Attached files
| file | filename |
|---|---|
| 8-K - FORM 8-K - Riot Blockchain, Inc. | appy_8k-101509.htm |
| EX-99.1 - SHAREHOLDER LETTER - Riot Blockchain, Inc. | appy_8k-ex99x2.htm |
Exhibit 99.2

October
2009

2
Safe
Harbor Statement
Certain statements
made in this presentation include historical
information and forward looking actions that AspenBio Pharma
anticipates based on certain assumptions. These statements are
indicated by words such as “expect”, “anticipate”, “should” and similar
words are indicating uncertainty in facts, figures and outcomes. While
AspenBio Pharma believes that the expectations reflected in such
forward-looking statements are reasonable, it can give no assurance
that such statements will prove to be correct. The risks associated with
the company are detailed in the Company’s Annual Report on Form 10-K
for the year ended December 31, 2008 and other reports filed by the
Company with the Securities and Exchange Commission.
information and forward looking actions that AspenBio Pharma
anticipates based on certain assumptions. These statements are
indicated by words such as “expect”, “anticipate”, “should” and similar
words are indicating uncertainty in facts, figures and outcomes. While
AspenBio Pharma believes that the expectations reflected in such
forward-looking statements are reasonable, it can give no assurance
that such statements will prove to be correct. The risks associated with
the company are detailed in the Company’s Annual Report on Form 10-K
for the year ended December 31, 2008 and other reports filed by the
Company with the Securities and Exchange Commission.

3
Appendicitis
Test Platform
Test Platform
•First of
kind
•Blood
based
•Rapid
•IP
Protected
• Dedicated
Instrument
and Consumables
and Consumables
Reproductive
Science
•Animals of
economic
importance
importance
•IP
Protected
•Recombinant Protein
Drug
platform
platform
•Enhances
reproductive
efficiency
efficiency
AspenBio Pharma’s
mission is to
be a leader in the development
and commercialization of
innovative products that address
unmet diagnostic and therapeutic
needs in both Human and Animal
Health.
be a leader in the development
and commercialization of
innovative products that address
unmet diagnostic and therapeutic
needs in both Human and Animal
Health.
Our
Animal Health Division is
focused on therapeutic
proteins that support
reproductive efficiency in non
-companion animals.
focused on therapeutic
proteins that support
reproductive efficiency in non
-companion animals.

Human
Health
4
5
5
What
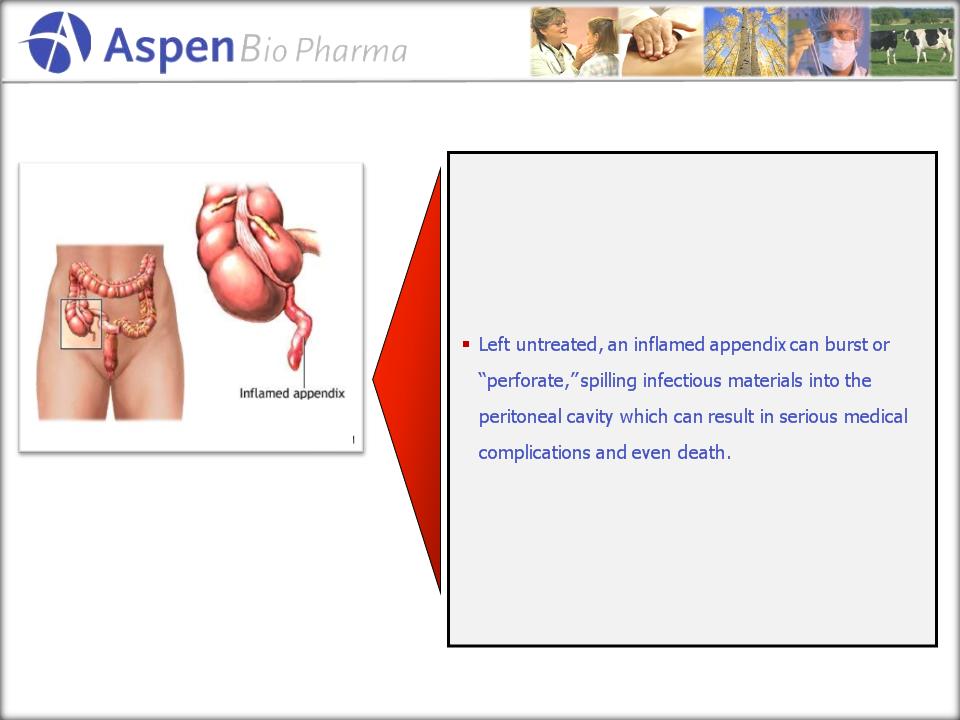
is Appendicitis?
§ Inflammation of the
appendix usually resulting from a
bacterial infection within the lumen.
bacterial infection within the lumen.
§ It is a
serious
medical emergency which
typically
requires surgery.
requires surgery.
§ Recent Good Morning
America story highlighted the
life threatening nature of appendicitis:
http://abcnews.go.com/video/playerIndex?id=8154374
life threatening nature of appendicitis:
http://abcnews.go.com/video/playerIndex?id=8154374
Appendicitis
is the Most Common Reason for Abdominal Surgery
Prevalence
Data
§ Effects
9% men and 7%
women in their lifetime.
women in their lifetime.
§ More
than 300K emergency
surgeries in US/year
surgeries in US/year

6
6
• Current
estimate ~320K appendectomies per year in
the US*
the US*
• Estimates
of up to 8-10% of surgeries remove a
normal appendix (~24,000)
normal appendix (~24,000)
• Avg.
18% of patients mis-diagnosed and sent home
with appendicitis**
with appendicitis**
• We
see ranges of 25%-30% of appendicitis cases not
diagnosed in time, resulting in perforated appendix &
emergency surgery
diagnosed in time, resulting in perforated appendix &
emergency surgery
• Gynecological
issues makes female diagnosis difficult
resulting in 2x as many appendectomies as men, with
~ 50% eventually confirmed NOT having appendicitis
resulting in 2x as many appendectomies as men, with
~ 50% eventually confirmed NOT having appendicitis
**Graff, et al,
study
Alvarado
Score
Symptoms
Score
Migratory right
iliac fossa pain
1
Nausea
/ Vomiting 1
Anorexia
1
Signs
Tenderness in right
iliac fossa
2
Rebound tenderness
in right iliac fossa 1
Elevated
temperature 1
Laboratory
findings
Elevated WBC 2
Increased
neutrophils 1
CT
Scan
ER…quick
decisions with imperfect information!
10M
patients/year enter ER
w/Abdominal
w/Abdominal
pain
*CDC
2006 data

