Attached files
Table of Contents
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
| x | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2009
or
| ¨ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission File Number: 000-50679
CORCEPT THERAPEUTICS INCORPORATED
(Exact Name of Corporation as Specified in Its Charter)
| Delaware | 77-0487658 | |
| (State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) |
149 Commonwealth Drive
Menlo Park, CA 94025
(Address of principal executive offices, including zip code)
(650) 327-3270
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12 (b) of the Act:
| Title of Each Class: |
Name of Each Exchange on which Registered: | |
| Common Stock, $0.001 par value | The NASDAQ Capital Market |
Securities registered pursuant to Section 12 (g) of the Act:
None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ¨ No x
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15 (d) of the Act. Yes ¨ No x
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes x No ¨
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes ¨ No ¨
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is not contained herein, and will not be contained, to the best of the Registrant’s knowledge, in definitive proxy or information statements incorporated by reference to Part III of this Form 10-K or any amendment to this Form 10-K. x
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer or a smaller reporting company. See the definitions of “large accelerated filer”, “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act.
| Large Accelerated Filer ¨ |
Accelerated Filer ¨ | |
| Non-accelerated filer ¨ (Do not check if a smaller reporting company) |
Smaller reporting company x |
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ¨ No x
The aggregate market value of voting and non-voting common equity held by non-affiliates of the Registrant was approximately $14,000,000 as of June 30, 2009 based upon the closing price on the Nasdaq Capital Market reported for such date. This calculation does not reflect a determination that certain persons are affiliates of the Registrant for any other purpose.
On March 15, 2010 there were 62,703,717 shares of common stock outstanding at a par value $.001 per share.
DOCUMENTS INCORPORATED BY REFERENCE
None.
Table of Contents
Form 10-K
For the year ended December 31, 2009
| Page | ||||
| PART I | ||||
| ITEM 1. | Business | 1 | ||
| ITEM 1A. | Risk Factors | 19 | ||
| ITEM 1B. | Unresolved Staff Comments | 40 | ||
| ITEM 2. | Properties | 40 | ||
| ITEM 3. | Legal Proceedings | 40 | ||
| ITEM 4. | (Removed and Reserved) | 40 | ||
| PART II | ||||
| ITEM 5. | 41 | |||
| ITEM 6. | 43 | |||
| ITEM 7. | Management’s Discussion and Analysis of Financial Condition and Results of Operations |
44 | ||
| ITEM 7A. | 56 | |||
| ITEM 8. | 56 | |||
| ITEM 9. | Changes in and Disagreements with Accountants on Accounting and Financial Disclosure |
56 | ||
| ITEM 9A (T). | 56 | |||
| ITEM 9B. | 57 | |||
| PART III | ||||
| ITEM 10. | 58 | |||
| ITEM 11. | 62 | |||
| ITEM 12. | Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters |
72 | ||
| ITEM 13. | Certain Relationships and Related Transactions, and Director Independence |
75 | ||
| ITEM 14. | 76 | |||
| PART IV | ||||
| ITEM 15. | Exhibits, Financial Statement Schedules | 77 | ||
| Signatures and Power of Attorney | 81 | |||
Table of Contents
This Annual Report on Form 10-K, or Form 10-K, contains forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended (Exchange Act), and Section 27A of the Securities Act of 1933, as amended (Securities Act). All statements contained in this Form 10-K, other than statements of historical fact, are forward-looking statements. When used in this report or elsewhere by management from time to time, the words “believe,” “anticipate,” “intend,” “plan,” “estimate,” “expect,” “may,” “will,” “should,” “seeks” and similar expressions are forward-looking statements. Such forward-looking statements are based on current expectations, but the absence of these words does not necessarily mean that a statement is not forward-looking. Forward-looking statements made in this Form 10-K include, but are not limited to, statements about:
| • | the progress of our research, development, clinical programs and the timing of regulatory activities; |
| • | our estimates of the dates by which we expect to report results of our clinical trials and the anticipated results of these trials; |
| • | the timing of market introduction of CORLUX® and future product candidates, including CORT 108297; |
| • | our ability to market, commercialize and achieve market acceptance for CORLUX or other future product candidates, including CORT 108297; |
| • | uncertainties associated with obtaining and enforcing patents; |
| • | our estimates for future performance; and |
| • | our estimates regarding our capital requirements and our needs for, and ability to obtain, additional financing. |
Forward-looking statements are not guarantees of future performance and involve risks and uncertainties. Actual events or results may differ materially from those discussed in the forward-looking statements as a result of various factors. For a more detailed discussion of such forward-looking statements and the potential risks and uncertainties that may impact upon their accuracy, see the “Risk Factors” section of this Form 10-K and the “Overview” and “Liquidity and Capital Resources” sections of the “Management’s Discussion and Analysis of Financial Condition and Results of Operations” section of this Form 10-K. These forward-looking statements reflect our view only as of the date of this report. Except as required by law, we undertake no obligations to update any forward-looking statements. Accordingly, you should also carefully consider the factors set forth in other reports or documents that we file from time to time with the Securities and Exchange Commission (SEC).
| ITEM 1. | BUSINESS |
Overview
We are a pharmaceutical company engaged in the discovery and development of drugs for the treatment of severe metabolic and psychiatric disorders. Our focus is on those disorders that are associated with a steroid hormone called cortisol. Elevated levels and abnormal release patterns of cortisol have been implicated in a broad range of human disorders. Since our inception in May 1998, we have been developing our lead product, CORLUX, a potent glucocorticoid receptor II (GR-II) antagonist that blocks the activity of cortisol. We have also discovered three series of novel selective GR-II antagonists and have moved one of these compounds, CORT 108297, into development.
Cushing’s Syndrome. Cushing’s Syndrome is a disorder caused by prolonged exposure of the body’s tissues to high levels of the hormone cortisol. Sometimes called “hypercortisolism,” it is relatively uncommon and most often affects adults aged 20 to 50. An estimated 10 to 15 of every one million people are newly diagnosed with this syndrome each year, resulting in approximately 3,000 new patients and an estimated prevalence of 20,000 patients with Cushing’s Syndrome in the United States.
1
Table of Contents
The Investigational New Drug application (IND) for the evaluation of CORLUX for the treatment of Cushing’s Syndrome was opened in September 2007. The United States Food and Drug Administration (FDA) has indicated that our single 50-patient open-label study may provide a reasonable basis for the submission of a New Drug Application (NDA) for this indication. We expect to complete enrollment in this Phase 3 study in April, as the requisite 50 patients have now been dosed or identified. We expect to announce results of this study in the fourth quarter of this year and to submit our NDA for the use of CORLUX in Cushing’s Syndrome by year-end 2010.
In July 2007, we received Orphan Drug Designation from the FDA for CORLUX for the treatment of endogenous Cushing’s Syndrome. Orphan Drug Designation is a special status granted by the FDA to encourage the development of treatments for diseases or conditions that affect fewer than 200,000 patients in the United States. Drugs that receive Orphan Drug Designation obtain seven years of marketing exclusivity from the date of drug approval, as well as tax credits for clinical trial costs, marketing application filing fee waivers and assistance from the FDA in the drug development process.
Psychotic depression. We are developing CORLUX for the treatment of the psychotic features of psychotic major depression under an exclusive patent license from Stanford University. Psychotic major depression will hereafter be referred to as psychotic depression. The FDA has granted “fast track” status to evaluate the safety and efficacy of CORLUX for the treatment of the psychotic features of psychotic depression.
In March of 2008, we began enrollment in Study 14, our ongoing Phase 3 trial in psychotic depression. The protocol for this trial incorporates what we have learned from our three previously completed Phase 3 trials. It attempts to address the established relationship between increased drug plasma levels and clinical response and to decrease the random variability observed in the results of the psychometric instruments used to measure efficacy. In one of the previously completed Phase 3 trials, Study 06, we prospectively tested and confirmed that patients whose plasma levels rose above a predetermined threshold statistically separated from both those patients whose plasma levels were below the threshold and those patients who received placebo; this threshold was established from data produced in earlier studies.
As expected, patients who took 1200 mg of CORLUX in Study 06 developed higher drug plasma levels than patients who received lower doses. Further, there was no discernable difference in the incidence of adverse events between patients who received placebo in Study 06 and those who received 300 mg, 600 mg or 1200 mg of CORLUX in that study. Based on this information, we are using a CORLUX dose of 1200 mg once per day for seven days in Study 14.
In addition, we also are utilizing a third party centralized rating service to independently evaluate the patients for entry into the study as well as to evaluate their level of response throughout their participation in the study. We believe the centralization of this process will improve the consistency of rating across clinical trial sites and reduce the background noise that was experienced in earlier studies and is endemic to many psychopharmacologic studies. We believe that this change in dose, as well as the other modifications to the protocol, should allow us to demonstrate the efficacy of CORLUX in the treatment of the psychotic symptoms of psychotic depression. In March 2009, we announced that, in order to conserve financial resources, we were scaling back our planned rate of spending on this trial and extended the timeline for its completion. As of early July 2009, we had completed the implementation of this strategy, which included reducing the number of clinical sites to eight.
Antipsychotic-induced Weight Gain Mitigation. In 2005, we published the results of studies in rats that demonstrated that CORLUX both reduced the weight gain associated with the ongoing use of olanzapine and mitigated the weight gain associated with the initiation of treatment with olanzapine (the active ingredient in Zyprexa). This study was paid for by Eli Lilly and Company (Eli Lilly).
2
Table of Contents
During 2007 we announced positive results from our clinical proof-of-concept study in lean healthy male volunteers evaluating the ability of CORLUX to mitigate weight gain associated with the use of Zyprexa. The results show a statistically significant reduction in weight gain in those subjects who took Zyprexa plus CORLUX compared to those who took Zyprexa plus placebo. Also, the addition of CORLUX to treatment with Zyprexa had a beneficial impact on secondary metabolic measures such as fasting insulin, triglycerides and abdominal fat, as indicated by waist circumference. Eli Lilly provided Zyprexa and financial support for this study. In January 2009 we announced positive results from a similar proof-of-concept study evaluating the ability of CORLUX to mitigate weight gain associated with the use of Johnson & Johnson’s Risperdal. This study, which began in 2008, confirmed and extended the earlier results seen with CORLUX and Zyprexa, demonstrating a statistically significant reduction in weight and secondary metabolic endpoints of fasting insulin, triglycerides and abdominal fat, as indicated by waist circumference. The results from the study of CORLUX and Risperdal were presented at several scientific conferences, including the American Diabetes Association meeting in June 2009.
The combination of Zyprexa or Risperdal and CORLUX is not approved for any indication. The purpose of these studies was to explore the hypothesis that GR-II antagonists, such as CORLUX and our next generation of selective GR-II antagonists, would mitigate weight gain associated with antipsychotic medications. The group of medications known as second generation antipsychotic medication, including Zyprexa, Risperdal, Clozaril and Seroquel, are widely used to treat schizophrenia and bipolar disorder. All medications in this group are associated with treatment emergent weight gain of varying degrees and carry a warning in their labels relating to treatment emergent hyperglycemia and diabetes mellitus.
We have completed IND enabling work with CORT 108297, which included preclinical studies in the rat in antipsychotic induced weight gain, diet induced weight gain and insulin sensitivity. In February 2010, we initiated a Phase 1 study to evaluate the tolerability of this compound in healthy volunteers. CORT 108297 is the lead compound from our three series of selective GR-II antagonists. Preclinical studies of CORT 108297, presented at scientific conferences during 2009, demonstrated a statistically significant mitigation in weight gain and other metabolic effects when added to olanzapine, the active ingredient in Eli Lilly’s medication Zyprexa. CORT 108297 also demonstrated the potential to mitigate weight gain caused by consumption of a high fat, high sucrose diet and improve insulin sensitivity in a preclinical mouse model.
Additional Indications. We have discovered and patented three series of next-generation selective GR-II receptor antagonists. As discussed above, the lead compound from these series, CORT 108297, is being developed for the prevention of weight gain induced by antipsychotic medication and is currently in a Phase 1 trial. There are numerous additional compounds in these three series that may be developed for weight gain mitigation or other diseases in which excess cortisol plays a role. The role of excess cortisol has been well established and documented in the scientific literature in diabetes, obesity, hypertension, osteoporosis, glaucoma, Alzheimer’s disease and various other neurodegenerative diseases, in addition to antipsychotic-induced weight gain.
The Role of Cortisol in Disease
Cortisol is a steroid hormone that plays a significant role in the way the body reacts to stressful conditions and is essential for survival. Cortisol significantly influences metabolism, exerts a clinically useful anti-inflammatory effect and contributes to emotional stability. Insufficient levels of cortisol may lead to dehydration, hypotension, shock, fatigue, low resistance to infection, trauma, stress and hypoglycemia. Excessive levels of cortisol may lead to edema, hypertension, fatigue and impaired glucose tolerance.
Elevated levels and abnormal release patterns of cortisol have also been linked to a broad range of metabolic and psychiatric conditions, such as weight gain, diabetes, hypertension, mood changes, psychosis and cognitive impairment.
3
Table of Contents
While excess release of cortisol may play a role in numerous diseases, Cushing’s Syndrome is the fundamental disease of excess cortisol, as patients have tumors that produce excess levels of the hormone cortisol or its precursor, adrenocorticotropic hormone (ACTH). Sometimes called “hypercortisolism”, the body’s exposure to high levels of cortisol can result in weight gain, diabetes, hypertension, infections, severe fatigue and psychosis.
Many studies have shown that patients with psychotic depression have elevated levels and abnormal release patterns of cortisol. This abnormal cortisol activity is not usually present in patients with nonpsychotic depression. More than 20 years ago, one of our scientific co-founders postulated that elevated levels of cortisol in patients with psychotic depression lead to elevated levels of dopamine, an important chemical substance found in the brain. Elevated levels of dopamine have been implicated in both delusional thinking and hallucinations. This hypothesis led to the concept that, by regulating the level and release patterns of cortisol, one could normalize dopamine levels in the brain, which may, in turn, ameliorate the symptoms of psychotic depression. In addition to cortisol’s effect on dopamine levels, research has shown that prolonged elevated cortisol may also play a direct role in causing the symptoms of psychotic depression.
The challenge in regulating levels of cortisol, however, is that it is needed for natural processes in the human body. Destroying the ability of the body to make cortisol or to drastically reduce its presence would result in serious detrimental effects. To have a viable therapeutic effect, a compound must be able to selectively modulate cortisol effects.
Glucocorticoid Receptor Antagonists
Cortisol is produced by the adrenal glands and is carried via the bloodstream to the brain, where it directly influences neuronal function. In the brain, cortisol binds to two receptors, Glucocorticoid Receptor I and Glucocorticoid Receptor II, also known as GR-I and GR-II. GR-I is a high-affinity receptor that is involved in the routine functions of cortisol in the brain. It has approximately ten times the affinity of GR-II for cortisol and its binding sites are filled with cortisol nearly all the time. In general, GR-II binding sites do not fill until levels of cortisol become elevated. Short-term activation of GR-II has benefits, which include helping the individual to be more alert and better able to function under stressful conditions. Long-term activation of GR-II, however, has been shown to have significant toxicity and appears to be linked to multiple metabolic and psychiatric disease states, such as Cushing’s Syndrome and psychotic depression. The action of cortisol can be moderated by the use of blockers, or antagonists, that prevent the binding of the hormone to its receptors. These antagonists, referred to as glucocorticoid, or cortisol, receptor antagonists, may prevent the undesirable effects of elevated levels and abnormal release patterns of cortisol.
CORLUX, also known as mifepristone, works by selectively blocking the binding of cortisol to GR-II; CORLUX is neither an antagonist nor agonist of GR-I. It also blocks the progesterone receptor (PR). Because of its selective affinity, we believe that CORLUX can have a therapeutic benefit by modulating the effects of abnormal levels and release patterns of cortisol without compromising the necessary normal functions of cortisol. We have also discovered three series of additional compounds, one of which includes our lead candidate CORT 108297, which, like CORLUX, potently block the GR-II receptor, but, unlike CORLUX, do not block the progesterone receptor.
Overview of Cushing’s Syndrome
Endogenous Cushing’s Syndrome is caused by prolonged exposure of the body’s tissues to high levels of the hormone cortisol due to a variety of pathologic conditions. In endogenous Cushing’s Syndrome, the production of excess cortisol is stimulated or directly produced by pituitary, adrenal or ectopic tumors. Cushing’s Syndrome is an orphan indication which most commonly affects adults aged 20 to 50. An estimated 10 to 15 of every one million people are newly diagnosed with this syndrome each year, resulting in over 3,000 new patients in the United States. An estimated 20,000 patients in the United States have been diagnosed with Cushing’s
4
Table of Contents
Syndrome. Symptoms vary, but most people have one or more of the following manifestations: high blood sugar, diabetes, high blood pressure, upper body obesity, rounded face, increased fat around the neck, thinning arms and legs, severe fatigue and weak muscles. Irritability, anxiety, cognitive disturbances and depression are also common. Cushing’s Syndrome can affect every organ system in the body and can be lethal if not treated effectively. There is no FDA-approved treatment for Cushing’s Syndrome.
Current Treatments for Cushing’s Syndrome
Current treatment depends on the specific cause of cortisol excess and may include surgery, radiation and chemotherapy. Patients sometimes may be treated with drugs that prevent the body from producing cortisol. Approximately 70% of the patients diagnosed with Cushing’s Syndrome are candidates for surgery. Depending on the type of tumor there are varying rates of success and complications related to removing the tumor. If the tumor is successfully removed in its entirety, the patient is essentially cured and will not require additional treatment for Cushing’s Syndrome. However, in approximately half of the patients, it is clear that surgery is not successful or, while surgery may appear to be successful initially, the patient later relapses. These patients currently have limited treatment options.
CORLUX for Cushing’s Syndrome
CORLUX represents a potentially attractive treatment option with the potential for long-term oral dosing. CORLUX is a GR-II antagonist that appears to mitigate the effects of the elevated levels of cortisol in patients suffering from Cushing’s Syndrome. We intend for CORLUX to be a once-daily chronic treatment in this indication. Mifepristone, the active ingredient in CORLUX, in addition to blocking GR-II, blocks the progesterone receptor and has been approved by the FDA for termination of early pregnancy.
We believe that CORLUX may significantly reduce a broad range of symptoms typically associated with Cushing’s Syndrome. These symptoms can include weight gain, diabetes, hypertension, poor tissue quality, fatigue and psychosis. Cushing’s Syndrome has a five-year 50% mortality rate if left untreated.
The FDA has granted Orphan Drug Designation for CORLUX for the treatment of endogenous Cushing’s Syndrome. “Orphan” drugs receive seven years of marketing exclusivity from the date of approval, as well as tax credits for clinical trial costs, marketing application filing fee waivers and assistance from the FDA in the drug development process.
CORLUX for Cushing’s Syndrome Clinical Experience
There have been reports in the scientific literature of more than 40 Cushing’s Syndrome patients who have been treated with mifipristone, the active ingredient in CORLUX. The clinical benefit supported by these data served as the rationale for our IND for CORLUX and design of our Phase 3 trial. While there have been no formal trials completed, the published results of the treatment of Cushing’s Syndrome patients with mifepristone include improvement in glucose tolerance and hemoglobin A1C levels, blood pressure, depression and psychosis, and improvement in the patient’s general quality of life.
CORLUX for Cushing’s Syndrome Phase 3 Study
We are conducting a Phase 3 trial with CORLUX for the treatment of endogenous Cushing’s Syndrome. The IND for the evaluation of CORLUX for the treatment of Cushing’s Syndrome was opened in September 2007. We are conducting a single 50-patient open-label study, in which patients’ dose is titrated to clinical benefit, and endpoints are focused on improvement in glucose tolerance and blood pressure, as well as broader measures of patient outcomes. The FDA has indicated that this trial may provide a reasonable basis for the submission of an NDA for this indication. We expect to complete enrollment in this Phase 3 study in April, as the requisite 50 patients have now been dosed or identified, and expect to announce results of this study in the fourth quarter of 2010.
5
Table of Contents
The primary endpoint in the trial is either 1) improvement in glucose tolerance (as measured by the area under the curve of an oral glucose tolerance test) at 24 weeks relative to baseline, or 2) if a patient is not glucose intolerant at baseline, improvement in diastolic blood pressure at 24 weeks relative to baseline. A patient in the glucose tolerance group is considered a responder if there is a 25% or greater improvement in the area under the curve of a standard oral glucose tolerance test over the 24-week course of the study. A patient in the hypertension group is considered a responder if there is a 5 millimeter or greater drop in diastolic blood pressure at 24 weeks relative to baseline. If a sufficient number of patients in either group are responders (such that the lower limit of the exact one-sided 95% binomial confidence interval for the responder rate is greater than 20%, or approximately 35% of the patients are responders, depending on the number of patients in each group) then the trial will have met its primary endpoint. The key secondary endpoint in the trial, global clinical improvement, is designed to capture the broader clinical benefit of CORLUX in this patient population.
Additional Trials and Preclinical Studies
In support of our planned NDA submission, we are conducting a long-term extension study in patients who completed the Phase 3 trial to assess safety of chronic dosing. We are conducting several small trials to evaluate how the drug acts on the human body, how the human body acts on the drug and the drug’s safety. In addition to our clinical trials, we have completed a standard 12-month toxicology study in dogs, a carcinogenicity study in rats, and a carcinogenicity study in mice. These studies are designed to meet FDA requirements and the guidelines of an international regulatory body called the International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use. We anticipate completing all of the additional trials required for our NDA submission for CORLUX for the treatment of Cushing’s Syndrome by the time of completion of our Phase 3 efficacy study.
Overview of Psychotic Depression
Psychotic depression is a serious psychiatric disease in which a patient suffers from severe depression accompanied by delusions, hallucinations or both. These psychotic features typically develop after the onset of a depressed mood, but may develop concurrently as well. Once psychotic symptoms occur, they usually reappear with each subsequent depressive episode. Of particular importance, when the patient’s mood returns to normal the psychosis also resolves.
Data from the National Institutes of Mental Health published in 2005 indicate that depressive disorders affect an estimated 9.5% of adults in the United States, or about 19 million people each year. Of these 19 million people, many published studies show that approximately 15-20%, or about three million people, have psychotic depression. Most patients with psychotic depression suffer their first episode of major depression between the ages of 30 and 40 and the majority will experience more than one episode in their lifetime. People with psychotic depression are approximately 70 times more likely to commit suicide in their lifetime than the general population and often require lengthy and expensive hospital stays.
Current Treatments for Psychotic Depression
There are two treatment approaches for psychotic depression currently used by psychiatrists: electroconvulsive therapy ECT and combination drug therapy, which is a combination of antidepressant and antipsychotic medication. Neither of these treatments has been approved by the FDA for psychotic depression and both approaches almost always have a slow onset of action, which may result in lengthy and costly hospitalization. Each of these treatments can have debilitating side effects. Of the two treatments, ECT is generally considered to be more effective.
| • | ECT involves passing an electrical current through the brain until the patient has a seizure. At least 100,000 patients receive ECT each year in the United States, with each patient requiring approximately six to twelve procedures over a period of three to five weeks. |
6
Table of Contents
| • | Combination drug therapy is an alternative treatment for psychotic depression that involves taking antipsychotic drugs such as olanzapine, haloperidol or chlorpromazine in combination with antidepressant drugs, such as fluoxetine, imipramine or venlafaxine. Patients on combination drug therapy often require three weeks or more to show improvement in their symptoms and treatment can take months before the symptoms are resolved entirely. Antipsychotic drugs can cause significant adverse side effects, including weight gain, diabetes, sedation, permanent movement disorders and sexual dysfunction. |
CORLUX for the Psychotic Features of Psychotic Depression
We are also developing CORLUX as an oral medication to treat the psychotic features of psychotic depression. As a GR-II antagonist, CORLUX appears to mitigate the effects of the elevated and abnormal release patterns of cortisol in patients suffering from psychotic depression. We intend CORLUX to be a once-daily treatment given to patients with psychotic depression over 7 consecutive days in a controlled setting, such as a hospital or physician’s office.
We believe that CORLUX may significantly reduce psychotic symptoms of psychotic depression in many patients within one week and allow patients to be more easily maintained on antidepressant therapy alone without the need for ECT or antipsychotic medication. We believe that CORLUX may be superior to currently available treatments because we believe that CORLUX will enable patients with psychotic depression to improve their quality of life more quickly and with fewer side effects than with ECT or combination drug therapy.
Completed Clinical Trials of CORLUX for Psychotic Depression
We have completed seven prior clinical trials evaluating CORLUX in psychotic depression, in addition to our ongoing Phase 3 trial. The trials include three Phase 3 trials conducted from 2004 through 2007, in addition to four earlier stage clinical trials with CORLUX. These completed trials generated important data confirming the safety profile of CORLUX (alone and in combination with commonly prescribed antipsychotic and antidepressant medications), demonstrated positive efficacy trends, and provided insights into the design of future clinical trials which might improve the probability of clinical success.
Completed Phase 3 Clinical Trials. In addition to Phase 1 and 2 studies, we have completed three randomized, double-blind, placebo-controlled Phase 3 clinical trials to further assess the safety and efficacy of CORLUX for the treatment of the psychotic features of psychotic depression. Two of these trials (Study 06 and Study 07) were conducted primarily in the United States. The third trial (Study 09) was conducted in Eastern Europe.
The primary endpoint for Study 06 and Study 07 was the proportion of patients with at least a 50% improvement in the Brief Psychiatric Rating Scale Positive Symptom Subscale (BPRS PSS) at both Day 7 and Day 56. The primary endpoint for Study 09 was the proportion of patients with at least a 50% improvement in the BPRS PSS, at both Day 7 and Day 28, with day 56 as a secondary endpoint. Patients must have had at least mild psychotic symptoms (BPRS PSS ³12) to enter the studies and were hospitalized if clinically necessary.
| • | Study 07: The first of these trials, which began in September 2004, enrolled 257 patients randomized one-to-one to either treatment or placebo. Patients in the treatment arm received 600 mg of CORLUX once daily for a period of seven days. Patients did not take any antidepressant or antipsychotic medication for at least one week before beginning the seven day treatment period. After the seven days of CORLUX treatment, all patients received antidepressant therapy through Day 56. Treatment with antipsychotic medications or ECT was not allowed at any time during the study. |
In this study patients receiving CORLUX did not have a statistically significant difference in response rate at the primary endpoint than did the patients receiving placebo. A retrospective analysis of the data showed that patients achieving drug plasma levels higher than 1800 nanograms per milliliter (ng/ml)
7
Table of Contents
had a statistically significant greater response rate than placebo. There was also a statistically significant site by treatment effect in this trial. Among the twenty sites who participated from the trial onset, patients who were given CORLUX had a significantly higher response rate than patients who received placebo. Among the sites added later in the trial, there was no significant difference in response rate between CORLUX and placebo patients. These findings were published in 2009 by Contemporary Clinical Trials.
| • | Study 09: This study, which commenced in May 2005, was a randomized, double-blind, placebo-controlled study in which 247 patients were enrolled at sites in Eastern Europe. The primary endpoint was the proportion of patients with at least a 50% improvement in the BPRS PSS score at both Day 7 and Day 28. The study did not demonstrate a significant difference in response between patients receiving CORLUX and patients receiving placebo as measured by the primary endpoint. The results at the two key secondary endpoints of Study 09 also were not statistically significant. Study 09 had an extremely high placebo response rate. |
| • | Study 06: This trial began in October 2004, and enrolled 443 patients. These patients were randomly assigned to three active dose groups (300 mg, 600 mg and 1200 mg) or a placebo group, with patients receiving once daily dosing for a period of seven days. The three dosing levels responded to the FDA’s request to supplement data on a range of doses to augment the data provided by our open label dose ranging study completed in 2001. |
The study did not achieve statistical significance with respect to the primary endpoint. However, there was a statistically significant correlation between plasma levels and clinical outcome achieved during treatment. Response rates for patients whose plasma levels rose above a predetermined threshold of 1661 ng/mL were statistically different than those patients whose plasma levels were below the threshold and those patients who received placebo. Further, the incidence of serious adverse events did not differ between placebo and any of the three CORLUX dose groups.
Ongoing Phase 3 trial – Study 14: We believe that the confirmation of a correlation between drug concentration and clinical response, as well as other observations from Study 06 and our two other completed Phase 3 clinical trials, served as a strong basis for the design of our ongoing Phase 3 study, which commenced in March 2008. The protocol for this trial incorporates information learned from the three completed Phase 3 trials in that it addresses the established relationship between increased drug plasma levels and clinical response, and it attempts to decrease the random variability observed in the results of the psychometric instruments used to confirm diagnosis and measure efficacy.
| • | Increased Signal: In this trial we are administering a CORLUX dose of 1200 mg once per day for seven days instead of 600 mg once per day for seven days. |
| • | Decreased “Noise”: We also are utilizing a third party centralized rating service to independently evaluate the patient’s diagnosis prior to entry into the study as well as to assess response. We believe the centralization of this process will improve the accuracy of diagnosis and the consistency of rating across clinical trial sites and reduce the background noise that is endemic to many psychopharmacologic studies and clearly visible in our earlier studies. |
We believe that these changes in the protocol should allow us to establish the efficacy of CORLUX in the treatment of the psychotic features of psychotic depression. Given the serious nature of psychotic depression, the lack of any approved drugs for the disorder and the data from our first clinical trial, the FDA granted a fast track designation for CORLUX for the treatment of the psychotic features of psychotic depression. In addition, the FDA has indicated that CORLUX will receive a priority review if no other treatment is approved for psychotic depression at the time we submit our NDA.
Clinical Trial Agreements. Many of our Phase 3 clinical trials are conducted through the use of clinical research organizations (CROs.) At our request, these organizations oversee clinical trials at various institutions to test the safety and efficacy of our product candidates for the targeted indications. Our ongoing Phase 3 clinical
8
Table of Contents
trial, Study 14, evaluating CORLUX for the treatment of the psychotic features of psychotic depression is being conducted under an agreement with ICON Clinical Research, LP (ICON). We may terminate this agreement with 60 days notice to ICON, or sooner based on mutual agreement of the parties. In addition, we entered into an agreement with MedAvante, Inc., in March 2008, to provide the centralized psychiatric diagnosis and rating services for patients being screened and enrolled in Study 14. We may terminate this agreement with 30 days notice to MedAvante.
CORLUX for Other Metabolic Disorders
In April 2005, we announced results from two preclinical studies conducted in a rat model of olanzapine-induced weight gain. These studies demonstrated that CORLUX’s GR-II antagonist action has the potential to both reduce the weight gain associated with olanzapine and to prevent the weight gain associated with the initiation of treatment with olanzapine, which led to our studies in humans.
In 2007, we announced results of our human clinical proof-of-concept study evaluating the ability of CORLUX to mitigate weight gain associated with the administration of Eli Lilly’s Zyprexa® (olanzapine). The results indicated a statistically significant reduction in weight gain in those subjects who took Zyprexa plus CORLUX compared to those who took Zyprexa plus placebo. Eli Lilly provided Zyprexa and financial support for this study. During 2009, we announced results from another proof-of-concept study evaluating the ability of CORLUX to mitigate weight gain associated with the administration of Johnson & Johnson’s Risperdal (risperidone). The results indicated a statistically significant reduction in weight gain in those subjects who took Risperdal plus CORLUX compared to those who took Risperdal plus placebo. Both Zyprexa and Risperdal are indicated for the treatment of schizophrenia and bipolar disorder.
In the study of CORLUX and Zyprexa, 57 lean, healthy men (body mass index of 25 or less) were randomized to receive either Zyprexa plus placebo (n=22), Zyprexa plus CORLUX (n=24) or CORLUX plus placebo (n=11). This study took place in an institutional setting where daily weights were recorded and a range of metabolic parameters were measured. In the two week study, subjects in the Zyprexa plus placebo group gained an average of 7.0 pounds and subjects in the Zyprexa plus CORLUX group gained an average of 4.4 pounds; which is a highly statistically significant difference (p<.001). Subjects in the CORLUX plus placebo group gained an average of 4.4 pounds. The difference in weight gain trajectory was apparent in the first days of the study, reaching statistical significance during the first week. The increase in waist circumference, a surrogate for abdominal fat, in subjects who received Zyprexa plus placebo was also significantly greater than subjects who received Zyprexa plus CORLUX (p<.01). The study was not designed to enroll a sufficient number of patients to have statistical power to detect significant effects on metabolic measures, including waist circumference. However, in addition to the finding about waist circumference, notable additional non-statistically significant group differences were observed. Patients taking Zyprexa plus placebo experienced greater increases from baseline to end of study in both triglycerides and fasting insulin compared to patients taking Zyprexa plus CORLUX. No unexpected study drug related adverse events were observed.
In the study of CORLUX and Risperdal, 75 lean, healthy men (body mass index of 23 or less) were randomized to receive either Risperdal plus placebo (n=30), Risperdal plus CORLUX (n=30) or CORLUX plus placebo (n=15). This study also took place in an institutional setting where daily weights were recorded and a range of metabolic parameters were measured. In this four-week randomized double-blind controlled study, subjects in the Risperdal plus placebo group gained an average of 9.2 pounds, compared to a gain of 5.1 pounds in the Risperdal plus CORLUX group. This difference was highly statistically significant (p<0.0001). Additional important metabolic parameters, including fasting insulin, triglycerides and abdominal fat, as reflected by waist circumference, were also measured. The addition of CORLUX to Risperdal resulted in a statistically significant reduction in fasting insulin levels, triglyceride levels, and abdominal fat (as measured by waist circumference). Consistent with prior studies, CORLUX appeared to be well tolerated.
9
Table of Contents
The combinations of Zyprexa and CORLUX or Risperdal and CORLUX are not approved for any indication. The purpose of these studies was to explore the hypothesis that GR-II antagonists would mitigate weight gain and other metabolic effects associated with antipsychotic medications. The group of medications sometimes referred to as “atypical antipsychotics,” including Zyprexa, Risperdal, Clozaril® (clozapine) and Seroquel® (quetiapine), are widely used to treat schizophrenia and bipolar disorder. All medications in this group are associated with treatment emergent weight gain of varying degrees and carry a warning in the label relating to treatment emergent hyperglycemia and diabetes mellitus.
CORT 108297 for the Prevention and Reversal of Antipsychotic Induced Weight Gain
In January 2009 we announced results from two preclinical studies of our next-generation selective GR-II receptor antagonist, CORT 108297 for the prevention and reversal of weight gain associated with olanzapine, which is marketed by Eli Lilly as Zyprexa. The data demonstrated that CORT 108297 has the potential to both reduce weight gain caused by olanzapine and to prevent weight gain caused by initiation of treatment with olanzapine. The two studies were conducted in the rat model of olanzapine induced weight gain described above, in which CORLUX was tested with olanzapine.
| • | One study evaluated the potential for CORT 108297 to reverse weight gain caused by treatment with olanzapine. In this study six groups (n = 12 per group) of rats were allowed to eat a normal diet for 56 days. Five groups were dosed orally with olanzapine daily. The sixth group received placebo. At day 35, the five groups receiving olanzapine had gained a statistically significant amount of weight compared to the group receiving placebo. The five olanzapine groups then began to receive daily oral doses either of CORT 108297 (at one of three dose levels), CORLUX or placebo through day 56. The data demonstrated that the rats administered olanzapine plus placebo continued to gain weight through day 56. In contrast, the rats given olanzapine along with CORT 108297 and those administered olanzapine with CORLUX did not. By day 56, there was a highly statistically significant difference between these groups and the group administered olanzapine plus placebo. In addition, the ameliorization of olanzapine induced weight gain by CORT 108297 was dose dependent. The rats that received the combination of olanzapine with CORT 108297, or with CORLUX, also had significantly less abdominal fat than the group dosed with olanzapine alone. |
| • | The other study evaluated the potential for CORT 108297 to prevent weight gain when administered concurrently with olanzapine. In this study six groups (n = 12 per group) of rats were allowed to eat a normal diet for 21 days. Five groups were dosed orally with olanzapine daily and one group was given placebo daily. Four of the groups that received olanzapine were also dosed orally with either CORT 108297 (at one of three dose levels) or CORLUX; one group received olanzapine plus placebo. The sixth group was dosed with only placebo. The data demonstrated that at day 21, the three groups dosed with the combination of olanzapine and CORT 108297 had gained significantly less weight compared to the group administered olanzapine alone. Rats administered olanzapine plus CORLUX also gained less weight than rats administered olanzapine alone, but this result did not reach statistical significance. |
These first two studies used dose levels of 20 milligrams per kilogram (mg/kg), 60 mg/kg and 120 mg/kg of CORT 108297. Eli Lilly provided olanzapine and funded the costs of these two studies.
| • | A third study in the rat further evaluated the dose response relationship of CORT 108297 in preventing olanzapine induced weight gain with doses from 2 mg/kg to 20 mg/kg. |
In summary, these studies in the rat demonstrated a constant dose response relationship from 2 mg/kg to 120 mg/kg.
CORT 108297 has also produced statistically significant results in the prevention of weight gain and insulin insensitivity in mice fed a high fat, high sucrose diet.
10
Table of Contents
If CORT 108297 or other GR-II antagonists prove to mitigate the weight gain and metabolic disturbances associated with the use of antipsychotic medication, they could be of benefit to the millions of people currently taking this important pharmacotherapy. We advanced CORT 108297 into a Phase 1 clinical trial during the first quarter of 2010. We plan to advance the compound into a Phase 2 trial in 2011, subject to availability of funds.
GR-II Antagonist Platform
We have assembled a patent portfolio covering a broad range of uses, as well as the composition of our new chemical entities.
| • | We have composition of matter claims on three patent families of novel selective glucocorticoid receptor (GR-II) antagonists. Applications for all of the three families have been allowed in Europe. In the United States, applications for two of the three families have been allowed. Examination has not yet begun in the United States on our third novel selective GR-II antagonist family. |
| • | We also have a portfolio of patents describing the use of drugs that block the GR-II receptor for the treatment of metabolic and psychiatric disorders. In addition to psychotic depression, we own or have exclusively licensed issued patents for the use of GR-II antagonists for treatment and / or prevention of: |
| • | weight gain following treatment with antipsychotic medication; |
| • | mild cognitive impairment; |
| • | stress disorders; |
| • | early dementia, including early Alzheimer’s disease; |
| • | delirium; |
| • | gastroesophageal reflux disease; |
| • | cognitive deterioration in adults with Down’s Syndrome; |
| • | psychosis associated with cocaine addiction and |
| • | increasing the therapeutic response to ECT. |
Discovery Research
In 2003, we initiated a discovery research program to identify and patent selective GR-II antagonists at a contract research organization in the United Kingdom. Through the research program, we identified and filed patent applications for three distinct series of GR-II antagonists. These compounds appear to be as potent as Corcept’s lead product CORLUX in blocking cortisol but, unlike CORLUX, they do not appear to block the progesterone or other steroid receptors. Currently, we are evaluating several compounds in our research programs, including CORT 108297, a lead compound from our discovery efforts. CORT 108297 has demonstrated attractive characteristics, with high plasma and brain concentrations in an animal model and promising results in a human microdosing study, including good bioavailability and potential for once-daily dosing. CORT 108297 has also demonstrated the ability to prevent and reduce olanzapine induced weight gain in a rat model, as well as to prevent weight gain from a high fat, high sugar diet and increase insulin sensitivity in a mouse model. CORT 108297 is being evaluated in a Phase 1 study.
Research and Development
We incurred approximately $14.4 million, $14.2 million and $7.9 million of research and development expenses, respectively, in the years ended December 31, 2009, 2008 and 2007, which accounted for approximately 71%, 71% and 62% of our total expenses in these respective fiscal years. For a further discussion, see Part II, Item 7, Management’s Discussion and Analysis of Financial Conditions and Results of Operations – Results of Operations.
11
Table of Contents
Medical Education and Commercialization
We are planning for the commercialization of CORLUX. To achieve commercial success for any approved product, we must either develop a marketing and sales force or enter into arrangements with others to market and sell our products. We intend to develop our own medical affairs and commercialization infrastructure in the United States for CORLUX because we believe that the initial markets for Cushing’s Syndrome and psychotic depression in the United States are highly concentrated and accessible. We intend to engage a partner to commercialize CORLUX in territories outside of the United States.
| • | If approved, we expect to hire a small, experienced field sales force, supported by medical affairs and other infrastructure, to sell CORLUX for the treatment of Cushing’s Syndrome. We intend to focus on patients who are in the care of an endocrinologist and in active treatment for their disease. We estimate that there are fewer than 1000 endocrinologists who would need to be targeted to reach the Cushing’s Syndrome population in active treatment. We plan to reach out directly to patients utilizing web-based initiatives and interactions with patient groups. We expect distribution and logistical support to be provided by specialty pharmacies. |
A large portion of the people who suffer from Cushing’s Syndrome remain unrecognized or undertreated. We intend to develop programs to educate the medical community about early diagnosis of this Syndrome and increase awareness regarding CORLUX.
| • | If approved for the treatment of psychotic depression, we plan to reach patients who are candidates for ECT by marketing to hospitals and psychiatrists that perform ECT. We estimate that there are approximately 900 hospitals with more than 30 in-patient psychiatric beds. Of these, we estimate that approximately 300 offer ECT. We believe that approximately 1000 psychiatrists administer most ECT procedures. Subsequently, we also intend to expand our commercialization efforts to address the larger set of patients with psychotic depression currently undergoing combination drug therapy, which would require an increase in the size of our initial sales force. |
As with Cushing’s Syndrome, a large portion of the people who suffer from psychotic depression remain unrecognized or undertreated. We intend to develop programs to educate the medical community about early diagnosis of psychotic depression and increase awareness regarding CORLUX as a treatment for this disorder.
Manufacturing
As a drug development entity, we intend to continue to utilize our financial resources to complete the development of CORLUX and advance other product candidates rather than diverting resources to establishing our own manufacturing facilities.
We intend to continue to rely on experienced contract manufacturers to produce our product candidates. We have entered into manufacturing agreements with two contract manufacturers, Produits Chimiques Auxiliaires et de Synthese SA (PCAS) and ScinoPharm Taiwan (ScinoPharm), to produce the active pharmaceutical ingredient (API) for CORLUX. The agreement with PCAS, which was executed in early November 2006, is for an initial period of five years with an automatic extension for one additional year unless either party gives twelve month’s prior notice that it does not want the extension. There is no guaranteed minimum purchase commitment under this agreement. If PCAS is unable to manufacture the product for a consecutive six-month period, we have the right to terminate the agreement. The agreement with ScinoPharm obligates us to purchase at least $1,000,000 of bulk mifepristone per year following the commercial launch of CORLUX. This agreement is terminable by either party at any time. We have also entered into an agreement with another contract manufacturer, PharmaForm, L.L.C., for the production of CORLUX tablets for use in clinical activities. To date, our need for CORLUX tablets has been limited to the amounts required to support our clinical trials.
12
Table of Contents
Competition
If approved for commercial use as a treatment for Cushing’s Syndrome or the psychotic features of psychotic depression, CORLUX will compete with established treatments, including other potential compounds under development for Cushing’s Syndrome or, in the case of psychotic depression, with ECT and combination drug therapy.
We are aware that Laboratoire HRA Pharma has received an Orphan Drug Designation in the United States and Europe for the use of mifepristone to treat a subtype of Cushing’s Syndrome and has begun a clinical trial in Europe and the United States. If this product is approved for commercialization before CORLUX, our potential future revenue could be reduced. We are also aware that Exelgyn Laboratories received Orphan Drug Designation for Cushing’s Syndrome in Europe, but they have stated that they have not yet conducted any clinical trials. We may also experience competition from Novartis, which is developing a somatostatin analogue, pasireotide, that is in Phase 3 trials for various endocrine disorders, including Cushing’s disease, which is a subset of the patients with Cushing’s Syndrome.
