discounts on these Notes of $1,807,542 was recognized as interest
expense. The following table shows activity in the Notes account during the year ended December 31, 2009. 15. RESTRUCTURING ACTIVITIES From May 29, 2008 to November 7, 2008, MMRIS had provided notices under the federal Worker Adjustment and Retraining
Notification Act to 142 employees, including six members of senior management, that it planned to conduct a workforce reduction at its facility
in San Diego, California and that their employment was expected to end on various dates between June 6, 2008 to November 7, 2008.
Immediately prior to the date of the Merger on January 27, 2009, the total severance liability relating to former MMRIS employees amounted
to $1,682,416. On January 27, 2009, immediately prior to the Merger, as part of the 9,999,992 warrants issued to creditors (see Note 12), the
Company issued warrants as settlement of $985,020 of these amounts. In addition, the Company signed promissory notes with certain former
executives totaling $76,783, which notes are payable in full on August 31, 2009 (see Note 13). As of December 31, 2009, the total remaining severance liabilities amounted to $620,613, which is reflected as severance liability on the
accompanying consolidated balance sheets. This consists of $571,362 payable to former non-executive employees in 18 monthly
installments starting on July 27, 2009, as well as $49,251 in estimated payroll tax. No payments were made during the year ended
December 31, 2009 on these severance liabilities. During the period from January 27, 2009 through June 30, 2009, the Company entered into a series of settlement agreements with
certain vendors of MMRIS pursuant to the Creditor Plan, in which the Company settled $302,982 of its outstanding accounts payable for an
aggregate settlement amount of $214,402, including promissory notes of $139,355 payable in 18 monthly installments starting on July 27,
2009 (see Note 13). This resulted in a gain of $89,170, which has been recorded as a gain on settlement of payables in the accompanying
consolidated statements of operations for the year ended December 31, 2009. 16. RELATED PARTY TRANSACTIONS The Company incurred $73,868 and $51,025 during the year ended December 31, 2009 and 2008, respectively, towards marketing
consulting services from Bernard Stolar, a director. Included in related party payables at December 31, 2009 and 2008 was $106,368 and
$76,025, respectively, with respect to these services. In addition, on August 6, 2009, the Company granted the Mr. Stolar 1,200,000 options
to purchase common stock in the company at an exercise price of $0.125 per share in consideration for consulting and other services. The
options vest in eight quarterly installments beginning on the grant date. F-31
The Company also incurred $76,000 and $50,000 during the year ended December 31, 2009 and 2008, respectively, towards marketing
consulting services from Hector Barreto, a director. Included in related party payables at December 31, 2009 and 2008 was $15,500 and
$75,000, respectively, with respect to these services. In the first quarter of 2009, the Company entered into an agreement with The Latino
Coalition, a non-profit organization in which Mr. Barreto is also the Chairman, to market the Company's product to its members. As of
December 31, 2009, the Company has paid The Latino Coalition $16,000 as part of the agreement. In addition, on August 6, 2009, the
Company granted Mr. Barreto 1,200,000 options to purchase common stock in the company at an exercise price of $0.125 per share in
consideration for consulting and other services. The options vest in eight quarterly installments beginning on the grant date. In August 2006, the Company entered into a month-to-month lease with The RHL Group for the use of furniture and art for the
Company's offices, for a total of $1,000 per month which terminated on August 15, 2008. The Company incurred expenses of $0 and $7,500
during the year ended December 31, 2009 and 2008, respectively. In August 2008, the Company entered into an eight month lease with its Chairman and Chief Executive Officer for the use of office space
for a total of $3,000 per month plus a share of utilities. This lease was renewed in April 2009 on a month-to-month basis and terminated in
May 2009. In February 2006, the Company entered into a letter agreement with MyMedicalRecords.com.au ("MMR AU") granting it a 10 year
exclusive right to market and sell the Company's products and services in Australia and New Zealand. The controlling shareholder of MMR
AU was, at the time of the signing of the initial letter agreement, a shareholder of the Company. This letter agreement was subsequently
formalized in the form of a ten year license agreement in October 2007, at which time the controlling shareholder of MMR AU was no longer a
shareholder of the Company. The Company terminated the license agreement on September 30, 2009. The Company received $50,000 from MMR AU upon the signing of the letter agreement in February 2006 towards set up costs of the
platform to be used by customers of MMR AU. The Company completed the setup of the platform for MMR AU in 2007, at which time it
started to recognize this amount into revenue. The Company also received $150,000 in 2007 from MMR AU towards the minimum guarantee
payment for years 2 and 3 of the license term. This amount has been deferred and is being recognized as income based on the number of
months elapsed. On September 30, 2009 the license agreement was terminated and the balance remaining in deferred revenue of $32,082
was recognized as license fee revenue. During the year ended December 31, 2009 and 2008, the Company amortized $48,332 and $84,375,
respectively, of these amounts as license fee and other revenues in the accompanying statement of operations. The Company has accounted
for this transaction under ASC 605-025 (formerly EITF 00-21, "Revenue Arrangements with Multiple Deliverables"). The Company contracts with a significant vendor for the development and maintenance of the software applications necessary to run our
MyMedicalRecords PHR, MyESafeDepositBox and MyMedicalRecords Pro products. Our outside developer supports our software
development needs through a team of software engineers, programmers, quality control personnel and testers, who work with our internal
product development team on all aspects of application development, design, integration and support of our products. This vendor is also a
shareholder. For the years ended December 31, 2009 and 2008, the total expenses relating to this shareholder amounted to $369,405 and
$132,877, respectively. In addition, the Company capitalized software development costs for the years ended December 31, 2009 and 2008,
of $272,532 and $0, respectively. As of December 31, 2009, the total amounts due to the shareholder and included in related party payables
amounted to $617,796. On January 6, 2010, the Company entered into a settlement agreement with an MMR-Asia investor to provide for full and final settlement
of any and all claims pertaining to any license rights for MMR-Asia to market, sell and sublicense MMRIS and MMR products and services.
Refer to the Guarantee provided by The RHL Group section within footnote 10, Commitments and Contingencies for further details. As
part of this settlement agreement, the Company granted 1,388,889 shares of common stock to the investor. The value of these shares, of
$138,889, was included in related party payables as of December 31, 2009. The Company incurred costs of $323,278 and $8,324 during the years ended December 31, 2009
and 2008, respectively, toward consulting services from Audit Prep Services, LLC. Michael T. Psomas, member of Audit
Prep Services, LLC, is a shareholder of MMRIS. At December 31, 2009 the Company had $64,568 due to Audit Prep
Services, LLC, which was recorded as a related party payable. At December 31, 2008, the Company had $8,324 due to
Audit Prep Services, LLC, which was included in accounts payable. On March 8, 2009, the Company satisfied the
payable of $8,324 to Audit Prep Services, LLC through the issuance of 100,000 options with a strike price of $0.175 to
Mr. Psomas. On July 29, 2009, the Company issued an additional 304,826 options with a strike price of $0.125 and a
value of $21,502 to Mr. Psomas in satisfaction of a portion of its liability to Audit Prep Services, LLC due at that time. All
options vested immediately on the grant date. Additionally, on July 4, 2009 and September 4, 2009, the Company
granted to Mr. Psomas 500,000 and 770,000 shares of common stock, respectively, valued at $137,000 in consideration
for amounts due to Audit Prep Services, LLC at the times of issuance. F-32
On September 15, 2009, the Company entered into a five year agreement with E-Mail Frequency, LLC and David Loftus, Managing
Partner of E-Mail Frequency, LLC, a significant shareholder of the Company. The Company will license an existing 80 million person direct
marketing database of street addresses, cellular phone numbers, e-mail addresses and other comprehensive data, or the Database, with
E-Mail Frequency. The agreement allows the Company to market, through the use of the Database, its MyMedicalRecords Personal Health
Record, MyEsafeDepositBox virtual vault, and MMRPro document management system to physicians and their patients.
Under the terms of
the Agreement, the Company, upon closing of the Agreement, paid $250,000 to David Loftus as a one-time consulting fee in the form of
2,777,778 shares of common stock. The $250,000 one-time licensee fee was recorded as a prepaid consulting fee and included in the
prepaid expenses and other current assets as of December 31, 2009, less amortization of $12,500 included in operating expensed for the
year ended December 31, 2009. In addition, the Company incurred a total of $62,577 and $0 during the year ended December 31, 2009 and
2008, respectively, towards marketing services and consulting services from E-Mail Frequency, LLC and David Loftus. Included in related
party payables at December 31, 2009 and December 31, 2008 was $50,577 and $0, respectively, in respect to these services. Furthermore,
during September and November 2009, the Company entered into 12% Convertible Promissory Notes with Mr. Loftus for a principal amount
totaling $600,000 and warrants to purchase the Company's common stock. On the dates of the investments, Mr. Loftus immediately
converted the 12% Convertible Promissory Notes into shares of common stock and exercised the attached warrants and received a total
13,410,962 shares of common stock. 17. SUBSEQUENT EVENTS On January 4, 2010, the Company and UNIS entered into a Cooperation Agreement and related documents, which we refer to as the
Cooperation Agreement. Under the Cooperation Agreement, the Company and UNIS agreed to form a joint venture in China for the purpose of
deploying the Company's personal health record services and document imaging and management solutions in China. The Company will own
40% of the joint venture and UNIS will own 60% and each party will have the right to designate two members of the joint venture's board of
directors, with the fifth member being a Chinese citizen mutually designated by the Company and UNIS. On January 6, 2010, the Company entered into a settlement agreement with an MMR-Asia investor to provide for full and final settlement
of any and all claims pertaining to any license rights for MMR-Asia to market, sell and sublicense MMRIS and MMR products and services.
As part of this settlement agreement, the Company granted 1,388,889 shares of common stock to the investor. On January 21, 2010, in connection with the Meeting of the Board of Directors, the Company's Board approved the following items: F-33
On January 26, 2010, the Company entered into an employment agreement with Ingrid Safranek as its Vice
President, Chief Financial Officer and Secretary. Under the employment agreement, Ms. Safranek receives a base salary, subject to
annual increases as determined by the board of directors, certain benefits as set forth in the employment agreements, and an annual
bonus at the discretion of the board of directors. The agreement is in effect until June 15, 2010, and shall be extended for successive
one-year periods, unless extended or otherwise terminated before expiration of the term. F-34
In January, February and March 2010, the Company entered into 12% Convertible Promissory Notes with two different unrelated
third-parties and with two related parties for a principal amount totaling $1,250,175 and warrants to purchase 8,050,875 shares of the
Company's commons stock. The 12% Convertible Promissory Notes mature on July 31 2010, and the Company may, at its sole
discretion, extend the maturity date to December 31, 2010. The 12% Convertible Promissory Notes bear an interest of 12% per annum
payable in cash or shares of common stock, or a combination of cash and shares of common stock. The decision whether to pay in cash,
shares of common stock or combination of both shall be at the sole discretion of the Company. The 12% Convertible Promissory Note are
convertible into shares of common stock by dividing (i) the then outstanding balance of such note by (ii) the product of eighty percent
(80%) multiplied by the arithmetic average of the VWAP of the common stock for the ten (10) consecutive trading
days ending on the day that is three (3) trading days prior to the applicable conversion date. All parties immediately exercised the
warrants and converted the 12% Convertible Promissory Notes into 16,712,304 shares of the Company's common stock. On March 16, 2010, the $125,000 principal and unpaid interest on the PM Creative Corporation Promissory Note was converted in
shares of the Company's common stock. On March 22, 2010, the Company entered into a Master Services Agreement with Chartis International LLC
("Chartis"). Chartis provides a wide range of insurance solutions and is one of the world's leading property-casualty and
general insurance organizations, with operations in over 160 countries and jurisdictions. The Company will provide MMR's secure
online Personal Health Record (PHR) to Chartis policyholders worldwide. The program, designed to be an integrated benefit to
policyholders, will be introduced through local Chartis insurance affiliates throughout the world. The PHR will initially be available in
English, Spanish, German, French, Italian, Japanese and Chinese. The Company was recently contacted by Ronald Levy, MD, Chief of the Oncology Division at Stanford University
School of Medicine. Dr. Levy requested certain data from Favrille's (the Company's pre-merger predecessor) vaccine trials for a project
that theoretically could be used to predict the outcomes of patients vaccinated with idiotype vaccines. The Company has also been
advised that results of such a project, if favorable, could have the potential to lead to a re-interpretation of the previous Favrille cancer
vaccine trials which could lead to a resumption of the Company's Vaccine trials. The Company acquired these intellectual property
assets, certain biological samples and materials and other related technologies through its reverse merger with Favrille, Inc., a
biopharmaceutical company, in January 2009. The Company also believes that these assets may also be of value to other
biopharmaceutical companies, academic institutions and other entities. The Company has engaged consultants to respond to this
opportunity and others that could maximize the value of the Company's pre-merger Favrille assets on a worldwide basis, the outcomes of
which may not be known for some time. The Company will remain focused on its primary business, which is specifically the development
and distribution of the MyMedicalRecords Personal Health Record, MMRPro, and other related solutions in Health Information
Technologies. F-35
MMR INFORMATION SYSTEMS, INC. The accompanying notes are an integral part of these
consolidated financial statements F-36
MMR INFORMATION SYSTEMS, INC. The accompanying notes are an integral part of these
consolidated financial statements F-37
The accompanying notes are an integral part of these
consolidated financial statements F-38
MMR INFORMATION SYSTEMS, INC. The accompanying notes are an integral part of these
consolidated financial statements F-39
MMR INFORMATION SYSTEMS, INC. NOTE 1 - NATURE OF OPERATIONS AND BASIS OF PRESENTATION On January 27, 2009, MMR Information Systems, Inc. ("MMRIS"), (formerly known
as Favrille, Inc. ("Favrille"), a Delaware corporation formed in January 2000,
and hereinafter referred to as the "Company") consummated a transaction
with MyMedicalRecords, Inc. (formerly known as mymedicalrecords.com, Inc., a
Delaware corporation formed in 2005) ("MMR") through a merger of a wholly-owned
subsidiary of MMRIS with and into MMR pursuant to the terms of the Agreement and
Plan of Merger and Reorganization dated November 8, 2008 (the "Merger").
