Attached files

1
Cowen and Company
Health Care Conference
March 10, 2010
NYSE Amex: PTX
EXHIBIT 99.1

2
The following presentation includes “forward-looking statements” within the meaning of the “safe harbor” provisions of
the Private Securities Litigation Reform Act of 1995. You should be aware that our actual results could differ materially
from those contained in the forward looking statements, which are based on management’s current expectations and
are subject to a number of risks and uncertainties, including, but not limited to, our failure to successfully commercialize
our product candidates; costs and delays in the development and/or FDA approval of our product candidates, including
as a result of the need to conduct additional studies, or the failure to obtain such approval of our product candidates,
including as a result of changes in regulatory standards or the regulatory environment with any of our product
candidates; our inability to maintain or enter into, and the risks resulting from our dependence upon, collaboration or
contractual arrangements necessary for the development, manufacture, commercialization, marketing, sales and
distribution of any products, including our inability to protect our patents or proprietary rights and obtain necessary rights
to third party patents and intellectual property to operate our business; our inability to operate our business without
infringing the patents and proprietary rights of others; general economic conditions; the failure of any products to gain
market acceptance; our inability to obtain any additional required financing; technological changes; government
regulation; changes in industry practice; and one-time events. More detailed information about the Company and the
risk factors that may affect the realization of forward-looking statements is set forth in the Company’s filings with the
Securities and Exchange Commission. Such documents may be read free of charge on the SEC’s web site at
www.sec.gov. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as
of the date hereof.
the Private Securities Litigation Reform Act of 1995. You should be aware that our actual results could differ materially
from those contained in the forward looking statements, which are based on management’s current expectations and
are subject to a number of risks and uncertainties, including, but not limited to, our failure to successfully commercialize
our product candidates; costs and delays in the development and/or FDA approval of our product candidates, including
as a result of the need to conduct additional studies, or the failure to obtain such approval of our product candidates,
including as a result of changes in regulatory standards or the regulatory environment with any of our product
candidates; our inability to maintain or enter into, and the risks resulting from our dependence upon, collaboration or
contractual arrangements necessary for the development, manufacture, commercialization, marketing, sales and
distribution of any products, including our inability to protect our patents or proprietary rights and obtain necessary rights
to third party patents and intellectual property to operate our business; our inability to operate our business without
infringing the patents and proprietary rights of others; general economic conditions; the failure of any products to gain
market acceptance; our inability to obtain any additional required financing; technological changes; government
regulation; changes in industry practice; and one-time events. More detailed information about the Company and the
risk factors that may affect the realization of forward-looking statements is set forth in the Company’s filings with the
Securities and Exchange Commission. Such documents may be read free of charge on the SEC’s web site at
www.sec.gov. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as
of the date hereof.
Note: All product sales data included herein are derived from data published by Wolters Kluwer Health for the 12
months ended December 2009.
months ended December 2009.
Trademarks referenced herein are the property of their respective owners. ©2010 Pernix Therapeutics, Inc.
Safe Harbor Statement

3
A Specialty Pharmaceutical Company Focused on Serving the
Pediatric Marketplace with Commercially-Proven Branded Products
• Pernix is defined as swift, nimble, and agile in Latin
• Sudden Change
– This allows Pernix to take advantage of opportunities that other companies
cannot
cannot
• Many Members of our Senior Management and Field Sales
Representatives are Former Collegiate Athletes
Representatives are Former Collegiate Athletes
– Hate to lose
– Understand how to come back from a loss
– Athletes that excel understand working when you are not asked to, which
separates you from the pack
separates you from the pack
– Only get rewarded for winning
– Get cut when they don’t perform
Pernix Philosophy

4
4
The executive management has experience with Pernix Therapeutics,
Pfizer, Biovail, Cornerstone Therapeutics, DJ Pharma, Amneal
Pharmaceuticals, and Sepracor.
Leaders that have helped build and enjoy building companies.
Cooper Collins, Chief Executive Officer
Tracy Clifford, Chief Financial Officer
Mike Venters, Executive VP - Operations
Dave Waguespack, VP - Internal Operations
Beth DeVille, VP - Communications & Training
Robert Cline , VP - Supply Chain Management
Shawn Doll, Director - U.S. Sales East
Brandon Belanger, Director - U.S. Sales West
Pernix Management