ECT has been shown to be the most effective treatment for psychotic depression, but it carries the risks of general anesthesia, potential memory loss and other adverse effects as well as the stigma associated with the procedure. Use of CORLUX does not require anesthesia and, in our clinical trials conducted to date, patients treated with CORLUX have not exhibited the adverse effects associated with ECT.
Other competitors include companies that market antipsychotic drugs that are used off-label as part of combination drug therapy for psychotic depression. To reduce the psychotic features of psychotic depression, these drugs generally are taken in combination with antidepressant medication over a period of weeks to several months. Unlike the use of CORLUX, this extended course of treatment may put patients at risk of significant adverse side effects, including weight gain, diabetes, sedation, permanent movement disorders and sexual dysfunction. Antipsychotics include Bristol-Myers Squibb’s Abilify, Novartis’ Clozaril, Pfizer’s Geodon and Navane, Ortho-McNeil’s Haldol, Janssen Pharmaceutica’s Risperdal, AstraZeneca’s Seroquel, GlaxoSmithKline’s Stelazine and Thorazine, Mylan’s Mellaril, Schering Corporation’s Trilafon and Eli Lilly’s Zyprexa.
We are aware of one clinical trial that has taken place, conducted by the pharmaceutical division of Akzo Nobel, a division of Schering Plough, for a new chemical entity for the treatment of psychotic depression. This medicine is a GR-II antagonist, the commercial use of which would be covered by our patent. In 2004, Akzo Nobel filed an observation in our exclusively licensed European patent application with claims directed to psychotic depression, in which Akzo Nobel challenged the claims of that patent application. In 2005, we filed a rebuttal to Akzo Nobel’s observation. In February 2006, the European Patent Office (EPO) allowed our patent application. In July 2006, the patent was issued. We are not aware of any public disclosures by any company, other than Akzo Nobel, regarding the development of new medicinal products to treat psychotic depression. However, other companies may be developing new drug products to treat psychotic depression and the other conditions we are exploring. Our present and potential competitors include major pharmaceutical companies, as well as specialty pharmaceutical firms. Most of our competitors have considerably greater financial, technical and marketing resources than we do. We expect competition to intensify as technical advances are made.
Many colleges, universities and public and private research organizations are also active in the human health care field. While these entities focus on education, they may develop or acquire proprietary technology that we may require for the development of our product candidates. We may attempt to obtain licenses to this proprietary technology.
Our ability to compete successfully will be based on our ability to develop proprietary products, attract and retain scientific personnel, obtain patent or other protection for our product candidates, obtain required regulatory approvals and manufacture and successfully market our future products either alone or through outside parties.
13
Table of Contents
Intellectual Property
Patents and other proprietary rights are important to our business. It is our policy to seek patent protection for our inventions, and to rely upon trade secrets, know-how, continuing technological innovations and licensing opportunities to develop and maintain our competitive position.
Under an agreement with Stanford University, we have licensed exclusive rights to the following issued U.S. patents and any corresponding foreign patents:
| U.S. Patent Number |
Subject Matter |
Expiration Date | ||
| 6,150,349 | Use of GR-II antagonists in the treatment of psychotic major depression | October 5, 2018 | ||
| 6,362,173 | Use of GR-II antagonists in the treatment of cocaine-induced psychosis | October 5, 2018 | ||
| 6,369,046 | Use of GR-II antagonists in the treatment of early dementia | February 4, 2019 | ||
We are required to make milestone payments and pay royalties to Stanford University on sales of products commercialized under any of the above patents. We are currently in compliance with our obligations under the agreement. If Stanford University were to terminate any of our exclusive licenses due to breach of the license on our part, we would not be able to commercialize CORLUX for the treatment of the psychotic features of psychotic depression, cocaine-induced psychosis or early dementia.
We also own issued U.S. patents for the use of GR-II antagonists in the treatment of mild cognitive impairment, for the treatment of weight gain following treatment with antipsychotic medication, for the prevention and treatment of stress disorders, for increasing the therapeutic response to ECT, for the treatment of delirium, for the treatment of gastroesophageal reflux disease and for inhibiting cognitive deterioration in adults with Down’s Syndrome.
In addition, we have eight U.S. method of use patent applications covering certain GR-II antagonists, including the treatment of:
| • | patients suffering from mental disorders by optimizing mifepristone levels in plasma serum; |
| • | postpartum psychosis; |
| • | neurological damage in premature infants; |
| • | catatonia; |
| • | migraine headaches; |
| • | psychosis associated with interferon-alpha therapy; |
| • | depression in patients taking Interleukin-2 (IL-2) and |
| • | amyotrophic lateral sclerosis (ALS). |
We have composition of matter claims on three patent families of novel selective GR-II antagonists. Applications for all of the three families have been allowed in Europe. In the United States, applications for two of the three families have been allowed. Examination has not yet begun in the United States on our third novel selective GR-II antagonist family.
14
Table of Contents
We have also filed, where we deemed appropriate, foreign patent applications corresponding to our U.S. patents and applications.
However, we cannot assure you that any of our patent applications will result in the issuance of patents, that any issued patent will include claims of the breadth sought in these applications or that competitors will not successfully challenge or circumvent our patents if they are issued.
Although two of our patents and one of our patent applications have claims directed to the composition of compounds, we do not have a patent with claims directed to the composition of mifepristone. Our rights under our issued patents related to mifepristone cover only the use of that compound in the treatment of specific diseases.
The patent covering the product mifepristone has expired. The only FDA-approved use of mifepristone is to terminate pregnancy. The FDA has imposed significant restrictions on the use of mifepristone to terminate pregnancy and may impose restrictions on CORLUX for the treatment of Cushing’s Syndrome and the psychotic features of psychotic depression. We plan to rely on (1) the scope of our use patent, (2) the restrictions imposed by the FDA on the use of mifepristone to terminate pregnancy and (3) the different patient populations, administering physicians and treatment settings between the use of mifepristone to terminate pregnancy and to treat Cushing’s Syndrome and psychotic depression.
The patent positions of companies in the pharmaceutical industry are highly uncertain, involve complex legal and factual questions and have been and continue to be the subject of much litigation. Our product candidates may give rise to claims that we infringe on the products or proprietary rights of others. If it is determined that our drug candidates infringe on others’ patent rights, we may be required to obtain licenses to those rights. If we fail to obtain licenses when necessary, we may experience delays in commercializing our product candidates while attempting to design around other patents, or determine that we are unable to commercialize our product candidates at all. If we do become involved in intellectual property litigation, we are likely to incur considerable costs in defending or prosecuting the litigation. We believe that we do not currently infringe any third party’s patents or other proprietary rights, and we are not obligated to pay royalties to any third party other than Stanford University.
In November 2003, McLean Hospital had alleged that it also had rights to the technology that led to the patent for the use of GR-II antagonists to treat the psychotic features of psychotic depression. McLean Hospital was a prior employer of one of our founders, Dr. Alan Schatzberg and it alleged that the invention of the technology underlying this patent was conceived by Dr. Schatzberg and/or Dr. Anthony Rothschild while the two were employed by McLean Hospital. We contended that the invention was actually conceived by Dr. Schatzberg and Dr. Joseph Belanoff while they were employed by Stanford University and that the patent was appropriately assigned by them to Stanford University. In October 2004, we announced a resolution of this issue in which we retained our exclusive rights under the patent and which required us to make no additional payments under the license, regardless of the resolution of the impending inventorship dispute. In January 2005, the inventorship issue was resolved in favor of Stanford University.
As discussed earlier under “Competition,” in 2004 Akzo Nobel filed an observation to the grant of our exclusively licensed European patent application with claims directed to psychotic depression. In February 2006, the EPO allowed our patent application. We are not aware of any other disputes related to patent issues.
License Agreement
Under our exclusive license agreement with Stanford University to patents covering the use of CORLUX to treat the psychotic features of psychotic depression and for the treatment of early dementia, we are required to pay Stanford $50,000 annually as a nonrefundable royalty payment. This payment is creditable against future royalties. We are also obligated to pay Stanford a $50,000 milestone upon the filing of the NDA for CORLUX
15
Table of Contents
for the treatment of psychotic depression and a further $200,000 milestone payment upon FDA approval of CORLUX. The milestone payments are also creditable against future royalties. This license agreement expires upon expiration of the related patents or upon notification by us to Stanford.
Government Regulation
Prescription pharmaceutical products are subject to extensive pre- and post-market regulation, including regulations that govern the testing, manufacturing, safety, efficacy, labeling, storage, record keeping, advertising, and promotion of the products under the Federal Food, Drug and Cosmetic Act. All of our product candidates will require regulatory approval by government agencies prior to commercialization. The process required by the FDA before a new drug may be marketed in the United States generally involves the following: completion of preclinical laboratory and animal testing; submission of an IND, which must become effective before clinical trials may begin; performance of adequate and well controlled human clinical trials to establish the safety and efficacy of the proposed drug or biologic’s intended use; and, in the case of a new drug, approval by the FDA of an NDA. The process of complying with these and other federal and state statutes and regulations in order to obtain the necessary approvals and subsequently complying with federal and state statutes and regulations involves significant time and expense.
Preclinical studies are generally conducted in laboratory animals to evaluate the potential safety and the efficacy of a product. Drug developers submit the results of preclinical studies to the FDA as a part of an IND, which must be approved before beginning clinical trials in humans. Typically, human clinical trials are conducted in three sequential phases that may overlap.
| • | Phase 1. Clinical trials are conducted with a small number of subjects to determine the early safety profile, maximum tolerated dose and pharmacokinetics of the product candidate in human volunteers. |
| • | Phase 2. Clinical trials are conducted with groups of patients afflicted with a specific disease to determine preliminary efficacy, optimal dosages and expanded evidence of safety. |
| • | Phase 3. Large-scale, multi-center, comparative trials are conducted with patients afflicted with a target disease to establish the overall risk/benefit ratio of the drug and to provide enough data to demonstrate with substantial evidence the efficacy and safety of the product, as required by the FDA. |
The FDA and the Institutional Review Boards closely monitor the progress of each of the three phases of clinical trials that are conducted in the United States and may reevaluate, alter, suspend or terminate the testing at any time for various reasons, including a belief that the subjects are being exposed to an unacceptable health risk. The FDA may also require that additional studies be conducted, such as studies demonstrating that the drug being tested does not cause cancer.
After Phase 3 trials are completed, drug developers submit the results of preclinical studies, clinical trials, formulation studies and data supporting manufacturing to the FDA in the form of an NDA for approval to commence commercial sales. The FDA reviews all NDAs submitted before it accepts them for filing. The FDA may request additional information rather than accept an NDA for filing. If the FDA accepts an NDA for filing, they may grant marketing approval, request additional information or deny the application if it determines that the application does not meet regulatory approval criteria. FDA approvals may not be granted on a timely basis, or at all.
If the FDA approves an NDA, the subject drug becomes available for physicians to prescribe in the United States. Once approved, the FDA may withdraw the product approval if compliance with pre- and post-market regulatory standards is not maintained. The drug developer must submit periodic reports to the FDA. Adverse experiences with the product must be reported to the FDA and could result in the imposition of marketing restrictions through labeling changes or product removal. Product approvals may be withdrawn if problems with
16
Table of Contents
safety or efficacy occur after the product reaches the marketplace. In addition, the FDA may require post- marketing studies, referred to as Phase 4 studies, to monitor the effect of approved products, and may limit further marketing of the product based on the results of these post-market studies.
Facilities used to manufacture drugs are subject to periodic inspection by the FDA and other authorities where applicable, and must comply with current Good Manufacturing Practices regulations (cGMP). Failure to comply with the statutory and regulatory requirements subjects the manufacturer to possible legal or regulatory action, such as suspension of manufacturing, seizure of product or voluntary recall of a product.
With respect to post-market product advertising and promotion, the FDA imposes a number of complex regulations on entities that advertise and promote pharmaceuticals, which include, among others, standards and regulations for direct-to-consumer advertising, off-label promotion, industry sponsored scientific and educational activities, and promotional activities involving the Internet. The FDA has very broad enforcement authority under the Federal Food, Drug and Cosmetic Act, and failure to abide by these regulations can result in penalties including the issuance of a warning letter directing a company to correct deviations from FDA standards, a requirement that future advertising and promotional materials be pre-cleared by the FDA, and state and federal civil and criminal investigations and prosecutions.
In addition to studies requested by the FDA after approval, a drug developer may conduct other trials and studies to explore use of the approved compound for treatment of new indications. The purpose of these trials and studies and related publications is to broaden the application and use of the drug and its acceptance in the medical community. Data supporting the use of a drug for these new indications must be submitted to the FDA in a new or supplemental NDA that must be approved by the FDA before the drug can be marketed for the new indications.
Approvals outside the United States. We have not started the regulatory approval process in any jurisdiction other than the United States and we are unable to estimate when, if ever, we will commence the regulatory approval process in any foreign jurisdiction. We or our partners will have to complete an approval process similar to the U.S. approval process in foreign target markets for our product candidates before we can commercialize our product candidates in those countries. The approval procedure and the time required for approval vary from country to country and can involve additional testing. Foreign approvals may not be granted on a timely basis, or at all. Regulatory approval of prices is required in most countries other than the United States. The prices approved may be too low to generate an acceptable return to us.
Orphan Drug Designation. The FDA has granted us Orphan Drug designation for CORLUX for the treatment of endogenous Cushing’s Syndrome. The designation provides special status to a product to treat a rare disease or condition providing that the product meets certain criteria. Orphan designation qualifies the sponsor of the product for the tax credit and marketing incentives of the Orphan Drug Act. A marketing application for a prescription drug product that has been designated as a drug for a rare disease or condition is not subject to a prescription drug user fee unless the application includes an indication for other than a rare disease or condition.
Fast Track Designation. The FDA sometimes grants “fast track” status under the Food and Drug Administration Modernization Act of 1997. The fast track mechanism was created to facilitate the development and approval of new drugs intended for the treatment of life-threatening conditions for which there are no effective treatments and which demonstrate the potential to address unmet medical needs for the condition. The fast track process includes scheduling of meetings to seek FDA input into development plans, the option of submitting an NDA serially in sections rather than submitting all components simultaneously, the option to request evaluation of studies using surrogate endpoints, and the potential for a priority review.
17
Table of Contents
We have been granted fast track status for CORLUX for the treatment of the psychotic features of psychotic depression. However, the fast track designation may be withdrawn by the FDA at any time. The fast track designation does not guarantee that we will qualify for or be able to take advantage of the expedited review procedures and does not increase the likelihood that CORLUX will receive regulatory approval.
Priority Review. The FDA has indicated to us that it will grant us a priority review of our NDA of CORLUX for the treatment of the psychotic features of psychotic depression if no other medications have been approved for this indication at the time of our submission.
Employees
We are managed by a core group of experienced pharmaceutical executives with a track record of bringing new drugs to market. To facilitate advancement of development programs, we also enlist the expertise of associates and advisors with extensive pharmaceutical development experience.
As of December 31, 2009, we had 16 full-time employees, three part-time employees and 11 long-term contract staff. Four of our employees are M.D.s. We consider our employee relations to be good. None of our employees is covered by a collective bargaining agreement.
General
We were incorporated in the State of Delaware on May 13, 1998. Our registered trademarks include Corcept® and CORLUX®. Other service marks, trademarks and trade names referred to in this document are the property of their respective owners.
Available Information
We are subject to the information requirements of the Securities Exchange Act of 1934 and we therefore file periodic reports, proxy statements and other information with the SEC relating to our business, financial statements and other matters. The reports, proxy statements and other information we file may be inspected and copied at prescribed rates at the SEC’s Public Reference Room, 100 F Street, N.E., Washington, D.C. 20549, on official business days during the hours of 10:00 A.M. to 3:00 P.M. You may obtain information on the operation of the SEC’s Public Reference Room by calling the SEC at 1-800-SEC-0330. The SEC also maintains an Internet site that contains reports, proxy statements and other information regarding issuers like us that file electronically with the SEC. The address of the SEC’s Internet site is www.sec.gov. For more information about us, please visit our website at www.corcept.com. You may also obtain a free copy of our annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K and amendments to those reports on the day the reports or amendments are filed with or furnished to the SEC by visiting our website at www.corcept.com. The information found on, or otherwise accessible through, our website, is not incorporated information, and does not form a part of, this Form 10-K.
18
Table of Contents
| ITEM 1A. | RISK FACTORS |
An investment in our common stock involves significant risks. You should carefully consider the risks described below and the other information in this Form 10-K, including our financial statements and related notes, before you decide to invest in our common stock. If any of the following risks or uncertainties actually occurs, our business, results of operations or financial condition could be materially harmed, the trading price of our common stock could decline and you could lose all or part of your investment. The risks and uncertainties described below are those that we currently believe may materially affect us; however, they may not be the only ones that we face. Additional risks and uncertainties of which we are unaware or currently deem immaterial may also become important factors that may harm our business. Except as required by law, we undertake no obligations to update any risk factors.
Risks Related to Our Business
We depend heavily on the success of our lead product candidate, CORLUX, currently being developed for the treatment of Cushing’s Syndrome and for the treatment of the psychotic features of psychotic depression. If we are unable to commercialize CORLUX for Cushing’s Syndrome or for psychotic depression, or experience significant delays in doing so, we may be unable to generate revenues and our stock price will likely decline.
We have invested a significant portion of our time and financial resources since our inception in the development of CORLUX for the treatment of Cushing’s Syndrome and the psychotic features of psychotic depression. We currently do not have any commercial products and we anticipate that for the foreseeable future our ability to generate meaningful revenues and achieve profitability will be solely dependent on the successful development, approval and commercialization of CORLUX for the treatment of Cushing’s Syndrome or for the psychotic features of psychotic depression. We are conducting a single Phase 3 trial in Cushing’s Syndrome and a Phase 3 clinical trial in psychotic depression. We have previously completed three Phase 3 clinical trials evaluating CORLUX for psychotic depression, all of which failed to achieve statistically significant results with regard to the primary or key secondary endpoints. Many factors could harm our efforts to develop and commercialize CORLUX, including:
| • | insufficient funding; |
| • | negative, inconclusive or otherwise unfavorable results from our pre-clinical or clinical development programs; |
| • | side effects that may be identified in the course of our clinical trials; |
| • | changes or delays in our clinical development program; |
| • | rapid technological change making CORLUX obsolete; |
| • | competition from companies with greater financial, technical and marketing resources than ours; |
| • | increases in the costs of our clinical trials; |
| • | an inability to obtain, or delay in obtaining, regulatory approval for the commercialization of CORLUX for the treatment of Cushing’s Syndrome or for the treatment of the psychotic features of psychotic depression; |
| • | an inability to manufacture CORLUX or the active ingredient in CORLUX in commercial quantities and at an acceptable cost; and |
| • | political concerns relating to other uses of mifepristone, or RU-486, that could limit the market acceptance of CORLUX. |
19
Table of Contents
Our clinical trials may not demonstrate that CORLUX is safe and effective. If our clinical program for CORLUX for the treatment of Cushing’s Syndrome, for the treatment of the psychotic features of psychotic depression or for any other indications does not demonstrate safety and efficacy, our business will be harmed.
To gain regulatory approval from the FDA to market CORLUX, our Phase 3 clinical trials must demonstrate the safety and efficacy of CORLUX for the particular indication. Our first three Phase 3 studies evaluating CORLUX for the treatment of the psychotic features of psychotic depression did not meet their primary or key secondary endpoints. In addition to the ongoing Phase 3 clinical trials of CORLUX for the treatment of Cushing’s Syndrome and for the treatment of the psychotic features of psychotic depression, we will need to conduct other studies in support of a potential NDA. Clinical development is a long, expensive and uncertain process and is subject to delays, and data obtained from clinical trials and supportive studies are susceptible to varying interpretations, which could delay, limit or prevent regulatory approval. While we obtained favorable results in our Phase 2 clinical trials in psychotic depression, these results were not replicated in a robust enough way in our completed Phase 3 clinical trials and are not sufficient to use by themselves as the pivotal clinical trials in an application for FDA approval of this indication. In addition, we cannot assure you that supportive studies and tests will produce favorable results.
The development plan for CORLUX, or any other compound, is not certain. If we decide to, or if the FDA or other regulatory agencies require us to pursue additional clinical trials or other studies, there may be a delay in the development of our compounds, which may have a negative impact on our business.
During the development of CORLUX, we have been engaged in dialogue with the FDA to determine an acceptable development plan which would enable the FDA to complete its review in a satisfactory manner. We anticipate continued dialogue with the FDA to define any additional data needed to complete an NDA.
We may decide, or the FDA or other regulatory authorities may require us, to pursue additional clinical, pre-clinical or manufacturing studies to satisfactorily complete our NDA for either Cushing’s Syndrome or psychotic depression. For example, the FDA may require us to perform a bioequivalance study comparing our recently reformulated CORLUX clinical trial materials to the materials used in our earlier clinical trials in psychotic depression. Additional trials or studies will require additional funding which is not assured. Also, it is possible that additional trials or studies that we decide are necessary or desirable will delay or prevent the completion of the development of CORLUX for treating Cushing’s Syndrome or the psychotic features of psychotic depression.
Many other factors could delay or result in termination of our clinical trials, including, but not limited to:
| • | availability of funding; |
| • | negative or inconclusive results; |
| • | slow patient enrollment; |
| • | patient noncompliance with the protocol; |
| • | adverse medical events or side effects among patients during the clinical trials; |
| • | negative or problematic FDA inspections of our clinical operations or our manufacturing operations; and |
| • | real or perceived lack of effectiveness or safety of CORLUX. |
Even after we conduct all of the clinical trials and supportive studies that we consider appropriate for an optimal NDA, we may not receive regulatory approval to market CORLUX.
20
Table of Contents
We will need additional capital in order to complete the development and commercialization of CORLUX and our other proprietary, selective GR-II antagonists, including CORT 108297. Additional capital may not be available to us at all or on favorable terms, which could adversely effect our business.
We may have to perform additional clinical trials prior to submission of an NDA for CORLUX for the treatment of Cushing’s Syndrome and for the treatment of the psychotic features of psychotic depression. We may need to raise additional funds to complete the development of CORLUX for the treatment of Cushing’s Syndrome and will need to raise additional funds to complete the development of CORLUX for the treatment of psychotic depression. In addition, we will need to raise additional funds to prepare for the commercialization of CORLUX for either of these indications, to develop a product for weight gain management associated with antipsychotic medications, and to continue and expand the development of our proprietary, selective GR-II antagonists, including CORT 108297.
We anticipate that our existing capital resources will be sufficient to fund our current operating plan into early 2011. However, our expectations are based on our currently planned clinical development and research programs for CORLUX and for certain of our proprietary, selective GR-II antagonists, including CORT 108297, which may change as a result of many factors, including:
| • | the costs, timing of site selection and enrollment of our clinical trials; |
| • | the results of our research efforts and clinical trials; |
| • | the need to perform additional clinical trials and other supportive studies; |
| • | the need to establish a second source for CORLUX tableting; |
| • | the timing of the approval by the FDA, if any, to market CORLUX for the treatment of Cushing’s Syndrome or for the treatment of the psychotic features of psychotic depression; |
| • | developments or disputes concerning patents or proprietary rights, including announcements of claims of infringement, interference or litigation against us or our licensors; |
| • | actual or anticipated fluctuations in our operating results; |
| • | changes in our growth rates; |
| • | changes in our research development plans for our proprietary, selective GR-II antagonists, including CORT 108297; |
| • | the timing of commercialization of CORLUX and future product candidates; and |
| • | changes in the reimbursement policies of third-party insurance companies or government agencies. |
Consequently, we may need additional funding sooner than anticipated. In addition, we may choose to raise additional capital due to market conditions or strategic considerations even if we believe we have sufficient funds for our current or future operating plans.
We cannot be certain that additional funding will be available on acceptable terms or at all. Even though we raised approximately $18.0 million in October 2009, through a private placement of our common stock and warrants, and generated an additional $875,000 between October 2009 and January 2010 under our Committed Equity Financing Facility (CEFF) with Kingsbridge Capital Limited (Kingsbridge), a private investment group, market and economic conditions may make it difficult for us to raise any additional capital. The sales of common stock and warrants during 2009 and the first quarter of 2010 have been dilutive to stockholders and any exercise of outstanding warrants and additional equity financing will cause further dilution to stockholders. Debt financing, if available, may involve restrictive covenants. If we obtain funds through collaborations with others, these arrangements may be on unfavorable terms or may require us to relinquish certain rights to our technologies or product candidates, including potentially our lead product candidate that we would otherwise
21
Table of Contents
seek to develop on our own. If adequate funds are not available, we may be required to delay, reduce the scope of or eliminate one or more of our research or development programs or we may be required to discontinue operations.
We have incurred losses since inception and anticipate that we will incur continued losses for at least the next few years.
We are a development stage company with no current source of product revenue. We have a limited history of operations and have focused primarily on clinical trials, and if the outcome of our clinical trials supports it, we plan to seek FDA regulatory clearance to market CORLUX for the treatment of Cushing’s Syndrome and for the treatment of the psychotic features of psychotic depression. Historically, we have funded our operations primarily from the sale of our equity securities. We have incurred losses in each year since our inception in 1998. As of December 31, 2009, we had an accumulated deficit of $150.2 million. We do not know when or if we will generate product revenue. Subject to our ability to raise additional funds, we expect our research and development expenses to increase in connection with the clinical trials and other development activities for CORLUX and for other product candidates. We expect to incur significant expenses related to the preparation for commercializing CORLUX and for the product’s launch, if the FDA approves our NDA. As a result, we expect that our losses will increase at least until CORLUX is launched and commercially available to patients. We are unable to predict the extent of any future losses or whether or when we will become profitable.
The Committed Equity Financing Facility (CEFF) that we entered into with Kingsbridge in March 2008 may not be available to us at certain times, may generate a lower level of funding than we anticipate, may require us to make additional “blackout” or other payments to Kingsbridge, and will result in dilution to our stockholders.
Kingsbridge will not be obligated to purchase shares under the CEFF unless certain conditions are met, which include a minimum price for our common stock, currently set at $1.50 per share and the effectiveness and continued effectiveness of the resale registration statements. The actual amount of funds that can be raised under this agreement will be dependent on the number of shares actually sold under the agreement and the market value of our stock during the pricing periods of each sale.
In June 2008, the SEC declared effective our registration statement with the SEC covering the resale of approximately 3.6 million of the shares issuable under the CEFF and the shares issuable upon the exercise of the warrant issued to Kingsbridge. This registration statement covers approximately 37% of the 9.6 million shares of our common stock issuable pursuant to the CEFF and all of the 330,000 shares of our common stock issuable upon exercise of the warrant issued to Kingsbridge. We intend to file an additional registration statement covering the resale of the remaining shares of our common stock issuable pursuant to the CEFF 60 days after Kingsbridge and its affiliates have resold substantially all of the securities covered by this initial registration statement; therefore, the timing of the submission of this subsequent registration statement is uncertain. This subsequent registration statement may be subject to review and comment by the Staff of the SEC, as well as consent by our independent registered accounting firm. We cannot assure you that these registration statements will be declared effective or, if declared effective, that they will remain continuously effective thereafter.
In addition, Kingsbridge is permitted to terminate the CEFF if it determines that a material and adverse event has occurred affecting our business, operations, properties or financial condition and if such condition continues for a period of 10 days from the date Kingsbridge provides us notice of such material and adverse event. If we are unable to access funds through the CEFF, or if the CEFF is terminated by Kingsbridge, we may be unable to access alternative capital on favorable terms or at all.
We are entitled in certain circumstances, to deliver a blackout notice to Kingsbridge to suspend the use of the resale registration statement and prohibit Kingsbridge from selling shares thereunder. If we deliver a blackout notice in the 15 trading days following the settlement of a draw down, or if the resale registration statement is not
22
Table of Contents
effective in circumstances not permitted by our agreement with Kingsbridge, then we may be required to make a payment to Kingsbridge, or issue Kingsbridge additional shares in lieu of the payment. If the trading price of our common stock declines during a suspension of the resale registration statement, the blackout or other payment could be significant.
Any shares that we may issue to Kingsbridge under the CEFF will have a dilutive effect on the holdings of our current stockholders, and may result in downward pressure on the price of our common stock. For each draw down under the CEFF, we will issue shares to Kingsbridge at a discount of up to 10% from the volume weighted average price of our common stock. If we draw down amounts under the CEFF when our share price is decreasing, we will need to issue more shares to raise the same amount than if our stock price was higher. Issuances in the face of a declining share price will have an even greater dilutive effect than if our share price were stable or increasing, and may further decrease our share price.
We may not be able to pursue all of our product research and development opportunities if we are unable to secure adequate funding for these programs.
The costs required to start or continue many of the programs that our intellectual property allow us to consider for further development are collectively greater than the funds currently available to us. For example, we have successfully discovered three series of compounds that are specific GR-II antagonists but, unlike CORLUX, do not appear to block the progesterone receptor. Further development of these proprietary compounds, including CORT 108297, or any further development stemming from our method of use patents may be delayed or cancelled if we determine that such development may jeopardize our ability to complete the clinical development of CORLUX for the treatment of Cushing’s Syndrome or psychotic depression.
Global economic conditions could adversely affect our liquidity and financial condition.
Global economic and market conditions were extremely unstable in 2009, with significantly tighter credit conditions. Any economic recovery during 2010 could be fragile and uneven. Continued concerns about the systemic impact of potential wide-spread and long-term recession, geopolitical issues, the availability and cost of credit, global financial markets and corporate, consumer and governmental debt levels have contributed to market volatility and diminished expectations for the U.S., European and other economies.
As a result of these conditions, the cost and availability of capital have been and may continue to be adversely affected. Concern about the stability of the markets generally, and the strength of counterparties specifically, has led many lenders and institutional investors to reduce, and in some cases, cease, to provide credit to businesses. Continued turbulence in the global markets and economies may adversely affect our liquidity and financial condition. If these market and economic conditions continue, they may limit our ability to fund our clinical trials and drug development programs.
In addition, our access to funds under our CEFF or any credit facility into which we may enter depends on the ability of the counterparties to such facilities to meet their funding commitments to us. We cannot assure you that continuing long-term disruptions in the global economy and tighter credit conditions will not have an adverse effect on such counterparties. If we do not have sufficient cash flow to continue operating our business and are unable to borrow funds, access our CEFF or raise equity capital, we may need to find alternative ways to increase our liquidity. Such alternatives may include, without limitation, curtailing clinical or drug development activity, which would have an adverse affect on our business and results of operation.
23
Table of Contents
We depend on clinical investigators and clinical sites to enroll patients in our clinical trials and other third parties to manage the trials and to perform related data collection and analysis, and, as a result, we may face costs and delays outside of our control.
We rely on clinical investigators and clinical sites to enroll patients and other third parties to manage our trials and to perform related data collection and analysis. However, we may not be able to control the timing of identification and selection of appropriate sites for our planned trials and the amount and timing of resources that the clinical sites that conduct the clinical testing may devote to our clinical trials. If our clinical investigators and clinical sites fail to enroll a sufficient number of patients in our clinical trials or fail to enroll them on our planned schedules, we will be unable to complete our trials or to complete them as planned, which could delay or prevent us from completing the clinical development of CORLUX or other development programs.
We have an agreement with a contract research organization (CRO) that is conducting our ongoing Phase 3 trial evaluating CORLUX for the treatment of the psychotic features of psychotic depression, Study 14, to supervise and monitor clinical site performance and to perform investigator supervision, data collection and analysis for this trial. We may not be able to maintain relationships with this or other CROs or with the clinical investigators and the clinical sites through the completion of all trial activities without delays in anticipated timing of trial activities or excessive expenditures. Our agreements place substantial responsibilities on these parties, which could result in excessive expenditures for our clinical trials if these parties fail to perform as expected. For example, if any of our clinical trial sites fail to comply with FDA-approved good clinical practices, we may be unable to use the data gathered at those sites. If these CROs, clinical investigators, clinical sites or other third parties do not carry out their contractual duties or obligations or fail to meet expected deadlines, or if the quality or accuracy of the clinical data they obtain is compromised due to their failure to adhere to our clinical protocols or for other reasons, we may be unable to obtain regulatory approval for, or successfully commercialize, CORLUX.
The conduct of any future clinical trials will likely also be conducted through the use of CROs and clinical research sites. The conduct, timing and cost of these trials will be subject to the same kinds of risks as discussed above.
Our use of MedAvante to provide centralized psychiatric rating services in Study 14, our ongoing clinical trial evaluating CORLUX for the psychotic features of psychotic depression, may not result in any improvement in the accuracy and consistency of the psychiatric assessments and may continue to slow the pace of enrollment in Study 14.
In connection with our ongoing Phase 3 trial evaluating CORLUX for the psychotic features of psychotic depression, Study 14, we engaged MedAvante to provide centralized psychiatric rating services. MedAvante is providing centralized psychometric assessments via high resolution video-conferencing. The use of MedAvante’s centralized rating services is expected to increase the accuracy and consistency of the psychiatric assessments.
MedAvante has provided similar centralized rating services to companies conducting clinical studies in various psychiatric disorders. However, they have not previously provided centralized rating services to any study in patients with psychotic depression. Although Corcept and MedAvante conducted a small pilot evaluation in patients with psychotic depression to assess patient receptivity, we cannot be certain that centralized rating will be successful in the patients enrolled in our study.
If patients are uncomfortable or unwilling to participate in the centralized rating process or if MedAvante is unable to provide services in a satisfactory manner over the course of the trial, we may not see any improvement in the accuracy or reliability of the psychiatric assessments. Such a result might diminish the likelihood of a successful trial or a definitive demonstration of the efficacy of CORLUX in treating the psychotic features of psychotic depression.
24
Table of Contents
During screening for Study 14, we have seen a higher than anticipated incidence of potential patients who do not meet appropriate criteria for entrance into the trial for diagnostic and other clinical reasons. We believe that this is the result of improved accuracy in the screening process resulting from the use of the MedAvante centralized rating services as an additional step in the selection of patients appropriate for inclusion in the study. In addition, in March 2009, we announced that, in order to lower variable and fixed expenses and to conserve financial resources, we scaled back our planned rate of spending on this trial and extended the timeline for its completion. We are currently using eight clinical sites in order to conserve capital. A continued lower enrollment rate could result in delays in the timing of anticipated completion of the trial and increased study costs over the longer term.
If we are unable to obtain or maintain regulatory approval, we will be limited in our ability to commercialize our product candidates, including CORLUX, and our business will be harmed.
The research, testing, manufacturing, selling and marketing of product candidates are subject to extensive regulation by the FDA and other regulatory authorities in the United States and other countries, in which regulations differ from country to country. Obtaining and maintaining regulatory approval typically is an uncertain process, is costly and takes many years. In addition, failure to comply with the FDA and other applicable foreign and U.S. regulatory requirements may subject us to administrative or judicially imposed sanctions. These include warning letters, civil and criminal penalties, injunctions, product seizure or detention, product recalls, total or partial suspension of production, and refusal to approve pending NDAs, or supplements to approved NDAs.
Regulatory approval of an NDA or NDA supplement is never guaranteed. Despite the time, resources and effort expended, failure can occur at any stage. The FDA has substantial discretion in the approval process for human medicines. The FDA can deny, delay or limit approval of a product candidate for many reasons including:
| • | the FDA may not find that the candidate is safe; |
| • | the FDA may not find data from the clinical or preclinical testing to be sufficient; or |
| • | the FDA may not approve our or our third party manufacturers’ processes or facilities. |
Future governmental action or changes in FDA policy or personnel may also result in delays or rejection of an NDA in the United States. In addition, because the only currently FDA-approved use of mifepristone is the termination of pregnancy, we expect that the label for CORLUX will include some limitations, including a warning that it should not be used by pregnant women or women seeking to become pregnant.
If we receive regulatory approval for our product candidates, including CORLUX, we will also be subject to ongoing FDA obligations and continued regulatory oversight and review, such as continued safety reporting requirements; and we may also be subject to additional FDA post-marketing obligations. If we are not able to maintain regulatory compliance, we may not be permitted to market our product candidates.
Any regulatory approvals that we receive for our product candidates may also be subject to limitations on the indicated uses for which the medicine may be marketed or contain requirements for potentially costly post-marketing follow-up studies. In addition, if the FDA approves any of our product candidates, the labeling, packaging, adverse event reporting, storage, advertising, promotion and record-keeping for the medicine will be subject to extensive regulatory requirements. The subsequent discovery of previously unknown problems with the medicine, including adverse events of unanticipated severity or frequency, may result in restrictions on the marketing of the medicine, and could include withdrawal of the medicine from the market.
25
Table of Contents
Failure to obtain regulatory approval in foreign jurisdictions will prevent us from commercializing our product candidates abroad.
We intend to commercialize our product candidates in international markets with the help of one or more partners. Outside the United States, we can commercialize a product only if we receive a marketing authorization and, in some cases, pricing approval, from the appropriate regulatory authorities. This foreign regulatory approval process includes all of the risks associated with the FDA approval process, and, in some cases, additional risks. The approval procedure varies among countries and can involve additional testing, and the time required to obtain approval may differ from that required to obtain FDA approval. We have not taken any actions to obtain foreign approvals. We may not develop our product candidates in the clinic in order to obtain foreign regulatory approvals on a timely basis, if at all.
Approval by the FDA does not ensure approval by regulatory authorities in other countries, and approval by one foreign regulatory authority does not ensure approval by regulatory authorities in other foreign countries or by the FDA. We may not be able to file for regulatory approvals and may not receive necessary approvals to commercialize our product candidates in any market.
The Orphan Drug Designation for CORLUX for the treatment of endogenous Cushing’s Syndrome may not provide protection from competition and other benefits as anticipated.
In July 2007, we received Orphan Drug Designation from the FDA for CORLUX for the treatment of endogenous Cushing’s Syndrome. Although we have received Orphan Drug Designation from the FDA, we cannot be assured that we will recognize the potential benefits of this designation.
For example, we are aware that Laboratoire HRA Pharma has received an Orphan Drug Designation in the United States and Europe for the use of mifepristone to treat a subtype of Cushing’s Syndrome and has begun a Phase 2 clinical trial in Europe and the United States for this indication. We are also aware that Exelgyn Laboratories recently received a recommendation for Orphan Drug Designation for Cushing’s Syndrome in Europe, but they have stated that they have not yet conducted any clinical trials.
If another drug is approved for this indication before CORLUX, we may not garner the seven years of marketing exclusivity from the date of drug approval and other benefits that we anticipate.
The “fast track” designation for the development program of CORLUX for the treatment of the psychotic features of psychotic depression may not lead to a faster development or regulatory review or approval process.
If a human medicine is intended for the treatment of a serious or life-threatening condition and the medicine demonstrates the potential to address unmet medical needs for this condition, the sponsor of an IND may apply for FDA “fast track” designation for a particular indication. Marketing applications submitted by sponsors of product candidates in fast track development may qualify for expedited FDA review under the policies and procedures offered by the FDA, but the fast track designation does not assure any such qualification. Although we have obtained a fast track designation from the FDA for CORLUX for the treatment of the psychotic features of psychotic depression, we may not experience a faster development process, review or approval compared to applications considered for approval under conventional FDA procedures. In addition, the FDA may withdraw our fast track designation at any time. If we lose our fast track designation, the approval process may be delayed. In addition, our fast track designation does not guarantee that we will qualify for or be able to take advantage of the expedited review procedures and does not increase the likelihood that CORLUX will receive regulatory approval for the treatment of the psychotic features of psychotic depression.
26
Table of Contents
Even if we receive approval for the marketing and sale of CORLUX for the treatment of Cushing’s Syndrome and / or psychotic depression, CORLUX may never be accepted as a treatment for the approved indications, which would adversely effect our financial results.
Many factors may affect the market acceptance and commercial success of CORLUX for the treatment of Cushing’s Syndrome and / or the psychotic features of psychotic depression or for any other approved indication.
Even if the FDA approves CORLUX for the treatment of Cushing’s Syndrome, for the treatment of the psychotic features of psychotic depression, or for any other indication, physicians may not adopt CORLUX. Physicians will recommend the use of CORLUX only if they determine, based on experience, clinical data, side effect profiles and other factors, that it is preferable to other products or treatments then in use. Acceptance of CORLUX among influential practitioners may be essential for market acceptance of CORLUX.
Other factors that may affect the market acceptance and commercial success of CORLUX include:
| • | the effectiveness of CORLUX, including any side effects, as compared to alternative treatment methods; |
| • | the product labeling or product insert required by the FDA for CORLUX; |
| • | the cost-effectiveness of CORLUX and the availability of third-party insurance coverage and reimbursement, in particular from government payors such as Medicare and Medicaid, for patients using CORLUX; |
| • | the timing of market entry of CORLUX relative to competitive products; |
| • | the intentional restriction of distribution of CORLUX to physicians treating the target patient population; |
| • | the extent and success of our sales and marketing efforts; |
| • | the rate of adoption of CORLUX by physicians and by target patient population; and |
| • | negative publicity concerning CORLUX, RU-486 or mifepristone. |
The failure of CORLUX to achieve market acceptance would prevent us from generating meaningful product revenue.
Public perception of the active ingredient in CORLUX, mifepristone or RU-486, may limit our ability to market and sell CORLUX.
The active ingredient in CORLUX, mifepristone (RU-486) is used to terminate pregnancy. As a result, mifepristone has been and continues to be the subject of considerable ethical and political debate in the United States and elsewhere. Public perception of mifepristone may limit our ability to engage alternative manufacturers and may limit the commercial acceptance of CORLUX by patients and physicians. Even though we intend to create measures to minimize the likelihood of the prescribing of CORLUX to a pregnant woman, physicians may decline to prescribe CORLUX to a woman simply to avoid altogether any risk of unintentionally terminating a pregnancy. We intend to create measures for controlling the distribution of CORLUX to reduce the potential for diversion. However, controlled distribution may negatively impact sales of CORLUX.
27
Table of Contents
We have no manufacturing capabilities and we currently depend on third parties to manufacture the active ingredient and the tablets for CORLUX. The tablet manufacturer is a single source supplier. If these suppliers are unable to continue manufacturing CORLUX and we are unable to contract quickly with alternative sources, our business will be harmed.
We currently have no experience in, and we do not own facilities for, nor do we plan to develop facilities for, manufacturing any products. We have agreements with two manufacturers of the active pharmaceutical ingredient (API) of mifepristone and an agreement with a tablet manufacturer for development quantities of CORLUX. The tablet manufacturer is a single source supplier to us. Our current arrangements with these manufacturers are terminable by either party at any time. Although we anticipate engaging our current tablet supplier to produce commercial quantities of CORLUX, we cannot guarantee that we will enter into an agreement with them on terms acceptable to us. If we are unable, for whatever reason, to obtain the active pharmaceutical ingredient or CORLUX tablets from our contract manufacturers, we may not be able to manufacture our required quantities or identify alternate manufacturers of mifepristone or CORLUX tablets in a timely manner or on reasonable terms, if at all.
If our third-party manufacturers of CORLUX fail to comply with FDA regulations or otherwise fail to meet our requirements, our product development and commercialization efforts may be delayed.
We depend on third party manufacturers to supply the active pharmaceutical ingredient in CORLUX and to manufacture CORLUX tablets. These suppliers and manufacturers must comply with the FDA’s current Good Manufacturing Practices (cGMP) regulations and guidelines. Our suppliers and manufacturers may encounter difficulties in achieving quality control and quality assurance and may experience shortages of qualified personnel. Their failure to follow cGMP or other regulatory requirements and to document their compliance with cGMP may lead to significant delays in the availability of products for commercial use or clinical study or the termination or hold on a clinical study, or may delay or prevent filing or approval of marketing applications for CORLUX.
Failure of our third party suppliers and manufacturers or us to comply with applicable regulations could result in sanctions being imposed on us, including fines, injunctions, civil penalties, failure of regulatory authorities to grant marketing approval of our product candidates, delays, suspension or withdrawal of approvals, license revocation, seizures or recalls of products, operating restrictions and criminal prosecutions, any of which could harm our business. If the operations of any current or future supplier or manufacturer were to become unavailable for any reason, commercialization of CORLUX could be delayed and our revenue from product sales could be reduced.