Pursuant to the terms of the Merger, all of the outstanding common and preferred
stock of MMR was cancelled and the former stockholders of MMR received an
aggregate of 79,812,116 shares of Favrille common stock. In addition, MMRIS
assumed the obligations of MMR under its outstanding stock options and warrants,
which, pursuant to the terms of the Merger, represented the right to receive an
aggregate 12,787,080 shares of MMRIS common stock at the effective time of the
Merger. In connection with the Merger, MMR became a wholly-owned subsidiary of
the Company, with the former stockholders of MMR collectively owning shares of
the Company's common and preferred stock representing approximately 60.3% of the
voting power of the Company's outstanding capital stock. For accounting purposes, the Merger was treated as a reverse acquisition with
MMR being the accounting acquirer. Accordingly, the historical financial results
prior to the Merger are those of MMR. The results of operations for MMRIS are
included in the Company's consolidated financial results beginning on January
27, 2009. The presentation of consolidated statements of stockholders' equity (deficit)
reflects the historical stockholders' equity (deficit) of MMR through January
26, 2009. The effect of the issuance of shares of MMRIS common stock in
connection with the Merger and the inclusion of MMRIS's outstanding shares of
common stock at the time of the Merger is reflected in the accompanying
consolidated financial statements. MMR was incorporated in Delaware in 2005 and is headquartered in Los Angeles,
CA. Effective February 9, 2009, MMR changed its corporate name to MMR
Information Systems, Inc., or MMRIS after completion of the reverse merger with
Favrille. The Company's Board approved an amendment to the Company's certificate
of incorporation to change the name of the Company and its trading symbol from
MMR Information Systems, Inc. to MMRGlobal, Inc. The change in name is pending
stockholder approval at the Company's shareholder meeting which will be held on
June 15, 2010. We provide secure and easy-to-use online Personal Health Records ("PHRs") and
electronic safe deposit box storage solutions, serving consumers, healthcare
professionals, employers, insurance companies, unions and professional
organizations and affinity groups. MyMedicalRecords enables individuals and
families to access their medical records and other important documents, such as
birth certificates, passports, insurance policies and wills, anytime from
anywhere using the Internet. The MyMedicalRecords Personal Health Record is
built on proprietary, patent pending, issued and applied for technologies to
allow documents, images and voicemail messages to be transmitted and stored in
the system using a variety of methods, including fax, phone, or file upload
without relying on any specific electronic medical record platform to populate a
user's account. The Company's professional offering, MMRPro, is designed to give
physicians' offices an easy and cost- effective solution to digitizing
paper-based medical records and sharing them with patients in real time. MMR is
an Independent Software Vendor Partner with Kodak to deliver an integrated
turnkey EMR solution for healthcare professionals. MMR is also an integrated
service provider on Google Health. Principles of Consolidation The consolidated financial statements include the accounts of MMRIS, and its
wholly-owned subsidiary MMR. All intercompany transactions have been eliminated
upon consolidation. Basis of Presentation The accompanying consolidated unaudited financial statements of the Company
have been prepared in accordance with accounting principles generally accepted
in the United States of America for interim financial statements and with
instructions to Form 10-Q pursuant to the rules and regulations of Securities
and Exchange Act of 1934, as amended (the "Exchange Act") and Article 8-03 of
Regulation S-X under the Exchange Act. Accordingly, they do not include all of
the information and footnotes required by U.S. Generally Accepted Accounting
Principles ("GAAP") for complete
F-40
financial statements. In the opinion of management, all adjustments
considered necessary (consisting of normal recurring adjustments) for a fair
presentation are included herein. Operating results for the three month period
ended March 31, 2010 are not indicative of the results that may be expected for
the fiscal year ending December 31, 2010. These unaudited consolidated financial
statements should be read in conjunction with the audited financial statements
and the notes thereto included in the Company's annual report on Form 10-K for
the year ended December 31, 2009. On July 1, 2009, MMRIS adopted Financial Accounting Standards
Board ("FASB") Accounting Standards Codification ("ASC") 105-10 (formerly
Statement of Financial Accounting Standards ("SFAS") No. 168, The FASB
Accounting Standards Codification and Hierarchy of Generally Accepted Accounting
Principles, a replacement of FASB Statement No. 162). ASC 105-10 establishes the
FASB ASC as the source of authoritative accounting principles recognized by the
FASB to be applied in preparation of financial statements in conformity with
generally accepted accounting principles in the United States of America. New
accounting standards issued subsequent to June 30, 2009 are communicated by
the FASB through Accounting Standards Updates ("ASU"s). The ASC is effective for
interim and annual reporting periods ending after September 15,
2009. The Company adopted ASC 105-10 during the year ended December
31, 2009 and revised its referencing of GAAP accounting standards in these
financial statements to reflect the new standards. Management's Plan As of March 31, 2010, the Company's current liabilities, of which $3.9
million represent non-cash derivative liabilities, exceeded its current assets
by $8.9 million. Furthermore, during the quarter ended March 31, 2010, the
Company incurred losses of $6.5 million. At March 31, 2010 and December 31, 2009, the Company had $878,442 and
$487,776, respectively, in cash and cash equivalents. Historically, the Company
issued capital stock, sold debt and equity securities and received funds from
The RHL Group, Inc. (a significant shareholder, wholly-owned by the Company's
Chairman and Chief Executive Officer) to operate its business. Although, the
Company received additional funding from The RHL Group pursuant to the Third
Amended and Restated Note dated April 29, 2009, it nevertheless will still be
required to obtain additional financing in order to meet installment payment
obligations resulting from settlement payments with various creditors, which we
refer to as the Creditor Plan and the previously existing obligations under the
subordinated secured indebtedness to The RHL Group, which note payable had a
balance of $1,473,934 at March 31, 2010. As a result of the above, there is
uncertainty about the Company's ability to continue as a going concern. Management's plan regarding this matter is to continue to utilize its
available line of credit with The RHL Group (see Note 3), if necessary. At March
31, 2010, there was approximately $1,500,000 available under The RHL Group line
of credit. Additionally, the Company plans to continue to sell additional debt
and equity securities, continue to settle its existing liabilities through
issuance of equity securities, explore other debt financing arrangements,
continue to increase its existing subscriber and affiliate customer base and
sell MMR Pro products to obtain additional cash flow over the next twelve
months. There can be no assurance that funds from these sources will be
available when needed or, if available, will be on terms favorable to us or to
our stockholders. If additional funds are raised by issuing equity securities,
the percentage ownership of our stockholders will be reduced, stockholders may
experience additional dilution or such equity securities may provide for rights,
preferences or privileges senior to those of the holders of our common
stock. If the Company is unable to utilize its available line of credit with The RHL
Group, or obtain suitable alternative debt financing, it may adversely affect
the Company's ability to execute its business plan and continue as a going
concern. NOTE 2 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (a) MANAGEMENT'S USE OF ESTIMATES The preparation of financial statements in accordance with GAAP requires
management to make estimates and assumptions that affect the reported amounts of
assets and liabilities at the date of the financial statements and the reported
amounts of net revenue and expenses during each reporting period. On an ongoing
basis, management evaluates its estimates, including those related to revenue
recognition, allowances for doubtful accounts, the valuation of deferred income
taxes, tax contingencies, long-lived and intangible assets, derivative
liabilities and stock-based compensation. These estimates are based on
historical experience and on various other factors that it believes to be
reasonable under the circumstances. Actual results could differ from those
estimates. F-41
(b) CASH AND CASH EQUIVALENTS The Company considers cash equivalents to be only those investments that are
highly liquid, readily convertible to cash and with maturities of 90 days or
less at the purchase date. The Company maintains its cash in bank deposit
accounts which, at times, may exceed federally insured limits. The Company has
not experienced any losses in such accounts and believes it is not exposed to
any significant credit risk on cash. The Company had cash and cash equivalents
of $878,442 and $487,766 as of March 31, 2010 and December 31, 2009,
respectively. (c) TRADE AND OTHER RECEIVABLES Receivables represent claims against third parties that will be settled in
cash. The carrying value of receivables, net of an allowance for doubtful
accounts, if any, represents their estimated net realizable value. The allowance
for doubtful accounts, if any, is estimated based on historical collection
trends, type of customer, the age of outstanding receivables and existing
economic conditions. If events or changes in circumstances indicate that
specific receivable balances may be impaired, further consideration is given to
the collectability of those balances and the allowance is adjusted accordingly.
Past due receivable balances are written-off when collection efforts have been
unsuccessful in collecting the amount due. (d) INVENTORIES Inventories are stated at the lower of standard cost (which approximates cost
determined on a first-in, first-out basis) or market. The Company writes down
the value of inventories for estimated excess and obsolete inventories based
upon assumptions about future demand and market conditions. Inventories consist
of finished goods. Inventory reserves, once established, are only reversed upon
sale or disposition of related inventories. (e) FAIR VALUE OF FINANCIAL INSTRUMENTS ASC 820-10 requires entities to disclose the fair value of financial
instruments, both assets and liabilities recognized and not recognized on the
balance sheet, for which it is practicable to estimate fair value. ASC 820-10
defines the fair value of a financial instrument as the amount at which the
instrument could be exchanged in a current transaction between willing parties.
As of December 31, 2009 and March 31, 2010, the carrying value of accounts
receivable, deposits, related party payables, compensation payable, severance
liabilities, and accounts payable and accrued expenses approximates fair value
due to the short-term nature of such instruments. The carrying value of
short-term debt approximates fair value as the related interest rates
approximate rates currently available to the Company. The Company utilizes ASC 820-10 for valuing financial assets and liabilities
measured on a recurring basis. ASC 820-10 defines fair value, establishes a
framework for measuring fair value and generally accepted accounting principles
and expands disclosures about fair value measurements. This standard applies in
situations where other accounting pronouncements either permit or require fair
value measurements. ASC 820-10 does not require any new fair value
measurements. Accounting guidance also establishes a fair value hierarchy which requires an
entity to maximize the use of observable inputs and minimize the use of
unobservable inputs when measuring fair value. The standard describes three levels of inputs that may be used to measure
fair value: Level 1: Quoted prices in active markets for identical or similar assets and
liabilities. Level 2: Quoted prices for identical or similar assets and liabilities in
markets that are not active or observable inputs other than quoted prices
in active markets for identical or similar assets and
liabilities. Level 3: Unobservable inputs that are supported by little or no market activity
and that are significant to the fair value of the assets or
liabilities. F-42
In determining the appropriate fair value of the option, warrant and
conversion feature liabilities, the Company used Level 3 inputs for its
valuation methodology. The Company applied the Black-Scholes model to value the
option, warrant and conversion feature liabilities. See Note 9 for the inputs
used in the Black-Scholes option valuation model. (f) PROPERTY AND EQUIPMENT Property and equipment are stated at cost. Equipment under capital leases is
stated at the present value of the minimum lease payments. Depreciation is
calculated using the straight-line method, based upon the following estimated
useful lives: Furniture and Fixtures: 5 Years Computer Equipment: 5 Years When items are retired or disposed of, income is charged or credited for the
difference between the net book value of the asset and the proceeds realized
thereon. Expenditures for maintenance and repairs are charged to operations as
incurred while renewals and betterments are capitalized. All property and equipment, along with all other assets of the Company, are
pledged as collateral for a line of credit from a related party (see Note 3 -
Related Party Note Payable). (g) INTANGIBLE ASSETS Intangible assets are comprised of website development costs, domain names
and patents. The Company accounts for website development costs in accordance
with ASC 350-50, Website Development Costs, and ASC 985-20, Costs of
Software to Be Sold, Leased or Marketed. Pursuant to ASC 350-50 and 985-20,
the Company capitalizes internally developed website costs when the website
under development has reached technological feasibility. These costs are
amortized, typically over an estimated life of five years, using the larger of
the amount calculated using the straight-line method or the amount calculated
using the ratio between current period gross revenues and the total of current
period gross revenues and estimated future gross revenues. At each balance sheet
date, the Company evaluates the unamortized capitalized website costs compared
to the net realizable value. The amount by which the unamortized capitalized
website costs exceed its net realizable value is written off. There were no
amounts written off during the three months ended March 31, 2010 and
2009. The Company accounts for domain names and patents in accordance with ASC
350-30, General Intangibles Other than Goodwill. The Company capitalizes
patent costs representing legal fees associated with filing patent applications
and amortizes them on a straight-line basis. The Company is in the process of
evaluating the patents' estimated useful life and will begin amortizing the
patents when they are brought to the market. Identifiable intangible assets are
amortized over their estimated useful lives as follows: Website Development Costs: 5 Years Domain Name: 5 Years F-43
(h) IMPAIRMENT OF LONG-LIVED ASSETS AND INTANGIBLES The Company accounts for long-lived assets, which include property and
equipment and identifiable intangible assets with finite useful lives (subject
to amortization), in accordance with ASC 350-30. ASC 350-30 requires that
long-lived assets be reviewed for impairment whenever events or changes in
circumstances indicate that the carrying amount of an asset may not be
recoverable. Recoverability is measured by comparing the carrying amount of an
asset to the expected future undiscounted net cash flows generated by the asset.
If it is determined that the asset may not be recoverable, or if the carrying
amount of an asset exceeds its estimated future undiscounted cash flows, an
impairment charge is recognized to the extent of the difference between the fair
value and the asset's carrying amount. The Company had no impairment charges
during the three months ended March 31, 2010 and 2009. (i) REVENUE RECOGNITION The Company's revenues are derived from services, which are comprised of
providing electronic access to consumer medical records and other vital
documents and from the licensing of its services. Revenue is recognized only
when the price is fixed or determinable, persuasive evidence of an arrangement
exists, the service is performed, and collectability of the resulting receivable
is reasonably assured. The Company's subscriber revenues consist of annual and monthly recurring
retail subscriptions and usage-based fees, which are primarily paid in advance
by credit card, and corporate accounts that are based on either an access-fee or
actual number of users, and in each case billed in advance at the beginning of
each month of service. The Company defers the portions of annual recurring
subscription fees collected in advance and recognizes them on a straight line
basis over the subscription period. The Company grants exclusive licenses for the sale and marketing of its
services in international territories in consideration of an up-front license
fee and an ongoing royalty. The royalty fee is usually a percentage of revenue
earned by the licensee and there usually are certain minimum guarantees. License
fee revenues received in advance from international licensees for the grant of
the license are deferred and recognized over the period covered by the
agreement. Minimum guaranteed royalty payments received in advance are deferred
and recognized over the period to which the royalty relates. All such revenues
are included under "License Fees and Other." In those cases where a license
agreement contains multiple deliverables, the agreement is accounted for in
accordance with ASC 605-25, Revenue Recognition - Multiple- Element
Arrangements. (j) INCOME TAXES AND UNCERTAIN TAX POSITIONS The Company accounts for income taxes in accordance with ASC 740-10,
Income Taxes. The Company recognizes deferred tax assets and liabilities
to reflect the estimated future tax effects, calculated at the tax rate expected
to be in effect at the time of realization. A valuation allowance related to a
deferred tax asset is recorded when it is more likely than not that some portion
of the deferred tax asset will not be realized. Deferred tax assets and
liabilities are adjusted for the effects of the changes in tax laws and rates of
the date of enactment. ASC 740-10 prescribes a recognition threshold that a tax position is required
to meet before being recognized in the financial statements and provides
guidance on recognition, measurement, classification, interest and penalties,
accounting in interim periods, disclosure and transition issues. The Company
classifies interest and penalties as a component of interest and other expenses.
