Attached files
| file | filename |
|---|---|
| EX-23.1 - Inspyr Therapeutics, Inc. | v162316_ex23-1.htm |
| EX-5.01 - Inspyr Therapeutics, Inc. | v162316_ex5-01.htm |
As
filed with the Securities and Exchange Commission on October 15,
2009
Registration
No. [________]
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington,
D.C. 20549
FORM
S-1
REGISTRATION
STATEMENT UNDER THE SECURITIES ACT OF 1933
GENSPERA,
INC.
(Exact
name of registrant as specified in its charter)
|
Delaware
|
2834
|
20-0438951
|
||
|
(State or jurisdiction of
incorporation or organization)
|
(Primary Standard Industrial
Classification Code Number)
|
(I.R.S. Employer Identification No.)
|
2511
N Loop 1604 W, Suite 204
San
Antonio, TX 78258
(210)
479-8112
FAX
(210) 479-8113
(Address,
including zip code, and telephone number,
including
area code, of registrant’s principal executive offices)
Agent
for Service:
National
Corporate Research
800
Brazos St., Suite 400
Austin,
TX 78701
800-345-4647
(Name,
address, including zip code, and telephone number,
including
area code, of agent for service)
Copy
to:
Raul
Silvestre
Silvestre
Law Group, P.C.
31200
Via Colinas, Suite 200
Westlake
Village, CA 91362
(818)
597-7552
Fax
(818) 597-7551
Approximate date of commencement of
proposed sale to the public: From time to time after this registration
statement becomes effective.
If any of
the securities being registered on this Form are to be offered on a delayed or
continuous basis pursuant to Rule 415 under the Securities Act of 1933 check the
following box.
x
If this
Form is filed to register additional securities for an offering pursuant to Rule
462(b) under the Securities Act, please check the following box and list the
Securities Act registration statement number of the earlier effective
registration statement for the same offering. ¨
If this
Form is a post-effective amendment filed pursuant to Rule 462(c) under the
Securities Act, check the following box and list the Securities Act registration
statement number of the earlier effective registration statement for the same
offering.
¨
If this
Form is a post-effective amendment filed pursuant to Rule 462(d) under the
Securities Act, check the following box and list the Securities Act registration
statement number of the earlier effective registration statement for the same
offering.
¨
Indicate
by check mark whether the registrant is a large accelerated filer, an
accelerated filer, a non-accelerated filer, or a smaller reporting company. See
the definitions of “large accelerated filer,” “accelerated filer” and “smaller
reporting company” in Rule 12b-2 of the Exchange Act.
|
Large accelerated filer
|
¨
|
|
Accelerated filer
|
¨
|
||
|
Non-accelerated filer
|
¨ (Do
not check if smaller reporting company)
|
|
Smaller reporting company
|
x
|
Pursuant
to Rule 429 under the Securities Act of 1933, the prospectus included in this
registration statement is a combined prospectus relating to:
(i)
the resale of 437,271 common shares by
the Seller Stockholders being registered herein;
(ii) the
resale of 6,012,400 common shares previously registered
on registration statement no. 333-153829, filed by the registrant on
Form S-1 and declared effective December 31, 2008; and
(iii) the
resale of 4,516,120 common shares previously registered
on registration statement no. 333-160949, filed by the registrant on
Form S-1 and declared effective August 14, 2009.
This
registration statement, which is a new registration statement, also constitutes
post-effective amendment no. 1 to the above referenced registration statements,
and such post-effective amendment shall hereafter become effective concurrently
with the effectiveness of this registration statement and in accordance with
Section 8(c) of the Securities Act of 1933.
CALCULATION
OF REGISTRATION FEE
|
Title of Each Class of
Securities to be Registered
|
Amount to be
Registered
|
Proposed Maximum
Offering Price
|
Proposed Maximum
Aggregate Offering
Price
|
Amount of
Registration Fee
|
||||||||
|
Common
Stock, par value $0.0001 per share
|
185,002
|
$
|
1.50
|
(1) |
$
|
277,503
|
$
|
15.48
|
||||
|
Common
Stock, par value $0.0001 per share(3)
|
92,269
|
$
|
3.00
|
(2) |
$
|
276,807
|
$
|
15.45
|
||||
|
Common
Stock, par value $0.0001 per share(4)
|
100,000
|
$
|
1.50
|
(2) |
$
|
150,000
|
$
|
8.37
|
||||
|
Common
Stock, par value $0.0001 per share(5)
|
60,000
|
$
|
0.50
|
(2) |
$
|
30,000
|
1.67
|
|||||
|
Subtotal
|
437,271
|
$
|
744,310
|
$ |
40.97
|
|||||||
|
Previously
registered on Registration Statement No. 333-153829:
|
||||||||||||
|
Common
Stock, par value $0.0001 per share
|
4,490,000
|
(7) |
N/A
|
(9)
|
||||||||
|
Common
Stock, par value $0.0001 per share(4)(10)
|
1,522,400
|
(7) |
N/A
|
(9)
|
||||||||
|
Subtotal
|
6,012,400
|
(7) |
N/A
|
(9)
|
||||||||
|
Previously
registered on Registration Statement No. 333-160949:
|
||||||||||||
|
Common
Stock, par value $0.0001 per share
|
2,558,686
|
(8) |
N/A
|
(9)
|
||||||||
|
Common
Stock, par value $0.0001 per share (4)
|
394,187
|
(8) |
N/A
|
(9)
|
||||||||
|
Common
Stock, par value $0.0001 per share (6)
|
50,000
|
(8) |
N/A
|
(9)
|
||||||||
|
Common
Stock, par value $0.0001 per share (3)
|
1,513,247
|
(8) |
N/A
|
(9)
|
||||||||
|
Sub
Total
|
4,516,120
|
N/A
|
(9)
|
|||||||||
|
Total
|
10,965,791
|
|
|
|
|
|||||||
|
(1)
|
Estimated
solely for the purpose of calculating the registration fee in accordance
with Rule 457 of the Securities Act based upon a per share amount of
$1.50, based on the price at which the securities were previously
sold. There is currently no trading market for the Registrant's
common stock. The price of $1.50 is a fixed price at which the selling
stockholders identified herein may sell their shares until a market
develops for the Registrant's common stock, if ever, at which time the
shares may be sold at prevailing market prices or privately negotiated
prices.
|
|
(2)
|
Fee
based on exercise price applicable to shares issuable upon exercise of
warrants in accordance with Rule
457(g).
|
|
(3)
|
Represents
shares of Common Stock issuable upon the exercise (at a price of
$3.00 per share) of outstanding
warrants.
|
|
(4)
|
Represents
shares of Common Stock issuable upon the exercise (at a price of
$1.50 per share) of outstanding
warrants.
|
|
(5)
|
Represents
shares of Common Stock issuable upon the exercise (at a price of
$0.50 per share) of outstanding
warrants.
|
|
(6)
|
Represents
shares of Common Stock issuable upon the exercise (at a price of
$2.00 per share) of outstanding
warrants.
|
|
(7)
|
These
securities were previously registered on registration statement no.
333-153829 filed
with the SEC on Form S-1 and declared effective on December 31,
2008. Pursuant to Rule 429 of SEC Regulation C, this
registration statement will act as a post-effective amendment with respect
to that prior registration
statement.
|
|
(8)
|
These
securities were previously registered on registration statement no.
333-160949 filed
with the SEC on Form S-1 and declared effective on August 14,
2009. Pursuant to Rule 429 of SEC Regulation C, this
registration statement will act as a post-effective amendment with respect
to that prior registration
statement.
|
2
|
(9)
|
No
fees due as the shares were previously registered in connection with the
filing of the registration statement described in notes (6) and (7) and
the fees were previously paid as part of that
registration.
|
| (10) | Initial exercise price was $2.00, subsequently repriced |
THE
REGISTRANT HEREBY AMENDS THIS REGISTRATION STATEMENT ON SUCH DATE OR DATES AS
MAY BE NECESSARY TO DELAY ITS EFFECTIVE DATE UNTIL THE REGISTRANT SHALL FILE A
FURTHER AMENDMENT WHICH SPECIFICALLY STATES THAT THIS REGISTRATION STATEMENT
SHALL THEREAFTER BECOME EFFECTIVE IN ACCORDANCE WITH SECTION 8(a) OF THE
SECURITIES ACT OF 1933, AS AMENDED, OR UNTIL THE REGISTRATION STATEMENT SHALL
BECOME EFFECTIVE ON SUCH DATE AS THE COMMISSION, ACTING PURSUANT TO SUCH SECTION
8(a), MAY DETERMINE.
3
SUBJECT
TO COMPLETION, DATED OCTOBER 15, 2009
The
information in this preliminary prospectus is not complete and may be changed.
These securities may not be sold until the registration statement filed with the
Securities and Exchange Commission is effective. This preliminary prospectus is
not an offer to sell nor does it seek an offer to buy these securities in any
jurisdiction where the offer or sale is not permitted.
PROSPECTUS
10,965,791
Shares
Common
Stock

This
prospectus relates to the resale of 10,965,791 shares of our common stock, by
the selling stockholders identified in the selling stockholders tables beginning
on page 37 of this prospectus (“Selling Stockholders”). We will not receive any
proceeds from the sale of these shares by the selling stockholders.
Our
common stock is not presently traded on any market or exchange. On
September 18, 2009, our common shares began quotation on the National
Association of Securities Dealers OTC Bulletin Board (“OTCBB”) under the trading
symbol of GNSZ. As of the date of this prospectus, the trading in our
common shares is limited and accordingly, no public market
exists. The Selling Stockholders may sell their shares at a price of
$1.50 per share or at prevailing market prices or privately negotiated
prices. Thereafter, the Selling Stockholders and any of their
pledgees, assignees and successors-in-interest may, from time to time, sell any
or all of their shares of common stock on any stock exchange in which a market
develops or trading facility on which the shares are traded or in private
transactions.
Our
principal executive offices are located at 2511 N Loop 1604 W,
Suite 204, San Antonio, Texas, 78258, telephone number
210-479-8112.
Investing
in our common stock is highly speculative and involves a high degree of risk.
You should consider carefully the risks and uncertainties in the section
entitled “Risk Factors” of this prospectus.
NEITHER
THE SECURITIES AND EXCHANGE COMMISSION NOR ANY STATE SECURITIES COMMISSION HAS
APPROVED OR DISAPPROVED OF THESE SECURITIES OR PASSED UPON THE ADEQUACY OR
ACCURACY OF THIS PROSPECTUS. ANY REPRESENTATION TO THE CONTRARY IS A CRIMINAL
OFFENSE.
The date
of this Prospectus is 15.
4
TABLE
OF CONTENTS
|
|
Page
|
|
FORWARD
LOOKING STATEMENTS
|
6
|
|
RISK
FACTORS
|
6
|
|
USE
OF PROCEEDS
|
11
|
|
DIVIDEND
POLICY
|
12
|
|
DETERMINATION
OF OFFERING PRICE
|
12
|
|
OUR
BUSINESS
|
12
|
|
PROPERTIES
|
19
|
|
MANAGEMENT’S
DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF
OPERATIONS
|
20
|
|
LEGAL
PROCEEDINGS
|
29
|
|
MANAGEMENT
|
29
|
|
EQUITY
COMPENSATION PLAN INFORMATION
|
33
|
|
CERTAIN
RELATIONSHIPS AND RELATED TRANSACTIONS
|
34
|
|
PRINCIPAL
STOCKHOLDERS
|
36
|
|
SELLING
STOCKHOLDERS
|
36
|
|
DESCRIPTION
OF SECURITIES
|
46
|
|
MARKET
FOR COMMON EQUITY & RELATED STOCKHOLDER MATTERS
|
47
|
|
SHARES
ELIGIBLE FOR FUTURE SALE
|
47
|
|
PLAN
OF DISTRIBUTION
|
49
|
|
INDEMNIFICATION
OF DIRECTORS AND OFFICERS
|
50
|
|
LEGAL
MATTERS
|
50
|
|
EXPERTS
|
50
|
|
INTERESTS
OF NAMED EXPERTS AND COUNSEL
|
50
|
|
WHERE
YOU CAN FIND MORE INFORMATION
|
51
|
|
FINANCIAL
STATEMENTS
|
F-1
|
You may
rely only on the information contained in this prospectus. We have not
authorized anyone to provide information or to make representations not
contained in this prospectus. This prospectus is neither an offer to sell nor a
solicitation of an offer to buy any securities other than those registered by
this prospectus, nor is it an offer to sell or a solicitation of an offer to buy
securities where an offer or solicitation would be unlawful. Neither the
delivery of this prospectus, nor any sale made under this prospectus, means that
the information contained in this prospectus is correct as of any time after the
date of this prospectus.
5
We
urge you to read this entire prospectus carefully, including the” Risk Factors”
section and the financial statements and related notes included in our Annual
Report on Form 10-K for the fiscal year ended December 31, 2008, filed with
the Securities and Exchange Commission (“SEC”) on March 30, 2009 as well as all
subsequent Quarterly Reports on Form 10-Q. As used in this prospectus,
unless context otherwise requires, the words “we,” “us,”“our,” “the Company” and
“GenSpera” refer to GenSpera, Inc. Also, any reference to “common
shares” or “common stock” refers to our $.0001 par value common
stock.
FORWARD
LOOKING STATEMENTS
This
prospectus contains forward-looking statements” within the meaning of Section
27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act
of 1934 (the "Exchange Act"), which are intended to convey our expectations or
predictions regarding the occurrence of possible future events or the existence
of trends and factors that may impact our future plans and operating results.
These forward-looking statements are derived, in part, from various assumptions
and analyses we have made in the context of our current business plan and
information currently available to us and in light of our experience and
perceptions of historical trends, current conditions and expected future
developments and other factors we believe are appropriate in the circumstances.
You can generally identify forward looking statements through words and phrases
such as “believe”, “expect”,
“seek”, “estimate”, “anticipate”, “intend”, “plan”, “budget”, “project”, “may
likely result”, “may be”, “may continue” and other similar
expressions.
When
reading any forward-looking statement you should remain mindful that actual
results or developments may vary substantially from those expected as expressed
in, or implied by, such statement for a number of reasons or factors, including
but not limited to:
|
|
·
|
our
research and development activities, the development of a viable product,
and the speed with which regulatory authorizations and product launches
may be achieved;
|
|
|
·
|
whether
or not a market for our product develops and, if a market develops, the
rate at which it develops;
|
|
|
·
|
our
ability to sell our products;
|
|
|
·
|
our
ability to attract and retain qualified personnel to implement our
business plan and growth
strategies;
|
|
|
·
|
our
ability to license or develop sales, marketing, and distribution
capabilities;
|
|
|
·
|
the
accuracy of our estimates and
projections;
|
|
|
·
|
our
ability to fund our short-term and long-term financing
needs;
|
|
|
·
|
changes
in our business plan and corporate growth strategies;
and
|
|
|
·
|
other
risks and uncertainties discussed in greater detail in the section
captioned “Risk Factors”
|
RISK
FACTORS
Investing
in our common stock involves a high degree of risk. You should carefully
consider the following risk factors and all other information contained in this
prospectus before purchasing our common stock. If any of the following events
were to occur, our business, financial condition or results of operations could
be materially and adversely affected. In these circumstances, the market price
of our common stock could decline, and you could lose your entire
investment.
Risks
Relating to Our Stage of Development
As a result of
our limited operating history, you cannot rely upon our historical performance
to make an investment decision.
Since
inception in 2003 and through June 30, 2009 we have raised approximately
$6,170,000 in capital. During this same period, we have recorded
accumulated losses totaling $7,590,844. As of June 30, 2009, we had working
capital of $1,750,163 and a deficiency in stockholders’ equity of $186,364. Our
net losses for the two most recent fiscal years ended December 31, 2007 and 2008
have been $691,199 and $3,326,261, respectively. Since inception, we have
generated no revenue.
Our
limited operating history means that there is a high degree of uncertainty in
our ability to: (i) develop and commercialize our technologies and proposed
products; (ii) obtain approval from the U.S. Food and Drug Administration
(“FDA”); (iii) achieve market acceptance of our proposed product, if developed;
(iv) respond to competition; or (v) operate the business, as management has not
previously undertaken such actions as a company. No assurances can be given as
to exactly when, if at all, we will be able to fully develop, commercialize,
market, sell and derive material revenues from our proposed products in
development.
6
We
will need to raise additional capital to continue operations.
We
currently generate no cash. We have relied entirely on external financing to
fund operations. Such financing has historically come from the sale of common
stock to third parties, loans from our Chief Executive Officer and the exercise
of warrants/options. We have expended and will continue to expend substantial
amounts of cash in the development, pre-clinical and clinical testing of our
proposed products. We will require additional cash to conduct drug development,
establish and conduct pre-clinical and clinical trials, support commercial-scale
manufacturing arrangements and provide for the marketing and distribution of our
products if developed. We anticipate that we will require an additional $7
million to take our lead drug through Phase II clinical evaluation, which is
currently anticipated to occur in the fourth quarter of 2011.
From June
through September of 2009, we raised an aggregate of approximately $3,278,000
from the private placement of our securities. We anticipate that our
cash as of September 30, 2009, and will be sufficient to satisfy contemplated
cash requirements through October of 2010, assuming we do not engage in an
extraordinary transaction or otherwise face unexpected events or contingencies,
any of which could affect cash requirements. As of September 30, 2009, we had
cash on hand of approximately $2,614,000 Presently, the Company has
an average monthly cash burn rate of approximately $195,000. We
expect this average monthly cash burn rate to remain constant over the next
thirteen months, assuming we do not engage in an extraordinary transaction or
otherwise face unexpected events or contingencies. Accordingly, we
will need to raise additional capital to fund anticipated operating expenses
after October of 2010. In the event we are not able to secure financing, we may
have to delay, reduce the scope of or eliminate one or more of our research,
development or commercialization programs or product launches or marketing
efforts. Any such change may materially harm our business, financial
condition and operations.
Our long
term capital requirements are expected to depend on many factors,
including:
|
·
|
our
development programs;
|
|
·
|
the
progress and costs of pre-clinical studies and clinical
trials;
|
|
·
|
the
time and costs involved in obtaining regulatory
clearance;
|
|
·
|
the
costs involved in preparing, filing, prosecuting, maintaining and
enforcing patent claims;
|
|
·
|
the
costs of developing sales, marketing and distribution channels and our
ability to sell our products;
|
|
·
|
competing
technological and market
developments;
|
|
·
|
market
acceptance of our proposed products, if
developed;
|
|
·
|
the
costs for recruiting and retaining employees, consultants and
professionals; and
|
|
·
|
the
costs for educating and training physicians about our
products.
|
We cannot
assure you that financing whether from external sources or related parties, will
be available if needed or on favorable terms. If additional financing is not
available when required or is not available on acceptable terms, we may be
unable to fund operations and planned growth, develop or enhance our
technologies, take advantage of business opportunities or respond to competitive
market pressures.
Raising
needed capital may be difficult as a result of our limited operating
history.
When
making investment decisions, investors typically look at a company’s historical
performance in evaluating the risks and operations of the business and the
business’s future prospects. Our limited operating history makes such evaluation
and an estimation of our future performance substantially more difficult. As a
result, investors may be unwilling to invest in us or such investment may be on
terms or conditions which are not acceptable. If we are unable to secure such
additional finance, we may need to cease operations.
Our
independent auditors have issued a qualified report for the year ended December
31, 2008 with respect to our ability to continue as a going
concern.
For the
year ended December 31, 2008, our independent auditor issued a report relating
to our audited financial statements which contains a qualification with respect
to our ability to continue as a going concern because, among other things, our
ability to continue as a going concern is dependent upon our ability to develop
a product and generate profits from operations in the future or to obtain the
necessary financing to meet our obligations and repay our liabilities when they
come due. However, during 2009 we have raised approximately $4,028,000 through
our private placements and we expect that our cash on hand at September 30, 2009
will be sufficient to fund operations through October of 2010.
7
We
may not be able to commercially develop our technologies.
We have
concentrated our research and development on our drug
technologies. Our ability to generate revenue and operate profitably
will depend on our being able to develop these technologies for human
applications. Our technologies are primarily directed toward the development of
cancer therapeutic agents. We cannot guarantee that the results obtained in
pre-clinical and clinical evaluation of our therapeutic agents will be
sufficient to warrant approval by the U.S. Food and Drug Administration (“FDA”).
Even if our therapeutic agents are approved for use by the FDA, there is no
guarantee that they will exhibit an enhanced efficacy relative to competing
therapeutic modalities such that they will be adopted by the medical community.
Without significant adoption by the medical community, our agents will have
limited commercial potential which will likely result in the loss of your entire
investment.
Inability
to complete pre-clinical and clinical testing and trials will impair our
viability.
On
September 4, 2009, we received approval from the FDA for our first
Investigational New Drug application (“IND”) in order to commence clinical
trials. Although we have received approval from the FDA to commence
trials, the outcome of the trials is uncertain and, if we are unable to
satisfactorily complete such trials, or if such trials yield unsatisfactory
results, we will be unable to commercialize our proposed products. No assurances
can be given that the clinical trials will be successful. The failure of such
trials could delay or prevent regulatory approval and could harm our ability to
generate revenues, operate profitably or remain a viable business.
Future
financing will result in dilution to existing stockholders.
We will
require additional financing in the future. We are authorized to issue 80
million shares of common stock and 10 million shares of preferred stock. Such
securities may be issued without the approval or consent of our
stockholders. The issuance of our equity securities in connection with a
future financing will result in a decrease of our current stockholders’
percentage ownership.
Risks
Relating to Intellectual Property and Government Regulation
We
may not be able to withstand challenges to our intellectual property
rights.
We rely
on our intellectual property, including our issued and applied for patents, as
the foundation of our business. Our intellectual property rights may come under
challenge. No assurances can be given that, even if issued, our
patents will survive claims alleging invalidity or infringement on other
patents. The viability of our business will suffer if such patent protection
becomes limited or is eliminated.
We
may not be able to adequately protect our intellectual property.
Considerable
research with regard to our technologies has been performed in countries outside
of the United States. The laws protecting intellectual property in some of those
countries may not provide protection for our trade secrets and intellectual
property. If our trade secrets or intellectual property are
misappropriated in those countries, we may be without adequate remedies to
address the issue. At present, we are not aware of any infringement of our
intellectual property. In addition to our patents, we rely on confidentiality
and assignment of invention agreements to protect our intellectual property.
These agreements provide for contractual remedies in the event of
misappropriation. We do not know to what extent, if any, these
agreements and any remedies for their breach will be enforced by a court. In the
event our intellectual property is misappropriated or infringed upon and an
adequate remedy is not available, our future prospects will greatly
diminish.
Our
proposed products may not receive FDA approval.
The FDA
and comparable government agencies in foreign countries impose substantial
regulations on the manufacture and marketing of pharmaceutical products through
lengthy and detailed laboratory, pre-clinical and clinical testing procedures,
sampling activities and other costly and time-consuming procedures. Satisfaction
of these regulations typically takes several years or more and varies
substantially based upon the type, complexity and novelty of the proposed
product. On September 4, 2009, we received approval from the FDA for
our first IND in order to commence clinical trials. We cannot
assure you that we will successfully complete any clinical trials in connection
with any such IND application. Further, we cannot yet accurately
predict when we might first submit any product license application for FDA
approval or whether any such product license application would be granted on a
timely basis, if at all. Any delay in obtaining, or failure to
obtain, such approvals could have a materially adverse effect on the
commercialization of our products and the viability of the company.
Risks
Relating to Competition
Our
competitors have significantly greater experience and financial
resources.
We
compete against numerous companies, many of which have substantially greater
financial and other resources than us. Several such enterprises have research
programs and/or efforts to treat the same diseases we target. Companies such as
Merck, Ipsen and Diatos, as well as others, have substantially greater resources
and experience than we do and are situated to compete with us
effectively. As a result, our competitors may bring competing
products to market that would result in a decrease in demand for our product, if
developed, which could have a materially adverse effect on the viability of the
company.
8
Risks
Relating to Reliance on Third Parties
We
intend to rely exclusively upon the third-party FDA-approved manufacturers and
suppliers for our products.
We
currently have no internal manufacturing capability, and will rely exclusively
on FDA-approved licensees, strategic partners or third party contract
manufacturers or suppliers. Should we be forced to manufacture our products, we
cannot give you any assurance that we will be able to develop internal
manufacturing capabilities or procure third party suppliers. In the event we
seek third party suppliers, they may require us to purchase a minimum amount of
materials or could require other unfavorable terms. Any such event would
materially impact our prospects and could delay the development and sale of our
products. Moreover, we cannot give you any assurance that any contract
manufacturers or suppliers that we select will be able to supply our products in
a timely or cost effective manner or in accordance with applicable regulatory
requirements or our specifications.
General
Risks Relating to Our Business
We
depend on Craig A. Dionne, PhD, our Chief Executive Officer, and Russell
Richerson PhD, our Chief Operating Officer, for our continued
operations.
The loss
of Craig A. Dionne, PhD, our Chief Executive Officer, or Russell Richerson, PhD,
our Chief Operating Officer, would be detrimental to us. We currently maintain a
one million dollar “key person” life insurance policy on the life of Dr. Dionne
but do not maintain a policy on Dr. Richerson. Our prospects and operations will
be significantly hindered upon the death or incapacity of either of these key
individuals.
We
will require additional personnel to execute our business plan.
Our
anticipated growth and expansion into areas and activities requiring additional
expertise, such as clinical testing, regulatory compliance, manufacturing and
marketing, will require the addition of new management personnel and the
development of additional expertise by existing management. There is intense
competition for qualified personnel in such areas. There can be no
assurance that we will be able to continue to attract and retain the qualified
personnel necessary for the development of our business.
Our
business is dependent upon securing sufficient quantities of a natural product
that currently grows in very specific locations outside of the United
States.
The
therapeutic component of our products, including our lead compound G-202, is
referred to as 12ADT. 12ADT functions by dramatically raising the levels of
calcium inside cells, which leads to cell death. 12ADT is derived from a
material called thapsigargin. Thapsigargin is derived from the seeds of a plant
referred to as Thapsia
garganica which grows along the coastal regions of the
Mediterranean. We currently secure the seeds from Thapsibiza, SL, a third party
supplier. There can be no assurances that the countries from which we can
secure Thapsia
garganica will continue to allow Thapsibiza, SL to collect such
seeds and/or to do so and export the seeds derived from Thapsia garganica
to the United States. In the event we are no longer able to import
these seeds, we will not be able to produce our proposed drug and our business
will be adversely affected.
The
current manufacturing process of G-202 requires acetonitrile.
The
current manufacturing process for G-202 requires the common solvent
acetonitrile. Beginning in late 2008, there has been a worldwide shortage of
acetonitrile for a variety of reasons, and this shortage is predicted to last at
least until the end of 2009 and possibly into future years. We have also
observed that the available supply of acetonitrile is of variable quality, some
of which is not suitable for our purposes. If we are unable to
successfully change our manufacturing methods to avoid the reliance upon
acetonitrile, we may incur prolonged production timelines and increased
production costs. In an extreme case this situation could adversely affect our
ability to manufacture G-202 altogether, thus significantly impacting our future
operations.
In
order to secure market share and generate revenues, our proposed products must
be accepted by the health care community.
Our
proposed products, if approved for marketing, may not achieve market acceptance
since hospitals, physicians, patients or the medical community in general may
decide not to accept and utilize them. We are attempting to develop products
that will likely be first approved for marketing in late stage cancer where
there is no truly effective standard of care. If approved for use in late stage,
the drugs will then be evaluated in earlier stage where they would represent
substantial departures from established treatment methods and will compete with
a number of more conventional drugs and therapies manufactured and marketed by
major pharmaceutical companies. It is too early in the development cycle of the
drugs for us to accurately predict our major
competitors. Nonetheless, the degree of market acceptance of any of
our developed products will depend on a number of factors,
including:
|
|
·
|
our demonstration to the medical
community of the clinical efficacy and safety of our proposed
products;
|
|
|
·
|
our ability to create products
that are superior to alternatives currently on the
market;
|
9
|
|
·
|
our ability to establish in the
medical community the potential advantage of our treatments over
alternative treatment methods;
and
|
|
|
·
|
the reimbursement policies of
government and third-party
payors.
|
If the
health care community does not accept our products for any of the foregoing
reasons, or for any other reason, our business will be materially
harmed.
We
may be required to secure land for cultivation and harvesting of Thapsia
garganica.
We
believe that we can satisfy our needs for clinical development of G-202 through
completion of Phase III clinical studies from Thapsia garganica that grows
naturally in the wild. In the event G-202 is approved for commercial
marketing, our current supply of Thapsia garganica may not be
sufficient for the anticipated demand. We estimate that in order to
secure sufficient quantities of Thapsia garganica for the
commercialization of a product comprising G-202, we will need to secure
approximately 100 acres of land to cultivate and grow Thapsia
garganica. We anticipate the cost to lease such land
would be $40,000 per year but have not yet fully assessed what other costs would
be associated with a full-scale farming operation. There can be no assurances
that we can secure such acreage, or that even if we are able to do so, that we
could adequately grow sufficient quantities of Thapsia garganica to satisfy
any commercial objectives that involve G-202. Our inability to secure adequate
seeds will result in us not being able to develop and manufacture our proposed
drug and will adversely impact our business.
Thapsia
garganica and Thapsigargin can cause severe skin irritation.
It has
been known for centuries that the plant Thapsia garganica can cause
severe skin irritation when contact is made between the plant and the
skin. In 1978, thapsigargin was determined to be the skin-irritating
component of the plant Thapsia
garganica. The therapeutic component of our products, including our lead
product G-202, is derived from thapsigargin. We obtain thapsigargin from the
above-ground seeds of Thapsia
garganica. These seeds are harvested by hand and those conducting the
harvesting must wear protective clothing and gloves to avoid skin contact.
Although we obtain the seeds from a third-party contractor located in Spain, and
although the contractor has contractually waived any and all liability
associated with collecting the seeds, it is possible that the contractor or
those employed by the contractor may suffer medical issues related to the
harvesting and subsequently seek compensation from us via, for example,
litigation. No assurances can be given, despite our contractual
relationship with the third party contractor, that we will not be the subject of
litigation related to the harvesting.
The
synthesis of 12ADT must be conducted in special facilities.
There are
a limited number of manufacturing facilities qualified to handle and manufacture
using toxic agents for therapeutic compounds. This limits the potential number
of possible manufacturing sites for our therapeutic compounds derived from Thapsia
garganica. No assurances can be provided that these
facilities will be available for the manufacture of our therapeutic compounds
under our time schedules or within the parameters of our manufacturing budget.
In the event facilities are not available for manufacturing our therapeutic
compounds, our business and future prospects will be adversely
affected.
Our
lead therapeutic compound, G-202, has not been subjected to large scale
manufacturing procedures.
To date,
G-202 has only been manufactured at a scale adequate to supply early stage
clinical trials. There can be no assurances that the current procedure for
manufacturing G-202 will work at a larger scale adequate for commercial
needs. In the event G-202 cannot be manufactured in sufficient
quantities, our future prospects could be significantly impacted.
We
do not have product liability insurance.
The
testing, manufacturing, marketing and sale of human therapeutic products entail
an inherent risk of product liability claims. We cannot assure you
that substantial product liability claims will not be asserted against
us. In the event we are forced to expend significant funds on
defending product liability actions beyond our current coverage, and in the
event those funds come from operating capital, we will be required to reduce our
business activities, which could lead to significant losses.
We
cannot assure you that adequate insurance coverage will be available in the
future on acceptable terms.
Insurance
availability, coverage terms and pricing continue to vary with market
conditions. We will endeavor to obtain appropriate insurance coverage for
insurable risks that we identify. We may not be able to obtain
appropriate insurance coverage. The occurrence of any claim may have
an adverse material effect on our business.
10
Risks
Relating To Our Common Stock
There
is no public market for our securities.
On
September 18, 2009, our common shares began quotation on the
OTCBB. Notwithstanding, there has been little or no trading in our
common shares. Accordingly, there is no public market for our
securities. An investment in our common stock should be considered
totally illiquid. No assurances can be given that a public market for
our securities will ever materialize. Additionally, even if a public market for
our securities develops and our securities become traded, the trading volume may
be limited, making it difficult for an investor to sell shares.
We
face risks related to compliance with corporate governance laws and financial
reporting standards.
The
Sarbanes-Oxley Act of 2002, as well as related new rules and regulations
implemented by the SEC and the Public Company Accounting Oversight Board,
require changes in the corporate governance practices and financial reporting
standards for public companies. These new laws, rules and regulations, including
compliance with Section 404 of the Sarbanes-Oxley Act of 2002 relating to
internal control over financial reporting (“Section 404”), will materially
increase the Company's legal and financial compliance costs and make some
activities more time-consuming and more burdensome. As a result, management will
be required to devote more time to compliance which could result in a reduced
focus on the development thereby adversely affecting the Company’s development
activities. Also, the increased costs will require the Company to seek financing
sooner that it may otherwise have had to.
Starting
in 2007, Section 404 of the Sarbanes-Oxley Act of 2002 requires that
our management assess the company’s internal control over financial
reporting annually and include a report on such assessment in our annual report
filed with the SEC. Effective December 15, 2009 for a smaller reporting company,
our independent registered public accounting firm is required to audit both the
design and operating effectiveness of our internal controls and management's
assessment of the design and the operating effectiveness of such internal
controls.
Because
of our limited resources, management has concluded that our internal control
over financial reporting may not be effective in providing reasonable assurance
regarding the reliability of financial reporting and the preparation of
financial statements for external purposes in accordance with U.S. generally
accepted accounting principles.
To
mitigate the current limited resources and limited employees, we rely heavily on
direct management oversight of transactions, along with the use of legal and
accounting professionals. As we grow, we expect to increase our number of
employees, which will enable us to implement adequate segregation of duties
within the Committee of Sponsoring Organizations of the Treadway Commission
internal control framework.
We do not intend
to pay cash dividends.
We do not
anticipate paying cash dividends in the foreseeable future. Accordingly, any
gains on your investment will need to come through an increase in the price of
our common stock. The lack of a market for our common stock makes
such gains highly unlikely.
Our
board of directors has broad discretion to issue additional
securities.
We are
entitled under our certificate of incorporation to issue up to 80,000,000 common
and 10,000,000 “blank check” preferred shares. Blank check preferred shares
provide the board of directors broad authority to determine voting, dividend,
conversion, and other rights. As of September 30, 2009, we have issued and
outstanding 15,292,281 common shares and we have 7,884,502 common shares
reserved for issuance upon the exercise of current outstanding options, warrants
and convertible securities. Accordingly, we will be entitled to issue up to
56,823,217 additional common shares and 10,000,000 additional preferred shares.
Our board may generally issue those common and preferred shares, or options or
warrants to purchase those shares, without further approval by our
shareholders. Any preferred shares we may issue will have such
rights, preferences, privileges and restrictions as may be designated from
time-to-time by our board, including preferential dividend rights, voting
rights, conversion rights, redemption rights and liquidation provisions. It is
likely that we will be required to issue a large amount of additional securities
to raise capital to further our development and marketing plans. It is also
likely that we will be required to issue a large amount of additional securities
to directors, officers, employees and consultants as compensatory grants in
connection with their services, both in the form of stand-alone grants or under
our various stock plans. The issuance of additional securities may cause
substantial dilution to our shareholders.
Our
Officers and Scientific Advisors beneficially own approximately 47% of our
outstanding common shares.
Our
Officers and Scientific Advisors own approximately 47% of our issued and
outstanding common shares. As a consequence of their level of stock ownership,
the group will substantially retain the ability to elect or remove members of
our board of directors, and thereby control our management. This group of
shareholders has the ability to significantly control the outcome of corporate
actions requiring shareholder approval, including mergers and other changes of
corporate control, going private transactions, and other extraordinary
transactions any of which may be in opposition to the best interest of the other
shareholders and may negatively impact the value of your
investment.
USE
OF PROCEEDS
We will
not receive any of the proceeds from the sale of the shares by any of the
selling stockholders, but we will receive up to $3,431,687 upon the exercise of
warrants in the event they are exercised for cash. We will use the proceeds
received from the exercise of warrants, if any, for working
capital.
11
DIVIDEND
POLICY
We have
never paid or declared cash dividends on our common stock, and we do not intend
to pay or declare cash dividends on our common stock in the foreseeable
future.
DETERMINATION
OF OFFERING PRICE
The
Selling Stockholders will initially offer their shares at $1.50 per share until
such time as a public market develops, if ever. Once traded, the
shares will be offered at prevailing market prices, privately negotiated prices,
or in any other fashion as described in the section of this Prospectus entitled
“Plan of
Distribution.” The selling price has no relationship to any
established criteria of value, such as book value or earnings per
share. The price was chosen arbitrarily.
OUR
BUSINESS
Our
History
We are a
biotechnology company focused on the discovery and development of pro-drug
cancer therapeutics, an emerging medical science. A pro-drug is an inactive
precursor of a drug that is converted into its active form only at the site of
the tumor. We were incorporated as a Delaware corporation in
2003.