We may use a different third-party manufacturer to produce commercial quantities of CORLUX than we are using in our clinical trials. The FDA may require us to conduct a study to demonstrate that the tablets used in our clinical trials are equivalent to the final commercial product. If we are unable to establish that the tablets are equivalent or if the FDA disagrees with the results of our study, commercial launch of CORLUX would be delayed.
If we or others identify side effects after our product candidates are on the market, we may be required to perform lengthy additional clinical trials, change the labeling of our future products or withdraw our future products from the market, any of which would hinder or preclude our ability to generate revenues.
If we or others identify side effects after any of our product candidates are on the market:
| • | regulatory authorities may withdraw their approvals; |
| • | we may be required to reformulate our future products, conduct additional clinical trials, make changes in labeling of such products or implement changes to or obtain re-approvals of our manufacturing facilities; |
28
Table of Contents
| • | we may experience a significant drop in the sales of the affected products; |
| • | our reputation in the marketplace may suffer; and |
| • | we may become the target of lawsuits, including class action lawsuits. |
Any of these events could harm or prevent sales of the affected products or could increase the costs and expenses of commercializing and marketing these product candidates.
If CORLUX or future product candidates conflict with the patents of others or if we become involved in other intellectual property disputes, we may have to engage in costly litigation or obtain a license and we may be unable to commercialize our product candidates.
Our success depends in part on our ability to obtain and maintain adequate patent protection for the use of CORLUX for the treatment of the psychotic features of psychotic depression and other potential uses of GR-II antagonists. If we do not adequately protect our intellectual property, competitors may be able to use our intellectual property and erode our competitive advantage.
To date, we own seven issued U.S. method of use patents and have exclusively licensed three issued U.S. method of use patents, with a number of corresponding foreign patents or patent applications. We have eight U.S. method of use patent applications for GR-II antagonists. We own two composition of matter patents and have one composition of matter patent application covering specific GR-II antagonists pending in the U.S. We have applied, and will continue to apply, for patents covering our product candidates as we deem appropriate.
We have exclusively licensed three issued U.S. patents from Stanford University for the use of GR-II antagonists in the treatment of psychotic major depression, which is commonly referred to as psychotic depression, cocaine-induced psychosis and early dementia, including early Alzheimer’s disease. We bear the costs of protecting and defending the rights to these patents. In order to maintain the exclusive license to these patents until their expiration, we are obligated to make milestone and royalty payments to Stanford University. We are currently in compliance with our obligations under this agreement. If we become noncompliant, we may lose the right to commercialize CORLUX for the treatment of psychotic depression, cocaine-induced psychosis and early dementia and our business would be materially harmed. In addition, if Stanford University were to terminate our CORLUX license due to breach of the license on our part, we would not be able to commercialize CORLUX for the treatment of the psychotic features of psychotic depression, cocaine-induced psychosis or early dementia.
Our patent applications and patents licensed or issued to us may be challenged by third parties and our patent applications may not result in issued patents. For example, in 2004, Akzo Nobel, which was subsequently acquired by Schering Plough, filed an observation challenging the claims of our exclusively licensed European patent application with claims directed to psychotic depression. In 2005, we filed a rebuttal to the EPO that responded to the points raised by Akzo Nobel. In February 2006, the EPO allowed our patent application and in July 2006, this patent was issued. In April 2007 we received notification that there will be no opposition proceedings in Europe in regards to this patent.
Our presently pending and future patent applications may not issue as patents, and any patent issued to us may be challenged, invalidated, held unenforceable or circumvented. For example, the arguments presented by Akzo Nobel could be raised in the United States either before the U.S. Patent and Trademark Office or in a court of law. Furthermore, the claims in patents which have been issued to us, or which may be issued to us in the future, may not be sufficiently broad to prevent third parties from producing competing products. In addition, the laws of various foreign countries in which we compete may not protect our intellectual property to the same extent as do the laws of the United States. If we fail to obtain adequate patent protection for our proprietary technology, our competitors may produce competing products based on our technology, which would impair our ability to compete.
29
Table of Contents
If a third party were successful in asserting an infringement claim against us, we could be forced to pay damages and prevented from developing, manufacturing or marketing our potential products. We do not have liability insurance for patent infringements. A third party could require us to obtain a license to continue to use their intellectual property, and we may not be able to do so on commercially acceptable terms, or at all. We believe that significant litigation will continue in our industry regarding patent and other intellectual property rights. If we become involved in litigation, it could consume a substantial portion of our resources. Regardless of the merit of any particular claim, defending a lawsuit takes significant time, is expensive and diverts management’s attention from other business.
If we are unable to protect our trade secrets and proprietary information, our ability to compete in the market could be diminished.
In addition to patents, we rely on a combination of confidentiality, nondisclosure and other contractual provisions, laws protecting trade secrets and security measures to protect our trade secrets and proprietary information. Nevertheless, these measures may not adequately protect our trade secrets or other proprietary information. If they do not adequately protect our rights, third parties could use our proprietary information, which could diminish our ability to compete in the market. In addition, employees, consultants and others who participate in the development of our product candidates may breach their agreements with us regarding our trade secrets and other proprietary information, and we may not have adequate remedies for the breach. We also realize that our trade secrets may become known through means not currently foreseen. Notwithstanding our efforts to protect our trade secrets and proprietary information, our competitors may independently develop similar or alternative products that are equal or superior to our product candidates without infringing on any of our proprietary information or trade secrets.
Our licensed patent covering the use of mifepristone to treat psychotic depression is a method of use patent rather than a composition of matter patent, which increases the risk that physicians will prescribe another manufacturer’s mifepristone for the treatment of Cushing’s Syndrome or psychotic depression rather than CORLUX or patients may acquire mifepristone from other sources, such as the internet or black market.
We have an exclusive license from Stanford University to a patent covering the use of GR-II antagonists, including mifepristone, for the treatment of psychotic depression. A method of use patent covers only a specified use of a particular compound, not a particular composition of matter. Ten of our 12 U.S. issued patents and eight of our nine U.S. patent applications are method of use patents. Because none of our issued patents covers the composition of mifepristone, we cannot prevent others from commercializing mifepristone in indications not covered by our method of use patents. If others receive approval to manufacture and market mifepristone or any other GR-II antagonist, physicians could prescribe mifepristone or any other GR-II antagonist for patients with psychotic depression instead of CORLUX. Although any such “off-label” use would violate our licensed patent, effectively monitoring compliance with our licensed patent may be difficult and costly. In addition, if others develop a treatment for psychotic depression that works through a mechanism which does not involve the GR-II receptor, physicians could prescribe that treatment instead of CORLUX.
In addition, we cannot be assured that patients will not obtain mifepristone from other sources. As with other pharmaceutical products, patients may be able to purchase mifepristone through the internet or black market. Mifepristone is also sold in the United States by Danco Laboratories for the termination of early pregnancy. While distribution is limited to a single dose provided in the physician’s office and covered by other restrictions, we cannot be certain that Cushing’s Syndrome patients may not be able to obtain mifepristone from this source.
30
Table of Contents
The composition of matter patents on our families of novel selective glucocorticoid antagonists may not be issued and we would not be able to prevent competition from others.
We have filed composition of matter patent claims on three families of novel selective glucocorticoid antagonists but not all of these have been issued. Applications for all of the three families have been allowed in Europe. In the United States, applications for two of the three families have been allowed. Examination has not yet begun in the United States on our third novel selective GR-II family. We cannot be certain that these patents will be issued to us. If these patents are not issued we may not be able to prevent others from developing competing compounds. The competing products could be prescribed by physicians instead of those developed by us.
Our efforts to discover, develop and commercialize new product candidates beyond CORLUX are at a very early stage. If we fail to identify and develop additional uses for GR-II antagonists, we may be unable to market additional products.
To develop additional potential sources of revenue, we believe that we must identify and develop additional product candidates. We own or have exclusively licensed issued U.S. patents covering the use of GR-II antagonists to treat psychotic depression, mild cognitive impairment, weight gain due to treatment with antipsychotic medication, stress disorders, early dementia, delirium, gastroesophageal reflux disease, Down’s Syndrome and psychosis associated with cocaine addiction, and to increase the therapeutic response to ECT. In addition, we have eight U.S. method of use patent applications covering GR-II antagonists for the treatment of a number of other metabolic and psychiatric disorders and three U.S. composition of matter patent applications covering specific GR-II antagonists. We have also filed patent applications in all of the major international markets.
We may not develop or continue to develop product candidates for any of the indications or compounds covered by our patents and patent applications. Typically, there is a high rate of attrition for product candidates in preclinical and clinical trials, so our product development efforts may not lead to commercially viable products. The use of GR-II antagonists may not be effective to treat these conditions or any other indications. In addition, we could discover that the use of GR-II antagonists in these patient populations has unacceptable side effects or is otherwise not safe.
We may elect to enter into collaboration arrangements with respect to one or more of our product candidates. If we do enter into such an arrangement, we would be dependent on a collaborative partner for the success of the product candidates developed under the arrangement. Any future collaborative partner may fail to successfully develop or commercialize a product candidate under a collaborative arrangement.
We only have significant clinical experience with CORLUX and we may determine that CORLUX is not desirable for uses other than for the treatment of Cushing’s Syndrome or the treatment of the psychotic features of psychotic depression. In that event, we would have to identify and may need to secure rights to a different GR-II antagonist. For example, we do not intend to develop CORLUX for mitigation of the weight gain associated with the use of Zyprexa, Risperdal or other atypical antipsychotics, even though we have reported positive results in the proof of concept studies described elsewhere in this Annual Report on Form 10-K. We are pursuing other GR-II antagonists for this use and may pursue additional compounds. The compounds developed pursuant to our preclinical and discovery research programs, including CORT 108297, may fail to generate commercially viable product candidates in spite of the resources we may dedicate to the program. Even if product candidates are identified, we may abandon further development efforts before we reach clinical trials or after expending significant expense and time conducting clinical trials due to financial constraints, concerns over safety, efficacy of the product candidates or for other reasons. Moreover, governmental authorities may enact new legislation or regulations that could limit or restrict our development efforts. If we are unable to successfully discover and commercialize new uses for GR-II antagonists, we may be unable to generate sufficient revenue to support our operations.
31
Table of Contents
We may have substantial exposure to product liability claims and may not have adequate insurance to cover those claims.
We may be subject to product liability or other claims based on allegations that the use of our products has resulted in adverse effects or that our product candidates are not effective, whether by participants in our clinical trials for CORLUX or other product candidates, or by patients using our future products. A product liability claim may damage our reputation by raising questions about our product candidates’ safety or efficacy and could limit our ability to sell a product by preventing or interfering with product commercialization. In some cases, less common adverse effects of a pharmaceutical product are not known until long after the FDA approves the product for marketing. The active ingredient in CORLUX is used to terminate pregnancy. Therefore, necessary and strict precautions must be taken by clinicians using the medicine in our clinical trials and, if approved by the FDA, physicians prescribing the medicine to women with childbearing potential, to insure that the medicine is not administered to pregnant women. The failure to observe these precautions could result in significant product claims.
We have only limited product liability insurance coverage, with limits that we believe to be customary for a development stage company. We intend to expand our product liability insurance coverage to any product candidates for which we obtain marketing approval. However, this insurance may be prohibitively expensive or may not fully cover our potential liabilities. Our inability to obtain adequate insurance coverage at an acceptable cost could prevent or inhibit the commercialization of our product candidates. Defending a lawsuit could be costly and significantly divert management’s attention from conducting our business. If a third party successfully sues us for any injury caused by our product candidates, our liability could exceed our total assets.
If CORLUX is approved and we are unable to obtain acceptable prices or adequate coverage and reimbursement for it from third-party payors, we will be unable to generate significant revenues.
There is significant uncertainty related to the availability of third-party insurance coverage and reimbursement for newly approved medications. The commercial success of our potential medications in both domestic and international markets is dependent on whether third-party coverage and reimbursement is available for them. Government payors, including Medicare and Medicaid, health maintenance organizations and other third-party payors are increasingly attempting to contain healthcare costs by limiting both coverage and the level of reimbursement of new medicines, and, as a result, they may not cover or provide adequate payment for our medications. The continuing efforts of government and other third-party payors to contain or reduce the costs of health care may limit our revenues. Our dependence on the commercial success of CORLUX alone makes us particularly susceptible to any cost containment or reduction efforts. Accordingly, even if CORLUX or future product candidates are approved for commercial sale, unless government and other third-party payors provide adequate coverage and reimbursement for our future products, physicians may not prescribe them. We intend to sell CORLUX directly to hospitals if we receive FDA approval. As a result, we will need to obtain approval from hospital formularies to receive wide-spread third-party coverage and reimbursement. If we fail to obtain that approval, we will be unable to generate significant revenues.
In some foreign markets, pricing and profitability of prescription pharmaceuticals are subject to government control. In the United States, we expect that there will continue to be federal and state proposals for similar controls. Also, the trends toward managed health care in the United States and proposed legislation intended to reduce the cost of government insurance programs could significantly influence the purchase of health care services and products and may result in lower prices for our future products or the exclusion of such products from reimbursement programs.
32
Table of Contents
We may face competition from other companies who attempt to develop mifepristone for the treatment of Cushing’s Syndrome, which could limit our future revenues from the commercialization of CORLUX for the treatment of that disorder and which could have a negative impact on future revenues from the commercialization of CORLUX for any indication.
As discussed above in the risk related to Orphan Drug Designation, we are aware that Laboratoire HRA Pharma has begun a Phase II clinical trial in Europe and the United States evaluating the use of mifepristone to treat a subtype of Cushing’s Syndrome. We are also aware that Novartis is developing a somatostatin analogue that is in Phase 3 trials for various endocrine disorders, including Cushing’s disease, which is a subset of the patients with Cushing’s Syndrome. If a product for treatment of Cushing’s Syndrome is approved for commercialization before CORLUX, our potential future revenue could be reduced.
We face competition from companies with substantial financial, technical and marketing resources, which could limit our future revenues from the commercialization of CORLUX for the treatment of psychotic depression or for other indications.
If approved for commercial use, CORLUX as a treatment for psychotic depression will compete with established treatments, including ECT and combination medicinal therapy.
Combination medicinal therapy consists of the use of antipsychotic and antidepressant medicines, not currently approved for the treatment of psychotic depression. The antipsychotics are prescribed for off-label use by physicians to treat the psychotic features of psychotic depression, which is the clinical target of CORLUX. Antipsychotics include Bristol-Myers Squibb’s Abilify, Novartis’ Clozaril, Pfizer’s Geodon and Navane, Ortho-McNeil’s Haldol, Janssen Pharmaceutica’s Risperdal, AstraZeneca’s Seroquel, GlaxoSmithKline’s Stelazine and Thorazine, Mylan’s Mellaril, Schering Corporation’s Trilafon and Eli Lilly’s Zyprexa. CORLUX may not compete effectively with these established treatments. We are aware of one clinical trial conducted by Organon, for a new chemical entity for the treatment of psychotic depression. Organon was the pharmaceutical division of Akzo Nobel, which was purchased by Schering Plough. Organon’s new chemical entity is a GR-II antagonist, the commercial use of which would be covered by our patent. As discussed above, in 2004, Akzo Nobel filed an observation in our exclusively licensed European patent application with claims directed to psychotic depression, in which it challenged the claims of that patent application. In 2005, we filed a rebuttal to the EPO that responded to the points raised by Akzo Nobel. In February 2006, the EPO allowed our patent application. In July 2006, the patent was issued. As of the time of filing of this report, we are not aware of any other public disclosures by any company, regarding the development of new products to treat psychotic depression.
Our present and potential competitors include major pharmaceutical companies, as well as specialized pharmaceutical firms, universities and public and private research institutions. Moreover, we expect competition to intensify as technical advances are made. These competitors, either alone or with collaborative parties, may succeed with the development and commercialization of medicinal products that are superior to and more cost- effective than CORLUX. Many of our competitors and related private and public research and academic institutions have greater experience, more financial resources and larger research and development staffs than we do. In addition, many of these competitors, either alone or together with their collaborative partners, have significantly greater experience than we do in developing human medicines, obtaining regulatory approvals, manufacturing and commercializing products.
Accordingly, CORLUX may not be an effective competitor against established treatments and our present or potential competitors may succeed in developing medicinal products that are superior to CORLUX or render CORLUX obsolete or non-competitive. If we are unable to establish CORLUX as a superior and cost-effective treatment for psychotic depression, or any future use, we may be unable to generate the revenues necessary to support our business.
33
Table of Contents
Rapid technological change could make our product candidates obsolete.
Pharmaceutical technologies have undergone rapid and significant change and we expect that they will continue to do so. Our future will depend in large part on our ability to maintain a competitive position with respect to these technologies. Any products and processes that we develop may become obsolete or uneconomical before we recover any or all expenses incurred in connection with their development. Rapid technological change could make our product candidates obsolete or uneconomical, which could materially adversely affect our business, financial condition and results of operations.
We have no sales staff and limited marketing activities and will need to develop sales and marketing capabilities to successfully commercialize CORLUX and any future uses of GR-II antagonists.
Our employees have limited experience in marketing or selling pharmaceutical products and we currently have no sales staff and limited marketing activities. To achieve commercial success for any approved product, we must either develop a sales and marketing force or enter into arrangements with others to market and sell our future products. We currently plan to establish small, specialty sales forces to market and sell CORLUX in the United States for the treatment of Cushing’s Syndrome and for the treatment of the psychotic features of psychotic depression, as each indication is approved for marketing by the FDA. However, our sales and marketing efforts may not be successful or cost-effective. In the event that the commercial launch of CORLUX is delayed due to FDA requirements or other reasons, we may establish a sales and marketing force too early relative to the launch of CORLUX. This may be expensive, and our investment would be lost if the sales and marketing force could not be retained. If our efforts to develop a sales and marketing force are not successful, cost-effective and timely, we may not achieve profitability.
We may need to increase the size of our organization, and we may experience difficulties in managing growth.
As we expand our research and development efforts and develop a sales and marketing organization, we expect to experience growth, which may strain our operations, product development and other managerial and operating resources. Future growth will impose significant added responsibilities on members of management, including the need to identify, recruit, maintain and integrate additional employees. To date, we have relied on a small management team, including a number of part-time contributors. Our future financial performance and our ability to compete effectively will depend, in part, on our ability to manage any future growth effectively.
To that end, we must be able to:
| • | manage our research and development efforts effectively; |
| • | manage our clinical trials effectively; |
| • | integrate additional management, clinical development, administrative and sales and marketing personnel; |
| • | expand the size and composition of our management team; |
| • | develop our administrative, accounting and management information systems and controls; and |
| • | hire and train additional qualified personnel. |
We may not be able to accomplish these tasks, and our failure to accomplish any of them could harm our business.
34
Table of Contents
If we lose our key personnel or are unable to attract and retain additional skilled personnel, we may be unable to pursue our product development and commercialization efforts.
We depend substantially on the principal members of our management and scientific staff, including Joseph K. Belanoff, M.D., our Chief Executive Officer, and Robert L. Roe, M.D., our President. We do not have agreements with any of our executive officers that provide for their continued employment with us or employment insurance covering any of our key personnel. Any officer or employee can terminate his or her relationship with us at any time and work for one of our competitors. The loss of these key individuals could result in competitive harm because we could experience delays in our product research, development and commercialization efforts without their expertise.
Our ability to operate successfully and manage our potential future growth depends significantly upon retaining key research, technical, sales, marketing, managerial and financial personnel, and attracting and retaining additional highly qualified personnel in these areas. We face intense competition for such personnel from numerous companies, as well as universities and nonprofit research organizations in the highly competitive northern California business area. Although we believe that we have been successful in attracting and retaining qualified personnel to date, we may not be able to attract and retain sufficient qualified personnel in the future. The inability to attract and retain these personnel could result in delays in the research, development and commercialization of our potential products.
If we acquire other GR-II antagonists or other technologies or potential products, we will incur a variety of costs and may never realize the anticipated benefits of the acquisition.
If appropriate opportunities become available, we may attempt to acquire other GR-II antagonists, particularly GR-II antagonists that do not terminate pregnancy. We may also be able to acquire other technologies or potential products that are complementary to our operating plan. We currently have no commitments, agreements or plans for any acquisitions. The process of acquiring rights to another GR-II antagonist or any other potential product or technology may result in unforeseen difficulties and expenditures and may absorb significant management attention that would otherwise be available for ongoing development of our business. In addition, we may fail to realize the anticipated benefits of any acquired potential product or technology. Future acquisitions could dilute our stockholders’ ownership interest in us and could cause us to incur debt, expose us to future liabilities and result in amortization or other expenses related to goodwill and other intangible assets.
The occurrence of a catastrophic disaster or other similar events could cause damage to our or our manufacturers’ facilities and equipment, which could require us to cease or curtail operations.
Because our executive offices are located in the San Francisco Bay Area and some of our current manufacturers are located in earthquake-prone areas, our business is vulnerable to damage from various types of disasters or other similarly disruptive events, including earthquake, fire, flood, power loss and communications failures. In addition, political considerations relating to mifepristone may put us and our manufacturers at increased risk for terrorist attacks, protests or other disruptive events. If any disaster or other similar event were to occur, we may not be able to operate our business and our manufacturers may not be able to produce our product candidates. Our insurance may not be adequate to cover, and our insurance policies may exclude coverage for, our losses resulting from disasters or other business interruptions.
35
Table of Contents
Risks Related to Our Stock
The market price of our common stock has been and is likely to continue to be highly volatile due to the limited number of shares of our common stock held by non-affiliates of the Company or factors influencing the stock market and opportunities for sale at any given time may be limited.
We cannot assure you that an active trading market for our common stock will exist at any time. Holders of our common stock may not be able to sell shares quickly or at the market price if trading in our common stock is not active. During the 52-week period ended March 15, 2010, our average daily trading volume has been approximately 36,000 shares and the intra-day sales prices per share of our common stock on the NASDAQ Capital Market has ranged from $0.73 to $3.22. As of March 15, 2010, our officers, directors and principal stockholders control approximately 60% of our common stock. The trading price of our common stock has been and is likely to continue to be highly volatile and could be subject to wide fluctuations in price in response to various factors, many of which are beyond our control, including:
| • | our cash and short-term investment position; |
| • | actual or anticipated timing and results of our clinical trials; |
| • | actual or anticipated regulatory approvals of our product candidates or of competing products; |
| • | changes in laws or regulations applicable to our product candidates or our competitors’ products; |
| • | changes in the expected or actual timing of our development programs or our competitors’ potential development programs; |
| • | actual or anticipated variations in quarterly operating results; |
| • | announcements of technological innovations by us, our collaborators or our competitors; |
| • | new products or services introduced or announced by us or our competitors; |
| • | general market and economic conditions, including those seen as a result of the recent worldwide financial credit crisis; |
| • | changes in financial estimates or recommendations by securities analysts; |
| • | conditions or trends in the biotechnology and pharmaceutical industries; |
| • | changes in the market valuations of similar companies; |
| • | announcements by us or our competitors of significant acquisitions, strategic partnerships, joint ventures or capital commitments; |
| • | additions or departures of key personnel; |
| • | disputes or other developments relating to proprietary rights, including patents, litigation matters and our ability to obtain patent protection for our technologies; |
| • | developments concerning collaborations; |
| • | trading volume of our common stock; |
| • | limited number of shares of our common stock held by our non-affiliates; |
| • | maintaining compliance with the listing requirements of the stock exchange on which we are listed; |
| • | announcement of, or expectation of, additional financing efforts; and |
| • | sales of our common stock by us or our stockholders. |
36
Table of Contents
In addition, the stock market in general, the Nasdaq Capital Market and the market for biotechnology and life sciences companies in particular have experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of those companies. These broad market and industry factors may seriously harm the market price of our common stock, regardless of our operating performance. In the past, following periods of volatility in the market, securities class-action litigation has often been instituted against companies. Such litigation, if instituted against us, could result in substantial costs and diversion of management’s attention and resources.
If we fail to continue to meet all applicable Nasdaq Capital Market requirements, our stock could be delisted by the Nasdaq Capital Market. If delisting occurs, it would adversely affect the market liquidity of our common stock and harm our business.
If we are unable to meet any of the Nasdaq listing requirements in the future, including, for example, if the closing bid price for our common stock is below $1 per share for 30 consecutive trading days, the Nasdaq Capital Market staff could determine to delist our common stock, the delisting could adversely affect the market liquidity of our common stock and the market price of our common stock could decrease. Such delisting could also adversely affect our ability to obtain financing for the continuation of our operations and could result in the loss of confidence by investors, suppliers and employees.
Securities analysts may not continue to provide or initiate coverage of our common stock or may issue negative reports, and this may have a negative impact on our common stock’s market price.
Securities analysts currently covering our common stock may discontinue research coverage. Additional securities analysts may elect not to provide research coverage of our common stock. A lack of research coverage may adversely affect our common stock’s market price. The trading market for our common stock may be affected in part by the research and reports that industry or financial analysts publish about us or our business. If one or more of the analysts who elects to cover us downgrades our stock, our stock price would likely decline rapidly and significantly. If one or more of these analysts ceases coverage of our company, we could lose visibility in the market, which in turn could cause our stock price to decline. In addition, rules mandated by the Sarbanes-Oxley Act of 2002, and a global settlement reached in 2003 between the SEC, other regulatory analysts and a number of investment banks have led to a number of fundamental changes in how analysts are reviewed and compensated. In particular, many investment banking firms are required to contract with independent financial analysts for their stock research. It may be difficult for companies such as ours with smaller market capitalizations to attract independent financial analysts that will cover our common stock. This could have a negative effect on our market price.
A sale of a substantial number of shares of our common stock may cause the price of our common stock to decline.
Sales of a substantial number of shares of our common stock in the public market could harm the market price of our common stock. As additional shares of our common stock become available for resale in the public market, the supply of our common stock will increase, which could decrease the price. Substantially all of the shares of our common stock are eligible for sale, subject to applicable volume and other resale restrictions.
We may be required to pay significant amounts if we are not able to meet our obligations under our outstanding registration rights agreements.
The registration rights agreement covering the approximately 8.9 million shares of our common stock issued in a private offering in March 2008 and an additional approximately 4.5 million shares of common stock underlying warrants issued in connection with the offering provided that if we failed to file or cause to be declared effective the registration statement covering the resale of these shares prior to a specified deadlines, or failed to maintain the effectiveness of such registration statement (subject to limited permissible suspension
37
Table of Contents
periods), we would be required to pay the holders of such shares and warrants liquidated damages at the rate of 1% of the purchase price of these shares and warrants per month, up to a total of 10%. The registration statement covering the resale of the shares and shares underlying the warrants sold in this transaction was declared effective by the SEC in November 2008. Since this registration statement was not declared effective within the time frame specified in the registration rights agreement, we became obligated to pay the investors in this financing liquidated damages of approximately $1.3 million in 2008. As noted above, if we fail to maintain the effectiveness of this registration statement, we may be obligated to pay additional liquidated damage amounts in the future.
See the discussion above under “Risks Related to our Business” regarding risks associated with the CEFF, including the risks regarding registration rights under that agreement.
If we are required to pay significant amounts under these or future registration rights agreements, it could have a material adverse effect on our financial condition and ability to finance our operations.
Our officers, directors and principal stockholders acting as a group, will be able to significantly influence corporate actions.
As of March 15, 2010, our officers, directors and principal stockholders control approximately 60% of our common stock. As a result, these stockholders, acting together, will be able to significantly influence all matters requiring approval by our stockholders, including the election of directors and the approval of mergers or other business combination transactions. The interests of this group of stockholders may not always coincide with our interests or the interests of other stockholders and may prevent or delay a change in control. This significant concentration of share ownership may adversely affect the trading price of our common stock because investors often perceive disadvantages to owning stock in companies with controlling stockholders.
Changes in laws and regulations may result in increased costs to us, which may harm our financial results.
New laws and regulations, as well as changes to existing laws and regulations, affecting public companies, including the provisions of the Sarbanes-Oxley Act of 2002 and rules adopted by the SEC and by The Nasdaq Capital Market, would likely result in increased costs to us as we respond to their requirements. We are investing resources to comply with evolving laws and regulations, and this investment may result in increased general and administrative expenses and a diversion of management’s time and attention from revenue-generating activities to compliance activities.
In addition, new rules and regulations could make it more difficult or costly for us to obtain certain types of insurance, including director and officer liability insurance, and we may be forced to accept reduced policy limits and coverage or incur higher costs to obtain the same or similar coverage. The impact of these events could also make it more difficult for us to attract and retain qualified persons to serve on our Board of Directors, or our board committees, or as executive officers. At present, we cannot predict or estimate the amount of the additional costs related to new rules and regulations or the timing of such costs.
Compliance with public company obligations, including the securities laws and regulations, is costly and requires significant management resources, and we may fail to comply.
We are a small company with limited resources.
The federal securities laws and regulations, including the corporate governance and other requirements of the Sarbanes-Oxley Act of 2002, impose complex and continually changing regulatory requirements on our operations and reporting. These requirements impose comprehensive reporting and disclosure requirements, set stricter independence and financial expertise standards for audit committee members, and impose civil and criminal penalties for companies, their chief executive officers, principal financial officers and directors for
38
Table of Contents
securities law violations. These requirements have increased and will continue to increase our legal compliance costs, increase the difficulty and expense in obtaining director and officer liability insurance, and make it harder for us to attract and retain qualified members of our Board of Directors and/or qualified executive officers. Such developments could harm our results of operations and divert management’s attention from business operations.
In addition, as directed by Section 404 of the Sarbanes-Oxley Act of 2002, the SEC adopted rules requiring public companies to include a report of management on the company’s internal control over financial reporting in their annual reports on Form 10-K. This requirement first applied to our annual report on Form 10-K for the year ended December 31, 2007. This same legislation also requires that the independent registered public accounting firm auditing our financial statements must attest to and report on the effectiveness of our internal controls over financial reporting. The SEC postponed the initial compliance date for this requirement for smaller reporting companies such that the requirement for the auditor’s attestation and report will first apply to our annual report on Form 10-K for our fiscal year ending December 31, 2010. Uncertainty exists regarding our ability to comply with these requirements by applicable deadlines and to maintain compliance in future years. If we are unable to complete the required assessment as to the adequacy of our internal control over financial reporting in 2009 or in future years or if our independent registered public accounting firm is unable to provide us with an unqualified report as to the effectiveness of our internal control over financial reporting as of the required deadline in 2010 and as of future year ends, investors could lose confidence in the reliability of our financial reporting.
Changes in or interpretations of accounting rules and regulations could result in unfavorable accounting charges or require us to change our accounting policies or operating practices.
Accounting methods and policies for business and marketing practices of pharmaceutical companies, including policies regarding are subject to continual review, interpretation and guidance from relevant accounting authorities, including the SEC. For example, in December 2004, the Financial Accounting Standards Board adopted Financial Accounting Standard 123R, “Share Based Payment.” This statement, which we adopted in 2006, requires the recording of expense for the fair value of stock options granted. As a result, our operating expenses have increased and are likely to continue to increase. We rely heavily on stock options to compensate existing employees and attract new employees. Because we are now required to expense stock options on a fair-value basis, we may choose to reduce our reliance on stock options as a compensation tool. If we reduce our use of stock options, it may be more difficult for us to attract and retain qualified employees. Although we believe that our accounting practices are consistent with current accounting pronouncements, changes to or interpretations of accounting methods or policies in the future may require us to reclassify, restate or otherwise change or revise our financial statements.
Anti-takeover provisions in our charter and bylaws and under Delaware law may make an acquisition of us or a change in our management more difficult, even if an acquisition or a management change would be beneficial to our stockholders.
Provisions in our charter and bylaws may delay or prevent an acquisition of us or a change in our management. Some of these provisions divide our board into three classes with only a portion of our directors subject to election at each annual meeting, allow us to issue preferred stock without any vote or further action by the stockholders, require advance notification of stockholder proposals and nominations of candidates for election as directors and prohibit stockholders from acting by written consent. In addition, a supermajority vote of stockholders is required to amend our bylaws. Our bylaws provide that special meetings of the stockholders may be called only by our Chairman, President or the Board of Directors and that the authorized number of directors may be changed only by resolution of the Board of Directors. These provisions may prevent or delay a change in our Board of Directors or our management, which is appointed by our board of directors. In addition, because we are incorporated in Delaware, we are governed by the provisions of Section 203 of the Delaware General Corporation Law. Section 203 may prohibit large stockholders, in particular those owning 15% or more of our outstanding voting stock, from merging or combining with us. These provisions in our charter, bylaws and
39
Table of Contents
under Delaware law could reduce the price that investors might be willing to pay for shares of our common stock in the future and result in the market price being lower than it would be without these provisions.
| ITEM 1B. | UNRESOLVED STAFF COMMENTS |
None.
| ITEM 2. | PROPERTIES |
We lease approximately 7,700 square feet of office space in Menlo Park, California for our corporate facilities. On October 20, 2008, we renewed our lease for office space for a two-year term commencing on January 1, 2009. The new lease provides us an option to extend the lease for an additional year upon 180 days notice. We expect that these facilities will accommodate our operations for the next year.
| ITEM 3. | LEGAL PROCEEDINGS |
We are not currently involved in any material legal proceedings.
| ITEM 4. | (Removed and Reserved) |
40
Table of Contents
| ITEM 5. | MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES |
Market Information
Our common stock is traded on The Nasdaq Capital Market under the symbol “CORT”. The following table sets forth the high and low intra-day sale prices per share of our common stock on The Nasdaq Capital Market for the periods indicated. These prices represent quotations among dealers without adjustments for retail mark-ups, markdowns or commissions, and may not represent prices of actual transactions.
| High | Low | |||||
| 2009 |
||||||
| First Quarter |
$ | 1.46 | $ | 0.75 | ||
| Second Quarter |
$ | 1.29 | $ | 0.73 | ||
| Third Quarter |
$ | 1.65 | $ | 0.75 | ||
| Fourth Quarter |
$ | 3.10 | $ | 1.36 | ||
| High | Low | |||||
| 2008 |
||||||
| First Quarter |
$ | 3.50 | $ | 2.25 | ||
| Second Quarter |
$ | 4.29 | $ | 1.85 | ||
| Third Quarter |
$ | 2.43 | $ | 1.19 | ||
| Fourth Quarter |
$ | 1.90 | $ | 0.80 | ||
Stockholders of Record and Dividends
As of March 15, 2010, we had 62,703,717 shares of common stock outstanding held by 144 stockholders of record. We have never declared or paid cash dividends on our capital stock. We currently intend to retain any future earnings to finance the growth and development of our business and therefore, do not anticipate paying any cash dividends in the foreseeable future.
Sale of Unregistered Securities
All sales of unregistered securities during the year ended December 31, 2009 have previously been disclosed in filings with the SEC. We have used, or will use, the net proceeds from these transactions to fund our research, development, and administrative activities, as well as for general corporate purposes, including working capital.
Sales and Repurchases of Securities
See Item 12, “Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters” for information with respect to our compensation plans under which equity securities are authorized for issuance.
Market Performance Graph
The graph and the accompanying text below is not “soliciting material,” is not deemed filed with the SEC and is not to be incorporated by reference in any filings by us under the Securities Act or the Exchange Act, whether made before or after the date hereof and irrespective of any general incorporation language in such filing.
41
Table of Contents
The rules of the SEC require that we include a line-graph comparing cumulative stockholder returns on our common stock with the NASDAQ Composite Index (which tracks the aggregate price performance of equity securities of companies traded on NASDAQ) and either a published industry or line-of-business standard index or an index of peer companies selected by us. We have elected to use the NASDAQ Biotechnology Index (consisting of a group of approximately 130 companies in the biotechnology sector, including us) for purposes of the performance comparison that appears below.
The graph shows the cumulative total stockholder return assuming the investment of $100.00 and the reinvestment of dividends and is based on the returns of the component companies weighted according to their market capitalizations as of the end of the period for which returns are indicated. No dividends have been declared on our common stock.
The stockholder return shown on the graph below is not necessarily indicative of future performance, and we do not make or endorse any predictions as to future stockholder returns.
COMPARISON OF 5 YEAR CUMULATIVE TOTAL RETURN* AMONG
CORCEPT THERAPEUTICS, THE NASDAQ CAPITAL MARKET (U.S.) INDEX
AND THE NASDAQ BIOTECHNOLOGY INDEX
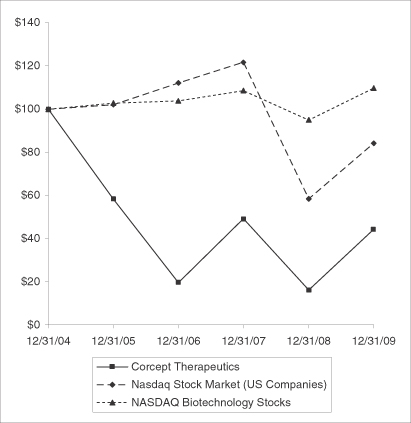
| * | $100 invested on December 31, 2004 including reinvestment of dividends. Fiscal year ending December 31. |
42
Table of Contents
| ITEM 6. | SELECTED FINANCIAL DATA |
SELECTED FINANCIAL DATA
(in thousands, except per share data)
The selected financial data set forth below are derived from our financial statements. The statement of operations data for the years ended December 31, 2007, 2008 and 2009 and for the period from inception (May 13, 1998) to December 31, 2009 and the balance sheet data as of December 31, 2008 and 2009 are derived from our audited financial statements included in this Annual Report on Form 10-K (Form 10-K). The statements of operations data for the years ended December 31, 2005 and 2006, and the balance sheet data as of December 31, 2006, 2005 and 2004 have been derived from our audited financial statements, which are not included in this Form 10-K. The selected financial data set forth below should be read in conjunction with our financial statements, the related notes and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” included elsewhere in this Form 10-K.
| Year Ended December 31, | Period from inception (May 13, 1998) to December 31, 2009 |
|||||||||||||||||||||||
| 2009 | 2008 | 2007 | 2006 | 2005 | ||||||||||||||||||||
| (In thousands, except per share data) | ||||||||||||||||||||||||
| Statement of Operations Data: |
||||||||||||||||||||||||
| Collaboration revenue |
$ | 29 | $ | 209 | $ | 482 | $ | 294 | $ | — | $ | 1,014 | ||||||||||||
| Operating expenses: |
||||||||||||||||||||||||
| Research and development* |
14,402 | 14,152 | 7,860 | 20,834 | 17,074 | 114,211 | ||||||||||||||||||
| General and administrative* |
5,877 | 5,746 | 4,867 | 5,042 | 4,084 | 40,762 | ||||||||||||||||||
| Total operating expenses |
20,279 | 19,898 | 12,727 | 25,876 | 21,158 | 154,973 | ||||||||||||||||||
| Loss from operations |
(20,250 | ) | (19,689 | ) | (12,245 | ) | (25,582 | ) | (21,158 | ) | (153,959 | ) | ||||||||||||
| Non-operating income (expense), net |
84 | (372 | ) | 672 | 709 | 1,065 | 3,721 | |||||||||||||||||
| Net loss |
$ | (20,166 | ) | $ | (20,061 | ) | $ | (11,573 | ) | $ | (24,873 | ) | $ | (20,093 | ) | $ | (150,238 | ) | ||||||
| Net loss per share: |
||||||||||||||||||||||||
| Basic and diluted |
$ | (0.38 | ) | $ | (0.43 | ) | $ | (0.34 | ) | $ | (1.09 | ) | $ | (0.89 | ) | |||||||||
| Weighted average shares—basic and diluted |
52,443 | 46,721 | 34,251 | 22,841 | 22,608 | |||||||||||||||||||
| * Includes non-cash stock-based compensation (recovery) of the following: |
|
|||||||||||||||||||||||
| Research and development |
$ | 263 | $ | 268 | $ | 213 | $ | 535 | $ | (26 | ) | $ | 5,276 | |||||||||||
| General and administrative |
1,552 | 1,360 | 846 | 1,013 | 799 | 9,562 | ||||||||||||||||||
| Total non-cash stock-based compensation |
$ | 1,815 | $ | 1,628 | $ | 1,059 | $ | 1,548 | $ | 773 | $ | 14,838 | ||||||||||||
| As of December 31, | |||||||||||||||
| 2009 | 2008 | 2007 | 2006 | 2005 | |||||||||||
| (In thousands) | |||||||||||||||
| Balance Sheet Data: |
|||||||||||||||
| Cash, cash equivalents and investments |
$ | 23,867 | $ | 18,309 | $ | 17,366 | $ | 9,456 | $ | 29,619 | |||||
| Working capital |
22,001 | 16,717 | 14,662 | 6,286 | 25,984 | ||||||||||
| Total assets |
24,511 | 19,775 | 17,744 | 9,902 | 30,156 | ||||||||||
| Long-term liabilities |
— | 6 | 16 | 29 | 42 | ||||||||||
| Total stockholders’ equity |
22,092 | 16,907 | 14,734 | 6,360 | 26,593 | ||||||||||
See our financial statements and related notes for a description of the calculation of the net loss per share and the weighted-average number of shares used in computing the per share amounts.
43
Table of Contents
| ITEM 7. | MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
Forward-Looking Statements
This Management’s Discussion and Analysis of Financial Condition and Results of Operations contains forward-looking statements within the meaning of Section 21E of the Exchange Act and Section 27A of the Securities Act and should be read in conjunction with the “Risk Factors” section of Part I of this Form 10-K. All statements contained in this Form 10-K other than statements of historical fact are forward-looking statements. When used in this report or elsewhere by management from time to time, the words “believe,” “anticipate,” “intend,” “plan,” “estimate,” “expect,” “may,” “will,” “should,” “seeks” and similar expressions are forward-looking statements. Such forward-looking statements are based on current expectations, but the absence of these words does not necessarily mean that a statement is not forward-looking. Forward-looking statements may include, but are not limited to, statements about:
| • | the progress and timing of our research, development and clinical programs and the timing of regulatory activities; |
| • | the timing of the market introduction of CORLUX® and future product candidates, including CORT 108297; |
| • | estimates of the dates by which we expect to report results of our clinical trials and the anticipated results of these trials; |
| • | our ability to market, commercialize and achieve market acceptance for CORLUX or other future product candidates, including CORT 108297; |
| • | uncertainties associated with obtaining and enforcing patents; |
| • | our estimates for future performance; and |
| • | our estimates regarding our capital requirements and our needs for, and ability to obtain, additional financing. |
Forward-looking statements are not guarantees of future performance and involve risks and uncertainties. Actual events or results may differ materially from those discussed in the forward-looking statements as a result of various factors. For a more detailed discussion of such forward-looking statements and the potential risks and uncertainties that may impact upon their accuracy, see “Risk Factors” included in Part I of this Form 10-K and the “Overview” and “Liquidity and Capital Resources” sections of this Management’s Discussion and Analysis of Financial Condition and Results of Operations. These forward-looking statements reflect our view only as of the date of this report. Except as required by law, we undertake no obligations to update any forward-looking statements. Accordingly, you should also carefully consider the factors set forth in other reports or documents that we file from time to time with the SEC.
Overview
We are a pharmaceutical company engaged in the discovery and development of drugs for the treatment of severe metabolic and psychiatric disorders. Since our inception in May 1998, we have been developing our lead product, CORLUX, a potent glucocorticoid receptor II (GR-II) antagonist. We are also developing three series of novel selective GR-II antagonists.
Cushing’s Syndrome
Cushing’s Syndrome is a disorder caused by prolonged exposure of the body’s tissues to high levels of the hormone cortisol. Sometimes called “hypercortisolism,” it is relatively uncommon and most often affects adults aged 20 to 50. An estimated 10 to 15 of every one million people are newly diagnosed with this syndrome each
44
Table of Contents
year, which equates to approximately 3,000 new patients in the United States. This results in an estimated prevalence of 20,000 patients with Cushing’s Syndrome in the United States.
