Attached files
| file | filename |
|---|---|
| EX-32.1 - Benda Pharmaceutical, Inc. | v167398_ex32-1.htm |
| EX-31.1 - Benda Pharmaceutical, Inc. | v167398_ex31-1.htm |
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
WASHINGTON,
D.C. 20549
_______________
FORM
10-Q
_______________
|
x
|
QUARTERLY
REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF
1934
|
For
the quarterly period ended September 30, 2009
|
o
|
TRANSITION
REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF
1934
|
For the transition period from
______to______.
BENDA PHARMACEUTICAL, INC.
(Exact
name of registrant as specified in Charter
|
Delaware
|
000-16397
|
41-2185030
|
||
|
(State
or other jurisdiction of
incorporation
or organization)
|
(Commission
File No.)
|
(IRS
Employee Identification No.)
|
Taibei
Mingju, 4th
Floor,
6
Taibei Road, Wuhan, Hubei Province, 430015, PRC
(Address
of Principal Executive Offices)
_______________
+86
(27) 85494916
(Issuer
Telephone number)
_______________
(Former
Name or Former Address if Changed Since Last Report)
Check
whether the issuer (1) has filed all reports required to be filed by Section 13
or 15(d) of the Exchange Act during the preceding 12 months (or for such shorter
period that the issuer was required to file such reports), and (2)has been
subject to such filing requirements for the past 90 days. Yes x No
o
Indicate
by check mark whether the registrant has submitted electronically and posted on
its corporate Web site, if any, every
Interactive
Data File required to be submitted and posted pursuant to Rule 405 of Regulation
S-T (§232.405 of this chapter) during the preceding 12 months (or for such
shorter period that the registrant was required to submit and post such
files).
Yes o No o
Indicate
by check mark whether the registrant is a large accelerated filer, an
accelerated filer, a non-accelerated filer or a smaller reporting company filer.
See definition of “accelerated filer” and “large accelerated filer” in
Rule 12b-2 of the Exchange Act (Check one):
Large
Accelerated Filer o Accelerated
Filer o Non-Accelerated
Filer o Smaller
Reporting Company x
Indicate
by check mark whether the registrant is a shell company as defined in Rule 12b-2
of the Exchange Act.
Yes o No
x
State the
number of shares outstanding of each of the issuer’s classes of common equity,
as of November 23, 2009: 105,155,355 shares of common stock.
BENDA
PHARMACEUTICAL, INC.
FORM
10-Q
September
30, 2009
INDEX
|
PART
I-- FINANCIAL INFORMATION
|
|||
|
Item
1.
|
Financial
Statements
|
3
|
|
|
Item
2.
|
Management’s
Discussion and Analysis of Financial Condition
|
4
|
|
|
Item
3.
|
Quantitative
and Qualitative Disclosures About Market Risk
|
9
|
|
|
Item
4T.
|
Control
and Procedures
|
9
|
|
|
PART
II-- OTHER INFORMATION
|
|||
|
Item
1
|
Legal
Proceedings
|
10
|
|
|
Item
2.
|
Unregistered
Sales of Equity Securities and Use of Proceeds
|
12
|
|
|
Item
3.
|
Defaults
Upon Senior Securities
|
12
|
|
|
Item
4.
|
Submission
of Matters to a Vote of Security Holders
|
13
|
|
|
Item
5.
|
Other
Information
|
13
|
|
|
Item
6.
|
Exhibits
|
13
|
|
|
SIGNATURE
|
14
|
||
2
Item 1. Financial
Information
BENDA
PHARMACEUTICAL, INC.
(an
exploration stage company)
FINANCIAL
STATEMENTS
AS
OF SEPTEMBER 30, 2009
CONTENTS
|
F1
|
CONDENSED
BALANCE SHEETS AS OF SEPTEMBER 30, 2009 (UNAUDITED) AND AS OF DECEMBER 31,
2008 (AUDITED).
|
|
|
F2
|
CONDENSED
STATEMENTS OF OPERATIONS FOR THE SIX MONTHS ENDED SEPTEMBER 30, 2009 AND
2008 AND FOR THE PERIOD FROM DECEMBER 17, 1999 (INCEPTION) TO SEPTEMBER
30, 2009 (UNAUDITED).
|
|
|
F5
|
CONDENSED
STATEMENT OF CHANGES IN STOCKHOLDERS’ EQUITY/(DEFICIENCY) FOR THE PERIOD
FROM DECEMBER 17, 1999 (INCEPTION) TO SEPTEMBER 30, 2009
(UNAUDITED).
|
|
|
F3
- F4
|
CONDENSED
STATEMENTS OF CASH FLOWS FOR THE SIX MONTHS ENDED SEPTEMBER 30, 2009 AND
2008 AND FOR THE PERIOD FROM DECEMBER 17, 1999 (INCEPTION) TO SEPTEMBER
30, 2009 (UNAUDITED).
|
|
|
F6
- F39
|
NOTES
TO CONDENSED FINANCIAL STATEMENTS
(UNAUDITED).
|
3
Benda
Pharmaceutical, Inc.
Consolidated
Balance Sheets
(Amounts
expressed in U.S. Dollars)
|
September
30
|
December
31
|
|||||||
|
2009
|
2008
|
|||||||
|
(Unaudited)
|
(Audited)
|
|||||||
|
Assets
|
||||||||
|
Current
Assets
|
||||||||
|
Cash
and cash equivalents
|
$ | 1,378,053 | $ | 569,019 | ||||
|
Trade
receivables, net (Note 5)
|
8,683,364 | 9,336,915 | ||||||
|
Other
receivables (Note 5)
|
678,528 | 504,349 | ||||||
|
Short-term
loan receivables
|
- | 146,685 | ||||||
|
Prepaid
tax (Note 23)
|
1,509,892 | 771,549 | ||||||
|
Due
from related parties (Note 18)
|
7,634 | 11,000 | ||||||
|
Inventories
(Note 7)
|
2,343,783 | 2,344,562 | ||||||
|
Prepaid
expenses and deposits (Note 5)
|
2,540,267 | 918,209 | ||||||
|
Total
current assets
|
17,141,521 | 14,602,288 | ||||||
|
Due
from related parties (Note 18)
|
3,063,153 | 2,961,744 | ||||||
|
Property
and equipments, net (Note 8)
|
27,901,030 | 29,057,225 | ||||||
|
Intangible
assets, net (Note 9)
|
5,454,525 | 6,003,102 | ||||||
|
Restricted
cash (Note 11)
|
5,847,894 | 6,162,849 | ||||||
|
Refundable
purchase price paid (Note 6)
|
1,200,000 | 1,200,000 | ||||||
|
Other
assets (Note 12)
|
1,830,556 | 1,830,634 | ||||||
|
Debt
issue costs (Note 24)
|
- | 55,485 | ||||||
|
Total
Assets
|
$ | 62,438,679 | $ | 61,873,327 | ||||
|
Liabilities
& Shareholders' Equity
|
||||||||
|
Current
Liabilities
|
||||||||
|
Bank
indebtedness (Note 11)
|
$ | 1,199,965 | $ | 1,288,257 | ||||
|
Bank
loans payable (current portion) (Note 13)
|
2,984,433 | 2,984,560 | ||||||
|
Short-term
loans payable
|
1,042,955 | - | ||||||
|
Long
term debt payable (current portion) (Note 14)
|
2,234,115 | 2,234,210 | ||||||
|
Accounts
payable and accrued liabilities (Note 15)
|
9,757,230 | 7,228,400 | ||||||
|
Commercial
notes payable (Note 11)
|
9,075,536 | 9,297,169 | ||||||
|
Taxes
payable
|
262,761 | 516,302 | ||||||
|
Acquisition
cost payable (Note 10)
|
1,426,430 | 1,426,491 | ||||||
|
Long-term
convertible promissory notes (current portion) (Note 24)
|
7,260,000 | 6,395,951 | ||||||
|
Wages
payable
|
1,180,027 | 630,475 | ||||||
|
Payable
under redeemable common stock (Note 19)
|
7,376,366 |
7,376,366
|
||||||
|
Due
to related parties (Note 18)
|
467,842 | 1,457,803 | ||||||
|
Total
current liabilities
|
44,267,660 | 40,835,984 | ||||||
|
Due
to related parties (Note 18)
|
960,406 | 997,593 | ||||||
|
Total
Liabilities
|
45,228,066 | 41,833,577 | ||||||
|
Equity
|
||||||||
|
Preferred
stock, $0.001 par value; 5,000,000 shares authorized;
|
||||||||
|
None
issued and outstanding
|
- | - | ||||||
|
Common
stock, $0.001 par value; 150,000,000 shares authorized;
|
||||||||
|
105,155,355
shares issued and outstanding as of 9/30/2009
|
||||||||
|
and
12/31/2008 respectively;
|
105,155 | 105,155 | ||||||
|
Additional
paid in capital (Note 25)
|
22,108,427 | 22,108,427 | ||||||
|
Retained
earnings (unrestricted)
|
(18,736,213 | ) | (16,047,561 | ) | ||||
|
Statutory
surplus reserve fund (Note 17)
|
2,642,775 | 2,642,775 | ||||||
|
Accumulative
other comprehensive income
|
6,344,117 | 6,347,547 | ||||||
|
Shares
issuable for acquisition and services
|
503,860 | 503,860 | ||||||
|
Total
Benda Pharmaceutical, Inc. Shareholders' Equity
|
12,968,121 | 15,660,203 | ||||||
|
Noncontrolling
interest
|
4,242,492 | 4,379,547 | ||||||
|
Total
Equity
|
17,210,613 | 20,039,750 | ||||||
|
Total
Liabilities & Equity
|
$ | 62,438,679 | $ | 61,873,327 | ||||
The
accompanying notes are an integral part of these consolidated financial
statements.
F-1
Benda
Pharmaceutical, Inc.
Consolidated
Statements of Operations
(Amounts
expressed in U.S. Dollars)
|
Nine
Months Ended September 30
|
Three
Months Ended September 30
|
|||||||||||||||
|
2009
|
2008
|
2009
|
2008
|
|||||||||||||
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
|||||||||||||
|
Revenue
|
$ | 17,345,065 | $ | 19,860,057 | $ | 5,904,653 | $ | 6,634,434 | ||||||||
|
Cost
of goods sold
|
(9,770,370 | ) | (12,610,394 | ) | (3,071,291 | ) | (4,213,540 | ) | ||||||||
|
Gross
profit
|
7,574,695 | 7,249,663 | 2,833,362 | 2,420,894 | ||||||||||||
|
Selling
expenses
|
(1,868,482 | ) | (2,122,689 | ) | (852,116 | ) | (691,477 | ) | ||||||||
|
General
and administrative expenses
|
||||||||||||||||
|
Amortization
of intangible assets
|
(119,118 | ) | (148,591 | ) | (39,718 | ) | (50,643 | ) | ||||||||
|
Amortization
of debt issue costs (Note 24)
|
(55,485 | ) | (198,360 | ) | - | (66,603 | ) | |||||||||
|
Depreciation
|
(707,622 | ) | (373,639 | ) | (328,327 | ) | (130,144 | ) | ||||||||
|
Bad
debts
|
(3,080,334 | ) | (534,124 | ) | (271,472 | ) | 239,769 | |||||||||
|
Write-down
of inventory to net realizable value (recovery)
|
(196,628 | ) | - | (72,389 | ) | - | ||||||||||
|
Director
remuneration
|
(67,500 | ) | (162,968 | ) | (22,500 | ) | (22,500 | ) | ||||||||
|
Penalty
to investors
|
- | (1,380,173 | ) | - | (266,768 | ) | ||||||||||
|
Other
general and administrative expenses (Note 21)
|
(1,885,994 | ) | (2,764,256 | ) | (898,062 | ) | (743,982 | ) | ||||||||
|
Total
general and administrative expenses
|
(6,112,681 | ) | (5,562,111 | ) | (1,632,468 | ) | (1,040,871 | ) | ||||||||
|
Research
and development expenses
|
(283,473 | ) | (249,357 | ) | (165,390 | ) | (79,576 | ) | ||||||||
|
Total
operating expenses
|
(8,264,636 | ) | (7,934,157 | ) | (2,649,974 | ) | (1,811,924 | ) | ||||||||
|
Operating
income / (loss)
|
(689,941 | ) | (684,494 | ) | 183,388 | 608,970 | ||||||||||
|
Interest
income / (expenses) (Note 24)
|
(2,104,510 | ) | (3,339,457 | ) | (471,263 | ) | (998,877 | ) | ||||||||
|
Other
income / (expenses) (Note 20)
|
106,161 | 10,140 | 60,388 | (237,918 | ) | |||||||||||
|
Government
subsidies / grants (Note 22)
|
26,386 | - | - | - | ||||||||||||
|
Loss
before provision for income taxes and noncontrolling
interest
|
(2,661,904 | ) | (4,013,811 | ) | (227,487 | ) | (627,825 | ) | ||||||||
|
Income
taxes (Note 23)
|
(163,803 | ) | (907,176 | ) | (79,391 | ) | (382,368 | ) | ||||||||
|
Net
loss
|
(2,825,707 | ) | (4,920,987 | ) | (306,878 | ) | (1,010,193 | ) | ||||||||
|
Net
loss attributable to the noncontrolling interest
|
137,055 | 256,486 | (65,519 | ) | 168,270 | |||||||||||
|
Net
loss attributable to Benda Pharmaceutical, Inc.
|
$ | (2,688,652 | ) | (4,664,501 | ) | $ | (372,397 | ) | $ | (841,923 | ) | |||||
|
Basic
and fully diluted loss per share
|
||||||||||||||||
|
Net
loss attributable to Benda Pharmaceutical, Inc. common
shareholders
|
$ | (0.03 | ) | (0.05 | ) | $ | (0.00 | ) | $ | (0.01 | ) | |||||
|
Weighted
average shares outstanding (Note 26)
|
105,155,355 | 100,887,853 | 105,155,355 | 101,556,376 | ||||||||||||
The accompanying notes are an integral part of these consolidated financial
statements.
F-2
Benda
Pharmaceutical, Inc.
Consolidated
Statements of Cash Flows
(Amounts
expressed in U.S. Dollars)
|
Nine
Months Ended September 30,
|
||||||||
|
2009
|
2008
|
|||||||
|
(Unaudited)
|
(Unaudited)
|
|||||||
|
Cash
Flows From Operating Activities
|
||||||||
|
Net
loss
|
$ | (2,825,707 | ) | $ | (4,920,987 | ) | ||
|
Adjustments
to reconcile net loss to net cash provided by operating
activities:
|
||||||||
|
Bad
Debt provision
|
3,080,334 | 534,125 | ||||||
|
Inventory
written down to net realizable value (recovery)
|
196,628 | - | ||||||
|
Depreciation
|
1,609,314 | 1,414,763 | ||||||
|
Amortization
of intangible assets
|
547,919 | 567,016 | ||||||
|
Amortization
of debt issue costs (Note 24)
|
55,485 | 198,360 | ||||||
|
Interest
expense (amortization of debt discount) (Note 24)
|
864,049 | 2,614,667 | ||||||
|
Penalty
to investors settled by issuance of common stock
|
- | 497,081 | ||||||
|
Directors
remuneration settled by issuance of common stock
|
- | 75,900 | ||||||
|
Changes
in operating assets and liabilities:
|
||||||||
|
Trade
receivables
|
(2,427,968 | ) | (2,312,392 | ) | ||||
|
Other
receivables
|
(174,180 | ) | 239,155 | |||||
|
Prepaid
expenses and deposits
|
(1,622,058 | ) | (85,211 | ) | ||||
|
Inventories
|
(195,849 | ) | (835,895 | ) | ||||
|
Accounts
payable and accrued liabilities
|
3,078,386 | 1,856,143 | ||||||
|
Taxes
payable
|
(991,888 | ) | (281,668 | ) | ||||
|
Net
cash provided by operating activities
|
1,194,465 | (438,943 | ) | |||||
|
Cash
Flows From Investing Activities
|
||||||||
|
Purchases
of property and equipment and construction-in-progress
|
(455,488 | ) | (210,254 | ) | ||||
|
Proceeds
and repayments of borrowings under short-term loan
receivables
|
146,685 | (146,304 | ) | |||||
|
Net
cash used in investing activities
|
(308,803 | ) | (356,558 | ) | ||||
|
Cash
Flows From Financing Actives
|
||||||||
|
Proceeds
and repayments of borrowings under related parties, net
|
(1,125,192 | ) | (1,247,617 | ) | ||||
|
Proceeds
and repayments of borrowings under government debts payable,
net
|
(88,236 | ) | 297,545 | |||||
|
Proceeds
and repayments of borrowings under commercial bank notes, net (Note
11)
|
93,266 | 1,876,424 | ||||||
|
Proceeds
and repayments of borrowings under bank loans, net
|
1,042,955 | (81,082 | ) | |||||
|
Net
cash provided by (used in) financing activities
|
(77,207 | ) | 845,270 | |||||
|
Effect
of exchange rate changes on cash
|
579 | 369,366 | ||||||
|
Net
increase in cash and cash equivalents
|
809,034 | 419,135 | ||||||
|
Cash
and cash equivalents, beginning of period
|
569,019 | 1,266,240 | ||||||
|
Cash
and cash equivalents, end of period
|
$ | 1,378,053 | $ | 1,685,375 | ||||
|
Supplemental
Disclosure of Cash Flow Information
|
||||||||
|
Cash
paid for interest
|
$ | 298,139 | $ | 823,037 | ||||
|
Cash
paid for income taxes
|
$ | 935,208 | $ | 954,431 | ||||
The accompanying notes are an
integral part of these consolidated financial statements.
F-3
Benda
Pharmaceutical, Inc.
Statements
of Consolidated Comprehensive Income
(Amounts
expressed in U.S. Dollars)
|
Nine
Months Ended June 30,
|
||||||||
|
2009
|
2008
|
|||||||
|
(Unaudited)
|
(Unaudited)
|
|||||||
|
Net
loss
|
$ | (2,825,707 | ) | $ | (4,920,987 | ) | ||
|
Other
comprehensive income (loss)
|
||||||||
|
Foreign
currency translation adjustment
|
(3,430 | ) | 2,997,021 | |||||
|
Comprehensive
loss
|
(2,829,137 | ) | (1,923,966 | ) | ||||
|
Comprehensive
loss attributable to the noncontrolling interest
|
137,055 | 256,486 | ||||||
|
Comprehensive
loss attributable to Benda Pharmaceutical, Inc.
|
$ | (2,692,082 | ) | $ | (1,667,480 | ) | ||
The accompanying notes are an integral part of these consolidated financial
statements.
F-4
Benda
Pharmaceutical, Inc.
Consolidated
Statement of Changes in Equity
(Amounts
expressed in U.S. Dollars)
|
Shares
|
||||||||||||||||||||||||||||||||||||||||
|
Issuable
|
||||||||||||||||||||||||||||||||||||||||
|
Common
|
Statutory
|
Accumulated
|
for
|
|||||||||||||||||||||||||||||||||||||
|
Shares
|
Additional
|
Retained
|
Surplus
|
Other
|
Acquisition
|
Non-
|
||||||||||||||||||||||||||||||||||
|
Issued
and
|
Comprehensive
|
Common
|
Paid-in
|
Earnings
|
Reserve
|
Comprehensive
|
and
|
Controlling
|
||||||||||||||||||||||||||||||||
|
Outstanding
|
Total
|
Income
|
Stock
|
Capital
|
(Unrestricted)
|
Fund
|
Income
|
Services
|
Interest
|
|||||||||||||||||||||||||||||||
|
Balance
at December 31, 2008
|
105,155,355 | $ | 20,039,750 | $ | - | $ | 105,155 | $ | 22,108,427 | $ | (16,047,561 | ) | $ | 2,642,775 | $ | 6,347,547 | $ | 503,860 | $ | 4,379,547 | ||||||||||||||||||||
|
Comprehensive
loss:
|
||||||||||||||||||||||||||||||||||||||||
|
Net
loss
|
(1,236,563 | ) | (1,236,563 | ) | - | - | (1,040,557 | ) | - | - | - | (196,006 | ) | |||||||||||||||||||||||||||
|
Other
comprehensive income (loss)
|
||||||||||||||||||||||||||||||||||||||||
|
Foreign
currency translation adjustment
|
(52,320 | ) | (52,320 | ) | - | - | - | - | (52,320 | ) | - | - | ||||||||||||||||||||||||||||
|
Comprehensive
loss
|
(1,288,883 | ) | $ | (1,288,883 | ) | |||||||||||||||||||||||||||||||||||
|
Dividend
paid
|
- | |||||||||||||||||||||||||||||||||||||||
|
Balance
at March 31, 2009 (unaudited)
|
105,155,355 | $ | 18,750,868 | $ | 105,155 | $ | 22,108,427 | $ | (17,088,117 | ) | $ | 2,642,775 | $ | 6,295,227 | $ | 503,860 | $ | 4,183,541 | ||||||||||||||||||||||
|
Comprehensive
loss:
|
||||||||||||||||||||||||||||||||||||||||
|
Net
loss
|
(1,282,266 | ) | (1,282,266 | ) | - | - | (1,275,698 | ) | - | - | - | (6,568 | ) | |||||||||||||||||||||||||||
|
Other
comprehensive income (loss)
|
||||||||||||||||||||||||||||||||||||||||
|
Foreign
currency translation adjustment
|
5,169 | 5,169 | - | - | - | - | 5,169 | - | - | |||||||||||||||||||||||||||||||
|
Comprehensive
loss
|
(1,277,098 | ) | $ | (1,277,097 | ) | |||||||||||||||||||||||||||||||||||
|
Dividend
paid
|
- | |||||||||||||||||||||||||||||||||||||||
|
Balance
at June 30, 2009 (unaudited)
|
105,155,355 | $ | 17,473,770 | $ | 105,155 | $ | 22,108,427 | $ | (18,363,816 | ) | $ | 2,642,775 | $ | 6,300,396 | $ | 503,860 | $ | 4,176,973 | ||||||||||||||||||||||
|
Comprehensive
loss:
|
||||||||||||||||||||||||||||||||||||||||
|
Net
loss
|
(306,878 | ) | (306,878 | ) | (372,397 | ) | 65,519 | |||||||||||||||||||||||||||||||||
|
Other
comprehensive income (loss)
|
||||||||||||||||||||||||||||||||||||||||
|
Foreign
currency translation adjustment
|
43,721 | 43,721 | 43,721 | |||||||||||||||||||||||||||||||||||||
|
Comprehensive
loss
|
(263,157 | ) | (263,157 | ) | ||||||||||||||||||||||||||||||||||||
|
Dividend
paid
|
- | |||||||||||||||||||||||||||||||||||||||
|
Balance
at September 30, 2009 (unaudited)
|
105,155,355 | $ | 16,947,456 | $ | 105,155 | $ | 22,108,427 | $ | (18,736,213 | ) | $ | 2,642,775 | $ | 6,344,117 | $ | 503,860 | $ | 4,242,492 | ||||||||||||||||||||||
The accompanying notes are an integral part of these consolidated
financial statements.