5
5
We are in a great position to take advantage of new business
opportunities.
opportunities.
Ø Debt-Free Balance Sheet
Ø Self-Financing to date and Strong Cash Position
Product candidates are evaluated against numerous criteria.
Ø High Gross Margins
Ø Low Product Development Risk
Pernix Today
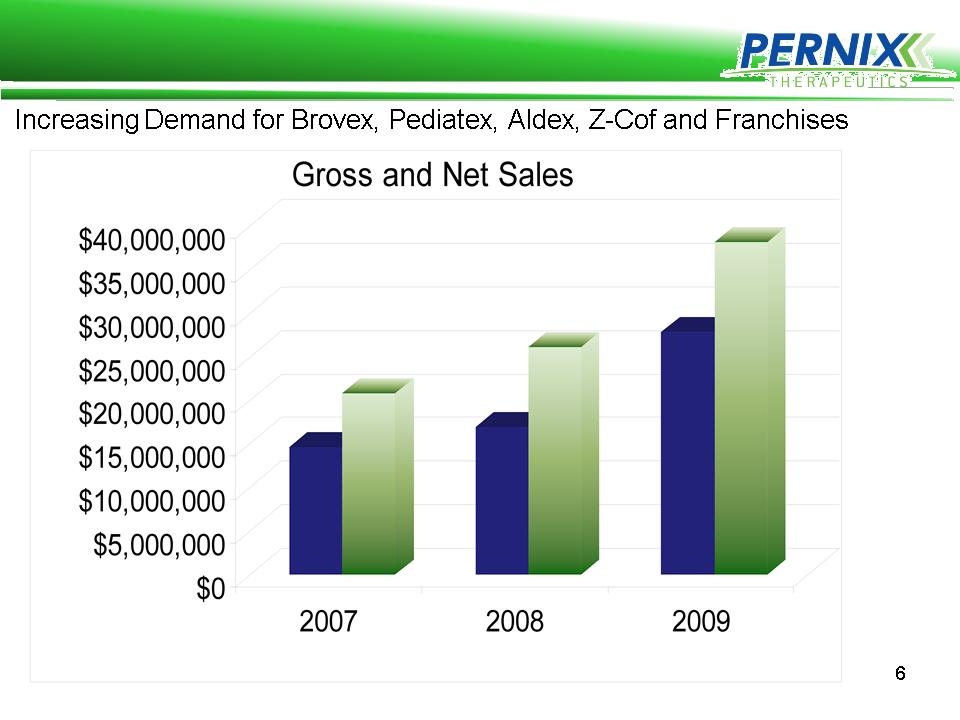
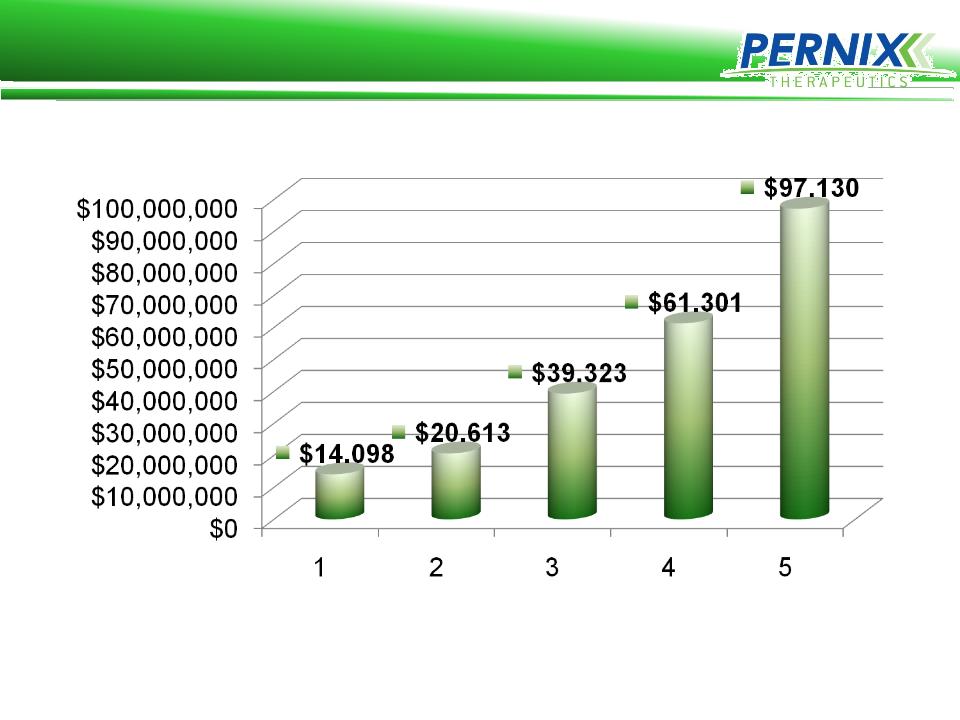
Pernix Revenue Growth
7
Pernix Revenue is Profitable