To date, there have been no interest or penalties assessed or paid. The Company measures and records uncertain tax positions by establishing a
threshold for the financial statement recognition and measurement of a tax
position taken or expected to be taken in a tax return. Only tax positions
meeting the more-likely-than-not recognition threshold at the effective date may
be recognized or continue to be recognized. (k) ADVERTISING The Company expenses advertising costs as incurred. Advertising expense for
the three months ended March 31, 2010 and 2009 amounted to $0, and $808,
respectively. F-44
(l) SHARE-BASED COMPENSATION The Company accounts for share-based compensation in accordance with ASC
718-20, Awards Classified as Equity. The Company applies ASC 718-20 in
accounting for stock-based awards issued to employees under the recognition of
compensation expense related to the fair value of employee share-based awards,
including stock options and restricted stock. Determining the fair value of
options at the grant date requires judgment, including estimating the expected
term that stock options will be outstanding prior to exercise, the associated
volatility and the expected dividends. Judgment is required in estimating the
amount of share-based awards expected to be forfeited prior to vesting. If
actual forfeitures differ significantly from these estimates, share-based
compensation expense could be materially impacted. Options and warrants issued to non-employees are accounted for in accordance
with ASC 505- 50, Equity-Based Payments to Non-Employees. Options and
warrants issued to non-employees are treated the same as those issued to
employees with the exception of determination of the measurement date. The
measurement date is the earlier of (1) the date at which a commitment for
performance by the counterparty to earn the equity instruments is reached, or
(2) the date at which the counterparty's performance is complete. Options and
warrants granted to consultants are valued at their respective measurement
dates, and recognized as expense based on the portion of the total consulting
services provided during the applicable period. As further consulting services
are provided in future periods, the Company will revalue the associated options
and warrants and recognize additional expense based on their then current
values. The fair value of each stock option is estimated on the date of grant using
the Black-Scholes option pricing model. Assumptions relative to volatility and
anticipated forfeitures are determined at the time of grant. Grants of stock
options and warrants during the three months ended March 31, 2010 and 2009 were
valued using the following assumptions. The assumptions used in the Black-Scholes models are based upon the following
data: (1) The Company uses the contractual life of the underlying non-employee
warrants as the expected life; the expected life of the employee options used in
this calculation is the period of time the options are expected to be
outstanding and has been determined based on historical exercise experience. (2)
In the absence of an extensive public market for the Company's shares, the
expected stock price volatility of the underlying shares over the expected term
of the option or warrant was taken at approximately the mid-point of the range
for similar companies at the various grant dates. (3) The risk free interest
rate is based on published U.S. Treasury Department interest rates for the
expected terms of the underlying options or warrants. (4) Expected dividends are
based on historical dividend data and expected future dividend activity. (5) The
expected forfeiture rate is based on historical forfeiture activity and
assumptions regarding future forfeitures based on the composition of current
grantees. (m) NET INCOME/LOSS PER SHARE The Company applies the guidance of ASC 260-10, Earnings Per Share for
calculating the basic and diluted loss per share. Basic loss per share is
calculated by dividing the net loss attributable to common shareholders by the
weighted average number of common shares outstanding. Diluted loss per share is
computed similar to basic loss per share except that the denominator is
increased to include the number of additional common shares that would have been
outstanding if the potential shares had been issued and if the additional shares
were dilutive. Common equivalent shares are excluded from the computation of net
loss per share if their effect is anti-dilutive. F-45
All potential common shares were excluded from the computation of diluted net
loss per common share for the three months ended March 31, 2010 and 2009 because
they were anti-dilutive due to the Company's net loss position. Stock options,
warrants and convertible notes excluded from the computation totaled 76,642,407
shares for the three months ended March 31, 2010 and 34,626,997 shares for the
three months ended March 31, 2009. (n) RESEARCH, DEVELOPMENT AND ENGINEERING Research, development and engineering costs are expensed as incurred and
presented as technology development in the accompanying consolidated statements
of operations. Costs for software development relating to our website incurred
subsequent to establishing technological feasibility, in the form of a working
model, are capitalized and amortized over their estimated useful lives. (o) CONCENTRATIONS For the three months ended March 31, 2010, our three largest customers
(Alexian at $38,970, Coverdell at $26,597 and Medicalert at $15,506) accounted
for approximately 90% of our total revenue. For the three months ended March 31,
2009, the then three largest customers (MMR Australia at $53,292, Alexian at
$38,616 and Medicalert at $19,515) accounted for approximately 76.4% of our
total revenue. (p) RECENT ACCOUNTING PRONOUNCEMENTS In October 2009, the FASB issued ASU No. 2009-13,
Multiple-Deliverable Revenue Arrangements-a consensus of the FASB Emerging
Issues Task Force, that provides amendments to the criteria for separating
consideration in multiple-deliverable arrangements. As a result of these
amendments, multiple-deliverable revenue arrangements will be separated in more
circumstances than under existing U.S. GAAP. The ASU does this by establishing a
selling price hierarchy for determining the selling price of a deliverable. The
selling price used for each deliverable will be based on vendor-specific
objective evidence if available, third-party evidence if vendor-specific
objective evidence is not available, or estimated selling price if neither
vendor-specific objective evidence nor third-party evidence is available. A
vendor will be required to determine its best estimate of selling price in a
manner that is consistent with that used to determine the price to sell the
deliverable on a standalone basis. This ASU also eliminates the residual method
of allocation and will require that arrangement consideration be allocated at
the inception of the arrangement to all deliverables using the relative selling
price method, which allocates any discount in the overall arrangement
proportionally to each deliverable based on its relative selling price. Expanded
disclosures of qualitative and quantitative information regarding application of
the multiple-deliverable revenue arrangement guidance are also required under
the ASU. The ASU does not apply to arrangements for which industry specific
allocation and measurement guidance exists, such as long-term construction
contracts and software transactions. The ASU is effective beginning
January 1, 2011. The Company is currently evaluating the impact of this
standard on the Company's financial statements. In October 2009, the FASB issued ASU No. 2009-14, Certain
Revenue Arrangements That Include Software Elements-a consensus of the FASB
Emerging Issues Task Force, that reduces the types of transactions that fall
within the current scope of software revenue recognition guidance. Existing
software revenue recognition guidance requires that its provisions be applied to
an entire arrangement when the sale of any products or services containing or
utilizing software when the software is considered more than incidental to the
product or service. As a result of the amendments included in ASU
No. 2009-14, many tangible products and services that rely on software will
be accounted for under the multiple-element arrangements revenue recognition
guidance rather than under the software revenue recognition guidance. Under the
ASU, the following components would be excluded from the scope of software
revenue recognition guidance: the tangible element of the product,
software products bundled with tangible products where the software components
and non-software components function together to deliver the product's essential
functionality, and undelivered components that relate to software that is
essential to the tangible product's functionality. The ASU also provides
guidance on how to allocate transaction consideration when an arrangement
contains both deliverables within the scope of software revenue guidance
(software deliverables) and deliverables not within the scope of that guidance
(non-software deliverables). The ASU is effective beginning January 1,
2011. The Company is currently evaluating the impact of this standard on the
Company's financial statements. F-46
In January 2010, the FASB issued ASU No. 2010-6, Improving Disclosures
About Fair Value Measurements, that amends existing disclosure requirements
under ASC 820 by adding required disclosures about items transferring into and
out of levels 1 and 2 in the fair value hierarchy; adding separate disclosures
about purchase, sales, issuances, and settlements relative to level 3
measurements; and clarifying, among other things, the existing fair value
disclosures about the level of disaggregation. This ASU is effective for the
first quarter of 2010, except for the requirement to provide level 3 activity of
purchases, sales, issuances, and settlements on a gross basis, which is
effective beginning the first quarter of 2011. The Company is currently
evaluating the impact of this standard on the Company's financial
statements. In February 2010, the FASB issued ASU No. 2010-09, Amendments to Certain
Recognition and Disclosure Requirements. Questions have arisen in practice
about Topic 855, Subsequent Events. Specifically, constituents have informed the
Board that the requirements to disclose the date that the financial statements
are issued potentially conflict with some of the Securities and Exchange
Commission's (SEC) guidance. This Update addresses both the interaction of the
requirements of Topic 855, Subsequent Events, specifically regarding the
requirements to disclose the date that the financial statements are issued, with
the SEC's reporting requirements and the intended breadth of the reissuance
disclosure provision related to subsequent events. The Company has evaluated
subsequent events through the date of issuance in accordance with ASU No
2010-09. In April 2010, the FASB issued ASU No. 2010-13, Effect of Denominating the
Exercise Price of a Share-Based Payment Award in the Currency of the Market in
Which the Underlying Equity Security Trades. This Update addresses the
classification of a share-based payment award with an exercise price denominated
in the currency of a market in which the underlying equity security trades.
Topic 718 is amended to clarify that a share-based payment award with an
exercise price denominated in the currency of a market in which a substantial
portion of the entity's equity securities trades shall not be considered to
contain a market, performance, or service condition. Therefore, such an award is
not to be classified as a liability if it otherwise qualifies as equity
classification. This update did not impact the Company. NOTE 3 - RELATED PARTY NOTE PAYABLE On July 31, 2007, MMR entered into a promissory note agreement and a security
agreement with The RHL Group, a corporation wholly owned by Robert H. Lorsch,
the Chairman and CEO of the Company, to borrow up to $100,000 under a revolving
line of credit. This agreement was subsequently superseded by an amended and
restated promissory note on August 23, 2007 under the terms of which the line
amount was increased to $1 million. On April 29, 2009, the Company agreed to
restructure MMR's secured credit facility with The RHL Group and entered into a
Secured Credit Restructuring Agreement with MMR, The RHL Group and Mr. Lorsch,
(the "Restructuring Agreement"). MMR issued The RHL Group a Third Amended and
Restated Note (the "Third Amended Note"), and the Company agreed to guaranty
MMR's obligations under the Third Amended Note (the "Guaranty"). The Third Amended Note amends and restates the previous The RHL Group note
("RHL Group Note"), matured November 30, 2009, and bears interest at the lesser
of 10% or the highest rate then permitted by law, and is secured (similar to the
previous note) by the Security Agreement. The RHL Group Note was renewed for an
additional six-month term. Although the reserve credit line has been increased
to $3,000,000, The RHL Group is only obligated to make a minimum of $100,000 of
loans, advances and guarantees under the Third Amended Note within 30 days of
execution pursuant to the Restructuring Agreement. The RHL Group Note payable
had a balance of $1,473,934 at March 31, 2010. The components of the Group Note
payable and the related balance sheet presentation as of March 31, 2010 are: (1)
$976,924, which is included in the line of credit, related party; (2) $283,694,
which is related to credit card charges and a vendor guaranty, is included in
accounts payable and accrued expenses; and (3) $213,316, which is related to
deferred salary and consulting expenses is included in related party payables.
The RHL Group was due to receive, as an origination fee ("Fee"), a promissory
note for $200,000, bearing interest at 10% per annum and due on demand. On
August 17, 2009, the Company paid the $200,000 Fee by granting 2,800,000 shares
of common stock to The RHL Group to the value of $364,000 and recorded a loss on
issuance of $164,000. The Fee was recorded as a deferred financing cost and is
being amortized over the Third Amended Note maturity period. As of March 31,
2010 and December 31, 2009, the remaining unamortized origination fee was $0.
Total interest expense on this note for the three months ended March 31, 2010
and 2009 amounted to $36,304 and $20,434, respectively. The unpaid interest
balance as of March 31, 2010 and December 31, 2009 was $81,458 and $45,155,
respectively. F-47
In conjunction with the loan agreement noted above, MMR was required to
maintain certain financial covenants, including the requirement that MMR have at
least $100,000 in cash in its bank accounts or such other amount as necessary to
maintain operations through the subsequent thirty (30) days and timely pay any
obligations due respecting payroll and all associated payroll taxes on and after
June 1, 2009. As of March 31, 2010, MMR was in compliance with its financial
covenants under the terms of the Third Amended Note. On May, 12, 2010, MMR issued The RHL Group a Fourth Amended and Restated Note
(the "Fourth Amended Note"), and the Company agreed to guaranty MMR's
obligations under the Fourth Amended Note (the "Guaranty"). Refer to Note 14 -
Subsequent Events. NOTE 4 - PREPAID EXENSES AND OTHER CURRENT ASSETS Prepaid expenses and other current assets include the following: NOTE 5 - ACCOUNTS PAYABLE AND ACCRUED EXPENSES Accounts payable and accrued expenses include the following: F-48
NOTE 6 - INCOME TAXES Under ASC 740-270, Income Taxes - Interim Reporting, the Company is
required to adjust its effective tax rate each quarter to be consistent with the
estimated annual effective tax rate. The Company is also required to record the
tax impact of certain discrete items, unusual or infrequently occurring,
including changes in judgment about valuation allowances and effects of changes
in tax laws or rates, in the interim period in which they occur. In addition,
jurisdictions with a projected loss for the year or a year-to-date loss where no
tax benefit can be recognized are excluded from the estimated annual effective
tax rate. The impact of such an exclusion could result in a higher or lower
effective tax rate during a particular quarter, based upon the mix and timing of
actual earnings versus annual projections. MMR, in its capacity as the operating company taking over MMRIS's income tax
positions in addition to its own positions after January 27, 2009 (see Note 1),
has estimated its annual effective tax rate to be zero. This is based on an
expectation that the combined entity will generate net operating losses in 2010,
and it is not likely that those losses will be recovered using future taxable
income. Therefore, no provision for income tax has been recorded as of and for
the three months ended March 31, 2010. NOTE 7 - COMMITMENTS AND CONTINGENCIES Leases The Company leases certain facilities and equipment under non-cancelable
capital and operating leases, which expire at various dates through 2011.
Effective May 1, 2009, the Company entered into a lease agreement to lease
additional office space in Beverly Hills, California. The lease is
month-to-month and requires a monthly payment of $7,667 commencing in June 2009.
