Attached files
U.S. SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 8-K
Current Report Pursuant to Section 13 or 15(d) of
the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported):
October 15, 2009
SUNSHINE BIOPHARMA, INC.
(Exact name of small business issuer as specified in its charter)
MOUNTAIN WEST BUSINESS SOLUTIONS, INC.
(Former Name)
|
Colorado |
000-0-52898 |
20-5566275 |
|
(State or other jurisdiction of incorporation) |
(Commission File Number) |
(IRS Employer ID No.) |
6100 Royalmount Ave.
Montreal, Quebec, Canada H4P 2R2
(Address of principal executive offices)
9844 W Powers Circle
Littleton, CO 80123
(Former Address)
(514) 496-5197
(Issuer's Telephone Number)
Item 2.01 Completion Of Acquisition Or Disposition Of Assets.
Effective October 15, 2009, we executed an agreement to acquire Sunshine Biopharma, Inc., a Colorado corporation (“SBI”), in exchange for the issuance of 21,962,000 shares of our Common Stock and 850,000 shares of Convertible Preferred Stock, each convertible into twenty (20) shares of our Common Stock (the “Agreement”). As a result of this transaction our officers and directors resigned their positions with us and were replaced by our current management. See Item 5.02, below. The effectiveness of the Agreement was conditional upon various conditions being satisfied, including the filing of our Form 10-K for our fiscal year ended August 31, 2009 and SBI changing its name to Sunshine Etopo, Inc. As of the date of this report, these conditions have been satisfied and Sunshine Etopo (formerly SBI) is now a wholly owned subsidiary of our Company. Also as a result of this transaction we have authorized changed our name to “Sunshine Biopharma, Inc.” We intend to file an amendment to this report upon receipt from the Colorado Secretary of State of an acknowledgment of this filing.
We entered into this transaction because of our former management’s belief that by doing so we will significantly increase our shareholder’s future opportunity to enhance the value of their respective ownership in our Company. In addition, our former Board of Directors approved a “spin-off” of our wholly owned subsidiary company Mountain West Beverage, Inc. The terms of this “spin-off” provide for a dividend to be issued to our shareholders of one share of common stock for every share that our shareholders owned as of October 14, 2009, the record date of the dividend.
Description of Business
As a result of the transaction described above we have revised our current business plan to that of a pharmaceutical company focused on the research, development and commercialization of drugs for the treatment of various forms of cancer. Our lead compound, Difluoro-Etoposidetm, a multi-purpose anti-tumor compound, is expected to enter Phase I clinical trials in 2010. We have licensed our technology on an exclusive basis from Advanomics Corporation, a privately held Canadian company (“Advanomics”), and we are planning to initiate our own R&D program as soon as practicable, once financing is in place. There are no assurances that we will obtain the financing necessary to allow us to implement this aspect of our business plan, or to enter clinical trials.
Carbon-Difluoride Technology
Many therapeutically important compounds contain diester bonds that link different parts of the molecule together. Diester bonds are naturally unstable often leading to suboptimal performance when the molecule is administered to patients. Diester bonds have specific three-dimensional, as well as electrostatic properties that cannot be easily mimicked by other bonds. Bonds that do not mimic the diester bond correctly invariably render the compound inactive. In collaboration with L’Institut National des Sciences Appliquées de Rouen in France (“INSA”), Advanomics has developed a way to replace the diester bond with a Carbon-Difluoride bond which acts as a diester isostere. An isostere is a different chemical structure that mimics the properties of the original. In the body, Carbon-Difluoride compounds are resistant to metabolic degradation but recognized similarly to the diester compounds (See Figure 1).
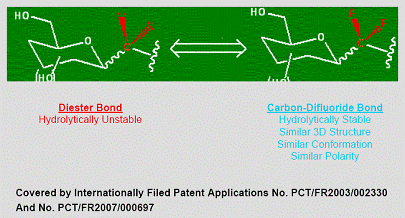
Figure 1
While no assurances can be provided, we are planning to expand our product line through acquisitions and/or in-licensing as well as in-house research & development.
