Attached files
| file | filename |
|---|---|
| EX-21.1 - LIST OF SUBSIDIARIES - INTEGRAMED AMERICA INC | exhibit21.htm |
| EX-23.1 - CONSENT OF AMPER, POLITZINER & MATTIA, P.C. - INTEGRAMED AMERICA INC | exhibit23.htm |
| EX-32.2 - CFO CERTIFICATION - INTEGRAMED AMERICA INC | exhibit32_2.htm |
| EX-32.1 - CEO CERTIFICATION - INTEGRAMED AMERICA INC | exhibit32_1.htm |
| EX-31.2 - CFO CERTIFICATION - INTEGRAMED AMERICA INC | exhibit31_2.htm |
| EX-31.1 - CEO CERTIFICATION - INTEGRAMED AMERICA INC | exhibit31_1.htm |
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington,
D.C. 20549
_________________
FORM
10-K
[X]
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d)
OF
THE SECURITIES EXCHANGE ACT OF 1934
For
the fiscal year ended December 31, 2009
OR
[ ]
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(D) OF THE
SECURITIES
EXCHANGE ACT OF 1934
For the
transition period
from _____ to _____
Commission
File No. 0-20260
INTEGRAMED AMERICA,
INC.
(Exact
name of registrant as specified in its charter)
|
Delaware
|
06-1150326
|
|
(State
or other jurisdiction of incorporation or organization)
|
(I.R.S. Employer Identification
No.)
|
|
Two
Manhattanville Road, Purchase, New York
|
10577
|
|
(Address
of principal executive offices)
|
(Zip
Code)
|
(914)
253-8000
(Registrant’s
telephone number, including area code)
Securities
registered pursuant to Section 12(b) of the Act:
|
Title of each class
|
Name of exchange on which
registered
|
|
Common
Stock, $.01 par value
|
NASDAQ
Global Market
|
Securities
registered pursuant to Section 12(g) of the Act:
None
Indicate by check mark if the
registrant is a well-known seasoned issuer, as defined in Rule 405 of the
Securities Act.
|
Yes
|
No X
|
Indicate by check mark if the registrant is not required to file reports
pursuant to Section 13 or Section 15 (d) of the Act.
|
Yes
|
No X
|
Indicate by check mark whether the registrant (1) has filed all reports required
to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during
the preceding 12 months (or for such shorter period that the registrant was
required to file such reports), and (2) has been subject to such filing
requirements for the past 90 days. Yes [X]No []
Indicate by check mark whether
registrant has submitted electronically and posted on its corporate website, if
any, every Interactive Data File required to be submitted and posted pursuant to
Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter
period that the registrant was required to submit and post such
files). Yes []No [ ]
Indicate
by check mark if disclosure of delinquent filers pursuant to Item 405 of
Regulation S-K (17 CFR 229.405) is not contained herein, and will not be
contained, to the best of registrant’s knowledge, in definitive proxy or
information statements incorporated by reference in Part III of this Form 10-K
or any amendment to this Form 10-K [ ]
Indicate
by check mark whether the registrant is a large accelerated filer, an
accelerated filer, a non-accelerated filer or a smaller reporting
company. See the definitions of “large accelerated filer”
“accelerated filer” and “smaller reporting company” in Rule 12b-2 of the
Exchange Act. (Check one):
|
Large
Accelerated Filer:
|
Accelerated
Filer X
|
|
Non-Accelerated
Filer: (Do not check if a smaller reporting
company)
|
Small
Reporting Company
|
Indicate by check mark whether the
registrant is a shell company (as defined in Rule 12b-2 of the Exchange
Act).
|
Yes
|
No X
|
Aggregate market value of voting stock (Common Stock, $.01 par value) held by
non-affiliates of the Registrant was approximately $64.0 million on June 30,
2009 based on the closing sales price of the Common Stock on such
date.
The
aggregate number of shares of the Registrant’s Common Stock, $.01 par value,
outstanding was approximately 11,703,400 on March 4, 2010.
DOCUMENTS
INCORPORATED BY REFERENCE
See Part
III hereof with respect to incorporation by reference from the Registrant’s
definitive proxy statement for the Registrant’s 2010 Annual Meeting of
Stockholders to be filed pursuant to Regulation 14A under the Securities
Exchange Act of 1934.
PART
I
ITEM
1. Business
Overview
–
We manage
highly specialized outpatient centers in emerging, technology-based, niche
medical markets. Currently, we are a leading manager of fertility centers and
vein clinics in the United States. We provide services and products through our
three operating divisions (Fertility Centers, Consumer Services and Vein
Clinics) and shared support services for providers through our corporate
offices. We provide our fertility centers and vein clinics with administrative
services such as finance, accounting, human resources, risk management, legal
and purchasing support; marketing and sales support; internet marketing and
website support; access to integrated information systems; in some instances,
non-physician practitioners; and access to capital for financing clinic
operations and expansion.
Fertility
Centers Division
Our
Fertility Centers Division provides business and management services to a
network of 14 contracted fertility centers in our Partner Program, serving
16 metropolitan markets across the United States. We believe these 14 Partner
centers are the largest managed network of fertility centers in the United
States, with 63 locations and 97 physicians and PhD scientists,
accounting for approximately 15% of the total in vitro fertilization
(“IVF”) procedures performed in the United States in 2008, which is the latest
period for which third-party data are available. The division supports fertility
centers’ operations and growth by providing access to information systems such
as our proprietary ARTworks electronic medical records software as well as
medical equipment and facilities, non-physician personnel and marketing and
financial support services. All fertility Partners have full access to our
Attain IVF programs, which are described below. We do not employ or control the
physicians who provide or direct the treatment of patients.
Our
fertility centers offer a range of diagnostic and fertility treatment options to
patients. The fertility centers’ physicians perform diagnostic tests on both
women and men to determine the cause of infertility and each fertility center
has an endocrine and andrology laboratory on site in order to perform and
expedite infertility analyses. Once the cause of infertility is identified,
several treatment options are offered to patients, including IVF treatment,
frozen embryo transfer, intrauterine insemination and minimally invasive surgery
to correct anatomical reproductive problems. All of our fertility centers have
on-site IVF laboratories in order to maintain the integrity of the IVF
processes. Fertility centers are typically staffed by six to seven physicians, a
scientist, embryologists, nurses, support staff and ultrasound
technicians.
Insurance
and managed care payors, depending on the plan under which a patient is covered,
reimburse certain services that our Partners provide, such as diagnostic
testing, surgeries and, in certain circumstances, fertility treatments. However,
the charges for assisted reproduction technology (“ART”) services our Partners
primarily provide are often paid directly by patients, including through
programs such as our Attain IVF programs. Several states mandate offering
certain benefits of varying degrees for ART services. For example, in
Massachusetts, Rhode Island, Maryland and Illinois, the mandate requires
coverage for many, but not necessarily all, ART services provided by our
Partners. Approximately 50% of our Partner centers’ payments were derived from
third-party payors for the twelve months ended December 31, 2009, all of which
was provided by private payors. Contractual arrangements with
2
third-party
payors typically are for a term of one year, may be terminated by either party
upon 90 days’ notice any time after the initial one-year term and contain
automatic annual renewal provisions. Contractual arrangements with third-party
payors also typically include payment terms and schedules of rates, although
those payment terms and schedules of rates are subject to renegotiation after
the initial term of the contract. During the twelve months ended December 31,
2009, in accordance with the terms of our contractual arrangements with them,
third-party payors paid approximately 51% of the charges billed to them by our
Partner Centers. We are aware of efforts to expand mandated coverage to
additional states, but it is unclear if these efforts will be
successful.
If in the
future mandates are enacted by additional states, we expect the impact on our
Partner fertility centers to be neutral to positive, as such mandates would
likely increase the market for fertility center services, but at payment rates
that are lower than the amounts typically paid directly by
patients.
When
establishing a Partner relationship, we typically acquire the assets of a
fertility center, enter into a long-term comprehensive business service
agreement with the center and assume most administrative and financial functions
of the center. The acquisition of a Partner agreement generally obligates us to
pay a fixed sum for the exclusive right to service the fertility center. These
agreements are typically for terms of 10 to 25 years and contain automatic
renewal provisions. Some of these agreements also contain provisions that allow
the Partner fertility center to terminate the agreement, upon 12 months’
prior notice, at any time after five years from the agreement’s effective date.
Partners typically have obligations upon termination in certain circumstances,
such as purchasing the assets used in operating the fertility center and making
payments based on recent revenues. Partners also agree to promote their
practices by, among other things, participating in marketing programs we develop
for them. Typically, the fertility center contracting with us is a professional
corporation in which the key physicians are the shareholders. Generally, no
shareholder of a Partner fertility center may assign his or her interest in the
Partner fertility center without our written consent.
We
require each professional corporation operating in our Partner fertility centers
to enter into employment agreements with all key physicians at that center.
These employment agreements typically have five-year terms and contain
provisions prohibiting the key physicians from practicing reproductive
endocrinology, infertility medicine or assisted reproductive technology in
competition with us, within a specified area, for the term of the agreement and
for 12 to 24 months thereafter. Although it is unclear whether these
non-competition provisions would be enforceable if challenged, we have not
experienced significant competition from physicians who formerly practiced at
our Partner centers. We also usually enter into a personal responsibility
agreement directly with each physician shareholder of the practice. The personal
responsibility agreement obligates a physician shareholder to repay us a
proportionate amount of the exclusive right to service fee payment received by
that physician shareholder if he or she leaves the practice sooner than five
years after the payment.
Under 13
of our current Partner agreements, as compensation for our services we receive a
three-part fee comprised of: a percentage of net revenues, generally between 3%
and 6%; reimbursed costs of services (costs incurred in providing services to a
fertility center and any costs paid on behalf of the fertility center); and
either a fixed amount or a percentage of the center’s earnings, which currently
ranges from 10% to 20%, but may be subject to limits. Our remaining Partner
agreement is a cost-plus arrangement in which we receive a fee for our
Reimbursed Cost of Services, plus an additional fee equal to 9.5% of those
costs.
Our
Fertility Centers Division also supports a Council of Physicians and Scientists
(the “Council”) for leading fertility providers, which we established
14 years ago. The Council is comprised mostly of representatives from our
fertility network and brings together leaders in reproductive medicine and
embryology with the goal of promoting a high quality clinical environment
throughout our fertility center network. The Council meets regularly and
conducts bi-monthly teleconferences on topics related to improving infertility
diagnosis, treatment and success rates. Additionally, the Council helps to
establish the principles of our culture of safety. We believe our centers follow
the Practice and Ethics Guidelines for clinical practice set forth by the
American Society for Reproductive Medicine. We have also achieved accreditation
from the American Association for Ambulatory Healthcare and the College of
American Pathologists, which demonstrates our commitment to compliance with
nationally recognized standards for laboratory services, patient safety and
quality patient care.
We
assisted in the organization of, and obtained a minority equity interest in, an
offshore captive insurance company called Assisted Reproductive Technology
Insurance Company (“ARTIC”), which is designed to moderate the cost of
malpractice insurance to members of our fertility network. Most of the equity of
ARTIC is owned by various physician practices that are members of our fertility
network, and we have no future obligations to provide additional funding to
ARTIC. On January 1, 2005, ARTIC began providing malpractice insurance
coverage to the majority of the physician practices within our Partner fertility
network.
3
Consumer
Services Division
Our
Consumer Services Division offers a family of programs, including our Attain IVF
Refund Program and our recently introduced Attain IVF Multi-Cycle Program,
collectively referred to as our Attain IVF programs, which are designed to help
patients attain their goal of starting a family. We offer our Attain IVF
programs directly to fertility patients, including patients of our Partner
centers and patients of the division’s contracted network of independent medical
providers under its Affiliate Program.
Our
Affiliate Program allows fertility centers to pay fees to receive selected
management services we provide to our Partners, such as internet marketing and
access to the Council. We also provide our Affiliates with access to our Attain
IVF programs. Historically, we provided services to our Affiliates on an
exclusive basis in the area in which the Affiliate operates, but Affiliates’
access to our Attain IVF programs is generally subject to achievement of certain
benchmarks, including with respect to Attain IVF Refund Program enrollments;
however, in July 2009 we began allowing access to our Attain IVF programs on a
non-exclusive basis in new markets. As of December 31, 2009, we had contracted
with 25 Affiliate fertility centers. During 2007, our Affiliate fertility
centers collectively provided 8% of the total IVF procedures in the United
States. Our Consumer Services Division does not provide, nor is it responsible
for providing, medical services or treatments to patients.
Our
Consumer Services Division re-launched its Shared Risk Refund Program under the
name “Attain IVF” in late 2008. This re-branding was done to reflect advantages
offered by the program beyond its packaged pricing features and to position the
program in a leadership role among smaller, similar programs offered by other
providers.
Beginning
in July 2009, we began referring to this program as our Attain IVF Refund
Program to differentiate it from our Attain IVF Multi-Cycle Program. As
described in more detail below, our Attain IVF Refund Program is an offer of
packaged pricing for a set of fertility treatments with a refund, equal to 70%
of the contract amount for patients using their own eggs, if treatment does not
result in a baby. Under circumstances where a patient uses donor eggs, 100% of
the contract amount is refunded if treatment does not result in a baby. For the
twelve months ended December 31, 2009, approximately 18% of the patients in our
Attain IVF Refund Program used donor eggs.
Patients
enrolling in our Attain IVF programs can select from various treatment and
financing options which are designed to appeal to patients at different stages
of their reproductive lives and with different financial needs and resources.
The average cost of one fresh IVF cycle as of December 2009 was approximately
$12,000 according to Marketdata Enterprises, Inc. According to our estimates,
the average cost of a frozen embryo transfer is approximately $3,000. The Attain
IVF Refund Program allows medically cleared patients to pay an up-front deposit
of approximately twice the average cost of a fresh IVF cycle in return for up to
six treatment cycles (consisting of three fresh IVF cycles and three frozen
embryo transfers) with a refund if treatment does not result in a baby. The
refund is equal to 70% of the contract amount for patients using their own eggs
and 100% of the contract amount if the patient uses donor eggs. The Attain IVF
Multi-Cycle Program allows all patients, including those who are not medically
cleared for our Attain IVF Refund Program, to pay a single fee, which is
slightly less than the average cost of two fresh IVF cycles, in return for up to
four treatment cycles (consisting of two fresh IVF cycles and two frozen embryo
transfers). The fertility treatment cycles are provided to patients by fertility
centers with which we contract for participation in the program. The benefits of
our Attain IVF programs to our fertility centers include: allowing for patients
to commit to multiple fertility treatments which improves treatment volume and
revenues; insulating the centers from refund risk; managing cash and
administrative details associated with our Attain IVF programs; and enabling
physicians to maintain a traditional fee for service arrangement without the
appearance of conflicts of interest that otherwise might arise from self
administering a refund program. The benefits of our Attain IVF programs to
patients include: improved success rates associated with multiple fertility
treatment cycles; increased financial certainty relating to the cost of the
fertility treatment process; and, in the case of our Attain IVF Refund Program,
a significant financial refund should the treatments be
unsuccessful.
4
Our
Attain IVF programs serve as patient recruitment and case management vehicles
where the patient contracts with us to provide the program services described
below. We bind our Partners and Affiliates to abide by the terms of the program
through participation agreements that support our packaged pricing. These
programs are designed to make the fertility treatment process easier for
patients by providing a continuum of services over an extended period, if
necessary. Our Attain IVF programs achieve this objective by offering the
following services:
|
•
|
Patient
recruitment via internet web portals and search engines, in-clinic
educational materials, in-clinic contact with fertility specialists and
on-line contact with patient service specialists;
|
|
|
•
|
Educating
patients as to the benefits of various treatment options offered by our
network of contracted medical providers which have been tailored to appeal
to patients at various stages of their reproductive lives and with various
medical conditions;
|
|
|
•
|
Explaining
the financial costs and patient responsibilities of the various treatment
options;
|
|
|
•
|
Educating
patients as to the various financing options offered by our Attain IVF
programs and referring them to sources of third-party financing when
requested;
|
|
|
•
|
Coordinating
an initial medical assessment required for entry into our Attain IVF
programs;
|
|
|
•
|
Arranging
treatment with an Affiliate or a Partner center for all treatment cycles
used by the patient; and
|
|
|
•
|
Providing
on-going case management, treatment plan monitoring and evaluation
services.
|
We
receive payment directly from patients who participate in our Attain IVF
programs. By contract, 30% of the Attain IVF Refund Program contract amount is
non-refundable (for the non-donor egg option) and is recognized ratably (on a
fair value basis) as revenues over the course of the patient’s treatment cycles.
If the patient achieves pregnancy prior to the completion of the last available
treatment cycle, then the remaining unamortized portion of the non-refundable
fee is immediately recognized as income. The remaining 70% of revenues are
recorded upon the patient becoming pregnant and achieving a fetal heartbeat. For
the donor egg option, for which 100% of the contract amount is refundable, all
revenues are recorded upon the patient becoming pregnant and achieving a fetal
heartbeat. We are able to record income at the time of pregnancy for our Attain
IVF Refund Program, as we have substantially completed our fertility obligation
to the patient and we can accurately estimate the amount of expenses or refunds
that will become due if there is a pregnancy loss. We are able to make these
estimates for pregnancy loss based upon reliable Company specific data with
respect to the large homogeneous population we have served for more than seven
years. Expenses prior to pregnancy related to the program are recorded as
incurred. All of the amounts shown on the balance sheets in our consolidated
financial statements included elsewhere in this report as Attain IVF Refund
Program deferred revenues and other patient deposits consist of unrecognized
program enrollment/service fees and potentially refundable contract amounts for
enrolled patients who have not had a successful pregnancy outcome and deposits
received from patients who have not yet commenced treatment under the
program.
Due to
the characteristics of our Attain IVF programs, we pay for a patient’s treatment
costs in excess of their contract amount should the initial treatment cycles be
unsuccessful. In order to moderate and manage the likelihood that we will need
to pay for these treatment costs, we have developed a sophisticated statistical
model and case management program in which Attain IVF Refund Program patients
are pre-approved prior to enrollment in the program. We also continuously review
patients’ clinical criteria as they undergo treatment. If, while undergoing
treatment, a patient’s clinical response falls outside our criteria for
participation in Attain IVF programs, we have the right to remove that
individual from the program, with an applicable refund to the patient. To date,
our case management process has been effective in managing the risks associated
with our Attain IVF Refund Program within expected limits. A patient has the
right to withdraw from our Attain IVF Refund Program at any time and will be
issued an applicable refund.
5
Vein
Clinics Division
Our Vein
Clinics Division was formed on August 8, 2007, with the purchase of Vein
Clinics of America, Inc. (“VCA”), a company that had been in business since
1981. Our Vein Clinics Division provides business and management services to a
network of 34 vein clinics located in 13 states. We believe our vein
clinics network is the largest single network of vein care providers in the
United States. These clinics provide specialized treatment for patients
suffering from vein diseases and other vein disorders, such as varicose veins,
spider veins and venous ulcers.
We offer
vein clinics services and support, including training for physicians, clinical
and financial information systems, revenue cycle management, yield management,
sales and marketing services, group purchasing, non-physician personnel,
facilities, site selection and development and other operational functions to
support the clinic. The division supports vein clinics’ operations and growth by
providing access to information systems such as our proprietary Virtual
Physician Assistant (“VPA”) information system, which is an end-to-end patient
and clinic operating system that provides decision support and revenue cycle
functions. A typical vein clinic averages 2,400 square feet and is located
in an affluent, growing community. Each clinic has a standardized operational
structure composed of a phlebologist (a physician specialized in the diagnosis
and treatment of disorders of venous origin), nurse, ultrasound technologist,
office manager and assistant. Medical services or treatments are provided to
vein clinic patients by physicians who are employed by professional
corporations, whose financial condition, results of operations and cash flows
are consolidated with our consolidated financial statements.
Our Vein
Clinics Division’s philosophy of patient care is based on complete disease
management, from initial screening to treatment to follow up. Our vein clinics
view each step in this process as critical to the patient’s successful outcome.
Our clinics currently use Endovenous Laser Treatment (“ELT”) as well as
sclerotherapy to treat varicose and spider veins. Our vein clinics use extensive
and sophisticated ultrasound mapping prior to treatment, which we believe
results in a more effective treatment plan. Rigorous post-treatment follow up is
meant to identify any residual or emerging issues so that they can be quickly
managed before the disease worsens.
Our Vein
Clinics Division depends upon third-party payors, including governmental and
private insurance programs, to pay for most treatments provided to patients. For
the twelve months ended December 31, 2009, approximately 60% of payments
received by our Vein Clinics Division were from managed care programs, 20% were
from commercial insurers, 15% were from Medicare and 5% were directly from
patients.
The
private third-party payors providing reimbursement to our vein clinics include
standard indemnity insurance programs as well as managed care programs, such as
preferred provider organizations and health maintenance organizations. These
third-party payors provide reimbursement to our vein clinics at negotiated
rates, which approximate 50% of the billed charges, for medically necessary
treatments. Most ELT treatments for varicose veins and venous leg ulcers
provided at our vein clinics are reimbursed by third-party payors. However,
third-party payors generally do not cover sclerotherapy or treatments they
determine are not medically necessary, such as the cosmetic treatment of spider
veins. In some cases, third-party payors require prior authorization of varicose
vein treatment to provide reimbursement. Contractual arrangements with
third-party payors typically are for a term of one year, may be terminated by
either party upon 60 to 90 days’ notice after the initial term and contain
automatic annual renewal provisions. Contractual arrangements with third-party
payors also typically include payment terms and schedules of rates that are
subject to change by the third-party payor upon as little as 30 days’
notice. Payments from Medicare are paid in accordance with a set fee schedule
and are subject to change or review by governmental authorities.
Once our
Vein Clinics Division has facilitated a vein clinic’s establishment, we enter
into a contract with the professional corporation operating in our clinic.
Unlike our Partner fertility centers, the physicians who are employed at our
vein clinics typically do not have an ownership interest in the medical
practice. A “friendly physician” model is often used for ownership, pursuant to
which we are the primary beneficiary and obligor of the vein clinic’s
operations; however, we also own and operate vein clinics through subsidiaries
in two states where we are not prohibited from doing so under applicable
corporate practice of medicine laws. Under the terms of our contracts with the
vein clinics, we have sole and exclusive responsibility to manage the
non-medical operations of the practice and the physicians have sole
responsibility for the medical and clinical aspects of the practice. Our
contracts with the vein clinics provide that we are responsible for the leasing
of space, obtaining all equipment and services needed, providing all billing and
collections functions, arranging for and supervising all non-physician personnel
and providing services
6
so they
can market their own practices. In exchange for our services, our contracts with
the vein clinics provide that the vein clinics pay us a fee equal to 150% of our
expenses of operating and managing the vein clinics. These fees have
historically exceeded the operating margin generated by any particular vein
clinic prior to payment of the management fee. Accordingly, each vein clinic
only pays the portion of the management fee that is equal to the amount of
revenue generated by the clinic annually up to the 150% amount. As a result, our
vein clinics (the professional corporation) do not generate any net profits at
year end. Our contracts with the vein clinics are typically for 25 years
with renewal rights. In the event of early termination, any accrued obligations
remain outstanding until satisfied. We also have the right at any time to cause
the “friendly physician” to transfer his or her ownership in a vein clinic to
another physician designated by us.
We
require each professional corporation operating in our vein clinics to enter
into an employment agreement with the physician practicing at that clinic. The
employment agreement typically has a term of one year and automatically renews
for additional one-year periods unless terminated by either party. The physician
generally is required to pay the clinic either $75,000 or $50,000 if the
agreement is terminated prior to three years from the physician’s first
employment with the clinic, with the amount due depending on the time of
termination. This requirement helps defray the training expenses we incur when
we assist the physician in establishing a practice. The physician also usually
covenants not to compete with the clinic or provide medical services in the
treatment of varicose veins or other venous diseases, within a specified area,
during the agreement’s term and for two years thereafter. Although it is unclear
whether these non-competition provisions would be enforceable if challenged, we
have not experienced significant competition from physicians who formerly
practiced at our vein clinics.
Since our
acquisition of VCA in August 2007, we have made significant investments in this
division’s infrastructure, which have been designed to allow us to open new
clinics at a more rapid and sustained pace utilizing a replicable model. These
investments include:
|
•
|
Physician
recruiting and training. The business model for our Vein Clinics
Division depends on being able to identify, recruit and train new
physicians to staff new clinics. We have invested in additional
professional personnel as well as other recruiting and training assets to
support scaled growth in the future.
|
|
|
•
|
Regional
management. We have established a regional management
infrastructure to manage the day-to-day operations of the expanding Vein
Clinics Division clinical network and anticipate continued investment in
regional management talent as our clinic base expands.
|
|
|
•
|
Revenue
cycle management. Over the past several years, the market for vein
care has undergone a shift from private out of pocket payment by patients
to an environment where most treatment is covered by insurance. This shift
has caused us to make heavy investments in physician credentialing,
working capital and improved billing and collections personnel, systems
and procedures. These investments will continue as the business
grows.
|
|
|
•
|
Quality
improvement. We established a more formal quality and
risk management oversight infrastructure with performance improvement
initiatives related to clinical processes. This includes
developing a formalized training and competency assessment process,
pursuing ICAVL accreditation for our diagnostic ultrasound examinations,
developing documentation review processes and standardized multi-lingual
patient education materials and consent forms.
|
|
|
•
|
New clinic
development. With our planned roll-out of new clinic openings, we
are making investments in personnel and procedures for identifying
opportunities and opening new clinics in existing and new
markets.
|
|
|
•
|
Marketing
and sales. We have established more formal, direct-to-consumer and
physician referral marketing
programs.
|
Shared
Services Group
Through
our Shared Services group, we provide the following support to our Fertility
Centers, Consumer Services and Vein Clinics Divisions:
7
|
•
|
Administrative
Services. Our Shared Services group provides our
contracted fertility centers and vein clinics with administrative
services, including: accounting and financial services, such as accounts
payable, payroll and financial reporting; human resources administration;
legal services; risk management; insurance; information systems and
services; and strategic planning.
|
|
|
•
|
Access to
Capital. We believe we provide our Partner fertility
centers and vein clinics with a competitive advantage through access to
capital for funding accounts receivable, expansion and growth. We provide
our Partner fertility centers and vein clinics with efficient access to
capital which allows them to obtain current technologies, equipment and
facilities that enable them to provide a full spectrum of services to
effectively compete for patients. For example, we have built new clinical
facilities housing state-of-the-art fertility laboratories for several
Partners, which enable them to expand their offerings to include a number
of services that they had previously outsourced, and have acquired
state-of-the-art ultrasound and laser technology for our vein clinics. We
believe this access to capital helps us to recruit Partner
practices.
|
|
|
•
|
Integrated
Information Systems. Using our established base of
treatment providers, we are continuously developing integrated information
systems to collect and analyze clinical, patient, financial and marketing
data, which we believe allow us to more effectively control expenses and
improve cash collections at our Partner fertility centers and vein
clinics. Our proprietary ARTworks clinical software provides
electronic medical records, treatment plan and success rate research
capabilities, decision support functionality and clinical risk management
services, which we believe makes our physicians more efficient and
improves quality of care. We provide our vein clinics access to
our proprietary VPA information system, which is an end-to-end patient and
clinic operating system that provides decision support and revenue cycle
functions.
|
|
|
•
|
Human
Resources. Our Shared Services group provides our
contracted fertility centers and vein clinics with human resources
services, including: policies and procedures; arranging for comprehensive
benefits and managing the implementation of those benefits; wage and hour
administration; performance reviews; job descriptions; and overall human
capital management.
|
|
Our
Industries
Reproductive
Medicine
Reproductive
medicine encompasses the medical discipline that focuses on male and female
reproductive systems and processes. According to a recent industry estimate,
approximately 10% of U.S. couples have trouble conceiving. There are many
reasons why couples have difficulty conceiving, and accurate identification of a
specific cause of infertility can be time consuming, expensive and requires
access to specialized diagnostic and treatment services. Reproductive
endocrinologists are specialized physicians who perform these more sophisticated
medical and surgical fertility diagnoses and treatments. Reproductive
endocrinologists generally have completed a minimum of four years of residency
training in obstetrics and gynecology and have at least two years of additional
training in an approved subspecialty fellowship program. There are approximately
1,400 practicing reproductive endocrinologists offering fertility services
across 480 fertility centers in the United States. According to Marketdata
Enterprises, Inc., expenditures relating to fertility services in the U.S.
market were estimated at approximately $4 billion for 2008. The fertility
services market is highly fragmented among providers in each major local market
as well as on a national basis.
Fertility
services include diagnostic tests performed on both the female and male.
Depending on the results of the diagnostic tests performed, treatment options
may include, among others, fertility drug therapy, artificial insemination and
fertility surgeries to correct anatomical problems. Procedures that require
gametes (sperm and eggs) to be handled in vitro (outside the body) are
classified as ART services. Current types of ART services include IVF, frozen
embryo transfers, donor egg programs as well as other more specialized
treatments. IVF treatments are the most frequently employed form of ART, with
107,095 fresh IVF cycles performed in the United States in 2008 (Society of Assisted Reproductive
Technology). Current techniques used in connection with IVF services
include intracytoplasmic sperm injection, assisted hatching, cryopreservation of
embryos, pre-implantation genetic diagnosis and blastocyst culture and
transfer.
8
Although
demand for advanced reproductive medicine and treatment is highly correlated
with larger demographic trends, we believe the market will continue to grow in
the future for the following reasons:
|
•
|
The
quality of treatment is improving, increasing pregnancy success
rates;
|
||
|
•
|
Improvements
in embryo culture media and implantation rates are leading to the
capability of reducing high order multiple births, which is one of the
greatest risk factors in this industry;
|
||
|
•
|
With
improving pregnancy rates, the cost of treatment is decreasing thereby
making these services more affordable;
|
||
|
•
|
Demand
for reproductive medical services is increasing through greater public
awareness and acceptance of these treatments; and
|
||
|
•
|
Couples
are delaying child birth until later in life. In 2006, approximately one
out of every 12 first births was to a woman age 35 or older, compared
with one out of every 100 first births in 1970, according to the
U.S. Centers for Disease Control and Prevention.
|
||
While
fertility market growth has moderated recently, in line with a demographic
trough of couples of family-bearing age, we believe that we are well positioned
to increase our share of the fertility market due to the following
factors:
|
•
|
The
benefits arising from consolidation, including the economies of scale that
can be realized by leveraging a corporate infrastructure like ours to
minimize general and administrative expenses as a percentage of fertility
center revenues;
|
|
|
•
|
The
need for greater efficiencies to offset rising costs and decreases in
revenue growth;
|
|
|
•
|
The
barriers to establishing new fertility centers, including the
capital-intensive nature of acquiring and maintaining state-of-the-art
medical equipment, laboratory and clinical facilities and the need to
develop and maintain specialized information systems to meet the demands
of patients and third-party payors;
|
|
|
•
|
The
need for support services like those we provide to address the need for
seven-days-a-week service to respond to patient demands and to
optimize the outcomes of patient treatments;
|
|
|
•
|
The
increased need for marketing services like those we provide to address
increasing competition among medical providers specializing in fertility
treatment; and
|
|
|
•
|
Our
track record of growing contracted fertility center Partners two to three
times faster than fertility centers that are not a part of our network,
based on the number of fresh IVF cycles
performed.
|
Vein
Disease
Phlebology
is the medical specialty concerned with the treatment of vein diseases. Common
vein diseases and their symptoms can take many forms, including:
|
•
|
Varicose
veins — which are caused when small valves designed to allow blood to
flow in only one direction fail or leak. This causes blood to flow
backwards under the force of gravity and pool inside the
vein;
|
|
|
•
|
Spider
veins — which are very small varicose veins. They are thin,
threadlike veins that lie close to the skin’s surface and are commonly red
or purple in appearance. Spider veins can be hormonally induced and are
often associated with pregnancy and menstruation;
|
|
|
•
|
Venous
Leg Ulcers — which are non-healing open wounds that are caused by
venous pump failure. It usually occurs near the inside of the ankle, but
can be found anywhere below the knee. It can occur with or without visible
varicose veins;
|
9
|
•
|
Klippel-Trenaunay
Syndrome — which is a rare, congenital disorder in which patients
usually have one hypertrophied leg, a port wine stain and large varicose
veins on the lateral aspect of the leg; and
|
|
|
•
|
Restless
Leg Syndrome — which may occur when valves fail, causing blood to
reflux, or flow backwards, causing it to pool and stagnate in the veins,
leading to aching, throbbing, cramping and fatigue in the
legs.
|
Although
there are both surgical as well as minimally invasive treatment protocols for
vein disease, we specialize in minimally invasive care. Conventional vein care
treatment under both protocols usually begins with an ultrasound assisted
mapping to determine the extent of the disease, generally followed by a surgical
or minimally invasive treatment protocol. Historically, the most common surgical
treatment has been a procedure referred to as vein stripping, which is the
surgical removal of surface veins. Vein stripping is generally done as an
outpatient procedure and is performed while the patient is under general
anesthesia. Vein stripping may leave scarring and require an extended recovery
time. More recent minimally invasive treatments include ELT and sclerotherapy,
which are the treatments offered by our clinics. ELT is a laser treatment which
does not involve hospitalization, general surgery or the potential for
significant scarring that is associated with vein stripping. With ELT, after
local anesthesia is administered, a small optical fiber is inserted through a
needle into the varicose vein under ultrasound guidance. The laser is activated
and, as the optic fiber is removed from the vein, it heats and closes the vein.
Once the vein is closed, the blood that was circulating through the vein is
naturally rerouted to other healthy veins. Over time, the varicose vein is
absorbed by the body. Sclerotherapy involves injecting abnormal veins with a
solution called a sclerosant. This immediately shrinks the vein and causes it to
dissolve over a period of weeks, allowing the body to naturally redirect the
blood flow to healthy veins. A typical sclerotherapy treatment may last for 15
to 20 minutes and consists of multiple microinjections.
Various
demographic trends are contributing to the growth in demand for vein care.
Annual expenditures related to vein care in the United States are approximately
$2 billion and are projected to grow 12% per year through 2010, according
to our estimates. The U.S. Food and Drug Administration’s approval of
lasers for thermal ablation of veins and subsequent establishment of an American
Medical Association Current Procedural Terminology code for reimbursement by the
Centers for Medicare and Medicaid Services has opened this market to rapid
growth and development over the last several years. We also believe that the
market for vein care will continue to grow in the future as awareness of
minimally invasive treatment protocols grows among people with vein disease and
as additional third-party payors recognize the medical necessity of treating
vein disease. We believe that approximately 25 million people are currently
affected by vein disease in the United States, but only approximately one
million receive treatment for such vein disease.
We
believe numerous market conditions in this industry produce business
opportunities for us, including:
|
•
|
The
level of specialized skills required for comprehensive patient
treatment;
|
|
|
•
|
Favorable
sociological trends including a growing demographic wave from an aging
population;
|
|
|
•
|
The
need to develop and maintain specialized management information systems to
meet the increasing demands of patient billing and third-party
payors;
|
|
|
•
|
The
current fragmented nature of the market, which is comprised of numerous
smaller, independent providers, allowing the opportunity for market
consolidation;
|
10
|
•
|
New
laser and medical technologies that make access to treatment less painful
and disfiguring, coupled with insurance company reimbursement for these
new technologies;
|
|
|
•
|
The
large number of people affected by vein disease in the United States in
relation to the relatively low percentage of people who actually receive
treatment for such vein disease; and
|
|
|
•
|
Our
experience recruiting and training physicians in treating varicose veins
and the ability to produce opportunities we believe are financially
attractive to physicians practicing in other areas, such as general
practice or emergency medicine.
|
Our
Strategy
Make
Selective Contract Acquisitions of Partner Fertility Centers
The
U.S. market for fertility services is highly fragmented and we believe that
it is ripe for consolidation. Recruitment into our Partner Program has
traditionally focused on fertility centers that first participate as Affiliates
serviced by our Consumer Services Division; as such, we had an established
pipeline of 25 fertility centers as of December 31, 2009. Affiliate practices
have the opportunity to become familiar with the offerings we provide and our
commitment to customer service, which allows the Affiliate practices to see our
value proposition first hand. We, in turn, have a chance to assess a practice’s
commitment to growth and utilization of our services without making a
significant up-front financial commitment. In addition to recruiting from
Affiliate centers, we have a development staff that targets leading physician
groups with established practices in selected metropolitan markets. These
candidates are then evaluated against our contract acquisition criteria, which
includes factors such as size of practice, physician reputation and the
physicians’ growth-oriented outlook. We believe that our competitors’ ability to
compete with us for contract acquisitions is currently limited due to our
experience acquiring Partner center contracts, our position as the manager of
what we believe is the largest network of fertility centers in the United States
and our developed infrastructure and experience in delivering valuable services
to support fertility center operations.