Trends
in Appendicitis Management
7
1. Historically,
appendicitis was a clinical
diagnosis with a negative appendectomy
rate (NAR) of ~16%.
diagnosis with a negative appendectomy
rate (NAR) of ~16%.
2. In 1998, the use of
CT was incorporated
into the diagnostic algorithm.
into the diagnostic algorithm.
3. Use of CT grows
exponentially but
incremental benefits are minimal.
incremental benefits are minimal.
4. Risks and
limitations of CT identified;
however few alternatives exist for ED
physicians and surgeons.
however few alternatives exist for ED
physicians and surgeons.
1
2
3
4
Source: American
Journal of Emergency Medicine (2008) 26, 39-44
CT
being over-prescribed
because of the lack of an
effective “screening”
alternative.
because of the lack of an
effective “screening”
alternative.
Despite
Advances…
8
Cost
Cost
for abdominal
CT ranges from
CT ranges from
$200 -
$2000****
Safety
0.4% -
2% of all
cancers in the US will
be caused by CT***
cancers in the US will
be caused by CT***
…There
is a Need for a More Effective Screening Tool!
Sources:
*American
Journal of Emergency Medicine 2008; 26, 39-44
**Surgery
2008;144:276-82
***N
Engl J Med 2007;357:2277-84
****Emerg Radiol
2008;15:23-28 and Company estimates
Indiscriminant
Use
CT in
diagnosing
appendicitis is
overused with
diminishing
appendicitis is
overused with
diminishing
benefits
.**
CT
initially reduced
Negative
Appendectomy Rate,
but trend flattened in
recent years.
Negative
Appendectomy Rate,
but trend flattened in
recent years.
Our
Science
9
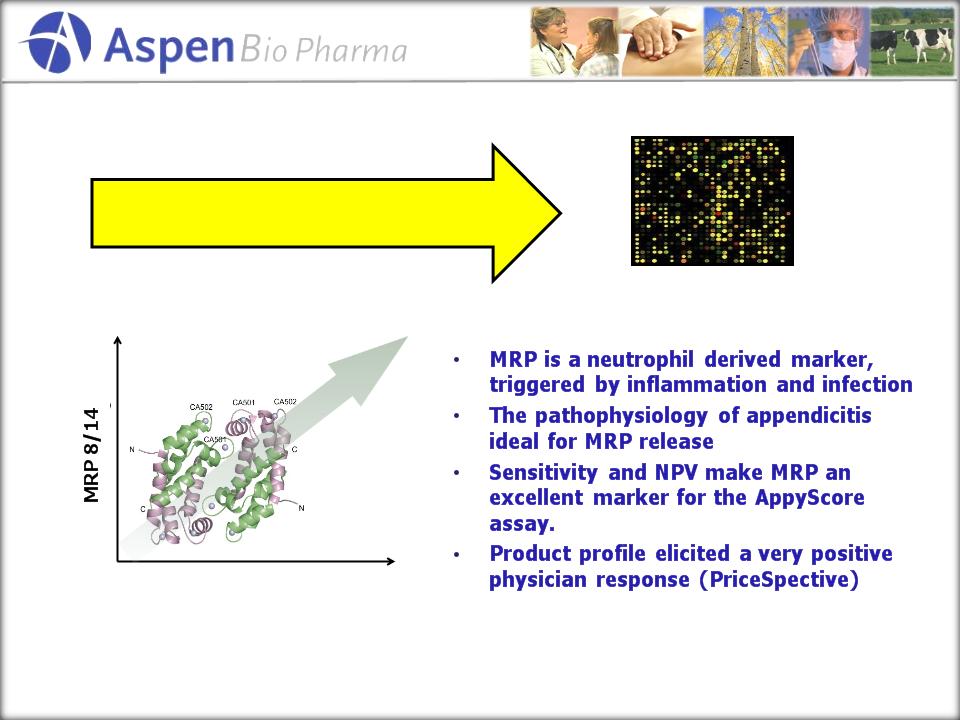
Appendicitis
Progression
Myeloid-related
protein
(MRP) 8/14
(MRP) 8/14
AspenBio
has patented this marker as a “aid in diagnosing Appendicitis”
AspenBio
did exhaustive proteomics differential
screens and identified >400 up-regulated proteins
in diseased appendix tissue.