F-5
Benda
Pharmaceutical, Inc.
Notes
to Consolidated Financial Statements
(Amounts
expressed in U.S. Dollars)
1.
Organization
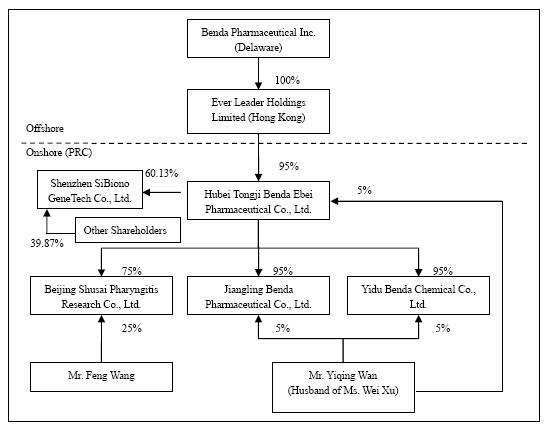
Benda
Pharmaceutical, Inc. (“Benda”) is a corporation organized under the Florida Laws
and headquartered in Hubei Province, the People’s Republic of China
(“PRC”).
Ever
Leader Holdings Limited (“Ever Leader”), a wholly owned subsidiary of Benda, is
a company incorporated under the laws of Hong Kong SAR.
Ever
Leader owns 95% of the issued and outstanding capital of Hubei Tongi Benda Ebei
Pharmaceutical Co. Ltd. (“Benda Ebei”), a Sino-Foreign Equity Joint Venture
company incorporated under the laws of PRC. Mr. Yiqing Wan owns 5% of the issued
and outstanding capital stock of Benda Ebei. Benda Ebei owns: (i) 95% of the
issued and outstanding capital stock of Jiangling Benda Pharmaceutical Co. Ltd.,
(“Jiangling Benda”) a company formed under the laws of PRC; (ii) 95% of the
issued and outstanding capital stock of Yidu Benda Chemical Co. Ltd., (“Yidu
Benda”) a company incorporated under the laws of PRC; and (iii) 75% of the
issued and outstanding capital stock of Beijing Shusai Pharyngitis Research Co.
Ltd., (“Beijing Shusai”) a company incorporated under the laws of PRC. Mr.
Yiqing Wan owns: (i) 5% of the issued and outstanding capital stock of Jingling
Benda; and (ii) 5% of the issued and outstanding capital stock of Yidu Benda.
Mr. Feng Wang owns 25% of the issued and outstanding capital stock of Beijing
Shusai.
On April
5, 2007, Benda Ebei entered into an Equity Transfer Agreements with Shenzhen
Yuanzheng Investment Development Co., Ltd. and Shenzhen Yuanxing Gene City
Development Co., Ltd., the then shareholders of Shenzhen SiBiono GeneTech Co.,
Ltd (“SiBiono”), to purchase 27.57% and 30% respectively of the shares of
SiBiono’s common stock for total consideration of Rmb60 million (or $7.88
million) due and payable on or before April 30, 2007. On June 11, 2007, Benda
Ebei entered into an Equity Transfer Agreements with Huimin Zhang and Yaojin
Wang, the individual shareholders of SiBiono, and to purchase 1.6% and 0.96%
respectively of the shares of SiBiono’s common stock for total consideration of
Rmb2.56 million (or $0.34 million) due and payable on or before June 30, 2007.
Altogether, the total consideration for 60.13% shares of SiBiono’s common stock
was Rmb62.56 million or $9.18 million. As of September 30, 2009, an accumulated
amount, approximately Rmb52.84 million or $7.76 million was paid and leaving a
balance of Rmb 9.72 million or $1.42 million (please refer to Note 10 for the
details).
Benda,
Ever Leader, Benda Ebei, Jingling Benda, Yidu Benda, Beijing Shusai and SiBiono
shall be referred to herein collectively as the “Group”. The Group is engaged
principally in the business of identifying, discovering, developing, and
manufacturing conventional medicines, active pharmaceuticals, bulk chemicals (or
pharmaceutical immediates), and Traditional Chinese Medicines (“TCM”) for the
treatment of some of the most widespread common ailments and diseases (e.g.
common cold, diabetes, and cancer).
F-6
As of
September 30, 2009, the organization and ownership structure of the Group is as
follows:
2.
Basis
of Preparation, Consolidation and Going Concern
The
unaudited consolidated financial statements of Benda and its subsidiaries have
been prepared in accordance with U.S. generally accepted accounting principles
for interim financial information and pursuant to the requirements for reporting
on Form 10-Q. Accordingly, they do not include all the information and footnotes
required by accounting principles generally accepted in the United States
of America for annual financial statements. However, the information included in
these interim financial statements reflects all adjustments (consisting solely
of normal recurring adjustments) which are, in the opinion of management,
necessary for the fair presentation of the consolidated financial position and
the consolidated results of operations. Results shown for interim periods are
not necessarily indicative of the results to be obtained for a full year. The
consolidated balance sheet information as of December 31, 2008 was derived from
the audited consolidated financial statements included in the Group’s Annual
Report on Form 10-K. These interim financial statements should be read in
conjunction with that report. We have reclassified amounts previously reported
in the Group’s financial statements to conform to the current
presentation related to redeemable common stock. The Group has
evaluated subsequent events through the date that the financial statements
were issued which was November 22, 2009, the date immediately preceding the
date of the Group’s Quarterly Report on Form 10-Q for the period ended September
30, 2009.
In the
Group’s Annual Report on Form 10-K for the fiscal year ended December 31, 2008,
the Report of the Independent Registered Public Accounting Firm includes an
explanatory paragraph that describes substantial doubt about the Group’s ability
to continue as a going concern. The Group’s interim financial statements for the
three and nine months ended September 30, 2009 have been prepared on a going
concern basis, which contemplates the realization of assets and the settlement
of liabilities and commitments in the normal course of business. The
Group reported a net loss of $2,825,707 for the nine months ended September 30,
2009 and has a working capital deficit of approximately $27,126,139 and
accumulated deficit of approximately $18,736,213 as of September 30,
2009.
Currently,
the Group does not have the ability to substantially increase its loan
indebtedness with any financial institution , nor can the Group provide any
assurance it will be able to enter into any loan agreements in the future, or be
able to raise funds through further issuance of debt or equity in the
Group. Moreover, the Group presently has little additional resources
with which to obtain or develop new operations.
These
factors raise substantial doubt about the Group’s ability to continue as a going
concern. The financial statements do not contain any adjustments
relating to the recoverability and classification of assets or the amounts and
classification of liabilities that might be necessary should the Group be unable
to continue as a going concern.
While the
Group is attempting to produce sufficient revenues, the Group’s cash position
may not be enough to support the Group’s daily operations. Management
intends to raise additional funds by way of a public or private
offering. Management believes that the actions presently being taking
to further implement its business plan and generate sufficient revenues provide
the opportunity for the Group to continue as a going concern. While
the Group believes in the viability of its strategy to increase revenues and in
its ability to raise additional funds, there can be no assurance to that
effect. The ability of the Group to continue as a going concern is
dependent upon the Group’s ability to further implement its business plan and
generate sufficient revenues. The financial statements do not include
any adjustments that might be necessary it the Group is unable to continue as a
going concern.
F-7
3.
Use
of Estimates
The
preparation of financial statements in conformity with US GAAP requires
management to make estimates and assumptions that affect the reported amounts of
assets and liabilities and disclosure of contingent assets and liabilities at
the date of the financial statements, and the reported amounts of revenue and
expenses during the reporting period. Actual results when ultimately realized
could differ from those estimates.
4.
Significant
Accounting Policies
Cash
and Cash Equivalents
Cash and cash equivalents include cash
on hand, cash on deposit with various financial institutions, and all highly-liquid
investments with original maturities of three months or less at the time of
purchase.
Estimates
Affecting Trade Receivables, Other Receivables, Prepaid and Deposits and
Inventories
The
preparation of our consolidated financial statements requires management to make
estimates and assumptions that affect our reporting of assets and liabilities
(and contingent assets and liabilities). However, it is explicated that the
changes in estimation were not material in the preparation of our consolidation
financial statements.
As of
September 30, 2009 and 2008, the Group provided a $6,867,978 and $1,552,068,
respectively for the allowance of doubtful accounts against trade receivables
(please refer to Note 5 for details). Management's estimate of the appropriate
allowance on those accounts receivable for the reporting periods was based on
the aged nature of these accounts. In making its judgment, management assessed
its customers' ability to continue to pay their outstanding invoices and the
collectibility of those accounts on a timely basis, and whether their financial
position might deteriorate significantly in the future, which would result in
their inability to pay their debts to the Company.
Inventories,
which are primarily comprised of raw materials, packaging materials, and
finished goods, are stated at the lower of cost or net realizable value. Cost
being determined on the basis of a moving average. The Group evaluates the need
for reserves associated with obsolete, slow-moving and non-salable inventory by
reviewing net realizable values on a periodic basis.
As of
September 30, 2009 and 2008, the Group provided a reserve against its obsolete,
slow-moving or non-salable inventory amounting to $ 4,925,299 and $4,191,726,
respectively (please refer to Note 7 for details).
F-8
Property
and Equipment
Property
and equipment are recorded at cost and depreciated using the straight-line
method, with an estimated 5% salvage value of original cost, over the estimated
useful lives of the assets as follows:
|
20-30years
|
|
|
Machinery
and equipment
|
10-15
years
|
|
5
years
|
|
|
Electronics
and office equipment
|
5
years
|
Expenditures
for repairs and maintenance, which do not improve or extend the expected useful
lives of the assets, are expensed as incurred while major replacements and
improvements are capitalized.
When
property or equipment is retired or disposed of, the cost and accumulated
depreciation are removed from the accounts, with any resulting gains or losses
being included in net income or loss in the year of disposition, Impairment of
Long-Lived Assets.
The Group
evaluates potential impairment of long-lived assets, in accordance with
Statement of FASB Accounting Standards Codification (“ASC”) 360, Propoerty, Plant and
Equipment, which
requires the Group to (a) recognize an impairment loss only if the carrying
amount of a long-lived asset is not recoverable from its undiscounted cash flows
and (b) measure an impairment loss as the difference between the carrying amount
and fair value of the asset. As of September 30, 2009, the Group has provided a
full reserve against its Goodwill in the amount of $7,904,613 (Please refer to
Note 10 for details).
Intangible
Assets
The
Group’s intangible assets are stated at cost less accumulated amortization and
are comprised of land-use rights, drug permits and licenses, patent and
technology formulas know-how. Land-use rights are related to land the Group
occupies in Hubei and Guangdong Province, PRC and are being amortized on a
straight-line basis over a period of 40 years. Other intangible assets are being
amortized on a straight-line basis over a period of 10 years.
Revenue
Recognition
Among the
most important accounting policies affecting the Group’s consolidated financial
statements is its policy of recognizing revenue in accordance with the ASC 650,
Under this policy, all of the following criteria must be met in order for us to
recognize revenue:
1. Persuasive evidence of an arrangement
exists;
2. Delivery has occurred or services have
been rendered;
3. The seller's price to the buyer is fixed
or determinable; and
4. Collectibility is reasonably
assured.
The
majority of the Group's revenue results from sales contracts
with distributors and revenue is recorded upon the shipment of goods.
Management conducts credit background checks for new customers as a means
to reduce the subjectivity of assuring collectibility. Based on these
factors, the Group believes that it can apply the provisions of ASC 650
with minimal subjectivity. Sales are presented net of value added tax (VAT). No
return allowance is made as products returns are insignificant based on
historical experience.
F-9
Research
and Development
Research
and development (“R&D”) costs are expensed as incurred and consist primarily
of salaries and related expenses of personnel engaged in research and
development activities. The Group spent $283,473 and $249,357 on R&D efforts
for the nine month ended September 30, 2009 and 2008, respectively.
Income
Taxes
The Group
accounts for income taxes under the liability method in accordance with
ASC 740, Income
Taxes. Deferred tax assets and liabilities are recorded for the estimated
future tax effects of temporary differences between the tax basis of assets and
liabilities and amounts reported in the accompanying consolidated balance
sheets. Deferred tax assets are reduced by a valuation allowance if current
evidence indicates that it is considered more likely than not that these
benefits will not be realized.
Comprehensive
Income
The Group
has adopted ASC 220, Comprehensive Income, which establishes standards
for reporting and displaying comprehensive income, its components, and
accumulated balances in a full-set of general-purpose financial statements.
Accumulated other comprehensive income represents the accumulated balance of
foreign currency translation adjustments.
Concentration
of Credit Risk
A
significant portion of the Group's cash at September 30, 2009 and 2008 is
maintained at various financial institutions in the PRC which do not provide
insurance for amounts on deposit.
The Group
has not experienced any losses in such accounts and believes it is not exposed
to significant credit risk in this area.
The Group
operates principally in the PRC and grants credit to its customers in this
geographic region. Although the PRC is economically stable, it is
always possible that unanticipated events in foreign countries could disrupt the
Group’s operations.
The
following table shows the individual customer’s revenue and account receivable
balance which was higher than 5% of total revenue and total account receivables
for the nine months and three months ended September 30, 2009 and
2008:
|
NINE
MONTHS ENDED
SEPTEMBER
30,
|
THREE
MONTH ENDED
SEPTEMBER
30,
|
||||||||||||||||||||||||||||||||||
|
2009
|
2008
|
2009
|
2008
|
||||||||||||||||||||||||||||||||
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
||||||||||||||||||||||||||||||||
|
Revenue
|
$ | 17,345,065 |
%
|
$ | 19,860,057 |
%
|
$ | 5,904,653 |
%
|
$ | 6,634,434 |
%
|
|||||||||||||||||||||||
|
Individual customer's
revenue
|
|||||||||||||||||||||||||||||||||||
| 1 |
Zhuhai
Gongbei Pharmaceutical Co, Ltd.
|
4,195,255 | 24 | % | 3,711,204 | 19 | % | 1,234,516 | 21 | % | 784,911 | 12 | % | ||||||||||||||||||||||
| 2 |
Shenyang
Pharmaceutical Co. Ltd.
|
2,234,332 | 13 | % | 1,985,584 | 10 | % | 661,749 | 11 | % | 711,963 | 11 | % | ||||||||||||||||||||||
| 3 |
Shenzhen
Huihua Pharmaceutical Co. Ltd.
|
2,005,451 | 12 | % | 2,191,049 | 11 | % | 565,257 | 10 | % | 800,653 | 12 | % | ||||||||||||||||||||||
| 4 |
Jiangxi
Huiren Pharmaceutical Co. Ltd.
|
1,149,220 | 7 | % | 1,602,885 | 8 | % | 263,982 | 4 | % | 647,020 | 10 | % | ||||||||||||||||||||||
| 5 |
Hubei
Hengchuan Health Products Co.,Ltd.
|
1,388,420 | 8 | % | 2,221,744 | 11 | % | 687,044 | 12 | % | 578,655 | 9 | % | ||||||||||||||||||||||
| 6 |
Huhan
Jiuding Pharmaceutical Co, Ltd.
|
1,106,645 | 6 | % | - | - | 676,870 | 11 | % | - | - | ||||||||||||||||||||||||
|
Account
receivable, gross
|
$ | 15,551,342 |
%
|
$ | 13,222,313 |
%
|
15,551,342 |
%
|
13,222,313 |
%
|
|||||||||||||||||||||||||
|
Individual customer's
account receivable gross balance
|
|||||||||||||||||||||||||||||||||||
| 1 |
Zhuhai
Gongbei Pharmaceutical Co, Ltd.
|
2,326,483 | 15 | % | 1,769,557 | 13 | % | 2,326,483 | 15 | % | 1,769,557 | 13 | % | ||||||||||||||||||||||
| 3 |
Shenzhen
Huihua Pharmaceutical Co. Ltd.
|
1,593,203 | 10 | % | 1,186,189 | 9 | % | 1,593,203 | 10 | % | 1,186,189 | 9 | % | ||||||||||||||||||||||
| 4 |
Shenyang
Pharmaceutical Co. Ltd.
|
1,429,213 | 9 | % | 963,855 | 7 | % | 1,429,213 | 9 | % | 963,855 | 7 | % | ||||||||||||||||||||||
| 5 |
Hubei
Hengchuan Health Products Co.,Ltd.
|
1,276,973 | 8 | % | 1,232,435 | 9 | % | 1,276,973 | 8 | % | 1,232,435 | 9 | % | ||||||||||||||||||||||
| 2 |
Jiangxi
Huiren Pharmaceutical Co. Ltd.
|
923,509 | 6 | % | 519,536 | 4 | % | 923,509 | 6 | % | 519,536 | 4 | % | ||||||||||||||||||||||
| 6 |
Huhan
Jiuding Pharmaceutical Co, Ltd.
|
944,909 | 6 | % | - | - | 944,909 | 6 | % | - | - | ||||||||||||||||||||||||
F-10
Basic
and Diluted Earnings Per Share
The Group
adopted ASC 260, Earnings Per
Share. ASC 260 requires the presentation of earnings per share (EPS) as
Basic and Diluted EPS. Basic earnings per share are calculated by taking net
income divided by the weighted average shares of common stock outstanding during
the period. Diluted earnings per share is calculated by taking basic weighted
average shares of common stock and increasing it for dilutive common stock
equivalents such as warrants that are in the money.
Foreign
Currency Translation
The
functional currency of the Group is the Renminbi (“RMB”), the PRC’s currency.
The Group maintains its financial statements using the functional
currency. Monetary assets and liabilities denominated in currencies
other than the functional currency are translated into the functional currency
at rates of exchange prevailing at the balance sheet dates. Transactions
denominated in currencies other than the functional currency are translated into
the functional currency at the exchange rates prevailing at the dates of the
transaction. Exchange gains or losses arising from foreign currency transactions
are included in the determination of net income (loss) for the respective
periods.
For
financial reporting purposes, the financial statements of the Group, which are
prepared using the RMB, are translated into the Group’s reporting currency,
United States Dollars. Balance sheet accounts are translated using the closing
exchange rate in effect at the balance sheet date and income and expense
accounts are translated using the average exchange rate prevailing during the
reporting period. Adjustments resulting from the translation, if any,
are included in accumulated other comprehensive income (loss) in stockholder’s
equity.
The
exchange rates in effect at September 30, 2009 and 2008 were stated as follows:
(for RMB 1.00):
|
September
30,
|
September
30,
|
|||||||
|
2009
|
2008
|
|||||||
|
(Unaudited)
|
(Unaudited)
|
|||||||
|
Fixed
rate
|
$ | 0.1467 | $ | 0.1463 | ||||
|
Average
rate
|
$ | 0.1466 | $ | 0.1434 | ||||
For the
nine months ended September 30, 2009 and 2008, the foreign exchange loss was
$6,594 and $7,060 respectively; for the three months ended September 30, 2009
and 2008, the foreign exchange loss was $4,360 and $4,904,
respectively.
Fair
Value of Financial Instruments
ASC 825,
Financial Instruments,
defines financial instruments and requires fair value disclosures of those
financial instruments. On January 1, 2008, the Group adopted ASC 820, Fair Value Measurements and
Disclosures, which defines fair value, establishes a three-level
valuation hierarchy for disclosures of fair value measurement and enhances
disclosures requirements for fair value measures. Current assets and current
liabilities qualified as financial instruments and management believes their
carrying amounts are a reasonable estimate of fair value because of the short
period of time between the origination of such instruments and their expected
realization and if applicable, their current interest rate is equivalent to
interest rates currently available. The three levels are defined as
follow:
|
|
·
|
Level
1 -- inputs to the valuation methodology are quoted prices (unadjusted)
for identical assets or liabilities in active
markets.
|
F-11
|
|
·
|
Level
2 -- inputs to the valuation methodology include quoted prices for similar
assets and liabilities in active markets, and inputs that are observable
for the assets or liability, either directly or indirectly, for
substantially the full term of the financial
instruments.
|
|
|
·
|
Level
3 -- inputs to the valuation methodology are unobservable and significant
to the fair value.
|
As of the
balance sheet date, the estimated fair values of the financial instruments were
not materially different from their carrying values as presented due to the
short maturities of these instruments and that the interest rates on the
borrowings approximate those that would have been available for loans of similar
remaining maturity and risk profile at respective period-ends. Determining which
category an asset or liability falls within the hierarchy requires significant
judgment. The Group evaluates the hierarchy disclosures each
quarter.