In early
2004, the intellectual property underlying our technologies was assigned from
Johns Hopkins University to the co-inventors, Dr. John Isaacs, Dr. Soren
Christensen, Dr. Hans Lilja and Dr. Samuel Denmeade. The Co-inventors
granted us an option to license the intellectual property in return for our
continued prosecution of the patent portfolio containing the intellectual
property. This option was exercised in early 2008 by reimbursement of past
patent prosecution costs previously incurred by Johns Hopkins University.
Subsequently, the co-inventors assigned us the intellectual property in April of
2008. Our activities during the period of 2004-2007 were limited to the
continued prosecution of the relevant patents.
Dr. John
Isaacs and Dr. Sam Denmeade serve on our Scientific Advisory Board as Chief
Scientific Advisor and Chief Medical Advisor, respectively. Dr Soren
Christensen and Dr. Hans Lilja also serve on the Company’s Scientific Advisory
Board. We currently have no oral or written agreements with
Johns Hopkins University with regard to any other intellectual
property or research activities.
The
Potential of Our Pro-Drug Therapies
Cancer
chemotherapy involves treating patients with cytotoxic drugs (compounds or
agents that are toxic to cells). Chemotherapy is often combined with surgery or
radiation in the treatment of early stage disease and it is the preferred, or
only, treatment option for many forms of cancer in later stages of the disease.
However, major drawbacks of chemotherapy include:
|
Side
effects
|
|
Non-cancer
cells in the body are also affected, often leading to serious side
effects.
|
|
Incomplete
tumor kill
|
|
Many
of the leading chemotherapeutic agents act during the process
of cell division - they might be effective with tumors comprised of
rapidly-dividing cells, but are much less effective for tumors that
contain cells that are slowly
dividing.
|
|
Resistance
|
|
Cancers
will often develop resistance to current drugs after repeated exposure,
limiting the number of times that a treatment can be effectively
applied.
|
Pro-drug
chemotherapy is a relatively new approach to cancer treatment that is being
investigated as a means to get higher concentrations of cytotoxic agents at the
tumor location while avoiding the toxicity of these high doses in the rest of
the body. An inactive form of a cytotoxin (referred to as the “pro-drug”) is
administered to the patient. The pro-drug is converted into the active cytotoxin
only at the tumor site.
We
believe that, if successfully developed, pro-drug therapies have the potential
to provide an effective therapeutic approach to a broad range of solid tumors.
We have proprietary technologies that we believe appears, in animal models, to
meet the requirements for an effective pro-drug. In addition, we believe that
our cytotoxin addresses two other issues prevalent with current cancer drugs -
it kills slowly- and non-dividing cancer cells as well as rapidly dividing
cancer cells, and does not appear to trigger the development of resistance to
its effects.
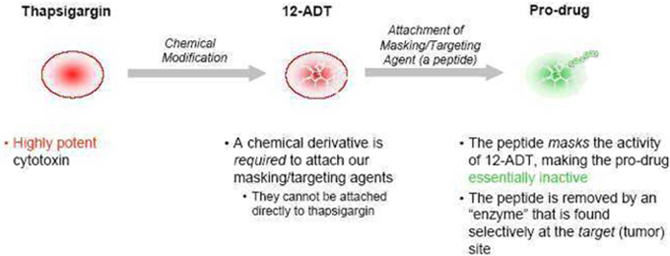
Our
Technology
Our
technology supports the creation of pro-drugs by attaching “masking/targeting
agents” (agents that simultaneously mask the toxicity of the cytotoxin and help
target the cytotoxin to the tumor) to the cytotoxin “12ADT”, and does so in a
way that allows conversion of the pro-drug to its active form selectively at the
site of tumors. We own patents that contain claims that cover 12ADT as a
composition of matter.
12
Cytotoxin
12ADT is
a chemically modified form of thapsigargin, a cytotoxin that kills fast-, slow-
and non-dividing cells. Our two issued core patents, both entitled “ Tissue Specific Prodrug, ”
contain claims which cover the composition of 12ADT.
Masking/Targeting
Agent
We use
peptides as our masking/targeting agents. Peptides are short strings of
amino-acids, the building blocks of many components found in cells. When
attached to 12ADT, they can make the cytotoxin inactive - once removed, the
cytotoxin is active again. Our technology takes advantage of the fact that the
masking peptides can be removed by chemical reactors in the body called enzymes,
and that the recognition of particular peptides by particular enzymes can be
very specific. The peptides also make 12ADT soluble in blood. When it is
removed, 12ADT returns to its natural insoluble state and precipitates directly
into nearby cells.
How
we make our pro-drugs
How
our pro-drugs work
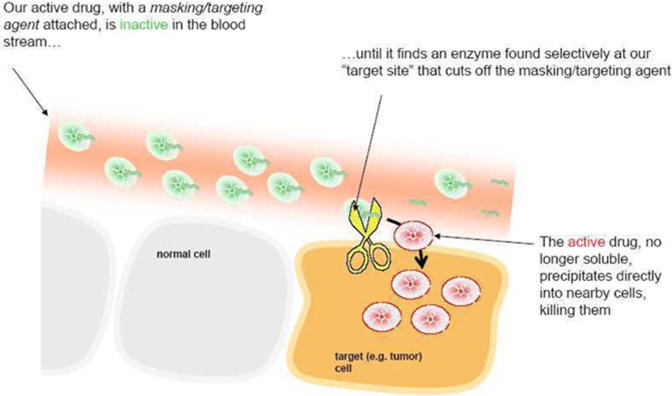
13
Our
Approach
Our
approach is to identify specific enzymes that are found at high levels in tumors
relative to other tissues in the body. Upon identifying these enzymes, we create
peptides that are recognized predominantly by those enzymes in the tumor and not
by enzymes in normal tissues. This double layer of recognition adds to the
tumor-targeting found in our pro-drugs. Because the exact nature of our
masking/targeting peptides is so refined and specific, they form the basis for
another set of our patents and patent applications on the combination of the
peptides and 12ADT.
Our
Pro-Drug Development Candidates
We
currently have four pro-drug candidates identified based on this technology, as
summarized in the table below (at this time we are only developing
G-202):
|
Pro-Drug Candidate
|
Activating enzyme
|
Target location of activation
enzyme
|
Status
|
||||
|
G-202
|
Prostate
Specific Membrane Antigen (PSMA)
|
The
blood vessels of all solid tumors
|
·
|
Investigational
New Drug Application planned is filed with the US Food and Drug
Administration. Currently on Clinical Hold pending our response to
questions.
|
|||
|
G-114
|
Prostate
Specific Antigen (PSA)
|
Prostate
cancers
|
·
|
Validated
efficacy in pre-clinical animal models (Johns Hopkins
University)
|
|||
|
G-115
|
Prostate
Specific Antigen (PSA)
|
Prostate
cancers
|
·
|
Validated
efficacy in pre-clinical animal models (Johns Hopkins
University)
|
|||
|
Ac-GKAFRR-L12ADT
|
Human
glandular kallikrein 2 (hK2)
|
Prostate
cancers
|
·
|
Validated
efficacy in pre-clinical animal models (Johns Hopkins
University)
|
|||
Strategy
Business
Strategy
We plan
to develop a series of therapies based on our pro-drug technology platform and
bring them through Phase I/II clinical trials.
Manufacturing
and Development Strategy
Under the
planning and direction of key personnel, we expect to outsource all of our Good
Laboratory Practices (“GLP”) preclinical development activities (e.g.,
toxicology) and Good Manufacturing Practices (“GMP”) manufacturing and clinical
development activities to contract research organizations (“CRO”) and contract
manufacturing organizations (“CMO”). Manufacturing will also be outsourced
to organizations with approved facilities and manufacturing
practices.
Commercialization
Strategy
We intend
to license our drug compounds to third parties after Phase I/II clinical trials.
It is expected that such third parties would then continue to develop, market,
sell, and distribute the resulting products.
Market
and Competitive Considerations
G-202
Our
primary focus is the opportunity offered by our lead pro-drug candidate, G-202.
We believe that we have validated G-202 as a drug candidate to treat various
forms of solid tumors; including breast, urinary bladder, kidney and prostate
cancer based on the ability of G-202 to cause tumor regression in animal models.
We filed our first IND for G-202 on June 23, 2009. On September 4,
2009, we received approval from the FDA for our first IND in order to commence
clinical trials. Although we have received approval from the FDA to
commence trials, the outcome of the trials is uncertain and, if we are unable to
satisfactorily complete such trials, or if such trials yield unsatisfactory
results, we will be unable to commercialize our proposed products. We
plan to begin the clinical evaluation of G-202 in late 2009. We hope to
eventually demonstrate that G-202 is more efficacious than current commercial
products that treat solid tumors by disrupting their blood
supply.
14
Potential
Markets for G-202
We
believe that if successfully developed, G-202 has the potential to treat a range
of solid tumors by disrupting their blood supply. It is too early in the
development process to determine target indications. The table below summarizes
a number of the potential United States patient populations which we believe may
be amenable to this therapy and represent potential target markets.
|
Estimated Number of
|
Probability of
Developing
(birth to death)
|
||||||||
|
Cancer
|
New Cases (2006)
|
Male
|
Female
|
||||||
|
Prostate
|
234,460 |
1
in 6
|
-
|
||||||
|
Breast
|
214,640 |
n/a
|
1
in 8
|
||||||
|
Urinary
Bladder
|
61,420 |
1
in 28
|
1
in 88
|
||||||
|
Kidney
Cancer
|
38,890 |
n/a
|
n/a
|
||||||
Source: CA Cancer J. Clin 2006;
56;106-130
The
clinical opportunity for G-202
We
believe that current anti-angiogenesis drugs (drugs that disrupt the blood
supply to tumors) validate the clinical approach and market potential of
G-202. Angiogenesis is the physiological process involving the growth
of new blood vessels from pre-existing vessels and is a normal process in growth
and development, as well as in wound healing. Angiogenesis is also a
fundamental step in the development of tumors from a clinically insignificant
size to a malignant state because no tumor can grow beyond a few millimeters in
size without the nutrition and oxygenation that comes from an associated blood
supply. Interrupting this process has been targeted as a point of intervention
for slowing or reversing tumor growth. A well known example of a successful
anti-angiogenic approach is the recently approved drug, Avastin TM , a monoclonal antibody that
inhibits the activity of Vascular Endothelial Growth Factor (“VEGF”), which is
important for the growth and survival of endothelial cells.
These
types of anti-angiogenic drugs have only a limited therapeutic effect with
increased median patient survival times of only a few months. Our approach is
designed to destroy both the existing and newly growing tumor vasculature,
rather than just block new blood vessel formation. We anticipate that this
approach will lead to a more immediate collapse of the tumors nutrient supply
and consequently an enhanced rate of tumor destruction.
G-202
destroys new and existing blood vessels in tumors

Competition
The
pharmaceutical, biopharmaceutical and biotechnology industries are very
competitive, fast moving and intense, and expected to be increasingly so in the
future. Although we are not aware of any competitor who is developing a
drug that is designed to destroy both the existing and newly growing tumor
vasculature in a manner similar to G-202, there are several marketed drugs and
drugs in development that attack tumor-associated blood vessels to some degree.
For example, Avastin TM
is a marketed product that acts predominantly as an anti-angiogenic
agent. Zybrestat TM
is another drug in development that is described as a
vascular-disrupting agent that inhibits blood flow to tumors. It is impossible
to accurately ascertain how well our drug will compete against these or other
products that may be in the marketplace until we have human patient data for
comparison.
15
Other
larger and well funded companies have developed and are developing drug
candidates that, if not similar in type to our drug candidates, are designed to
address the same patient or subject population. Therefore, our lead
product, other products in development, or any other products we may acquire or
in-license may not be the best, the safest, the first to market, or the most
economical to make or use. If a competitor’s product or product in
development is better than ours, for whatever reason, then our ability to
license our technology could be diminished and our sales could be lower than
that of competing products, if we are able to generate sales at
all.
Patents
and Proprietary Rights
Our
success will likely depend upon our ability to preserve our proprietary
technologies and operate without infringing on the proprietary rights of other
parties. However, we may rely on certain proprietary technologies and know-how
that are not patentable or that we determine to keep as trade secrets. We
protect our proprietary information, in part, by the use of confidentiality and
assignment of invention agreements with our officers, directors, employees,
consultants, significant scientific collaborators and sponsored researchers that
generally provide that all inventions conceived by the individual in the course
of rendering services to us shall be our exclusive property.
The
intellectual property underlying our technology is covered by certain patents
and patent applications previously owned by the Johns Hopkins University
("JHU"). In early 2004, the intellectual property underlying the Company’s
technologies was assigned from JHU to the co-inventors, Dr. John Isaacs, Dr.
Soren Christensen, Dr. Hans Lilja and Dr. Samuel Denmeade, who in turn granted
us an option to license the intellectual property in return for our continued
prosecution of the patent portfolio. This option was exercised in early 2008 by
payment to the co-inventors of past patent prosecution costs previously incurred
by Johns Hopkins University (approximately $122,000) and additional fees
(approximately $62,000) to cover the tax consequences of such payments to the
co-inventors. Subsequently, the co-inventors assigned us the intellectual
property in April of 2008 and we recorded these assignments in the United
States Patent & Trademark Office. By virtue of the April 2008 assignments,
we have no further financial obligations to the inventors or to JHU with regard
to the assigned intellectual property. JHU retains a paid-up, royalty-free,
non-exclusive license to use the intellectual property for non-profit
purposes. Each of the co-inventors remains affiliated with the Company as
a member of the Scientific Advisory Board.
|
Number
|
Country
|
Filing
Date
|
Issue Date
|
Expiration
Date
|
Title
|
||||||
|
Patents
Issued
|
|||||||||||
|
6,504,014
|
US
|
6/7/00
|
1/7/2003
|
6/6/2020
|
Tissue
specific pro-drug (TG)
|
||||||
|
6,545,131
|
US
|
7/28/00
|
4/8/2003
|
7/27/2020
|
Tissue
specific pro-drug (TG)
|
||||||
|
6,265,540
|
US
|
5/19/98
|
7/24/2001
|
5/18/2018
|
Tissue
specific pro-drug (PSA)
|
||||||
|
6,410,514
|
US
|
6/7/00
|
6/25/2002
|
6/6/2020
|
Tissue
specific pro-drug (PSA)
|
||||||
|
7,053,042
|
US
|
7/28/00
|
5/30/2006
|
7/27/2020
|
Activation
of peptide pro-drugs by HK2
|
||||||
|
7,468,354
|
US
|
11/30/01
|
12/23/08
|
11/29/2021
|
Tissue
specific pro-drug
(G-202,
PSMA)
|
||||||
|
Patents
Pending
|
|||||||||||
|
US
2008/0247950
|
US
|
3/15/07
|
Pending
|
N/A
|
Activation
of peptide pro-drugs by HK2
|
||||||
|
US
2007/0160536
|
US
|
1/6/2006
|
Pending
|
N/A
|
Tumor
Activated Pro-drugs (PSA,G-115)
|
||||||
|
US
2009/0163426
|
US
|
11/25/08
|
Pending
|
N/A
|
Tumor
specific pro-drugs (PSMA)
|
16
When
appropriate, we will continue to seek patent protection for inventions in our
core technologies and in ancillary technologies that support our core
technologies or which we otherwise believe will provide us with a competitive
advantage. We will accomplish this by filing and maintaining patent applications
for discoveries we make, either alone or in collaboration with scientific
collaborators and strategic partners. Typically, we plan to file patent
applications in the United States. In addition, we plan to obtain licenses or
options to acquire licenses to patent filings from other individuals and
organizations that we anticipate could be useful in advancing our research,
development and commercialization initiatives and our strategic business
interest.
Manufacturing
& Development
12ADT is
manufactured by chemically modifying the cytotoxin thapsigargin, which is
isolated from the seeds of
Thapsia garganica, a plant found in the Mediterranean. Our pro-drug,
G-202, is then manufactured by attaching a specific peptide to
12ADT.
Outsource
Manufacturing
To
leverage our experience and available financial resources, we do not plan to
develop company-owned or company-operated manufacturing facilities. We plan to
outsource all drug manufacturing to contract manufacturers that operate in
compliance with GMP. We may also seek to refine the current manufacturing
process and final drug formulation to achieve improvements in storage
temperatures and the like.
Supply
of Raw Materials – Thapsibiza SL
To our
knowledge, there is only one commercial supplier of Thapsia garganica seeds. In
April 2007, we obtained the proper permits from the United States Department of
Agriculture (“USDA”) for the importation of Thapsia garganica
seeds. In January 2008, we entered into a sole source agreement with this
supplier, Thapsibiza, SL. The material terms of the agreement are as
follows:
|
Term
|
|
The
term of the agreement is for 5 years.
|
|
Exclusivity
|
Thapsibiza
shall exclusively provide Thapsia garganica
seeds to the Company. The Company has the ability to seek addition
suppliers to supplement the supply from Thapsibiza, SL.
|
|
|
Pricing
|
The
price shall be 300 Euro/kg. Thapsibiza may, from time to time, without
notice, increase the price to compensate for any increased governmental
taxes.
|
|
|
Minimum
Order
|
Upon
successfully securing $5,000,000 of equity financing, and for so long as
the Company continues to develop drugs derived from thapsigargin, the
minimum purchase shall be 50kg per harvest period year.
|
|
|
Indemnification
|
Once
the product is delivered to an acceptable carrier, the Company shall be
responsible for an injury or damage result from the handling of the
product. Prior to delivery, Thapsibiza shall be solely
responsible.
|
Government
Regulation
The FDA,
as well as drug regulators in state and local jurisdictions, imposes substantial
requirements upon the clinical development, manufacturing and marketing of
pharmaceutical products. The process we are required by the FDA to
complete before our drug compound may be marketed in the U.S. generally involves
the following:
|
·
|
Preclinical
laboratory and animal tests;
|
|
|
·
|
Submission
of an IND, which must become effective before human clinical trials may
begin;
|
|
|
·
|
Adequate
and well-controlled human clinical trials to establish the safety and
efficacy of the product candidate for its intended use;
|
|
|
·
|
Submission
to the FDA of an New Drug Application (“NDA”); and
|
|
|
·
|
FDA
review and approval of an
NDA.
|
The
testing and approval process requires substantial time, effort, and financial
resources, and we cannot be certain that any approval will be granted on an
expeditious basis, if at all. Preclinical tests include laboratory
evaluation of the drug candidate, its chemistry, formulation and stability, as
well as animal studies to assess the potential safety and efficacy of the drug
candidate. Certain preclinical tests must be conducted in compliance with
good laboratory practice regulations. Violations of these regulations can, in
some cases, lead to invalidation of the studies, requiring such studies to be
replicated. In some cases, long-term preclinical studies are conducted
while clinical studies are ongoing.
17
The
results of the preclinical tests, together with manufacturing information and
analytical data, are submitted to the FDA as part of an IND, which must become
effective before we may begin human clinical trials. The IND automatically
becomes effective 30 days after receipt by the FDA, unless the FDA, within the
30-day time period, raises concerns or questions about the conduct of the trials
as outlined in the IND and imposes a clinical hold. In such a case, the
IND sponsor and the FDA must resolve any outstanding concerns before clinical
trials can begin. Our submission of an IND may not result in FDA
authorization to commence clinical trials for any particular compound. All
clinical trials must be conducted under the supervision of a qualified
investigator in accordance with good clinical practice regulations. These
regulations include the requirement that all prospective patients provide
informed consent. Further, an independent Institutional Review Board (“IRB”) at
each medical center proposing to conduct the clinical trials must review and
approve any clinical study. The IRB also monitors the study and must be
kept informed of the study’s progress, particularly as to adverse events and
changes in the research. Progress reports detailing the results of the
clinical trials must be submitted at least annually to the FDA and more
frequently if adverse events occur.
Human
cancer drug clinical trials are typically conducted in three sequential phases
that may overlap:
|
·
|
Phase
I: The drug candidate is initially introduced into cancer patients and
tested for safety and tolerability at escalating
dosages,
|
|
·
|
Phase
II: The drug candidate is studied in a limited cancer patient population
to further identify possible adverse effects and safety risks, to evaluate
the efficacy of the drug candidate for specific targeted diseases and to
determine dosage tolerance and optimal
dosage.
|
|
·
|
Phase
III: When Phase II evaluations demonstrate that a dosage range of the drug
candidate may be effective and has an acceptable safety profile, Phase III
trials are undertaken to further evaluate dose response, clinical efficacy
and safety profile in an expanded patient population, often at
geographically dispersed clinical study
sites.
|
Our
business strategy is to bring our drug candidates through Phase I/II clinical
trials before licensing them to third parties who would then further develop the
drugs and seek marketing approval. Once the drug is approved, the third party
licensee will be expected to market, sell, and distribute the products in
exchange for some combination of up-front payments, royalty payments, and
milestone payments. Management cannot be certain that we, or our licensees, will
successfully initiate or complete Phase I, Phase II, or Phase III testing of our
product candidates within any specific time period, if at all.
Furthermore, the FDA or the IRB or the IND sponsor may suspend clinical trials
at any time on various grounds, including a finding that the patients are being
exposed to an unacceptable health risk.
Concurrent
with clinical trials and pre-clinical studies, we also must develop information
about the chemistry and physical characteristics of the drug and finalize a
process for manufacturing the product in accordance with GMP requirements.
The manufacturing process must be capable of consistently producing quality
batches of the experimental drug, and management must develop methods for
testing the quality, purity, and potency of the final experimental drugs.
Additionally, appropriate packaging must be selected and tested.
The
results of the drug development efforts and pre-clinical and clinical studies
are then submitted to the FDA as part of an NDA for approval of the marketing
and commercial shipment of the product. The FDA reviews each NDA submitted
and may request additional information, rather than accepting the NDA for
filing. In this event, the application must be resubmitted with the additional
information included. The resubmitted application is also subject to
review before the FDA accepts it for filing. Once the FDA accepts the NDA
for filing, the agency begins an in-depth review of the NDA. The FDA has
substantial discretion in the approval process and may disagree with our, or our
licensees’, interpretation of the data submitted.
The
review process may be significantly extended by FDA requests for additional
information or clarification regarding information already provided. Also,
as part of this review, the FDA may refer the application to an appropriate
advisory committee, typically a panel of clinicians, for review, evaluation and
a recommendation. The FDA is not bound by the recommendation of the
advisory committee. Manufacturing establishments are also
subject to inspections prior to NDA approval to assure compliance with GMPs and
with manufacturing commitments made in the relevant marketing
application.
Under the
Prescription Drug User Fee Act (“PDUFA”), submission of an NDA with clinical
data requires payment of a fee to the FDA, which is adjusted annually. For
fiscal year 2009, that fee is $1,247,200. In return, the FDA assigns a
goal (often months) for standard NDA reviews from acceptance of the application
to the time the agency issues its “complete response,” in which the FDA may
approve the NDA, deny the NDA if the applicable regulatory criteria are not
satisfied, or require additional clinical data. Even if this data is submitted,
the FDA may ultimately decide that the NDA does not satisfy the criteria for
approval. If the FDA approves the NDA, the product becomes available for
physicians to prescribe. Even if the FDA approves the NDA, the agency may
decide later to withdraw product approval if compliance with regulatory
standards is not maintained or if safety problems are recognized after the
product reaches the market. The FDA may also require post-marketing
studies, also known as Phase IV studies, as a condition of approval to develop
additional information regarding the efficacy and safety of a product. In
addition, the FDA requires surveillance programs to monitor approved products
that have been commercialized, and the agency has the power to require changes
in labeling or to prevent further marketing of a product based on the results of
these post-marketing programs.
18
Satisfaction
of the above FDA requirements or requirements of state, local and foreign
regulatory agencies typically takes several years. Government regulation
may delay or prevent marketing of potential products for a considerable period
of time and impose costly procedures upon our activities. Management
cannot be certain that the FDA or any other regulatory agency will grant
approval for our lead product G-202 (or any other products we may develop,
acquire, or in-license) under development on a timely basis, if at all.
Success in preclinical or early-stage clinical trials does not assure success in
later-stage clinical trials. Data obtained from preclinical and clinical
activities are not always conclusive and may be susceptible to varying
interpretations that could delay, limit or prevent regulatory approval.
Even if a product receives regulatory approval, the approval may be
significantly limited to specific indications or uses. Further, even after
regulatory approval is obtained, later discovery of previously unknown problems
with a product may result in restrictions on the product or even complete
withdrawal of the product from the market. Delays in obtaining, or
failures to obtain regulatory approvals would have a material adverse effect on
our business.
Any
products manufactured or distributed by us, or our licensees, pursuant to the
FDA clearances or approvals are subject to pervasive and continuing regulation
by the FDA, including record-keeping requirements, reporting of adverse
experiences with the drug, submitting other periodic reports, drug sampling and
distribution requirements, notifying the FDA and gaining its approval of certain
manufacturing or labeling changes, complying with certain electronic records and
signature requirements, and complying with the FDA promotion and advertising
requirements. Failure to comply with these regulations could result, among
other things, in suspension of regulatory approval, recalls, suspension of
production or injunctions, seizures, or civil or criminal sanctions.
Management cannot be certain that our present or future subcontractors or
licensees will be able to comply with these regulations and other FDA regulatory
requirements.
Our
product candidates are also subject to a variety of state laws and regulations
in those states or localities where our lead product G-202 (and any other
products we may develop, acquire, or in-license) will be marketed. Any
applicable state or local regulations may hinder our ability to market our lead
product G-202 (and any other products we may develop, acquire, or in-license) in
those states or localities. In addition, whether or not FDA approval has
been obtained, approval of a pharmaceutical product by comparable governmental
regulatory authorities in foreign countries must be obtained prior to the
commencement of clinical trials and subsequent sales and marketing efforts in
those countries. The approval procedure varies in complexity from country
to country, and the time required may be longer or shorter than that required
for FDA approval. We may incur significant costs to comply with these laws
and regulations now or in the future.
The FDA’s
policies may change, and additional government regulations may be enacted which
could prevent or delay regulatory approval of our potential products.
Moreover, increased attention to the containment of health care costs in the
U.S. and in foreign markets could result in new government regulations that
could have a material adverse effect on our business. Management cannot
predict the likelihood, nature or extent of adverse governmental regulation that
might arise from future legislative or administrative action, either in the U.S.
or abroad.
Other
Regulatory Requirements
The U.S.
Federal Trade Commission and the Office of the Inspector General of the U.S.
Department of Health and Human Services (“HHS”) also regulate certain
pharmaceutical marketing practices. Also, reimbursement practices and HHS
coverage of medicine or medical services are important to the success of
procurement and utilization of our product candidates, if they are ever approved
for commercial marketing.
We are
also subject to numerous federal, state and local laws relating to such matters
as safe working conditions, manufacturing practices, environmental protection,
fire hazard control, and disposal of hazardous or potentially hazardous
substances. We may incur significant costs to comply with these laws and
regulations now or in the future. Management cannot assure you that any
portion of the regulatory framework under which we currently operate will not
change and that such change will not have a material adverse effect on our
current and anticipated operations.
Employees
As of
September 30, 2009 we employed 2 individuals who are also our 2 executive
officers.
PROPERTIES
Our
executive offices
are located at 2511 N Loop 1604 W, Suite 204, San Antonio, TX, 78258. We lease
this facility consisting of approximately 850 square feet, for $1425 per month
Our lease expires on September 30, 2012.
We also
rent a virtual office at 12100 Wilshire Blvd, 8th Floor, Los Angeles, CA 90025
to maintain a business presence in that state and for meetings with participants
who are located within travel distance to Los Angeles so as not to require
travel exclusively to our executive office in San Antonio. This contract carries
forward on a month by month basis at a charge of $210 per month.
The
aforesaid properties are in good condition and we believe they will be suitable
for our purposes for the next 12 months. There is no affiliation between us or
any of our principals or agents and our landlords or any of their principals or
agents.
19
MANAGEMENT’S
DISCUSSION AND ANALYSIS OF FINANCIAL
CONDITION
AND RESULTS OF OPERATIONS
This
Management’s Discussion and Analysis of Financial Condition and Results of
Operations section (“MD&A”) contains statements and information about
management’s view of our future expectations, plans and prospects, that
constitute forward-looking statements. These statements are subject to risks and
uncertainties that could cause actual results and events to differ materially
from those we anticipate. We undertake no obligation to update any
forward-looking statements, whether as a result of new information, future
events or otherwise.
Our
MD& is organized as follows:
|
•
|
Overview —
Discussion of our business and plan of operations, overall analysis
of financial and other highlights affecting the company in order to
provide context for the remainder of
MD&A.
|
|
•
|
Significant Accounting
Policies — Accounting policies that we believe are important to
understanding the assumptions and judgments incorporated in our reported
financial results and forecasts.
|
|
•
|
Results of Operations
— Analysis of our financial results comparing: (i) three and
six months ended June 30 of 2009 to the comparable period in 2008; and
(ii) our financial results for 2008 to
2007.
|
|
•
|
Liquidity and Capital
Resources — A discussion of our financial condition and
potential sources of liquidity.
|
The
various sections of this MD&A contain a number of forward-looking
statements. Words such as “expects,” “goals,” “plans,” “believes,” “continues,”
“may,” and variations of such words and similar expressions are intended to
identify such forward-looking statements. In addition, any statements that refer
to projections of our future financial performance, our anticipated growth and
trends in our businesses, and other characterizations of future events or
circumstances are forward-looking statements. Such statements are based on our
current expectations and could be affected by the uncertainties and risk factors
described throughout this filing and particularly in the Risk Factors section of
this Prospectus. Our actual results may differ materially.
Overview
We are a
development stage company focused on the development of targeted cancer
therapeutics for the treatment of cancerous tumors, including breast, prostate,
bladder, and kidney cancer. Our operations are based in San Antonio,
TX.
Management's
Plan of Operation
We are
pursuing a business plan related to the development of targeted cancer
therapeutics for the treatment of cancerous tumors, including breast, prostate,
bladder, and kidney cancer. We are considered to be in development
stage as defined by SFAS No. 7, “Accounting and reporting by
Development Stage Enterprises”.
Business
Strategy
Our
business strategy is to develop a series of therapeutics based on our
target-activated pro-drug technology platform and bring them through Phase I/II
clinical trials. At that point, we plan to license the rights to further
development of the drug candidates to major pharmaceutical companies. We believe
that major pharmaceutical companies see significant value in drug candidates
that have passed one or more phases of clinical trials, and these organizations
have the resources and expertise to finalize drug development and market the
drugs.
Plan
of Operation
We have
made significant progress in key areas such as drug manufacture, toxicology, and
pre-clinical activities for our lead compound G-202.
For the
manufacture of G-202, we have secured a stable supply of source material (Thapsia garganica seeds) from
which thapsigargin is isolated, have a sole source agreement with a European
supplier, Thapsibiza, SL, and have obtained the proper import permits from the
USDA for these materials. We have also identified a clinically and commercially
viable formulation for G-202 and have manufactured sufficient G-202 to supply
our Phase I clinical needs.
On June
23, 2009, we submitted our first IND for G-202 to FDA. On September
4, 2009, we received approval from the FDA for our IND in order to commence
clinical trials. Although we have received approval from the FDA to
commence trials, the outcome of the trials is uncertain and, if we are unable to
satisfactorily complete such trials, or if such trials yield unsatisfactory
results, we will be unable to commercialize our proposed products. Over the next
twelve months we plan to focus on clinical trials of G-202 in cancer
patients.
20
Additionally,
we will continue to protect our intellectual property position particularly with
regard to the outstanding claims contained within the core PSMA-pro-drug patent
application in the United States. We will also continue to prosecute the claims
contained in our other patent applications in the United States.
We
anticipate that during 2009, and much of 2010, we will be engaged in conducting
the Phase I clinical trial of G-202, and, if appropriate, extension into a Phase
II clinical trial of G-202. The purpose of a Phase I study of G-202 is to
evaluate safety, understand the pharmacokinetics (the process by which a
compound is absorbed, distributed, metabolized, and eliminated by the body) of
the drug candidate in humans, and to determine an appropriate dosing regime for
the subsequent clinical studies. We currently plan to conduct the Phase I study
in refractory cancer patients (those who have relapsed after former treatments)
with any type of solid tumors. This strategy is intended to facilitate
enrollment and perhaps give us a glimpse of safety across a wider variety of
patients. We expect to enroll up to 30 patients in this Phase I study at: (i)
Johns Hopkins Oncology Center (Michael Carducci, MD as Principal Investigator);
and (ii) University of Wisconsin Comprehensive Cancer Center (George Wilding, MD
as Principal Investigator). We are currently negotiating contracts for conduct
of the Phase I studies. The final terms of the contracts have not yet
been determined.
Assuming
successful completion of the Phase I clinical trial, we expect to conduct a
Phase II clinical trial to determine the therapeutic efficacy of G-202 in cancer
patients. Although we believe that G-202 will be useful across a wide variety of
cancer types, it is usually most efficient and medically prudent to evaluate a
drug candidate in a single tumor type within a single trial. It is currently too
early in the pre-clinical development process to determine which single tumor
type will be evaluated, but we expect that over 40 patients will be required for
an appropriate evaluation over a total time span of 18 months.
We have
identified 4 pro-drug candidates: G-202, G-114, G-115 and Ac-GKAFRR-L12ADT. At
this time, we are engaged solely in the development of G-202. It is anticipated
that the development of the remaining candidates will not commence until we have
sufficient resources to devote to their development and in all likelihood this
will not occur until after the development of G-202.
From
inception through June 30, 2009, the vast majority of costs incurred have been
devoted to G-202. We estimate that we have incurred costs of approximately
$235,000 related to G-114, G-115 and Ac-GKAFRR-L12ADT. All of these costs were
incurred prior to December 2007, at which time we began focusing solely on
G-202. The balance of our costs, aggregating approximately $4,124,000, was
incurred in the development of G-202. For the six months ended June 30, 2009 and
2008, approximately $934,000 and $519,000, respectively, was incurred in the
development of G-202.
It is
estimated that the development of G-202 will occur as follows:
It is
estimated that the Phase I clinical trial will cost approximately $2,000,000 and
will be completed in the fourth quarter of 2010.
Phase II
clinical studies will cost an additional $4,200,000 and will be completed in the
fourth quarter of 2011.
We
anticipate that we will license G-202 to a third party during Phase
I/II. In the event we are not able to license G-202, we will proceed
with Phase III Clinical trials. We estimate that Phase III Clinical
trials will cost approximately $25,000,000 and will be completed in the fourth
quarter of 2014. If all goes as planned, we may expect marketing approval in the
second half of 2015 with an additional $3,000,000 spent to get the NDA approved.
We do not expect material net cash inflows before late 2015. The
Phase III estimated costs are subject to major revision simply because we have
not yet entered clinical testing of our drug in patients. The estimates will
become more refined as we obtain clinical data.
At this
time, we have suspended the development of our other drug candidates, G-114,
G-115 and Ac-GKAFRR-L12ADT. As a result we are unable to reasonably estimate the
nature, timing and estimated costs and completion dates of those projects at
this time.
We
currently have budgeted $2,395,000 in cash expenditures for the twelve month
period following the date of this report, including (1) $1,095,000 to cover our
projected general and administrative expense during this period; and (2)
$1,300,000 for research and development activities. Our cash on hand as of
September 30, 2009 is sufficient to fund our operations for the next thirteen
months after which time we will need to undertake additional
financings.
The
amounts and timing of our actual expenditures may vary significantly from our
expectations depending upon numerous factors, including our results of
operation, financial condition and capital requirements. Accordingly, we will
retain the discretion to allocate the available funds among the identified uses
described above, and we reserve the right to change the allocation of available
funds among the uses described above.
Significant
Accounting Policies
Our
financial statements have been prepared in accordance with accounting principles
generally accepted in the United States of America. The preparation of these
financial statements requires management to make estimates and assumptions that
affect the reported amounts of assets, liabilities, revenues and expenses.
Note 1 of the Notes to Financial Statements describes the significant
accounting policies used in the preparation of the financial statements. Certain
of these significant accounting policies are considered to be critical
accounting policies, as defined below. We do not believe that there have been
significant changes to our accounting policies during the six months ended June
30, 2009, as compared to those policies disclosed in the December 31, 2008
financial statements except as disclosed in the notes to financial
statements.
21
A
critical accounting policy is defined as one that is both material to the
presentation of our financial statements and requires management to make
difficult, subjective or complex judgments that could have a material effect on
our financial condition and results of operations. Specifically, critical
accounting estimates have the following attributes: 1) we are required to
make assumptions about matters that are highly uncertain at the time of the
estimate; and 2) different estimates we could reasonably have used, or
changes in the estimate that are reasonably likely to occur, would have a
material effect on our financial condition or results of
operations.
Estimates
and assumptions about future events and their effects cannot be determined with
certainty. We base our estimates on historical experience and on various other
assumptions believed to be applicable and reasonable under the circumstances.