Expand
our Network of Affiliate Fertility Centers
As of
December 31, 2009, we had Affiliates in 25 metropolitan markets and intend to
expand our network of Affiliate fertility centers to other metropolitan markets
across the United States. We primarily focus our network development activities
on metropolitan markets with populations in excess of 500,000. Because of the
relatively low percentage of the population that seeks fertility treatment, a
large population base is required to support a sophisticated fertility center.
Our fertility centers are capable of drawing consumers from a large geographic
area and, as such, our development staff is focused on the top 100 largest
metropolitan markets, where we expect the highest demand for fertility centers
to occur.
Develop
De Novo Vein Clinics
We intend
to develop new vein clinics in targeted markets. Our past experience suggests
that the vein clinics business can generally support one clinic per one million
population in metropolitan markets. Our new clinic development staff focuses on
the following:
|
•
|
Developing
new clinics in markets where we already have existing clinics that have
not fully penetrated their market to take advantage of existing
investments in regional management, managed care contracts, personnel and
marketing capabilities;
|
11
|
•
|
Identifying
attractive new markets in states that already have a vein clinic location
and states contiguous to existing vein clinic locations to leverage
regional management, personnel and other infrastructure
assets; and
|
|
|
•
|
Identifying
locations where we believe there are attractive demographics, reasonable
media costs and a favorable reimbursement
environment.
|
We
believe our vein clinic model can be predictably and profitably replicated in
new markets. Our ability to develop de novo vein clinics is demonstrated by the
five new vein clinics we opened during 2008 and the new vein clinics we opened
in Cincinnati and Cleveland, Ohio in 2009. We expect to accelerate and increase
the number of de novo vein clinics opened in 2010, however this pace could be
affected by challenges in physician recruitment. De novo vein clinics offer an
attractive return on capital as they require relatively little capital
investment, typically $300,000, and usually reach break-even in nine months or
less after opening of the clinic.
Increase
the Total Number of Patients Treated
We intend
to work with our fertility centers and vein clinics to increase the total number
of patients they treat. To achieve this objective we intend to:
|
•
|
Offer
products and services to centers and clinics that help them attract
patients, including access to state-of-the-art equipment, access to our
Attain IVF programs and access to our clinical and information technology
applications;
|
|
|
•
|
Enable
fertility centers to enhance their ability to provide superior care
through use of our proprietary ARTworks software, which provides
electronic medical records, treatment plan and success rate research
capabilities, decision support functionality and clinical risk management
auditing services;
|
|
|
•
|
Enable
vein clinics to enhance their ability to provide superior care through use
of our proprietary VPA information system, which is an end-to-end patient
and clinic operating system that provides decision support and revenue
cycle functions;
|
|
|
•
|
Help
our fertility centers and vein clinics drive additional patient volume
through our sales and marketing efforts, including our direct-to-consumer
advertising, internet marketing, physician referral development and
providing marketing materials and programs to our fertility centers and
vein clinics for their use; and
|
|
|
•
|
Convert
initial potential patient contacts into patients treated at our centers
and clinics. We believe we can accomplish this through the protocols we
established for our call center professionals and contact follow up
procedures our center and clinic staff employ to ensure patients attend
their consultation and all scheduled
treatments.
|
Increase
Penetration of Our Attain IVF Programs
Currently,
many third-party payors provide limited coverage for the diagnosis and treatment
of infertility. Our Attain IVF programs, which are offered directly to patients,
have been designed to offer attractive financial options to prospective
patients. For the twelve months ended December 31, 2009, approximately 12.4% of
self-pay patients in our Partner and Affiliate network utilized our Attain IVF
Refund Program. We formally introduced our Attain IVF Multi-Cycle Program in
July 2009. We believe that the penetration of our Attain IVF programs can be
meaningfully increased by educating patients on the improved success rates
associated with multiple treatment cycles and the packaged pricing features of
our Attain IVF programs, which allow for multiple treatment cycles and, in the
case of our Attain IVF Refund Program, a significant financial refund if the
treatments are unsuccessful. We also believe we can increase overall market
penetration of our Attain IVF programs by demonstrating to physicians at
potential Affiliate and Partner practices the benefit of increased patient
volume and retention that we believe result from offering our Attain IVF
programs. We have demonstrated the ability to increase Attain IVF Refund Program
penetration because certain of our fertility centers had Attain IVF Refund
Program penetration rates in excess of 25% during the twelve months ended
December 31, 2009.
12
Continue
Improving Operating Efficiencies
We
continuously seek opportunities to lower costs and realize operating
efficiencies through the implementation of a centralized infrastructure focused
on improved accounts receivable management, along with leveraging economies of
scale in support functions such as procurement, finance, information technology,
human resources, risk management and legal services. We expect to further
leverage our corporate infrastructure as we expand our network of Partner
fertility centers and vein clinics.
Sales
and Marketing
The
marketing departments for our Fertility Centers, Consumer Services and Vein
Clinics Divisions specialize in the development of sophisticated marketing and
sales programs that give contracted fertility centers and vein clinics access to
business-building techniques designed to facilitate growth and development.
Although we believe these marketing and sales efforts are often too expensive
for many individual physician practices, affiliation with us provides access to
greater marketing and sales capabilities than would otherwise be available. In
addition, our Consumer Services Division is focused on direct-to-consumer
marketing, which we believe represents a competitive advantage over
non-affiliated fertility centers. Our marketing services focus on referral
enhancement, relationships with local physicians, media and public
relations.
We
operate web portals that: allow visitors access to educational material
concerning infertility and vein care issues; provide links to our fertility
Partner and Affiliate practices; provide links to our vein clinics; allow
prospective patients to request fertility and vein care appointments or follow
up contact; and allow prospective patients to request information on our Attain
IVF programs and apply for treatment financing.
Competition
Our
business divisions operate in highly competitive areas. Our fertility centers
compete with national, regional and local physician practice fertility centers,
hospitals and university medical centers, some of which have programs that
compete with our Attain IVF programs. Our fertility centers may also compete
with fertility centers located outside of the United States, due to the self-pay
nature of IVF treatment. Our vein clinics compete with other vein care clinic
providers, dermatologist and surgical clinics that provide ELT and sclerotherapy
as an ancillary offering, vascular surgeons and interventional radiologists.
Barriers to entry in the vein care industry are low. We do not believe that we
currently face significant competition providing managerial services to
fertility centers and vein clinics. We believe that the fertility centers and
vein clinics we work with are well positioned to compete in our markets based on
the reputations of the physicians providing services at those centers and
clinics; however, there can be no assurance that these centers and clinics will
be able to compete effectively with existing providers in our markets or that
new competitors will not enter into our markets. These existing and new
competitors may have greater financial and other resources than we or our
fertility centers or vein clinics do. See “Risk Factors — We face increased
competition from existing providers, as well as new providers entering our
markets.”
Health
Care Regulation
The
health care industry is highly regulated. Our ability to operate profitably will
depend in part upon our ability, and the ability of our affiliated physicians
and physician practice groups, to obtain and maintain all licenses and other
approvals necessary to comply with applicable health care regulations. We
believe that health care regulations will continue to change. Therefore, we
monitor developments in health care law, and we are likely to be required to
modify our operations from time to time as our business and the regulatory
environment changes. Many aspects of our current and anticipated business
operations have not been the subject of specific judicial or regulatory
interpretation. A review of our business by courts or regulatory authorities may
result in a determination that could adversely affect our operations. In
addition, the health care regulatory environment may change in a way that
restricts our operations. Future changes in health care regulation are difficult
to predict and may constrain or require us to restructure our operations, which
could negatively impact our business and operating results.
13
Physician
Licensure Laws
The
practice of medicine is subject to state licensure laws, regulations and
approvals. We have established a system designed to ensure that the physicians
at our fertility centers and vein clinics are appropriately licensed under
applicable state law. If physicians at the centers or clinics fail to renew
their licenses on an annual basis or fail to maintain an unrestricted license,
our business, financial condition and results of operations may be negatively
impacted.
Corporate
Practice of Medicine
Some
states have laws that prevent business entities like us from practicing
medicine, employing physicians and other individuals licensed in the healing
arts or other learned profession and exercising control over their decisions,
also known, collectively, as the corporate practice of medicine. In some states
these prohibitions are expressly stated in a statute or regulation, whereas in
other states the prohibition is a matter of judicial or regulatory
interpretation. Additionally, in those states which apply such prohibitions to
individuals licensed in the healing arts or other learned profession, it is not
clear whether physician assistants or nurse practitioners would be subject to
such prohibitions.
In states
that prohibit the corporate practice of medicine, we operate by maintaining
long-term management contracts with affiliated medical practices, which are each
owned and operated by physicians and which employ or contract with additional
physicians. Under such an arrangement, the laws of most states focus on the
extent to which the corporation exercises control over the physicians and on the
ability of the physicians to use their own professional judgment as to diagnosis
and treatment. We do not represent to the public that we offer medical services,
and we do not exercise influence or control over the practice of medicine by
physicians or by their affiliated medical practices. In each of these states,
the affiliated fertility center or vein clinic is the sole employer of the
physicians, and the affiliated fertility center or vein clinic retains the full
authority to direct the medical, professional and ethical aspects of its medical
practice. Our fertility centers and vein clinics are duly licensed or qualified
as a medical practice or foreign corporation in the states where such license or
qualification is required.
Corporate
practice of medicine laws and their interpretations are continually evolving and
may change in the future. Moreover, these laws and their interpretations are
generally enforced by state courts and regulatory agencies that have broad
discretion in their enforcement. Although we neither employ physicians nor
provide medical services in those states that prohibit the corporate practice of
medicine, a state court or enforcement agency may conclude that we are engaged
in the corporate practice of medicine in those states where we employ nurse
practitioners and physician assistants who work under the supervision of the
physicians at our fertility centers and vein clinics or because we provide
services to physicians in connection with their performance of professional
medical services through our management contracts.
Although
we have not received notification from any state regulatory or similar authority
asserting that we are engaged in the corporate practice of medicine, to the
extent any act or service to be performed by us is construed by a court or
enforcement agency as constituting the practice of medicine in a particular
jurisdiction, we cannot be sure that such court or enforcement agency would not
construe our arrangements as violating that jurisdiction’s corporate practice of
medicine doctrine. If such a claim were successful, we could be subject to civil
and criminal penalties, could be required to restructure or terminate our
applicable contractual arrangements and managed physicians could have
restrictions imposed upon their licenses to practice medicine. Additionally, a
physician shareholder of a managed practice might successfully avoid
restrictions on the practice’s ability to operate independent of our management
on the grounds that the managed practice’s management arrangement with us
violates the state’s prohibition on the corporate practice of medicine. Such
results or the inability of us or the managed practices to restructure our
relationships to comply with such prohibitions could have an adverse effect on
our business, financial condition and results of operations.
14
Fee
Splitting
The laws
of some states prohibit physicians from splitting with anyone, other than
providers who are part of the same group practice, any professional fee,
commission, rebate or other form of compensation for any services not actually
and personally rendered. The precise language and judicial interpretation of
fee-splitting prohibitions varies from state to state.
Courts in
a number of states, including Illinois, have interpreted fee-splitting statutes
as prohibiting all percentage of gross revenue and percentage of net profit
management fee arrangements, despite the performance of legitimate management
services. Prior to November 1, 2009, our management agreement with our Partner
fertility center in Illinois provided that we be paid a base fee equal to a
fixed percentage of the revenues of the fertility center that declined to zero
to the extent the costs relating to the management of the fertility center
increase as a percentage of total revenues. Due to the substantial risk that
this compensation arrangement, being based on a percentage of revenues, would
not be upheld if challenged under Illinois law, we entered into a new
compensation agreement with this fertility Partner effective November 1, 2009.
Under the terms of our new cost-plus agreement our fees are now equal to
reimbursement of our costs incurred in operating the center, plus
9.5%.
Fee-splitting
laws and their interpretations vary from state to state and are enforced by
state courts and regulatory authorities that have broad discretion in their
enforcement. For example, our Attain IVF Refund Program could be interpreted by
one or more state regulators as a prohibited fee split to the extent that we
retain a portion of the payments patients pay directly to us for their medical
treatment by our fertility centers. There can be no assurance that these laws
will be interpreted in a manner consistent with our practices or that other laws
or regulations will not be enacted in the future that could have a material
adverse effect on our business, financial condition and operating results.
Penalties for violating fee-splitting statutes or regulations may include the
medical license revocation, suspension, probation or other disciplinary action
of the providers affiliated with our fertility centers or vein clinics who have
been found to violate the fee-splitting statutes or regulations.
In
addition, courts have refused to enforce contracts found to violate state
fee-splitting prohibitions. To the extent any of our contractual arrangements
are construed by a court or enforcement agency as violating a jurisdiction’s
fee-splitting laws, there is a possibility that some provisions of our
agreements may not be enforceable. For example, a physician shareholder of a
managed practice might successfully avoid payment of a management fee on the
grounds that the management arrangement violates the state’s fee-splitting
prohibition. We also may be required to redesign or terminate our arrangements,
including our Attain IVF programs, and our relationships with our fertility
centers or vein clinics in order to bring their activities into compliance with
such laws. The termination of, or our failure to, successfully restructure, any
such relationship could have a material adverse affect on our business,
financial condition and operating results. In particular, a forced restructuring
of our management fee could have a material impact on us. In addition, expansion
of our operations to new jurisdictions could require structural and
organizational modifications of our relationships with our fertility centers in
order to comply with additional statutes.
Further,
although our management agreements with our vein clinics provide that each vein
clinic pay us a fee equal to 150% of our expenses of operating and managing the
vein clinic, these fees have historically exceeded the operating margin
generated by any particular vein clinic prior to payment of the management fee.
Accordingly, each vein clinic only pays the portion of the management fee that
is equal to the amount of revenue generated by the clinic annually up to the
150% amount. As a result, our vein clinics do not generate any net profits at
year end. In those states that have interpreted fee-splitting statutes as
prohibiting a percentage of net revenue management fee arrangements where we
have vein clinics, there is material risk that a regulator could recharacterize
our management fee as 100% of net revenue in violation of such states’
fee-splitting statutes which would subject physicians affiliated with our vein
clinics to disciplinary actions or civil penalties.
Courts in
other states, including Maryland, where we have two vein clinics, have
interpreted their fee-splitting statutes to prohibit non-physicians from
receiving fees in connection with the management of a physician practice that do
not bear a reasonable relationship to the services being rendered based on the
fair market value of such services. A state regulator could conclude that 150%
of our expenses does not bear a reasonable relationship to the services
15
being
rendered because none of our vein clinics generate sufficient revenues to pay
the full management fee. If our management fee were to be challenged in a state
such as Maryland, there is substantial risk that this compensation method would
not be upheld, which could subject the providers who are affiliated with the
vein clinics in Maryland to disciplinary action as well as civil penalties. We
also have vein clinics in Wisconsin, Tennessee and Kansas which have a similar
requirement that the management fee reflect the fair market value of the
services being rendered and impose disciplinary actions, civil penalties and
criminal penalties on the physicians who are affiliated with our vein clinics in
those locations if such fee does not reflect the fair market value of the
services being rendered.
Federal
and State Anti-Kickback Prohibitions
Various
federal and state laws govern financial arrangements among health care
providers. The federal anti-kickback law prohibits the knowing and willful
offer, payment, solicitation or receipt of any form of remuneration in return
for, or with the purpose to induce, the referral of Medicare, Medicaid or other
federal health care program patients, or in return for, or with the purpose to
induce, the purchase, lease or order of items or services that are covered by
Medicare, Medicaid or other federal health care programs. Similarly, many state
laws prohibit the solicitation, payment or receipt of remuneration in return
for, or to induce, the referral of patients to private as well as government
programs in violation of these statutes.
Federal
and state anti-kickback statutes are very broad and it is possible that a court
could conclude that the marketing services we offer in exchange for a management
fee based on a percentage of net profits constitutes a payment in violation of
these statutes. Our fertility centers and vein clinics are also subject to these
statutes, but we do not oversee and are not responsible for their compliance
with these laws.
Violations
of these anti-kickback laws may result in substantial civil or criminal
penalties for individuals or entities. These laws may be enforced by the
government or by private individual whistleblowers. If we or our fertility
centers or vein clinics are found to have violated federal or state
anti-kickback laws, our business, operations or financial condition could be
adversely affected.
Physician
Self-Referral Prohibitions
The
federal physician self-referral statute, known as the “Stark” statute, prohibits
a physician from making a referral for certain designated health services to any
entity with which the physician has a financial relationship, unless there is an
exception in the statute that allows the referral. The entity that receives a
prohibited referral from a physician may not submit a bill to Medicare for that
service. Federal courts have ruled that a violation of the Stark statute, as
well as a violation of the federal anti-kickback law described above, can serve
as the basis for a Federal False Claims Act suit. Many state laws prohibit
physician referrals to entities with which the physician has a financial
interest, or require that the physician provide the patient notice of the
physician’s financial relationship before making the referral. Violation of the
Stark statute and state laws prohibiting physician referrals can result in
substantial civil penalties for both the referring physician and any entity that
submits a claim for a health care service made pursuant to a prohibited
referral. Although we have structured our arrangements with our fertility
centers and vein clinics to comply with the Stark statute and state laws
prohibiting certain physician referrals, because of the complexity of these
laws, these laws could be interpreted in a manner inconsistent with our
operations. In addition, our fertility centers and vein clinics are themselves
subject to these laws, but we do not oversee and are not responsible for their
compliance with these laws. Federal or state self-referral regulation could
adversely impact our arrangements with certain customers, and our ability to
market our services directly to physicians in a position to refer patients to
our fertility centers and vein clinics.
False
Claims
Under
separate federal statutes, submission of false or fraudulent claims to
government payors may lead to civil monetary penalties, criminal fines and
imprisonment and/or exclusion from participation in the Medicare, Medicaid and
other federally-funded health care programs. These false claims statutes include
the Federal False Claims Act, which allows any person to bring suit alleging
false or fraudulent Medicare or Medicaid claims or other violations of the
statute and to share in any amounts paid by the entity to the government in
fines or settlement. In some jurisdictions, even claims that were accurately
submitted for medically necessary health care services have been held by courts
16
to be
“false” where the provider was not in compliance with federal anti-kickback or
Stark laws, or applicable Medicare regulations. These private actions have
increased significantly in recent years and have increased the risk that we or
our vein clinics will have to defend a false claims action, pay fines or be
excluded from participation in the Medicare and/or Medicaid programs as a result
of an investigation involving our fertility centers or vein clinics arising out
of such an action.
Business
of Insurance
Laws and
regulatory approaches to insurance are state specific and vary widely from state
to state. Although most states supply statutory definitions of “insurance,”
these definitions are subject to disparate interpretation by state courts,
attorney generals and regulators. Our Attain IVF programs have several
characteristics that are present in an insurance contract. We view our Attain
IVF programs as guaranties or warranties of our fertility centers’ performance;
however, it is possible that an insurance regulator in a state where we conduct
business could take the position that our Attain IVF programs are insurance and
should be regulated as such by the state. If we are found to have engaged in the
business of insurance without a license, we could be subject to criminal and
civil penalties or be forced to comply with burdensome reserve requirements or
restructure the programs.
Health
Insurance Portability and Accountability Act of 1996
Health
care providers, health care clearinghouses and operators of health plans
(collectively, “covered entities”) are significantly affected by certain health
information requirements contained in HIPAA. HIPAA and its implementing
regulations established national standards for, among other things, certain
electronic health care transactions, the use and disclosure of certain
individually identifiable patient health information and the security of the
electronic systems maintaining this information. These are commonly known as the
HIPAA transaction and code set standards, privacy standards and security
standards, respectively.
HIPAA
allows covered entities to disclose protected health information to “business
associates” if the covered entities obtain satisfactory assurances that the
business associate will use the information only for the purposes for which it
was engaged by the covered entity, will safeguard the information from misuse
and will help the covered entity comply with some of the covered entity’s duties
under HIPAA. We are a “business associate” under HIPAA because we perform
services for or on behalf of covered entities, such as our fertility centers or
vein clinics, that involve the use or disclosure of protected health
information. We enter into business associate agreements with covered entities
and are contractually obligated to comply with the requirements of those
agreements.
The
American Recovery and Reinvestment Act of 2009, specifically the portion known
as the Health Information Technology for Economic and Clinical Health Act (the
“HITECH Act”), expanded the scope and application of HIPAA, including, among
other things, applying the security and certain privacy provisions of HIPAA
directly to business associates. Application of these rules to business
associates is a significant change. Previously, liability under HIPAA rested
exclusively with the covered entity. Under the HITECH Act, the business
associate now has responsibility and liability directly for a
breach.
On
February 17, 2010, certain administrative, physical and technical
safeguards and policy, procedure, and documentation requirements of the security
standards under HIPAA began applying to a business associate in the same manner
that they apply to a covered entity. For example, breaches of the security of
electronic health records may require disclosure to affected individuals, news
media and the Secretary of the U.S. Department of Health and Human
Services. Such requirements must be incorporated into the business associate
agreement between the business associate and the covered entity.
Under the
HITECH Act, business associates will face criminal and civil liabilities for
failure to comply with HIPAA. Criminal penalties may be imposed against persons
who obtain or disclose protected health information without authorization. In
addition, a state’s attorney general can bring civil actions against a person on
behalf of residents of the state that are adversely affected by violations of
either HIPAA or the HITECH Act. The attorney general can
17
either
seek to enjoin further violations or obtain money damages on behalf of the
residents harmed. The U.S. Department of Health and Human Services is also
beginning to perform periodic audits of health care providers to ensure that
required policies under the HITECH Act are in place. In addition, individuals
harmed by violations will be able to recover a percentage of monetary penalties
or a monetary settlement based upon methods established by the
U.S. Department of Health and Human Services for this private recovery.
HIPAA also authorizes the imposition of civil monetary penalties against
entities that employ or enter into contracts with individuals or entities that
have been excluded from participation in the Medicare or Medicaid programs,
which means that we could be subject to penalties if our fertility centers, vein
clinics or employees are excluded from participation in the Medicare or Medicaid
programs. Any failure to comply with these laws could have an adverse impact on
our business, operations or financial condition.
Antitrust
Laws
In
connection with the corporate practice of medicine laws referred to above, our
fertility centers and vein clinics are organized as separate legal entities. As
such, our fertility centers and vein clinics may be deemed to be persons
separate from both us and each other under antitrust laws and, accordingly,
subject to a wide range of laws that prohibit anti-competitive conduct among
separate legal entities. There can be no assurance that a review of our business
by courts or regulatory authorities would not have a material adverse effect on
our operation or the operation of our fertility centers or vein
clinics.
Future
Legislation and Regulation
Health
care providers are subject to federal, state and local laws and regulations, and
sanctions imposed under or changes to such laws or regulations could adversely
affect our operations or financial results. The federal fiscal year 2010 budget
establishes a reserve fund of more than $630 billion over the next
10 years to finance fundamental reform of the United States’ health care
system, in an effort to reduce costs and expand health care coverage. The fund
will be paid for by a combination of tax revenue and reductions in Medicare and
Medicaid spending.
In
addition, the White House announced in July 2009 that it had reached agreement
with leading hospital groups, including the American Hospital Association, to
cut federal payments under Medicare and Medicaid by $155 billion over
10 years as part of a plan to offset a portion of the cost of a national
health insurance and health reform proposal. Much of these savings are reported
to be derived from across-the-board cuts in Medicare hospital payments, with at
least $50 billion in the cuts linked directly to increases in the number of
uninsured who would be provided coverage under the proposed national health
insurance proposal.
There are
currently numerous proposals on the federal and state levels for comprehensive
reforms relating to health care that could affect payment and reimbursement for
health care services in the United States. The U.S. Congress is considering
legislation that could dramatically overhaul the health care system, including
the possibility of a government health care plan. If national reform legislation
is enacted, we may benefit from certain provisions thereof, and, conversely, may
be adversely affected by other provisions. For example, because our Attain IVF
programs are self-pay programs for patients that do not have insurance coverage
for fertility treatments, health care reform that increases insurance coverage
for fertility treatments could lead to a decrease in demand for our Attain IVF
programs. We cannot predict whether any such reforms will ultimately be adopted
or the impact that such reforms may have on the demand or payment for our
services.
Employees
As of
December 31, 2009, we had 1,322 employees. Of these, 1,073 were employed
by our Fertility Centers Division, 15 by our Consumer Services Division, 202 by
our Vein Clinics Division and 32 were employed at our corporate headquarters,
including 8 who were executive management. Of the 1,322 employees, 145
were employed on a part-time basis and 101 were employed on a per diem
basis. We are not a party to any collective bargaining agreement and
we believe that our employee relationships are good.
Segment
Information
We follow
the requirements contained in ASC 280, with respect to identifying and reporting
business segments. This statement requires that segment reporting reflect our
organizational structure, major revenue sources, lines of
18
responsibility
and senior management’s perspective of an organization. With the
acquisition of Vein Clinics of America (VCA) during the third quarter of 2007,
we reorganized our service offerings into three major product
lines: Fertility Centers, Consumer Services and Vein
Clinics. Each of the operating segments includes an element of
overhead specifically associated with it. Such overhead costs were previously
reported as General and Administrative costs, and have been reclassified in all
periods presented to better reflect the operating results of our business
segments.
Performance
by segment, for the three years ended December 31 2009, 2008 (restated) and 2007
(restated) are presented below (000’s omitted):
|
Fertility
Centers
|
Consumer
Services
|
Vein
Clinics
(1)
|
Corp.
G&A
|
Consolidated
|
||||||||||||||||
|
For
the Year ended December 31, 2009
|
||||||||||||||||||||
|
Revenues
|
$ | 145,309 | 20,826 | 50,625 | $ | — | $ | 216,760 | ||||||||||||
|
Cost of Services
|
$ | 133,706 | 15,639 | 46,525 | — | 195,870 | ||||||||||||||
|
Contribution
|
11,603 | 5,187 | 4,100 | — | 20,890 | |||||||||||||||
|
Operating Margin
|
8.0 | % | 24.9 | % | 8.1 | % | — | 9.6 | % | |||||||||||
|
General and
administrative
|
0 | 0 | 0 | 12,155 | 12,155 | |||||||||||||||
|
Interest (income) expense,
net
|
(149 | ) | 0 | 0 | 1,059 | 910 | ||||||||||||||
|
Income (loss) before income
taxes
|
$ | 11,752 | $ | 5,187 | $ | 4,100 | $ | (13,214 | ) | $ | 7,825 | |||||||||
|
Depreciation expense included
above
|
4,076 | 0 | 873 | 873 | 5,822 | |||||||||||||||
|
Capital expenditures,
net
|
4,173 | 0 | 947 | 790 | 5,910 | |||||||||||||||
|
Total assets
|
39,086 | 104 | 49,845 | 35,274 | 124,309 | |||||||||||||||
|
For
the Year ended December 31, 2008
|
||||||||||||||||||||
|
Revenues
|
$ | 138,440 | $ | 19,763 | $ | 39,950 | $ | — | $ | 198,153 | ||||||||||
|
Cost of Services
|
128,224 | 14,344 | 37,299 | — | 179,867 | |||||||||||||||
|
Contribution
|
10,216 | 5,419 | 2,651 | — | 18,286 | |||||||||||||||
|
Operating Margin
|
7.4 | % | 27.4 | % | 6.6 | % | — | 9.2 | % | |||||||||||
|
General and
administrative
|
0 | 0 | 0 | 10,654 | 10,654 | |||||||||||||||
|
Interest (income) expense,
net
|
(181 | ) | 0 | 8 | 1,353 | 1,180 | ||||||||||||||
|
Income (loss) before income
taxes
|
$ | 10,397 | $ | 5,419 | $ | 2,643 | $ | (12,007 | ) | $ | 6,452 | |||||||||
|
Depreciation expense included
above
|
$ | 4,327 | $ | 3 | $ | 761 | $ | 898 | $ | 5,989 | ||||||||||
|
Capital expenditures,
net
|
$ | 4,053 | $ | — | $ | 1,057 | $ | 585 | $ | 5,695 | ||||||||||
|
Total assets
|
$ | 36,885 | $ | 331 | $ | 46,750 | $ | 37,475 | $ | 121,441 | ||||||||||
|
For
the Year ended December 31, 2007
|
||||||||||||||||||||
|
Revenues
|
$ | 121,078 | $ | 16,460 | $ | 14,284 | $ | — | $ | 151,822 | ||||||||||
|
Cost of Services
|
111,059 | 12,336 | 13,304 | — | 136,699 | |||||||||||||||
|
Contribution
|
10,019 | 4,124 | 980 | — | 15,123 | |||||||||||||||
|
Operating Margin
|
8.3 | % | 25.1 | % | 6.9 | % | — | 10.0 | % | |||||||||||
|
General and
administrative
|
— | — | — | 10,537 | 10,537 | |||||||||||||||
|
Interest (income) expense,
net
|
(203 | ) | — | 2 | 81 | (120 | ) | |||||||||||||
|
Income (loss) before income
taxes
|
$ | 10,222 | $ | 4,124 | $ | 978 | $ | (10,618 | ) | $ | 4,706 | |||||||||
|
Depreciation expense included
above
|
$ | 4,003 | $ | 3 | $ | 255 | $ | 846 | $ | 5,107 | ||||||||||
|
Capital expenditures,
net
|
$ | 4,654 | $ | — | $ | 906 | $ | 662 | $ | 6,222 | ||||||||||
|
Total assets
|
$ | 42,586 | $ | 888 | $ | 44,786 | $ | 25,911 | $ | 114,171 | ||||||||||
(1) Acquired
August 8, 2007.
Our
Fertility Centers and Vein Clinics
For the
years ended December 31, 2009, 2008 and 2007, the following contracted
fertility centers each individually provided greater than 10% of our revenues,
net and/or contribution as follows:
19
|
Percent
of Company
Revenues,
net
|
Percent
of
Contribution
|
|||||||||||||||||||||||
|
2009
|
2008
|
2007
|
2009
|
2008
|
2007
|
|||||||||||||||||||
|
R.S.C.
of New England
|
7.2 | 7.2 | 8.9 | 9.1 | 8.7 | 10.5 | ||||||||||||||||||
|
Fertility
Centers of Illinois
|
13.1 | 16.3 | 19.2 | 10.3 | 15.2 | 17.6 | ||||||||||||||||||
|
Shady
Grove Fertility Center
|
17.6 | 18.0 | 21.3 | 15.4 | 17.2 | 21.4 | ||||||||||||||||||
Under our
fertility Partner agreements with the R.S.C. of New England and Shady Grove
Fertility Center, we receive as compensation for our services a three-part fee
comprised of: a percentage of the fertility center’s net revenues;
reimbursed costs of services (costs incurred in servicing a fertility center and
any costs paid on behalf of the fertility center); and either a fixed
percentage, or a fixed dollar amount, of the fertility center’s earnings after
services fees, which may be subject to further limits.
The third
tier of our fee structure under these significant Partner agreements is as
follows:
|
•
|
RSC
of New England — a fixed annual percentage of the center’s
earnings.
|
|
|
•
|
Shady
Grove — a fixed dollar amount of the center’s earnings subject to a
fixed percentage of the center’s earnings limitation. The upper boundary
of the calculation is $1,071,000 and the lower boundary of the calculation
is $540,000.
|
Under our
current Partner agreement with FCI, however, we do not receive a three-part
fee. Rather, effective as of November 1, 2009, we receive a fee that
is generally equal to the operating expenses associated with managing FCI’s
medical practice plus 9.5% of such expenses. Our revenues from FCI
prior to November 1, 2009 were, pursuant to our current partner agreement with
FCI, set at a fixed annual amount paid monthly.
A
complete listing of our fertility Partner agreements and vein clinic locations
is presented below.
Fertility
Partner Agreements
|
|
||||||||||||||
|
Name
|
State
|
Year
Contract
Acquired
|
Remaining
Contract
Years
|
No.
of
M.D.s
|
No.
of
PhDs
|
|||||||||
|
Foulk
& Whitten Nevada Center for Reproductive Medicine,
P.C.
|
NV
|
December
2009
|
24
|
2
|
0
|
|||||||||
|
Idaho
Center for Reproductive Medicine, P.C.
|
ID
|
December
2009
|
24
|
2
|
0
|
|||||||||
|
Utah
Fertility Center, P.C.
|
UT
|
December
2009
|
24
|
3
|
0
|
|||||||||
|
Arizona
Reproductive Medicine Specialists, Ltd.
|
AZ
|
July
2008
|
23
|
4
|
1
|
|||||||||
|
Southeastern
Fertility Centers, P.A.
|
SC
|
April
2008
|
23
|
3
|
1
|
|||||||||
|
Center
for Reproductive Medicine, P.A.
|
FL
|
August
2007
|
22
|
4
|
1
|
|||||||||
|
Reproductive
Partners Medical Group, Inc.
|
CA
|
January
2005
|
19
|
9
|
0
|
|||||||||
|
Seattle
Reproductive Medicine, Inc., P.S.
|
WA
|
January
2004
|
7
|
7
|
1
|
|||||||||
|
Reproductive
Endocrine Associates of Charlotte, P.C.
|
NC
|
September
2003
|
8
|
6
|
1
|
|||||||||
|
Northwest
Center for Infertility & Reproductive
Endocrinology
|
FL
|
April
2002
|
7
|
7
|
1
|
|||||||||
|
Shady
Grove Fertility Reproductive Science
Center, P.C.
|
MD,
VA & DC
|
March
1998
|
13
|
21
|
2
|
|||||||||
|
Fertility
Centers of Illinois, S.C.
|
IL
|
February
1997
|
12
|
11
|
2
|
|||||||||
|
Bay
Area Fertility & Gynecology Medical Group,
Inc.
|
CA
|
January
1997
|
11
|
6
|
1
|
|||||||||
|
MPD
Medical Associates (MA), P.C. (doing business as RSC of New
England)
|
MA,
NH & RI
|
July
1988
|
2
|
7
|
1
|
|||||||||
20
Vein
Clinic Locations
|
Location
|
Date Clinic Opened
|
|
Cleveland,
OH
|
April
2009
|
|
Cincinnati,
OH
|
January
2009
|
|
Pittsburgh,
PA
|
December
2008
|
|
Skokie,
IL
|
December
2008
|
|
Marietta,
GA
|
June
2008
|
|
Alexandria,
VA
|
April
2008
|
|
Boca
Raton, FL
|
February
2008
|
|
Sterling,
VA
|
December
2007
|
|
Ft. Lauderdale,
FL
|
July
2007
|
|
St. Louis,
MO
|
January
2007
|
|
Merrillville,
IN
|
August
2006
|
|
Kansas
City, MO
|
June
2006
|
|
West
Palm Beach, FL
|
December
2005
|
|
Alpharetta,
GA
|
October
2005
|
|
Gurnee,
IL
|
September
2005
|
|
Naperville,
IL
|
September
2004
|
|
Lawrenceville,
GA
|
September
2001
|
|
Indianapolis,
IN
|
April
2001
|
|
Knoxville,
TN
|
March
2001
|
|
Raleigh,
NC
|
March
2000
|
|
Greensboro,
NC
|
January
2000
|
|
Madison,
WI
|
March
1999
|
|
Rockville,
MD
|
November
1998
|
|
Milwaukee,
WI
|
March
1998
|
|
Charlotte,
NC
|
February
1998
|
|
Orland
Park, IL
|
November
1996
|
|
Fairfax,
VA
|
March
1992
|
|
Overland
Park, KS
|
April
1991
|
|
Owings
Mills, MD
|
July
1990
|
|
Buffalo
Grove, IL
|
August
1989
|
|
Atlanta,
GA
|
June
1988
|
|
Oak
Brook, IL
|
Pre-1985
|
|
Chicago,
IL
|
Pre-1985
|
|
Schaumburg,
IL
|
Pre-1985
|
21
ITEM
1A. Risk
Factors
RISK
FACTORS
In
evaluating our business, every investor should carefully consider the following
risks. Our business, financial condition or results of operations
could be materially adversely affected by any of the following
risks.