Recent
Issued Accounting Pronouncements
In
June 2009, the Financial Accounting Standards Board (FASB) issued a
standard that established the FASB Accounting Standards Codification (ASC) and amended the
hierarchy of generally accepted accounting principles (ASC) and amended the
hierarchy of generally accepted accounting principles (GAAP) such that the ASC
became the single source of authoritative nongovernmental U.S. GAAP. The ASC did
not change current U.S. GAAP, but was intended to simplify user access to all
authoritative U.S. GAAP by providing all the authoritative literature related to
a particular topic in one place. All previously existing accounting standard
documents were superseded and all other accounting literature not included in
the ASC is considered non-authoritative. New accounting standards issued
subsequent to June 30, 2009 are communicated by the FASB through Accounting
Standards Updates (ASUs). The Company adopted the ASC on July 1, 2009. This
standard did not have an impact on the Company’s consolidated results of
operations or financial condition. However, throughout the notes to the
consolidated financial statements references that were previously made to
various former authoritative U.S. GAAP pronouncements have been changed to
coincide with the appropriate section of the ASC.
In
September 2006, the FASB issued an accounting standard codified in ASC 820,
Fair Value Measurements and
Disclosures. This standard established a single definition of fair value
and a framework for measuring fair value, set out a fair value hierarchy to be
used to classify the source of information used in fair value measurements, and
required disclosures of assets and liabilities measured at fair value based on
their level in the hierarchy. This standard applies under other accounting
standards that require or permit fair value measurements. One of the amendments
deferred the effective date for one year relative to nonfinancial assets and
liabilities that are measured at fair value, but are recognized or disclosed at
fair value on a nonrecurring basis. This deferral applied to such items as
nonfinancial assets and liabilities initially measured at fair value in a
business combination (but not measured at fair value in subsequent periods) or
nonfinancial long-lived asset groups measured at fair value for an impairment
assessment. The adoption of the fair value measurement standard did not
have a material impact on the Company’s consolidated results of operations or
financial condition.
F-12
In
December 2007, the FASB issued and, in April 2009, amended a new
business combinations standard codified within ASC 805, which changed the
accounting for business acquisitions. Accounting for business combinations under
this standard requires the acquiring entity in a business combination to
recognize all (and only) the assets acquired and liabilities assumed in the
transaction and establishes the acquisition-date fair value as the measurement
objective for all assets acquired and liabilities assumed in a business
combination. Certain provisions of this standard impact the determination of
acquisition-date fair value of consideration paid in a business combination
(including contingent consideration); exclude transaction costs from acquisition
accounting; and change accounting practices for acquisition-related
restructuring costs, in-process research and development, indemnification
assets, and tax benefits. The Company adopted the standard for business
combinations and adjustments to an acquired entity’s deferred tax asset and
liability balances and it had no immediate impact on the Company’s consolidated
financial position or results of operations.
In
December 2007, the FASB issued a new standard which established the
accounting for and reporting of noncontrolling interests (NCIs) in partially
owned consolidated subsidiaries and the loss of control of subsidiaries. Certain
provisions of this standard indicate, among other things, that NCIs (previously
referred to as minority interests) be treated as a separate component of equity,
not as a liability (as was previously the case); that increases and decreases in
the parent’s ownership interest that leave control intact be treated as equity
transactions, rather than as step acquisitions or dilution gains or losses; and
that losses of a partially owned consolidated subsidiary be allocated to the NCI
even when such allocation might result in a deficit balance. This standard also
required changes to certain presentation and disclosure requirements. The
Company adopted the standard beginning January 1, 2009. The provisions of
the standard were applied to all NCIs prospectively, except for the presentation
and disclosure requirements, which were applied retrospectively to all periods
presented. As a result, upon adoption, the Company retroactively reclassified
the “Minority interest in subsidiaries” balance previously included in the
“Other liabilities” section of the consolidated balance sheet to a new component
of equity with respect to NCIs in consolidated subsidiaries. The adoption also
impacted certain captions previously used on the consolidated statement of
income, largely identifying net income including NCI and net income attributable
to the Company. The adoption of this standard did not have a material
impact on the Company’s consolidated financial position or results of
operations.
In
April 2009, the FASB issued an accounting standard which provides guidance
on (1) estimating the fair value of an asset or liability when the volume
and level of activity for the asset or liability have significantly declined and
(2) identifying transactions that are not orderly. The standard also
amended certain disclosure provisions for fair value measurements and
disclosures in ASC 820 to require, among other things, disclosures in interim
periods of the inputs and valuation techniques used to measure fair value as
well as disclosure of the hierarchy of the source of underlying fair value
information on a disaggregated basis by specific major category of investment.
For 3M, this standard was effective prospectively beginning April 1, 2009.
The adoption of this standard did not have a material impact on the Company’s
consolidated results of operations or financial condition.
In
April 2009, the FASB issued an accounting standard which modifies the
requirements for recognizing other-than-temporarily impaired debt securities and
changes the existing impairment model for such securities. The standard also
requires additional disclosures for both annual and interim periods with respect
to both debt and equity securities. Under the standard, impairment of debt
securities will be considered other-than-temporary if an entity (1) intends
to sell the security, (2) more likely than not will be required to sell the
security before recovering its cost, or (3) does not expect to recover the
security’s entire amortized cost basis (even if the entity does not intend to
sell). The standard further indicates that, depending on which of the above
factor(s) causes the impairment to be considered other-than-temporary,
(1) the entire shortfall of the security’s fair value versus its amortized
cost basis or (2) only the credit loss portion would be recognized in
earnings while the remaining shortfall (if any) would be recorded in other
comprehensive income. The standard requires entities to initially apply its
provisions to previously other-than-temporarily impaired debt securities
existing as of the date of initial adoption by making a cumulative-effect
adjustment to the opening balance of retained earnings in the period of
adoption. The cumulative-effect adjustment potentially reclassifies the
noncredit portion of a previously other-than-temporarily impaired debt security
held as of the date of initial adoption from retained earnings to accumulated
other comprehensive income. The adoption of this standard did not have a
material impact on the Company’s consolidated results of operations or financial
condition.
F-13
In
April 2009, the FASB issued an accounting standard regarding interim
disclosures about fair value of financial instruments. The standard essentially
expands the disclosure about fair value of financial instruments that were
previously required only annually to also be required for interim period
reporting. In addition, the standard requires certain additional disclosures
regarding the methods and significant assumptions used to estimate the fair
value of financial instruments. The adoption of this standard did not have a
material impact on the Company’s consolidated results of operations or financial
condition.
In
May 2009, the FASB issued a new accounting standard regarding subsequent
events. This standard incorporates into authoritative accounting literature
certain guidance that already existed within generally accepted auditing
standards, with the requirements concerning recognition and disclosure of
subsequent events remaining essentially unchanged. This guidance addresses
events which occur after the balance sheet date but before the issuance of
financial statements. Under the new standard, as under previous practice, an
entity must record the effects of subsequent events that provide evidence about
conditions that existed at the balance sheet date and must disclose but not
record the effects of subsequent events which provide evidence about conditions
that did not exist at the balance sheet date. This standard added an additional
required disclosure relative to the date through which subsequent events have
been evaluated and whether that is the date on which the financial statements
were issued. For the Company,
this standard was effective beginning April 1, 2009.
In
June 2009, the FASB issued a new standard regarding the accounting for
transfers of financial assets amending the existing guidance on transfers of
financial assets to, among other things, eliminate the qualifying
special-purpose entity concept, include a new unit of account definition that
must be met for transfers of portions of financial assets to be eligible for
sale accounting, clarify and change the derecognition criteria for a transfer to
be accounted for as a sale, and require significant additional disclosure. The
standard is effective for new transfers of financial assets beginning
January 1, 2010. The adoption of this standard is not expected to have a
material impact on the Company’s consolidated results of operations or financial
condition.
In
June 2009, the FASB issued an accounting standard that revised the
consolidation guidance for variable-interest entities. The modifications include
the elimination of the exemption for qualifying special purpose entities, a new
approach for determining who should consolidate a variable-interest entity, and
changes to when it is necessary to reassess who should consolidate a
variable-interest entity. The standard is effective January 1, 2010. The
Company is currently evaluating the impact of this standard, but would not
expect it to have a material impact on the Company’s consolidated results of
operations or financial condition.
F-14
In
August 2009, the FASB issued ASU No. 2009-05, Measuring Liabilities at Fair
Value, which provides additional guidance on how companies should measure
liabilities at fair value under ASC 820. The ASU clarifies that the quoted price
for an identical liability should be used. However, if such information is not
available, a entity may use, the quoted price of an identical liability when
traded as an asset, quoted prices for similar liabilities or similar liabilities
traded as assets, or another valuation technique (such as the market or income
approach). The ASU also indicates that the fair value of a liability is not
adjusted to reflect the impact of contractual restrictions that prevent its
transfer and indicates circumstances in which quoted prices for an identical
liability or quoted price for an identical liability traded as an asset may be
considered level 1 fair value. This ASU is effective October 1, 2009. The
Company is currently evaluating the impact of this standard, but would not
expect it to have a material impact on the Company’s consolidated results of
operations or financial condition.
In
September 2009, the FASB issued ASU No. 2009-12, Investments in Certain Entities That
Calculate Net Asset Value per Share (or Its Equivalent), that amends ASC
820 to provide guidance on measuring the fair value of certain alternative
investments such as hedge funds, private equity funds and venture capital funds.
The ASU indicates that, under certain circumstance, the fair value of such
investments may be determined using net asset value (NAV) as a practical
expedient, unless it is probable the investment will be sold at something other
than NAV. In those situations, the practical expedient cannot be used and
disclosure of the remaining actions necessary to complete the sale is required.
The ASU also requires additional disclosures of the attributes of all
investments within the scope of the new guidance, regardless of whether an
entity used the practical expedient to measure the fair value of any of its
investments. This ASU is effective October 1, 2009. The Company is
currently evaluating the impact of this standard, but would not expect it to
have a material impact on the Company’s consolidated results of operations or
financial condition.
In
October 2009, the FASB issued ASU No. 2009-13, Multiple-Deliverable Revenue
Arrangements—a consensus of the FASB Emerging Issues Task Force, that
provides amendments to the criteria for separating consideration in
multiple-deliverable arrangements. As a result of these amendments,
multiple-deliverable revenue arrangements will be separated in more
circumstances than under existing U.S. GAAP. The ASU does this by establishing a
selling price hierarchy for determining the selling price of a deliverable. The
selling price used for each deliverable will be based on vendor-specific
objective evidence if available, third-party evidence if vendor-specific
objective evidence is not available, or estimated selling price if neither
vendor-specific objective evidence nor third-party evidence is available. A
vendor will be required to determine its best estimate of selling price in a
manner that is consistent with that used to determine the price to sell the
deliverable on a standalone basis. This ASU also eliminates the residual method
of allocation and will require that arrangement consideration be allocated at
the inception of the arrangement to all deliverables using the relative selling
price method, which allocates any discount in the overall arrangement
proportionally to each deliverable based on its relative selling price. Expanded
disclosures of qualitative and quantitative information regarding application of
the multiple-deliverable revenue arrangement guidance are also required under
the ASU. The ASU does not apply to arrangements for which industry specific
allocation and measurement guidance exists, such as long-term construction
contracts and software transactions. The ASU is effective beginning
January 1, 2011. The Company is currently evaluating the impact of this
standard on 3M’s consolidated results of operations and financial
condition.
F-15
In
October 2009, the FASB issued ASU No. 2009-14, Certain Revenue Arrangements That
Include Software Elements—a consensus of the FASB Emerging Issues Task
Force, that reduces the types of transactions that fall within the
current scope of software revenue recognition guidance. Existing software
revenue recognition guidance requires that its provisions be applied to an
entire arrangement when the sale of any products or services containing or
utilizing software when the software is considered more than incidental to the
product or service. As a result of the amendments included in ASU
No. 2009-14, many tangible products and services that rely on software will
be accounted for under the multiple-element arrangements revenue recognition
guidance rather than under the software revenue recognition guidance. Under the
ASU, the following components would be excluded from the scope of software
revenue recognition guidance: the tangible element of the product,
software products bundled with tangible products where the software components
and non-software components function together to deliver the product’s essential
functionality, and undelivered components that relate to software that is
essential to the tangible product’s functionality. The ASU also provides
guidance on how to allocate transaction consideration when an arrangement
contains both deliverables within the scope of software revenue guidance
(software deliverables) and deliverables not within the scope of that guidance
(non-software deliverables). The ASU is effective beginning January 1,
2011. The Company is currently evaluating the impact of this standard on the
Company’s consolidated results of operations and financial
condition.
5.
Trade
Receivables, Other Receivables, Prepaid expenses and Deposits
As
mentioned in Note 4 that the company estimates of the appropriate allowance on
those accounts receivable for the reporting periods was based on the aged nature
of these accounts. The table below shows the allowance for doubtful debts of the
Group’s trade receivables, other receivables and prepaid expenses and
deposits as of September 30, 2009 and December 31, 2008:
|
September
30,
|
December
31,
|
|||||||
|
2009
|
2008
|
|||||||
|
(Unaudited)
|
||||||||
|
Trade
receivables, gross
|
15,551,342 | $ | 13,123,374 | |||||
|
Allowance
for doubtful debts
|
(6,867,978 | ) | (3,786,459 | ) | ||||
|
Trade
receivables, net
|
8,683,364 | $ | 9,336,915 | |||||
|
September
30,
|
December
31,
|
|||||||
|
2009
|
2008
|
|||||||
|
Other receivables,
gross
|
678,528 | $ | 504,349 | |||||
|
Allowance
for doubtful debts
|
- | - | ||||||
|
Other receivables,
net
|
678,528 | $ | 504,349 | |||||
|
September
30,
|
December
31,
|
|||||||
|
2009
|
2008
|
|||||||
|
Prepaid
and deposits, gross
|
2,540,267 | $ | 918,209 | |||||
|
Allowance
for doubtful debts
|
- | |||||||
|
Prepaid
and deposits, net
|
2,540,267 | $ | 918,209 | |||||
F-16
The
change of the allowance for doubtful debts between the reporting periods, as of
September 30, 2009 and 2008, is displayed as follows:
|
NINE
MONTHS ENDED
SEPTEMBER
30,
|
||||||||
|
2009
|
2008
|
|||||||
|
(Unaudited)
|
(Unaudited)
|
|||||||
|
Balance
at beginning of period
|
$ | (3,786,459 | ) | $ | (943,647 | ) | ||
|
Provision
during the period
|
(3,080,334 | ) | (534,124 | ) | ||||
|
Foreign
exchange difference
|
(1,185 | ) | (74,297 | ) | ||||
|
Balance
at end of period
|
$ | (6,867,978 | ) | $ | (1,552,068 | ) | ||
The Group
set full allowance for trade receivables aged over 120 days which is general
credit term granted to the customers. After deducting those allowances from the
gross trade receivable, based on the management’s past experience, there would
be no collectability issue on the net amount.
6.
Refundable
Purchase Price Paid
On,
December 7, 2006, Benda Ebei paid $1.2 million to SECO (Shenzhen) Biotech Co.,
Ltd. (“SECO”) pursuant to a purchase agreement signed between SECO and Benda
Ebei on December 3, 2006 to acquire a technology know-how and drug
specifications and technical parameters in producing a Gastropathy drug owned by
SECO. According to the signed contract:
|
·
|
The
amount paid is refundable only if SECO fails to pass the examination
conducted by Benda Ebei;
|
|
·
|
Altogether,
there are three phases for the examination; however there is no specific
time-table for the examination;
|
|
·
|
The
contract is valid for 10 years upon the contract
signed.
|
As at
September 30, 2009, the deal has not been closed as the product is still under
the process of development. As the date when could SECO pass the examination
conducted by Benda Ebei cannot be determined, thus the amount paid is reported
as non-current assets as of September 30, 2009.
7.
Inventories,
Net
The
Group’s inventories at September 30, 2009 and December 31, 2008 comprised as
follows:
|
September
30,
|
December
31,
|
|||||||
|
2009
|
2008
|
|||||||
|
(Unaudited)
|
||||||||
|
Raw
materials
|
$ | 899,885 | $ | 874,180 | ||||
|
Packing
materials
|
161,938 | 481,724 | ||||||
|
Other
materials / supplies
|
83,510 | 88,412 | ||||||
|
Finished
goods
|
626,561 | 754,838 | ||||||
|
Work-in-process
|
5,497,187 | 5,086,780 | ||||||
|
Total
inventories at cost
|
7,269,082 | 7,285,934 | ||||||
|
Less:
Reserves on inventories
|
(4,925,299 | ) | (4,941,372 | ) | ||||
|
Total
inventories, net
|
$ | 2,343,783 | $ | 2,344,562 | ||||
F-17
A reserve
for obsolete, slow-moving or non-salable inventory was made on raw materials,
packing materials, other material and suppliers, finished goods and
work-in-process at the amount of $82,026, $39,786, $54,563, $6,507 and
$4,742,417 respectively as of September 30, 2009. As of December 31, 2008, there
was a reserve made on raw materials, packing materials, finished goods and
work-in-process at the amount of $122,433, $7,118, $269,749 and $4,542,072
respectively.
The
provision of reserve on work-in-process was resulted from the manufacturing
process of Gendicine, SiBiono’s sole product and SiBiono was acquired by the
company in April 2007.
The
following chart shows the manufacturing process of Gendicine
(Ad-p53):
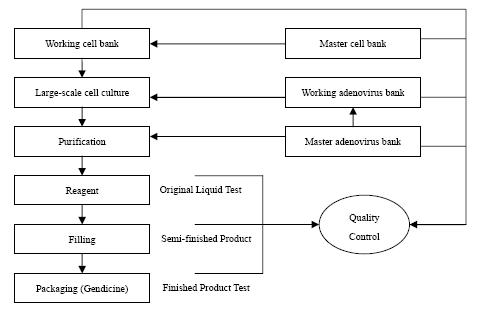
In the
production process of finished goods, Gendicine, several working steps are
needed: (i) large-scale culturing of adenovirus from master adenovirus bank;
(ii) culturing of cell from master cell bank; (iii) purification. The whole
process including step (i) to step (iii) takes approximately twenty-four days to
make reagent (“original liquid”). This particular liquid can only be stored for
approximately five years. It takes approximately another seven days for mixing
and bottling original liquid to finished goods which is known as
Gendicine.
Therefore,
up to the stage of reagent, all the related production costs are treated as
work-in-process. The major components of those production costs are: (i) direct
labor; (ii) direct materials; (iii) power; (iv) supplies and other materials and
(v) manufacturing overheads.
Before
acquisition, as of March 31, 2007, the accumulated units of original liquid
produced was 198,075 and which could be converted to approximately 226,736 vials
of Gendicine. However, the accumulated vials of Gendicine sold throughout the
years 2004 to three-months period ended March 31, 2007 were only approximately
18,424 vials. The accumulated production costs of $4,080,644 were remained as
work-in-process as of three-month period ended March 31, 2007.
F-18
Furthermore,
due to the special feature of the original liquid which can only be stored for
five years, and most of the original liquid was produced in the year of 2004,
and the provision of reserve on work in process was $3,696,083 as of three-month
period ended March 31, 2007.
After the
acquisition with the effective date April 1, 2007, the same accounting treatment
was adopted for the treatment of the provision of reserve on work-in-process. As
of September 30, 2009, the provision of reserve on work in process was
$4,742,417.
8.
Property
and Equipment, Net
The Group’s property and equipment at September 30, 2009 and December 31, 2008 were comprised as
follows:
|
December
31, 2008
|
Addition
|
Disposal
|
Foreign
Currency Translation Difference
|
September
30,
2009
|
||||||||||||||||
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
|||||||||||||||||
|
Buildings
|
$ | 9,692,441 | - | - | 3,261 | 9,695,702 | ||||||||||||||
|
Machinery
and equipment
|
16,769,328 | 10,444 | - | (890 | ) | 16,778,882 | ||||||||||||||
|
Office
equipment
|
40,728 | 3,356 | - | (3,779 | ) | 40,305 | ||||||||||||||
|
Motor
Vehicles
|
252,163 | - | - | (14 | ) | 252,149 | ||||||||||||||
|
Cost
|
26,754,660 | 13,800 | - | (1,421 | ) | 26,767,039 | ||||||||||||||
|
Less:
Accumulated Depreciation
|
||||||||||||||||||||
|
Buildings
|
$ | (1,559,511 | ) | (341,070 | ) | - | (367 | ) | (1,900,948 | ) | ||||||||||
|
Machinery
and equipment
|
(4,160,667 | ) | (1,221,246 | ) | - | (480 | ) | (5,382,393 | ) | |||||||||||
|
Office
equipment
|
(30,839 | ) | (17,970 | ) | - | (9 | ) | (48,818 | ) | |||||||||||
|
Motor
Vehicles
|
(89,863 | ) | (29,028 | ) | - | (14 | ) | (118,905 | ) | |||||||||||
|
Accumulated
Depreciation
|
(5,840,880 | ) | (1,609,314 | ) | - | (870 | ) | (7,451,064 | ) | |||||||||||
|
Construction
in progress
|
$ | 8,143,445 | 441,765 | 77 | (78 | ) | 8,585,055 | |||||||||||||
|
Total
property and equipment, net
|
$ | 29,057,225 | 27,901,030 | |||||||||||||||||
As
mentioned in Note 11, Benda Ebei entered into a commercial bank note issuance
agreement with Shanghai Pudong Development Bank on August 14, 2007 and a
supplementary agreement on January 21, 2008. Under the agreements the credit
facility is secured by the buildings, machinery and equipment of Benda Ebei and
Jiangling Benda. As of September 30, 2009, the net book value of pledged
property and equipment was approximately Rmb123.40 million (or $18.1 million) in
total.