screens and identified >400 up-regulated proteins
in diseased appendix tissue.
AppyScore
ELISA Assay
10
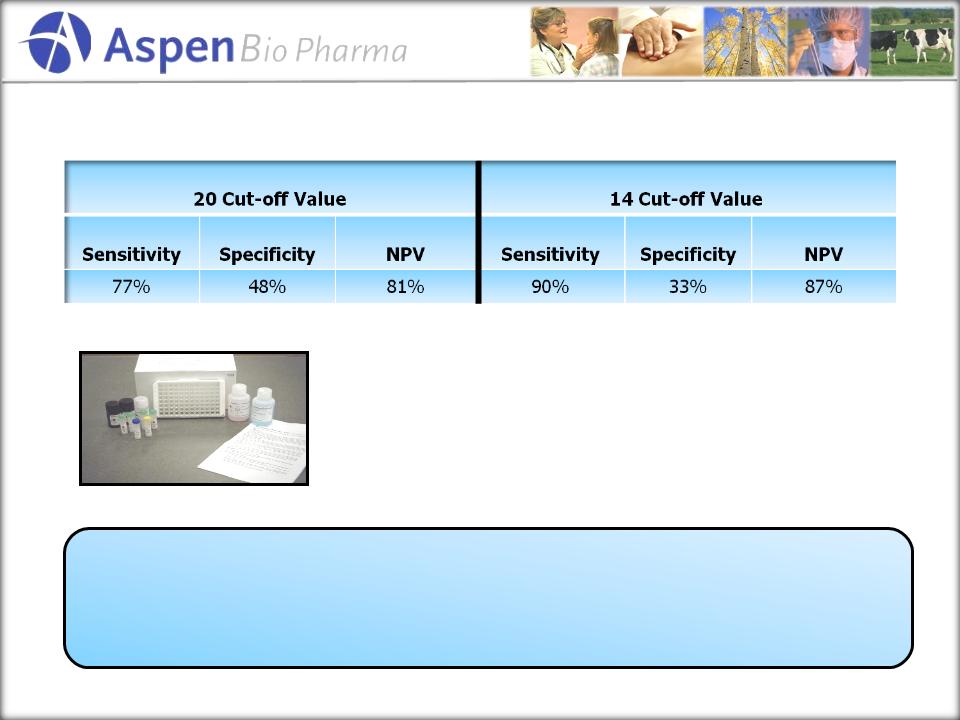
Analysis
of 15 other diagnostic tests
approved for the “aid in the diagnosis
of…”
approved for the “aid in the diagnosis
of…”
•Sensitivity
Ranges of 60-96%
•Specificity
Ranges of 32-100%
•NPV
Ranges of 80-96%
Our
primary research through PriceSpective, utilizing the
product specifications above, indicated strong acceptance
of the clinical utility of AppyScore.
product specifications above, indicated strong acceptance
of the clinical utility of AppyScore.
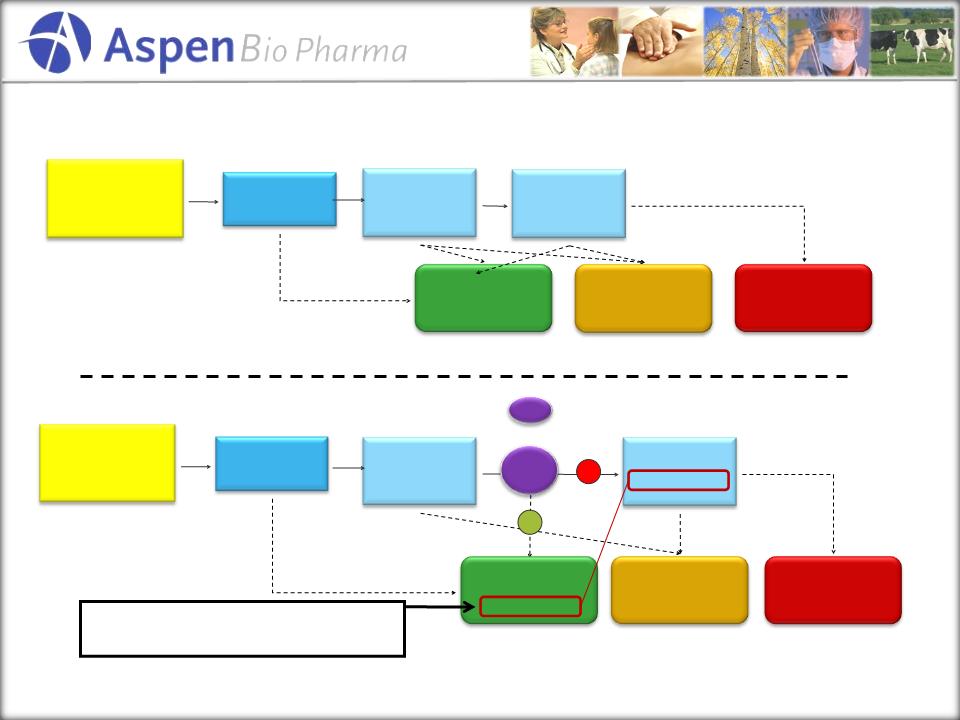
How
AppyScore Fits in the Workflow
11
Patient
experiencing acute
abdominal pain
experiencing acute
abdominal pain
Emergency
Room
Room
10.3
M Patients
X
AppyScore
Standard
Lab
Workup
Workup
5.6
M Patients
Abdominal
CT
1.5
M Patients
Appendectomies
320k
Patients
256
k CT’s
Other
Diagnosis
6.4M
Patients
1.2
M CT’s
X
+
Idiopathic
3.6M
Patients
0
CT’s
Source:
Calendar year 2005-2006 visits from the National Hospital Ambulatory Care Survey
(NHAMCS).
Counts
are annualized over the two-year period.
Patient
experiencing acute
abdominal pain
experiencing acute
abdominal pain
Emergency
Room
Room
10.3
M Patients
Standard
Lab
Workup
Workup
5.6
M Patients
Abdominal
CT
2.5
M Patients
Appendectomies
320k
Patients
256
k CT’s
Other
Diagnosis
6.4M
Patients
1.2
M CT’s
Idiopathic
3.6M
Patients
1.0
M CT’s
Current
Standard of Care
New
Standard of Care with AppyScore
Opportunity to take out significant
portion of these CT Scans!
portion of these CT Scans!
-
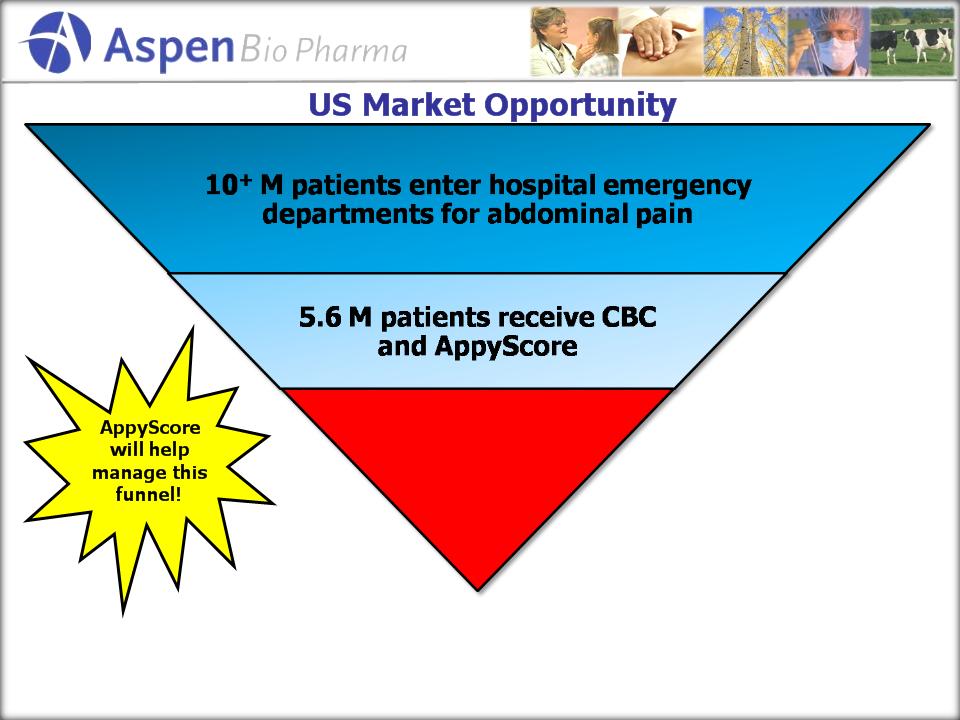
12
US
Market Estimates for Hospital Segment is 5.6 M Tests!
(Excludes
Primary Physician, OB/GYN, and Urgent Care Channels)
CT
scan given only to
patients who
need it
patients who
need it