The Investigational New Drug application (IND) for the evaluation of CORLUX for the treatment of Cushing’s Syndrome was opened in September 2007. The United States Food and Drug Administration (FDA) has indicated that our single 50-patient open-label study may provide a reasonable basis for the submission of a New Drug Application (NDA) for this indication. We expect to complete enrollment in this Phase 3 study in April, as the requisite 50 patients have now been dosed or identified. We expect to announce results of this study in the fourth quarter of this year and to submit our NDA for the use of CORLUX in Cushing’s Syndrome by year-end 2010.
In July 2007, we received Orphan Drug Designation from the FDA for CORLUX for the treatment of endogenous Cushing’s Syndrome. Orphan Drug Designation is a special status granted by the FDA to encourage the development of treatments for diseases or conditions that affect fewer than 200,000 patients in the United States. Drugs that receive Orphan Drug Designation obtain seven years of marketing exclusivity from the date of drug approval, as well as tax credits for clinical trial costs, marketing application filing fee waivers and assistance from the FDA in the drug development process.
Psychotic Depression
We are also developing CORLUX for the treatment of the psychotic features of psychotic major depression under an exclusive patent license from Stanford University. Psychotic major depression will hereinafter be referred to as psychotic depression. The FDA has granted “fast track” status to evaluate the safety and efficacy of CORLUX for the treatment of the psychotic features of psychotic depression.
In March 2008, we commenced enrollment in Study 14, our ongoing Phase 3 trial in psychotic depression. The protocol for this trial incorporates what we have learned from our three previously completed Phase 3 trials to address the established relationship between increased drug plasma levels and clinical response and attempts to decrease the random variability observed in the results of the psychometric instruments used to measure efficacy. In one of the previously completed Phase 3 trials, Study 06, we prospectively tested and confirmed that patients whose plasma levels rose above a predetermined threshold statistically separated from both those patients whose plasma levels were below the threshold and those patients who received placebo; this threshold was established from data produced in earlier studies. As expected, patients who took 1200 mg of CORLUX in Study 06 developed higher drug plasma levels than patients who received lower doses. Further, there was no discernable difference in the incidence of adverse events between placebo and any of the three CORLUX dose groups in Study 06. Based on this information, we are using a CORLUX dose of 1200 mg once per day for seven days in Study 14. In addition, we also are utilizing a third party centralized rating service to independently evaluate the patients for entry into the study as well as for their level of response. We believe the centralization of this process will improve the consistency of rating across clinical trial sites and reduce the background noise that was illustrated in earlier studies and is endemic to many psychopharmacologic studies. We believe that this change in dose, as well as the other modifications to the protocol, should allow us to demonstrate the efficacy of CORLUX in the treatment of the psychotic symptoms of psychotic depression. In March 2009, we announced that, in order to lower variable and fixed expenses and to conserve financial resources, we were scaling back our planned rate of spending on this trial and extended the timeline for its completion. As of early July 2009, we completed the implementation of this strategy, which included reducing the number of clinical sites to eight.
Management of Weight Gain Induced by Antipsychotics
In 2005, we published the results of studies in rats that demonstrated that CORLUX both reduced the weight gain associated with the ongoing use of olanzapine and mitigated the weight gain associated with the initiation of treatment with olanzapine (the active ingredient in Zyprexa). This study was paid for by Eli Lilly and Company (Eli Lilly.)
45
Table of Contents
During 2007 we announced positive results from our clinical proof-of-concept study in lean healthy male volunteers evaluating the ability of CORLUX to mitigate weight gain associated with the use of Zyprexa. The results show a statistically significant reduction in weight gain in those subjects who took Zyprexa plus CORLUX compared to those who took Zyprexa plus placebo. Also, the addition of CORLUX to treatment with Zyprexa had a beneficial impact on secondary metabolic measures such as fasting insulin, triglycerides and abdominal fat, as indicated by waist circumference. Eli Lilly provided Zyprexa and financial support for this study. In January 2009 we announced positive results from a similar proof-of-concept study evaluating the ability of CORLUX to mitigate weight gain associated with the use of Johnson & Johnson’s Risperdal. This study, which began in 2008, confirmed the earlier results seen with CORLUX and Zyprexa, demonstrating a statistically significant reduction in weight and secondary metabolic endpoints of fasting insulin, triglycerides and abdominal fat, as indicated by waist circumference. The results from the study of CORLUX and Risperdal were presented at several scientific conferences, including the American Diabetes Association meeting in June 2009.
The combination of Zyprexa or Risperdal and CORLUX is not approved for any indication. The purpose of these studies was to explore the hypothesis that GR-II antagonists would mitigate weight gain associated with atypical antipsychotic medications. The group of medications known as atypical antipsychotics, including Zyprexa, Risperdal, Clozaril and Seroquel, are widely used to treat schizophrenia and bipolar disorder. All medications in this group are associated with treatment emergent weight gain of varying degrees and carry a warning in the label relating to treatment emergent hyperglycemia and diabetes mellitus.
Research
In 2003, we initiated a discovery research program to identify and patent selective GR-II antagonists to develop a pipeline of products for proprietary use. Three distinct series of GR-II antagonists were identified. These compounds appear to be as potent as our lead product CORLUX in blocking cortisol but, unlike CORLUX, they do not appear to block the PR (progesterone), ER (estrogen), AR (androgen) or GR-I (mineralocorticoid) receptors. Composition of matter patents on all of the three series have been granted in Europe. The patent on two series have issued in the United States. Examination has not yet begun in the United States on the third.
New Chemical Entity—CORT 108297
In 2007, we commenced a human microdosing study of one of our newly identified selective GR-II antagonists, CORT 108297, with Xceleron Limited utilizing their Accelerator Mass Spectrometry technology. In this microdosing study, we evaluated CORT 108297, a compound which develops particularly high plasma and brain concentrations in an animal model. On May 1, 2008, we announced the results from this study, which demonstrated that CORT 108297 was extremely well absorbed, demonstrated good bioavailability and had a half-life that appears compatible with once-a-day oral dosing. In addition, further pharmacokinetic testing of CORT 108297 in a rat model indicated that a ten-fold increase in oral dose (5 milligrams per kilograms to 50 milligrams per kilograms) led to a proportional increase in the amount of compound detected in plasma.
In September 2008, we signed a second agreement with Eli Lilly, under which Eli Lilly agreed to provide funding and provide olanzapine for two studies to test the effectiveness of CORT 108297 in rat models of olanzapine induced weight gain. In January 2009 we announced top-line results from these studies of CORT 108297 and olanzapine. The results from the studies of both the prevention and reversal of antipsychotic-induced weight gain were positive and statistically significant. The results of these studies were presented at the International Society of Psychoneuroendocrinology and the World Congress of Biological Psychiatry conferences in July 2009.
At the American Diabetes Association conference in June 2009 there was also a presentation of preclinical data from another study of CORT 108297 conducted at Stanford University. This study demonstrated that CORT
46
Table of Contents
108297 suppresses body weight gain and improves insulin sensitivity in healthy mice fed a 60% fat diet and high sucrose liquid.
The manufacturing and pre-clinical development of CORT 108297 began late in 2008 and continued through 2009 as preparatory steps to the submission of an IND with the FDA, which we submitted in December 2009. On February 22, 2010, we began dosing patients in the Phase 1 study with this compound.
General
Our activities to date have included:
| • | product development; |
| • | designing, funding and overseeing clinical trials; |
| • | regulatory affairs; and |
| • | intellectual property prosecution and expansion. |
Historically, we have financed our operations and internal growth primarily through private placements of our preferred and common stock and the public sale of common stock rather than through collaborative or partnership agreements. Therefore, we have no research funding or collaborative payments payable to us, except for the limited revenue that has been collected under the agreements with Eli Lilly discussed above.
We are in the development stage and have incurred significant losses since our inception. We have not generated any revenue other than the revenue under the agreements with Eli Lilly, and do not expect to generate significant revenue until CORLUX has been approved by the FDA for marketing in the United States, if at all. As of December 31, 2009, we had an accumulated deficit of $150.2 million. Our historical operating losses have resulted principally from our research and development activities, including clinical trial activities for CORLUX, discovery research, non-clinical activities such as toxicology and carcinogenicity studies, manufacturing process development and regulatory activities, as well as general and administrative expenses. We expect to continue to incur net losses over at least the next several years as we continue our CORLUX clinical development program, apply for regulatory approvals, initiate development of newly identified GR-II antagonists for various indications, continue our discovery research program, acquire and develop treatments in other therapeutic areas, establish sales and marketing capabilities and expand our operations.
Our business is subject to significant risks, including the risks inherent in our research and development efforts, the results of our CORLUX clinical trials, uncertainties associated with securing financing, uncertainties associated with obtaining and enforcing patents, our investment in manufacturing set-up, the lengthy and expensive regulatory approval process and competition from other products. Our ability to successfully generate revenues in the foreseeable future is dependent upon our ability, alone or with others, to finance our operations and develop, obtain regulatory approval for, manufacture and market our lead product.
Results of Operations
Collaboration revenue. Collaboration revenue relates to services rendered in connection with our agreements with Eli Lilly discussed above under the caption “Overview-Management of Weight Gain induced by Antipsychotics.” Under these agreements, Eli Lilly agreed to supply the Zyprexa and olanzapine and pay for the costs of the studies. We were required to perform development activities as specified in the agreements and we were reimbursed based on the costs associated with the conduct of the trial and the preparation and packaging of clinical trial materials. Revenue was recognized as the services are rendered in accordance with the agreements.
47
Table of Contents
During the years ended December 31, 2009, 2008 and 2007, we recognized approximately $29,000, $209,000 and $482,000, respectively, under these agreements. There will be no revenue under the agreements in the future as all of the activities were completed by mid-2009.
Research and development expenses. Research and development expenses include the personnel costs related to our development activities, including facilities costs and non-cash stock-based compensation, as well as the costs of discovery research, pre-clinical studies, clinical trial preparations, enrollment and monitoring expenses, regulatory costs, the costs of manufacturing development and the costs of manufacture and / or acquisition of clinical trial materials.
Research and development expenses increased 2% to $14.4 million for the year ended December 31, 2009 from $14.2 million for the year ended December 31, 2008. During 2009, as compared to 2008, there were increases of approximately $1.9 million in costs related to research, manufacturing and IND-enabling work with our selective GR-II antagonist, CORT 108297, $830,000 related to the Phase 3 trial and the long-term extension study in Cushing’s Syndrome, $242,000 related to the conduct of other NDA-supportive studies, $260,000 in staffing costs, $130,000 of consulting expenses and $90,000 related to the cost of a carcogenicity study. Offsetting these increases were decreases of approximately $1.5 million in manufacturing expenses related to CORLUX due to the acquisition and manufacture during 2008 of the initial supply of materials for the CORLUX clinical trials and completion of certain manufacturing process development activities related to CORLUX, $840,000 related to the scaling back of our Phase 3 trial of CORLUX in psychotic depression that was announced in March 2009, $610,000 in basic research on our selective GR-II antagonists as CORT 108297 moved into the IND-enabling phase and $210,000 related to our clinical trial of CORLUX for the mitigation of weight gain caused by Risperdal that was completed early in 2009.
Research and development expenses increased 80% to $14.2 million for the year ended December 31, 2008, from $7.9 million for the year ended December 31, 2007. The increase in expenses reflects clinical trial cost increases of approximately $5.1 million related to trials in psychotic depression, Cushing’s Syndrome and the mitigation of weight gain caused by Risperdal that commenced in 2008, which were partially offset by decreases of approximately $3.1 million due to the completion of our earlier Phase 3 clinical trials for psychotic depression, our cardiac study and our earlier human proof of concept study in the mitigation of Zyprexa induced weight gain in 2007. During 2008, we also performed two smaller clinical studies to test formulation development and drug-drug interaction at a cost of approximately $705,000. During the year ended December 31, 2008 as compared to 2007, there were also increases in contract research expenses of approximately $750,000 due to basic research work on new chemical compounds, approximately $495,000 related to other research and preclinical work with our selective new GR-II antagonist, CORT 108297, and approximately $209,000 of costs associated with the rat studies using this compound in combination with olanzapine, which was conducted in connection with the agreement with Eli Lilly discussed above. In addition, during the year ended December 31, 2008, there was an increase in manufacturing expenses of approximately $925,000 due to the acquisition and manufacture of materials for the clinical trials and manufacturing process development. There were also increases of approximately $565,000 in staffing costs, $530,000 in consulting expenses, $135,000 in travel costs and $115,000 in the allocation of facilities costs during 2008 as compared to 2007.
Research and development expenses discussed above included stock based compensation charges related to option grants to individuals performing these functions of approximately $265,000, $270,000 and $240,000, respectively, for the years ended December 31, 2009, 2008 and 2007. The increase in expense between years was primarily the result of expense related to new option grants calculated on a straight-line basis, which was partially offset by the decrease of expense due to the declining scale of expense related to earlier option grants that were being expensed using the graded vesting method. In addition, during 2007, upon the termination of an employee who worked in a development function, we recorded a reversal of approximately $25,000 of previously reported stock-based compensation expense, which represents the difference between the expense recorded and the expense that would have been recorded based upon the rights to options that vested during the service of these individuals as employees. There were no terminations or conversions to consultant during 2008 or 2009.
48
Table of Contents
Below is a summary of our research and development expenses by major project:
| Year Ended December 31, | |||||||||
| Project |
2009 | 2008* | 2007* | ||||||
| (in thousands) | |||||||||
| CORLUX |
|||||||||
| Psychotic Depression |
$ | 5,030 | $ | 5,948 | $ | 2,993 | |||
| Cushing’s Syndrome |
2,952 | 2,316 | 553 | ||||||
| Weight Gain Mitigation |
565 | 1,588 | 677 | ||||||
| Selective GR-II antagonists |
3,940 | 2,186 | 917 | ||||||
| Unallocated activities, including NDA supportive studies and manufacturing, regulatory, pre-clinical activities |
1,652 | 1,846 | 2,507 | ||||||
| Stock-based compensation |
263 | 268 | 213 | ||||||
| Total research and development expense |
$ | 14,402 | $ | 14,152 | $ | 7,860 | |||
| * | The data in the table above for the years ended December 31, 2007 and 2008 has been reorganized to be consistent with the presentation for 2009 to recognize that certain costs such as NDA supportive studies, some manufacturing activities, regulatory and pre-clinical activities are not readily allocable to any one product or indication as these activities benefit multiple products and/or indications. |
We expect that research and development expenditures will increase during 2010 as compared to 2009 due to the continuation of our Phase 3 studies in Cushing’s Syndrome and psychotic depression, and the continued development of CORT 108297 and our other proprietary selective GR-II antagonists. Research and development expenses in 2011 and future years will be largely dependent on the availability of additional funds to finance clinical development plans. See also, “Liquidity and Capital Resources”.
Many factors can affect the cost and timing of our trials including inconclusive results requiring additional clinical trials, slow patient enrollment, adverse side effects in study patients, insufficient supplies for our clinical trials and real or perceived lack of effectiveness or safety of the drug in our trials. The cost and timing of development of our selective GR-II antagonists will be dependent on our success in the effort and any difficulties that may be encountered. In addition, the development of all of our product candidates will be subject to extensive governmental regulation. These factors make it difficult for us to predict the timing and costs of the further development and approval of our product candidates.
General and administrative expenses. General and administrative expenses consist primarily of the costs of administrative personnel and related facility costs along with legal, accounting and other professional fees.
For the year ended December 31, 2009, general and administrative expenses increased 2% to $5.9 million from $5.7 million for the year ended December 31, 2008. This increase reflects higher staffing costs of approximately $445,000, due primarily to the recruitment of our new chief financial officer during the fourth quarter of 2008, which includes a net increase in stock-based compensation of $180,000 that reflects the expense related to stock options granted to our new chief financial officer, other employees and directors late in 2008 and during 2009. During this period, there were also increases in professional fees and consultancy costs of approximately $195,000, primarily related to the costs associated with periodic filings with the SEC and the preparations for the initial year of auditor attestation under SOX section 404, which requirement was deferred until 2010 by the SEC in a ruling announced in October 2009. These increases were partially offset by a decrease of approximately $575,000 in legal expenses due primarily to the reduction in patent related legal costs in 2009 as compared to the prior year.
General and administrative expenses increased 18% to $5.7 million for the year ended December 31, 2008 from $4.9 million for the year ended December 31, 2007. The increase in costs between years was primarily an
49
Table of Contents
increase of approximately $510,000 of legal costs related to patent activities and combined increases in staffing and consultancy costs of approximately $415,000. The changes in staffing costs include increases in non-cash stock-based compensation of approximately $510,000 and in recruiting costs of approximately $130,000, which were partially offset by a decrease of approximately $320,000 related to bonus compensation. The increase in stock-based compensation was the net result of approximately $315,000 of costs associated with additional stock options being expensed under the straight-line method, decreases of approximately $200,000 associated with the declining scale of expense of options accounted for under the graded vesting method and the inclusion in 2007 of a reversal of approximately $395,000 of stock-compensation expense in connection with the resignation of an officer, which represented the excess of expense under the graded vesting method as compared with the expense associated with stock options that actually vested prior to this termination.
General and administrative expenses included stock-based compensation expense related to option grants to individuals performing these functions of approximately $1.5 million, $1.4 million and $1.2 million, respectively, for the years ended December 31, 2009, 2008 and 2007.
The amount of general and administrative expenses in 2010 and future years will be largely dependent on our assessment of the staff necessary to support our continued clinical development activities, potential product commercialization support and the availability of additional funds. See also, “Liquidity and Capital Resources”.
Interest and other income, net. Interest and other income, net of investment management fees, was approximately $101,000 for the year ended December 31, 2009 as compared to $945,000 for the same period in 2008 and $690,000 in 2007. Interest income in 2009 and 2008 included approximately $60,000 and $410,000, respectively, related to the note receivable in connection with the March 2008 Financing, which was collected in February 2009. In addition, there were also decreases in yields and balances of invested funds as compared to 2008. The increase in net interest income in 2008 as compared to 2007 is the net result of the interest on this note being partially offset by decreased yields and balances of invested funds as compared to 2007.
Other expense. Other expense for the year ended December 31, 2009 was approximately $17,000 as compared to $1.3 million in 2008 and $15,000 in 2007. The other expense for 2008 was primarily related to the cost of liquidated damages due to the delay in the effectiveness of the registration statement of the securities sold in the March 2008 Financing. Other expense includes interest expense on capitalized leases and state tax on capital, which is based on our capital and asset positions as of each year-end.
Liquidity and Capital Resources
We have incurred operating losses since inception, and at December 31, 2009, we had a deficit accumulated during the development stage of $150.2 million. Since our inception, we have relied primarily on the proceeds from public and private sales of our equity securities to fund our operations.
At December 31, 2009, we had cash, cash equivalents and investments balances of $23.9 million, compared to $18.3 million at December 31, 2008. Net cash used in operating activities for the years ended December 31, 2009, 2008 and 2007, was $18.0 million, $18.4 million and $11.0 million, respectively. The use of cash in each period was primarily a result of our research and development activities and amounts incurred to develop our administrative infrastructure.
During 2009 we sold common stock and warrants in a private placement generating gross proceeds of approximately $18.0 million and we sold common stock to Kingsbridge Capital Limited (Kingsbridge), a private investment group, under a Committed Equity Financing Facility (CEFF) generating an additional $250,000. Issuance costs for these transactions totaled approximately $725,000. In February 2009, we collected a note receivable of $6.0 million that had been issued in March 2008 in connection with the March 2008 Financing. In January 2010, we sold additional stock to Kingsbridge under the CEFF for proceeds of $625,000. The net proceeds of all these transactions have been, or will be, used for general corporate purposes.
50
Table of Contents
We expect cash used in operating activities to increase during 2010 as the increased spending on the continuation of our Cushing’s Syndrome study and the development of our selective GR-II antagonists, including CORT 108297, is expected to be only partially offset by the decreased spending on psychotic depression, as compared to spending levels during 2009. We expect our funding requirements for operating activities will increase during later years due to the continuation and expansion of our development programs for Cushing’s Syndrome, psychotic depression and our selective GR-II antagonists, research activities, commercialization activities and general and administrative expenses.
We believe that we have sufficient capital resources to maintain our operations into early 2011, including the planned completion of enrollment of our Phase 3 Cushing’s Syndrome trial, continuation of our long-term extension study in this indication, and other activities in preparation of the submission of an NDA for CORLUX for the treatment of Cushing’s Syndrome, the continuation of enrollment in our Phase 3 psychotic depression trial, the early clinical development of CORT 108297, one of our proprietary, selective GR-II antagonists, and research activities aimed at the selection of additional compounds for further study.
We will need to raise additional funds to continue the development of CORLUX for the treatment of Cushing’s Syndrome or the psychotic features of psychotic depression beyond early 2011, to prepare for the commercialization of CORLUX for either of these indications and to continue and expand the development of our proprietary selective GR-II antagonists.
We cannot be certain that additional funding will be available on acceptable terms or at all. Further, any additional equity financing may be dilutive to stockholders, and any debt financing, if available, may involve restrictive covenants. If we obtain funds through collaborations with others, these arrangements may be on unfavorable terms or may require us to relinquish certain rights to our technologies or product candidates, including potentially our lead product candidate that we would otherwise seek to develop on our own. If adequate funds are not available, we may be required to delay, reduce the scope of or eliminate one or more of our research or development programs or we may be required to discontinue operations.
In March 2008, we entered into a CEFF with Kingsbridge. Under the terms of the agreement, Kingsbridge committed to provide up to $60 million of capital in exchange for newly-issued shares of our common stock for a period of up to three years after the SEC declares effective the registration statements filed by us covering the resale of the shares of common stock issuable in connection with the CEFF and the shares of common stock underlying the warrant issued to Kingsbridge. In June 2008, the SEC declared effective our registration statement with the SEC covering the resale of approximately 3.6 million of the shares issuable under the CEFF and the shares issuable upon the exercise of the warrant issued to Kingsbridge. This registration statement covers approximately 37% of the 9.6 million shares of our common stock issuable pursuant to the CEFF and all of the 330,000 shares of our common stock issuable upon exercise of the warrant issued to Kingsbridge. As of the filing of this report, approximately 2.9 million shares remain available for sale under the initial registration statement. We intend to file an additional registration statement covering the resale of the remaining 6.0 million shares of our common stock issuable pursuant to the CEFF approximately 60 days after Kingsbridge and its affiliates have resold substantially all of the securities registered for sale under this initial registration statement.
Under the terms of the agreement, the determination of the exact timing and amount of any CEFF financings will be made solely by us, subject to certain conditions. The agreement currently requires a minimum stock price of $1.50 per share to allow us to issue shares to Kingsbridge under the CEFF. Through January 2010, we have raised a total of approximately $1.6 million from the sales of stock under the CEFF. Based on the volume weighted average price on the NASDAQ Capital Market for our common stock for the period from March 25, 2008, the date of the signing of the Kingsbridge CEFF, through March 15, 2010, the maximum amount of additional funds that could be raised under the CEFF is approximately $16 million. The actual amount of funds that can be raised under this agreement will be dependent on the number of shares actually sold under the agreement and the market value of our stock during the pricing periods of each sale.
51
Table of Contents
While we monitor the cash balance in our checking account and transfer the funds in only as needed, these cash balances could be impacted if the underlying financial institution were to fail or could be subject to other adverse conditions in the financial markets. On October 23, 2008, the Federal Deposit Insurance Corporation implemented its Temporary Liquidity Guarantee Program. Under this program, non-interest bearing commercial accounts are insured to an unlimited amount through June 30, 2010, thus mitigating our exposure to any possible bank failure. To date, we have experienced no loss or lack of access to cash in our checking accounts.
As a result of volatile market conditions, the cost and availability of capital has been and may continue to be adversely affected by illiquid capital markets. Concern about the stability of the markets generally and the strength of counterparties specifically has led many lenders and institutional investors to reduce, and in some cases, cease to provide credit to businesses and consumers. Continued turbulence in the U.S. and international markets and economies and prolonged declines in business consumer spending may adversely affect our liquidity and financial condition, including our ability to access the capital markets to meet liquidity needs.
Contractual Obligations and Commercial Commitments
The following table presents our estimates of obligations under contractual agreements as of December 31, 2009:
| Payments Due by Period |
Less than 1 year |
1-3 Years |
3-5 Years |
More than 5 Years | ||||||||
| (in thousands) | ||||||||||||
| Research and development studies (1) through (4) |
$ | 4,156 | $ | 7,894 | $ | — | $ | — | ||||
| Operating lease (5) |
241 | — | — | — | ||||||||
| Capital leases (6) |
6 | — | — | — | ||||||||
| Minimum royalty payments (7) (8) |
150 | 100 | 100 | 50 per year | ||||||||
| Total |
$ | 4,553 | $ | 7,994 | $ | 100 | $ | 50 per year | ||||
| (1) | Amounts reflected for research and development studies exclude amounts included in accounts payable and accrued clinical costs reflected on the balance sheet as of December 31, 2009. |
| (2) | During 2007 and 2008, we signed agreements for services in connection with our trials for CORLUX for the treatment of Cushing’s Syndrome. The total commitment under these agreements is approximately $2.1 million. Approximately $900,000 of costs under these agreements have been expensed as of December 31, 2009, with the remainder to be expensed over the remainder of the trials. Under the master agreement with these vendors, the agreements may be terminated upon sixty days notice to the vendors. If terminated early, we would be responsible for the costs incurred by the vendor through the effective date of the termination plus cancellation charges as stipulated in the various agreements. |
| (3) | During 2008, we signed agreements for services in connection with our ongoing Phase 3 trial to confirm the utility of CORLUX for the treatment of the psychotic features of psychotic depression. The total commitment under these original agreements was approximately $21.1 million. In June 2009, we amended these agreements to reduce the amounts of commitments with these organizations by approximately $5.0 million in accordance with the reduction in the near-term scope of activities under this trial. However, we view the reduction in these commitments as a temporary measure as it is our intent to continue the conduct of this trial to its conclusion, when sufficient capital is available for this purpose. Approximately $7.0 million of these costs was expensed through December 31, 2009, with the remainder to be incurred over the course of the trial. Under the master services agreements with these vendors, the project contracts may be terminated upon thirty to sixty day’s notice. If terminated early, we would be responsible for the costs incurred by the vendors through the effective date of termination plus cancellation charges as stipulated in the agreements. |
| (4) | During the period from October to December 2009, we signed agreements with contract research organizations and other vendors for the conduct of four of the NDA-supportive studies for aggregate commitments of approximately $2.2 million. Approximately $870,000 of costs under these agreements has been expensed as of December 31, 2009, with the remainder to be incurred during 2010. |
| (5) | In October 2008, we renewed the operating lease agreement for our office facility for a two-year term commencing on January 1, 2009. The new lease provides us an option to extend the lease for an additional year upon 180 days notice. |
| (6) | During 2005, we entered into capital leases for the acquisition of certain pieces of office furniture. The obligations under these leases will be fully satisfied during 2010. |
| (7) | Under our cancellable license agreement with Stanford University, we are obligated to make nonrefundable minimum royalty payments of $50,000 annually for as long as we maintain our licenses with Stanford; however, these payments are creditable against future royalties. |
52
Table of Contents
| (8) | Under the agreement with our contract research company, we are obligated to make a payment of $100,000 related to the commencement in February 2010 of the Phase 1 clinical trial evaluating CORT 108297. |
We also have other contractual payment obligations, the timing of which is contingent on future events.
| (a) | Under our license agreement with Stanford University related to the patent covering the use of GR-II antagonists to treat the psychosis associated with psychotic depression and early dementia, we are obligated to make milestone payments to Stanford of $50,000 upon filing of an NDA covering the licensed product and $200,000 upon FDA approval of the licensed product. The milestone payments payable to Stanford under these licenses are creditable against future royalties. |
| (b) | Under the agreement with our contract research company we may be obligated to make milestone payments upon the occurrence of certain events, including: (i) patent filings in connection with the project; (ii) entries into Phase 1 clinical trials; and (iii) national regulatory approval of each product arising from work performed under the agreement, provided that sales of the product by the Company or any future licensees reach $5,000,000. These obligations remain in force after the conclusion of work under the agreement. There are no royalty obligations associated with this contract. |
| (c) | Our agreement with ScinoPharm Taiwan that provides for the manufacture and supply of the active pharmaceutical ingredient for CORLUX includes a minimum purchase commitment of $1,000,000 per year following the commercial launch of CORLUX. This agreement may be terminated by us at any time without penalty. |
| (d) | On November 8, 2006, we signed an agreement with Produits Chimiques Auxiliaires et de Synthese SA (PCAS), for the manufacture of mifepristone, the active pharmaceutical ingredient in CORLUX, for our development and commercial needs for an initial period of five years. The agreement provides for an automatic extension for one additional year unless either party gives twelve month’s prior notice that it does not want the extension. There is no guaranteed minimum purchase commitment under this agreement. If PCAS is unable to manufacture the product for a consecutive six-month period, we have the right to terminate the agreement without penalty. |
In February 2010, we signed an agreement with a clinical research organization for the conduct of a Phase 1 clinical study to evaluate CORT 108297 for a commitment of approximately $690,000 and, in March 2010, we signed agreements with two contract research organizations and a clinical research site for the conduct of two NDA-supportive studies for CORLUX for aggregate commitments of approximately $1.2 million. All of these amounts are expected to be expended in 2010.
Net Operating Loss Carryforwards
At December 31, 2009 we had approximately $76.4 million of federal net operating loss carryforwards and approximately $4.9 million in federal research and development tax credit carryforwards, as well as approximately $76.7 million of California net operating loss carryforwards and approximately $1.3 million in California research and development tax credit carryforwards, available to offset any future taxable income we may generate. The federal and California net operating loss and tax credit carryforwards will expire beginning in 2019 and 2012, respectively. Our deferred tax assets have been offset by a full valuation allowance as the realization of such assets is uncertain. The Internal Revenue Code of 1986, as amended, places certain limitations on the annual amount of net operating loss and tax credit carryforwards that can be utilized in any particular year if certain changes in our ownership occur.
Off-Balance Sheet Arrangements
None.
53
Table of Contents
Critical Accounting Policies and Estimates
Our financial statements have been prepared in accordance with U.S. generally accepted accounting principles. The preparation of these financial statements requires us to make estimates and judgments that affect the reported amounts of assets, liabilities and expenses. We base our estimates on historical experience and on various other assumptions that we believe to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions.
Accruals of Research and Development Costs. We recorded accruals for estimated costs of research, pre-clinical and clinical studies, and manufacturing development of approximately $710,000 and $990,000 as of December 31, 2009 and 2008, respectively. These costs are a significant component of our research and development expenses. We make significant judgments and estimates in determining the accrual balance in each reporting period. Accrued clinical trial costs are based on estimates of the work completed under the service agreements, milestones achieved, patient enrollment and past experience with similar contracts and service providers. Our estimate of the work completed, and associated costs to be accrued, includes our assessment of the information received from our third-party contract research organizations and the overall status of our clinical trial activities. In the past, we have not experienced any material deviations between accrued clinical trial expenses and actual clinical trial expenses. However, actual services performed, number of patients enrolled and the rate of patient enrollment may vary from our estimates, resulting in adjustments to clinical trial expense in future periods.
Stock-based compensation. Stock-based compensation arises from the granting of stock options to employees and directors, as well as to non-employees.
Employees and directors
Our accounting practices and the estimates and judgments that are considered in determining fair value in regard to stock option grants to employees and directors are as follows:
| • | Options granted subsequent to January 1, 2006: |
| ¡ | The grant date fair value for all new grants issued after January 1, 2006 is being amortized to expense using the straight-line method over the vesting period of the options. |
| ¡ | The expected term used in determining the fair value for options is based on the “simplified” method prescribed by the SEC that considers the weighted average of the vesting period and contractual life of the options. There has been no adjustment made to the expected term to adjust for employees’ expected exercise and expected post-vesting termination behavior because we have a limited employee base and do not have sufficient historical information to determine such an adjustment. |
| ¡ | The expected volatility of our common stock used in determining the fair value of option grants is based on a weighted-average combination of the volatility of our own stock price and that of a group of peer companies since we do not have sufficient historical data from which to base an appropriate volatility assumption. |
| • | Options granted prior to January 1, 2006: |
| ¡ | For options granted prior to our initial public offering (IPO) in 2004, we have continued to account for the portion of these grants that were non-vested as of January 1, 2006 based on the intrinsic value of these grants. |
| ¡ | For the options granted after the IPO, we began, as of January 1, 2006, to record non-cash stock-based compensation expense in the financial statements in amounts that represent the remaining fair value of the non-vested portion of these grants. |
54
Table of Contents
| ¡ | For all options granted prior to January 1, 2006, we are continuing to utilize the graded-vesting attribution method for amortization of the relevant compensation amounts. |
| • | Since we have a limited employee base, we do not have sufficient historical information to determine a reasonable forfeiture rate for options that might not vest because of employee terminations and, therefore, do not apply a forfeiture rate. When an employee terminates, we will record a change in accounting estimate that represents the difference between the expense recorded in the financial statements and the expense that would have been recorded based upon the rights to options that vested during the individual’s service as an employee. |
As of December 31, 2009, the Company had approximately $4.1 million of unrecognized compensation expense for employee options outstanding as of that date, which had a remaining weighted-average vesting period of 2.3 years.
Non-employees
Stock-based compensation related to option grants to non-employees is charged to expense on a straight line basis over the vesting period of the options, based on the fair value of the options, which approximates the period over which the related services are rendered, using the Black-Scholes option pricing model. The assumptions used in these calculations are similar to those used for the determination of fair value for options granted to employees, with the exception that, for non-employee options, we are required to use the remaining contractual term as the life of the option and the fair value related to unvested non-employee options is re-measured quarterly, based on the then current stock price as reflected on the Nasdaq Capital Market.
Recently Adopted Accounting Standards
In October 2009, the Financial Accounting Standards Board (FASB) adopted a change in the accounting standards applicable to revenue arrangements with multiple deliverables. This change eliminates the residual method of allocation and the requirement to use the relative selling price method when allocating revenue in a multiple deliverable arrangement. When applying the relative selling price method, the selling price for each deliverable needs to be determined using vendor specific objective evidence of selling price, if it exists, otherwise third-party evidence of selling price. If neither vendor specific objective evidence nor third-party evidence of selling price exists for a deliverable, it is permissible to use the best estimate of the selling price for that deliverable when applying the relative selling price method. This change is to be applied on a prospective basis for revenue arrangements entered into or materially modified in fiscal years beginning on or after June 15, 2010, with earlier application permitted. As the Company does not have any such agreements at this time, this change is not expected to have an impact on our financial statements.
Recently Issued Accounting Standards
In September 2009, the Emerging Issues Task Force of the Financial Accounting Standards Board (EITF) discussed possible changes to the standard regarding the milestone method of revenue recognition. Under the milestone method, the additional consideration earned from achievement of the milestone is viewed as being indicative of the value provided to the customer through either (a) the efforts performed by the vendor or (b) a specific outcome resulting from the vendor’s performance to achieve that specific milestone. Under the milestone method an entity recognizes contingent arrangement consideration earned from the achievement of a milestone in its entirety in the period in which the milestone is achieved. The proposed model requires that a milestone be “substantive” before this method can be applied; that is, there is a substantial uncertainty about the achievement of the milestone, substantive effort is required to achieve the milestone, and none of the payment for the milestone is refundable. The EITF did not reach a consensus on this issue. The task force will continue deliberations on this issue at future meetings. The adoption of such a standard is not expected to have an impact on our financial statements as we currently have no such arrangements.
55
Table of Contents
| ITEM 7A. | QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK |
Quantitative and Qualitative Disclosures About Market Risk
Market Risk
The primary objective of our investment activities is to preserve principal while at the same time maximizing the income we receive from our investments without significantly increasing risk of loss. As of December 31, 2009, our cash and cash equivalents consisted primarily of money market funds maintained at major U.S. financial institutions To minimize our exposure to interest rate risk, we have limited the maturities of our investments to less than two years with an average maturity not to exceed one year. Due to the short-term nature of these instruments, a 1% increase or decrease in market interest rates would not have a material impact on the total value of our portfolio as of December 31, 2009.
| ITEM 8. | FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA |
The financial statements required by this item are set forth beginning at page F-1 of this report and are incorporated herein by reference.
| ITEM 9. | CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE |
None.
ITEM 9A(T). CONTROLS AND PROCEDURES
| (a) | Conclusion Regarding the Effectiveness of Disclosure Controls and Procedures |
Evaluation of disclosure controls and procedures. We maintain disclosure controls and procedures that are designed to ensure that information required to be disclosed in our periodic and current reports that we file with the SEC is recorded, processed, summarized and reported within the time periods specified in the SEC’s rules and forms, and that such information is accumulated and discussed with our management, including our Chief Executive Officer, Chief Financial Officer and Chief Accounting Officer, as appropriate, to allow timely decisions regarding required disclosure. In designing and evaluating the disclosure controls and procedures, management recognized that any controls and procedures, no matter how well designed and operated, can provide only reasonable and not absolute assurance of achieving the desired control objectives. In reaching a reasonable level of assurance, management necessarily was required to apply its judgment in evaluating the cost-benefit relationship of possible controls and procedures. In addition, the design of any system of controls also is based in part upon certain assumptions about the likelihood of future events, and there can be no assurance that any design will succeed in achieving its stated goals under all potential future conditions; over time, control may become inadequate because of changes in conditions, or the degree of compliance with policies or procedures may deteriorate. Because of the inherent limitations in a cost-effective control system, misstatements due to error or fraud may occur and not be detected.
As of December 31, 2009, our Chief Executive Officer, Chief Financial Officer and Chief Accounting Officer have evaluated our disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) of the Exchange Act) which were designed to ensure that the information required to be disclosed by us in this Annual Report on Form 10-K was recorded, processed, summarized and reported within the time periods specified in the SEC’s rules and Form 10-K. Our disclosure controls and procedures are designed to provide reasonable, not absolute, assurance that the objectives of our disclosure control system are met. Based on the evaluation, our Chief Executive Officer, Chief Financial Officer and Chief Accounting Officer have concluded that our disclosure controls and procedures are effective.
56
Table of Contents
| (b) | Management’s Report on Internal Control Over Financial Reporting |
Our management is responsible for establishing and maintaining adequate internal control over financial reporting, as such term is defined in Exchange Act Rule 13a-15(f). Our internal control system is designed to provide reasonable assurance regarding the preparation and fair presentation of financial statements for external purposes in accordance with generally accepted accounting principles. All internal control systems, no matter how well designed, have inherent limitations and can provide only reasonable assurance that the objectives of the internal control system are met.
Under the supervision and with the participation of our management, including our Chief Executive Officer, Chief Financial Officer and Chief Accounting Officer, we conducted an evaluation of the effectiveness of our internal control over financial reporting, based on criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO). Based on our evaluation, we concluded that our internal control over financial reporting was effective as of December 31, 2009.
This annual report does not include an attestation report of our independent registered public accounting firm regarding internal control over financial reporting. Management’s report on internal controls over financial reporting was not subject to attestation by our independent registered public accounting firm pursuant to temporary rules of the Securities and Exchange Commission that permit us to provide only management’s report in this annual report.
(c) Changes in internal controls. There were no changes in our internal controls over financial reporting during the quarter ended December 31, 2009 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
| ITEM 9B. | OTHER INFORMATION |
None.
57
Table of Contents
| ITEM 10. | DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE |
Board of Directors
The following table sets forth, as of December 31, 2009, the name, age and occupation of each member of our Board of Directors:
| Name |
Age | Occupation | ||
| James N. Wilson(3) |
66 | Chairman of the Board of the Company | ||
| Joseph K. Belanoff, M.D. |
52 | Chief Executive Officer of the Company | ||
| G. Leonard Baker, Jr.(2) |
67 | Venture Capitalist | ||
| Joseph C. Cook, Jr.(1) (3) |
67 | Investor | ||
| Patrick G. Enright(1) |
48 | Venture Capitalist | ||
| James A. Harper(2) |
62 | Retired Pharmaceutical Executive | ||
| David L. Mahoney(1)(2) |
55 | Private Equity Investor | ||
| Edward E. Penhoet, Ph. D.(4) |
69 | Venture Capitalist |
| (1) | Member of audit committee |
| (2) | Member of compensation committee |
| (3) | Member of nominating and corporate governance committee |
| (4) | Dr. Penhoet resigned from our Board of Directors, effective January 5, 2010 |
The directors are elected at each annual meeting of stockholders, or special meeting in lieu thereof. The directors serve for a one-year term until the next annual meeting of stockholders and until their successors are elected and qualified. In addition to the information presented below regarding each director’s specific experience, qualifications, attributes and skills that led our Board to the conclusion that each individual should serve as a director, we also believe that all of our director nominees have a reputation for integrity, honesty and adherence to high ethical standards. They each have demonstrated business acumen and an ability to exercise sound judgment, as well as a commitment of service to us and our Board of Directors. The Board of Directors believes that the backgrounds and qualifications of the directors, considered as a group, provides a significant composite mix of experience, knowledge and abilities that allows the Board of Directors to fulfill its responsibilities.
James N. Wilson has served as a director and as Chairman of the Board since 1999. In addition, since 2005, Mr. Wilson has been the Chairman of the Board of NuGEN Technologies, Inc., a provider of systems for genomic analysis. From 2002 to 2009, he served as a director of Amylin Pharmaceuticals, Inc., a biopharmaceutical company, and from 1996 to 2001 Mr. Wilson was Chairman of the Board of Amira Medical, Inc., which was acquired by Hoffmann-La Roche A.G. From 1991 to 1994, he was Chief Operating Officer of Syntex Corporation, which was acquired by Roche Holding, Ltd. From 1989 to 1990, Mr. Wilson was Chairman and Chief Executive Officer of Neurex Corporation, which was acquired by Elan Corporation plc, and from 1982 to 1988, Mr. Wilson was Chief Executive Officer of LifeScan, Inc., which was acquired by Johnson & Johnson Company. Mr. Wilson received his B.A. and M.B.A. from the University of Arizona. Mr. Wilson brings to the Board of Directors extensive experience in the biotechnology industry, evidenced by nearly 30 years of representing biotechnology companies as a director or officer.
Joseph K. Belanoff, M.D. is a co-founder and has served as a member of the Board and as our Chief Executive Officer since 1999. Dr. Belanoff is currently a clinical faculty member and has held various positions in the Department of Psychiatry and Behavioral Sciences at Stanford University since 1992. From 1997 to 2001, he served as the Director of Psychopharmacology at the outpatient division of the Palo Alto Veterans Affairs Hospital. Dr. Belanoff received his B.A. from Amherst College and his M.D. from Columbia University’s College of Physicians & Surgeons. As our Chief Executive Officer, Dr. Belanoff brings expertise and knowledge regarding our business and operations to our Board of Directors. Dr. Belanoff also has expertise in clinical medicine and psychopharmacology.
58
Table of Contents
G. Leonard Baker, Jr. has served as a member of the Board of Directors since 1999. Since 1973, Mr. Baker has been a Managing Director of the General Partner of Sutter Hill Ventures, a venture capital firm. Mr. Baker currently serves on the boards of a number of private companies. Mr. Baker received his B.A. from Yale University and his M.B.A. from Stanford University. Mr. Baker has broad experience in advising companies, including expertise in capital raising, strategic transactions and operations.