The
loss of one or more of our Partner fertility centers would lead to a decline in
our revenues and profit.
The
contracts that we enter into with our Partner fertility centers typically have
terms that range from 10 to 25 years and contain automatic renewal
provisions. Some of these agreements also contain provisions that allow the
Partner fertility center to terminate the agreement, upon 12 months’ prior
notice, at any time after five years from the agreement’s effective date. Our
two largest Partner fertility centers provided approximately 46% of our
Fertility Centers Division revenues for the year ended December 31, 2009.
If either of these Partner fertility centers, or any of our other Partner
fertility centers, were to terminate its agreement with us, we would lose all of
the revenues associated with such Partner fertility center, but would not
experience any meaningful reduction in our infrastructure costs.
We
may not be able to find suitable Partner candidates or successfully integrate
the operations of the fertility centers with which we enter into Partner
contracts.
A key
part of our business strategy is to enter into additional Partner contracts. We
cannot assure you that we will be able to find suitable Partner candidates or
that the fertility centers that we enter into Partner contracts with will be
successful. Even if suitable Partner candidates are identified, negotiation over
suitable terms and conditions may be protracted and unsuccessful, and we may not
be able to achieve planned increases in the number of Partner centers. Further,
achieving the anticipated benefits of current and possible future Partner
contracts will depend in part upon whether we can integrate the operations of
those fertility centers with our operations in a timely and cost-effective
manner. The process of integrating the operations of Partner fertility centers
with our operations is complex, expensive and time consuming and involves a
number of risks, including, but not limited to:
|
•
|
difficulties
in integrating or retaining key medical providers of the Partner fertility
center;
|
|
|
•
|
difficulties
in integrating the operations of the Partner fertility center, such as
information technology resources and financial and operational
data;
|
|
|
•
|
diversion
of our management’s attention; and
|
|
|
•
|
potential
incompatibility of cultures.
|
We
are dependent on the medical providers in our fertility centers and vein clinics
to successfully execute our business strategy.
Although
we manage our fertility centers and vein clinics, the medical providers at those
centers and clinics provide medical services directly to patients and we do not
have control over their medical activities. We cannot guarantee any medical
provider’s ability to generate positive patient outcomes, build a positive
reputation for their practice or to comply with our expectations. If the medical
providers in our fertility centers and vein clinics act negligently or
unethically, allow their medical practices to deteriorate or do not meet our
growth expectations, it could diminish the value of our brand and our results of
operations could be adversely affected.
22
We
may have difficulty attracting and retaining physicians for our fertility
centers and vein clinics.
A key
part of our business strategy is to enter into additional Partner contracts and
open new vein clinics. The success of our fertility centers is dependent upon
our ability to retain the key medical providers associated with those centers.
If one or more key medical providers were to depart from a fertility center, our
business could suffer. Our ability to open new vein clinics is dependent upon
identifying, recruiting and retaining qualified physicians to perform procedures
at these clinics. We have had difficulties staffing new vein clinics because
some third-party payors require that the physicians performing procedures at
these clinics have certain specified credentials. We will not be able to
implement successfully our business strategy if we are unable to properly staff
our fertility centers and vein clinics.
A
reduction in reimbursements or an inability to negotiate attractive
reimbursement rates from third-party payors for the services that our Partner
centers or vein clinics provide could adversely affect our revenues and
growth.
A
significant portion of our fertility Partner and vein clinic revenues depends on
reimbursements to the underlying physician practices from third-party payors.
These third parties include private health insurers and other organizations,
such as health maintenance organizations, as well as government authorities.
Third parties are systematically challenging prices charged for medical
treatment. A third-party payor may deny or reduce reimbursement if it determines
that a prescribed treatment is not used in accordance with cost-effective
treatment methods, as determined by the payor, or is experimental, unnecessary
or inappropriate. In addition, although third parties may approve reimbursement,
such approvals may be under terms and conditions that discourage use of our
services, even if those services are safer or more effective than alternative
services. A reduction in reimbursements from third-party payors, whether in the
form of changes to reimbursement contracts, such as by limiting reimbursement
for certain procedures to specialists, loss of reimbursement contracts, solvency
issues on the part of the payors, or in the case of our vein clinics, changes in
Medicare reimbursement, would cause patients to reduce their treatments or
obtain services from other providers and could reduce our revenues and
profitability. Our ability to profitably open vein clinics in new markets also
significantly depends on our ability to obtain attractive reimbursement rates
from third-party payors in those new markets. If we are unable to obtain
satisfactory reimbursement rates from third-party payors for vein clinics in new
markets, our growth would suffer.
In early
2009, one of our top fertility centers in the Midwest terminated a reimbursement
contract with an important third-party payor. Contribution from this fertility
center in 2008 was approximately $2.3 million and this third-party payor
represented approximately 20% of this contribution, or $460,000.
Health
care reform could impact the demand for our services.
There are
currently numerous proposals on the federal and state levels for comprehensive
reforms relating to health care that could affect payment and reimbursement for
health care services in the United States. The U.S. Congress is considering
legislation that could dramatically overhaul the health care system, including
the possibility of a government health care plan. We cannot predict whether any
such reforms will ultimately be adopted or the impact that such reforms may have
on the demand or payment for our services. Because our Attain IVF programs are
self-pay programs for patients that do not have insurance coverage for fertility
treatments, health care reform that increases insurance coverage for fertility
treatments could lead to a decrease in demand for our Attain IVF
programs.
We
face competition from existing providers, as well as new providers entering our
markets.
Our
business divisions operate in highly competitive areas. Our fertility centers
compete with national, regional and local physician practice fertility centers,
hospitals and university medical centers, some of which have programs that
compete with our Attain IVF programs. Our fertility centers may also compete
with fertility centers located outside of the United States, due to the self-pay
nature of IVF treatment. Our vein clinics compete with other vein care clinic
providers, dermatologist and surgical clinics that provide ELT and sclerotherapy
as an ancillary offering, vascular surgeons and interventional radiologists.
Barriers to entry in the vein care industry are low. New health care providers
23
that
enter our markets impact our market share, patient volume and growth rates.
Increased competitive pressures require us to commit resources to marketing
efforts, which impacts our margins and profitability. There can be no assurance
that our fertility centers or vein clinics will be able to compete effectively
with existing providers in our markets or that new competitors will not enter
into our markets. These existing and new competitors may have greater financial
and other resources than we or our fertility centers or vein clinics do.
Increased competition could also make it more difficult for us to expand our
business by entering into new contracts with fertility centers or opening new
vein clinics.
The
development of alternative treatments could diminish demand for our
services.
The
fertility and vein care industries are dynamic, and new, technologically
intensive treatments are constantly under development. New treatments that are
more effective or provide better reimbursement could decrease patient demand or
profitability for the treatments that our fertility centers or vein clinics
currently offer. If our fertility centers or vein clinics do not adopt new
treatments as they are developed, patients could seek treatment
elsewhere.
If
we are found not to be in compliance with applicable laws and regulations, we
could be subject to significant fines or penalties, be forced to curtail certain
of our operations or rearrange material agreements to our
detriment.
We, and
each of our fertility centers and vein clinics, are subject to numerous federal
and state laws and regulations, including, but not limited to, federal and state
anti-kickback laws, controlled substances laws, the federal Stark law and state
self-referral laws, false claims laws, the Health Insurance Portability and
Accountability Act of 1996 (“HIPAA”), Medicare and Medicaid regulations and laws
regulating the business of insurance. These laws and regulations are extremely
complex and could be subject to various interpretations. Our fertility centers
and vein clinics are also subject to these statutes, but we do not oversee, nor
are we responsible for, their compliance with these laws. Many aspects of our
business, to date, have not been the subject of federal or state regulatory
review and we, and any of our fertility centers or vein clinics, may not have
been in compliance at all times with all applicable laws and regulations. If we,
or our fertility centers or vein clinics, are found by a court or regulatory
authority to have violated any applicable laws or regulations, we could be
subject to significant fines or penalties or be forced to curtail certain of our
operations.
Further,
the laws of many states prohibit physicians from splitting fees with
non-physicians, or other physicians, and prohibit non-physician entities from
practicing medicine. These laws vary from state to state and are enforced by the
courts and by regulatory authorities with broad discretion. Many aspects of our
business, to date, have not been the subject of judicial or regulatory
interpretation; thus, a review of our business by courts or regulatory
authorities may result in determinations that could adversely affect our
operations. In addition, the health care regulatory environment could change so
as to restrict our existing operations or their expansion. State corporate
practice of medicine laws may be interpreted as prohibiting corporations or
associations from exercising control over physicians or employing nurse
practitioners or physician assistants and may prohibit physicians from
practicing medicine in partnership with, or as employees of, any person not
licensed to practice medicine.
State
regulators may seek to challenge the arrangements that we have with our
fertility centers and vein clinics. A determination in any state that we are
engaged in the corporate practice of medicine or any unlawful fee-splitting
arrangement could render any management agreement between us and a practice
located in such state unenforceable or subject to modification, which could have
an adverse effect on our financial condition and results of operations.
Regulatory authorities or other parties may assert that we are or a practice is
engaged in the corporate practice of medicine or that the management fees paid
to us by the managed practices constitute unlawful fee-splitting or the
corporate practice of medicine. If such a claim were asserted successfully, we
could be subject to civil and criminal penalties, managed physicians could have
restrictions imposed upon their licenses to practice medicine, parts or all of
our existing management agreements could be rendered unenforceable and we could
be required to restructure our contractual arrangements with the managed
practices, all of which could have an adverse effect on our financial condition
and results of operations.
Although
we view our Attain IVF programs as a guaranty or warranty of our fertility
centers’ performance, the Attain IVF programs have several characteristics that
are present in an insurance contract. As such, an insurance regulator in a
particular state may find that we have been and are engaged in the business of
insurance without a license, which could subject us to criminal and civil
liabilities and would subject our Attain IVF programs to substantial
24
regulation
in that state as an insurance contract, including burdensome reserve
requirements. In addition, in states that prohibit physicians from splitting
professional fees with non-physicians, we could be required to restructure our
Attain IVF programs if a state concluded that our Attain IVF programs
constituted fee splitting because we retain a portion of the payments patients
pay directly to us for their medical treatment by our fertility centers. The
imposition of any such liabilities and any such changes in our method of doing
business would likely reduce revenues and contribution from our Consumer
Services Division.
Additionally,
our management agreements with our vein clinics provide that the vein clinics
will pay us a fee equal to 150% of our expenses of operating and managing the
vein clinics. These fees have historically exceeded the operating margin
generated by any particular vein clinic prior to payment of the management fee.
Accordingly, each vein clinic only pays the portion of the management fee that
is equal to the amount of revenue generated by the clinic annually up to the
150% amount. As a result, our vein clinics do not generate any net profits at
year end. A state regulator could find that such a compensation model is
actually based on a percentage of the revenue of a particular vein clinic or
that our management fee is not commensurate with the services we provide, in
which case our management agreements would be violating fee-splitting laws of
certain states where we operate vein clinics. We could be forced to restructure
the fee structure under the management agreements to our material financial
detriment or the providers affiliated with our vein clinics who have been found
to violate the fee-splitting statutes or regulations may be subject to
disciplinary action or criminal sanctions, which could lead to the closure of
one or more of our vein clinics.
Our
arrangements with our fertility centers and vein clinics may trigger the
application of federal and various state franchise laws. We have never sought to
comply with any such franchise laws, nor have we ever sought any exemptions from
such laws. The U.S. Federal Trade Commission could bring an enforcement
action against us for failure to comply with federal franchise laws and could
impose significant fines against us, order us to pay restitution to the
fertility centers and vein clinics that are found to be franchisees (and the
physicians that own or operate them) and/or seek criminal sanctions against us.
Under the laws of certain of the states in which we operate, the physicians that
own or operate our fertility centers and vein clinics may bring private causes
of actions against us for violating such laws. In many of these jurisdictions,
in addition to a judgment for actual damages, a court could award the physicians
rescission, attorney’s fees and costs and treble damages. Additionally, we could
be subject to fines and criminal sanctions. Even if we were to comply with these
federal and state franchise laws, we would still be potentially liable for prior
violations that occurred prior to the time we came into compliance with such
laws.
New
or enhanced laws and regulations affecting the fertility industry could increase
our costs of compliance and force us to alter certain of our
operations.
A number
of high profile events have occurred recently related to ART and fertility
practices generally, such as the implantation of a greater than recommended
number of embryos, resulting in extraordinarily high-order multiple births, or
the implantation of incorrect patient embryos. Federal and state regulators may
more carefully scrutinize the fertility industry as a result of these events,
and may adopt more stringent laws and regulations that could increase our
compliance costs or force us to alter certain of our operations.
We
and our Partner fertility centers and vein clinics may not have sufficient
liability insurance to cover potential claims.
The
medical procedures performed by physicians and other medical personnel in our
network of fertility centers and vein clinics can involve significant
complications, including genetically defective births, embryo loss and patient
death. We are likely to be, and from time to time have been, named as a party in
legal proceedings involving medical malpractice or other injuries that occur at
one of our fertility centers or vein clinics, particularly in those fertility
centers where we provide the services of a physician assistant or nurse
practitioner. A successful malpractice claim could exceed the limits of
insurance that we maintain, in which case we would have to fund any settlement
in excess of our insurance coverage. We also maintain medical malpractice
insurance coverage for our Partner fertility centers and vein clinics, and a
successful malpractice claim against one of those centers or clinics in excess
of the coverage we maintain for them would adversely affect the revenues we
derive from those centers and clinics. In addition, the captive insurance
company that provides a portion of our insurance coverage does not maintain
reserves in amounts that would be required of other, larger insurers, and
therefore may not have adequate capital to fund a claim against us or the
Partner fertility centers covered by the captive insurance company. A
malpractice claim, whether or not successful, could be costly to defend, could
consume management resources and could adversely affect our reputation and
business and the reputations and businesses of our Partner fertility centers and
vein clinics. We also cannot assure you that we or our Partner fertility centers
or vein clinics will be able to obtain insurance coverage in the future on
commercially reasonable terms, or at all.
25
Our
success depends on retaining key members of our management team.
The
success of our business strategy depends on the continued contribution of key
members of our management team. The loss of key members of this team could
disrupt our growth plans and our ability to implement our business
strategy.
We
rely on a limited number of third-party vendors for medicine and
supplies.
Our
fertility centers and vein clinics rely on a limited number of third-party
vendors that produce medications and supplies vital to patient treatment, such
as AngioDynamics, Inc., which provides the only U.S. Food and Drug
Administration approved solution used in sclerotherapy. If any of these vendors
were to experience a supply shortage or cease doing business, and we were unable
to find an alternative third-party vendor, we might not be able to properly
serve our patients.
Our
credit agreement contains covenants that impose restrictions on us that may
limit our operating flexibility, prevent us from entering into extraordinary
transactions that benefit our stockholders and limit our growth.
Our
credit agreement contains covenants that restrict our flexibility to conduct
business. These covenants prohibit or limit, among other things:
|
•
|
the
payment of dividends to our stockholders;
|
|
|
•
|
the
incurrence of additional indebtedness;
|
|
|
•
|
the
making of certain types of restricted payments and
investments;
|
|
|
•
|
sales
of assets; and
|
|
|
•
|
consolidations,
mergers and transfers of all or substantially all of our
assets.
|
The
credit agreement also requires that we maintain certain leverage and fixed
charge ratios and minimum levels of earnings before interest, taxes,
depreciation and amortization. Our failure to comply with any of these covenants
could cause the lenders to declare a default and accelerate amounts due to them
under the credit agreement.
In
addition, our credit agreement places certain restrictions on our ability to
acquire the business, assets or capital stock of fertility centers. For example,
our credit agreement prevents us from acquiring a fertility center for a
purchase price in excess of $5.5 million (increasing to $6.0 million
after August 31, 2010) without the prior written consent of our
lender. In addition, our credit agreement prevents us from making acquisitions
of fertility centers that aggregate in excess of $11 million for the period
from August 1, 2009 through July 31, 2010, or that exceed
$12 million for the period after August 1, 2010. If we identify
fertility centers that we want to acquire in excess of limits in our credit
agreement and do not obtain the consent of our lender to those acquisitions, we
may not be able to execute on our strategy.
Our
failure to maintain effective internal control over financial reporting could
lead to inaccuracies in our reported financial results.
A
material weakness is a significant deficiency, or combination of significant
deficiencies, that results in more than a remote likelihood that a material
misstatement of the annual or interim financial statements will not be prevented
or detected. If our independent registered public accounting firm
were to determine that a significant deficiency were to exist, or if we were
otherwise unable to achieve and maintain effective internal controls on a timely
basis, management would not be able to conclude that we have effective internal
control over financial reporting for purposes of Section 404 of the
Sarbanes-Oxley Act of 2002. In addition, our independent registered
public accounting firm
26
would not
be able to certify as to the effectiveness of our internal control over
financial reporting. Moreover, any failure to establish and maintain
effective systems of internal control and procedures may impair our ability to
accurately report our financial results. Such failures and the
reporting that our system of internal controls over financial reporting was not
effective could result in a restatement of our financial statements and cause
investors to lose confidence in the reliability of our financial statements,
which could result in a decline in our stock price.
We
may not have adequate protection for our intellectual property
rights.
Trade
secrets and other proprietary information not protected by patents are critical
to our business. Our sole means of protecting this information is to utilize
confidentiality agreements with employees, third parties and consultants. If
these agreements are breached, another entity could obtain our trade secrets and
proprietary information and attempt to replicate our business model, which could
have an adverse effect on our business.
We
could be subject to additional income tax liabilities.
We are
subject to income taxes in various states within the United States. Judgment is
often required in evaluating our provision for income taxes. During the ordinary
course of business, there are certain transactions for which the ultimate tax
determination is uncertain. For example, certain taxing authorities may take the
position that we are providing services in jurisdictions where our Partner
fertility centers operate. The final determination of tax audits and any related
litigation could be materially different from our historical income tax
provisions and accruals. The results of an audit or litigation could have a
material effect on our operating results or cash flows.
ITEM
1B. Unresolved
Staff Comments
None.
ITEM 2. Properties
Our
headquarters and executive offices are located in Purchase, New York, where we
occupy approximately 18,500 square feet under a lease expiring in 2012. Future
lease payments will approximate $51,100 per month.
We also
lease or sublease locations for our fertility centers and vein clinics. Costs
associated with the fertility agreements are reimbursed to us as part of our fee
agreement with the applicable clinic whereas costs associated with vein clinic
locations are not reimbursed.
We
believe that our executive offices and the space occupied by our clinics are
adequate for our operations.
ITEM 3. Legal
Proceedings
From time
to time, we and our Partner fertility centers and vein clinics and their
physicians are parties to legal proceedings in the ordinary course of
business. We are exposed to claims of professional negligence based
on services performed by our employees, including physician assistants and nurse
practitioners, we well as based on our relationships with physicians providing
treatments at our Partner fertility centers and vein clinics. None of
these proceedings is expected to have a material adverse effect on our financial
position, results of operations or cash flow.
ITEM
4. Reserved
27
PART
II
|
ITEM
5.
|
Market
for Registrant's Common Equity, Related Stockholder Matters and Issuer
Purchases of Equity Securities
|
Our
common stock is traded on the NASDAQ Global Market under the symbol “INMD” The
following table sets forth the high and low closing sales price for our common
stock, as reported on the NASDAQ Global Market.
|
Common
Stock
|
||||||||
|
High
|
Low
|
|||||||
|
2009
|
||||||||
|
Fourth
Quarter
|
$ | 9.37 | $ | 7.95 | ||||
|
Third
Quarter
|
$ | 10.25 | $ | 7.03 | ||||
|
Second
Quarter
|
$ | 7.99 | $ | 5.81 | ||||
|
First
Quarter
|
$ | 7.45 | $ | 5.60 | ||||
|
2008
|
||||||||
|
Fourth
Quarter
|
$ | 6.97 | $ | 4.80 | ||||
|
Third
Quarter
|
$ | 8.17 | $ | 6.01 | ||||
|
Second
Quarter
|
$ | 10.23 | $ | 7.07 | ||||
|
First
Quarter
|
$ | 11.95 | $ | 8.50 | ||||
On March
4, 2010, there were approximately 111 holders of record of the Common Stock and
approximately 926 beneficial owners of shares registered in nominee or
street name.
Dividend
Policy
We have
not paid cash dividends on our common stock during the last two fiscal years,
and we currently anticipate retaining all available funds for use in the
operation and expansion of the business. Therefore, we do not anticipate paying
any cash dividends on our common stock in the foreseeable future.
Securities
Authorized for Issuance Under Equity Compensation Plans
We have
three stock option plans which have been approved by our
shareholders. The following table sets forth certain information
relative to these stock option plans.
|
Plan
Category
|
Number
of securities to be issued upon exercise of outstanding
options
|
Weighted
average exercise price of outstanding options
|
Number
of securities remaining available for future issuance under equity
compensation plans (excluding securities reflected in column
(a)
|
|||||||||
|
(a)
|
(b)
|
(c)
|
||||||||||
|
Equity
compensation plans approved by security holders
|
205,685 | $ | 6.20 | 464,933 | ||||||||
|
Equity
compensation plans not approved by security holders
|
¾ | ¾ | ¾ | |||||||||
|
Total
|
205,685 | $ | 6.20 | 464,933 | ||||||||
During
2009, 2008 and 2007, we issued approximately 142,000, 99,000 and 78,000 shares,
respectively, of restricted common stock as deferred compensation to several of
our officers and directors with an aggregate value of $978,000 $889,000 and
$956,000 respectively. These shares were valued at their fair value on the date
of grant, and are amortized to expense over their vesting period, which
approximates the service period.
During
2008, we also issued approximately 128,000 incentive stock options to certain
members of our management team. These options have a ten year life, vest over
four years and had a fair value at date of grant of approximately
$741,000.
28
During
2007, we issued 1,628,907 shares respectively, of Common Stock as a stock split
effected in the form of a stock dividend to our then current stockholders. The
additional shares represent splits at 25% for each year. The issuance of these
shares had no direct financial impact on our results of operations or financial
position and did not alter the market capitalization of our common shares
outstanding. All earnings per share amounts were restated to reflect
the splits.
In 2007,
the Company issued an aggregate of 336,700 shares of unregistered restricted
shares of Common Stock in reliance of Section 4(2) of the Securities Act of 1933
to Kush K. Agarwal and Brian D. McDonagh in connection with the Company’s
acquisition of Vein Clinics of America, Inc. on August 8, 2007. These shares had
a market value of $4 million on the date of issuance.
29
Performance
Graph
The
following graph compares the cumulative 5-year total return provided to
shareholders on IntegraMed America, Inc.'s common stock relative to the
cumulative total returns of the NASDAQ Composite index and the NASDAQ Health
Services index. An investment of $100 (with reinvestment of all dividends) is
assumed to have been made in our common stock and in each of the indexes on
December 31, 2004 and its relative performance is tracked through December 31,
2009.
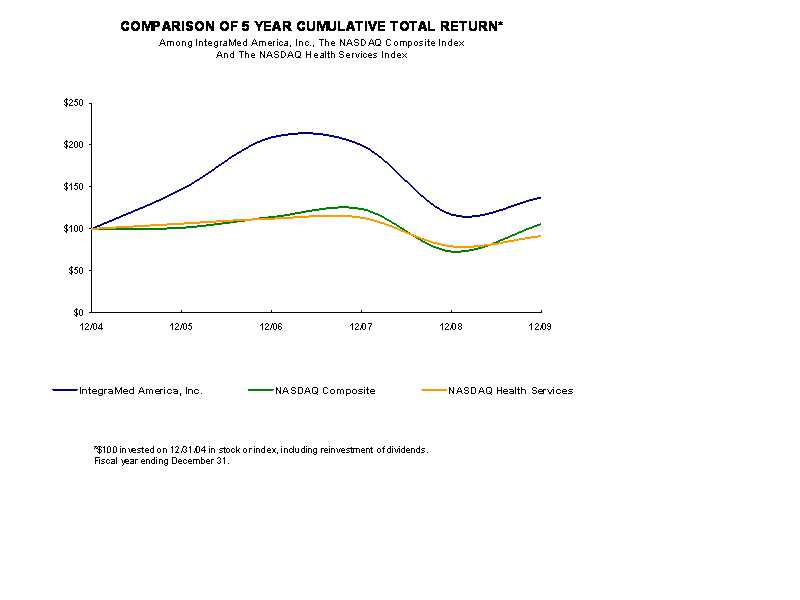
|
12/04
|
12/05
|
12/06
|
12/07
|
12/08
|
12/09
|
||||||
|
IntegraMed
America, Inc.
|
100.00
|
147.10
|
209.01
|
199.64
|
117.18
|
137.31
|
|||||
|
NASDAQ
Composite
|
100.00
|
101.33
|
114.01
|
123.71
|
73.11
|
105.61
|
|||||
|
NASDAQ
Health Services
|
100.00
|
106.30
|
112.25
|
113.33
|
79.24
|
91.44
|
|||||
The
stock price performance included in this graph is not necessarily indicative of
future stock price performance.
30
ITEM
6. Selected Financial Data -
The
following selected financial data (for the years ended December 31, 2009, 2008,
2007, 2006, and 2005) are derived from our consolidated financial statements and
should be read in conjunction with the financial statements, related notes, and
other financial information included elsewhere in this Annual Report on Form
10-K. Financial information for our Vein Clinics division is included only for
the period subsequent to its August 8, 2007 acquisition. Earnings per
share and average share values for the years 2006, 2005, and 2004 have been
restated to reflect the 25% stock split effected in the form of a stock dividend
declared in March 2007, the 25% stock split effected in the form of a stock
dividend declared in May 2006, and the 30% stock split effected in the form of a
stock dividend declared in May 2005.
Statement
of Operations Data:
|
Year
Ended December 31,
|
||||||||||||||||||||
|
2009
|
2008
|
2007
|
2006
|
2005
|
||||||||||||||||
|
(in
thousands, except per share amounts)
|
||||||||||||||||||||
|
Revenues,
net
|
$ | 216,760 | $ | 198,153 | $ | 151,822 | $ | 126,320 | $ | 128,961 | ||||||||||
|
Costs
of services incurred
|
195,870 | 179,867 | 136,699 | 113,778 | 116,396 | |||||||||||||||
|
Contribution
|
20,890 | 18,286 | 15,123 | 12,542 | 12,565 | |||||||||||||||
|
General
and administrative expenses
|
12,155 | 10,654 | 10,537 | 9,380 | 9,973 | |||||||||||||||
|
Total
other (income) expense, net
|
910 | 1,180 | (120 | ) | (378 | ) | (192 | ) | ||||||||||||
|
Income
before taxes
|
7,825 | 6,452 | 4,706 | 3,540 | 2,784 | |||||||||||||||
|
Provision
for income taxes
|
3,331 | 2,537 | 1,662 | 470 | 1,053 | |||||||||||||||
|
Net
income applicable to Common Stock
|
$ | 4,494 | $ | 3,915 | $ | 3,044 | $ | 3,070 | $ | 1,731 | ||||||||||
|
Basic
EPS
|
$ | 0.51 | $ | 0.45 | $ | 0.37 | $ | 0.38 | $ | 0.21 | ||||||||||
|
Diluted
EPS
|
$ | 0.51 | $ | 0.45 | $ | 0.36 | $ | 0.37 | $ | 0.21 | ||||||||||
|
Weighted
average shares - basic
|
8,773 | 8,618 | 8,310 | 8,090 | 8,090 | |||||||||||||||
|
Weighted
average shares - diluted
|
8,834 | 8,691 | 8,410 | 8,194 | 8,194 | |||||||||||||||
Balance
Sheet Data:
|
December
31,
|
||||||||||||||||||||
|
2009
|
2008
|
2007
|
2006
|
2005
|
||||||||||||||||
|
(in
thousands)
|
||||||||||||||||||||
|
Working
capital
(1)
|
$ | (2,703 | ) | $ | (2,447 | ) | $ | (3,435 | ) | $ | 11,685 | $ | 6,148 | |||||||
|
Total
assets
|
124,309 | 121,441 | 114,171 | 76,323 | 67,190 | |||||||||||||||
|
Total
indebtedness
|
26,166 | 30,219 | 25,460 | 8,774 | 10,147 | |||||||||||||||
|
Retained
Earnings (Accumulated deficit)
|
2,314 | (2,180 | ) | (6,095 | ) | (9,139 | ) | (12,209 | ) | |||||||||||
|
Total
shareholders' equity
|
58,193 | 52,264 | 47,634 | 40,178 | 36,298 | |||||||||||||||
|
(1)
|
Represents
current assets less current
liabilities.
|
31
ITEM
7. Management’s Discussion and Analysis of Financial Condition and
Results of Operations
The
following discussion and analysis should be read in conjunction with the
historical consolidated financial statements and related notes and the other
financial information appearing elsewhere in this report. This discussion and
analysis contains forward-looking statements that involve risks, uncertainties
and assumptions. Our actual results and timing of events could differ materially
from those anticipated in the forward-looking statements as a result of many
factors, including those discussed under the caption “Risk Factors” and
elsewhere in this report.
Forward
Looking Statements
This Form
10-K contains certain forward-looking statements regarding events and/or
anticipated results within the meaning of the "safe harbor" provisions of the
Private Securities Litigation Reform Act of 1995, the attainment of which
involves various risks and uncertainties. Forward-looking statements may be
identified by the use of forward-looking terminology such as, "may", "will",
"expect", "believe", "estimate", "anticipate", "continue", or similar terms,
variations of those terms or the negative of those terms. Our actual results may
differ materially from those described in these forward-looking statements due
to the following factors: our ability to acquire additional Fertility Partner
agreements or open additional vein clinics, our ability to raise additional debt
and/or equity capital to finance future growth, the loss of significant Partner
agreement(s), the profitability or lack thereof at fertility centers or vein
clinics serviced by us, increases in overhead due to expansion, the exclusion of
fertility services or vein care from insurance coverage, government laws and
regulation regarding health care, changes in managed care contracting, the
timely development of and acceptance of new fertility or vein treatment
technologies and techniques. We are under no obligation (and expressly disclaim
any such obligation) to update or alter any forward-looking statements whether
as a result of new information, future events or otherwise.
Overview
We manage
highly specialized outpatient centers in emerging, technology-based, niche
medical markets. Currently, we are a leading manager of fertility centers and
vein clinics in the United States. We provide services and products through our
three operating divisions (Fertility Centers, Consumer Services and Vein
Clinics) and shared support services for providers through our corporate
offices. Each of our operating divisions is presented as a separate segment for
financial reporting purposes.
Our
Fertility Centers Division is a provider network of 14 contracted fertility
centers, referred to as our Partner Program, serving 16 metropolitan
markets across the United States. We offer products and services to these
providers designed to support the fertility center’s growth. All fertility
Partners also have full access to our Consumer Services Division offerings. The
division also sponsors a Council of Physicians and Scientists for fertility
providers. Physicians affiliated with our Partner fertility centers obtain a
portion of their malpractice insurance through ARTIC — Assisted
Reproductive Technology Insurance Company, a captive insurance company which we
helped organize in 2005.
Our
Consumer Services Division offers our Attain IVF programs to fertility patients.
The division’s Attain IVF programs are designed to make the treatment process
easier and more affordable for patients. Currently, this division maintains a
contracted network of 25 independent fertility centers (22 as of
December 31, 2008) under its Affiliate Program, which is designed to
distribute the division’s products and services to a wider group of patients
than those serviced by our Partner locations.
Our Vein
Clinics Division began operations on August 8, 2007, with the purchase of
Vein Clinics of America, Inc. (“VCA”), a company that had been in business since
1981. The Vein Clinics Division currently manages a network of 34 clinics (32 as
of December 31, 2008) located in 13 states, which specialize in
the treatment of vein disease and other vein disorders.
32
The
primary elements of our business strategy include:
|
•
|
Making
selective contract acquisitions of Partner fertility
centers;
|
|
|
•
|
Expanding
our network of Affiliate fertility centers;
|
|
|
•
|
Developing
de novo vein clinics;
|
|
|
•
|
Increasing
the total number of patients treated;
|
|
|
•
|
Increasing
the penetration of our Attain IVF programs; and
|
|
|
•
|
Continuing
to improve operating efficiencies.
|
Major
Events Impacting Financial Condition and Results of Operations
Restatement
Of Revenue Recognition For Attain IVF Refund Program:
The
following information is presented here for historical reference and does not
appear in the financial statements in the 2009 Form 10-K as this information has
been previously presented on Form 10-K Amendment No. 3 for 2008 filed February
3, 2010 and on Form S-1 Amendment No. 2 filed February 8, 2010. These
restatements were the result of a material weakness in internal control over
financial reporting as of December 31, 2008. We have remediated this
material weakness.
Restatement No. 2 – The
Correction of an Error
On
October 28, 2009, management concluded and subsequently reported to the audit
committee of our board of directors that our audited consolidated financial
statements as of and for the years ended December 31, 2006, 2007, and 2008
should no longer be relied upon and will be restated due to an understatement in
revenue recognized in connection with its Attain IVF Refund
Program.
We had
previously restated (see Restatement No. 1- Correction of an Error in Revenue
Recognition Policy for the Attain IVF Refund Program below) certain financial
statements after determining that they could not be relied
upon. Specifically, we restated our prior financial statements
with respect to the correction of an error in the timing of revenue and profit
recognition of $3,477,000 of the revenue related to our Attain IVF Refund
Program within our Consumer Services Division for the years 2001 through
2008. That restatement did not impact the cash flows from operations
of this program or the ultimate profits to be recognized, only the timing of the
revenue and profit recognition.
Subsequent
to Restatement No. 1, we developed, programmed and tested a new patient
management and revenue recognition system for our Attain IVF Refund
Program. The new system recently reached sufficient operating
proficiency to allow input of the patient information related to our Attain IVF
Refund Program. As a result, we identified that the deferred revenue
amount of $3,477,000, related to the years 2001 through 2008, that had been
previously restated should have been only $822,000. We determined
that while the previous system properly accounted for the recognition of the
proportional fair value revenue related to the non-refundable portion of the
patient fee (which was the subject of a comment and review process by the Staff
of the Division of Corporation Finance of the Securities and Exchange Commission
(SEC)), it assumed all patients would either achieve pregnancy or utilize all
services available to them under the program. It is a fact that most
of the patients that seek a refund, do so prior to utilizing all services
available under the program. As a result, our revenue recognition
model failed to recognize as revenue the remaining deferred revenue portion of
the non-refundable fee on that class of patients.
The
change in our Consolidated Balance Sheet and Consolidated Statement
of Operations as of and for the twelve months ended December 31, 2008 and
2007 related to Restatement No. 2 is presented below (000’s, except per
share amounts):
33
|
Twelve Months
Ending
December
31,
|
||||||||
|
2008
|
2007
|
|||||||
|
Revenue
recognized from prior period
|
$
|
750
|
$
|
1,406
|
||||
|
Revenue
deferred to future period
|
0
|
(750
|
)
|
|||||
|
Net
change in period revenue
|
$
|
750
|
$
|
656
|
||||
|
Revenue
as reported
|
$
|
197,403
|
$
|
151,166
|
||||
|
Net
change in reported revenue
|
750
|
656
|
||||||
|
Revenue
as restated
|
$
|
198,153
|
$
|
151,822
|
||||
|
Income
before income taxes as reported
|
$
|
5,715
|
$
|
4,062
|
||||
|
Net
change in reported revenue
|
750
|
656
|
||||||
|
Net
change in reserve for medical costs
|
(13
|
)
|
(12
|
)
|
||||
|
Income
before income taxes as restated
|
$
|
6,452
|
$
|
4,706
|
||||
|
Income
tax provision as reported
|
$
|
2,227
|
$
|
1,391
|
||||
|
Net
change in income taxes from above adjustments
|
310
|
271
|
||||||
|
Income
tax provision as restated
|
$
|
2,537
|
$
|
1,662
|
||||
|
Net
Income as reported
|
$
|
3,488
|
$
|
2,670
|
||||
|
Summary
of above adjustments
|
427
|
374
|
||||||
|
Net
income as restated
|
$
|
3,915
|
$
|
3,044
|
||||
|
Diluted
earnings per share as reported
|
$
|
0.40
|
$
|
0.32
|
||||
|
Change
in earnings per share from above adjustments
|
$
|
0.05
|
$
|
0.04
|
||||
|
Diluted
earnings per share as restated
|
$
|
0.45
|
$
|
0.36
|
||||
|
Current
liabilities as reported
|
$
|
51,126
|
$
|
44,005
|
||||
|
Cumulative
effect of restatement on current liabilities
|
(1,513
|
)
|
(1,086
|
)
|
||||
|
Current
liabilities as restated
|
$
|
49,613
|
$
|
42,919
|
||||
|
Shareholders
Equity as reported
|
$
|
50,753
|
$
|
46,549
|
||||
|
Cumulative
effect of restatement on Shareholders Equity
|
1,511
|
1,085
|
||||||
|
Shareholders
Equity as restated
|
$
|
52,264
|
$
|
47,634
|
||||
There was
no change to cash flow as a result of this restatement.