Our Lead Compound (Adva-27a)
Our initial drug candidate is Difluoro-Etoposidetm (Adva-27a), a Carbon-Difluoride derivative of Etoposide, targeted for various forms of cancer. If sufficient funding can be obtained, Difluoro-Etoposidetm is expected to enter Phase I clinical trials in Canada during 2010. Etoposide is currently on the market and has been for over 20 years. It is sold under different brand names by various drug companies including, VePesid, VP-16, Etopophos and Vumon or Teniposide (Bristol-Myers Squibb, the original developer), Toposar (Sicor/Pfizer), Lastet (Nippon Kayaku Ltd) and Etoposide (TEVA, Bedford Laboratories, Supergen, American Pharmaceutical Partners, Watson Pharmaceuticals, and Genpharm). Etoposide is an effective anti-tumor compound and is currently in use to treat various types of cancer including leukemia, lymphoma, testicular cancer, breast cancer, lung cancer, brain cancer, prostate cancer, bladder cancer, colon cancer, ovarian cancer, liver cancer and several other forms of cancer. It is also being tested in clinical trials against other types of cancer, such as Kaposi's sarcoma. Etoposide is administered both intravenously and orally as liquid capsules.
This Etoposide compound which is currently in use suffers from molecular instability leading to reduced efficacy and high toxicity. Using its Carbon-Difluoride platform technology (see Figure 1), Advanomics has constructed several Difluoro derivatives of Etoposide by replacing the labile diester bond between the sugar and the toxin moieties of the existing Etoposide molecule with a Carbon-Difluoride bond (Figure 1). All Difluoro substituted constructs were found to be completely stable. Advanomics subsequently tested these constructs for their ability to kill cancer cells in vitro by conducting side-by-side experiments against the standard Etoposide compound. The results of these studies, which have been published in our patent application PCT/FR2007/000697, are summarized in Table 1. One of the constructs, Adva-27a, showed enhanced cancer cell killing activity over the existing Etoposide molecule (see Table 1).
This new compound, which we call Difluoro-Etoposidetm, is entering Phase I clinical trials in Canada in 2010. Subject to receipt of financing, we anticipate the Phase I clinical trials to be completed by late 2010 at which time we will apply for limited marketing approval (see Clinical Trials below).
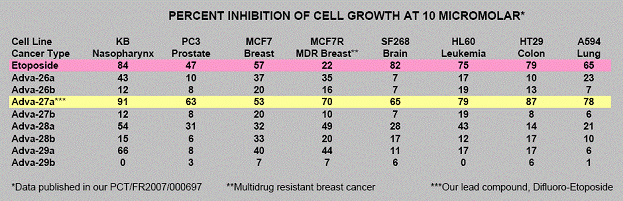
Table 1
Clinical Trials
Advanomics is entering Phase I clinical trials for our lead compound, Difluoro-Etoposidetm, in Canada in 2010. The planned clinical trials for small-cell lung cancer indication will be carried out at the Jewish General Hospital, one of the McGill University Hospital Centers in Montreal (Canada). In addition, Advanomics is planning to conduct Phase I clinical trials on Multi-Drug-Resistant breast cancer patients at Hotel Dieu Hospital, one of the University of Montreal Hospital Centers. All aspects of the planned clinical trials in Canada will employ FDA standards at all levels. We anticipate the clinical trials to begin in early 2010 and be completed by late 2010, at which time we together with our licensor will file for limited marketing approval with the regulatory authorities in Canada and the FDA in the U.S. (see Marketing below).
Marketing
According to the American Cancer Society, nearly 1.5 million new cases of cancer are diagnosed in the U.S. each year. Given the terminal and limited treatment options available for the indications Advanomics is planning to study, we anticipate being granted limited marketing approval for our Difluoro-Etoposidetm following receipt of funding and a successful Phase I. There are no assurances that either will occur. Such limited approval will allow us to make the drug available to various hospitals and health care centers for experimental therapy/further studies, thereby generating some revenues. As with the existing Etoposide, our Difluoro-Etoposidetm is anticipated to be usable to treat virtually all forms of cancer and supervising physicians are at liberty to prescribe it once it is available on the market. Similarly to the existing Etoposide, our Difluoro-Etoposidetm product will be a single-treatment blister-pack comprised of 20 gel-caps each containing 50 milligrams of Difluoro-Etoposidetm for a total of 1 gram per pack.
Intellectual Property
We are the exclusive licensee for the US territory of Advanomics’ Difluoro-Etoposidetm which is covered by international patent applications filed on April 25, 2006 (PCT/FR2007/000697). This patent, which is issued in France and still pending elsewhere, is owned by L’Institut National des Sciences Appliquées de Rouen (France) and is licensed exclusively on a worldwide basis to Advanomics Corporation, who has subsequently issued to us an exclusive license for the US.
Our Lead Anti-Cancer Compound in 3D
Property
We have moved our principal place of business to 6100 Royalmount Ave., Montreal, Quebec, Canada H4P 2R2. This is also the location of our licensor, Advanomics Corporation, who is providing this space to us on a rent free basis as of the date of this report. If and when we are able to secure financing we expect that we will pay rent commensurate with rents for similar locations in Montreal, Canada. Our current space consists of approximately 100 square feet of executive office space and 250 square feet of laboratory space. We anticipate that this will be sufficient for our needs for the foreseeable future.