Total rent expense for the three months ended March 31, 2010 and 2009 was
$17,225 and $9,000, respectively. Future minimum lease payments as of March 31,
2010, under non-cancelable operating leases (with initial or remaining lease
terms in excess of one year) and future minimum capital lease payments are as
follows. Guarantee provided by The RHL Group Under the terms of an agreement with an investor who purchased $500,000 of
MMR's Series B Preferred shares in 2006, the Company entered into an agreement
to invest $250,000 in a joint venture with the investor to establish an entity
to market and sell the Company's services in the countries of Japan, China,
Korea, Taiwan and Thailand. The Company has paid $100,000 to date of this amount
which the Company has expensed during the year ended December 31, 2009 as the
Company has since terminated its relationship with the investor. To date, no
joint venture has been formalized or incorporated and no operations have
commenced. In September 2007, The RHL Group provided the investor with a
guarantee that the Company would meet its obligations under this agreement in
exchange for 300,000 shares of restricted MMR common stock valued at $39,000. As
consideration for renewing the guarantee when it expired in September 2008, in
January 2009, MMR issued The RHL Group 100,000 shares of MMR common stock, which
became 328,174 shares of MMRIS common stock upon the closing of the Merger,
valued at approximately $5,000. This expense has been reflected in general and
administrative expenses in the accompanying consolidated statement of operations
for the year ended December 31, 2009. On January 6, 2010, the Company entered
into a settlement agreement with the investor to provide for full and final
settlement of any and all claims pertaining to any license rights for MMR-Asia
to market, sell and sublicense MMRIS and MMR products and services. As part of
this settlement agreement, the Company granted 1,388,889 shares of common stock
to the investor in January 2010. The value of these shares amounted to $138,889,
and has been recorded as an expense in the accompanying consolidated statement
of operations during the year ended December 31, 2009. F-49
Guarantee provided by Robert H. Lorsch On August 17, 2009, Robert H. Lorsch, the Company's Chairman, Chief Executive
Officer and President, agreed to guarantee $282,641 in payments due to a vendor
for services rendered to the Company. On August 17, 2009, in consideration of a
personal guaranty given by Mr. Lorsch, the Company granted Mr. Lorsch (i) a
warrant to purchase 706,605 shares of Company common stock, at an exercise price
of $0.13 per share, the closing price of the Company's common stock on the date
immediately preceding the date of grant, and (ii) 141,321 shares of Company
common stock. Employment Agreements The Company has employment agreements with its Chairman, President and Chief
Executive Officer, Robert H. Lorsch, its Vice President of Finance and Chief
Financial Officer, Ingrid Safranek, and its Executive Vice President of
Technology and Product Development, Naj Allana. Under each employment agreement,
the executive officers receive a base salary, subject to annual increases as
determined by the board of directors, certain benefits as set forth in the
employment agreements, and an annual bonus at the discretion of the board of
directors. The current term of Mr. Lorsch's employment agreement is effective until
December 31, 2011 and will automatically renew for successive 12 month periods
unless terminated at least 60 days prior to the end of the term. The employment
agreement may be terminated by the Company without cause (as defined in the
agreement) upon 30 days written notice or for cause if Mr. Lorsch fails to cure
the acts or omissions constituting cause within 30 days. If Mr. Lorsch's
employment is terminated by the Company for cause or voluntarily by Mr. Lorsch
without good reason, he will not be entitled to receive any severance payments
or benefits under the employment agreement. If Mr. Lorsch's employment is
terminated by the Company without cause or voluntarily by Mr. Lorsch for good
reason, Mr. Lorsch will be entitled to one year of salary at his then current
rate of pay, including all monthly benefits, and the pro rata portion of the
annual bonus otherwise due Mr. Lorsch. Mr. Lorsch's employment agreement includes provisions that prohibit Mr.
Lorsch from disclosing the Company's confidential information and trade secrets
and competing with the Company during the term of his employment agreement or
soliciting the Company's employees for 18 months following termination of
employment. The Company also approved an employment agreement with our former Chief
Financial Officer and Senior Vice President and current Executive Vice President
of Technology and Product Development, Naj Allana. The agreement provides for a
base salary of $15,845 per month, subject to an upward increase. In addition to
his base salary, Mr. Allana is entitled to receive a commission with respect to
certain of our accounts. Mr. Allana's employment agreement was effective until February 15, 2010 and
automatically renews for successive 12 month periods unless terminated at least
120 days prior to the end of the term. If we terminate Mr. Allana's employment,
other than for misconduct (as defined in the employment agreement), we would
continue to pay Mr. Allana his monthly salary for the remainder of the then
current term, or if done 120 days preceding the end of the term, then through
the end of the next 12 month term. On January 26, 2010, the Company entered into an employment agreement with
Ingrid Safranek as our Vice President, Chief Financial Officer and Secretary.
Under the employment agreement, Ms. Safranek receives a base salary, subject to
annual increases as determined by the board of directors, certain benefits as
set forth in the employment agreements, and an annual bonus at the discretion of
the board of directors. Ms. Safranek's employment agreement is in effect until
June 15, 2010, and shall automatically renew for successive one-year periods,
unless extended or otherwise terminated at least 60 days prior to the end of the
term. If the Company terminates Ms. Safranek's employment, unless due to misconduct
(as defined in the employment agreement), the Company must continue to pay Ms.
Safranek her monthly salary for two (2) months, if such termination occurs
within the first year of employment; for six (6) months, if such termination
occurs between the first year and the last day of the second year of employment;
and for twelve (12) months, if such termination occurs after the last day of the
second year of employment. For purposes of clarification, a change in title or
diminution of responsibilities, regardless of the level of materiality, shall
not be considered a termination under the provisions of this Agreement. F-50
Litigation Matters From time to time, the Company is involved in various legal proceedings
generally incidental to its business. While the result of any litigation
contains an element of uncertainty, management presently believes that the
outcome of any known, pending or threatened legal proceeding or claim,
individually or combined, will not have a material adverse effect on the
Company's financial statements. On April 30, 2010, the Company filed a petition in the New York Supreme Court
against the Lymphoma Research Foundation ("LRF"), a California nonprofit
corporation, to enforce the Company's rights to a portion of certain
intellectual property and biotechnology assets acquired as a part of the reverse
merger with Favrille. LRF has claimed ownership of some of these assets
including samples and data which were transferred to LRF in the summer of 2008.
Also on April 30, 2010, LRF filed a declaratory relief action in the New York
Supreme Court asserting its rights over the aforementioned samples and data.
Both the Company and LRF have agreed to mediate the matter. NOTE 8 - STOCKHOLDERS' DEFICIT Preferred Stock As of December 31, 2008, there were 3,332,694; 1,263,750; and 1,580,082
shares of MMR's Series A, Series B and Series C Preferred Stock, respectively,
issued and outstanding. Immediately prior to the Merger, the outstanding MMR
preferred stock was converted to MMR common stock, representing an aggregate of
6,176,526 shares, which converted into MMRIS common stock in the Merger on
January 27, 2009. As of March 31, 2010 and December 31, 2009, there were no
shares of MMR preferred stock issued and outstanding. The Company has 5,000,000 shares of MMRIS preferred stock authorized. As of
March 31, 2010 and December 31, 2009, there were no shares of MMRIS preferred
stock issued and outstanding. Common Stock As of March 31, 2010, the Company was authorized to issue 650,000,000 shares
of common stock. Immediately prior to the Merger, there were 41,254,550 shares
of MMRIS common stock issued and outstanding. On July 10, 2009, the stockholders
of the Company approved an increase to the shares of common stock it is
authorized to issue. As of July 10, 2009, the Company was authorized to issue
650,000,000 shares of common stock. Pursuant to the Merger Agreement, MMRIS agreed to issue (or reserve for
issuance) an aggregate 92,599,196 shares of MMRIS common stock to the
stockholders of MMR and the holders of options and warrants to acquire MMR
common stock at the time of the Merger. Immediately prior to the Merger, and including the conversion of 6,176,526
shares of MMR preferred stock into MMR common stock, the issuance of 100,000
shares of MMR common stock to The RHL Group for renewal of a guarantee (see Note
7), MMR had 24,320,100 shares of common stock issued and outstanding. At the
effective time of the Merger, and in accordance with the terms of the Merger
Agreement, these shares of outstanding MMR common stock automatically converted
into 79,812,087 shares of MMRIS common stock. As of March 31, 2010, the total shares of the Company's common stock issued
and outstanding amounted to 204,409,509. Warrants for Purchase of Preferred B shares On April 1, 2007, MMR granted 200,000 warrants to purchase Series B Preferred
Stock of MMR at an exercise price of $2.00. The warrants were issued to the
largest distributor of employee assistance services in the US as an incentive to
market and sell MMR's services into their customer base. The related services
agreement was for five years, and the customer never bought any services from
MMR. F-51
Upon closing of the Merger on January 27, 2009, these warrants converted into
warrants to purchase 656,346 shares of MMRIS common stock at an exercise price
of $0.61 per share. There was no additional expense recorded during the nine
months ended September 30, 2009 resulting from the modification of these
warrants. NOTE 9 - EQUITY ISSUANCES Stock Option Activity On January 21, 2010, the Company's Board of Directors approved an increase to
the number of shares authorized for issuance under the 2001 Equity Incentive
Plan (the "Plan") from 12,000,000 to 27,000,000 shares as the original Plan was
not deemed adequate to retain key directors, executives and managers. A summary of option activity for the three months ended March 31, 2010 is
presented below. Options granted by MMR prior to the date of the Merger of
January 27, 2009 have been retroactively restated to reflect the conversion
ratio of MMR to MMRIS shares. The aggregate intrinsic value in the table above is before applicable income
taxes and is calculated based on the difference between the exercise price of
the options and the quoted price of the common stock as of the reporting
date. Total stock option expense recorded during the three months ended March 31,
2010 and 2009 amounted to $656,539 and $112,709, respectively, and is reflected
in operating expenses in the accompanying consolidated statements of operations.
The following table summarizes information about stock options outstanding
and exercisable at March 31, 2010. F-52
Warrants On November 7, 2007, in conjunction with the registered direct offering
of an aggregate of 7.4 million shares, the Company issued warrants to purchase
up to 4.4 million shares of the Company's common stock at an exercise price of
$2.77 per share. The warrants are exercisable as of the date of grant through
November 7, 2012. The Company valued the warrants using a Black-Scholes
pricing model. ASC 815-40, Contracts in Entity's Own Equity, requires
freestanding contracts that are settled in a Company's own stock, including
common stock warrants to be designated as an equity instrument, asset or
liability. A contract designated as an asset or a liability must be carried at
fair value until exercised or expired, with any changes in fair value recorded
in the results of operations. ASC 815-40 was applied to determine that the
liability is accounted for as a derivative (see annual report on Form 10-K for
the year ended December 31, 2008, filed on April 15, 2009 for a more detailed
discussion). The valuation of these warrants amounted to $266,994 and $88,998 as
of March 31, 2010 and December 31, 2009, respectively. The increase in fair
value of $177,996 was presented as change in valuation of derivative liabilities
in the accompanying consolidated statements of operations. Immediately prior to the Merger, MMRIS issued warrants to acquire 9,999,992
shares of MMRIS's common stock at an exercise price of $0.12 per share, which
expire on January 26, 2014, to certain former officers, former directors and
their affiliates who were willing to take such equity as partial or full payment
for outstanding liabilities. These warrants vested immediately and were valued
on the date of grant using the Black-Scholes option pricing model. As this
transaction occurred immediately prior to the Merger, the resulting gain on the
issuance of warrants was reflected in the statement of operations of MMRIS.
There is no impact of this transaction recorded on the accompanying consolidated
statement of operations for the three months ended March 31, 2009. In January and March 2010, the Company granted to one employee, two different
related parties and two different unrelated third-parties (the "Holders") a
total of 8,050,875 warrants to purchase shares of the Company's common stock in
connection with the 12% Convertible Promissory Note ("the Convertible Notes") at
an exercise price equal to the lesser of (i) the product of 50% multiplied by
the arithmetic average of the dollar volume-weighted average price ("VWAP") of
the common stock for the 10 consecutive trading days ending on the day that is 3
Trading Days prior to the applicable exercise date or (ii) $0.15. The total
value of these warrants amounted to $845,482, which was bifurcated from the
Convertible Notes and accounted for as a discount on debt. On the date of
investment, all the aforementioned parties converted their promissory notes and
exercised the attached warrants. As a result of the warrant exercise, the
$845,482 discount was recorded to interest expense during the three months ended
March 31, 2010 with an offset to additional paid in capital. See Note 11 below.
On January 26, 2010, the Company issued warrants to Alexian Brothers
Foundation's building and capital fund consisting of two warrants: (a) an
immediate grant of a warrant to acquire up to 500,000 shares of Common Stock,
exercisable at any time on or before December 31, 2011, with an exercise price
of $0.25 per share payable in cash or through a cashless exercise at the option
of the holder and (b) an immediate grant of a warrant to acquire up to 500,000
shares of Common Stock, exercisable at any time on or before December 31, 2013,
with an exercise price of $0.35 per share payable in cash or through a cashless
exercise at the option of the holder. These warrants had an aggregate value of
$62,251, and were recorded in sales and marketing expenses during the three
month ended March 31, 2010. In March 2010, the Company granted an aggregate total of 650,000 warrants to
purchase shares of the Company's common stock as payment for services. The
warrants had exercise prices of $0.12 per share, with contractual lives through
March 1, 2015. Of the warrants issued, 150,000 vested immediately, and 500,000
begin vesting in September 2010 after the consultant has completed six months of
service. From the vesting commencement date, these warrants vest in eight
quarterly installments through September 30, 2012. The warrants had an aggregate
value of $63,451, of which $15,017 was recorded as operating expenses during the
three months ended March 31, 2010. F-53
The following summarizes the total warrants outstanding and exercisable as of
March 31, 2010. The weighted average fair value of warrants granted during the three months
ended March 31, 2010 amounted to $0.05 per warrant. Shares Issued for Services or Reduction to Liabilities During the three months ended March 31, 2010, the Company issued 4,584,696
shares of common stock with a value of $457,054 to non-employees and charged to
the appropriate accounts for the following reasons: (1) Includes 147,124 shares issued to two members of the Board of Directors
valued at $21,000. The 4,584,696 shares were not contractually restricted, however as they were
unregistered they were restricted from sale until the requirements of Rule 144
under the Securities Act permit such sale. All such shares were issued at the
trading closing price on the date of issuance and such value was calculated
therefrom. Restricted Stock Program Under the Restricted Stock Program, a restricted stock award is an offer by
the Company to sell to an eligible person shares that are subject to
restrictions relating to the sale or transfer of the shares. A committee
appointed by the Board to administer the program or the Board itself shall
determine to whom an offer will be made, the number of shares the person may
purchase, the price to be paid and the restriction to which the shares shall be
subject. The offer must be accepted by the eligible person within thirty days
from the date of the offer evidenced by the Restricted Stock Purchase Agreement.
The purchase price of shares shall not be less than 85% of the fair market value
of such shares on the issue date, with the provision that the purchase price for
a 10% stockholder shall not be less than 110% of such fair market value. Shares
are either fully and immediately vested upon issuance, or may vest in
installments upon attainment of specified performance objectives. No shares were
issued under the Restricted Stock Program during the three months ended March
31, 2010 or 2009. F-54
Stock Bonus Program Under the Stock Bonus Program, shares are issued as a bonus for past services
rendered pursuant to the Stock Bonus Agreement. Stock bonuses may be awarded
upon satisfaction of specified performance goals pursuant to the Performance
Stock Bonus Agreement. No shares were issued under the Stock Bonus Program
during the three months ended March 31, 2010 or 2009. Derivative Liabilities In accordance with ASC 815-40, on January 27, 2009, the Company performed an
analysis as if all holders of options and warrants exercised their rights under
those contracts. This analysis resulted in the conclusion that the Company had
inadequate authorized shares to settle 100% of these contracts. Therefore, as
shareholder approval would be necessary to increase the number of authorized
shares, settlement of these obligations would not be within the control of the
Company. Consequently certain non-employee options and warrants were accounted
for as derivative liabilities in accordance with ASC 815-40. The event giving rise to this condition was the merger between Favrille, Inc.
and MyMedicalRecords.com, Inc. on January 27, 2009. On that date, the fair value
of 3,436,458 vested non-employee options and 15,218,890 vested warrant contracts
subject to this analysis was reclassified from equity to derivative liabilities.