Joseph C. Cook, Jr. has served as a member of the Board of Directors since 2002. Mr. Cook served as Chairman of the Board of Amylin Pharmaceuticals, Inc. from 2003 to 2009 and as its Chief Executive Officer from 1998 to 2003. Mr. Cook is a founder and currently serves as Chairman of the Board of Ironwood Pharmaceuticals, Inc. Mr. Cook is an officer of Mountain Ventures, Inc. and a founder of Clinical Products, Inc. and Mountain Group Capital, LLC. Mr. Cook also serves as a director of Mercy Ministries International and as a member and past Chairman of the University of Tennessee College of Engineering Advisory Board. Mr. Cook retired as Group Vice President of Eli Lilly in 1993 after more than 28 years of service. Mr. Cook received his B.S. from the University of Tennessee. Mr. Cook brings to our Board of Directors extensive experience in the pharmaceutical industry.
Patrick G. Enright has served as a member of the Board of Directors since April 2008. He is a founder of Longitude Capital Management Co., LLC, a venture capital firm focused on investments in biotechnology and has served as its Managing Director since 2006. From 2002 through 2006, Mr. Enright was a Managing Director of Pequot Ventures where he co-led the life sciences investment practice. Prior to Pequot, he was a Managing Member responsible for the Delta Opportunity Fund, where he invested in privately-held and publicly-traded biotechnology companies, such as SUGEN, Inc. and Cephalon, Inc. Mr. Enright began his investment career at PaineWebber Development Corporation, a direct investment group focused primarily on biotechnology companies. Mr. Enright also has significant life sciences operations experience. He was CFO and Senior Vice President Business Development of Valentis, Inc. (now Urigen Pharmaceuticals, Inc.) and Senior Vice President Finance and Business Development of Boehringer Mannheim Pharmaceuticals (now Hoffmann-La Roche). Mr. Enright began his life sciences career 23 years ago at Sandoz (now Novartis). He currently serves on the boards of Corcept and a number of privately-held companies. Mr. Enright has served within the last five years, on the Board of Directors of Infacare Pharmaceuticals, Xanodyne Pharmaceuticals, Jazz Pharamceuticals, Threshold Pharmaceuticals, Sequenom and Valentis. Mr. Enright holds an M.B.A. from the Wharton School of Business at the University of Pennsylvania and a B.S. in Biological Sciences from Stanford University. Mr. Enright has extensive knowledge of finance and experience in the biotechnology industry.
James A. Harper has served as a member of the Board of Directors since October 2004. Mr. Harper held various positions with Eli Lilly, from which he retired in 2004. Mr. Harper served as Group Vice President and Chief Marketing Officer from 2001 to 2004 and as President, Diabetes and Growth Disorders Business Unit / Product Group from 1994 to 2001. He was a Vice President, Global Pharmaceutical Marketing, from 1993 to 1994 and was President and CEO, Advanced Cardiovascular Systems, Inc. from 1991 to 1993. Mr. Harper also serves on the Board of Directors of Zymogenetics, Inc., including membership on the Audit and Compensation Committees, and the Board of Directors of Phenomix Corporation, where he serves as the Chairman of the Board and a member of the Compensation Committee. Zymogenetics, Inc. and Phenomix Corporation are both biotechnology companies. Mr. Harper served on the Board of Directors of Anesiva, Inc., a biotechnology company, from 2007 through 2008, including as a member of the Compensation Committee. He is also an advisor for Nomura Phase4 Ventures. Mr. Harper received his B.A. from Vanderbilt University and his M.B.A. from The Wharton School of Business. He has spent over 30 years in the pharmaceutical and healthcare industries and expertise in marketing,
David L. Mahoney is a private equity investor who has served as a member of the Board since July 2004. From 1999 to 2001, Mr. Mahoney served as co-CEO of McKesson HBOC, Inc., a healthcare supply management and information technology company and as CEO of iMcKesson LLC, a healthcare management and connectivity company. He joined McKesson Corporation in 1990 as Vice President for Strategic Planning. Prior to joining McKesson, Mr. Mahoney was a principal with McKinsey & Company where he worked from 1981 to 1990. He also serves on the Board of Directors of Symantec Corporation, Adamas Pharmaceuticals,
59
Table of Contents
San Francisco Museum of Modern Art, Mercy Corps and NCPB, Inc., a public television and radio operator. Mr. Mahoney served on the Board of Directors of Tercica, Inc., a pharmaceutical company, from 2004 through 2008, including as a member of the Audit and Compensation committees. Tercica was acquired by the Ipsen Group in 2008. Mr. Mahoney received his B.A. from Princeton University and his M.B.A. from Harvard University. Mr. Mahoney brings to our Board of Directors extensive experience in operating and advising technology companies.
Edward E. Penhoet served as a member of the Board from June 2008 to January 2010. He has served as a Director of Alta Partners, LLP, a venture capital firm, since 2000. He is a member of the Boards of Directors of ChemoCentryx, Chimerix, Immune Design Corp, Scynexis, and ZymoGenetics, and serves as the Chairman of the Board for Metabolex. A co-founder of Chiron, he served as the company’s President and Chief Executive Officer from its formation in 1981 until April 1998. Dr. Penhoet is a member of the Independent Citizens Oversight Committee for the California Institute of Regenerative Medicine (CIRM), and recently served as the as President of the Gordon and Betty Moore Foundation. For 10 years prior to founding Chiron, he was a faculty member of the Biochemistry Department of the University of California, Berkeley. Dr. Penhoet is the immediate past Dean of the School of Public Health at the University of California, Berkeley. He is a member of the Institute of Medicine of the National Academy of Sciences and has co-authored more than 50 scientific articles and papers. Dr. Penhoet was recently appointed to President Obama’s Council of Advisors on Science and Technology (PCAST). Due to time pressures from this appointment to PCAST, Dr. Penhoet resigned from our Board of Directors, effective January 5, 2010,
There are no family relationships among any of our directors or executive officers.
Executive Officers
The following table sets forth, as of December 31, 2009, information about our executive officers:
| Name |
Age | Position | ||
| Joseph K. Belanoff, M.D. |
52 | Chief Executive Officer and Director | ||
| Robert L. Roe, M.D. |
69 | President and Secretary | ||
| Caroline M. Loewy |
43 | Chief Financial Officer | ||
| Anne M. LeDoux |
62 | Vice President, Controller and Chief Accounting Officer |
Joseph K. Belanoff, M.D.’s background is discussed above.
Robert L. Roe, M.D. joined us as President in October 2001. Dr. Roe has spent more than 30 years in the pharmaceutical and biotechnology industries. From 1999 to 2001, he served as President and Chief Executive Officer of Allergenics, Inc. From 1996 to 1999, he was Executive Vice President, Chief Operating Officer and a director of Cytel Corporation. From 1995 to 1996, he was Executive Vice President, Chief Operating Officer and a director of Chugai Biopharmaceuticals, Inc. From 1992 to 1995, Dr. Roe served as President of the Development Research Division and Senior Vice President of Syntex Corporation. Dr. Roe received his B.A. from Stanford University and his M.D. from the University of California, San Francisco.
Caroline M. Loewy joined us as Chief Financial Officer in November 2008. From 2006-2008, Ms. Loewy served as Chief Financial Officer of Poniard Pharmaceuticals, a publicly traded biopharmaceutical company. From 2004-2006 she acted as an independent consultant to a variety of biopharmaceutical companies advising on corporate strategy, business development, and financing. Ms. Loewy spent 14 years in equity research and corporate finance. From 2000-2004 she was an Executive Director in biotechnology equity research at Morgan Stanley, providing fundamental analysis and recommendations to investors, as well as strategic advisory services to corporate clients. She was also a Managing Director in biotechnology equity research at Prudential Securities and held positions in corporate finance at BankAmerica. Ms. Loewy holds a BA degree from the University of California, Berkeley, and an MBA/MS degree from Carnegie Mellon University.
60
Table of Contents
Anne M. LeDoux joined the company as Controller in 2004 and was promoted to the position of Vice President, Controller and Chief Accounting Officer in April 2007. Ms. LeDoux has over 15 years of financial and accounting management experience with public pharmaceutical and biotechnology companies. Prior to joining Corcept in 2004, Ms. LeDoux served in various financial positions at Aviron, Roche Biosciences and Syntex Corporation. She was also Vice President and Chief Financial Officer at the Northern California Health Center and Vice President, Finance for the Children’s Hospital of San Francisco. Ms. LeDoux is a Certified Public Accountant and has over 13 years of experience in public accounting, primarily at Coopers and Lybrand. Ms. LeDoux received her Bachelor of Arts degree in Business from the University of Massachusetts and a law degree from Western New England College, School of Law.
Board Meetings and Committees
The Board met five times during 2009. The Audit Committee met four times and the Compensation Committee met twice. The Nominating and Corporate Governance Committee did not meet during 2009. Each member of the Board attended 75% or more of the total number of Board meetings and meetings of Board committees on which such Board member served.
The Board has standing Audit, Compensation and Nominating and Corporate Governance Committees as described below.
Audit Committee. The Audit Committee currently consists of David L. Mahoney (chairman), Joseph C. Cook, Jr. and Patrick G. Enright. The Board has determined that all members of the Audit Committee are independent directors under the rules of the Nasdaq Capital Market and each of them is able to read and understand fundamental financial statements. In addition, the Board has determined that each member of the Audit Committee also satisfies the independence requirements of Rule 10A-3(b)(1) of the Exchange Act. The Board has determined that David L. Mahoney qualifies as an “Audit Committee financial expert” as defined by Item 407(d)(5) of Regulation S-K of the Securities Act and the Exchange Act. The purpose of the Audit Committee is to oversee the accounting and financial reporting processes and financial statements audits. The responsibilities of the Audit Committee include appointing and providing the compensation of the independent accountants to conduct the annual audit of our accounts, reviewing the scope and results of the independent audits, reviewing and evaluating internal accounting policies, and approving all professional services to be provided to us by our independent auditors.
Compensation Committee. The Compensation Committee currently consists of G. Leonard Baker, Jr. (chairman), James A. Harper and David L. Mahoney. Allen Andersson was also a member of this committee until his term as a director was concluded at the Annual Meeting on June 11, 2009. The Board has determined that all members of the Compensation Committee are independent directors under the rules of the Nasdaq Capital Market. The Compensation Committee administers our benefit plans, reviews and administers all compensation arrangements for executive officers, and establishes and reviews general policies relating to the compensation and benefits of our officers and employees.
Nominating and Corporate Governance Committee. Our Nominating and Corporate Governance Committee consists of Joseph C. Cook, Jr. (chairman), and James N. Wilson. The Nominating and Governance Committee is responsible for identifying individuals qualified to serve as members of the Board, recommending to the independent members of the Board nominees for election as our directors and providing oversight with respect to corporate governance and ethical conduct. Although Mr. Wilson is our employee and therefore not an “independent director” for NASDAQ purposes, our director nomination process meets applicable NASDAQ requirements because our director nominees are selected by the independent members of the Board.
Communications with Directors
Stockholders or other interested parties may communicate with any director or committee of the Board by writing to them c/o Secretary, Corcept Therapeutics, 149 Commonwealth Drive, Menlo Park, California 94025.
61
Table of Contents
Comments or questions regarding our accounting, internal controls or auditing matters will be referred to members of the Audit Committee. Comments or questions regarding the nomination of directors and other corporate governance matters will be referred to members of the Nominating and Governance Committee.
We encourage our directors to attend the annual stockholder meetings. Seven of our directors attended the 2009 annual meeting.
Code of Ethics
We have adopted a code of ethics that applies to all officers and employees, including its principal executive officer, principal financial officer and controller. This code of ethics has been filed as Exhibit 14.1 to our Registration Statement on From Form S-1/A (Registration No. 333-112676) filed with the SEC on March 19, 2004. We will also deliver a copy of our code of ethics to any stockholder, without charge, upon written request to Corcept Therapeutics, 149 Commonwealth Drive, Menlo Park, California 94025, Attention: Secretary, or upon oral request by calling (650) 327-3270.
Section 16(a) Beneficial Ownership Reporting Compliance
Under Section 16(a) of the Exchange Act and SEC rules, our directors, executive officers and beneficial owners of more than 10% of any class of equity security are required to file periodic reports of their ownership, and changes in that ownership, with the SEC. Based solely on our review of copies of these reports and representations of such reporting persons, we believe that during 2009, such SEC filing requirements were satisfied
| ITEM 11. | EXECUTIVE COMPENSATION |
Compensation Discussion and Analysis
Compensation Objectives
For Joseph K. Belanoff, M.D., our Chief Executive Officer, Robert L. Roe, M.D., our President, Caroline M. Loewy, our Chief Financial Officer, and Anne LeDoux, our Vice President and Controller (Chief Accounting Officer), our named executive officers (NEOs), compensation is intended to be performance-based, with the exception of such NEOs’ base salary. The Compensation Committee believes that compensation paid to NEOs should be closely aligned with our performance on both a short-term and long-term basis, linked to specific, measurable results intended to create value for stockholders, and that such compensation should assist us in attracting and retaining key executives critical to our long-term success.
In establishing compensation for executive officers, the following are the Compensation Committee’s objectives:
| • | Attract and retain individuals of superior managerial talent; |
| • | Ensure senior officer compensation is aligned with the Company’s corporate strategies, business objectives and the long-term interests of the Company’s stockholders; |
| • | Increase the incentive to achieve key strategic and financial performance measures by linking incentive award opportunities to the achievement of performance goals in these areas; and |
| • | Align officer and shareholder interests, as well as promote retention of key people, by providing a portion of total compensation opportunities for senior management in the form of direct ownership in the Company through stock options. |
Our overall compensation program is structured to attract, motivate and retain highly qualified executive officers by paying them competitively, consistent with the Company’s success and their contribution to that
62
Table of Contents
success. We believe compensation should be structured to ensure that a portion of compensation opportunity will be directly related to Company stock performance and other factors that directly and indirectly influence stockholder value. Accordingly, we set goals designed to link each NEO’s compensation to our performance, such as the attainment of clinical goals and meeting agreed upon financial targets.
We provide a base salary to our executive officers. Additionally, consistent with our performance-based philosophy, we reserve the largest potential compensation awards for performance- and incentive-based programs for our senior executive management team, comprised of the Chief Executive Officer, President, Chief Financial Officer and Chief Accounting Officer. Such programs include stock option grants, designed to provide compensation opportunities if milestones that increase our value, such as positive results in clinical trials, are attained. Incentive-based programs provide compensation in the form of both cash and equity, to reward for both short-term and long-term performance. The Compensation Committee allocates total compensation between cash and equity compensation based on the Compensation Committee members’ knowledge of compensation practices in the biotechnology and specialty pharmaceutical industries. The balance between equity and cash compensation among members of the senior executive management team, all four of whom are NEOs, is evaluated annually to align the interests of management with stockholders through both short and long term incentives.
The Chairman of the Board and the members of the Compensation Committee are seasoned executives of, consultants to or venture capitalists with investments in the biotechnology and specialty pharmaceutical industry. Collectively they have served as board and compensation committee members of many public and privately held companies including Amylin Pharmaceuticals, Inc., NuGen Technologies, Inc., Neurex Corporation, Praecis Pharmaceuticals, Inc., Tercica, Inc., and Zymogenetics Inc. As a result of this extensive involvement in the compensation of executives in these and other companies, the Chairman of the Board and the members of the Compensation Committee collectively have developed a clear understanding and knowledge of the compensation structures that are necessary to attract, motivate and retain management talent.
Determination of Compensation
The Compensation Committee is provided with the primary authority to determine and recommend the compensation awards available to our executive officers for approval by the Board of Directors. Based on the Compensation Committee members’ collective understanding of compensation practices in similar companies in the biotechnology and specialty pharmaceutical industry, our executive compensation package consists of the following elements, in addition to the employee benefit plans in which all employees may participate:
| • | Base salary: compensation for ongoing performance throughout the year. |
| • | Periodic performance-based cash compensation: awards to recognize and reward achievement of performance goals. |
| • | Long-term performance-based equity incentive program: equity compensation to provide an incentive to the NEOs to manage us from the perspective of an owner with an equity stock in the business. |
| • | Severance and change of control benefits: remuneration paid to executives in the event of a change of control or involuntary employment termination. |
To aid the Compensation Committee in making its determination, our Chief Executive Officer provides recommendations annually to the Compensation Committee regarding the compensation of all other executive officers. Each NEO in turn, participates in an annual performance review with our Chief Executive Officer to provide input about their individual contributions to our success for the period being assessed. The overall performance of our senior executive management team is reviewed annually by the Compensation Committee.
We set base salary structures and any grants of stock options based on the Compensation Committee members’ collective understanding of compensation practices in the biotechnology and specialty pharmaceutical
63
Table of Contents
industry and such members’ experiences as seasoned executives, consultants, board and compensation committee members, or investors in similar biotechnology and specialty pharmaceutical industry companies.
Tax Considerations
A goal of the Compensation Committee is to comply with the requirements of Internal Revenue Code Section 162(m) of the Internal Revenue Code of 1986, as amended, which limits the tax deductibility by us of annual compensation in excess of $1,000,000 paid to our Chief Executive Officer and any of our three other most highly compensated executive officers, other than our Chief Financial Officer. However, performance-based compensation that has been approved by our stockholders is excluded from the $1,000,000 limit if, among other requirements, the compensation is payable only upon the attainment of pre-established, objective performance goals and the committee of our Board of Directors that establishes such goals consist only of “outside directors.” All members of the Compensation Committee qualify as outside directors.
While the tax impact of any compensation arrangement is one factor to be considered, such impact is evaluated in light of the Compensation Committee’s overall compensation philosophy and objectives. The Compensation Committee will consider ways to maximize the deductibility of executive compensation, while retaining the discretion it deems necessary to compensate officers in a manner commensurate with performance and the competitive environment for executive talent. From time to time, the Compensation Committee may award compensation to our executive officers which is not fully deductible if it determines that such award is consistent with its philosophy and is in our and our stockholders’ best interests.
Certain option grants made under our equity plans are intended to be structured so that any compensation deemed paid upon the exercise of those options is intended to qualify as performance-based compensation that is not subject to the $1,000,000 limitation.
Elements of Executive Compensation
Base Compensation
We pay base salaries to provide fixed compensation based on the Compensation Committee’s assessment of competitive market practices. Due to the Compensation Committee’s collective experience with similar companies in the biotechnology and specialty pharmaceutical industry, the Compensation Committee has intricate knowledge and understanding of what the industry demands in order to motivate and retain our executive officers. We provide each NEO with a base salary that was established by extensive negotiations with each NEO when such individual first joined us as an employee or was promoted to the position of executive officer. Base salaries have not changed in 2009 as compared to 2008 other than for annual merit adjustments of 3% per year that were approved by the Compensation Committee and applied equally to all employees. While base salaries are not considered by the Internal Revenue Service to constitute performance-based compensation, each year the Compensation Committee reviews the CEO’s base salary to determine if a change is appropriate based on Company performance, such as our progress on research and development programs. Similarly, the CEO reviews the base salary of the other NEOs and has the ability to propose a change in base salary based on performance to the Compensation Committee. Other than the annual merit increases that the Compensation Committee has approved, no formulaic base salary increases are provided to the NEOs.
Performance-Based Compensation
Performance Goals and Periodic Performance-Based Cash Compensation
We structure our compensation programs to reward executive officers based on the Company’s performance. This allows executive officers to receive bonus compensation in the event certain specified corporate performance measures are achieved. To date, we have not instituted an annual performance-based cash compensation or annual performance-based equity compensation program because the Compensation Committee believes that the compensation objective to ensure that executive officers’ compensation is aligned with our
64
Table of Contents
corporate strategies, business objectives and the long-term interests of our stockholders is achieved when milestone successes are met, such as meeting the predetermined endpoints in our clinical trials. The achievement of these milestones does not necessarily correspond with annual performance periods.
Performance-based cash compensation has been awarded in past years primarily to recognize the attainment of certain accomplishments of value enhancing milestones such as successful financing transactions and positive results in clinical trials. The Compensation Committee believes that performance-based compensation should be based on achievement of certain milestone successes, such as the attainment of predetermined end-points in our clinical trials, successful financing transactions and commencement of certain clinical trials. No bonuses were awarded to the NEOs for 2009.
Long-Term Performance-Based Equity Incentive Program
Our executive officers, along with all of our employees, are eligible to participate in our awarding of stock options under our 2004 Equity Incentive Plan. As discussed above, we believe, with our performance-based approach to compensation, that equity ownership in the Company is important to tie the ultimate level of an executive officer’s compensation to the performance of our stock and stockholder gains while creating an incentive for sustained growth. We have, thus far, only used stock options as the long-term performance-based equity incentive vehicle because the Compensation Committee believes that stock options maximize executive officers’ incentive to increase our stock price and maximize stockholder value (i.e., there is no financial gain to an executive officer unless our stock price appreciates).
Equity compensation in the form of incentive or non-qualified stock options is awarded by the Compensation Committee from time to time. The size and the timing of each grant is based on a number of factors, including the executive officer’s salary, such executive officer’s contributions to the achievement of our financial and strategic objectives, the value of the stock option at the time of grant, the possible value of the option if we achieve our objectives and industry practices and norms from the collective knowledge of the Compensation Committee as seasoned executives of, consultants to, board and compensation members of, and venture capitalists with investments in similar companies in the industry. The relative weight given to each of these factors varies among individuals at the Compensation Committee’s discretion. There is no set formula for the granting of stock options to individual executives and employees. Grants also may be made following a significant change in job responsibility or in recognition of a significant achievement.
Stock options granted to NEOs under the various stock plans generally have a four- or five-year vesting schedule in order to provide an incentive for continued employment and generally expire ten years from the date of the grant. This provides a reasonable time frame in which to provide the executive officer with the possibility of price appreciation of our shares. The exercise price of options granted under the stock plans is 100% of the fair market value of the underlying stock on the date of grant.
During 2009, the Compensation Committee and Board of Directors approved the award of stock option grants to Joseph K. Belanoff, our CEO, Robert L. Roe, our President, and Anne M. LeDoux, our Vice President, Controller and Chief Accounting Officer. A portion of the vesting of the awards to Dr. Belanoff and Dr. Roe in the amounts of 500,000 shares and 200,000 shares, respectively, is subject to a performance-based condition under which these options will vest in their entirety upon the approval of the NDA for the Company’s first product by the FDA. The remainder of the option awards in the amounts of 500,000 shares, 200,000 shares, and 125,000 shares granted respectively to Dr. Belanoff, Dr. Roe and Mrs. LeDoux vest monthly on a pro-rata basis over a four-year period from the date of grant.
We grant all stock option awards based on the fair market as of the date of grant. We do not have a policy of granting stock option awards at other than the fair market value. The exercise price for stock option grants is determined by looking at the fair market value of the last quoted price per share on the Nasdaq Capital Market on the date of grant. We do not have a policy and do not intend to have a policy or practice to select option grant dates for executive officers in coordination with the release of material non-public information.
65
Table of Contents
Severance and Change in Control Arrangements
We entered into Severance and Change in Control Agreements with each of our named executive officers to encourage continued attention and dedication to duties without distraction arising from the possibility of a change in control of our company and provide the business with a smooth transition in the event of a change in control. The terms of the agreements are identical. For a detailed description of the Severance and Change in Control Agreements, see the “Potential Payments Upon Termination or Change in Control – Severance and Change in Control Agreements,” below.
Other Elements of Compensation and Perquisites
401(k) Plan. The Company has a Section 401(k) Savings/Retirement Plan (the “401(k) Plan”) to cover eligible employees of the Company and any designated affiliate. The 401(k) Plan permits our eligible employees to defer up to 100% of their annual compensation, subject to certain limitations imposed by the Internal Revenue Code. The employees’ elective deferrals are immediately vested and non-forfeitable upon contribution to the 401(k) Plan. We currently make no matching contributions to the 401(k) Plan. Employees of the Company are eligible to participate in the 401(k) Plan on the first day of the month coinciding with or immediately following the first day of employment.
Medical Insurance. We, at our sole cost, provide to each employee (including each NEO), and his or her spouse and children such health, dental and optical insurance as we may from time to time make available to our other employees of the same level of employment. Such insurance programs are part of an overall broad-based total compensation program designed to facilitate our ability to attract and retain employees as we compete for talented individuals in the marketplace where such benefits are commonly offered.
Life and Disability Insurance. We provide each employee (including each NEO) such disability and/or life insurance as we in our sole discretion may from time to time make available to our other employees of the same level of employment. Such insurance programs are part of an overall broad-based total compensation program designed to facilitate our ability to attract and retain employees as we compete for talented individuals in the marketplace where such benefits are commonly offered.
The following tables and descriptive materials set forth information concerning compensation earned for services rendered to us by our Chief Executive Officer (the CEO), President, Chief Financial Officer (the CFO) and Chief Accounting Officer (the CAO). The data for Caroline Loewy is included from November 28, 2008, the date of her appointment as our CFO. Collectively these are the “named executive officers” for the respective years.
66
Table of Contents
Summary Compensation Table
The following table provides compensation information for the years ended December 31, 2009, 2008 and 2007 for each of our named executive officers.
| Name and Principal Position |
Year | Salary ($) |
Bonus ($) |
Option Awards(1) ($) |
All Other Compensation ($) |
Total ($) | ||||||||||||
| Joseph K. Belanoff, M.D., |
2009 | $ | 440,272 | — | $ | 909,350 | (2) | $ | 1,349,622 | |||||||||
| 2008 | $ | 427,448 | — | — | — | $ | 427,448 | |||||||||||
| 2007 | $ | 411,008 | $ | 102,752 | $ | 1,130,000 | — | $ | 1,643,760 | |||||||||
| Caroline M. Loewy, |
2009 | $ | 300,000 | — | — | — | $ | 300,000 | ||||||||||
| 2008 | $ | 25,000 | — | $ | 616,000 | — | $ | 641,000 | ||||||||||
| Robert L. Roe, M.D., |
2009 | $ | 405,745 | — | $ | 363,740 | (2) | $ | 1,800 | $ | 771,285 | |||||||
| 2008 | $ | 393,927 | — | — | $ | 900 | $ | 394,827 | ||||||||||
| 2007 | $ | 378,776 | $ | 95,294 | $ | 791,000 | $ | 2,400 | $ | 1,267,470 | ||||||||
| Anne LeDoux,(4) |
2009 | $ | 214,240 | — | $ | 113,588 | $ | 327,828 | ||||||||||
| 2008 | $ | 208,000 | $ | 20,800 | — | — | $ | 228,800 | ||||||||||
| 2007 | $ | 191,777 | $ | 47,944 | $ | 141,250 | — | $ | 380,971 | |||||||||
| (1) | Amounts shown do not reflect compensation actually received by the named executive officers or the actual value that may be recognized by the named executive officers with respect to these awards in the future. Instead, the amounts shown represent the grant date fair value of the awards as of the date of grant. The relevant assumptions used to calculate the value of the option awards are set forth under Part II—Item 8—Financial Statements Notes 1—the Notes to Financial Statements—“Accounting Policies and Estimates—Stock-Based Compensation”. |
| (2) | The stock option grants awarded to Joseph K. Belanoff, M.D. and Robert L. Roe, M.D., are each comprised of 2 parts. One-half of the shares of each award (500,000 shares for Dr. Belanoff and 200,000 shares for Dr. Roe) is a service-based award that vests pro rata over a four-year period at the rate of 2.0834% on the monthly anniversary of the date of grant, until fully vested. The remaining one-half of each award (500,000 shares for Dr. Belanoff and 200,000 shares for Dr. Roe) will vest in its entirety upon the occurrence of the approval of the NDA for the Company’s first product by the FDA. The grant date fair value for these performance grants are $455,000 for the 500,000 share performance award to Dr. Belanoff and $182,000 for the 200,000 share performance award to Dr. Roe. |
| (3) | Caroline Loewy joined us in November 2008 as Chief Financial Officer. |
| (4) | Anne LeDoux serves as Chief Accounting Officer. |
Grants of Plan-Based Awards During 2009
The following table summarizes the grants of stock and option awards we made to the named executive officers in 2009.
| Estimated Future Payouts Under Equity Incentive Plan Awards |
All Other Option Awards: Number of Securities Underlying Options(1) (#) |
Exercise or Base Price of Option Awards ($/Sh) |
Grant Date Fair Value of Stock and Option Awards(2) ($) |
||||||||||||||||
| Name |
Grant Date |
Threshold (#) |
Target (#) |
Maximum (#) |
|||||||||||||||
| Joseph K. Belanoff, M.D. |
3/26/09 | — | 500,000 | (3) | — | 500,000 | (3) | $ | 1.19 | $ | 909,350 | (3) | |||||||
| Caroline M. Loewy |
— | — | — | — | — | — | — | ||||||||||||
| Robert L. Roe, M.D. |
3/26/09 | — | 200,000 | (3) | — | 200,000 | (3) | $ | 1.19 | $ | 363,740 | (3) | |||||||
| Anne LeDoux |
3/26/09 | — | — | — | 125,000 | (4) | $ | 1.19 | $ | 113,588 | |||||||||
| (1) | The options were granted under our 2004 Equity Incentive Plan. |
| (2) | The value of the option award is based on the fair value as of the grant date of the award multiplied by the number of shares. Refer to Notes 1—“Accounting Policies and Estimates—Stock-Based Compensation” included in the Part II—Item 8—Financial Statements in this Annual Report on Form 10-K for the relevant assumptions used to determine the valuation of our option awards. |
| (3) | The stock option grants awarded to Joseph K. Belanoff, M.D. and Robert L. Roe, M.D., are each comprised of 2 parts. One-half of the shares of each award (500,000 shares for Dr. Belanoff and 200,000 shares for Dr. Roe) is a service-based award that vests pro rata over a four-year period at the rate of 2.0834% on the monthly anniversary of the date of grant, until fully vested. The remaining one-half of each |
67
Table of Contents
| award (500,000 shares for Dr. Belanoff and 200,000 shares for Dr. Roe) will vest in its entirety upon the occurrence of the approval of the NDA for the Company’s first product by the FDA. The grant date fair value for these performance grants are $455,000 for the 500,000 share performance award to Dr. Belanoff and $182,000 for the 200,000 share performance award to Dr. Roe. |
| (4) | The stock option grants awarded to Ms. LeDoux is a service-based award that vests pro rata over a four-year period at the rate of 2.0834% on the monthly anniversary of the date of grant, until fully vested. |
Outstanding Equity Awards At Fiscal Year-End
The following table summarizes unexercised options that have not vested and related information for each of our named executive officers as of December 31, 2009.
| Option Awards | ||||||||||||||
| Name |
Number of Securities Underlying Unexercised Options Exercisable (#) |
Number of Securities Underlying Unexercised Options Unexercisable (#) |
Equity Incentive Plan Awards: Number of Securities Underlying Unexercised Unearned Options (#) |
Option Exercise Price ($) |
Option Expiration Date | |||||||||
| Joseph K. Belanoff, M.D. |
666,688 93,753 |
(3) (4) |
333,312 406,247 |
(3) (4) |
— 500,000 |
(4) |
$ $ |
1.50 1.19 |
4/16/2017 3/26/2019 | |||||
| Caroline M. Loewy |
216,673 | (2) | 583,327 | (2) | — | $ | 1.02 | 11/28/2018 | ||||||
| Robert L. Roe, M.D. |
10,000 100,000 96,820 46,876 466,681 37,501 |
(1) (1) (1) (2) (3) (4) |
— — 3,180 3,124 233,319 162,499 |
(1) (2) (3) (4) |
— — — — — 200,000 |
(4) |
$ $ $ $ $ $ |
0.10 7.00 4.82 4.95 1.50 1.19 |
10/1/2010 11/23/2013 2/10/2015 3/2/2016 4/16/2017 3/26/2019 | |||||
| Anne M. LeDoux |
17,500 42,500 12,769 83,336 23,438 |
(1) (1) (1) (3) (3) |
— — 2,231 41,664 101,562 |
(1) (1) (3) |
— — — — — |
|
$ $ $ $ $ |
12.00 7.73 5.70 1.50 1.19 |
4/16/2014 10/6/2014 9/23/2015 4/16/2017 3/26/2019 | |||||
| (1) | The option vests at the rate of 20% at the first anniversary of the grant date and, thereafter, at the rate of 1.67% per month, until fully vested. |
| (2) | The option vests at the rate of 25% at the first anniversary of the grant date and, thereafter, at the rate of 2.0834% per month, until fully vested. |
| (3) | The option vests at the rate of 2.0834% per month until fully vested. |
| (4) | The stock option grants awarded to Joseph K. Belanoff, M.D. and Robert L. Roe, M.D., are each comprised of 2 parts. One-half of the shares of each award (500,000 shares for Dr. Belanoff and 200,000 shares for Dr. Roe) is a service-based award that vests prorata over a four-year period at the rate of 2.0834% on the monthly anniversary of the date of grant, until fully vested. The remaining one-half of each award (500,000 shares for Dr. Belanoff and 200,000 shares for Dr. Roe) will vest in its entirety upon the occurrence of the approval of the NDA for the Company’s first product by the FDA. |
Option Exercises and Stock Vested
None of our named executive officers exercised stock options during 2009. To date, no stock awards have been granted to any of our named executive officers.
Pension Benefits
None of our named executive officers participate in or have account balances in qualified or non-qualified defined benefit plans sponsored by us.
68
Table of Contents
Nonqualified Deferred Compensation
None of our named executives participate in or have account balances in non-qualified defined contribution plans or other deferred compensation plans maintained by us.
Potential Payments Upon Termination or Change of Control
Severance and Change of Control Agreements
We entered into Severance and Change in Control Agreements with each of our named executive officers: Joseph K. Belanoff, M.D., Chief Executive Officer; Robert L. Roe, M.D., President; Caroline M. Loewy, our Chief Financial Officer, and Anne M. LeDoux, Chief Accounting Officer. The terms of the agreements are identical. The agreements provide that, if employment is terminated without cause or for good reason regardless of whether it is in connection with a change in control, the executive will be eligible for 12 months of his or her then current base salary and continued health insurance coverage for such 12-month period. In addition, the agreements provide for the full vesting of all outstanding equity awards in the event the executive employment is terminated without cause or for good reason within 18 months following a change in control. The receipt of any severance will be subject to the executive signing and not revoking a separation agreement and release of claims in a form reasonably acceptable to the Company within sixty (60) days following executive’s termination of employment. No severance pursuant will be paid or provided until the separation agreement and release of claims becomes effective.
The following table reflects compensation payable to each named executive officer under a change in control or various employment termination events. The amounts shown below assume that (i) a change in control of the Company or (ii) each named executive officer terminated employment with the Company, was effective as of December 31, 2009, and estimates the value to the named executive officer as a result of each triggering event.
| Name |
Benefit |
Termination Without Cause |
Involuntary Termination Other Than for Death, Disability or Cause Within 18 Months of Change of Control |
||||||
| Joseph K. Belanoff, M.D. |
Base Salary Accelerated Vesting, of Stock Options(1) Health Benefit |
$
$ |
440,272 — 22,892 |
$ $ $ |
440,272 1,867,572 22,892 |
(2)
| |||
| Caroline M. Loewy |
Base Salary Accelerated Vesting, of Stock Options(1) Health Benefit |
$
$ |
300,000 — 20,246 |
$ $ $ |
300,000 1,026,656 20,246 |
(2)
| |||
| Robert L. Roe, M.D. |
Base Salary Accelerated Vesting, of Stock Options(1) Health Benefit |
$
$ |
405,745 — 17,846 |
$ $ $ |
405,745 875,022 17,846 |
(2)
| |||
| Anne M. LeDoux |
Base Salary Accelerated Vesting, of Stock Options(1) Health Benefit |
$
$ |
214,240 — 28,009 |
$ $ $ |
214,240 214,814 28,009 |
(2)
| |||
| (1) | Assumes that the stock options were not assumed or substituted by the successor entity to the Company or a parent or subsidiary of the successor entity. |
| (2) | For unvested options held by named executive officers as of December 31, 2009, the value ascribed to the change in control acceleration features under the Severance and Change of Control Agreements is calculated as follows: |
| a. | For option grants to these individuals where the closing stock price for the Company’s common stock on the Nasdaq Capital Market as of December 31, 2009 had exceeded the exercise price of the option grant, the value of the acceleration benefit on change of control would has been calculated as the difference between these factors multiplied by the number of unvested shares in each of these option awards as of that date. |
| b. | There is no value ascribed to any unvested shares for any option grants to these individuals where the exercise price of the option grant equaled or exceeded the closing stock price for the Company’s common stock on the Nasdaq Capital Market as of December 31, 2009. |
69
Table of Contents
DIRECTOR COMPENSATION
The following table provides compensation information for the one year period ended December 31, 2009, for each member of our Board of Directors.
| Name |
Fees Earned or Paid in Cash ($) |
Option Awards ($)(1) |
All Other Compensation ($) |
Total ($) | ||||||||||
| James N. Wilson(2) |
— | $ | 716,000 | (2) | $ | 132,081 | (2) | $ | 848,081 | |||||
| Joseph K. Belanoff, M.D.(3) |
— | — | — | — | ||||||||||
| Allen Andersson(4)(5) |
$ | 6,675 | — | — | $ | 6,675 | ||||||||
| G. Leonard Baker, Jr.(4) |
$ | 15,000 | $ | 21,738 | — | $ | 36,738 | |||||||
| Joseph C. Cook, Jr.(4) |
$ | 25,000 | $ | 21,738 | — | $ | 46,738 | |||||||
| Patrick G. Enright(4) |
$ | 25,000 | $ | 21,738 | $ | 46,738 | ||||||||
| James A. Harper(4) |
$ | 15,000 | $ | 21,738 | — | $ | 36,738 | |||||||
| David L. Mahoney(4) |
$ | 25,000 | $ | 36,230 | — | $ | 61,230 | |||||||
| Edward E. Penhoet, Ph.D.(4)(6) |
$ | 15,000 | $ | 21,738 | — | $ | 36,738 | |||||||
| (1) | Amounts shown do not reflect compensation actually received by the directors or the actual value that may be recognized by the directors with respect to these awards in the future. Instead, the amounts shown represent the grant date fair value of the awards. The relevant assumptions used to calculate the value of the option awards are set forth under Notes 1—“Accounting Policies and Estimates—Stock-Based Compensation” included in Part II—Item 8—Financial Statements in this Annual Report on Form 10-K. |
| (2) | Mr. Wilson is an employee director. He receives compensation in his role as an employee providing advice and business insight. The entire amount shown as Other Compensation for Mr. Wilson is salary paid in regard to his services as an employee. He receives no additional compensation in his capacity as a director. During 2009, Mr. Wilson was granted an option for 400,000 shares that vests prorata over a four-year period at the rate of 2.0834% on the monthly anniversary of the date of grant, until fully vested. Including this grant, Mr. Wilson had been granted option awards totaling 650,000 shares as of December 31, 2009. |
| (3) | Dr. Belanoff is a full time employee and a named executive officer and is compensated in that capacity. He receives no additional compensation in his capacity as a director. Dr. Belanoff is a named executive officer. See “Outstanding Equity Awards At Fiscal Year-End” table above for the aggregate number of shares represented by option awards outstanding that have been granted to Dr. Belanoff. |
| (4) | During 2009, Mr. Mahoney, as chairman of the Audit Committee, was granted an option for 50,000 shares. Messrs Baker, Cook, Enright, Harper and Penhoet were each granted an award for 30,000 shares. All of these awards vest prorata over a one-year period at the rate of 8.3334% on the monthly anniversary of the date of grant, until fully vested. Including these grants, the following are the aggregate number of shares represented by option awards outstanding that have been granted to each of our non-employee directors as of December 31, 2009, the last day of the 2009 fiscal year: Mr. Baker: 90,000; Mr. Cook: 135,000; Mr. Enright: 100,000; Mr. Harper: 120,000; Mr. Mahoney: 180,000. Mr. Penhoet’s aggregate options are discussed below. |
| (5) | The term on the Board for Mr. Andersson was completed in June 2009 at the time of our annual meeting. |
| (6) | Dr. Penhoet resigned from our Board of Directors, effective January 5. 2010, due to time pressures from his appointment as a member of President Obama’s Council of Advisors on Science and Technology. Mr. Penhoet had originally been granted option awards for a total of 100,000 shares. As of the date of his resignation, the rights to exercise 41,250 shares had vested and the remainder of the option award was cancelled. |
Non-employee directors receive a director fee from us for their services as members of the Board in the amount of $15,000 per year. Members of the Audit Committee receive an additional $10,000 per year. New directors receive an initial stock option grant of 70,000 shares of our common stock in connection with their initial election to the Board. The initial director options will vest with respect to 25% of the shares on the first anniversary of the date of the grant and, thereafter, at the rate of 2.0834% per month, until fully vested. Non-employee directors who are reelected at the Annual Shareholder Meeting each receive a stock option grant that vests over the one year term as director at the rate of 8.3334% per month from the date of the Annual Meeting until fully vested. The chairmen of the Audit Committee and the Compensation Committee each receive additional grant of our common stock with a similar one-year vesting provision. The amounts of these annual grants are determined each year.
During 2009, the chairman of the Audit Committee received a stock option grant for 50,000 shares of our stock and all other non-employee directors that were reelected in June 2009 received grants of 30,000 shares of our common stock. Directors are reimbursed for certain expenses in connection with attending Board and committee meetings.
70
Table of Contents
We entered into a Severance and Change in Control Agreement with James N. Wilson, Chairman of the Board of Directors. The agreement with Mr. Wilson provides that if his employment or service on the Board terminates involuntarily without cause or good reason within eighteen months of a change in control all of his outstanding equity awards shall become fully vested. Mr. Wilson will only receive severance under this agreement if he signs and does not revoke a separation agreement and release of claims in a form reasonably acceptable to the Company within 60 days following termination of employment. No severance will be provided to Mr. Wilson until the separation agreement and release of claims becomes effective.
Compensation Committee Interlocks and Insider Participation
No interlocking relationship exists, or in the past fiscal year has existed, between any member of the Company’s Compensation Committee and any member of any other company’s board of directors or compensation committee.
Compensation Committee Report
The Compensation Committee of the Board of Directors (the “Compensation Committee”) has furnished this report on executive compensation. None of the members of the Compensation Committee is currently our officer or employee and all are “non-employee directors” for purposes of Rule 16b-3 under the Securities Exchange Act of 1934 and “outside directors” for purposes of Section 162(m) of the Internal Revenue Code. The Compensation Committee is responsible for designing, recommending to the Board of Directors for approval and evaluating our compensation plans, policies and programs and reviewing and approving the compensation of the Chief Executive Officer and other officers and directors.
This report, filed in accordance with Item 407(e)(5) of Regulation S-K, should be read in conjunction with the other information relating to executive compensation which is contained elsewhere in this Annual Report on Form 10-K and is not repeated here.
In this context, the Compensation Committee hereby reports as follows:
1. The Compensation Committee has reviewed and discussed the Compensation Discussion and Analysis section contained herein with management.
2. Based on the review and discussions referred to in paragraph (1) above, the Compensation Committee recommended to our Board of Directors, and our Board of Directors has approved, that the Compensation Discussion and Analysis be included in this Annual Report on Form 10-K for filing with the SEC.