The
depreciation expense for the nine months ended September 30, 2009 was calculated
as follows:
|
Original
Cost
|
||||||||||||||||||||||||||||||||
|
December
31,
|
September
30
|
Depreciation
|
Deprecation
|
|||||||||||||||||||||||||||||
|
2008
|
2009
|
Average
|
Salvage
|
Estimate
|
Calculated
|
Reported
|
Difference
|
|||||||||||||||||||||||||
|
(Unaduited)
|
(Unaduited)
|
Value
|
Useful
Lives
|
(Unaduited)
|
(Unaduited)
|
(Unaduited)
|
||||||||||||||||||||||||||
|
Building
|
$ | 9,692,441 | 9,695,702 | 9,694,071 | 5 | % | 20 | 345,351 | 341,070 | 4,281 | ||||||||||||||||||||||
|
Property
and equipment
|
16,769,328 | 16,778,882 | 16,774,105 | 5 | % | 11 | 1,138,243 | 1,221,246 | (83,003 | ) | ||||||||||||||||||||||
|
Office
equipment
|
40,728 | 40,305 | 40,517 | 5 | % | 5 | 5,774 | 17,970 | (12,196 | ) | ||||||||||||||||||||||
|
Motor
vehicle
|
252,163 | 252,149 | 252,156 | 5 | % | 5 | 35,932 | 29,028 | 6,904 | |||||||||||||||||||||||
|
Total
property and equipments
|
$ | 26,754,660 | 26,767,039 | 26,760,849 | 1,525,300 | 1,609,314 | (84,014 | ) | ||||||||||||||||||||||||
The above
table shows the calculation of depreciation expenses for the nine months ended
September 30, 2009. The difference between the depreciation calculated and
depreciation reported was due to the changes of foreign exchange
translation.
F-19
The
estimated useful lives for buildings and property and equipments vary from 20 to
30 years and 10 to 15 years, respectively. For the nine months ended September
30, 2009, the majority of the building subjected to depreciation had useful
lives of 20 years and property and equipment had useful lives of 11
years.
The total
depreciation expense was $1,609,314 and $490,915 for the nine months and three
months ended September 30, 2009, respectively, and is broken down as
follows:
|
NINE
MONTHS ENDED
SEPTEMBER
30,
|
THREE
MONTHS ENDED
SEPTEMBER
30,
|
|||||||||||||||
|
2009
|
2008
|
2009
|
2008
|
|||||||||||||
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
|||||||||||||
|
Cost
of sales
|
$ | 901,692 | $ | 1,041,124 | $ | 300,598 | $ | 381,947 | ||||||||
|
Operating
expenses
|
707,622 | 373,639 | 328,327 | 130,144 | ||||||||||||
|
Balance
at end of period
|
$ | 1,609,314 | $ | 1,414,763 | $ | 628,925 | $ | 512,091 | ||||||||
9.
Intangible
Assets, Net
The
Group’s intangible assets at September 30, 2009 and December 31, 2008 were
comprised as follows:
|
December
31,
2008
|
Addition
|
Foreign
Currency Translation Difference
|
September
30,
2009
|
|||||||||||||
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
||||||||||||||
|
Land-use
rights
|
$ | 3,117,135 | - | (157 | ) | $ | 3,116,978 | |||||||||
|
Drugs
permits and licenses
|
2,780,254 | - | (140 | ) | 2,780,114 | |||||||||||
|
Technology
formulas
|
1,012,129 | - | (43 | ) | 1,012,086 | |||||||||||
|
Patent
|
1,803,829 | - | (77 | ) | 1,803,752 | |||||||||||
|
Cost
|
8,713,347 | - | (417 | ) | 8,712,930 | |||||||||||
|
Land-use
rights
|
$ | (279,127 | ) | (46,556 | ) | (20 | ) | $ | (325,703 | ) | ||||||
|
Drugs
permits and licenses
|
(1,664,541 | ) | (206,261 | ) | (72 | ) | (1,870,874 | ) | ||||||||
|
Technology
formulas
|
(254,681 | ) | (75,860 | ) | (38 | ) | (330,579 | ) | ||||||||
|
Patent
|
(511,896 | ) | (219,242 | ) | (111 | ) | (731,249 | ) | ||||||||
|
Accumulated
amortization
|
(2,710,245 | ) | (547,919 | ) | (241 | ) | (3,258,405 | ) | ||||||||
|
Total
intangible assets, net
|
$ | 6,003,102 | (547,919 | ) | (658 | ) | $ | 5,454,525 | ||||||||
The
amortization expense for the nine months ended September 30, 2009 was calculated
as follows:
|
Original
Cost
|
||||||||||||||||||||||||||||
|
December
31,
|
September
30
|
Amortization
|
Amortization
|
|||||||||||||||||||||||||
|
2008
|
2009
|
Average
|
Estimated
|
Calculated
|
Reported
|
Difference
|
||||||||||||||||||||||
|
(Unaudited)
|
(Unaudited)
|
Useful
lives
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
|||||||||||||||||||||||
|
Land-use
rights
|
$ | 3,117,136 | 3,116,978 | 3,117,057 | 40 | $ | 58,445 | 46,556 | 11,889 | |||||||||||||||||||
|
Drugs
permits and licenses
|
2,780,254 | 2,780,114 | 2,780,184 | 10 | 208,514 | 206,261 | 2,253 | |||||||||||||||||||||
|
Technology
formulas
|
1,012,129 | 1,012,086 | 1,012,108 | 10 | 75,908 | 75,860 | 48 | |||||||||||||||||||||
|
Patent
|
1,803,829 | 1,803,752 | 1,803,791 | 6 | 225,474 | 219,242 | 6,232 | |||||||||||||||||||||
|
Total
Intangible assets
|
$ | 8,713,348 | 8,712,930 | 8,713,139 | $ | 568,341 | 547,919 | 20,422 | ||||||||||||||||||||
The above
table shows the calculation of amortization expenses for the nine months ended
September 30, 2009. The difference between the amortization calculated and
amortization reported was due to the changes of foreign exchange
translation.
F-20
The total
amortization expense for the nine months and the three months ended September
30, 2009, and 2008 are broken down as follows:
|
NINE
MONTHS ENDED
SEPTEMBER
30,
|
THREE MONTHS
ENDED
SEPTEMBER
30,
|
|||||||||||||||
|
2009
|
2008
|
2009
|
2008
|
|||||||||||||
|
(Unaudited)
|
(Unaudited)
|
|||||||||||||||
|
Cost
of sales
|
$ | 428,801 | $ | 418,425 | $ | 142,976 | $ | 142,758 | ||||||||
|
Operating
expenses
|
119,118 | 148,591 | 39,718 | 50,643 | ||||||||||||
|
Balance
at end of period
|
$ | 547,919 | $ | 567,016 | $ | 182,694 | $ | 193,401 | ||||||||
10.
Goodwill
and Acquisition Cost Payable
ASC 805,
Business Combinations,
requires all acquisitions must be accounted for by allocating the acquisition
consideration to the assets acquired based upon the fair market value of those
assets. Consideration value that cannot be allocated to the acquired assets must
be assigned to goodwill. In addition, it also requires eliminating the pooling
treatment and eliminating the amortization of goodwill. ASC 350, Intangibles –
Goodwill and Others, requires that a company carrying goodwill on its books must
revalue the assets acquired in a business combination. If there is an overall
decline in the value of the acquired assets, then earlier booked goodwill is
deemed “impaired” and must be written down. ASC 350 requires a two step
impairment test. The fair value of a reporting unit is first compared to its
carrying value, including goodwill. Then the implied fair value of the goodwill
is compared to the carrying value of the goodwill. If the fair value is lower,
it is considered to be impaired.
As of
September 30, 2009, there was an amount approximately $7.9 million was recorded
as goodwill. Meanwhile, the Group made an impairment charge at full
amount on the goodwill due to the following facts (please read in conjunction
with Note 13):
|
|
a.
|
SiBiono
has a bank loan matured and was in default, which has been sued and the
court has issued a judgment on November 23, 2008 to SiBiono which required
SiBiono to repay all loan balance plus penalty and interest approximately
Rmb24 million immediately. As SiBiono has not paid anything yet, thus the
land use right of SiBiono on its new production plant may be foreclosed
for sales by the lender bank has been frozen unless the amount has been
settled between the lender bank and SiBiono;
|
|
|
b.
|
SiBiono
has serious working capital deficit situation and has reported accumulated
loss of $3.4 million as of September 30, 2009;
|
|
|
c.
|
The
management has not yet located or confirmed any feasible re-financing plan
and / or equity placement as of the report date. The follow up action is
still under the discussion with the lender
bank.
|
As of
September 30, 2009, out of the total acquisition cost Rmb62.56 million (or $9.18
million), the following payments were made:
In the
year of 2006, Benda, through its subsidiaries Everleader and Benda Ebei, paid
Rmb19.52 million and Rmb13.03 million or totaling Rmb32.55 million to the
selling shareholders of SiBiono and reported in the consolidated balance sheet
and cash flow statement as “refundable purchase price paid”. It was recorded as
refundable assets due to the fact that the deal was not concluded as of December
31, 2006. The acquisition was closed on April 5, 2007, and thus the total
refundable amount of Rmb32.55 or $4.17 million was reclassified as investment
cost.
F-21
In the
year of 2007, an additional amount Rmb20.28 million was paid. The remaining
balance was reported as “acquisition cost payable” on the balance sheet. As of
September 30, 2009, the total amount paid was Rmb52.83 million (or $7.76
million) and the outstanding balance was Rmb9.72 million (or $1.42
million).
The Group
has already obtained the oral consent from the selling shareholders of SiBiono
that the remaining balance could be settled within the year of
2009.
11.
Restricted
Cash, Bank Indebtedness and Commercial Notes Payable
The
Group’s restricted cash at September 30, 2009 and December 31, 2008 was
comprised as follows:
|
September
30,
|
December
31,
|
|||||||
|
2009
|
2008
|
|||||||
|
(Unaudited)
|
||||||||
|
Deposits
for issuance of commercial notes
|
$ | 4,544,447 | $ | 4,859,346 | ||||
|
Funds
from government technolgy agencies
|
1,303,447 | 1,303,503 | ||||||
|
Total
restricted cash
|
$ | 5,847,894 | $ | 6,162,849 | ||||
|
A)
|
On
August 14, 2007, Benda Ebei entered into a commercial bank note issuable
agreement with Shanghai Pudong Development Bank. Pursuant to this
agreement, the following terms are
included:
|
|
a)
|
Duration
of the agreement is three years;
|
|
|
b)
|
It is non-interest bearing; | |
|
c)
|
The repayment period of each commercial note payable is six months; | |
|
d)
|
The total commercial note issuable limit is Rmb 60 million; however 50% of deposit should be made into the bank in order to secure the issuance of commercial bank note, thus the net available amount is Rmb 30 million; | |
|
e)
|
If the net amount of each commercial bank note payable is not settled on the due date, the penalty will be the penalty rate of the PRC bank loan on daily and compound basis. | |
|
f)
|
As mentioned in Note 8, this credit facility is guaranteed by SiBiono and secured by the buildings, machinery and equipment of Benda Ebei. On December 15, 2007, Benda Ebei received a consent letter from Pudong Bank that Pudong Bank agreed to cancel SiBiono’s guarantee toward this credit facility. |
On
January 21, 2008, Benda Ebei entered into a supplementary agreement with
Shanghai Pudong Development Bank, to supplement the commercial bank note
issuance agreement dated on August 14, 2007. According to this supplementary
agreement, the credit facility is further secured by the buildings, machinery
and equipment of Jiangling Benda.
As of
September 30, 2009, Benda Ebei and Jiangling Benda deposited an amount $
4,544,447 in Shanghai Pudong Development Bank as deposit for the issuance of
commercial bank notes. Such deposits will be released when the commercial bank
notes are cleared. As of September 30, 2009, the balance of the commercial bank
notes payable was $ 9,075,536. Thus the net
commercial bank notes payable was $4,531,089 as of September 30,
2009.
F-22
|
B)
|
The
bank indebtedness was resulted from the acquisition of SiBiono with the
effective date April 1, 2007. The reasons for causing bank indebtedness
were stated as follows:
|
|
|
a)
|
Among
the cash and cash equivalents balances of SiBiono were composed of two
parts; (i) unrestricted cash, which were generated from either operations,
or loans from bank and financial institutions, or invested capital; (ii)
restricted cash, which were obtained from the various government
technology agencies as long term debt payable (see note 14 for the related
details).
|
|
|
b)
|
The
cash obtained from the various government technology agencies as long term
debt payable could only be dedicated to the related project’s research and
development activities and purchase of fixed assets and construction in
progress, therefore the cash balances for that part will be classified as
restricted cash.
|
|
|
c)
|
Due
to the above reasons, SiBiono relocated the balances of restricted cash
from the cash and cash equivalents balances for the reporting
periods.
|
|
|
d)
|
However,
since the balance of the restricted cash was larger than the balance of
cash and cash equivalents balances, thus bank indebtedness were resulted
for the reporting periods.
|
Due to
the above reasons, SiBiono relocated the balances of restricted cash from the
cash and cash equivalents balances with an amount $1,303,447 as of September 30,
2009. However, since the balances of the restricted cash were larger than the
balance of cash and cash equivalents balances, bank indebtedness were resulted
with an amount $1,199,965 as of September 30, 2009.
12.
Other
Assets
On
November 23, 2006, Benda Ebei entered into an Equity Transfer Agreement with
Xiaozhi Zhang (“Zhang”), to purchase approximately 6.24% of SiBiono’s common
stock for a total consideration of Rmb12.48 million (Rmb6.24 million in cash and
shares of our common stock equal to Rmb6.24 million) (or $1.71 million) which
was due and payable on or before March 31, 2007.
Due to
the fact that the signed agreement on November 23, 2006 was not practically
executable according to the PRC regulations, Benda Ebei asked Zhang to terminate
the signed agreement and sign a new agreement that was feasible under PRC
regulations with essentially the same terms.
However,
Zhang refused to sign the new agreement and applied to the Shenzhen Arbitration
Commission (the “Commission”) in April 2007 for enforcement of the original
agreement. Zhang requested the Commission to require Benda Ebei to pay for the
total consideration, penalty for late payment and the related legal and
arbitration expenses.
On
November 27, 2007, Shenzhen Arbitration Commission determined that:
|
1.
|
Benda
Ebei should pay for the consideration of Rmb6.24 million, equal to 50% of
the total consideration set forth in the Equity Transfer Agreement. For
the other 50% of the total consideration which was supposed to be settled
in the form of issuing common stock, since Zhang did not make an
arbitration request on how to execute the arrangement, the Arbitration
Commission did not make an award on this particular
part.
|
|
2.
|
Benda
Ebei should pay for the penalty of Rmb46,800;
|
|
3.
|
Benda
Eebi should pay for legal and arbitration expenses of
Rmb268,971.
|
F-23
Following
this arbitration decision, Benda Ebei recognized the liability as total
acquisition cost payable of Rmb12.48 million, plus the penalty and related legal
and arbitration expenses, totaling approximately Rmb12.80 million or ($1.87
million) (Note 15). Accordingly Benda Eebi recognized the right to purchase the
6.24% equity shares in SiBiono and recorded as other assets at Rmb12.48 million
or $1.83 million.
On May
22, 2008, Benda Ebei applied to Shenzhen People Court to terminate above
mentioned arbitration. The termination is based on the ground that Xiaozhi Zhang
does not own all 6.24% of SiBiono’s common stock. In fact, he only owns 3.28% of
SiBiono’s stock. The application has been accepted by Shenzhen People Court and
is waiting for its further investigation. Please also see the note descriptions
in Note 29, Subsequent Events.
13.
Bank
Loans Payable
As of
September 30, 2009 and December 31, 2008, SiBiono, had one outstanding bank loan
with an amount of $ 2,984,433 and $2,984,560
respectively, which was used primarily to fund construction in progress projects
and for general working capital purposes. This loan carries annual
interest rate of 6.34% and matures on April 29, 2008. This loan is
personal guaranteed by Zhaohui Peng, the former Chairman and a shareholder of
SiBiono.
For the
nine months ended September 30, 2009, SiBiono has been sued for default on the
bank loans and judgment has been made as follows:
|
a.
|
On
or before November 23, 2008 (10 days after the judgment date of
November 13, 2008), SiBiono needs to repay the bank for the principal of
Rmb20,346,672, accrued interest and penalty up to August 15, 2008 for
Rmb1,506,994 and accrued interest from August 16, 2008 through repayment
date. However, as SiBiono has not yet repaid anything yet, the accrued
interest for the later period will be estimated at Rmb1,176,362, or
total accrued interest as of September 30, 2009 was
Rmb2,683,358;
|
|
b.
|
Should
SiBiono did not make the full repayment; the lender bank can apply for
selling the land use rights owned by SiBiono (as the collateral of the
loan). Any balance after full repayment of the bank loan principal and
accrued interest and penalty from the net proceeds will be returned to
SiBiono;
|
|
c.
|
Zhaohui
Peng should have liability to repay any insufficient amount should the net
proceeds of selling the above land use rights are insufficient to
repay the bank debts and then reclaimed from SiBiono;
|
|
d.
|
SiBiono
and Zhaohui Peng have the obligation to repay the legal fees for this
lawsuit of Rmb156,068 immediately.
|
As of
November 15, 2009, the Group is still under the negotiation with the bank on
arranging the settlement of the bank loan. Furthermore, as of November 15, 2009,
the bank has not taken any further actions to against SiBiono.
Total
interest expense paid related to the Group’s outstanding bank loans was $157,482
and $140,547 for the nine months ended September 30, 2009 and 2008,
respectively.
F-24
14.
Long
Term Debt Payable
As of
September 30, 2009, long term debt payable was raised due to the fact that
various technology funds were obtained from various government technology
agencies to support its gene therapy research and development activities during
the past years and recorded as long term debt payable.
The
obligations will be discharged once the examination by the various government
technology agencies is conducted and most of the examination will be carried out
and completed.
During
the nine months ended September 30, 2009, there was no long term debt payable
discharged and recorded as government subsidies (see Note 22 for the related
details).
15.
Accounts
Payable and Accrued Liabilities
The
Group’s accounts payable and accrued liabilities as of September 30, 2009
and December 31, 2008 were comprised as follows:
|
September
30,
|
December
31,
|
|||||||
|
2009
|
2008
|
|||||||
|
(Unaudited)
|
||||||||
|
Trade
payable
|
2,402,254 | $ | 605,316 | |||||
|
Deposits
paid by customer
|
218,453 | 1,055 | ||||||
|
Acquisition
cost payable following the arbitration (Note 12)
|
1,876,873 | 1,876,953 | ||||||
|
Accrued
liabilities
|
4,908,004 | 3,999,155 | ||||||
|
Miscellaneous
payables
|
351,646 | 745,921 | ||||||
|
Total
account payables and accrued liabilities
|
9,757,230 | $ | 7,228,400 | |||||
16.
Welfare
and Employment Liabilities
As
stipulated by the relevant laws and regulations for enterprises operating in the
PRC, the Group’s PRC entities are required to maintain a welfare plan for all of
its employees who are residents of the PRC. Based on the wages
payable and according to the labor law of the PRC, the Group accrued 14% on a
monthly basis, for employees’ welfare, labor union fees, and education and
training programs, respectively. As of September 30, 2009 and December 31, 2008,
the Group accrued approximately $387,360 and $486,000 for the employees’ welfare
respectively.
17.
Statutory
Surplus Reserve Fund
As
stipulated by the relevant laws and regulations for enterprises operating in the
PRC, the Group is required to make annual appropriations to a statutory surplus
reserve fund for each of its PRC subsidiaries. Specifically, the
Group is required to allocate 15% its profits after taxes at the fiscal year
end, as determined in accordance with the PRC accounting standards applicable to
the Group’s PRC subsidiaries, to a statutory surplus reserve until such reserve
reaches 50% of the registered capital of the Group’s PRC subsidiaries. As of
September 30, 2009 and December 31, 2008, the registered capital of the Group’s
PRC subsidiaries was $22,108,427.
F-25
18.