8
Solid Organic Growth supplemented by
Acquisitions/In-Licensings/Co-Promotions
Acquisitions/In-Licensings/Co-Promotions
• With emphasis on promotion sensitive products for
the pediatric market to leverage cost of sales force.
All acquisitions are targeted to be accretive to
earnings
the pediatric market to leverage cost of sales force.
All acquisitions are targeted to be accretive to
earnings
The Pernix Model

9
• Cold/Cough Sector-Annual Sales in
Excess of $2 Billion with 75MM
Prescriptions Annually
Excess of $2 Billion with 75MM
Prescriptions Annually
• Currently Very Limited Brand
Competition in Prescription Sector
of Anti-Infective & Cold/Cough
Markets
Competition in Prescription Sector
of Anti-Infective & Cold/Cough
Markets
• Competitors include: Cornerstone,
Hawthorn, Lupin, Meda, & GSK.
Hawthorn, Lupin, Meda, & GSK.
• Prescription Medical Foods are a
high growth market
high growth market
|
Products
|
Active Categories
|
|
Aldex Family
|
Cold/Cough
|
|
Brovex Family
|
Cold/Cough
|
|
Cedax
|
Antibiotic
|
|
Hylatopic
|
Dermatology
|
|
Pediatex Family
|
Cold/Cough
|
|
QuinZyme*
|
Cardiovascular
|
|
ReZyst IM
|
Gastro-Probiotic
|
|
TussiNAC*
|
Cold/Cough
|
|
Z-Cof Family
|
Cold/Cough
|
* Partnering Opportunities
Product Portfolio
10
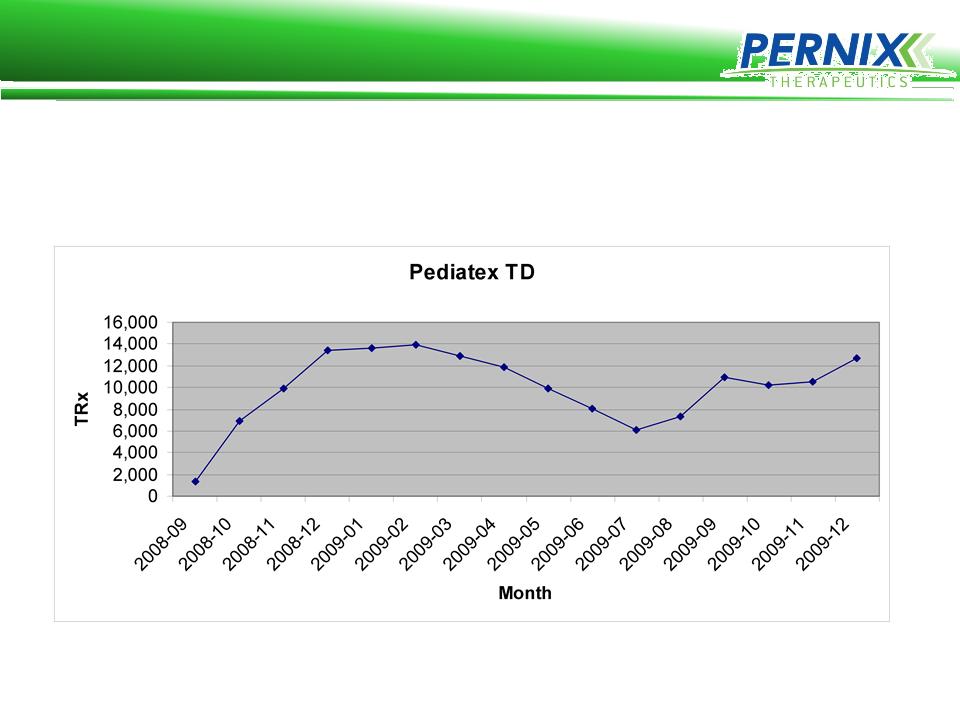
Pediatex TD Launch
Pernix’s sales force averaged almost 560 RX’s per rep for the