Total
Available Market (TAM)Estimates
Based
on 5.6M Tests in the US and Global IVD Market Share (Kalorama)
5.6
3.3
1.4
2.4
12.7
13

14
Aspenbio
engaged
PriceSpective, experts in
biopharmaceutical pricing
and value capturing
strategies
PriceSpective, experts in
biopharmaceutical pricing
and value capturing
strategies
Utilizing the Appy
product
specifications PS conducted
32 interviews in the US and
15 in main European
countries
specifications PS conducted
32 interviews in the US and
15 in main European
countries
Extensive
“blind”
Interviews
“blind”
Interviews
Interviewees included
many highly published key opinion leaders in their
respective area of expertise including appendicitis management.
respective area of expertise including appendicitis management.

Market
Research in the US and
EU Confirms the Need for AppyScore
EU Confirms the Need for AppyScore
15
“There
are problems with the whole system; CT scan with contrast is taking too long; it
is a 5
hour ordeal. CT without contrast is much faster, but the sensitivity is low; most of
radiologists don’t do that. There is a need for a more specific rule in/out test.”
hour ordeal. CT without contrast is much faster, but the sensitivity is low; most of
radiologists don’t do that. There is a need for a more specific rule in/out test.”
- ER
Physician
“The
current unmet need is in accuracy of diagnosis; it takes time to perform CT scan
and
the cost of CT is high.”
the cost of CT is high.”
-
Surgeon
“Principal
advantages of [test] will be better discrimination of patients who require
surgical
intervention from those who do not; reduced healthcare costs in achieving this better
discrimination from fewer unnecessary surgeries, and less chance of medical errors.”
intervention from those who do not; reduced healthcare costs in achieving this better
discrimination from fewer unnecessary surgeries, and less chance of medical errors.”
- Lab
Director
“If
they are accepted [by physicians] as necessary tests, we will cover
them.”
-
Payer
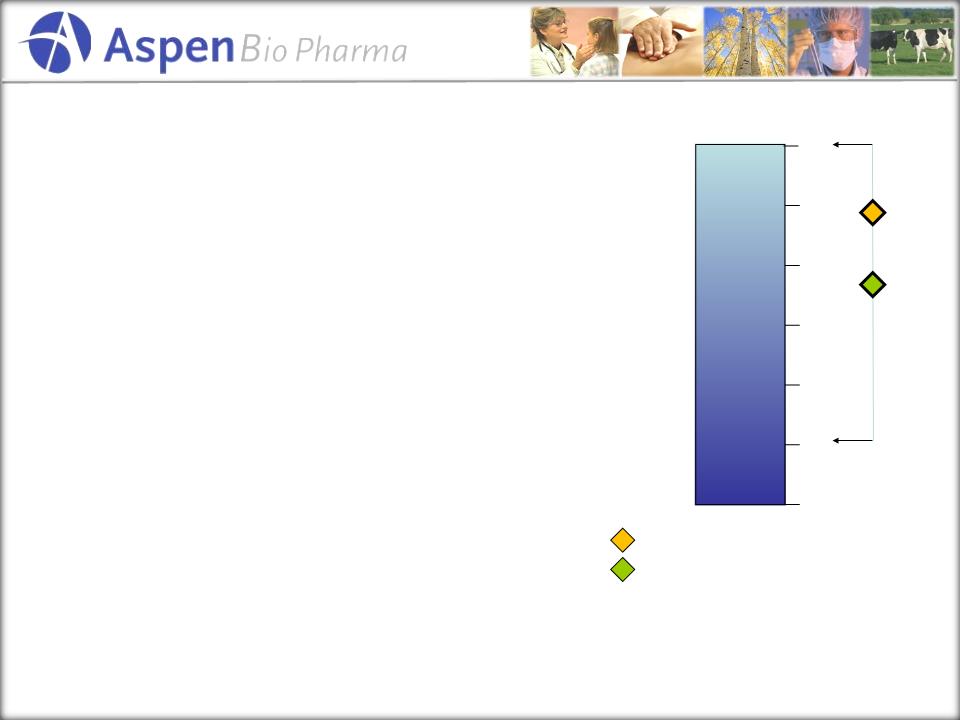
Findings
from PriceSpective’s Work
• Physician value
rating range from 2.5 to 7, with
an average of 5.7 on the 1 to 7 scale
an average of 5.7 on the 1 to 7 scale
• The high ratings
are based on convenience, a blood
-based assay and clinical specificity
-based assay and clinical specificity
• The two lower
ratings, were based on the desire for
more information on patient demographics
more information on patient demographics
• Physicians say they
would initially use it in
patients that are more difficult to diagnose,
…young children, women, and the elderly
patients that are more difficult to diagnose,
…young children, women, and the elderly
• Payers’ value
rating ranged from 2 to 6.5 with an
average of 4.5 on a 1 to 7 scale
average of 4.5 on a 1 to 7 scale
• Unprompted,
payers
state that they would
not manage the use of the AppyScore™
not manage the use of the AppyScore™
• It would be
included in bundled DRG payments
• Managing emergency
care utilization is difficult
because payers are reluctant to interfere in life-
threatening situations
because payers are reluctant to interfere in life-
threatening situations
Very
Valuable
Valuable
Not
Valuable
1
7
Physician Average
(5.7)
4
Please
rank the value of this product on a scale of 1-7, where 1 is
not valuable at all and 7 is very valuable.
not valuable at all and 7 is very valuable.
Max
Min
Payer
Average (4.5)
“AppyScore
…second highest initial product approval we have ever seen!”
16
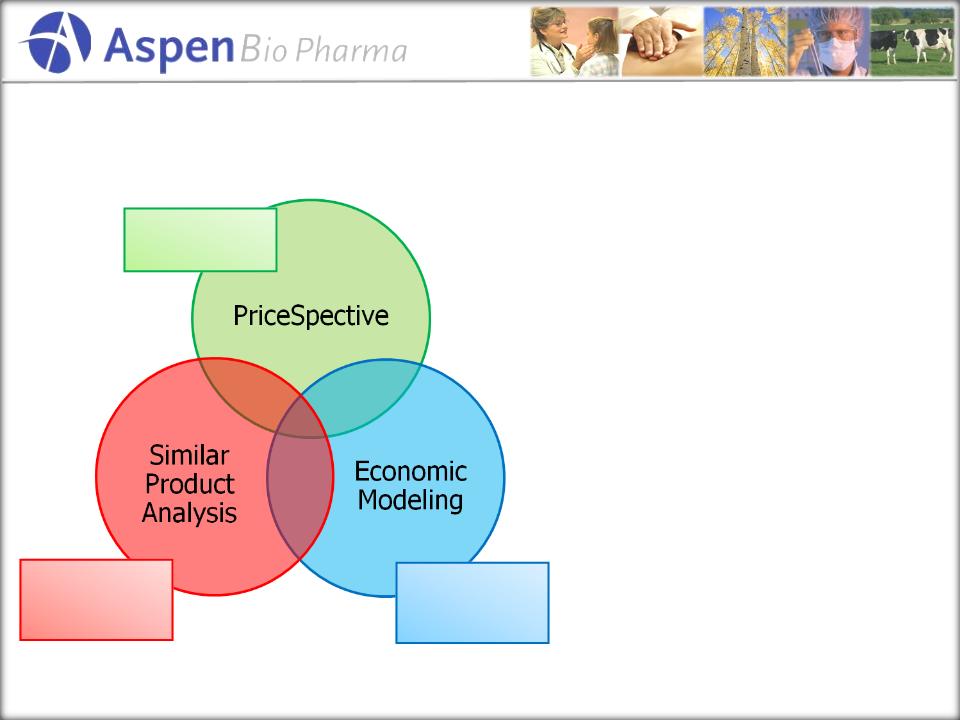
Pricing…
a triangulation approach
17
1.
PriceSpective
Analysis
supports AppyScore should
follow “pharmaceutical”
versus IVD model.
supports AppyScore should
follow “pharmaceutical”
versus IVD model.
2.
Our
own economic modeling
on value to the health care
supports this theory.
on value to the health care
supports this theory.
3.
As
the first, blood based
test, with a dedicated
instrument and cassette,
specifically FDA approved as
an “aid in the diagnosis of
appendicitis,” we believe the
product will support premium
pricing.
test, with a dedicated
instrument and cassette,
specifically FDA approved as
an “aid in the diagnosis of
appendicitis,” we believe the
product will support premium
pricing.
Independent,
Expert, value-
based analysis
Expert, value-
based analysis
Analysis
of
Products with
similar value
characteristic
Products with
similar value
characteristic
Value
based on
savings to the
healthcare cost
structure
savings to the
healthcare cost
structure

Policy
Makers &
Influencers
Influencers
Physician
Community
Community
Payers
Patients
18
•FDA
•MAB
•Publication
•Emergency
Physicians
Physicians
•Surgeons
•Pricing
Study
•Pricing
Study
“Build
a Company”
• Market own
brand
• Build/Lease sales
force
License
• Assay for adoption
on
existing platforms
existing platforms
Sales
& Distribution
Partnership
Partnership
• Marketing
partnerships
19
Commercial
Options
Value
Drivers
Support
Option A
Drivers
Support
Option A
A
B
C
• Target Market
easily
reachable with
moderate sized sales force.
moderate sized sales force.
• Top 30
US metropolitan areas
represent >40% of US Market.
represent >40% of US Market.