Related
Party Transactions
Due from
related parties at September 30, 2009 and December 31, 2008 were comprised as
follows:
|
September
30,
|
||||||||||
|
2009
|
December
31,
|
|||||||||
|
Relationship
|
(Unaudited)
|
2008
|
||||||||
|
Current
|
||||||||||
|
Qin
Yu
|
Vice
president
|
|||||||||
|
Due
to Shenzhen SiBiono Gene Tech Co. Ltd.
|
1,736 | $ | 4,314 | |||||||
|
Xiaoji
Zhang
|
Minority
shareholder
|
|||||||||
|
Due
to Shenzhen SiBiono Gene Tech Co. Ltd.
|
5,438 | 5,439 | ||||||||
|
Hsieh,
Chang
|
Supervisor,
VP, HR & Admin.
|
|||||||||
|
Due
to Shenzhen SiBiono Gene Tech Co. Ltd.
|
5 | - | ||||||||
|
Rong
He
|
Manager
|
|||||||||
|
Due
to Shenzhen SiBiono Gene Tech Co. Ltd.
|
455 | 1,247 | ||||||||
| $ | 7,634 | $ | 11,000 | |||||||
|
Non
current
|
||||||||||
|
Yiqing
Wan
|
CEO
& Director
|
|||||||||
|
Due
to Ever Leader Holdings Co. Ltd.
|
$ | 650,760 | $ | 650,791 | ||||||
|
Due
to Hubei Tongji Benda Ebei Phamacetucial Co. Ltd.
|
491,767 | 439,719 | ||||||||
|
Hubei
Benda Science and Technology Co. Ltd
|
Controlled
by CEO
|
|||||||||
|
Due
to Yidu Benda Chemicals Co. Ltd.
|
1,607,105 | 1,607,173 | ||||||||
|
Due
to Ever Leader Holdings Co. Ltd.
|
231,701 | 231,713 | ||||||||
|
Feng
Wang
|
Minority
shareholder
|
|||||||||
|
Due
to Beijing Shusai Pharyngitis Research Co. Ltd.
|
32,345 | 32,348 | ||||||||
|
Hua
Xu
|
||||||||||
|
Due
to Hubei Tongji Benda Ebei Phamacetucial Co. Ltd.
|
49,475 | - | ||||||||
| $ | 3,063,153 | $ | 2,961,744 | |||||||
Due to
related parties at September 30, 2009 and December 31, 2008 were comprised as
follows:
|
September
30,
|
||||||||||
|
2009
|
December
31,
|
|||||||||
|
Current
|
Relationship
|
(Unaudited)
|
2008
|
|||||||
|
Wei
Xu
|
Vice
President, CEO's Spouse & Director
|
|||||||||
|
Due
from Shenzhen SiBiono Gene Tech Co. Ltd.
|
$ | 230,378 | $ | 188,065 | ||||||
|
Due
from Everleader Holding Ltd.
|
967,999 | |||||||||
|
Hua
Xu
|
General
Manager's Sister
|
|||||||||
|
Due
from Shenzhen SiBiono Gene Tech Co. Ltd.
|
21,219 | 25,523 | ||||||||
|
Yiqing,
Wan
|
CEO
& Director
|
|||||||||
|
Due
from Shenzhen SiBiono Gene Tech Co. Ltd.
|
211,426 | 237,780 | ||||||||
|
Hua
Shen
|
Vice
president
|
|||||||||
|
Due
from Shenzhen SiBiono Gene Tech Co. Ltd.
|
- | 33,253 | ||||||||
|
Dongyi
Tien
|
Manager
|
|||||||||
|
Due
from Shenzhen SiBiono Gene Tech Co. Ltd.
|
- | 364 | ||||||||
|
Pong
Tsaiohuei
|
Minority
shareholder
|
|||||||||
|
Due
from Shenzhen SiBiono Gene Tech Co. Ltd.
|
4,819 | 4,818 | ||||||||
|
Total
due to related parties
|
$ | 467,842 | $ | 1,457,803 | ||||||
|
Non
current
|
||||||||||
|
Hubei
Benda Science and Technology Co. Ltd
|
Controlled
by CEO
|
|||||||||
|
Due
from Hubei Tongji Benda Ebei Phamacetucial Co. Ltd.
|
$ | 28,602 | $ | 28,602 | ||||||
|
Due
from Jiangliang Benda Pharamaceutical Co. Ltd.
|
795,923 | 811,073 | ||||||||
|
Due
from Beijing Shusai Pharyngitis Research Co. Ltd.
|
14,148 | 14,262 | ||||||||
|
Wei
Xu
|
||||||||||
|
Due
from Hubei Tongji Benda Ebei Phamacetucial Co. Ltd.
|
Vice
President, CEO's Spouse & Director
|
27,183 | 48,922 | |||||||
|
Due
from Beijing Shusai Pharyngitis Research Co. Ltd.
|
65,508 | 65,691 | ||||||||
|
Yiqing,
Wan
|
CEO
& Director
|
|||||||||
|
Due
from Yidu Benda Chemicals Co. Ltd.
|
560 | 560 | ||||||||
|
Hui
Xu
|
Manager
|
|||||||||
|
Due
from Hubei Tongji Benda Ebei Phamacetucial Co. Ltd.
|
28,483 | 28,483 | ||||||||
| $ | 960,406 | $ | 997,593 | |||||||
Except
for the loans from the shareholder Wei Xu by Everleader which bears interest
rate at 12% per annum, unsecure and matures within six months, the above
advances bear no interest and the above loans due to related parties are
unsecured, non-interest bearing and are not convertible into equity. Proceeds
from the above loans were used primarily for general working capital purposes,
among which the current portion does not have definitive terms and for
those portions which are long-term debts in nature, due on December 31,
2012.
F-26
19.
Redeemable
Common Stock
On April
5, 2007, Benda Ebei entered into Equity Transfer Agreements with certain
shareholders of SiBiono to purchase a total of approximately 57.57% of the
shares of SiBiono’s common stock for total consideration of RMB 60,000,000 due
and payable on or before April 30, 2007.
On June
11, 2007, Benda Ebei entered into additional Equity Transfer Agreements with
Yaojin Wang (“Wang”) and Huimin Zhang “(Zhang”), also shareholders of SiBiono,
for the purchase of an additional 2.56% of the shares of SiBiono’s common stock
for total consideration of RMB 2,560,000 due and payable on or before June 30,
2007.
In
connection with the above Equity Transfer Agreements, Benda entered into a
Financial Consultancy Agreement with Super Pioneer International Limited (“Super
Pioneer”) and Technical Consultancy Agreements with Wang and Zhang for financial
and technical consultancy services to be rendered. Pursuant to the
Financial and Technical Consultancy Agreements (the “Agreements”), Benda agreed
to issue an aggregate of 2,189,560 shares of its common stock to Super Pioneer
(2.1 million shares, out of which 1.96 million shares is redeemable), Wang
(33,585 shares, redeemable) and Zhang (55,975 share, redeemable) within three
months from the date of the Agreements. Since the issuance of common shares to
Super Pioneer, Wang and Zhang was in the form of financial and technical
consultancy services to be rendered, thus the corresponding amount $7,882,416
was recorded as consulting and professional fees.
Super
Pioneer, Wang and Zang also agreed to refrain from selling shares of Benda’s
common stock for a period of twelve months from the date of the issuance of the
shares (the “Lock-up Period”). Within three months from expiration of
the Lock-up Period, in the event that the public trading price of Benda’s common
stock has not reach $3.60 per share and Benda’s common stock has not been listed
on either the NASDAQ or AMEX stock exchanges, Super Pioneer, Wang, and Zang will
have the option to require Benda to redeem an aggregate 2,049,560 shares of
Benda’s common stock owned by Super Pioneer, Wang, and Zhang at a price of $3.60
per share.
In
accordance ASC 480, Distinguishing Liabilities from
Equity,, as the Agreements governing the issuance of the 2,189,560 shares
of common stock to Super Pioneer, Wang, and Zang contain provisions requiring
Benda to repurchase 2,049,560 of these shares at a redemption price of $3.60 per
share at the option of the holders (if certain events outside of control of the
Group do not occur), these 2,049,560 shares have been classified as redeemable
common stock, at their redemption price of $3.60 per share or totaling
$7,376,366, outside of permanent equity at December 31, 2009.
According
to the signed agreements mentioned in above, the redemption would be required to
perform in September 2008. However the company has already obtained an oral
consent from Super Pioneer, Wang and Zhang that the redemption would be deferred
to the year of 2009. Therefore, the redeemable common stock was reclassified as
current liability as of September 30, 2009.
F-27
20.
Other
Income (expenses)
In
accordance with ASC 825, Financial Instruments,, the
Group recorded $1,113,405 in Registration Delay Expense, for the nine month
ended September 30, 2008. Out of the total penalty, $230,312 was settled by
issuance of $523,438 shares of common stock. There was no penalty to investors
incurred for the nine months ended September 30, 2009.
21.
Other
General and Administrative Expenses
For the
nine months and three months ended September 30, 2009 and 2008, the amount of
other general and administrative expenses mainly composed of the following
events:
|
NINE
MONTHS ENDED SEPTEMBER 30,
|
THREE MONTHS
ENDED SEPTEMBER 30,
|
|||||||||||||||
|
2009
|
2008
|
2009
|
2008
|
|||||||||||||
|
(Unaudited)
|
(Unaudited)
|
|||||||||||||||
|
Audit
and accounting
|
$ | 102,013 | $ | 232,509 | $ | 32,330 | $ | 30,680 | ||||||||
|
Legal
fees
|
194,084 | 166,800 | 56,860 | 52,096 | ||||||||||||
|
Office
expenses
|
174,122 | 451,979 | 109,735 | 159,449 | ||||||||||||
|
Salaries
and wages
|
646,570 | 712,810 | 207,431 | 197,817 | ||||||||||||
|
Consulting
fee
|
320,421 | 637,409 | 245,619 | 202,636 | ||||||||||||
|
Rent
& Utilities
|
38,036 | 75,848 | 35,343 | 20,018 | ||||||||||||
|
Investor
relation, Transfer agent and filing fees
|
2,720 | 30,843 | - | 2,720 | ||||||||||||
|
Travel
and transportation
|
149,228 | 185,379 | 102,071 | 25,045 | ||||||||||||
|
Repair
and maintanence
|
21,131 | 136,890 | 9,774 | 17,791 | ||||||||||||
|
Miscellaneous
|
237,669 | 133,789 | 98,901 | 35,730 | ||||||||||||
|
Total
other general & administrative
|
$ | 1,885,994 | $ | 2,764,256 | $ | 898,062 | $ | 743,982 | ||||||||
22. Government Subsidies and Grants
As
mentioned in the Note 14, long term debt payable, various technology funds were
obtained from various government technology agencies to support its gene therapy
research and development activities during the past years and recorded as long
term debt payable. According to the technology fund agreements, the
various government technology agencies will examine the results of research and
development according to the status of the projects. Once the examination takes
place, the obligation of a particular debt payable is discharged
accordingly.
According
to US GAAP, once the obligation of a particular debt payable is discharged, the
amount of this particular debt payable should be treated as government subsidies
/ grants. During the nine months ended September 30, 2009, there were
no long term debt payable discharged and government subsidies / grants
recognized as revenue
Besides,
during the nine months ended September 30, 2009, there was Rmb180,000 or $26,386
received by Jiangling Benda from the local financial bureau as subsidy for
support of the enterprise with small and medium size.
23.
Income
Taxes
Benda is
subject to Delaware, United State of America tax, but no provision for income
taxes were made for the nine months ended September 30, 2009 and 2008 as Benda
did not have reportable taxable income for the period.
F-28
Ever
Leader, a wholly owned subsidiary of Benda, is subject to Hong Kong tax, but no
provisions for income taxes were made for the nine months ended September 30,
2009 and 2008 as Ever Leader did not have reportable taxable income for the
periods.
Benda
Ebei was registered as a Sino-Foreign Equity Joint Venture on May 26, 2004 and
is subject to the tax laws applicable to Sino-Foreign Equity Joint Ventures in
the PRC. Benda Ebei, starting from 2005, is fully exempt from PRC
enterprise income tax for two years starting from the first profit-making year,
followed by a 50% redsuction in income taxes, for the following three
years.
Jiangling
Benda and Yidu Benda are cross-municipal investment entities and enjoy the same
tax treatment as Sino-Foreign Joint Ventures, starting from 2005, and were
therefore exempt from PRC enterprise income tax for two years starting from the
first profit-making year, followed by a 50% reduction in income taxes, for the
following three years. Cross-municipal investments entities refer to entities
that are incorporated in one municipal region but have investments in another
municipal region.
The
exemption periods for Benda Ebei, Jiangling Benda and Yidu Benda expired in the
year of 2006, after which they are subject to 50% reduction in income taxes;
whereas the full income tax rate is 33%. The remaining tax holidays will be
expired in 2009.
However,
starting and effective from January 1, 2008, the full income tax rate would be
changed from 33% to 25% according to the new PRC taxation regulations. Therefore
these subsidiaries will be subject to the regular full income tax rate at 25%
after the tax holidays expire.
According
to the new taxation regulations starting and effective from January 1, 2008,
Beijing Shuhai is subject to the full income tax rate of 25%.
According
to the new taxation regulations starting and effective from January 1, 2008,
SiBiono, which is located in Shenzhen, a Special Economic District of PRC, is
subject to the full income tax rate of 25% gradually in five years as
following:
|
Year
|
Tax
rate
|
|||
|
2008
|
18 | % | ||
|
2009
|
20 | % | ||
|
2010
|
22 | % | ||
|
2011
|
24 | % | ||
|
2012
and thereafter
|
25 | % | ||
Benda
Ebei recorded $163,803 income tax for the nine months ended September 30, 2009.
The prepaid income tax by the Group as of September 30, 2009 at amount of $1.66
million was recorded as taxes recoverable.
F-29
The
provision for taxes on earnings consisted of:
|
NINE
MONTHS ENDED SEPTEMBER 30,
|
THREE
MONTHS ENDED SEPTEMBER 30,
|
|||||||||||||||
|
2009
|
2008
|
2009
|
2008
|
|||||||||||||
|
(Unaudited)
|
(Unaudited)
|
|||||||||||||||
|
Crurrent income
taxes expenses:
|
||||||||||||||||
|
PRC
Enterprise Income
|
$ | 163,803 | $ | 907,176 | $ | 79,391 | $ | 382,368 | ||||||||
|
United
States Federal Income Tax
|
- | - | - | - | ||||||||||||
| $ | 163,803 | $ | 907,176 | $ | 79,391 | $ | 382,368 | |||||||||
A
reconciliation between the income tax computed at the U.S statutory rate and the
Groups provision for Income tax for the nine month ended September 30, 2009 and
2008 are as follows:
|
NINE
MONTHS ENDED SEPTEMBER 30,
|
THREE
MONTHS ENDED SEPTEMBER 30,
|
|||||||||||||||
|
2009
|
2008
|
2009
|
2008
|
|||||||||||||
|
U.S
Statutory rate
|
34.0 | % | 34.0 | % | 34.0 | % | 34.0 | % | ||||||||
|
Foreign
income not recognized in the U.S
|
-34.0 | % | -34.0 | % | -34.0 | % | -34.0 | % | ||||||||
|
PRC
statuory rate
|
25.0 | % | 25.0 | % | 25.0 | % | 25.0 | % | ||||||||
|
Tax
relief and holidy granted to the Subsidary
|
0.0 | % | -12.5 | % | 0.0 | % | -12.5 | % | ||||||||
|
Provision
for income tax
|
25.0 | % | 12.5 | % | 25.0 | % | 12.5 | % | ||||||||
24.
Long
Term Convertible Promissory Notes
Our
financial statements include enhanced note disclosure addressing the fair value
of the convertible promissory notes and warrants and the appropriate accounting
treatment for the overall transaction.
Additionally,
the enhanced note disclosure includes a detailed discussion of the accounting
for the debt discount resulting from the relative fair value of the warrants
issued in conjunction with the convertible promissory notes (calculated in
accordance with ASC 470, Debt) and the debt discount resulting from the
beneficial conversion feature / conversion discount associated with the
convertible promissory notes (calculated in accordance with ASC 470) as
follows:
In March
and April of 2007, the Group entered into Investment Agreements (“Agreements”)
with certain accredited and institutional investors (“Investors”) pursuant to
which the Investors purchased from the Group a total of 252 Units (“Units”),
resulting in aggregate gross proceeds to the Group $7,560,000, with each Unit
consisting of: (i) a Convertible Promissory Note (“Note”) in the principal
amount of $30,000 and convertible into 54,087 shares of the Group's common
stock; and (ii) a Common Stock Warrant (“Warrant”) to acquire 54,087 shares
of the Group’s common stock at an exercise price of $0.555 per share and
expiring on November 14, 2011. The Notes are immediately convertible at the
option of each Investor, bear interest at a rate of 4% per annum and mature on
March 28, 2009.
The Group
has accounted for the Warrants issued in conjunction with the Notes in
accordance with the provisions of ASC 470, Debt. Accordingly, the
Warrants were valued using a Black-Scholes option pricing model with the
following assumptions: (i) a risk free interest rate of 4.63%, (ii) a
contractual life of 4.6 years, (iii) an expected volatility of 25%, and (iv) a
dividend yield of zero. The relative fair value of the Warrants, based on an
allocation of the value of the Notes and the value of the Warrants issued in
conjunction with the Notes, was recorded as a debt discount (with a
corresponding increase to additional paid-in capital) in the amount of
$5,174,911, and is being amortized to interest expense over the expected term of
the Notes.
F-30
Additionally,
the difference between the effective conversion price of the Notes into shares
of the Group’s common stock, and the fair value of the Group’s common stock on
the date of issuance of the Notes, resulted in a beneficial conversion feature
of $2,385,089 (capped at the $7,560,000 of gross proceeds raised less the
previously calculated $5,174,911 debt discount associated with Warrants issued
in conjunction with the Notes) and was calculated in accordance with ASC 470.
This beneficial conversion feature was recorded as an additional debt discount
(with a corresponding increase to additional paid-in capital) and is being
amortized to interest expense over the expected term of the Notes.
On July
10, 2008, 10 units of were converted to shares of 540,870. Therefore, as of
September 30, 2009, the aggregate $7,260,000 principal balance of the notes
remained outstanding and for the nine months ended September 30, 2009, the Group
recorded $ 816,750 of interest expense related to the Notes.
During
the nine months ended September 30, 2009, the Group recorded $864,049 in
interest expense related to the amortization of the debt discount associated
with the Warrants and the debt discount associated with the beneficial
conversion feature.
The Group
also incurred $529,200 in placement agent commissions related to the issuance of
the Notes and Warrants. This amount was recorded by the Group as debt issue
costs and is being amortized over expected term of the Notes. For the
nine months ended September 30, 2009, the Group recorded $55,485 in amortization
expense related to debt issue costs.
The
securities underlying the Notes and Warrants issued to the Investors are also
subject to the terms of a Make Good Agreement entered into in connection with a
financing the Group executed in November of 2006 (the “Make Good Agreement”).
The Group further represented to the Investors that its target net income for
fiscal year 2007 (“FY07 Net Income”) will be greater than or equal to $10.0
million (adjusted for a variety of non-cash charges) (the “Performance
Threshold”). In the event the Performance Threshold is not attained,
the Group is required to issue to the Investors a pro rata portion of 1,000,000
shares of the Group’s common stock for every one (1) cent by which the Group’s
earnings per share, determined on a fully diluted basis, is less than $0.065.
During the year ended December 31, 2008 the Group issued shares 3,810,976 shares
of common stock in lieu of the non-achievement of the Performance Threshold at
$0.07 per share, totaling to $266,768 and none for the nine months ended
September 30, 2009.
In the
accounting for the Long Term Convertible Promissory Note, the Group’s analysis
of the Performance Threshold had no effect because of the cap of the debt
discounted and beneficial conversion feature as calculated in accordance with
ASC 470.
The Group
is also required to register for resale: (i) the shares of common stock
underlying the Notes; and (ii) 150% of the shares of common stock underlying the
Warrants, on a registration statement to be filed with the Securities and
Exchange Commission (“Registration Statement”) and such Registration Statement
is required to be declared effective by August 15, 2007 (the “Effectiveness
Deadline”). If the Registration Statement does not become effective by the
Effectiveness Deadline, or if the Group fails to maintain the effectiveness of
the Registration Statement, for any reason, the Group is required to pay the
Investors in cash an amount equal to 1% of the purchase price of each Unit held
by the Investors on such Effectiveness Deadline or the first day of such failure
to maintain the Registration Period, as applicable, and for every 30 day period
(or part) thereafter, in each case until cured (“Registration Delay Payments”),
provided that the Registration Delay Payments will not exceed 10% of the
purchase price of the Units. In the event that the Registration Delay Payments
are not made in a timely manner, such Registration Delay Payments will bear
interest at a rate of 1.5% per month until paid in full.
F-31
The
calculated fair value of the warrants (issued in conjunction with the
convertible promissory notes) was recorded as a debt discount (reduction to the
carrying value of the convertible promissory notes) with a corresponding
increase (credit) to additional paid in capital.
In
accordance with the registration rights granted to investors who purchased the
convertible promissory notes and warrants, we are required to file a
registration statement with the Securities and Exchange Commission
(“Registration Statement”) attempting to register the potentially issuable
shares of common stock underlying the convertible promissory
notes and warrants. There is no cash settlement requirement, in the
event the Company cannot deliver Registered Shares.
Specifically,
in the event the Registration Statement is not declared effective by August 15,
2007 (“Effectiveness Deadline”), we are required to pay to the investors in cash
an amount equal to 1% of the purchase price paid by each investor for the
convertible promissory notes and warrants (“Registration Delay
Payments”). Additionally, for every 30 day period (or part)
thereafter that the Registration Statement is not declared effective, we are
required to continue to make Registration Delay Payments, provided that such
Registration Delay Payments shall not exceed 10% of the purchase price paid by
each investor for the convertible promissory notes and warrants.
In the
event the Registration Statement is never declared effective, the Registration
Delay Payments are capped at 10% (of the purchase price paid by each investor
for the convertible promissory notes and warrants). As a result, the
initial requirement for the Company to deliver registered shares upon conversion
of the convertible promissory notes or exercise of the warrants is negated and
paragraphs 14 – 18 of EITF 00-19 are not applicable (for purposes of classifying
the fair value of the warrants issued in conjunction with the convertible
promissory notes as a liability with changes in fair value recorded in
earnings).
25.