34
Restatement No. 1 –
Correction of an Error in Revenue Recognition Policy for the Attain IVF
Program.
The
accompanying 2008 and 2007 consolidated financial statements have been restated
to reflect the correction of an error in the revenue recognition policy for our
Attain IVF Refund Program. Our previous revenue recognition policy
had generally recognized the non-refundable patient fees (generally 30% of the
contract amount) as revenue upon the completion of the first treatment cycle and
we now recognize the non-refundable fees based on the relationship of the
relative fair value of each treatment to the total fair value of the treatment
package available to each patient. We also recognize a “warranty
reserve” representing the estimated cost of services to be provided in the event
a qualified patient miscarries. This restatement does not impact the
cash flows from the operations of this program or the ultimate profits to be
recognized, only the timing of the revenue recognition for a portion of the fees
that we collect from our customers.
The
impact of the correction of the error in the timing of revenue recognition
for Restatement No. 1 only, is as follows:
|
2008
|
2007
|
|||||||
|
Revenue
as reported
|
$
|
198,084
|
$
|
151,998
|
||||
|
Net
Change in reported revenue
|
(681
|
)
|
(832
|
)
|
||||
|
Revenue
as restated
|
$
|
197,403
|
$
|
151,166
|
||||
|
Income
before income taxes as reported
|
$
|
6,454
|
$
|
4,952
|
||||
|
Net
change in reported revenue
|
(681
|
)
|
(832
|
)
|
||||
|
Net
change in reserve for medical costs
|
(58
|
)
|
(58
|
)
|
||||
|
Income
before income taxes as restated
|
$
|
5,715
|
$
|
4,062
|
||||
|
Income
tax provision as reported
|
$
|
2,514
|
$
|
1,695
|
||||
|
Net
change in income taxes from above adjustments
|
(287
|
)
|
(304
|
)
|
||||
|
Income
tax provision as restated
|
$
|
2,227
|
$
|
1,391
|
||||
|
Net
income as reported
|
$
|
3,940
|
$
|
3,257
|
||||
|
Summary
of above adjustments
|
(452
|
)
|
(586
|
)
|
||||
|
Net
income as restated
|
$
|
3,488
|
$
|
2,671
|
||||
|
Diluted
earnings per share as reported
|
$
|
0.45
|
$
|
0.39
|
||||
|
Change
in earnings per share from above adjustments
|
(0.05
|
)
|
(0.07
|
)
|
||||
|
Diluted
earnings per share as restated
|
$
|
0.40
|
$
|
0.32
|
||||
|
Current
liabilities as reported
|
$
|
47,329
|
$
|
40,946
|
||||
|
Cumulative
effect of restatement on liabilities
|
3,797
|
3,059
|
||||||
|
Current
liabilities as restated
|
$
|
51,126
|
$
|
44,005
|
||||
|
Shareholders’
Equity as reported
|
$
|
53,158
|
$
|
48,503
|
||||
|
Cumulative
effect of restatement on Shareholders’ Equity
|
(2,405
|
)
|
(1,954
|
)
|
||||
|
Shareholders’
Equity as restated
|
$
|
50,753
|
$
|
46,549
|
||||
The Cumulative Impact of
Both Restatements
The
cumulative effect of the corrections of errors is to decrease net income in 2008
and 2007 by ($25,000) and ($213,000), respectively. The change in our
financial statements for the years ended December 31, 2008 and 2007 for the
effects of both restatements is illustrated below (000’s).
35
|
Twelve Months
Ending
December
31,
|
||||||||
|
2008
|
2007
|
|||||||
|
Net
Revenue as initially recognized from prior period, first
restatement
|
$
|
2,796
|
$
|
1,964
|
||||
|
Revenue
deferred to future period, first restatement
|
(3,477
|
)
|
(2,796)
|
|||||
|
Revenue
initially recognized from prior period, second restatement
|
750
|
1,406
|
||||||
|
Revenue
deferred to future period, second restatement
|
—
|
(750)
|
||||||
|
Net
change in period revenue
|
$
|
69
|
$
|
(176)
|
||||
|
Revenue
as initially reported
|
$
|
198,084
|
$
|
151,998
|
||||
|
Net
change in reported revenue, first restatement
|
(681
|
)
|
(832)
|
|||||
|
Net
change in reported revenue, second restatement
|
750
|
656
|
||||||
|
Revenue
as restated
|
$
|
198,153
|
$
|
151,822
|
||||
|
Income
before income taxes as initially reported
|
$
|
6,454
|
$
|
4,952
|
||||
|
Net
change in reported revenue, first restatement
|
(681
|
)
|
(832)
|
|||||
|
Net
change in reserve for medical costs, first restatement
|
(58
|
)
|
(58)
|
|||||
|
Net
change in reported revenue, second restatement
|
750
|
656
|
||||||
|
Net
change in reserve for medical costs, second restatement
|
(13
|
)
|
(12)
|
|||||
|
Income
before income taxes as restated
|
$
|
6,452
|
$
|
4,706
|
||||
|
Income
tax provision as initially reported
|
$
|
2,514
|
$
|
1,695
|
||||
|
Net
change in income taxes from above adjustments, first
restatement
|
(287
|
)
|
(304)
|
|||||
|
Net
change in income taxes from above adjustments, second
restatement
|
310
|
271
|
||||||
|
Income
tax provision as restated
|
$
|
2,537
|
$
|
1,662
|
||||
|
Net
income as initially reported
|
$
|
3,940
|
$
|
3,257
|
||||
|
Summary
of above adjustments, first restatement
|
(452
|
)
|
(586)
|
|||||
|
Summary
of above adjustments, second restatement
|
427
|
373
|
||||||
|
Net
income as restated
|
$
|
3,915
|
$
|
3,044
|
||||
|
Diluted
earnings per share as initially reported
|
$
|
0.45
|
$
|
0.39
|
||||
|
Change
in Earnings per share from above adjustments, first
restatement
|
(0.05
|
)
|
(0.07)
|
|||||
|
Change
in earnings per share from above adjustments, second
restatement
|
0.05
|
0.04
|
||||||
|
Diluted
earnings per share as restated
|
$
|
0.45
|
$
|
0.36
|
||||
|
Current
liabilities as initially reported
|
$
|
47,329
|
$
|
40,946
|
||||
|
Cumulative
effect of restatement on current liabilities, first
restatement
|
3,797
|
3,059
|
||||||
|
Cumulative
effect of restatement on current liabilities, second
restatement
|
(1,513
|
)
|
(1,086)
|
|||||
|
Current
liabilities as restated
|
$
|
49,613
|
$
|
42,919
|
||||
|
Shareholders
Equity as initially reported
|
$
|
53,158
|
$
|
48,503
|
||||
|
Cumulative
effect of restatement on Shareholders Equity, first
restatement
|
(2,405
|
)
|
(1,954)
|
|||||
|
Cumulative
effect of restatement on Shareholders Equity, second
restatement
|
1,511
|
1,085
|
||||||
|
Shareholders
Equity as restated
|
$
|
52,264
|
$
|
47,634
|
||||
Our new
revenue recognition policy, as more fully explained in Note 2 to our Form 10-K –
“Summary of Significant Accounting Policies” changes the timing of revenue
recognition for non-refundable fees, and it aligns it more closely to the
underlying treatment cycles delivered to the patients.
36
The
financial statements, schedules and related footnotes reflect included herein
reflect these restatements.
2009
On
December 1, 2009, we acquired the rights to supply a complete range of business,
marketing and facility services to three new Partner fertility centers in the
western United States; the Idaho Center for Reproductive Medicine, the Nevada
Center for Reproductive Medicine and the Utah Fertility Center, based in Boise,
Idaho, Reno, Nevada and Prove, Utah respectively. The Idaho and Nevada fertility
centers are established centers with the Utah center scheduled to begin seeing
patients in early 2010. Under the terms of these 25-year agreements, our service
fees are comprised of a fixed percentage of revenues, reimbursed costs of
services, and an additional fixed percentage of each center’s earnings. We also
committed up to $1.0 million to fund any necessary capital needs of the
practices.
On
October 28, 2009, management concluded and subsequently reported to the audit
committee of our board of directors that our audited consolidated financial
statements as of December 31, 2007 and 2008 and for the years ended December 31,
2006, 2007 and 2008 should no longer be relied upon and should be restated for
the correction of errors due to an understatement in revenue recognized in
connection with our Attain IVF Refund Program (formerly our Shared Risk Refund
Program). As a result, we restated our audited consolidated financial
statements as of December 31, 2007 and 2008 and for the years ended December 31,
2006, 2007 and 2008 with respect to the revenue recognized for our Attain IVF
Refund Program within our Consumer Services Division. The financial
data included in this Form 10-K reflects this restatement.
On
April 20, 2009, we announced the opening of a new vein clinic in Cleveland,
Ohio. This represents the 34th clinic in our Vein Clinics Division, our
entry into the Cleveland market and the expansion of our presence in the State
of Ohio.
On
April 1, 2009, we elected to exercise the option contained in our business
service agreement with Arizona Reproductive Medicine Specialists, based in
Phoenix, Arizona, and expand our service offerings from a limited range of
services to those offered to our other fertility Partners.
On
January 20, 2009, we announced the opening of a new vein clinic in
Cincinnati, Ohio. This represents the 33rd clinic in our Vein Clinics
Division and our first entry into the State of Ohio and the Cincinnati
market.
2008
From June
2008 through March 2009, our 2007 annual and our 2008 periodic interim
Securities and Exchange Commission reports were the subject of a standard
comment and review process by the Staff of the Division of Corporation Finance
of the Securities and Exchange Commission. The application of generally accepted
accounting principles to our Attain IVF Refund Program’s multiple element
revenue arrangements is complex and management’s interpretation of the
applicable authoritative literature related to the timing of the recognition of
the fair value of revenues for the non-refundable portion of the Attain IVF
Refund Program fees differed from that of the Securities and Exchange
Commission, which caused us to re-evaluate our revenue recognition policies. As
a result, we restated our prior financial statements for the correction of an
error with respect to the timing of revenue recognition for our Attain IVF
Refund Program within our Consumer Services Division. Our previous revenue
recognition policy had generally recognized the non-refundable patient fees
(generally 30% of the contract amount) as revenues upon the completion of the
first treatment cycle. We now recognize the non-refundable fees based on the
relationship of the fair value of each treatment to the total fair value of the
treatment package available to each patient. We also recognize a “warranty
reserve” representing the estimated cost of services to be provided in the event
a qualified patient miscarries, as well as a reserve for potential refunds
should a patient elect to discontinue participation in the program prior to full
treatment. This restatement does not impact our cash flows from operations or
the ultimate profits from our Attain IVF Refund Program, only the timing of the
revenue recognition for the non-refundable portion of the Attain IVF Refund
Program fees paid by patients. The financial data included in this
Form 10-K reflects this restatement.
37
On
December 17, 2008, we announced the opening of a new vein clinic in Skokie,
Illinois. This clinic represents our ninth vein care clinic in the greater
Chicago metropolitan area and benefits from the operational and marketing
leverage we have developed in that market.
On
December 8, 2008, we announced the opening of a new vein clinic in
Monroeville, Pennsylvania. This clinic is our first vein clinic in Pennsylvania
and is designed to provide state-of-the-art vein care to patients in the greater
Pittsburgh area.
On
July 9, 2008, we entered into a business services agreement to provide
discrete business services to Arizona Reproductive Medicine Specialists, based
in Phoenix, Arizona. Under the terms of this 25-year agreement, our service fees
were initially comprised of a fixed percentage of the fertility practice’s net
revenues. We also had the exclusive option, which we exercised on April 1,
2009, at any point during the life of the contract to expand our service
offerings into a complete range of business, marketing and financial services.
After we exercised the option on April 1, 2009, our fees also included a
fixed percentage of the fertility practice’s earnings.
On
June 23, 2008, we announced that we entered into a new Affiliate services
contract with the University of North Carolina (“UNC”) School of Medicine’s
Department of Obstetrics and Gynecology in Chapel Hill, North Carolina. As an
Affiliate, UNC School of Medicine’s Department of Obstetrics and Gynecology
receives distribution rights to our consumer products and services. In addition,
UNC School of Medicine’s Department of Obstetrics and Gynecology has the
right to receive other products and services uniquely designed to support the
business needs of successful, high-growth fertility centers.
On
June 5, 2008, we announced the opening of a new vein clinic in Marietta,
Georgia. This clinic was our fourth vein clinic in Georgia.
On
April 29, 2008, we announced the opening of a new vein clinic in
Alexandria, Virginia. This addition to our Vein Clinics Division provides
focused vein care treatment solutions to the Washington, D.C. metropolitan
area.
On
April 24, 2008, we entered into a business service agreement to supply a
complete range of business, marketing and facility services to Southeastern
Fertility Centers, P.A., located in Mount Pleasant, South Carolina. Under
the terms of this 25-year agreement, our service fees are comprised of
reimbursed costs of services, a tiered percentage of revenues and an additional
fixed percentage of the practice’s earnings. We also committed up to $600,000 to
fund any necessary capital needs of the practice.
On
April 1, 2008, we entered into an Affiliate services contract with OU
Physicians Reproductive Health in Oklahoma City, Oklahoma. As a result of this
agreement, OU Physicians Reproductive Health provides another opportunity for
our Consumer Services Division to distribute its product offerings.
2007
On
August 30, 2007, we entered into a business service agreement to supply a
complete range of business, marketing and facility services to the Center for
Reproductive Medicine in Orlando, Florida. The Center for Reproductive Medicine
is a fertility practice comprised of four physicians. Under the terms of this
25-year agreement, our service fees are comprised of reimbursed costs of
services, a tiered percentage of revenues and an additional fixed percentage of
the Center for Reproductive Medicine’s earnings. We also committed up to
$1.0 million to fund any necessary capital needs of the
practice.
On
August 8, 2007, we acquired all of the outstanding stock of VCA for a total
cost of approximately $29 million in cash and common stock. The results of
VCA are included in our financial statements from the date of the
acquisition.
38
Also on
August 8, 2007, we entered into an amended credit agreement with Bank of
America, N.A. (“Bank of America”). The new term loan under the amended credit
agreement is in the amount of $25 million (the proceeds of which were
applied to repay our original term loan and finance, in part, the VCA
transaction). Interest on the new term loan is, at our option, at the prime rate
less up to 0.50% or at LIBOR plus 2.00% to 2.75%, depending upon the level of
the ratio of consolidated debt to earnings before interest, taxes depreciation
and amortization (“EBITDA”). The amended credit agreement also contains
provisions for a revolving line of credit in the amount of $10 million.
Interest on the revolving line of credit is at the prime rate less up to 0.50%
or at LIBOR plus 1.5% to 2.5%, depending on the level of the ratio of
consolidated debt to EBITDA. Effective July 1, 2007, we expanded our
fertility center Partner service arrangement with Shady Grove Fertility
Reproductive Science Center, P.C. (“Shady Grove”) with the addition of the
Fertility Center of the Greater Baltimore Medical Center (the “Center”) in
Baltimore, Maryland, where we now provide a full range of business, marketing
and financial services. Under the terms of this agreement, we purchased the
assets of the Center from Greater Baltimore Medical Center and have committed
additional resources to support further growth and development of the Center.
Under the terms of this agreement, we are paid service fees comprised of
reimbursed costs of services and a fixed percentage of revenues, plus an
additional fixed amount of the Center’s earnings.
Effective
July 1, 2007, we expanded our fertility center Partner service arrangement
with Shady Grove Fertility Reproductive Science Center, P.C. (“Shady
Grove”) with the addition of the Fertility Center of the Greater Baltimore
Medical Center (the “Center”) in Baltimore, Maryland, where we now provide a
full range of business, marketing and financial services. Under the terms of
this agreement, we purchased the assets of the Center from Greater Baltimore
Medical Center and have committed additional resources to support further growth
and development of the Center. Under the terms of this agreement, we are paid
service fees comprised of reimbursed costs of services and a fixed percentage of
revenues, plus an additional fixed amount of the Center’s earnings.
On
March 19, 2007, we declared a 25% common stock split effected in the form
of a common stock dividend for all holders of record as of April 13, 2007.
As a result of this dividend, 1,628,907 new shares of common stock were issued
on the payment date of May 4, 2007. No fractional shares were issued as all
fractional amounts were rounded up to the next whole share. All weighted average
shares outstanding and earnings per share calculations in this Form 10-K have
been restated to reflect this common stock dividend.
Subsequent
Events
On
February 12, 2010, we announced that we priced a public offering of 2,800,000
(including the over allotment) shares of common stock at a price to the public
of $7.50 per share, of which 2,300,000 shares were offered through Piper Jaffray
& Co., which acted as book-running manager and Dougherty & Company LLC,
which acted as co-manager; and 500,000 shares were sold directly to IAT
Reinsurance Company Ltd., our largest stockholder.
Approximately
$19 million of net proceeds, after deducting underwriting discounts, commissions
and estimated offering expenses, are intended to be used to accelerate the
addition of new partner fertility centers, accelerate the pace of new vein
clinic openings in 2010, and for general working capital and other corporate
purposes. The offering closed on February 18, 2010.
On
February 4, 2010, we announced the addition of Tennessee Reproductive Medicine
as an affiliate and that they will begin offering our Attain™ IVF
programs.
On
January 12, 2010, we announced plans to open a new vein clinic in Chevy
Chase, Maryland in early May 2010. This will be the 36th clinic in our Vein
Clinics Division and our seventh clinic in the greater Baltimore/Washington D.C.
region.
On
January 8, 2010, we announced plans to open a new vein clinic in Columbia,
Maryland in mid-2010. This will be the 35th clinic in our Vein Clinics
Division and will add interventional radiology treatments to the full range of
vein treatments provided at our existing vein clinics, enabling patients to
undergo a host of additional procedures. Interventional radiology involves
minimally invasive procedures performed using image guidance. Adding
interventional radiology will allow this vein clinic to offer patients more high
value and complex vascular procedures including uterine fibroid embolization, as
well as procedures for varicoceles and pelvic congestion, among
others.
39
Significant
Accounting Policies and Use of Estimates
Our
significant accounting policies are described in Note 2 of our consolidated
financial statements included elsewhere in this Form 10-K.
The
preparation of financial statements in conformity with accounting principles
generally accepted in the United States, including our significant accounting
policies, requires management to make estimates and assumptions that affect the
reported amounts of assets and liabilities and disclosure of contingent assets
and liabilities at the date of the financial statements and the reported amounts
of revenues and expenses during the reporting periods. On an ongoing basis, we
evaluate our estimates and assumptions, including those related to revenue
recognition, allowance for uncollectible accounts and contractual allowance
reserves, contingencies and income taxes. We base our estimates on historical
experience and on various other assumptions that we believe are reasonable under
the circumstances. The results of our analysis form the basis for making
assumptions about the carrying values of assets and liabilities that are not
readily apparent from other sources. Actual results may differ from these
estimates under different assumptions or conditions, and the impact of such
differences may be material to our consolidated financial statements. The most
significant use of estimates and assumptions in the preparation of our
consolidated financial statements relates to the determination of net revenues
and accounts receivable and reserves for estimated refunds due to pregnancy
losses in our Attain IVF Refund Program.
Contractual
allowance and uncollectible reserve amounts are determined based on historical
collection performance data and are reviewed and adjusted monthly as necessary.
We make periodic estimates for pregnancy loss based upon Company specific
data.
Results
of Operations
The
following table shows the percentage of net revenues represented by various
expenses and other income items reflected in our statements of operations for
the years ended December 31, 2009, 2008 and 2007:
|
2009
|
2008
|
2007
|
|||
|
Revenues,
net:
|
|||||
|
Fertility
Centers
|
67.0%
|
69.9%
|
79.8%
|
||
|
Consumer
Services
|
9.6%
|
10.0%
|
10.8%
|
||
|
Vein Clinics
|
23.4%
|
20.1%
|
9.4%
|
||
|
Total revenues
|
100.0%
|
100.0%
|
100.0%
|
||
|
Costs
of services incurred:
|
|||||
|
Fertility
Centers
|
61.7%
|
64.7%
|
73.2%
|
||
|
Consumer
Services
|
7.2%
|
7.3%
|
8.1%
|
||
|
Vein Clinics
|
21.5%
|
18.8%
|
8.7%
|
||
|
Total costs of
service
|
90.4%
|
90.8%
|
90.0%
|
||
|
Contribution:
|
|||||
|
Fertility
Centers
|
5.3%
|
5.2%
|
6.6%
|
||
|
Consumer
Services
|
2.4%
|
2.7%
|
2.7%
|
||
|
Vein Clinics
|
1.9%
|
1.3%
|
0.7%
|
||
|
Total
contribution
|
9.6%
|
9.2%
|
10.0%
|
||
|
General
and administrative expenses
|
5.6%
|
5.4%
|
6.9%
|
||
|
Interest
income
|
(.1)%
|
(.2)%
|
(.8)%
|
||
|
Interest
expense
|
.5%
|
.8%
|
.8%
|
||
|
Total other
expenses
|
6.0%
|
6.0%
|
6.9%
|
||
|
Income
from operations before income taxes
|
3.6%
|
3.2%
|
3.1%
|
||
|
Income
tax provision
|
1.5%
|
1.2%
|
1.1%
|
||
|
Net
income
|
2.1%
|
2.0%
|
2.0%
|
(1) Our
Vein Clinics Division began operations on August 8, 2007 with our purchase
of VCA.
40
Year
Ended December 31, 2009 as Compared to Year Ended December 31, 2009
For the
year ended December 31, 2009, total revenues of $216.8 million increased
approximately $18.6 million, or 9.4%, from the same period in 2008.
Approximately $10.6 million of this increase was generated by our Vein
Clinics Division, $6.9 million from our Fertility Centers Division and
$1.1 million from our Consumer Services Division. The increase in revenues
in each of our divisions is attributed to the following:
In our
Fertility Division: The increase was the result of a $2.8 million rise in
same-center revenues as well as $4.1 million of incremental revenues from
the addition of new Partner contracts in April, July 2008 and December
2009.
In Our
Consumer Division: The increase of $1.1 million is due primarily to a
combination of increased cycle volumes, slightly higher success rates in 2009 as
compared to 2008.
In our
Vein Clinics Division: Two new vein clinics opened during the first quarter of
2009, which, accounted for $1.7 million of our Vein Clinics Division’s
increase, the five clinics opened throughout 2008 accounted for $6.1 million of
the increase with the remaining $2.8 million generated by legacy
clinics.
For the
year ended December 31, 2009, contribution increased from $18.3 million in the
comparable period of 2008 to $20.9 million in 2009, or an increase of 14.2%,
driven primarily by growth in our Fertility Centers and Vein Clinics
divisions.
Year
Ended December 31, 2008 as Compared to Year Ended December 31, 2007
For the
year ended December 31, 2008, total revenues of $198.2 million
increased approximately $46.3 million, or 30.5%, from the year ended
December 31, 2007. We experienced year-over-year organic revenue increases
in both of our fertility business segments. Our Fertility Centers Division
revenues increased as a result of growth within our existing medical practices,
as well as the addition of two new Partner arrangements and the full year of
results of a Partner added in the third quarter of 2007. Expansion continued in
our Consumer Services Division, driven by the continued expansion of our Attain
IVF Refund Program. In addition to growth in our two fertility segments, our
performance for the year ended December 31, 2008 included full year results
from our Vein Clinics Division which was purchased on August 8,
2007.
For the
year ended December 31, 2008, total contribution of $18.3 million was
up approximately $3.2 million, or 20.9%, from the year ended
December 31, 2007. Increased contribution in our Fertility Centers Division
in 2008 was the result of increased profitability in our platform of existing
centers as well as the addition of contract acquisition related results. The
continued growth of our Attain IVF Refund Program and full year results from our
Vein Clinics Division were also major contributors to the
improvement.
|
|
A
Segment-by-Segment Discussion is Presented
Below:
|
Fertility
Centers Segment
In
providing clinical care to patients, each of our Partner fertility centers
generates patient revenues which we do not report in our financial statements.
Although we do not consolidate the Partner fertility center practice financials
with our own, these financials do directly affect our revenues.
The
components of our revenues from most of the Partner fertility centers
are:
|
•
|
A
base service fee calculated as a percentage of patient revenues as
reported by the Partner fertility center (this percentage generally varies
from 6% down to 3% depending on the agreement and the level of patient
revenues);
|
|
|
•
|
Cost
of services equal to reimbursement for the expenses which we advanced to
the Partner fertility center during the month (representing substantially
all of the expenses incurred by the center, except physician
compensation); and
|
|
|
•
|
Our
additional fees which represent our share of the net income of the Partner
fertility center (which varies from 10% to 20% or a fixed amount depending
on the underlying center, subject to limits in some
circumstances).
|
41
However,
our revenues from our Fertility Centers of Illinois, S.C. (“FCI”) Partner
fertility center are not based on this three-part structure. Rather, effective
as of November 1, 2009, our revenues from FCI are generally equal to the
operating expenses associated with managing FCI’s medical practice plus 9.5% of
such expenses. Our revenues from FCI prior to November 1, 2009 were,
pursuant to our current Partner agreement with FCI, set at a fixed annual amount
paid monthly.
In
addition to these revenues generated from our fertility centers, we often
receive miscellaneous other revenues related to providing services to medical
practices. From the total of our revenues, we subtract the annual amortization
of our business service rights under most agreements, which are the rights to
provide business services to each of the centers.
During
2009, our Fertility Centers Division revenues increased by $6.9 million, or
5.0%, relative to the same period in the prior year. This increase was the
result of a $2.8 million rise in same-center revenues as well as
$4.1 million of incremental revenues from the addition of new Partner
contracts in April 2008, July 2008 and December 2009. These increases are net of
the reduction in business at one of our top fertility centers in the Midwest as
a result of termination of a contract with one of the center’s third-party
payors, as well as a slight moderation in demand that we believe is attributable
to the prolonged recession. Contribution from this facility in 2008 was
approximately $2.3 million and this third-party payor represented
approximately 20% of this contribution or approximately $460,000.
Fertility
Centers Division contribution for the year ended December 31, 2009 rose $1.4
million, or 13.6%, from the prior year period based on a 5.0% rise in revenues.
During 2009, $0.3 million of contribution was attributable to our new Partner
Fertility centers and $1.1 million was attributable to our legacy centers. The
rise in contribution, along with improved operating margins, are the result of
division wide cost control measures implemented early in 2009.
Fertility
Centers Division revenues in the year ended December 31, 2008 increased by
$17.4 million, or 14.3%, from the year ended December 31, 2007. This
compares to an increase of $8.3 million, or 7.4%, for the year ended
December 31, 2007 versus the year ended December 31, 2006. During 2008
and 2007, growth was largely attributable to same center year-over-year growth
in our network of underlying medical practices. Influencing this growth is our
focus on increasing patient revenues through effective multi-faceted marketing
programs, as well as our continued focus on expense management, which drives
operational efficiency and higher contribution margins. Revenues for the year
ended December 31, 2008 also benefited from:
|
•
|
the
inclusion of a new fertility Partner in Mount Pleasant, South Carolina,
which contributed $3.5 million to our net revenues from its addition
in April 2008 through December 31, 2008;
|
|
|
•
|
full
year results from our Orlando, Florida Partner added in September
2007; and
|
|
|
•
|
the
full year impact from the expansion of Shady Grove into the Baltimore,
Maryland market in July 2007.
|
In July
2008, we also entered into a 25-year contract with Arizona Reproductive Medicine
Specialists, based in Phoenix, Arizona. Under the terms of this agreement, we
phased in the implementation of our full suite of Partner services over time.
Under this arrangement, our fees were originally calculated as a fixed
percentage of the center’s revenues with an option to expand into our standard
three-tiered fee structure, in line with expanded Partner services, based upon
the center meeting certain performance targets. We exercised this option on
April 1, 2009.
Fertility
Centers Division contribution of $10.2 million for the year ended
December 31, 2008 increased approximately $200,000, or 2.0%, from prior
year levels. Although this segment experienced revenue growth of 14.3% in 2008,
versus the prior year, margin growth was tempered by additional division level
infrastructure investments. These investments, which totaled $1.2 million
during 2008, were designed to support continuing growth and new contract
acquisitions within this segment.
42
The table
below illustrates the components of the Fertility Centers Division revenues in
relation to the Partner fertility center practice financials for the years ended
December 31, 2009, 2008 and 2007 (in thousands):
|
Year
ended December 31,
|
|||||||||||||
|
2009
|
2008
|
2007
|
|||||||||||
|
Partner Fertility Center
Financials
|
|||||||||||||
|
(a)
|
Patient
revenue
|
$ | 203,898 | $ | 192,380 | $ | 168,653 | ||||||
|
(b)
|
Cost
of services
|
130,615 | 125,156 | 109,132 | |||||||||
|
(c)
|
Base
service fee
|
9,562 | 8,798 | 7,791 | |||||||||
|
(d)
|
Practice
contribution (a-b-c)
|
63,721 | 58,426 | 51,730 | |||||||||
|
(e)
|
Physician
compensation
|
57,419 | 52,863 | 46,678 | |||||||||
|
(f)
|
IntegraMed
additional fee
|
6,302 | 5,563 | 5,052 | |||||||||
|
IntegraMed Financials
|
|||||||||||||
|
(g)
|
IntegraMed
gross revenue (b+c+f)
|
146,479 | 139,517 | 121,975 | |||||||||
|
(h)
|
Amortization
of business service rights
|
(1,295 | ) | (1,300 | ) | (1,343 | ) | ||||||
|
(i)
|
Other
revenue
|
125 | 223 | 446 | |||||||||
|
(j)
|
IntegraMed
fertility services revenue (g+h+i)
|
$ | 145,309 | $ | 138,440 | $ | 121,078 | ||||||
(i) Other
revenue includes administrative fees we receive from ARTIC, the captive
insurance company, fees from Arizona Reproductive Medicine Specialists as well
as other miscellaneous fees.
The
following summarized quarterly data for the years ended December 31, 2009,
2008 and 2007 is presented for additional analysis and demonstration of the
slight seasonality of our Fertility Centers Division. New patients visits are an
indicator of initial patient interest in fertility treatment and IVF cases
completed are an indicator of billable charges. IVF cases completed in the
fourth quarter of each year are typically lower, as many patients do not wish to
undergo the IVF procedure during the year end holidays. Contributing to the
lower number of IVF cases completed are voluntary laboratory closures at year
end at several of our labs in order to undergo normal maintenance (in thousands,
except new patient visits and IVF cases completed).
|
Period
Ended December 31, 2009
|
||||||||||||||||||||
| Q1 | Q2 | Q3 | Q4 |
Total
|
||||||||||||||||
|
Revenues,
Net
|
$ | 36,284 | $ | 37,290 | $ | 35,964 | $ | 35,771 | $ | 145,309 | ||||||||||
|
Contribution
|
$ | 2,640 | $ | 3,057 | $ | 2,979 | $ | 2,927 | $ | 11,603 | ||||||||||
|
New
Patient Visits
|
7,554 | 7,089 | 7,063 | 6,460 | 28,166 | |||||||||||||||
|
IVF
Cases Completed
|
3,533 | 3,547 | 3,424 | 3,231 | 13,735 | |||||||||||||||
|
Period
Ended December 31, 2008
|
||||||||||||||||||||
| Q1 | Q2 | Q3 | Q4 |
Total
|
||||||||||||||||
|
Revenues,
Net
|
$ | 32,746 | $ | 35,051 | $ | 36,505 | $ | 34,138 | $ | 138,440 | ||||||||||
|
Contribution
|
$ | 2,304 | $ | 2,570 | $ | 2,743 | $ | 2,599 | $ | 10,216 | ||||||||||
|
New
Patient Visits
|
6,765 | 7,093 | 7,186 | 7,173 | 28,217 | |||||||||||||||
|
IVF
Cases Completed
|
3,141 | 3,314 | 3,474 | 3,219 | 13,148 | |||||||||||||||
|
Period
Ended December 31, 2007
|
||||||||||||||||||||
| Q1 | Q2 | Q3 | Q4 |
Total
|
||||||||||||||||
|
Revenues,
Net
|
$ | 29,092 | $ | 29,728 | $ | 31,046 | $ | 31,212 | $ | 121,078 | ||||||||||
|
Contribution
|
$ | 2,315 | $ | 2,526 | $ | 2,714 | $ | 2,464 | $ | 10,019 | ||||||||||
|
New
Patient Visits
|
5,917 | 5,867 | 5,930 | 6,279 | 23,993 | |||||||||||||||
|
IVF
Cases Completed
|
3,038 | 3,088 | 3,069 | 2,971 | 12,166 | |||||||||||||||
43
Consumer
Services Segment
Revenues
from our Consumer Services Division increased by 5.4%, or $1.1 million, for
the year ended December 31, 2009 versus the same period in the prior year.
Attain IVF Refund Program revenues accounted for approximately 93.8% of the
division’s revenues during 2009, up slightly from approximately 93.1% for the
same period in 2008. Patients enrolled in our Attain IVF Refund Program pay us
an up-front fee (deposit) in return for up to six treatment cycles (consisting
of three fresh IVF cycles and three frozen embryo transfers). Any non-refundable
portion of these fees is recognized as revenue, based on the relative fair value
of each treatment cycle completed relative to the total fair value of the
contracted treatment package available to the patient. The refundable portion of
the program contract amount is recognized as revenue when the patient becomes
pregnant. At the time of pregnancy, we establish a reserve for future medical
costs should the patient miscarry and require additional contracted treatment
cycles. The two main factors that impact Attain IVF Refund Program financial
performance are:
|
•
|
the
number of patients enrolled and receiving
treatment, and
|
|
|
•
|
clinical
pregnancy rates.
|
Patients
enrolled in our Attain IVF Multi-Cycle Program pay us a single fee, which is
slightly less than the average cost of two fresh IVF cycles, in return for up to
four treatment cycles (consisting of two fresh IVF cycles and two frozen embryo
transfers). Our Attain IVF Multi-Cycle Program offers a refund ranging from 10%
to 85% of the contract amount depending on where in the process either we or the
patient elects to terminate the program, as long as termination is prior to a
second fresh IVF cycle. With respect to our Attain IVF Multi-Cycle Program, we
recognize a pro rata share of the contract amount as revenue as each treatment
cycle is completed. The refundable portion of the program contract amount is
recognized as revenue when the patient becomes pregnant. Under such revenue
recognition methodology, we never recognize more revenue than the potential
refundable amount under the program. At the time of pregnancy, we establish a
reserve for future medical costs should the patient miscarry and
require
additional contracted treatment cycles. The main factor that impacts Attain IVF
Multi-Cycle Program financial performance is the number of patients enrolled and
receiving treatment.
During
the second quarter of 2009, the loss of a primary third-party lender that
provided financing programs for Attain IVF patients and a general tightening of
credit standards and higher interest rates caused a decline in new patient
enrollments that adversely affected the program. In October 2009, we
entered into a referral agreement with a new third-party lender to provide
financing options for Attain IVF patients, as well as for certain other
fertility treatments.
Contribution
from our Consumer Services Division for the year ended December 31, 2009 was
$5.2 million versus $5.4 million in the year earlier period. This
decrease reflects slowing enrollments into the program resulting in part from
disruptions in the credit markets which occurred during 2009, which made it more
difficult for patients to finance treatments.
Our
Affiliate Program generated revenues of $1.2 million during 2009, about even
with that of the prior year period. Although our Affiliate Program produces
revenues on a stand alone basis, the primary value of the Affiliate Program is
to serve as a distribution channel for our Attain IVF programs and as an
introduction to our services for medical practices that may become full
fertility Partners. As of December 31, 2009, this network was comprised of 25
independent fertility centers, 3 more than the year earlier period.