Pediatex TD product in the 4th month of the launch
Pediatex TD product in the 4th month of the launch
11
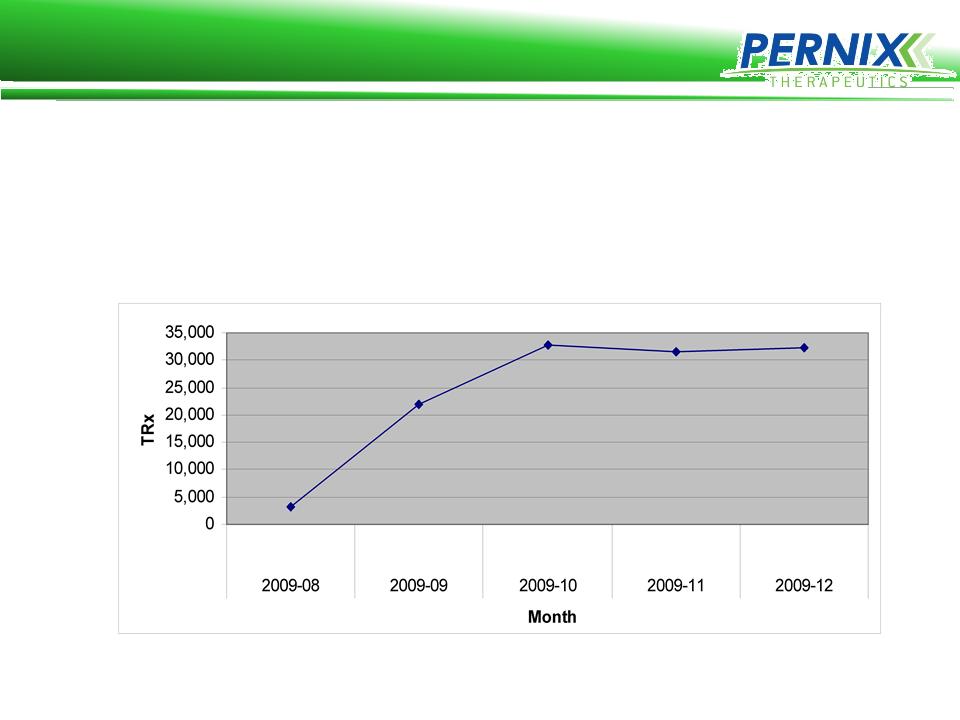
Brovex Acquisition
• Pernix acquired the Brovex product line for $450,000 in June 2009
and Launched in August 2009
and Launched in August 2009
• Pernix’s sales force achieved approximately $7,000,000 in sales
and averaged over 1600 Rx’s per Rep for this time period
and averaged over 1600 Rx’s per Rep for this time period

12
CEDAX© (ceftibuten)
• Third-generation cephalosporin indicated for Upper and Lower
Respiratory tract infections
Respiratory tract infections
• Launched by Schering-Plough with first year sales of approximately
$75,000,000
$75,000,000
• Acquired U.S Rights from Shionogi in 2010
• Patent Protection(Orange Book)
• Suspension expires in 2014
• Capsule - expires in 2013
• 2009 net sales: ~$6.0 Million
Cedax Acquisition

13
Cedax Opportunity
CEDAX© (ceftibuten)
• 2011 Cedax will likely be the only 3rd generation
patented cephalosporin promoted in pediatrics
patented cephalosporin promoted in pediatrics
• Antibiotics are promotion sensitive and highly
prescribed in pediatrics
prescribed in pediatrics
• Sales force expansion will be aligned to Cedax
Targets
Targets
• Life cycle management
•New strength of the Suspension (TBD)
14
Relaunched Branded Cephalosporin
Five Year Growth