Common
Stock, Preferred Stock, Additional Paid-in Capital, Warrants and
Options
As of
September 30, 2009, and December 31, 2008, the Group had an aggregate
of:
|
(a)
|
None
shares of preferred stock issued and
outstanding;
|
|
(b)
|
The
share of common stock issued and outstanding was 105,155,355 as of
September 30, 2009 and December 31, 2008
respectively.
|
F-32
|
(c)
|
The
following tables shows the events occurred in additional paid-in
capital:
|
|
Events
|
||||||||||||
|
occurred
|
||||||||||||
|
during
the
|
||||||||||||
|
reporting
|
Septebmer
30,
|
|||||||||||
|
December
31,
|
period
|
2009
|
||||||||||
|
Events
|
2008
|
(Unaudited)
|
(Unaudited)
|
|||||||||
|
To
record the changes of par value from $0.01 to $0.001
|
||||||||||||
|
of
the outstanding common stock as of 11/17/2005
|
$ | 584,481 | $ | - | $ | 584,481 | ||||||
|
To
adjust the par value of outstanding common stock as of
3/31/2006
|
(5,354 | ) | - | (5,354 | ) | |||||||
|
Waiver
of shareholder loan on 9/5/2006
|
2,298,434 | - | 2,298,434 | |||||||||
|
To
eliminate the common stock and additional paid-in capital of the former
shell
|
||||||||||||
|
company
"Applied Spectrum Technologies, Inc." on 11/15/2006
|
16,215,770 | - | 16,215,770 | |||||||||
|
To
eliminate the accumulated deficit of the former shell
company
|
||||||||||||
|
"Applied
Spectrum Technologies, Inc." on 11/15/2006
|
(16,209,962 | ) | - | (16,209,962 | ) | |||||||
|
Issuance
of common stock, 25,961,760 shares at $0.4622 per share, on
11/15/2006
|
11,974,038 | - | 11,974,038 | |||||||||
|
Issuance
of common stock, 64,942,369 shares at $0.001 per share on
11/15/2006
|
(64,942 | ) | - | (64,942 | ) | |||||||
|
To
relocate the original common stock of Ever Leader on
11/15/2006
|
1,285 | - | 1,285 | |||||||||
|
To
record the placement agent commission and transaction related
fee
|
||||||||||||
|
of
reverse merger on 11/15/2006
|
(1,694,326 | ) | - | (1,694,326 | ) | |||||||
|
Issuance
of common stock 706,195 at $0.4622 per share
|
325,697 | - | 325,697 | |||||||||
|
Debt
discount on beneficial conversion feature on convertible promissory
notes
|
2,385,089 | - | 2,385,089 | |||||||||
|
Debt
discount on warrants issued with convertible promissory
notes
|
5,174,911 | - | 5,174,911 | |||||||||
|
Placement
agent exercised 849,007 warrants at strike price through cashless
arrangement
|
470,347 | - | 470,347 | |||||||||
|
Issuance
of common stock to PIPE investors as penalty for late submission of
10KSB
|
92,461 | - | 92,461 | |||||||||
|
Issuance
of common stock to directors in lieu of their remuneration
|
75,790 | - | 75,790 | |||||||||
|
Issuance
of common stock to PIPE investors as penalty for registeration delay
expenses
|
229,789 | - | 229,789 | |||||||||
|
Conversion
of convertible promissory notes
|
(8,039 | ) | - | (8,039 | ) | |||||||
|
Issuance
of common stock to investors under "Make Good Agreement"
|
262,957 | - | 262,957 | |||||||||
|
Additional
paid-in capital, balance for the period ended
|
$ | 22,108,427 | $ | - | $ | 22,108,427 | ||||||
|
(d)
|
As
of September 30, 2009 and December 31, 2008, 30,264,249 Warrants, each
convertible into one (1) share of the Group’s Common Stock, issued and
outstanding.
|
|
(e)
|
None
options issued and outstanding.
|
26.
Weighted
Average Shares Outstanding
Basic net
income (loss) per share is computed by dividing net income (loss) by the
weighted-average number of shares outstanding during the period.
Diluted
net income (loss) per share is computed by using the weighted-average number of
shares of common stock outstanding and, when dilutive, potential shares from
stock options and warrants to purchase common stock, using the treasury stock
method.
F-33
The
following table illustrates the computation of basic and dilutive net income
(loss) per share and provides a reconciliation of the number of weighted-average
basic and diluted shares outstanding:
|
NINE
MONTHS ENDED SEPTEMBER 30,
|
THREE MONTHS
ENDED SEPTEMBER 30,
|
|||||||||||||||
|
2008
|
||||||||||||||||
|
2009
|
2008
|
2009
|
Restated
|
|||||||||||||
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
|||||||||||||
|
Numerator:
|
||||||||||||||||
|
Net
income (loss)
|
$ | (2,688,652 | ) | $ | (4,664,501 | ) | $ | (372,397 | ) | $ | (841,923 | ) | ||||
|
Denominator:
|
||||||||||||||||
|
Basic
weighted-average shares outstanding
|
105,155,355 | 100,887,853 | 105,155,355 | 101,556,376 | ||||||||||||
|
Effect
of dilutive stock options & warrants
|
- | - | - | - | ||||||||||||
|
Diluted
weighted-average shares outstanding
|
105,155,355 | 100,887,853 | 105,155,355 | 100,803,509 | ||||||||||||
|
Net
income (loss) per share:
|
||||||||||||||||
|
Basic
|
$ | (0.03 | ) | $ | (0.05 | ) | $ | (0.00 | ) | $ | (0.01 | ) | ||||
|
Diluted
|
$ | (0.03 | ) | $ | (0.05 | ) | $ | (0.00 | ) | $ | (0.01 | ) | ||||
27.
Commitments
and Contingencies
As of
September 30, 2009, there was an operating lease commitment and the amounts are
stated as follows;
|
September
30
|
||||
|
2009
|
||||
|
(Unaudited)
|
||||
|
Rental
and Property Management Fee
|
||||
|
Within
one year
|
$ | 50,764 | ||
|
One
to two year
|
35,203 | |||
|
Two
to three year
|
35,203 | |||
|
Over
three years
|
52,805 | |||
|
Total
commitments payable
|
$ | 173,975 | ||
28.
Segment
Information
The Group
states the segment information according to the requirement stated in ASC 280,
Segment Reporting. The Group produces
five different categories of products and each category of product is produced
in different subsidiaries or operation plants. The details are stated as
follows:
|
1.
|
Benda
Ebei produces conventional medicines which including banded and generic
medicines;
|
|
2.
|
Jiangling
Benda produces active pharmaceutical ingredients,
APIs;
|
|
3.
|
Yidu
Benda produces bulk
chemicals;
|
|
4.
|
Beijing
Shusai produces pharyngitis killer therapy;
and
|
|
5.
|
SiBiono
produces gene therapy medicines,
Gendicine.
|
Since
each subsidiary produces the corresponding products by using the same production
facilities of each subsidiary, therefore according to the requirement stated in
ASC 280, the Group reports the segment information according to the un-identical
products that produced in each subsidiary.
F-34
Selected
financial information for each of these segments for the nine months and three
months ended September 30, 2009 and 2008 were as follows:
|
NINE
MONTHS ENDED SEPTEMBER 30,
|
THREE
MONTHS ENDED SEPTEMBER 30,
|
|||||||||||||||
|
(Unaudited)
|
(Unaudited)
|
|||||||||||||||
|
Branded/Generic
medicine segment
|
2009
|
2008
|
2009
|
2008
|
||||||||||||
|
Revenue
from external customers
|
$ | 13,617,252 | 17,439,205 | $ | 4,887,566 | $ | 5,973,012 | |||||||||
|
Cost
of sales
|
(8,600,569 | ) | (11,479,479 | ) | (3,051,518 | ) | (3,831,922 | ) | ||||||||
|
Gross
profit
|
5,016,683 | 5,959,726 | 1,836,047 | 2,141,090 | ||||||||||||
|
Gross
margin
|
37 | % | 34 | % | 38 | % | 36 | % | ||||||||
|
Research
and development
|
- | - | 2,301 | - | ||||||||||||
|
Selling
expense
|
(942,604 | ) | (1,093,119 | ) | (529,292 | ) | (319,636 | ) | ||||||||
|
General
and administrative expense
|
(2,848,745 | ) | (516,513 | ) | (762,307 | ) | (29,908 | ) | ||||||||
|
Segment
contribution
|
$ | 1,225,334 | 4,350,094 | $ | 546,750 | $ | 1,791,546 | |||||||||
|
Contribution
margin
|
9 | % | 25 | % | 11 | % | 30 | % | ||||||||
|
Total
assets, segment
|
$ | 30,252,879 | 39,164,262 | $ | 39,164,262 | $ | 38,243,467 | |||||||||
|
Active
pharmaceutical ingredients segment
|
2009
|
2008
|
2009
|
2008
|
||||||||||||
|
Revenue
from external customers
|
$ | 817,137 | 609,139 | $ | 27,075 | $ | 28,807 | |||||||||
|
Cost
of sales
|
(901,431 | ) | (669,313 | ) | 66,164 | (30,784 | ) | |||||||||
|
Gross
profit
|
(84,294 | ) | (60,174 | ) | 93,238 | (1,977 | ) | |||||||||
|
Gross
margin
|
-10 | % | -10 | % | 344 | % | -7 | % | ||||||||
|
Research
and development
|
(143 | ) | - | (70 | ) | - | ||||||||||
|
Selling
expense
|
(16,576 | ) | (3,716 | ) | (1,851 | ) | -3,359 | |||||||||
|
General
and administrative expense
|
(526,094 | ) | (1,154,333 | ) | (205,085 | ) | (127,767 | ) | ||||||||
|
Segment
contribution
|
$ | (627,107 | ) | (1,218,223 | ) | $ | (113,768 | ) | $ | (133,103 | ) | |||||
|
Contribution
margin
|
-77 | % | -200 | % | -420 | % | -462 | % | ||||||||
|
Total
assets, segment
|
$ | 9,654,558 | 8,545,875 | $ | 8,545,875 | $ | 8,436,445 | |||||||||
|
Bulk
chemicals segment
|
2009
|
2008
|
2009
|
2008
|
||||||||||||
|
Revenue
from external customers
|
$ | - | - | $ | - | $ | - | |||||||||
|
Cost
of sales
|
- | - | - | - | ||||||||||||
|
Gross
profit
|
- | - | - | - | ||||||||||||
|
Gross
margin
|
- | - | - | - | ||||||||||||
|
Research
and development
|
- | - | - | - | ||||||||||||
|
Selling
expense
|
- | - | - | - | ||||||||||||
|
General
and administrative expense
|
(469,567 | ) | (438,355 | ) | (249,393 | ) | (132,387 | ) | ||||||||
|
Segment
contribution
|
$ | (469,567 | ) | (438,355 | ) | $ | (249,393 | ) | $ | (132,387 | ) | |||||
|
Contribution
margin
|
- | - | - | - | ||||||||||||
|
Total
assets, segment
|
$ | 6,990,800 | 7,553,516 | $ | 7,553,516 | $ | 7,694,510 | |||||||||
|
Pharynigitis
killer therapy segment
|
2009
|
2008
|
2009
|
2008
|
||||||||||||
|
Revenue
from external customers
|
$ | - | 3,106 | $ | - | $ | 978 | |||||||||
|
Cost
of sales
|
- | (3,397 | ) | - | (3,280 | ) | ||||||||||
|
Gross
profit
|
- | (291 | ) | - | (2,302 | ) | ||||||||||
|
Gross
margin
|
- | (0 | ) | - | -235 | % | ||||||||||
|
Research
and development
|
- | - | - | - | ||||||||||||
|
Selling
expense
|
(733 | ) | (4,346 | ) | (733 | ) | (1,692 | ) | ||||||||
|
General
and administrative expense
|
(19,609 | ) | (24,548 | ) | (5,134 | ) | (4,321 | ) | ||||||||
|
Segment
contribution
|
$ | (20,342 | ) | (29,185 | ) | $ | (5,867 | ) | $ | (8,315 | ) | |||||
|
Contribution
margin
|
- | (9 | ) | - | -850 | % | ||||||||||
|
Total
assets, segment
|
$ | 76,653 | 99,744 | $ | 99,744 | $ | 108,332 | |||||||||
|
Gendicine
(Ad-p53) segment
|
2009
|
2008
|
2009
|
2008
|
||||||||||||
|
Revenue
from external customers
|
$ | 2,910,676 | 1,808,607 | $ | 990,013 | $ | 631,637 | |||||||||
|
Cost
of sales
|
(268,370 | ) | (458,205 | ) | (85,937 | ) | (347,554 | ) | ||||||||
|
Gross
profit
|
2,642,306 | 1,350,402 | 904,076 | 284,083 | ||||||||||||
|
Gross
margin
|
91 | % | 75 | % | 91 | % | 45 | % | ||||||||
|
Research
and development
|
(283,330 | ) | (249,357 | ) | (167,621 | ) | (79,576 | ) | ||||||||
|
Selling
expense
|
(908,569 | ) | (1,021,508 | ) | (320,240 | ) | (366,790 | ) | ||||||||
|
General
and administrative expense
|
(1,582,201 | ) | (518,651 | ) | (216,189 | ) | (33,311 | ) | ||||||||
|
Segment
contribution
|
$ | (131,794 | ) | (439,114 | ) | $ | 200,026 | $ | (195,594 | ) | ||||||
|
Contribution
margin
|
-5 | % | -24 | % | 20 | % | -31 | % | ||||||||
|
Total
assets, segment
|
$ | 12,169,195 | 13,808,709 | $ | 13,808,709 | $ | 14,048,551 | |||||||||
|
NINE
MONTHS ENDED SEPTEMBER 30,
|
THREE
MONTHS ENDED SEPTEMBER 30,
|
|||||||||||||||
|
2009
|
2009
|
2008
|
||||||||||||||
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
||||||||||||||
|
Total
revenue from external customers
|
$ | 17,345,065 | 19,860,057 | $ | 5,904,653 | $ | 6,634,434 | |||||||||
|
Cost
of sales
|
(9,770,370 | ) | (12,610,394 | ) | (3,071,291 | ) | (4,213,540 | ) | ||||||||
|
Gross
profit
|
7,574,695 | 7,249,663 | 2,833,362 | 2,420,894 | ||||||||||||
|
Gross
margin
|
44 | % | 37 | % | 48 | % | 36 | % | ||||||||
|
Research
and development
|
(283,473 | ) | (249,357 | ) | (165,390 | ) | (79,576 | ) | ||||||||
|
Selling
expense
|
(1,868,482 | ) | (2,122,689 | ) | (852,116 | ) | (691,477 | ) | ||||||||
|
General
and administrative expense
|
(5,446,216 | ) | (2,652,400 | ) | (1,438,108 | ) | (327,694 | ) | ||||||||
|
Segment
contribution
|
$ | (23,476 | ) | 2,225,217 | $ | 377,748 | $ | 1,322,147 | ||||||||
|
Contribution
margin
|
0 | % | 11 | % | 6 | % | 20 | % | ||||||||
|
Total
assets, segment
|
$ | 59,144,085 | 69,172,106 | $ | 69,172,106 | $ | 68,531,305 | |||||||||
F-35
The
results of the total consolidated net profit before income taxes for the
reporting periods are as follows:
|
NINE
MONTHS ENDED SEPTEMBER 30,
|
THREE
MONTHS ENDED SEPTEMBER 30,
|
|||||||||||||||
|
2009
|
2008
|
2009
|
2008
|
|||||||||||||
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
|||||||||||||
|
Total
segment contribution
|
$ | (23,476 | ) | 2,225,217 | $ | 377,748 | $ | 1,322,147 | ||||||||
|
Unallocated
amounts:
|
||||||||||||||||
|
Interest
income/(expenses)
|
(2,104,510 | ) | (471,263 | ) | - | |||||||||||
|
Government
subsidy
|
26,386 | - | (237,918 | ) | ||||||||||||
|
Other
income/(expenses)
|
106,161 | 10,140 | 60,388 | - | ||||||||||||
|
Other
corporate expenses
|
(666,465 | ) | (6,249,168 | ) | (194,360 | ) | (1,712,054 | ) | ||||||||
|
Total
income / (loss) before noncontrolling interest and income
taxes
|
$ | (2,661,904 | ) | (4,013,811 | ) | $ | (227,487 | ) | $ | (627,825 | ) | |||||
The other
corporate expenses for the nine months and three months ended September 30, 2009
and 2008 composed of the following events:
|
NINE
MONTHS ENDED SEPTEMBER 30,
|
THREE
MONTHS ENDED SEPTEMBER 30,
|
|||||||||||||||
|
2009
|
2009
|
2008
|
||||||||||||||
|
(Unaudited)
|
(Unaudited)
|
(Unaudited)
|
||||||||||||||
|
Penalty
to investor
|
$ | - | 1,380,173 | $ | - | $ | 266,768 | |||||||||
|
Wages
and salaries
|
283,817 | 295,421 | 88,801 | 102,746 | ||||||||||||
|
Audit
and accounting
|
74,995 | 130,735 | 24,998 | 24,672 | ||||||||||||
|
Amortization
of debt issue cost
|
55,485 | 198,360 | - | 66,603 | ||||||||||||
|
Consulting
fee
|
9,336 | 560,040 | 3,120 | 188,080 | ||||||||||||
|
Investor
relation, transfer agent and filing fees
|
2,720 | 30,843 | - | 2,720 | ||||||||||||
|
Director
renumeration
|
67,500 | 162,968 | 22,500 | 22,500 | ||||||||||||
|
Legal
fee
|
158,134 | 107,315 | 53,567 | 30,640 | ||||||||||||
|
Travel
and transportation
|
2,446 | 4,844 | 303 | - | ||||||||||||
|
Office
expense
|
- | 1,120 | - | - | ||||||||||||
|
Interest
expense
|
- | 3,339,457 | - | 998,877 | ||||||||||||
|
Miscellaneous
|
12,029 | 37,892 | 1,070 | 8,448 | ||||||||||||
|
Total
|
$ | 666,465 | 6,249,168 | $ | 194,360 | $ | 1,712,054 | |||||||||
For the
details of information of this particular, it should be read in conjunction with
the management discussion and analysis section.
The
following table shows the reconciliation between the segments assets and the
total assets as of September 30, 2009 and 2008:
F-36
|
September
30,
|
September
30,
|
|||||||
|
2009
|
2008
|
|||||||
|
(Unaudited)
|
(Unaudited)
|
|||||||
|
Total
assets, segment
|
$ | 59,144,085 | 69,172,106 | |||||
|
Total
assets of corporate:
|
||||||||
|
Cash
and cash equivalent
|
12,168 | 39,615 | ||||||
|
Bank
indebtedness
|
1,199,965 | 1,259,525 | ||||||
|
Prepaid
expesnes and deposit
|
- | 5,672 | ||||||
|
Refundable
purchase price paid
|
1,200,000 | 1,200,000 | ||||||
|
Due
from related parties
|
882,461 | 670,541 | ||||||
|
Debit
issue costs:
|
||||||||
|
placement
agent commission
|
- | 129,585 | ||||||
|
Total
assets
|
$ | 62,438,679 | 72,477,044 | |||||
The
following table shows how the noncontrolling interest for nine months ended
September 30, 2009 and 2008 was derived:
|
Nine
Months Ended September 30, 2009 (Unaudited)
|
||||||||||||||||||||||||
|
Benda
|
Jiangling
|
Yidu
|
Beijing
|
|||||||||||||||||||||
|
Ebei
|
Benda
|
Benda
|
Shusai
|
SiBiono
|
Total
|
|||||||||||||||||||
|
Segment
operating profit / (loss)
|
$ | 1,225,334 | (627,107 | ) | (469,567 | ) | (20,342 | ) | (131,794 | ) | $ | (23,476 | ) | |||||||||||
|
Interest
income/ (expenses)
|
(186,293 | ) | (253 | ) | 4 | - | (150,390 | ) | (336,932 | ) | ||||||||||||||
|
Other
income / (expenses)
|
(15,329 | ) | (175 | ) | 2,968 | - | 118,697 | 106,161 | ||||||||||||||||
|
Government
subsidy
|
- | 26,386 | - | - | - | 26,386 | ||||||||||||||||||
|
Income
taxes
|
(163,803 | ) | - | - | - | - | (163,803 | ) | ||||||||||||||||
|
Income
/ (loss) before minority interest
|
$ | 859,909 | (601,149 | ) | (466,595 | ) | (20,342 | ) | (163,487 | ) | $ | (391,664 | ) | |||||||||||
|
Noncontrolling
interest percentage
|
5.00 | % | 9.75 | % | 9.75 | % | 28.75 | % | 42.88 | % | ||||||||||||||
|
Noncontrolling
interest
|
$ | 42,995 | (58,612 | ) | (45,493 | ) | (5,848 | ) | (70,097 | ) | $ | (137,055 | ) | |||||||||||
|
Nine
Months Ended September 30, 2008 (Unaudited)
|
||||||||||||||||||||||||
|
Benda
|
Jiangling
|
Yidu
|
Beijing
|
|||||||||||||||||||||
|
Ebei
|
Benda
|
Benda
|
Shusai
|
SiBiono
|
Total
|
|||||||||||||||||||
|
Segment
operating profit / (loss)
|
$ | 4,350,094 | (1,218,223 | ) | (438,355 | ) | (29,185 | ) | (439,114 | ) | $ | 2,225,217 | ||||||||||||
|
Interest
income/ (expenses)
|
(339,014 | ) | 1,010 | 6 | (8 | ) | (142,689 | ) | (480,695 | ) | ||||||||||||||
|
Other
income / (expenses)
|
(13,874 | ) | 228 | 2,953 | 6,820 | 14,013 | 10,140 | |||||||||||||||||
|
Income
taxes
|
(907,176 | ) | - | - | - | - | (907,176 | ) | ||||||||||||||||
|
Income
/ (loss) before minority interest
|
$ | 3,090,030 | (1,216,985 | ) | (435,396 | ) | (22,373 | ) | (567,790 | ) | $ | 847,486 | ||||||||||||
|
MI
percentage
|
5.00 | % | 9.75 | % | 9.75 | % | 28.75 | % | 42.88 | % | ||||||||||||||
|
MI
interest
|
$ | 154,502 | (118,656 | ) | (42,451 | ) | (6,432 | ) | (243,448 | ) | $ | (256,486 | ) | |||||||||||
29.
Subsequent
Events
|
(a)
|
SiBiono
patents - on January 29, 2007, SiBiono entrusted Grandall Legal Group
Shenzhen Law Firm to issue a legal letter to Zhaohui Peng, one of the
shareholders of Sibiono and the inventor of Gendicine, requesting him to
transfer all the title of patents to
SiBiono.
|
On June
18, 1999, during the formation of SiBiono, Zhaohui Peng transferred the rights
to the patent “A new method for manufacturing recombinant adenovirus” and
related research results to SiBiono as a payment for the registered capital. In
return, Zhaohui Peng was granted 32.03% of the common stock of
SiBiono.