Pharmaceutical
revenues for the year ended December 31, 2009 were $40,000 down from $150,000
for the same period in the prior year. Our pharmaceutical revenues are comprised
of marketing support fees we earn based upon underlying product margin as
reported by a third-party pharmaceutical distributor. Over the past several
years we have seen flat or declining revenues due to pharmaceutical cost
increases which the distributor has been unable to pass on to the consumer as a
result of competitive pressures. We view these pricing and margin developments
as longer-term structural elements within the pharmaceutical market and do not
expect significant improvement during 2009. As such we did not renew our
contract with the third-party distributor when it expired on June 30, 2009
and we anticipate no further revenues from this source during 2010 and
beyond.
For the
year ended December 31, 2008, revenues of $18.4 million from our
Attain IVF Refund Program represented approximately 93.1% of our Consumer
Services Division revenues. This compares to revenues of $15.1 million, or
91.8%, of Consumer Services Division revenues in 2007. Revenue growth in our
Attain IVF Refund Program of $3.3 million, or 21.8%, in 2008 compared to
2007 was the result of enrolling more patients into the program and increasing
patient throughput by maintaining high pregnancy success rates. From the
beginning of 2005 through the end of 2009, while pregnancy success rates have
either been maintained or increased, enrollments in our Attain IVF Refund
44
Program
grew at a compound annual rate of 33.3%. Because the patients in our Attain IVF
Refund Program prepay for their suite of services, and a significant portion of
the fees received by us are not recognized until the patient achieves pregnancy,
our Attain IVF Refund Program deposits and deferred revenue balance continues to
grow each year the number of enrolled patients grows. Our Affiliate Program
generated revenues of $1.2 million for the year ended December 31,
2008, which is approximately unchanged from the years ended December 31,
2007 and 2006. During 2008, four independent fertility centers joined our
Affiliate Program and two left in Partner related transactions for a net
increase of two centers. For the year ended December 31, 2007, our
Affiliate Program had a net reduction of one center as one practice moved to our
Partner Program.
Our
Affiliate Program generated revenues of $1.2 million for the year ended
December 31, 2008, which is approximately unchanged from the years ended
December 31, 2007 and 2006. During 2008, four independent fertility centers
joined our Affiliate Program and two left in Partner related transactions for a
net increase of two centers. For the year ended December 31, 2007, our
Affiliate Program had a net reduction of one center as one practice moved to our
Partner Program.
Pharmaceutical
revenues of $154,000 for the year ended December 31, 2008 were
approximately equal to pharmaceutical revenues for the year ended
December 31, 2007, and down from $400,000 in 2006 due to the pharmaceutical
cost increases discussed above.
Contribution
from our Consumer Services Division grew by $1.3 million to
$5.4 million for the year ended December 31, 2008 versus a
contribution of $4.1 million in the year ended December 31, 2007. This
growth was driven by our Attain IVF Refund Program in which the two key
profitability metrics, the number of patients receiving treatment and pregnancy
success rates, showed year-over-year improvement in 2008 versus
2007.
The
following summarized Consumer Services Division quarterly data for the years
ended December 31, 2009, 2008 and 2007 is presented for additional analysis
and demonstration of the fluctuations of enrollments and pregnancies in our
Attain IVF Programs (in thousands, except enrollments and
pregnancies).
|
Period
Ended December 31, 2009
|
||||||||||||||||||||
| Q1 | Q2 | Q3 | Q4 |
Total
|
||||||||||||||||
|
Revenues,
Net
|
$ | 5,226 | $ | 5,004 | $ | 5,013 | $ | 5,583 | $ | 20,826 | ||||||||||
|
Contribution
|
$ | 1,513 | $ | 1,161 | $ | 1,068 | $ | 1,445 | $ | 5,187 | ||||||||||
|
Enrollments
|
253 | 239 | 288 | 283 | 1,063 | |||||||||||||||
|
Pregnancies
|
212 | 199 | 175 | 194 | 780 | |||||||||||||||
|
Period
Ended December 31, 2008
|
||||||||||||||||||||
| Q1 | Q2 | Q3 | Q4 |
Total
|
||||||||||||||||
|
Revenues,
Net
|
$ | 4,196 | $ | 4,807 | $ | 5,364 | $ | 5,396 | $ | 19,763 | ||||||||||
|
Contribution
|
$ | 1,235 | $ | 1,446 | $ | 1,352 | $ | 1,386 | $ | 5,419 | ||||||||||
|
Enrollments
|
212 | 280 | 307 | 250 | 1,049 | |||||||||||||||
|
Pregnancies
|
167 | 189 | 217 | 205 | 778 | |||||||||||||||
|
Period
Ended December 31, 2007
|
||||||||||||||||||||
| Q1 | Q2 | Q3 | Q4 |
Total
|
||||||||||||||||
|
Revenues,
Net
|
$ | 3,235 | $ | 4,179 | $ | 4,557 | $ | 4,489 | $ | 16,460 | ||||||||||
|
Contribution
|
$ | 751 | $ | 1,125 | $ | 1,171 | $ | 1,077 | $ | 4,124 | ||||||||||
|
Enrollments
|
250 | 241 | 247 | 222 | 960 | |||||||||||||||
|
Pregnancies
|
114 | 167 | 173 | 183 | 637 | |||||||||||||||
45
Vein
Clinics Segment
Revenues
in this segment are generated from direct billings to patients or their insurer
for vein disease treatment services and these revenues are consolidated directly
into our financials.
Revenues
for the year ended December 31, 2009 were $50.6 million, up 26.7%, or
$10.7 million from 2008. During the 2009, we opened new vein clinic
locations in Cincinnati and Cleveland, marking our entry into the State of Ohio
and these two markets. These additional clinics brought our total number of vein
clinics to 34. The two new vein clinics opened in 2009 accounted for
$1.7 million of our Vein Clinics Division’s revenue increase, the five
clinics opened throughout 2008 accounted for $6.1 million of the increase with
the remaining $2.8 million generated by legacy clinics.
For 2009,
Vein Clinics Division contribution of $4.1 million, or 8.1% of Vein Clinics
Division revenues, compared to contribution of $2.7 million, or 6.7% of
Vein Clinics Division revenues, for 2008. The improved performance for the 2009
is largely attributable to the additional operational and marketing
infrastructure put in place during 2008, combined with increased revenue from
both new and previously operated clinics. The infrastructure improvements
allowed the division to conduct ongoing direct-to-consumer marketing initiatives
and provided the resources necessary to service the resulting increase in
patient flow.
Revenues
for the year ended December 31, 2008 were approximately $40.0 million
versus partial year revenues of $14.3 million in 2007. Revenues for the
year ended December 31, 2007 represent operating results only since VCA was
purchased on August 8, 2007.
For the
year ended December 31, 2008, contribution from our Vein Clinics Division
of $2.7 million was up $1.7 million from 2007. Year versus year
comparisons for this segment are not directly comparable as 2007 results only
include contribution generated since we acquired this segment in August
2007.
We
continue to target the opening of additional new vein clinics in locations
across the United States during 2010 and future years, however this pace
could be affected by challenges in physician recruitment. To address this issue
we have assembled a physician recruitment task force to develop a strategy and
plan to raise the profile of the vein care career opportunity to high-quality
physicians across the United States.
New
consultations, which are an indication of patient interest in vein care
treatment, rose 43% for the year ended December 31, 2009 versus the year earlier
period. First leg starts, which signify the beginning of a billable treatment
cycle, rose 31.8% for the year ended December 31, 2009 versus the year earlier
period. We have included the results of VCA in our financial statements since
the date of its acquisition on August 8, 2007. Vein Clinics Division
quarterly data for the years ended December 31, 2009, 2008 and 2007 appear
below (in thousands, except first leg starts).
|
Period
Ended December 31, 2009
|
||||||||||||||||||||
| Q1 | Q2 | Q3 | Q4 |
Total
|
||||||||||||||||
|
Revenues,
Net
|
$ | 10,846 | $ | 13,821 | $ | 12,621 | $ | 13,337 | $ | 50,625 | ||||||||||
|
Contribution
|
$ | 754 | $ | 1,282 | $ | 995 | $ | 1,069 | $ | 4,100 | ||||||||||
|
First
Leg Starts
|
1,574 | 2,085 | 1,959 | 1,590 | 7,208 | |||||||||||||||
|
Total
procedures
|
2,578 | 3,499 | 3,215 | 2,847 | 12,319 | |||||||||||||||
|
Period
Ended December 31, 2008
|
||||||||||||||||||||
| Q1 | Q2 | Q3 | Q4 |
Total
|
||||||||||||||||
|
Revenues,
Net
|
$ | 8,842 | $ | 10,062 | $ | 10,360 | $ | 10,686 | $ | 39,950 | ||||||||||
|
Contribution
|
$ | 322 | $ | 713 | $ | 892 | $ | 724 | $ | 2,651 | ||||||||||
|
First
Leg Starts
|
1,208 | 1,572 | 1,500 | 1,187 | 5,467 | |||||||||||||||
|
Total
procedures
|
2,035 | 2,615 | 2,489 | 2,116 | 9,273 | |||||||||||||||
46
|
Period
Ended December 31, 2007
|
||||||||||||||||||||
| Q1 | Q2 | Q3 | Q4 |
Total
|
||||||||||||||||
|
Revenues,
Net
|
$ | N/A | $ | N/A | $ | 8,283 | $ | 9,704 | $ | 17,987 | ||||||||||
|
Contribution
|
$ | N/A | $ | N/A | $ | (1,704 | ) | $ | 438 | $ | (2,166 | ) | ||||||||
|
First
Leg Starts
|
N/A | N/A | 1,266 | (1) | 1,127 | 2,393 | ||||||||||||||
|
Total
procedures
|
N/A | N/A | 1,916 | (1) | 1,878 | 3,794 | ||||||||||||||
|
(1)
|
Includes
the period from July 1, 2007 through August 7, 2007, which is
prior to the VCA acquisition.
|
General
and Administrative Expenses
General
and administrative expenses are comprised of salaries and benefits,
administrative, regulatory compliance and operational support costs defined as
our Shared Services group, which are not specifically related to individual
center or clinic operations or other product offerings.
General
and administrative expenses totaled $12.2 million for the year
ended December 31, 2009, an increase from the $10.7 million recognized in
the prior year. The increased general and administrative expenses in 2009 is
attributable to higher service and infrastructure activities designed to provide
operational support to our three growing business segments. We measure our
performance in part by relating general and administrative expenses to operating
contribution. For the year ended December 31, 2009, general and administrative
expenses were 58.2% of contribution compared to a ratio of 58.3% for the year
ended December 31, 2008.
General
and administrative expenses totaled $10.7 million in 2008 and
$10.5 million in 2007. For the year ended December 31, 2008, general
and administrative expenses were 58.3% of contribution as compared to
69.7% in 2007. We continue to actively manage general and
administrative expenses in an effort to leverage our Shared Services group and
extract economies of scale as those opportunities arise.
Interest
Net
interest expense was $0.9 million for the year ended December 31, 2009 as
compared to net interest expense of $1.2 million for 2008. The reduction in net
interest expense for the year is the result of scheduled debt repayments which
reduced our outstanding loan balances combined with lower market interest rates
on certain portions of the remaining balances.
Net
interest expense for the year ended December 31, 2008 totaled
$1.2 million, compared to net interest income of $0.1 million for the year
ended December 31, 2007. The change in net interest income/expense between
2008 and 2007 is primarily the result of utilizing cash on hand and additional
borrowings as the principal means of financing our acquisition of VCA in August
2007. This acquisition used approximately $14 million of cash from our
balance sheet in addition to $17 million of new borrowings.
Subject
to interest rate fluctuations and any changes to our credit arrangements, we
anticipate interest expense to decrease gradually in the coming quarters as
scheduled debt repayments reduce our outstanding principal
balances.
Income
Tax Provision
Our
provision for income tax was approximately $3.3 million, $2.5 million and $1.7
million for the three years ended December 31, 2009, 2008 and 2007 respectively,
or 42.6%, 39.3% and 35.3% of pre-tax income, respectively. Our effective tax
rates for all years reflect provisions for both federal and state income taxes.
The lower effective tax rate of 35.3% for the year ended December 31, 2007 was
mainly due to benefits received from tax-exempt interest income. We expect the
effective tax rate to approximate 42% in 2010.
47
In July
2006, guidance on accounting for uncertainty in income taxes clarified the
accounting for uncertainty in income taxes recognized in an entity’s financial
statements and prescribes a recognition threshold and measurement attribute for
financial statement disclosure of tax positions taken or expected to be taken on
a tax return. We adopted this guidance on January 1, 2007, and the
adoption did not have a material impact on our financial
statements.
We file
income tax returns in the U.S. federal jurisdiction and various states. For
federal income tax purposes, our 2007 through 2009 tax years remain open for
examination by the tax authorities under the normal three year statute of
limitations. A federal income tax examination for tax years through 2006 was
completed during 2008 resulting in no adjustment to our income tax liability.
For state tax purposes, our 2004 through 2009 tax years remain open for
examination by the tax authorities under a four year statute of limitations for
many of the jurisdictions in which we operate.
Off-Balance
Sheet Arrangements
Current
accounting guidance addresses how a business enterprise should evaluate whether
it has a controlling financial interest in an entity through means other than
voting rights and accordingly should consolidate the entity. In June 2009, the
Financial Accounting Standards Board ("FASB") amended its guidance on accounting
for variable interest entities ("VIE"). The new accounting guidance is effective
for reporting periods after January 1, 2010. Both sets of guidance require
continuous assessments of whether an enterprise is the primary beneficiary of a
VIE and requires consolidation if the enterprise has the power to direct the
activities of the VIE that most significantly impact the entity's economic
performance, and is the primary beneficiary or obligor of the VIE.As of December
31, 2009, through the acquisition of Vein Clinics of America, Inc, we have
interests in the individual vein clinics, where we are the primary beneficiary
and obligor of their financial results (our contract provides for us to receive
any excess or deficit profits from the vein clinics). As such we have
consolidated these vein clinic operations in our financial
statements. Since we do not have any financial interest in the
individual fertility centers and we are not the primary beneficiary or obligor
of their financial results (our contracts provide for the physician owners of
the clinics to receive any excess or deficit profits), we do not consolidate the
results of the fertility centers in our accounts. Also, since we do not have a
controlling interest in the captive insurance provider and we are not the
primary beneficiary, we do not consolidate the results of the captive insurance
company in our accounts.
Liquidity
and Capital Resources
As of
December 31, 2009, we had approximately $28.9 million in cash and cash
equivalents on hand as compared to $28.3 million at December 31, 2008. We had a
working capital deficit of approximately $2.7 million, at December 31, 2009,
approximately in line with the deficit of $2.4 million as of December 31,
2008.
On
February 12, 2010, we announced that we priced an underwritten public offering
of 2,800,000 shares of our common stock at an offering price of $7.50 per
share. This offering raised approximately $17 million in proceeds after
underwriting discounts and expenses related to the offering. The
offering closed on February 18,2010.
Attain
IVF deferred revenue and other patient deposits, which are reflected as a
current liability, represent funds received from patients in advance of
treatment cycles and are an indication of potential future revenues. These
deposits totaled approximately $13.4 million and $11.2 million as December 31,
2009 and 2008, respectively. The change in deposit level is a function of
patient enrollments into the program, less patient through-put into our
treatment programs. These deposits are a significant source of cash flow and
represent interest-free financing for us.
As of
December 31, 2009, we did not have any significant contractual commitments for
the acquisition of fixed assets or construction of leasehold
improvements. However we anticipate capital acquisition totaling $4.9
million during the upcoming twelve months. These expenditures are
primarily related to medical equipment, information system infrastructure and
leasehold improvements. We believe that working capital, specifically cash and
cash equivalents, including an additional $17 million raised in February 2010
from our underwritten offering, remains at adequate levels to fund our
operations and our commitments for fixed asset acquisitions. We also believe
that the cash flows from our operations plus our available credit facility will
be sufficient to provide for our future liquidity needs over the next twelve
months.
In
August, 2007, we entered into a new financing arrangement with Bank of America
and secured a $25 million five-year term loan. After deducting the outstanding
balance of $7.7 million on our previous loan amount, interest and fees, our net
funding from Bank of America was $17.0 million. In order to mitigate the
interest rate risk associated with this term loan, we also entered into an
interest rate swap agreement on 50% of the principal amount effectively
48
fixing
the interest rate at 5.39%, plus the applicable margin, on that portion of the
loan. Our swap agreement matures in August 2010 and our term loan matures in
August 2012. We deem this hedge to be highly effective over its term to maturity
as it shares the same amortization schedule as the underlying debt subject to
the hedge and any change in fair value inversely mimics the appropriate portion
of the hedged item. At the expiration of the swap agreement, or amendment to the
term loan, which ever occurs first, we will re-evaluate our hedge position and
modify our disclosures as necessary.
Each
component of our amended credit facility bears interest by reference, at our
option, to Bank of America’s prime rate minus a margin or to LIBOR plus a
margin. The margin is dependent upon a leverage test, ranging from 2.00% to
2.75% in the case of LIBOR-based term loans and 0.00% to 0.50% in the case of
prime-based term loans. Interest on the revolving line of credit is at the prime
rate less up to 0.50% or at LIBOR plus 1.50% to 2.50%, depending on a leverage
test. Interest on the prime-based loans became payable quarterly beginning on
November 8, 2007 and interest on LIBOR-based loans is payable on the last
day of each applicable interest period. As of December 31, 2009, interest
on the term loan was payable at a rate of approximately 2.71%.
Other
features of this credit facility include a $10 million five-year revolving line
of credit. Availability of borrowings under the working capital revolver is
based on eligible accounts receivable, as defined in the credit
agreement. As of December 31, 2009 under the revolving line of credit
the full amount of $10.0 million was available, of which $7.5 million was
outstanding. Unused amounts under the working capital revolver bear a commitment
fee of 0.25% and are payable quarterly.
Our Bank
of America credit facility is collateralized by substantially all of our
assets. As of December 31, 2009, we were in full compliance with all
applicable debt covenants. We also continuously review our credit agreements and
may renew, revise or enter into new agreements from time to time as deemed
necessary.
Significant
Contractual Obligations and Other Commercial Commitments
The
following summarizes our contractual obligations and other commercial
commitments at December 31, 2009, and the effect such obligations are
expected to have on our liquidity and cash flows in future periods.
|
Payments
Due by Period
|
||||||||||||||||||||
|
Total
|
Less
Than
1 Year
|
1 —
3 Years
|
4 —
5 Years
|
After
5 Years
|
||||||||||||||||
|
(in
thousands)
|
||||||||||||||||||||
|
Notes
payable
|
$
|
18,142
|
$
|
3,755
|
$
|
14,387
|
$
|
—
|
$
|
—
|
||||||||||
|
Line
of credit outstanding
|
7,500
|
7,500
|
—
|
—
|
—
|
|||||||||||||||
|
Capital
lease obligations
|
218
|
89
|
129
|
—
|
—
|
|||||||||||||||
|
Interest
on debt
|
2,
187
|
1,142
|
1,045
|
—
|
—
|
|||||||||||||||
|
Operating
leases
|
68,584
|
10,339
|
29,746
|
7,050
|
21,449
|
|||||||||||||||
|
Fertility
Partners capital and other obligations (1)
|
4,901
|
4,901
|
—
|
—
|
—
|
|||||||||||||||
|
Total
contractual cash obligations
|
$
|
101,532
|
$
|
27,726
|
$
|
45,307
|
$
|
7,050
|
$
|
21,449
|
||||||||||
|
Amount
of Commitment Expiration Per Period
|
||||||||||||||||||||
|
Total
|
Less
Than
1 Year
|
1 —
3 Years
|
4 —
5 Years
|
After
5 Years
|
||||||||||||||||
|
(in
thousands)
|
||||||||||||||||||||
|
Unused
lines of credit
|
$
|
2,500
|
$
|
—
|
$
|
2,500
|
$
|
—
|
$
|
—
|
||||||||||
|
|
Includes
planned, but not yet legally committed capital expenditures at our
Fertility Partner Practices
|
49
We also
have commitments to provide working capital financing to member centers in our
Fertility Centers Division that are not included in the above table. A
significant portion of these commitments relate to our transactions with the
medical practices themselves. Our responsibilities to the these medical
practices are to provide financing for their accounts receivable and to hold
patient deposits on their behalf, as well as undistributed physician earnings.
Disbursements to the medical practices generally occur monthly. The medical
practice’s repayment hierarchy consists of the following:
|
•
|
We
provide a cash credit to the practice for billings to patients and
insurance companies;
|
|
|
•
|
We
reduce the cash credit for center expenses that we have incurred on behalf
of the practice;
|
|
|
•
|
We
reduce the cash credit for the base portion of our service fee which
relates to the Partner revenues;
|
|
|
•
|
We
reduce the cash credit for the variable portion of our service fee which
relates to the Partner earnings; and
|
|
|
•
|
We
disburse to the medical practice the remaining cash amount which
represents the physician’s undistributed
earnings.
|
We are
also responsible for the collection of the Partner accounts receivables. We
continuously fund these needs from our cash flows from operations, the
collection of prior months’ receivables and deposits from patients in advance of
treatment. If delays in repayment are incurred, which have not as yet been
encountered, we could draw on our existing revolving line of credit. We also
make payments on behalf of the Partner for which we are reimbursed in the
short-term. Other than these payments, as a general course, we do not make other
advances to the medical practices. We have no other funding commitments to the
Partner centers.
Recently
Issued Accounting Pronouncements
Please
see Note 2 (under the subheading “Recently Issued Accounting
Pronouncements”) of the notes to our consolidated financial statements included
elsewhere in this Form 10-K for a discussion on recently issued accounting
pronouncements.
ITEM
7a. Quantitative and Qualitative Disclosures About Market
Risk
In the
normal course of business our interest income and expense items are sensitive to
changes in the general level of interest rates. During the third quarter of 2007
we entered into an interest rate swap agreement designed to hedge 50% of our
$25 million variable interest rate term loan maturing in 2012. As a result
of this swap transaction we have partially shielded ourselves from a portion of
the interest rate risks associated with that portion of the term loan, as the
swap transaction essentially converts that portion of the term loan to a fixed
rate instrument at 5.39% plus the applicable margin through the maturity of the
swap agreement in August 2010. We are currently subject to interest rate risks
associated with the remaining 50% of our term loan, as well as our short term
investments and certain advances to our fertility centers, all of which are tied
to either short term interest rates, LIBOR or the prime rate. As of both
December 31, 2008 and 2009, a 1% change in interest rates would have
impacted our pre-tax income by approximately $100,000 annually.
ITEM
8. Financial Statements and Supplementary Data
See Index
to Financial Statements on Page F-1.
ITEM
9. Changes in and disagreements with accountants on Accounting and
financial Disclosure.
None.
50
ITEM
9A. Controls and Procedures
Evaluation
of Disclosure Controls and Procedures
We
carried out an evaluation, under the supervision and with the participation of
our management, including our Chief Executive Officer and Chief Financial
Officer, of the effectiveness of our disclosure controls and procedures (as
defined in Rules 13a-15(e) and 15d-15(e) under the Securities Exchange Act of
1934, as amended). Based upon that evaluation, the Chief Executive Officer and
Chief Financial Officer have concluded that our disclosure controls and
procedures were effective as of the end of the period covered by this
report.
Management’s
Annual Report on Internal Control Over Financial Reporting
Management
is responsible for establishing and maintaining adequate internal control over
financial reporting as defined in Rule 13a-15(f) under the Exchange
Act.
Internal
control over financial reporting refers to a process designed by, or under the
supervision of, our Chief Executive Officer and Chief Financial Officer and
effected by our board of directors, management and other personnel, to provide
reasonable assurance regarding the reliability of financial reporting and the
preparation of financial statements for external purposes in accordance with
generally accepted accounting principles and includes those policies and
procedures that:
|
•
|
pertain
to the maintenance of records that in reasonable detail accurately and
fairly reflect the transactions and dispositions of our
assets;
|
|
•
|
provide
reasonable assurance that transactions are recorded as necessary to permit
preparation of financial statements in accordance with generally accepted
accounting principles, and that our receipts and expenditures are being
made only in accordance with authorizations of our management and members
of our board of directors; and
|
||
|
|
•
|
provide
reasonable assurance regarding prevention or timely detection of
unauthorized acquisition, use, or disposition of our assets that could
have a material effect on our financial
statements.
|
Internal
control over financial reporting cannot provide absolute assurance of achieving
financial reporting objectives because of its inherent limitations. Internal
control over financial reporting is a process that involves human diligence and
compliance and is subject to lapses in judgment and breakdowns resulting from
human failures. Internal control over financial reporting also can be
circumvented by collusion or improper override. Because of such limitations,
there is a risk that material misstatements may not be prevented or detected on
a timely basis by internal control over financial reporting. However, these
inherent limitations are known features of the financial reporting process, and
it is possible to design into the process safeguards to reduce, though not
eliminate, this risk.
Management
conducted the above-referenced assessment of the effectiveness of our internal
control over financial reporting as of December 31, 2009 using the
framework set forth in the report entitled, “Internal Control—Integrated
Framework,” issued by the Committee of Sponsoring Organizations of the
Treadway Commission, or the COSO Report. Based on management’s
evaluation and the criteria set forth in the COSO Report, management concluded
that our internal control over financial reporting was effective as of
December 31, 2009.
The
Company’s independent registered certified public accounting firm, Amper,
Politziner & Mattia LLP, has audited our internal control over financial
reporting as of December 31, 2009 as stated in their report which appears
on page F-2 of this Annual Report on Form 10-K for the fiscal year ended
December 31, 2009.
(b) Changes in internal
controls.
There
were no changes made in our internal control over financial reporting during the
period covered by this report that have materially affected, or are reasonably
likely to materially affect, our internal control over financial
reporting.
ITEM
9B. Other Information
None.
51
PART
III
ITEM
10. Directors,
Executive Officers and Corporate Governance
Our Board
of Directors has adopted a Code of Business Conduct that is applicable to all of
our directors, officers and employees, a copy of which has previously been filed
with the SEC. Any material changes made to our Code of Business
Conduct or any waivers granted to any of our directors and executive officers
will be publicly disclosed by filing a current report on Form 8-K. A
copy of our Code of Business Conduct as well as charters for our Audit
Committee, Compensation Committee and Nominating and Corporate Governance
Committee, which comply with the corporate governance rules of NASDAQ, are
available on our website at www.integramed.com. In
addition, copies of such documents are also available to our shareholders upon
request by contacting our Investor Relations Department at 914-253-8000 or
through an e-mail request from our website at www.integramed.com.
The other
information required by this Item is incorporated by reference to the applicable
information in the definitive proxy statement for our 2009 annual meeting of
shareholders, which is to be filed with the SEC within 120 days after our fiscal
year end, including the information set forth under the captions “Election of
Directors for a Term of One Year”, “Section 16 (a) Beneficial Ownership
Reporting Compliance” and “Committees of the Board”.
ITEM
11. Executive
Compensation
The
information required by this Item is incorporated by reference to the applicable
information in the definitive proxy statement for our 2010 annual meeting of
shareholders, which is to be filed with the SEC within 120 days after our fiscal
year end, including the information set forth under the captions “Executive
Compensation”, “Director Compensation” and “Compensation Committee Interlocks
and Insider Participation”.
|
ITEM
12.
|
Security
Ownership of Certain Beneficial Owners and Management, and Related
Stockholder Matters
|
The
information required by this Item is incorporated by reference to the applicable
information in the definitive proxy statement for our 2010 annual meeting of
shareholders, which is to be filed with the SEC within 120 days after our fiscal
year end, including the information set forth under the caption “Security
Ownership”.
ITEM
13. Certain
Relationships, Related Transactions and Director Independence
The
information required by this Item is incorporated by reference to the applicable
information in the definitive proxy statement for our 2010 annual meeting of
shareholders, which is to be filed with the SEC within 120 days after our fiscal
year end, including the information set forth under the caption “Certain
Relationships and Related Transactions”.
ITEM
14. Principal Accountant Fees and Services
The
information required by this Item is incorporated by reference to the applicable
information in the definitive proxy statement for our 2010 annual meeting of
shareholders, which is to be filed with the SEC within 120 days after our fiscal
year end, including the information set forth under the caption “Independent
Public Registered Accounting Firm”.
PART IV
ITEM
15. Exhibits and Financial Statement Schedule
|
(a)
|
(1)
|
Financial
Statements
|
|
(2)
|
The
exhibits that are listed on the Index to Exhibits herein which are filed
as 21.1; 23.1; 31.1; 31.2; 32.1 and 32.2
|
|
|
(b)
|
Exhibits. The
list of exhibits required to be filed with Annual Report on Form 10-K is
set forth in the Index to Exhibits
herein.
|
52
FINANCIAL STATEMENTS
Item
8 and 15 (a)(1)
Contents
|
IntegraMed
America, Inc
|
Page
|
|
Report of Independent
Registered Public Accounting Firm
|
F-2
|
|
Consolidated
Balance Sheets as of December 31, 2009 and 2008
|
F-3
|
|
Consolidated
Statements of Operations for the years ended December 31, 2009, 2008 and
2007
|
F-4
|
|
Consolidated
Statements of Shareholders' Equity for the years ended December 31, 2009,
2008 and 2007
|
F-5
|
|
Consolidated
Statements of Cash Flows for the years ended December 31, 2009, 2008 and
2007
|
F-6
|
|
Notes to Consolidated Financial Statements
|
F-7
|
|
Financial
Statement Schedule
|
|
|
Report
of Independent Registered Public Accounting Firms on Financial Statement
Schedule II
|
S-1
|
|
Valuation and Qualifying
Accounts
|
S-2
|
F-1
REPORT OF
INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the
Board of Directors and
Shareholders
of IntegraMed America, Inc.
We have
audited the accompanying consolidated balance sheets of IntegraMed America, Inc.
as of December 31, 2009 and 2008 and the related consolidated statements of
operations, shareholders’ equity and cash flows for each of the years in the
three-year period ended December 31, 2009. We also have audited
IntegraMed America, Inc.‘s internal control over financial reporting as of
December 31, 2009, based on criteria established in Internal Control-Integrated
Framework issued by the Committee of Sponsoring Organizations of the
Treadway Commission (COSO). IntegraMed America, Inc.’s management is
responsible for these financial statements, for maintaining effective internal
control over financial reporting, and for its assessment of the effectiveness of
internal control over financial reporting, included in the accompanying
consolidated financial statements. Our responsibility is to express
an opinion on these financial statements and an opinion on the company’s
internal control over financial reporting based on our audits.
We
conducted our audits in accordance with the standards of the Public Company
Accounting Oversight Board (United States). Those standards require
that we plan and perform the audits to obtain reasonable assurance about whether
the financial statements are free of material misstatement and whether effective
internal control over financial reporting was maintained in all material
respects. Our audits of the financial statements included examining,
on a test basis, evidence supporting the amounts and disclosures in the
financial statements, assessing the accounting principles used and significant
estimates made by management, and evaluating the overall financial statement
presentation. Our audit of internal control over financial reporting
included obtaining an understanding of internal control over financial
reporting, assessing the risk that a material weakness exists, and testing and
evaluating the design and operating effectiveness of internal control based on
the assessed risk. Our audits also included performing such other
procedures as we considered necessary in the circumstances. We
believe that our audits provide a reasonable basis for our
opinions.
A
company’s internal control over financial reporting is a process designed to
provide reasonable assurance regarding the reliability of financial reporting
and the preparation of financial statements for external purposes in accordance
with generally accepted accounting principles. A company’s internal
control over financial reporting includes those policies and procedures that (1)
pertain to the maintenance of records that, in reasonable detail, accurately and
fairly reflect the transactions and dispositions of the assets of the company;
(2) provide reasonable assurance that transactions are recorded as necessary to
permit preparation of financial statements in accordance with generally accepted
accounting principles, and that receipts and expenditures of the company are
being made only in accordance with authorizations of management and directors of
the company; and (3) provide reasonable assurance regarding prevention or timely
detection of unauthorized acquisition, use, or disposition of the company’s
assets that could have a material effect on the financial statements. Because of
its inherent limitations, internal control over financial reporting may not
prevent or detect misstatements. Also, projections of any evaluation
of effectiveness to future periods are subject to the risk that controls may
become inadequate because of changes in conditions, or that the degree of
compliance with the policies or procedures may deteriorate.
In our
opinion, the financial statements referred to above present fairly, in all
material respects, the financial position of IntegraMed America, Inc. as
of December 31, 2009 and 2008, and the results of its operations and
its cash flows for each of the years in the three-year period ended December 31,
2009 in conformity with accounting principles generally accepted in the United
States of America. Also in our opinion, IntegraMed America, Inc.
maintained, in all material respects, effective internal control over financial
reporting as of December 31, 2009, based on criteria established in Internal Control-Internal
Framework issued by the Committee of Sponsoring Organizations of the
Treadway Commission (COSO).
/s/Amper,
Politziner & Mattia, LLP.
Edison,
New Jersey
March 10,
2010
F-2
PART
I — FINANCIAL INFORMATION
Item
1. Consolidated
Financial Statements
INTEGRAMED
AMERICA, INC.
CONSOLIDATED
BALANCE SHEETS
(all
dollars in thousands, except share amounts)
|
December
31,
|
December
31,
|
|||||||
|
2009
|
2008
|
|||||||
|
ASSETS
|
||||||||
|
Current
assets:
|
||||||||
|
Cash
and cash equivalents
|
$ | 28,865 | $ | 28,275 | ||||
|
Patient
and other receivables, net
|
6,964 | 6,681 | ||||||
|
Deferred
income taxes
|
2,883 | 5,744 | ||||||
|
Prepaids
and other current assets
|
7,653 | 6,466 | ||||||
|
Total
current assets
|
46,365 | 47,166 | ||||||
|
Fixed
assets, net
|
16,705 | 16,618 | ||||||
|
Intangible
assets, Business Service Rights, net
|
24,210 | 21,956 | ||||||
|
Goodwill
|
30,334 | 29,478 | ||||||
|
Trademarks
|
4,442 | 4,442 | ||||||
|
Other
assets
|
2,253 | 1,781 | ||||||
|
Total
assets
|
$ | 124,309 | $ | 121,441 | ||||
|
LIABILITIES
AND SHAREHOLDERS' EQUITY
|
||||||||
|
Current
liabilities:
|
||||||||
|
Accounts
payable
|
$ | 2,846 | $ | 2,853 | ||||
|
Accrued
liabilities
|
15,119 | 17,818 | ||||||
|
Current
portion of long-term notes payable and other obligations
|
11,317 | 11,351 | ||||||
|
Due
to Fertility Medical Practices
|
6,424 | 6,354 | ||||||
|
Attain
IVF Refund Program deferred revenue and other Patient
Deposits
|
13,362 | 11,237 | ||||||
|
Total
current liabilities
|
49,068 | 49,613 | ||||||
|
Long-term
notes payable and other obligations
|
14,849 | 18,868 | ||||||
|
Deferred
income and other tax liabilities
|
2,199 | 696 | ||||||
|
Total
Liabilities
|
66,116 | 69,177 | ||||||
|
Commitments
and Contingencies
|
||||||||
|
Shareholders'
equity:
|
||||||||
|
Common
Stock, $.01 par value – 15,000,000 shares authorized at December 31, 2009
and 2008, 8,831,558 and 8,668,376 shares issued and outstanding on
December 31, 2009 and 2008 respectively
|
88 | 87 | ||||||
|
Capital
in excess of par
|
56,354 | 54,943 | ||||||
|
Other
comprehensive loss
|
(188 | ) | (375 | ) | ||||
|
Treasury
stock, at cost – 46,408 and 22,682 shares in 2009 and 2008,
respectively
|
(375 | ) | (211 | ) | ||||
|
Retained
Earnings (Accumulated deficit)
|
2,314 | (2,180 | ) | |||||
|
Total
shareholders' equity
|
58,193 | 52,264 | ||||||
|
Total
liabilities and shareholders' equity
|
$ | 124,309 | $ | 121,441 | ||||
See accompanying notes to the
consolidated financial statements.
F-3
INTEGRAMED
AMERICA, INC.