Regulatory
Update
• 800
patient Pivotal trial completed in Dec 2008. (Utilizing ELISA
format)
• Trial
Demonstrated:
• Fundamental
analytical performance to be robust and repeatable.
• Clinical
utility that supports the intended use claim
• 510(k)
filed in June 2009
• Probable
label intended use: “to be used as an aid to rule out the diagnosis of
appendicitis”
• FDA
response received August, 2009: Supplemental trial underway
• Internal
schedule for clearance is late Q1 - early Q2 2010 (assuming de
novo classification
of device)
of device)
• Instrument/Cassette
trials will commence in Q2 2010, with ELISA as predicate device
• AppyScore
Instrument/Cassette 510(k) to be filed late 2010, with commercialization
to
follow FDA clearance.
follow FDA clearance.
20
21
21
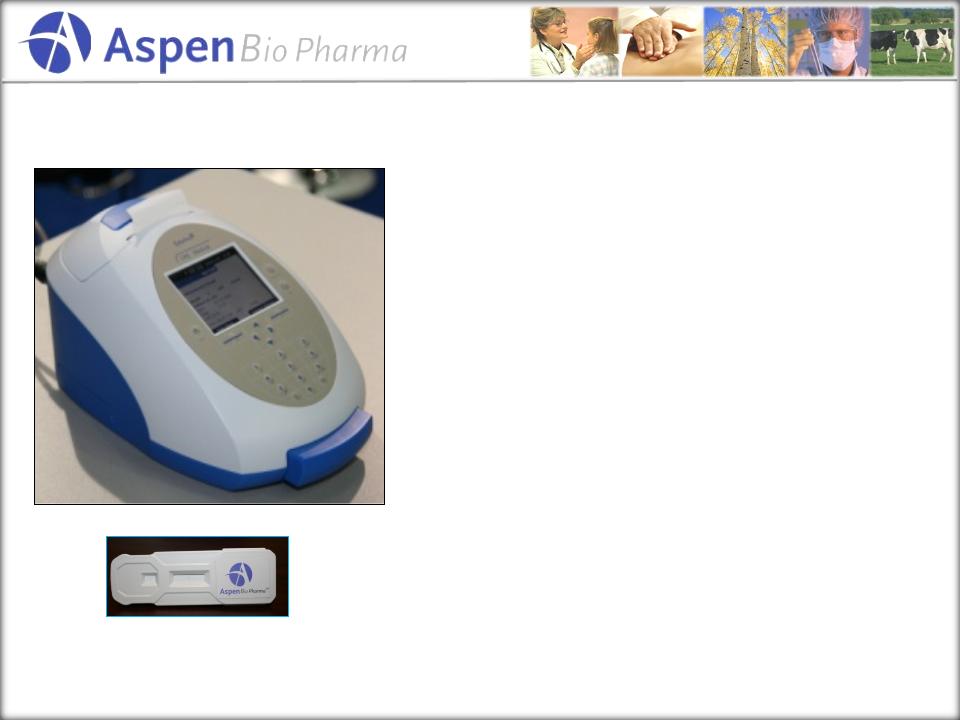
Product
Profile
• Fully-integrated
assay system for
near patient testing
near patient testing
• Results
in ~15 minutes
• Industrial
product designed to
significantly reduce operator
dependence
significantly reduce operator
dependence
• Hospital
LIS interface
• Feasibility
completed and final specs
being set
being set
• Rapid
assay clinical trial estimated to
commence Q1 2010
commence Q1 2010

Getting
to Market…Key Milestones
22
Submit
510(k)
to FDA for ELISA
to FDA for ELISA
Expected
FDA
clearance on
ELISA
clearance on
ELISA
Q2
09
Q4
09
Late
Q1-
Early Q2 '10
Early Q2 '10
Commence
rapid
assay clinical
trial
assay clinical
trial
Q1
10
Complete
rapid
assay product
development
assay product
development
Submit
510(k)
to FDA for rapid
assay
to FDA for rapid
assay
Q3
10
Q1
2011
Rapid
assay
clearance and
product launch
clearance and
product launch
Completed

Veterinary
Science
23
Milk
Production
24
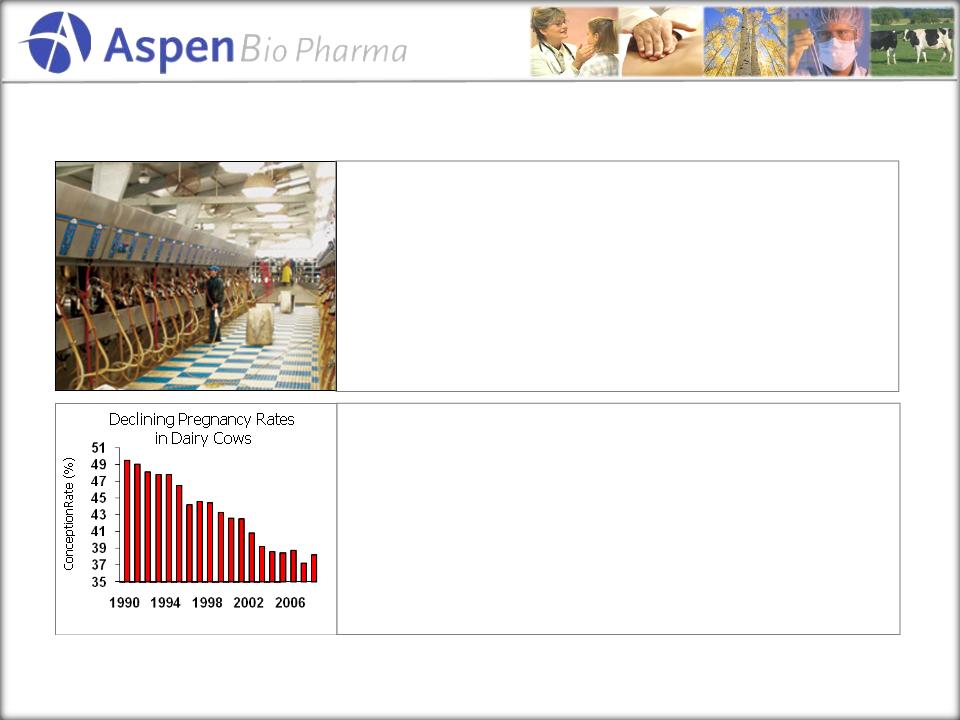
Significantly
impacts economics for producers!
Basic
Facts:
§ There
are ~ 9 Million dairy cows in the U.S. (125 million
worldwide)
worldwide)
§ Their
pregnancy optimizes milk production
§ Breeding
is costly: Every year, dairy cows receive ~20 million
AIs (Artificial Inseminations) at $42 - $105 each
AIs (Artificial Inseminations) at $42 - $105 each
Problem:
§ Intensive
dairy herd management has negatively impacted
pregnancy rates
pregnancy rates
§ Pregnancy
rates have dropped from 80% to mid 30’s per AI
§ This
wastes critical time, money, and productivity
§ Cost
of culling cows due to poor reproduction: $1.2B annually
(est.)
(est.)
Source: DHI-Provo

Animal
Health Reproductive Science
25
•Exclusive
license of patented technology
•Single-chain
recombinant gonadotropins
•Demonstrated
highly robust stability
•Delivers
precise activity for improved pregnancy maintenance
AspenBio’s
platform technology uses a proprietary
linker that enables recombinant production of single
chain proteins that are replacements for depleted
gonadotropins LH and FSH.
linker that enables recombinant production of single
chain proteins that are replacements for depleted
gonadotropins LH and FSH.