These estimates may change as new events occur, as additional information is
obtained and as our operating environment changes. These changes have
historically been minor and have been included in the financial statements as
soon as they became known. Based on a critical assessment of our accounting
policies and the underlying judgments and uncertainties affecting the
application of those policies, management believes that our financial statements
are fairly stated in accordance with accounting principles generally accepted in
the United States, and present a meaningful presentation of our financial
condition and results of operations. We believe the following critical
accounting policies reflect our more significant estimates and assumptions used
in the preparation of our financial statements:
Use of
Estimates — These financial statements have been prepared in accordance
with accounting principles generally accepted in the United States and,
accordingly, require management to make estimates and assumptions that affect
the reported amounts of assets and liabilities at the date of the financial
statements and the reported amounts of revenues and expenses during the
reporting period. Specifically, our management has estimated the expected
economic life and value of our licensed technology, our net operating loss for
tax purposes and our stock, option and warrant expenses related to compensation
to employees and directors and consultants. Actual results could differ from
those estimates.
Fair Value of
Financial Instruments — For certain of our financial instruments,
including accounts payable, accrued expenses and notes payable, the carrying
amounts approximate fair value due to their relatively short
maturities.
Cash and
Equivalents — Cash equivalents are comprised of certain highly liquid
investments with maturity of three months or less when purchased. We maintain
our cash in bank deposit accounts, which at times, may exceed federally insured
limits. We have not experienced any losses in such accounts.
Intangible and
Long-Lived Assets — We follow SFAS No. 144, "Accounting for Impairment of
Disposal of Long-Lived Assets ", which established a "primary asset"
approach to determine the cash flow estimation period for a group of assets and
liabilities that represents the unit of accounting for a long lived asset to be
held and used. Long-lived assets to be held and used are reviewed for impairment
whenever events or changes in circumstances indicate that the carrying amount of
an asset may not be recoverable. The carrying amount of a long-lived asset is
not recoverable if it exceeds the sum of the undiscounted cash flows expected to
result from the use and eventual disposition of the asset. Long-lived assets to
be disposed of are reported at the lower of carrying amount or fair value less
cost to sell. We have not recognized any impairment losses.
Research and
Development Costs — Research and development costs include expenses
incurred by the Company for research and development of therapeutic agents for
the treatment of cancer and are charged to operations as incurred.
Stock Based
Compensation — We account for our share-based compensation under the
provisions of FASB Statement No. 123(R), “Share-Based Payment”, (“FAS
123R”). We adopted FAS 123R as of January 1, 2006, using the modified
prospective application method. Prior to January 1, 2006 we applied the
provisions of FAS 123, “Accounting for Stock-Based
Compensation”.
Fair Value of
Financial Instruments — Our
short-term financial instruments, including cash, accounts payable and other
liabilities, consist primarily of instruments without extended maturities, the
fair value of which, based on management’s estimates, reasonably approximate
their book value. The fair value of long term convertible notes is based on
management estimates and reasonably approximates their book value after
comparison to obligations with similar interest rates and maturities. The fair
value of the Company’s derivative instruments is determined using option pricing
models.
Recent
Accounting Pronouncements
In
December 2007, the FASB issued SFAS No. 160, “Non-controlling Interests in
Consolidated Financial Statements-an amendment of ARB No. 51” (SFAS 160).
SFAS 160 requires that non-controlling (or minority) interests in subsidiaries
be reported in the equity section of the company’s balance sheet, rather than in
a mezzanine section of the balance sheet between liabilities and equity. SFAS
160 also changes the manner in which the net income of the subsidiary is
reported and disclosed in the controlling company’s income statement. SFAS 160
also establishes guidelines for accounting or changes in ownership percentages
and for deconsolidation. SFAS 160 is effective for financial statements for
fiscal years beginning on or after December 15, 2008 and interim periods within
those years. The adoption of SFAS 160 did not have a material impact on our
financial position, results of operations or cash flows.
22
In March
2008, the FASB issued SFAS No. 161, “Disclosures about Derivative Instruments and
Hedging Activities-an amendment of FASB Statement No. 133” (SFAS 161).
SFAS 161 requires enhanced disclosures about an entity’s derivative and hedging
activities and thereby improves the transparency of financial reporting. SFAS
161 is effective for financial statements issued for fiscal years and interim
periods beginning after November 15, 2008, with early application encouraged.
The adoption of SFAS 161 did not have a material impact on our financial
position, results of operations or cash flows.
Effective
January 1, 2009, the Company adopted the Financial Accounting Standards Board's
Staff Position (FSP) on the Emerging Issues Task Force (EITF) Issue No. 03-6-1,
“Determining Whether
Instruments Granted in Share-Based Payment Transactions are Participating
Securities”. The FSP required that all unvested share-based payment
awards that contain nonforfeitable rights to dividends should be included in the
basic Earnings Per Share (EPS) calculation. This standard did not
affect the financial position or results of operations.
In April
2009, the FASB issued FSP FAS No. 115-2 and FAS No. 124-2, “Recognition and Presentation of
Other-Than-Temporary Impairments” (“FSP FAS No. 115-2”). FSP FAS No.
115-2 provides guidance in determining whether impairments in debt securities
are other than temporary, and modifies the presentation and disclosures
surrounding such instruments. This FSP is effective for interim periods ending
after June 15, 2009, but early adoption is permitted for interim periods ending
after March 15, 2009. The adoption of this standard during the second quarter of
2009 had no impact on the Company’s financial position or results of
operations.
In April
2009, the FASB issued FSP FAS No. 157-4, “Determining Fair Value When the
Volume and Level of Activity for the Asset or Liability Have Significantly
Decreased and Identifying Transactions That Are Not Orderly” (“FSP FAS
No. 157-4”). FSP FAS No. 157-4 provides additional guidance in determining
whether the market for a financial asset is not active and a transaction is not
distressed for fair value measurement purposes as defined in SFAS No. 157, “Fair
Value Measurements.” FSP FAS No. 157-4 is effective for interim periods ending
after June 15, 2009, but early adoption is permitted for interim periods ending
after March 15, 2009. The adoption of this standard during the second quarter of
2009 had no impact on the Company’s financial position or results of
operations.
In April
2009, the FASB issued FSP FAS No. 107-1 and APB 28-1, “Interim Disclosures about Fair Value
of Financial Instruments” (“FSP FAS No. 107-1 and APB 28-1”). This FSP
amends FASB Statement No. 107, “Disclosures about Fair Values of Financial
Instruments,” to require disclosures about fair value of financial instruments
in interim financial statements as well as in annual financial statements. APB
28-1 also amends APB Opinion No. 28, “Interim Financial Reporting,” to require
those disclosures in all interim financial statements. This standard is
effective for interim periods ending after June 15, 2009, but early adoption is
permitted for interim periods ending after March 15, 2009. The adoption of this
standard during the second quarter of 2009 had no impact on the Company’s
financial position or results of operations.
In May
2009, the FASB issued SFAS No. 165, “Subsequent Events”
(“SFAS No. 165”). SFAS No. 165 establishes general standards
of accounting for and disclosure of events that occur after the balance sheet
date but before financial statements are issued or are available to be issued.
SFAS No. 165 will be effective for interim and annual financial
periods ending after June 15, 2009. The Company adopted
SFAS No. 165 during the three months ended June 30, 2009 and
evaluated subsequent events through the issuance date of the financial
statements. SFAS No. 165 had no impact on the Company’s financial
position or results of operations.
In June
2009, the FASB issued SFAS No. 166, “Accounting for Transfers and
Servicing of Financial Assets – an amendment of SFAS Statement No. 140”
(“SFAS No. 166”). SFAS No. 166 will require more information about transferred
of financial assets and where companies have continuing exposure to the risks
related to transferred financial assets. SFAS 166 is effective at the start of a
company’s first fiscal year beginning after November 15, 2009, or January 1,
2010 for companies reporting earnings on a calendar-year basis.
In June 2009, the FASB
issued SFAS No. 167, “Amendments to FASB Interpretation
No. 46(R)” (“SFAS No. 167”). SFAS No. 167 will change how a company
determines when an entity that is insufficiently capitalized or is not
controlled through voting (or similar rights) should be consolidated. Under SFAS
No. 167, determining whether a company is required to consolidate an entity will
be based on, among other things, an entity's purpose and design and a company's
ability to direct the activities of the entity that most significantly impact
the entity's economic performance. SFAS 167 is effective at the start of a
company’s first fiscal year beginning after November 15, 2009, or January 1,
2010 for companies reporting earnings on a calendar-year basis.
In June
2009, the FASB issued SFAS No. 168, “The FASB Accounting Standards
Codification and the Hierarchy of Generally Accepted Accounting Principals – a
replacement of FAS No.162” (“SFAS No. 168”). This statement establishes
the Codification as the source of authoritative U.S. accounting and reporting
standards recognized by the FASB for use in the preparation of financial
statements of nongovernmental entities that are presented in conformity with
GAAP. Rules and interpretive releases of the SEC under authority of federal
securities laws are also sources of authoritative GAAP for SEC registrants. The
Codification was the result of a project of FASB to organize and simplify all
authoritative GAAP literature into one source. This statement is effective for
interim reporting and annual periods ending after September 15, 2009.
Accordingly, the Company will adopt SFAS No. 168 during the quarter ended
September 30, 2009. The Company does not anticipate this statement to have
a material impact on its consolidated financial position, results of operations
or cash flows.
23
Other
recent accounting pronouncements issued by the FASB (including its Emerging
Issues Task Force), the AICPA, and the SEC did not, or are not believed by
management to, have a material impact on the Company's present or future
financial statements.
Result
of Operations
Our
results of operations have varied significantly from year to year and quarter to
quarter and may vary significantly in the future.
Second
Quarter of 2009 Compared to Second Quarter of 2008
Revenue
The
company did not have revenue for the three months ended June 30, 2009 and 2008,
respectively. We do not anticipate any revenues for 2009.
Operating
Expenses
Operating
expense totaled $856,244 and $506,939 for the three months ended June 30, 2009
and 2008, respectively. The increase in operating expenses is the
result of the following factors.
|
Three Months Ended June
30,
|
||||||||
|
2009
|
2008
|
|||||||
|
Operating
expenses
|
||||||||
|
General
and administrative expenses
|
$
|
231,255
|
$
|
138,745
|
||||
|
Research
and development
|
624,989
|
368,194
|
||||||
|
Total
expense
|
$
|
856,244
|
$
|
506,939
|
||||
General
and Administrative Expenses
G&A
expenses totaled $231,255 for the three months ended June 30, 2009 compared to
$138,745 for the same period of 2008. The increase of $92,510 or 67%
for the three months ended June 30, 2009 compared to the comparable period in
2008 was primarily attributable to increases of approximately $27,000 in
compensation and consulting expense, $12,000 in insurance expense and $47,000 in
professional fees.
Research
and Development Expenses
Research
and development expenses totaled $624,989 for the three months ended June 30,
2009 compared to $368,194 for the same period of 2008. The increase
of $256,795 or 70% for the three months ended June 30, 2009 compared to the
comparable period in 2008 was attributable to an increase of approximately
$268,000 in costs associated with manufacture and other expenses related to our
lead drug.
Our
research and development expenses consist primarily of expenditures for
toxicology and other studies, manufacturing, and compensation and consulting
costs.
Under the
planning and direction of key personnel, we expect to outsource all of our GLP
preclinical development activities (e.g., toxicology) and GMP manufacturing and
clinical development activities to CROs and CMOs. Manufacturing will be
outsourced to organizations with approved facilities and manufacturing
practices.
Other
Expenses
Other
expenses totaled $118,348 and $1,605 for the three months ended June 30, 2009
and 2008, respectively.
|
Three Months Ended June 30,
|
||||||||
|
2009
|
2008
|
|||||||
|
Other
expense:
|
||||||||
|
Finance
Cost
|
$
|
(5,155
|
)
|
$
|
—
|
|||
|
Change
in fair value of derivative liability
|
(110,326
|
)
|
—
|
|||||
|
Interest
income (expense)
|
(2,867
|
)
|
(1,605
|
)
|
||||
|
Total
other expenses
|
$
|
(118,348
|
)
|
$
|
(1,606
|
)
|
||
Finance
Cost
Finance
Cost totaled $5,155 for the three months ended June 30 compared to $0 for the
same period of 2008. This cost consists of the amortization of debt
discount.
24
Change
in fair value of derivative liability
Change in
fair value of derivative liability totaled $110,326 for the three months ended
June 30, 2009 compared to $0 for the same period of 2008. The increase of
$110,326 for the three months ended June 30, 2009 compared to the comparable
period in 2008 was the result of a change to the accounting treatment of our
issued and outstanding warrants which contain certain anti-dilution
provisions.
At June
30, 2009 we recalculated the fair value of our warrants subject to derivative
accounting and have determined that their fair value at June 30, 2009 is
$1,833,703. The fair value of the warrants was determined using the
Black-Scholes method based on the following
assumptions: (1) risk free interest rate of 0.875%;
(2) dividend yield of 0%; (3) volatility factor of the expected market
price of our common stock of 175%; and (4) an expected life of the
warrants of 2 years. We have recorded an expense of $110,326 during the
three months ended June 30, 2009 related to the change in fair value during that
period.
Interest
expense
Interest
expense totaled $2,867 for the three months ended June 30, 2009 compared to
$1,605 for the same period of 2008. The increase of $1,262 for the three months
ended June 30, 2009 compared to the comparable period in 2008 was attributable
to an increase in debt outstanding in 2009, partially offset by interest earned
on deposits.
Six
months ended June 30, 2009 Compared to Six months ended June 30,
2008
Revenue
The
company did not have revenue for the six months ended June 30, 2009 and 2008,
respectively. We do not anticipate any revenues for 2009.
Operating
Expenses
Operating
expense totaled $1,365,463 and $1,032,531 for the six months ended June 30, 2009
and 2008, respectively. The increase in operating expenses is the
result of the following factors.
|
Six Months Ended June
30,
|
||||||||
|
2009
|
2008
|
|||||||
|
Operating
expenses
|
||||||||
|
General
and administrative expenses
|
$
|
430,972
|
$
|
513,536
|
||||
|
Research
and development
|
934,491
|
518,995
|
||||||
|
Total
expense
|
$
|
1,365,463
|
$
|
1,032,531
|
||||
General
and Administrative Expenses
G&A
expenses totaled $430,972 for the six months ended June 30, 2009 compared to
$513,536 for the same period of 2008. The decrease of $82,564 or 16%
for the six months ended June 30, 2009 compared to the comparable period in 2008
was primarily attributable to a decrease of approximately $199,000 in
compensation and consulting expense, offset by increases of approximately
$92,000 in professional fees and $11,000 in travel expenses.
Research
and Development Expenses
Research
and development expenses totaled $934,491 for the six months ended June 30, 2009
compared to $518,995 for the same period of 2008. The increase of
$415,496 or 80% for the six months ended June 30, 2009 compared to the
comparable period in 2008 was attributable to increases of approximately $88,000
in compensation expense and approximately $343,000 in costs associated with
manufacture and other expenses related to our lead drug.
Our
research and development expenses consist primarily of expenditures for
toxicology and other studies, manufacturing, and compensation and consulting
costs.
Under the
planning and direction of key personnel, we expect to outsource all of our GLP
preclinical development activities (e.g., toxicology) and GMP manufacturing and
clinical development activities to CROs and CMOs. Manufacturing will be
outsourced to organizations with approved facilities and manufacturing
practices.
Other
Expenses
Other
expenses totaled $1,166,679 and $3,211 for the six months ended June 30, 2009
and 2008, respectively.
25
|
Six Months Ended June
30,
|
||||||||
|
2009
|
2008
|
|||||||
|
Other
expense:
|
||||||||
|
Finance
Cost
|
$
|
(478,093
|
)
|
$
|
—
|
|||
|
Change
in fair value of derivative liability
|
(683,111
|
)
|
—
|
|||||
|
Interest
income (expense)
|
(5,475
|
)
|
(3,211
|
)
|
||||
|
Total
other expenses
|
$
|
(1,166,679
|
)
|
$
|
(3,211
|
)
|
||
Finance
Cost
Finance
Cost totaled $478,093 for the six months ended June 30, 2009 compared to $0 for
the same period of 2008. The increase of $478,093 for the six months ended June
30, 2009 compared to the comparable period in 2008 was primarily attributable to
a $415,976 charge for the fair value of additional warrants issued when the
anti-dilution provisions in our warrants issued during the July and August 2008
financing were triggered plus a $51,864 charge for the fair value of
additional warrants issued as consideration for the extension of the maturity
dates of notes payable. The balance of the cost consists of the amortization of
debt discount
Change
in fair value of derivative liability
Change in
fair value of derivative liability totaled $683,111 for the six months ended
June 30, 2009 compared to $0 for the same period of 2008. The increase of
$683,111 for the six months ended June 30, 2009 compared to the comparable
period in 2008 was the result of a change to the accounting treatment of our
issued and outstanding warrants which contain certain anti-dilution
provisions.
In June
2008, the FASB issued Emerging Issues Task Force No. 07-5 (EITF 07-5),
Determining Whether an
Instrument (or Embedded Feature) is Indexed to an Entity’s Own Stock.
EITF 07-5 requires entities to evaluate whether an equity-linked
financial instrument (or embedded feature) is indexed to its own stock by
assessing the instrument’s contingent exercise provisions and settlement
provisions. Instruments not indexed to their own stock fail to meet the scope
exception of Statement of Financial Accounting Standards No. 133, Accounting for
Derivative Instruments and Hedging Activities, paragraph 11(a), and
should be classified as a liability and marked-to-market. The Company has
assessed its outstanding equity-linked financial instruments and has concluded
that warrants issued during 2008 with a fair value of $734,617 on
January 1, 2009 are required to be reclassified from equity to a liability.
Fair value at January 1, 2009 was determined using the Black-Scholes method
based on the following assumptions: (1) risk free interest rate
of 0.875%; (2) dividend yield of 0%; (3) volatility factor of the
expected market price of our common stock of 144%; and (4) an expected
life of the warrants of 2 years.
As a
result of our February offering described in Note 2 to our financials statements
contained in this Report, the anti-dilution provisions in our warrants issued
during the July and August 2008 financing were triggered. These
anti-dilution provisions resulted in the exercise price of these warrants being
reduced from $2.00 from $1.50. Additionally, we are obligated to
issue holders of these warrants an additional 506,754 warrants, and we are
obligated to file a registration statement for the common stock underlying such
warrants pursuant to the registration rights agreement entered into in
connection with the July and August 2008 financing. We have recorded the fair
value of the additional warrants as a derivative liability upon issue. The fair
value of the warrants of $415,976 was determined using the Black-Scholes method
based on the following assumptions: (1) risk free interest rate
of 0.875%; (2) dividend yield of 0%; (3) volatility factor of the
expected market price of our common stock of 149%; and (4) an expected
life of the warrants of 2 years.
At June
30, 2009 we recalculated the fair value of our warrants subject to derivative
accounting and have determined that their fair value at June 30, 2009 is
$1,833,703. The fair value of the warrants was determined using the
Black-Scholes method based on the following
assumptions: (1) risk free interest rate of 0.875%;
(2) dividend yield of 0%; (3) volatility factor of the expected market
price of our common stock of 175%; and (4) an expected life of the
warrants of 2 years. We have recorded an expense of $683,111 during the six
months ended June 30, 2009 related to the change in fair value during that
period.
Interest
expense
Interest
expense totaled $5,475 for the six months ended June 30, 2009 compared to $3,211
for the same period of 2008. The increase of $2,264 for the six months ended
June 30, 2009 compared to the comparable period in 2008 was attributable to an
increase in debt outstanding in 2009, partially offset by interest earned on
deposits.
26
Year
Ended December 31, 2008 Compared to the Year Ended December 31,
2007
Revenue
The
company did not have revenue for the years ended December 31, 2008 and
2007 We do not anticipate any revenues for 2009.
|
|
Years ended December 31,
|
|||||||
|
|
2008
|
2007
|
||||||
|
|
|
|||||||
|
Operating
expenses:
|
|
|
||||||
|
General
and administrative expenses
|
$ | 854,294 | $ | 389,799 | ||||
|
Research
and development
|
2,432,991 | 294,440 | ||||||
|
Total
operating expenses
|
3,287,285 | 684,239 | ||||||
|
Loss
from operations
|
$ | (3,287,285 | ) | $ | (684,239 | ) | ||
Operating
losses increased from $684,239 in 2007 to $3,287,285 in 2008. The increase of
$2,603,046 was the result of an increase of $464,495 in general and
administrative expenses, from $389,799 in 2007 to $854,294 in 2008, and an
increase in research and development expenses of $2,138,551, from $294,440 in
2007 to $2,432,991 in 2008. The increase in expenses is directly attributable to
the availability of cash resulting from our private placements in 2008 and late
2007, enabling us to implement our business plan.
Research
and Development Expenses
Research
and development expenses for the years ended December 31, 2008 and 2007 were
$2,432,991 and $294,440 respectively. The increase in 2008 is directly
attributable to the availability of cash resulting from our private placements
in 2008 and late 2007 and consequent implementation of the development program
for G-202. The 2008 increase results primarily from increases in expenditures
for toxicology and other studies and manufacturing.
Our
research and development expenses consist primarily of expenditures for
toxicology and other studies, manufacturing, and compensation and consulting
costs.
Under the
planning and direction of key personnel, we expect to outsource all of our GLP
preclinical development activities (e.g., toxicology) and GMP manufacturing and
clinical development activities to CROs and CMOs. Manufacturing will be
outsourced to organizations with approved facilities and manufacturing
practices.
Prior to
the assignment of the patents to GenSpera in 2008, we had an exclusive option to
license 5 issued patents and 3 patent applications pending worldwide. The
previous owner of the intellectual property, John Hopkins University, agreed to
assign the patents underlying the technology to our co-founders (the “Assignee
Co-Founders”) in return for their assumption of future patent fees and costs,
and patent attorney fees and costs, associated with all of the assigned
technology. In exchange for us continuing to pay for these future costs, the
Assignee Co-Founders entered into world-wide, exclusive option agreements with
us. Therefore, we have continued to pay these costs as we have used the
technology prior to the actual assignment.
General
and Administrative Expenses
General
and administrative expenses for the years ended December 31, 2008 and 2007 were
$854,294 and $389,799 respectively. The increase in 2008 results from increases
in compensation and consulting expense of approximately $106,000, professional
fees of approximately $215,000, rent expense of approximately $38,000, insurance
of approximately $22,000 and various other expenses of approximately $83,000 and
is directly attributable to the availability of cash resulting from our private
placements in 2008 and late 2007, enabling us to implement our business
plan.
Other
Expense
Other
expense for the years ended December 31, 2008 and 2007 was $38,976 and $6,960,
respectively, consisting of interest expense and finance costs. The increase in
2008 results primarily from a charge of $30,160 related to a penalty for the
late filing of our registration statement.
Net
Loss
Net
losses for the years ended December 31, 2008 and 2007 were $3,326,261 and
$691,199 respectively, resulting from the expenses described
above.
27
Liquidity
and Capital Resources
Since our
inception, we have financed our operations through the private placement of our
securities and loans from Craig Dionne, our Chief Executive Officer. At
September 30, 2009 we have cash on hand of approximately $2,614,000. Our
currently average monthly cash burn rate is approximately $195,000. We
anticipate that our available cash will be sufficient to finance most of our
current activities for at least the next thirteen months from July 31,
2009, assuming we do not engage in an extraordinary transaction or
otherwise face unexpected events or contingencies.
|
Six Months Ended June
30,
|
||||||||
|
2009
|
2008
|
|||||||
|
Cash
& Cash Equivalents
|
$
|
2,100,212
|
$
|
251,781
|
||||
|
Net
cash used in operating activities
|
$
|
(1,172,930
|
)
|
$
|
(654,486
|
)
|
||
|
Net
cash used in investing activities
|
—
|
(184,168
|
)
|
|||||
|
Net
cash provided by financing activities
|
2,738,852
|
500,000
|
||||||
Net
Cash Used in Operating Activities
In our
operating activities we used $1,172,930 for the six months ended June 30, 2009
compared to $654,486 for the same period of 2008. An increase in net loss of
approximately $1,496,000 was partially offset by increases in noncash expenses
of approximately $954,000.
Net
Cash Used in Investing Activities
In our
investment activities we used $0 for the six months ended June 30, 2009 compared
to $184,168 for the same period of 2008. The decrease in investment activities
of $184,168 for the six months ended June 30, 2009 compared to the comparable
period in 2008 was attributable to the $184,168 cost associated with acquisition
of key intellectual property in 2008.
Net
Cash Provided by Financing Activities
During
the six months ended June 30, 2009 we raised approximately $2,739,000 through
the sale of common stock units. During the comparable period of 2008 we received
proceeds of $500,000 upon the exercise of warrants.
Listed
below are key financing transactions we have entered
into.
|
•
|
During
November of 2007, we sold an aggregate of 1,300,000 common shares
resulting in gross proceeds of
$650,000.
|
|
•
|
During
March of 2008, we issued 1,000,000 common shares upon the exercise of
outstanding warrants which resulted in gross proceeds to us of
$500,000.
|
|
•
|
During
July and August of 2008, we sold an aggregate of 2,320,000 units resulting
in gross proceeds of $2,320,000.
|
|
•
|
In
February and April of 2009, we sold 500,000 units resulting in gross
proceeds of approximately $750,000.
|
|
•
|
In
June and July of 2009, we sold 2,025,344 units resulting in gross proceeds
of approximately $3,038,000.
|
|
•
|
In
September of 2009, we sold 160,002 units resulting in gross proceeds of
approximately $240,000.
|
We have
incurred significant operating losses and negative cash flows since inception.
We have not achieved profitability and may not be able to realize sufficient
revenue to achieve or sustain profitability in the future. We do not expect to
be profitable in the next several years, but rather expect to incur additional
operating losses. We have limited liquidity and capital resources and must
obtain significant additional capital resources in order to sustain our product
development efforts, for acquisition of technologies and intellectual property
rights, for preclinical and clinical testing of our anticipated products,
pursuit of regulatory approvals, acquisition of capital equipment, laboratory
and office facilities, establishment of production capabilities, for general and
administrative expenses and other working capital requirements. We rely on cash
balances and the proceeds from the offering of our securities, exercise of
outstanding warrants and grants to fund our operations.
The
source, timing and availability of any future financing will depend principally
upon market conditions, interest rates and, more specifically, on our progress
in our exploratory, preclinical and future clinical development programs.
Funding may not be available when needed, if at all, or on terms acceptable to
us. Lack of necessary funds may require us, among other things, to delay, scale
back or eliminate some or all of our research and product development programs,
planned clinical trials, and/or our capital expenditures or to license our
potential products or technologies to third parties.
28
LEGAL
PROCEEDINGS
As of the
date of this prospectus, there are no material pending legal or governmental
proceedings relating to our company or properties to which we are a party, and
to our knowledge there are no material proceedings to which any of our
directors, executive officers or affiliates are a party adverse to us or which
have a material interest adverse to us.
MANAGEMENT
Directors
The
following sets forth the current members of our board of directors (“Board”) and
information concerning their ages and background. All directors hold office
until the next annual meeting of stockholders or until their respective
successors are elected, except in the case of death, resignation or
removal:
|
Name
|
|
Principal Occupation
|
|
Age
|
|
Director
Since
|
|
Craig
A. Dionne, PhD
|
Chief
Executive Officer, Chief Financial Officer, President and Director of
GenSpera
|
52
|
11/03
|
|||
|
John
M. Farah, Jr., PhD
|
Vice
President Intercontinental Operations at Cephalon (NASDAQ:
CEPH)
|
57
|
02/08
|
|||
|
Scott
Ogilvie
|
President
and CEO of Gulf Enterprises International, Ltd.
|
55
|
02/08
|
Craig A. Dionne,
PhD, age 52, has over 21 years experience in the pharmaceutical industry,
including direct experience of identifying promising oncology treatments and
bringing them through the clinic. For example, he served for five years as VP
Discovery Research at Cephalon, Inc. where he was responsible for its oncology
and neurobiology drug discovery and development programs. Dr. Dionne has also
recently served as EVP at the Prostate Cancer Research Foundation. In addition
to extensive executive experience, Dr. Dionne’s productive scientific career has
led to 6 issued patents and co-authorship of many scientific
papers.
John M. Farah,
Jr., Ph.D., age 57, is VP Intercontinental Operations at Cephalon
(Nasdaq:CEPH), which he joined in 1992 after six years as a discovery research
scientist at G.D. Searle and Co. He is responsible for ensuring corporate
support and managing sales performance of international partners in the Americas
and Asia Pacific with specific growth initiatives for Cephalon in China and
Japan. His prior roles included the responsibility for promoting and negotiating
R&D and commercial alliances with multinational and regional pharmaceutical
firms, and responsibilities in scientific affairs, product licensing and
academic collaborations. He currently serves on the board of directors of Aeolus
Pharmaceuticals (AOLS.OB).
Scott
Ogilvie, age 55, is President and CEO of Gulf Enterprises
International, Ltd, (“Gulf”) a company that brings strategic partners, expertise
and investment capital to the Middle East and North Africa. He has held this
position since August of 2006. Mr. Ogilvie also serves as Chief Operating
Officer of CIC Group, Inc., an investment manager, a position he has held for
the last five years. He began his career as a corporate and securities lawyer
with Hill, Farrer & Burrill, and has extensive public and private corporate
board experience in finance, real estate, and technology companies. Mr. Ogilvie
currently serves on the board of directors of Neuralstem, Inc. (NYSE AMEX:CUR),
Innovative Card Technologies, Inc. (OTCBB:INVC) and Preferred Voice Inc,
(OTCBB:PRFV).
Executive
Officers and Significant Employees
The
following sets forth our current executive officers and information concerning
their age and background:
|
Name
|
|
Position
|
|
Age
|
|
Position
Since
|
|
Craig
A. Dionne, PhD
|
Chief
Executive Officer, Chief Financial Officer and President
|
52
|
11/03
|
|||
|
Russell
Richerson, PhD
|
Chief
Operating Officer and Secretary
|
57
|
07/08
|
Craig A. Dionne, PhD. –
See Bio in Directors Section
Russell
Richerson, PhD, age 57, has over 25 years experience in the
Biotechnology/Diagnostics industry, including 11 years at Abbott Laboratories in
numerous management roles. Most recently, he has served as Vice President of
Diagnostic Research and Development at Prometheus Laboratories (2001-2004) and
then as Chief Operating Officer of the Molecular Profiling Institute
(2005-2008).
29
Family
Relationships
There are
no family relationships between any director, executive officer, or person
nominated or chosen by the registrant to become a director or executive
officer.
Code
of Ethics
We have
adopted a "Code of Ethics” that applies to our principal executive officer,
principal financial officer, principal accounting officer or controller, or
persons performing similar functions. A copy of our code can be
viewed on our website at www.genspera.com.
Committees
The board
of directors currently does not have an audit, nomination, or compensation
committee. Due to our size and limited resources and employees, our board has
determined that the functions of such committees, including the compensation
committee, will be undertaken by the entire board. Upon securing additional
financing and the hiring of additional employees, the board of directors
anticipates the creation of free standing committees. Executive compensation is
determined by the entire board.
Independent
Directors
For
purposes of determining independence, the Company has adopted the definition of
independence as contained in NASDAQ Market Place Rules 4200. Pursuant to the
definition, the Company has determined that Messrs. Ogilvie and Farah qualify as
independent.
Executive
Compensation
Summary
Compensation
The
following table sets forth information for our most recently completed fiscal
year concerning the compensation of (i) the Principal Executive Officer and
(ii) all other executive officers of GenSpera, Inc. who earned over
$100,000 in salary and bonus during the last most recently completed fiscal
years ended December 31, 2008 and 2007 (together the “Named
Executive Officers”). No other employees earned a salary over
$100,000 in the last completed fiscal years.
|
Name and
principal
position
(a)
|
Year
(b)
|
Salary
($)
(c)
|
Bonus
($)
(d)
|
Stock
Awards
($)
(e)
|
Option
Award
($)
(f)(2)
|
Nonequity
Incentive
Plan
compensation
($)
(g)
|
Non-qualified
deferred
compensation
earning
($)
(h)
|
All other
Compensation
($)
(i)(1)
|
Total
($)
(j)
|
||||||||||||
|
Craig
Dionne, PhD
|
|||||||||||||||||||||
|
Chief
Executive
|
|||||||||||||||||||||
|
Officer/Chief
Financial Officer
|
2008
|
$ | 240,000 |
|
|
|
|
|
|
$ | 240,000 | ||||||||||
|
2007
|
$ | 20,000 | (1) |
|
|
|
|
|
|
$ | 20,000 | ||||||||||
|
Russell
Richerson, PhD
|
|||||||||||||||||||||
|
Chief
Operating Officer
|
|||||||||||||||||||||
|
Secretary
|
2008
|
$ | 100,000 | (2) |
|
|
|
|
|
|
$ | 100,000 | |||||||||
|
(1)
|
During
2007 Dr. Dionne forwent compensation until such time as we secured
financing. In December of 2007, upon securing financing, Dr. Dionne began
receiving a salary.
|
|
(2)
|
During
2008, Dr. Richerson forwent second quarter compensation in the amount of
$50,000. Dr. Richerson began receiving a salary in July of
2008
|
Employment
Agreements and Arrangements
Mr.
Craig Dionne
On
September 2, 2009, we entered into the following written agreements in
connection with Mr. Dionne’s employment: (i) an employment agreement; (ii) a
severance agreement; (iii) a proprietary information, inventions and competition
agreement; and (iv) an indemnification agreement.
30
(i) Pursuant
to the terms of the employment agreement, we will employ Craig Dionne as our
Chief Executive Officer for a term of 5 years. As compensation for
his services, Mr. Dionne shall receive a base salary of $240,000 per
year. In addition, Mr. Dionne is eligible to receive annual and
discretionary bonuses as determined by the Board. Mr. Dionne is also
entitled to receive certain payments and acceleration of outstanding equity
awards in the event his employment is terminated. As part of the
agreement, Mr. Dionne was also granted options to purchase 1,000,000 shares of
Common Stock with an exercise price of $1.65 per share. The options
were issued pursuant to the 2009 Executive Compensation Plan and vest upon the
achievement of certain milestones as more fully described in the employment
agreement which is incorporated herein by reference. The options have
a term of 7 years.
(ii) The
severance agreement provides for certain payments, as described below, in the
event Mr. Dionne’s employment is terminated in connection with a change in
control.
(iii) The
proprietary information, inventions and competition agreement requires Mr.
Dionne to maintain the confidentiality of our intellectual property as well as
the assignment of any inventions made by Mr. Dionne during his
employment. The agreement also limits Mr. Dionne’s ability to compete
within certain fields of interest, as defined in the agreement, for a period of
18 months following the end of his employment.
(iv) The
indemnification agreement provides for the indemnification and defense of Mr.
Dionne, in the event of litigation, to the fullest extent permitted by
law. We have adopted the form of indemnification agreement for use
with our other executive officers, employees and directors.
As part
of the agreements, Mr. Dionne shall be entitled to the following payments in the
event of termination:
|
|
|
|
|
Accelerated
Vesting of
|
|
|||||||||||||||
|
Officer
|
Salary
|
Bonus
|
Health
|
Options*
|
Total
|
|||||||||||||||
|
Craig
Dionne
|
||||||||||||||||||||
|
Terminated
without cause (1)
|
$ | 720,000 | (2) | $ | 0 | (3) | $ | 54,000 | (4) | $ | 0 | (5) | $ | 774,000 | ||||||
|
Terminated,
change of control (6)
|
$ | 1,440,000 | $ | 0 | (3) | $ | 54,000 | (4) | $ | 0 | (5) | $ | 1,494,000 | |||||||
|
Disability
|
$ | 240,000 | — | — | — | $ | 240,000 | |||||||||||||
|
Other
|
— | — | — | — | — | |||||||||||||||
|
(1)
|
Also
includes termination by Mr. Dionne with Good
Reason
|
|
(2)
|
Represents
36 months of Mr. Dionne’s base
salary.
|
|
(3)
|
There
has been no bonus established for the current
year.
|
|
(4)
|
Represents
36 months of Mr. Dionne’s monthly health care reimbursement of
$1,500.
|
|
(5)
|
There
does not presently exist a market for the Company’s
securities. In the event of termination, Mr. Dionne’s 1,000,000
common stock options would vest and would remain exercisable for their
term.
|
|
(6)
|
Assumes
termination without cause or good
reason.
|
The
foregoing summary of Mr. Dionne’s: (i) employment agreement; (ii)
severance agreement; (iii) proprietary information, inventions and competition
agreement; and (iv) indemnification agreement are qualified in their
entirety by reference to the full text of the agreements which have been filed
with the SEC as Exhibits hereto, and which are incorporated herein in their
entirety by reference.
Mr.
Russell Richerson
On
September 2, 2009, we entered into the following written agreements in
connection with Mr. Richerson’s employment: (i) an employment agreement; (ii) a
proprietary information, inventions and competition agreement; and (iii) an
indemnification agreement.
(i) Pursuant
to the terms of the employment agreement, we will employ Russell Richerson as
our Chief Operating Officer for a term of 3 years. As
compensation for his services, Mr. Richerson shall receive a base salary of
$200,000 per year. In addition, Mr. Richerson is eligible to receive
annual and discretionary bonuses as determined by the Board. Mr.