From 1999
to 2007, SiBiono successfully obtained various technology funds from various
government technology agencies to support the further research and development
activities of Gendicine. Due to this significant funding obtained by SiBiono,
Sibiono developed five additional patents which are summarized as
follows:
F-37
|
Item
|
Patent
name
|
Countries
/ Date
|
Application
Number
(1)
|
Publication
Number
(2)
|
Approved
Patent
Number (3)
|
Name
of Patent Inventor (6)
|
Name
of Applicant (6)
|
Patent
Assignees
|
||||||||
|
1
|
A
new method for manufacturing recombinant adenovirus
|
|||||||||||||||
|
A
|
China
|
98123346.5
|
CN1228474A
|
ZL98123346.5
|
Peng
|
Peng
|
SiBiono
|
|||||||||
|
Date
|
12/14/1998
|
9/15/1999
|
7/3/2002
|
|||||||||||||
|
2
|
A
recombinant constructed by a virus vector and a human tumor
suppressor gene and its use
|
|||||||||||||||
|
A
|
China
|
02115228.4
|
CN1401778A
|
ZL02115228.4
|
Peng
/ Zhang
|
Peng
/ Zhang
|
Peng
/ Zhang
|
|||||||||
|
Date
|
5/8/2002
|
3/12/2003
|
11/24/2004
|
|||||||||||||
|
B
|
PCT
(4)
|
PCT/CN/2004/000465
|
WO2004/078987A1
|
Not
Approved
|
Peng
/ Zhang
|
Peng
/ Zhang
|
N/A
|
|||||||||
|
Date
|
3/8/2004
|
9/16/2004
|
N/A
|
|||||||||||||
|
3
|
Recombinant
gene medicine of adenovirus vector and and gene p54 for treating
proloferative diseases
|
|||||||||||||||
|
A
|
China
|
03125129.3
|
CN1471977A
|
ZL03125129.3
|
Peng
/ Zhang
|
Peng
/ Zhang
|
Peng
/ Zhang
|
|||||||||
|
Date
|
5/10/2003
|
2/4/2004
|
7/25/2007
|
|||||||||||||
|
B
|
PCT
(4)
|
PCT/CN/2004/000458
|
WO2004/104204A1
|
Not
Approved
|
Peng
/ Zhang
|
Peng
/ Zhang
|
N/A
|
|||||||||
|
Date
|
5/9/2004
|
12/2/2004
|
N/A
|
|||||||||||||
|
4
|
The
application of recombinant adenoviral p53 as cancer vaccine
(tentative title)
|
|||||||||||||||
|
A
|
China
|
200510002779.1
|
CN1679641A
|
ZL200510002779.1
|
Peng
/ Zhang
|
Peng
/ Zhang
|
Peng
/ Zhang
|
|||||||||
|
Date
|
1/26/2005
|
10/12/2005
|
8/29/2007
|
|||||||||||||
|
B
|
PCT
(4)
|
PCT/CN/2005/000111
|
WO2006/079244A1
|
Not
Approved
|
Peng
/ Zhang
|
Peng
/ Zhang
|
N/A
|
|||||||||
|
Date
|
1/26/2005
|
8/3/2006
|
N/A
|
|||||||||||||
|
C
|
US
(5)
|
11/075035
|
2005/0281785A1
|
Not
Approved
|
Peng
/ Zhang
|
Unidentified
Yet
|
N/A
|
|||||||||
|
Date
|
3/7/2005
|
12/22/2005
|
N/A
|
|||||||||||||
|
5
|
Human
Embryonic Kidney (HEK) sub-clone cell line
|
|||||||||||||||
|
A
|
China
|
03126889.7
|
CN1513985A
|
Not
Approved
|
Peng
/ Zhang
|
Peng
/ Zhang
|
N/A
|
|||||||||
|
Date
|
6/13/2003
|
7/21/2004
|
N/A
|
|||||||||||||
|
B
|
PCT
(4)
|
PCT/CN/2004/000457
|
WO2004/111239
|
Not
Approved
|
Peng
/ Zhang
|
Peng
/ Zhang
|
N/A
|
|||||||||
|
Date
|
5/9/2004
|
12/23/2004
|
N/A
|
|||||||||||||
|
6
|
The
complex of polypeptide liposome and human VGEF gene, and its use and
human VGEF gene, and its use
|
|||||||||||||||
|
A
|
China
|
02134321.7
|
CN1389269A
|
Not
Approved
|
Peng
/ Zhang / Zhu
|
Peng
/ Zhang / Zhu
|
N/A
|
|||||||||
|
Date
|
7/4/2002
|
1/8/2003
|
N/A
|
F-38
Note:
|
|
(1)
|
Application
number is obtained when application is
submitted;
|
|
|
(2)
|
Publication
number is obtained after the first phase
examination;
|
|
|
(3)
|
Approved
patent number is obtained after the final
examination;
|
|
|
(4)
|
PCT
is referred to an International Patent Organization in
Paris;
|
|
|
(5)
|
US
is referred to the application is made in United States of America
alone;
|
|
|
(6)
|
Peng
is referred to Zhaohui Peng; Zhang is referred to Xiaozhi Zhang; Zhu is
referred to Jinya Zhu.
|
As
indicated in the above table, Item 1, the patent “A new method for manufacturing
recombinant adenovirus” had been assigned to SiBiono; however, the other
approved patents (item 2 through item 4) in PRC still have not been assigned to
SiBiono. The Group believes that all the above mentioned patents should be
rightfully transferred to SiBiono, a subsidiary of the Group. Accordingly, the
above mentioned legal letter was issued on this ground.
On August
27, 2008, the Group through its subsidiary, SiBiono filed an application to the
Guongdong Province Shenzhen City (Middle) Peoples’ Court to demand Zhaozhu Peng
to transfer back all the above mentioned patents to SiBiono. The case has been
accepted by the Court and is waiting for its further investigation. As of
September 30, 2009, no further progress has occurred.
|
(b)
|
As
mentioned in Note 12, following this arbitration decision, Benda Ebei has
the obligation to pay the total acquisition cost payable of Rmb12.48
million, plus the penalty and related legal and arbitration expenses,
totaling approximately Rmb12.80 million (or $1.88
million).
|
On May
22, 2008, Benda Ebei applied to Shenzhen People Court to terminate above
mentioned arbitration. The termination is based on the ground that Xiaozhi Zhang
does not own all 6.24% of SiBiono’s common stock. In fact, he only owns 3.28% of
SiBiono’s stock. The application has been accepted by Shenzhen People Court and
is waiting for its further investigation. As of September 30, 2009, no further
progress has occurred.
|
(c)
|
The
Company has become aware that Excalibur Limited Partnership and Excalibur
Limited Partnership II (the "Plaintiffs") filed a motion for summary
judgment in lieu of a complaint pursuant to CPLR § 3213 (the "Motion")
with the Supreme Court of the State of New York (the "Court"), alleging
that the Company has been delinquent on the payment of an aggregate sum of
$600,000 and accrued interest and costs arising from the Convertible
Promissory Notes that were issued to the Plaintiffs in April 2007 in
connection with a $7,560,000 private placement. Pursuant to the motion,
the Plaintiffs requested that the Court (1) enter summary judgment in
favor of Excalibur Limited Partnership (“Excalibur Limited”) in the amount
of $390,000 plus all accrued interest and costs, and, (2) enter summary
judgment in favor of Excalibur Small Cap Opportunities LP (“Excalibur
Small Cap”) in the amount of $210,000 and accrued interest and costs. As
of September 30, 2009, the Company has not received service of such
notice, and therefore, the Company does not have details regarding the
content of the complaint made by the
Plaintiffs.
|
|
(d)
|
On
March 4, 2009, the Company received a Notice and Default and Payment
Demand letter (the "Default Letter") from Pope Investments LLC ("Pope") in
connection with its convertible promissory note in the amount of
$5,520,000 (the "Note") purchased in our April 2007 private placement
offering. The Default Letter provided notice of default based on the
Company's failure to make a required interest payment on the Note by
February 20, 2009. The Default Letter further demanded full payment of all
interest, liquidated damages and accrued interest thereon in the amount of
$130,364.37 by March 14, 2009, or Pope will accelerate the maturity date
of the full principal amount of the Note. On April 7, 2009, the Company
received further Default Letters and Payment Demand from Pope, Excalibur
Limited and Excalibur Small Cap demanding payment in full of the balance
of the Notes, which matured on March 28, 2009. The Group has been
negotiating with Pope and all of the other noteholders listed above since
then and no conclusions have been reached as of September 30,
2009.
|
F-39
Item 2. Management’s Discussion and Analysis
or Plan of Operation
Critical Accounting
Policies
Accounting
policies discussed in this section are those that we consider to be most
critical to an understanding of our financial statements because they inherently
involve significant judgment and uncertainties. For all of these
estimates, we caution that future events rarely develop exactly as forecast, and
the best estimates routinely require adjustment.
Revenue
Recognition
Among the
most important accounting policies affecting the Group’s consolidated financial
statements is its policy of recognizing revenue in accordance with the SEC's
Staff Accounting Bulletin ("SAB") No. 104. Under this policy, all of the
following criteria must be met in order for us to recognize
revenue:
|
1.
|
Persuasive
evidence of an arrangement exists;
|
|
2.
|
Delivery
has occurred or services have been
rendered;
|
|
3.
|
The
seller's price to the buyer is fixed or determinable;
and
|
|
4.
|
Collectibility
is reasonably assured.
|
The
majority of the Group's revenue results from sales contracts
with distributors and revenue is recorded upon the shipment of goods.
Management conducts credit background checks for new customers as a means
to reduce the subjectivity of assuring collectibility. Based on these
factors, the Group believes that it can apply the provisions of SAB 104
with minimal subjectivity. Sales are presented net of value added tax (VAT). No
return allowance is made as products returns are insignificant based on
historical experience.
Estimates
Affecting Accounts Receivable and Inventories
The
preparation of our consolidated financial statements requires management to make
estimates and assumptions that affect our reporting of assets and liabilities
(and contingent assets and liabilities). However, it is explicated that the
changes in estimation were not material in the preparation of our consolidation
financial statements.
As of
September 30, 2009 and 2008, the Group provided a $6,867,978 and $1,552,068,
respectively for the allowance of doubtful accounts against trade receivables.
Management's estimate of the appropriate allowance on those accounts receivable
for the reporting periods was based on the aged nature of these accounts. In
making its judgment, management assessed its customers' ability to continue to
pay their outstanding invoices and the collectibility of those accounts on a
timely basis, and whether their financial position might deteriorate
significantly in the future, which would result in their inability to pay their
debts to the Company.
Inventories,
which are primarily comprised of raw materials, packaging materials, and
finished goods, are stated at the lower of cost or net realizable value. Cost
being determined on the basis of a moving average. The Group evaluates the need
for reserves associated with obsolete, slow-moving and non-salable inventory by
reviewing net realizable values on a periodic basis.
For the
nine months ended September 30, 2009 and 2008, the Group provided a reserve
against its obsolete, slow-moving or non-salable inventory amounting to
$4,925,299 and $4,191,726, respectively.
The
provision of reserve was mainly resulted from the manufacturing process of
Gendicine, SiBiono’s sole product and SiBiono was acquired by the company in
April 2007.
The
following chart shows the manufacturing process of Gendicine
(Ad-p53):
4

In the
production process of finished goods, Gendicine, several working steps are
needed: (i) large-scale culturing of adenovirus from master adenovirus bank;
(ii) culturing of cell from master cell bank; (iii) purification. The
whole process including step (i) to step (iii) takes approximately twenty-four
days to make reagent (“original liquid”). This particular liquid can
only be stored for approximately five years. It takes approximately
another seven days for mixing and bottling original liquid to finished goods
which is known as Gendicine.
Therefore,
up to the stage of reagent, all the related production costs are treated as
work-in-progress. The major components of those production costs are: (i) direct
labor; (ii) direct materials; (iii) power; (iv) supplies and other materials;
and (v) manufacturing overheads.
Before
acquisition, as of March 31, 2007, the accumulated units of original liquid
produced was 198,075 and which could be converted to approximately 226,736 vials
of Gendicine. However, the accumulated vials of Gendicine sold throughout the
years 2004 to three-month period ended March 31, 2007 were only approximately
18,424 vials. Thus the accumulated production costs of $4,080,644 were remained
as work-in-process as of March 31, 2007.
Furthermore,
due to the special feature of the original liquid which can only be stored for
five years, and most of the original liquid was produced in the year of 2004,
and the provision of reserve on work- in-progress was $3,696,083 as of March 31,
2007.
After the
acquisition with the effective date April 1, 2007, the same accounting treatment
was adopted for the treatment of the provision of reserve on work-in-progress.
As of September 30, 2009, the provision of reserve on work-in-process
was$4,742,417.
A reserve
for obsolete, slow-moving or non-salable inventory was made on raw materials,
packing materials, other material and suppliers, finished goods and
work-in-process at the amount of $82,026, $39,786, $54,563, $6,507 and
$4,742,417 respectively as of September 30, 2009.
Management
determination of this allowance was based on potential impairments to the
current carrying value of the inventories due to potential obsolescence of aged
inventories. In making its estimate, management considered the probable demand
for our products in the future and historical trends in the turnover of our
inventories. While the Company currently believes that there is little
likelihood that actual results will differ materially from these current
estimates.
Operational
Results
Nine months and three months
ended September 30, 2009 Compared to Nine months and three months ended
September 30, 2008
The
following table provides key components of our operational results for the nine
months and three months ended September 30, 2009 and 2008 for Benda
Pharmaceutical, Inc.
Error! Not a valid
link.
Net
Revenue:
The
Company has five core operating segments: Benda Ebei, Jiangling Benda, Yidu
Benda, Beijing Shusai and SiBiono. Benda Ebei manufactures branded/generic
medicines; Jiangling Benda manufactures active pharmaceutical ingredients (API);
Yidu
5
Benda
manufactures bulk chemicals; Beijing Shusai operates and distributes Pharyngitis
Killer Therapy; and SiBiono is a gene therapy company dedicated to the
development, manufacturing and commercialization of gene therapy product,
Gedicine.
Net
revenue decreased by $2.51 million (or 12.6%) to $17.35 million for the nine
months ended September 30, 2009 from $19.86 million for the nine months ended
September 30, 2008, while the net revenue decreased by $0.73 million (or 11.0%)
to $5.90 million for three months ended September 30, 2009 from $6.63 million
for three months ended September 30, 2008 Decrease in revenue is principally
attributed by the following factors:
|
1.
|
One
of the Benda’s subsidiaries, Benda Ebei’s net revenue decreased $3.82
million (or 21.9%) and $1.09 million (or 18.2%) to $13.62 million and
$4.89 million for the nine months and three months ended September 30,
2009 respectively from $17.44 million and $5.97 million for the nine
months and three months ended September 30, 2008 respectively. It was
mainly due to the keen competition in generic medicine and the seasonal
factors; however the management believes that the overall year revenue of
2009 will remain the same level as year
2008.
|
|
2.
|
One
of the Benda’s subsidiaries, Jiangling Benda which resumed its production
in October 2007 and achieved $0.8 million and $0.6 million for the nine
months ended September 30, 2009 and 2008,
respectively.
|
Jiangling
Benda plans to produce four types of active pharmaceutical ingredients and they
are Ribavirin, Asarin, Levofloxacin and Ribose whereas the production of Ribose
does not require the GMP certificate, but the production of the other three
products do require the GMP certificate.
On April 9, 2008, Jiangling Benda
received the approved GMP Certificate from the Chinese State Food and
Drug Administration ("SFDA") which authorizing the production of
Ribavrin. The other two products, Asarin and Levolfozacin, are still
under the stage of GMP certificate approving process. The management could not
estimate the exact timing for obtaining those certificates.
|
3.
|
One
of the Benda’s subsidiaries, Yidu Benda ceased operation due to the fact
that the plant was closed since mid January of 2007 to upgrade its waster
water treatment system to comply with new environmental standards enforced
by PRC local government.
|
Yidu
Benda has completed its upgrading of the waster water system and passed the
government’s verification and testing of equipments in October 2007. It is now
permitted for the testing on actual production process. Once the actual products
are produced, then the environmental government bodies will re-test the
production results. The management could not estimate the exact timing for
obtaining the final approval on the actual production process. Furthermore, the
management is searching for new products to be produced in Yidu Benda which with
higher profit margin.
|
4.
|
One
of the Benda’s subsidiaries, Beijing Shusai which incorporated on July 15,
2006. China’s State Food and Drug Administration (SFDA) recently
experienced an overhaul in its policies and regulatory systems in an
effort to fight against corruption in Chinese pharmaceutical industry.
Beijing Shusai’s operation has been adversely affected by this recent
policy changes which prohibits some state-owned hospitals from forming
alliances with private companies. The management could not estimate that
such situation could be resolved in the coming
future.
|
|
5.
|
One
of the Benda’s subsidiaries, SiBiono, acquired and effective since April
1, 2007, net revenue increased $1.10 million (or 60.9%) and $0.36 million
(or 56.7%) to $2.91 million and $0.99 million for the nine months and
three months ended September 30, 2009 respectively from $1.81 million and
$0.63 million for the nine months and three months ended September 30,
2008 respectively. The main reason for the increase in net
revenue is mainly due to the fact that SiBiono was previously underwent a
process of re-engineering of the production department during the year of
2008 which improved its production process and also the company put more
effort in the marketing
|
SiBiono
GMP - On October 16, 2003, SiBiono successfully obtained a New Drug License from
the State Food & Drug Administration of China (SFDA), and then, in April 4,
2004, SiBiono obtained “Manufacture Certificate” and “Certificate of GMP for
Pharmaceutical Product”, so far being fully qualified for the market launch of
Recombinant Human Ad-p53 Injection, trademarked as Gendicine
® in China. Gendicine ® is the
commercialized gene therapy product approved in the PRC government
agency. On May 19, 2008, SiBiono received an official notice from the
PRC State of SFDA in which it mentioned that during the random inspection
performed by the PRC State of SFDA on April 8 to April 10, 2008, the PRC State
of SFDA discovered there were several production procedures that did not meet
the requirement stated in GMP, thus it required SiBiono to perform necessary
improvements in order to fulfill the GMP requirements and the PRC State of SFDA
collected back the distributed GMP certificate until the necessary improvements
being carried out and passed the examination that conducted by
SFDA. On June 10, 2008, SiBiono received another official notice from
Guangdong Province SFDA and they demanded the same requirements as stated in the
official notice which issued by the PRC State of SFDA dated on May 19,
2008. On November 24, 2008, SiBiono received another official notice
from Guangdong Province SFDA which mentioned that after the examination
conducted by Shenzhen City SFDA, the Guangdong Province SFDA consent SiBiono to
carry out production on a trial basis. It further required SiBiono
strictly to follow the requirements of GMP to organize trail production and
follow the procedures to apply for GMP Certificate verification.
On July
14, 2009, SiBiono obtained the final approved GMP Certificate, in order words,
the SFDA allows SiBiono to resume its production and sales.
6
Cost
of Goods Sold
Cost of
goods sold decreased $2.84 million (or 22.5%) and $1.14 million (or 27.1%) to
$9.8 million and $3.1 million for the nine months and three months ended
September 30, 2009 respectively from $12.6 million and $4.2 million for the nine
months and three months ended September 30, 2008 respectively, primarily due to
the decreased in the sales volume in Benda Ebei.
Gross
Profit
Gross
profit increased $0.32 million (or 4.4%) and $0.41 million (or 16.9%) to $7.57
million and $2.83 million for the nine months and three months ended September
30, 2009 respectively from $7.25 million and $2.42 million for the nine months
and three months ended September 30, 2008 respectively, which was mainly due to
improved material management causing a slightly decrease in cost.
Selling
Expenses:
Selling
expenses slightly decreased $0.25 million (or 11.80%) to $1.87 million for the
nine months ended September 30, 2009 from $2.12 million for the nine months
ended September 30, 2008, but increased slightly $0.16 million to $0.85 million
for the three months ended September 30, 2009 from $0.69 million for the three
months ended September 30, 2008, primarily due to the management performed cost
control in the reporting period.
General
and Administrative Expenses:
General
and administrative increased $0.55 million (or 9.9%) and $0.59 million (or 45%)
to $6.11 million and $1.63 million for the nine months and three months ended
September 30, 2009 respectively from $5.56 million and $1.04 million for the
nine months and three months ended September 30, 2008. It was primary
due to the fact that the company was facing a global financial crisis, thus an
amount approximately $3.08 million and $0.27 million was provided for bad debt
provision, for the nine months and three months ended September 30, 2009,
increase by $2.5 million and $0.5 million respectively, compared with the same
period in 2008. However, the company will put more efforts to improve and manage
the situation onwards.
Operating
Income / (Loss):
The
Company resulted an operating loss $0.69 million for the nine months
ended September 30, 2009, at the same level as for the nine months ended
September 30, 2008. But the Company reported an operating income $0.18 million
for the three months, ended September 30, 2009, while the operating income from
comparative period for 2008 was $0.61 million. which was mainly due to increase
in net profit during the reporting periods.
Interest
Expenses:
Interest
expenses was $2.1 million and $0.47 million for the nine months and three months
ended September 30, 2009 respectively while the interest expenses was $3.34
million and $1.00 million for the comparative period of 2008 respectively. The
main component of it was the financing cost associated with the issuance of
convertible promissory note.
For the
three months ended September 30, 2009, $0.27 million was incurred for the
interest of the notes
Other
Income / (Expenses)
Due to
the Registration Delay Penalty, the Group incurred an expense for the nine
months ended September 30, 2008. Out of the total penalty, $230,312 was settled
by issuance of 523,438 shares of common stock. There was no penalty to investors
incurred for the nine months ended September 30, 2009.
Income
Taxes:
Benda is
subject to Delaware, United State of America tax, but no provision for income
taxes were made for the nine months and three months ended September 30, 2009
and 2008 as Benda did not have reportable taxable income for the
period.