COMPENSATION COMMITTEE
G. LEONARD BAKER, JR., CHAIRMAN
JAMES A. HARPER
DAVID L. MAHONEY
71
Table of Contents
| ITEM 12. | SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS |
Securities Authorized for Issuance under Equity Compensation Plans
The following table provides information as of December 31, 2009 with respect to the shares of our common stock that may be issued under all of our existing equity compensation plans, including the 2004 Equity Incentive Plan and the 2000 Stock Option Plan.
| Plan Category |
(a) Number of Securities to Be Issued upon Exercise of Outstanding Options |
(b) Weighted Average Exercise Price of Outstanding Options |
(c) Number of Securities Remaining Available for Future Issuance under Equity Compensation Plans (Excluding Securities Reflected in Column(a))(2) |
|||||
| Equity compensation plans approved by stockholders |
7,346,636 | $ | 2.28 | 201,044 | (1)(2) | |||
| Equity compensation plans not approved by stockholders |
— | — | — | |||||
| Total |
7,346,636 | $ | 2.28 | 201,044 | ||||
| (1) | Represents shares of common stock remaining available for future issuance under our 2004 Equity Incentive Plan as of December 31, 2009. |
| (2) | The 2004 Equity Incentive Plan contains an “evergreen” provision that allows for increases on the first business day of each fiscal year beginning January 1, the lesser of an additional (i) 4,000,000 shares of our common stock, (ii) 4% of the outstanding shares of common stock on the immediately preceding December 31, or (iii) an amount determined by the Board. None of our other plans has an “evergreen” provision. On December 3, 2009, the Board of Directors authorized an “evergreen” increase in the shares available for grant under the 2004 Plan in the amount of 2,498,987 shares. This increase, which was effective on January 1, 2010, represented 4% of the shares of our common stock outstanding on December 31, 2009. |
72
Table of Contents
Security Ownership of Certain Beneficial Owners and Management
The following table sets forth information regarding ownership of our common stock as of March 15, 2010 or earlier date for information based on filings with the SEC by (a) each person known to us to own more than 5% of the outstanding shares of our common stock, (b) our directors, (c) our Chief Executive Officer and each other executive officer named in the compensation tables appearing earlier in this Form 10-K and (d) all directors and executive officers as a group. The information in this table is based solely on statements in filings with the SEC or other information we believe to be reliable. Percentage of ownership is based on 62,703,717 shares of common stock outstanding as of March 15, 2010. Beneficial ownership is determined in accordance with the rules of the SEC, and includes voting and investment power with respect to the shares. Shares of common stock subject to outstanding options and warrants exercisable within 60 days of March 15, 2010 are deemed outstanding for computing the percentage of ownership of the person holding such options or warrants, but are not deemed outstanding for computing the percentage of any other person.
| Name of Beneficial Owner(1) |
Number of Shares Beneficially Owned(2) |
Percentage of Shares Beneficially Owned |
|||
| 5% Stockholders |
|||||
| Entities and individuals affiliated with Longitude Venture Partners, LP(3) |
14,152,306 | 21.5 | % | ||
| Entities and individuals affiliated with Sutter Hill Ventures(4) |
12,957,146 | 20.1 | % | ||
| Entities affiliated with Alta Partners, LLP(5) |
6,316,212 | 10.0 | % | ||
| Directors and Named Executive Officers |
|||||
| Patrick G. Enright(3) |
14,152,306 | 21.5 | % | ||
| G. Leonard Baker, Jr.(6) |
9,017,553 | 14.1 | % | ||
| Joseph K. Belanoff(7) |
3,649,640 | 5.7 | % | ||
| James N. Wilson(8) |
3,267,902 | 5.2 | % | ||
| Joseph C. Cook, Jr.(9) |
2,980,409 | 4.7 | % | ||
| David L. Mahoney(10) |
1,359,733 | 2.2 | % | ||
| Robert L. Roe(11) |
988,074 | 1.6 | % | ||
| Caroline M. Loewy(12) |
283,342 | * | |||
| Anne M. LeDoux(13) |
201,379 | * | |||
| James A. Harper(14) |
184,900 | * | |||
| All directors and executive officers as a group (10 persons)(15) |
36,085,238 | 51.3 | % | ||
| * | Less than 1% of our outstanding common stock. |
| (1) | Unless otherwise indicated, the address of each of the named individuals is c/o Corcept Therapeutics, 149 Commonwealth Drive, Menlo Park, California 94025. |
| (2) | Beneficial ownership of shares is determined in accordance with the rules of the SEC and generally includes any shares over which a person exercises sole or shared voting or investment power, or of which a person has the right to acquire ownership within 60 days after March 15, 2010. Except as otherwise noted, each person or entity has sole voting and investment power with respect to the shares shown. |
| (3) | Includes (a) 10,830,959 shares held of record by Longitude Venture Partners, LP, and 3,091,479 shares that may be acquired by the entity within 60 days of March 15, 2010 pursuant to warrants (b) 139,326 shares held of record by Longitude Capital Associates, L.P. and 26,583 shares that may be acquired by that entity within 60 days of March 15, 2010 pursuant to warrants, and (c) 63,959 shares issuable within 60 days of March 15, 2010, pursuant to an option to Patrick Enright exercisable within 60 days of March 15, 2010. Mr. Enright disclaims beneficial ownership of all such shares, except to the extent of his pecuniary interest therein. The address for Longitude Capital is 800 El Camino Real, Suite 220, Menlo Park, California 94025. Mr. Enright is a member of our Board of Directors and is a managing member of Longitude Capital Partners, LLC. |
| (4) | Consists of: (a) 5,217,464 shares held by Sutter Hill Ventures, A California Limited Partnership (Sutter Hill Ventures), and 707,752 shares that may be acquired by the entity within 60 days of March 15, 2010 pursuant to warrants, (b) 29,273 shares held by Sutter Hill Entrepreneurs Fund (AI), L.P. (SHAI), (c) 74,113 shares held by Sutter Hill Entrepreneurs Fund (QP), L.P. (SHQP), (d) 205,439 shares of Common Stock owned by G. Leonard Baker, Jr., one of our directors, (e) 1,180,231 shares held by Mr. Baker, as Trustee of The Baker Revocable Trust, and 228,765 shares that may be acquired by the Trust within 60 days of March 15, 2010 pursuant to warrants, (f) 792,268 shares held by Saunders Holdings, L.P. of which Mr. Baker is a General Partner, and 115,015 shares that may be acquired |
73
Table of Contents
| by the entity within 60 days of March 15, 2010 pursuant to warrants, (g) 281,284 shares held by the Sutter Hill Ventures Profit Sharing Plan, for the benefit of Mr. Baker, and 98,449 shares that may be acquired by the entity within 60 days of March 15, 2010 pursuant to a warrant, (h) 87,500 shares issuable within 60 days of March 15, 2010 pursuant to options granted to Mr. Baker and (i) 3,456,808 shares held by individuals other than Mr. Baker who are affiliated with Sutter Hill Ventures and entities affiliated with such individuals, and 482,785 shares that may be acquired by such individuals and entities within 60 days of March 15, 2010 pursuant to warrants. Mr. Baker shares voting and dispositive power with respect to the shares held by Sutter Hill Ventures, SHAI and SHQP with the following natural persons: David L. Anderson, William H. Younger, Jr., Tench Coxe, Gregory P. Sands, James C. Gaither, James N. White, Jeffrey W. Bird, David E. Sweet, Andrew T. Sheehan and Michael L. Speiser. As a result of the shared voting and dispositive powers referenced herein, Messrs. Baker, David L. Anderson, William H. Younger, Jr., Tench Coxe, Gregory P. Sands, James C. Gaither, James N. White, Jeffrey W. Bird, David E. Sweet, Andrew T. Sheehan and Michael L. Speiser may each be deemed to beneficially own the shares held by Sutter Hill Ventures, SHAI and SHQP. Mr. Baker has shared voting and dispositive power with respect to the shares and warrants held by The Baker Revocable Trust and Saunders Holdings, L.P. Mr. Baker, Sutter Hill Ventures, SHAI and SHQP do not have any voting or dispositive power with respect to the shares held by individuals affiliated with Sutter Hill Ventures and entities affiliated with such individuals referenced under part (i) of this note. The address for Sutter Hill Ventures and affiliates is 755 Page Mill Road, Suite A-200, Palo Alto, CA 94304. G. Leonard Baker, Jr., a member of our Board of Directors, is also managing director of the general partner of Sutter Hill Ventures. |
| (5) | Consists of: (a) 5,484,063 shares held of record by Alta BioPharma Partners II, L.P., and 640,996 shares that may be acquired by the entity within 60 days of March 15, 2010 pursuant to warrants, and (b) 180,204 shares held of record by Alta Embarcadero BioPharma Partners II, LLC, and 10,949 shares that may be acquired by the entity within 60 days of March 15, 2010 pursuant to warrants. Alta Parents II, Inc. provides investment advisory services to several venture capital funds including Alta BioPharma Partners II, L.P. and Alta Embarcadero BioPharma Partners II, LLC. The managing directors of Alta BioPharma Partners II, L.P. and the managers of Alta Embarcadero BioPharma Partners II, LLC exercise sole voting and investment power with respect to shares owned by such funds. Certain principals of Alta Partners II, Inc. are managing directors of Alta BioPharma Management II, LLC (which is the general partner of Alta BioPharma Partners II, L.P.), and managers of Alta Embarcadero BioPharma Partners II, LLC. As managing directors and managers of such entities, they may be deemed to share voting and investment powers for the shares held by the funds. The principals of Alta Partners II, Inc. disclaim beneficial ownership of all such shares held by the foregoing funds, except to the extent of their proportionate pecuniary interests therein. Edward E. Penhoet, Ph.D., is a director of Alta BioPharma Management II, LLC (which is a general partner of Alta BioPharma Partners II, L.P.) and a manager of Alta Embarcadero BioPharma Partners II, LLC. Dr. Penhoet disclaims beneficial ownership of all such shares held by all of the foregoing funds, except to the extent of his proportionate pecuniary interests therein. Dr. Penhoet resigned from our Board of Directors, effective January 5. 2010, due to time pressures from his appointment as a member of President Obama’s Council of Advisors on Science and Technology. As of the date of his resignation, 41,250 shares had vested pursuant to options and remain exercisable as of March 15, 2010. These shares pursuant to Mr. Penhoet’s options are not included above in the beneficial ownership of Alta BioPharma Partners II, L.P. The address of Alta Partners II, Inc. is One Embarcadero Center, Suite 3700, San Francisco, California 94111. |
| (6) | Includes all shares referenced in footnote (4) other than the 3,456,808 shares held by individuals affiliated with Sutter Hill Ventures and entities affiliated with such individuals, and 482,785 shares that may be acquired by the individuals and entities within 60 days of March 15, 2010 pursuant to warrants, as referenced under part (i) of footnote (4.) |
| (7) | Includes 300,000 shares held as custodian for Julia E. Belanoff under the California Uniform Transfers to Minors Act over which Dr. Belanoff has voting control, 300,000 shares held as custodian for Edward G. Belanoff and 885,445 shares issuable pursuant to options exercisable within 60 days of March 15, 2010. |
| (8) | Includes 2,074,511 shares held of record by the James N. Wilson and Pamela D. Wilson Trust and 951,774 shares held of record by the James and Pamela Wilson Family Partners, over all of which Mr. Wilson has voting control pursuant to voting agreements. Mr. Wilson disclaims beneficial ownership of such shares, except to the extent of his pecuniary interests in the entities holding such shares. Mr. Wilson’s beneficial interest also includes 17,652 shares that may be acquired by the James and Pamela Wilson Family Partners within 60 days of March 15, 2010 pursuant to a warrant, and 223,965 shares issuable pursuant to options exercisable within 60 days of March 15, 2010. |
| (9) | Includes (a) 1,130,000 shares held of record by Farview Management, Co. L.P., a Texas limited partnership and 14,402 shares that may be acquired by that entity within 60 days of March 15, 2010 pursuant to warrants (b) 414,826 shares held of record by the Joseph C. Cook, Jr., Roth IRA, (c) 86,839 shares that may be acquired by the Joseph C. Cook, Jr., IRA Rollover within 60 days of March 15, 2010, and (d) 132,500 shares issuable pursuant to options exercisable within 60 days of March 15, 2010. Joseph C. Cook, Jr. is a member of our Board of Directors. Data presented does not include holdings of an entity for which Mr. Cook has shared voting power but disclaims beneficial interest in 350,000 shares and 13,995 shares that may be acquired by the entity within 60 days of March 15, 2010 pursuant to warrants. |
| (10) | Includes 1,069,110 shares held of record by the David L. Mahoney and Winnifred C. Ellis 1998 Family Trust, and 114,790 shares that may be acquired by the Trust within 60 days of March 15, 2010 pursuant to warrants, and 175,833 shares issuable pursuant to options exercisable within 60 days of March 15, 2010. David L. Mahoney is a member of our Board of Directors. |
| (11) | Includes 839,184 shares issuable pursuant to options exercisable within 60 days of March 15, 2010. |
| (12) | Includes 283,342 shares issuable pursuant to an option exercisable within 60 days of March 15, 2010. |
74
Table of Contents
| (13) | Includes 201,379 shares issuable pursuant to options exercisable within 60 days of March 15, 2010. |
| (14) | Includes (a) 25,000 shares held of record by the James A. Harper 2008 Annuity Trust over which Mr. Harper is Trustee and has voting control, (b) 25,000 shares held of record by the Zo P. Harper 2008 Annuity Trust over which Mr. Harper’s spouse, Zo P. Harper, is Trustee and has voting control, and (c) 117,500 shares issuable pursuant to options exercisable within 60 days of March 15, 2010. |
| (15) | Total number of shares includes common stock held by entities affiliated with directors and executive officers. See footnotes 1 through 14 above. |
| ITEM 13. | CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE |
On October 12, 2009, we entered into a definitive agreement with certain accredited investors for the private placement of 12,596,475 shares of our common stock and warrants to purchase 4,408,773 shares of our common stock, which we refer to as the October 2009 Financing. The securities were sold at a purchase price of $1.43 per unit, which consisted of one share of common stock and a warrant to purchase 0.35 shares of common stock. The warrants have a three-year term and an exercise price of $1.66 per share. The October 2009 Financing, which closed on October 16, 2009, generated approximately $17.3 million, after deducting costs of issuance.
Investors participating in the October 2009 Financing include funds managed by or investors affiliated with existing stockholders, Longitude Venture Partners, L.P., Sutter Hill Ventures and Alta Partners, LLP, and several other institutional or accredited investors, including Ingalls & Snyder and Federated Kaufmann Funds. The investors also include trusts and other entities related to members of our Board of Directors, including G. Leonard Baker, Jr., Joseph C. Cook, Patrick G. Enright, David L. Mahoney and Edward E. Penhoet, Ph.D., who was a member of our Board of Directors at the time of the October 2009 Financing. Mr. Enright is a managing director of Longitude Venture Partners, L.P. Mr. Baker is a partner and managing director of Sutter Hill Ventures. Dr. Penhoet is a director of Alta Partners, LLP.
In connection with the October 2009 Financing, we entered into a Registration Rights Agreement (the October 2009) Registration Rights Agreement, with the investors participating in the October 2009 Financing. Pursuant to the October 2009 Registration Rights Agreement, we agreed to prepare and file a registration statement with the SEC to register the resale of the shares, the shares of common stock issuable upon exercise of the warrants, and any shares of common stock issued as a dividend or other distribution with respect to the shares or shares underlying the warrants. This registration statement was filed on November 16, 2009 and declared effective by the SEC on January 26, 2010. We also agreed, among other things, to indemnify the selling stockholders under the registration statements from certain liabilities and to pay all fees and expenses (excluding underwriting discounts and selling commissions and all legal fees of any selling stockholder) incident to our obligations under the October 2009 Registration Rights Agreement.
On February 6, 2009, we collected a note receivable of $6.0 million from Paperboy Ventures, LLC that had been issued in March 2008 in connection with the March 2008 Financing. The note was collected in full, including all accrued interest to that date and expenses associated with the note. Allen Andersson, the chairman of Paperboy Ventures, LLC. was a member of our Board of Directors from June 2007 to June 2009.
We have entered into an agreement with Robert L. Roe, M.D., our President, dated October 18, 2001. Pursuant to such letter agreement, Dr. Roe received an option to purchase 250,000 shares of our common stock with an exercise price of $0.75 per share and a loan in the amount of $187,250, subject to interest rate of 6.5% and evidenced by a full-recourse promissory note to us to finance the exercise of the option. Through December 2009, Dr. Roe had repaid $99,705 of the principal of the loan plus accrued interest, leaving a total remaining balance of $87,545 plus accrued interest in the amount of $52,340 for a total combined balance of $ 139,885.
During 2008, we entered into Amended and Restated Severance and Change in Control Agreements with each of our executive officers: Joseph K. Belanoff, M.D., Chief Executive Officer; Robert L. Roe, M.D., President; Caroline M. Loewy, Chief Financial Officer, and Anne M. LeDoux, Chief Accounting Officer. The
75
Table of Contents
terms of the agreements are identical. The agreements provide that, if employment is terminated without cause or for good reason regardless of whether it is in connection with a change in control, the executive will be eligible for 12 months of his or her then current base salary and continued health insurance coverage for this same period. In addition, the agreements provide for the full vesting of all outstanding equity awards in the event the executive’s employment is terminated without cause or for good reason within 18 months following a change in control.
During 2008, we also entered into an Amended and Restated Severance and Change in Control Agreement with James N. Wilson, Chairman of the Board of Directors. The agreement with Mr. Wilson provides that if his employment or service on the Board terminates involuntarily without cause or good reason within eighteen months of a change in control all of his outstanding equity awards shall become fully vested.
We have entered into indemnification agreements with our directors and executive officers. Such agreements require us, among other things, to indemnify its officers and directors, other than for liabilities arising from willful misconduct of a culpable nature, and to advance their expenses incurred as a result of any proceedings against them as to which they could be indemnified.
On May 23, 2008 the Board of Directors approved the engagement of Abbrah Publishing LLC (Abbrah), a firm in which the son of James N. Wilson, the Company’s chairman, is a principal, to assist the Company in the preparation and placement of materials to facilitate the recruitment of patients in its Cushing’s Syndrome trial, based on, among other things, the special qualifications of Abbrah and its willingness to accept performance-based compensation of its services. Compensation to Abbrah is based the number of patients actually enrolled in the study based on their materials. An initial payment is due upon patient enrollment, with an additional amount due if the patient completes the study. During the term of the agreement, the Company recorded expense of $42,000 as compensation to Abbrah in connection with these services assuming patient completion in the study. No additional amounts are expected to be incurred under this agreement.
The Board has determined that the following directors were “independent” during 2009 under current NASDAQ rules:
G. Leonard Baker, Jr.
Joseph C. Cook, Jr.
Patrick G. Enright
James A. Harper
David L. Mahoney
Edward E. Penhoet
See “Director Compensation” for a discussion of our director compensation policy.
| ITEM 14. | PRINCIPAL ACCOUNTING FEES AND SERVICES |
Audit Fees
Fees for audit services totaled approximately $417,000 in 2009 and $447,000 in 2008, including fees for professional services provided in connection with the annual audit of the Company’s financial statements and review of our quarterly financial statement and audit services provided in connection with other statutory or regulatory filings.
Audit- Related Fees, Tax Fees, and All Other Fees
There were no fees paid to our principal accounting firm during 2009 or 2008 for any of these services.
76
Table of Contents
Pre-approval of audit-related and non-audit services
The Audit Committee has delegated to the Chair of the Audit Committee the authority to pre-approve audit-related and non-audit services not prohibited by law to be performed by the Company’s independent registered public accounting firm and associated fees, provided that the Chair shall report any decision to pre-approve such audit-related or non-audit services and fees to the full Audit Committee at its next regular meeting.
| ITEM 15. | EXHIBITS, FINANCIAL STATEMENT SCHEDULES |
The following documents are filed as part of this Form 10-K
| (1) | Financial Statements: |
| Page | ||
| F-2 | ||
| Audited Financial Statements |
||
| F-3 | ||
| F-4 | ||
| Statement of Convertible Preferred Stock and Stockholders’ Equity (Net Capital Deficiency) |
F-5 | |
| F-11 | ||
| F-12 |
(2) Financial Statement Schedules:
All schedules have been omitted because the information required to be set forth therein is not applicable or is shown in the financial statements or notes thereto.
(3) Exhibits:
Item 601 of Regulation S-K requires the exhibits listed below. Each management contract or compensatory plan or arrangement required to be filed as an exhibit to this Form 10-K has been identified.
(A) EXHIBITS
| Exhibit |
Description of Document | |
| 3.1 | Amended and Restated Certificate of Incorporation (incorporated by reference to Exhibit 3.1 to the registrant’s Registration Statement on Form S-1/A (File No. 333-112676) filed on March 19, 2004). | |
| 3.2 | Amended and Restated Bylaws (incorporated by reference to Exhibit 3.1 to the registrant’s Current Report on Form 8-K filed on September 27, 2007). | |
| 4.1 | Specimen Common Stock Certificate (incorporated by reference to Exhibit 4.1 to the registrant’s Registration Statement on Form S-1 (Registration No. 333-112676) filed on February 10, 2004). | |
| 4.2 | Amended and Restated Information and Registration Rights Agreement by and among Corcept Therapeutics Incorporated and certain holders of preferred stock, dated as of May 8, 2001 (incorporated by reference to Exhibit 4.2 to the registrant’s Registration Statement on Form S-1 (Registration No. 333-112676) filed on February 10, 2004). | |
77
Table of Contents
| Exhibit |
Description of Document | |
| 4.3 | Amendment No. 1 to Amended and Restated Information and Registration Rights Agreement by and among Corcept Therapeutics Incorporated and certain holders of preferred stock, dated as of March 16, 2004 (incorporated by reference to Exhibit 4.3 to the registrant’s Registration Statement on Form S-1/A (File No. 333-112676) filed on March 19, 2004). | |
| 4.4 | Registration Rights Agreement by and among Corcept Therapeutics Incorporated and the investors signatory thereto, dated March 14, 2008 (incorporated by reference to Exhibit 10.25 to the registrant’s Annual Report on Form 10-K filed on March 31, 2008). | |
| 4.5 | Registration Rights Agreement by and between Corcept Therapeutics Incorporated and Kingsbridge Capital Limited, dated as of March 25, 2008 (incorporated by reference to Exhibit 10.27 to the registrant’s Annual Report on Form 10-K filed on March 31, 2008). | |
| 4.6 | Registration Rights Agreement by and among Corcept Therapeutics Incorporated and the investors signatory thereto, dated October 12, 2009 (incorporated by reference to Exhibit 4.2 to the registrant’s Quarterly Report on Form 10-Q filed on November 12, 2009). | |
| 10.1* | 2000 Stock Option Plan (incorporated by reference to Exhibit 10.1 to the registrant’s Registration Statement on Form S-1 (Registration No. 333-112676) filed on February 10, 2004). | |
| 10.2* | Promissory Note and Pledge Agreement by and between Corcept Therapeutics Incorporated and Robert L. Roe, M.D., dated as of October 22, 2001 (incorporated by reference to Exhibit 10.4 to the registrant’s Registration Statement on Form S-1 (Registration No. 333-112676) filed on February 10, 2004). | |
| 10.3# | License Agreement by and between The Board of Trustees of the Leland Stanford Junior University and Corcept Therapeutics Incorporated, dated as of July 1, 1999 (incorporated by reference to Exhibit 10.6 to the registrant’s Registration Statement on Form S-1 (Registration No. 333-112676) filed on February 10, 2004). | |
| 10.4 | Master Clinical Development Agreement by and between Corcept Therapeutics Incorporated and Scirex Corporation, dated as of July 12, 2001 (incorporated by reference to Exhibit 10.8 to the registrant’s Registration Statement on Form S-1 (Registration No. 333-112676) filed on February 10, 2004). | |
| 10.5# | Memorandum of Understanding, Supply and Services Agreement, by and between Corcept Therapeutics Incorporated and ScinoPharm Taiwan, dated as of June 12, 2000 (incorporated by reference to Exhibit 10.9 to the registrant’s Registration Statement on Form S-1/A (File No. 333-112676) filed on March 19, 2004). | |
| 10.6 | Master Services Agreement by and between Corcept Therapeutics Incorporated and PPD Development, LP, dated as of January 17, 2003 (incorporated by reference to Exhibit 10.12 to the registrant’s Registration Statement on Form S-1/A (File No. 333-112676) filed on March 19, 2004). | |
| 10.7 | Master Services Agreement by and between Corcept Therapeutics Incorporated and i3 Research, a division of Ingenix Pharmaceuticals Services (UK) Limited, dated as of October 28, 2004 (incorporated by reference to Exhibit 10.13 to the registrant’s Annual Report on Form 10-K filed on March 29, 2005). | |
| 10.8## | Manufacturing Agreement with Produits Chimgues Auxiliaries et de Synthese SA, dated November 8, 2006 (incorporated by reference to Exhibit 10.15 to the registrant’s Annual Report on Form 10-K filed on April 2, 2007). | |
| 10.9 | Common Stock Purchase Agreement by and among Corcept Therapeutics Incorporated and each of the Purchasers listed on Exhibit A thereto, dated November 14, 2006 (incorporated by reference to Exhibit 10.1 to the registrant’s Current Report on Form 8-K filed on November 16, 2006). | |
78
Table of Contents
| Exhibit |
Description of Document | |
| 10.10 | Common Stock Purchase Agreement by and among Corcept Therapeutics Incorporated and each of those persons and entities listed on the Schedule of Purchasers thereto, dated as of March 30, 2007 (incorporated by reference to Exhibit 10.1 to the registrant’s Current Report on Form 8-K filed on April 3, 2007). | |
| 10.11 | Common Stock Purchase Agreement by and among Corcept Therapeutics Incorporated and each of those persons and entities listed on the Schedule of Purchasers thereto, dated as of August 16, 2007 (incorporated by reference to Exhibit 10.1 to the registrants Current Report on Form 8-K filed on August 21, 2007). | |
| 10.12* |
Form of Indemnification Agreement for directors and officers approved by the Board of Directors on September 24, 2007 (incorporated by reference to Exhibit 10.7 to the registrant’s Quarterly Report on Form 10-Q filed on November 14, 2007). | |
| 10.13 | Securities Purchase Agreement by and among Corcept Therapeutics Incorporated and the purchasers named therein, dated March 14, 2008 (incorporated by reference to Exhibit 10.24 to the registrant’s Annual Report on Form 10-K filed on March 31, 2008). | |
| 10.14 | Form of Warrant issued in connection with the Securities Purchase Agreement by and among Corcept Therapeutics Incorporated and the purchasers named therein, dated March 14, 2008 (incorporated by reference to Exhibit 4.4 to the registrant’s Annual Report on Form 10-K filed on March 31, 2008). | |
| 10.15 | Common Stock Purchase Agreement by and between Kingsbridge Capital Limited and Corcept Therapeutics Incorporated dated as of March 25, 2008 (incorporated by reference to Exhibit 10.26 to the registrant’s Annual Report on Form 10-K filed on March 31, 2008). | |
| 10.16 | Warrant, dated March 25, 2008 issued to Kingsbridge Capital Limited (incorporated by reference to Exhibit 4.5 to the registrant’s Annual Report on Form 10-K filed on March 31, 2008). | |
| 10.17# | Master Service Agreement by and among Corcept Therapeutics Incorporated and ICON Clinical Research, L.P., signed on June 4, 2008 (incorporated by reference to Exhibit 10.5 to the registrant’s Quarterly Report on Form 10-Q filed on August 14, 2008). | |
| 10.18* | Amended and Restated Severance and Change in Control Agreement by and between Corcept Therapeutics Incorporated and Joseph K. Belanoff, M. D., dated September 19, 2008 (incorporated by reference to Exhibit 10.25 to the registrant’s Annual Report on Form 10-K filed on March 31, 2009). | |
| 10.19* | Amended and Restated Severance and Change in Control Agreement by and between Corcept Therapeutics Incorporated and Robert L. Roe, M. D., dated September 19, 2008 (incorporated by reference to Exhibit 10.26 to the registrant’s Annual Report on Form 10-K filed on March 31, 2009). | |
| 10.20* | Amended and Restated Severance and Change in Control Agreement by and between Corcept Therapeutics Incorporated and Anne M. LeDoux, dated September 19, 2008 (incorporated by reference to Exhibit 10.27 to the registrant’s Annual Report on Form 10-K filed on March 31, 2009). | |
| 10.21* | Amended and Restated Severance and Change in Control Agreement by and between Corcept Therapeutics Incorporated and James N. Wilson, dated September 19, 2008 (incorporated by reference to Exhibit 10.28 to the registrant’s Annual Report on Form 10-K filed on March 31, 2009). | |
| 10.22* | Employment offer letter to Caroline M. Loewy, dated October 21, 2008 (incorporated by reference to Exhibit 10.29 to the registrant’s Annual Report on Form 10-K filed on March 31, 2009). | |
79
Table of Contents
| Exhibit |
Description of Document | |
| 10.23 | Amendment to Registration Rights Agreement by and among Corcept Therapeutics Incorporated and the investors signatory thereto, dated November 11, 2008 (incorporated by reference to Exhibit 10.30 to the registrant’s Annual Report on Form 10-K filed on March 31, 2009). | |
| 10.24* | Severance and Change in Control Agreement by and between Corcept Therapeutics Incorporated and Caroline M. Loewy, dated November 28, 2008 (incorporated by reference to Exhibit 10.31 to the registrant’s Annual Report on Form 10-K filed on March 31, 2009). | |
| 10.25 | Form of Warrant issued in connection with the Securities Purchase Agreement by and among Corcept Therapeutics Incorporated and the purchasers named therein, dated October 12, 2009 (incorporated by reference to Exhibit 4.1 to the registrant’s Quarterly Report on Form 10-Q filed on September 30, 2009). | |
| 10.26 | Securities Purchase Agreement by and among Corcept Therapeutics Incorporated and the purchasers named therein, dated October 12, 2009 (incorporated by reference to Exhibit 10.1 to the registrant’s Quarterly Report on Form 10-Q filed on September 30, 2009). | |
| 10.27* | Amended and Restated 2004 Equity Incentive Plan (incorporated by reference to Exhibit 10.1 to the registrant’s Quarterly Report on Form 10-Q filed on June 30, 2009). | |
| 14.1 | Code of Ethics (incorporated by reference to Exhibit 99.1 to the registrant’s Registration Statement on Form S-1/A (File No. 333-112676) filed on March 19, 2004). | |
| 23.1 | Consent of Independent Registered Public Accounting Firm | |
| 24.1 | Power of Attorney (See signature page) | |
| 31.1 | Certification pursuant to Rule 13a-14(a) under the Securities Exchange Act of 1934 of Joseph K. Belanoff, M.D. | |
| 31.2 | Certification pursuant to Rule 13a-14(a) under the Securities Exchange Act of 1934 of Caroline M. Loewy | |
| 32.1 | Certification pursuant to 18 U.S.C. Section 1350 of Joseph K. Belanoff, M.D. | |
| 32.2 | Certification pursuant to 18 U.S.C. Section 1350 of Caroline M. Loewy | |
| # | Confidential treatment granted |
| ## | Confidential treatment requested |
| * | Management contract or compensatory plan or arrangement |
80
Table of Contents
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized.
| CORCEPT THERAPEUTICS INCORPORATED | ||
| By: | /S/ JOSEPH K. BELANOFF | |
| Joseph K. Belanoff, M.D., Chief Executive Officer | ||
| Date: | March 26, 2010 | |
POWER OF ATTORNEY
KNOW ALL PERSONS BY THESE PRESENTS, that each person whose signature appears below hereby constitutes and appoints Joseph K. Belanoff and Caroline M. Loewy, and each of them acting individually, as his true and lawful attorneys-in-fact and agents, each with full power of substitution, for him in any and all capacities, to sign any and all amendments to this report on Form 10-K and to file the same, with exhibits thereto and other documents in connection therewith, with the Securities and Exchange Commission, granting unto said attorneys-in-fact and agents, with full power of each to act alone, full power and authority to do and perform each and every act and thing requisite and necessary to be done in connection therewith, as fully for all intents and purposes as he might or could do in person, hereby ratifying and confirming all that said attorneys-in-fact and agents, or his or their substitute or substitutes, may lawfully do or cause to be done by virtue hereof.
Pursuant to the requirements of the Exchange Act, this Annual Report on Form 10-K has been signed by the following persons on behalf of the registrant and in the capacities and on the dates indicated:
| Signature |
Title |
Date | ||
| /S/ JOSEPH K. BELANOFF Joseph K. Belanoff, M.D. |
Chief Executive Officer and Director (Principal Executive Officer) |
March 26, 2010 | ||
| /s/ CAROLINE M. LOEWY Caroline M. Loewy |
Chief Financial Officer (Principal Financial Officer) |
March 26, 2010 | ||
| /s/ ANNE M. LEDOUX Anne M. LeDoux |
Vice President and Controller (Principal Accounting Officer) |
March 26, 2010 | ||
| /s/ JAMES N. WILSON James N. Wilson |
Director and Chairman of the Board of Directors |
March 26, 2010 | ||
| /s/ G. Leonard Baker, Jr. G. Leonard Baker, Jr. |
Director | March 26, 2010 | ||
| /s/ JOSEPH C. COOK, JR. Joseph C. Cook, Jr. |
Director | March 26, 2010 | ||
| /s/ PATRICK G. ENRIGHT Patrick G. Enright |
Director | March 26, 2010 | ||
81
Table of Contents
| Signature |
Title |
Date | ||
| /s/ JAMES A. HARPER James A. Harper |
Director | March 26, 2010 | ||
| /s/ DAVID L. MAHONEY David L. Mahoney |
Director | March 26, 2010 | ||
82
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
INDEX TO FINANCIAL STATEMENTS
| Page | ||
| F-2 | ||
| Audited Financial Statements |
||
| F-3 | ||
| F-4 | ||
| Statement of Convertible Preferred Stock and Stockholders’ Equity (Net Capital Deficiency) |
F-5 | |
| F-11 | ||
| F-12 |
F-1
Table of Contents
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
The Board of Directors and Stockholders of Corcept Therapeutics Incorporated
We have audited the accompanying balance sheets of Corcept Therapeutics Incorporated (a development stage company) as of December 31, 2009 and 2008, and the related statements of operations, convertible preferred stock and stockholders’ equity (net capital deficiency), and cash flows for each of the three years in the period ended December 31, 2009, and for the period from inception (May 13, 1998) to December 31, 2009. These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States.) Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. We were not engaged to perform an audit of the Company’s internal control over financial reporting. Our audit included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion. An audit also includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, and evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the financial statements referred to above present fairly, in all material respects, the financial position of Corcept Therapeutics Incorporated (a development stage company) at December 31, 2009 and 2008, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2009 and for the period from inception (May 13, 1998) to December 31, 2009, in conformity with U.S. generally accepted accounting principles.
/s/ Ernst & Young LLP
Palo Alto, California
March 26, 2010
F-2
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
BALANCE SHEETS
(in thousands, except per share amounts)
| December 31, | ||||||||
| 2009 | 2008 | |||||||
| Assets |
||||||||
| Current assets: |
||||||||
| Cash and cash equivalents |
$ | 23,867 | $ | 14,716 | ||||
| Short-term investments |
— | 3,593 | ||||||
| Prepaid expenses and other current assets |
553 | 1,270 | ||||||
| Total current assets |
24,420 | 19,579 | ||||||
| Property and equipment, net of accumulated depreciation |
10 | 20 | ||||||
| Other assets |
81 | 176 | ||||||
| Total assets |
$ | 24,511 | $ | 19,775 | ||||
| Liabilities and Stockholders’ Equity |
||||||||
| Current liabilities: |
||||||||
| Accounts payable |
$ | 1,270 | $ | 1,304 | ||||
| Accrued clinical expenses |
709 | 989 | ||||||
| Accrued compensation |
210 | 243 | ||||||
| Obligations under capital lease, short-term |
6 | 10 | ||||||
| Other liabilities |
224 | 316 | ||||||
| Total current liabilities |
2,419 | 2,862 | ||||||
| Obligations under capital lease, long-term |
— | 6 | ||||||
| Total liabilities |
2,419 | 2,868 | ||||||
| Commitments |
||||||||
| Stockholders’ equity: |
||||||||
| Preferred stock, $0.001 par value, 10,000 shares authorized and no shares outstanding at December 31, 2009 or 2008 |
— | — | ||||||
| Common stock, $0.001 par value; 140,000 shares authorized and 62,475 and 49,763 shares issued and outstanding at December 31, 2009 and 2008, respectively |
62 | 50 | ||||||
| Additional paid-in capital |
172,369 | 153,031 | ||||||
| Notes receivable from stockholders |
(101 | ) | (6,101 | ) | ||||
| Deficit accumulated during the development stage |
(150,238 | ) | (130,072 | ) | ||||
| Accumulated other comprehensive loss |
— | (1 | ) | |||||
| Total stockholders’ equity |
22,092 | 16,907 | ||||||
| Total liabilities and stockholders’ equity |
$ | 24,511 | $ | 19,775 | ||||
See accompanying notes.
F-3
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
STATEMENTS OF OPERATIONS
(in thousands, except per share amounts)
| Year ended December 31, | Period from inception (May 13, 1998) to December 31, 2009 |
|||||||||||||||
| 2009 | 2008 | 2007 | ||||||||||||||
| Collaboration revenue |
$ | 29 | $ | 209 | $ | 482 | $ | 1,014 | ||||||||
| Operating expenses: |
||||||||||||||||
| Research and development* |
14,402 | 14,152 | 7,860 | 114,211 | ||||||||||||
| General and administrative* |
5,877 | 5,746 | 4,867 | 40,762 | ||||||||||||
| Total operating expenses |
20,279 | 19,898 | 12,727 | 154,973 | ||||||||||||
| Loss from operations |
(20,250 | ) | (19,689 | ) | (12,245 | ) | (153,959 | ) | ||||||||
| Interest and other income, net |
101 | 944 | 688 | 5,326 | ||||||||||||
| Other expense |
(17 | ) | (1,316 | ) | (16 | ) | (1,605 | ) | ||||||||
| Net loss |
$ | (20,166 | ) | $ | (20,061 | ) | $ | (11,573 | ) | $ | (150,238 | ) | ||||
| Basic and diluted net loss per share |
$ | (0.38 | ) | $ | (0.43 | ) | $ | (0.34 | ) | |||||||
| Shares used in computing basic and diluted net loss per share |
52,443 | 46,721 | 34,251 | |||||||||||||
| * Includes non-cash stock-based compensation of the following: |
|
|||||||||||||||
| Research and development |
$ | 263 | $ | 268 | $ | 213 | $ | 5,276 | ||||||||
| General and administrative |
1,552 | 1,360 | 846 | 9,562 | ||||||||||||
| Total non-cash stock-based compensation |
$ | 1,815 | $ | 1,628 | $ | 1,059 | $ | 14,838 | ||||||||
See accompanying notes.