Richerson is also entitled to receive certain payments and acceleration of
outstanding equity awards in the event his employment is terminated and as
described below. As part of the agreement, Mr. Richerson was also
granted options to purchase 775,000 shares of Common Stock with an exercise
price of $1.50 per share. The options were issued pursuant to the
2009 Executive Compensation Plan and vest upon the achievement of certain
milestones as more fully described in the agreement. The options have
a term of 7 years.
(ii) The
proprietary information, inventions and competition agreement requires Mr.
Richerson to maintain the confidentiality of our intellectual property as well
as the assignment of any inventions made by Mr. Richerson during his
employment. The agreement also limits Mr. Richerson’s ability to
compete within certain fields of interest, as defined in the agreement, for a
period of 18 months following end of his employment.
31
(iii) The
indemnification agreement provides for the indemnification and defense of Mr.
Richerson, in the event of litigation, to the fullest extent permitted by
law.
As part
of the agreements, Mr. Richerson shall be entitled to the following payments in
the event of termination:
|
|
|
|
|
Accelerated
Vesting of
|
|
|||||||||||||||
|
Officer
|
Salary
|
Bonus
|
Health
|
Options*
|
Total
|
|||||||||||||||
|
Russell
Richerson
|
||||||||||||||||||||
|
Terminated
without cause (1)
|
$ | 300,000 | (2) | $ | 0 | (3) | $ | 27,000 | (4) | $ | 0 | (5) | $ | 327,000 | ||||||
|
Terminated,
change of control
|
— | — | — | — | — | |||||||||||||||
|
Disability
|
$ | 200,000 | — | — | — | $ | 200,000 | |||||||||||||
|
Other
|
— | — | — | — | — | |||||||||||||||
|
(1)
|
Also
includes termination by Mr. Richerson with Good
Reason
|
|
(2)
|
Represents
18 months of Mr. Richerson’s base
salary.
|
|
(3)
|
There
has been no bonus established for the current
year.
|
|
(4)
|
Represents
18 months of Mr. Richerson’s monthly health care reimbursement of
$1,500.
|
|
(5)
|
There
does not presently exist a market for the Company’s
securities. In the event of termination, Mr. Richerson’s
775,000 common stock options would vest and would remain exercisable for
their term.
|
The
foregoing summary of Mr. Richerson’s: (i) employment agreement; (ii)
proprietary information, inventions and competition agreement; and (iii)
indemnification agreement are qualified in their entirety by
reference to the full text of the agreements which have been filed with the SEC
as Exhibits hereto, and which are incorporated herein in their entirety by
reference.
Outstanding
Equity Awards at Fiscal Year-End
There
were no unexercised options; stock that has not vested; equity incentive; or
awards that were outstanding as of the end of the last completed fiscal year
with regard to our Named Executive Officer.
Director
Compensation
The
following table summarizes the compensation for our board of directors for the
fiscal year ended December 31, 2008:
|
Name
|
Fees Earned
or Paid in
Cash
($)
|
Stock
Awards
($)
|
Option
Awards
($)
|
Non-Equity
Incentive Plan
Compensation
($)
|
Nonqualified
Deferred
Compensation
Earnings
($)
|
All Other
Compensation
($)
|
Total
($)
|
|||||||||||||
|
(a)
|
(b)
|
(c)
|
(d)
|
(e)
|
(f)
|
(g)
|
(h)
|
|||||||||||||
|
Scott
Ogilvie
|
$ | 25,225 | (1) |
|
|
$ | 25,225 | |||||||||||||
|
|
|
|
||||||||||||||||||
|
John
Farah
|
$ | 18,052 | (2) |
|
|
$ | 18,052 | |||||||||||||
|
|
|
|
||||||||||||||||||
|
Richard
Burgoon
|
$ | 50,000 | (3) | 16,548 | (4)(5) |
|
|
4,203
|
(5) | $ | 70,751 | |||||||||
|
(1)
|
Includes:
(i) options to purchase 100,000 common shares at $0.50 per share of which
50,000 options vested immediately with the balance vesting quarterly over
a two year period commencing on March 31, 2008;
and (ii) fully vested options to purchase 15,000
common shares at $1.00 per share. As of December 31,
2008, Mr. Ogilvie had an aggregate of 115,000 options, 90,000 of which
were vested.
|
32
|
(2)
|
Includes
options to purchase 100,000 common shares at $0.50 per share of which
50,000 options vested immediately with the balance vesting quarterly over
a two year period commencing on March 31, 2008. As of
December 31, 2008, Mr. Farah had an aggregate of 100,000 options, 75,000
of which were vested.
|
|
(3)
|
Includes
100,000 shares of common stock issued as compensation for
services.
|
|
(4)
|
Includes
options to purchase 100,000 common shares at $0.50 per share of which
50,000 options vested immediately with the balance vesting quarterly over
a two year period commencing on March 31, 2008. As of
December 31, 2008, Mr. Burgoon had an aggregate of 100,000 options, 75,000
of which were vested.
|
|
(5)
|
On
September 30, 2008, Mr. Burgoon resigned as a director. As a
result, we have reported his compensation as follows: (i) Option Awards of
$16,548 attributed to 68,750 options vested through his termination date;
and (ii) Other Compensation of $4,203 consisting of 31,250 options that
will vest as compensation for services rendered subsequent to his
resignation.
|
EQUITY
COMPENSATION PLAN INFORMATION
The
following table sets forth information with respect to our equity compensation
plans as of December 31, 2008.
|
(a)
|
(b)
|
(c)
|
||||||||||
|
Number of Securities
to be Issued
upon Exercise of
Outstanding
Options, Warrants
and Rights
|
Weighted-Average
Exercise Price of
Outstanding
Options,
Warrants and
Rights
|
Number of Securities
Remaining Available for
Future Issuance under
Equity Compensation Plans
(Excluding Securities
Reflected in Column (a))
|
||||||||||
|
Equity
compensation plans approved by security holders
|
|
|
|
|||||||||
|
2007
Stock Plan, as amended
|
575,000 | $ | 0.51 | 925,000 | (1) | |||||||
|
Equity
compensation plans not approved by security holders
|
N/A | N/A | N/A | |||||||||
|
Total
|
575,000 | $ | 0.00 | 925,000 | (1) | |||||||
(1) Our
2007 Stock Plan, as amended, is considered an “Evergreen
Plan.” Pursuant to the terms of the plan, on the 1 st day of each
fiscal year, the number of shares issuable under the plan is increased to
1,500,000. As of January 1, 2009, the number of securities remaining
available for future issuance under the plan was 1,500,000.
2009
Executive Compensation Plan
On
September 2, 2009, our Board of Directors approved the 2009 Executive
Compensation Plan (“2009 Plan”). The 2009 Plan permits the granting
of up to 1,775,000 shares of our common stock through the issuance of Incentive
Stock Options, Nonstatutory Stock Options, Restricted Stock, Stock Appreciation
Rights, Restricted Stock Units, Performance Units, Performance Shares and Other
Stock Based Awards to our executive officers. As of the date of
this prospectus, we have granted stock options to Messrs Dionne and Richerson to
purchase 1,775,000 common shares. Accordingly, there are no
additional shares available for future grants.
The
purpose of our 2009 Plan is to advance the interests of GenSpera and our
stockholders by attracting, retaining and rewarding persons performing services
for us and to motivate such persons to contribute to our growth and
profitability.
Issuance of
Awards. The issuance of awards under our 2007 Plan is at the discretion
of the Committees, which has the authority to determine the persons to whom any
awards shall be granted and the terms, conditions and restrictions applicable to
any award. Under our 2009 Plan, we may grant stock options and restricted stock
to employees, directors and consultants. Our 2009 Plan authorizes the issuance
of up to 1,775,000 shares of our common stock for the foregoing awards. As of
September 30, 2009, we had made awards under our 2009 Plan of 1,775,000 and
there are 0 shares available for future awards.
Exercise Price
for Options. The exercise price of Nonqualified Stock Options shall not
be less than 85% of the fair market value per share on the date of grant. The
exercise price per share for Incentive Stock Option grants must be no less than
100% of the fair market value per share on the date of grant. The exercise price
per share for an incentive stock option grant to an employee who, at the time of
grant, owns stock representing more than 10% of the voting power of all classes
of stock of GenSpera or any parent or subsidiary, must be no less than 110% of
the fair market value per share on the date of grant.
33
Payment of
Exercise Price. Generally, the option exercise price may be paid in cash,
by check, by cashless exercise, by net exercise or by tender or attestation of
ownership of shares having a fair market value not less than the exercise price
and that either (A) have been owned by the optionee for more than six
months and not used for another exercise by tender or attestation, or
(B) were not acquired, directly or indirectly, from us.
Exercisability
and Vesting. At the time an award is granted, the Committee must fix the
period within which the award may be exercised and determine any conditions that
must be satisfied before the award may be exercised. The Committee
may accelerate the exercisability of any or all outstanding options at any time
for any reason.
Term of
Options. The maximum term of an option granted under our 2009 Plan is ten
years.
Transferability
of Awards. Grants are nontransferable by the grantee other than by will
or by the laws of descent and distribution and are exercisable during the
grantee’s lifetime only by the grantee.
Change in
Control. Our 2009 Plan provides that in the event of our merger with or
into another corporation, the sale of substantially all of our assets, or the
sale or exchange of more than 50% of our voting stock, each outstanding award
shall be assumed or an equivalent award substituted by the surviving,
continuing, successor or purchasing corporation or a parent thereof. The
Committee may also deem an award assumed if the award confers the right to the
award-holder to receive, for each share of stock subject to an award immediately
prior to the change in control, the consideration that a stockholder is entitled
on the effective date of the change in control. Upon a change in control, all
outstanding options shall automatically accelerate and become fully exercisable
and all restrictions and conditions on all outstanding restricted stock grants
shall immediately lapse.
The
summary information related to our 2009 Plan is not included in the summary
table above.
CERTAIN
RELATIONSHIPS AND RELATED TRANSACTIONS
We
believe the terms and conditions set forth in such agreements are reasonable and
customary for transactions of these types. Information regarding
disclosure of an employment relationship or transaction involving an executive
officer and any related compensation solely resulting from that employment
relationship or transaction is incorporated by reference from the section of
this Prospectus entitled “Executive
Compensation.”
Information
regarding disclosure of compensation to a director is incorporated by reference
from the section of this Prospectus entitled “Director
Compensation.”
|
|
·
|
Between
December 2003 and December 2006, we entered into five convertible notes
with Craig Dionne, our Chief Executive Officer, pursuant to which we have
borrowed an aggregate of $155,000. The notes bear an interest rate of 4.2%
and mature at various dates through December 6, 2011. Interest accrued
through February 29, 2008 was $15,859. On March 7, 2008 we issued 31,718
shares of common stock as payment of this
amount.
|
|
|
·
|
On
November 10, 2006, we issued options to purchase an aggregate of 150,000
common shares to the Company’s officers, directors and certain
shareholders as compensation for services provided to the Company. The
options vested at grant and have a term of 10 years. The options were
granted as follows: (i) 111,250 to Mr. Burgoon, a former director; (ii)
8,875 to Mr. Dionne, a director and Chief Executive Officer; (iii) 10,000
to Mr. Richerson, our Chief Operating Officer; and (iv) 10,000 to each of
Messrs. Isaacs and Denmeade, each an advisor to the Company and a
beneficial owner of 5% or more of the Company’s common shares. The options
had an exercise price of $0.01 and a term of 10 years. The
options were exercised in 2007 pursuant to their
terms
|
|
|
·
|
On
May 14, 2007, our Board of Directors approved the acceleration of
outstanding common stock options that were previously issued to Messrs
Isaacs and Denmeade, our Scientific Advisors, as compensation. As a result
of the acceleration, 10,500 common stock options because immediately
vested. The options have an exercise price of $0.0016 and a term of 10
years.
|
|
|
·
|
On
January 7, 2008, we granted 100,000 shares of common stock, valued at
$50,000, to a Mr. Burgoon, a former director, as compensation for serving
on the board. The shares vested upon
grant.
|
34
|
|
·
|
On
February 1, 2008, we granted each of Messrs Isaacs and Denmeade, our
Scientific Advisors, common stock purchase options to purchased 60,000
shares, as compensation for joining the Company’s scientific advisory
board. The options have an exercise price of $0.50 per share. The
options vest in equal installments quarterly over a period of three years
commencing March 31, 2008, and lapse if unexercised on January
31, 2018.
|
|
|
·
|
In
March of 2008, we granted options to purchase an aggregate of 300,000
(100,000 each) common shares to our directors Messrs Farah and Ogilvie as
well as our former director Mr. Burgoon. Each director received options to
purchase 100,000 common shares at an exercise price of $0.50 per share.
Each director’s grant vests 50,000 upon grant with the balance vesting
quarterly over a period of two years commencing March 31, 2008, and lapses
if unexercised on April 1, 2018.
|
|
|
·
|
On
March 11, 2008 we exercised our option to license certain intellectual
property from Messrs Isaacs and Denmeade. As consideration for the option
exercise, we paid each of Isaacs and Denmeade: (i) $37,995.90 which they
immediately transferred to John Hopkins University as repayment of past
patent costs; and (ii) $18,997 as a “gross-up” to pay for relevant tax
consequences of the option exercise
payment.
|
|
|
·
|
In
April of 2008, Messrs Isaacs and Denmeade transferred to the Company their
interest in the intellectual property licensed on March 11,
2008.
|
|
|
·
|
In
October of 2008, we granted options to purchase an aggregate of
15,000 common shares to our director Scott Ogilvie at an exercise
price of $1.00 per share. The options vested on the date of grant and
lapse if unexercised on October 16,
2018.
|
|
|
·
|
On
February 17, 2009, we entered into a modification with Craig Dionne, our
Chief Executive Officer and Chairman with regard to our 4% Convertible
Promissory Note issued to Mr. Dionne in the amount of
$35,000. Pursuant to the modification, Mr. Dionne agreed to
extend the maturity date of the Note from December 2, 2008 to December 2,
2009. Mr. Dionne had previously deferred repayment of the
note. As consideration for the modification, we issued Mr.
Dionne a common stock purchase warrant entitling Mr. Dionne to purchase
11,000 shares of our common stock at a per share purchase price of
$1.50. The warrant has a five year term and contains certain
anti-dilution provisions requiring us to adjust the exercise price and
number of shares upon the occurrence of a stock split, stock dividends or
stock consolidation.
|
35
PRINCIPAL
STOCKHOLDERS
The
following table sets forth, as of September 30, 2009, information regarding
beneficial ownership of our capital stock by:
|
·
|
each
person, or group of affiliated persons, known by us to be the beneficial
owner of 5% or more of any class of our voting
securities;
|
|
·
|
each
of our current directors and
nominees;
|
|
·
|
each
of our current named executive officers;
and
|
|
·
|
all
current directors and named executive officers as a
group.
|
Beneficial
ownership is determined according to the rules of the SEC. Beneficial ownership
means that a person has or shares voting or investment power of a security and
includes any securities that person or group has the right to acquire within 60
days after the measurement date. This table is based on information supplied by
officers, directors and principal stockholders. Except as otherwise indicated,
we believe that each of the beneficial owners of the common stock listed below,
based on the information such beneficial owner has given to us, has sole
investment and voting power with respect to such beneficial owner’s shares,
except where community property laws may apply.
|
Common Stock
|
|
|||||||||
|
Name and Address of Beneficial Owner(1)
|
Shares
|
Shares
Underlying
Convertible
Securities(2)
|
|
Total
|
|
Percent of
Class(2)
|
||||
|
Directors
and named executive officers
|
|
|||||||||
|
Craig
Dionne, PhD
|
2,438,662
|
959,432
|
3,398,094
|
20.9
|
%
|
|||||
|
Russell
B. Richerson, PhD(3)
|
925,000
|
512,500
|
1,437,500
|
9.1
|
%
|
|||||
|
John
M. Farah, PhD
|
|
93,750
|
93,750
|
*
|
||||||
|
Scott
Ogilvie
|
|
102,500
|
102,500
|
*
|
||||||
|
All
directors and executive officers as a group
(4 persons)
|
3,363,662
|
1,668,182
|
5,031,844
|
29.7
|
%
|
|||||
|
Beneficial
Owners of 5% or more
|
|
|||||||||
|
John
T. Isaacs, PhD(4)
|
1,271,528
|
35,000
|
1,306,528
|
8.5
|
%
|
|||||
|
Samuel
R. Denmeade, M.D (5)
|
1,271,528
|
35,000
|
1,306,528
|
8.5
|
%
|
|||||
|
*
|
Less than one
percent.
|
|
(1)
|
Except
as otherwise indicated, the persons named in this table have sole voting
and investment power with respect to all shares of common stock shown as
beneficially owned by them, subject to community property laws where
applicable and to the information contained in the footnotes to this
table. Unless otherwise indicated, the address of the beneficial owner is
GenSpera, Inc., 2511 N Loop 1604 W, Suite 204, San Antonio, TX
78258.
|
|
(2)
|
Pursuant
to Rules 13d-3 and 13d-5 of the Exchange Act, beneficial ownership
includes any shares as to which a shareholder has sole or shared voting
power or investment power, and also any shares which the shareholder has
the right to acquire within 60 days, including upon exercise of common
shares purchase options or warrant. There are 15,292,281 shares of common
stock issued and outstanding as of September 30,
2009.
|
|
(3)
|
5050
East Gleneagles Drive, Tucson, AZ
85718
|
|
(4)
|
13638
Poplar Hill Road, Phoenix, MD 21131
|
|
(5)
|
5112
Little Creek Drive, Ellicott City, MD
21043
|
SELLING
STOCKHOLDERS
This
prospectus relates to the offering and sale, from time to time, of up to
10,985,791 shares of our common stock held by the stockholders named in the
tables below (“Selling Stockholders”), which amount, includes common shares
issuable upon the exercise of warrants held by the Selling
Stockholders. Of the shares being sold, 10,528,520 have been
previously registered and this prospectus will constitute a post effective
amendment to the respective registration statements.
36
Initial
Registration
September
2, 2009 Offering
The
majority of the shares being initially registered in this prospectus relate to
our September 2, 2009 offering. Pursuant to the terms of the
offering, we sold an aggregate of 160,000 units resulting in gross proceeds of
$240,000 or $1.50 per unit. Each unit consists of: (i) one share of
common stock; and (ii) one half common stock purchase warrant. The
warrants have a term of five years and allow the investors to purchase our
common shares at a price per share of $3.00. The warrants also
contain anti-dilution protection in the event of stock splits, stock dividends
and other similar transactions. The warrants are callable by us in
the event our shares are publically traded in the future and certain price and
volume conditions are met. We paid a total of $23,100 in fees and
expenses incurred in connection with the offering. We also issued
warrants to purchase 12,267, common shares, with identical terms to the warrant,
as a partial finder’s fee in connection with the
offering. We are registering herein the: (i) 160,002
common shares, and (ii) 92,269 common shares underlying the warrants, issued
pursuant to the offering.
Consultant
Warrants and Common Shares
In
addition to the shares issued or to be issued in connection with the September
2, 2009, we are registering 185,000 shares issued to consultants and service
providers in exchange for services and reimbursement of expenses. We
are hereby registering: (i) 25,000 common shares, (ii) and 160,000 common shares
underlying warrants.
The
Selling Stockholders may exercise their warrants at any time in their sole
discretion. All of the Selling Stockholders named below acquired their common
stock and warrants directly from us in private transactions.
Set forth
below is information, to the extent known to us, setting forth the name of each
Selling Shareholder and the amount and percentage of Common Stock owned by each
(including shares that can be acquired on the exercise of outstanding warrants)
prior to the offering, the shares to be sold in the offering, and the amount and
percentage of Common Stock to be owned by each (including shares that can be
acquired on the exercise of outstanding warrants) after the offering assuming
all shares are sold. The footnotes provide information about persons who have
investment voting power for the Selling Shareholders and about material
transactions between the Selling Shareholders and the Company.
The
Selling Stockholders may sell all or some of the shares of common stock they are
offering, and may sell shares of our common stock otherwise than pursuant to
this prospectus. The table below assumes that each Selling Stockholder exercises
all of its warrants and sells all of the shares issued upon exercise thereof,
and that each selling stockholder sells all of the shares offered by it in
offerings pursuant to this prospectus, and does not acquire any additional
shares. We are unable to determine the exact number of shares that will actually
be sold or when or if these sales will occur.
The
Selling Stockholders may sell all, some or none of their shares in this
offering. See “Plan of Distribution.”
The total
number of common shares sold under this prospectus may be adjusted to reflect
adjustments due to stock dividends, stock distributions, splits, combinations or
recapitalizations with regard to the common stock and warrants.
Unless
otherwise stated below in the footnotes, to our knowledge, no Selling
Stockholder nor any affiliate of such stockholder: (i) has held any position or
office with, been employed by or otherwise has had any material relationship
with us or our affiliates during the three years prior to the date of this
prospectus; or (ii) is a broker-dealer, or an affiliate of a
broker-dealer.
We may
amend or supplement this prospectus from time to time in the future to update or
change this list and shares which may be resold.
We may
amend or supplement this prospectus from time to time in the future to update or
change this list and shares which may be resold.
|
Common
Shares Owned Before Sale (1)
|
Common
Shares Owned
After
Sale (2)
|
|||||||||||||||||||||||||||
|
Held
Outright
|
Warrants/
Options
|
Amount
|
%
of
class
|
Shares
being
registered
|
Amount
|
%
of Class
|
||||||||||||||||||||||
|
The
Alec and Evelyn Sabo Trust(3)(5)
|
133,334 | 66,667 | 200,001 | 1 | % | 200,001 | - | * | ||||||||||||||||||||
|
Walter
H. Bass, LLC (3)(6)
|
20,000 | 32,934 | 52,934 | * | 42,267 | 10,667 | * | |||||||||||||||||||||
|
Robert
Scherne(3)
|
3,334 | 51,667 | 55,001 | * | 5,001 | 50,000 | * | |||||||||||||||||||||
|
Craig
Petralia(3)
|
1,667 | 834 | 2,501 | * | 2,501 | - | * | |||||||||||||||||||||
|
Todd
Shapiro(3)
|
1,667 | 834 | 2,501 | * | 2,501 | - | * | |||||||||||||||||||||
|
The
Verrazano Group, LLC(4)(7)
|
184,000 | 184,000 | 1 | % | 100,000 | 84,000 | * | |||||||||||||||||||||
|
Cataracta
Aps(4)(8)
|
60,000 | 60,000 | * | 60,000 | - | * | ||||||||||||||||||||||
|
Treskerby
Clinical Solutions, LLC(4)(9)
|
25,000 | 25,000 | * | 25,000 | - | * | ||||||||||||||||||||||
|
Total
|
185,002 | 396,936 | 581,938 | 4 | % | 437,271 | 144,667 | 1 | % | |||||||||||||||||||
|
(1)
|
Pursuant
to Rules 13d-3 and 13d-5 of the Exchange Act, beneficial ownership
includes any common shares as to which a shareholder has sole or shared
voting power or investment power, and also any common shares which the
shareholder has the right to acquire within 60 days, including upon
exercise of common shares purchase options or warrants. There were
15,292,281 common shares outstanding as of September 30,
2009.
|
|
(2)
|
Assumes
the sale of all common shares initially registered pursuant to this
registration statement but does not include shares previously registered
which may be part of the Previously Registered Shares contained
below.
|
37
|
(3)
|
Issued
pursuant to September 2, 2009
Offering.
|
|
(4)
|
Consultant
Warrants and Common Shares.
|
|
(5)
|
Evelyn
P. Sabo, Trustee, has voting and dispositive control with respect to the
securities being offered.
|
|
(6)
|
Walter
Bass has voting and dispositive control with respect to the securities
being offered.
|
|
(7)
|
Steven
Chizzik, Managing Director has voting and dispositive control with respect
to the securities being offered.
|
|
(8)
|
Soren
Brogger Christensen, Ph.D. has voting and dispositive control with respect
to the securities being offered. Dr. Christensen is one of our
founders.
|
|
(9)
|
Suzanne
Wilson has voting and dispositive control with respect to the securities
being offered.
|
Previously
Registered Shares
The
shares previously registered related primarily to: (i) shares underlying
warrants issued as a result of anti-dilution provisions (“Anti-Dilution
Warrants”); (ii) shares and shares underlying warrants issued during our
February 2009 offering (“2009 February Offering”); (iii) shares and shares
underlying warrants issued during our June and July 2009 offering (“2009 June
Offering”); (iv) shares and shares underlying warrants issued to consultants for
services provided (“Consultant Warrants”); and (v) shares and shares underlying
warrants issued during our July and August 2008 offering (“2008 July
Offering”).
Anti-Dilution
Warrants
In our
2008 Offering, we sold an aggregate of 2,320,000 units resulting in gross
proceeds of $2,320,000 or $1.00 per unit. Each unit consisted of: (i) 1 share of
common stock; and (ii) ½ common stock purchase warrant. The warrants have a term
of 5 years and an exercise price of $2.00 per shares subject to certain
anti-dilution adjustments. During February of 2009 the Company
completed the 2009 February Offering and sold an aggregate of $750,000 units at
a price per unit of $1.50. As a result of the 2009 February Offering,
certain anti-dilution provisions in the warrants held by investors who
participated in our July and August, 2008 offering were
triggered. The result of these anti-dilution provisions was that the
exercise price of warrants issued during our July and August 2008 offering were
reduced from $2.00 to $1.50; and we issued the investors in the offering
warrants to purchase an additional 506,754 common shares at $1.50. We
are including herein the shares underlying 394,187 of the addition 506,754
warrants which we issued.
2009
February Offering
In
February and April of 2009 we completed the offering of 500,000 units resulting
in gross proceeds of $750,000 or $1.50 per unit. Each unit consists
of: (i) one share of common stock; and (ii) one half common stock purchase
warrant. The warrants have a term of five years and allow the
investors to purchase our common shares at a price per share of
$3.00. The Warrants also contain anti-dilution protection in the
event of stock splits, stock dividends and other similar
transactions. The warrants are callable by us in the event the
Company’s shares are publically traded in the future and certain price and
volume conditions are met. We are including herein the: (i) 500,008
common shares, and (ii) 250,006 common shares underlying the warrants, issued
pursuant to the 2009 February Offering.
2009
June Offering
In June
and July of 2009 we completed the offering of 2,025,344 units resulting in gross
proceeds of $3,038,000 or $1.50 per unit. Each unit consists of: (i)
one share of common stock; and (ii) one half common stock purchase
warrant. The warrants have a term of five years and allow the
investors to purchase our common shares at a price per share of
$3.00. The Warrants also contain anti-dilution protection in the
event of stock splits, stock dividends and other similar
transactions. The warrants are callable by us in the event the
Company’s shares are publically traded in the future and certain price and
volume conditions are met. We are registering herein the: (i)
2,025,344 common shares, and (ii) 1,012,679 common shares underlying the
warrants, issued pursuant to the June Offering. We are including
herein the: (i) 33,334 common shares; and (ii) 100,562 common shares underlying
warrants, that were issued to finders in connection with the
offering.
Consultant
Warrants and Common Shares
We have
issued shares and warrants to consultants and service providers in exchange for
services and reimbursement of expenses. We are registering 200,000
common shares underlying warrants that have been issued to consultants as
compensation or for reimbursement of expenses.
38
2008
July Offering
In July
and August of 2008 we completed the offering of 2,320,000 units resulting in
gross proceeds of $2,320,000 or $1.00 per unit. Each unit consists of: (i) 1
share of common stock; and (ii) ½ common stock purchase warrant. The warrants
have a term of 5 years and an exercise price of $2.00 per shares subject to
certain anti-dilution adjustments. The warrants are also callable by the Company
in the event the Company’s shares are publically traded in the future and
certain price and volume conditions are met. As part of the offering, TR Winston
& Company, LLC acted as the Company’s placement agent with respect to the
transaction. Pursuant to a placement agent agreement with TR Winston &
Company, LLC we agreed to the following compensation: (i) cash fee equal to 8%
of gross proceeds raised, including any payments made to the Company upon the
exercise of the warrants; (ii) the issuance of a warrant to purchase 8% of all
securities issued; and (iii) payment of legal expenses totaling $20,000.
Accordingly, we issued to TR Winston & Company, LLC a warrant to purchase
278,400 common shares of which 22,500 were assigned to Mercer Capital, Ltd. and
associated persons for their assistance in the offering. The warrant has an
exercise price per common shares of $2.00 and a term of 5 years. Also, as an
accommodation to the Company, TR Winston & Company, LLC agreed to receive a
convertible debenture in the principal amount of $163,600 and warrants to
purchase an additional 81,800 common shares, in lieu of its $163,600 cash
fee. The convertible debenture accrues interest at 5% per annum and has a
maturity date of July 14, 2009. It is convertible into the shares of the
Company’s common stock, at the sole discretion of the holder, at $1.00 per share
subject to certain anti-dilution adjustments. The warrant has the same terms as
those issued to investors in the offering.
The
exercise price of the warrants issued as part of the 2008 July Offering have
previously been adjusted to $1.50.
The
Selling Stockholders may exercise their warrants at any time in their sole
discretion. All of the Selling Stockholders named below acquired their common
stock and warrants directly from us in private transactions.
Set forth
below is information, to the extent known to us, setting forth the name of each
Selling Shareholder and the amount and percentage of Common Stock owned by each
(including shares that can be acquired on the exercise of outstanding warrants)
prior to the offering, the shares to be sold in the offering, and the amount and
percentage of Common Stock to be owned by each (including shares that can be
acquired on the exercise of outstanding warrants) after the offering assuming
all shares are sold. The footnotes provide information about persons who have
investment voting power for the Selling Shareholders and about material
transactions between the Selling Shareholders and the Company.
The
Selling Stockholders may sell all or some of the shares of common stock they are
offering, and may sell shares of our common stock otherwise than pursuant to
this prospectus. The table below assumes that each Selling Stockholder exercises
all of its warrants and sells all of the shares issued upon exercise thereof,
and that each selling stockholder sells all of the shares offered by it in
offerings pursuant to this prospectus, and does not acquire any additional
shares. We are unable to determine the exact number of shares that will actually
be sold or when or if these sales will occur.
The
Selling Stockholders may sell all, some or none of their shares in this
offering. See “Plan of Distribution.”
The total
number of common shares sold under this prospectus may be adjusted to reflect
adjustments due to stock dividends, stock distributions, splits, combinations or
recapitalizations with regard to the common stock and warrants.
Unless
otherwise stated below in the footnotes, to our knowledge, no Selling
Stockholder nor any affiliate of such stockholder: (i) has held any position or
office with, been employed by or otherwise has had any material relationship
with us or our affiliates during the three years prior to the date of this
prospectus; or (ii) is a broker-dealer, or an affiliate of a
broker-dealer.
We may
amend or supplement this prospectus from time to time in the future to update or
change this list and shares which may be resold.
We may
amend or supplement this prospectus from time to time in the future to update or
change this list and shares which may be resold.
39
|
Common
Shares Owned Before Sale (1)
|
Common
Shares Owned After Sale (2)
|
|||||||||||||||||||||||||||
|
Held
Outright
|
Warrants/
Options
|
Amount
|
%
of class
|
Shares
being
registered
|
Amount
|
%
of Class
|
||||||||||||||||||||||
|
Bristol
Investment Fund, Ltd.(3)
|
500,000 | 166,667 | 666,667 | 4 | % | 666,667 | — | * | ||||||||||||||||||||
|
The
JD Group, LLC(4)
|
500,000 | — | 500,000 | 3 | % | 500,000 | — | * | ||||||||||||||||||||
|
G.
Tyler Runnels or Jasmine Niklas Runnels TTEES The Runnel Family Trust dtd
1-11-20(5)
|
375,000 | 83,334 | 458,334 | * | 83,334 | 375,000 | 2 | % | ||||||||||||||||||||
|
Richard
Hull, PhD
|
295,000 | 33,334 | 328,334 | 2 | % | 83,334 | 245,000 | 2 | % | |||||||||||||||||||
|
IRA
FBO J. Steven Emerson Rollover II Pershing LLC as
Custodian
|
250,000 | 166,667 | 416,667 | 3 | % | 416,667 | — | * | ||||||||||||||||||||
|
T.R.
Winston & Company, LLC (6)
|
337,700 | 337,700 | 2 | % | 255,900 | 81,800 | 1 | % | ||||||||||||||||||||
|
Steven
Michell Sack PSP U/A DTD 01/01/1994
|
200,000 | 66,667 | 266,667 | 2 | % | 266,667 | — | * | ||||||||||||||||||||
|
Steven
Chizzik
|
245,000 | — | 245,000 | 2 | % | 245,000 | — | * | ||||||||||||||||||||
|
Kathryn
F. Hopper
|
130,000 | 20,000 | 150,000 | 1 | % | 150,000 | — | * | ||||||||||||||||||||
|
Robert
O'Mara
|
153,334 | 43,334 | 196,668 | 1 | % | 196,668 | — | * | ||||||||||||||||||||
|
Subhash
C. Gulati
|
110,000 | 6,667 | 116,667 | 1 | % | 116,667 | — | * | ||||||||||||||||||||
|
Ajax
Partners (7)
|
100,000 | 66,667 | 166,667 | 1 | % | 166,667 | — | * | ||||||||||||||||||||
|
JAG
MULTI INVESTMENTS LLC (8)
|
100,000 | 66,667 | 166,667 | 1 | % | 166,667 | — | * | ||||||||||||||||||||
|
Robert
R. Kauffman
|
100,000 | 66,667 | 166,667 | 1 | % | 166,667 | — | * | ||||||||||||||||||||
|
Steven
Mitchell Sack
|
100,000 | 66,667 | 166,667 | 1 | % | 166,667 | — | * | ||||||||||||||||||||
|
New
Giles, LLC (9)
|
75,000 | 50,000 | 125,000 | 1 | % | 125,000 | — | * | ||||||||||||||||||||
|
Samax
Family Limited Partnership (10)
|
75,000 | 50,000 | 125,000 | 1 | % | 125,000 | — | * | ||||||||||||||||||||
|
Jay
R. Solan
|
75,000 | 16,667 | 91,667 | 1 | % | 91,667 | — | * | ||||||||||||||||||||
|
Core
Fund, L.P. (11)
|
100,000 | — | 100,000 | 1 | % | 100,000 | — | * | ||||||||||||||||||||
|
Bruce
N. Barron & Jacqueline A. Barron
|
100,000 | — | 100,000 | 1 | % | 100,000 | — | * | ||||||||||||||||||||
|
Thomas
E. Genna
|
100,000 | — | 100,000 | 1 | % | 100,000 | — | * | ||||||||||||||||||||
|
Richard
W. Green
|
75,000 | 16,667 | 91,667 | 1 | % | 91,667 | — | * | ||||||||||||||||||||
|
D.
Carl Lustig, III
|
75,000 | 50,000 | 125,000 | 1 | % | 125,000 | — | * | ||||||||||||||||||||
|
The
Verrazano Group, LLC (12)
|
— | 84,000 | 84,000 | 1 | % | 84,000 | — | * | ||||||||||||||||||||
|
Windermere
Insurance Co. Ltd. (13)
|
50,000 | 33,334 | 83,334 | 1 | % | 83,334 | — | * | ||||||||||||||||||||
|
Christopher
Miglino
|
50,000 | 33,334 | 83,334 | 1 | % | 83,334 | — | * | ||||||||||||||||||||
|
Doris
Sutz Roth IRA
|
50,000 | 33,334 | 83,334 | 1 | % | 83,334 | — | * | ||||||||||||||||||||
|
Dr.