Ever
Leader, a wholly owned subsidiary of Benda, is subject to Hong Kong tax, but no
provisions for income taxes were made for the nine months and three months ended
September 30, 2009 and 2008 as Ever Leader did not have reportable taxable
income for the periods.
Benda
Ebei was registered as a Sino-Foreign Equity Joint Venture on May 26, 2004 and
is subject to the tax laws applicable to Sino-Foreign Equity Joint Ventures in
the PRC. Benda Ebei, starting from 2005, is fully exempt from PRC
enterprise income tax for two years starting from the first profit-making year,
followed by a 50% reduction in the state income taxes, for the following three
years, commencing from the first profitable year.
Jiangling
Benda and Yidu Benda are cross-municipal investment entities and enjoy the same
tax treatment as Sino-Foreign Joint Ventures, starting from 2005, and were
therefore exempt from PRC enterprise income tax for two years starting from the
first profit-making year, followed by a 50% reduction in the state income taxes,
for the following three years, commencing from the first profitable year.
Cross-municipal investments entities refer to entities that are incorporated in
one municipal region but have investments in another municipal
region.
7
The
exemption periods for Benda Ebei, Jiangling Benda and Yidu Benda expired in the
year of 2006, after which they are subject to a 50% reduction in state income
taxes, at 18%; whereas the full income tax rate is 33%. The remaining tax
holidays will be expired in 2010.
However,
starting and effective from January 1, 2008, the full income tax rate would be
changed from 33% to 25% according to the new PRC taxation regulations. Therefore
these subsidiaries will be subject to the regular full income tax rate at 25%
after the tax holidays expire in 2010.
According
to the new taxation regulations starting and effective from January 1, 2008,
Beijing Shusai is subject to the full income tax rate of 25%.
According
to the new taxation regulations starting and effective from January 1, 2008,
SiBiono, which is located in Shenzhen, a Special Economic District of PRC, is
subject to the full income tax rate of 25% gradually in five years as
following:
|
Year
|
Tax
rate
|
|
|
2008
|
18%
|
|
|
2009
|
20%
|
|
|
2010
|
22%
|
|
|
2011
|
24%
|
|
|
2012
and thereafter
|
25%
|
Benda
Ebei recorded $163,803 income tax for the nine months ended September 30, 2009.
The prepaid income tax by the Group as of September 30, 2009 at amount of $1.66
million was recorded as taxes recoverable.
LIQUIDITY
AND CAPITAL RESOURCES
Net cash
provided by the operating activities was $1.19 million for the nine months ended
September 30, 2009, while for the nine months ended September 30, 2008 was
negative $0.44 million
|
a)
|
Non-cash
operating activities, reconciliation items to the net
income
|
For the
nine months ended September 30, 2008, an amount about $4.48 million non-cash
operating activities was reconciled back to the net income and which mainly
included amortization of debt discount and debt issue cost, penalty payment made
in form of share issuance, bad debt provision, amortization of intangible
assets, and depreciation.
However,
for the nine months ended September 30, 2009, about $4.02 million of non-cash
operating activities was reconciled back to the net income and summarized as
follows:
|
1.
|
$0.86
million that incurred as interest expenses related to the amortization of
discount on long-term debt and issuance cost associated with the warrants
and the beneficial conversion features (please refer to Note 24 of the
Notes to Consolidated Financial Statements for details);
and
|
|
2.
|
Other
factors: $3.08 million incurred on bad debt provision; $1.61 million
incurred on depreciation; and $0.55 million incurred on amortization of
intangible assets.
|
|
b)
|
Trade
receivables
|
The net
change of trade receivable was increased by $2.43 million for the nine months
ended September 30, 2009. The management also noticed that the net balance of
the trade receivable, as of September 30, 2009, was a significant asset to the
company. However, the management believes that the above situation is
temporarily due to the following reasons:
Trade
receivables
|
a)
|
Customers
whom have sales relationship with our company are all relatively big
business wholesale enterprises and they have all passed the examination of
GMP Certificate so that the collectibility from those is out of
question;
|
|
b)
|
The
management realized that it did affect the cash flow situation of the
company; therefore the company will put more efforts to reduce the balance
of trade receivables.
|
For the
nine months ended September 30, 2009 and 2008, the amount spent in investing
activities were $0.46 million and $0.21 million respectively. It was mainly
because most of the settlements were made in the year of 2007, thus the
investing activities were relatively small for the reporting
periods.
Financing
cash outflow was $0.07 million for the reporting period nine months ended
September 30, 2009 while the financing cash inflow was $0.70 million for the
reporting period nine months ended September 30, 2008. The major of
it was a net amount
8
$1.8
million was received from the commercial bank notes in the nine months ended
September 30, 2008. (Please refer to the Note 11 of the Notes to Consolidated
Financial Statements for the details)
Item
3. Quantitative and Qualitative Disclosures About Market
Risk
The
Company is subject to certain market risks, including changes in interest rates
and currency exchange rates. The Company does not undertake any specific
actions to limit those exposures.
Item
4T. Evaluation of Disclosure Controls and Procedures
a)
Evaluation of Disclosure
Controls. Our Chief Executive Officer and Chief Accounting Officer
evaluated the effectiveness of our disclosure controls and procedures as of the
end of our third fiscal quarter 2009 pursuant to Rule 13a-15(b) of the
Securities and Exchange Act. Disclosure controls and procedures are
controls and other procedures that are designed to ensure that information
required to be disclosed by us in the reports that we file or submit under the
Exchange Act is recorded, processed, summarized and reported within the time
periods specified in the SEC’s rules and forms. Disclosure controls and
procedures include, without limitation, controls and procedures designed to
ensure that information required to be disclosed by us in the reports that we
file under the Exchange Act is accumulated and communicated to our management,
as appropriate to allow timely decisions regarding required disclosure. Based on
his evaluation, our Chief Executive Officer and Chief Accounting Officer
concluded that our disclosure controls and procedures were effective as of
September 30, 2009.
It should
be noted that any system of controls, however well designed and operated, can
provide only reasonable, and not absolute, assurance that the objectives of the
system are met. In addition, the design of any control system is based in part
upon certain assumptions about the likelihood of future events. Because of these
and other inherent limitations of control systems, there can be no assurance
that any design will succeed in achieving its stated goals under all potential
future conditions
(b)
Changes in internal
control over financial reporting. There have been no changes in our
internal control over financial reporting that occurred during the third fiscal
quarter that has materially affected, or is reasonably likely to materially
affect, our internal control over financial reporting. Our management team will
continue to evaluate our internal control over financial reporting in 2009 as we
implement our Sarbanes Oxley Act testing.
9
PART
II - OTHER INFORMATION
Item
1. Legal Proceedings.
The
Company has become aware that Excalibur Limited Partnership and Excalibur
Limited Partnership II (the "Plaintiffs") filed a motion for summary judgment in
lieu of a complaint pursuant to CPLR § 3213 (the "Motion") with the Supreme
Court of the State of New York (the "Court"), alleging that the Company has been
delinquent on the payment of an aggregate sum of $600,000 and accrued interest
and costs arising from the Convertible Promissory Notes that were issued to the
Plaintiffs in April 2007 in connection with a $7,560,000 private placement.
Pursuant to the motion, the Plaintiffs requested that the Court (1) enter
summary judgment in favor of Excalibur Limited Partnership (“Excalibur Limited”)
in the amount of $390,000 plus all accrued interest and costs, and, (2) enter
summary judgment in favor of Excalibur Small Cap Opportunities LP (“Excalibur
Small Cap”) in the amount of $210,000 and accrued interest and
costs. On July 29, 2009, the Court entered a judgment against the
Company in favor of the Plaintiffs in the amount of $674,251.65 in connection
with the Convertible Promissory Notes issued to the Plaintiffs in April 2007 in
a private placement.
On March
4, 2009, the Company received a Notice and Default and Payment Demand letter
(the "Default Letter") from Pope Investments LLC ("Pope") in connection with its
convertible promissory note in the amount of $5,520,000 (the "Note") purchased
in our April 2007 private placement offering. The Default Letter provided notice
of default based on the Company's failure to make a required interest payment on
the Note by February 20, 2009. The Default Letter further demanded full payment
of all interest, liquidated damages and accrued interest thereon in the amount
of $130,364.37 by March 14, 2009, or Pope will accelerate the maturity date of
the full principal amount of the Note. On April 7, 2009, the Company
received further Default Letters and Payment Demand from Pope, Excalibur Limited
and Excalibur Small Cap demanding payment in full of the balance of the Notes,
which matured on March 28, 2009. We were notified on June 15, 2009,
that on May 11, 2009 Pope filed a motion for summary judgment in lieu of a
complaint against us pursuant to CPLR § 3213 (the “Motion”) with the Supreme
Court of the State of New York (the “Court”), alleging that we have been
delinquent on the payment of an aggregate sum of $5,520,000 and accrued interest
and costs arising from the Note that we issued to the Pope in April
2007. Pursuant to the motion, the Plaintiffs requested that the Court
enter summary judgment in favor of Plaintiff in the amount of $5,994,617.53
constituting principal and interest, plus costs.
On June
23, 2009, we filed an Affidavit in Opposition to Motion for Summary Judgment in
Lieu of Complaint with the Court requesting that Plaintiff’s Motion be denied.
On October 14, 2009, this motion was denied, and the court entered a judgment in
favor of Pope in the amount of $5,520,000 plus interest.
On July
30, 2009, we received a Notice of Default from three additional investors in the
April 2007 private placement offering holding Notes totaling
$90,000.
|
2.
|
On
November 23, 2006, Benda Ebei entered into an Equity Transfer Agreement
with Xiaozhi Zhang (“Zhang”), to purchase approximately 6.24% of SiBiono’s
common stock for a total consideration of Rmb12.48 million (Rmb6.24
million in cash and shares of our common stock equal to Rmb6.24 million)
(or $1.71 million) which was due and payable on or before March 31,
2007.
|
Due to
the fact that the signed agreement on November 23, 2006 was not practically
executable according to the PRC regulations, Benda Ebei asked Zhang to terminate
the signed agreement and sign a new agreement that was feasible under PRC
regulations with essentially the same terms.
However,
Zhang refused to sign the new agreement and applied to the Shenzhen Arbitration
Commission (the “Commission”) in April 2007 for enforcement of the original
agreement. Zhang requested the Commission to require Benda Ebei to pay for the
total consideration, penalty for late payment and the related legal and
arbitration expenses.
On
November 27, 2007, Shenzhen Arbitration Commission determined that:
|
1.
|
Benda
Ebei should pay for the consideration of Rmb 6.24 million, equal to 50% of
the total consideration set forth in the Equity Transfer Agreement. For
the other 50% of the total consideration which was supposed to be settled
in the form of issuing common stock, since Zhang did not make an
arbitration request on how to execute the arrangement,
the Arbitration Commission did not make an award on this particular
part.
|
|
| 2. | Benda Ebei should pay for the penalty of Rmb 46,800; | |
|
3.
|
Benda Eebi should pay for legal and arbitration expenses of Rmb 268,971. |
10
On May
22, 2008, Benda Ebei applied to Shenzhen People Court to terminate above
mentioned arbitration. The termination is based on the ground that Xiaozhi Zhang
does not own all 6.24% of SiBiono’s common stock. In fact, he only owns 3.28% of
SiBiono’s stock. The application has been accepted by Shenzhen People Court and
is waiting for its further investigation..
|
3.
|
SiBiono
patents - on January 29, 2008, SiBiono entrusted Grandall Legal Group
Shenzhen Law Firm to issue a legal letter to Zhaohui Peng, one of the
shareholders of Sibiono and the inventor of Gendicine, demanding that he
transfer all the title of patents to
SiBiono.
|
On June
18, 1999, during the formation of SiBiono, Zhaohui Peng transferred the rights
to the patent “A new method for manufacturing recombinant adenovirus” and
related research results to SiBiono as a payment for the registered capital. In
return, Zhaohui Peng was granted 32.03% of the common stock of
SiBiono.
From 1999
to 2007, SiBiono successfully obtained various technology funds from various
government technology agencies to support the further research and development
activities of Gendicine. Due to this significant funding obtained by SiBiono,
Sibiono developed five additional patents which are summarized as
follows:
|
Item
|
Patent
name
|
Countries
/ Date
|
Application
Number
(1)
|
Publication
Number
(2)
|
Approved
Patent Number (3)
|
Name
of Patent Inventor (6)
|
Name
of
Applicant
(6)
|
Patent
Assignees
|
||||||||
|
1
|
A
new method for manufacturing recombinant adenovirus
|
|||||||||||||||
|
A
|
China
|
98123346.5
|
CN1228474A
|
ZL98123346.5
|
Peng
|
Peng
|
SiBiono
|
|||||||||
|
Date
|
1998/12/14
|
1999/9/15
|
2002/7/3
|
|||||||||||||
|
2
|
A
recombinant constructed by a virus vector and a human tumor
suppressor gene and its use
|
|||||||||||||||
|
A
|
China
|
02115228.4
|
CN1401778A
|
ZL02115228.4
|
Peng
/ Zhang
|
Peng
/ Zhang
|
Peng
/ Zhang
|
|||||||||
|
Date
|
2002/5/8
|
2003/3/12
|
2004/11/24
|
|||||||||||||
|
B
|
PCT
(4)
|
5
|
WO2004/078987A1
|
Not
Approved
|
Peng
/ Zhang
|
Peng
/ Zhang
|
N/A
|
|||||||||
|
Date
|
2004/3/8
|
2004/9/16
|
N/A
|
|||||||||||||
|
3
|
Recombinant
gene medicine of adenovirus vector and and gene p54 for treating
proloferative diseases
|
|||||||||||||||
|
A
|
China
|
03125129.3
|
CN1471977A
|
ZL03125129.3
|
Peng
/ Zhang
|
Peng
/ Zhang
|
Peng
/ Zhang
|
|||||||||
|
Date
|
2003/5/10
|
2004/2/4
|
2007/7/25
|
|||||||||||||
|
B
|
PCT
(4)
|
8
|
WO2004/104204A1
|
Not
Approved
|
Peng
/ Zhang
|
Peng
/ Zhang
|
N/A
|
|||||||||
|
Date
|
2004/5/9
|
2004/12/2
|
N/A
|
|||||||||||||
|
4
|
The
application of recombinant adenoviral p53 as cancer vaccine
(tentative title)
|
|||||||||||||||
|
A
|
China
|
200510002779.1
|
CN1679641A
|
ZL200510002779.1
|
Peng
/ Zhang
|
Peng
/ Zhang
|
Peng
/ Zhang
|
|||||||||
|
Date
|
2005/1/26
|
2005/10/12
|
2007/8/29
|
|||||||||||||
|
B
|
PCT
(4)
|
1
|
WO2006/079244A1
|
Not
Approved
|
Peng
/ Zhang
|
Peng
/ Zhang
|
N/A
|
|||||||||
|
Date
|
2005/1/26
|
2006/8/3
|
N/A
|
|||||||||||||
|
C
|
US
(5)
|
11/075035
|
2005/0281785A1
|
Not
Approved
|
Peng
/ Zhang
|
Unidentified
Yet
|
N/A
|
|||||||||
|
Date
|
2005/3/7
|
2005/12/22
|
N/A
|
|||||||||||||
|
5
|
Human
Embryonic Kidney (HEK) sub-clone cell line
|
|||||||||||||||
|
A
|
China
|
03126889.7
|
CN1513985A
|
Not
Approved
|
Peng
/ Zhang
|
Peng
/ Zhang
|
N/A
|
|||||||||
|
Date
|
2003/6/13
|
2004/7/21
|
N/A
|
|||||||||||||
|
B
|
PCT
(4)
|
7
|
WO2004/111239
|
Not
Approved
|
Peng
/ Zhang
|
Peng
/ Zhang
|
N/A
|
|||||||||
|
Date
|
2004/5/9
|
2004/12/23
|
N/A
|
|||||||||||||
|
6
|
The
complex of polypeptide liposome and human GEF gene, and its use and
human VGEF gene, and its use
|
|||||||||||||||
|
A
|
China
|
02134321.7
|
CN1389269A
|
Not
Approved
|
Peng
/ Zhang / Zhu
|
Peng
/ Zhang / Zhu
|
N/A
|
|||||||||
|
Date
|
2002/7/4
|
2003/1/8
|
N/A
|
11
Note:
|
(1)
|
Application
number is obtained when application is submitted;
|
|
(2)
|
Publication
number is obtained after the first phase examination;
|
|
(3)
|
Approved
patent number is obtained after the final examination;
|
|
(4)
|
PCT
is referred to an International Patent Organization in
Paris;
|
|
(5)
|
US
is referred to the application is made in United States of America
alone;
|
|
(6)
|
Peng
is referred to Zhaohui Peng; Zhang is referred to Xiaozhi Zhang; Zhu is
referred to Jinya Zhu.
|
As
indicated in the above table, Item 1, the patent “A new method for manufacturing
recombinant adenovirus” had been assigned to SiBiono; however, the other
approved patents (item 2 through item 4) in PRC still have not been
assigned to SiBiono. The Group believes that all the above mentioned patents
should be rightfully transferred to SiBiono, a subsidiary of the Group.
Accordingly, the above mentioned legal letter was issued on this
ground.
On August
27, 2008, the Group through its subsidiary, SiBiono filed an application to the
Guongdong Province Shenzhen City (Middle) Peoples’ Court and
demand Zhaozhu Peng to transfer back all the mentioned patents that mentioned in
above to SiBiono. The case has been accepted by the Court and is waiting for its
further investigation.
Item
2. Unregistered Sales of Equity Securities and Use of Proceeds
None.
Item
3. Defaults Upon Senior Securities.
12
The
Company has become aware that Excalibur Limited Partnership and Excalibur
Limited Partnership II (the "Plaintiffs") filed a motion for summary judgment in
lieu of a complaint pursuant to CPLR § 3213 (the "Motion") with the Supreme
Court of the State of New York (the "Court"), alleging that the Company has been
delinquent on the payment of an aggregate sum of $600,000 and accrued interest
and costs arising from the Convertible Promissory Notes that were issued to the
Plaintiffs in April 2007 in connection with a $7,560,000 private placement.
Pursuant to the motion, the Plaintiffs requested that the Court (1) enter
summary judgment in favor of Excalibur Limited Partnership (“Excalibur Limited”)
in the amount of $390,000 plus all accrued interest and costs, and, (2) enter
summary judgment in favor of Excalibur Small Cap Opportunities LP (“Excalibur
Small Cap”) in the amount of $210,000 and accrued interest and
costs. On July 29, 2009, the Court entered a judgment against the
Company in favor of the Plaintiffs in the amount of $674,251.65 in connection
with the Convertible Promissory Notes issued to the Plaintiffs in April 2007 in
a private placement.
On March
4, 2009, the Company received a Notice and Default and Payment Demand letter
(the "Default Letter") from Pope Investments LLC ("Pope") in connection with its
convertible promissory note in the amount of $5,520,000 (the "Note") purchased
in our April 2007 private placement offering. The Default Letter provided notice
of default based on the Company's failure to make a required interest payment on
the Note by February 20, 2009. The Default Letter further demanded full payment
of all interest, liquidated damages and accrued interest thereon in the amount
of $130,364.37 by March 14, 2009, or Pope will accelerate the maturity date of
the full principal amount of the Note. On April 7, 2009, the Company
received further Default Letters and Payment Demand from Pope, Excalibur Limited
and Excalibur Small Cap demanding payment in full of the balance of the Notes,
which matured on March 28, 2009. We were notified on June 15, 2009,
that on May 11, 2009 Pope filed a motion for summary judgment in lieu of a
complaint against us pursuant to CPLR § 3213 (the “Motion”) with the Supreme
Court of the State of New York (the “Court”), alleging that we have been
delinquent on the payment of an aggregate sum of $5,520,000 and accrued interest
and costs arising from the Note that we issued to the Pope in April
2007. Pursuant to the motion, the Plaintiffs requested that the Court
enter summary judgment in favor of Plaintiff in the amount of $5,994,617.53
constituting principal and interest, plus costs.
On
June 23, 2009, we filed an Affidavit in Opposition to Motion for Summary
Judgment in Lieu of Complaint with the Court requesting that Plaintiff’s Motion
be denied. On
October 14, 2009, this motion was denied, and the court entered a judgment in
favor of Pope in the amount of $5,520,000 plus interest.
On July
30, 2009, we received a Notice of Default from three additional investors in the
April 2007 private placement offering holding Notes totaling
$90,000.
Item
4. Submission of Matters to a Vote of Security Holders.
None.
Item
5. Other Information.
None
Item
6. Exhibits and Reports of Form 8-K.
|
(a)
|
Exhibits
|
|
31.1
|
Certifications pursuant to Section 302 of Sarbanes Oxley Act of 2002 |
|
32.1
|
Certifications pursuant to Section 906 of Sarbanes Oxley Act of 2002 |
|
(b)
|
Reports on Form 8-K |
|
|
On July 7, 2009, the Company filed a Form 8-K updating the status of litigation. |
| On August 19, 2009, the Company filed a Form 8-K disclosing the Company’s new address. | |
| On August 19, 2009, the Company filed a Form 8-K disclosing a judgment against the Company. | |
| On September 22, 2009, the Company filed a Form 8-K disclosing the resignation of an officer. |
13
SIGNATURES
In
accordance with Section 13 or 15(d) of the Exchange Act, the registrant caused
this report to be signed on its behalf by the undersigned, there unto duly
authorized.
|
BENDA
PHARMACEUTICAL, INC.
|
|||
|
Registrant
|
|||
|
Date:
November 23, 2009
|
By:
|
/s/ Yiqing Wan | |
|
Yiqing
Wan
|
|||
|
President,
Chief Executive Officer,
Chief
Financial and Accounting
Officer
|
|||
|
Chairman
of Board of Directors
|
|||
14