15
• Exclusive Patented Natural Cough
Suppressant
Suppressant
• As efficacious as Codeine without the side
effects
effects
• US cough suppressant market is
approximately $2 Billion
approximately $2 Billion
• Plan to partner the development, which will
reduce our risk and is consistent with the
Pernix business model
reduce our risk and is consistent with the
Pernix business model
Product Development
16
2009 Total Employees: 49
2009 Sales Representatives: 35
2010 Q2 Recruiting Class:10-12
2010 Q3 Recruiting Class: 10-12
Current/Future Sales Force

17
17
Ø Execution-Oriented Business Model
Ø Highly Productive, Competitive Sales Force
Ø Minimal Product Development Risk
Ø Outsourced Contract Manufacturing & Logistics
Ø Industry Time-To-Market Launch Leader
Ø Lean Corporate Expense Infrastructure
Ø We are not “Waiting to Hear”
Investment Highlights

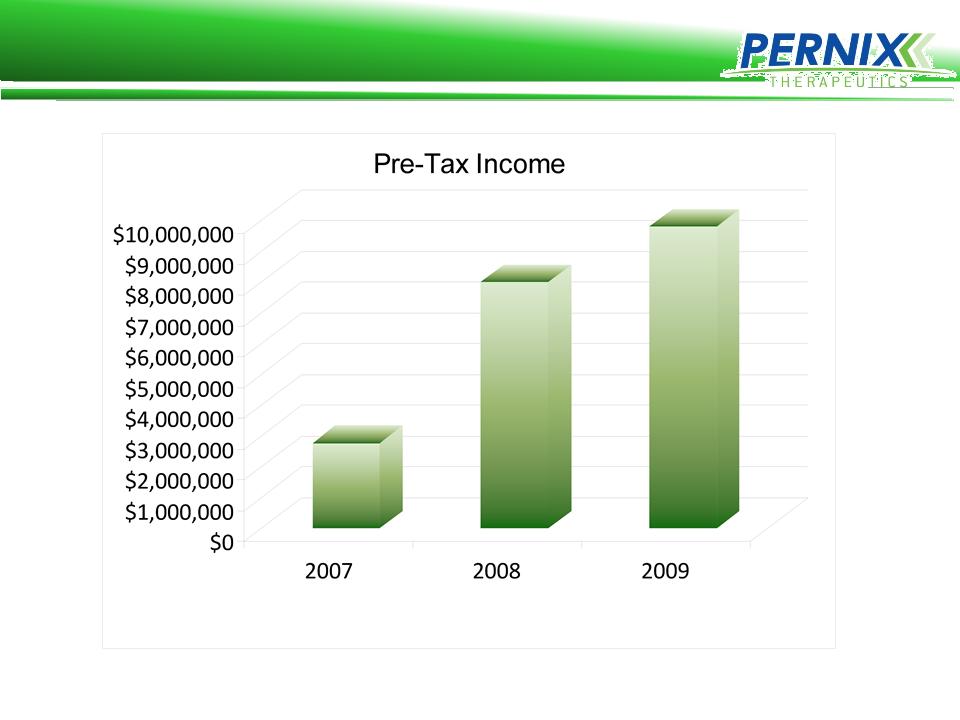
Cash Generation Focus:
2009 Pre-Tax Income: $9.2 million
Low Cap Ex, High Gross Margin
Model
Model
Debt-Free Balance Sheet
Self-Financing and a Strong Cash
Position
Position
Selected Data:
Symbol: PTX
Exchange: NYSE Amex
Recent Price: $4.00
52 Week Range: $5.74- $1.82
Market Cap: $99,754,328
Shares Outstanding: 24,938,582
Employees:45
Founded:1999
CAGR 32.3% - Total Sales (6 years)
CAGR 40.2% - Pre-Tax Income (6 years)
18
Corporate Snapshot

Cooper Collins, CEO
Pernix Therapeutics, Inc.
33219 Forest West Drive
Magnolia, TX 77354
Tel: (800) 793-2145
Email: tclifford@pernixtx.com
Internet: www.pernixtx.com
NYSE Amex: PTX
19
Corporate Information