Arnold Yoskowitz and Regina Yoskowitz
|
50,000 | 33,334 | 83,334 | 1 | % | 83,334 | — | * | ||||||||||||||||||||
|
Gerald
B. Lichtenberger
|
50,000 | 33,334 | 83,334 | 1 | % | 83,334 | — | * | ||||||||||||||||||||
|
John
Peter Christensen
|
50,000 | 33,334 | 83,334 | 1 | % | 83,334 | — | * | ||||||||||||||||||||
|
Joseph
Giamanco
|
50,000 | 33,334 | 83,334 | 1 | % | 83,334 | — | * | ||||||||||||||||||||
|
Philip
S. Sassower
|
50,000 | 33,334 | 83,334 | 1 | % | 83,334 | — | * | ||||||||||||||||||||
40
|
Common
Shares Owned Before Sale (1)
|
Common
Shares Owned After Sale (2)
|
|||||||||||||||||||||||||||
|
Held
Outright
|
Warrants/
Options
|
Amount
|
%
of class
|
Shares
being
registered
|
Amount
|
%
of Class
|
||||||||||||||||||||||
| Mitchell J. Sassower | 50,000 | 33,334 | 83,334 | 1 | % | 83,334 | — | * | ||||||||||||||||||||
|
Jerry
A. Lubliner, M.D.
|
50,000 | 33,334 | 83,334 | 1 | % | 83,334 | — | * | ||||||||||||||||||||
|
Beatrice
Slomiuc
|
50,000 | — | 50,000 | * | 50,000 | — | * | |||||||||||||||||||||
|
Chaim
Slomiuc
|
50,000 | — | 50,000 | * | 50,000 | — | * | |||||||||||||||||||||
|
David
N. Baker
|
50,000 | — | 50,000 | * | 50,000 | — | * | |||||||||||||||||||||
|
Equireal
Leasing, Inc. (14)
|
50,000 | — | 50,000 | * | 50,000 | — | * | |||||||||||||||||||||
|
Jeff
Strauss & Mindy Schultheis
|
50,000 | — | 50,000 | * | 50,000 | — | * | |||||||||||||||||||||
|
John
Curley & Patricia Jennings Curley
|
50,000 | — | 50,000 | * | 50,000 | — | * | |||||||||||||||||||||
|
Marie
A. Karanfilian
|
50,000 | — | 50,000 | * | 50,000 | — | * | |||||||||||||||||||||
|
Sheila
Sugerman
|
50,000 | — | 50,000 | * | 50,000 | — | * | |||||||||||||||||||||
|
Steven
E. Holzel
|
50,000 | — | 50,000 | * | 50,000 | — | * | |||||||||||||||||||||
|
Steven
Shum
|
50,000 | — | 50,000 | * | 50,000 | — | * | |||||||||||||||||||||
|
Alan
Schwartz
|
25,000 | 16,667 | 41,667 | * | 41,667 | — | * | |||||||||||||||||||||
|
Arthur
Dunkin
|
25,000 | 16,667 | 41,667 | * | 41,667 | — | * | |||||||||||||||||||||
|
Faith
Griffin & John A. Lenhart JTWROS
|
25,000 | 16,667 | 41,667 | * | 41,667 | — | * | |||||||||||||||||||||
|
IRA
FBO John Curley, Pershing LLC as Custodian
|
25,000 | 16,667 | 41,667 | * | 41,667 | — | * | |||||||||||||||||||||
|
Patrick
Hund
|
25,000 | 16,667 | 41,667 | * | 41,667 | — | * | |||||||||||||||||||||
|
Rhonda
Wesolak, Individual Retirement Account, RBC Capital Markets Corp
Cust
|
25,000 | 16,667 | 41,667 | * | 41,667 | — | * | |||||||||||||||||||||
|
John
G. Korman
|
25,000 | 16,667 | 41,667 | * | 41,667 | — | * | |||||||||||||||||||||
|
A.C.
Providenti
|
25,000 | 16,667 | 41,667 | * | 41,667 | — | * | |||||||||||||||||||||
|
Benjamin
Hill
|
21,667 | 13,334 | 35,001 | * | 35,001 | — | * | |||||||||||||||||||||
|
John
Toedtman
|
20,000 | — | 20,000 | * | 20,000 | — | * | |||||||||||||||||||||
|
Donald
L. Stahl
|
12,500 | 8,334 | 20,834 | * | 20,834 | — | * | |||||||||||||||||||||
|
Leslie
M. James
|
12,500 | 8,334 | 20,834 | * | 20,834 | — | * | |||||||||||||||||||||
|
Nathan
Sugerman
|
12,500 | 8,334 | 20,834 | * | 20,834 | — | * | |||||||||||||||||||||
|
Robert
B. Greene
|
12,500 | 8,334 | 20,834 | * | 20,834 | — | * | |||||||||||||||||||||
|
Klaus
Peter Eichner
|
12,500 | 8,334 | 20,834 | * | 20,834 | — | * | |||||||||||||||||||||
|
Gary
J. Faden
|
12,500 | 8,334 | 20,834 | * | 20,834 | — | * | |||||||||||||||||||||
|
Mercer
Capital, Ltd. (15)
|
— | 20,667 | 20,667 | * | 20,667 | — | * | |||||||||||||||||||||
|
Andrew
B. Dorman
|
— | 5,667 | 5,667 | * | 5,667 | — | * | |||||||||||||||||||||
|
David
S. Lustig
|
— | 2,774 | 2,774 | * | 2,774 | — | * | |||||||||||||||||||||
|
Nicole
H. Tavernier
|
— | 667 | 667 | * | 667 | — | * | |||||||||||||||||||||
41
|
Common
Shares Owned Before Sale (1)
|
Common
Shares Owned After Sale (2)
|
|||||||||||||||||||||||||||
|
Held
Outright
|
Warrants/
Options
|
Amount
|
%
of class
|
Shares
being
registered
|
Amount
|
%
of Class
|
||||||||||||||||||||||
|
Mark
P. Eichner
|
—
|
227 | 227 | * | 227 | — | * | |||||||||||||||||||||
| Jeffrey J. Kraws |
—
|
25,000 | 25,000 | * | 25,000 | — | * | |||||||||||||||||||||
|
Karen
B. Goldfarb
|
—
|
25,000 | 25,000 | * | 25,000 | — | * | |||||||||||||||||||||
|
Kemmerer
Resources Corp. (16)
|
—
|
150,000 | 150,000 | 1 | % | 150,000 | — | * | ||||||||||||||||||||
|
John
L. Kemmerer, JR. 1970 Trust (17)
|
690,000 | 345,000 | 1,035,000 | 7 | % | 1,035,000 | — | * | ||||||||||||||||||||
|
Photon
Global Ltd. (18)
|
333,334 | 166,667 | 500,001 | 3 | % | 500,001 | — | * | ||||||||||||||||||||
|
Gregory
P. Zeller
|
166,667 | 83,334 | 250,001 | 2 | % | 250,001 | — | * | ||||||||||||||||||||
|
Far
Hills Capital, LLC (19)
|
133,334 | 66,667 | 200,001 | 1 | % | 200,001 | — | * | ||||||||||||||||||||
|
Cynthia
P. Stafford Trust
|
133,334 | 66,667 | 200,001 | 1 | % | 200,001 | — | * | ||||||||||||||||||||
|
Jonathan
Meyers
|
66,667 | 33,334 | 100,001 | 1 | % | 100,001 | — | * | ||||||||||||||||||||
|
Gene
Mulvihill
|
66,667 | 33,334 | 100,001 | 1 | % | 100,001 | — | * | ||||||||||||||||||||
|
Robert
L. and Natacha Stafford
|
66,667 | 33,334 | 100,001 | 1 | % | 100,001 | — | * | ||||||||||||||||||||
|
Horemheb
Investments, LLC (20)
|
66,667 | 33,334 | 100,001 | 1 | % | 100,001 | — | * | ||||||||||||||||||||
|
Robert
L. Stafford Jr. Trust
|
66,667 | 33,334 | 100,001 | 1 | % | 100,001 | — | * | ||||||||||||||||||||
|
Equity
Trust Company, d.b.a. Sterling Trust, Custodian FBO: John
Sanderford
|
46,667 | 23,334 | 70,001 | * | 70,001 | — | * | |||||||||||||||||||||
|
Peter
Cunningham
|
33,334 | 16,667 | 50,001 | * | 50,001 | — | * | |||||||||||||||||||||
|
Gordon
W. Clark, Jr.
|
33,334 | 16,667 | 50,001 | * | 50,001 | — | * | |||||||||||||||||||||
|
Edward
J. Foley, III
|
33,334 | 16,667 | 50,001 | * | 50,001 | — | * | |||||||||||||||||||||
|
Held
Under Will of Joan P. Foley FBO Edward J. Foley, III - Lifetime
Trust
|
33,334 | 16,667 | 50,001 | * | 50,001 | — | * | |||||||||||||||||||||
|
Edward
J. Foley, III, 2009 GRAT
|
33,334 | 16,667 | 50,001 | * | 50,001 | — | * | |||||||||||||||||||||
|
Francis
O'Connor
|
33,334 | 16,667 | 50,001 | * | 50,001 | — | * | |||||||||||||||||||||
|
Frank
E. Walsh, III
|
33,334 | 16,667 | 50,001 | * | 50,001 | — | * | |||||||||||||||||||||
|
Bass
Family Trust
|
33,334 | 16,667 | 50,001 | * | 50,001 | — | * | |||||||||||||||||||||
|
Brian
Thebault
|
33,334 | 16,667 | 50,001 | * | 50,001 | — | * | |||||||||||||||||||||
|
Voiceified
Technologies, LLC (21)
|
33,334 | 16,667 | 50,001 | * | 50,001 | — | * | |||||||||||||||||||||
|
Kihong
Kwon, M.D.
|
33,334 | 16,667 | 50,001 | * | 50,001 | — | * | |||||||||||||||||||||
|
Iroquois
Master Fund, Ltd. (22)
|
33,334 | 16,667 | 50,001 | * | 50,001 | — | * | |||||||||||||||||||||
|
Roxy
Transportation Co., Inc. (23)
|
25,000 | 12,500 | 37,500 | * | 37,500 | — | * | |||||||||||||||||||||
|
William
Stewart
|
22,223 | 11,112 | 33,335 | * | 33,335 | — | * | |||||||||||||||||||||
|
Bernard
B. Markey
|
22,223 | 11,112 | 33,335 | * | 33,335 | — | * | |||||||||||||||||||||
|
Diamond
II Investments, LLC (24)
|
22,223 | 11,112 | 33,335 | * | 33,335 | — | * | |||||||||||||||||||||
|
Ravenwood
Partners, LLC (25)
|
20,000 | 10,000 | 30,000 | * | 30,000 | — | * | |||||||||||||||||||||
42
|
Common
Shares Owned Before Sale (1)
|
Common
Shares Owned After Sale (2)
|
|||||||||||||||||||||||||||
|
Held
Outright
|
Warrants/
Options
|
Amount
|
%
of class
|
Shares
being
registered
|
Amount
|
%
of Class
|
||||||||||||||||||||||
| John Sanderford | 20,000 | 10,000 | 30,000 | * | 30,000 | — | * | |||||||||||||||||||||
|
Terry
Sholty Strada
|
16,667 | 8,334 | 25,001 | * | 25,001 | — | * | |||||||||||||||||||||
|
John
Amorosa
|
16,667 | 8,334 | 25,001 | * | 25,001 | — | * | |||||||||||||||||||||
|
Glen
Bliwise
|
16,667 | 8,334 | 25,001 | * | 25,001 | — | * | |||||||||||||||||||||
|
Joseph
C. Roselle
|
16,667 | 8,334 | 25,001 | * | 25,001 | — | * | |||||||||||||||||||||
|
Schuyler
L. Merrihew
|
16,667 | 8,334 | 25,001 | * | 25,001 | — | * | |||||||||||||||||||||
|
Kihong
Kwon, M.D., Custodian, UGMA for Connor Merrihew
|
16,000 | 8,000 | 24,000 | * | 24,000 | — | * | |||||||||||||||||||||
|
Kihong
Kwon, M.D., Custodian, UGMA for Mason Kwon
|
16,000 | 8,000 | 24,000 | * | 24,000 | — | * | |||||||||||||||||||||
|
Timothy
V. O'Connor
|
10,000 | 5,000 | 15,000 | * | 15,000 | — | * | |||||||||||||||||||||
|
Jarmila
Cunningham
|
8,334 | 4,167 | 12,501 | * | 12,501 | — | * | |||||||||||||||||||||
|
Bernard
B. Markey, Individual 401(K) ETRADE Custodian
|
6,667 | 3,334 | 10,001 | * | 10,001 | — | * | |||||||||||||||||||||
|
Timothy
V. O'Connor Roth IRA
|
6,667 | 3,334 | 10,001 | * | 10,001 | — | * | |||||||||||||||||||||
|
Brandon
Hill
|
3,334 | 1,667 | 5,001 | * | 5,001 | — | * | |||||||||||||||||||||
|
Sequoia
Global Partners, LLC (26)
|
— | 73,228 | 73,228 | * | 73,228 | — | * | |||||||||||||||||||||
|
Walter
H. Bass, LLC (26)
|
— | 10,667 | 10,667 | * | 10,667 | — | * | |||||||||||||||||||||
| 7,668,686 | 3,561,634 | 11,230,320 | 70 | % | 10,528,520 | 701,800 | 5 | % | ||||||||||||||||||||
43
*
Represents less than 1%
**Unless
otherwise stated, the individuals with voting and dispositive control of
securities offered on behalf of trusts or custodial accounts is the individual
or entity referenced in the name of such accounts.
(1) Pursuant
to Rules 13d-3 and 13d-5 of the Exchange Act, beneficial ownership includes any
common shares as to which a shareholder has sole or shared voting power or
investment power, and also any common shares which the shareholder has the right
to acquire within 60 days, including upon exercise of common shares purchase
options or warrants. There were 15,292,281 common shares outstanding as of
September 30, 2009.
(2) Assumes
the sale of all common shares previously registered and included herein but does
not include shares initially being registered which may be part of the Initial
Registration shares contained above.
(3) Bristol
Capital Advisors, LLC (“BCA”) is the investment advisor to Bristol Investment
Fund, Ltd. (“Bristol”). Paul Kessler is the manager of BCA and as such has
voting and investment control over the securities held by Bristol. Mr. Kessler
disclaims beneficial ownership of these securities.
(4) John
Davies, Manager, is the person with voting and dispositive control with respect
to the securities being offered.
(5) G.
Tyler Runnels and Jasmine Niklas Runnels, as Trustees, have voting and
dispositive control with respect to the securities being offered.
(6) G.
Tyler Runnels, as President, has voting and dispositive control with respect to
the securities being offered. Mr. Runnels is an associated person of TR Winston
& Company, LLC.
(7) Richard
Stone, Managing Partner, has voting and dispositive control with respect to the
securities being offered.
(8) James
Coren, member has voting and dispositive control with respect to the securities
being offered.
(9) Leonard
Pearlman, Managing Director, has voting and dispositive control with respect to
the securities being offered.
(10) Andrew
Margulies, General Partner, has voting and dispositive control with respect to
the securities being offered.
(11) Steve
Shum, Managing Director, has voting and dispositive control with respect to the
securities being offered.
(12) Stephen
Chizzik has voting and dispositive control with respect to the securities being
offered.
44
(13) John
Scardino, Director, has voting and dispositive control with respect to the
securities being offered.
(14) Andrew
Margulies, Vice President, has voting and dispositive control with respect to
the securities being offered.
(15) Anthony
Salino has voting and dispositive control with respect to the securities being
offered by Mercer Capital, Ltd.
(16) John
L Kemmerer, III has voting and dispositive control with respect to the
securities being offered.
(17) Peter
F. Nejes and Dennis Powers, as trustees, have voting and dispositive control
with respect to the securities being offered.
(18) Rene
de Villiers has voting and dispositive control with respect to the securities
being offered.
(19) Steve
Sciaretta has voting and dispositive control with respect to the securities
being offered.
(20) Robert
Stafford, Jr. has voting and dispositive control with respect to the securities
being offered.
(21) Robert
L. Magaletta has voting and dispositive control with respect to the securities
being offered.
(22) Joshua
Silverman has voting and dispositive control with respect to the securities
being offered by Iroquois Master Fund Ltd. Mr. Silverman disclaims
beneficial ownership of the shares.
(23) Peter
C. Cunningham, has voting and dispositive control with respect to the securities
being offered.
(24) Lorrie
Allegra has voting and dispositive control with respect to the securities being
offered.
(25) Greg
Bolloten has voting and dispositive control with respect to the securities being
offered.
(26) Greg
Bolloten has voting and dispositive control with respect to the securities being
offered.
45
DESCRIPTION
OF SECURITIES
General
As of
September 30, 2009, our authorized capital stock consisted of:
|
|
·
|
80,000,000
shares of common stock, par value $0.0001;
and
|
|
|
·
|
10,000,000
shares of “blank check” preferred stock, par value
$0.0001.
|
As of
September 30, 2009, 15,292,281 shares of common stock were issued and
outstanding and 0 shares of preferred stock were issued and outstanding. All of
our currently issued and outstanding shares of capital stock were validly
issued, fully paid and non-assessable under the Delaware General Corporation
Law, as amended, or the DGCL.
Set forth
below is a summary description of all the material terms of our common stock and
warrants. This description is qualified in its entirety by reference to our
amended and restated certificate of incorporation, bylaws and form of warrants,
each of which is filed as an exhibit to this registration
statement.
Common
Stock
The
holders of our common stock are entitled to one vote per share on each matter
submitted to a vote at a meeting of our stockholders, except to the extent that
the voting rights of our shares of any class or series of stock are determined
and specified as greater or lesser than one vote per share in the manner
provided by our certificate of incorporation. Our stockholders have no
pre-emptive rights to acquire additional shares of our common stock or other
securities. Our common stock is not subject to redemption rights and carries no
subscription or conversion rights. In the event of liquidation of our company,
the shares of our common stock are entitled to share equally in corporate assets
after satisfaction of all liabilities. All shares of our common stock now
outstanding are fully paid and non-assessable. Our bylaws authorize the board of
directors to declare dividends on our outstanding shares. As of
September 30, 2009, 15,292,281 shares of common stock were issued and
outstanding.
Preferred
Stock
We may
issue our preferred shares from time to time in one or more series as determined
by our board of directors. The voting powers and preferences, the relative
rights of each series, and the qualifications, limitations and restrictions
thereof may be established by our board of directors without any further vote or
action by our shareholders. As of July 31, 2009 there were no shares of our
preferred stock issued and outstanding.
Warrants
Anti-Dilution
Warrants
The
warrants have a term of 5 years and an exercise price of $1.50 per shares
subject to anti-dilution adjustments. The anti-dilution adjustments
provide for adjustment of the warrant exercise price and number of shares
issuable upon exercise in the event the Company: (i) declares a stock dividend
or split; (ii) undertakes subsequent equity sales are a price below $1.50 per
share; (iii) undertakes a rights offering; (iv) there is a pro-rata distribution
to the Company’s shareholders; or (v) in the event of a fundamental transaction.
The warrants are also callable by the Company in the event: (a) the Company’s
shares are publically traded in the future; (b) the shares trade above $4.00 per
share for 20 consecutive trading days; (c) the average daily volume over the 20
consecutive trading days exceeds 75,000; and (d) there is an effective
registration statement covering the underlying shares.
2009
February and June Offering Warrants
In
connection with our February and June Offerings we issued the investors in those
offerings warrants. The warrants have a term of 5 years and an
exercise price of $3.00 per shares subject to anti-dilution
adjustments. The warrants also contain anti-dilution protection in
the event of stock splits, stock dividends and other similar
transactions. The provisions do not provide for any adjustment
in the event of subsequent equity sales or transactions. The
warrants are also callable by the Company in the event: (a) the Company’s shares
are publically traded in the future; (b) the shares trade above $5.00 per share
for 20 consecutive trading days; (c) the average daily volume over the 20
consecutive trading days exceeds 75,000; and (d) there is an effective
registration statement covering the underlying shares.
Consultant
Warrants and Common Shares
The terms
of the consultant warrants are typically negotiated on an individual
basis. Notwithstanding, the warrants typically have a term of five
years and an exercise price equal to or greater than the last previously
completed offering. The warrants also contain anti-dilution
protection in the event of stock splits, stock dividends and other similar
transactions. The provisions do not provide for any adjustment
in the event of subsequent equity sales or
transactions.
46
2008
June Offering Warrants
The
warrants have a term of 5 years and an exercise price of $1.50 per shares
subject to anti-dilution adjustments. The anti-dilution adjustments
provide for adjustment of the warrant exercise price and number of shares
issuable upon exercise in the event the Company: (i) declares a stock dividend
or split; (ii) undertakes subsequent equity sales are a price below $1.50 per
share; (iii) undertakes a rights offering; (iv) there is a pro-rata distribution
to the Company’s shareholders; or (v) in the event of a fundamental transaction.
The warrants are also callable by the Company in the event: (a) the Company’s
shares are publically traded in the future; (b) the shares trade above $4.00 per
share for 20 consecutive trading days; (c) the average daily volume over the 20
consecutive trading days exceeds 75,000; and (d) there is an effective
registration statement covering the underlying shares.
The
warrants previously had an exercise price of $2.00. Upon the closing
of the 2009 February and June Offerings, the warrant exercise price adjusted to
$1.50.
Registration
Rights
Since
inception, we have completed a number of private offerings. As part
of the private placement, we entered into a registration rights agreements with
the investors under which we agreed to file the registration statement of which
this prospectus is a part in order to register (1) the common shares issued in
the private placement; and (2) the common shares issuable upon the exercise of
the warrants.
The
registration rights agreement required us to use our best efforts
to:
|
|
·
|
file
the registration statement with in certain period of time after a
triggering event, usually the first closing (“Filing
Deadline”);
|
|
|
·
|
have
the registration agreement declared effective within a certain amount of
days after the Filing Deadline (“Effectiveness Deadline”);
and
|
|
|
·
|
maintain
the registration statement continuously effective until the date that the
shares covered by this prospectus may be sold pursuant to Rule 144 of the
Securities Act without any manner of sale or volume
restrictions.
|
If we
fail to file the registration statement by the Filing Deadline, have the
registration statement declared effective by the Effectiveness Deadline, or the
registration statement does not stay effective, we are obligated to pay the
holder a penalty.
The exact
Filing Deadline, Effective Deadline, amount of penalty and the form of payment
is determined on a transaction by transaction basis. For a more
complete description of our obligations under each registration statement, we
refer you to the form of each such registration rights agreements filed as
exhibits to this registration statement.
MARKET FOR COMMON EQUITY &
RELATED STOCKHOLDER MATTERS
Market
Information & Holders
On
September 18, 2009, our common shares began quotation on the OTCBB under the
trading symbol of GNSZ. As of the date of this prospectus, the
trading in our common shares is limited and accordingly, no public market
exists. Private sales or transfers are permitted under the respective
state and Federal securities laws, subject to compliance with exemptions set
forth under the respective statutory guidelines. As of September 30, 2009,
we had 124 common shareholders of record.
Options,
Warrants and Convertible Securities
As of
September 30, 2009, there were outstanding common share purchase options,
warrants and convertible securities entitling the holders to purchase up to
7,884,502 common shares at exercise prices between $0.50 and $3.00 with an
average weighted exercise price of $1.63 per share.
SHARES ELIGIBLE FOR FUTURE
SALE
To date,
there has been no market for our common stock. In the event a public market for
our shares develops, future sales of substantial amounts of our common stock in
the public market could adversely affect prevailing market prices from time to
time. Further, since only a limited number of shares will be available for sale
shortly after this offering because of certain contractual and legal
restrictions on resale described below, sales of substantial amounts of our
common stock in the public market after the restrictions lapse could adversely
affect the prevailing market price and our ability to raise equity capital in
the future.
47
Sale
of Restricted Shares
As of
September 30,2009 we will have 15,292,281 shares of common stock
outstanding. Of these shares, the shares sold in this offering will
be freely tradable without restriction under the Securities Act, except for any
shares purchased by our “affiliates” as that term is defined in Rule 144
under the Securities Act. In general, affiliates include executive officers,
directors, and 10% stockholders. Shares purchased by affiliates will remain
subject to the resale limitations of Rule 144.
Upon
completion of this offering, 8,870,958 shares of common stock will be
“restricted securities,” as that term is defined in Rule 144 under the
Securities Act and as a result of Lock-Up Agreements as discussed below. These
restricted securities are eligible for public sale only if they are registered
under the Securities Act or if they qualify for an exemption from registration
under rules 144 or 701 under the Securities Act, which are summarized
below.
As a
result of the lock-up agreements described below and the provisions of
Rule 144 and Rule 701 of the Securities Act, the shares of our common
stock (excluding the shares sold in this offering or issuable upon the exercise
or conversion of a security) will be available for sale in the public market as
follows:
|
Date
|
Number of Shares
|
|||
|
On
the date of this prospectus
|
6,421,323 | |||
|
Within
90 days after the date of this prospectus
|
8,980,001 | |||
|
Between
90 and 360 days after the date of this prospectus(1)
|
15,292,281 | |||
|
365
days after the effective date of this prospectus(1)
|
15,292,281 | |||
(1)
shares may be subject to volume limitations as a result of certain shareholders
affiliate status.
Lock-Up
Agreements
Our
directors, executive officers, and certain other stockholders are subject to
lock up agreements, generally providing that they will not offer, sell, contract
to sell, or grant any option to purchase or otherwise dispose of our common
stock or any securities exercisable for or convertible into our common stock
owned by them until December 31, 2009. Despite possible earlier
eligibility for sale under the provisions of Rules 144 and 701, shares
subject to lock-up agreements will not be salable until these agreements expire
or are waived by prior investors. The Lock-Up Agreements were entered
into in connection with our July and August 2008
offering. Approximately 6,312,280 of our outstanding shares of common
stock will be subject to such lock-up agreements.
Rule 144
Generally,
Rule 144 (as amended effective February 15, 2008) provides that
an affiliate who has beneficially owned “restricted” shares of our common stock
for at least six months will be entitled to sell on the open market in brokers’
transactions, within any three-month period, a number of shares that does not
exceed the greater of:
|
|
•
|
1%
of the number of shares of common stock then
outstanding; or
|
|
|
•
|
the
average weekly trading volume of the common stock during the four calendar
weeks preceding the filing of a notice on Form 144 with respect to
such sale.
|
In
addition, sales under Rule 144 are subject to requirements with respect to
manner of sale, notice, and the availability of current public information about
us.
In the
event that any person who is deemed to be our affiliate purchases shares of our
common stock in this offering or acquires shares of our common stock pursuant to
one of our employee benefits plans, sales under Rule 144 of the shares held
by that person are subject to the volume limitations and other restrictions
described in the preceding two paragraphs.
The
volume limitation, manner of sale and notice provisions described above will not
apply to sales by non-affiliates. For purposes of Rule 144, a non-affiliate is
any person or entity who is not our affiliate at the time of sale and has not
been our affiliate during the preceding three months. Once we have been a
reporting company for 90 days, a non-affiliate who has beneficially owned
restricted shares of our common stock for six months may rely on Rule 144
provided that certain public information regarding us is available. The six
month holding period increases to one year in the event we have not been a
reporting company for at least 90 days. However, a non-affiliate who has
beneficially owned the restricted shares proposed to be sold for at least one
year will not be subject to any restrictions under Rule 144 regardless of how
long we have been a reporting company.
Rule 701
Under
Rule 701, each of our employees, officers, directors, and consultants who
purchased shares pursuant to a written compensatory plan or contract is eligible
to resell these shares 90 days after the effective date of this offering in
reliance upon Rule 144, but without compliance with specific restrictions.
Rule 701 provides that affiliates may sell their Rule 701 shares
under Rule 144 without complying with the holding period requirement and that
non-affiliates may sell their shares in reliance on Rule 144 without
complying with the holding period, public information, volume limitation, or
notice provisions of Rule 144.
48
Form S-8 Registration
Statements
We intend
to file one or more registration statements on Form S-8 under the
Securities Act as soon as practicable after the completion of this offering for
shares issued upon the exercise of options and shares to be issued under our
employee benefit plans. As a result, any shares acquired upon the exercise of
such options will be freely tradable in the public market.
PLAN
OF DISTRIBUTION
The
Selling Stockholders may sell their shares at a price of $1.50 per share or at
prevailing market prices or privately negotiated prices. Thereafter,
the Selling Stockholders and any of their pledgees, assignees and
successors-in-interest may, from time to time, sell any or all of their shares
of common stock on any stock exchange in which a market develops or trading
facility on which the shares are traded or in private transactions. These sales
may be at fixed or negotiated prices. A Selling Stockholder may use any one or
more of the following methods when selling shares:
|
·
|
ordinary
brokerage transactions and transactions in which the broker-dealer
solicits purchasers;
|
|
·
|
block
trades in which the broker-dealer will attempt to sell the shares as agent
but may position and resell a portion of the block as principal to
facilitate the transaction;
|
|
·
|
purchases
by a broker-dealer as principal and resale by the broker-dealer for its
account;
|
|
·
|
an
exchange distribution in accordance with the rules of the applicable
exchange;
|
|
·
|
privately
negotiated transactions;
|
|
·
|
settlement
of short sales entered into after the effective date of the registration
statement of which this prospectus is a
part;
|
|
·
|
broker-dealers
may agree with the Selling Stockholders to sell a specified number of such
shares at a stipulated price per
share;
|
|
·
|
through
the writing or settlement of options or other hedging transactions,
whether through an options exchange or
otherwise;
|
|
·
|
a
combination of any such methods of sale;
or
|
|
·
|
any
other method permitted pursuant to applicable
law.
|
The
Selling Stockholders may also sell shares under Rule 144 under the Securities
Act of 1933, as amended (the “ Securities Act”), if available, rather than under
this prospectus.
Broker-dealers
engaged by the Selling Stockholders may arrange for other brokers-dealers to
participate in sales. Broker-dealers may receive commissions or discounts from
the Selling Stockholders (or, if any broker-dealer acts as agent for the
purchaser of shares, from the purchaser) in amounts to be negotiated, but,
except as set forth in a supplement to this Prospectus, in the case of an agency
transaction not in excess of a customary brokerage commission in compliance with
FINRA Rule 2440; and in the case of a principal transaction a markup or markdown
in compliance with FINRA IM-2440.
In
connection with the sale of the common stock or interests therein, the Selling
Stockholders may enter into hedging transactions with broker-dealers or other
financial institutions, which may in turn engage in short sales of the common
stock in the course of hedging the positions they assume. The Selling
Stockholders may also sell shares of the common stock short and deliver these
securities to close out their short positions, or loan or pledge the common
stock to broker-dealers that in turn may sell these securities. The Selling
Stockholders may also enter into option or other transactions with
broker-dealers or other financial institutions or the creation of one or more
derivative securities which require the delivery to such broker-dealer or other
financial institution of shares offered by this prospectus, which shares such
broker-dealer or other financial institution may resell pursuant to this
prospectus (as supplemented or amended to reflect such
transaction).
The
Selling Stockholders and any broker-dealers or agents that are involved in
selling the shares may be deemed to be “underwriters” within the meaning of the
Securities Act in connection with such sales. In such event, any commissions
received by such broker-dealers or agents and any profit on the resale of the
shares purchased by them may be deemed to be underwriting commissions or
discounts under the Securities Act. Each Selling Stockholder has informed the
Company that it does not have any written or oral agreement or understanding,
directly or indirectly, with any person to distribute the Common Stock. In no
event shall any broker-dealer receive fees, commissions and markups which, in
the aggregate, would exceed eight percent (8%).
49
Any
securities acquired by an underwriter or related persons during 180 days prior
to the filing date of this prospectus or after the filing date of this
registration statement shall be deemed to be underwriting compensation by
FIINRA. Securities excluded from underwriting compensation pursuant
to FINRA Rule 2710(d)(5), shall not be directly or indirectly be offered, sold,
transferred, assigned, pledged, or hypothecated, or be the subject of any
hedging, short sale, derivative, put, or call transaction that would result in
the effective economic disposition of the securities by any person agree to
offer or sell, transfer, assign, pledge, hypothecate or subject to hedging,
short sale, derivative, put or call transaction such shares for a period of 180
days after the effective date of this registration statement except as provided
for in FINRA Rule 2710(g)(2).
The
Company is required to pay certain fees and expenses incurred by the Company
incident to the registration of the shares. The Company has agreed to indemnify
the Selling Stockholders against certain losses, claims, damages and
liabilities, including liabilities under the Securities Act.
Because
Selling Stockholders may be deemed to be “underwriters” within the meaning of
the Securities Act, they will be subject to the prospectus delivery requirements
of the Securities Act including Rule 172 thereunder. In addition, any securities
covered by this prospectus which qualify for sale pursuant to Rule 144 under the
Securities Act may be sold under Rule 144 rather than under this prospectus.
There is no underwriter or coordinating broker acting in connection with the
proposed sale of the resale shares by the Selling Stockholders.
We agreed
to keep this prospectus effective until the earlier of (i) the date on which the
shares may be resold by the Selling Stockholders without registration and
without regard to any volume or manner-of-sale limitations by reason of Rule
144, without the requirement for the Company to be in compliance with the
current public information under Rule 144 under the Securities Act or any other
rule of similar effect or (ii) all of the shares have been sold pursuant to this
prospectus or Rule 144 under the Securities Act or any other rule of similar
effect. The resale shares will be sold only through registered or licensed
brokers or dealers if required under applicable state securities laws. In
addition, in certain states, the resale shares may not be sold unless they have
been registered or qualified for sale in the applicable state or an exemption
from the registration or qualification requirement is available and is complied
with.
Under
applicable rules and regulations under the Exchange Act, any person engaged in
the distribution of the resale shares may not simultaneously engage in market
making activities with respect to the common stock for the applicable restricted
period, as defined in Regulation M, prior to the commencement of the
distribution. In addition, the Selling Stockholders will be subject to
applicable provisions of the Exchange Act and the rules and regulations
thereunder, including Regulation M, which may limit the timing of purchases and
sales of shares of the common stock by the Selling Stockholders or any other
person. We will make copies of this prospectus available to the Selling
Stockholders and have informed them of the need to deliver a copy of this
prospectus to each purchaser at or prior to the time of the sale (including by
compliance with Rule 172 under the Securities Act).
INDEMNIFICATION
OF DIRECTORS AND OFFICERS
The
Corporation Laws of the State of Delaware and the Company's Bylaws provide for
indemnification of the Company's Directors for expenses actually and necessarily
incurred by them in connection with the defense of any action, suit or
proceeding in which they, or any of them, are made parties, or a party, by
reason of having been Director(s) or Officer(s) of the corporation, or of such
other corporation, except, in relation to matter as to which any such Director
or Officer or former Director or Officer or person shall be adjudged in such
action, suit or proceeding to be liable for negligence or misconduct in the
performance of duty. Furthermore, the personal liability of the Directors
is limited as provided in the Company's Articles of Incorporation.
Insofar
as indemnification for liabilities arising under the Securities Act of 1933 may
be permitted to directors, officers or persons controlling the Company pursuant
to the foregoing provisions, the Company has been informed that in the opinion
of the Securities and Exchange Commission such indemnification is against public
policy as expressed in the Securities Act of 1933 and is therefore
unenforceable.
LEGAL
MATTERS
The
Silvestre Law Group, P.C. will issue a legal opinion as to the validity of the
issuance of the shares of common stock offered under this
prospectus.
EXPERTS
The
financial statements as of December 31, 2008 and 2007 and for each interim
period since December 31, 2008, included in this prospectus and in the
registration statement of which it forms a part, have been so included in
reliance on the report of RBSM LLP, our independent registered public accounting
firm, appearing elsewhere in this prospectus and the registration statement of
which it forms a part, given on the authority of said firm as experts in
auditing and accounting.
INTERESTS
OF NAMED EXPERTS AND COUNSEL
No expert
or counsel named in this prospectus as having prepared or certified any part of
this prospectus or having given an opinion upon the validity of the securities
being registered or upon other legal matters in connection with the registration
or offering of the shares of common stock was employed on a contingency basis or
had, or is to receive, in connection with the offering, a substantial interest,
directly or indirectly, in the registrant nor was any such person connected with
the registrant as a promoter, managing or principal underwriter, voting trustee,
director, officer or employee.
50
WHERE
YOU CAN FIND MORE INFORMATION
We will
file annual, quarterly and other reports, proxy statements and other information
with the SEC. You may read and copy any document we file at the public reference
facilities of the SEC at 100 F Street, NE, Washington, D.C. 20549. Please call
the SEC at 1-800-SEC-0330 for further information on the operation of the public
reference rooms. Our SEC filings are also available to the public at the SEC’s
web site at http://www.sec.gov and at our website at http://www.genspera.com. We
will furnish our stockholders with annual reports containing audited financial
statements.
This
prospectus is part of a registration statement on Form S-1 that we filed with
the SEC. Certain information in the registration statement has been omitted from
this prospectus in accordance with the rules and regulations of the SEC. We have
also filed exhibits and schedules with the registration statement that are
excluded from this prospectus. For further information you may:
|
·
|
read
a copy of the registration statement, including the exhibits and
schedules, without charge at the SEC’s public reference rooms;
or
|
|
·
|
obtain
a copy from the SEC upon payment of the fees prescribed by the
SEC.
|
51
INDEX
TO FINANCIAL STATEMENTS
|
Page
|
||
|
Report
of Independent Registered Public Accounting Firm
|
F-2 | |
|
Balance
Sheets at December 31, 2008 and 2007
|
F-3 | |
|
Statements
of Losses for the years ended December 31, 2008 and 2007 and for the
period from Inception (November 21, 2003) to December 31, 2008
|
F-4 | |
|
Statements
of Stockholders’ Equity for the period from Inception (November 21, 2003)
to December 31, 2008
|
F-5 | |
|
Statements
of Cash Flows for the years ended December 31, 2008 and 2007 and for the
period from Inception (November 21, 2003) to December 31,
2008
|
F-6 | |
|
Notes
to Financial Statements
|
F-7 | |
F-1
REPORT
OF INDEPENDENT REGISTERED CERTIFIED PUBLIC ACCOUNTING FIRM
Board of
Directors
GenSpera
Inc.
San
Antonio, TX
We have
audited the accompanying balance sheets of GenSpera Inc., a development stage
company, as of December 31, 2008 and 2007, and the related statements of losses,
statement of stockholders' equity, and cash flows for each of the two years in
the period ended December 31, 2008 and the period November 21, 2003 (date of
inception) through December 31, 2008. These financial statements are the
responsibility of the company's management. Our responsibility is to express an
opinion on the financial statements based upon our audits.