F-4
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
STATEMENT OF CONVERTIBLE PREFERRED STOCK AND STOCKHOLDERS’ EQUITY (NET CAPITAL DEFICIENCY)
(in thousands, except per share amounts)
| Convertible Preferred Stock |
Common Stock | Additional Paid-in Capital |
Notes Receivable from Stockholders |
Deferred Compensation |
Deficit Accumulated During the Development Stage |
Accumulated Other Comprehensive Income (Loss) |
Total Stockholders’ Equity (Net Capital Deficiency) |
||||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | ||||||||||||||||||||||||||||||||||
| Balance at inception (May 13, 1998) |
— | $ | — | — | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | |||||||||||||||||||
| Issuance of common stock to directors for cash in June and July 1998 |
— | — | 7,500 | 8 | (5 | ) | — | — | — | — | 3 | ||||||||||||||||||||||||||
| Issuance of common stock to a director for cash in May 1999 |
— | — | 1,771 | 2 | 63 | — | — | — | — | 65 | |||||||||||||||||||||||||||
| Issuance of common stock to Stanford and directors in conjunction with a license agreement in October 1999 |
— | — | 30 | — | 1 | — | — | — | — | 1 | |||||||||||||||||||||||||||
| Issuance of Series A convertible preferred stock to institutional and individual investors at $1.08 per share for cash and conversion of notes payable, net of issuance costs of $34 in May 1999 |
608 | 623 | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||
| Common stock issued to attorneys and consultants in exchange for services in May 1999 |
— | — | 49 | — | 2 | — | — | — | — | 2 | |||||||||||||||||||||||||||
| Issuance of common stock upon option exercise |
— | — | 60 | — | — | — | — | — | — | — | |||||||||||||||||||||||||||
| Repurchase of common stock held by director in March 1999 |
— | — | (750 | ) | (1 | ) | — | — | — | — | — | (1 | ) | ||||||||||||||||||||||||
| Deferred compensation related to options granted to non-employees |
— | — | — | — | 65 | — | (65 | ) | — | — | — | ||||||||||||||||||||||||||
| Amortization of deferred compensation |
— | — | — | — | — | — | 7 | — | — | 7 | |||||||||||||||||||||||||||
| Net loss from inception to December 31, 1999 |
— | — | — | — | — | — | — | (321 | ) | — | (321 | ) | |||||||||||||||||||||||||
| Balance at December 31, 1999 |
608 | 623 | 8,660 | 9 | 126 | — | (58 | ) | (321 | ) | — | (244 | ) | ||||||||||||||||||||||||
| Issuance of Series B convertible preferred stock to institutional and individual investors at $3.00 per share for cash, net of issuance costs of $19 in January 2000 |
400 | 1,180 | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||
| Deferred compensation related to options granted to an employee and non-employees |
— | — | — | — | 248 | — | (248 | ) | — | — | — | ||||||||||||||||||||||||||
| Amortization of deferred compensation |
— | — | — | — | — | — | 91 | — | — | 91 | |||||||||||||||||||||||||||
| Net loss |
— | — | — | — | — | — | — | (1,846 | ) | — | (1,846 | ) | |||||||||||||||||||||||||
| Balance at December 31, 2000 |
1,008 | 1,803 | 8,660 | 9 | 374 | — | (215 | ) | (2,167 | ) | — | (1,999 | ) | ||||||||||||||||||||||||
| Issuance of Series B convertible preferred stock to consultants in exchange for services in January and April 2001 |
12 | 205 | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||
| Issuance of Series BB convertible preferred stock to institutional and individual investors at $4.033 per share upon conversion of promissory notes in May 2001 |
268 | 1,081 | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||
| Issuance of Series C convertible preferred stock to institutional and individual investors at $7.066 per share for cash, net of issuance costs of approximately $95 in May and June 2001 |
3,807 | 26,805 | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||
| Issuance of Series C convertible preferred stock to consultants in exchange for services in October 2001 |
1 | 20 | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||
| Issuance of common stock to a consultant for cash below fair value in April 2001 |
— | — | 50 | — | 50 | — | — | — | 50 | ||||||||||||||||||||||||||||
| Issuance of common stock upon option exercises |
— | — | 768 | — | 438 | (438 | ) | — | — | — | — | ||||||||||||||||||||||||||
| Issuance of common stock in conjunction with a license agreement |
— | — | 1 | — | 15 | — | — | — | — | 15 | |||||||||||||||||||||||||||
| Deferred compensation related to options granted to employees and non-employees |
— | — | — | — | 10,226 | — | (10,226 | ) | — | — | — | ||||||||||||||||||||||||||
| Amortization of deferred compensation |
— | — | — | — | — | — | 1,849 | — | — | 1,849 | |||||||||||||||||||||||||||
| Net loss |
— | — | — | — | — | — | — | (7,454 | ) | — | (7,454 | ) | |||||||||||||||||||||||||
| Balance at December 31, 2001 (carried forward) |
5,096 | 29,914 | 9,479 | 9 | 11,103 | (438 | ) | (8,592 | ) | (9,621 | ) | — | (7,539 | ) | |||||||||||||||||||||||
F-5
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
STATEMENT OF CONVERTIBLE PREFERRED STOCK AND STOCKHOLDERS’ EQUITY (NET CAPITAL DEFICIENCY), (Continued)
(in thousands, except per share amounts)
| Convertible Preferred Stock |
Common Stock | Additional Paid-in Capital |
Notes Receivable from Stockholders |
Deferred Compensation |
Deficit Accumulated During the Development Stage |
Accumulated Other Comprehensive Income (Loss) |
Total Stockholders’ Equity (Net Capital Deficiency) |
||||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | ||||||||||||||||||||||||||||||||||
| Balance at December 31, 2001 (brought forward) |
5,096 | $ | 29,914 | 9,479 | $ | 9 | $ | 11,103 | $ | (438 | ) | $ | (8,592 | ) | $ | (9,621 | ) | $ | — | $ | (7,539 | ) | |||||||||||||||
| Issuance of Series C convertible preferred stock to institutional and individual investors at $7.066 per share for cash, net of issuance costs of approximately $19 in December 2002 |
1,673 | 11,802 | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||
| Issuance of common stock upon option exercises |
— | — | 62 | — | — | — | — | — | — | — | |||||||||||||||||||||||||||
| Amortization of deferred compensation |
— | — | — | — | — | — | 4,085 | — | — | 4,085 | |||||||||||||||||||||||||||
| Reduction of deferred compensation related to the unamortized portion of deferred stock compensation related to a terminated employee |
— | — | — | — | (239 | ) | — | 239 | — | — | — | ||||||||||||||||||||||||||
| Reversal of previously expensed deferred compensation related to a terminated employee based on the straight line method |
— | — | — | — | (50 | ) | — | — | — | — | (50 | ) | |||||||||||||||||||||||||
| Stock-based compensation related to lapsing repurchase right of stock held by a non-employee |
68 | 68 | |||||||||||||||||||||||||||||||||||
| Net loss |
— | — | — | — | — | — | — | (18,504 | ) | — | (18,504 | ) | |||||||||||||||||||||||||
| Balance at December 31, 2002 |
6,769 | 41,716 | 9,541 | 9 | 10,882 | (438 | ) | (4,268 | ) | (28,125 | ) | — | (21,940 | ) | |||||||||||||||||||||||
| Deferred compensation related to options granted to employees and non-employees |
— | — | — | — | 1,159 | — | (1,159 | ) | — | — | — | ||||||||||||||||||||||||||
| Amortization of deferred compensation |
— | — | — | — | — | — | 1,559 | — | — | 1,559 | |||||||||||||||||||||||||||
| Reduction of deferred compensation related to the unamortized portion of deferred stock compensation related to terminated employees |
— | — | — | — | (1,588 | ) | — | 1,588 | — | — | — | ||||||||||||||||||||||||||
| Reversal of previously expensed deferred compensation related to terminated employees |
— | — | — | — | (1,384 | ) | — | — | — | — | (1,384 | ) | |||||||||||||||||||||||||
| Repurchase of common stock and reduction of note payable upon termination of employees |
— | — | (206 | ) | — | (155 | ) | 155 | — | — | — | — | |||||||||||||||||||||||||
| Repayment of note receivable from stockholder |
— | — | — | — | — | 37 | — | — | — | 37 | |||||||||||||||||||||||||||
| Stock-based compensation related to lapsing repurchase right of stock held by a non-employee |
68 | 68 | |||||||||||||||||||||||||||||||||||
| Net loss |
— | — | — | — | — | — | — | (9,812 | ) | — | (9,812 | ) | |||||||||||||||||||||||||
| Unrealized loss on short-term investments |
— | — | — | — | — | — | — | — | (1 | ) | (1 | ) | |||||||||||||||||||||||||
| Total comprehensive loss |
(9,813 | ) | |||||||||||||||||||||||||||||||||||
| Balance at December 31, 2003 (carried forward) |
6,769 | 41,716 | 9,335 | 9 | 8,982 | (246 | ) | (2,280 | ) | (37,937 | ) | (1 | ) | (31,473 | ) | ||||||||||||||||||||||
F-6
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
STATEMENT OF CONVERTIBLE PREFERRED STOCK AND STOCKHOLDERS’ EQUITY (NET CAPITAL DEFICIENCY), (Continued)
(in thousands, except per share amounts)
| Convertible Preferred Stock |
Common Stock | Additional Paid-in Capital |
Notes Receivable from Stockholders |
Deferred Compensation |
Deficit Accumulated During the Development Stage |
Accumulated Other Comprehensive Income (Loss) |
Total Stockholders’ Equity (Net Capital Deficiency) |
|||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | |||||||||||||||||||||||||||||||||
| Balance at December 31, 2003 (brought forward) |
6,769 | $ | 41,716 | 9,335 | $ | 9 | $ | 8,982 | $ | (246 | ) | $ | (2,280 | ) | $ | (37,937 | ) | $ | (1 | ) | $ | (31,473 | ) | |||||||||||||
| Sale of Shares in IPO at $12.00 per share for cash, net of issuance costs of approximately $4,974 |
— | — | 4,500 | 5 | 49,020 | — | — | — | — | 49,025 | ||||||||||||||||||||||||||
| Conversion of preferred shares in IPO |
(6,769 | ) | (41,716 | ) | 8,807 | 9 | 41,707 | — | — | — | — | 41,716 | ||||||||||||||||||||||||
| Conversion of note payable |
— | — | 45 | — | 534 | — | — | — | — | 534 | ||||||||||||||||||||||||||
| Issuance of common stock upon option exercises |
— | — | 7 | — | 1 | — | — | — | — | 1 | ||||||||||||||||||||||||||
| Deferred compensation related to options granted to employees and non-employees |
— | — | — | — | 1,447 | — | (1,447 | ) | — | — | — | |||||||||||||||||||||||||
| Amortization of deferred compensation |
— | — | — | — | — | — | 1,854 | — | — | 1,854 | ||||||||||||||||||||||||||
| Reduction of deferred compensation related to the unamortized portion of deferred stock compensation related to terminated employees and consultants |
— | — | — | — | (155 | ) | — | 155 | — | — | — | |||||||||||||||||||||||||
| Reversal of previously expensed deferred compensation related to employees terminated or converted to consultant |
— | — | — | — | (243 | ) | — | — | — | — | (243 | ) | ||||||||||||||||||||||||
| Repayment of note receivable from stockholder |
— | — | — | — | — | 62 | — | — | — | 62 | ||||||||||||||||||||||||||
| Stock-based compensation related to lapsing repurchase right of stock held by a non-employee |
68 | 68 | ||||||||||||||||||||||||||||||||||
| Net loss |
— | — | — | — | — | — | — | (15,535 | ) | — | (15,535 | ) | ||||||||||||||||||||||||
| Change in unrealized loss on investments |
— | — | — | — | — | — | — | — | (61 | ) | (61 | ) | ||||||||||||||||||||||||
| Total comprehensive loss |
(15,596 | ) | ||||||||||||||||||||||||||||||||||
| Balance at December 31, 2004 |
— | — | 22,694 | 23 | 101,361 | (184 | ) | (1,718 | ) | (53,472 | ) | (62 | ) | 45,948 | ||||||||||||||||||||||
| Issuance of common stock upon option exercise for cash in June 2005 at a price of $0.10 per share |
— | — | 9 | — | 1 | — | — | — | — | 1 | ||||||||||||||||||||||||||
| Deferred compensation related to options granted to employees and non-employees |
— | — | — | — | (94 | ) | — | 94 | — | — | — | |||||||||||||||||||||||||
| Amortization of deferred compensation |
— | — | — | — | 35 | — | 912 | — | — | 947 | ||||||||||||||||||||||||||
| Reduction of deferred compensation related to the unamortized portion of deferred stock compensation related to unvested shares at termination of employees |
— | — | — | — | (109 | ) | — | 109 | — | — | — | |||||||||||||||||||||||||
| Reversal of previously expensed deferred compensation related to employees terminated or converted to consultant |
— | — | — | — | (250 | ) | — | — | — | — | (250 | ) | ||||||||||||||||||||||||
| Repayment of note receivable from stockholder |
— | — | — | — | — | 16 | — | — | — | 16 | ||||||||||||||||||||||||||
| Stock-based compensation related to lapsing repurchase right of stock held by a non-employee |
— | — | — | — | 68 | — | — | — | — | 68 | ||||||||||||||||||||||||||
| Issuance of common stock for services |
— | — | 1 | — | 2 | — | — | — | — | 2 | ||||||||||||||||||||||||||
| Net loss |
— | — | — | — | — | — | — | (20,093 | ) | — | (20,093 | ) | ||||||||||||||||||||||||
| Change in unrealized loss on investments |
— | — | — | — | — | — | — | — | (46 | ) | (46 | ) | ||||||||||||||||||||||||
| Total comprehensive loss |
(20,139 | ) | ||||||||||||||||||||||||||||||||||
| Balance at December 31, 2005 (carried forward) |
— | — | 22,704 | 23 | 101,014 | (168 | ) | (603 | ) | (73,565 | ) | (108 | ) | 26,593 | ||||||||||||||||||||||
F-7
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
STATEMENT OF CONVERTIBLE PREFERRED STOCK AND STOCKHOLDERS’ EQUITY (NET CAPITAL DEFICIENCY), (Continued)
(in thousands, except per share amounts)
| Convertible Preferred Stock |
Common Stock | Additional Paid-in Capital |
Notes Receivable from Stockholders |
Deferred Compensation |
Deficit Accumulated During the Development Stage |
Accumulated Other Comprehensive Income (Loss) |
Total Stockholders’ Equity (Net Capital Deficiency) |
|||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | |||||||||||||||||||||||||||||||||
| Balance at December 31, 2005 (brought forward) |
— | $ | — | 22,704 | $ | 23 | $ | 101,014 | $ | (168 | ) | $ | (603 | ) | $ | (73,565 | ) | $ | (108 | ) | $ | 26,593 | ||||||||||||||
| Sale of common stock in December 2006 at $1.00 per share for cash, net of issuance costs of approximately $83 |
— | — | 3,000 | 3 | 2,914 | — | — | — | — | 2,917 | ||||||||||||||||||||||||||
| Issuance of common stock upon option exercises at various times for cash at weighted-average exercise price of $0.73 per share |
— | — | 26 | — | 19 | — | — | — | — | 19 | ||||||||||||||||||||||||||
| Issuance of common stock at various times for services in lieu of cash compensation at an average value of $4.93 per share |
— | — | 2 | — | 12 | — | — | — | — | 12 | ||||||||||||||||||||||||||
| Amortization of deferred compensation related to options granted to employees prior to the IPO |
— | — | — | — | — | — | 375 | — | — | 375 | ||||||||||||||||||||||||||
| Stock-based compensation related to employee options granted after the IPO |
— | — | — | — | 1,118 | — | — | — | — | 1,118 | ||||||||||||||||||||||||||
| Stock-based compensation related to options to consultants at various times at prices ranging from $0.10 to $10.06 |
— | — | — | — | 75 | — | — | — | — | 75 | ||||||||||||||||||||||||||
| Reversal of previously expensed compensation related to employees terminated or converted to consultant |
— | — | — | — | (50 | ) | — | — | — | — | (50 | ) | ||||||||||||||||||||||||
| Repayments of notes receivable from stockholders in October and December of 2006 |
— | — | — | — | — | 43 | — | — | — | 43 | ||||||||||||||||||||||||||
| Stock-based compensation related to lapsing repurchase right of stock held by a non-employee |
23 | 23 | ||||||||||||||||||||||||||||||||||
| Net loss |
— | — | — | — | — | — | — | (24,873 | ) | — | (24,873 | ) | ||||||||||||||||||||||||
| Change in unrealized loss on investments |
— | — | — | — | — | — | — | — | 108 | 108 | ||||||||||||||||||||||||||
| Total comprehensive loss |
(24,765 | ) | ||||||||||||||||||||||||||||||||||
| Balance at December 31, 2006 (carried forward) |
— | — | 25,732 | 26 | 105,125 | (125 | ) | (228 | ) | (98,438 | ) | — | 6,360 | |||||||||||||||||||||||
F-8
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
STATEMENT OF CONVERTIBLE PREFERRED STOCK AND STOCKHOLDERS’ EQUITY (NET CAPITAL DEFICIENCY), (Continued)
(in thousands, except per share amounts)
| Convertible Preferred Stock |
Common Stock | Additional Paid-in Capital |
Notes Receivable from Stockholders |
Deferred Compensation |
Deficit Accumulated During the Development Stage |
Accumulated Other Comprehensive Income (Loss) |
Total Stockholders’ Equity (Net Capital Deficiency) |
|||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | |||||||||||||||||||||||||||||||||
| Balance at December 31, 2006 (brought forward) |
— | $ | — | $ | 25,732 | $ | 26 | $ | 105,125 | $ | (125 | ) | $ | (228 | ) | $ | (98,438 | ) | $ | — | $ | 6,360 | ||||||||||||||
| Sale of common stock in March 2007 at $1.00 per share for cash, net of issuance costs of approximately $151 |
— | — | 9,000 | 9 | 8,840 | — | — | — | — | 8,849 | ||||||||||||||||||||||||||
| Sale of common stock in August & September 2007 at $2.10 per share for cash, net of issuance costs of approximately $64 |
— | — | 4,790 | 5 | 9,991 | — | — | — | — | 9,996 | ||||||||||||||||||||||||||
| Issuance of common stock upon option exercises at various times for cash at weighted-average exercise price of $0.79 per share |
— | — | 26 | — | 21 | — | — | — | — | 21 | ||||||||||||||||||||||||||
| Amortization of deferred compensation related to options granted to employees prior to the IPO |
— | — | — | — | — | — | 96 | — | — | 96 | ||||||||||||||||||||||||||
| Stock-based compensation related to employee options granted after the IPO |
— | — | — | — | 1,334 | — | — | — | — | 1,334 | ||||||||||||||||||||||||||
| Stock-based compensation related to options to consultants at various times at prices ranging from $0.10 to $10.06 |
— | — | — | — | 48 | — | — | — | — | 48 | ||||||||||||||||||||||||||
| Reduction of deferred compensation related to the unamortized portion of deferred stock compensation related to unvested shares at termination of employees |
— | — | — | — | (119 | ) | — | 119 | — | — | — | |||||||||||||||||||||||||
| Reversal of previously expensed compensation related to employees terminated |
— | — | — | — | (418 | ) | — | — | — | — | (418 | ) | ||||||||||||||||||||||||
| Repayments of notes receivable from stockholders in March and October 2007 |
— | — | — | — | — | 18 | — | — | — | 18 | ||||||||||||||||||||||||||
| Net loss |
— | — | — | — | — | — | — | (11,573 | ) | — | (11,573 | ) | ||||||||||||||||||||||||
| Change in unrealized gain on investments |
— | — | — | — | — | — | — | — | 3 | 3 | ||||||||||||||||||||||||||
| Net comprehensive loss |
(11,570 | ) | ||||||||||||||||||||||||||||||||||
| Balance at December 31, 2007 |
— | — | 39,548 | 40 | 124,822 | (107 | ) | (13 | ) | (110,011 | ) | 3 | 14,734 | |||||||||||||||||||||||
See accompanying notes.
F-9
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
STATEMENT OF CONVERTIBLE PREFERRED STOCK AND STOCKHOLDERS’ EQUITY (NET CAPITAL DEFICIENCY), (Continued)
(in thousands, except per share amounts)
| Convertible Preferred Stock |
Common Stock | Additional Paid-in Capital |
Notes Receivable from Stockholders |
Deferred Compensation |
Deficit Accumulated During the Development Stage |
Accumulated Other Comprehensive Income (Loss) |
Total Stockholders’ Equity (Net Capital Deficiency) |
||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | ||||||||||||||||||||||||||||||||
| Balance at December 31, 2007 (brought forward) |
— | $ | — | 39,548 | $ | 40 | $ | 124,822 | $ | (107 | ) | $ | (13 | ) | $ | (110,011 | ) | $ | 3 | $ | 14,734 | ||||||||||||||
| Sale of common stock and issuance of warrants in March 2008 at $2.83 per unit for cash and note receivable, net of issuance costs of approximately $382 |
— | — | 8,924 | 9 | 24,783 | (6,000 | ) | — | — | — | 18,792 | ||||||||||||||||||||||||
| Sales of common stock in August and September 2008 under Committed Equity Financing Facility (CEFF), at an average discounted price of $1.85 per share, net of costs associated with the registration of shares under the CEFF of $216 |
— | — | 405 | — | 533 | — | — | — | — | 533 | |||||||||||||||||||||||||
| Issuance of common stock in November 2008 in settlement of liquidated damages, net of issuance costs of $5 |
— | — | 883 | 1 | 1,274 | — | — | — | — | 1,275 | |||||||||||||||||||||||||
| Issuance of common stock upon option exercise in September 2008 for cash at exercise price of $1.50 per share |
— | — | 2 | — | 4 | — | — | — | — | 4 | |||||||||||||||||||||||||
| Issuance of common stock in February for services in lieu of cash compensation at a value of $2.73 per share |
— | — | 1 | — | 4 | — | — | — | — | 4 | |||||||||||||||||||||||||
| Amortization of deferred compensation related to options granted to employees prior to the IPO |
— | — | — | — | — | — | 13 | — | — | 13 | |||||||||||||||||||||||||
| Stock-based compensation related to employee options granted after the IPO |
— | — | — | — | 1,580 | — | — | — | — | 1,580 | |||||||||||||||||||||||||
| Stock-based compensation related to options to consultants at various times at prices ranging from $4.90 to $10.06 |
— | — | — | — | 31 | — | — | — | — | 31 | |||||||||||||||||||||||||
| Repayment of note receivable from shareholder in May 2008 |
— | — | — | — | — | 6 | — | — | — | 6 | |||||||||||||||||||||||||
| Net loss |
— | — | — | — | — | — | — | (20,061 | ) | — | (20,061 | ) | |||||||||||||||||||||||
| Change in unrealized loss on investments |
— | — | — | — | — | — | — | — | (4 | ) | (4 | ) | |||||||||||||||||||||||
| Total comprehensive loss |
(20,065 | ) | |||||||||||||||||||||||||||||||||
| Balance at December 31, 2008 |
— | $ | — | 49,763 | $ | 50 | $ | 153,031 | $ | (6,101 | ) | $ | — | $ | (130,072 | ) | $ | (1 | ) | $ | 16,907 | ||||||||||||||
| Sale of common stock and issuance of warrants in October 2009 at $1.43 per unit for cash, net of issuance costs of approximately $720 |
— | — | 12,597 | 12 | 17,280 | — | — | — | — | 17,292 | |||||||||||||||||||||||||
| Sales of common stock in October 2009 under CEFF, at an average discounted price of $2.45 per share, net of issuance costs of approximately $7 |
— | — | 102 | — | 243 | — | — | — | — | 243 | |||||||||||||||||||||||||
| Issuance of common stock in August – October 2009 for services in lieu of cash compensation at an average value of $1.22 per share |
— | — | 13 | — | 16 | — | — | — | — | 16 | |||||||||||||||||||||||||
| Stock-based compensation related to employee options |
— | — | — | — | 1,789 | — | — | — | — | 1,789 | |||||||||||||||||||||||||
| Stock-based compensation related to options to consultants at various times at prices ranging from $4.90 to $10.06 |
— | — | — | — | 10 | — | — | — | — | 10 | |||||||||||||||||||||||||
| Repayment of note receivable from shareholder in February 2009 |
— | — | — | — | — | 6,000 | — | — | — | 6,000 | |||||||||||||||||||||||||
| Net loss |
— | — | — | — | — | — | — | (20,166 | ) | — | (20,166 | ) | |||||||||||||||||||||||
| Change in unrealized loss on investments |
— | — | — | — | — | — | — | — | 1 | 1 | |||||||||||||||||||||||||
| Total comprehensive loss |
(20,165 | ) | |||||||||||||||||||||||||||||||||
| Balance at December 31, 2009 |
— | $ | — | 62,475 | $ | 62 | $ | 172,369 | $ | (101 | ) | $ | — | $ | (150,238 | ) | $ | — | $ | 22,092 | |||||||||||||||
F-10
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
STATEMENTS OF CASH FLOWS
(in thousands)
| Year ended December 31, | Period from inception (May 13, 1998) to December 31, 2009 |
|||||||||||||||
| 2009 | 2008 | 2007 | ||||||||||||||
| Operating activities |
||||||||||||||||
| Net loss |
$ | (20,166 | ) | $ | (20,061 | ) | $ | (11,573 | ) | $ | (150,238 | ) | ||||
| Adjustments to reconcile net loss to net cash used in operations: |
||||||||||||||||
| Depreciation and amortization of property and equipment |
10 | 12 | 13 | 110 | ||||||||||||
| Stock-based compensation, net of recoveries |
1,799 | 1,624 | 1,060 | 14,463 | ||||||||||||
| Expense related to stock issued for services |
16 | 4 | — | 64 | ||||||||||||
| Settlement of liquidated damages in stock |
— | 1,281 | — | 1,281 | ||||||||||||
| Expense related to stock issued in conjunction with license agreement |
— | — | — | 31 | ||||||||||||
| Expense related to stock issued below fair value |
— | — | — | 522 | ||||||||||||
| Interest accrued on convertible promissory notes |
— | — | — | 104 | ||||||||||||
| Changes in operating assets and liabilities: |
||||||||||||||||
| Prepaid expenses and other current assets |
717 | (980 | ) | 53 | (553 | ) | ||||||||||
| Other assets |
95 | (113 | ) | 2 | (81 | ) | ||||||||||
| Accounts payable |
(34 | ) | 189 | 199 | 1,270 | |||||||||||
| Accrued clinical |
(280 | ) | 110 | (1,345 | ) | 709 | ||||||||||
| Other liabilities |
(125 | ) | (428 | ) | 627 | 434 | ||||||||||
| Net cash used in operating activities |
(17,968 | ) | (18,362 | ) | (10,964 | ) | (131,884 | ) | ||||||||
| Investing activities |
||||||||||||||||
| Purchases of property and equipment |
— | (7 | ) | — | (61 | ) | ||||||||||
| Purchases of short-term and long-term investments |
— | (3,594 | ) | (6,380 | ) | (118,320 | ) | |||||||||
| Maturities of short-term investments |
3,594 | 5,930 | 1,000 | 118,320 | ||||||||||||
| Net cash provided by (used in) investing activities |
3,594 | 2,329 | (5,380 | ) | (61 | ) | ||||||||||
| Financing activities |
||||||||||||||||
| Proceeds from issuance of common stock and warrants, including collection of note receivable, net of cash paid for issuance costs |
23,535 | 19,329 | 18,884 | 113,944 | ||||||||||||
| Proceeds from issuance of convertible notes |
— | — | — | 1,543 | ||||||||||||
| Principal payments of obligations under capital leases |
(10 | ) | (13 | ) | (13 | ) | (53 | ) | ||||||||
| Proceeds from issuance of convertible preferred stock, net of cash paid for issuance costs |
— | — | — | 40,378 | ||||||||||||
| Net cash provided by financing activities |
23,525 | 19,316 | 18,871 | 155,812 | ||||||||||||
| Net increase in cash and cash equivalents |
9,151 | 3,283 | 2,527 | 23,867 | ||||||||||||
| Cash and cash equivalents at beginning of period |
14,716 | 11,433 | 8,906 | — | ||||||||||||
| Cash and cash equivalents at end of period |
$ | 23,867 | $ | 14,716 | $ | 11,433 | $ | 23,867 | ||||||||
| Supplemental disclosure of cash flow information |
||||||||||||||||
| Interest paid |
$ | 1 | $ | 5 | $ | 3 | $ | 16 | ||||||||
| Supplemental disclosure of non-cash financing activities |
||||||||||||||||
| Conversion of convertible promissory notes and accrued interest |
||||||||||||||||
| —to convertible preferred stock |
$ | — | $ | — | $ | — | $ | 1,111 | ||||||||
| —to common stock |
$ | — | $ | — | $ | — | $ | 534 | ||||||||
| Issuance of warrant in connection with financing agreement |
$ | — | $ | 653 | $ | — | $ | 653 | ||||||||
| Issuance of common stock in settlement of liquidated damages |
$ | — | $ | 1,281 | $ | — | $ | 1,281 | ||||||||
| Purchase of equipment under capital leases |
$ | — | $ | — | $ | — | $ | 59 | ||||||||
See accompanying notes.
F-11
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
NOTES TO FINANCIAL STATEMENTS
1. Basis of Presentation and Summary of Significant Accounting Policies
Description of Business
Corcept Therapeutics Incorporated (the “Company” or “Corcept”) was incorporated in the state of Delaware on May 13, 1998, and its facilities are located in Menlo Park, California. Corcept is a pharmaceutical company engaged in the development of drugs for the treatment of severe metabolic and psychiatric diseases.
The Company’s primary activities since incorporation have been establishing its offices, recruiting personnel, conducting research and development, performing business and financial planning, raising capital, and overseeing clinical trials. Accordingly, the Company is considered to be in the development stage.
Management Plans
In the course of its development activities, the Company has sustained operating losses and expects such losses to continue for at least the next few years. The Company plans to continue to finance its operations through the sale of its equity and/or debt securities or by engaging in strategic relationships with potential partners. The accompanying financial statements have been prepared on a going concern basis, which contemplates the realization of assets and the settlement of liabilities and commitments in the normal course of business. The Company’s ability to continue its operations through the complete development and commercialization of its products is dependent upon the successful execution of its financing and/or any partnership strategies. The Company’s most advanced programs are the two Phase 3 trials of CORLUX in Cushing’s Syndrome and in psychotic depression.
As reflected in the accompanying financial statements as of December 31, 2009, the Company had cash, cash equivalents and investments balances of $23.9 million, working capital of $22.0 million and an accumulated deficit of $150.2 million. In January 2010, the Company sold additional shares of its common stock for proceeds of $625,000. (See Note 14 – Subsequent Events.) The Company has sufficient funds to maintain its operations into early 2011, including the planned completion of enrollment of its Phase 3 Cushing’s Syndrome trial, the submission of a New Drug Application (NDA) for CORLUX, the Company’s lead product, to the United States Food and Drug Administration (FDA) for this indication, the continuation of enrollment in its Phase 3 psychotic depression trial, and the conduct of a Phase 1 trial for CORT 108297, one of its proprietary, selective GR-II antagonists.
The Company will need to raise additional funds in order to sustain its operations at anticipated levels beyond early 2011. Although the Company’s management recognizes the need to raise funds in the future, there can be no assurance that the Company will be successful in consummating any such transaction, or, if the Company does consummate such a transaction, that the terms and conditions of such financing or any partnership will not be unfavorable to it. Any failure by the Company to obtain additional funding will have a material effect upon it and will likely result in the Company’s inability to continue its operations as currently planned beyond early 2011.
Use of Estimates
The preparation of financial statements in conformity with U.S. generally accepted accounting principles requires management to make estimates and assumptions that affect the amounts reported in the financial statements and accompanying notes. Actual results could differ materially from those estimates. Any changes in estimates are recorded in the period of the change.
F-12
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
NOTES TO FINANCIAL STATEMENTS, Continued
Cost accruals for clinical trials are based upon estimates of work completed under service agreements, milestones achieved, patient enrollment and past experience with similar contracts. The Company’s estimates of work completed and associated cost accruals include its assessments of information received from third-party contract research organizations and the overall status of clinical trial activities. The estimates are updated on a recurring basis as new information becomes available.
Subsequent Events
The Company has reviewed and evaluated material subsequent events from the balance sheet date of December 31, 2009 through the time of issuance of these financial statements by the filing of this Form 10-K with the Securities and Exchange Commission (SEC). See Note 14 – Subsequent Events for subsequent events that have been identified for disclosure.
Cash, Cash Equivalents and Short-term Investments
The Company invests its excess cash in bank deposits, money market accounts, corporate debt securities, and obligations of the U.S. government and U.S. government sponsored entities. The Company considers all highly liquid investments purchased with maturities of three months or less from the date of purchase to be cash equivalents. Cash equivalents are carried at fair value, which approximates cost, and primarily consist of money market funds maintained at major U.S. financial institutions and commercial paper issued by major corporations with maturities of less than 90 days from date of purchase.
Short-term investments at December 31, 2008, which primarily represented readily marketable debt securities, were classified as “available-for-sale.” Short-term investments included debt securities with maturities of one year or less from the balance sheet date. Debt securities with maturities of greater than 12 months from the balance sheet dates would be classified as long-term investments. Purchased premiums or discounts on debt securities were amortized to interest income through the stated maturities of the debt securities. The differences between amortized cost and fair values of the debt securities were recorded as a component of accumulated other comprehensive loss. Management determines the appropriate classification of its investments in debt securities at the time of purchase and evaluates such designation as of each balance sheet date. Unrealized gains and losses are included in accumulated other comprehensive loss and reported as a separate component of stockholders’ equity. Realized gains and losses and declines in value judged to be other-than-temporary, if any, on available-for-sale securities would be included in other expenses. The cost of securities sold is based on the specific identification method. Interest earned on short-term and long-term investments is included in interest income.
Credit Risks and Concentrations
The Company’s concentration of credit risk consists of cash, cash equivalents, and short-term investments. The Company is exposed to credit risk in the event of default by the financial institutions holding the cash, cash equivalents, and short-term investments to the extent of the amount recorded on the balance sheet. This risk is mitigated by investing in securities with high credit ratings from the major rating services and by limiting the amount of investment in any one issuer. As of December 31, 2009 and 2008, the Company had no investments in mortgage-backed securities or auction rate securities.
While the Company monitors the cash balance in its checking account and transfers the funds in only as needed, these cash balances could be impacted if the underlying financial institution were to fail or could be
F-13
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
NOTES TO FINANCIAL STATEMENTS, Continued
subject to other adverse conditions in the financial markets. On October 23, 2008, the Federal Deposit Insurance Corporation (FDIC) implemented its Temporary Liquidity Guarantee Program. Under this program, non-interest bearing commercial accounts are insured to an unlimited amount through June 30, 2010, thus mitigating our exposure to any possible bank failure. To date, the Company has experienced no loss or lack of access to cash in its operating accounts.
The Company also has a concentration of risk in regard to the manufacture of its product. As of December 31, 2009, the Company has a single source supplier for its tablet manufacture. If this supplier is unable to prepare the CORLUX tablets in the quantities and time frame required, the Company may not be able to manufacture its product in a timely manner.
Fair Value Measurements
Financial instruments are categorized in a fair value hierarchy that prioritizes the information used to develop assumptions for measuring fair value and expands disclosures about fair value measurements. The fair value hierarchy gives the highest priority to quoted prices in active markets for identical assets or liabilities (Level 1 input), then to quoted prices (in non-active markets or in active markets for similar assets or liabilities), inputs other than quoted prices that are observable for the asset or liability, and inputs that are not directly observable, but that are corroborated by observable market data for the asset or liability (Level 2 input), then the lowest priority to unobservable inputs, for example, the Company’s own data about the assumptions that market participants would use in pricing an asset or liability (Level 3 input). Fair value is a market-based measurement, not an entity-specific measurement, and a fair value measurement should therefore be based on the assumptions that market participants would use in pricing the asset or liability.
No assets or liabilities in the Company’s financial statements are required to be measured at fair value other than the Company’s investment portfolio.
Revenue Recognition
Collaboration revenue relates to services rendered in connection with agreements signed with Eli Lilly and Company (Eli Lilly), in which Eli Lilly agreed to support certain of the Company’s pre-clinical and clinical proof-of-concept studies evaluating the ability of the Company’s product candidates to mitigate or prevent weight gain associated with the use of Zyprexa (olanzapine), an atypical antipsychotic medication. Under the agreements, Eli Lilly agreed to supply the Zyprexa and olanzapine and pay for the studies. The Company was required to perform development activities as specified in these agreements and was reimbursed based on the costs associated with the conduct of the trial and the preparation and packaging of clinical trial materials. Revenue was recognized as services were rendered in accordance with the agreement.
Research and Development
Research and development expenses consist of costs incurred for Company-sponsored research and development activities. These costs include direct expenses (including nonrefundable payments to third parties) and research-related overhead expenses, as well as the cost of funding clinical trials, pre-clinical studies, manufacturing development and the contract development of second-generation compounds, and are expensed as incurred. Costs to acquire technologies and materials that are utilized in research and development and that have no alternative future use are expensed when incurred (see Note 2).
F-14
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
NOTES TO FINANCIAL STATEMENTS, Continued
Segment Reporting
Operating segments are determined based on the way management organizes its business for making operating decisions and assessing performance. The Company has only one operating segment, which is involved in the development of pharmaceutical products.
Property and Equipment
Property and equipment are stated at cost less accumulated depreciation. Property and equipment are depreciated using the straight-line method over the estimated useful lives of the assets, ranging from three to five years.
Assets acquired under capital leases are amortized over the term of their useful lives or the lease period, whichever is shorter.
Stock-Based Compensation
Stock-based compensation for employee and director options
Since January 1, 2006, the Company has accounted for stock-based compensation related to option grants to employees and directors under the fair value method, based on the fair value of the award at the grant date. For service awards, expense is recognized over the requisite service period, for all options granted after the Company’s initial public offering (IPO) in 2004. For option awards granted prior to the IPO, the Company expensed the stock-based compensation based on the intrinsic value method.
For service awards, expense is recognized over the requisite service period. For performance-based awards, expense will be recognized at such time as there is a high degree of probability (i.e., greater than 70%) of achieving the required objective.
Stock-based compensation expense related to non-employees
Expense is recognized for options granted to non-employees based on the fair-value of the option grants at the time of vesting.
See Note 9 for a detailed discussion of stock-compensation expense and related accounting.
Income Taxes
Deferred tax assets and liabilities are determined based on the differences between the financial reporting and tax bases of assets and liabilities and are measured using the enacted tax rates that will be in effect when the differences are expected to reverse. A valuation allowance is recorded when it is more likely than not that the deferred tax asset will not be recovered.
No amounts have been recognized as interest or penalties on income tax related matters. The determination of an accounting policy as to the classification of such costs has been deferred until such time as any such costs are incurred.
F-15
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
NOTES TO FINANCIAL STATEMENTS, Continued
Recently Adopted Accounting Standards
In October 2009, the Financial Accounting Standards Board (FASB) adopted a change in the accounting standards applicable to revenue arrangements with multiple deliverables. This change eliminates the residual method of allocation and the requirement to use the relative selling price method when allocating revenue in a multiple deliverable arrangement. When applying the relative selling price method, the selling price for each deliverable needs to be determined using vendor specific objective evidence of selling price, if it exists, otherwise third-party evidence of selling price. If neither vendor specific objective evidence nor third-party evidence of selling price exists for a deliverable, it is permissible to use the best estimate of the selling price for that deliverable when applying the relative selling price method. This change is to be applied on a prospective basis for revenue arrangements entered into or materially modified in fiscal years beginning on or after June 15, 2010, with earlier application permitted. As the Company does not have any such agreements at this time, this change is not expected to have an impact on our financial statements.
Recently Issued Accounting Standards
In September 2009, the Emerging Issues Task Force of the Financial Accounting Standards Board (EITF) discussed possible changes to the standard regarding the milestone method of revenue recognition. Under the milestone method, the additional consideration earned from achievement of the milestone is viewed as being indicative of the value provided to the customer through either (a) the efforts performed by the vendor or (b) a specific outcome resulting from the vendor’s performance to achieve that specific milestone. Under the milestone method an entity recognizes contingent arrangement consideration earned from the achievement of a milestone in its entirety in the period in which the milestone is achieved. The proposed model requires that a milestone be “substantive” before this method can be applied; that is, there is a substantial uncertainty about the achievement of the milestone, substantive effort is required to achieve the milestone, and none of the payment for the milestone is refundable. The EITF did not reach a consensus on this issue. The task force will continue deliberations on this issue at future meetings. The adoption of such a standard is not expected to have an impact on our financial statements as we currently have no such arrangements.
2. Significant Agreements
Stanford License Agreements
In October 1998, the Company entered into an agreement with The Board of Trustees of Leland Stanford Junior University (Stanford) in which Stanford granted the Company an exclusive option to acquire an exclusive license for inventions and patents related to “Mifepristone for Psychotic Major Depression” and “Mifepristone and Alzheimer’s Disease” owned by Stanford.
In October 1999, the Company exercised its option to acquire an exclusive license to patents covering the use of glucocorticoid receptors antagonists for the treatment of psychotic major depression, early dementia, and cocaine-induced psychosis, as specified in the license agreement. This license agreement expires upon the expiration of the related patents or upon notification by the Company to Stanford. In exchange for the license, the Company paid Stanford $47,000 and immediately issued 30,000 shares of the Company’s common stock to Stanford. The Company is further required to pay Stanford $50,000 per year as a nonrefundable royalty payment. The annual royalty payments are creditable against future royalties. The Company is also obligated to pay a $50,000 milestone upon filing of the first NDA with the FDA for CORLUX in one of the indications covered by the license and a $200,000 milestone upon FDA approval of the related drug. The milestone payments are also creditable against future royalties. The Company has expensed the $47,000 payment made up front, the $50,000
F-16
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
NOTES TO FINANCIAL STATEMENTS, Continued
annual nonrefundable royalty payments and the value of the common stock issued to Stanford as research and development costs.
Manufacturing Agreements
In June 2000, the Company entered into a Memorandum of Understanding with a pharmaceutical manufacturer, ScinoPharm Taiwan, in which the manufacturer agreed to produce the active pharmaceutical ingredient (API) in CORLUX for the Company. In exchange, the Company agreed to share initial research and development costs related to the manufacturing process, which consisted of the acquisition of starting materials and equipment, as well as personnel costs, to complete the technology transfer, process development, and scale-up studies. Further, the Company has committed to purchase $1,000,000 per year of the API in CORLUX from the manufacturer following the receipt of marketing approval and initiation of sales of CORLUX.
On November 8, 2006, the Company signed an agreement with Produits Chimiques Auxiliaires et de Synthese SA (PCAS) for the manufacture of the API in CORLUX, for its development and commercial needs for an initial period of five years. The agreement provides for an automatic extension for one additional year unless either party gives twelve month’s prior notice that it does not want the extension. If PCAS is unable to manufacture the product for a consecutive six-month period, the Company has the right to terminate the agreement. There is no guaranteed minimum purchase commitment under this agreement.
Research and Development Agreements
In 2003, the Company entered into a contract research agreement with Argenta Discovery Limited (Argenta) in which Argenta agreed to conduct research toward identifying a novel small molecule glucocorticoid receptor antagonist for the treatment of psychotic depression, Alzheimer’s disease, and other metabolic and psychiatric disorders. The project was expected to last at least two years, during which time the Company would make payments to Argenta based upon agreed-upon FTE (full-time equivalent) rates. By December 31, 2008, work under the initial agreement with Argenta and major subsequent amendments had been concluded. The Company continues its relationship with Argenta, requesting them to conduct research projects from time to time. Under the agreements with Argenta, the Company may be obligated to make milestone payments upon the occurrence of certain events, including: (i) patent filings in connection with the project; (ii) entries into Phase 1 clinical trials; and (iii) national regulatory approval of each product arising from work performed under the agreement, provided that sales of the product by the Company or any future licensees reach $5,000,000. These obligations remain in force after the conclusion of work under the agreement.
During 2007 through 2009, the Company signed agreements for services in connection with the trials for CORLUX for the treatment of Cushing’s Syndrome and for the long-term use of the drug in this indication. The total commitment under these agreements is approximately $2.1 million. Approximately $900,000 has been expensed under these agreements through December 31, 2009, with the remainder to be expensed over the remainder of the trials. Under the master agreement with these vendors, the agreements may be terminated upon sixty days notice to the vendors. If terminated early, the Company would be responsible for the costs incurred by the vendor through the effective date of the termination plus cancellation charges as stipulated in the various agreements.
In 2008, the Company executed a Master Service Agreement (MSA) and a Project Contract (Contract), with ICON Clinical Research, L.P. (ICON) to assist the Company in various clinical trial activities, including the selection of clinical sites, supervision and monitoring of clinical site performance, data collection and analysis in connection with Study 14, the Company’s current Phase 3 trial to confirm the utility of CORLUX for the
F-17
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
NOTES TO FINANCIAL STATEMENTS, Continued
treatment of the psychotic features of psychotic depression. In June 2009, the Company amended its agreements with this vendor to reduce the amounts of commitments under these agreements in accordance with the reduction in the near-term scope of activities in this trial. However, the Company views the reduction in these commitments as a temporary measure as it is the Company’s intent to continue the conduct of this trial to its conclusion, assuming the availability of sufficient capital for this purpose. The total commitment under this agreement, including amendments executed through 2009 is estimated to be approximately $12.3 million over the course of the trial. Approximately $4.5 million of costs under this agreement has been expensed through December 31, 2009. The actual amount and timing of expense recognition and payments will depend upon various factors, including the timing of site initiation, the pace of patient enrollment, the fees negotiated with site investigators, the timing of other trial activities and the timing of payments of pass-through costs, such as grants to investigators and laboratory services. The Contract may be terminated by the Company at any time upon sixty days’ written notice, or sooner based on mutual agreement of the parties. Upon termination, the Company would be obligated to pay ICON for services performed and pass-through costs incurred to the date of termination plus a cancellation fee to compensate the CRO for staff reallocation costs.
The Company entered into an agreement with MedAvante, Inc. (MedAvante), effective March 17, 2008, under which MedAvante will provide centralized psychiatric rating services of patients to be screened and enrolled in Study 14. In June 2009, the Company amended its agreements with this vendor to reduce the amounts of commitments under these agreements in accordance with the reduction in the near-term scope of activities in this trial. However, the Company views the reduction in these commitments as a temporary measure as it is the Company’s intent to continue the conduct of this trial to its conclusion, assuming the availability of sufficient capital for this purpose. The total commitment under this agreement, including amendments executed through 2009, is approximately $4.0 million. Approximately $2.3 million of costs under this agreement has been expensed through December 31, 2009, with the remainder of any actual costs to be incurred over the remainder of the course of the trial. This agreement may be terminated by Corcept with 30 days notice to MedAvante. In the event of termination, the Company is obligated to pay certain costs including costs incurred to date, costs associated with any non-cancellable commitments for video service connectivity and costs of staff assigned to the project for a period of three months or until such time as they can be assigned to other projects, whichever is less.
During the period from October to December 2009, we signed agreements with contract research organizations and other vendors for the conduct of four of the NDA-supportive studies for aggregate commitments of approximately $2.2 million. Approximately $870,000 of costs under these agreements has been expensed as of December 31, 2009, with the remainder to be incurred in 2010.
See footnote 14 – Subsequent Events for discussion of agreements signed during the first quarter of 2010.
3. Fair Value
As of December 31, 2009, our financial assets were invested in a money market fund, which can be converted to cash at par on demand. These funds, which totaled $23.0 million, were measured at fair value as of December 31, 2009 and were classified as Level 1 assets in the fair value hierarchy for financial assets.
As of December 31, 2008, the Company’s financial assets were invested in money market funds, which can be converted to cash at par on demand, and in commercial paper and corporate debt securities with maturities of less than nine months. The following table represents the Company’s fair value hierarchy for its financial assets
F-18
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
NOTES TO FINANCIAL STATEMENTS, Continued
(cash equivalents and short-term investments) measured at fair value on a recurring basis as of December 31, 2008:
| Level 1 | Level 2 | |||||
| ( in thousands) | ||||||
| Money market funds |
$ | 13,282 | $ | — | ||
| Commercial paper |
— | 1,392 | ||||
| Corporate debt securities |
— | 2,201 | ||||
| Total |
$ | 13,282 | $ | 3,593 | ||
All cash equivalents and short-term investments held as of December 31, 2009 and 2008 were in active markets.
4. Financial Instruments
The following tables present a summary of cash, cash equivalents and short-term investments. All amounts are in thousands.
| Cost | Unrealized Gain |
Unrealized Loss |
Fair Value | |||||||||
| December 31, 2009 |
||||||||||||
| Cash |
$ | 887 | $ | — | $ | — | $ | 887 | ||||
| Money market fund |
22,980 | — | — | 22,980 | ||||||||
| $ | 23,867 | $ | — | $ | — | $ | 23,867 | |||||
| Reported as: |
||||||||||||
| Cash and cash equivalents |
$ | 23,867 | $ | — | $ | — | $ | 23,867 | ||||
| Cost | Unrealized Gain |
Unrealized Loss |
Fair Value | ||||||||||
| December 31, 2008 |
|||||||||||||
| Cash |
$ | 1,434 | $ | — | $ | — | $ | 1,434 | |||||
| Money market funds |
13,282 | — | — | 13,282 | |||||||||
| Commercial paper |
1,390 | 2 | — | 1,392 | |||||||||
| Corporate debt securities |
2,204 | — | (3 | ) | 2,201 | ||||||||
| $ | 18,310 | $ | 2 | $ | (3 | ) | $ | 18,309 | |||||
| Reported as: |
|||||||||||||
| Cash and cash equivalents |
$ | 14,716 | $ | — | $ | — | $ | 14,716 | |||||
| Short-term investments |
3,594 | 2 | (3 | ) | 3,593 | ||||||||
| $ | 18,310 | $ | 2 | $ | (3 | ) | $ | 18,309 | |||||
F-19
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
NOTES TO FINANCIAL STATEMENTS, Continued
As of December 31, 2009 and 2008, all cash, cash equivalents and investments were classified as available-for-sale securities. There were no mortgage-backed securities and no auction rate securities in the portfolio at any time during 2008 or 2009.