The condition was remedied on June 13, 2009 when certain of the option and
warrant holders waived their rights under those contracts until such time that
the Company had 250 million or more shares authorized. On March 31, 2009, these
contracts were again valued using the Black-Scholes option valuation model and
the $1,794,837 difference between the original value at January 27, 2009 of
$871,052 and the value at March 31, 2009 of $2,665,889 was recorded as a loss on
change in value of derivatives for the three months ended March 31, 2009. On July 16, 2009, the Company entered into a 12% Convertible Promissory Note
with an unrelated third-party for a principal amount totaling $30,000.
Subsequently, the Company entered into identical 12% Convertible Promissory
Notes ("the Convertible Notes") with four other unrelated third-parties for an
additional $530,000. Each Convertible Note is convertible into shares of common
stock by dividing (i) the then outstanding balance of such note by (ii) the
product of eighty percent (80%) multiplied by the arithmetic average of the
VWAP of the common stock for the ten (10)
consecutive trading days ending on the day that is three (3) trading days prior
to the applicable conversion date. In addition, the Company granted a total of
3,110,000 warrants to purchase shares of the Company's common stock in
connection with the Convertible Notes at an exercise price equal to the lesser
of (i) the product of 50% multiplied by the arithmetic VWAP of the common stock
for the 10 consecutive trading days ending on the day that is 3 Trading Days
prior to the applicable exercise date or (ii) $0.15. Such arrangement resulted
in the possibility that the Company may not have enough authorized shares to
settle these obligations using common stock. Instead, there is the possibility,
although remote, that it might need to settle one or more of these contracts
with cash. Consequently certain non-employee options and warrants were accounted
for as derivative liabilities as of July 16, 2009. The event giving rise to this condition was the first Convertible Note the
Company entered into on July 16, 2009. As of July 16, 2009 and for all
subsequent options and warrants issued prior to March 31, 2010, the fair value
of vested non-employee options and warrant contracts subject to this analysis
was reclassified from equity to derivative liabilities. On March 31, 2010, these
contracts were again valued using the Black-Scholes option valuation model and
the difference between the value at December 31, 2009 of $1,534,824 plus the
value of options and warrants granted to non-employees during the three months
ended March 31, 2010 of 89,770, including the cancellation of exercised warrants
and options of $19,814 and the value at March 31, 2010 of $3,576,430, was
recorded as a loss on change in value of derivatives. The loss recorded related
to this matter for the three months ended March 31, 2010 was $1,971,650 and is
included in the change in valuation of derivative liabilities. None of the derivatives liabilities were designated as hedging
instruments. F-55
The following is a reconciliation of all derivative liabilities: The inputs used for the Black-Scholes option valuation model were as follows.
On May 4, 2010, the $3,576,430 derivative liability was
reclassified back into equity and will no longer be accounted for as liability.
As of March 31, 2010, two unrelated third-parties had a principal balance of
$100,000 and 250,000 warrants to purchase common stock outstanding in connection
with the 12% Convertible Promissory Notes. On May 4, 2010, the two parties
converted the $100,000 balance and exercised the 250,000 warrants and received
886,943 shares of common stock. As a result, the condition leading to the
$3,576,430 derivative liability was remediated and the derivative liability was
reclassified back into equity. NOTE 10 - NOTES PAYABLE F-56
On March 16, 2010, the $125,000 principal and unpaid interest on the PM
Creative Corporation Promissory Note was converted into a 12% Convertible
Promissory Note. See Note 11. NOTE 11 - CONVERTIBLE PROMISSORY NOTES In January and March 2010, the Company entered into 12% Convertible
Promissory Notes with one employee, two related parties and two unrelated
third-parties for a principal amount totaling $1,250,175. Under the terms of the
agreements, principal amounts owed under the Notes become due and payable on
June 30, 2010, August 31, 2010 and September 30, 2010, provided that, upon ten
(10) days' prior written notice to the Holder, the Company may, in its sole
discretion, extend the maturity date to December 31, 2010, January 31, 2011 and
March 31, 2011.The 12% Convertible Promissory Notes bear an interest of 12% per
annum payable in cash or shares of common stock, or a combination of cash and
shares of common stock. The Notes are convertible at the option of the holders into common stock at a
variable conversion price of eighty percent (80%) multiplied by the arithmetic
average of the volume-weighted average trading price of the Common Stock for the
ten (10) consecutive trading days ending on the day that is three (3) trading
days prior to the applicable conversion date, subject to anti-dilution and other
customary adjustments. In connection with the Notes, the Company also issued warrants to purchase an
aggregate of 8,050,875 shares of common stock. The term of the warrants is three
years and the exercise price is the lesser of (i) the product of fifty percent
(50%) multiplied by the arithmetic average of the volume-weighted average
trading price of the Common Stock for the ten (10) consecutive trading days
ending on the day that is three (3) trading days prior to the applicable
exercise date, or (ii) $0.15, subject to anti-dilution and other customary
adjustments. By virtue of the Notes' variable conversion ratio, a possibility exists,
although remote, that the Notes may be convertible into shares of Common Stock
that exceed the currently authorized limit. Consequently, a possibility exists
that the Company might be placed in a position of net settling the Notes for an
amount that exceeds their face value. This resulted in the conversion feature
being a derivative under ASC 815-10. Additionally, as a result of the above, all outstanding options and warrants
issued to non-employees, including the warrants issued in connection with the
Notes, are also derivatives as they too could require net settlement in the
absence of adequate authorized shares. See Note 9 Derivative Liabilities.
The fair value of the conversion feature of the Notes and related warrants
was derived using the Black-Scholes option valuation model, resulting in an
aggregate fair value of $1,958,300. For five of the Notes issued during the
quarter ended March 31, 2010, the fair value of the conversion feature of the
Notes and related warrants exceeded the face value of the Notes by $710,093 and
such amount was recorded as a deferred financing cost. The fair value of the
conversion feature of the Notes and related warrants, net of the deferred
financing cost, was deducted from the Notes, resulting in an initial value
ascribed to the Notes of $1,968. The initial value of the Notes is being
accreted to their face value over the initial term of the Notes using the
interest method. Upon conversion, the entire unamortized discount from the face
value of the Notes is recognized as interest expense. The following table summarizes the Convertible Promissory Notes and discounts
upon issuance: On the same day the $1,250,175 in principal amount of Notes entered into in
January and March 2010 were issued, all of the Note holders converted their
Notes into 16,712,304 shares of common stock and exercised the 8,050,875
warrants. On these dates the entire conversion feature and warrant discounts on
these Notes of $1,958,300 was recognized as interest expense for the three
months ended March 31, 2010. F-57
In addition, on February 24, 2010 and February 26, 2010, two Note holders
with principal amounts of $80,000 outstanding at December 31, 2009, converted
their Notes into 1,026,160 shares of common stock. One of these note holders
also exercised 100,000 warrants issued in connection with the Notes. On these
dates the unamortized values of the conversion feature and warrant discounts on
these Notes of $13,063 was recognized as interest expense for the three months
ended March 31, 2010. The following table shows activity in the Notes account during the three
months ended March 31, 2010: NOTE 12 - RESTRUCTURING ACTIVITIES From May 29, 2008 to November 7, 2008, MMRIS had provided notices under
the federal Worker Adjustment and Retraining Notification Act to 142 employees,
including six members of senior management, that it planned to conduct a
workforce reduction at its facility in San Diego, California and that their
employment was expected to end on various dates between June 6, 2008 to
November 7, 2008. Immediately prior to the date of the Merger on January 27,
2009, the total severance liability relating to former MMRIS employees amounted
to $1,682,416. On January 27, 2009, immediately prior to the Merger, as part of
the 9,999,992 warrants issued to creditors (see Note 9), the Company issued
warrants as settlement of $985,020 of these amounts. In addition, the Company
signed promissory notes with certain former executives totaling $76,783, which
notes are payable in full on August 31, 2009 (see Note 10). As of March 31, 2010, the total remaining severance liabilities amounted to
$620,613, which is reflected as severance liability on the accompanying
consolidated balance sheets. This consists of $571,362 payable to former
non-executive employees in 18 monthly installments starting on July 27, 2009, as
well as $49,251 in estimated payroll tax. No payments were made during the three
months ended March 31, 2010 on these severance liabilities. During the period from January 27, 2009 through March 31, 2009, the Company
entered into a series of settlement agreements with certain vendors of MMRIS
pursuant to the Creditor Plan, in which the Company settled approximately
$245,853 of its outstanding accounts payable for an aggregate settlement amount
of $193,628. This resulted in a gain of $52,225, which has been recorded as a
gain on settlement of payables in the accompanying consolidated statement of
operations for the three months ended March 31, 2009. There was no gain recorded
during the three months ended March 31, 2010. NOTE 13 - RELATED PARTY TRANSACTIONS Our President, Chairman and Chief Executive Officer, Robert H. Lorsch, is
also the Chief Executive Officer of The RHL Group, Inc. and owns all of the
capital stock of The RHL Group, Inc. Mr. Lorsch directly, and indirectly through
The RHL Group, Inc., beneficially owns approximately 28.0% our total outstanding
voting stock. The RHL Group, Inc. has loaned money to MMR pursuant to a secured
note. See Note 3 - Related Party Note Payable. The RHL Group is an investment holding company which provides consulting,
operational and technical services to the Company, which we refer to as the RHL
Services. As part of the RHL Services, the RHL Group provides the Company with
unrestricted access to its internal business and relationship contact database
of more than 10,000 persons and entities, which includes clients of the RHL
Group and other individuals which may hold value to the Company. The RHL Group
also provides infrastructure support to the Company, including allowing the
Company unlimited access to its facilities, equipment, and data, information
management and server systems. In addition to allowing the Company the use of
its office support personnel, the RHL Group also has consented to allow the
Company to utilize the full-time services of Mr. Lorsch as the Company's
President, Chairman and Chief Executive Officer, which requires substantial time
and energy away from his required duties as the RHL Group's Chairman and Chief
Executive Officer. In addition, the RHL Group has made its President, Kira Reed
Lorsch, available as the Company's spokesperson, for personal appearances,
tradeshows and hosting the Company's product videos. Ms. Reed Lorsch also helps
manage the Company's social networking activities. F-58
The Company incurred $12,500 and $17,500 during the three months ended March
31, 2010 and 2009, respectively, towards marketing consulting services from
Bernard Stolar, a director. Included in related party payables at March 31, 2010
and December 31, 2009 was $116,417 and $106,368, respectively, with respect to
these services. In addition, on January 27, 2010, the Company granted the Mr.
Stolar 4,000,000 options to purchase common stock in the company at an exercise
price of $0.10 per share in consideration for director services. The options
vest in three annual installments beginning January 27, 2011. The Company also incurred $12,500 during the three months ended March 31,
2010 and 2009, towards marketing consulting services from Hector Barreto, a
director. Included in related party payables at March, 31, 2010 and December 31,
2009 was $42,500 and $23,500, respectively, with respect to these services. In
addition, on January 27, 2010, the Company granted the Mr. Barreto 4,000,000
options to purchase common stock in the company at an exercise price of $0.10
per share in consideration for director services. The options vest in three
annual installments beginning January 27, 2011. Additionally, on January 21,
2010, the Company granted to Mr. Barreto 50,000 shares of common stock, valued
at $5,500 in consideration for sales and marketing services. In the first
quarter of 2009, the Company entered into an agreement with The Latino
Coalition, a non-profit organization in which Mr. Barreto is also the Chairman,
to market the Company's product to its members. During the three months ended
March 31, 2010 and 2009, the Company paid $0 and $2,000, respectively, to The
Latino Coalition for services. The Company also incurred $31,172 and $0 during the three months ended March
31, 2010 and 2009, respectively, towards finder's fees from George Rebensdorf, a
director. Included in related party payables at March, 31, 2010 and December 31,
2009 was $56,633 and $71,333, respectively, with respect to these services. In
addition, on January 27, 2010, the Company granted the Mr. Rebensdorf 4,000,000
options to purchase common stock in the company at an exercise price of $0.10
per share in consideration for director services. The options vest in three
annual installments beginning January 27, 2011. The Company contracts with a significant vendor for the development and
maintenance of the software applications necessary to run our MyMedicalRecords
PHR, MyESafeDepositBox and MyMedicalRecords Pro products. Our outside developer
supports our software development needs through a team of software engineers,
programmers, quality control personnel and testers, who work with our internal
product development team on all aspects of application development, design,
integration and support of our products. This vendor is also a shareholder. For
the three months ended March 31, 2010 and 2009, the total expenses relating to
this shareholder amounted to $12,533 and $30,825, respectively. In addition, the
Company capitalized $20,005 of software development costs for the three months
ended March 31, 2009. As of March 31, 2010 and December 31, 2009, the total
amounts due to the shareholder and included in related party payables amounted
to $630,359 and $617,796, respectively. On January 6, 2010, the Company entered into a settlement agreement with an
MMR-Asia investor to provide for full and final settlement of any and all claims
pertaining to any license rights for MMR-Asia to market, sell and sublicense
MMRIS and MMR products and services. Refer to the Guarantee provided by The
RHL Group section in Note 7, Commitments and Contingencies for further
details. As part of this settlement agreement, the Company granted 1,388,889
shares of common stock to the investor. The Company incurred costs of $37,895 and $116,122 during the three months
ended March 31, 2010 and 2009, respectively, toward consulting services from
Audit Prep Services, LLC. Michael T. Psomas, member of Audit Prep Services, LLC,
is a shareholder of MMRIS. At March 31, 2010 and December 31, 2009 the Company
had $71,884 and $64,568, respectively, due to Audit Prep Services, LLC, which
was recorded as a related party payable. Additionally, on February 11, 2010, the
Company granted to Mr. Psomas 674,000 shares of common stock, valued at $60,660
in consideration for amounts due to Audit Prep Services, LLC at the time of
issuance. On September 15, 2009, the Company entered into a five year agreement with
E-Mail Frequency, LLC and David Loftus, Managing Partner of E-Mail Frequency,
LLC, a significant shareholder of the Company. The Company will license an
existing 80 million person direct marketing database of street addresses,
cellular phone numbers, e-mail addresses and other comprehensive data (the
"Database") with E-Mail Frequency. The agreement allows the Company to market,
through the use of the Database, its MyMedicalRecords Personal Health Record,
MyEsafeDepositBox virtual vault, and MMRPro document management system to
physicians and their patients. Under the terms of the Agreement, the Company,
upon closing of the Agreement, paid $250,000 to David Loftus as a one-time
consulting fee in the form of 2,777,778 shares of common stock. The $250,000
one-time licensee fee was recorded as a prepaid consulting fee and included in
the prepaid expenses and other current assets as of December 31, 2009, less
amortization of $12,500 included in operating expensed for the year ended
December 31, 2009 and for the three months ended March 31, 2010.