CONSOLIDATED
STATEMENTS OF OPERATIONS
(all
amounts in thousands, except per share amounts)
|
For
the years ended
|
||||||||||||
|
2009
|
2008
|
2007
|
||||||||||
|
Revenues,
net
|
||||||||||||
|
Fertility
Centers
|
$ | 145,309 | $ | 138,440 | $ | 121,078 | ||||||
|
Consumer
Services
|
20,826 | 19,763 | 16,460 | |||||||||
|
Vein Clinics
|
50,625 | 39,950 | 14,284 | |||||||||
|
Total revenues
|
216,760 | 198,153 | 151,822 | |||||||||
|
Costs
of services and sales:
|
||||||||||||
|
Fertility
Centers
|
133,706 | 128,224 | 111,059 | |||||||||
|
Consumer
Services
|
15,639 | 14,344 | 12,336 | |||||||||
|
Vein Clinics
|
46,525 | 37,299 | 13,304 | |||||||||
|
Total costs of services and
sales
|
195,870 | 179,867 | 136,699 | |||||||||
|
Contribution
|
||||||||||||
|
Fertility
Centers
|
11,603 | 10,216 | 10,019 | |||||||||
|
Consumer
Services
|
5,187 | 5,419 | 4,124 | |||||||||
|
Vein Clinics
|
4,100 | 2,651 | 980 | |||||||||
|
Total
contribution
|
20,890 | 18,286 | 15,123 | |||||||||
|
General
and administrative expenses
|
12,155 | 10,654 | 10,537 | |||||||||
|
Interest
income
|
(250 | ) | (383 | ) | (1,256 | ) | ||||||
|
Interest
expense
|
1,160 | 1,563 | 1,136 | |||||||||
|
Total other
expenses
|
13,065 | 11,834 | 10,417 | |||||||||
|
Income
before income taxes
|
7,825 | 6,452 | 4,706 | |||||||||
|
Income
tax provision
|
3,331 | 2,537 | 1,662 | |||||||||
|
Net
income
|
$ | 4,494 | $ | 3,915 | $ | 3,044 | ||||||
|
Basic
and diluted net earnings per share:
|
||||||||||||
|
Basic earnings per
share
|
$ | 0.51 | $ | 0.45 | $ | 0.37 | ||||||
|
Diluted earnings per
share
|
$ | 0.51 | $ | 0.45 | $ | 0.36 | ||||||
|
Weighted average shares -
basic
|
8,773 | 8,618 | 8,310 | |||||||||
|
Weighted average shares -
diluted
|
8,834 | 8,691 | 8,410 | |||||||||
See
accompanying notes to the consolidated financial statements.
F-4
INTEGRAMED AMERICA,
INC.
CONSOLIDATED STATEMENTS OF
SHAREHOLDERS’ EQUITY
(all amounts in
thousands)
|
Accumulated
|
Retained
Earnings
|
|||||||||||||||||||||||||||||||
|
Common
Stock
|
Capital
in
|
Comprehensive
|
Treasury
Stock
|
(Accumulated
|
Total
|
|||||||||||||||||||||||||||
|
Shares
|
Amount
|
Excess
of Par
|
Income
|
Shares
|
Amount
|
Deficit)
|
Equity
|
|||||||||||||||||||||||||
|
BALANCE
AT DECEMBER 31, 2006
|
8,127 | 81 | 49,245 | (9 | ) |
|
|
(9,139 | ) | 40,178 | ||||||||||||||||||||||
|
Stock
grants issued, net
|
78 |
|
|
|
19 | (228 | ) |
|
(228 | ) | ||||||||||||||||||||||
|
Stock
based compensation expense
|
|
|
558 |
|
|
|
|
558 | ||||||||||||||||||||||||
|
Exercise
of common stock options and related
tax benefits
|
35 | 1 | 154 |
|
|
|
|
155 | ||||||||||||||||||||||||
|
Treasury
stock transactions, net
|
(5 | ) |
|
(63 | ) |
|
(5 | ) | 63 |
|
|
|||||||||||||||||||||
|
Issuance
of common stock upon acquisition of
Vein Clinics of America,
Inc.
|
337 | 4 | 3,996 |
|
|
|
|
4,000 | ||||||||||||||||||||||||
|
Unrealized
loss on hedging transaction
|
|
|
|
(73 | ) |
|
|
|
(73 | ) | ||||||||||||||||||||||
|
Net
income for the year ended 12/31/07
|
|
|
|
|
|
|
3,044 | 3,044 | ||||||||||||||||||||||||
|
BALANCE
AT DECEMBER 31, 2007
|
8,572 | 86 | 53,890 | (82 | ) | 14 | (165 | ) | (6,095 | ) | 47,634 | |||||||||||||||||||||
|
Stock
grants issued, net
|
99 | 1 | (1 | ) |
|
|
|
|
|
|||||||||||||||||||||||
|
Stock
based compensation expense
|
|
|
858 |
|
|
|
|
858 | ||||||||||||||||||||||||
|
Exercise
of common stock options and related
tax benefits
|
11 | 1 | 360 |
|
2 | (23 | ) |
|
338 | |||||||||||||||||||||||
|
Treasury
stock transactions, net
|
(14 | ) | (1 | ) | (164 | ) |
|
7 | (23 | ) |
|
(188 | ) | |||||||||||||||||||
|
Unrealized
loss on hedging transaction
|
|
|
|
(293 | ) |
|
|
|
(293 | ) | ||||||||||||||||||||||
|
Net
income for the year ended 12/31/08
|
|
|
|
|
|
|
3,915 | 3,915 | ||||||||||||||||||||||||
|
BALANCE
AT DECEMBER 31, 2008
|
8,668 | $ | 87 | $ | 54,943 | $ | (375 | ) | 23 | $ | (211 | ) | $ | (2,180 | ) | $ | 52,264 | |||||||||||||||
|
Stock
grants issued, net
|
142 | 1 | (1 | ) | 23 | (164 | ) | (164 | ) | |||||||||||||||||||||||
|
Stock
based compensation expense
|
1,337 | 1,337 | ||||||||||||||||||||||||||||||
|
Exercise
of common stock options and related
tax benefits
|
22 |
|
75 |
|
|
|
|
75 | ||||||||||||||||||||||||
|
Unrealized
gain on hedging transaction
|
|
|
|
187 |
|
|
|
187 | ||||||||||||||||||||||||
|
Net
income for the year ended 12/31/09
|
|
|
|
|
|
|
4,494 | 4,494 | ||||||||||||||||||||||||
|
BALANCE
AT DECEMBER 31, 2009
|
8,832 | $ | 88 | $ | 56,354 | $ | (188 | ) | 46 | $ | (375 | ) | 2,314 | $ | 58,193 | |||||||||||||||||
See
accompanying notes to the consolidated financial statements
F-5
INTEGRAMED
AMERICA, INC.
CONSOLIDATED
STATEMENTS OF CASH FLOWS
(all
amounts in thousands)
|
For
the year ended
December
31,
|
||||||||||||
|
2009
|
2008
|
2007
|
||||||||||
|
(restated)
|
(restated)
|
|||||||||||
|
Cash
flows from operating activities:
|
||||||||||||
|
Net
income
|
$ | 4,494 | $ | 3,915 | $ | 3,044 | ||||||
|
Adjustments
to reconcile net income to net cash provided
|
||||||||||||
|
operating
activities:
|
||||||||||||
|
Depreciation and
amortization
|
7,119 | 7,288 | 6,450 | |||||||||
|
Deferred income tax
provision
|
3,433 | (1,068 | ) | 469 | ||||||||
|
Deferred or stock-based
compensation
|
1,337 | 858 | 558 | |||||||||
|
Changes
in assets and liabilities —
|
||||||||||||
|
Decrease (increase) in assets, net
of assets acquired from VCA
|
||||||||||||
|
Patient and other accounts
receivables
|
(283 | ) | (1,170 | ) | (378 | ) | ||||||
|
Prepaids and other current
assets
|
(1,187 | ) | (643 | ) | (1,040 | ) | ||||||
|
Other assets
|
(472 | ) | (162 | ) | (122 | ) | ||||||
|
(Decrease) increase in
liabilities, net of liabilities acquired from VCA
|
||||||||||||
|
Accounts payable
|
(7 | ) | 958 | (271 | ) | |||||||
|
Accrued
liabilities
|
(2,865 | ) | (1,097 | ) | 285 | |||||||
|
Due to medical
practices
|
70 | (2,689 | ) | 4,744 | ||||||||
|
Attain IVF Refund Program
deferred revenue and other patient
deposits
|
2,125 | 677 | 2,217 | |||||||||
|
Net
cash provided by operating activities
|
13,764 | 6,867 | 15,956 | |||||||||
|
Cash
flows from investing activities:
|
||||||||||||
|
Purchase
of business service rights
|
(3,550 | ) | (950 | ) | (2,653 | ) | ||||||
|
Cash
paid to purchase VCA, net of cash acquired
|
(119 | ) | (25,409 | ) | ||||||||
|
Purchase
of other intangibles, net
|
50 | (40 | ) | |||||||||
|
Purchase
of fixed assets and leasehold improvements, net
|
(5,909 | ) | (5,695 | ) | (6,222 | ) | ||||||
|
Net
cash used in investing activities
|
(9,459 | ) | (6,714 | ) | (34,324 | ) | ||||||
|
Cash
flows from financing activities:
|
||||||||||||
|
Proceeds from issuance of
debt
|
7,880 | 25,000 | ||||||||||
|
Debt repayments
|
(3,750 | ) | (3,648 | ) | (15,163 | ) | ||||||
|
Common Stock transactions,
net
|
37 | 150 | 87 | |||||||||
|
Net
cash provided by (used in) financing activities
|
(3,713 | ) | 4,382 | 9,924 | ||||||||
|
Net
increase (decrease) in cash and cash equivalents
|
590 | 4,535 | (8,444 | ) | ||||||||
|
Cash
and cash equivalents at beginning of year
|
28,275 | 23,740 | 32,184 | |||||||||
|
Cash
and cash equivalents at end of year
|
$ | 28,865 | $ | 28,275 | $ | 23,740 | ||||||
|
Supplemental
Information:
|
||||||||||||
|
Interest paid
|
$ | 1,067 | $ | 1,632 | $ | 1,024 | ||||||
|
Income taxes paid
|
$ | 3,896 | $ | 1,526 | $ | 1,130 | ||||||
See
accompanying notes to the consolidated financial statements.
F-6
INTEGRAMED
AMERICA, INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
NOTE
1 — THE COMPANY:
IntegraMed
America, Inc. is a specialty healthcare services company offering products and
services to patients and providers in the fertility and vein care segments of
the health industry.
As of
December 31, 2009, our fertility line of business encompassed two of our
reporting segments and was comprised of 39 contracted fertility centers in major
markets across the United States, with products and services designed to support
fertility center growth, assist patients in obtaining third-party treatment
financing, an Attain IVF Refund Program and Attain IVF Multi-Cycle Program and
captive insurance offerings. We offer a comprehensive array of defined business
services to 14 of these contracted fertility centers under our Partner program,
and a more discrete menu of services to 25 other fertility centers under our
Affiliate program. All 39 centers have access to our consumer services offerings
which are comprised of our Attain IVF program and patient financing
products.
In late
2008, our Consumer Services Division re-launched its successful Shared Risk
Refund program under the name “Attain IVF”. This re-branding was done to reflect
advantages offered by the program beyond its basic risk sharing features and to
position the program in a leadership role among smaller, similar programs
offered by other providers.
Our Vein
Clinic Division, which began operations in August, 2007, is currently comprised
of 34 (32 as of December 31, 2008) vein clinics in major markets, which
primarily provide advanced treatment for vein diseases. We offer a comprehensive
array of defined business services to these clinics which are designed to
support their operations and growth.
NOTE
2 ¾ SUMMARY OF
SIGNIFICANT ACCOUNTING POLICIES:
|
|
Basis of consolidation
—
|
The
consolidated financial statements comprise the accounts of IntegraMed America,
Inc. and its wholly owned subsidiaries. With the acquisition of Vein Clinics of
America (VCA) in the third quarter of 2007, we reorganized our service offerings
into three major product lines, Fertility Centers, Consumer Services and Vein
Clinics. In our Fertility Centers Segment, we derive our revenues from business
service contracts with independent fertility centers. Our Consumer Services
Segment derives its revenues from fees assessed to patients enrolling in our
Attain IVF Programs, fees assessed to affiliated fertility clinics, and referral
fees derived from fertility patient financing products. Our Vein Clinics Segment
derives revenues from billings to patients and third party payers for treatment
services rendered based upon the amount billed to the patient or their payer
less any expected contractual allowances resulting from specified rates
contained within payer contracts.
We
evaluate whether we should report the results of the clinical operations in
which we have management service contracts in accordance with ASC 810. Since we
do not have a controlling financial interest in any of the fertility medical
practices to which we provide services, and we are not the primary beneficiary
or obligor of their financial results (our contracts provide for the physician
owners of the clinics to receive any excess or deficit of profits), we do not
consolidate their results. This is further supported by the facts that the
physician owners of the clinics have the voting rights with respect to the
entity and sufficient equity interests to fund their entity. We do
have effective voting control and a controlling financial interest in the
operations of each of the vein clinics, where we are the primary beneficiary and
obligor of their financial results (our contracts provide for us to receive any
excess or deficit of profits) and therefore consolidate the results of those
clinic operations. Accordingly, we report the revenue for patient services only
from the vein clinic segment and those fertility patients who enroll in the
Attain IVF Refund Program or Attain IVF Multi-Cycle Program (included in our
consumer services segment).
F-7
INTEGRAMED
AMERICA, INC.
NOTES
TO FINANCIAL STATEMENTS (Continued)
|
|
Stock split effected in the
form of a stock dividend —
|
In May
2007, we effected a 25% stock split in the form of a stock dividend. Where
applicable, we have restated our capital accounts, shares outstanding, weighted
average shares and earnings per share calculations for all years in these
financial statements and related footnotes to reflect these transactions.
Revenue Recognition —
Fertility
Centers - Partner service fees
Generally
under our current fertility Partner agreements, we receive as compensation for
our services a three-part fee comprised of: (i) a tiered percentage of the
fertility center’s net revenues, (ii) reimbursed costs of services (costs
incurred in servicing a fertility center and any costs paid on behalf of the
fertility center) and (iii) either a fixed percentage ranging from 10% to
20%, or a fixed dollar amount (limited to $1,071,000 for the year ended
December 31, 2008 at our largest fertility center) of the fertility
center’s earnings after service fees, which may be subject to further limits.
However, under our current Partner agreement with Fertility Centers of Illinois,
we do not receive a three-part fee. Rather, effective as of November 1,
2009, we receive a fee that is generally equal to the operating expenses
associated with managing Fertility Centers of Illinois’ medical practice plus
9.5% of such expenses. Our revenues from Fertility Centers of Illinois prior to
November 1, 2009 were, pursuant to our current Partner agreement with
Fertility Centers of Illinois, set at a fixed annual amount paid
monthly.
All
revenues from Partner contracts are recorded in the period services are
rendered. Direct costs incurred by us in performing our services and costs
incurred on behalf of the medical practices are reported as costs of services.
Revenue and costs are recognized in the same period in which the related
services have been performed.
Consumer
Services - Affiliate Service Fee
Under our
Affiliate agreements, we receive as compensation for our services a fixed fee
dependent upon the level of service provided. All revenues and costs from
Affiliate contracts are recorded in the period services are
rendered.
Consumer
Services – Attain IVF Programs
The
Attain IVF Programs consist of fertility treatment packages that
includes a fixed number of treatment cycles for one fixed price. We receive
payment directly from consumers who qualify for the program and the patient
contracts with us to provide the medical treatment. We discharge the
obligation of patient treatment by arranging with affiliated fertility clinics
for the provision of patient care. We pay contracted fertility
centers a defined reimbursement for each treatment cycle performed. Since the
Company is the primary obligor in the arrangement, has latitude in establishing
the price, performs a portion of the contracted service, has discretion in
supplier selection, the amount earned by the Company per transaction is not
fixed and the patient looks to the Company as the contracting party, these
arrangements qualify for gross accounting under ASC 605. We have
revised our revenue recognition policy and have restated all periods presented
to reflect the revised revenue recognition policy described below. We
have two principal programs in this program, the Attain IVF Refund Program and
the Attain IVF Multi-Cycle Program.
Consumer
Services – Attain IVF Refund Program
Under our
Attain IVF Refund Program, by contract, a portion of the enrollment fee
(generally 30%) is non-refundable and is recognized as revenue based on the
relative fair value of each treatment cycle completed relative to the total fair
value of the contracted treatment package available to the patient, following
the guidance of ASC 605. The remaining revenue, which consists of the 70%
refundable portion as well as any part of the 30% non-refundable portion
F-8
not yet
recognized as revenue, is recorded upon the patient becoming pregnant and
achieving a fetal heartbeat (most of the patients that are pregnant at this
point go on to deliver a baby). We are able to record income at the
time of pregnancy as we have substantially completed our obligation to the
patient, discharged the patient from the care of the fertility
specialists, and we can accurately estimate the amount of expenses
and refunds that will become due if there is a pregnancy loss. We are
able to make these estimates for pregnancy loss based upon reliable Company
specific data with respect to the large homogeneous population we have served
for more than seven years. Expenses prior to pregnancy related to the program
and principally paid to the affiliated fertility clinic are recorded as
incurred.
Accordingly,
at each balance sheet date, we have established a liability for patients in the
Attain IVF Refund Program for the following:
|
1.
|
Deposits
for customers who have not yet begun treatment and for whom no revenue has
been recognized (we expect such amounts to be recognized as income or
refunded within twelve to eighteen
months)
|
|
2.
|
Refund
reserve for those patients who became pregnant, but may not
deliver a baby (See Note 12)
|
|
3.
|
Medical
costs associated with additional treatments to a patient who became
pregnant, did not deliver a baby and still has additional treatments
available under their treatment package. (See Note
12)
|
The table
below presents the balances of each of these liabilities as of the respective
dates (000’s):
|
December
31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Deposits
or refundable fees
|
$ | 11,648 | $ | 9,981 | ||||
|
Refund
reserve for pregnant patients
|
300 | 397 | ||||||
|
Medical
cost reserve
|
344 | 331 | ||||||
Due to
the characteristics of the program, we assume the risk for a patient’s treatment
cost in excess of their enrollment fee should initial treatment cycles be
unsuccessful. In order to moderate and manage this risk, we have
developed a sophisticated statistical model and case management program in which
Attain IVF patients are medically pre-approved prior to enrollment in the
program. We also continuously review their clinical criteria as they undergo
treatment. If, while undergoing treatment, a patient’s clinical
response falls outside our criteria for participation in the Attain IVF program,
we have the right to remove that individual from the program, with an applicable
refund to the patient. To date, our case management process has been effective
in managing the risks associated with our Attain IVF program within expected
limits. A patient may withdraw from the program at anytime and will
be issued a refund.
Consumer
Services — Attain IVF Multi-Cycle Program
Patients
enrolled in our Attain IVF Multi-Cycle Program pay us a single fee, which is
slightly less than the average cost of two fresh IVF cycles, in return for up to
four treatment cycles (consisting of two fresh IVF cycles and two frozen embryo
transfers). Our Attain IVF Multi-Cycle Program offers a refund ranging from 10%
to 85% of the contract amount depending on where in the process either we or the
patient elects to terminate the program, as long as termination is prior to a
second fresh IVF cycle. With respect to our Attain IVF Multi-Cycle Program, we
recognize a pro rata share of the contract amount as revenue as each treatment
cycle is completed. The refundable portion of the program contract amount is
recognized as revenue when the patient becomes pregnant. Under such revenue
recognition methodology, we never recognize more revenue than the potential
refundable amount under the program. At the time of pregnancy, we establish a
reserve for future medical costs should the patient miscarry and require
additional contracted treatment cycles.
Consumer
Services - Pharmaceutical Sales
Marketing
fees associated with third-party pharmaceutical sales are recorded upon shipment
to customers. Our revenues for the periods presented are comprised of these
marketing fees and not from the sales of actual pharmaceuticals.
Consumer
Services - Patient Financing
A
fertility treatment cycle can be an expensive process for which many patients do
not have full medical insurance coverage. As a service to these patients, we can
arrange financing to qualified patients of our network at rates significantly
lower than credit cards and other finance companies. Our financing operations
are administered by a third-party vendor and loans are made to qualified
patients by an independent bank or finance organization. We are
F-9
not at
risk for loan losses and receive a placement fee from the lender
involved. Since many financing transactions are closely associated
with our Attain IVF Programs, financing revenues, which we receive and record at
the time the loans are closed, are reported as part of those
programs.
Vein
Clinics – Patient Revenues and Accounts Receivable and Allowance for
Uncollectible Accounts
Our
relationship with the individual medical practices comprising our vein care
division meets the test for consolidation under ASC 810. Among these tests is
the fact that we hold a controlling financial interest in the medical practices,
we are the primary beneficiary of the results of the practices and we are
obligated to absorb any losses of the practices. As a result of these
relationships, we consolidate the medical practice’s patient revenues in our
financial statements. These revenues are derived from the treatment of
individual patients and revenue is recognized when the services are performed,
net of estimated contractual allowances.
The
medical practices have agreements with third-party payers that provide for
payments at amounts different from established rates. Payment arrangements
include prospectively determined rates for reimbursed cost and discounted
charges. Revenue is reported at the estimated net realizable amounts from
patients and third-party payers.
A summary
of the payment arrangements with major third-party payers follows:
|
·
|
Medicare: All
outpatient services related to Medicare beneficiaries are paid based on a
fixed physician fee schedule per service which is updated
annually.
|
|
|
|
|
·
|
Other: Estimates
for contractual allowances under managed care health plans are based
primarily on the payment terms of contractual arrangements, such as
predetermined rates per diagnosis, per diem rates or discounted fee for
service rates.
|
|
|
|
Approximately 15% and 17%
respectively, of gross patient revenues of the Vein Clinics Division for the
years ended December 31, 2009 and 2008, respectively, related to services
rendered to patients covered by the Medicare program.
Laws and
regulations governing the Medicare program are complex and subject to
interpretation. Management believes that it is in compliance with all
applicable laws and regulations and is not aware of any pending or threatened
investigations involving allegations of potential wrongdoing. While no
such regulatory inquiries have been made, compliance with such laws and
regulations can be subject to future government review and
interpretation.
Our
accounts receivable are primarily comprised of patient and third-party
receivables arising from services provided by our vein care division.
Receivables due from third-party payers are carried at an estimated collectible
value determined by the original charge for the service provided, less an
estimate for contractual allowances or discounts provided to the third-party
payers. Receivables due directly from patients are carried at the original
charge for the service provided less an estimated allowance for uncollectible
amounts. Contractual allowance and uncollectible reserve amounts are determined
based on historical collection performance data and are reviewed and adjusted
monthly as necessary.
Vein Clinics – Deferred Compensation
Arrangements
The
Professional Corporations providing medical services at the clinics have entered
into employment agreements with physicians at clinic sites providing for
multi-year bonus compensation to be accumulated over a physician’s first five
years of employment. Accumulated balances are paid out during the years
following this period, or after specific performance targets have been
met. These obligations are funded in physician designated investment
accounts on a quarterly basis. At December 31, 2009 and 2008, these
balances, which are included in other assets, totaled approximately $1,118,000
and $938,000 respectively.
F-10
INTEGRAMED
AMERICA, INC.
NOTES
TO FINANCIAL STATEMENTS (Continued)
Intangible and Long-Lived
Assets –
Our
intangible assets are comprised of Business Service Rights associated with our
fertility Partner contracts, Goodwill associated with our acquisition of Vein
Clinics of America, Inc., and Trademarks, also principally associated with our
Vein Clinic acquisition.
Business
service rights represent payments we made for the right to service certain
fertility centers. We amortize our non-refundable Business Service Rights on a
straight-line basis over the life of the underlying contract, usually ten to
twenty five years. Our refundable Business Service Rights are not amortized as
they are contractually reimbursable from the medical practice upon termination
of the underlying contract. Our Goodwill and Trademark assets associated with
the Vein Clinics of America, Inc. acquisition are deemed to have indefinite
lives and are therefore not amortized.
We test
all of our intangible and long-lived assets for impairment on a regular basis in
accordance with ASC 360. If we record an impairment loss, it may have
a material adverse effect on our results of operations for the year in which the
impairment is recorded. As of December 31, 2009 and 2008, none of our long lived
assets were deemed to be impaired.
Use
of Estimates –
The
preparation of financial statements in conformity with accounting principles
generally accepted in the United States of America requires management to make
estimates and assumptions that affect the reported amounts of assets and
liabilities and disclosure of contingent assets and liabilities at the date of
the financial statements and the reported amounts of revenues and expenses
during the reporting periods. Actual results could differ from those estimates.
The use of estimates and assumptions in the preparation of the accompanying
consolidated financial statements is primarily related to the determination of
accounts receivable reserves as well as refunds due to pregnancy losses and
subsequent medical costs within our Attain IVF Refund Programs.
Due to Medical Practices
—
Due to
Medical Practices represents the net amounts owed by us by contracted medical
practices in our Fertility Partner Program. This balance is comprised of amounts
due to us by the medical practices for funds which we advanced for use in
financing their accounts receivable, less balances owed to the medical practices
by us for undistributed physician earnings and patient deposits we hold on
behalf of the medical practices.
Cash
and cash equivalents ¾
Cash and
cash equivalents primarily include all highly liquid debt instruments with
original maturities of three months or less, recorded at cost, which
approximates market.
Prepaid
Marketing¾
Prepaid
marketing consists of two items, those expenses which relate to the following
period which were paid prior to the end of the current period, and direct
response advertising costs, which we capitalize and then expense over the life
of the expected benefit from the advertising costs in accordance with ASC
720. Direct response advertising costs reported as part of “Prepaid
marketing” are $1,342,000 and $0 as of December 31, 2009 and 2008,
respectively. Total advertising expenses were $5,852,000 and
$3,683,000 in 2009 and 2008, respectively.
Concentrations of credit risk
—
Financial
instruments, which potentially expose us to concentrations of credit risk,
consist primarily of trade receivables from patients and third-party payers
which totaled approximately $13.2 million as of both December 31, 2009 and 2008.
Our related reserves for uncollectible accounts and contractual allowances
totaled $6.2 million and $6.5 million as of December 31, 2009 and 2008,
respectively (see Note 6).
F-11
INTEGRAMED
AMERICA, INC.
NOTES
TO FINANCIAL STATEMENTS (Continued)
Income
taxes ¾
We
account for income taxes utilizing the asset and liability approach in
accordance with FASB ASC 740 “Income Taxes.” Deferred tax assets and
liabilities are recognized on differences between the book and tax basis of
assets and liabilities using presently enacted tax rates. The income tax
provision is the sum of the amount of income tax paid or payable for the year as
determined by applying the provisions of enacted tax laws to the taxable income
for that year and the net change during the year in our deferred tax assets and
liabilities. (See Notes 15 and 16).
Earnings per share —
We
determine earnings per share in accordance with ASC 260. Basic earnings per
share is calculated by dividing net income by the weighted average number of
common shares outstanding during the reporting period. Diluted
earnings per share is calculated by dividing net income by the weighted average
number of common shares, and potential common shares, outstanding during the
reporting period. (See Note 17)
Fair value of financial
instruments —
The fair
value of a financial instrument, such as a notes payable, represents the amount
at which the instrument could be exchanged in a current transaction between
willing parties, other than in a forced sale or liquidation. Significant
differences can arise between the fair value and carrying amounts of financial
instruments that are recorded at historical cost amounts. We believe that the
carrying amounts of cash and cash equivalents, our accounts receivable and
accounts payable approximate fair value due to their short-term
nature.
As of
December 31, 2009 and 2008, the carrying amount of our long-term liabilities
approximates the fair value of such instruments based upon our best estimate of
interest rates that would be available to us for similar debt obligations with
similar maturities.
New
accounting pronouncements ¾
Recently
Issued Accounting Pronouncements
ASU
2009-13: In October 2009, the FASB issued FASB ASU 2009-13,
Revenue Recognition (Topic 605) – Multiple-Deliverable Revenue Arrangements. The
consensus in ASU 2009-13 supersedes certain guidance in ASC 605-25, Revenue Recognition – Multiple
Element Arrangements, and requires an entity to allocate arrangement
consideration at the inception of an arrangement to all of its deliverables
based on their relative selling prices (i.e., the relative-selling-price
method). The consensus eliminates the use of the residual method of
allocation (i.e., in which the undelivered element is measured at its estimated
selling price and the delivered element is measured as the residual of the
arrangement consideration) and requires the relative-selling-price method in all
circumstances in which an entity recognizes revenue for an arrangement with
multiple deliverables subject to ASC 605-25.
When
applying the relative-selling-price method, the determination of the selling
price for each deliverable must be consistent with the objective of determining
vendor-specific objective evidence of fair value (VSOE); that is, the price at
which the entity does or would sell the element on a stand-alone
basis. This determination requires the use of a hierarchy designed to
maximize the entity’s use of available, objective evidence to support its
selling price. The entity must consider market conditions as well as
entity-specific factors when estimating this selling price.
The
amendments in ASU 2009-13 require both ongoing disclosures regarding an entity’s
multiple-element revenue arrangements as well as certain transitional
disclosures during periods after adoption. The objective of the
ongoing disclosures is to provide information regarding the significant
judgments and estimates made and their impact on revenue
recognition. Additionally, disclosures will be made when changes in
either those judgments or the application of the relative-selling-price method
may significantly affect the timing or amount of revenue
recognition. An entity will be required to aggregate these
disclosures for similar types of arrangements.
F-12
INTEGRAMED
AMERICA, INC.
NOTES
TO FINANCIAL STATEMENTS (Continued)
All
entities must have adopted the guidance in ASU 2009-13 no later than the
beginning of their first fiscal year beginning on or after June 15,
2010. We are currently evaluating the impact, if any, ASU 2009-13
will have on our financial statements.
Adopted
Accounting Pronouncements:
Effective
July 1, 2009, the Company adopted The “FASB Accounting Standards
Codification” and the Hierarchy of Generally Accepted Accounting
Principles (ASC 105). This standard establishes only two
levels of U.S. generally accepted accounting principles (“GAAP”), authoritative
and nonauthoritative. The FASB Accounting Standards Codification (the
“Codification”) became the source of authoritative, nongovernmental GAAP, except
for rules and interpretive releases of the SEC, which are sources of
authoritative GAAP for SEC registrants. All other non-grandfathered, non-SEC
accounting literature not included in the Codification became nonauthoritative.
The Company began using the new guidelines and numbering system prescribed by
the Codification when referring to GAAP in the third quarter of fiscal 2009. As
the Codification was not intended to change or alter existing GAAP, it did not
have any impact on the Company’s Consolidated Financial Statements.
Effective
April 1, 2009, the Company adopted three accounting standard updates which
were intended to provide additional application guidance and enhanced
disclosures regarding fair value measurements and impairments of securities.
They also provide additional guidelines for estimating fair value in accordance
with fair value accounting. The first update, as codified in ASC 820-10-65,
provides additional guidelines for estimating fair value in accordance with fair
value accounting. The second accounting update, as codified in ASC 320-10-65,
changes accounting requirements for other-than-temporary-impairment (OTTI) for
debt securities by replacing the current requirement that a holder have the
positive intent and ability to hold an impaired security to recovery in order to
conclude an impairment was temporary with a requirement that an entity conclude
it does not intend to sell an impaired security and it will not be required to
sell the security before the recovery of its amortized cost basis. The third
accounting update, as codified in ASC 825-10-65, requires fair value disclosures
in the interim periods as well as in the annual financial statements. These
updates were effective for fiscal years and interim periods ended after
June 15, 2009. The adoption of these accounting updates did not have any
impact on the Company’s Consolidated Financial Statements.
Effective
April 1, 2009, the Company adopted a new accounting standard for subsequent
events, as codified in ASC 855-10. The update modifies the names of the two
types of subsequent events either as recognized subsequent events (previously
referred to in practice as Type I subsequent events) or non-recognized
subsequent events (previously referred to in practice as Type II subsequent
events). In addition, the standard modifies the definition of subsequent events
to refer to events or transactions that occur after the balance sheet date, but
before the financial statements are issued (for public entities) or available to
be issued (for nonpublic entities). It also requires the disclosure of the date
through which subsequent events have been evaluated. The update did not result
in significant changes in the practice of subsequent event disclosures, and
therefore the adoption did not have any impact on the Company’s Consolidated
Financial Statements (see Note 24 for disclosure).
Subsequent Events
—
The
Company has evaluated subsequent events through the date the financial
statements were issued, and determined that any events or transactions occurring
during this period that would require revenue recognition or disclosure are
appropriately addressed in these financial statements.
F-13
INTEGRAMED
AMERICA, INC.
NOTES
TO FINANCIAL STATEMENTS (Continued)
NOTE
3 ¾ SIGNIFICANT
SERVICE CONTRACTS:
For one
or more of the years ended December 31, 2009, 2008 and 2007, the following
contracted fertility centers each individually provided greater than 10% of our
revenues, net and/or contribution as follows:
|
Percent
of Company
Revenues,
net
|
Percent
of
Contribution
|
|||||||||||||||||||||||
|
2009
|
2008
|
2007
|
2009
|
2008
|
2007
|
|||||||||||||||||||
|
R.S.C.
of New England
|
7.2 | 7.2 | 8.9 | 9.1 | 8.7 | 10.5 | ||||||||||||||||||
|
Fertility
Centers of Illinois
|
13.1 | 16.3 | 19.2 | 10.3 | 15.2 | 17.6 | ||||||||||||||||||
|
Shady
Grove Fertility Center
|
17.6 | 18.0 | 21.3 | 15.4 | 17.2 | 21.4 | ||||||||||||||||||
Under
most of our fertility Partner agreements, we receive as compensation for our
services a three-part fee comprised of: a percentage of the fertility
center’s net revenues; reimbursed costs of services (costs incurred in servicing
a fertility center and any costs paid on behalf of the fertility center); and
either a fixed percentage, or a fixed dollar amount, of the fertility center’s
earnings after services fees, which may be subject to further
limits.
The third
tier of our fee structure under these significant Partner agreements is as
follows:
|
•
|
RSC
of New England — a fixed annual percentage of the center’s
earnings.
|
|
|
•
|
Shady
Grove — a fixed dollar amount of the center’s earnings subject to a
fixed percentage of the center’s earnings limitation. The upper boundary
of the calculation is $1,351,000 and the lower boundary of the calculation
is $540,000.
|
Under our
current Partner agreement with Fertility Centers of Illinois, however, we do not
receive a three-part fee. Rather, effective as of November 1, 2009, we
receive a fee that is generally equal to the operating expenses associated with
managing Fertility Centers of Illinois’ medical practice plus 9.5% of such
expenses. Our revenues from Fertility Centers of Illinois prior to
November 1, 2009 were, pursuant to our current Partner agreement with
Fertility Centers of Illinois, set at a fixed annual amount paid
monthly.
NOTE
4 ¾ SEGMENT
INFORMATION:
We follow
the requirements contained in ASC 280, with respect to identifying and reporting
business segments. This statement requires that segment reporting reflect our
organizational structure, major revenue sources, lines of responsibility and
senior management’s perspective of an organization. With the
acquisition of Vein Clinics of America (VCA) during the third quarter of 2007,
we reorganized our service offerings into three major product
lines: Fertility Centers, Consumer Services and Vein
Clinics. Each of the operating segments includes an element of
overhead specifically associated with it. Such overhead costs were previously
reported as General and Administrative costs, and have been reclassified in all
periods presented to better reflect the operating results of our business
segments.
F-14
INTEGRAMED
AMERICA, INC.
NOTES
TO FINANCIAL STATEMENTS (Continued)
Performance
by segment, for the three years ended December 31, 2009, 2008 (restated) and
2007 (restated) are presented below (000’s omitted):
|
Fertility
Centers
|
Consumer
Services
|
Vein
Clinics
(1)
|
Corp.