The net realized loss on sales of available-for-sale investments was not material for any period presented.
5. Property and Equipment
Property and equipment, including assets purchased under capitalized leases, consists of the following:
| December 31, | ||||||||
| 2009 | 2008 | |||||||
| (in thousands) | ||||||||
| Furniture and equipment |
$ | 51 | $ | 51 | ||||
| Less: accumulated depreciation and amortization |
(41 | ) | (31 | ) | ||||
| $ | 10 | $ | 20 | |||||
Amortization expense related to assets under capital lease was approximately $9,000, $12,000 and $13,000, respectively for the years ended December 31, 2009, 2008 and 2007. As of December 31, 2009, approximately $44,000 of furniture remained subject to lien under a capital lease.
6. Other Liabilities
Other accrued liabilities consisted of the following:
| December 31, | ||||||
| 2009 | 2008 | |||||
| (in thousands) | ||||||
| Accrued professional fees |
$ | 186 | $ | 150 | ||
| Accrued legal fees |
— | 149 | ||||
| Other |
38 | 17 | ||||
| $ | 224 | $ | 316 | |||
7. Lease Obligations
On October 20, 2008, the Company renewed its lease for office space for a two-year term commencing on January 1, 2009 at a monthly cost of approximately $20,000 plus operating expenses. The new lease provides an option to the Company to extend the lease for an additional year upon 180 days notice. At December 31, 2009, the remaining minimum rental payments under this operating lease were approximately $241,000.
During 2005, the Company acquired office furniture of approximately $44,000 under a capital lease that remains outstanding as of December 31, 2009. The lease is payable over 60 months at regular monthly payments of $941. The estimated principal portion of payments within the next year under this lease is approximately $6,000, which is classified as a short-term obligation.
Rent expense amounted to approximately $240,000, $265,000, $185,000, and $1.9 million for the years ended December 31, 2009, 2008 and 2007, and the period from inception (May 13, 1998) to December 31, 2009, respectively.
F-20
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
NOTES TO FINANCIAL STATEMENTS, Continued
8. Related Party Transactions
See discussion below in Note 9, Preferred Stock and Stockholders’ Equity, Stockholder Notes Receivable, regarding the collection of a note receivable from Paperboy Ventures on February 6, 2009 and, in Common Stock, regarding the sale of securities in October 2009 to various investors, including members of the Board of Directors.
On May 23, 2008 the Board of Directors approved the engagement of Abbrah Publishing LLC (Abbrah), a firm in which the son of James N. Wilson, the Company’s chairman, is a principal, to assist the Company in the preparation and placement of materials to facilitate the recruitment of patients in its Cushing’s Syndrome trial, based on, among other things, the special qualifications of Abbrah and its willingness to accept performance-based compensation of its services. Compensation to Abbrah is based the number of patients actually enrolled in the study based on their materials. An initial payment is due upon patient enrollment, with an additional amount due if the patient completes the study. During the year ended December 31, 2009, the Company recorded expense of $40,000 as compensation to Abbrah.
9. Preferred Stock and Stockholders’ Equity
Preferred Stock
The board of directors is authorized, subject to any limitations prescribed by law, without stockholder approval, to issue up to an aggregate of 10,000,000 shares of preferred stock at $0.001 par value in one or more series and to fix the rights, preferences, privileges and restrictions granted to or imposed upon the preferred stock, including voting rights, dividend rights, conversion rights, redemption privileges and liquidation preferences. The rights of the holders of common stock will be subject to the rights of holders of any preferred stock that may be issued in the future.
As of December 31, 2009 and 2008, the Company has no outstanding shares of preferred stock.
Common Stock
The Company’s authorized capital stock includes 140,000,000 shares of common stock at $0.001 par value. Holders of common stock are entitled to one vote per share on all matters to be voted upon by the stockholders of the Company.
In October 2009, the Company sold approximately 12.6 million shares of its common stock and warrants to purchase approximately 4.4 million shares of its common stock in a private placement, which we refer to as the October 2009 Financing. The securities were sold at a price of $1.43 per unit, which consisted of one share of common stock and a warrant to purchase 0.35 shares of common stock. The warrants have a three year term and an exercise price of $1.66 per share. The October 2009 Financing generated gross proceeds of approximately $18.0 million, before the deduction of issuance costs of approximately $720,000. Investors participating in the October 2009 Financing include funds managed by or investors affiliated with existing stockholders, Longitude Venture Partners, L.P., Sutter Hill Ventures and Alta Partners, LLP, and several other institutional or accredited investors. The investors also include trusts and other entities related to members of our Board of Directors, including G. Leonard Baker, Jr., Joseph C. Cook, Patrick G. Enright, David L. Mahoney and Edward E. Penhoet, Ph.D., who was a member of our Board of Directors at the time of the October 2009 Financing. Mr. Enright is a managing director of Longitude Venture Partners, L.P. Mr. Baker is a partner and managing director of Sutter Hill Ventures. Dr. Penhoet is a director of Alta Partners, LLP. Approximately 49% of the securities sold in this transaction were purchased by these venture capital funds, trusts and other entities affiliated with members of our Board of Directors for an aggregate purchase price of approximately $7.4 million.
F-21
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
NOTES TO FINANCIAL STATEMENTS, Continued
During 2009, the Company also sold 102,149 shares of common stock to Kingsbridge under the Committed Equity Financing Facility (CEFF) at an average price of $2.45 per share, for proceeds of $250,000, before the deduction of issuance costs of approximately $7,000.
Under the terms of the CEFF, which was executed in March 2008, Kingsbridge has committed to provide up to $60 million of capital in exchange for newly-issued shares of Corcept’s common stock for a period of up to three years after the SEC declares effective the registration statement covering the resale of the shares of common stock issuable in connection with the CEFF and the shares of common stock underlying the warrant discussed below. The maximum number of shares that can be sold by Corcept under this agreement is approximately 9.6 million shares. Through December 31, 2009, approximately 507,000 shares of common stock had been sold to Kingsbridge under this CEFF, for aggregate gross proceeds of approximately $1.0 million. As discussed in Note 14 – Subsequent Events, in January 2010, we sold an additional 229,031 shares of common stock to Kingsbridge under the CEFF, for total gross proceeds of $625,000. Based on the volume weighted average price on the NASDAQ Capital Market for the Company’s common stock for the period from March 25, 2008, the date of the signing of the Kingsbridge CEFF, through March 15, 2010, the maximum amount of additional funds that could be raised under the CEFF is projected to be approximately $16 million. Under the terms of the agreement, the determination of the exact timing and amount of any CEFF financings will be made solely by Corcept, subject to certain conditions. The actual amount of funds that can be raised under this agreement will be dependent on the number of shares actually sold under the agreement and the market value of Corcept’s stock during the pricing periods of each sale.
Certain details of the CEFF are as follows:
| • | Corcept can access capital under the CEFF in tranches of up to 1.25% of Corcept’s market capitalization at the time of the initiation of the draw down period, or, at Corcept’s option, the lesser of (a) 2.5% of Corcept’s market capitalization at the time of the initiation of the draw down period, and (b) an alternative draw down amount as defined in the agreement; provided, however, that in no event may the maximum draw down amount exceed $10 million per tranche, subject to certain conditions. |
| • | Each tranche will be issued and priced over an eight-day pricing period. Kingsbridge will purchase shares of common stock pursuant to the CEFF at discounts ranging from 6% to 10%, depending on the volume weighted average price of the common stock during the eight-day pricing period, provided that the minimum acceptable purchase price for any shares to be issued to Kingsbridge during the eight-day period is determined by the higher of $1.50 or 90% of Corcept’s common stock closing price the day before the commencement of each draw down. |
| • | Throughout the term of the agreement, Kingsbridge has agreed it will not, and will not cause any other person to, enter into or execute a short sale of any of Corcept’s securities. |
| • | Corcept is not obligated to utilize any of the $60 million available under the CEFF and there are no minimum commitments or minimum use penalties. The CEFF agreement does not contain any restrictions on Corcept’s operating activities, automatic pricing resets or minimum market volume restrictions. |
| • | The agreement does not prohibit Corcept from conducting additional debt or equity financings, other than financings similar to the CEFF and other future priced securities. |
| • | In connection with the CEFF, Corcept issued a warrant to Kingsbridge to purchase up to 330,000 shares of common stock at an exercise price of $3.525 per share, which represents 125% of the average of the closing bid prices of Corcept’s common stock during the 5 trading days preceding the signing of the agreement. The warrant became exercisable on September 25, 2008 and will remain exercisable, subject to certain exceptions, until five years after that date. The warrant was valued at approximately |
F-22
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
NOTES TO FINANCIAL STATEMENTS, Continued
| $655,000 using the Black-Scholes pricing model using the following assumptions: a contractual term of five and one-half years, risk-free interest rate of 2.71%, volatility of 89%, and the closing price of our stock price on the Nasdaq Capital Market on the date of signing the commitment, March 25, 2008, of $2.84 per share. The warrant value was recorded in Additional Paid In Capital with an offsetting amount recorded as issuance cost in Additional Paid In Capital. |
At the time of the signing of the CEFF agreements, the warrant issued to Kingsbridge and the shares of common stock issuable under the CEFF, and the shares issuable upon the exercise of the warrant, were not registered under the Securities Act, or state securities laws, and may not be offered or sold in the United States without being registered with the SEC or through an applicable exemption from SEC registration requirements. On June 10, 2008, the SEC declared effective the Company’s initial registration statement covering the resale of approximately 3.9 million shares, which includes approximately 3.6 million of the shares issuable under the CEFF and the shares issuable upon the exercise of the warrant. This registration statement covers approximately 37% of the 9.6 million shares of our common stock issuable pursuant to the CEFF and all of the 330,000 shares of our common stock issuable upon exercise of the warrant issued to Kingsbridge. The Company intends to file an additional registration statement covering the resale of the remaining 6.0 million shares of our common stock issuable pursuant to the CEFF approximately 60 days after Kingsbridge and its affiliates have resold substantially all of the securities registered for sale under the initial registration statement.
In March 2008, the Company sold approximately 8.9 million shares of its common stock and warrants to purchase approximately 4.5 million shares of its common stock in a private placement (the March 2008 Financing). The registration rights agreement covering securities issued in the March 2008 Financing provides that if the Company failed to file or cause to be declared effective the registration statement or registration statements covering the resale of these shares prior to specified deadlines, or fails to maintain the effectiveness of such registration statements (subject to limited permissible suspension periods), the Company may be required to pay the holders of such shares and warrants liquidated damages at the rate of 1% per month of the purchase price of these shares and warrants, up to a total of 10%. The Company filed the registration statement covering the resale of the shares sold and shares underlying the warrants sold in this transaction with the Securities and Exchange Commission (SEC) on April 11, 2008, within the time period required by the agreement. However, this registration statement was not declared effective by the SEC until November 10, 2008. During 2008, the Company recorded approximately $1.3 million in liquidated damages because of the delay in the effectiveness of the registration statement, which represented approximately 5% of the purchase price.
No dividends have been declared or paid by the Company.
Shares of common stock reserved for future issuance as of December 31, 2009 are as follows:
| (in thousands) | ||
| Common stock: |
||
| Exercise of outstanding options |
7,347 | |
| Exercise of warrants |
9,200 | |
| Shares available for grant under stock option plans |
201 | |
| 16,748 | ||
In December 2009, the Board of Directors authorized an increase of 2,498,987 shares in the shares available under the 2004 Plan to be effective on January 1, 2010, which amount is based on 4% of the shares of the Company’s common stock outstanding as of December 31, 2009, pursuant to the terms of the 2004 Plan.
F-23
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
NOTES TO FINANCIAL STATEMENTS, Continued
Stock Option Plans
In October 2000, the Company adopted the 2000 Stock Option Plan (the 2000 Plan), which was amended in May 2001, to provide for the issuance of option grants for up to 2,000,000 shares of the Company’s common stock to eligible participants. Under the 2000 Plan, options to purchase common stock could be granted at no less than 100% of fair value on the date of grant for incentive stock options and 85% of fair value on the date of grant for nonqualified options, as determined by the board of directors. Options became exercisable at such times and under such conditions as determined by the board of directors. As of December 31, 2009, all option grants under this plan were fully vested, with grants covering approximately 360,000 shares remaining outstanding with contractual lives expiring in 2010 through 2014. Vested shares under this plan that are not exercised within the remaining contractual life will expire on that date and not be added to the pool of shares available for future grant.
In 2004, the Company’s board of directors and stockholders approved the 2004 Equity Incentive Plan (the 2004 Plan), which became effective upon the completion of the IPO, after which time, no additional options are being issued under the 2000 Plan. Under the 2004 Plan, options, stock purchase and stock appreciation rights and restricted stock awards can be issued to employees, officers, directors and consultants of the Company. The 2004 Plan provides that the exercise price for incentive stock options will be no less than 100% of the fair value of the Company’s common stock, as of the date of grant. Options granted under the 2004 Plan vest over periods ranging from one to five years. The vesting period of the options is generally equivalent to the requisite service period.
Upon exercise, new shares are issued.
In December 2008, the Board of Directors authorized an increase of 995,264 shares in the shares available under the 2004 Plan that was effective on January 1, 2009. In June 2009, the shareholders approved a proposal that (a) increased the number of shares available under the plan by 1,000,000 shares, and increased the parameters for annual increase to be the least of (a) 4% of the number of the Company’s shares issued and outstanding at the preceding December 31, (b) 4,000,000 shares and (c) a number of shares set by the Board of Directors. As of December 31, 2009, the total number of shares authorized for issuance under the 2004 Plan was 7,208,818, of which 201,044 shares remained available for future grants. See discussion above under Common Stock regarding an additional increase to the shares available for grant under the 2004 Plan that was authorized by the Board of Directors in December 2009 and effective as of January 1, 2010.
F-24
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
NOTES TO FINANCIAL STATEMENTS, Continued
Option activity during 2007, 2008 and 2009
The following table summarizes all stock plan activity:
| Stock Options | |||||||||
| Shares Available |
Options Outstanding |
Weighted- Average Exercise Price | |||||||
| (in thousands, except per share data) | |||||||||
| Balance at December 31, 2006 |
2,680 | 1,810 | $ | 5.80 | |||||
| Increase in shares authorized under 2004 Plan |
514 | — | — | ||||||
| Shares granted |
(2,502 | ) | 2,502 | $ | 1.61 | ||||
| Shares exercised |
— | (26 | ) | $ | 0.79 | ||||
| Shares cancelled and forfeited under 2004 Plan |
195 | (195 | ) | $ | 5.40 | ||||
| Shares cancelled and forfeited under 2000 Plan |
— | (200 | ) | $ | 7.00 | ||||
| Balance at December 31, 2007 |
887 | 3,891 | $ | 3.10 | |||||
| Increase in shares authorized under 2004 Plan |
791 | — | — | ||||||
| Shares granted |
(1,273 | ) | 1,273 | $ | 1.48 | ||||
| Shares exercised |
— | (2 | ) | $ | 1.50 | ||||
| Shares issued for services |
(1 | ) | — | $ | 2.73 | ||||
| Shares cancelled and forfeited under 2004 Plan |
30 | (30 | ) | $ | 2.70 | ||||
| Balance at December 31, 2008 |
434 | 5,132 | $ | 2.70 | |||||
| Increase in shares authorized under 2004 Plan |
1,995 | — | — | ||||||
| Shares granted |
(2,300 | ) | 2,300 | $ | 1.36 | ||||
| Shares issued for services |
(13 | ) | — | $ | 1.17 | ||||
| Shares cancelled and forfeited under 2004 Plan |
85 | (85 | ) | $ | 2.62 | ||||
| Balance at December 31, 2009 |
201 | 7,347 | $ | 2.28 | |||||
All stock option grants are service-based awards, with the exception of the performance-based awards granted during 2009 to Joseph K. Belanoff, the Company’s Chief Executive Officer, and Robert L. Roe, the Company’s President, in the amounts of 500,000 shares and 200,000 shares, respectively. These performance-based grants will vest in their entirety upon the approval of the New Drug Application for the Company’s first product by the U.S. Food and Drug Administration.
The total intrinsic value of options exercised during the years ended December 31, 2007 and 2008 was approximately $37,000 and $1,000, respectively based on the difference between the closing price of the Company’s common stock on the date of exercise of the options and the exercise price. There were no exercises of options during the year ended December 31, 2009.
F-25
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
NOTES TO FINANCIAL STATEMENTS, Continued
The following table presents the total fair value of options to employees that vested during the years ended December 31, 2009, 2008 and 2007. All amounts are in thousands.
| Year ended December 31, | |||||||||
| 2009 | 2008 | 2007 | |||||||
| Pre-IPO options, using minimum value method |
$ | — | $ | 197 | $ | 342 | |||
| Options granted after IPO through 2005, using fair value |
318 | 475 | 487 | ||||||
| Options granted after January 1, 2006, using fair values |
1,849 | 1,330 | 1,052 | ||||||
| Total |
$ | 2,167 | $ | 2,002 | $ | 1,881 | |||
As of December 31, 2009, the Company had approximately $4.1 million of unrecognized compensation expense for employee options outstanding as of that date, which had a remaining weighted-average vesting period of 2.3 years.
The following is a summary of options outstanding and options exercisable at December 31, 2009. All options outstanding at December 31, 2009 are either exercisable or expected to become exercisable.
| Options Outstanding | Options Exercisable | |||||||||||||||||
| Number of Shares |
Weighted Average Remaining Contractual Life |
Weighted Average Exercise Price |
Aggregate Intrinsic Value |
Options Exercisable |
Weighted Average Exercise Price |
Aggregate Intrinsic Value | ||||||||||||
| (in thousands) | (in years) | (in thousands) | (in thousands) | (in thousands) | ||||||||||||||
| $ 0.10 - $ 1.19 |
2,741 | 9.0 | $ | 1.11 | $ | 4,577 | 545 | $ | 1.01 | $ | 963 | |||||||
| $ 1.50 - $ 2.98 |
3,257 | 7.8 | $ | 1.75 | $ | 3,356 | 1,855 | $ | 1.67 | 2,063 | ||||||||
| $ 4.00 - $ 7.73 |
1,213 | 5.3 | $ | 5.26 | — | 1,130 | $ | 5.33 | — | |||||||||
| $ 10.06 - $ 15.00 |
136 | 4.4 | $ | 12.07 | — | 136 | $ | 12.07 | — | |||||||||
| 7,347 | 7.8 | $ | 2.28 | $ | 7,933 | 3,666 | $ | 3.09 | $ | 3,026 | ||||||||
The aggregate intrinsic value in the table above represents the total pre-tax intrinsic value that option holders would have received had all option holders exercised their options on December 31, 2009. The aggregate intrinsic value is the difference between the Company’s closing stock price on December 31, 2009 and the exercise price, multiplied by the number of in-the-money options.
Stock-Based Compensation related to Employee Options
Accounting Practices
For options granted prior to the Company’s initial public offering (IPO) in 2004, the Company accounted for options granted to employees and directors based on the intrinsic value of these grants. This treatment was followed even after the change in accounting pronouncements affecting stock-based compensation in January 2006 because the Company had used the “minimum” value method for these options for purposes of pro-forma disclosure under the previous guidance. During the period from inception (May 13, 1998) to September 30, 2008, the Company recorded $10.1 million in deferred compensation for employee stock options to purchase common stock granted at exercise prices deemed to be below the fair value of common stock at the date of grant. The Company amortized the deferred stock-based compensation to expense utilizing the graded-vesting method over the vesting periods of the applicable stock options, generally five years. As of September 30, 2008, the deferred compensation related to these options was fully amortized.
F-26
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
NOTES TO FINANCIAL STATEMENTS, Continued
For options granted after the IPO, the Company began, as of January 1, 2006, to account for stock-based compensation related to option grants to employees and directors under the fair value method. Following is a synopsis of the Company’s accounting practices in regard to these stock option grants:
| • | Options granted after the IPO but prior to January 1, 2006: |
| ¡ | Prior to the January 1, 2006, the Company had accounted for employee options under the intrinsic value method, which did not require the recognition of any compensation expense as the grants had been issued at the market value on the date of grant. |
| ¡ | The Company began, as of January 1, 2006, to record non-cash stock-based compensation expense related to these grants in the financial statements based on the remaining fair value, as of January 1, 2006, of the non-vested portion of these grants, utilizing the assumptions and fair value per share information as of the original grant date that the Company has been using for pro forma disclosure purposes. The Company is continuing to utilize the graded-vesting attribution method for amortization of the relevant compensation amounts, which will be fully expensed early in 2010. |
| • | Options granted on or after January 1, 2006: |
| ¡ | Compensation expense is being recorded in the financial statements based on the fair value on the date of grant. |
| ¡ | For service based awards, the grant date fair value being amortized to expense using the straight-line attribution method over the vesting period of the options, which is commensurate with the service period. |
| ¡ | For performance-based awards, expense will be recognized at such time as there is a high degree of probability (i.e., greater than 70%) of achieving the required objective. |
Assumptions used in determining fair value for options granted to employees
The following table summarizes the weighted-average assumptions and resultant fair value for options granted to employees.
| Year Ended December 31, | ||||||||||||
| 2009 | 2008 | 2007 | ||||||||||
| Weighted average assumptions for stock options granted: |
||||||||||||
| Risk-free interest rate |
2.24 | % | 2.63 | % | 4.59 | % | ||||||
| Expected term |
6.0 years | 6.0 years | 6.0 years | |||||||||
| Expected volatility of stock price |
94.2 | % | 90.6 | % | 87.2 | % | ||||||
| Dividend rate |
0 | % | 0 | % | 0 | % | ||||||
| Weighted average grant date fair value |
$ | 1.05 | $ | 1.12 | $ | 1.21 | ||||||
The expected term for options granted since January 1, 2006 is based on the “simplified” method prescribed by the SEC, and considers the weighted average of the vesting period and contractual life of the options. There has been no adjustment made to the expected term to adjust for employees’ expected exercise and expected post-vesting termination behavior because the Company has a limited employee base and does not have sufficient historical information to determine such an adjustment.
The expected volatility of the Company’s stock used in determining the fair value of option grants is based on a weighted-average combination of the volatility of the Company’s own stock price and that of a group of
F-27
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
NOTES TO FINANCIAL STATEMENTS, Continued
peer companies since the Company does not have sufficient historical data from which to base an appropriate valuation assumption.
Since the Company has a limited employee base, it does not have sufficient historical information to determine a reasonable forfeiture rate for options that might not vest because of employee terminations and does not, therefore, apply a forfeiture rate. When an employee terminates, the Company will record a change in accounting estimate that represents the difference between the expense recorded in the financial statements and the expense that would have been recorded based upon the rights to options that vested during the individual’s service as an employee.
Summary of compensation expense related to employee options
Compensation expense of approximately $1.8 million, $1.6 million, $1.0 million, and $13.6 million was recognized for employee options during the years ended December 31, 2009, 2008 and 2007 and for the period from inception (May 13, 1998) to December 31, 2009, respectively, net of recoveries.
During the year ended December 31, 2007, the Company recorded recoveries of previously reported stock-based compensation expense of approximately $420,000 upon the termination of employees. These amounts represent the difference between the expense recorded under the graded-vesting method and the expense that would have been recorded based upon the rights to options that vested during the service of these individuals as employees. The recoveries recorded during 2007 included approximately $395,000 related to an officer and $25,000 related to a development employee. There were no similar recoveries recorded during 2008 or 2009.
In addition, the Company reversed approximately $120,000 during the year ended December 31, 2007, from deferred compensation related to outstanding options forfeited by an employee who terminated during the year, as the rights to the underlying shares had not fully vested by the date of conversion or termination of service as employees. There was no similar reversal in 2008 or 2009.
Stock Options to Consultants
Stock-based compensation related to option grants to non-employees is charged to expense on a straight line basis over the vesting period of the options, based on the fair value of the options, which approximates the period over which the related services are rendered, using the Black-Scholes option pricing model. The assumptions used in these calculations are similar to those used for the determination of fair value for options granted to employees, with the exception that, for non-employee options, the remaining contractual term is utilized as the expected term of the option and the fair value related to unvested non-employee options is re-measured quarterly, based on the then current stock price as reflected on the Nasdaq Capital Market.
The Company recorded charges in the statement of operations for stock options granted to consultants using the straight-line vesting method of approximately $11,000, $30,000, $50,000 and $980,000 for the years ended December 31, 2009, 2008, 2007 and the period from inception (May 13, 1998) to December 31, 2009, respectively. The straight-line method is commensurate with the services being provided by such consultants.
As of December 31, 2009, all options that had been granted to consultants were fully vested.
Stockholder Notes Receivable
In 2001, the Company recorded notes receivable from stockholders in the aggregate amount of $438,165 in connection with the exercise of 585,000 shares of common stock options issued under the 2000 Plan. The notes
F-28
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
NOTES TO FINANCIAL STATEMENTS, Continued
are secured by the related shares of common stock and are full recourse notes, with interest compounded annually at the rate of 6.5% per year. The notes mature ten years from the date of issuance. As of December 31, 2009, the amounts outstanding under these notes included principal in the amount of approximately $100,000 and interest in the amount of approximately $60,000.
One of the investors in the March 2008 Financing financed the purchase of its securities in this transaction with a promissory note to the Company in the amount of $6.0 million. On February 6, 2009, the Company collected this note in full, including all accrued interest to that date and expenses associated with the note. Upon receipt of the funds, the Company released its interest in the collateral that had been held as security for the note.
Warrants
On October 12, 2009, the Company sold warrants to purchase approximately 4.4 million shares of its common stock in the October 2009 Financing. See discussion above under the caption Common Stock.
Outstanding warrants at December 31, 2009 were as follows:
| Number of shares | Exercise Price | Expiration Date | |||||
| March 2008 Financing |
4,461,599 | $ | 2.77 | 3/25/15 | |||
| Kingsbridge CEFF |
330,000 | $ | 3.525 | 9/25/13 | |||
| October 2009 Financing |
4,408,773 | $ | 1.66 | 10/16/12 | |||
10. Net Loss Per Share
Basic and diluted net loss per share is computed by dividing the net loss by the weighted-average number of common shares outstanding during the period less outstanding shares subject to repurchase. The computation of net loss per share for each period, including the number of weighted-average shares outstanding, is shown on the face of the statements of operations.
The Company has excluded the impact of common stock equivalents from the calculation of diluted net loss per common share because all such securities are antidilutive for all periods presented. In addition, for all periods presented, the Company excluded additional shares that might have been issued under stock option grants.
The following table presents information on securities outstanding as of the end of each period that could potentially dilute the per share data in the future.
| December 31, | ||||||
| 2009 | 2008 | 2007 | ||||
| (in thousands) | ||||||
| Stock options outstanding |
7,347 | 5,132 | 3,891 | |||
| Warrants outstanding |
9,200 | 4,792 | — | |||
| Total |
16,547 | 9,924 | 3,891 | |||
F-29
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
NOTES TO FINANCIAL STATEMENTS, Continued
11. Income Taxes
Deferred income taxes reflect the net tax effects of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for income tax purposes. Significant components of the Company’s deferred tax assets are as follows:
| December 31, | ||||||||
| 2009 | 2008 | |||||||
| (in thousands) | ||||||||
| Deferred tax assets: |
||||||||
| Federal and state net operating losses |
$ | 30,463 | $ | 24,406 | ||||
| Capitalized research and patent costs |
22,734 | 22,158 | ||||||
| Stock-based compensation costs |
1,756 | 1,319 | ||||||
| Research credits |
5,743 | 4,408 | ||||||
| Total deferred tax assets |
60,696 | 52,291 | ||||||
| Valuation allowance |
(60,696 | ) | (52,291 | ) | ||||
| Net deferred tax assets |
$ | — | $ | — | ||||
Realization of deferred tax assets is dependent upon future earnings, if any, the timing and amount of which are uncertain. Accordingly, the net deferred tax assets have been fully offset by a valuation allowance. The valuation allowance increased by $8.4 million, $8.9 million and $6.1 million for the years ended December 31, 2009, 2008 and 2007, respectively.
As of December 31, 2009, the Company had net operating loss carryforwards for federal income tax purposes of approximately $76.4 million, which expire in the years 2019 through 2029. The Company also has California net operating loss carryforwards of approximately $76.7 million, which expire in the years 2012 through 2029. The Company also has federal and California research and development tax credits of approximately $4.9 million and $1.3 million, respectively. The federal research credits will expire in the years 2019 through 2029 and the California research credits have no expiration date. All tax years from inception remain open to examination by the Internal Revenue Service and the California Franchise Tax Board until such time as the net operating losses and research credits are either fully utilized or expire.
Utilization of the Company’s net operating loss may be subject to substantial annual limitation due to the ownership change limitations provided by the Internal Revenue Code and similar state provisions. Such an annual limitation could result in the expiration of the net operating loss before utilization.
A reconciliation from the statutory federal income tax rate to the effective rate is as follows:
| Year ended December 31, | ||||||||||||
| 2009 | 2008 | 2007 | ||||||||||
| (in thousands) | ||||||||||||
| U.S. federal taxes (benefit) at statutory rate |
$ | (6,856 | ) | $ | (6,821 | ) | $ | (3,935 | ) | |||
| State tax |
— | — | — | |||||||||
| Unutilized, net operating loss |
6,649 | 6,597 | 3,814 | |||||||||
| Non-deductible stock based compensation |
207 | 168 | 112 | |||||||||
| Other |
— | 56 | 9 | |||||||||
| Total |
$ | — | $ | — | $ | — | ||||||
F-30
Table of Contents
CORCEPT THERAPEUTICS INCORPORATED
(A DEVELOPMENT STAGE COMPANY)
NOTES TO FINANCIAL STATEMENTS, Continued
12. Commitments
During 2007 through 2009 the Company executed a number of agreements to conduct clinical trials and pre-clinical studies for further development of its lead product, CORLUX, targeted for the treatment of psychotic depression and Cushing’s Syndrome, as well as other product indications. See the discussion in Note 2—Significant Agreements—Research Agreements and Development Agreements—for further discussion regarding these agreements.
In the ordinary course of its business, the Company makes certain indemnities, commitments and guarantees under which it may be required to make payments in relation to certain transactions. These include indemnities of clinical investigators and contract research organizations involved in the development of the Company’s clinical stage product candidates, indemnities of contract manufacturers and indemnities to directors and officers of the Company to the maximum extent permitted under the laws of the State of Delaware. The duration of these indemnities, commitments and guarantees varies, and in certain cases, is indefinite. The majority of these indemnities, commitments and guarantees do not provide for any limitation of the maximum potential future payments the Company could be obligated to make. The Company has not recorded any liability for these indemnities, commitments and guarantees in the accompanying balance sheets. However, the Company accrues for losses for any known contingent liability, including those that may arise from indemnification provisions, when future payment is probable. No such losses have been recorded to date.
13. Quarterly Financial Data (Unaudited)
The following table is in thousands, except per share amounts:
| Quarter Ended |
March 31 | June 30 | September 30 | December 31 | ||||||||||||
| 2009 |
||||||||||||||||
| Net revenue |
$ | 24 | $ | 5 | — | — | ||||||||||
| Net loss |
$ | (5,450 | ) | $ | (4,877 | ) | $ | (4,668 | ) | $ | (5,170 | ) | ||||
| Basic and diluted net loss per share |
$ | (0.11 | ) | $ | (0.10 | ) | $ | (0.09 | ) | $ | (0.09 | ) | ||||
| 2008 |
||||||||||||||||
| Net revenue |
— | — | $ | 66 | $ | 143 | ||||||||||
| Net loss |
$ | (3,930 | ) | $ | (4,396 | ) | $ | (5,565 | ) | $ | (6,170 | ) | ||||
| Basic and diluted net loss per share |
$ | (0.10 | ) | $ | (0.09 | ) | $ | (0.11 | ) | $ | (0.13 | ) | ||||
14. Subsequent Events
In January 2010, the Company sold 229,031 shares of common stock to Kingsbridge under the CEFF, at an average cost of $2.73 per share, for total gross proceeds of $625,000.
In February 2010, the Company signed an agreement with a clinical research organization for the conduct of a Phase 1 clinical study to evaluate CORT 108297 for a commitment of approximately $690,000 and, in March 2010, additional agreements were signed with two contract research organizations and a clinical research site for the conduct of two NDA-supportive studies for CORLUX for aggregate commitments of approximately $1.2 million. All of these amounts are expected to be expended in 2010.
F-31
Table of Contents
Exhibit Index
| Exhibit |
Description of Document | |
| 3.1 | Amended and Restated Certificate of Incorporation (incorporated by reference to Exhibit 3.1 to the registrant’s Registration Statement on Form S-1/A (File No. 333-112676) filed on March 19, 2004). | |
| 3.2 | Amended and Restated Bylaws (incorporated by reference to Exhibit 3.1 to the registrant’s Current Report on Form 8-K filed on September 27, 2007). | |
| 4.1 | Specimen Common Stock Certificate (incorporated by reference to Exhibit 4.1 to the registrant’s Registration Statement on Form S-1 (Registration No. 333-112676) filed on February 10, 2004). | |
| 4.2 | Amended and Restated Information and Registration Rights Agreement by and among Corcept Therapeutics Incorporated and certain holders of preferred stock, dated as of May 8, 2001 (incorporated by reference to Exhibit 4.2 to the registrant’s Registration Statement on Form S-1 (Registration No. 333-112676) filed on February 10, 2004). | |
| 4.3 | Amendment No. 1 to Amended and Restated Information and Registration Rights Agreement by and among Corcept Therapeutics Incorporated and certain holders of preferred stock, dated as of March 16, 2004 (incorporated by reference to Exhibit 4.3 to the registrant’s Registration Statement on Form S-1/A (File No. 333-112676) filed on March 19, 2004). | |
| 4.4 | Registration Rights Agreement by and among Corcept Therapeutics Incorporated and the investors signatory thereto, dated March 14, 2008 (incorporated by reference to Exhibit 10.25 to the registrant’s Annual Report on Form 10-K filed on March 31, 2008). | |
| 4.5 | Registration Rights Agreement by and between Corcept Therapeutics Incorporated and Kingsbridge Capital Limited, dated as of March 25, 2008 (incorporated by reference to Exhibit 10.27 to the registrant’s Annual Report on Form 10-K filed on March 31, 2008). | |
| 4.6 | Registration Rights Agreement by and among Corcept Therapeutics Incorporated and the investors signatory thereto, dated October 12, 2009 (incorporated by reference to Exhibit 4.2 to the registrant’s Quarterly Report on Form 10-Q filed on November 12, 2009). | |
| 10.1* | 2000 Stock Option Plan (incorporated by reference to Exhibit 10.1 to the registrant’s Registration Statement on Form S-1 (Registration No. 333-112676) filed on February 10, 2004). | |
| 10.2* | Promissory Note and Pledge Agreement by and between Corcept Therapeutics Incorporated and Robert L. Roe, M.D., dated as of October 22, 2001 (incorporated by reference to Exhibit 10.4 to the registrant’s Registration Statement on Form S-1 (Registration No. 333-112676) filed on February 10, 2004). | |
| 10.3# | License Agreement by and between The Board of Trustees of the Leland Stanford Junior University and Corcept Therapeutics Incorporated, dated as of July 1, 1999 (incorporated by reference to Exhibit 10.6 to the registrant’s Registration Statement on Form S-1 (Registration No. 333-112676) filed on February 10, 2004). | |
| 10.4 | Master Clinical Development Agreement by and between Corcept Therapeutics Incorporated and Scirex Corporation, dated as of July 12, 2001 (incorporated by reference to Exhibit 10.8 to the registrant’s Registration Statement on Form S-1 (Registration No. 333-112676) filed on February 10, 2004). | |
| 10.5# | Memorandum of Understanding, Supply and Services Agreement, by and between Corcept Therapeutics Incorporated and ScinoPharm Taiwan, dated as of June 12, 2000 (incorporated by reference to Exhibit 10.9 to the registrant’s Registration Statement on Form S-1/A (File No. 333-112676) filed on March 19, 2004). | |
Table of Contents
| Exhibit |
Description of Document | |
| 10.6 | Master Services Agreement by and between Corcept Therapeutics Incorporated and PPD Development, LP, dated as of January 17, 2003 (incorporated by reference to Exhibit 10.12 to the registrant’s Registration Statement on Form S-1/A (File No. 333-112676) filed on March 19, 2004). | |
| 10.7 | Master Services Agreement by and between Corcept Therapeutics Incorporated and i3 Research, a division of Ingenix Pharmaceuticals Services (UK) Limited, dated as of October 28, 2004 (incorporated by reference to Exhibit 10.13 to the registrant’s Annual Report on Form 10-K filed on March 29, 2005). | |
| 10.8## | Manufacturing Agreement with Produits Chimgues Auxiliaries et de Synthese SA, dated November 8, 2006 (incorporated by reference to Exhibit 10.15 to the registrant’s Annual Report on Form 10-K filed on April 2, 2007). | |
| 10.9 | Common Stock Purchase Agreement by and among Corcept Therapeutics Incorporated and each of the Purchasers listed on Exhibit A thereto, dated November 14, 2006 (incorporated by reference to Exhibit 10.1 to the registrant’s Current Report on Form 8-K filed on November 16, 2006). | |
| 10.10 | Common Stock Purchase Agreement by and among Corcept Therapeutics Incorporated and each of those persons and entities listed on the Schedule of Purchasers thereto, dated as of March 30, 2007 (incorporated by reference to Exhibit 10.1 to the registrant’s Current Report on Form 8-K filed on April 3, 2007). | |
| 10.11 | Common Stock Purchase Agreement by and among Corcept Therapeutics Incorporated and each of those persons and entities listed on the Schedule of Purchasers thereto, dated as of August 16, 2007 (incorporated by reference to Exhibit 10.1 to the registrants Current Report on Form 8-K filed on August 21, 2007). | |
| 10.12* |
Form of Indemnification Agreement for directors and officers approved by the Board of Directors on September 24, 2007 (incorporated by reference to Exhibit 10.7 to the registrant’s Quarterly Report on Form 10-Q filed on November 14, 2007). | |
| 10.13 | Securities Purchase Agreement by and among Corcept Therapeutics Incorporated and the purchasers named therein, dated March 14, 2008 (incorporated by reference to Exhibit 10.24 to the registrant’s Annual Report on Form 10-K filed on March 31, 2008). | |
| 10.14 | Form of Warrant issued in connection with the Securities Purchase Agreement by and among Corcept Therapeutics Incorporated and the purchasers named therein, dated March 14, 2008 (incorporated by reference to Exhibit 4.4 to the registrant’s Annual Report on Form 10-K filed on March 31, 2008). | |
| 10.15 | Common Stock Purchase Agreement by and between Kingsbridge Capital Limited and Corcept Therapeutics Incorporated dated as of March 25, 2008 (incorporated by reference to Exhibit 10.26 to the registrant’s Annual Report on Form 10-K filed on March 31, 2008). | |
| 10.16 | Warrant, dated March 25, 2008 issued to Kingsbridge Capital Limited (incorporated by reference to Exhibit 4.5 to the registrant’s Annual Report on Form 10-K filed on March 31, 2008). | |
| 10.17# | Master Service Agreement by and among Corcept Therapeutics Incorporated and ICON Clinical Research, L.P., signed on June 4, 2008 (incorporated by reference to Exhibit 10.5 to the registrant’s Quarterly Report on Form 10-Q filed on August 14, 2008). | |
| 10.18* | Amended and Restated Severance and Change in Control Agreement by and between Corcept Therapeutics Incorporated and Joseph K. Belanoff, M. D., dated September 19, 2008 (incorporated by reference to Exhibit 10.25 to the registrant’s Annual Report on Form 10-K filed on March 31, 2009). | |
| 10.19* | Amended and Restated Severance and Change in Control Agreement by and between Corcept Therapeutics Incorporated and Robert L. Roe, M. D., dated September 19, 2008 (incorporated by reference to Exhibit 10.26 to the registrant’s Annual Report on Form 10-K filed on March 31, 2009). | |
Table of Contents
| Exhibit |
Description of Document | |
| 10.20* | Amended and Restated Severance and Change in Control Agreement by and between Corcept Therapeutics Incorporated and Anne M. LeDoux, dated September 19, 2008 (incorporated by reference to Exhibit 10.27 to the registrant’s Annual Report on Form 10-K filed on March 31, 2009). | |
| 10.21* | Amended and Restated Severance and Change in Control Agreement by and between Corcept Therapeutics Incorporated and James N. Wilson, dated September 19, 2008 (incorporated by reference to Exhibit 10.28 to the registrant’s Annual Report on Form 10-K filed on March 31, 2009). | |
| 10.22* | Employment offer letter to Caroline M. Loewy, dated October 21, 2008 (incorporated by reference to Exhibit 10.29 to the registrant’s Annual Report on Form 10-K filed on March 31, 2009). | |
| 10.23 | Amendment to Registration Rights Agreement by and among Corcept Therapeutics Incorporated and the investors signatory thereto, dated November 11, 2008 (incorporated by reference to Exhibit 10.30 to the registrant’s Annual Report on Form 10-K filed on March 31, 2009). | |
| 10.24* | Severance and Change in Control Agreement by and between Corcept Therapeutics Incorporated and Caroline M. Loewy, dated November 28, 2008 (incorporated by reference to Exhibit 10.31 to the registrant’s Annual Report on Form 10-K filed on March 31, 2009). | |
| 10.25 | Form of Warrant issued in connection with the Securities Purchase Agreement by and among Corcept Therapeutics Incorporated and the purchasers named therein, dated October 12, 2009 (incorporated by reference to Exhibit 4.1 to the registrant’s Quarterly Report on Form 10-Q filed on September 30, 2009). | |
| 10.26 | Securities Purchase Agreement by and among Corcept Therapeutics Incorporated and the purchasers named therein, dated October 12, 2009 (incorporated by reference to Exhibit 10.1 to the registrant’s Quarterly Report on Form 10-Q filed on September 30, 2009). | |
| 10.27* | Amended and Restated 2004 Equity Incentive Plan (incorporated by reference to Exhibit 10.1 to the registrant’s Quarterly Report on Form 10-Q filed on June 30, 2009). | |
| 14.1 | Code of Ethics (incorporated by reference to Exhibit 99.1 to the registrant’s Registration Statement on Form S-1/A (File No. 333-112676) filed on March 19, 2004). | |
| 23.1 | Consent of Independent Registered Public Accounting Firm | |
| 24.1 | Power of Attorney (See signature page) | |
| 31.1 | Certification pursuant to Rule 13a-14(a) under the Securities Exchange Act of 1934 of Joseph K. Belanoff, M.D. | |
| 31.2 | Certification pursuant to Rule 13a-14(a) under the Securities Exchange Act of 1934 of Caroline M. Loewy | |
| 32.1 | Certification pursuant to 18 U.S.C. Section 1350 of Joseph K. Belanoff, M.D. | |
| 32.2 | Certification pursuant to 18 U.S.C. Section 1350 of Caroline M. Loewy | |
| # | Confidential treatment granted |
| ## | Confidential treatment requested |
| * | Management contract or compensatory plan or arrangement |