Strategic
Partnership
• License
and Development relationship focused on product for bovine
reproduction
• Collaboration
covers bLH and bFSH which has a US market potential of $230M
• Novartis
and Aspen share development expenses 65%/35% respectively
26
A
Perfect Partner for Advancing Aspen’s Single Chain Technology to
Market
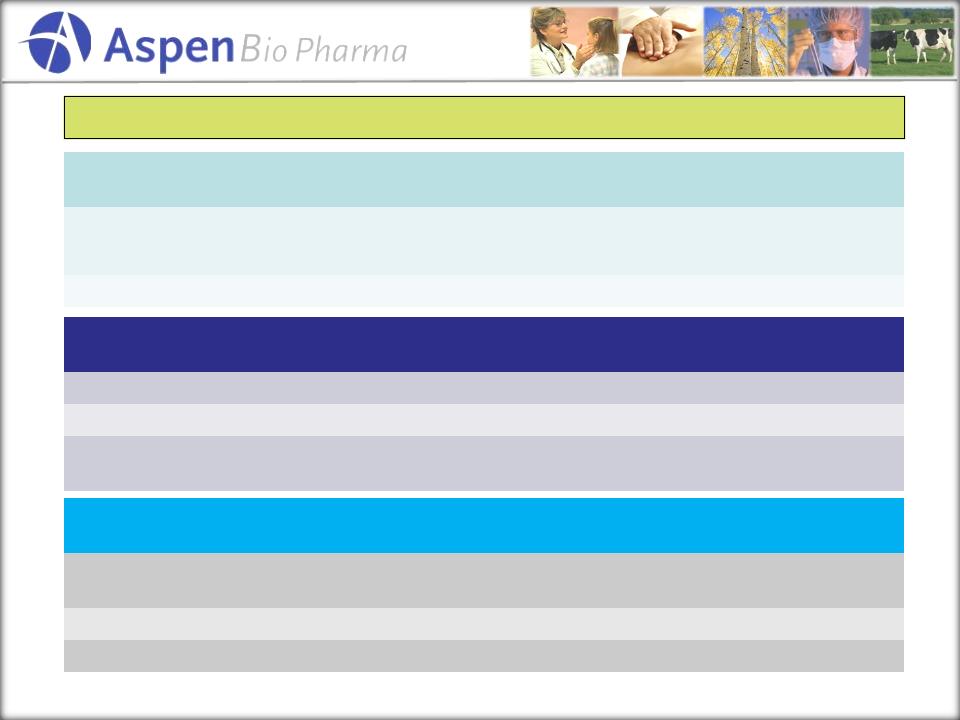
Animal
Health Portfolio
|
Products
in Partnership
|
Solves
What?
|
|
US
Revenue
Potential |
|
BoviPure-LH
|
Ovulation,
dairy pregnancy
rates |
|
$100-200
M
|
|
BoviPure-FSH
|
Superovulation
|
|
$20-30
M
|
|
In
Development
|
Solves
What?
|
|
US
Revenue
Potential |
|
EquiPure-LH
|
Timed
Breeding
|
|
TBD
|
|
EquiPure-FSH
|
Superovulation,
transition
|
|
TBD
|
|
OviPure-LH,
FSH
|
Off-season
Breeding
Superovulation
|
|
TBD
|
|
Early
Development
|
Solves
What?
|
|
US
Revenue
Potential |
|
rhCG/rPMSG
for Swine
|
Timed
Breeding, cycling in
Swine |
|
TBD
|
|
rhCG
|
Ovulation-all
species
|
|
TBD
|
|
rPMSG
|
Timed
Breeding, cycling
|
|
TBD
|
27

28
Key
Statistics: APPY
(NASDAQ
CM)
Mrq
estimates as of June 2009
|
Stock
Price (10/9/09)
|
$2.01
|
|
Avg. Daily
Vol. (3mo.)
|
137,000
|
|
Shares
Out. (mrq)
|
32.2M
|
|
Shares
Out. (*)
|
37.4M
|
|
Fully-diluted
(mrq)
|
37.0M
|
|
Public Float,
est.
(mrq)
|
29.4M
|
|
Institutional
Holdings,
est. |
40%
|
|
Insider
Holdings,
est.
|
23%
|
|
Cash
& Equiv. (est.
10-09*)
|
~$18M
|
|
Total Assets
(mrq)
|
$18.5M
|
|
Total
Liabilities (mrq)
|
$6.0M
|
|
|
|
|
Monthly
Burn-Rate
(avg.)
|
~$880K
|
mrq (most recent
quarter)
* -
Includes recently announced offering
of 5,155,000 common shares
of 5,155,000 common shares