G&A
|
Consolidated
|
||||||||||||||||
|
For
the Year ended December 31, 2009
|
||||||||||||||||||||
|
Revenues
|
$ | 145,309 | $ | 20,826 | $ | 50,625 | $ | — | $ | 216,760 | ||||||||||
|
Cost of Services
|
$ | 133,706 | 15,639 | 46,525 | — | 195,870 | ||||||||||||||
|
Contribution
|
11,603 | 5,187 | 4,100 | — | 20,890 | |||||||||||||||
|
Operating Margin
|
8.0 | % | 24.9 | % | 8.1 | % | — | 9.6 | % | |||||||||||
|
General and
administrative
|
— | — | — | 12,155 | 12,155 | |||||||||||||||
|
Interest (income) expense,
net
|
(149 | ) | — | — | 1,059 | 910 | ||||||||||||||
|
Income (loss) before income
taxes
|
$ | 11,752 | $ | 5,187 | $ | 4,100 | $ | (13,214 | ) | $ | 7,825 | |||||||||
|
Depreciation expense included
above
|
$ | 4,076 | $ | 0 | $ | 873 | $ | 873 | $ | 5,822 | ||||||||||
|
Capital expenditures,
net
|
$ | 4,173 | $ | 0 | $ | 947 | $ | 790 | $ | 5,910 | ||||||||||
|
Total assets
|
$ | 39,086 | $ | 104 | $ | 49,845 | $ | 35,274 | $ | 124,309 | ||||||||||
|
For
the Year ended December 31, 2008, restated
|
||||||||||||||||||||
|
Revenues
|
$ | 138,440 | $ | 19,763 | $ | 39,950 | $ | — | $ | 198,153 | ||||||||||
|
Cost of Services
|
128,224 | 14,344 | 37,299 | — | 179,867 | |||||||||||||||
|
Contribution
|
10,216 | 5,419 | 2,651 | — | 18,286 | |||||||||||||||
|
Operating Margin
|
7.4 | % | 27.4 | % | 6.6 | % | — | 9.2 | % | |||||||||||
|
General and
administrative
|
— | — | — | 10,654 | 10,654 | |||||||||||||||
|
Interest (income) expense,
net
|
(181 | ) | — | 8 | 1,353 | 1,180 | ||||||||||||||
|
Income (loss) before income
taxes
|
$ | 10,397 | $ | 5,419 | $ | 2,643 | $ | (12,007 | ) | $ | 6,452 | |||||||||
|
Depreciation expense included
above
|
$ | 4,327 | $ | 3 | $ | 761 | $ | 898 | $ | 5,989 | ||||||||||
|
Capital expenditures,
net
|
$ | 4,053 | $ | — | $ | 1,057 | $ | 585 | $ | 5,695 | ||||||||||
|
Total assets
|
$ | 36,885 | $ | 331 | $ | 46,750 | $ | 37,475 | $ | 121,441 | ||||||||||
|
For
the Year ended December 31, 2007, restated
|
||||||||||||||||||||
|
Revenues
|
$ | 121,078 | $ | 16,460 | $ | 14,284 | $ | — | $ | 151,822 | ||||||||||
|
Cost of Services
|
111,059 | 12,336 | 13,304 | — | 136,699 | |||||||||||||||
|
Contribution
|
10,019 | 4,124 | 980 | — | 15,123 | |||||||||||||||
|
Operating Margin
|
8.3 | % | 25.1 | % | 6.9 | % | — | 10.0 | % | |||||||||||
|
General and
administrative
|
— | — | — | 10,537 | 10,537 | |||||||||||||||
|
Interest (income) expense,
net
|
(203 | ) | — | 2 | 81 | (120 | ) | |||||||||||||
|
Income (loss) before income
taxes
|
$ | 10,222 | $ | 4,124 | $ | 978 | $ | (10,618 | ) | $ | 4,706 | |||||||||
|
Depreciation expense included
above
|
$ | 4,003 | $ | 3 | $ | 255 | $ | 846 | $ | 5,107 | ||||||||||
|
Capital expenditures,
net
|
$ | 4,654 | $ | — | $ | 906 | $ | 662 | $ | 6,222 | ||||||||||
|
Total assets
|
$ | 42,586 | $ | 888 | $ | 44,786 | $ | 25,911 | $ | 114,171 | ||||||||||
(1) Acquired
August 8, 2007.
F-15
INTEGRAMED
AMERICA, INC.
NOTES
TO FINANCIAL STATEMENTS (Continued)
NOTE
5 – CASH AND CASH EQUIVALENTS:
Cash and
cash equivalents consist of cash and short term marketable securities. To the
extent that cash balances exceed short term operating needs, excess cash is
invested in short term interest bearing instruments. It is our policy to
restrict our investments to high-quality securities with fixed maturity dates
and principle amounts. The composition of our cash and short term investments is
as follows (000’s omitted):
|
December
31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Cash
|
$ | 27,393 | $ | 26,807 | ||||
|
Money
market funds
|
58 | 58 | ||||||
|
Certificates
of deposit
|
1,400 | 1,400 | ||||||
|
Accrued
interest income
|
14 | 10 | ||||||
|
Total cash and cash
equivalents
|
$ | 28,865 | $ | 28,275 | ||||
NOTE
6 – PATIENT AND OTHER RECEIVABLES, NET:
Patient
and other receivables are principally comprised of gross patient and insurance
receivables from our Vein Clinics segment which represent outstanding balances
due for patient treatments less estimated allowances for insurance contractual
agreements and uncollectible balances. Insurance contractual
allowances are calculated based on recent allowance trends stratified by major
payer category and uncollectible reserves are based on both historical trends
and specific identification of specific accounts. For the periods ended December
31, 2009 and 2008, we believe that our receivable reserves were adequate to
provide for any contractual or collection issues.
The
composition of our patient and other receivables is as follows (000’s
omitted):
|
December
31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Vein
Clinic patient and insurance receivables
|
$ | 13,073 | $ | 12,865 | ||||
|
Reserve
for insurance contractual allowance
|
(3,300 | ) | (3,866 | ) | ||||
|
Reserve
for uncollectible accounts
|
(2,913 | ) | (2,648 | ) | ||||
|
Subtotal Vein Clinic
receivables, net
|
6,860 | 6,351 | ||||||
|
Other
receivables
|
104 | 330 | ||||||
|
Total Patient and other
receivables, net
|
$ | 6,964 | $ | 6,681 | ||||
NOTE
7 ¾ FIXED
ASSETS, NET:
Fixed
assets, net of depreciation at December 31, 2009 and 2008 consisted of the
following (000’s omitted):
|
2009
|
2008
|
|||||||
|
Furniture,
office and computer equipment
|
$ | 20,923 | $ | 18,311 | ||||
|
Medical
equipment
|
8,626 | 7,396 | ||||||
|
Leasehold
improvements
|
23,112 | 21,059 | ||||||
|
Construction
in progress
|
0 | 63 | ||||||
|
Assets
under capital leases
|
427 | 427 | ||||||
|
Total
|
53,088 | 47,256 | ||||||
|
Less
¾
Accumulated depreciation and amortization
|
(36,383 | ) | (30,638 | ) | ||||
| $ | 16,705 | $ | 16,618 | |||||
F-16
INTEGRAMED
AMERICA, INC.
NOTES
TO FINANCIAL STATEMENTS (Continued)
Our fixed
assets are depreciated on a straight line basis. We generally assign useful
lives of five years to assets classified as furniture, fixtures, office and
medical equipment. Assets classified as computer hardware and software are
generally assigned a three year useful life and leasehold improvements are
depreciated over the lesser of their useful life, or the term of the
lease.
Depreciation
expense on fixed assets for the years ended December 31, 2009 and 2008 was
$5,822,000, and $5,989,000, respectively. Assets under capital lease
are comprised of various medical equipment. Accumulated amortization
related specifically to capital leases at December 31, 2009 and 2008 was
$209,000 and $126,000, respectively.
NOTE
8 ¾ BUSINESS
SERVICE RIGHTS, NET:
Business
Service Rights, net at December 31, 2009 and 2008 consisted of the following
(000’s omitted):
|
2009
|
2008
|
|||||||
|
Business
Service rights, net
|
$ | 37,755 | $ | 34,205 | ||||
|
Less
accumulated amortization
|
(13,545 | ) | (12,249 | ) | ||||
|
Total
|
$ | 24,210 | $ | 21,956 | ||||
Business
Service Rights are negotiated one-time payments we generally make to physician
practices joining our fertility Partner program. These payments are made to
secure the right to provide business services to the practices for contracted
terms generally ranging from ten to twenty five years. Depending upon the
negotiated terms, these payments may be refundable at the termination of the
contract or non-refundable. We amortize our non-refundable Business Service
Rights over the life of the applicable contract. Refundable Business Service
Rights, which totaled approximately $9.6 million as of December 31, 2009, are
not amortized because these amounts will be repaid to us upon termination of the
contract.
For the
twelve months ended December 31, 2009 and 2008, amortization expense related to
our Business Service Rights totaled approximately $1.3 million and $1.3 million,
respectively.
|
|
Amortization
expense of our Business Service Rights in future years are as follows
(000’s omitted):
|
|
2010
|
$ | 1,296 | ||
|
2011
|
1,296 | |||
|
2012
|
1,296 | |||
|
2013
|
1,296 | |||
|
2014
|
1,296 | |||
|
Thereafter
|
8,080 | |||
|
Total
payments
|
$ | 14,560 |
We test
our Business Service Rights for impairment on a regular basis in accordance with
ASC 360. To date, no impairment charges have been recognized.
NOTE
9 – GOODWILL:
On August
8, 2007, IntegraMed acquired 100 percent of the outstanding common shares of
Vein Clinics of America, Inc. (VCA). With this acquisition, IntegraMed became
the country’s leading provider of services to the vein disease segment in the
health care market. At the date of acquisition, VCA operated 27 clinics in 11
states. This acquisition also provided the opportunity for
operational efficiencies in the form of cost reductions through economies of
scale and resource sharing for both organizations. Purchase
accounting principles in accordance with ASC 805 were applied and accordingly,
only the results of VCA operations subsequent to its acquisition are included in
the accompanying financial statements.
F-17
INTEGRAMED
AMERICA, INC.
NOTES
TO FINANCIAL STATEMENTS (Continued)
The
goodwill of $30.3 million arising from this acquisition consists largely of the
market potential expected from the operations and enhanced resources of VCA. All
of this goodwill was assigned to VCA’s vein care operations, with none of the
goodwill expected to be deductible for income tax purposes.
The
following pro forma data reflects the consolidated revenue and earnings of
IntegraMed America, Inc, and Subsidiaries had the VCA acquisition date been
January 1, 2007 (000’s omitted):
|
Revenue
|
Net
Income
|
Basic
Earnings
Per
share
|
||||||||||
|
Supplemental
pro forma results for 01/01/2007 to 12/31/2007
|
$ | 171,925 | $ | 3,464 | $ | 0.41 | ||||||
We test
our goodwill for impairment in accordance with the provisions of ASC 350. This
test consists of a two-step process. The first step is to identify potential
impairment by comparing the fair value of the underlying asset with its carrying
amount. If the fair value, which is based on future cash flows, exceeds the
carrying amount, the intangible asset is not considered impaired. If the
carrying amount exceeds the fair value, the second step must be performed to
measure the amount of the impairment loss, if any. The second step compares the
implied fair value of the intangible with the carrying amount of that
intangible. If the implied fair value is less than the carrying amount, an
impairment loss would be recognized in an amount equal to the excess of the
carrying amount of the intangible over its implied fair value. To date we have
not recorded any impairment losses.
NOTE
10 ¾
TRADEMARKS:
Trademarks
and other intangibles, net at December 31, 2009 and 2008 consisted of the
following trademark items (000’s omitted)
|
2009
and 2008
|
||||
|
IntegraMed
America, Inc.
|
$ | 42 | ||
|
Vein
Clinics of America, Inc.
|
4,400 | |||
|
Total
|
$ | 4,442 | ||
We do not
amortize our trademarks as they have an indefinite useful life. We do test our
trademarks for impairment on a regular basis in accordance with ASC 360. To
date, no impairment charges have been recognized.
NOTE
11 ¾ OTHER
ASSETS:
As of
December 31, 2009 and 2008, other assets consisted of the following (000’s
omitted):
|
2009
|
2008
|
|||||||
|
Physician
investment accounts – supporting VCA
deferred compensation
arrangements
|
$ | 1,000 | $ | 863 | ||||
|
Security
deposits
|
847 | 858 | ||||||
|
Deferred
offering costs
|
346 | — | ||||||
|
Other
|
60 | 60 | ||||||
| $ | 2,253 | $ | 1,781 | |||||
F-18
Deferred
offering costs totaled $0.3 million and $0 as of December 31, 2009 and 2008,
respectively. These costs primarily represent legal, accounting and other direct
costs related to our efforts to raise capital through a secondary offering and
have been deferred based on the guidance provided in SEC Staff Accounting
Bulletin (SAB) Topic 5A “Deferred Offering Costs”. We completed this offering
during the first quarter of 2010, at which time these costs will be reclassified
to additional paid-in capital as a reduction of the offering proceeds (see Note
24).
NOTE
12 ¾ ACCRUED
LIABILITIES:
Accrued
liabilities at December 31, 2009 and 2008 (restated) consisted of the following
(000’s omitted):
|
2009
|
2008
|
|||||||
|
Accrued
payroll
|
$ | 2,251 | $ | 1,665 | ||||
|
Accrued
employee incentives and benefits
|
4,177 | 3,072 | ||||||
|
Accrued
vacation
|
139 | 151 | ||||||
|
Accrued
physician incentives (VCA)
|
3,160 | 2,754 | ||||||
|
New
physician recruitment
|
50 | 113 | ||||||
|
Accrued
costs on behalf of medical practices
|
1,454 | 1,894 | ||||||
|
Accrued
rent
|
1,108 | 1,166 | ||||||
|
Accrued
professional fees
|
878 | 250 | ||||||
|
Accrued
insurance
|
78 | 1,246 | ||||||
|
Reserves
for estimated Attain IVF patient refunds
|
300 | 397 | ||||||
|
Reserve
for Attain IVF post-pregnancy medical costs
|
344 | 331 | ||||||
|
Accrued
federal and state taxes
|
157 | 2,899 | ||||||
|
Other
accrued taxes
|
180 | 300 | ||||||
|
Other
(1)
|
843 | 1,580 | ||||||
|
Total
accrued liabilities
|
$ | 15,119 | $ | 17,818 | ||||
(1)
Individually represents less than 5% of total accrued liabilities.
NOTE
13 – DUE TO MEDICAL PRACTICES:
Due to
Medical Practices is comprised of the net amounts owed by us to fertility
medical practices contracted as Partners. This balance is comprised of amounts
due to us by the medical practices for funds which we advanced for use in
financing their accounts receivable, less balances owed to the medical practices
by us for undistributed physician earnings and patient deposits we hold on
behalf of the medical practices.
While we
are responsible for the management and collection of the Partner’s accounts
receivable, as part of the business services we provide, the credit and
collection risk for these receivables remains with the medical practice. We
finance the receivables with full recourse. Amounts financed relating to
uncollectible accounts are recovered from the medical practice in the month
uncollectible reserves are established or accounts are written-off.
As of
December 31, 2009 and 2008, Due to Medical Practices was comprised of the
following balances (000’s omitted):
|
2009
|
2008
|
|||||||
|
Advances
to Partners for receivable financing
|
$ | (14,653 | ) | $ | (17,121 | ) | ||
|
Undistributed
physician earnings
|
2,270 | 3,205 | ||||||
|
Physician
practice patient deposits
|
18,807 | 20,270 | ||||||
|
Due
to Medical Practices, net
|
$ | 6,424 | $ | 6,354 | ||||
F-19
Our
responsibilities to the these medical practices are to provide financing for
their accounts receivable and to hold patient deposits on their behalf as well
as undistributed physician earnings.
We are
also responsible for the collection of the Partner accounts receivables, which
we finance with full recourse. We have no other funding commitments to the
Partner.
NOTE
14 ¾ NOTES
PAYABLE AND OTHER OBLIGATIONS:
Notes
payable and other obligations at December 31, 2009 and 2008 consisted of the
following (000’s omitted):
|
2009
|
2008
|
|||||||
|
Note
payable to bank
|
$ | 18,142 | $ | 21,809 | ||||
|
Credit
facility payable to bank
|
7,500 | 29,309 | ||||||
|
Derivative
fair valuation adjustment
|
306 | 609 | ||||||
|
Obligations
under capital lease
|
218 | 301 | ||||||
|
Total
notes payable and other obligations
|
$ | 26,166 | $ | 30,219 | ||||
|
Less
¾ Current
portion
|
(11,317 | ) | (11,351 | ) | ||||
|
Long-term
notes payable and other obligations
|
$ | 14,849 | $ | 18,868 | ||||
Note payable to Bank ¾
In August
2007, as part of our acquisition of Vein Clinics of America, we secured a new
$25.0 million 5-year term loan. Our previous term loan of $7.7 million was paid
off in its entirety as part of this agreement. After deducting the previous loan
amount, interest and fees, our net funding from Bank of America was $17.0
million. Other features of this credit facility include a $10 million three-year
revolving line of credit. Availability of borrowings under the working capital
revolver is based on eligible accounts receivable, as defined in the credit
agreement. As of December 31, 2009, the full amount of $10.0 million was
available under the revolving line of credit, of which $7.5 million was
outstanding.
Each
component of our amended credit facility bears interest by reference, at our
option, to Bank of America’s prime rate minus a margin or to LIBOR plus a
margin. The margin is dependent upon a leverage test, ranging from 2.00% to
2.75% in the case of LIBOR-based term loans and 0.0% to 0.50% in the case of
prime-based term loans. Interest on the revolving line of credit is at the prime
rate less up to 0.50% or at LIBOR plus 1.50% to 2.50% depending on a leverage
test. Interest on the prime-based loans is payable quarterly beginning
November 8, 2007 and interest on LIBOR-based loans is payable on the last
day of each applicable interest period. As of December 31, 2008 and
September 30, 2009, interest on the term loan was payable at a rate of
approximately 2.71% and 2.57%, respectively. Unused amounts under the revolving
line of credit bear a commitment fee of 0.25% and are payable
quarterly.
In order
to mitigate the interest rate risk associated with this term loan, we also
entered into an interest rate swap agreement on 50% of the principal amount
effectively fixing the interest rate at 5.39%, plus the applicable margin, on
that portion of the loan.. Our swap agreement matures in August 2010 and our
term loan matures in August 2012. We deem this hedge to be highly effective over
its useful life as it shares the same amortization schedule as the underlying
debt subject to the hedge and any change in fair value inversely mimics the
appropriate portion of the hedged item. At the expiration of the swap agreement,
or amendment to the term loan, which ever occurs first, we will re-evaluate our
hedge position and modify our disclosures as necessary. See Note
15.
Our Bank
of America credit facility is collateralized by substantially all of our assets.
As of December 31, 2009, we were in full compliance with all applicable debt
covenants. We also continuously review our credit agreements and may renew,
revise or enter into new agreements from time to time as deemed
necessary.
F-20
Debt Maturities ¾
At
December 31, 2009, aggregate note payments, including capital lease obligation
payments, in future years were as follows (000’s omitted):
|
2010
|
$ | 11,317 | ||
|
2011
|
3,816 | |||
|
2012
|
3,766 | |||
|
2013
|
7,267 | |||
|
Total
payments
|
$ | 26,166 |
Leases ¾
Our
capital lease obligation relates to medical equipment acquired for certain vein
clinics.
We
maintain operating leases for our corporate headquarters and for medical office
space for our Partner and our vein clinic centers. We also have
operating leases covering certain medical equipment. Aggregate rental
expense under operating leases was approximately $12.6 million, $11.5 million,
and $10.7 million, for the years ended December 31, 2009, 2008 and 2007,
respectively.
At
December 31, 2009, the minimum lease payments for assets under capital and
non-cancelable operating leases in future years were as follows (000’s
omitted):
|
Capital
|
Operating
|
|||||||
|
2010
|
$ | 102 | $ | 10,340 | ||||
|
2011
|
102 | 10,806 | ||||||
|
2012
|
33 | 10,176 | ||||||
|
2013
|
— | 8,763 | ||||||
|
2014
|
— | 7,050 | ||||||
|
Thereafter
|
¾ | 21,449 | ||||||
|
Total
minimum lease payments
|
$ | 237 | $ | 68,584 | ||||
|
Less
¾ Amount
representing interest
|
19 | |||||||
|
Present
value of minimum lease payments
|
$ | 218 | ||||||
NOTE 15 – OTHER COMPREHENSIVE
LOSS:
We are
exposed to the risk that our earnings and cash flows could be adversely impacted
by market driven fluctuations in the level of interest rates. It is our policy
to manage these risks by using a mix of fixed and floating rate debt and
derivative instruments.
During
the third quarter of 2007, we entered into a revised financing agreement with
Bank of America. This agreement contained an interest rate swap provision
designed to hedge risks associated with $12.5 million of our then
$25.0 million term loan through the maturity date of the swap agreement in
August 2010. As a result of this agreement, our net income for the twelve months
ended December 31, 2008 and 2009 includes additional financing costs of
approximately $260,000 and $390,000, respectively, and we expect to record
additional financing costs of approximately $300,000 related to the swap
agreement over the coming twelve months given current interest rate forecasts
(these financing costs are expected to be offset by lower interest expense on
the portion of the term loan that was not hedged over this same time
frame).
This
agreement qualifies as a cash flow hedge under ASC 820 and as such our
accounting and disclosures will be in accordance with this
Statement.
F-21
As a
result of this agreement, our net income for the twelve months ended December
31, 2009 includes additional financing costs of approximately $390,000, and we
expect to record additional financing costs of approximately $280,000 - $320,000
related to the swap agreement over its remaining life given current interest
rate forecasts. In addition to the costs included in our reported net income,
this hedge has also generated a non-recognized, tax-effected gain of
approximately $188,000 as of December 31, 2009 which is reported as part of our
comprehensive income. The fair value of this hedge was calculated in accordance
with ASC 820, utilizing Level 2 inputs of quoted prices for similar liabilities
in active markets.
We deem
this hedge to be highly effective as it shares the same amortization (at 50% of
the notional amount) schedule as the underlying debt subject to the hedge and
any change in fair value inversely mimics the appropriate portion of the hedged
item. As of December 31, 2009 and 2008, we had no other hedge or derivative
transactions.
As of
December 31, 2009, we had no other hedge or derivative
transactions.
The
following table summarizes total comprehensive income (loss) for the applicable
periods (000's omitted):
|
For
the twelve-month period ending
December
31,
|
||||||||||||
|
2009
|
2008
|
2007
|
||||||||||
|
Net
income as reported
|
$ | 4,494 | $ | 3,915 | $ | 3,044 | ||||||
|
Net
non-recognized gain (loss) on derivative transactions
|
187 | (293 | ) | (73 | ) | |||||||
|
Total
comprehensive income
|
$ | 4,681 | $ | 3,622 | $ | 2,971 | ||||||
NOTE
16 ¾ INCOME
TAXES:
We are
subject to income taxes in various states within the United
States. Judgment is often required in evaluating our provision for
income taxes. During the ordinary course of business, there are
certain transactions for which the ultimate tax determination is
uncertain. For example, certain taxing authorities where we do not
file tax returns may take the position that we are providing services in
jurisdictions where our Partner fertility centers operate and therefore are
subject to the state’s tax laws. We have made provision for estimated
liabilities in such states in accordance with ASC 450. The final
determination of tax audits and any related litigation could be materially
different from our historical income tax provisions and accruals. The
results of an audit or litigation could have a material effect on our operating
results or cash flows.
The
provision for income taxes consisted of the following (000's
omitted):
|
For
the years ended
December
31,
|
||||||||||||
|
2009
|
2008
|
2007
|
||||||||||
|
Current
taxes:
|
||||||||||||
|
Federal
|
$ | 665 | $ | 2,699 | $ | 1,049 | ||||||
|
State
|
396 | 644 | 391 | |||||||||
|
Total current tax
expense
|
$ | 1,061 | $ | 3,343 | $ | 1,440 | ||||||
|
Deferred
taxes:
|
||||||||||||
|
Federal
|
$ | 1,868 | $ | (645 | ) | $ | 178 | |||||
|
State
|
402 | (161 | ) | 44 | ||||||||
|
Total deferred tax expense
(benefit)
|
$ | 2,270 | $ | (806 | ) | $ | 222 | |||||
|
Total
tax provision
|
$ | 3,331 | $ | 2,537 | $ | 1,662 | ||||||
F-22
The
financial statement income tax provision differed from income taxes determined
by applying the statutory federal income tax rate to the financial statement
income before income taxes for the years ended December 31, 2009, 2008 and 2007
primarily as a result of the following (000's omitted):
|
For
the years ended
December
31,
|
||||||||||||
|
2009
|
2008
|
2007
|
||||||||||
|
Provision
at U.S. federal statutory rate
|
$ | 2,661 | $ | 2,192 | $ | 1,599 | ||||||
|
State
income taxes, net of federal tax effect
|
366 | 306 | 135 | |||||||||
|
Non-deductible
expenses
|
118 | 27 | 62 | |||||||||
|
Tax-exempt
interest income
|
|
(14 | ) | (199 | ) | |||||||
|
Other
|
|
|
39 | |||||||||
|
Change
in ASC 740 liability
|
186 | 26 | 27 | |||||||||
|
Income
tax expense
|
$ | 3,331 | $ | 2,537 | $ | 1,663 | ||||||
Significant
components of the deferred tax assets (liabilities) at December 31, 2009 and
2008 were as follows (000's omitted):
|
December
31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Deferred
tax assets
|
||||||||
|
Temporary book to tax
differences
|
$ | 3,426 | $ | 6,055 | ||||
|
Total deferred tax
assets
|
3,426 | 6,055 | ||||||
|
Deferred
tax liabilities
|
||||||||
|
Depreciation and
amortization
|
(2,137 | ) | (737 | ) | ||||
|
Other
|
(307 | ) | (18 | ) | ||||
|
Total deferred tax
liabilities
|
(2,444 | ) | (755 | ) | ||||
|
Net
total deferred tax asset
|
$ | 982 | $ | 5,300 | ||||
We assess
the realizability of our deferred tax assets at each interim and annual balance
sheet date based on actual and forecasted operating results in order to
determine the proper amount, if any, required for a valuation allowance. As a
result of this assessment, we believe that it is more likely than not, given the
weight of available evidence, that all of our deferred tax assets will be
realized. We will continue to assess the realizability of our deferred tax
assets at each interim and annual balance sheet date in order to determine the
proper amount, if any, required for a valuation allowance.
Effective
January 1, 2007, we adopted ASC 740, "Accounting for Uncertainty in Income
Taxes," which clarifies the accounting and disclosure for uncertainty in income
taxes. The adoption of this interpretation did not have a material impact on our
financial statements.
We file
income tax returns in the U.S. federal jurisdiction and various states. For
federal income tax purposes, our 2007, 2008 and 2009 tax years remain open for
examination by the tax authorities under the normal three year statute of
limitations. A federal income tax examination for tax years through 2006 was
completed during 2008 resulting in no adjustment to our income tax liability.
For state tax purposes, our 2005 through 2009 tax years remain open for
examination by the tax authorities under a four year statute of limitations for
many of the states in which we file.
A
reconciliation of the unrecognized tax benefits for the years ended December 31,
2009 and 2008 follows:
F-23
INTEGRAMED
AMERICA, INC.
NOTES
TO FINANCIAL STATEMENTS (Continued)
|
Unrecognized
Tax
Benefits (000s)
|
||||||||
|
2009
|
2008
|
|||||||
|
Balance
as of January 1,
|
$ | 175 | $ | 149 | ||||
|
Additions for current year tax
positions
|
69 | 46 | ||||||
|
Additions for prior year tax
positions
|
117 |
|
||||||
|
Reductions for prior year tax
positions
|
|
(31 | ) | |||||
|
Reductions related to
expirations of statute of limitations
|
(7 | ) |
|
|||||
|
Additional
interest
|
14 | 11 | ||||||
|
Balance
as of December 31,
|
$ | 368 | $ | 175 | ||||
As of
December 31, 2009 and 2008, all of the unrecognized tax benefits could affect
our tax provision and effective tax rate.
In
accordance with our accounting policy, interest expense and penalties related to
income taxes are included in the income tax expense line of our consolidated
statement of operations. For the years ended December 31, 2009 and
2008, we recognized $14,000 and $11,000, respectively, for interest expense
related to uncertain tax positions. As of December 31, 2009 and 2008,
we had recorded liabilities for interest expense related to uncertain tax
positions in the amounts of $77,000 and $33,000, respectively. We
made no accrual for penalties related to income tax positions.
In
conjunction with the preparation of the December 31, 2009 financial statements,
we determined that approximately $856,000 was improperly recognized as a
deferred tax asset when we allocated the purchase price associated with the
acquisition of our Vein Clinics Division (which occurred in August 2007.)
We have concluded that the $856,000 should be recognized as additional goodwill
associated with the acquisition transaction and as of December 31, 2009 has been
recorded as such. The Company concluded that this misclassification
was not material, both qualitatively or quantitatively, to the Company’s
financial position and that this misclassification had no effect on the
statement of operations or cash flows.
NOTE
17 - EARNINGS PER SHARE:
The
reconciliation of the numerators and denominators of the basic and diluted EPS
computations for the years ended December 31, 2009, 2008 (restated) and 2007
(restated) is a follows (000's omitted, except for per share
amounts):
|
For
the years ended
December
31,
|
||||||||||||
|
2009
|
2008
|
2007
|
||||||||||
|
Numerator
|
||||||||||||
|
Net
Income
|
$ | 4,494 | $ | 3,915 | $ | 3,044 | ||||||
|
Denominator
|
||||||||||||
|
Weighted
average shares outstanding
|
8,773 | 8,618 | 8,310 | |||||||||
|
Effect
of dilutive options
|
61 | 73 | 100 | |||||||||
|
Weighted
average shares and dilutive potential Common shares
|
8,834 | 8,691 | 8,410 | |||||||||
|
Basic
earnings per common share
|
$ | 0.51 | $ | 0.45 | $ | 0.37 | ||||||
|
Diluted
earnings per common share
|
$ | 0.51 | $ | 0.45 | $ | 0.36 | ||||||
F-24
INTEGRAMED
AMERICA, INC.
NOTES
TO FINANCIAL STATEMENTS (Continued)
For the
years ended December 31, 2009 and 2008, options to purchase approximately 61,000
and 124,000 shares of common stock, respectively, were excluded from the
computation of diluted earnings per share as the exercise price of the options
was above the average market price of the shares of common stock. For the year
ended December 31, 2007, there were no outstanding options to purchase shares of
common stock which were excluded from the computation of the diluted earnings
per share amount as the exercise price of all outstanding options was less than
the average market price of the shares of common stock.
NOTE
18 ¾
SHAREHOLDERS’ EQUITY:
During
2009, 2008 and 2007, we issued approximately 142,000, 99,000 and 78,000 shares,
respectively, of restricted common stock as deferred compensation to several
officers and directors with an aggregate value of $978,000, $899,000 and
$956,000 respectively. These shares were valued at their fair value on the date
of grant, and are amortized to expense over their vesting period which generally
is a three year period.
During
2008, we issued incentive stock options to purchase approximately 128,000 shares
of common stock to several officers of the company. These options had
an aggregate fair value of approximately $741,000 on the date of issue. These
options have a term of ten years and vest ratably over a four year
period.
Our Board
of Directors has authorized the retirement of common stock held as Treasury
Shares on a periodic basis. As such we retired approximately 14,000 and 5,000
shares of Treasury Stock during the years ended December 31, 2008 and 2007
respectively. No shares were retired during 2009. As of December 31,
2009 there were 46,408 shares of common stock held as Treasury
shares.
In May
2007, we effected a 25% stock split in the form of a stock dividend. Where
applicable we have restated our capital accounts, shares outstanding, weighted
average shares and earnings per share calculations for all years in these
financial statements and related footnotes to reflect these
transactions.
NOTE
19 ¾
STOCK-BASED EMPLOYEE COMPENSATION:
We
account for our stock based employee compensation plans in accordance with ASC
718. ASC 718 addresses the accounting for share based payment transactions in
which an enterprise receives employee services in exchange for equity
instruments of the enterprise or liabilities that are based on the fair value of
the enterprise’s equity instruments or that may be settled by the issuance of
such equity instruments. ASC 718 requires that such transactions be accounted
for using a fair value based method.
In
considering the fair value of the underlying stock when we grant options or
issue restricted stock, we consider several factors including the fair values
established by market transactions. Stock-based compensation includes
significant estimates and judgments of when stock options might be exercised,
forfeiture rates and stock price volatility. The timing of option exercises is
out of our control and depends upon a number of factors including our market
value and the financial objectives of the option holders. These estimates can
have a material impact on our stock compensation expense but will have no impact
on our cash flows.
We
currently have three stock option plans which have been previously approved by
the stockholders. Under the 1992 Incentive and Non-Incentive Stock Option Plan
(the “1992 Plan”), the 2000 Long-term Compensation Plan (the “2000
Plan”) and the 2007 Long-term Compensation Plan (the “2007 Plan”), 500,000,
700,000 and 500,000 shares, subject to adjustment, of common stock,
respectively, were reserved for issuance of incentive and non-incentive stock
options and stock grants. The 1992 Plan expired in May 2002, and
although some options are still outstanding, no further awards may be made under
that plan. Under the 2000 and 2007 Plans, stock options and stock grants may be
F-25
awarded
to employees, directors and such other persons as the Board of Directors
determines will contribute to our success. Vesting periods are set by the Board
of Directors and stock options are generally exercisable during a ten-year
period following the date of award, with stock grants generally vesting in three
to five years. The Board of Directors has the authority to accelerate the
maturity of any stock option or grant at its discretion, and all stock options
and grants have anti-dilution provisions. Under all of our plans,
options expire three months from the date of the holder’s termination of
employment or twelve months in the event of disability or death. As
of December 31, 2009, there were 464,933 shares available for granting under
these plans. We recognize compensation cost for stock option plans over the
vesting period based on the fair value of the option as of the date of the
grant.
The
following table sets forth information about the weighted-average fair value of
options granted during the periods below,
and the assumptions used for each grant:
|
For
the twelve-month period ending December 31,
|
||||||||||||
|
2009
|
2008
|
2007
|
||||||||||
|
Fair
Value of Options
|
N/A | $ | 8.45 | N/A | ||||||||
|
Dividend
yield
|
N/A | 0.0 | % | N/A | ||||||||
|
Expected
volatility
|
N/A | 51.8 | % | N/A | ||||||||
|
Risk
free interest rate
|
N/A | 4.0 | % | N/A | ||||||||
|
Expected
term in years
|
N/A | 6.3 | N/A | |||||||||
Our
dividend yield assumptions on the underlying common stock upon which the options
were granted anticipate that all earnings will be retained for use in the
operation and expansion of the company and no dividends will be paid to
shareholders. Our expected volatility is based on historic trading patterns of
our common stock. The risk free interest rate is based on the yield of short
term U.S. Treasury securities in effect at the time of the grant. The expected
term of the options reflects our historic exercise and forfeiture experience
with similar option grants.
Stock
option activity under these plans is summarized below:
|
Number
of
shares
of
Common
Stock underlying options
|
Weighted
Average exercise price
|
|||||||
|
Options
outstanding at December 31, 2006
|
146,394 | $ | 2.35 | |||||
|
Granted
|
— | $ | 0.00 | |||||
|
Exercised
|
(42,146 | ) | $ | 2.38 | ||||
|
Canceled
|
(2,029 | ) | $ | 2.94 | ||||
|
Options
outstanding at December 31, 2007
|
102,219 | $ | 2.33 | |||||
|
Granted
|
127,844 | $ | 8.45 | |||||
|
Exercised
|
(3,047 | ) | $ | 2.68 | ||||
|
Options
outstanding at December 31, 2008
|
227,016 | $ | 5.78 | |||||
|
Granted
|
— | — | ||||||
|
Exercised
|
(21,331 | ) | $ | 1.72 | ||||
|
Options
outstanding at December 31, 2009
|
205,685 | $ | 5.78 | |||||
Options
exercisable at:
|
December
31, 2007
|
102,219 | $ | 2.33 | |||||
|
December
31, 2008
|
99,171 | $ | 2.34 | |||||
|
December
31, 2009
|
119,771 | $ | 4.64 |
F-26
As of
December 31, 2009, stock options outstanding and exercisable by price range were
as follows:
|
OPTIONS
OUTSTANDING
|
OPTIONS
EXERCISABLE
|
|||||||||||||||||||||
|
Range
of
Exercise Prices
|
Outstanding
As
of
12/31/2009
|
Weighted
Average
Remaining
Contractual Life
|
Weighted
Average
Exercise Price
|
Exercisable
As
of
12/31/2009
|
Weighted
Average
Exercise Price
|
|||||||||||||||||
| $ | 0.00 - $2.55 | 48,575 | 1.2 | $ | 2.25 | 48,575 | $ | 2.25 | ||||||||||||||
| $ | 2.56 - $5.00 | 29,265 | 2.3 | $ | 2.95 | 29,265 | $ | 2.95 | ||||||||||||||
| $ | 5.01 - $20.00 | 127,845 | 8.5 | $ | 8.49 | 41,931 | $ | 8.58 | ||||||||||||||
| 205,685 | 5.9 | $ | 6.20 | 119,771 | $ | 4.64 | ||||||||||||||||
The total
intrinsic value of options exercised during the years ended December 31, 2009,
2008 and 2007 was approximately $125,000, $57,000 and $100,000
respectively.The aggregate
intrinsic value of options outstanding and exercisable as of December 31, 2009,
2008 and 2007 was approximately $354,000, $333,000, and $238,000,
respectively.