We
conducted our audits in accordance with standards of the Public Company
Accounting Oversight Board (United States of America). Those standards require
that we plan and perform the audit to obtain reasonable assurance about whether
the financial statements are free of material misstatements. The Company is not
required to have, nor were we engaged to perform, an audit of its internal
control over financial reporting. Our audit included consideration of internal
control over financial reporting as a basis for designing audit procedures that
are appropriate in the circumstances, but not for the purpose of expressing an
opinion on the effectiveness of the Company's internal control over financial
reporting. Accordingly we express no such opinion. An audit includes examining,
on a test basis, evidence supporting the amounts and disclosures in the
financial statements. An audit also includes assessing the accounting principles
used and significant estimates made by management, as well as evaluating the
overall financial statement presentation. We believe our audits provide a
reasonable basis for our opinion.
In our
opinion, the financial statements referred to above present fairly, in all
material respects, the financial position of GenSpera Inc., a development stage
company, at December 31, 2008 and 2007 and the results of its operations and its
cash flows for each of the two years in the period ended December 31, 2008 and
the period November 21, 2003 (date of inception) through December 31, 2008 in
conformity with accounting principles generally accepted in the United States of
America.
The
accompanying financial statements have been prepared assuming that the Company
will continue as a going concern. As discussed in Note 1, the Company has a
generated negative cash outflows from operating activities, experienced
recurring net operating losses, and is dependent on securing additional equity
and debt financing to support its business efforts. These factors raise
substantial doubt about the Company's ability to continue as a going concern.
Management's plans in regard to this matter are described in Note 1. The
financial statements do not include any adjustments that might result from the
outcome of this uncertainty.
|
/s/ RBSM LLP
|
|
|
New
York, New York
|
RBSM
LLP
|
|
March
30, 2009
|
F-2
GENSPERA
INC.
(A
Development Stage Company)
BALANCE
SHEETS
DECEMBER
31,2008 and 2007
|
2008
|
2007
|
|||||||
|
Assets
|
||||||||
|
Current
assets:
|
||||||||
|
Cash
|
$ | 534,290 | $ | 590,435 | ||||
|
Total
current assets
|
534,290 | 590,435 | ||||||
|
Intangible
assets, net of accumulated amortization of $11,511
|
172,657 | - | ||||||
|
Total
assets
|
$ | 706,947 | $ | 590,435 | ||||
|
Liabilities
and stockholders' equity
|
||||||||
|
Current
liabilities:
|
||||||||
|
Accounts
payable and accrued expenses
|
$ | 238,817 | $ | 3,874 | ||||
|
Accrued
interest - stockholder
|
5,399 | 14,800 | ||||||
|
Convertible
note payable - stockholder, current portion
|
50,000 | 35,000 | ||||||
|
Total
current liabilities
|
294,216 | 53,674 | ||||||
|
Convertible
note payable, net of discount of $11,046
|
152,554 | - | ||||||
|
Convertible
notes payable - stockholder, long term portion
|
105,000 | 120,000 | ||||||
|
Total
liabilities
|
551,770 | 173,674 | ||||||
|
Commitments
and contingencies
|
||||||||
|
Stockholders'
equity:
|
||||||||
|
Preferred
stock, par value $.0001 per share; 10,000,000 shares
authorized,
|
||||||||
|
none
issued and outstanding
|
- | - | ||||||
|
Common
stock, par value $.0001 per share; 80,000,000 shares
authorized,
|
||||||||
|
12,486,718
and 9,035,000 shares issued and outstanding
|
1,249 | 904 | ||||||
|
Additional
paid-in capital
|
4,922,174 | 1,857,842 | ||||||
|
Deficit
accumulated during the development stage
|
(4,768,246 | ) | (1,441,985 | ) | ||||
|
Total
stockholders' equity
|
155,177 | 416,761 | ||||||
|
Total
liabilities and stockholders' equity
|
$ | 706,947 | $ | 590,435 | ||||
See
accompanying notes to financial statements.
F-3
GENSPERA,
INC.
(A
Development Stage Company)
STATEMENTS
OF LOSSES
FOR THE
YEARS ENDED DECEMBER 31, 2008 AND 2007
AND FOR
THE PERIOD FROM INCEPTION (NOVEMBER 21, 2003) TO DECEMBER 31, 2008
|
Cumulative Period
|
||||||||||||
|
from November 21, 2003
|
||||||||||||
|
(date of inception) to
|
||||||||||||
|
Years ended December 31,
|
December 31,
|
|||||||||||
|
2008
|
2007
|
2008
|
||||||||||
|
Operating
expenses:
|
||||||||||||
|
General
and administrative expenses
|
$ | 854,294 | $ | 389,799 | $ | 1,289,542 | ||||||
|
Research
and development
|
2,432,991 | 294,440 | 3,424,407 | |||||||||
|
Total
operating expenses
|
3,287,285 | 684,239 | 4,713,949 | |||||||||
|
Loss
from operations
|
(3,287,285 | ) | (684,239 | ) | (4,713,949 | ) | ||||||
|
Finance
cost
|
(39,789 | ) | - | (39,789 | ) | |||||||
|
Interest
income (expense), net
|
813 | (6,960 | ) | (14,508 | ) | |||||||
|
Loss
before provision for income taxes
|
(3,326,261 | ) | (691,199 | ) | (4,768,246 | ) | ||||||
|
Provision
for income taxes
|
- | - | - | |||||||||
|
Net
loss
|
$ | (3,326,261 | ) | $ | (691,199 | ) | $ | (4,768,246 | ) | |||
|
Net
loss per common share, basic and diluted
|
$ | (0.30 | ) | $ | (0.10 | ) | ||||||
|
Weighted
average shares outstanding
|
11,030,657 | 7,103,904 | ||||||||||
See
accompanying notes to financial statements.
F-4
GENSPERA,
INC.
(A
Development Stage Company)
STATEMENT
OF STOCKHOLDERS' EQUITY
FROM DATE
OF INCEPTION (NOVEMBER 21, 2003) TO DECEMBER 31, 2008
|
Deficit
|
||||||||||||||||||||
|
Accumulated
|
||||||||||||||||||||
|
Additional
|
During the
|
|||||||||||||||||||
|
Common Stock
|
Paid-in
|
Development
|
Stockholders'
|
|||||||||||||||||
|
Shares
|
Amount
|
Capital
|
Stage
|
Equity
|
||||||||||||||||
|
Balance,
November 21, 2003
|
- | $ | - | $ | - | $ | - | $ | - | |||||||||||
|
Sale
of common stock to founders at $0.0001
|
||||||||||||||||||||
|
per
share in November, 2003
|
6,100,000 | 610 | (510 | ) | - | 100 | ||||||||||||||
|
Contributed
services
|
- | - | 120,000 | - | 120,000 | |||||||||||||||
|
Net
loss
|
- | - | - | (125,127 | ) | (125,127 | ) | |||||||||||||
|
Balance,
December 31, 2003
|
6,100,000 | 610 | 119,490 | (125,127 | ) | (5,027 | ) | |||||||||||||
|
Contributed
services
|
- | - | 192,000 | - | 192,000 | |||||||||||||||
|
Stock
based compensation
|
- | - | 24,102 | - | 24,102 | |||||||||||||||
|
Net
loss
|
- | - | - | (253,621 | ) | (253,621 | ) | |||||||||||||
|
Balance,
December 31, 2004
|
6,100,000 | 610 | 335,592 | (378,748 | ) | (42,546 | ) | |||||||||||||
|
Contributed
services
|
- | - | 48,000 | - | 48,000 | |||||||||||||||
|
Stock
based compensation
|
- | - | 24,100 | - | 24,100 | |||||||||||||||
|
Net
loss
|
- | - | - | (126,968 | ) | (126,968 | ) | |||||||||||||
|
Balance,
December 31, 2005
|
6,100,000 | 610 | 407,692 | (505,716 | ) | (97,414 | ) | |||||||||||||
|
Contributed
services
|
- | - | 144,000 | - | 144,000 | |||||||||||||||
|
Stock
based compensation
|
- | - | 42,162 | - | 42,162 | |||||||||||||||
|
Net
loss
|
- | - | - | (245,070 | ) | (245,070 | ) | |||||||||||||
|
Balance,
December 31, 2006
|
6,100,000 | 610 | 593,854 | (750,786 | ) | (156,322 | ) | |||||||||||||
|
Shares
sold for cash at $0.50 per share
|
||||||||||||||||||||
|
in
November, 2007
|
1,300,000 | 130 | 649,870 | - | 650,000 | |||||||||||||||
|
Shares
issued for services
|
735,000 | 74 | 367,426 | - | 367,500 | |||||||||||||||
|
Contributed
services
|
- | - | 220,000 | - | 220,000 | |||||||||||||||
|
Stock
based compensation
|
- | - | 24,082 | - | 24,082 | |||||||||||||||
|
Exercise
of options for cash at $0.003 per share
|
||||||||||||||||||||
|
in
March and June, 2007
|
900,000 | 90 | 2,610 | - | 2,700 | |||||||||||||||
|
Net
loss
|
- | - | - | (691,199 | ) | (691,199 | ) | |||||||||||||
|
Balance,
December 31, 2007
|
9,035,000 | 904 | 1,857,842 | (1,441,985 | ) | 416,761 | ||||||||||||||
|
Exercise
of options for cash at $0.50 per share
|
||||||||||||||||||||
|
on
March 7,2008
|
1,000,000 | 100 | 499,900 | - | 500,000 | |||||||||||||||
|
Sale
of common stock and warrants at $1.00 per
|
||||||||||||||||||||
|
share
- July and August 2008
|
2,320,000 | 232 | 2,319,768 | - | 2,320,000 | |||||||||||||||
|
Cost
of sale of common stock and warrants
|
- | - | (205,600 | ) | - | (205,600 | ) | |||||||||||||
|
Shares
issued for accrued interest
|
31,718 | 3 | 15,856 | - | 15,859 | |||||||||||||||
|
Shares
issued for services
|
100,000 | 10 | 49,990 | - | 50,000 | |||||||||||||||
|
Stock
based compensation
|
- | - | 313,743 | - | 313,743 | |||||||||||||||
|
Contributed
services
|
- | - | 50,000 | - | 50,000 | |||||||||||||||
|
Beneficial
conversion feature of convertible debt
|
- | - | 20,675 | - | 20,675 | |||||||||||||||
|
Net
loss
|
- | - | - | (3,326,261 | ) | (3,326,261 | ) | |||||||||||||
|
Balance,
December 31, 2008
|
12,486,718 | $ | 1,249 | $ | 4,922,174 | $ | (4,768,246 | ) | $ | 155,177 | ||||||||||
See
accompanying notes to financial statements.
F-5
GENSPERA,
INC.
(A
Development Stage Company)
STATEMENTS
OF CASH FLOWS
FOR THE
YEARS ENDED DECEMBER 31, 2008 AND 2007
AND FOR
THE PERIOD FROM INCEPTION (NOVEMBER 21, 2003) TO DECEMBER 31, 2008
|
Cumulative Period
|
||||||||||||
|
from November 21, 2003
|
||||||||||||
|
(date of inception) to
|
||||||||||||
|
Years ended December 31,
|
December 31,
|
|||||||||||
|
2008
|
2007
|
2008
|
||||||||||
|
Cash
flows from operating activities:
|
||||||||||||
|
Net
loss
|
$ | (3,326,261 | ) | $ | (691,199 | ) | $ | (4,768,246 | ) | |||
|
Adjustments
to reconcile net loss to net
|
||||||||||||
|
cash
used in operating activities:
|
||||||||||||
|
Amortization
|
11,511 | - | 11,511 | |||||||||
|
Stock
based compensation
|
363,743 | 391,582 | 845,689 | |||||||||
|
Contributed
services
|
50,000 | 220,000 | 774,000 | |||||||||
|
Amortization
of debt discount
|
9,629 | - | 9,629 | |||||||||
|
Increase
in accounts payable and accrued expenses
|
241,401 | 1,589 | 260,075 | |||||||||
|
Cash
used in operating activities
|
(2,649,977 | ) | (78,028 | ) | (2,867,342 | ) | ||||||
|
Cash
flows from investing activities:
|
||||||||||||
|
Acquisition
of intangibles
|
(184,168 | ) | - | (184,168 | ) | |||||||
|
Cash
used in investing activities
|
(184,168 | ) | - | (184,168 | ) | |||||||
|
Cash
flows from financing activities:
|
||||||||||||
|
Proceeds
from sale of common stock and warrants
|
2,778,000 | 652,700 | 3,430,800 | |||||||||
|
Proceeds
from convertible notes - stockholder
|
- | - | 155,000 | |||||||||
|
Cash
provided by financing activities
|
2,778,000 | 652,700 | 3,585,800 | |||||||||
|
Net
increase (decrease) in cash
|
(56,145 | ) | 574,672 | 534,290 | ||||||||
|
Cash,
beginning of period
|
590,435 | 15,763 | - | |||||||||
|
Cash,
end of period
|
$ | 534,290 | $ | 590,435 | $ | 534,290 | ||||||
|
Supplemental
cash flow information:
|
||||||||||||
|
Cash
paid for interest
|
$ | - | $ | - | ||||||||
|
Cash
paid for income taxes
|
$ | - | $ | - | ||||||||
|
Non-cash
financial activities:
|
||||||||||||
|
Accrued
interest paid with common stock
|
$ | 15,859 | $ | - | ||||||||
|
Convertible
note issued as payment of placement fees
|
163,600 | - | ||||||||||
|
Fair
value of warrants issued with convertible debt recorded
|
||||||||||||
|
as
debt discount
|
20,675 | - | ||||||||||
See
accompanying notes to financial statements.
F-6
GENSPERA,
INC.
(A
Development Stage Company)
NOTES
TO FINANCIAL STATEMENTS
FOR
THE YEARS ENDED DECEMBER 31, 2008 AND 2007
AND
FOR THE PERIOD FROM NOVEMBER 21, 2003
(INCEPTION)
TO DECEMBER 31, 2008
NOTE
1 - SUMMARY OF ACCOUNTING POLICIES
A summary
of the significant accounting policies applied in the preparation of the
accompanying financial statements follows.
Business
and Basis of Presentation
GenSpera
Inc. (“we”, “us”, “
our company “, “our”, “GenSpera” or the “Company” )
was formed under the laws of the State of Delaware in 2003. We are a development
stage company, as defined by Statement of Financial Accounting Standards
(“SFAS”) No. 7. GenSpera, Inc. is a pharmaceutical company focused on the
development of targeted cancer therapeutics for the treatment of cancerous
tumors, including breast, prostate, bladder and kidney cancer. Our operations
are based in San Antonio, Texas.
To date,
we have generated no sales revenues, have incurred significant expenses and have
sustained losses. Consequently, our operations are subject to all the risks
inherent in the establishment of a new business enterprise. For the period from
inception on November 21, 2003 through December 31, 2008, we have accumulated
losses of $4,768,246.
Going
Concern
The
accompanying financial statements have been prepared on a going concern basis,
which contemplates the realization of assets and the satisfaction of liabilities
in the normal course of business. As shown in the accompanying financial
statements during years ended December 31, 2008 and 2007 the Company incurred
losses of $3,326,261 and $691,199 respectively. These factors among others may
indicate that the Company will be unable to continue as a going concern for a
reasonable period of time.
In order
to address our capital requirements, we intend to seek to raise additional cash
for working capital purposes through the public or private sales of debt or
equity securities, the procurement of advances on contracts or licenses, funding
from joint-venture or strategic partners, debt financing or short-term loans, or
a combination of the foregoing. We may also seek to satisfy indebtedness without
any cash outlay through the private issuance of debt or equity securities. There
can be no assurance the Company will be successful in its effort to secure
additional equity financing.
If
operations and cash flows continue to improve through these efforts, management
believes that the Company can continue to operate. However, no assurance can be
given that management's actions will result in profitable operations or the
resolution of its liquidity issues.
The
accompanying financial statements do not include any adjustments that might
result should the Company be unable to continue as a going concern.
Use
of Estimates
The
preparation of financial statements in conformity with generally accepted
accounting principles requires management to make estimates and assumptions that
affect the amounts reported in the financial statements and accompanying
disclosures. Although these estimates are based on management's best knowledge
of current events and actions the Company may undertake in the future, actual
results may differ from those estimates.
Research
and Development
Research
and development costs include expenses incurred by the Company for research and
development of therapeutic agents for the treatment of cancer and are charged to
operations as incurred. Our research and development expenses consist primarily
of expenditures for toxicology and other studies, manufacturing, and
compensation and consulting costs.
F-7
NOTE
1 - SUMMARY OF ACCOUNTING POLICIES (cont’d)
GenSpera
incurred research and development expenses of $2,432,991, $294,440 and
$3,424,407 for the years ended December 31, 2008 and 2007, and from November 21,
2003 (inception) through December 31, 2008, respectively.
Cash
Equivalents
For
purposes of the statements of cash flows, we consider all highly liquid debt
instruments purchased with a maturity date of three months or less to be cash
equivalents. We maintain our cash in bank deposit accounts which, at times, may
exceed insured limits. We have not experienced any losses in our
accounts.
Concentrations
of Credit Risk
Financial
instruments and related items, which potentially subject the Company to
concentrations of credit risk, consist primarily of cash and cash equivalents.
The Company places its cash and temporary cash investments with credit quality
institutions. At times, such investments may be in excess of applicable
government mandated insurance limits. At December 31, 2008, deposits exceeded
current insurance limits by approximately $284,000.
Fair
Value of Financial Instruments
SFAS No.
107, "Disclosures About Fair Value of Financial Instruments," requires
disclosure of the fair value of certain financial instruments. The carrying
value of cash and cash equivalents, accounts payable and short-term borrowings,
as reflected in the balance sheets, approximate fair value because of the
short-term maturity of these instruments.
Intangible
Assets
Intangible
assets consist of 5 issued patents and 3 patent applications pending worldwide
(see Note 4). These patents and patent applications cover the intellectual
property underlying our technology. The assets are recorded at cost. The patents
are being amortized on the straight line basis over their estimated useful lives
of twelve years.
Loss
Per Share
We use
SFAS No. 128, “Earnings Per Share” for calculating the basic and diluted
loss per share. We compute basic loss per share by dividing net loss and net
loss attributable to common shareholders by the weighted average number of
common shares outstanding. Basic and diluted loss per share are the same, in
that any potential common stock equivalents would have the effect of being
anti-dilutive in the computation of net loss per share. There were 3,713,598
common share equivalents at December 31, 2008 and 339,600 at December 31,
2007. For the years ended December 31, 2008 and 2007, these potential
shares were excluded from the shares used to calculate diluted earnings per
share as their inclusion would reduce net loss per share.
Income
Taxes
We have
adopted the provisions of SFAS No. 109 "Accounting for Income Taxes"("SFAS
109"). SFAS 109 requires recognition of deferred tax liabilities and assets for
the expected future tax consequences of events that have been included in the
financial statements or tax returns. Under this method, deferred tax liabilities
and assets are determined based on the differences between the financial
statement and tax basis of assets and liabilities using enacted tax rates in
effect for the year in which the differences are expected to reverse. The
Company has incurred losses that can be carried forward to offset future
earnings if conditions of the Internal Revenue Codes are met. A valuation
allowance is recorded to reduce a deferred tax asset to that portion that is
expected to more likely than not be realized.
In July
2006, the FASB issued FIN 48, "Accounting for Uncertainty in Income Taxes-an
interpretation of FASB Statement No. 109" ("FIN 48"). FIN 48 clarifies the
recognition threshold and measurement of a tax position taken on a tax return.
FIN 48 is effective for fiscal years beginning after December 15, 2006. FIN 48
also requires expanded disclosure with respect to the uncertainty in income
taxes. The adoption of FIN 48 did not have any effect on the Company’s financial
position or results of operations.
F-8
NOTE
1 - SUMMARY OF ACCOUNTING POLICIES (cont’d)
Stock-Based
Compensation
We
account for our share-based compensation under the provisions of FASB Statement
No. 123(R), “Share-Based Payment”, (“FAS 123R”). We adopted FAS 123R as of
January 1, 2006, using the modified prospective application method. Prior to
January 1, 2006 we applied the provisions of FAS 123, “Accounting for
Stock-Based Compensation”.
Recent
Accounting Pronouncements
In
September 2006, the Financial Accounting Standards Board issued Statement of
Financial Accounting Standards No.157, Fair Value Measurements
(“SFAS 157”). SFAS 157 defines fair value to be the price that would be
received to sell an asset or paid to transfer a liability in an orderly
transaction between market participants at the measurement date and emphasizes
that fair value is a market-based measurement, not an entity-specific
measurement. It establishes a fair value hierarchy and expands disclosures
about fair value measurements in both interim and annual periods. SFAS 157 is
effective for fiscal years beginning after November 15, 2007 and interim periods
within those fiscal years. The Company adopted SFAS 157 on January 1, 2008 which
did not have a material impact on its consolidated financial position and
results of operations. The Company also adopted the deferral provisions of the
Financial Accounting Standards Board Staff Position No. 157-2, which delays
the effective date of SFAS No. 157 for all nonrecurring fair value
measurements of non-financial assets and liabilities until fiscal years
beginning after November 15, 2008.
Fair
value is defined as the exchange price that would be received for an asset or
paid to transfer a liability (an exit price) in the principal or most
advantageous market for the asset or liability in an orderly transaction between
market participants on the measurement date. SFAS No. 157 also
expands disclosures about instruments measured at fair value and establishes a
fair value hierarchy which requires an entity to maximize the use of observable
inputs and minimize the use of unobservable inputs when measuring fair
value. The standard describes three levels of inputs that may be used to
measure fair value:
Level 1 —
Quoted prices for identical assets and liabilities in active
markets;
Level 2 —
Quoted prices for similar assets and liabilities in active markets; quoted
prices for identical or similar assets and liabilities in markets that are not
active; and model-derived valuations in which all significant inputs and
significant value drivers are observable in active markets;
and
Level 3 —
Valuations derived from valuation techniques in which one or more significant
inputs or significant value drivers are unobservable.
The
Company designates cash equivalents as Level 1. As of December 31, 2008 and
2007, the Company did not have any cash equivalents, therefore there were no
assets measured at fair value.
In
February 2007, the FASB issued Statement of Financial Accounting Standards
(“SFAS”) No. 159, The Fair
Value Option for Financial Assets and Financial Liabilities-Including an
Amendment of FASB Statement No. 115 (“SFAS 159”). SFAS 159
permits entities to measure eligible assets and liabilities at fair
value. Unrealized gains and losses on items for which the fair value
option has been elected are reported in earnings. SFAS 159 is
effective for fiscal years beginning after November 15, 2007. The
Company adopted SFAS 159 on January 1, 2008 and did not elect the fair value
option which did not have a material impact on its financial position and
results of operations.
In
December 2007, the Financial Accounting Standards Board issued Statement of
Financial Accounting Standards No. 141R, Business Combinations , and
Statement of Financial Accounting Standards No. 160, Noncontrolling Interests in
Consolidated Financial Statements, an amendment of ARB No. 51
. These new standards significantly change the accounting for and
reporting of business combination transactions and noncontrolling interests
(previously referred to as minority interests) in consolidated financial
statements. Both standards are effective for fiscal years beginning on or
after December 15, 2008, with early adoption prohibited. These Statements are
effective for the Company beginning on January 1, 2009. The Company is
currently evaluating the provisions of FAS 141(R) and FAS 160.
In
March 2008, the Financial Accounting Standards Board issued Statement of
Financial Accounting Standards No. 161, Disclosures About Derivative
Instruments and Hedging Activities, an amendment of FASB Statement No.
133 . This new standard enhances the disclosure requirements related
to derivative instruments and hedging activities required by FASB Statement No. 133
. This standard is effective for fiscal years and interim periods
beginning after November 15, 2008, with early adoption encouraged. The Company
adopted the required provisions of SFAS 161 on January 1, 2008 and the adoption
did not have a significant impact on its financial position and results of
operations.
F-9
NOTE
1 - SUMMARY OF ACCOUNTING POLICIES (cont’d)
In June
2008, the FASB issued Emerging Issues Task Force No. 07-5 (EITF 07-5),
Determining Whether an
Instrument (or Embedded Feature) is Indexed to an Entity’s Own Stock.
EITF 07-5 requires entities to evaluate whether an equity-linked financial
instrument (or embedded feature) is indexed to its own stock by assessing the
instrument’s contingent exercise provisions and settlement provisions.
Instruments not indexed to their own stock fail to meet the scope exception of
Statement of Financial Accounting Standards No. 133, Accounting for Derivative
Instruments and Hedging Activities, paragraph 11(a), and should be
classified as a liability and marked-to-market. The statement is effective for
fiscal years beginning after December 15, 2008 and is to be applied to
outstanding instruments upon adoption with the cumulative effect of the change
in accounting principle recognized as an adjustment to the opening balance of
retained earnings. The Company has assessed its outstanding equity-linked
financial instruments and has concluded that effective January 1, 2009,
warrants issued during 2008 with a fair value of $734,617 on December 31,
2008 will need to be reclassified from equity to a liability. The cumulative
effect of the change in accounting principle on January 1, 2009 will be a
$290,455 increase to the deficit accumulated during development
stage.
NOTE
2 - CAPITAL STOCK AND STOCKHOLDER’S EQUITY
We are
authorized to issue 80,000,000 shares of common stock with a par value of $.0001
per share and 10,000,000 shares of preferred stock with a par value of $.0001
per share.
Our Chief
Executive Officer has provided his services without compensation from inception
through November 2007. We have recorded compensation expense for these
contributed services, with the corresponding credit to additional paid-in
capital. For the year ended December 31, 2007, we have recorded compensation
expense of $220,000. Our Chief Operating Officer provided his services without
compensation for the second quarter of 2008. We have recorded compensation
expense for these contributed services in the amount of $50,000, with the
corresponding credit to additional paid-in capital. For the period from November
21, 2003 to December 31, 2008, compensation expense for contributed services
aggregated $774,000.
On
January 1, 2008, we granted a total of 1,000,000 common stock warrants to
consultants for financial services. The warrants have an exercise price
of $0.50 per share. The warrants vested upon grant. We have recorded an
expense of $89,680 during 2008 related to the fair value of the warrants that
vested during that period, using the Black-Scholes method based on the following
assumptions: (1) risk free interest rate of 3.2%;
(2) dividend yield of 0%; (3) volatility factor of the expected market
price of our common stock of 89%; and (4) an expected life of the
warrants of .25 years. The warrants were exercised during March and we
received proceeds of $500,000.
On
January 7, 2008, we granted 100,000 shares of common stock, valued at $50,000,
to a director as payment for services. The shares were vested upon
grant.
On
February 1, 2008, we granted a total of 240,000 common stock options to members
of our Scientific Advisory Board. The options have an exercise price
of $0.50 per share. The options vest in equal installments quarterly over a
period of three years commencing March 31, 2008, and lapse if unexercised on
January 31, 2018. We have recorded an expense of $35,643 during 2008
related to the fair value of the options that vested during that period, using
the Black-Scholes method based on the following weighted average
assumptions: (1) risk free interest rate of 2.2%;
(2) dividend yield of 0%; (3) volatility factor of the expected market
price of our common stock of 111%; and (4) an expected life of the
options of 2 years.
On
February 11, 2008, we granted a total of 100,000 common stock options to a
consultant for investor relation services. The options have an exercise price
of $0.50 per share and expire if unexercised on February 11, 2013. The
options vest 20,000 upon grant and 80,000 upon the attainment of certain
financial milestones. Any options not vesting by June 30, 2008 terminate on that
date. Of the 80,000 options subject to the attainment of financial milestones,
64,000 vested on June 30, 2008 and the balance were terminated. We have recorded
an expense of $21,906 during 2008 related to the fair value of the options that
vested during that period, using the Black-Scholes method based on the following
weighted average assumptions: (1) risk free interest rate
of 2.7%; (2) dividend yield of 0%; (3) volatility factor of the
expected market price of our common stock of 97%; and (4) an expected
life of the options of 2 years.
On
February 11, 2008, we granted a total of 20,000 common stock options to a
consultant for professional services. The options have an exercise price
of $0.50 per share. The options vest in equal installments quarterly over a
period of one year commencing March 31, 2008, and lapse if unexercised on
February 11, 2018. We have recorded an expense of $8,910 during 2008
related to the fair value of the options that vested during that period, using
the Black-Scholes method based on the following weighted average
assumptions: (1) risk free interest rate of 2.2%;
(2) dividend yield of 0%; (3) volatility factor of the expected market
price of our common stock of 111%; and (4) an expected life of the
options of 2 years.
F-10
NOTE
2 - CAPITAL STOCK AND STOCKHOLDER’S EQUITY (cont’d)
On March
6, 2008, we granted a total of 1,000,000 common stock warrants to consultants
for financial services. The warrants have an exercise price of $1.00 per
share. The warrants vested upon grant and expire if unexercised on March 6,
2011. We have recorded an expense of $76,338 during 2008 related to the fair
value of the warrants that vested during that period, using the Black-Scholes
method based on the following assumptions: (1) risk free
interest rate of 2%; (2) dividend yield of 0%; (3) volatility
factor of the expected market price of our common stock of 89%; and
(4) an expected life of the warrants of 1 year.
During
March 2008, we granted to each of three new members of our board of directors,
as compensation for serving on our board of directors, options to
purchase 100,000 common shares at $0.50 per share, reflecting the fair
market value of the shares as of that date. The options vest 50,000 each upon
grant with the balance vesting quarterly over a period of two years commencing
March 31, 2008, and lapse if unexercised on April 1, 2018. The 300,000 options
have been valued at $72,208 at the date of grant using the Black-Scholes method
based on the following assumptions: (1) risk free interest rate
of 2%; (2) dividend yield of 0%; (3) volatility factor of the
expected market price of our common stock of 100%; and (4) an expected
life of the options of 2 years. We have recorded an expense of $52,652
during 2008 based on the options that vested during that period. On September
30, 2008 one of the directors resigned from the board, but will continue to
perform services as a consultant. His remaining options, totaling 31,250
options, will be valued and recorded as compensation as they vest. During the
fourth quarter of 2008, 6,250 options vested. We have recorded an expense of
$4,203 related to the fair value of these options, using the Black-Scholes
method based on the following assumptions: (1) risk free
interest rate of 0.9%; (2) dividend yield of 0%; (3) volatility
factor of the expected market price of our common stock of 144%; and
(4) an expected life of the options of 2 years.
On March
7, 2008, we issued 31,718 shares of common stock to our president and chief
executive officer as payment of accrued interest in the amount of $15,859. Of
this amount, $14,800 had been accrued at December 31, 2007.
During
July and August of 2008, we sold an aggregate of 2,320,000 units resulting in
gross proceeds of $2,320,000 or $1.00 per unit. Net cash received was
$2,278,000. Each unit consists of 1 share of common stock and ½ common stock
purchase warrant. The warrants have a term of 5 years and an exercise price of
$2.00 per share subject to certain anti-dilution adjustments. The fair value of
the warrants has been recorded as permanent equity since the warrants are
exercisable into unregistered shares of our common stock and do not contain any
net cash settlement provisions. The warrants are also callable by the Company in
the event the Company’s shares are publically traded in the future and certain
price and volume conditions are met.
TR
Winston & Company, LLC (“TR Winston”) acted as the Company’s placement agent
with respect to the transaction. Pursuant to a placement agent agreement with TR
Winston we agreed to the following compensation: (i) cash fee equal to 8% of
gross proceeds raised, including any payments made to the Company upon the
exercise of the warrants; (ii) the issuance of a warrant to purchase 8% of all
securities issued; and (iii) payment of legal expenses totaling $20,000. As an
accommodation to the Company, TR Winston agreed to receive a convertible
debenture and warrants to purchase an additional 81,800 common shares in lieu of
$163,600 of its cash fee.
On
October 16, 2008, we granted a total of 50,000 common stock warrants to a
consultant for marketing services. The warrants have an exercise price
of $2.00 per share and expire if unexercised on October 16, 2013. The
warrants vested upon grant. We have recorded an expense of $17,238 during 2008
related to the fair value of the options, using the Black-Scholes method based
on the following assumptions: (1) risk free interest rate
of 2.9%; (2) dividend yield of 0%; (3) volatility factor of the
expected market price of our common stock of 111%; and (4) an expected
life of the options of 2 years.
On
October 16, 2008, we granted a total of 15,000 common stock options to a
director as compensation for serving on a special committee. The options have an
exercise price of $1.00 per share and expire if unexercised on October 16,
2018. The options vested upon grant. We have recorded an expense of $7,173
during 2008 related to the fair value of the options, using the Black-Scholes
method based on the following assumptions: (1) risk free
interest rate of 2.9%; (2) dividend yield of 0%; (3) volatility
factor of the expected market price of our common stock of 111%; and
(4) an expected life of the options of 2 years.
On June
7, 2004, we granted a total of 750,000 common stock options to members of our
Scientific Advisory Board. The options vested over a four year period on
December 31 of each year. On May 15, 2007 our board approved a resolution to
accelerate the vesting of the remaining 187,500 unvested options. At that time,
all of the options were exercised and we received proceeds of $1,200. We have
recorded compensation expense of $24,082 for the nine months ended September 30,
2007 related to the fair value of the options that vested during that period,
using the Black-Scholes method based on the following assumption
ranges: (1) risk free interest rate of 3% - 4.9%;
(2) dividend yield of 0%; (3) estimated volatility factor of the
expected market price of our common stock of 89%; and (4) an expected
life of the options of .5 - 3 years.
F-11
NOTE
2 - CAPITAL STOCK AND STOCKHOLDER’S EQUITY (cont’d)
On
November 10, 2006, we granted a total of 150,000 common stock options to
employees and advisors. The options vested upon grant. All of these options were
exercised during 2007 and we received proceeds of $1,500.
During
2007, we issued an aggregate of 735,000 shares of common stock, valued at
$367,500, as compensation for consulting and financial and legal advisory
services.
During
November 2007, we sold an aggregate of 1,300,000 common shares in a private
placement at $0.50 per share, for proceeds of $650,000.
NOTE
3 -CONVERTIBLE NOTES PAYABLE
We have
executed five convertible notes with our president and chief executive officer
pursuant to which we have borrowed an aggregate of $155,000. The notes bear an
interest rate of 4.2% and mature at various dates through December 6, 2011.
Interest accrued through February 29, 2008 was $15,859. On March 7, 2008 we
issued 31,718 shares of common stock as payment of this amount. Accrued interest
at December 31, 2008 was $5,399. The notes and accrued interest are convertible,
at the option of the holder, into shares of our common stock at a conversion
price of $0.50 per share.
As an
accommodation to the Company, TR Winston & Company, LLC, our placement
agent, agreed to receive a convertible debenture in the principal amount of
$163,600 plus warrants to purchase an additional 81,800 common shares in lieu of
$163,600 of its cash fee. The convertible debenture accrues interest at 5% per
annum and has a maturity date of July 14, 2010 (extended from July 14, 2009 –
see Note 7, Subsequent Events). It is convertible into the shares of the
Company’s common stock, at the sole discretion of the holder, at $1.00 per share
subject to certain anti-dilution adjustments.
In
accordance with Emerging Issues Task Force (“EITF”) No.00-27, “ Application of EITF Issue
No. 98-5, ‘Accounting for Convertible Securities with Beneficial Conversion
Features or Contingently Adjustable Conversion Rates’, to Certain Convertible
Instruments ”, a
portion of the proceeds were allocated to the warrants based on their relative
fair value, which totaled $20,675 using the Black Scholes option pricing
model. This amount has been recorded as a debt discount and will be amortized
over the term of the debenture. We determined that there was no beneficial
conversion feature attributable to the convertible debenture since the effective
conversion price was greater than the value of our common shares on the date of
issuance. The assumptions used in the Black Scholes model are as
follows: (1) dividend yield of 0%; (2) expected volatility
of 100%, (3) risk-free interest rate of 2.9%, and
(4) expected life of 2 year.
Principal
amounts of the notes mature as follows:
|
Years
ended December 31,
|
||||
|
2009
|
$
|
50,000
|
||
|
2010
|
198,600
|
|||
|
2011
|
70,000
|
|||
|
$
|
318,600
|
|||
NOTE
4 – INTELLECTUAL PROPERTY
We have
acquired know-how, pre-clinical data, development data and related patent
portfolios for a series of technologies that relate to targeted, potentially
curative treatments for a variety of human cancers. We currently own 5 issued
patents and 3 patent applications pending worldwide. The previous owner of
the intellectual property, John Hopkins University, agreed to assign the patents
underlying the technology to our co-founders (the “Assignee Co-Founders”) in
return for their assumption of future patent fees and costs, and patent attorney
fees and costs, associated with all of the assigned technology. In exchange for
us continuing to pay for these future costs, the Assignee Co-Founders entered
into world-wide, exclusive option agreements with us. In April 2008, upon the
reimbursement of approximately $122,778 in previously-paid patent costs, fees
and expenses to John Hopkins University, the Assignee Co-Founders assigned to
GenSpera all right, title, and interest in and to the intellectual property, and
GenSpera subsequently recorded these assignments in the United States
Patent & Trademark Office. By virtue of the April 2008 assignments, GenSpera
has no further financial obligations to the Assignee Co-Founders or to John
Hopkins University with regard to the assigned intellectual property. These
reimbursement costs were required to be paid by the Assignee Co-Founders to
Johns Hopkins University. As part of our agreements with the Assignee
Co-Founders, we have provided these reimbursement costs directly to the Assignee
Co-Founders specifically for reimbursement to Johns Hopkins University. Because
these payments have been made by us to the
F-12
NOTE
4 – INTELLECTUAL PROPERTY (cont’d)
Assignee
Co-Founders, this may trigger a taxable event such that the Assignee Co-Founders
may be required to pay Federal and state taxes (if any) based upon our payment
of the reimbursement costs to the Assignee Co-Founders. Therefore, as part
of our agreements with the Assignee Co-Founders, we have further provided
additional funds to cover applicable Federal and state taxes (if any)
associated with the reimbursement payments. Under our agreement with the
Assignee Co-Founders, we will not be required to make any other future payments,
including fees, milestone or royalty fees, to either Johns Hopkins University or
the Assignee Co-Founders.