F-59
In addition, the Company incurred a total of $0 during the three months ended
March 31, 2010 and 2009, respectively, towards marketing services and consulting
services from E-Mail Frequency, LLC and David Loftus. Included in related party
payables at March 31, 2010 and December 31, 2009 was $12,533 and $50,577,
respectively, in respect to these services. Furthermore, on January 6, 2010, the
Company entered into 12% Convertible Promissory Notes with Mr. Loftus for a
principal amount totaling $400,000 and warrants to purchase the Company's common
stock. On the date of the investment, Mr. Loftus immediately converted the 12%
Convertible Promissory Notes into shares of common stock and exercised the
attached warrants and received a total 8,860,606 shares of common stock. NOTE 14 - SUBSEQUENT EVENTS On April 30, 2010, the Company filed a petition in the New York Supreme Court
against the Lymphoma Research Foundation ("LRF"), a California nonprofit
corporation, to enforce the Company's rights to a portion of certain
intellectual property and biotechnology assets acquired as a part of the reverse
merger with Favrille. LRF has claimed ownership of some of these assets
including samples and data which were transferred to LRF in the summer of 2008.
Also on April 30, 2010, LRF filed a declaratory relief action in the New York
Supreme Court asserting its rights over the aforementioned samples and data.
Both the Company and LRF have agreed to mediate the matter. On May 4, 2010, the $3,576,430 derivative liability (see Note
9) and $70,379 conversion feature liability was reclassified back into equity
and will no longer be accounted for as liability. As of March 31, 2010, two
unrelated third-partied had a principal balance of $100,000 and 250,000 warrants
to purchase common stock outstanding in connection with the 12% Convertible
Promissory Notes. On May 4, 2010, the two parties converted the $100,000 balance
and exercised the 250,000 warrants and received 886,943 shares of common stock.
As a result, the condition leading to the $3,576,430 derivative liability and
$70,379 conversion feature liability was remediated and the derivative
liabilities were reclassified back into equity. On May 12, 2010, MMR issued The RHL Group and Robert H. Lorsch, our Chairman,
Chief Executive Officer and President, a Fourth Amended and Restated Secured
Promissory Note (the "Fourth Amended Note"), and the Company agreed to guaranty
MMR's obligations under the Fourth Amended Note (the "Guaranty"). The Fourth
Amended Note amends and restates the April 29, 2009 Third Amended and Restated
Secured Promissory Note Agreement (the "RHL Note"). The Fourth Amended Note
matures April 29, 2011, and bears interest at the lesser of 10% or the highest
rate then permitted by law, and is secured (similar to the previous RHL Note) by
the Security Agreement. The reserve credit line of the Fourth Amended Noted
remained at $3,000,000. In connection with the Fourth Amended Note, The RHL
Group received a $200,000 loan origination fee ("Fee") due on May 12, 2010. In
the event the Fourth Amended Note is renewed for an additional six-month term,
The RHL Group is due an additional $200,000 loan origination fee. In addition,
The RHL Group received 2,614,684 warrants to purchase shares of the Company's
common stock at an exercise price of $0.21 per share. The warrants are fully
vested and be exercisable in cash or in a cashless exercise at any time prior to
their fourth anniversary of issuance. The Fourth Amended Note and Guaranty are
filed as Exhibits to this current report on Form 10-Q. F-60
60,000,000 SHARES COMMON STOCK PROSPECTUS Through and including , 2010 (the 25th day after the date of this prospectus), all
dealers effecting transaction in these securities, whether or not
participating in this offering, may be required to deliver a prospectus. This is in addition to the dealers' obligation to deliver a prospectus when acting
as underwriters and with respect to their unsold allotments or subscriptions.
PART II INFORMATION NOT REQUIRED IN PROSPECTUS Item 13. Other Expenses of Issuance and Distribution The following table sets forth expenses in connection with the issuance and distribution of the securities being registered. All amounts
shown are estimated, except the SEC registration fee. SEC registration fee
$ 713 Legal fees and expenses
20,000 Accountants' fees and expenses
5,000 Miscellaneous fees
5,000
Total
$ 30,713
Item 14. Indemnification of Directors and Officers Section 145 of the Delaware General Corporation Law, or DGCL, empowers a Delaware corporation to
indemnify any persons who are, or are threatened to be made, parties to any threatened, pending or completed legal action,
suit or proceeding, whether civil, criminal, administrative or investigative (other than an action by or in the right of such
corporation), by reason of the fact that such person was an officer or director of such corporation, or is or was serving at the
request of such corporation as a director, officer, employee or agent of another corporation or enterprise. The indemnity may
include expenses (including attorneys' fees), judgments, fines and amounts paid in settlement actually and reasonably
incurred by such person in connection with such action, suit or proceeding, provided that such officer or director acted in
good faith and in a manner he reasonably believed to be in or not opposed to the corporation's best interests, and, for
criminal proceedings, had no reasonable cause to believe his conduct was illegal. A Delaware corporation may indemnify
officers and directors in an action by or in the right of the corporation under the same conditions, except that no
indemnification is permitted without judicial approval if the officer or director is adjudged to be liable to the corporation in the
performance of his duty. Where an officer or director is successful on the merits or otherwise in the defense of any action
referred to above, the corporation must indemnify him against the expenses which such officer or director actually and
reasonably incurred. Our amended and restated certificate of incorporation and amended and restated bylaws provide that we will indemnify
our directors and officers to the fullest extent not prohibited by Delaware law, except that that we shall not be required to
indemnify any director or executive officer in connection with any proceeding (or part thereof) initiated by such person unless
(i) such indemnification is expressly required to be made by law, (ii) the proceeding was authorized by the Board of Directors
of the corporation, (iii) such indemnification is provided by us, in our sole discretion, pursuant to the powers vested in the
corporation under the DGCL or any other applicable law or (iv) such indemnification is required to be made under separate
contract. The bylaws also provide that the right of directors and officers to indemnification shall be a contract right and shall
not be exclusive of any other right now possessed or hereafter acquired under any statute, provision of our certificate of
incorporation, bylaws, agreements, vote of stockholders or disinterested directors or otherwise. The bylaws also permit us to
secure insurance on behalf of any officer, director, employee or other agent for any liability arising out of his or her actions in
such capacity. In accordance with Section 102(b)(7) of the DGCL, our amended and restated certificate of
incorporation provides that directors shall not be personally liable for monetary damages for breaches of their fiduciary duty
as directors to the fullest extent permitted under applicable law. The effect of this provision is to eliminate the personal
liability of directors for monetary damages for actions involving a breach of their fiduciary duty of care, including any actions
involving gross negligence. Notwithstanding this provision the DGCL does not permit us to eliminate personal liability for
(i) breaches of their duty of loyalty to us or our stockholders, (ii) acts or omissions not in good faith or
which involve intentional misconduct or knowing violations of law, (iii) certain transactions under
Section 174 of the DGCL (unlawful payment of dividends or unlawful stock purchases or redemptions) or
(iv) transactions from which a director derives an improper personal benefit. II-1
Insofar as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers or persons
controlling us pursuant to the foregoing provisions, or otherwise, we have been advised that in the opinion of the Securities
and Exchange Commission, such indemnification is against public policy as expressed in the Securities Act and is, therefore,
unenforceable. Item 15. Recent Sales of Unregistered Securities The following is a summary of transactions by us within the past three years involving sales of our
securities that were not registered under the Securities Act. Each offer and sale was exempt from registration under either
Section 4(2) of the Securities Act or Rule 506 under Regulation D of the Securities Act because (i) the securities were
offered and sold only to accredited investors; (ii) there was no general solicitation or general advertising related to the
offerings; (iii) each investor was given the opportunity to ask questions and receive answers concerning the terms of and
conditions of the offering and to obtain additional information; (iv) the investors represented that they were acquiring the
securities for their own account and for investment; and (v) the securities were issued with restrictive legends. II-2
II-3
II-4
II-5
II-6
In addition to the Note, the Purchaser received a warrant to purchase initially up to 1,200,000 shares of our common
stock at a per share price equal to the lesser of (i) the product of fifty percent (50%) multiplied by the arithmetic average of
the VWAP of the shares for the ten (10) consecutive trading days ending on the day that is three (3) trading days prior to the
applicable exercise date, or (ii) $0.15, whichever is less (the "Warrant"). The Warrant expires on September 15, 2012. II-7
In the past three years through the date of this registration statement, we granted stock options and stock purchase
rights to acquire an aggregate of 101,412,779 shares of our Common Stock We generally used the proceeds of the foregoing sales of securities for repayment of indebtedness, working capital
and other general corporate purposes. Item 16. Exhibits and Financial Statement Schedules (a) The exhibits set forth under the caption "Exhibit Index" below are included herein and incorporated by
reference. (b) Financial Statement Schedules. The financial statement schedules have been omitted because they are not
applicable, not required, or the information is included in the consolidated financial statements or notes thereto. Item 17. Undertakings The undersigned registrant hereby undertakes: 1. To file, during any period in which offers or sales are being made, a post-effective amendment to this registration
statement: i. To include any prospectus required by section 10(a)(3) of the Securities Act of 1933; ii. To reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the
most recent post-effective amendment thereof) which, individually or in the aggregate, represent a fundamental change in
the information set forth in the registration statement. Notwithstanding the foregoing, any increase or decrease in volume of
securities offered (if the total dollar value of securities offered would not exceed that which was registered) and any deviation
from the low or high end of the estimated maximum offering range may be reflected in the form of prospectus filed with the
Commission pursuant to Rule 424(b) if, in the aggregate, the changes in volume and price represent no
more than 20% change in the maximum aggregate offering price set forth in the "Calculation of Registration Fee" table in the
effective registration statement. iii. To include any material information with respect to the plan of distribution not previously disclosed in the
registration statement or any material change to such information in the registration statement; II-8
2. That, for the purpose of determining any liability under the Securities Act of 1933, each such post-effective
amendment shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of
such securities at that time shall be deemed to be the initial bona fide offering thereof. 3. To remove from registration by means of a post-effective amendment any of the securities being registered which
remain unsold at the termination of the offering. 4. That, for the purpose of determining liability under the Securities Act of 1933 to any purchaser, each prospectus
filed pursuant to Rule 424(b) as part of a registration statement relating to an offering, other than
registration statements relying on Rule 430B or other than prospectuses filed in reliance on Rule 430A,
shall be deemed to be part of and included in the registration statement as of the date it is first used after effectiveness.
Provided, however, that no statement made in a registration statement or prospectus that is part of the registration statement
or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus that
is part of the registration statement will, as to a purchaser with a time of contract of sale prior to such first use, supersede or
modify any statement that was made in the registration statement or prospectus that was part of the registration statement or
made in any such document immediately prior to such date of first use. Insofar as indemnification for liabilities arising under the Securities Act of 1933, may be permitted to directors, officers
and controlling persons of the registrant pursuant to the foregoing provisions, or otherwise, the registrant has been advised
that, in the opinion of the SEC, such indemnification is against public policy as expressed in the Securities Act and is,
therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the
registrant of expenses incurred or paid by a director, officer or controlling person of the registrant in the successful defense
of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities
being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent,
submit to a court of appropriate jurisdiction the question whether such indemnification by them is against public policy as
expressed in the Securities Act and will be governed by the final adjudication of such issue. II-9
Pursuant to the requirements of the Securities Act of 1933, the registrant has duly caused this Registration Statement on Form S-1 to
be signed on its behalf by the undersigned, thereunto duly authorized, in the City of Los Angeles, State of California,
on June 18, 2010. MMRGLOBAL, INC. By: /s/ Robert H. Lorsch Name: Robert H. Lorsch Title: Chief Executive Officer II-10
We, the undersigned officers and directors of MMRGlobal, Inc., do hereby constitute and appoint Robert H. Lorsch and
Ingrid Safranek, and each of them individually, our true and lawful attorney-in-fact and agent with full power of substitution and re-substitution, for him and
in his name, place and stead, in any and all capacities, to sign any and all amendments (including post-effective amendments) to this Registration
Statement, or any related registration statement that is to be effective upon filing pursuant to Rule 462(b) under the Securities Act of
1933, as amended, and to file the same, with exhibits thereto, and other documents in connection therewith, with the SEC, granting unto said
attorney-in-fact and agent, full power and authority to do and perform each and every act and thing requisite or necessary to be done in and about
the premises, as fully to all intents and purposes as he might or could do in person, hereby ratifying and confirming all that said attorney-in-fact and
agent, or his substitute, may lawfully do or cause to be done by virtue hereof. Pursuant to the requirements of the Securities Act of 1933, this Registration Statement on Form S-1 has been signed below by the
following persons in the capacities and on the dates indicated. Name Title Date /s/ Robert H. Lorsch Chief Executive Officer and Director June 18, 2010 Robert H. Lorsch (Principal Executive Officer) /s/ Ingrid Safranek Chief Financial Officer, Chief Accounting Officer June 18, 2010 Ingrid Safranek (Principal Financial and Accounting Officer) /s/ Hector V. Barreto, Jr. Director June 18, 2010 Hector V. Barreto, Jr. /s/ David A. Boyden Director June 18, 2010 David A. Boyden /s/ Douglas H. Helm Director June 18, 2010 Douglas H. Helm /s/ George Rebensdorf Director June 18, 2010 George Rebensdorf /s/ Bernard Stolar Director June 18, 2010 Bernard Stolar /s/ Jack Zwissig Director June 18, 2010 Jack Zwissig II-11
EXHIBIT INDEX Exhibit Exhibit Description 2.1 2.2 3.1 3.2 3.3 3.4 3.5 4.1 4.2 4.3 4.4 4.5 4.6 4.7 4.8 4.9 4.10 4.11 4.12 4.13 4.14 4.15 5.1
* Opinion of Stradling Yocca Carlson & Rauth, a Professional Corporation.
PDF file as a courtesy 10.1 10.2 10.3 10.4 10.5 10.6 10.7 10.8 10.9 10.10 10.11 10.12 10.13 10.14 10.15 10.16 10.17 10.18 10.19 10.20 10.21 10.22 10.23 10.24 10.25 10.26 10.27 10.28 10.29 10.30 10.31 10.32 10.33 10.34 10.35 10.36 10.37 10.38 16.1 21.1 23.1
* Consent of Rose, Snyder & Jacobs, Independent Registered Public Accounting Firm
PDF file as a courtesy 24.1 +
The Company has requested confidential treatment with respect to portions of this exhibit. *
Filed herewith.