Government Regulations
Our existing and proposed business operations are subject to extensive and frequently changing federal, state, provincial and local laws and regulations. We will be subject to significant regulations in the US in order to obtain the approval of the Federal Drug Administration (“FDA”) to offer our product. The approximate procedure for obtaining FDA approval involves an initial filing of an IND (Innovative New Drug) application or simply an NDA (New Drug Application) following which the FDA would give the go ahead with Phase I clinical (human) trials. Following completion of Phase I, the results are filed with the FDA and a request is made to proceed to Phase II. Similarly, following completion of Phase II the data are filed with the FDA and a request is made to proceed to Phase III. Following completion of Phase III, a request is made for marketing approval. Depending on various issues and considerations, the FDA could provide limited marketing approval on a humanitarian basis if the drug treats terminally ill patients with limited treatment options available. Part of what may be required to be included in the IND are animal studies (xenografts and toxicity studies). Such studies have not as yet been carried out. We also have not as of this date made any filings with the FDA or other regulatory bodied in other jurisdictions. We have however had extensive discussions with clinicians at the Jewish General Hospital in Montreal where we plan to undertake our initial Phase I study and they believe that Health Canada is likely to grant us a so-called fast-track process on the basis of the terminal nature of the cancer which we will be treating.
Employees
As of the date of this report we have three (3) employees, our management. We anticipate that if we receive financing we will hire additional employees in the areas of accounting, regulatory affairs, marketing and laboratory personnel.
Competition
We will be competing with publicly and privately held companies engaged in developing a cure for cancer. There are numerous other entities engaged in this business that have greater resources, both financial and otherwise, than the resources presently available to us. Nearly all major pharmaceutical companies including Amgen, Genentech, Pfizer, Bristol-Myers Squibb and, Novartis, to name just a few, have on-going anti-cancer drug development program and some of the drug they may develop could be in direct competition with our drug. Also, a number of small companies are also working in the area of cancer and could develop drugs that may be in competition with ours. However, none of these competitor companies can use molecules similar to ours as they would be infringing our patents.
Executive Compensation
We do not expect to pay salaries to any of our executive officers or directors until such time as we are able to secure adequate funding for our operations.
Item 3.02 Unregistered Sales of Equity Securities
Effective October 15, 2009, we executed an agreement to acquire Sunshine Biopharma, Inc., a Colorado corporation, in exchange for the issuance of 21,962,000 shares of our Common Stock and 850,000 shares of Convertible Preferred Stock, each convertible into twenty (20) shares of our Common Stock.
Item 5.01 Changes in Control of the Registrant
The following table sets forth certain information regarding the ownership of Common Stock as of the date of this report by (i) each of our directors, (ii) each of our executive officers, and (iii) all of our directors and executive officers as a group. Unless otherwise indicated, all shares are owned directly and the indicated person has sole voting and investment power.
|
Title of Class |
|
Name and Address Of Beneficial Owner |
|
Amount and Nature Of Beneficial Ownership |
|
Percent Of Class |
|
|
|
|
|
|
|
|
|
Common |
|
Dr. Steve N. Slilaty(1) 579 rue Lajeunesse Street Laval, Quebec Canada H7X 3K4 |
|
34,343,567(2)) |
|
73.7% |
|
|
|
|
|
|
|
|
|
Common |
|
Michele Di Turi(1) 3100 Boulevard Des Gouverneurs Laval, Quebec Canada H7E 5J3 |
|
234,373 |
|
* |
|
|
|
|
|
|
|
|
|
Common |
|
Camille Sebaaly(1) 14464 Gouin W, #B Montreal, Quebec Canada H9H 1B1 |
|
234,373 |
|
* |
|
|
|
|
|
|
|
|
|
Common |
|
All Officers and Directors As a Group (3 persons) |
|
34,812,313(2) |
|
74.7% |
|
|
|
|
|
|
|
|
|
* |
Less than 1% |
|
(1) |
Officer and Director of our Company. |
|
(2) |
Includes 17,109,194 shares held in the name of Advanomics Corporation and 850,000 shares of Series “A” Convertible Preferred Stock that is convertible into 17,000,000 shares of Common Stock held in the name of Advanomics Corporation. Dr. Slilaty is an officer, director and principal shareholder of Advanomics Corporation. |
Item 5.02 Departure of Directors or Certain Officers; Election of Directors: Appointment of Certain Officers; Compensatory Arrangements of Certain Officers.