On March
10, 2008, we paid an aggregate of $184,167 to acquire the 5 issued patents and 3
patent applications pending worldwide described above. Amortization expense
recorded during the period ended December 31, 2008 was $11,511.
Amortization
expense for each on the next five fiscal years is estimated to be $15,348 per
year.
NOTE
5- STOCK OPTIONS AND WARRANTS
We have
one equity incentive plan, our 2007 Equity Compensation Plan (“2007 Plan”). Our
2007 Plan is administered by a committee of non-employee directors who are
appointed by our board of directors (“Committee”). The purpose of our 2007 Plan
is to advance the interests of GenSpera and our stockholders by attracting,
retaining and rewarding persons performing services for us and to motivate such
persons to contribute to our growth and profitability.
Under our
2007 Plan, we may grant stock options and restricted stock to employees,
directors and consultants. Our 2007 Plan authorizes the issuance of up to
1,500,000 shares of our common stock for the foregoing awards. The exercise
price of Nonqualified Stock Options shall not be less than 85% of the fair
market value per share on the date of grant. The exercise price per share for
Incentive Stock Option grants must be no less than 100% of the fair market value
per share on the date of grant. The exercise price per share for an incentive
stock option grant to an employee who, at the time of grant, owns stock
representing more than 10% of the voting power of all classes of stock of
GenSpera or any parent or subsidiary, must be no less than 110% of the fair
market value per share on the date of grant.
Generally,
the option exercise price may be paid in cash, by check, by cashless exercise,
by net exercise or by tender or attestation of ownership of shares having a fair
market value not less than the exercise price and that either (A) have been
owned by the optionee for more than six months and not used for another exercise
by tender or attestation, or (B) were not acquired, directly or indirectly,
from us.
At the
time an award is granted, the Committee must fix the period within which the
award may be exercised and determine any conditions that must be satisfied
before the award may be exercised. Notwithstanding, options shall vest over a
period of not more than five years and at a rate of not less than 20% per year.
The Committee may accelerate the exercisability of any or all outstanding
options at any time for any reason. The maximum term of an option granted under
our 2007 Plan is ten years.
Our 2007
Plan provides that in the event of our merger with or into another corporation,
the sale of substantially all of our assets, or the sale or exchange of more
than 50% of our voting stock, each outstanding award shall be assumed or an
equivalent award substituted by the surviving, continuing, successor or
purchasing corporation or a parent thereof. The Committee may also deem an award
assumed if the award confers the right to the award-holder to receive, for each
share of stock subject to an award immediately prior to the change in control,
the consideration that a stockholder is entitled on the effective date of the
change in control. Upon a change in control, all outstanding options shall
automatically accelerate and become fully exercisable and all restrictions and
conditions on all outstanding restricted stock grants shall immediately
lapse.
The
Committee may at any time amend, suspend or terminate our 2007 Plan.
Notwithstanding the forgoing, the Committee shall not amend the Plan without
shareholder approval if such approval is required by section 422 of the Internal
Revenue Code or section 162(m) therein.
Transactions
involving our stock options are summarized as follows:
|
2008
|
2007
|
|||||||||||||||
|
Number
|
Weighted
Average
Exercise Price
|
Number
|
Weighted
Average Exercise
Price
|
|||||||||||||
|
Outstanding
at beginning of the period
|
— | $ | — | 900,000 | $ | 0.003 | ||||||||||
|
Granted
during the period
|
675,000 | 0.51 | — | — | ||||||||||||
|
Exercised
during the period
|
— | — | (900,000 | ) | 0.003 | |||||||||||
|
Terminated
during the period
|
(16,000 | ) | 0.50 | — | — | |||||||||||
|
Outstanding
at end of the period
|
659,000 | $ | 0.51 | — | $ | — | ||||||||||
|
Exercisable
at end of the period
|
424,000 | $ | 0.52 | — | $ | — | ||||||||||
The
intrinsic value of options exercised during 2007 was approximately
$114,000.
At
December 31, 2008 employee options outstanding totaled 215,000 with a weighted
average exercise price of $0.53. These options had an intrinsic value of $74,000
and a remaining contractual term of 9.3 years. Of these options, 165,000 are
exercisable at December 31, 2008, with an intrinsic value of $55,500 and a
remaining weighted average contractual term of 9.3 years. Compensation cost
related to the unvested employee options not yet recognized is $12,036 at
December 31, 2008. This amount will be recognized during 2009.
The
weighted average remaining life of the options is 8.6 years.
Transactions
involving our stock warrants are summarized as follows:
|
2008
|
2007
|
|||||||||||||||
|
Number
|
Weighted
Average
Exercise Price
|
Number
|
Weighted
Average Exercise
Price
|
|||||||||||||
|
Outstanding
at beginning of the period
|
— | $ | — | — | $ | — | ||||||||||
|
Granted
during the period
|
3,570,200 | 1.30 | — | — | ||||||||||||
|
Exercised
during the period
|
(1,000,000 | ) | 0.50 | — | — | |||||||||||
|
Terminated
during the period
|
— | — | — | — | ||||||||||||
|
Outstanding
at end of the period
|
2,570,200 | $ | 1.61 | — | $ | — | ||||||||||
|
Exercisable
at end of the period
|
2,570,200 | $ | 1.61 | — | $ | — | ||||||||||
The
weighted average remaining life of the warrants is 3.6 years.
F-13
NOTE
6 - INCOME TAXES
During
the year ended December 31, 2007, we adopted Financial Accounting Standards
Board (FASB) Interpretation No. 48, "Accounting for Uncertainty in Income Taxes"
(FIN 48), which supplements SFAS No. 109, "Accounting for Income Taxes," by
defining the confidence level that a tax position must meet in order to be
recognized in the financial statements. The Interpretation requires that the tax
effects of a position be recognized only if it is "more-likely-than-not" to be
sustained based solely on its technical merits as of the reporting date. The
more-likely-than-not threshold represents a positive assertion by management
that a company is entitled to the economic benefits of a tax position. If a tax
position is not considered more-likely- than-not to be sustained based solely on
its technical merits no benefits of the tax position are to be recognized.
Moreover, the more-likely-than-not threshold must continue to be met in each
reporting period to support continued recognition of a benefit. With the
adoption of FIN 48, companies are required to adjust their financial statements
to reflect only those tax positions that are more-likely-than-not to be
sustained. Any necessary adjustment would be recorded directly to retained
earnings and reported as a change in accounting principle.
SFAS No.
109 requires the recognition of deferred tax liabilities and assets for the
expected future tax consequences of events that have been included in the
financial statement or tax returns. Under this method, deferred tax liabilities
and assets are determined based on the difference between financial statements
and tax bases of assets and liabilities using enacted tax rates in effect for
the year in which the differences are expected to reverse. Temporary differences
between taxable income reported for financial reporting purposes and income tax
purposes are insignificant.
Net
operating losses for tax purposes of approximately $3,171,000 at December 31,
2008 are available for carryover. The net operating losses will expire from 2013
through 2028. We have provided a 100% valuation allowance for the deferred tax
benefits resulting from the net operating loss carryover and our tax credits due
to our limited operating history. In addressing the realizability of deferred
tax assets, management considers whether it is more likely than not that some
portion or all of the deferred tax assets will not be realized. The ultimate
realization of deferred tax assets is dependent upon the generation of future
taxable income during the periods in which those temporary differences are
deductible. The valuation allowance increased by $1,089,000 and $31,000 during
the years ended December 31, 2008 and 2007, respectively. A reconciliation of
the statutory Federal income tax rate and the effective income tax rate for the
years ended December 31, 2008 and 2007 follows.
Significant
components of deferred tax assets and liabilities are as follows:
|
2008
|
2007
|
|||||||
|
Deferred
tax assets:
|
||||||||
|
Net
operating loss carryforward
|
1,078,000 | 94,000 | ||||||
|
Tax
credits
|
105,000 | — | ||||||
|
Valuation
allowance
|
(1,183,000 | ) | (94,000 | ) | ||||
|
Net
deferred tax assets
|
$ | - | $ | - | ||||
|
Statutory
federal income tax rate
|
-34 | % | -34 | % | ||||
|
State
income taxes, net of federal taxes
|
-0 | % | -7 | % | ||||
|
Non-deductible
items
|
4 | % | 30 | % | ||||
|
Tax
credits
|
3 | % | — | |||||
|
Valuation
allowance
|
27 | % | 11 | % | ||||
|
Effective
income tax rate
|
0 | % | 0 | % | ||||
F-14
NOTE
7 – SUBSEQUENT EVENTS
On
February 17, 2009, we entered into a modification with Dr. Dionne with regard to
our 4% Convertible Promissory Note issued to Dionne in the amount of $35,000
(“Note”). Pursuant to the modification, Dr. Dionne agreed to extend
the maturity date of the Note from December 2, 2008 to December 2, 2009. As
consideration for the modification, the Company issued Dr. Dionne a common stock
purchase warrant entitling him to purchase 11,000 shares of our common stock at
a per share purchase price of $1.50. The warrant has a five year
term. The warrants also contain anti-dilution protection in the event of stock
splits, stock dividends and other similar transactions.
On
February 17, 2009, we entered into a modification with TR Winston & Company,
LLC (“TRW”) with regard to the Company’s 5% Convertible Debenture issued to TRW
in the amount of $163,600. Pursuant to the modification, TRW agreed
to extend the maturity date of the debenture from July 14, 2009 to July 14,
2010. As consideration for the modification, the we issued TRW a
common stock purchase warrant entitling TRW to purchase 50,000 shares of our
common stock at a per share purchase price of $1.50. The warrant has
a five year term. The warrants also contain anti-dilution protection in the
event of stock splits, stock dividends and other similar
transactions.
On
February 19, 2009, we entered into a Securities Purchase Agreement with a number
of accredited investors. Pursuant to the terms of the Securities
Purchase Agreement, we sold the investors units aggregating approximately
$700,000 “Offering”. The price per unit was $1.50. Each
unit consists of: (i) one share of the Company’s common stock; and (ii) one half
Common Stock Purchase Warrant. The Warrants have a term of five years
and allow the investors to purchase our common shares at a price per share of
$3.00. The warrants also contain anti-dilution protection in the
event of stock splits, stock dividends and other similar
transactions.
As a
result of offering, the anti-dilution provisions in our warrants issued during
the July and August 2008 financing were triggered. These
anti-dilution provisions resulted in the exercise price of these warrants being
reduced from $2.00 from $1.50. Additionally, we are obligated to
issue holders of these warrants an additional 506,754 warrants, and we are
obligated to file a registration statement for the common stock underlying such
warrants pursuant to the registration rights agreement entered into in
connection with the July and August 2008 financing.
F-15
GENSPERA
INC.
(A Development Stage Company)CONDENSED BALANCE SHEETS
|
June
30,
|
December
31,
|
|||||||
|
2009
|
2008
|
|||||||
|
Assets
|
(Unaudited)
|
|||||||
|
Current
assets:
|
||||||||
|
Cash
|
$ | 2,100,212 | $ | 534,290 | ||||
|
Total
current assets
|
2,100,212 | 534,290 | ||||||
|
Intangible
assets, net of accumulated amortization of $19,184 and
$11,511
|
164,983 | 172,657 | ||||||
|
Total
assets
|
$ | 2,265,195 | $ | 706,947 | ||||
|
Liabilities
and stockholders' (deficit) equity
|
||||||||
|
Current
liabilities:
|
||||||||
|
Accounts
payable and accrued expenses
|
$ | 291,456 | $ | 238,817 | ||||
|
Accrued
interest - stockholder
|
8,593 | 5,399 | ||||||
|
Convertible
note payable - stockholder, current portion
|
50,000 | 50,000 | ||||||
|
Total
current liabilities
|
350,049 | 294,216 | ||||||
|
Convertible
note payable, net of discount of $793 and $11,046
|
162,807 | 152,554 | ||||||
|
Convertible
notes payable - stockholder, long term portion
|
105,000 | 105,000 | ||||||
|
Derivative
liabilities
|
1,833,703 | - | ||||||
|
Total
liabilities
|
2,451,559 | 551,770 | ||||||
|
Commitments
and contingencies
|
||||||||
|
Stockholders'
(deficit) equity:
|
||||||||
|
Preferred
stock, par value $.0001 per share; 10,000,000 shares
authorized,
|
||||||||
|
none
issued and outstanding
|
- | - | ||||||
|
Common
stock, par value $.0001 per share; 80,000,000 shares
authorized,
|
||||||||
|
14,469,496
and 12,486,718 shares issued and outstanding
|
1,447 | 1,249 | ||||||
|
Additional
paid-in capital
|
7,403,033 | 4,922,174 | ||||||
|
Deficit
accumulated during the development stage
|
(7,590,844 | ) | (4,768,246 | ) | ||||
|
Total
stockholders' (deficit) equity
|
(186,364 | ) | 155,177 | |||||
|
Total
liabilities and stockholders' (deficit) equity
|
$ | 2,265,195 | $ | 706,947 | ||||
See
accompanying notes to unaudited condensed financial
statements.
4
GENSPERA,
INC.
(A
Development Stage Company)
CONDENSED
STATEMENTS OF LOSSES
FOR THE
THREE AND SIX MONTHS ENDED JUNE 30, 2009 AND 2008
AND FOR
THE PERIOD FROM INCEPTION (NOVEMBER 21, 2003) TO JUNE 30, 2009
(Unaudited)
|
Cumulative
Period
|
||||||||||||||||||||
|
from
November 21, 2003
|
||||||||||||||||||||
|
(date
of inception) to
|
||||||||||||||||||||
|
Three
Months ended June 30,
|
Six
Months ended June 30,
|
June
30,
|
||||||||||||||||||
|
2009
|
2008
|
2009
|
2008
|
2009
|
||||||||||||||||
|
Operating
expenses:
|
||||||||||||||||||||
|
General
and administrative expenses
|
$ | 231,255 | $ | 138,745 | $ | 430,972 | $ | 513,536 | $ | 1,720,514 | ||||||||||
|
Research
and development
|
624,989 | 368,194 | 934,491 | 518,995 | 4,358,898 | |||||||||||||||
|
Total
operating expenses
|
856,244 | 506,939 | 1,365,463 | 1,032,531 | 6,079,412 | |||||||||||||||
|
Loss
from operations
|
(856,244 | ) | (506,939 | ) | (1,365,463 | ) | (1,032,531 | ) | (6,079,412 | ) | ||||||||||
|
Finance
cost
|
(5,155 | ) | - | (478,093 | ) | - | (517,882 | ) | ||||||||||||
|
Change
in fair value of derivative liability
|
(110,326 | ) | (683,111 | ) | (973,567 | ) | ||||||||||||||
|
Interest
income (expense), net
|
(2,867 | ) | (1,605 | ) | (5,475 | ) | (3,211 | ) | (19,983 | ) | ||||||||||
|
Loss
before provision for income taxes
|
(974,592 | ) | (508,544 | ) | (2,532,142 | ) | (1,035,742 | ) | (7,590,844 | ) | ||||||||||
|
Provision
for income taxes
|
- | - | - | - | - | |||||||||||||||
|
Net
loss
|
$ | (974,592 | ) | $ | (508,544 | ) | $ | (2,532,142 | ) | $ | (1,035,742 | ) | $ | (7,590,844 | ) | |||||
|
Net
loss per common share, basic and diluted
|
$ | (0.07 | ) | $ | (0.05 | ) | $ | (0.20 | ) | $ | (0.11 | ) | ||||||||
|
Weighted
average shares outstanding
|
13,026,971 | 10,166,718 | 12,864,048 | 9,789,282 | ||||||||||||||||
See
accompanying notes to unaudited condensed financial statements.
5
GENSPERA,
INC.
(A
Development Stage Company)
CONDENSED
STATEMENT OF STOCKHOLDERS' (DEFICIT) EQUITY
FROM DATE
OF INCEPTION (NOVEMBER 21, 2003) TO JUNE 30, 2009
(Unaudited)
|
Deficit
|
||||||||||||||||||||
|
Additional
|
Accumulated During
the |
|||||||||||||||||||
|
Common
Stock
|
Paid-in
|
Development
|
Stockholders'
|
|||||||||||||||||
|
Shares
|
Amount
|
Capital
|
Stage
|
Equity
|
||||||||||||||||
|
Balance,
November 21, 2003
|
- | $ | - | $ | - | $ | - | $ | - | |||||||||||
|
Sale
of common stock to founders at $0.0001
|
||||||||||||||||||||
|
per
share in November, 2003
|
6,100,000 | 610 | (510 | ) | - | 100 | ||||||||||||||
|
Contributed
services
|
- | - | 120,000 | - | 120,000 | |||||||||||||||
|
Net
loss
|
- | - | - | (125,127 | ) | (125,127 | ) | |||||||||||||
|
Balance,
December 31, 2003
|
6,100,000 | 610 | 119,490 | (125,127 | ) | (5,027 | ) | |||||||||||||
|
Contributed
services
|
- | - | 192,000 | - | 192,000 | |||||||||||||||
|
Stock
based compensation
|
- | - | 24,102 | - | 24,102 | |||||||||||||||
|
Net
loss
|
- | - | - | (253,621 | ) | (253,621 | ) | |||||||||||||
|
Balance,
December 31, 2004
|
6,100,000 | 610 | 335,592 | (378,748 | ) | (42,546 | ) | |||||||||||||
|
Contributed
services
|
- | - | 48,000 | - | 48,000 | |||||||||||||||
|
Stock
based compensation
|
- | - | 24,100 | - | 24,100 | |||||||||||||||
|
Net
loss
|
- | - | - | (126,968 | ) | (126,968 | ) | |||||||||||||
|
Balance,
December 31, 2005
|
6,100,000 | 610 | 407,692 | (505,716 | ) | (97,414 | ) | |||||||||||||
|
Contributed
services
|
- | - | 144,000 | - | 144,000 | |||||||||||||||
|
Stock
based compensation
|
- | - | 42,162 | - | 42,162 | |||||||||||||||
|
Net
loss
|
- | - | - | (245,070 | ) | (245,070 | ) | |||||||||||||
|
Balance,
December 31, 2006
|
6,100,000 | 610 | 593,854 | (750,786 | ) | (156,322 | ) | |||||||||||||
|
Shares
sold for cash at $0.50 per share
|
||||||||||||||||||||
|
in
November, 2007
|
1,300,000 | 130 | 649,870 | - | 650,000 | |||||||||||||||
|
Shares
issued for services
|
735,000 | 74 | 367,426 | - | 367,500 | |||||||||||||||
|
Contributed
services
|
- | - | 220,000 | - | 220,000 | |||||||||||||||
|
Stock
based compensation
|
- | - | 24,082 | - | 24,082 | |||||||||||||||
|
Exercise
of options for cash at $0.003 per share
|
||||||||||||||||||||
|
in
March and June, 2007
|
900,000 | 90 | 2,610 | - | 2,700 | |||||||||||||||
|
Net
loss
|
- | - | - | (691,199 | ) | (691,199 | ) | |||||||||||||
|
Balance,
December 31, 2007
|
9,035,000 | 904 | 1,857,842 | (1,441,985 | ) | 416,761 | ||||||||||||||
|
Exercise
of options for cash at $0.50 per share
|
||||||||||||||||||||
|
on
March 7,2008
|
1,000,000 | 100 | 499,900 | - | 500,000 | |||||||||||||||
|
Sale
of common stock and warrants at $1.00 per
|
||||||||||||||||||||
|
share
- July and August 2008
|
2,320,000 | 232 | 2,319,768 | - | 2,320,000 | |||||||||||||||
|
Cost
of sale of common stock and warrants
|
- | - | (205,600 | ) | - | (205,600 | ) | |||||||||||||
|
Shares
issued for accrued interest
|
31,718 | 3 | 15,856 | - | 15,859 | |||||||||||||||
|
Shares
issued for services
|
100,000 | 10 | 49,990 | - | 50,000 | |||||||||||||||
|
Stock
based compensation
|
- | - | 313,743 | - | 313,743 | |||||||||||||||
|
Contributed
services
|
- | - | 50,000 | - | 50,000 | |||||||||||||||
|
Beneficial
conversion feature of convertible debt
|
- | - | 20,675 | - | 20,675 | |||||||||||||||
|
Net
loss
|
- | - | - | (3,326,261 | ) | (3,326,261 | ) | |||||||||||||
|
Balance,
December 31, 2008
|
12,486,718 | 1,249 | 4,922,174 | (4,768,246 | ) | 155,177 | ||||||||||||||
|
Cumulative
effect of change in accounting principle
|
- | - | (444,161 | ) | (290,456 | ) | (734,617 | ) | ||||||||||||
|
Warrants
issued for extension of debt maturities
|
- | - | 51,864 | - | 51,864 | |||||||||||||||
|
Stock
based compensation
|
- | - | 59,633 | - | 59,633 | |||||||||||||||
|
Common
stock issued for services
|
61,875 | 7 | 74,862 | - | 74,869 | |||||||||||||||
|
Sale
of common stock and warrants at $1.50 per
|
||||||||||||||||||||
|
share
- February 2009
|
466,674 | 46 | 699,939 | - | 699,985 | |||||||||||||||
|
Sale
of common stock and warrants at $1.50 per
|
||||||||||||||||||||
|
share
- April 2009
|
33,334 | 3 | 49,997 | - | 50,000 | |||||||||||||||
|
Sale
of common stock and warrants at $1.50 per
|
||||||||||||||||||||
|
share
- June 2009
|
1,420,895 | 142 | 1,988,725 | - | 1,988,867 | |||||||||||||||
|
Net
loss
|
- | - | - | (2,532,142 | ) | (2,532,142 | ) | |||||||||||||
|
Balance,
June 30, 2009
|
14,469,496 | $ | 1,447 | $ | 7,403,033 | $ | (7,590,844 | ) | $ | (186,364 | ) | |||||||||
See
accompanying notes to unaudited condensed financial statements.
6
GENSPERA,
INC.
(A
Development Stage Company)
CONDENSED
STATEMENTS OF CASH FLOWS
FOR THE
SIX MONTHS ENDED JUNE 30, 2009 AND 2008
AND FOR
THE PERIOD FROM INCEPTION (NOVEMBER 21, 2003) TO JUNE 30, 2009
(Unaudited)
|
Cumulative
Period
|
||||||||||||
|
from
November 21, 2003
|
||||||||||||
|
(date
of inception) to
|
||||||||||||
|
Six
months ended June 30,
|
June
30,
|
|||||||||||
|
2009
|
2008
|
2009
|
||||||||||
|
Cash
flows from operating activities:
|
||||||||||||
|
Net
loss
|
$ | (2,532,142 | ) | $ | (1,035,742 | ) | $ | (7,590,844 | ) | |||
|
Adjustments
to reconcile net loss to net
|
||||||||||||
|
cash
used in operating activities:
|
||||||||||||
|
Amortization
|
7,674 | 3,837 | 19,185 | |||||||||
|
Stock
based compensation
|
134,501 | 295,748 | 980,190 | |||||||||
|
Warrants
issued for financing costs
|
467,840 | - | 467,840 | |||||||||
|
Change
in fair value of derivative liability
|
683,111 | - | 973,567 | |||||||||
|
Contributed
services
|
- | 50,000 | 774,000 | |||||||||
|
Amortization
of debt discount
|
10,253 | - | 19,882 | |||||||||
|
Increase
in accounts payable and accrued expenses
|
55,833 | 31,671 | 315,908 | |||||||||
|
Cash
used in operating activities
|
(1,172,930 | ) | (654,486 | ) | (4,040,272 | ) | ||||||
|
Cash
flows from investing activities:
|
||||||||||||
|
Acquisition
of intangibles
|
- | (184,168 | ) | (184,168 | ) | |||||||
|
Cash
used in investing activities
|
- | (184,168 | ) | (184,168 | ) | |||||||
|
Cash
flows from financing activities:
|
||||||||||||
|
Proceeds
from sale of common stock and warrants
|
2,738,852 | 500,000 | 6,169,652 | |||||||||
|
Proceeds
from convertible notes - stockholder
|
- | - | 155,000 | |||||||||
|
Cash
provided by financing activities
|
2,738,852 | 500,000 | 6,324,652 | |||||||||
|
Net
increase (decrease) in cash
|
1,565,922 | (338,654 | ) | 2,100,212 | ||||||||
|
Cash,
beginning of period
|
534,290 | 590,435 | - | |||||||||
|
Cash,
end of period
|
$ | 2,100,212 | $ | 251,781 | $ | 2,100,212 | ||||||
|
Supplemental
cash flow information:
|
||||||||||||
|
Cash
paid for interest
|
$ | 79 | $ | - | ||||||||
|
Cash
paid for income taxes
|
$ | - | $ | - | ||||||||
|
Non-cash
financial activities:
|
||||||||||||
|
Accrued
interest paid with common stock
|
$ | - | $ | 15,859 | ||||||||
See
accompanying notes to unaudited condensed financial statements.
7
GENSPERA,
INC.
NOTES
TO CONDENSED FINANCIAL STATEMENTS
FOR
THE SIX MONTH PERIODS ENDED JUNE 30, 2009 AND 2008
(Unaudited)
NOTE
1 - SUMMARY OF ACCOUNTING POLICIES
A summary
of the significant accounting policies applied in the preparation of the
accompanying financial statements follows.
Business
and Basis of Presentation
GenSpera
Inc. (“we”, “us”,“our company “, “our”, “GenSpera” or the “Company” )
was formed under the laws of the State of Delaware in 2003. We are a development
stage company, as defined by Statement of Financial Accounting Standards
(“SFAS”) No. 7. GenSpera, Inc. is a pharmaceutical company focused on the
development of targeted cancer therapeutics for the treatment of cancerous
tumors, including breast, prostate, bladder and kidney cancer. Our operations
are based in San Antonio, Texas.
To date,
we have generated no sales revenues, have incurred significant expenses and have
sustained losses. Consequently, our operations are subject to all the risks
inherent in the establishment of a new business enterprise. For the period from
inception on November 21, 2003 through June 30, 2009, we have accumulated losses
of $7,590,844.
The
accompanying unaudited condensed financial statements as of June 30, 2009 and
for the three and six month periods ended June 30, 2009 and 2008 and from date
of inception as a development stage enterprise (November 21, 2003) to June 30,
2009 have been prepared by GenSpera pursuant to the rules and regulations of the
Securities and Exchange Commission, including Form 10-Q and Regulation S-X. The
information furnished herein reflects all adjustments (consisting of normal
recurring accruals and adjustments) which are, in the opinion of management,
necessary to fairly present the operating results for the respective periods.
Certain information and footnote disclosures normally present in annual
financial statements prepared in accordance with accounting principles generally
accepted in the United States of America have been omitted pursuant to such
rules and regulations. The company believes that the disclosures provided are
adequate to make the information presented not misleading. These financial
statements should be read in conjunction with the audited financial statements
and explanatory notes for the year ended December 31, 2008 as disclosed in the
company's 10-K for that year as filed with the SEC, as it may be
amended.
The
results of the six months ended June 30, 2009 are not necessarily indicative of
the results to be expected for the pending full year ending December 31,
2009.
Use
of Estimates
The
preparation of financial statements in conformity with generally accepted
accounting principles requires management to make estimates and assumptions that
affect the amounts reported in the financial statements and accompanying
disclosures. Although these estimates are based on management's best knowledge
of current events and actions the Company may undertake in the future, actual
results may differ from those estimates.
8
NOTE
1 - SUMMARY OF ACCOUNTING POLICIES (cont’d)
Research
and Development
Research
and development costs include expenses incurred by the Company for research and
development of therapeutic agents for the treatment of cancer and are charged to
operations as incurred. Our research and development expenses consist primarily
of expenditures for toxicology and other studies, manufacturing, and
compensation and consulting costs.
GenSpera
incurred research and development expenses of $624,989 and $368,194 for the
three months ended June 30, 2009 and 2008, respectively, and $934,491 and
$518,995 for the six months ended June 30, 2009 and 2008, respectively, and
$4,358,898 from November 21, 2003 (inception) through June 30,
2009.
Loss
Per Share
We use
SFAS No. 128, “Earnings Per Share” for calculating the basic and diluted
loss per share. We compute basic loss per share by dividing net loss and net
loss attributable to common shareholders by the weighted average number of
common shares outstanding. Basic and diluted loss per share are the same, in
that any potential common stock equivalents would have the effect of being
anti-dilutive in the computation of net loss per share. There were 5,248,197
common share equivalents at June 30, 2009 and 1,958,306 at June 30,
2008. For the three and six month periods ended June 30, 2009 and 2008,
these potential shares were excluded from the shares used to calculate diluted
earnings per share as their inclusion would reduce net loss per
share.
Fair value of financial
instruments
In April
2009, we adopted FASB Staff Position (“FSP”) No. FAS 107-1 and APB
28-1, “Interim Disclosures
about Fair Value of Financial Instruments” (“FSP FAS 107-1 and APB
28-1”) for its interim period ended June 30, 2009. FSP FAS 107-1 and
APB 28-1 requires disclosures about fair value of financial instruments as
defined by SFAS No. 107, “Disclosures About Fair Value of
Financial Instruments” (“SFAS 107”), for interim reporting periods
of publicly traded companies as well as in annual financial
statements.
Our
short-term financial instruments, including cash, accounts payable and other
liabilities, consist primarily of instruments without extended maturities, the
fair value of which, based on management’s estimates, reasonably approximate
their book value. The fair value of long term convertible notes is based on
management estimates and reasonably approximates their book value after
comparison to obligations with similar interest rates and maturities. The fair
value of the Company’s derivative instruments is determined using option pricing
models.
Fair value
measurements
Effective
January 1, 2008, we adopted SFAS No. 157, “Fair Value Measurements”
(“SFAS No. 157”). This standard establishes a framework for
measuring fair value and expands disclosure about fair value measurements. The
Company did not elect fair value accounting for any assets and liabilities
allowed by SFAS No. 159, “The Fair Value Option for Financial
Assets and Financial Liabilities”. Effective January 1, 2009, the
Company adopted the provisions of SFAS No. 157 that relate to
non-financial assets and liabilities that are not required or permitted to be
recognized or disclosed at fair value on a recurring basis. Effective April 1,
2009, the Company adopted FSP No. FAS 157-4, “Determining Fair Value When the
Volume and Level of Activity for the Asset or Liability Have Significantly
Decreased and Identifying Transactions That Are Not Orderly” (“FSP
FAS 157-4”). FSP FAS 157-4 provides additional guidance for estimating
fair value in accordance with SFAS No. 157, when the volume and level
of activity for the asset or liability have significantly decreased. The
adoptions of the provisions of SFAS No. 157 that relate to
non-financial assets and liabilities and FSP FAS 157-4 did not have a
material impact on our financial position or results of operations.
9
NOTE
1 - SUMMARY OF ACCOUNTING POLICIES (cont’d)
SFAS No. 157
defines fair value as the amount that would be received for an asset or paid to
transfer a liability (i.e., an exit price) in the principal or most advantageous
market for the asset or liability in an orderly transaction between market
participants on the measurement date. SFAS No. 157 also establishes a
fair value hierarchy that requires an entity to maximize the use of observable
inputs and minimize the use of unobservable inputs when measuring fair value.
SFAS No. 157 describes the following three levels of inputs that may
be used:
Level 1:
Quoted prices (unadjusted) in active markets that are accessible at the
measurement date for identical assets and liabilities. The fair value hierarchy
gives the highest priority to Level 1 inputs.
Level 2:
Observable prices that are based on inputs not quoted on active markets but
corroborated by market data.
Level 3:
Unobservable inputs when there is little or no market data available, thereby
requiring an entity to develop its own assumptions. The fair value hierarchy
gives the lowest priority to Level 3 inputs.
The table
below summarizes the fair values of our financial liabilities as of
June 30, 2009:
|
Fair
Value at
|
Fair Value Measurement
Using
|
|||||||||||||||
|
June 30,
2009
|
Level 1
|
Level 2
|
Level 3
|
|||||||||||||
|
Warrant
derivative liability
|
$
|
1,833,703
|
—
|
—
|
$
|
1,833,703
|
||||||||||
|
$
|
1,833,703
|
$
|
—
|
$
|
—
|
$
|
1,833,703
|
|||||||||
The
following is a description of the valuation methodologies used for these
items:
Warrant derivative
liability — these instruments consist of certain of our warrants
with anti-dilution provisions. These instruments were valued using pricing
models which incorporate the Company’s stock price, volatility, U.S. risk
free rate, dividend rate and estimated life.
Change
in Accounting Principle
In June
2008, the FASB issued Emerging Issues Task Force No. 07-5 (EITF 07-5),
Determining Whether an
Instrument (or Embedded Feature) is Indexed to an Entity’s Own Stock.
EITF 07-5 requires entities to evaluate whether an equity-linked financial
instrument (or embedded feature) is indexed to its own stock by assessing the
instrument’s contingent exercise provisions and settlement provisions.
Instruments not indexed to their own stock fail to meet the scope exception of
Statement of Financial Accounting Standards No. 133, Accounting for Derivative
Instruments and Hedging Activities , paragraph 11(a), and should be
classified as a liability and marked-to-market. The statement is effective for
fiscal years beginning after December 15, 2008 and is to be applied to
outstanding instruments upon adoption with the cumulative effect of the change
in accounting principle recognized as an adjustment to the opening balance of
retained earnings. The Company has assessed its outstanding equity-linked
financial instruments and has concluded that effective January 1, 2009,
warrants issued during 2008 with a fair value of $734,617 on January 1,
2009 will need to be reclassified from equity to a liability. The cumulative
effect of the change in accounting principle on January 1, 2009 is an
increase in our derivative liability related to the fair value of the warrants
of $734,617, a decrease in additional paid-in capital of $444,161, based on the
fair value of the warrants at date of issue, and a $290,456 increase to the
deficit accumulated during development stage to reflect the change in fair value
of the derivative liability from date of issue to January 1, 2009.
10
NOTE
1 - SUMMARY OF ACCOUNTING POLICIES (cont’d)
Recent
Accounting Pronouncements
In
December 2007, the FASB issued SFAS No. 160, “Non-controlling Interests in
Consolidated Financial Statements-an amendment of ARB No. 51” (SFAS 160).
SFAS 160 requires that non-controlling (or minority) interests in subsidiaries
be reported in the equity section of the company’s balance sheet, rather than in
a mezzanine section of the balance sheet between liabilities and equity. SFAS
160 also changes the manner in which the net income of the subsidiary is
reported and disclosed in the controlling company’s income statement. SFAS 160
also establishes guidelines for accounting or changes in ownership percentages
and for deconsolidation. SFAS 160 is effective for financial statements for
fiscal years beginning on or after December 15, 2008 and interim periods within
those years. The adoption of SFAS 160 did not have a material impact on our
financial position, results of operations or cash flows.
In March
2008, the FASB issued SFAS No. 161, “Disclosures about Derivative
Instruments and Hedging Activities-an amendment of FASB Statement No.
133” (SFAS 161). SFAS 161 requires enhanced disclosures about an entity’s
derivative and hedging activities and thereby improves the transparency of
financial reporting. SFAS 161 is effective for financial statements issued for
fiscal years and interim periods beginning after November 15, 2008, with early
application encouraged. The adoption of SFAS 161 did not have a material impact
on our financial position, results of operations or cash flows.
Effective
January 1, 2009, the Company adopted the Financial Accounting Standards Board's
Staff Position (FSP) on the Emerging Issues Task Force (EITF) Issue No. 03-6-1,
“Determining Whether
Instruments Granted in Share-Based Payment Transactions are Participating
Securities.” The FSP required that all unvested share-based
payment awards that contain nonforfeitable rights to dividends should be
included in the basic Earnings Per Share (EPS) calculation. This
standard did not affect the financial position or results of
operations.
In April
2009, the FASB issued FSP FAS No. 115-2 and FAS No. 124-2, “Recognition and Presentation of
Other-Than-Temporary Impairments” (“FSP FAS No. 115-2”). FSP FAS No.
115-2 provides guidance in determining whether impairments in debt securities
are other than temporary, and modifies the presentation and disclosures
surrounding such instruments. This FSP is effective for interim periods ending
after June 15, 2009, but early adoption is permitted for interim periods ending
after March 15, 2009. The adoption of this standard during the second quarter of
2009 had no impact on the Company’s financial position or results of
operations.