29
Daryl
J. Faulkner
Chief Executive Officer, Executive Chairman and Director
Daryl Faulkner has more than 25 years experience in developing and commercializing medical devices, drug and drug delivery systems, life science research tools
and molecular diagnostics. He most recently served as president, CEO and member of the board of directors of Digene Corporation, acquired by Qiagen in July
2008. He has continued to serve as a consultant to Qiagen. Prior to joining Digene, Mr. Faulkner spent eight years with Invitrogen (now merged as Life
Technologies Corp., a Nasdaq-traded company) . Mr. Faulkner’s career also includes15 years with the Fortune 100 company Abbott Laboratories. Mr. Faulkner
currently serves as a member of the board of directors of Osmetech, an emerging molecular diagnostics company.
Daryl Faulkner has more than 25 years experience in developing and commercializing medical devices, drug and drug delivery systems, life science research tools
and molecular diagnostics. He most recently served as president, CEO and member of the board of directors of Digene Corporation, acquired by Qiagen in July
2008. He has continued to serve as a consultant to Qiagen. Prior to joining Digene, Mr. Faulkner spent eight years with Invitrogen (now merged as Life
Technologies Corp., a Nasdaq-traded company) . Mr. Faulkner’s career also includes15 years with the Fortune 100 company Abbott Laboratories. Mr. Faulkner
currently serves as a member of the board of directors of Osmetech, an emerging molecular diagnostics company.
Greg
Bennett Vice
President of Product Development and Manufacturing
Mr. Bennett brings to AspenBio Pharma 25 years of experience in product design and development focused in cassette and instrument test formats, including point-of-
care (“POC”) and home test products. He recently served as general manager of Cholestech’s operations following the company’s acquisition by Inverness Medical
Innovations, Inc., in 2007. At Cholestech, Mr. Bennett served as vice president of research and development where he was responsible for the development and
launch of its cholesterol instrument and cartridge system for POC use. Prior to his six years with Cholestech / Inverness, Bennett spent 12 years with LifeScan, Inc., a
Johnson & Johnson Company, where he served in increasing levels of responsibility and lastly as director of process development engineering. At LifeScan, he led the
group responsible for the process development, scale-up and commercialization of several blood glucose monitoring devices. Bennett earned his B.S. in Mechanical
Engineering from the University of Wisconsin, and has received specialized training in Process Excellence/Six Sigma and Stanford Executive Training - Corporate Finance
and Portfolio Management.
Mr. Bennett brings to AspenBio Pharma 25 years of experience in product design and development focused in cassette and instrument test formats, including point-of-
care (“POC”) and home test products. He recently served as general manager of Cholestech’s operations following the company’s acquisition by Inverness Medical
Innovations, Inc., in 2007. At Cholestech, Mr. Bennett served as vice president of research and development where he was responsible for the development and
launch of its cholesterol instrument and cartridge system for POC use. Prior to his six years with Cholestech / Inverness, Bennett spent 12 years with LifeScan, Inc., a
Johnson & Johnson Company, where he served in increasing levels of responsibility and lastly as director of process development engineering. At LifeScan, he led the
group responsible for the process development, scale-up and commercialization of several blood glucose monitoring devices. Bennett earned his B.S. in Mechanical
Engineering from the University of Wisconsin, and has received specialized training in Process Excellence/Six Sigma and Stanford Executive Training - Corporate Finance
and Portfolio Management.
Robert
F. Caspari, MD
Chief Operating Officer / Chief Medical Officer
Robert Caspari brings to AspenBio Pharma more than 25 years of experience in drug and diagnostic product development and commercialization. He most recently
served as CEO of Living Cell Technologies, a publicly traded biotech company focused on cellular therapy for Type I diabetes and neurological disorders. He was
previously president and CEO of Aurogen, a privately held biotech company involved in drug development for neurological disorders. Dr. Caspari has also served as
senior vice president of commercial operations and medical affairs at Myogen (now a unit of Gilead Sciences, traded on the Nasdaq), and as vice president and
general manager of biopharmaceuticals at Novo Nordisk Pharmaceuticals. Dr. Caspari received a B.A. in psychology from UCLA and his medical degree from
Georgetown University.
Robert Caspari brings to AspenBio Pharma more than 25 years of experience in drug and diagnostic product development and commercialization. He most recently
served as CEO of Living Cell Technologies, a publicly traded biotech company focused on cellular therapy for Type I diabetes and neurological disorders. He was
previously president and CEO of Aurogen, a privately held biotech company involved in drug development for neurological disorders. Dr. Caspari has also served as
senior vice president of commercial operations and medical affairs at Myogen (now a unit of Gilead Sciences, traded on the Nasdaq), and as vice president and
general manager of biopharmaceuticals at Novo Nordisk Pharmaceuticals. Dr. Caspari received a B.A. in psychology from UCLA and his medical degree from
Georgetown University.
Jeffrey
G. McGonegal
Chief Financial Officer
Jeffrey McGonegal joined AspenBio Pharma as CFO in 2003. He has more than 30 years experience in accounting and developing public companies. Mr. McGonegal
serves in limited capacities both as Senior Vice President — Finance of Cambridge Holdings, Ltd., a holding company with limited activities and as Chief Financial Officer
of PepperBall Technologies, Inc. For 23 years he was in accounting with BDO Seidman LLP and his last position with that firm was managing partner of the Denver,
Colorado office. Mr. McGonegal graduated from Florida State University with a B.A. in accounting
Jeffrey McGonegal joined AspenBio Pharma as CFO in 2003. He has more than 30 years experience in accounting and developing public companies. Mr. McGonegal
serves in limited capacities both as Senior Vice President — Finance of Cambridge Holdings, Ltd., a holding company with limited activities and as Chief Financial Officer
of PepperBall Technologies, Inc. For 23 years he was in accounting with BDO Seidman LLP and his last position with that firm was managing partner of the Denver,
Colorado office. Mr. McGonegal graduated from Florida State University with a B.A. in accounting
Gregory
Pusey Vice
Chairman / Vice President Investor Relations
Gregory Pusey became a director of AspenBio Pharma, Inc. in February 2002, Chairman in May 2003 and in January 2009 became the Vice Chairman , a newly created
position. Mr. Pusey has been helping develop and advance small public companies for approximately 30 years. Mr. Pusey is a director of PepperBall Technologies, Inc.,
a publicly held provider of non-lethal technology. Since 1988, Mr. Pusey has been the President and a director of Cambridge Holdings, Ltd. which has limited activities.
Mr. Pusey is secretary and a director of Bactolac Pharmaceutical, Inc., a privately held company engaged in manufacturing and marketing of vitamins and nutritional
supplements. Mr. Pusey graduated from Boston College with a BS degree in finance.
Gregory Pusey became a director of AspenBio Pharma, Inc. in February 2002, Chairman in May 2003 and in January 2009 became the Vice Chairman , a newly created
position. Mr. Pusey has been helping develop and advance small public companies for approximately 30 years. Mr. Pusey is a director of PepperBall Technologies, Inc.,
a publicly held provider of non-lethal technology. Since 1988, Mr. Pusey has been the President and a director of Cambridge Holdings, Ltd. which has limited activities.
Mr. Pusey is secretary and a director of Bactolac Pharmaceutical, Inc., a privately held company engaged in manufacturing and marketing of vitamins and nutritional
supplements. Mr. Pusey graduated from Boston College with a BS degree in finance.
Mark
Colgin, PhD
Chief Scientific Officer
Mark Colgin was appointed Chief Scientific Officer of the Company in February 2009. Dr. Colgin joined the Company in September 2000 and served as Director of
Recombinant Technology until he was promoted to Chief Scientist in January 2003. Prior to joining the Company, his areas of research included the characterization
and artificial synthesis of spider silk proteins, regulation of gene expression, neurovirology and gene delivery systems. Dr. Colgin received a B.S. in Biochemistry and a
Ph.D. in Molecular Biology from the University of Wyoming.
Mark Colgin was appointed Chief Scientific Officer of the Company in February 2009. Dr. Colgin joined the Company in September 2000 and served as Director of
Recombinant Technology until he was promoted to Chief Scientist in January 2003. Prior to joining the Company, his areas of research included the characterization
and artificial synthesis of spider silk proteins, regulation of gene expression, neurovirology and gene delivery systems. Dr. Colgin received a B.S. in Biochemistry and a
Ph.D. in Molecular Biology from the University of Wyoming.
30
Newly
Formed Medical Advisory Board
David
Flum, MD, MPH is a leading
gastrointestinal surgeon and outcomes researcher at the University of
Washington. He holds the rank of