Effective October 15, 2009, Ms. Lynn M. Vagi did resign her positions as President, Treasurer and as a director of our Company. There were no disagreements between Ms. Vagi and our Company regarding the operation, policies or practices of our Company. Simultaneous therewith, Dr. Steve N. Slilaty was appointed as our CEO and President, Mr. Michele Di Turi was appointed our Chief Operating Officer and Mr. Camille Sebaaly was appointed as our Chief Financial Officer and Secretary of our Company. Each of the aforesaid persons was also appointed as directors of our Company. They will hold such positions until the next annual meeting of our Board of Directors or shareholders, as applicable, their resignation, removal or death, whichever occurs first.
|
|
The following is a biographical summary of the business experience of our new management: |
Dr. Steve N. Slilaty, age 57, was appointed as our CEO, President and Chairman of our Board of Directors on October 15, 2009. In addition, since February 2002, Dr. Slilaty has been President and Chief Scientific Officer of Advanomics Corporation, Montreal, Canada, a privately held company engaged in the research, development and commercialization of drugs for the treatment of various forms of cancer. Advanomics Corporation is the third in a line of biotechnology companies that Dr. Slilaty founded and managed through their early and mid stages of development. The first, Quantum Biotechnologies Inc. later known as Qbiogene Inc., was founded in 1991 and grew to over $60 million in annual sales. Today, Qbiogene is a member of a family of companies owned by MP Biomedicals, one of the largest international suppliers of biotechnology reagents with a catalogue containing over 55,000 products. The second company which Dr. Slilaty founded, Genomics One Corporation, now known as Alert B&C Corporation, conducted an initial public offering (IPO) of its capital stock in 1999 and, on the basis of its ownership of Dr. Slilaty’s patented TrueBlue® Technology, Genomics One became one of the handful of participants in the Human Genome Project. Formerly a research team leader of the Biotechnology Research Institute, a division of the National Research Council of Canada, Dr. Slilaty also served as a consultant in a management and advisory capacity for a major Canadian biotechnology company between 1995 and 1997 during which time the company completed one of the largest biotechnology IPO‘s in Canada raising over $34 million. Dr. Slilaty received his Ph.D degree from the University of Arizona in 1983 and a Bachelor of Science degree from Cornell University in 1976. In addition, Dr. Slilaty holds a position as Adjunct Professor at Université du Québec in the Department of Microbiology and Biotechnology. He intends to devote approximately 50% of his time to our business affairs.
Michele Di Turi, age 32, was appointed as our Chief Operating Officer and a Director of our Company on October 15, 2009. Since November 2008, Mr. Di Turi has been President of Sunshine Bio Investments, Inc., a privately held Canadian corporation engaged in the sale of nonregulated biotechnology and medical products. Prior, from February 2003 through November 2008, he was employed by Mazda President, Inc., Montreal, Canada, as a sales representative and director of customer service. He intends to devote approximately 60% of his time to our business affairs.
Camille Sebaaly, age 50, was appointed as our Chief Financial Officer, Secretary and a Director of our Company on October 15, 2009. Since 2001, Mr. Sebaaly was self-employed as a business consultant, primarily in the biotechnology and biopharmaceutical sectors, as well as in the hydrogen generation and energy savings fields. He was a co-founder of Advanomics Corporation with Dr. Slilaty. He received a Bachelor of Science degree in electrical and computer engineering from the State University of New York at Buffalo in 1987. He intends to devote all of his time to our business affairs.
Item 7.01 Regulation FD Disclosure
Our Press Release relating to our acquisition described above is attached as Exhibit 99.1 and is hereby incorporated.
Item 9.01 Financial Statements and Exhibits
(a) Financial Statements. We intend to file an amendment to this report within seventy days from the date of this report that will include the audited financial statements of SBI from the date of inception (August 17, 2009) through September 30, 2009, along with an unaudited consolidated pro forma financial statement.
|
|
(b) |
Exhibits. |
|
|
No. |
Description |
|
|
10.1 |
Share Exchange Agreement with Sunshine Biopharma, Inc. |
|
|
99.1 |
Press Release dated October 20, 2009, announcing closing of transaction with |
Sunshine Biopharma, Inc.
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
|
Dated: October 19, 2009 |
SUNSHINE BIOPHARMA, INC. |
|
|
(Registrant) |
|
|
By:s/Steve N. Slilaty______________________ |
|
|
Dr. Steve N. Slilaty |
|
|
Chief Executive Officer |