In April
2009, the FASB issued FSP FAS No. 157-4, “Determining Fair Value When the
Volume and Level of Activity for the Asset or Liability Have Significantly
Decreased and Identifying Transactions That Are Not Orderly” (“FSP FAS
No. 157-4”). FSP FAS No. 157-4 provides additional guidance in determining
whether the market for a financial asset is not active and a transaction is not
distressed for fair value measurement purposes as defined in SFAS No. 157, “Fair
Value Measurements.” FSP FAS No. 157-4 is effective for interim periods ending
after June 15, 2009, but early adoption is permitted for interim periods ending
after March 15, 2009. The adoption of this standard during the second quarter of
2009 had no impact on the Company’s financial position or results of
operations.
In April
2009, the FASB issued FSP FAS No. 107-1 and APB 28-1, “Interim Disclosures about Fair Value
of Financial Instruments” (“FSP FAS No. 107-1 and APB 28-1”). This FSP
amends FASB Statement No. 107, “Disclosures about Fair Values of Financial
Instruments,” to require disclosures about fair value of financial instruments
in interim financial statements as well as in annual financial statements. APB
28-1 also amends APB Opinion No. 28, “Interim Financial Reporting,” to require
those disclosures in all interim financial statements. This standard is
effective for interim periods ending after June 15, 2009, but early adoption is
permitted for interim periods ending after March 15, 2009. The adoption of this
standard during the second quarter of 2009 had no impact on the Company’s
financial position or results of operations.
11
NOTE
1 - SUMMARY OF ACCOUNTING POLICIES (cont’d)
In May
2009, the FASB issued SFAS No. 165, “Subsequent Events”
(“SFAS No. 165”). SFAS No. 165 establishes general standards
of accounting for and disclosure of events that occur after the balance sheet
date but before financial statements are issued or are available to be issued.
SFAS No. 165 will be effective for interim and annual financial
periods ending after June 15, 2009. The Company adopted
SFAS No. 165 during the three months ended June 30, 2009 and
evaluated subsequent events through the issuance date of the financial
statements. SFAS No. 165 had no impact on the Company’s financial
position or results of operations.
In June
2009, the FASB issued SFAS No. 166, “Accounting for Transfers and
Servicing of Financial Assets – an amendment of SFAS Statement No. 140”
(“SFAS No. 166”). SFAS No. 166 will require more information about transferred
of financial assets and where companies have continuing exposure to the risks
related to transferred financial assets. SFAS 166 is effective at the start of a
company’s first fiscal year beginning after November 15, 2009, or January 1,
2010 for companies reporting earnings on a calendar-year basis.
In June
2009, the FASB issued SFAS No. 167, “Amendments to FASB Interpretation
No. 46(R)” (“SFAS No. 167”). SFAS No. 167 will change how a company
determines when an entity that is insufficiently capitalized or is not
controlled through voting (or similar rights) should be consolidated. Under SFAS
No. 167, determining whether a company is required to consolidate an entity will
be based on, among other things, an entity's purpose and design and a company's
ability to direct the activities of the entity that most significantly impact
the entity's economic performance. SFAS 167 is effective at the start of a
company’s first fiscal year beginning after November 15, 2009, or January 1,
2010 for companies reporting earnings on a calendar-year basis.
In June
2009, the FASB issued SFAS No. 168, “The FASB Accounting Standards
Codification and the Hierarchy of Generally Accepted Accounting Principals – a
replacement of FAS No.162” (“SFAS No. 168”). This statement establishes
the Codification as the source of authoritative U.S. accounting and reporting
standards recognized by the FASB for use in the preparation of financial
statements of nongovernmental entities that are presented in conformity with
GAAP. Rules and interpretive releases of the SEC under authority of federal
securities laws are also sources of authoritative GAAP for SEC registrants. The
Codification was the result of a project of FASB to organize and simplify all
authoritative GAAP literature into one source. This statement is effective for
interim reporting and annual periods ending after September 15, 2009.
Accordingly, the Company will adopt SFAS No. 168 during the quarter ended
September 30, 2009. The Company does not anticipate this statement to have
a material impact on its consolidated financial position, results of operations
or cash flows.
Other
recent accounting pronouncements issued by the FASB (including its Emerging
Issues Task Force), the AICPA, and the SEC did not, or are not believed by
management to, have a material impact on the Company's present or future
financial statements.

10,965,791
Shares
of Common Stock
Prospectus
October
[___], 2009
52
PART
II
INFORMATION
NOT REQUIRED IN PROSPECTUS
Item 13. Other Expenses of Issuance and
Distribution.
The
following table sets forth the estimated costs and expenses in connection with
the sale and distribution of the securities being registered, other than
underwriting discounts and commissions. All expenses incurred will be paid by
the Company. All of the amounts shown are estimates except the
Securities and Exchange Commission, or SEC, registration fees.
|
To be Paid
by the
Registrant
|
||||
|
SEC
registration fees
|
$
|
316
|
||
|
Legal
fees and expenses
|
$
|
30,000
|
||
|
Accounting
fees and expenses
|
$
|
10,000
|
||
|
Printing
and engraving expenses
|
$
|
5,000
|
||
|
Transfer
agent’s fees
|
$
|
2,000
|
||
|
Miscellaneous
fees and expenses
|
$
|
5,000
|
||
|
Total
|
$
|
52,316
|
||
Item 14. Indemnification of Directors and
Officers.
Section 102
of the Delaware General Corporation Law, as amended, or DGCL, allows a
corporation to eliminate the personal liability of directors to a corporation or
its stockholders for monetary damages for a breach of a fiduciary duty as a
director, except where the director breached his duty of loyalty, failed to act
in good faith, engaged in intentional misconduct or knowingly violated a law,
authorized the payment of a dividend or approved a stock repurchase or
redemption in violation of Delaware corporate law or obtained an improper
personal benefit.
Section 145
of the DGCL provides, among other things, that a corporation may indemnify any
person who was or is a party or is threatened to be made a party to any
threatened, pending or completed action, suit or proceeding (other than an
action by or in the right of the corporation) by reason of the fact that the
person is or was a director, officer, employee or agent of the corporation, or
is or was serving at the corporation’s request as a director, officer, employee
or agent of another corporation, partnership, joint venture, trust or other
enterprise, against expenses, including attorneys’ fees, judgments, fines and
amounts paid in settlement actually and reasonably incurred by the person in
connection with the action, suit or proceeding. The power to indemnify applies
if (i) such person is successful on the merits or otherwise in defense of
any action, suit or proceeding or (ii) such person acted in good faith and
in a manner he or she reasonably believed to be in or not opposed to the best
interests of the corporation, and with respect to any criminal action or
proceeding, had no reasonable cause to believe his conduct was unlawful. The
power to indemnify applies to actions brought by or in the right of the
corporation as well, but only to the extent of defense expenses (including
attorneys’ fees but excluding amounts paid in settlement) actually and
reasonably incurred and not to any satisfaction of judgment or settlement of the
claim itself, and with the further limitation that in such actions no
indemnification shall be made in the event of any adjudication of negligence or
misconduct in the performance of his duties to the corporation, unless a court
believes that in light of all the circumstances indemnification should
apply.
Section 174
of the DGCL provides, among other things, that a director who willfully and
negligently approves of an unlawful payment of dividends or an unlawful stock
purchase or redemption may be held liable for such actions. A director who was
either absent when the unlawful actions were approved or dissented at the time,
may avoid liability by causing his or her dissent to such actions to be entered
in the books containing the minutes of the meetings of the board of directors at
the time the action occurred or immediately after the absent director receives
notice of the unlawful acts.
Our
certificate of incorporation states that, to the fullest extent permitted by the
DGCL, no director shall be personally liable to us or our stockholders for
monetary damages for breach of fiduciary duty as director.
Our
bylaws provide that we shall, to the fullest extent authorized by the DGCL,
indemnify any person who was or is made a party or threatened to be made a party
to or is involved in any action, suit or proceeding, whether civil, criminal,
administrative or investigative, by reason of the fact that he or she or a
person of whom he or she is the legal representative, is or was our director or
officer or is or was serving at our request as a director or officer of another
corporation, or as a controlling person of a partnership, joint venture, trust
or other enterprise, including service with respect to employee benefit plans,
whether the basis of such proceeding is alleged action in an official capacity
as a director or officer, or in any other capacity while serving as a director
or officer, against all expenses, liability or loss reasonably incurred or
suffered by such person in connection with such action, suit or proceeding. Our
bylaws also provide that we may enter into one or more agreements with any
director, officer, employee or agent of ours, or any person serving at our
request as a director, officer, employee or agent of another corporation,
partnership, joint venture, trust or other enterprise, including employee
benefit plans, that provides for indemnification rights equivalent to or, if our
board of directors so determines, greater than, those provided for in such
bylaws.
53
We
maintain a liability insurance policy for our directors and officers, subject to
certain exclusions.
Item 15. Recent Sales of Unregistered
Securities.
The
following information is given with regard to unregistered securities sold
during the preceding three years including the dates and amounts of securities
sold, the persons or class of persons to whom we sold the securities, the
consideration received in connection with such sales and, if the securities were
issued or sold other than for cash, the description of the transaction and the
type and amount of consideration received. The descriptions contained
below are a summary and qualified by the agreements, if applicable, included as
Exhibits to this Registration Statement. The following securities were issued in
private offerings pursuant to the exemption from registration contained in
Section 4(2) of the Securities Act and the rules promulgated
thereunder:
|
·
|
On
June 7, 2004, we granted a total of 750,000 common stock options to
members of our Scientific Advisory Board. The options vested over a four
year period on December 31 of each year and have an exercise price of
$0.0016.
|
|
·
|
On
September 29, 2004, we issued one of our shareholders a convertible note
in the amount of $15,000. The consideration paid by the shareholder was
cash. The note accrues interest at 4.2% per annum and is convertible into
common shares at $0.50 per share. The note is due and payable on September
28, 2009.
|
|
·
|
On
December 23, 2005, we issued one of our shareholders a convertible note in
the amount of $35,000. The consideration paid by the shareholder was cash.
The note accrues interest at 4.2% per annum and is convertible into common
shares at $0.50 per share. The note is due and payable on December 22,
2010.
|
|
·
|
On
May 19, 2006, we issued one of our shareholders a convertible note in the
amount of $40,000. The consideration paid by the shareholder was cash. The
note accrues interest at 4.2% per annum and is convertible into common
shares at $0.50 per share. The note is due and payable on May 18,
2011.
|
|
·
|
On
November 10, 2006, we granted a total of 150,000 common stock options to
employees and advisors. The options vested upon grant, have a term of 10
years and an exercise price of $0.01. All of these options were exercised
during 2007.
|
|
·
|
On
December 6, 2006, we issued one of our shareholders a convertible note in
the amount of $30,000. The consideration paid by the shareholder was cash.
The note accrues interest at 4.2% per annum and is convertible into common
shares at $0.50 per share. The note is due and payable on December 6,
2011.
|
|
·
|
In
March and June of 2007 we issued a total of 900,000 common shares to
employees and advisors in connection with the exercise of the options
granted on June 7, 2004 and November 10,
2006.
|
|
·
|
During
2007, we issued an aggregate of 735,000 shares of common stock, valued at
$367,500, as compensation for consulting and financial and legal advisory
services. The compensation cost was based on the fair value of our common
stock as determined by the Company’s board of
directors.
|
|
·
|
During
November 2007, we sold an aggregate of 1,300,000 common shares in a
private placement to accredited investors at $0.50 per share, for gross
proceeds of $650,000.
|
|
·
|
On
January 1, 2008, we granted a total of 1,000,000 common stock warrants to
consultants for financial services. Of this total, 500,000 stock warrants
were granted to JD Group, LLC and an additional 500,000 common stock
warrants were granted to G. Tyler Runnels or Jasmine Niklas Runnels TTEES
The Runnel Family Trust dtd 1-11-20. The warrants have an
exercise price of $0.50 per share. The warrants vested upon
grant.
|
|
·
|
On
January 7, 2008, we granted 100,000 shares of common stock, valued at
$50,000, to a former director, Richard P. Burgoon, Jr., as compensation
for serving on the board. The shares were vested upon
grant.
|
|
·
|
On
February 1, 2008, we granted a total of 240,000 common stock options to
members of our Scientific Advisory Board. Of this total, 60,000 options
were granted to each of the following individuals: Dr. John T. Isaacs, Dr.
Samuel R. Denmeade, Dr. Soren Brogger Christensen and Dr. Hans Lilja. The
options have an exercise price of $0.50 per share. The options vest
in equal installments quarterly over a period of three years commencing
March 31, 2008, and lapse if unexercised on January 31,
2018.
|
54
|
·
|
On
February 11, 2008, we granted a total of 100,000 common stock options to
the Verrazano Group, LLC, a consultant, for investor relation services.
The options have an exercise price of $0.50 per share and expire if
unexercised on February 11, 2013. Subsequent to the issuance, options to
purchase 16,000 shares lapsed.
|
|
·
|
On
February 11, 2008, we granted a total of 20,000 common stock options to a
consultant, Robert C. Scherne, CPA, PC for professional services. The
options have an exercise price of $0.50 per share. The options vest
in equal installments quarterly over a period of one year commencing March
31, 2008, and lapse if unexercised on February
11, 2018.
|
|
·
|
On
March 6, 2008, we granted a total of 1,000,000 common stock warrants to
consultants for financial services. Of this total, 500,000 stock warrants
were granted to Pangaea Partners, LLC and an additional 500,000 common
stock warrants were granted to High Tide, LLC. The warrants have an
exercise price of $1.00 per
share.
|
|
·
|
On
March 7, 2008, we issued 1,000,000 common shares upon the exercise of
1,000,000 common stock warrants at $.50 per share. Five hundred thousand
warrants were each exercised by JD Group, LLC and G. Tyler Runnels or
Jasmine Niklas Runnels TTEES The Runnel Family Trust dtd 1-11-20. We
received gross proceeds of
$500,000.
|
|
·
|
During
March 2008, we granted to our board of directors, as compensation for
serving on our board of directors, options to purchase an aggregate
of 300,000 common shares at $0.50 per share, reflecting the fair
market value of the shares as of that date. Each Director (Scott Ogilvie,
Richard P. Burgoon, Jr. and John M. Farah, Jr., Ph.D.) received 50,000
options that vested upon grant with the remaining 50,000 options vesting
quarterly over a period of two years commencing March 31, 2008, and lapse
if unexercised on April 1, 2018.
|
|
·
|
On
March 7, 2008, we issued 31,718 shares of common stock to our Chief
Executive Officer and President, Craig A. Dionne, Ph.D., as payment of
accrued interest in the amount of
$15,859.
|
|
·
|
During
July and August of 2008, we sold an aggregate of 2,320,000 units resulting
in gross proceeds of $2,320,000 or $1.00 per unit. Each unit consists of:
(i) 1 share of common stock; and (ii) ½ common stock purchase warrant. The
warrants have a term of 5 years and an exercise price of $2.00 per shares
subject to certain anti-dilution adjustments. The warrants are also
callable by the Company in the event the Company’s shares are publically
traded in the future and certain price and volume conditions are
met.
|
TR
Winston & Company, LLC acted as the Company’s placement agent with respect
to the transaction. Pursuant to a placement agent agreement with TR Winston
& Company, LLC we agreed to the following compensation: (i) cash fee equal
to 8% of gross proceeds raised, including any payments made to the Company upon
the exercise of the warrants; (ii) the issuance of a warrant to purchase 8% of
all securities issued; and (iii) payment of legal expenses totaling $20,000.
Accordingly, we issued to TR Winston & Company, LLC a warrant to purchase
278,400 common shares of which they assigned 22,500 to Mercer Capital for
assisting in the transaction. The warrant has an exercise price per common
shares of $2.00 and a term of 5 years.
Also, as
an accommodation to the Company, TR Winston & Company, LLC agreed to receive
a convertible debenture and warrants to purchase an additional 81,800 common
shares in lieu of $163,600 of its cash fee. The warrant is in addition to any
warrants received as compensation for acting as placement agent. The
convertible debenture accrues interest at 5% per annum and has a maturity date
of July 14, 2009. It is convertible into the shares of the Company’s common
stock, at the sole discretion of the holder, at $1.00 per share subject to
certain anti-dilution adjustments. The warrant has the same terms as those
issued to investors in the offering.
|
·
|
In
October of 2008, we granted warrants to purchase an aggregate of
50,000 common shares to Crystal Research Associates, LLC for
marketing services. The warrants have an exercise price of $2.00 per
share.
|
|
·
|
In
October of 2008, we granted options to purchase an aggregate of
15,000 common shares to our director Scott Ogilvie at an exercise
price of $1.00 per share. The options vested on the date of grant and
lapse if unexercised on October 16,
2018.
|
|
·
|
On
February 17, 2009, we entered into a modification with TRW with regard to
our 5% Convertible Debenture issued to TRW in the amount of
$163,600. Pursuant to the modification, TRW agreed to extend
the maturity date of the debenture from July 14, 2009 to July 14,
2010. As consideration for the modification, we issued TRW a
common stock purchase warrant entitling TRW to purchase 50,000 shares of
our common stock at a per share purchase price of $1.50. The
warrant has a five year term and contains certain anti-dilution provisions
requiring us to adjust the exercise price and number of shares upon the
occurrence of a stock split, stock dividends or stock
consolidation.
|
55
|
·
|
On
February 17, 2009, we entered into a modification with Craig Dionne, our
Chief Executive Officer and Chairman with regard to our 4% Convertible
Promissory Note issued to Mr. Dionne in the amount of
$35,000. Pursuant to the modification, Mr. Dionne agreed to
extend the maturity date of the Note from December 2, 2008 to December 2,
2009. Mr. Dionne had previously deferred repayment of the
note. As consideration for the modification, we issued Mr.
Dionne a common stock purchase warrant entitling Mr. Dionne to purchase
11,000 shares of our common stock at a per share purchase price of
$1.50. The warrant has a five year term and contains certain
anti-dilution provisions requiring us to adjust the exercise price and
number of shares upon the occurrence of a stock split, stock dividends or
stock consolidation.
|
|
·
|
On
February 19, 2009, we entered into a securities purchase agreement with a
number of accredited investors. Pursuant to the terms of
the securities purchase agreement, we sold the investors 500,000 units in
the aggregate amount of approximately $750,000. The price per
unit was $1.50. Each unit consists of: (i) one share of common
stock; and (ii) one-half common stock purchase warrant. The
warrants have a term of five years and allow the holder to purchase our
common shares at a price per share of $3.00. The warrants also
contain anti-dilution protection in the event of stock splits, stock
dividends and other similar transactions. The provisions
do not provide for any adjustment in the event of subsequent equity sales
or transactions. The warrants are also callable by the Company
in the event the Company’s shares are publically traded in the future and
certain price and volume conditions are
met.
|
As a
result of offering, the anti-dilution provisions in our warrants issued during
the July and August 2008 financing were triggered. These
anti-dilution provisions resulted in the exercise price of these warrants being
reduced from $2.00 to $1.50. Additionally, we issued holders of these
warrants an additional 506,754 additional warrants. We are obligated
to file a registration statement for the common stock underlying such warrants
pursuant to the registration rights agreement entered into in connection with
the July and August 2008 financing.
We also
entered into registration rights agreements with the investors granting certain
registration rights with regard to the shares and the shares underlying the
warrants. The registration rights Agreement provides for penalties to
be paid in restricted shares in the event the Company: (i) fails to file a
registration statement or have such registration statement declared effective
within a certain period of time; or (ii) fails to maintain the registration
statement effective until all the securities registered therein are sold or are
eligible for resale pursuant to Rule 144 without manner of sale or volume
restrictions. Subsequent to the issuance, a majority of the investors
agreed to waive the date by which the registration statement needed to be
filed. As a result of the waiver, a registration statement covering
the shares and shares underlying the warrants must be filed on or before July
31, 2009.
|
·
|
On
May 8, 2009, we issued 61,875 common shares to Lyophilization Services of
New England, Inc. as payment for services valued at $92,812.50 provided in
connection with manufacturing of our first drug
compound. The shares were valued at $1.50 per
share.
|
|
·
|
In
June and July of 2009 we entered into a series of securities purchase
agreements with a number of accredited and institutional
investors. Pursuant to the terms of the agreements, we offered
and sold an aggregate of 2,025,344 units resulting in gross proceeds to us
of approximately $3,038,000. The price per unit was
$1.50. Each unit consists of: (i) one share of common stock;
and (ii) one half common stock purchase warrant. The warrants
have a term of five years and entitle the investors to purchase our common
shares at a price per share of $3.00. The warrants also contain
provisions providing for an adjustment in the underlying number of shares
and exercise price in the event of stock splits or stock dividends and
fundamental transactions. The provisions do not provide for any
adjustment in the event of subsequent equity sales or
transactions. The warrants are also callable in the event our
common stock becomes publically traded and certain other conditions, as
described in the warrants, are met. The Company incurred a
total of $220,050 in fees and expenses incurred in connection with the
transaction. Of this amount, $50,000 was paid through the
issuance of 33,334 units. We also issued a total of 83,895 additional
common stock purchase warrants as compensation to certain
finders. The warrants have the same terms as the investor
warrants. The securities purchase agreements are substantially
similar other than the agreement entered into on June 29, 2009 extended
the expiration of most favored nation treatment from 90 days until
December 31, 2009.
|
The
Company also entered in two registration rights Agreements with the investors
granting the Investors certain registration rights with regard to the Shares and
the shares underlying the Warrants. The Registration Rights Agreement
provides for penalties to be paid in restricted shares in the event the Company:
(i) fails to file a registration statement or have such registration statement
declared effective within a certain period of time; or (ii) fails to maintain
the registration statement effective until all the securities registered therein
are sold or are eligible for resale pursuant to Rule 144 without manner of sale
or volume restrictions. Both Registration Rights Agreements are
substantially similar other than the agreement entered into on June 29, 2009
requires the Company to: (i) file a registration statement covering the Shares
and common stock underlying the Warrants by July 31, 2009 as compared to 120
days after closing; and (ii) requires the registration statement to be declared
effective 150 days after filing as compared to 270 days after
filing. Notwithstanding the forgoing, the Company anticipates
filing a registration statement covering all the shares and common stock
underlying the warrants issued in connection with the June 29 and June 30th
closing by July 31, 2009.
56
|
·
|
On
July 10, 2009, we issued Kemmerer Resources Corp. a common stock
purchase warrant to purchase 150,000 common shares as reimbursement of due
diligence expenses. The warrants have a term of five years and entitle the
investors to purchase our common shares at a price per share of
$3.00. The warrants also contain provisions providing for an
adjustment in the underlying number of shares and exercise price in the
event of stock splits or stock dividends and fundamental
transactions. The provisions do not provide for any adjustment
in the event of subsequent equity sales or
transactions.
|
|
·
|
On
September 2, 2009 we entered a securities purchase agreement with a number
of accredited investors. Pursuant to the terms of the
agreement, the Company sold units in the aggregate of 160,000 units
resulting in gross proceeds of $240,000. The price per unit was
$1.50. Each unit consists of: (i) one share of the Company’s
common stock; and (ii) one half common stock purchase
warrant. The warrants have a term of five years and entitle the
Investors to purchase the Company’s common shares at a price per share of
$3.00. The warrants also contain provisions providing for an
adjustment in the underlying number of shares and exercise price in the
event of stock splits or stock dividends and fundamental
transactions. The provisions do not provide for any adjustment
in the event of subsequent equity sales or transactions. The
warrants are also callable in the event the our common stock becomes
publically traded and certain other conditions, as described in the
warrants, are met. In connection with the offering, we paid a
total of $23,100 in fees and expenses incurred in connection with the
transaction. We also issued a warrants to purchase 12,267,
common shares, with identical terms to the warrant, as a partial finder’s
fee in connection with the
offering.
|
We also
entered in a Registration Rights Agreement with regard to the registration of
the shares and the shares underlying the warrants. The Registration
Rights Agreement provides for penalties to be paid in restricted shares in the
event we: (i) fail to file a registration statement or have such registration
statement declared effective within a certain period of time; or (ii) fail to
maintain the registration statement effective until all the securities
registered therein are sold or are eligible for resale pursuant to Rule 144
without manner of sale or volume restrictions.
|
·
|
On
September 2, 2009, we issued Messrs Dionne and Richerson common stock
purchase options to purchase an aggregate of 1,775,000 common
shares. For a further description of the grant, refer to the
section of this registration statement entitled “Employment Agreements and
Arrangements.”
|
|
·
|
On
September 2, 2009, we issued certain consultants options to purchase an
aggregate of 125,000 common shares. The options were granted
pursuant to our 2007 Equity Compensation Plan, have an exercise price of
$1.50 per share and are fully vested at the grant date. The
options have a term of 5 years and can be exercisable at any time during
their term.
|
|
·
|
On
September 2, 2009, we issued a consultant a warrant to purchase 20,000
common shares. The warrant was issued as compensation for
services related to our clinical trials. The warrant has an
exercise price of $1.50 per share and is fully vested at the grant
date. The warrant has a term of 5
years.
|
|
·
|
On
September 2, 2009, we issued a consultant a warrant to purchase 100,000
common shares. The warrant was issued as compensation for
investor relations services. The warrant has an exercise price
of $1.50 per share and is fully vested at the grant date. The
warrant has a term of 5
years.
|
|
·
|
On
September 2, 2009, we issued one of our service providers 25,000 common
shares as compensation for services relating to the coordination of
clinical trial sites and CROs services. The shares were valued
at $1.50 per share or an aggregate consideration of
$37,500.
|
Item 16. Exhibits.
See
Exhibit Index beginning on page 60 of this registration
statement.
Item 17. Undertakings.
Insofar
as indemnification by the Registrant for liabilities arising under the
Securities Act may be permitted to directors, officers and controlling persons
of the Registrant pursuant to the provisions referenced in Item 15 of this
registration statement or otherwise, the Registrant has been advised that in the
opinion of the SEC such indemnification is against public policy as expressed in
the Securities Act, and is therefore unenforceable. In the event that a claim
for indemnification against such liabilities (other than the payment by the
Registrant of expenses incurred or paid by a director, officer, or controlling
person of the Registrant in the successful defense of any action, suit or
proceeding) is asserted by such director, officer or controlling person in
connection with the securities being registered hereunder, the Registrant will,
unless in the opinion of its counsel the matter has been settled by controlling
precedent, submit to a court of appropriate jurisdiction the question whether
such indemnification by it is against public policy as expressed in the
Securities Act and will be governed by the final adjudication of such
issue.
57
The
undersigned Registrant hereby undertakes:
|
(1)
|
To
file, during any period in which offers or sales are being made, a
post-effective amendment to this registration
statement:
|
|
(i)
|
To
include any prospectus required by section 10(a)(3) of the Securities
Act;
|
|
(ii)
|
To
reflect in the prospectus any facts or events arising after the effective
date of the registration statement (or the most recent post-effective
amendment thereof) which, individually or in the aggregate, represent a
fundamental change in the information set forth in the registration
statement. Notwithstanding the foregoing, any increase or decrease in
volume of securities offered (if the total dollar value of securities
offered would not exceed that which was registered) and any deviation from
the low or high end of the estimated maximum offering range may be
reflected in the form of prospectus filed with the Commission pursuant to
Rule 424(b) if, in the aggregate, the changes in volume and price
represent no more than a 20% change in the maximum aggregate offering
price set forth in the “Calculation of Registration Fee” table in the
effective registration statement;
and
|
|
(iii)
|
To
include any material information with respect to the plan of distribution
not previously disclosed in the registration statement or any material
change to such information in the registration
statement;
|
|
(2)
|
That,
for the purpose of determining any liability under the Securities Act,
each such post-effective amendment shall be deemed to be a new
registration statement relating to the securities offered therein, and the
offering of such securities at that time shall be deemed to be the initial
bona fide offering thereof.
|
|
(3)
|
To
remove from registration by means of a post-effective amendment any of the
securities being registered which remain unsold at the termination of the
offering.
|
58
SIGNATURES
Pursuant
to the requirements of the Securities Act, the Registrant has duly caused this
Registration Statement to be signed on its behalf by the undersigned, thereunto
duly authorized in the City of San Antonio, State of Texas, on October 15,
2009.
|
GENSPERA,
INC.
|
||
|
By:
|
||
|
/S/ Craig Dionne,
PhD
|
||
|
Craig
Dionne, PhD
|
||
|
Chief
Executive Officer
|
||
POWER
OF ATTORNEY
KNOW ALL
MEN BY THESE PRESENTS, that each person whose signature appears below
constitutes and appoints Craig Dionne and Russell Richerson, and each of them
acting alone, with full power of substitution and resubstitution and full power
to act without the other, his true and lawful attorney-in-fact and agent, with
full power of substitution and resubstitution for him and in his name, place and
stead, in any and all capacities, to sign any and all amendments to this
Registration Statement, and all documents in connection therewith (including all
post-effective amendments and any subsequent registration statement filed
pursuant to Rule 462(b) under the Securities Act), with the SEC, granting unto
said attorney-in-fact and agent, full power and authority to do and perform each
and every act and thing requisite and necessary to be done in and about the
premises, as fully to all intents and purposes as he might or could do in
person, hereby ratifying and confirming all that said attorney-in-fact and
agent, or his substitute or substitutes, may lawfully do or cause to be done by
virtue hereof.
Pursuant
to the requirements of the Securities Act, this registration statement has been
signed by the following persons in the capacities and on the dates
indicated.
|
Signature
|
Title
|
Date
|
||
|
/S/ CRAIG
DIONNE
|
Chief
Executive Officer, President and Director
|
October
15, 2009
|
||
|
Craig
Dionne
|
(Principal
Executive Officer)
|
|||
|
/S/ CRAIG
DIONNE
|
Chief
Financial Officer
|
October
15, 2009
|
||
|
Craig
Dionne
|
(Principal
Financial and Accounting Officer)
|
|||
|
/S/ JOHN M. FARAH,
JR
|
Director
|
October
15, 2009
|
||
|
John
M. Farah, Jr.
|
||||
|
/S/ SCOTT
OGILVIE
|
Director
|
October
15, 2009
|
||
|
Scott
Ogilvie
|
59
INDEX
TO EXHIBITS
|
|
Incorporated by Reference
|
||||||||||||||
|
Exhibit
No.
|
Description
|
Filed
Herewith
|
Form
|
Exhibit
No.
|
File No.
|
Filing Date
|
|||||||||
|
3.01
|
Amended
and Restated Certificate of Incorporation
|
S-1 | 3.01 | 333-153829 |
10/03/08
|
||||||||||
|
3.02
|
Bylaws
|
S-1 | 3.02 | 333-153829 |
10/03/08
|
||||||||||
|
4.01
|
Specimen
of Common Stock certificate
|
S-1 | 4.01 | 333-153829 |
10/03/08
|
||||||||||
|
4.02**
|
Amended
and Restated GenSpera 2007 Equity Compensation Plan adopted on April 20,
2009
|
8-K | 4.01 | 333-153829 |
4/24/09
|
||||||||||
|
4.03**
|
GenSpera
Form of 2007 Equity Compensation Plan Grant and 2009 Executive
Compensation Plan
|
8-K | 4.02 | 333-153829 |
9/09/09
|
||||||||||
|
4.04
|
Form
of 4.0% convertible note issued to shareholder
|
S-1 | 4.05 | 333-153829 |
10/03/08
|
||||||||||
|
4.05
|
Form
of Subscription Agreement for November 2007 offering
|
S-1 | 4.06 | 333-153829 |
10/03/08
|
||||||||||
|
4.06
|
Form
of Warrant dated March 6, 2008 issued to consultant for financial
consulting services.
|
S-1 | 4.07 | 333-153829 |
10/03/08
|
||||||||||
|
4.07
|
Form
of Securities Purchase Agreement—July and August 2008 private
placement
|
S-1 | 4.08 | 333-153829 |
10/03/08
|
||||||||||
|
4.08
|
Form
of Registration Rights Agreement – July and August 2008 private
placement
|
S-1 | 4.09 | 333-153829 |
10/03/08
|
||||||||||
|
4.09
|
Form
of Warrant – July and August 2008 private placement
|
S-1 | 4.10 | 333-153829 |
10/03/08
|
||||||||||
|
4.10
|
Form
of insider Lock-Up Agreement – July and August 2008 private
placement
|
S-1 | 4.11 | 333-153829 |
10/03/08
|
||||||||||
|
4.11
|
Form
of 5.0% convertible debenture issued to TR Winston & Company,
LLC
|
S-1 | 4.12 | 333-153829 |
10/03/08
|
||||||||||
|
4.12
|
Form
of 5.0% convertible debenture modification between TR Winston
& Company, LLC and GenSpera, Inc.
|
8-K | 10.01 | 333-153829 |
2/20/09
|
||||||||||
|
4.13
|
Form
of 4.0% convertible debenture modification between GenSpera,
Inc. and shareholder
|
8-K | 10.02 | 333-153829 |
2/20/09
|
||||||||||
60
|
4.14
|
Form
of Common Stock Purchase Warrant issued on 2/17/09 to TR Winston &
Company, LLC
|
8-K | 10.05 | 333-153829 |
2/20/09
|
||||||||||
|
4.15
|
Form
of Common Stock Purchase Warrant issued on 2/17/09 to Craig
Dionne
|
8-K | 10.06 | 333-153829 |
2/20/09
|
||||||||||
|
4.16
|
Form
of Securities Purchase Agreement dated 2/19/09
|
8-K | 10.01 | 333-153829 |
2/20/09
|
||||||||||
|
4.17
|
Form
of Common Stock Purchase Warrant dated 2/19/09
|
8-K | 10.02 | 333-153829 |
2/20/09
|
||||||||||
|
4.18
|
Form
of Registration Rights Agreement dated 2/19/09
|
8-K | 10.03 | 333-153829 |
2/20/09
|
||||||||||
|
4.19
|
Form
of Securities Purchase Agreement dated 6/29/09
|
8-K | 10.01 | 333-153829 |
7/06/09
|
||||||||||
|
4.20
|
Form
of Securities Purchase Agreement dated 6/30/09
|
8-K | 10.02 | 333-153829 |
7/06/09
|
||||||||||
|
4.21
|
Form
of Common Stock Purchase Warrant dated June of 2009
|
8-K | 10.03 | 333-153829 |
7/06/09
|
||||||||||
|
4.22
|
Form
of Registration Rights Agreement dated 6/29/09
|
8-K | 10.04 | 333-153829 |
7/06/09
|
||||||||||
|
4.20
|
Form
of Registration Rights Agreement dated 6/30/09
|
8-K | 10.05 | 333-153829 |
7/06/09
|
||||||||||
|
4.21**
|
2009
Executive Compensation Plan
|
8-K | 4.01 | 333-153829 |
9/09/09
|
||||||||||
|
4.22
|
Form
of Securities Purchase Agreement – September 2, 2009
|
8-K | 10.01 | 333-153829 |
9/09/09
|
||||||||||
|
4.23
|
Form
of Common Stock Purchase Warrant – September 2, 2009
|
8-K | 10.02 | 333-153829 |
9/09/09
|
||||||||||
|
4.24
|
Form
of Registration Rights Agreement—September 2, 2009
|
8-K | 10.03 | 333-153829 |
9/09/09
|
||||||||||
|
5.01
|
Opinion
of Silvestre Law Group, P.C.
|
*
|
|||||||||||||
|
10.01
|
Form
of Transactional Fee Agreement between the Company and TR Winston &
Company, LLC dated March 17, 2008
|
S-1 | 10.01 | 333-153829 |
10/03/08
|
61
|
10.02
|
Exclusive
Supply Agreement between GenSpera and Thapsibiza dated January 22,
2008
|
S-1 | 10.02 | 333-153829 |
10/03/08
|
||||||||||
|
10.03**
|
Craig
Dionne Employment Agreement
|
8-K | 10.04 | 333-153829 |
9/09/09
|
||||||||||
|
10.04**
|
Craig
Dionne Severance Agreement
|
8-K | 10.05 | 333-153829 |
9/09/09
|
||||||||||
|
10.05**
|
Craig
Dionne Proprietary Information, Inventions And Competition
Agreement
|
8-K | 10.06 | 333-153829 |
9/09/09
|
||||||||||
|
10.06**
|
Form
of Indemnification Agreement
|
8-K | 10.07 | 333-153829 |
9/09/09
|
||||||||||
|
10.07**
|
Russell
Richerson Employment Agreement
|
8-K | 10.08 | 333-153829 |
9/09/09
|
||||||||||
|
10.08**
|
Russell
Richerson Proprietary Information, Inventions And Competition
Agreement
|
8-K | 10.09 | 333-153829 |
9/09/09
|
||||||||||
|
10.04**
|
Terms
of verbal employment agreement with Russell Richerson dated July 1,
2008
|
S-1 | 10.04 | 333-153829 |
10/03/08
|
||||||||||
|
23.01
|
Consent
of RBSM LLP
|
*
|
|||||||||||||
|
23.02
|
Consent
of Silvestre Law Group, P.C. (contained in opinion filed as Exhibit 5.01
to this registration statement)
|
*
|
|||||||||||||
|
24.01
|
Power
of Attorney – Included on the signature page
|
*
|
**Management
contracts or compensation plans or arrangements in which directors or executive
officers are eligible to participate.
62