MMR Information Systems, Inc.
A Corporation of Certified Public Accountants
CONSOLIDATED BALANCE SHEETS
December 31,
December 31,
2009
2008
ASSETS
Current assets:
Cash and cash equivalents
$
487,766
$
75,779
Accounts receivable
29,402
6,928
Related party receivables
-
22,057
Prepaid expenses and other current assets
612,137
82,471
Total current assets
1,129,305
187,235
Property and equipment, net
36,737
47,050
Deposits
7,665
1,885
Advances due from related party
-
100,000
Intangible assets, net
450,457
128,484
Total assets
$
1,624,164
$
464,654
LIABILITIES AND STOCKHOLDERS' DEFICIT
Current liabilities:
Line of credit, related party
$
940,620
$
822,520
Advances from affiliate
-
701,322
Related party payables
1,537,605
513,688
Compensation payable
10,943
58,188
Severance liability
620,613
-
Accounts payable and accrued expenses
2,979,222
944,438
Deferred revenue
-
189,824
Warrant liability
88,998
-
Derivative liability
1,534,824
-
Conversion feature liability
91,828
-
Convertible notes payable
166,937
-
Notes payable, current portion
325,343
-
Capital leases payable, current portion
10,302
6,929
Total current liabilities
8,307,235
3,236,909
Notes payable, less current portion
125,000
-
Capital leases payable, less current portion
8,484
16,151
Total liabilities
8,440,719
3,253,060
Commitments and contingencies (See Note 10)
Stockholders' deficit:
Preferred stock - $0.001 per value, 5,000,000 shares authorized
-
-
Common stock, $0.001 par value, 650,000,000 shares authorized, 173,689,343 and
80,629,266 shares issued and outstanding as of December 31, 2009 and 2008, respectively
173,689
80,629
Treasury stock, at cost, 0 and 1,145,324, shares as of December 31, 2009 and 2008, respectively
-
(16,860)
Additional paid-in capital
14,458,052
8,300,460
Accumulated deficit
(21,448,296)
(11,152,635)
Total stockholders' deficit
(6,816,555)
(2,788,406)
Total liabilities and stockholders' deficit
$
1,624,164
$
464,654
CONSOLIDATED STATEMENTS OF OPERATIONS
Years Ended
December 31,
2009
2008
Revenues
Subscriber
$
397,798
$
280,572
License fees and other
221,451
170,783
Total revenues
619,249
451,355
Cost of revenues
456,456
409,900
Gross profit
162,793
41,455
General and administrative expenses
5,440,545
2,108,063
Sales and marketing expenses
1,402,870
914,162
Technology development
269,270
143,031
Loss from operations
(6,949,892)
(3,123,801)
Gain on settlement of payables
89,170
-
Change in valuation of derivative liabilities
633,626
-
Interest and other finance charges, net
(4,068,565)
(398,675)
Net loss
$
(10,295,661)
$
(3,522,476)
Deemed dividend to preferred shareholders
-
(35,620)
Net loss available to common shareholders
$
(10,295,661)
$
(3,558,096)
Net loss available to common shareholders per share:
Basic and Diluted
$
(0.08)
$
(0.06)
Weighted average common shares outstanding:
Basic and Diluted
132,311,386
60,911,929
CONSOLIDATED STATEMENTS OF STOCKHOLDERS' DEFICIT
FOR THE YEAR ENDED DECEMBER 31, 2008 AND 2009
Preferred Stock
Common Stock
Treasury Stock
Additional
Accumulated
Shares
Amount
Shares
Amount
Shares
Amount
Paid-In Capital
Deficit
Total
Balance at December 31, 2007, as restated in terms of Favrille, Inc.
-
$
-
60,155,691
$
60,155
(14,482,453)
$
-
$
7,183,683
$
(7,594,539)
$
(350,701)
Issuance of Common Stock for cash, net of expenses of $2,548
1,538,702
1,539
619,508
621,047
Issuance of Common Stock as an incentive
1,538,702
1,539
34,081
(35,620)
-
Common shares issued for services
15,469,969
15,470
382,469
397,939
Issuance of shares, including Treasury Stock, to related party
as consideration for renewal of line of credit
1,926,202
1,926
14,482,453
-
48,074
50,000
Issuance of options replacing shares previously issued
(1,145,324)
(16,860)
16,860
-
Stock based compensation
15,785
15,785
Net loss
(3,522,476)
(3,522,476)
Balance at December 31, 2008, as restated in terms of Favrille, Inc.
-
-
80,629,266
80,629
(1,145,324)
(16,860)
8,300,460
(11,152,635)
(2,788,406)
Reverse merger with Favrille, Inc.
41,254,550
41,255
441,346
482,601
Shares issued to RHL Group for extension of guarantee
328,174
328
4,672
5,000
Stock options exercised by RHL Group as a reduction of line of credit
2,461,298
2,461
110,759
113,220
Shares and warrants issued to Robert Lorsch and RHL Group
2,941,321
2,941
1,511,283
1,514,224
Common stock sold
84,326
84
3,795
3,879
Reclassification of warrants and options to derivative liabilities
(2,222,698)
(2,222,698)
Shares issued to replace options previously issued
57,263
57
1,145,324
16,860
(15,451)
1,466
Shares issued for services, liability deferral and reduction of liabilities
13,049,774
13,050
1,314,950
1,328,000
Convertible debt issuance, conversion and warrant exercises
26,219,265
26,219
3,359,756
3,385,975
Stock option exercises
6,289,106
6,290
306,794
313,084
Warrant exercises
375,000
375
33,000
33,375
Stock based compensation
887,577
887,577
Warrants issued for services
421,809
421,809
Net loss
(10,295,661)
(10,295,661)
Balance at December 31, 2009
-
$
-
173,689,343
$
173,689
-
$
-
$
14,458,052
$
(21,448,296)
$
(6,816,555)
CONSOLIDATED STATEMENTS
OF CASH FLOWS
Year Ended
December 31,
2009
2008
Operating activities:
Net loss
$
(10,295,661)
$
(3,522,476)
Adjustments to reconcile net loss to net cash used in operating activities:
Depreciation and amortization
78,163
148,777
Impairment of intangible assets
-
197,000
Common stock issued as consideration for extension of guarantee
5,000
-
Common stock issued as replacement of options previously issued
1,466
-
Change in valuation of derivative liabilities
(633,626)
-
Warrants issued for services
1,491,781
-
Restricted common stock issued for services
440,279
397,939
Share based compensation
887,577
15,785
Interest expense on convertible debt
1,951,522
-
Gain on settlement of payables
(89,170)
50,000
Loss from disposal of property and equipment
2,237
2,996
Loss on stock settlement of liabilities
220,792
-
Loss on write-off of advances due from related party
100,000
-
Loan commitment fee amortization
200,000
-
Effect of changes in:
Accounts receivable
(22,474)
17,428
Related party receivables
22,057
(21,057)
Prepaid expenses and other current assets
180,105
57,042
Deposits
(5,780)
51,452
Related party payables
1,304,916
258,047
Compensation payable
13,139
20,375
Accounts payable and accrued expenses
1,920,521
483,537
Deferred revenue
(189,824)
59,638
Deferred rent
-
(6,344)
Net cash used in operating activities
(2,416,980)
(1,789,861)
Investing activities:
Purchase of property and equipment
(9,441)
(3,566)
Cash acquired from reverse merger with Favrille, Inc.
1,050,506
-
Patents
(107,087)
-
Investment in MMRPro
(275,533)
-
Proceeds from sales of property and equipment
-
2,226
Net cash provided by (used in) investing activities
658,445
(1,340)
Financing activities:
Payments on capital lease
(6,929)
(6,261)
Proceeds from line of credit, related party
211,000
539,422
Proceeds from issuance of note payable
125,000
-
Proceeds from issuance of convertible notes payable
1,373,000
-
Proceeds from advances from affiliate
-
701,322
Proceeds from exercise of stock options
76,316
-
Proceeds from warrant exercises
388,256
-
Issuance of common stock, net of issuance costs
3,879
621,047
Net cash provided by financing activities
2,170,522
1,855,530
Net increase in cash
411,987
64,329
Cash, beginning of period
75,779
11,450
Cash, end of period
$
487,766
$
75,779
Supplemental disclosures of cash flow information:
Cash paid for interest
$
901
$
18,616
Cash paid for income taxes
$
1,989
$
800
Supplemental disclosure of non-cash investing and financing activities:
Payment of accounts payable through issuance of notes payable
$
139,355
$
-
Capitalized loan commitment fees payable
$
230,000
$
-
Payment of accounts payable and loan origination fee through issuance of warrants
$
61,880
$
-
Conversion of convertible notes payable into common stock
$
1,193,000
$
-
Establishment of derivative liabilities
$
2,523,159
$
-
Payment of related party line of credit and accounts payable through issuance of common stock
$
1,115,983
$
50,000
Prepayment for services through issuance of common stock
$
499,130
$
-
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
Fair Value
Fair Value Measurements at December 31, 2009
as of
Using Fair Value Heirarchy
Financial Instruments
December 31, 2009
Level 1
Level 2
Level 3
Liabilities:
Warrant liability
$
88,998
$
-
$
-
$
88,998
Derivative liability
1,534,824
-
-
1,534,824
Conversion feature liability
91,828
-
-
91,828
Total
$
1,715,650
$
-
$
-
$
1,715,650
Years Ended December 31,
2009
2008
Expected life in years
4.58 - 5.00
0.38
Stock price volatility
104.43 - 104.72%
65.93%
Risk free interest rate
2.16% - 2.73%
0.49%
Expected dividends
None
None
Forfeiture rate
0%
0%
December 31,
December 31,
2009
2008
Prepaid consulting fees from issuance of common stock
$
380,395
$
-
Prepaid insurance
183,710
41,183
Deferred financing costs
-
29,167
Loan origination fee settled through issuance of warrants
21,250
-
Prepaid trade shows
18,451
8,750
Prepaid rent from issuance of common stock
6,900
-
Prepaid other
1,431
3,371
Total prepaid expenses and other current assets
$
612,137
$
82,471
December 31,
December 31,
2009
2008
Furnitures and fixtures
$
3,170
$
3,170
Computers and related equipment
85,395
81,708
88,565
84,878
Less: accumulated depreciation and amortization
(51,828)
(37,828)
$
36,737
$
47,050
Years Ended
December 31,
December 31,
2009
2008
Website development
$
644,212
$
644,212
MMR Pro website development
275,533
-
Patents
107,087
-
Domain name
10,000
10,000
1,036,832
654,212
Less: accumulated amortization
(389,375)
(328,728)
Reserve for impairment
(197,000)
(197,000)
$
450,457
$
128,484
Year Ending
December 31,
2010
$
76,157
2011
84,841
2012
62,974
2013
55,107
2014
55,107
Thereafter
116,271
Total
$
450,457
December 31,
December 31,
2009
2008
Accounts payable and accruals
$
2,414,438
$
916,596
Accounts payable and accruals from Favrille Merger
532,505
-
Accrued vacation
18,502
27,842
Interest payable
13,777
-
Total accounts payable and accrued expenses
$
2,979,222
$
944,438
Years Ended December 31,
2009
2008
Federal statutory rate
-34.00%
-34.00%
State tax, net of federal benefit
-5.20%
-5.71%
Non-deductible items
3.68%
0.62%
Valuation allowance
35.52%
39.12%
Effective income tax rate
0.00%
0.03%
Years Ended December 31,
2009
2008
Net operating loss carryforwards
$
7,693,049
$
4,565,404
Depreciation and amortization
(27,058)
(1,924)
Share based compensation
870,409
39,793
State tax and other
(595,704)
(320,033)
Deferred tax assets, net
7,940,696
4,283,240
Less: valuation allowance
(7,940,696)
(4,283,240)
$
-
$
-
Year Ended
Operating
Capital
December 31,
Leases
Leases
2010
$
-
$
11,594
2011
-
8,959
Total minimum lease payments
$
-
20,553
Less interest portion
(1,767)
$
18,786
Weighted-
Average
Weighted-
Remaining
Average
Contractual
Aggregate
Exercise
Life
Intrinsic
Options
Price
(Years)
Value
Outstanding at December 31, 2007
11,453,269
$
0.05
3.88
$
-
Granted
1,333,863
0.01
Exercised
(656,348)
0.05
Outstanding at December 31, 2008
12,130,784
0.01
1.76
-
Options acquired through Merger
1,731,444
3.44
Granted
16,170,000
0.13
Exercised
(8,750,478)
0.05
Forfeited
(579,935)
3.55
Cancelled
(3,240,166)
1.21
Outstanding at December 31, 2009
17,461,649
0.12
4.00
54,582
Vested and expected to vest
at December 31, 2009
17,461,649
0.12
4.00
54,582
Exercisable at December 31, 2009
6,173,402
0.11
3.46
54,582
Weighted
Average
Grant Date
Shares
Fair Value
Nonvested at December 31, 2007
2,655,398
$
0.01
Granted
1,333,863
$
0.003
Vested
(2,732,311)
$
0.01
Forfeited
(328,173)
$
0.01
Nonvested at December 31, 2008
928,777
$
0.01
Options acquired through Merger
279,039
$
2.44
Granted
16,170,000
$
0.10
Forfeited
(5,888,482)
$
0.16
Vested
(201,087)
$
2.28
Nonvested at December 31, 2009
11,288,247
$
0.09
Options Outstanding
Options Exercisable
Weighted
Weighted
Weighted
Average
Average
Average
Exercise
Number
Remaining
Exercise
Number
Exercise
Price
of Shares
Life (Years)
Price
of Shares
Price
$
0.05
1,591,649
1.11
$
0.05
1,591,649
$
0.05
0.13
14,300,000
4.28
0.13
4,100,000
0.13
0.18
1,570,000
4.39
0.18
481,753
0.18
17,461,649
6,173,402
Warrants Outstanding
Warrants Exercisable
Weighted
Weighted
Weighted
Average
Average
Average
Exercise
Number
Remaining
Exercise
Number
Exercise
Price
of Shares
Life (Years)
Price
of Shares
Price
$
0.05 - 0.61
26,782,147
4.27
$
0.14
26,486,257
$
0.14
1.80 - 2.77
4,459,910
2.86
2.77
4,459,910
2.77
3.98 - 6.33
3,371,787
1.26
5.14
3,371,787
5.14
34,613,844
34,317,954
Value at December 31, 2007
$
-
Establishment of derivative liability, net
-
Change in Value
-
Reclassification of value of warrants and options exercised
-
Value at December 31, 2008
-
Establishment of derivative liability, net
2,523,159
Change in Value
(633,626)
Reclassification of value of warrants and options exercised
(138,883)
Value at December 31, 2009
$
1,750,650
January 27, 2009
March 31, 2009
June 13, 2009
July 16, 2009
September 30, 2009
December 31, 2009
Expected life in years
0.25 - 5.0 years
0.07 - 5.59 years
0.13 - 5.4 years
0.13 - 4.83 years
0.00 - 4.83 years
0.02 - 4.63 years
Stock price volatility
102.42% - 183.93%
103.45% - 178.61%
104.54% - 152.74%
104.67% - 152.29%
94.68% - 147.10%
111.89% - 187.44%
Risk free Interest rate
0.13% - 1.59%
0.17% - 1.67%
0.19% - 2.81%
0.19% - 2.46%
0.06% - 2.31%
0.04% - 2.69%
Expected dividends
None
None
None
None
None
None
Forfeiture rate
0%
0%
0%
0%
0%
0%
December 31,
December 31,
2009
2008
Promissory notes payable due to the former officers of MMRIS as part of
severance packages, due in full on August 31, 2009 with no stated interest
$
76,783
$
-
Promissory notes payable due to the two remaining officers of MMRIS
pursuant to the Resignation and Post-Merger Employment
Arrangement, due in full on August 31, 2009 with no stated interest
25,444
-
Promissory notes payable due to vendors relating to settlement of
certain outstanding accounts payable, payable in 18 equal monthly
installments commencing on July 27, 2009 and ending on January 27, 2011,
with no stated interest
223,116
-
Promissory notes payable due to PM Creative Corporation, due in full
on July 30, 2011, with a 12% per annum stated interest.