During
the years ended December 31, 2009, 2008 and 2007, we issued restricted stock
grants to selected officers and members of the Board of Directors. These stock
grants vest over a three or five year period for officers, with grants to
directors vesting immediately. These grants are valued at the closing market
price on the date granted with the associated compensation expense is recognized
ratably over the applicable period.
Compensation
expense recognized in connection with stock options for the years ended December
31, 2009, 2008 and 2007 was $189,000, $105,000 and $0 respectively. Compensation
expense recognized in connection with stock grants for the years ended December
31, 2009, 2008 and 2007 was $1,147,000, $753,000 and $558,000
respectively. As of December 31, 2009, remaining unamortized stock
compensation expense for both stock options and stock grants was approximately $
1.6 million and will be recognized as follows (000’s):
|
Stock
Options
|
Stock
Grants
|
|||||||
|
2010
|
189 | 524 | ||||||
|
2011
|
189 | 358 | ||||||
|
2012
|
70 | 141 | ||||||
|
2013
|
— | 49 | ||||||
|
2014
|
— | 40 | ||||||
|
Thereafter
|
— | 40 | ||||||
|
Unamortized
stock compensation costs at December 31, 2009
|
$ | 448 | $ | 1,152 | ||||
NOTE
20 ¾ QUARTERLY
FINANCIAL DATA (UNAUDITED):
Summarized
quarterly financial data 2009, 2008 , and 2007 appear below (in thousands,
except per share data):
F-27
INTEGRAMED
AMERICA, INC.
NOTES
TO FINANCIAL STATEMENTS (Continued)
|
Revenues
Net
|
Contribution
|
Net
Income
|
Diluted
Net
Income
Per Share (1)
|
|||||||||||||||||||||||||||||||||||||||||||||
|
2009
|
2008
|
2007
|
2009
|
2008
|
2007
|
2009
|
2008
|
2007
|
2009
|
2008
|
2007
|
|||||||||||||||||||||||||||||||||||||
|
First
quarter
|
$ | 52,355 | $ | 45,784 | $ | 32,327 | $ | 4,907 | $ | 3,861 | $ | 3,066 | $ | 920 | $ | 720 | $ | 555 | $ | 0.10 | $ | 0.08 | $ | 0.07 | ||||||||||||||||||||||||
|
Second
quarter
|
56,115 | 49,920 | 33,907 | 5,500 | 4,729 | 3,651 | 1,114 | 1,015 | 728 | 0.13 | 0.12 | 0.09 | ||||||||||||||||||||||||||||||||||||
|
Third
quarter
|
53,598 | 52,229 | 40,183 | 5,043 | 4,987 | 4,427 | 1,228 | 1,099 | 933 | 0.14 | 0.13 | 0.10 | ||||||||||||||||||||||||||||||||||||
|
Fourth
quarter
|
54,692 | 50,220 | 45,405 | 5,440 | 4,709 | 3,979 | 1,232 | 1,081 | 828 | 0.14 | 0.12 | 0.1.0 | ||||||||||||||||||||||||||||||||||||
|
Total
year
|
$ | 216,760 | $ | 198,153 | $ | 151,822 | $ | 20,890 | $ | 18,286 | $ | 15,123 | $ | 4,494 | $ | 3,915 | $ | 3,044 | $ | 0.51 | $ | 0.45 | $ | 0.36 | ||||||||||||||||||||||||
(1) The
sum of the quarterly earnings per share may not equal the full year earnings per
share as the computations of the weighted average shares outstanding for each
quarter and the full year are made independently.
NOTE 21 ¾ COMMITMENTS AND
CONTINGENCIES:
Capital
Leases ¾
Refer to
Note 14 for a summary of capital lease commitments.
Reliance on Third Party
Vendors ¾
Our
fertility and vein clinics are dependent on a limited number of primary
third-party vendors that produce supplies and medications vital to treating
infertility and vein disease. Should any of these vendors experience
a supply shortage, it may have an adverse impact on the operations of our
clinical locations and network members. To date, no shortage or
disruption has been experienced.
Employment
Agreements
We have
an employment agreement with our President and Chief Executive Officer. Pursuant
to that agreement, we may terminate the President and Chief Executive Officer’s
employment without cause on thirty days notice, in which event severance pay
equal to twelve months’ base salary plus an annual bonus, calculated
without regard to the condition precedents established under the bonus plan,
will be payable in a lump sum. Under the employment agreement, if we had
terminated Mr. Higham effective December 31, 2009, based on his 2009
compensation, he would have been paid an aggregate of $1,237,500, $750,000 of
which represents twice his 2009 base salary and $487,500 of which represents
twice his accrued 2009 bonus.
The
employment agreement further provides that in the event that within one year
after a “Change of Control” (as defined therein) of the Company occurs,
and the President and Chief Executive Officer’s employment is
terminated, the President and Chief Executive Officer will be paid a lump sum
amount equal to their base salary for a 24-month period following termination,
plus twice the full amount of their annual bonus based on their then current
salary, without regard to the condition precedents established for the bonus
payment. Based on this change of control provision, if there had been a change
of control of the Company in 2009 and the President and Chief Executive
Officer’s employment had terminated effective December 31, 2009, either for
“Good Reason” or without cause, then the President and Chief Executive Officer
would be entitled to termination pay equal to $750,000 representing his then
annualized base salary for 24-months, plus $487,500 representing twice the
amount to which he was eligible under our Executive Incentive Compensation Plan
for 2009.
F-28
We have
also entered into indemnification and change in control severance agreements
with certain of our management employees, which include, among other terms,
noncompetitive provisions and salary and benefits continuation. Our
minimum aggregate commitment under these agreements at December 31, 2009 was
approximately $3.3 million.
Commitments
to Partners ¾
In
accordance with the majority of our Partner agreements, we are obligated to: (i)
on an ongoing basis, advance funds to the fertility centers to fund operations
and provide services; and (ii) on a monthly basis, transfer to the fertility
centers funds equal to the net accounts receivable generated that month to
finance those receivables less any amounts owed to us for services fees and/or
advances.
Litigation
¾
We are
party, from time to time, to legal proceedings in the ordinary course of
business and are required to maintain compliance with extensive health care
regulations. None of these proceedings or potential issues associated with
health care regulation compliance are expected to have a material adverse effect
on our financial position, results of operations or cash flow.
Insurance ¾
As of
December 31, 2009 and December 31, 2008, we and our affiliated fertility and
vein care centers were insured with respect to medical malpractice risks on a
claims made basis. We believe, either through this captive insurance
company, or on the open market, we will be able to obtain renewal coverage for
both our fertility and vein care physicians in the future. We are not aware of
any claims against us or our affiliated medical practices, which would expose
us, or our affiliated medical practices to liabilities in excess of insured
amounts.
As of
December 31, 2009 and 2008, we also carried policies to insure against
liability, theft, property loss, business interruption and a variety of other
business risks. We also maintain an appropriate insurance reserve to cover
estimated deductible amounts should a claim be filed under our
policies. Premiums paid to the captive insurance company, which we
manage for the benefit of our Fertility Partners are $2,140,000, $2,245,000 and
$2,533,000 for the years ended December 31, 2009, 2008 and 2007,
respectively.
NOTE
22 ¾ RELATED
PARTY TRANSACTIONS:
In
accordance with our Partner agreement with Shady Grove, Michael J. Levy, M.D.,
an employed shareholder physician of the P.C., became a member of our Board of
Directors in March 1998. In 2004, Dr. Levy became an advisory
director and was no longer a voting member of the Board of Directors. The
medical practice at Shady Grove paid us service fees of $3,225,172, $3,145,000
and $2,916,000 in 2009, 2008 and 2007, respectively.
In
accordance with our Partner agreement with FCI (the Illinois practice), Aaron
Lifchez, M.D., an employed shareholder physician of FCI, became a member of our
Board of Directors in August 1997. In 2004, Dr. Lifchez became an
advisory director and was no longer a voting member of the Board of Directors.
The medical practice FCI paid us service fees of $2,147,445, $2,787,000 and
$2,649,000 in 2009, 2008 and 2007, respectively.
The
Company has a Consulting Agreement with its Chairman of the Board. The agreement
provided for compensation of $125,000 for the twelve months ending December 31,
2008. This consulting agreement expired on December 31, 2008 and was replaced
with a new one-year agreement providing for $36,000 in compensation in
2009. This one-year agreement expired on December 31, 2009 and was
not renewed.
F-29
INTEGRAMED
AMERICA, INC.
NOTES
TO FINANCIAL STATEMENTS (Continued)
NOTE
23 ¾
SUPPLEMENTAL DISCLOSURES OF CASH FLOW INFORMATION AND NON-CASH
TRANSACTIONS:
Equity
transactions related to common stock, principally arising from stock grants,
option exercises and related tax benefits disclosed on our
Consolidated Statements of Cash Flows are comprised of the following (000’s
omitted):
|
For
the
Twelve
months ended
|
||||||||||||
|
2009
|
2008
|
2007
|
||||||||||
|
Common
stock options and grants
|
$ | 37 | $ | 30 | $ | 35 | ||||||
|
Tax
benefit related to stock transactions
|
39 | 332 | 67 | |||||||||
|
Treasury
Stock, net and other
|
(39 | ) | (211 | ) | (67 | ) | ||||||
| $ | 37 | $ | 151 | $ | 35 | |||||||
NOTE
24 ¾ SUBSEQUENT
EVENTS:
On
February 12, 2010, we announced that we priced a public offering of 2,800,000
(including the over allotment) shares of common stock at a price to the public
of $7.50 per share, of which 2,300,000 shares were offered through Piper Jaffray
& Co., which acted as book-running manager and Dougherty & Company LLC,
which acted as co-manager; and 500,000 shares were sold directly to IAT
Reinsurance Company Ltd., our largest stockholder.
Approximately
$19 million of net proceeds, after deducting underwriting discounts, commissions
and expected offering expenses payable by us, are intended to be used to
accelerate the addition of new partner fertility centers, accelerate the pace of
new vein clinic openings in 2010, and for general working capital and other
corporate purposes. The offering closed on February 18,
2010.
F-30
REPORT
OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM ON
FINANCIAL
STATEMENT SCHEDULE
To the
Board of Directors and Shareholders
of
IntegraMed America, Inc.:
Our
audits of the consolidated financial statements referred to in our report dated
March 10, 2010 appearing in the 2009 Annual Report to Shareholders of IntegraMed
America, Inc. (which report and consolidated financial statement are
incorporated by reference in this Annual Report on Form 10-K) also included an
audit of the financial statement schedule listed in Item 8 and 15 (a) (1) of
this Form 10-K. In our opinion, this financial statement schedule presents
fairly, in all material respects, the information set forth therein when read in
conjunction with the related consolidated financial statements.
/s/Amper,
Politziner & Mattia, LLP.
Edison,
New Jersey
March 10,
2010
S-1
SCHEDULE
II
INTEGRAMED
AMERICA, INC.
VALUATION
AND QUALIFYING ACCOUNTS
For
the Years Ended December 31, 2009, 2008, 2007
|
Balance
at
Beginning
of Period
|
Additions
|
Deductions
|
End
of
Period
|
|||||||||||||
|
Year
Ended December 31, 2009
|
||||||||||||||||
|
Allowance for doubtful accounts
receivable
|
$ | 2,648 | $ | 4,820 | $ | 4,555 | $ | 2,913 | ||||||||
|
Reserve for Attain IVF
refunds
|
397 | 206 | 303 | 300 | ||||||||||||
|
Reserve for attain IVF medical
costs
|
331 | 72 | 59 | 344 | ||||||||||||
|
Year
Ended December 31, 2008
|
||||||||||||||||
|
Allowance for doubtful accounts
receivable
|
$ | 3,386 | $ | 3,613 | $ | 4,351 | $ | 2,648 | ||||||||
|
Reserve for Attain IVF
refunds
|
326 | 427 | 356 | 397 | ||||||||||||
|
Reserve for Attain IVF medical
costs
|
272 | 355 | 296 | 331 | ||||||||||||
|
Year
Ended December 31, 2007
|
||||||||||||||||
|
Allowance for doubtful accounts
receivable
|
$ | 13 | $ | 3,524 | (1) | $ | 151 | $ | 3,386 | |||||||
|
Reserve for Attain IVF
refunds
|
257 | 355 | 286 | 326 | ||||||||||||
|
Reserve for Attain IVF medical
costs
|
215 | 296 | 239 | 272 | ||||||||||||
|
(1)
|
Includes
$3,224 acquired in connection with the Vein Clinics of America, Inc.
transaction.
|
S-2
SIGNATURES
Pursuant
to the requirements of Section 13 or 15(d) of the Securities Exchange Act of
1934, the Registrant has duly caused this report to be signed on its behalf by
the undersigned thereunto duly authorized.
|
INTEGRAMED
AMERICA, INC.
|
|||
|
March
10, 2010
|
By:
|
/s/JOHN
HLYWAK, JR.
|
|
|
John
W. Hlywak, Jr.
Executive
Vice President
and
Chief Financial Officer
(Principal
Financial and Accounting Officer)
|
Pursuant
to the requirements of the Securities Exchange Act of 1934, this report has been
signed below by the following persons on behalf of the Registrant and in the
capacities and on the dates indicated.
|
Signature
|
Title
|
Date
|
|
|
/s/Jay
Higham
|
|||
|
Jay Higham
|
President
and Chief Executive Officer and Director
(Principal
Executive Officer)
|
March
10, 2010
|
|
|
/s/John
W. Hlywak, Jr.
|
|||
|
John W. Hlywak,
Jr.
|
Executive
Vice President and
Chief
Financial Officer
(Principal
Financial and
Accounting
Officer)
|
March
10, 2010
|
|
|
/s/Kush
K. Agarwal
|
|||
|
Kush K. Agarwal
|
Director
|
March
10, 2010
|
|
|
/s/Gerardo
Canet
|
|||
|
Gerardo Canet
|
Director
|
March
10, 2010
|
|
|
/s/Wayne
R. Moon
|
|||
|
Wayne R. Moon
|
Director
|
March
10, 2010
|
|
|
/s/Lawrence
J. Stuesser
|
|||
|
Lawrence J.
Stuesser
|
Director
|
March
10, 2010
|
|
|
/s/Elizabeth
E. Tallett
|
|||
|
Elizabeth E.
Tallett
|
Director
|
March
10, 2010
|
|
|
/s/Yvonne
S. Thornton, M.D.
|
|||
|
Yvonne S. Thornton,
M.D.
|
Director
|
March
10, 2010
|
EXHIBIT INDEX
|
Exhibit
|
||||
|
Number
|
Description
|
|||
|
1
|
.1
|
Form
of Underwriting Agreement
|
||
|
3
|
.1
|
Restated
Certificate of Incorporation of IntegraMed filed as an Exhibit to
IntegraMed’s Quarterly Report on Form 10-Q for the quarterly period
ended June 30, 2004 and incorporated herein by reference
thereto
|
||
|
3
|
.2
|
By-laws
of IntegraMed filed as an Exhibit to IntegraMed’s Quarterly Report on
Form 10-Q for the quarterly period ended June 30, 2009 and
incorporated herein by reference thereto
|
||
|
4
|
.1
|
Specimen
certificate for shares of common stock
|
||
|
5
|
.1
|
Opinion
of Dorsey & Whitney LLP regarding the validity of the securities
being registered
|
||
|
10
|
.1
|
Stock
Purchase Agreement, dated August 8, 2007, by and among IntegraMed,
IDVC Acquisition Co., the Sellers named therein, the Guarantors named
therein and VCA filed as an Exhibit to IntegraMed’s Current Report on
Form 8-K dated August 8, 2007 and incorporated herein by
reference thereto
|
||
|
10
|
.2
|
1992
Stock Option Plan, including form of option, filed as an Exhibit to
IntegraMed’s Registration Statement on Form S-1 (Registration
No. 333-47046) filed with the Securities and Exchange Commission on
April 9, 1992 and incorporated herein by reference
thereto
|
||
|
10
|
.3
|
Amended
and Restated 1992 Incentive and Non-Incentive Stock Option Plan, dated
April 16, 1998, filed as an Exhibit to IntegraMed’s Definitive Proxy
Statement on Schedule 14A filed with the Securities and Exchange
Commission on May 5, 1998 and incorporated herein by reference
thereto
|
||
|
10
|
.4
|
Agreement
of Lease, dated September 27, 1994, between Purchase Corporate Park
Associates and IntegraMed filed as an Exhibit to IntegraMed’s Annual
Report on Form 10-K for the year ended December 31, 1994 and
incorporated herein by reference thereto
|
||
|
10
|
.5
|
Amendment,
dated January 1995, to Agreement of Lease between Purchase Corporate Park
Associates and Integramed
|
||
|
10
|
.6
|
Second
Amendment, dated August 23, 1999, to Agreement of Lease between
Purchase Corporate Park Associates, L.P. and IntegraMed
|
||
|
10
|
.7
|
Third
Amendment, dated October 15, 2002, to Agreement of Lease between
Purchase Corporate Park Associates, L.P. and IntegraMed
|
||
|
10
|
.8
|
Business
Service Agreement, dated August 30, 2007, by and between IntegraMed
and the Center for Reproductive Medicine, P.A. filed as an Exhibit to
IntegraMed’s Current Report on Form 8-K dated September 6, 2007
and incorporated herein by reference thereto
|
||
|
10
|
.9
|
Consulting
Agreement, dated December 24, 2008, between Gerardo Canet and
IntegraMed filed as an Exhibit to IntegraMed’s Annual Report on
Form 10-K for the year ended December 31, 2008 and incorporated
herein by reference thereto
|
||
|
10
|
.10
|
Employment
Agreement, dated October 10, 2005, between IntegraMed and Jay Higham
filed as an Exhibit to IntegraMed’s Quarterly Report on Form 10-Q for
the quarterly period ended September 30, 2005 and incorporated herein
by reference thereto
|
||
|
10
|
.11
|
Management
Agreement, dated January 7, 1997, by and between IntegraMed and Bay
Area Fertility and Gynecology Medical Group, Inc. filed as an Exhibit to
IntegraMed’s Current Report on Form 8-K dated January 20, 1997
and incorporated herein by reference thereto
|
|||
|
10
|
.12
|
Amendment
No. 1, dated April 5, 1998, to Management Agreement between
IntegraMed and Bay Area Fertility and Gynecology Medical Group, Inc. filed
as an Exhibit to IntegraMed’s Quarterly Report on Form 10-Q for the
quarterly period ended March 31, 1998 and incorporated herein by
reference thereto
|
|||
|
10
|
.13
|
Amendment
No. 2, dated July 21, 1998, to Management Agreement between
IntegraMed and Bay Area Fertility and Gynecology Medical Group, Inc. filed
as an Exhibit to IntegraMed’s Quarterly Report on Form 10-Q for the
quarterly period ended March 31, 1999 and incorporated herein by
reference thereto
|
|||
|
10
|
.14
|
Amendment
No. 3, dated April 1, 2000, to Management Agreement between
IntegraMed and Bay Area Fertility and Gynecology Group, Inc. filed as an
Exhibit to IntegraMed’s Quarterly Report on Form 10-Q for the
quarterly period ended June 30, 2000 and incorporated herein by
reference thereto
|
|||
|
10
|
.15
|
Amendment
No. 4, dated January 1, 2001, to Management Agreement between
IntegraMed and Bay Area Fertility and Gynecology Medical Group, Inc. filed
as an Exhibit to IntegraMed’s Quarterly Report on Form 10-Q for the
quarterly period ended September 30, 2001 and incorporated herein by
reference thereto
|
|||
|
10
|
.16
|
Amendment
No. 5, dated September 19, 2001, to Management Agreement between
IntegraMed and Bay Area Fertility and Gynecology Medical Group, Inc. filed
as an Exhibit to IntegraMed’s Quarterly Report on Form 10-Q for the
quarterly period ended September 30, 2001 and incorporated herein by
reference thereto
|
|||
|
10
|
.17
|
Amendment
No. 6, dated December 2003, to Service Agreement between IntegraMed
and Reproductive Science Center of the San Francisco Bay Area, a
California medical corporation, filed as an Exhibit to IntegraMed’s Annual
Report on Form 10-K for the year ended December 31, 2003 and
incorporated herein by reference thereto
|
|||
|
10
|
.18
|
Management
Agreement, dated February 28, 1997, between IntegraMed and Fertility
Centers of Illinois, S.C. filed as an Exhibit to IntegraMed’s Registration
Statement on Form S-1 (Registration No. 333-26551) filed with
the Securities and Exchange Commission on May 6, 1997 and
incorporated herein by reference thereto
|
|||
|
10
|
.19
|
Amendment,
dated May 2, 1997, to Management Agreement between IntegraMed and
Fertility Centers of Illinois, S.C. filed as an Exhibit to Amendment
No. 1 to IntegraMed’s Registration Statement on Form S-1
(Registration No. 333-26551) filed with the Securities and Exchange
Commission on June 20, 1997 and incorporated herein by reference
thereto
|
|||
|
10
|
.20
|
Amendment
No. 2, dated June 18, 1997, to Management Agreement between
IntegraMed and Fertility Centers of Illinois, S.C. filed as an Exhibit to
Amendment No. 1 to IntegraMed’s Registration Statement on
Form S-1 (Registration No. 333-26551) filed with the Securities
and Exchange Commission on June 20, 1997 and incorporated herein by
reference thereto
|
|||
|
10
|
.21
|
Amendment
No. 3, dated August 19, 1997, to Management Agreement between
IntegraMed and Fertility Centers of Illinois, S.C. filed as an Exhibit to
IntegraMed’s Quarterly Report on Form 10-Q for the quarterly period
ended September 30, 1997 and incorporated herein by reference
thereto
|
|||
|
10
|
.22
|
Amendment
No. 4, dated January 9, 1998, to Management Agreement between
IntegraMed and Fertility Centers of Illinois, S.C. filed as an Exhibit to
a Schedule 13D dated February 11, 1998 filed with the Securities
and Exchange Commission by Gerardo Canet and incorporated herein by
reference thereto
|
|||
|
10
|
.23
|
Amendment
No. 5, dated March 5, 1998, to Management Agreement between
IntegraMed and Fertility Centers of Illinois, S.C. filed as an Exhibit to
IntegraMed’s Annual Report on Form 10-K for the year ended
December 31, 1997 and incorporated herein by reference
thereto
|
||||
|
10
|
.24
|
Amendment
No. 6, dated July 1, 1999, to Management Agreement between
IntegraMed and Fertility Centers of Illinois, S.C. filed as an Exhibit to
IntegraMed’s Quarterly Report on Form 10-Q for the quarterly period
ended June 30, 1999 and incorporated herein by reference
thereto
|
||||
|
10
|
.25
|
Amendment
No. 7, dated April 1, 2000, to Management Agreement between
IntegraMed and Fertility Centers of Illinois, S.C. filed as an Exhibit to
IntegraMed’s Quarterly Report on Form 10-Q for the quarterly period
ended June 30, 2000 and incorporated herein by reference
thereto
|
||||
|
10
|
.26
|
Amendment
No. 8, dated September 24, 2001, to Management Agreement between
IntegraMed and Fertility Centers of Illinois, S.C. filed as an Exhibit to
IntegraMed’s Quarterly Report on Form 10-Q for the quarterly period
ended September 30, 2001 and incorporated herein by reference
thereto
|
||||
|
10
|
.27
|
Amendment
No. 9, dated December 2003, to Service Agreement between IntegraMed
and Fertility Centers of Illinois, S.C. filed as an Exhibit to
IntegraMed’s Annual Report on Form 10-K for the year ended
December 31, 2003 and incorporated herein by reference
thereto
|
||||
|
10
|
.28
|
Amendment
No. 10, dated January 1, 2005, to Service Agreement between
IntegraMed and Fertility Centers of Illinois, S.C. filed as an Exhibit to
IntegraMed’s Annual Report on Form 10-K for the year ended
December 31, 2005 and incorporated herein by reference
thereto
|
||||
|
10
|
.29
|
Service
Agreement, dated May 25, 2001, between IntegraMed and MPD Medical
Associates (MA), P.C. filed as an Exhibit to IntegraMed’s Quarterly
Report on Form 10-Q for the quarterly period ended June 30, 2001
and incorporated herein by reference thereto
|
||||
|
10
|
.30
|
Amendment
No. 1, dated March 5, 2002, to Service Agreement between
IntegraMed and MPD Medical Associates (MA), P.C. filed as an Exhibit
to IntegraMed’s Annual Report on Form 10-K for the year ended
December 31, 2001 and incorporated herein by reference
thereto
|
||||
|
10
|
.31
|
Management
Agreement, dated March 11, 1998, between Shady Grove Fertility
Centers, P.C. and Levy, Sagoskin and Stillman, M.D., P.C.
filed as an Exhibit to IntegraMed’s Annual Report on Form 10-K for
the year ended December 31, 1997 and incorporated herein by reference
thereto
|
||||
|
10
|
.32
|
Amendment
No. 1, dated April 16, 1998, to Management Agreement between
Shady Grove Fertility Centers, Inc. and Levy, Sagoskin and
Stillman, M.D., P.C filed as an Exhibit to IntegraMed’s Quarterly
Report on Form 10-Q for the quarterly period ended March 31,
1998 and incorporated herein by reference thereto
|
||||
|
10
|
.33
|
Amendment
No. 2, dated May 6, 1998, to Management Agreement between Shady
Grove Fertility Centers, Inc. and Levy, Sagoskin and
Stillman, M.D., P.C. filed as an Exhibit to IntegraMed’s Annual
Report on Form 10-K for the year ended December 31, 1998 and
incorporated herein by reference thereto
|
||||
|
10
|
.34
|
Amendment
No. 3, dated September 1, 1999, to Management Agreement between
IntegraMed and Shady Grove Reproductive Science Center, P.C. filed as
an Exhibit to IntegraMed’s Quarterly Report on Form 10-Q for the
quarterly period ended September 30, 1999 and incorporated herein by
reference thereto
|
||||
|
10
|
.35
|
Amendment
No. 4, dated April 1, 2000, to Management Agreement between
IntegraMed and Shady Grove Reproductive Science Center, P.C. filed as
an Exhibit to IntegraMed’s Quarterly Report on Form 10-Q for the
quarterly period ended June 30, 2000 and incorporated herein by
reference thereto
|
||||
|
10
|
.36
|
Amendment
No. 5 to Management Agreement between IntegraMed and Shady Grove
Reproductive Science Center, P.C. filed as an Exhibit to IntegraMed’s
Quarterly Report on Form 10-Q for the quarterly period ended
September 30, 2001 and incorporated herein by reference
thereto
|
||||
|
10
|
.37
|
Amendment
No. 6, dated September 18, 2001, to Management Agreement between
IntegraMed and Shady Grove Reproductive Science Center, P.C. filed as
an Exhibit to IntegraMed’s Quarterly Report on Form 10-Q for the
quarterly period ended September 30, 2001 and incorporated herein by
reference thereto
|
||||
|
10
|
.38
|
Amendment
No. 7, dated November 2003, to Service Agreement between IntegraMed
and Shady Grove Reproductive Science Center, P.C. filed as an Exhibit
to IntegraMed’s Annual Report on Form 10-K for the year ended
December 31, 2003 and incorporated herein by reference
thereto
|
||||
|
10
|
.39
|
Amendment
No. 8, dated February 16, 2006, to Service Agreement between
IntegraMed and Shady Grove Reproductive Science Center, P.C. filed as
an Exhibit to IntegraMed’s Annual Report on Form 10-K for the year
ended December 31, 2005 and incorporated herein by reference
thereto
|
||||
|
10
|
.40
|
Amendment
No. 9, dated March 22, 2007, to Service Agreement between
IntegraMed and Shady Grove Fertility Reproductive Science
Center, P.C. filed as an Exhibit to IntegraMed’s Quarterly Report on
Form 10-Q for the quarterly period ended March 31, 2007 and
incorporated herein by reference thereto
|
||||
|
10
|
.41
|
Second
Amended and Restated Loan Agreement, dated August 8, 2007, by and
among IntegraMed and Bank of America, N.A. filed as an Exhibit to
IntegraMed’s Current Report on Form 8-K dated August 8, 2007 and
incorporated herein by reference thereto
|
||||
|
10
|
.42
|
Form
of Retention Agreement filed as an Exhibit to IntegraMed’s Quarterly
Report on Form 10-Q for the quarterly period ended June 30, 1999
and incorporated herein by reference thereto
|
||||
|
10
|
.43
|
Form
of Amendment to Retention Agreement relating to Section 409A of the
U.S. Internal Revenue Code of 1986, as amended, filed as an Exhibit to
IntegraMed’s Annual Report on Form 10-K for the year ended
December 31, 2008 and incorporated herein by reference
thereto
|
||||
|
10
|
.44
|
Form
of Indemnification Agreement filed as an Exhibit to IntegraMed’s Quarterly
Report on Form 10-Q for the quarterly period ended June 30, 2000
and incorporated herein by reference thereto
|
||||
|
10
|
.45
|
Form
of Amendment to Indemnification Agreement relating to Section 409A of
the U.S. Internal Revenue Code of 1986, as amended, filed as an Exhibit to
IntegraMed’s Annual Report on Form 10-K for the year ended
December 31, 2008 and incorporated herein by reference
thereto
|
||||
|
10
|
.46
|
Service
Agreement, dated April 26, 2002, between IntegraMed and Northwest
Center for Infertility and Reproductive Endocrinology filed as an Exhibit
to IntegraMed’s Quarterly Report on Form 10-Q for the quarterly
period ended March 31, 2002 and incorporated herein by reference
thereto
|
||||
|
10
|
.47
|
Amendment
No. 1, dated June 14, 2002, to Service Agreement between
IntegraMed and Northwest Center for Infertility and Reproductive
Endocrinology filed as an Exhibit to IntegraMed’s Quarterly Report on
Form 10-Q for the quarterly period ended September 30, 2002 and
incorporated herein by reference thereto
|
||||
|
10
|
.48
|
Amendment
No. 2, dated November 1, 2002, to Service Agreement between
IntegraMed and Northwest Center for Infertility and Reproductive
Endocrinology filed as an Exhibit to IntegraMed’s Annual Report on
Form 10-K for the year ended December 31, 2002 and incorporated
herein by reference thereto
|
||||
|
10
|
.49
|
Amendment
No. 3, dated January 1, 2003, to Service Agreement between
IntegraMed and Northwest Center for Infertility and Reproductive
Endocrinology filed as an Exhibit to IntegraMed’s Annual Report on
Form 10-K for the year ended December 31, 2003 and incorporated
herein by reference thereto
|
|||
|
10
|
.50
|
2000
Long-Term Compensation Plan filed as an Exhibit to IntegraMed’s Quarterly
Report on Form 10-Q for the quarterly period ended June 30, 2002
and incorporated herein by reference thereto
|
|||
|
10
|
.51
|
Form
of Incentive Stock Option Agreement filed as an Exhibit to IntegraMed’s
Quarterly Report on Form 10-Q for the quarterly period ended
June 30, 2005 and incorporated herein by reference
thereto
|
|||
|
10
|
.52
|
Form
of Non-Qualified Stock Option Agreement filed as an Exhibit to
IntegraMed’s Quarterly Report on Form 10-Q for the quarterly period
ended June 30, 2005 and incorporated herein by reference
thereto
|
|||
|
10
|
.53
|
Form
of Restricted Stock Unit Award Agreement filed as an Exhibit to
IntegraMed’s Annual Report on Form 10-K for the year ended
December 31, 2008 and incorporated herein by reference
thereto
|
|||
|
10
|
.54
|
Service
Agreement, dated September 1, 2003, between IntegraMed and
Reproductive Endocrine Associates of Charlotte, P.C. filed as an
Exhibit to IntegraMed’s Quarterly Report on Form 10-Q for the
quarterly period ended September 30, 2003 and incorporated herein by
reference thereto
|
|||
|
10
|
.55
|
Service
Agreement, dated January 1, 2004, between IntegraMed and Seattle
Reproductive Medicine, Inc., P.S. filed as an Exhibit to IntegraMed’s
Quarterly Report on Form 10-Q for the quarterly period ended
March 31, 2004 and incorporated herein by reference
thereto
|
|||
|
10
|
.56
|
Submanagement
Agreement, dated January 1, 2005, between Reproductive Partners, Inc.
and IntegraMed filed as an Exhibit to IntegraMed’s Quarterly Report on
Form 10-Q for the quarterly period ended March 31, 2005 and
incorporated herein by reference thereto
|
|||
|
10
|
.57
|
2007
Long-Term Compensation Plan filed as an Exhibit to IntegraMed’s Annual
Report on Form 10-K for the year ended December 31, 2007 and
incorporated herein by reference thereto
|
|||
|
10
|
.58
|
Business
Service Agreement, dated April 24, 2008, between IntegraMed and
Southeastern Fertility Centers, P.A. filed as an Exhibit to IntegraMed’s
Quarterly Report on Form 10-Q for the quarterly period ended
March 31, 2008 and incorporated herein by reference
thereto
|
|||
|
10
|
.59
|
Business
Service Agreement, dated July 9, 2008, between IntegraMed and Arizona
Reproductive Medicine Specialists Ltd. filed as an Exhibit to IntegraMed’s
Annual Report on Form 10-K for the year ended December 31, 2008
and incorporated herein by reference thereto
|
|||
|
10
|
.60
|
Amendment
No. 11, dated November 30, 2009, to Service Agreement between
IntegraMed and Fertility Centers of Illinois, S.C.
|
|||
|
10
|
.61
|
First
Amended and Restated Management Agreement, dated December 1, 2009, by
and among IntegraMed, Idaho Center for Reproductive Medicine, P.C. and
Idaho Reproductive Labs, Inc.
|
|||
|
10
|
.62
|
First
Amended and Restated Management Agreement, dated December 1, 2009, by
and among IntegraMed, Foulk & Whitten Nevada Center for Reproductive
Medicine, P.C. and Nevada Reproductive Labs, Inc.
|
|||
|
10
|
.63
|
Management
Agreement, dated December 1, 2009, by and between IntegraMed and Utah
Fertility Center, P.C.
|
|||
|
10
|
.64
|
Form
of Purchase Agreement between IntegraMed and IAT Reinsurance Company
Ltd.
|
|||
|
21
|
.1
**
|
List
of subsidiaries of IntegraMed America, Inc.
|
|||
|
23
|
.1
**
|
Consent
of Amper, Politziner & Mattia, LLP, Independent Registered Public
Accounting Firm
|
||
|
23
|
.2
|
Consent
of Dorsey & Whitney LLP (included in
Exhibit 5.1)
|
||
|
24
|
.1
|
Powers
of Attorney
|
||
|
31
|
.1
**
|
CEO
Certification Pursuant to 18 U.S.C. § 1350 as Adopted Pursuant to Section
302 of the Sarbanes Oxley Act of 2002 dated March 31,
2009.
|
||
|
31
|
.2
**
|
CFO
Certification Pursuant to 18 U.S.C. § 1350 as Adopted Pursuant to Section
302 of the Sarbanes Oxley Act of 2002 dated March 31,
2009.
|
||
|
32
|
.1
**
|
CEO
Certification Pursuant to 18 U.S.C. § 1350 as Adopted Pursuant to Section
906 of the Sarbanes Oxley Act of 2002 dated March 31,
2009.
|
||
|
32
|
.2
**
|
CFO
Certification Pursuant to 18 U.S.C. § 1350 as Adopted Pursuant to Section
906 of the Sarbanes Oxley Act of 2002 dated March 31,
2009.
|
|
**
|
Filed
herewith.
|