Professor in the Schools of Medicine and Public Health and serves as the Director of the Surgical Outcomes Research Center
(SORCE) at the University of Washington. He has a Masters in Public Health in the field of health services research. Dr. Flum serves as
Medical Director of the Surgical Care and Outcomes Assessment Program (SCOAP), a quality of care improvement program providing hospital-
specific data feedback and best practices regarding processes of care and outcomes across the Pacific Northwest. He is also one of the Principal
Investigators of the Longitudinal Assessment of Bariatric Surgery (LABS) study-the first NIH-funded study in bariatric surgery aimed at addressing
fundamental issues in the field. He is the contributing editor for surgery at the Journal of the American Medical Association, serves on the
editorial board of the journal Surgery for Obesity and Related Disease, and is a member of the Executive Leadership of the American College of
Surgeons Research Committee.
Professor in the Schools of Medicine and Public Health and serves as the Director of the Surgical Outcomes Research Center
(SORCE) at the University of Washington. He has a Masters in Public Health in the field of health services research. Dr. Flum serves as
Medical Director of the Surgical Care and Outcomes Assessment Program (SCOAP), a quality of care improvement program providing hospital-
specific data feedback and best practices regarding processes of care and outcomes across the Pacific Northwest. He is also one of the Principal
Investigators of the Longitudinal Assessment of Bariatric Surgery (LABS) study-the first NIH-funded study in bariatric surgery aimed at addressing
fundamental issues in the field. He is the contributing editor for surgery at the Journal of the American Medical Association, serves on the
editorial board of the journal Surgery for Obesity and Related Disease, and is a member of the Executive Leadership of the American College of
Surgeons Research Committee.
Douglas
K. Owens, MD, MS
is a
general
internist and a Professor of Medicine and of Health Research and Policy at
Stanford
University, where he directs the Program on Clinical Decision Making and Guideline Development at the Center for Primary Care and Outcomes
Research (PCOR). Dr. Owens also directs the Stanford University-UCSF Evidence-Based Practice Center funded by the Agency for Healthcare
Research and Quality (AHRQ). Dr. Owens’ research interests include diagnostic test evaluation, evidence synthesis, technology assessment, cost-
effectiveness analysis and guideline development. Dr. Owens has been principal investigator on grants funded by the National Institutes of
Health, the Agency for Healthcare Research and Quality, and the Centers for Disease Control and Prevention (CDC), among others. From 2005 to
2009, Dr. Owens was Chair of the Clinical Efficacy Assessment Subcommittee (CEAS) of the American College of Physicians (ACP), which
develops clinical practice guidelines for the ACP. Dr. Owens is a past President of the Society for Medical Decision Making. He was elected to the
American Society for Clinical Investigation (ASCI), and the Association of American Physicians (AAP), societies that recognize excellence in clinical
research.
University, where he directs the Program on Clinical Decision Making and Guideline Development at the Center for Primary Care and Outcomes
Research (PCOR). Dr. Owens also directs the Stanford University-UCSF Evidence-Based Practice Center funded by the Agency for Healthcare
Research and Quality (AHRQ). Dr. Owens’ research interests include diagnostic test evaluation, evidence synthesis, technology assessment, cost-
effectiveness analysis and guideline development. Dr. Owens has been principal investigator on grants funded by the National Institutes of
Health, the Agency for Healthcare Research and Quality, and the Centers for Disease Control and Prevention (CDC), among others. From 2005 to
2009, Dr. Owens was Chair of the Clinical Efficacy Assessment Subcommittee (CEAS) of the American College of Physicians (ACP), which
develops clinical practice guidelines for the ACP. Dr. Owens is a past President of the Society for Medical Decision Making. He was elected to the
American Society for Clinical Investigation (ASCI), and the Association of American Physicians (AAP), societies that recognize excellence in clinical
research.
David
A. Talan, MD, FACEP, FIDSA is
Chairman of the Department of Emergency Medicine and faculty in the Division
of
Infectious Diseases at Olive View-UCLA Medical Center, and Professor of Medicine in Residence at UCLA School of Medicine. He is a Fellow of
the American College of Emergency Physicians and the Infectious Diseases Society of America, and a member of the Society for Academic
Emergency Medicine and the American Society for Microbiology. Dr. Talan received his medical degree from the University of Illinois Medical
College in Chicago. He completed his residencies in Internal and Emergency Medicine and fellowship in Infectious Diseases at the University of
California Los Angeles (UCLA) and its associated medical centers. He is board certified in Internal Medicine, Emergency Medicine, and Infectious
Diseases. Dr. Talan serves on the editorial boards of the Annals of Emergency Medicine, Emergency Medicine News, and Pediatric Emergency
Care and is a reviewer for Clinical Infectious Diseases, Journal of the American Medical Association, and The Medical Letter.
Infectious Diseases at Olive View-UCLA Medical Center, and Professor of Medicine in Residence at UCLA School of Medicine. He is a Fellow of
the American College of Emergency Physicians and the Infectious Diseases Society of America, and a member of the Society for Academic
Emergency Medicine and the American Society for Microbiology. Dr. Talan received his medical degree from the University of Illinois Medical
College in Chicago. He completed his residencies in Internal and Emergency Medicine and fellowship in Infectious Diseases at the University of
California Los Angeles (UCLA) and its associated medical centers. He is board certified in Internal Medicine, Emergency Medicine, and Infectious
Diseases. Dr. Talan serves on the editorial boards of the Annals of Emergency Medicine, Emergency Medicine News, and Pediatric Emergency
Care and is a reviewer for Clinical Infectious Diseases, Journal of the American Medical Association, and The Medical Letter.
Steven
E. Wolf, MD
is the
Bob
and Betty
Kelso Distinguished Chair in Burn and Trauma Surgery, Vice Chairman for Research
and
Professor in the Department of Surgery at the University of Texas Health Science Center at San Antonio (UTHSCSA). He serves as
Chair for the Institutional Review Board UTHSCSA. He is Burn Surgeon and Chief of Clinical Research at the United States Army Institute of
Surgical Research at Fort Sam Houston and Pediatric Burn Program Director at University Hospital. Dr. Wolf received his medical degree from
UTMB Galveston and completed his post graduate training at the University of Missouri-Kansas City and at Shriners Hospital for Children in
Galveston. Dr. Wolf has previously held the position of Director of the US Army Institute of Surgical Research Burn Center at Brooke Army
Medical Center and Assistant Chief of Staff at Shriners Hospital for Children. He has authored over 150 peer reviewed publications and is
nationally and internationally recognized for his work in burn and trauma care and regenerative medicine. Dr. Wolf received the 2009 Health
Care Heroes Outstanding Physician Award. Dr. Wolf is Editor-in-Chief of Burns and on the editorial boards of Journal of Burn Care and
Rehabilitation and Surgery News.
Professor in the Department of Surgery at the University of Texas Health Science Center at San Antonio (UTHSCSA). He serves as
Chair for the Institutional Review Board UTHSCSA. He is Burn Surgeon and Chief of Clinical Research at the United States Army Institute of
Surgical Research at Fort Sam Houston and Pediatric Burn Program Director at University Hospital. Dr. Wolf received his medical degree from
UTMB Galveston and completed his post graduate training at the University of Missouri-Kansas City and at Shriners Hospital for Children in
Galveston. Dr. Wolf has previously held the position of Director of the US Army Institute of Surgical Research Burn Center at Brooke Army
Medical Center and Assistant Chief of Staff at Shriners Hospital for Children. He has authored over 150 peer reviewed publications and is
nationally and internationally recognized for his work in burn and trauma care and regenerative medicine. Dr. Wolf received the 2009 Health
Care Heroes Outstanding Physician Award. Dr. Wolf is Editor-in-Chief of Burns and on the editorial boards of Journal of Burn Care and
Rehabilitation and Surgery News.

Secured
Industry Leading Expertise
31
Committed
to Success!
•Regulatory
Strategy
and Execution
and Execution
•Pricing
and
Reimbursement
Reimbursement
•Global
Market Analysis
•Market
Strategies

32
Key
Milestones
• Finalize
product specs for Instrument
and Cassette product
and Cassette product
• Finalize
Manufacturing Agreements
• Complete
Supplemental Study and
advance response to FDA
advance response to FDA
• Finalize
marketing/commercial plans
• Determine
best source of funds
• Advance
Animal Business Opportunity
• Achieve
FDA clearance ELISA
• Commence
Device/Cassette
Pivotal Trial
Pivotal Trial
• File
FDA 510K for
Device/Cassette
Device/Cassette
• Determine
final pricing and
reimbursement strategy
reimbursement strategy
• Build
Commercial Capability
Balance
‘09
Next
Year…
Executing
towards commercialization!

“AspenBio…a
small company with BIG opportunities”