125,000
-
450,343
-
Less: current portion
325,343
-
Notes payable, less current portion
$
125,000
$
-
Year Ended
December 31,
2010
$
325,343
2011
125,000
Total
$
450,343
Convertible Promissory Notes at face value
$
1,373,000
Conversion feature derivative discount
(1,125,574)
Warrant derivative discount
(839,012)
Deferred financing costs
778,520
Initial value of Notes
$
186,934
Initial value of Notes
$
186,934
Conversion to common stock
(1,193,000)
Accretion of converted Notes
1,029,023
Accretion of un-converted Notes
143,980
Value of Notes at December 31, 2009
$
166,937
CONSOLIDATED
BALANCE SHEETS
March 31,
December 31,
2010
2009
(Unaudited)
ASSETS
Current assets:
Cash and cash
equivalents
$
878,442
$
487,766
Accounts
receivable
26,227
29,402
Inventory
29,022
-
Prepaid expenses and other
current assets
524,565
612,137
Total current assets
1,458,256
1,129,305
Property and equipment, net
32,225
36,737
Deposits
7,665
7,665
Intangible assets, net
454,603
450,457
Total assets
$
1,952,749
$
1,624,164
LIABILITIES AND STOCKHOLDERS'
DEFICIT
Current liabilities:
Line of credit, related
party
$
976,924
$
940,620
Related party
payables
1,381,760
1,537,605
Compensation
payable
11,246
10,943
Severance
liability
620,613
620,613
Accounts payable and
accrued expenses
3,036,574
2,979,222
Warrant
liability
266,994
88,998
Derivative
liability
3,576,430
1,534,824
Conversion feature
liability
70,379
91,828
Convertible notes
payable
100,000
166,937
Notes payable, current
portion
325,351
325,343
Capital leases payable,
current portion
10,499
10,302
Total current liabilities
10,376,770
8,307,235
Notes
payable, less current portion
-
125,000
Capital leases payable, less current
portion
6,443
8,484
Total liabilities
10,383,213
8,440,719
Commitments and contingencies (See Note
7)
Stockholders' deficit:
Preferred stock - $0.001
per value, 5,000,000 shares authorized
-
-
Common stock, $0.001 par
value, 650,000,000 shares authorized, 204,409,509 and
173,689,343 shares issued and outstanding as
of March 31, 2010 (unaudited) and
December 31,
2009, respectively
204,410
173,689
Additional paid-in
capital
19,276,780
14,458,052
Accumulated
deficit
(27,911,654)
(21,448,296)
Total stockholders' deficit
(8,430,464)
(6,816,555)
Total liabilities and stockholders' deficit
$
1,952,749
$
1,624,164
CONSOLIDATED
STATEMENTS OF OPERATIONS
Three Months Ended
March 31,
2010
2009
(Unaudited)
(Unaudited)
Revenues
Subscriber
$
90,164
$
92,495
License fees and other
-
53,292
Total revenues
90,164
145,787
Cost
of revenues
113,036
106,852
Gross profit
(loss)
(22,872)
38,935
General and administrative expenses
1,219,315
1,294,405
Sales
and marketing expenses
930,132
235,886
Technology development
82,139
43,748
Loss from
operations
(2,254,458)
(1,535,104)
Gain
on settlement of payables
-
52,225
Change
in valuation of derivative liabilities
(2,168,480)
(2,150,831)
Interest and other finance charges, net
(2,040,420)
(242,680)
Net loss
$
(6,463,358)
$
(3,876,390)
Net
loss available to common shareholders per share:
Basic
and Diluted
$
(0.03)
$
(0.04)
Weighted average common shares
outstanding:
Basic
and Diluted
188,263,878
109,748,626
Preferred Stock
Common Stock
Additional
Accumulated
Shares
Amount
Shares
Amount
Paid-In Capital
Deficit
Total
Balance as of December 31, 2009
-
$
-
173,689,343
$
173,689
$
14,458,052
$
(21,448,296)
$
(6,816,555)
Convertible debt issuances and warrants
exercises
-
-
25,889,340
25,889
3,684,032
-
3,709,921
Reclassification of warrants and options to
derivative liabilities
-
-
-
-
(69,957)
-
(69,957)
Shares issued for services or reduction to
liabilities
-
-
4,584,696
4,585
452,469
-
457,054
Stock option exercises
-
-
246,130
247
11,081
-
11,328
Stock based compensation
-
-
-
-
655,940
-
655,940
Warrants issued for services
-
-
-
-
85,163
-
85,163
Net loss
-
-
-
-
-
(6,463,358)
(6,463,358)
Balance as of March 31, 2010
(unaudited)
-
$
-
$
204,409,509
$
204,410
$
19,276,780
$
(27,911,654)
$
(8,430,464)
CONSOLIDATED
STATEMENTS OF CASH FLOWS
Three Months Ended
March 31,
2010
2009
(Unaudited)
(Unaudited)
Operating activities:
Net
loss
$
(6,463,358)
$
(3,876,390)
Adjustments to reconcile net loss to net cash used
in operating activities:
Depreciation and
amortization
20,371
27,199
Change in allowance for
doubtful accounts
-
16,667
Common stock issued as
consideration for extension of guarantee
-
5,000
Common stock issued as
replacement of options previously issued
-
1,466
Change in valuation of
derivative liabilities
2,168,480
2,150,831
Share-based
compensation
656,539
112,709
Warrants issued for
services
85,163
205,604
Restricted common stock
issued for services
352,437
-
Interest expense on
convertible debt
1,971,363
-
Loan commitment fee
amortization
21,250
-
Gain on settlement of
payables
-
(52,225)
Effect
of changes in:
Restricted cash
-
(165,000)
Accounts
receivable
3,175
(22,427)
Inventory
(29,022)
-
Related party
receivables
-
7,753
Prepaid expenses and other
current assets
108,322
(90,021)
Deposits
-
-
Related party
payables
(119,541)
2,306
Compensation
payable
303
1,392
Accounts payable and
accrued expenses
136,769
615,266
Deferred revenue
-
(45,491)
Net cash used in operating activities
(1,087,749)
(1,105,361)
Investing activities:
Purchase of property and
equipment
-
(6,924)
Website
development
(20,005)
(6,370)
Cash acquired from reverse
merger with Favrille, Inc.
-
1,050,506
Net cash provided by (used in) investing activities
(20,005)
1,037,212
Financing activities:
Payments on capital
lease
(1,844)
(1,667)
Proceeds from line of
credit, related party
-
19,533
Proceeds from issuance of
convertible notes payable
1,142,300
-
Proceeds from exercise of
stock options
3,779
-
Proceeds from warrant
exercises
354,195
-
Net cash provided by financing activities
1,498,430
17,866
Net
increase (decrease) in cash
390,676
(50,283)
Cash,
beginning of period
487,766
75,779
Cash,
end of period
$
878,442
$
25,496
Supplemental disclosures of cash flow
information:
Cash paid for
interest
$
389
$
901
Cash paid for income
taxes
$
1,600
$
-
Supplemental disclosure of non-cash investing
and financing activities:
Payment of accounts
payable through issuance of notes payable
$
-
$
245,853
Exercise of warrants as
reduction of accounts payable and notes payable
$
26,969
$
-
Payment of accounts
payable and loan origination fee through issuance of warrants
$
61,880
$
-
Conversion of convertible
notes payable into common stock
$
1,330,175
$
-
Establishment of
derivative liabilities, net
$
60,096
$
-
Payment of prepaid
expenses through issuance of common stock
$
42,000
$
-
Prepayment for services
through issuance of common stock
$
70,165
$
-
NOTES TO
CONSOLIDATED FINANCIAL STATEMENTS (UNAUDITED)
March 31, 2010
Fair Value Measurements at March 31,
2010
Fair Value
as of
Using Fair Value Heirarchy
Financial Instruments
March 31, 2010
Level 1
Level 2
Level 3
Liabilities:
Warrant
liability
$
266,994
$
-
$
$
266,994
Derivative
liability
3,576,430
-
3,576,430
Conversion feature
liability
70,379
-
70,379
Total
$
3,913,803
$
-
$
-
$
3,913,803
Three Months Ended
March 31,
March 31,
2010
2009
(Unaudited)
(Unaudited)
Expected life in years
2-10 years
5 years
Stock price volatility
111.85% - 163.60%
102.18% - 103.28%
Risk free interest rate
1.02% - 3.66%
1.86% - 1.90%
Expected dividends
None
None
Forfeiture rate
0%
0%
March 31,
December 31,
2010
2009
(Unaudited)
Prepaid consulting fees from issuance of common
stock
$
337,000
$
380,395
Prepaid insurance
167,659
183,710
Loan
origination fee settled through issuance of warrants
-
21,250
Prepaid trade shows
17,375
18,451
Prepaid rent from issuance of common
stock
2,300
6,900
Prepaid other
231
1,431
Total
prepaid expenses and other current assets
$
524,565
$
612,137
March 31,
December 31,
2010
2009
Legal
and accounting fees
$
2,114,680
$
1,991,526
Accounts payable and accruals from Favrille
Merger
499,295
532,505
Trade
payables
243,247
286,758
Consulting services
156,282
136,154
Accrued vacation
20,119
18,502
Interest payable
2,951
13,777
Total accounts payable and accrued
expenses
$
3,036,574
$
2,979,222
Year
Ending
Operating
Capital
December
31,
Leases
Leases
2010 (remainder of)
$
-
$
9,354
2011
-
8,959
Total
minimum lease payments
$
-
18,313
Less interest portion
(1,371)
$
16,942
Weighted-
Average
Weighted-
Remaining
Aggregate
Average
Contractual
Intrinsic
Exercise
Life
Value
Options
Price
(Years)
Outstanding at
December 31, 2009
17,461,649
$
0.12
4.00
$
54,582
Granted
22,027,891
0.10
Exercised
(246,130)
0.05
Cancelled
(164,087)
0.05
Outstanding at March
31, 2010 (unaudited)
39,079,323
0.11
6.87
1,903,255
Vested and expected to
vest
at March 31, 2010
(unaudited)
39,079,323
0.11
6.87
1,903,255
Exercisable at March
31, 2010 (unaudited)
13,074,817
0.12
3.72
533,119
Options
Outstanding
Options
Exercisable
Weighted
Weighted
Weighted
Average
Average
Average
Exercise
Number
Remaining
Exercise
Number
Exercise
Price
of Shares
Life (Years)
Price
of Shares
Price
$
0.05
1,181,432
1.20
$
0.05
1,181,432
$
0.05
0.09 -
0.13
36,327,891
7.17
0.11
11,218,070
0.12
0.18
1,570,000
4.14
0.18
675,315
0.18
39,079,323
13,074,817
Warrants Outstanding
Warrants Exercisable
Weighted
Weighted
Weighted
Average
Average
Average
Exercise
Number
Remaining
Exercise
Number
Exercise
Price
of Shares
Life (Years)
Price
of Shares
Price
$
0.05 -
0.61
28,332,147
4.01
$
0.14
27,597,900
$
0.14
1.80 - 2.77
4,459,910
2.61
2.77
4,459,910
2.77
3.98 - 6.33
3,341,956
1.26
5.13
3,341,956
5.13
36,134,013
35,399,766
Purpose
Shares
Value
Services provided
1,128,942
(1)
$
116,000
Settlement with MMR Asia
1,388,889
138,889
Capital contribution fund
1,000,000
90,000
Reduction of payables
766,865
70,165
Prepaid trade conferences
300,000
42,000
Totals
4,584,696
$
457,054
Value at December 31, 2009
$
1,715,650
Change
in value of derivative liability
2,168,480
Establishment of derivative liability for
non-employee options and warrants granted in 2010
89,770
Reclassification back to equity for non-employee
options and warrants exercised or cancelled in 2010
(60,097)
Value at March 31, 2010
(unaudited)
$
3,913,803
March 31, 2010
December 31, 2009
Expected life in years
0.02 - 4.92 yrs
0.02 - 4.63 yrs
Stock price volatility
115.13% - 163.56%
111.89% - 187.44%
Risk free interest rate
0.15% - 2.55%
0.04% - 2.69%
Expected dividends
None
None
Forfeiture rate
0%
0%
March 31,
December 31,
2010
2009
(Unaudited)
Promissory notes payable due to the former officers
of MMRIS as part of severance
packages, due in full on August 31, 2009
with no stated interest
$
76,783
$
76,783
Promissory notes payable due to the two remaining
officers of MMRIS pursuant to the
Resignation and Post-Merger
Employment Arrangement, due in full on August 31, 2009
with no stated
interest
25,444
25,444
Promissory notes payable due to vendors relating to
settlement of certain
outstanding accounts payable, payable in 18 equal
monthly installments
commencing on July 27, 2009 and ending on January
27, 2011, with no stated interest
223,124
223,116
Promissory notes payable due to PM Creative
Corporation, due in full on July 30, 2011,
with a 12% per annum stated
interest.
-
125,000
325,351
450,343
Less:
current portion
325,351
325,343
Notes
payable, less current portion
$
-
$
125,000
Convertible Promissory Notes at face
value
$
1,250,175
Conversion feature derivative discount
(845,482)
Warrant derivative discount
(1,112,818)
Deferred financing costs
710,093
Initial value of Notes
$
1,968
Value
of Notes at December 31, 2009
$
166,937
Conversion to common stock
(1,330,175)
Accretion of converted Notes
1,263,238
Accretion of un-converted Notes
-
Value
of Notes at March 31, 2010
$
100,000

![]()
![]()
Number
